Abstract
The escalating crisis of global plastic pollution has intensified the demand for eco-friendly and biodegradable materials. Polylactic acid (PLA) stands as a prominent candidate in this regard, valued for its biodegradability and biocompatibility. However, incorporating fillers to reduce cost and modify properties often introduces challenges in interfacial compatibility. In this study, a polycaprolactone-based polyurethane prepolymer (PCLPU) was synthesized as a compatibilizer to improve the poor interfacial adhesion between PLA and mung bean starch (MBS). PLA/MBS/PCLPU composites were prepared by melt extrusion, and the influence of the soft segment molecular weight of PCLPU on the structure and properties of the composites was systematically investigated. Results indicate that PCLPU enhances the toughness of the composites through a combination of chemical bonding and physical cross-linking. Although PCLPUs with different soft segment molecular weights all contributed to toughening, a lower molecular weight promoted more complete chemical cross-linking, resulting in more pronounced improvements in mechanical performance. In particular, the composite compatibilized with the lowest-molecular-weight prepolymer (PLAPSPU-500) exhibited the most significant enhancement, with a 27.14% increase in tensile strength, 162.67% in elongation at break, 175.00% in impact strength, and 31.62% in flexural strength, compared to PLA/MBS composites without compatibilizer. This work provides a viable strategy for developing high-performance PLA-based biodegradable composites, thereby facilitating the broader adoption of sustainable materials.
Keywords
Introduction
In recent years, the proliferation of non-degradable plastic products has exerted escalating pressure on the global environment. 1 This issue has catalyzed a significant shift in research focus toward replacing conventional plastics with more environmentally friendly, bio-based, or biodegradable alternatives, which have become a central theme in materials science. 2 Polylactic acid (PLA) stands out as a prominent bio-based polymer with considerable ecological value and potential as a sustainable resource substitute. 3 Sourced from plant sugars such as those derived from corn, sugarcane, and cassava, PLA is mass-produced through bio-fermentation and chemical synthesis, providing a viable alternative to petroleum-based monomers. 4 Studies show that the carbon emissions associated with PLA production are approximately 60% lower than those of conventional petroleum-based plastics.5,6 Therefore, modifying PLA to improve its performance for a variety of industrial and consumer applications represents a highly feasible strategy for advancing the use of biodegradable plastics. 7
However, PLA suffers from several inherent drawbacks compared to traditional plastics. Mechanically, its low elongation at break (typically around 3%) leads to pronounced brittleness and a high susceptibility to fracture. 8 Economically, the high production cost of PLA continues to hinder its widespread market adoption.9,10
A promising approach to overcome these issues is the incorporation of natural, bio-based fillers into PLA. 11 Starch is an ideal candidate due to its low cost, wide availability, and safety. However, the strong hydrophilicity of starch results in poor compatibility with the hydrophobic PLA matrix. 12 A compatibilizer addresses this by forming chemical bonds with both components, thereby enhancing interfacial adhesion. 13 It encapsulates the filler to form a cross-linked layer, which can physically interact with both phases. This process reduces interfacial gaps and improves the composite’s mechanical properties.14,15
Prior research has explored similar issues in other biopolymer systems. For example, Mouren et al. 16 employed methylene diphenyl diisocyanate (MDI) as a key component in the synthesis of sustainable thermoplastic polyurethanes (TPUs) using the bio-based cycloaliphatic diol betulin. Their study systematically investigated the reactivity between isocyanate (-NCO) groups and the hydroxyl (-OH) groups of betulin, demonstrating that controlled NCO/OH reactions are crucial for forming urethane linkages and enhancing interfacial adhesion within the polymer matrix. In another study, Ivanič et al. 17 tested urea as a compatibilizer in PBAT/TPS blends, though interfacial adhesion was still suboptimal. Both strategies rely on reactions between isocyanate (-NCO) and hydroxyl (-OH) groups to promote cross-linking at the interface. 18
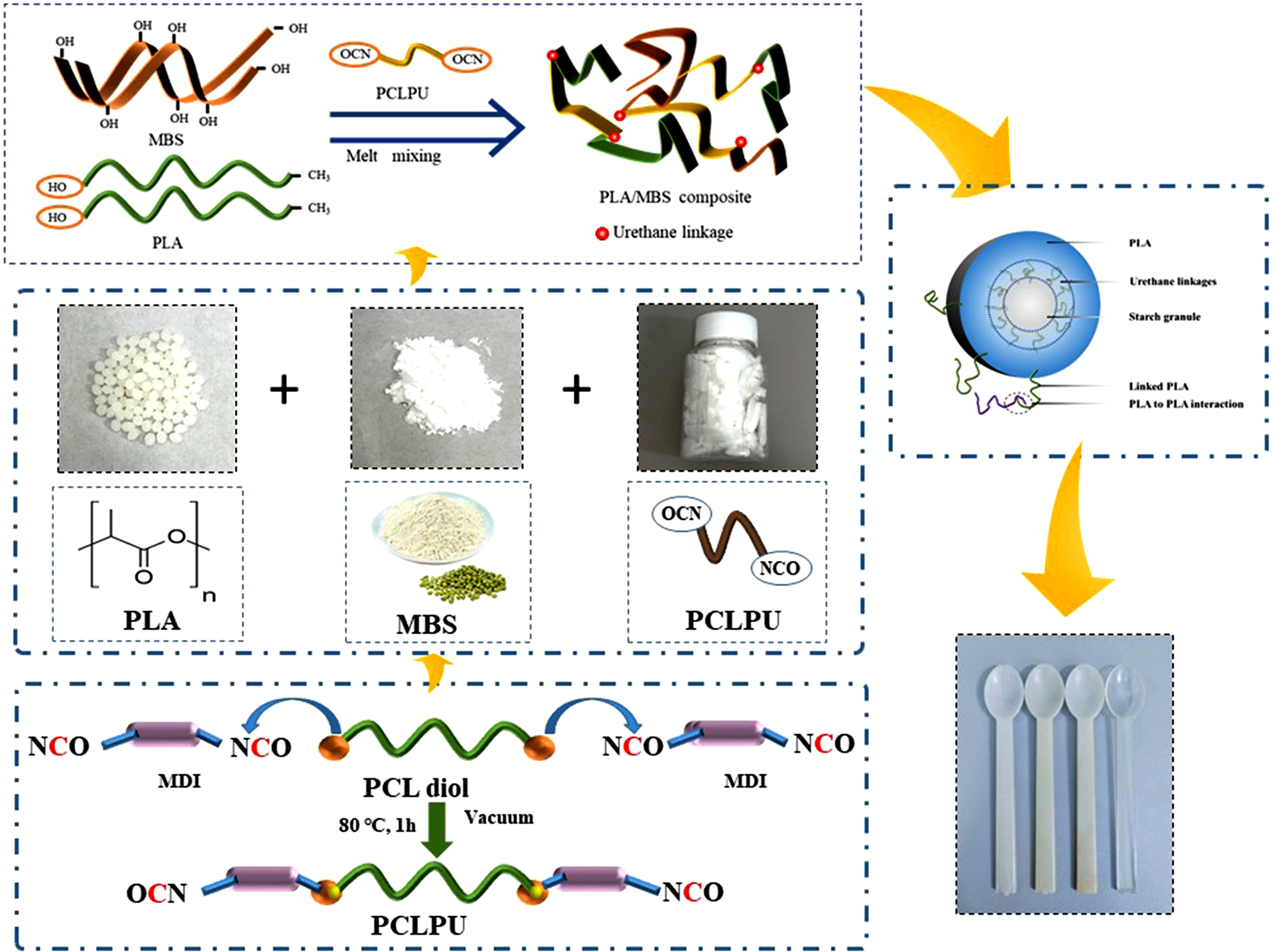
Building on the established reactivity of isocyanate groups, this study introduces a polycaprolactone-based polyurethane (PCLPU) compatibilizer to address the critical interfacial incompatibility between polylactic acid (PLA) and mung bean starch (MBS). A series of PCLPUs with varying soft-segment molecular weights were synthesized to systematically investigate their role in interfacial design. The composites were fabricated via melt extrusion and hot-pressing. To elucidate the compatibilization mechanism, two control groups were established: an uncompatibilized composite and one with -NCO capped PCLPU. This work aims to elucidate the influence of soft-segment molecular weight on the interfacial structure, mechanical properties, and shape-memory behavior of the composites. Furthermore, the practical applicability of the optimized material is demonstrated through the successful injection molding of prototype spoons, highlighting its commercial potential for biodegradable products.
