Abstract
Advanced membrane technologies with enhanced antifouling resistance and long-term stability are crucial for addressing the escalating issues of water pollution and resource scarcity. In this work, silver (Ag), copper (Cu), and iron (Fe) nanoparticles were systematically incorporated over a polysulfone (PSF) membrane via in situ reduction within a polyethyleneimine (PEI) layer, followed by interfacial polymerization using trimesoyl chloride (TMC). Unlike previous studies that evaluate individual metals in different polymer systems, this study provides a direct comparative assessment of Ag, Cu, and Fe within the same PSF–PEI–TMC composite, enabling clear evaluation of metal-specific effects on membrane performance and stability. Nanoparticle incorporation significantly modified surface wettability, pore structure, and roughness relative to pristine PSF. Iron-embedded membranes exhibited the highest pure water permeance (up to 276.46 LMH/Bar), representing more than a twofold increase compared to PSF; however, they showed the poorest antifouling performance due to strong protein–metal interactions. Silver-modified membrane achieved the highest COD rejection (17.44%) among all but suffered from substantial nanoparticle leaching (up to 370.4 µg/m2h), limiting their long-term applicability. In contrast, copper-embedded membranes demonstrated a balanced performance, demonstrating an antifouling performance with flux recovery ratio of 82.02% with negligible metal leaching while maintaining enhanced permeability compared to pristine PSF. These findings demonstrate that although Ag improves rejection and Fe enhances flux, in situ copper nanomaterial provides the optimal performance between permeability, fouling resistance, and environmental stability.
Highlights
• In situ formation of nanomaterial by reduction in the top layer of the membrane • Variation in surface morphology and performance by change in reducing agent concentration • Embedding nanoparticles increased hydrophilicity, altered the pore sizes, and surface roughness compared to the PSF membrane. • Silver-embedded membranes exhibited the highest COD rejection (17.44%) among the membranes • Highest leaching of nanomaterial was observed in Silver-embedded membrane (up to 370.4 µg/m2h) • Iron-embedded membranes had the highest flux (276.46 LMH/Bar) • Copper-embedded membrane had promising antifouling performance with FRR 82.02% and no significant leaching.
Introduction
According to the United Nations (UN), nearly 4 billion people worldwide face a water crisis by 2025. 1 The severity of this issue is expected to worsen even further in the years to come because of population expansion, climate change, and ineffective water management techniques. 2 The need for energy-efficient wastewater treatment technologies is necessary to guarantee sustainable water management, reducing operational costs, minimizing greenhouse gas emissions, and tackling the worldwide issues of water scarcity and climate change. 3
The membrane separations technology has grown much in recent years due to its high potential in operational simplicity, eco-friendliness (low carbon footprint), low-energy usage, selective separation and scalability.4,5 However, since it is being used in a wider range of applications, these membranes also present significant fouling threats because of the adherence of organic debris, inorganic salts, and microorganisms that might impair performance efficiency. 6 Therefore, researchers have coated/modified the membranes to improve thermal resistance, hydrophilicity, chemical abrasion, anticorrosion and longevity.7–11 Polyethylene Glycol (PEG), Polydopamine, Polyvinyl Alcohol (PVA), and many other organic-inorganic substances have been used for coating.
Polysulfone (PSF) membranes are hydrophobic in nature and have high mechanical, thermal, and chemical stability while being inexpensive.12,13 These properties of PSF membrane make it a robust candidate for application as the base layer for desalination membranes. 14 Polysulfone, being hydrophobic in nature, is susceptible to fouling and will deteriorate its performance in high fouling environment because of its hydrophobicity. 15 Thus, it is essential to modify its surface with suitable particles or functional groups to increase its hydrophilicity. 16
Embedding nanoparticles can improve the hydrophilic nature, antimicrobial performance, fouling resistance and mechanical properties of membranes significantly. Such membranes are more efficient and robust for the wastewater treatment applications, where the propensity to fouling is higher. Many nanoparticles, like Ag, Cu, Fe, Zn, Ti, and many others, are commonly used. The ability to modify the surface characteristics of polymer membranes to achieve desired performance without compromising permeability and selectivity is one of the key advantages of adding nanoparticles. 17 Silver nanoparticles have optical, electrical, catalytic, and antimicrobial properties; especially antimicrobial property makes silver nanoparticles widely used in nanocomposite membranes. 18 Copper nanoparticles have excellent hydrophilic and antimicrobial characteristics.19,20 Iron (Fe) nanoparticles have magnetic properties and potential for catalytic degradation of organics.21,22 It has been reported that nanomaterials like TiO2, CNTs, and BiVO4 can be utilized to increase PVDF membranes’ resistance to fouling in dairy wastewater treatment applications. 23
Comparison of this work with other reported works
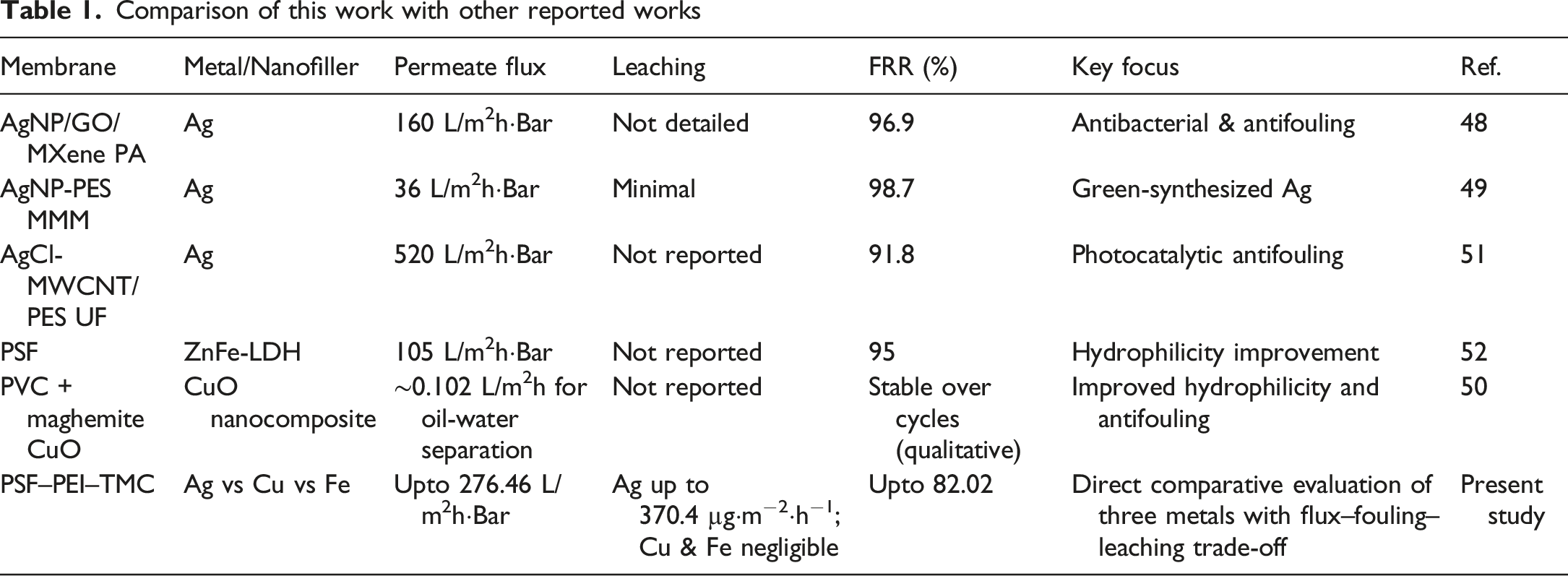
Recent studies attempted to discover more stable and antifouling polymeric membranes that can work for prolonged periods, without significant leaching. 29 Numerous publications have studied on most commonly used Ag, Cu, and Fe nanoparticles, but a comparative study of these particles in the same polymer matrix is - a research gap, which is being addressed in this paper.
Individual metal-modified PSF membranes are widely reported, a systematic comparison of Ag, Cu, and Fe nanoparticles fabricated via the same PEI–TMC interfacial polymerization and in situ reduction route has not been extensively explored. The variation in the performances of the modified membranes with the change in concentrations of the reducing agent and the physical and chemical changes, particularly with respect to leaching stability and fouling behavior under identical operating conditions are studied. Thus, the present study demonstrates the fabrication of nanocomposite membranes with in situ formation of metal nanoparticles, resulting in enhanced permeance suitable for high-flux ultrafiltration applications.
Materials
Polysulfone pellets (Solvay), Nonwoven technical fabric- Polyester (AWA paper), Dimethyl Formamide (Central Drug House (P) Ltd), Polyethyleneimine (Tokyo Chemicals Industry Co. Ltd) Sodium Borohydride (Central Drug House (P) Ltd), n-Hexane (Loba Chemie Pvt. Ltd), 1,3,5-Benzenetricarbonyl Trichloride (Tokyo Chemical Industry Co. Ltd), Silver Sulphate (Qualigens Fine Chemicals), Copper Sulfate pentahydrate (Central Drug House (P) Ltd), Iron Sulphate heptahydrate (Thermo Fisher Scientific India Pvt. Ltd), Egg Albumin (Sisco Research laboratory Pvt. Limited) and deionized water.
Methods
Polysulfone membrane preparation
A non-solvent-induced phase inversion method was used to create the polysulfone (PSF) membrane. PSF pellets were dried at 70°C until all of the moisture was removed completely. These dried PSF pellets were slowly added to N, N′ Dimethylformamide (DMF), which was preheated at 60°C with continuous stirring to prepare a 14% (w/v) solution. The membrane casting machine was set up by rolling a polyester non-woven fabric over its rollers. A precision knife with a desired gap was positioned over the roller to maintain the thickness of the cast membrane. Deionized water was used as the non-solvent in a gelation bath, while maintaining a linear speed of 2 m/min for the rollers. The prepared membrane was used as the base membrane for further coating.