Materials and methods
Experimental materials
Polylactic Acid (PLA, 4032D, Industrial grade) was supplied by Shanghai Tongjie Liang Biomedical Materials Co, Ltd (Shanghai, China). Mung bean starch (MBS, 200 mesh, Food grade) was purchase from Henan Xinliang Grain and Oil Processing Limited Liability Company. 4, 4′-methylenedi-p-phenyldiisocyanate (MDI, 98%, industrial grade, Sigma) was supplied by Anhui Zesheng Technology Co., Ltd (Shanghai, China). Polycaprolactone diol (PCL diol, M w = 500, 1000, 2000, 4000 g•mol−1, hydroxyl value = 57 mg•KOH•g−1) was kindly provided by Perstorp UK Ltd (Cheshire, UK).
Experimental methods
Synthesis of PCL-based polyurethane prepolymer (PCLPU)
The PCL-based polyurethane prepolymer (PCLPU) with varying soft segment molecular weights was synthesized via a two-step reaction. To ensure the prepolymer chain ends were functionalized with isocyanate groups, PCL diol was reacted with a stoichiometric excess of 4,4′-diphenylmethane diisocyanate (MDI) at a molar ratio of n-OH: n-NCO = 1:2. The overall reaction scheme is illustrated in Figure 1, with comprehensive synthetic details available in the Supporting Information. The resulting PCLPUs, synthesized from PCL diols with molecular weights of 500, 1000, 2000, and 4000 g·mol−1, were designated as PCLPU-500, PCLPU-1000, PCLPU-2000, and PCLPU-4000, respectively. The overall reaction scheme of PCLPU.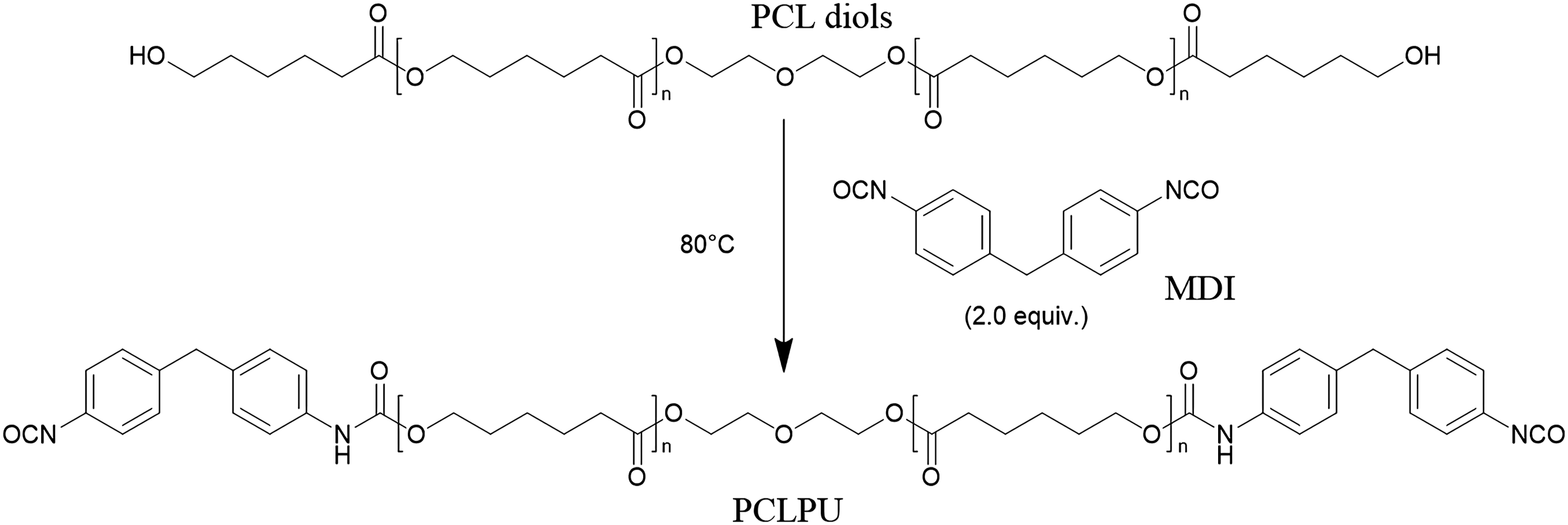
Synthesis of non-reactive polyurethane (NPCLPU)
Polycaprolactone-based polyurethane prepolymer (PCLPU-2000, 20 g) was dissolved in 120 mL of acetone in a beaker. After complete dissolution, 20 mL of anhydrous ethanol was introduced, and the reaction was allowed to proceed at 45°C for 30 minutes to cap the terminal isocyanate groups. The product was then precipitated