Coating of a layer with in situ formed nanoparticles
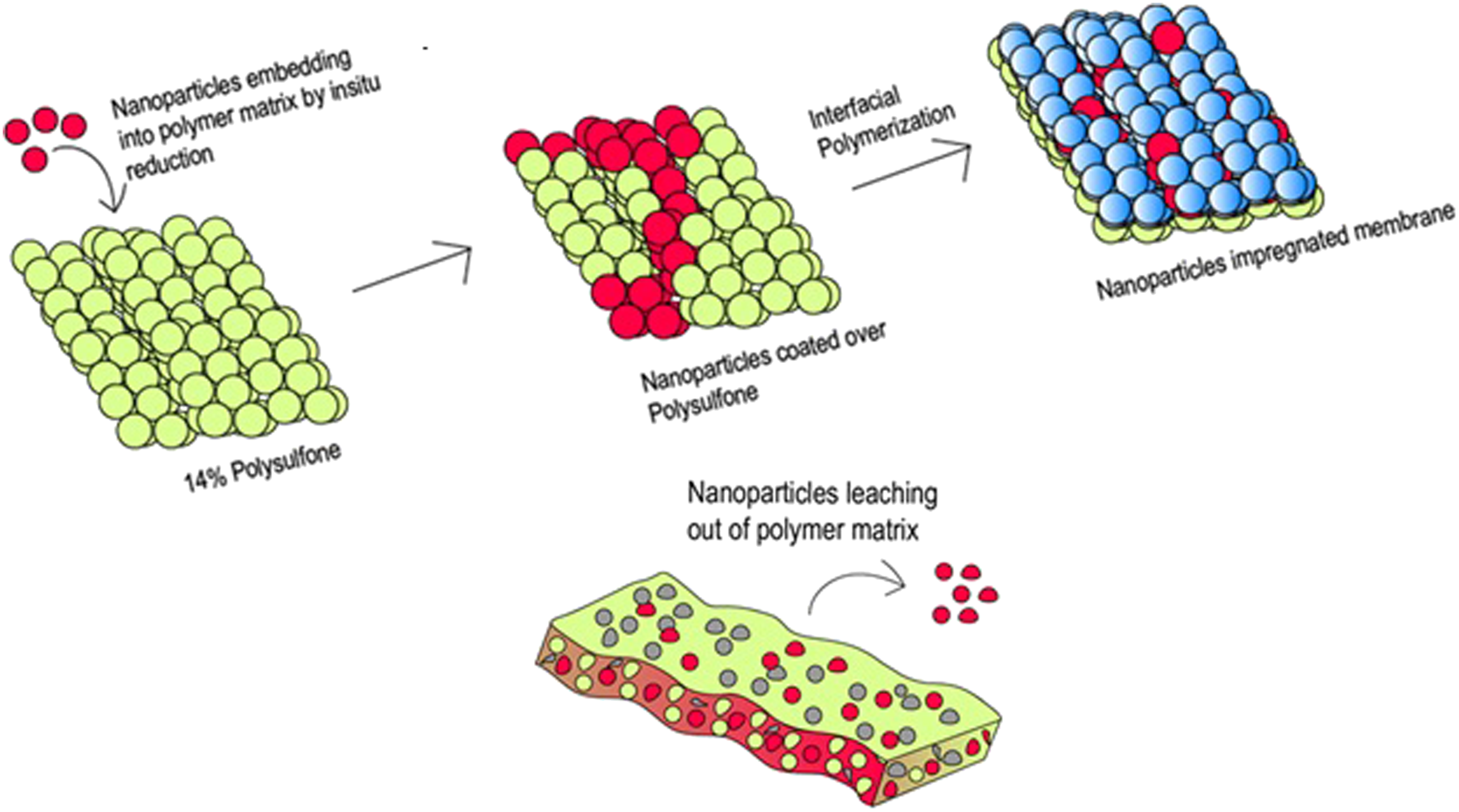
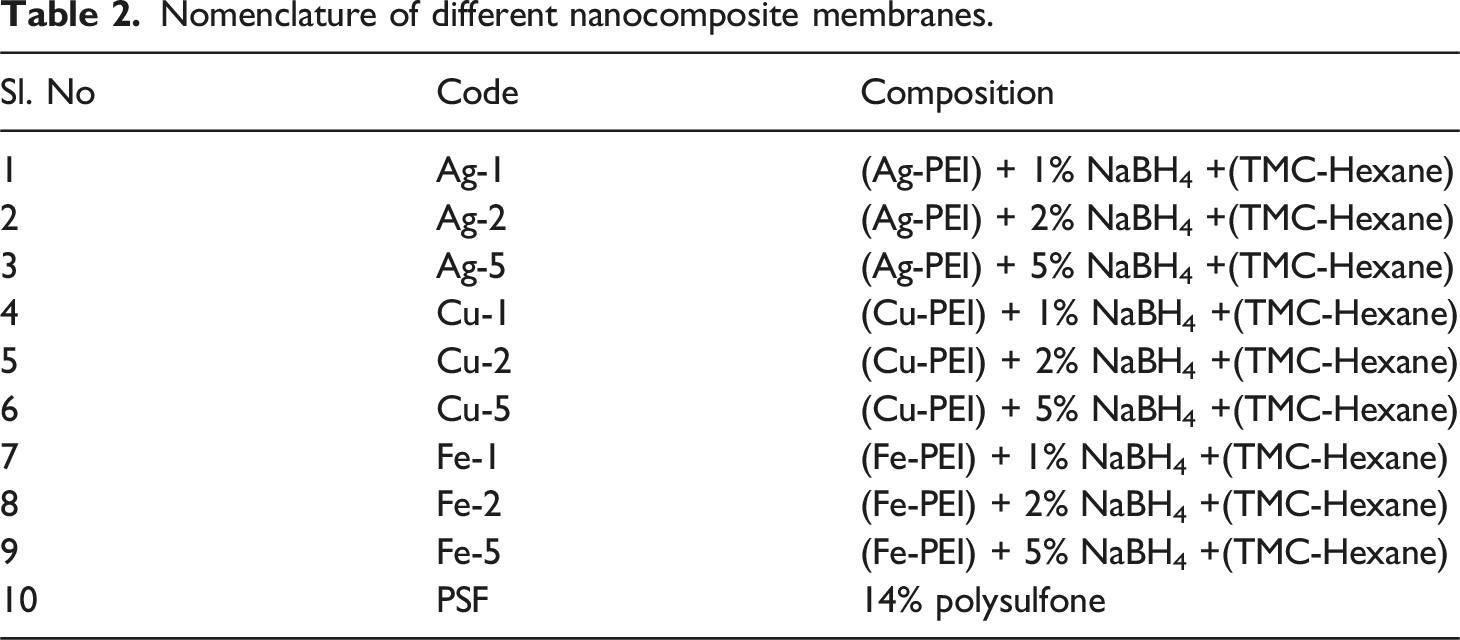
To prepare (Ag, Fe, Cu) nanoparticle-coated PSF membranes, 25 mmol of metal salt (Ag2SO4/FeSO4/CuSO4) was dissolved in deionized water, then 1.5% (v/v) polyethyleneimine (PEI) was added with thorough mixing. Sodium borohydride (NaBH4) solutions at 1%, 2%, and 5% (w/v) were prepared separately in DI water for each solution. The NaBH4 reduces the nanoparticle –PEI solution to produce nanoparticles. Simultaneously, Tri-mesoyl chloride (TMC) was liquefied by placing it in a water bath at 50°C, after which 720 µL of the liquefied TMC was dissolved in 600 mL of hexane. Three 14% polysulfone (PSF) membranes were then coated with 200 mL of the nanoparticle-PEI aqueous solution for 10 min. After draining out the excess solution, NaBH4 solution was added to different membranes and allowed to react for 10 min to reduce metal salt, forming nanoparticles stabilized by PEI. Following this reduction process, the NaBH4 solutions were drained, and the TMC-hexane solution was applied to each membrane surface to facilitate interfacial polymerization for 5 min, completing the membrane preparation process. The membranes were dried at 60°C after draining out the excess solution. Figure 1 shows the schematic of the experimental process followed to coat the nanoparticleimpregnated top layer. Table 2 shows the nomenclature of different nanocomposite membranes. Preparation of nanoparticle-impregnated membranes. Nomenclature of different nanocomposite membranes.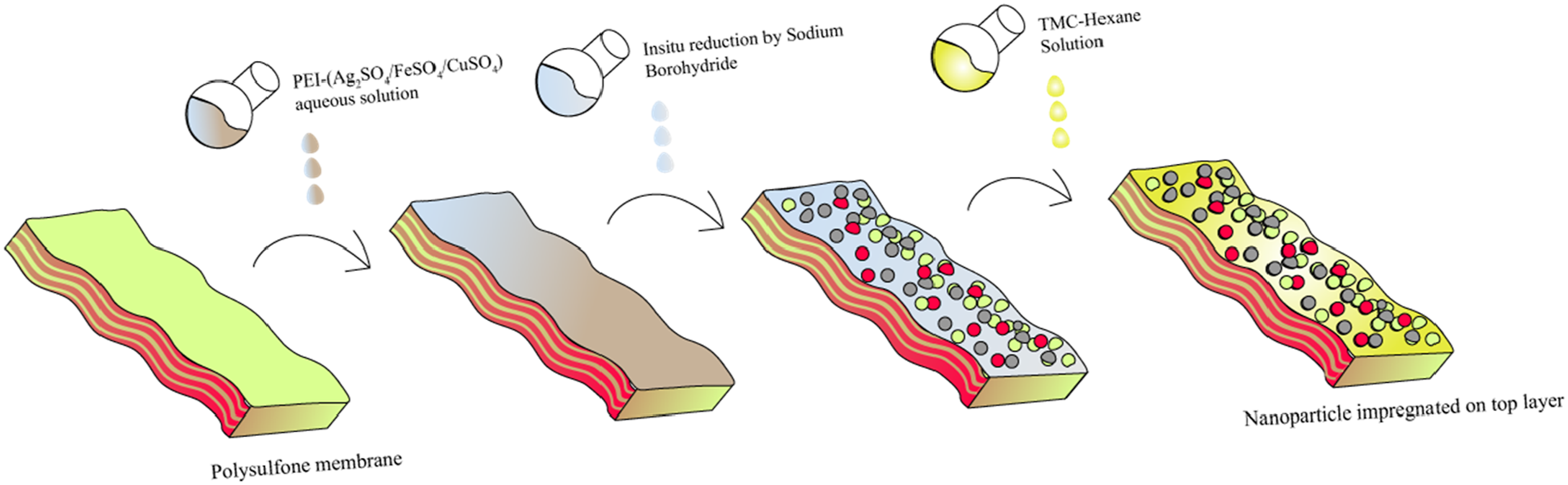
Characterization
Pure Water Permeability (PWP)
The PWP of the membranes was tested by a cross-flow membrane filtration setup with reverse osmosis (RO) water as the feed. The cross-sectional membrane area for each sample was 14.5 cm2. Before permeate collection, the membranes underwent a compaction process at 10.34 Bar for 30 min to ensure optimal operation. Thereafter, the permeate samples were collected at an applied pressure of 6.89 Bar.
Scanning electron microscope/energy dispersive X-ray
A Field emission scanning electron microscope (FE-SEM, JEOL JSM-7100F) was used to observe the morphology of the top surface.
Atomic force microscope
An atomic force microscope (NT-MDT, Ntegra Aura) was utilized to analyze the surface properties, including roughness.
Attenuated total reflectance-fourier transform infrared
The chemical structure and functional groups of the membranes were examined using ATR-FTIR spectra (Thermo Fisher Scientific).
Chemical oxygen demand
Artificial wastewater was prepared according to the specific composition given below: Glucose (1 g/L), MgSO4·7H2O (1 g/L), FeSO4·7H2O (1 g/L), Urea (1 g/L), and Polyethylene Glycol-PEG (1 g/L). The membranes were compacted at 10.34 Bar using the prepared wastewater, and permeate was collected at 6.89 Bar.
The collected permeate was then analyzed for Chemical Oxygen Demand (COD). Before analysis, the permeate samples were diluted ten times with deionized water. To prepare the sample and blank separately, add 2 mL of diluted permeate and 2 mL of deionized water to the COD reagents. They were then digested at 150°C for 2 h. After digestion, the samples were cooled, and COD was measured using a photometer.
The Palintest Photometer 7500 Bluetooth was used to find the COD of the samples. The percentage rejection was calculated with the following equation, where CODF: Feed COD in mg/L,
CODP: Permeate COD in mg/L.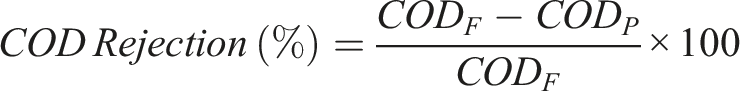
Leaching
Membrane samples were cut into uniform pieces measuring 3 cm × 3 cm and placed in beakers containing deionized water. Each membrane piece was affixed to the inner wall of the beaker to ensure full contact with the water. The beakers were then placed on a magnetic stirrer and stirred continuously at 570 revolutions per minute for 24 h.
The concentration of any leached silver, iron, or copper elements in samples was determined using an Inductively Coupled Plasma (ICP)-Mass Spectrometry. The leaching rate (µg/m2h) was calculated using the following equation: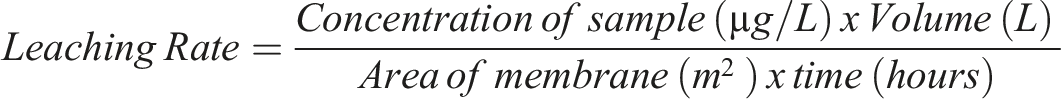
Inductively coupled plasma-mass spectroscopy
ICP-MS (S8 Tiger Model Bruker Company) was used to detect and analyze elements in leached-out samples.
Zeta potential
Malvern Nanoseries Nano-ZS was used for zeta potential measurement.
Fouling study
The Flux Recovery Ratio is a common way to study membrane fouling. Take the initial pure water flux of each membrane at 6.89 Bar before fouling. Thereafter, a 3000 mg/L egg albumin solution was utilized to foul membranes for 6 h at 10.34 Bar pressure, and flux was measured every 30 min. Membranes were gently cleaned after fouling, and the final pure water flux was collected at 6.89 Bar. Use the initial and final PWP data to find the flux recovery ratio (FRR) by the formula given below. This ratio indicates up to what extent the membrane flux can be recovered after fouling.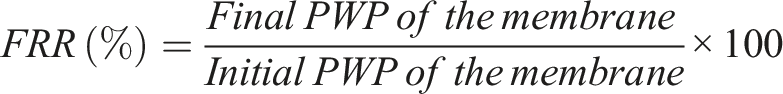
Results and discussions
Scanning Electron Microscope (SEM) imaging
The pristine polysulfone membrane was smooth with no particle distribution or agglomeration (Figure 2(a) (i)). The effect of increasing sodium borohydride concentration on pore size was investigated for silver, copper, and iron-embedded membranes. With increasing sodium borohydride concentration, the rate of reduction of the respective salt to the metal nanoparticle increases and the agglomeration of the nanoparticle increases. The agglomerated nanoparticle escapes from the membrane matrix, leaving the larger pore size at higher concentration of sodium borohydride. The pore size distribution of all membranes is given in Figure 2(c) with virgin PSF had 101.01 nm, while Fe-5 (148.85 nm) has the highest among all membranes. (a) SEM images (i) PSF (top) (ii) Ag-1 (iii) Ag-2 (iv) Ag-5 (v) (Second row, left to right) Cu-1 (vi) Cu-2 (vii) Cu-5 (Third row, left to right) (viii) Fe-1 (ix) Fe-2 (x) Fe-5 (Fourth row, left to right). (b) Energy dispersive X-ray (i) PSF (top) (ii) Ag-1 (iii) Ag-2 (iv) Ag-5 (v) (Second row, left to right) Cu-1 (vi) Cu-2 (vii) Cu-5 (Third row, left to right) (viii) Fe-1 (ix) Fe-2 (x) Fe-5 (Fourth row, left to right). (c) EDx (i) PSF (ii) Ag-1 (iii) Ag-2 (iv) Ag-5 (v) Cu-1 (vi) Cu-2 (vii) Cu-5 (viii) Fe-1 (ix) Fe-2 (x) Fe-5. (d) Pore-size distribution (i) PSF (ii) Ag-1 (iii) Ag-2 (iv) Ag-5 (v) Cu-1 (vi) Cu-2 (vii) Cu-5 (viii) Fe-1 (ix) Fe-2 (x) Fe-5.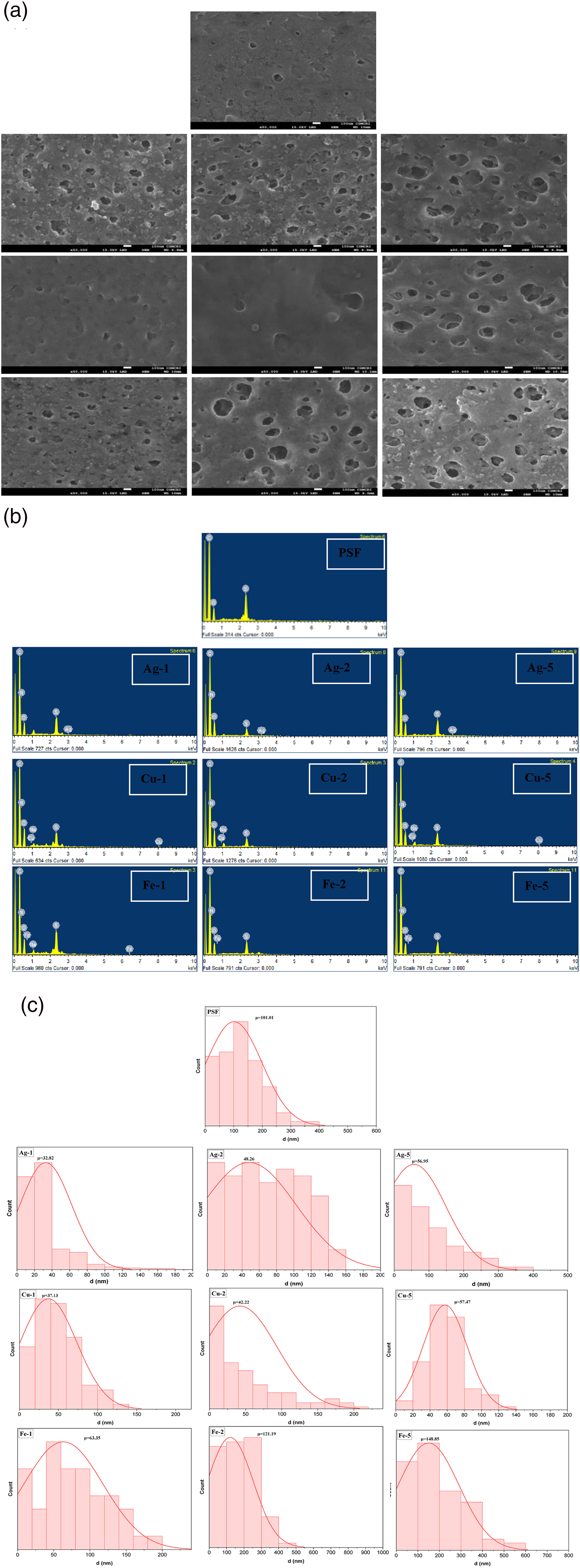
Nanoparticle loading, oxygen content, and average pore diameter of membrane.