by adding the reaction solution into 1000 mL of anhydrous ethanol, which resulted in the formation of a white solid. The mixture was left to stand for 24 hours, after which the precipitate was collected by filtration. The obtained solid was vacuum-dried at 90°C for 12 h to yield the final product, defined as NPCLPU-2000, which is theoretically devoid of active -NCO groups. (Figure 2). The overall reaction scheme of NPCLPU.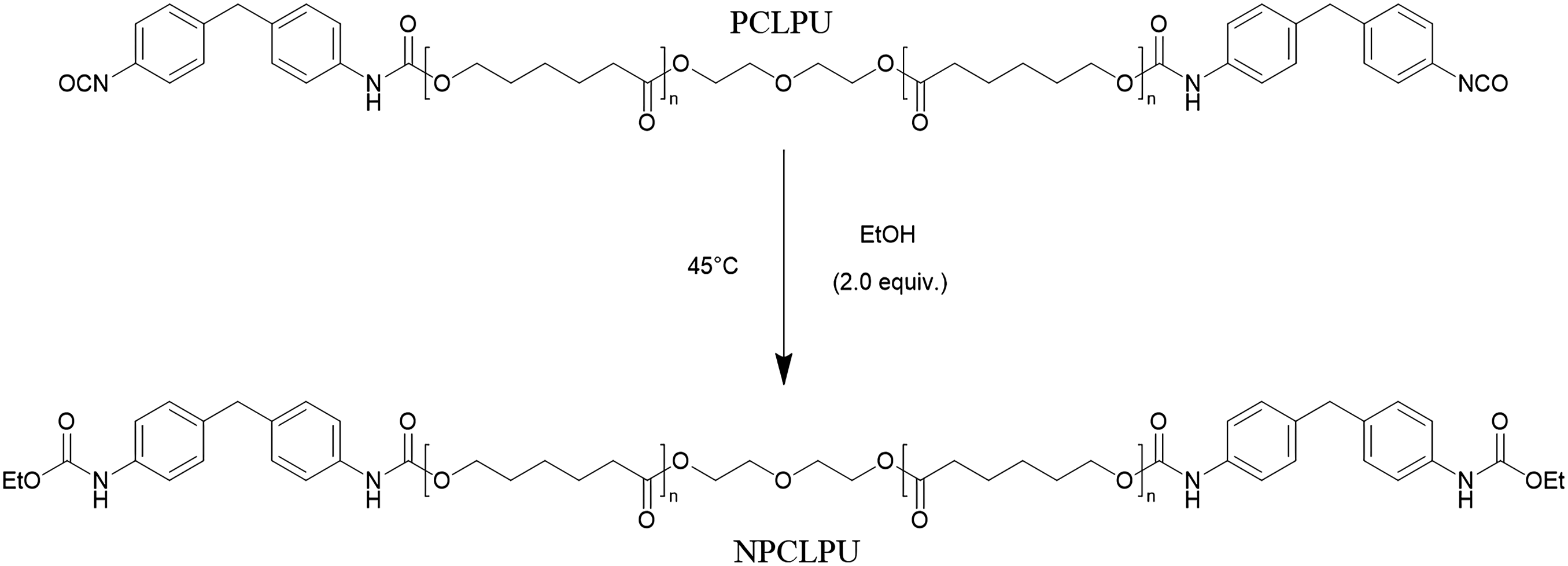
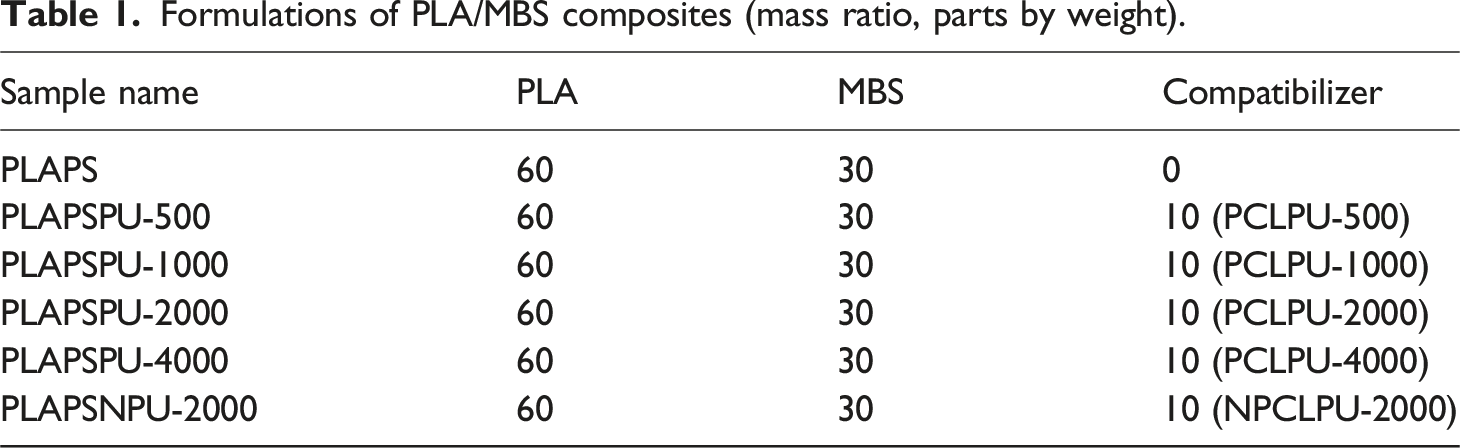
Preparation of composites with different soft segments
Formulations of PLA/MBS composites (mass ratio, parts by weight).
Injection molding process
Standardized specimens for mechanical testing were injection-molded using a Qixin M1400 system. The process was conducted at a barrel temperature of 180°C, with a mold closing time of 3 s, an injection time of 15 s, and a cooling time of 3 s.
Characterization
Fourier transform infrared (FTIR) spectroscopy
The chemical structures of PCLPU, MBS, and a series of composite materials were characterized by Fourier transform infrared (AVATAR 360E.S.P, MA, USA) with test wavelengths in the range of 500 ∼ 4000 cm−1.
Scanning electron microscope (SEM)
The interfacial morphology of composite materials was systematically investigated using a scanning electron microscope (Quanta 200 FEG SEM, FEI Netherlands) under optimized parameters (15 kV accelerating voltage, 10 mm working distance).
Nuclear magnetic resonance (1H NMR) spectroscopy
The molecular structures of MDI, PCL diol, and the synthesized PCLPU were characterized by 1H NMR spectroscopy. Each sample was dissolved in dimethyl sulfoxide-d6 (DMSO-d6) to prepare a solution with an appropriate concentration. The 1H NMR spectra were then acquired using a nuclear magnetic resonance spectrometer.
Mechanical properties
Tensile test
Tensile properties, including tensile strength, elastic modulus, and elongation at break, were measured using a CMT6503 universal testing machine (Shenzhen Gaopin Test Machine Co., Ltd, China). The test was conducted on dumbbell-shaped specimens at a crosshead speed of 10 mm/min, conforming to the GB/T 1040.2-2006 standard.
Impact test
Impact strength was determined according to GB/T 1043-93-A (equivalent to ASTM D256) using a ZBC1400-1 impact tester (Shenzhen Xinsansi Measurement Co., Ltd, China) equipped with a 7.5 J hammer. The calculated impact strength (σk) was derived using the following equation (1):
Flexural property
The flexural properties of the composites were evaluated using a universal testing machine (CMT6503, Shenzhen Gaopin Testing Equipment Co., Ltd, China) in accordance with the GB/T 9341-2008 standard. Rectangular specimens were tested in a three-point bending mode with a support span of 64 mm and a crosshead speed of 2.00 mm·min−1. The resulting data were subsequently analyzed for statistical significance.
Melt flow rate
The melt flow rate (MFR) of the composites was measured using a melt flow indexer (Model GC-MI-BP, Dongguan Guangce Automation Equipment Co., Ltd, China). The test was conducted at 190°C under a total load of 2160 g (including a 325 g plunger and 1835 g of NIST-traceable standard weights). Following a 4-min preheating period, eight consecutive melt segments were collected at 10-s intervals, with the initial unstable flow portion being discarded. The melt flow rate was then calculated using equation (2):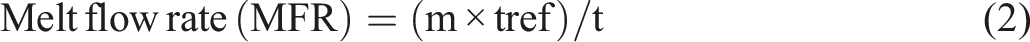
Water absorption
The water absorption behavior of the composite materials was investigated in accordance with ASTM D570. Five specimens (dimensions: 20 × 10 × 4 mm3) were first dried at 110°C for 24 h to obtain their initial dry mass (m
0
). Subsequently, the samples were immersed in distilled water at a constant temperature of 23 ± 2°C. The mass (m
t
) of each specimen was measured at 24-h intervals for a total duration of 6 days. The water absorption percentage was then calculated using equation (3):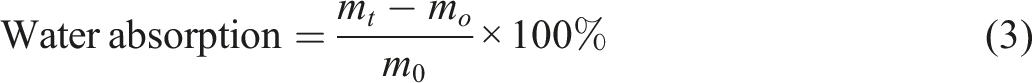
Injection molding and comparative evaluation of PLA/MBS composites
To assess the practical processability and application potential of the developed composite, the PLAPSPU-500 formulation was processed into sample spoons using an injection molding machine. For comparison, spoons were also molded from neat PLA under identical processing conditions. The barrel temperature was set at 180°C, with a mold closing time of 3 s, an injection time of 20 s, and a cooling time of 3 s. Three replicate spoons were fabricated for each material. The performance of the composite and neat PLA spoons was subsequently compared through a series of observational and quantitative measurements to evaluate the potential of the composite for practical applications.
Results and discussion
FTIR spectroscopy analysis
As shown in Figure 3(a), the FTIR spectra of PCLPU with different soft segment molecular weights exhibit similar characteristic peaks. The peak at 3340 cm−1 corresponds to the N–H stretching vibration. Together with the carbonyl stretching vibration at 1730 cm−1, this indicates that MDI has reacted with PCL diol to form urethane bonds (–NH–COO–). The absorption at 2270 cm−1 is attributed to the stretching vibration of unreacted isocyanate groups (–NCO), suggesting residual isocyanate in the product and further confirming the reaction mechanism. These results are consistent with the FTIR data reported by Dai et al.
19
for waterborne polyurethane. The presence of –NCO peaks confirms the successful synthesis of PCLPU bearing active isocyanate groups. In contrast, the absence of the peak at 2270 cm−1 in the spectrum of NPCLPU-2000 indicates complete consumption of –NCO groups, demonstrating that NPCLPU-2000 is no longer reactive toward hydroxyl groups. According to Figure 3(b), the FTIR profiles of samples with different soft segment molecular weights are nearly identical. Therefore, representative spectra are compared in Figure 3(c), it can be observed that the characteristic –NCO peak at 2270 cm−1 present in PCLPU-2000, is absent in both NPCLPU-2000 and PLAPSPU-2000. This result confirms the successful preparation of NPCLPU and suggests that during melt blending, the isocyanate groups react with hydroxyl groups from starch and PLA, forming new chemical linkages. This behavior aligns with the findings reported by Weng et al.
20
FTIR of (a) PCLPU synthesized with different soft segment molecular weights, (b) PLA composite materials compatibilized with different PCLPU, (c) composite materials.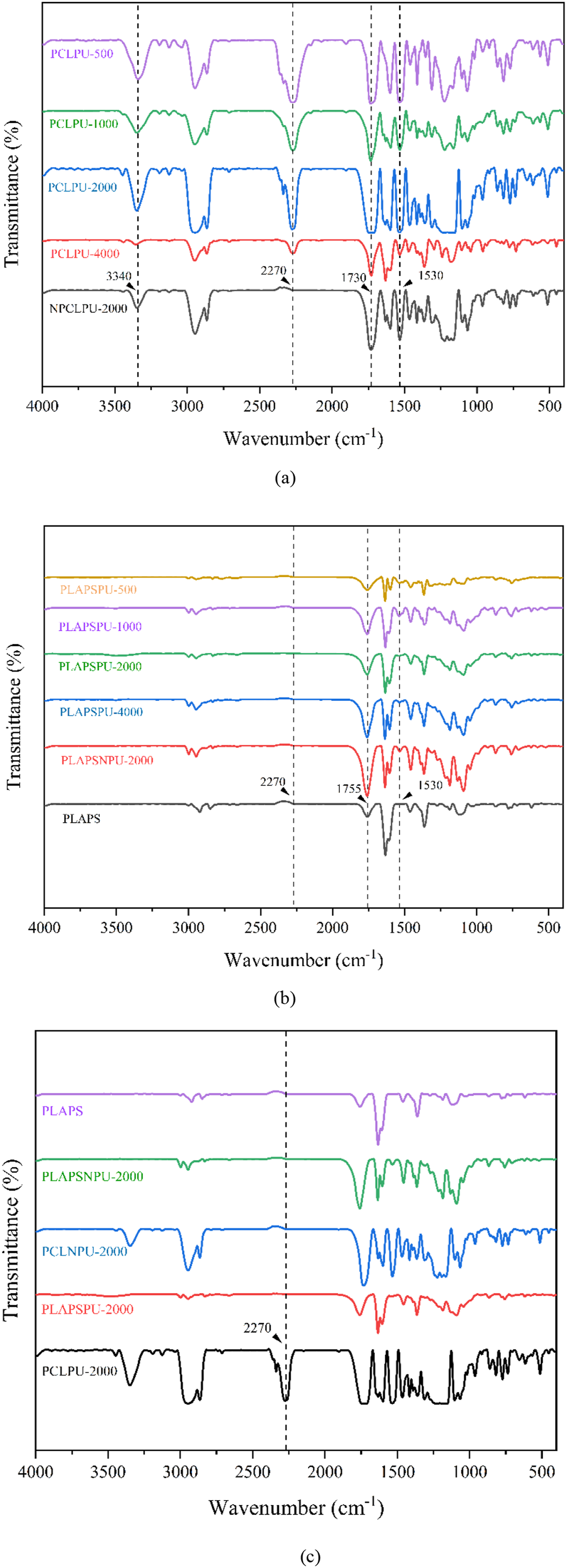
Analysis of 1H NMR spectra
The 1H NMR spectrum of PCLPU (Figure 4) confirms its chemical structure. The characteristic signals observed at δ 7.04–7.36 ppm are attributed to the aromatic protons of the MDI phenyl ring. A weak signal at approximately δ 6.90 ppm corresponds to the N–H proton of the urethane group. Multiplets in the range of δ 4.0–4.1 ppm are assigned to the methylene protons (–O–CH2–) adjacent to the oxygen atom, while the triplet at δ 2.20–2.35 ppm arises from the methylene protons (–CH2–C=O) alpha to the carbonyl group. The presence of characteristic peaks associated with both the MDI phenyl ring and the PCL diol ester segments, along with the appearance of the urethane-related N–H signal, indicates the successful reaction between PCL diol and MDI to form PCLPU. These findings are consistent with the FTIR results and align with the NMR data reported by Tang et al.