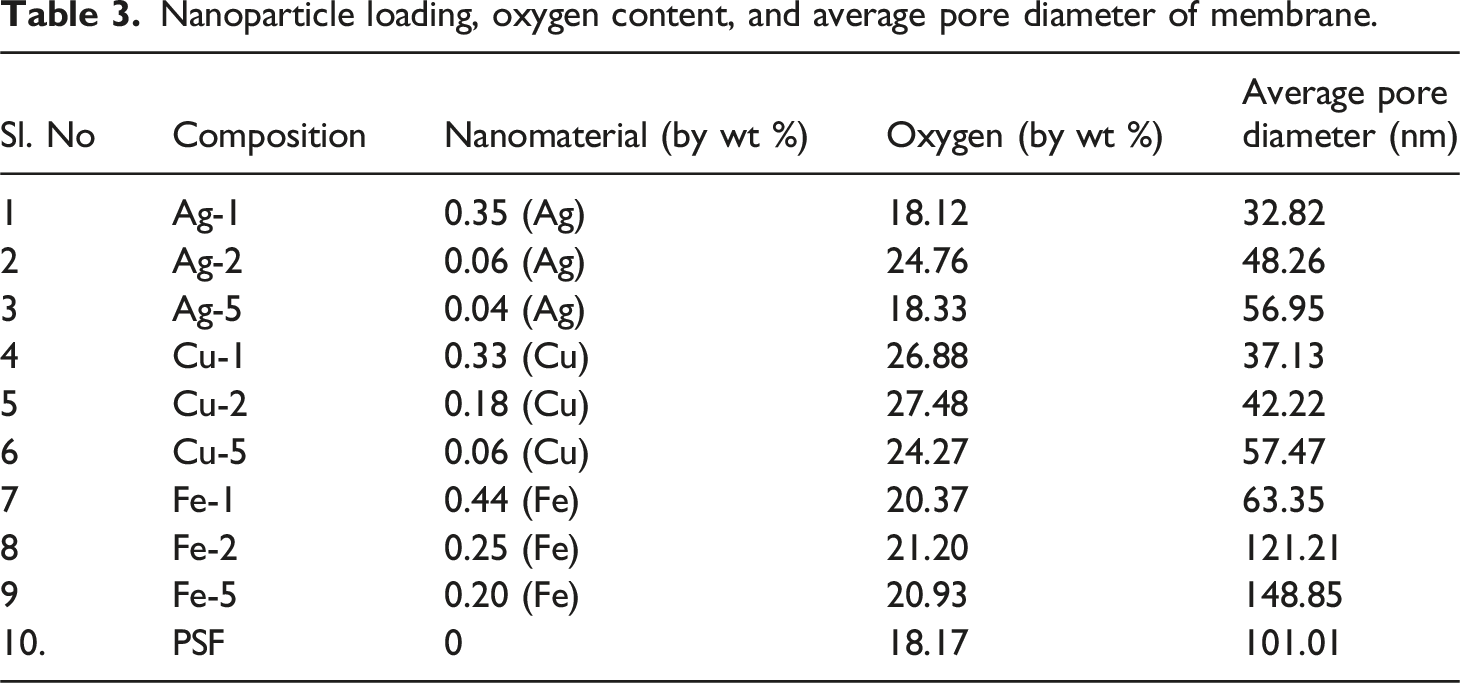
Contact angle
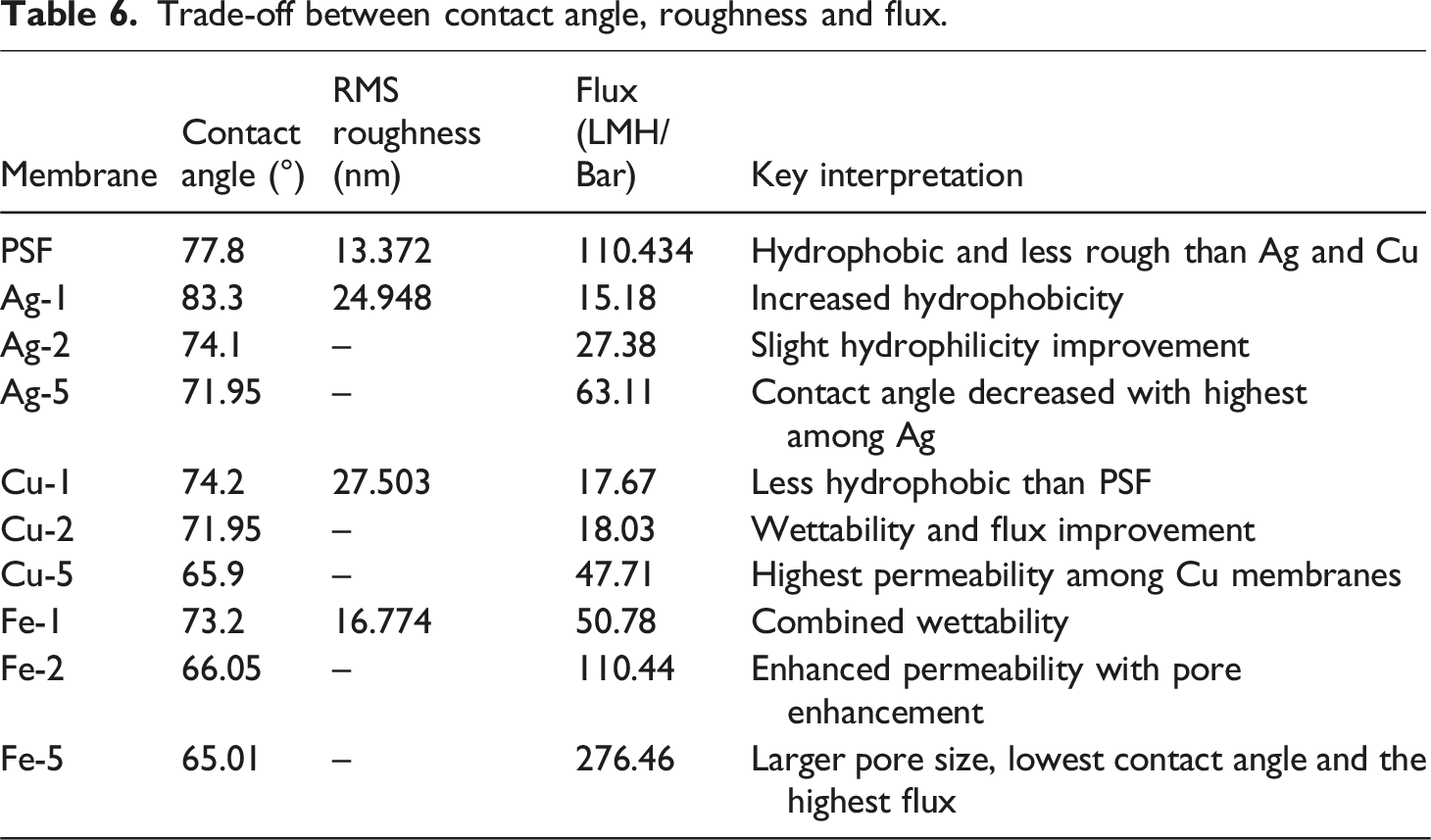
The virgin PSF had an average contact angle of 77.8°. Silver embedded with 1% NaBH4 exhibited the highest average contact angle (83.3°) out of all the modified nanocomposite membranes, reflecting its resistance to pass water through the membrane. For all modified membranes, the contact angle decreased with higher NaBH4 concentration. Contact angle shows membrane wettability,
30
and all membranes with higher NaBH4 concentration have shown to have higher wettability, which also proves the higher flux performance by the membranes. The average contact angle value of each nanocomposite membrane can be seen in Figure 3. Average contact angle of all membranes.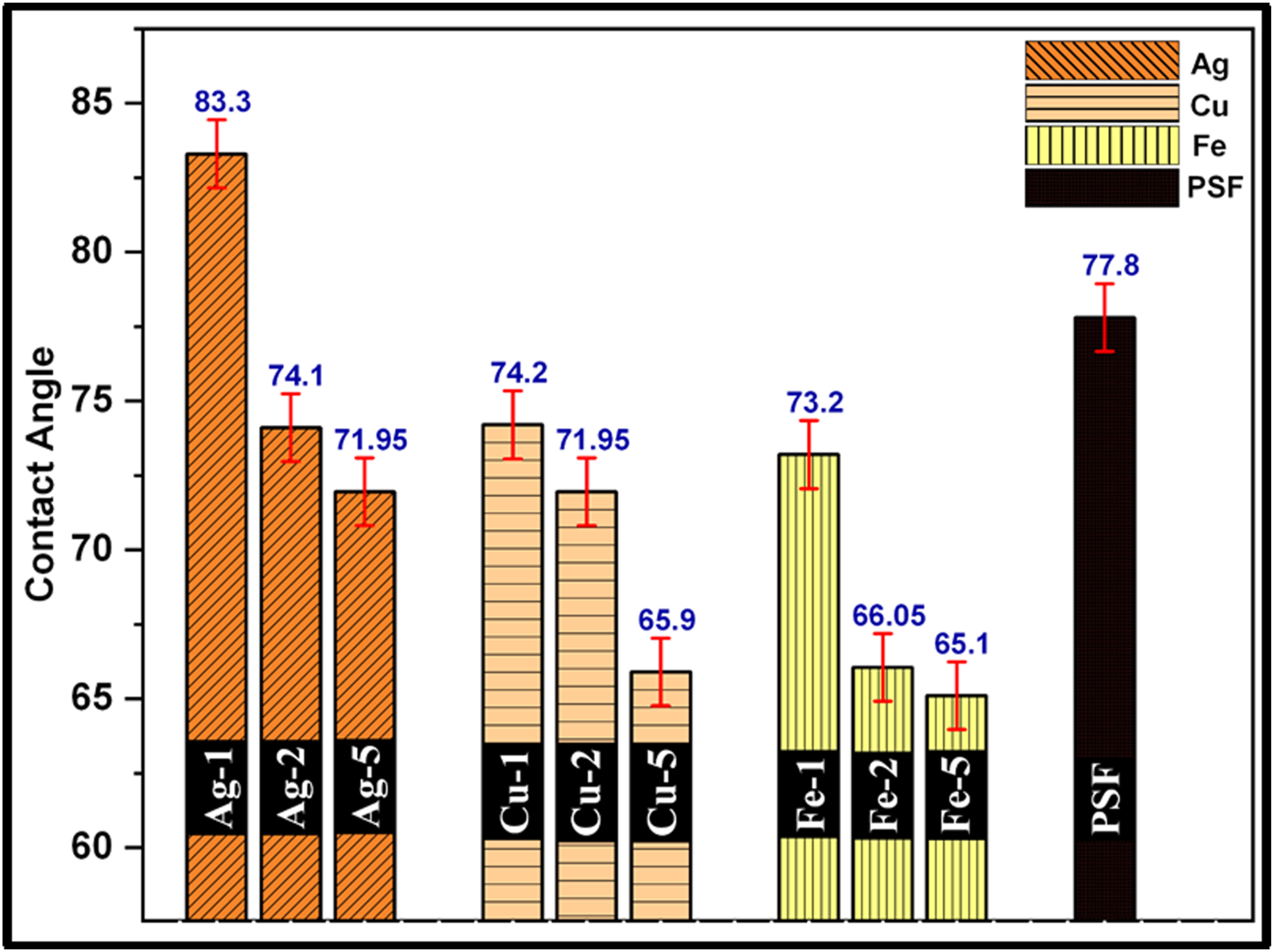
Atomic force microscopy
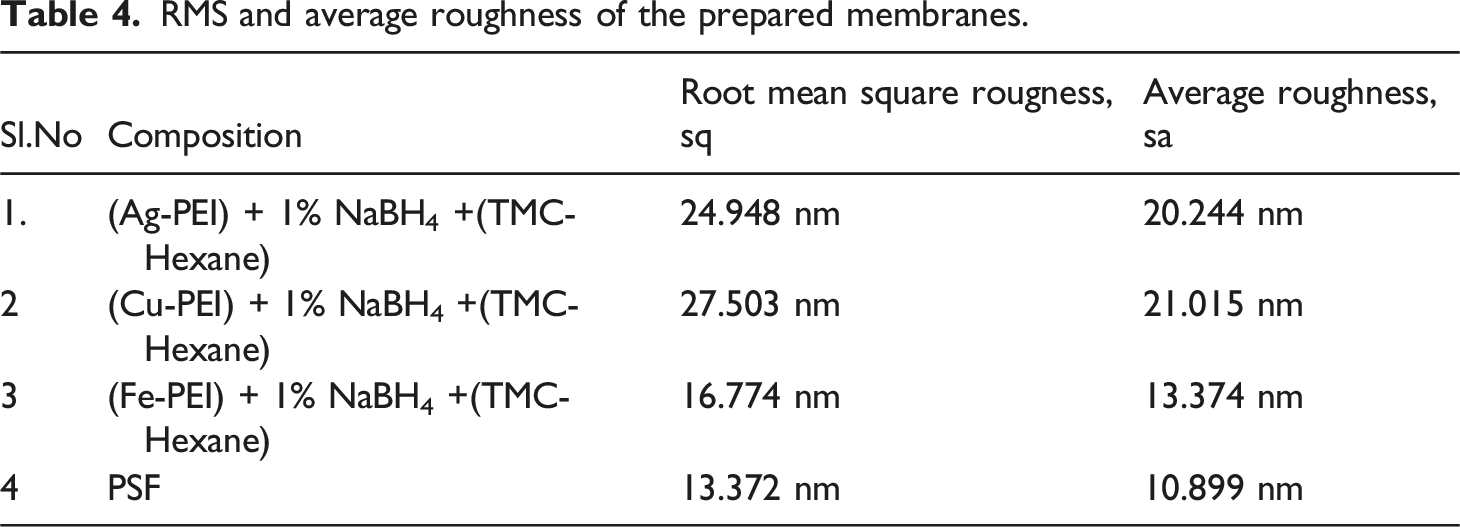
The atomic force microscopic images of PSF and the membranes reduced with 1% NaBH4 are shown in Figure 4. The average roughness and the root mean square roughness of the membranes are given in Table 4. The PSF membrane had an average roughness of approximately 10.899 nm and an RMS value of approximately 13.372 nm. All modified membranes showed a higher roughness value because of the distribution of nanoparticles in the membrane matrix. Copper-embedded membranes showed the highest roughness, while the iron-embedded membrane showed the lowest roughness amongst the nanocomposite membranes. It is clearly visible in ridge and valley structures, as visible in AFM images. AFM images. RMS and average roughness of the prepared membranes.
X-ray photoelectron spectroscopy
The surface chemical composition and oxidation states of the prepared membranes were investigated by X-ray photoelectron spectroscopy (XPS) given in Figure 5. The survey spectrum confirms the presence of C, O and S elements characteristic of the polysulfone (PSF) backbone. Deconvolution of C1s spectrum exhibits a dominant peak at 