21
1H NMR spectra of PCLPU-2000.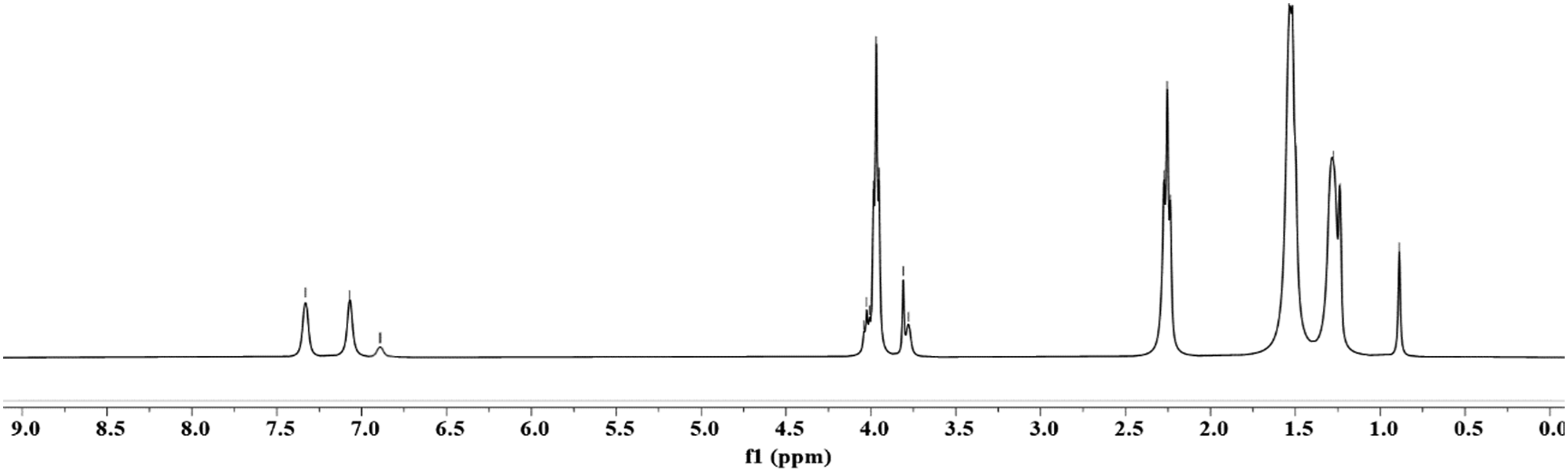
Microscopic morphology analysis of composite
Figure 5 compares the microstructural morphologies of PLA composites with and without the PCLPU compatibilizer. As shown in Figure 5(a), the fracture surface of the composite without PCLPU (PLAPS) exhibits typical brittle fracture characteristics, featuring numerous grooves, pores, and granular structures, which can be attributed to the rigid ester-based chains of PLA.
22
A distinct interfacial gap between starch granules and the PLA matrix is observed, consistent with the findings reported by Kim et al.
23
In contrast, the cross-section of the PCLPU-modified composite (PLAPSPU-2000, Figure 5(b)) appears relatively smooth, with almost no exposed starch granules. This suggests that the isocyanate groups in PCLPU react with the hydroxyl groups of starch, forming urethane linkages that chemically cross-link the interface. As a result, a cross-linked layer encapsulates the starch and connects it to the PLA matrix. This layer acts as a physical “mediator,” enhancing interfacial adhesion and improving the overall compatibility between the two phases. These results underscore the effectiveness of the chemical compatibilization strategy in enhancing the microstructure and performance of the composite. SEM image of (a1) PLAPS (1000×), (a2) PLAPS (3000×), (b1) PLAPSPU-2000 (1000×), (b2) PLAPSPU-2000 (3000×).
Mechanical properties of the composites
Mechanical property test data.