284.7 eV attributed to the C–C bond of PSF, along with a secondary component centered at 286.2 eV corresponding to C–O bonds from ether linkages in the polymer structure. The S2p spectrum shows the characteristic doublet at 167.79 and 168.88 eV, assigned to the sulfone (–SO2–) group, confirming the chemical integrity of the PSF backbone. The O1s spectrum was deconvoluted into two components centered at 532.18 and 533.7 eV. The lower binding energy peak (532.18 eV) is attributed to oxygen in the sulfone (O = S = O) group, while the higher binding energy component (533.7 eV) corresponds to ether (C–O–C) oxygen within the PSF structure.
31
The XPS survey spectra confirm the successful impregnation of nanoparticles in the modified membranes as shown in Figure 6(a)–(c). Also, Nitrogen from interfacial polymerization of both PEI and TMC, along with Carbon and Oxygen from PSF, is visible. XPS (i) survey of PSF and deconvoluted peaks of (ii) C1s (iii) O1s (iv) S2p. XPS survey of (a) Ag-1 (b) Cu-1 (c) Fe-1 and deconvoluted peaks (d) Ag-1 (e) Cu-1 (f) Fe-1.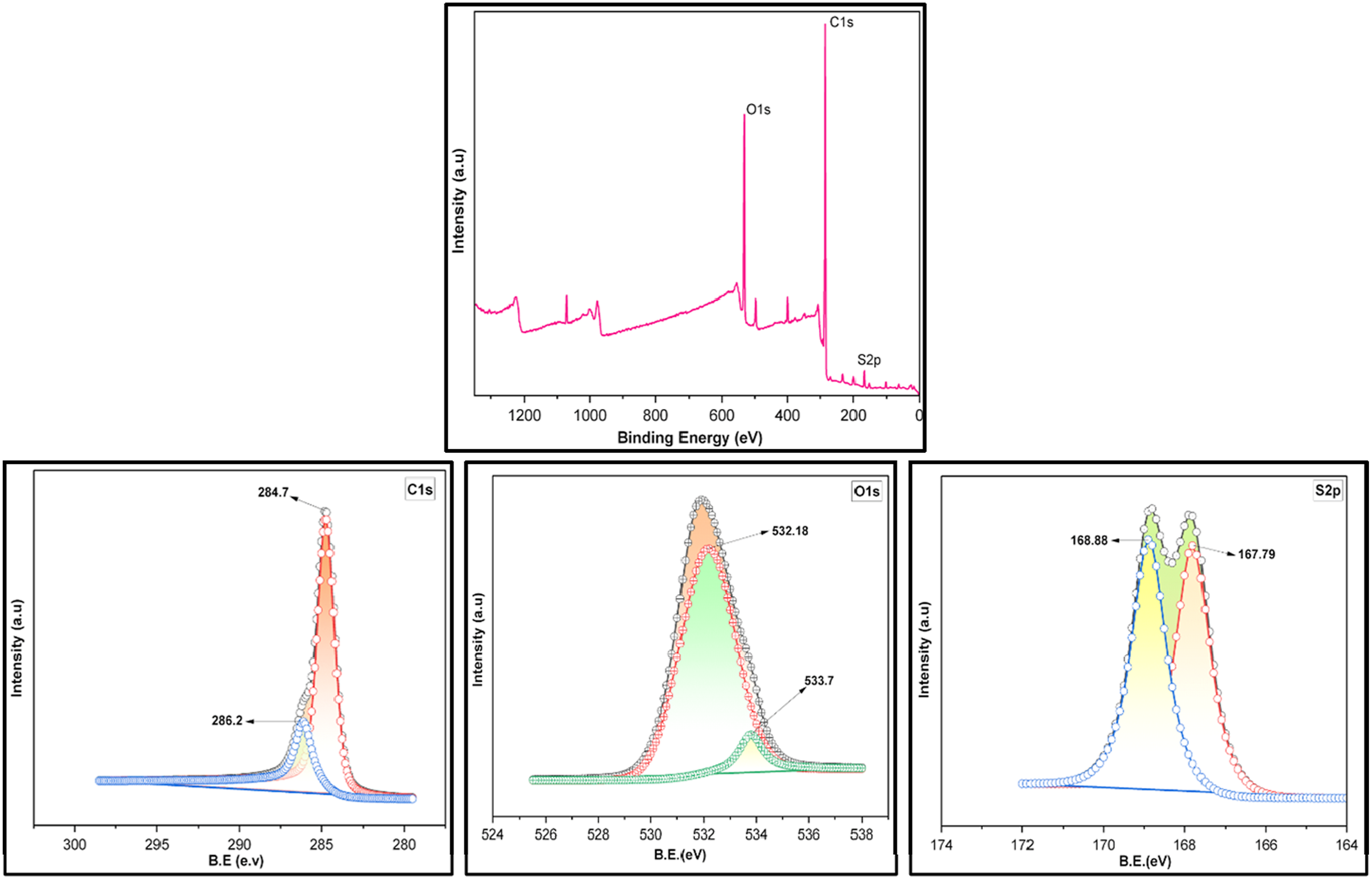
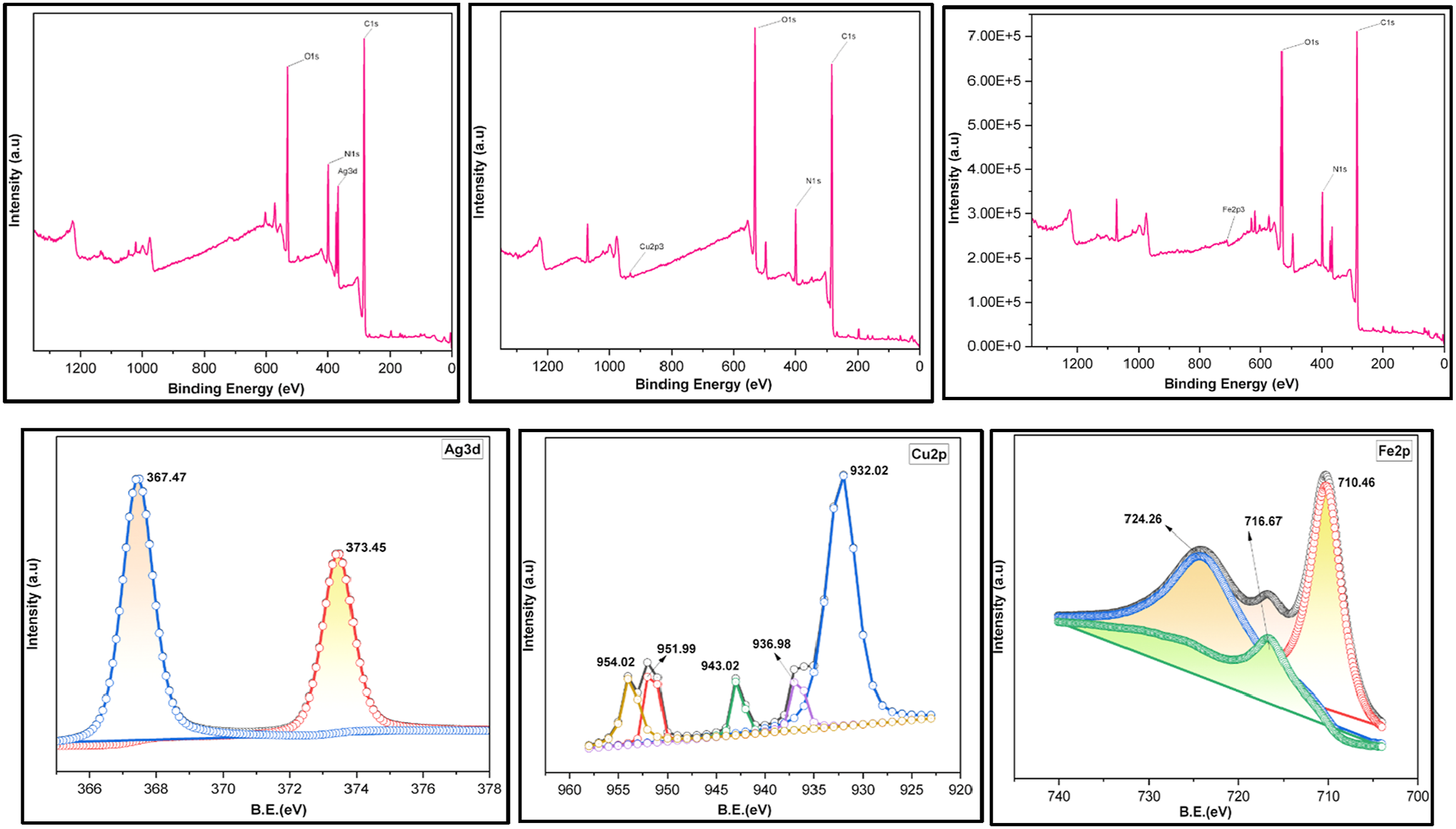
The Ag3d spectrum displays two well-defined peaks at 367.47 eV (FWHM 1.13 eV) and 373.45 eV corresponding to Ag3d5/2 and Ag3d3/2, respectively, characteristic of silver (AgO, mixed Ag+). No evident satellite features associated with oxidized silver species were observed. 32 The Cu2p spectrum shows a primary Cu2p3/2 peak at 932.02 eV (reduced copper) and Cu2p1/2 at 951.99 eV. The presence of shake-up satellite peaks at higher binding energies (936.98 eV and 943.02 eV) confirms the existence of Cu2+ species, indicating partial oxidation of copper within the membrane. The FWHM of 4.29 eV for Cu2p3/2 peak indicates the convolution of multiple oxidation states.33,34 Similarly, the Fe2p spectrum exhibits Fe2p3/2 and Fe2p1/2 peaks at 710.46 eV and 724.26 eV, respectively, along with a satellite feature at 716.67 