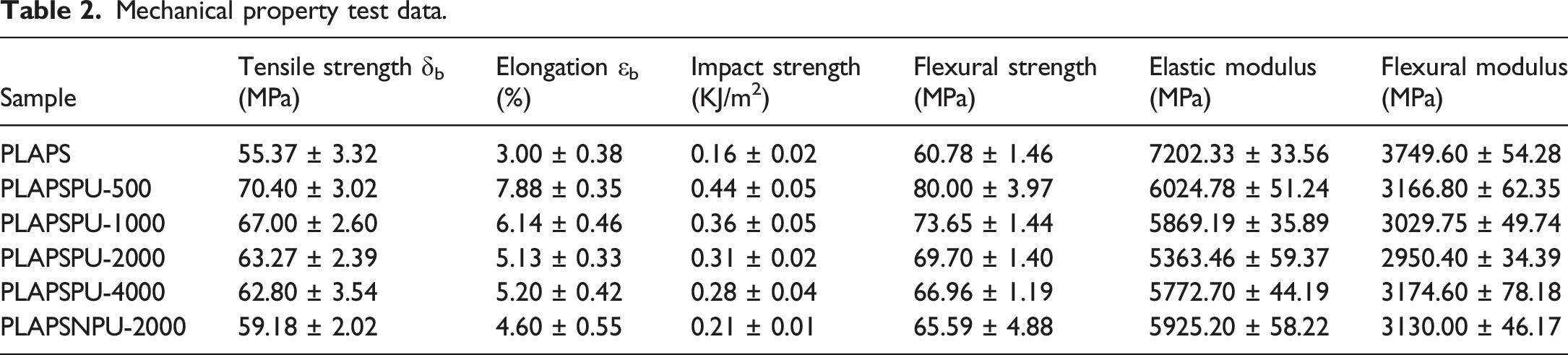
As illustrated in Figure 6, PCLPU-500 imparted the most substantial reinforcement and toughening effect among all the compatibilizers tested. Compared to the uncompatibilized PLAPS, the composite with PCLPU-500 exhibited a 27.14% increase in tensile strength, a 162.67% increase in elongation at break, a 175.00% increase in impact strength, and a 31.62% increase in flexural strength, demonstrating a comprehensive enhancement of mechanical performance, reflecting an optimal balance between stiffness and toughness. Furthermore, the data indicate that the mechanical properties of the composites are influenced by the soft-segment molecular weight of PCLPU. A lower soft-segment molecular weight corresponds to a higher concentration of isocyanate groups per unit mass, leading to more extensive chemical cross-linking. Consequently, PCLPU with a lower molecular weight, such as PCLPU-500, results in superior composite properties, a trend that is consistent with the findings of Iskender et al.
25
Histogram of (a) tensile properties, (b) impact and bending properties, (c) stress-strain curves of the composites.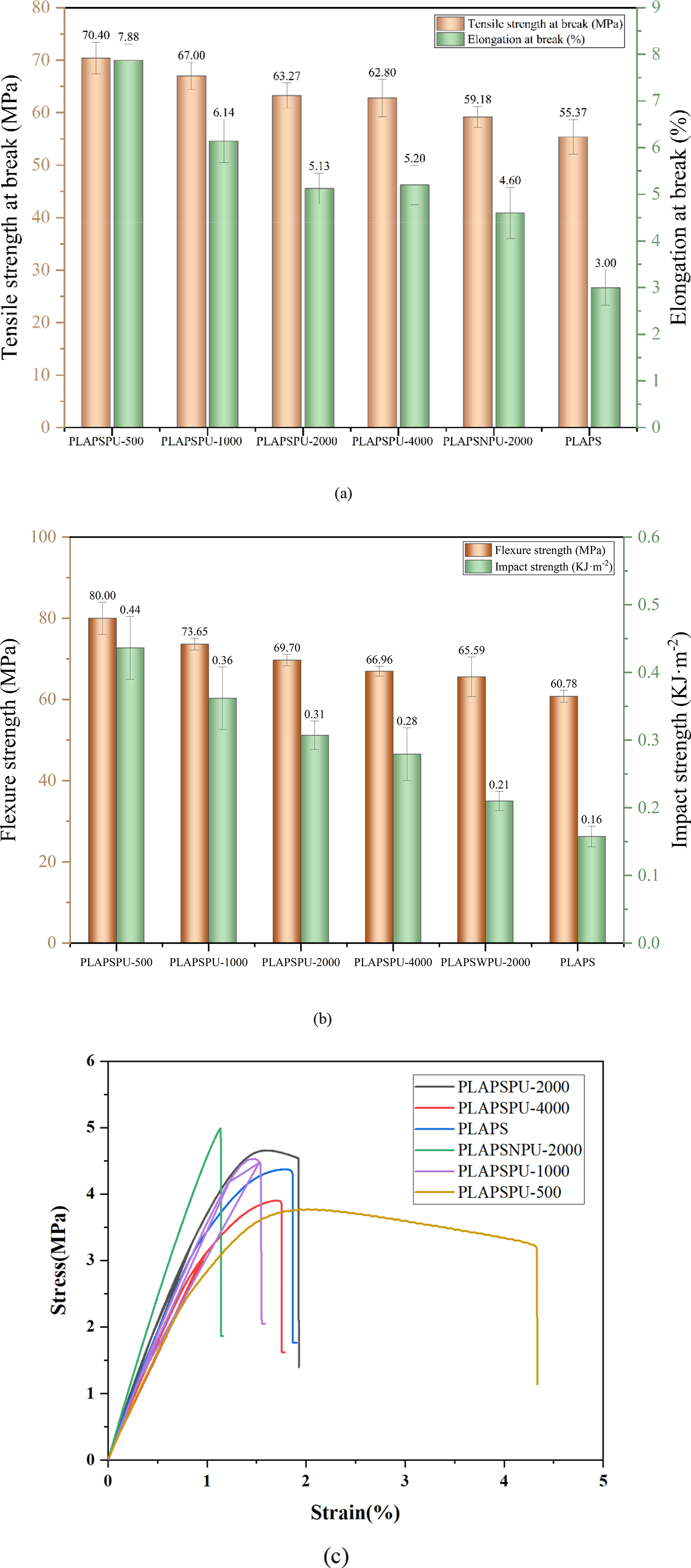
Melt flow index analysis
Melt flow index data.
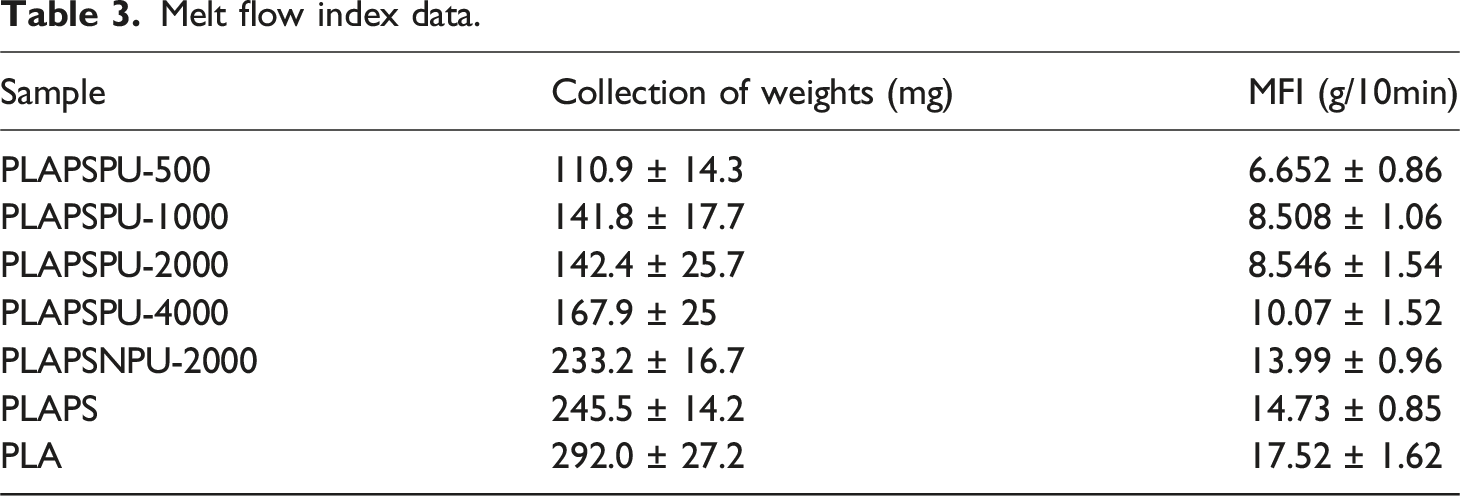
Additionally, the MFI values vary with the soft-segment molecular weight of PCLPU. Composites with lower soft-segment molecular weight (e.g., PLAPSPU-500) show lower MFI, indicating poorer melt fluidity. This trend is consistent with a higher concentration of isocyanate groups per unit mass, leading to more extensive chemical cross-linking and stronger network formation. These results corroborate the previous mechanical and structural analyses, confirming that PCLPU with lower soft-segment molecular weight promotes a more robust cross-linked structure and yields composites with superior performance. 27
Water absorption behavior
Figure 7 illustrates the water absorption behavior of the prepared composites over time. As water uptake is a prerequisite for initiating enzymatic degradation in biodegradable plastics, the water absorption rate and hydrophilicity are key indicators of a material’s degradability.