eV, characteristic of Fe3+ species. The high FWHM value (3.93 eV) indicates that the peak is very broad and Fe has mixed oxidation states. This broadness, along with the binding energy, is characteristic of the existence of shake-up satellite peaks, which are a definitive signature of oxidized iron species e.g., Fe3+. 35 Thus, the nanoparticles embedded in membranes are in mixed oxidation states.
Attenuated Total Reflectance (ATR)−Fourier Transform Infrared (FTIR)
Attenuated total reflectance Fourier transform Infrared Spectroscopy of the membrane samples was done. Characteristic peaks of polysulfone membrane have been reported.
30
The ATR-FTIR spectra of all the membranes are shown in Figure 7, and characteristic peaks are shown below: • 2968 cm−1: Alkyl C–H stretching, • 1585 cm−1 and 1487 cm−1: Indicating the presence of aromatic groups through C = C and C–C stretching vibrations of the aromatic ring, • 1240 cm−1: Asymmetric C–O–C stretching (aryl-ether), • 1150 cm−1: Symmetric O = S = O stretching from the sulfone group • 1106 cm−1: Benzene ring vibrations, • 1013 cm−1: Aromatic C–H in-plane bending, • 557 cm−1: C–S stretching, • 1325 cm−1 and 1295 cm−1: Asymmetric S = O stretching ATR-FTIR (i) PSF (ii) Ag-1 (iii) Ag-2 (iv) Ag-5 (v) Cu-1 (vi) Cu-2 (vii) Cu-5 (viii) Fe-1 (ix) Fe-2 (x) Fe-5.
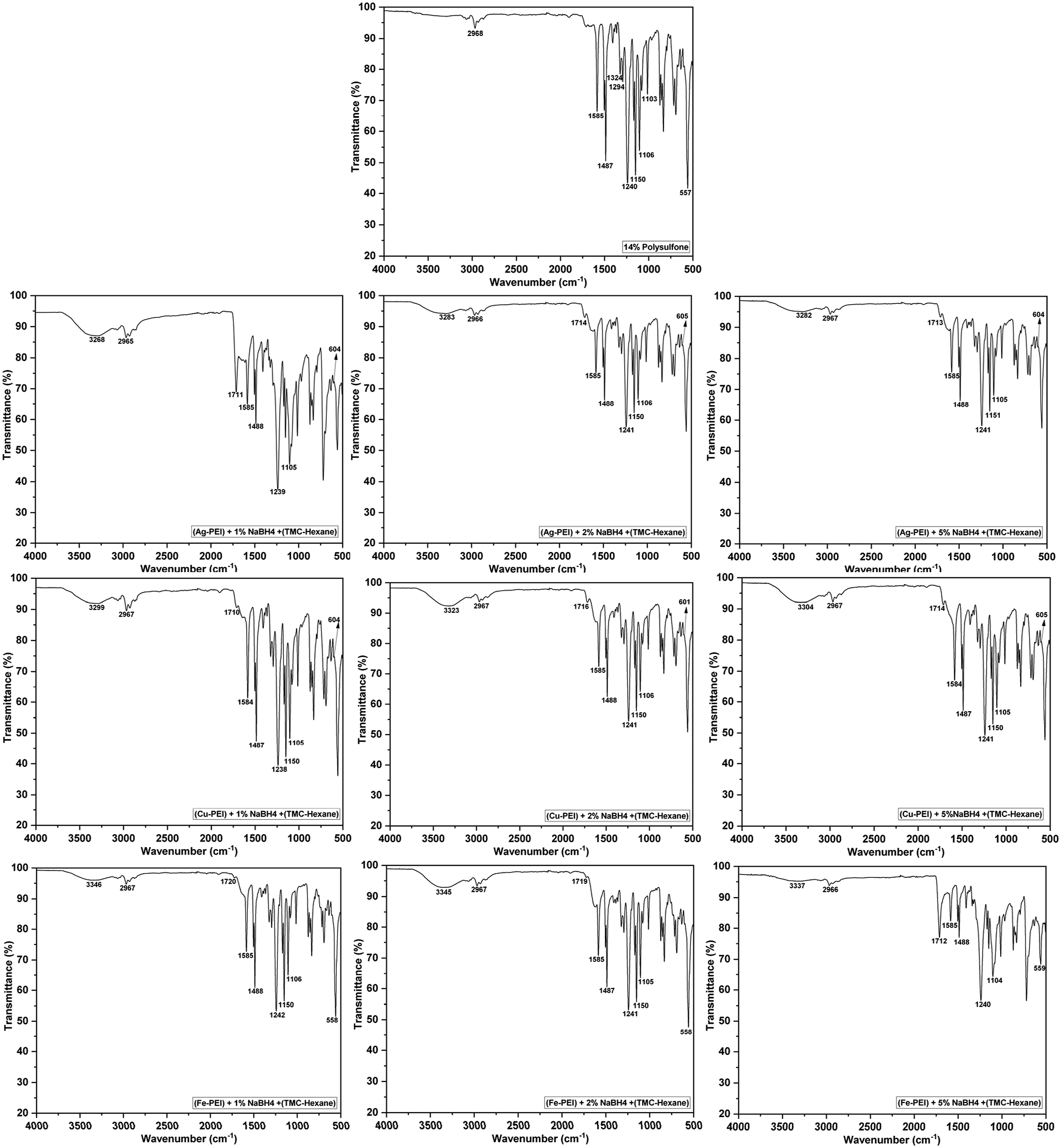
All modified membranes displayed these characteristic polysulfone peaks. In addition, new peaks appeared at 1710–1723 cm−1 (C = O stretching) and 3200–3400 cm−1 (N–H stretching), confirming the amide bond formation by reaction of polyethyleneimine (PEI) and trimesoyl chloride (TMC). 36 According to established studies, Silver oxides peaks appear in the range of (585 cm−1 - 620 cm−1), while Iron oxides (540 cm−1- 562 cm−1) and Copper oxides (596 cm−1- 611 cm−1).37–40 Due to spectral overlap with PSF it is difficult to predict metal oxides with FTIR alone, surface chemical states were verified using XPS analysis.