28
Generally, higher water absorption capacity correlates with faster degradation rates. As shown in Figure 7, the water absorption curves begin to plateau after 216 hours of immersion. The uncompatibilized composite (PLAPS) demonstrates the most rapid moisture uptake and the highest equilibrium water absorption. This is attributed to its poor interfacial adhesion, which leaves numerous starch granules exposed and readily accessible to water. The incorporation of deactivated PCLPU (PLAPSNPU-2000) reduces the water absorption compared to PLAPS, indicating that physical cross-linking promoted by NPCLPU leads to a more coherent structure and better encapsulation of starch. A further significant reduction in water uptake is observed for the composite containing reactive PCLPU (PLAPSPU-2000). This enhanced moisture resistance stems from the chemical cross-links formed by the isocyanate groups, which create a denser network that more effectively shields the hydrophilic starch from water. Water absorption rate of composite.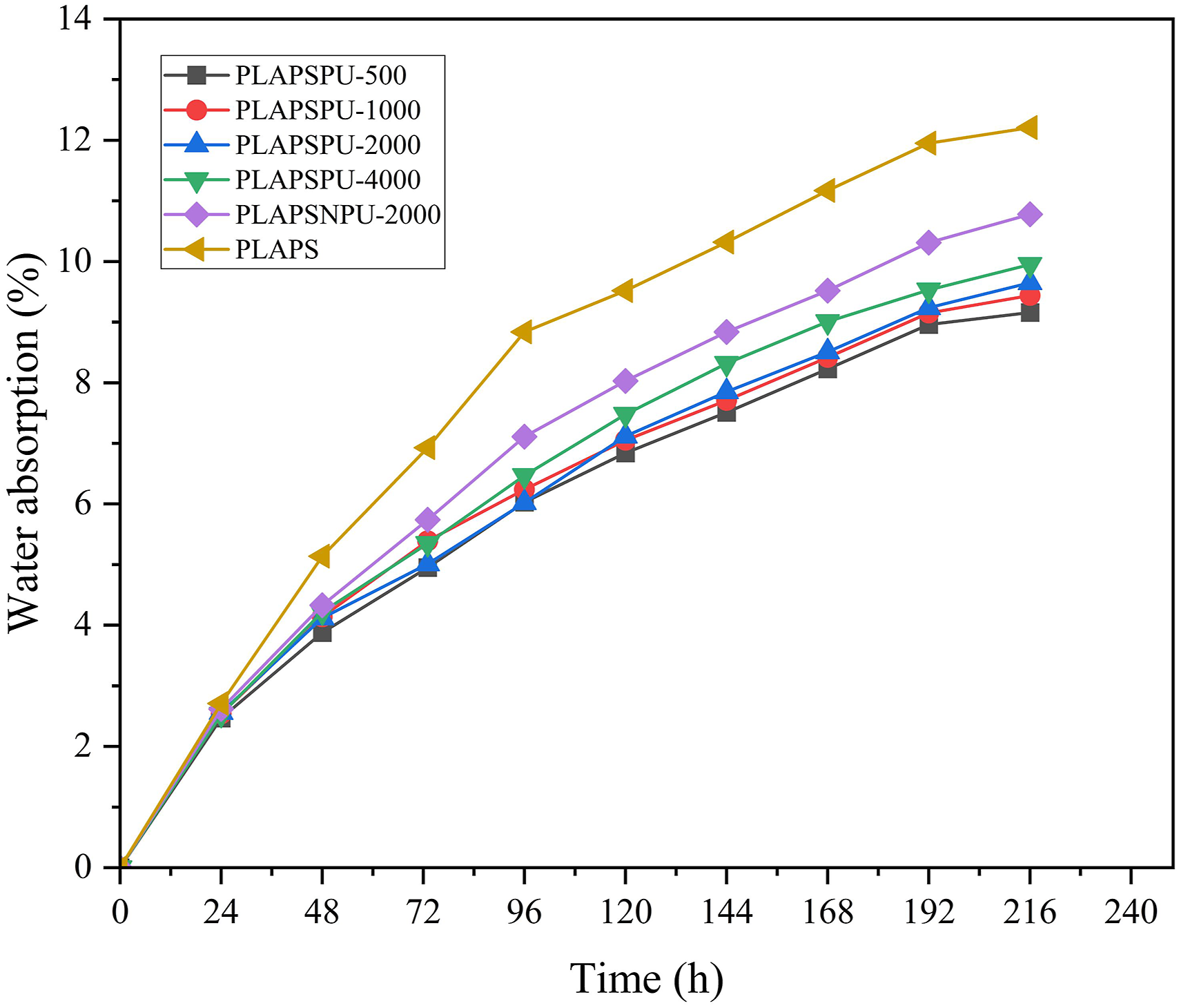
While the influence of PCLPU’s soft-segment molecular weight on water absorption is less pronounced, the overall trend confirms that the compatibilizer contributes through a combination of physical and chemical cross-linking mechanisms. 29 These findings are consistent with the conclusions drawn from the mechanical and melt-flow analyses.