Pure water permeability
The pure water permeance/flux (in LMH/Bar) of the synthesized membranes are displayed in Figure 8. The pristine PSF membrane showed permeance of about 110.434 LMH/Bar. The flux for nanoparticle embedded membranes increases as the concentration of NaBH4 increases from 1% to 5%. Ag-5 has the highest flux among the silver embedded membranes (63.11 LMH/Bar), Cu-5 (47.71 LMH/Bar) for copper embedded, and Fe-5 has the highest flux for iron membranes (276.46 LMH/Bar). This trend may be explained by the fact that, as the concentration of NaBH4 in these membranes increases, agglomerated nanoparticles are being removed from the membrane matrix as explained earlier, leaving larger pores as shown in Figure 2(a). Moreover, in one case (Fe-5), the pure water flux is even higher than polysulfone membrane because of very low contact angle and hydrophilic surface formation with a very porous layer formation on top, which imposes no additional resistance. Overall, among all the membranes, those reduced with 5% NaBH4 showed the highest flux, with Fe-5 having 276.46 LMH/Bar. Literature also reported the high flux membrane with iron nanoparticles.41,42 Pure water permeance in LMH/Bar.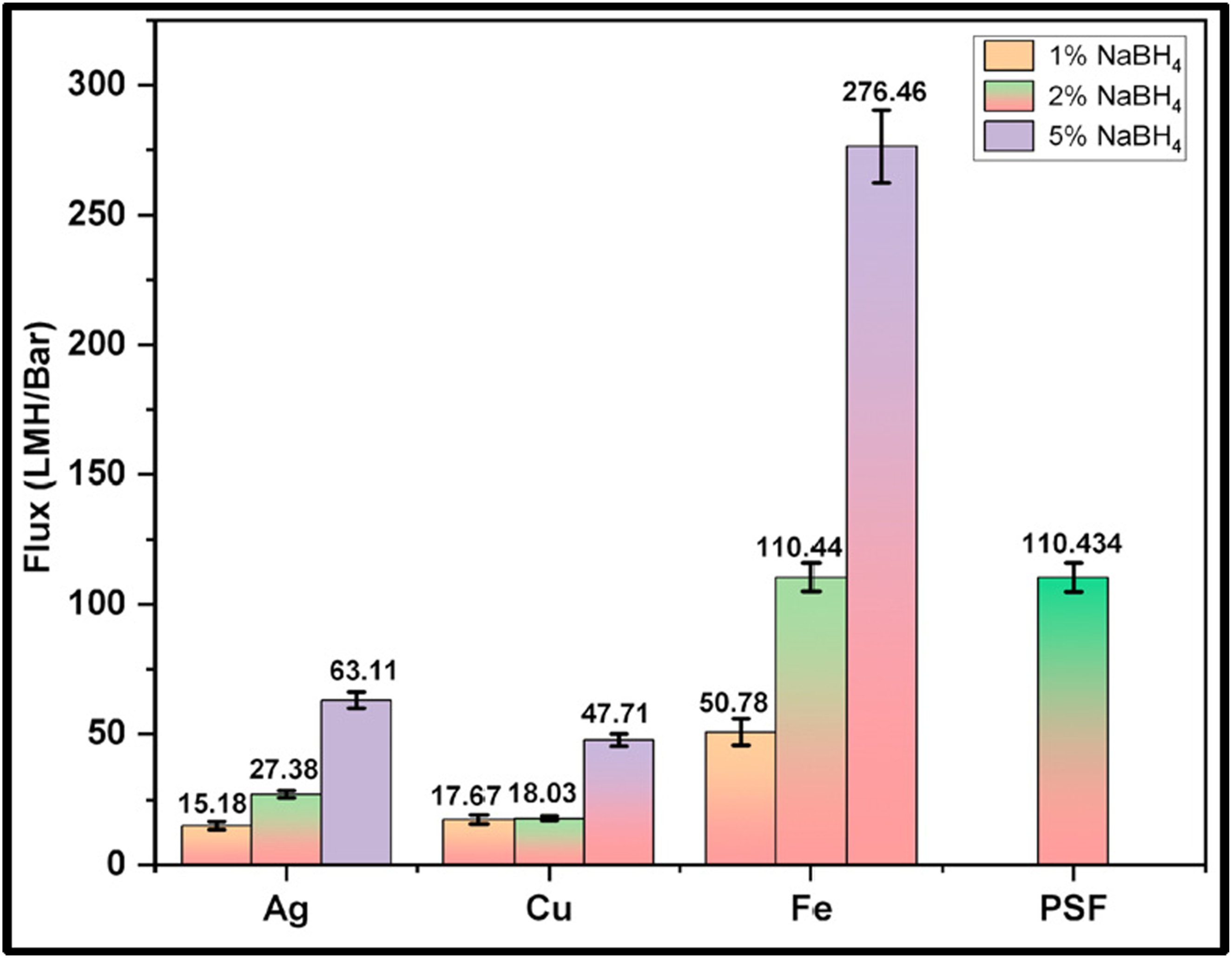
Chemical oxygen demand
The COD rejection (%) is displayed in Figure 9. Compared to the other membranes, the silver membrane, reduced by 1% NaBH4, had the lowest permeate COD and the highest rejection of 17.44%. PSF had the highest permeate COD and the lowest rejection rate of 1.16%. Rejection was determined by the difference in Feed and permeate COD divided by feed COD in percentage terms. Surface charge and adsorption ability to retain organics of membranes can have a significant effect on the COD removal.
43
The membranes with nanomaterials have shown higher COD removal because of the nanomaterials in the top surface and the charge effect, facilitating the adsorption of organics to retain them. The iron membrane showed moderate rejection of COD, although the pore size and flux are higher, primarily due to organic adsorption over the surface. Chemical Oxygen Demand (COD) rejection.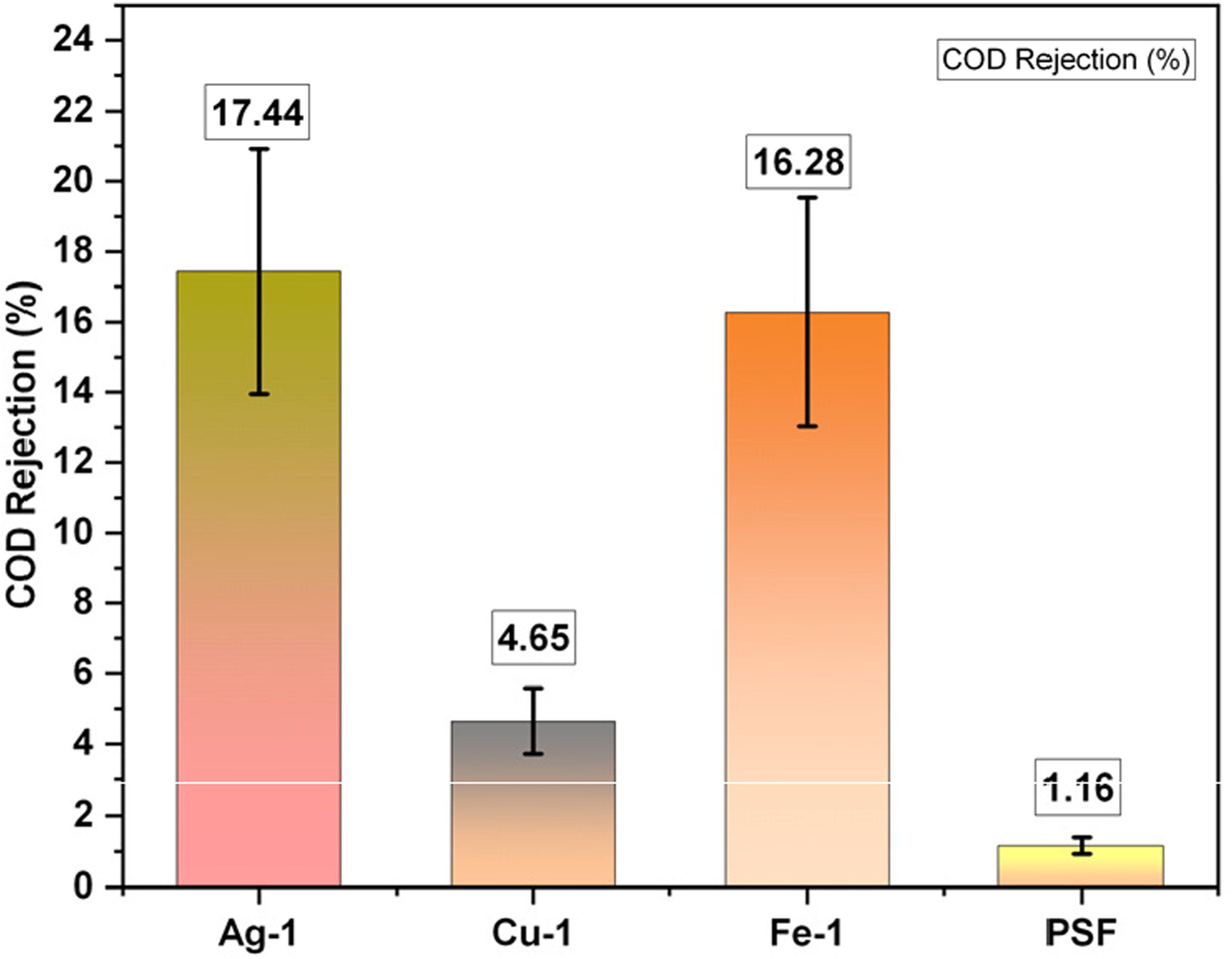
Leaching study
Leaching rate of the membranes.