Formation of interface structure
Figure 8 schematically illustrates the proposed interfacial compatibilization mechanism, which is supported by multi-scale experimental evidence. FTIR and 1H NMR confirm the reaction between terminal –NCO groups of PCLPU and hydroxyl groups from starch, forming covalent urethane linkages. SEM reveals a clear morphological transition: the uncompatibilized PLA/MBS exhibits distinct interfacial gaps and exposed starch granules, whereas PCLPU-modified composites show a smooth, continuous fracture surface with starch granules tightly encapsulated. These chemical and morphological improvements directly translate into enhanced mechanical properties, lower MFI, and reduced water uptake (Tables 2 and 3, Figures 6 and 7). The effect strongly correlates with the soft-segment molecular weight of PCLPU: lower molecular weight (e.g., 500 g·mol−1) provides higher –NCO density, enabling more extensive chemical cross-linking and stronger interfacial adhesion. Thus, the compatibilization mechanism combines dominant chemical cross-linking with secondary physical entanglement, forming a “many-to-many” interfacial network that bridges hydrophilic starch and hydrophobic PLA across length scales from covalent bonds (∼0.1 nm) to starch granules (∼20 μm). Schematic illustration of the proposed interfacial compatibilization mechanism in PLA/MBS/PCLPU composites.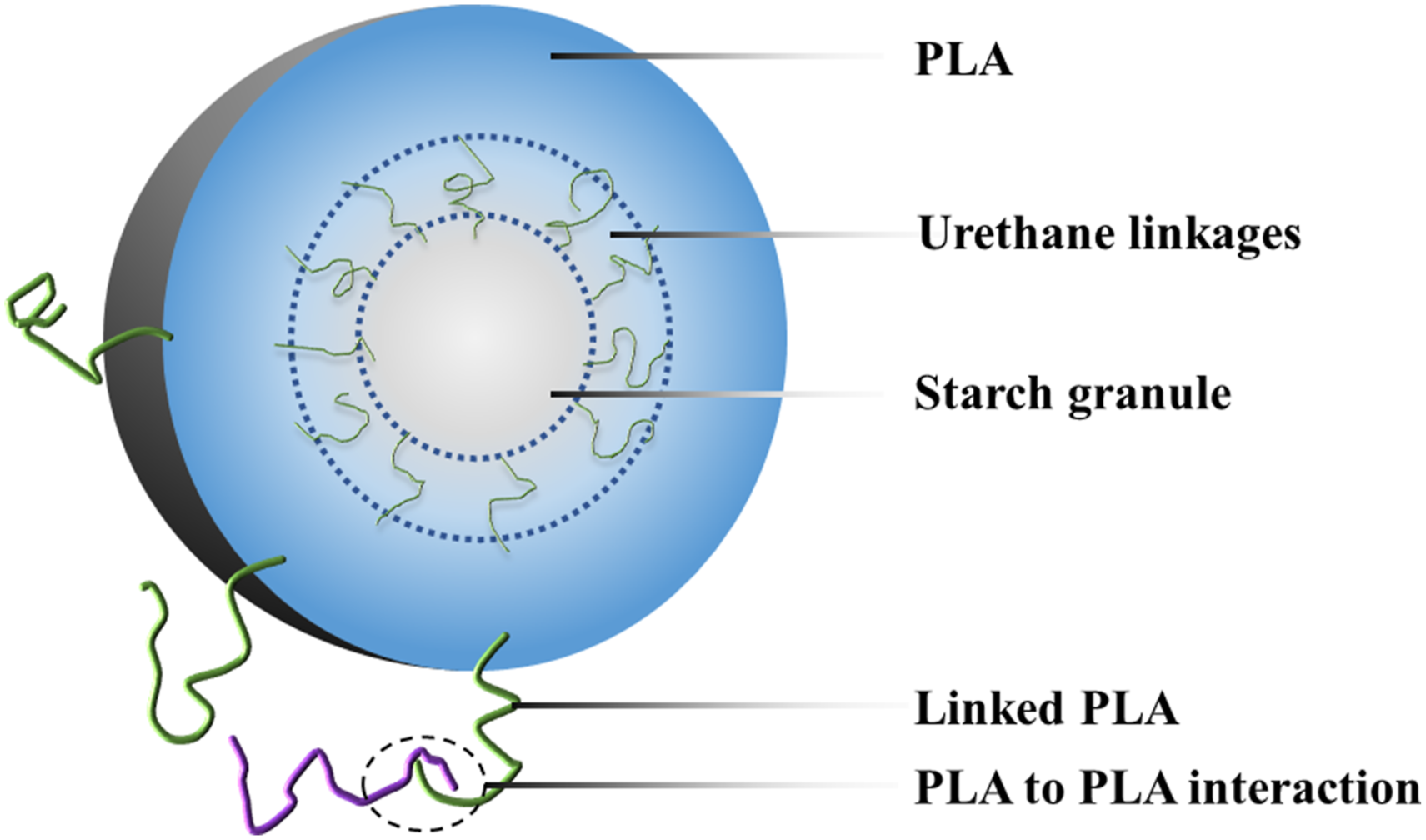
Conclusion
In this study, a series of polycaprolactone-based polyurethane prepolymers (PCLPU) with varying soft‐segment molecular weights were synthesized and employed as interfacial compatibilizers for PLA/mung bean starch (MBS) composites prepared via melt blending. It was demonstrated that the interfacial compatibility was significantly enhanced through a dual mechanism involving both physical interactions and chemical cross-linking, where a lower soft-segment molecular weight of PCLPU provided a higher concentration of reactive isocyanate groups, leading to more extensive chemical cross-linking and superior mechanical properties. The optimal composite containing PCLPU-500 exhibited remarkable improvements, with increases of 27.14% in tensile strength, 162.67% in elongation at break, 175.00% in impact strength, and 31.62% in flexural strength compared to the uncompatibilized composite (PLAPS). Although this work primarily demonstrated the concept through laboratory-scale prototypes, the findings clearly establish the feasibility of developing high-performance, bio-based composites with enhanced interfacial properties and mechanical performance, showing promising potential for sustainable plastic applications.
Footnotes
Acknowledgements
We gratefully acknowledge the Program of Introducing Talents of Discipline to the Universities of China (Award Number: B17019), the Hubei Key Laboratory of Novel Reactor and Green Chemical Technology, School of Chemical Engineering and Pharmacy, Wuhan Institute of Technology (Grant No. NRG202405), and China Scholarship Council (File No. 202508420213).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We gratefully acknowledge the Program of Introducing Talents of Discipline to the Universities of China (Award Number: B17019), the Hubei Key Laboratory of Novel Reactor and Green Chemical Technology, School of Chemical Engineering and Pharmacy, Wuhan Institute of Technology (Grant No. NRG202405), and China Scholarship Council (File No. 202508420213).
Data Availability Statement
The data sets generated and analyzed during the current study are available from the corresponding author upon reasonable request.