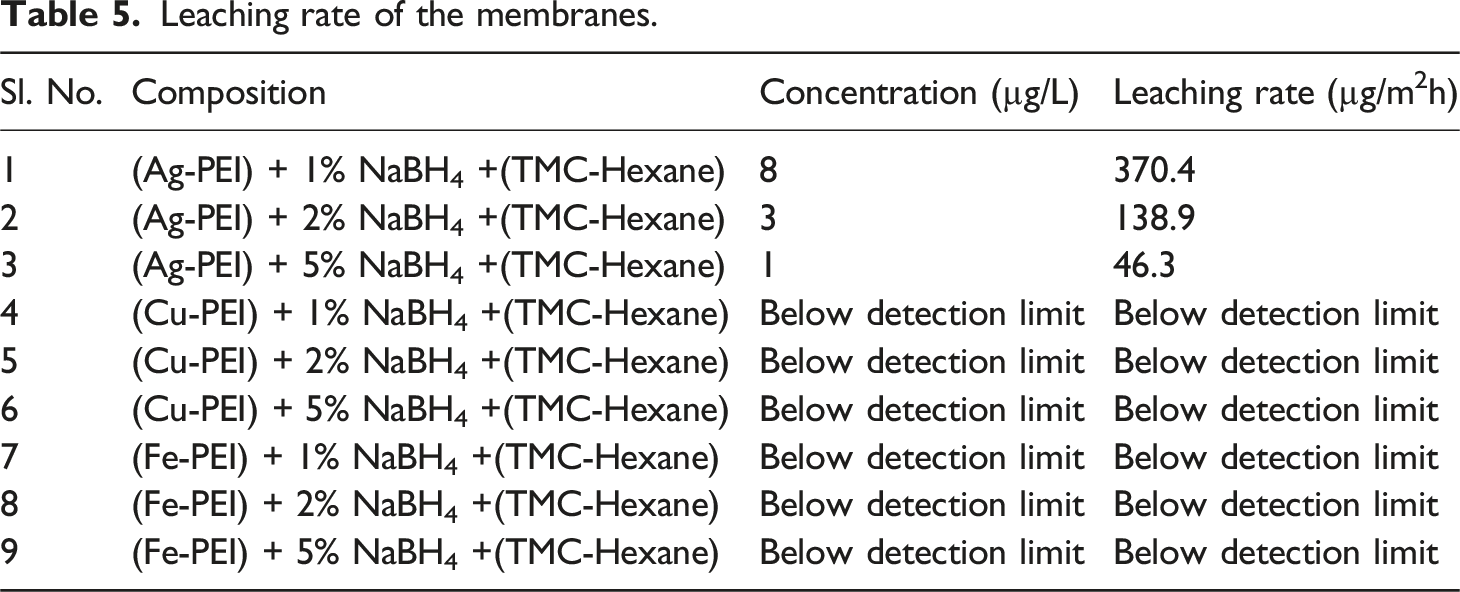
The presence of metal oxides may result from exposure to air or water post-modification, promoting oxidation of the embedded metals. As previously reported, silver nanoparticles embedded in a polysulfone matrix tend to leach out, especially under aqueous conditions.
44
In contrast, the lower leaching observed for Cu- and Fe-modified membranes can be attributed to their higher oxide stability and stronger interaction with the membrane matrix.
45
XPS analysis and EDX data prove the presence of metal oxide. The XPS analysis confirmed that the metal nanoparticles are in mixed oxidation states. As shown in Figure 10, the membranes reduced by a higher concentration of sodium borohydride had shown lower leaching because of escape of nanomaterials during membrane formation and lower net metal concentration. Whereas the membrane reduced by a lower concentration of sodium borohydride had a higher silver composition and therefore a higher leaching rate. Leaching rate (µg/m2h) with reference to silver composition in the membrane.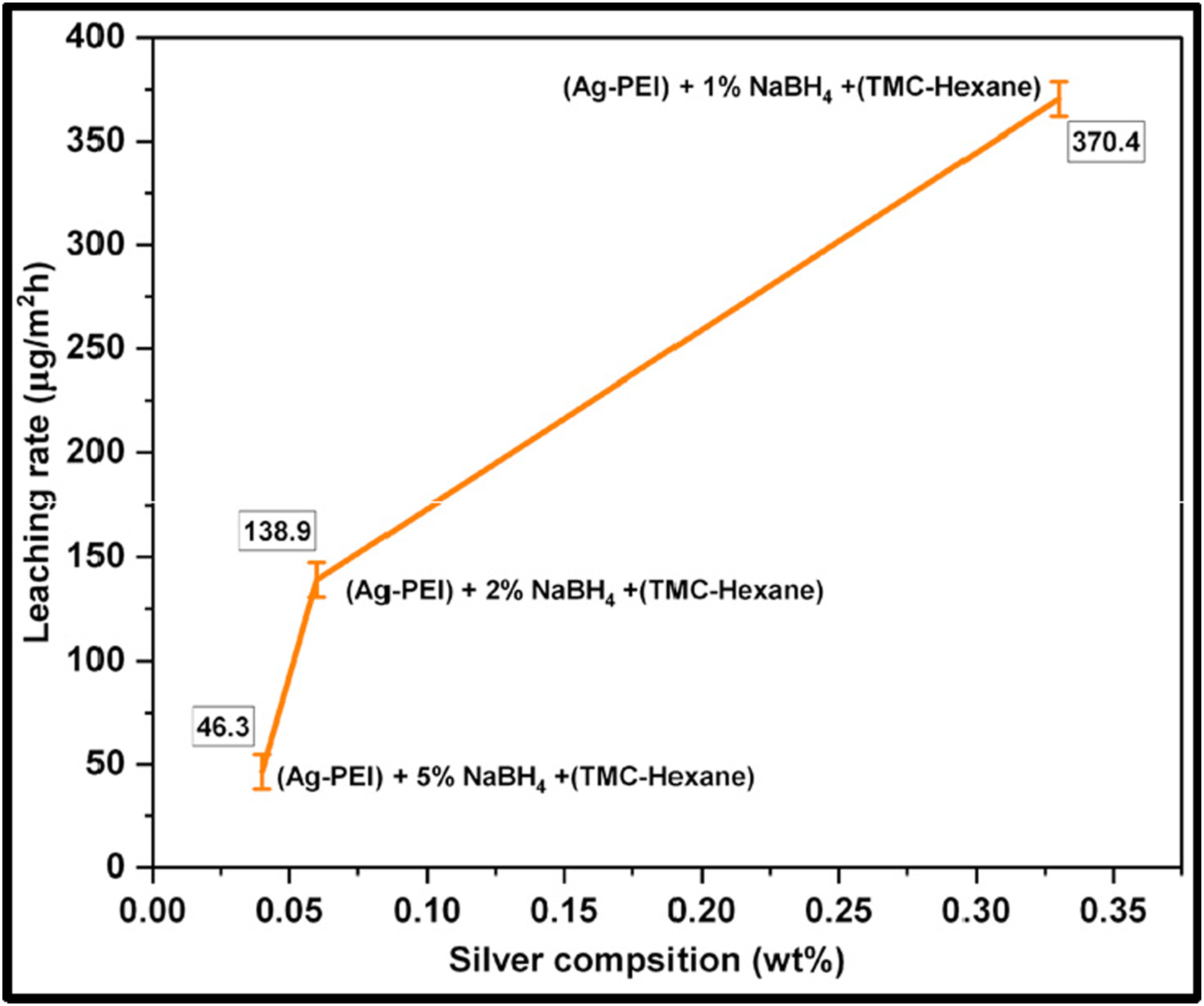
Flux recovery ratio
The flowrate decline curve and FRR data for all the membranes is given below (Figure 11(a) and (b)). Copper embedded membranes showed better flux recovery ratio compared to others, with copper reduced with 1% NaBH4 having the best antifouling performance (FRR:82.02%), followed by copper embedded with 2% NaBH4. All Iron-embedded membranes showed poor antifouling performance compared to PSF, Ag, and Cu; the control polysulfone membrane had an FRR% of 12.71%. Iron-embedded membranes had higher porosity and larger pore size. Also, iron and iron oxides enhance protein adhesion and fouling in iron-embedded membranes by interacting with proteins via coordination bonds and electrostatic interactions.46,47 The control system exhibited a zeta potential of −12.5 mV, which shifted markedly toward neutrality (−0.06 mV) in the presence of iron at higher concentration. This substantial increase in value of surface charge indicates pronounced electrostatic neutralization, promoting protein aggregation with Iron and increased fouling behavior. The zeta potential revealed adhesion promoting protein aggregation and increased fouling behavior. Thus, a higher adhesion tendency resulted in a lower FRR of all iron membranes as compared to the PSF membrane. (a) Volumetric flowrate decline curve. (b) Flux recovery ratio (%).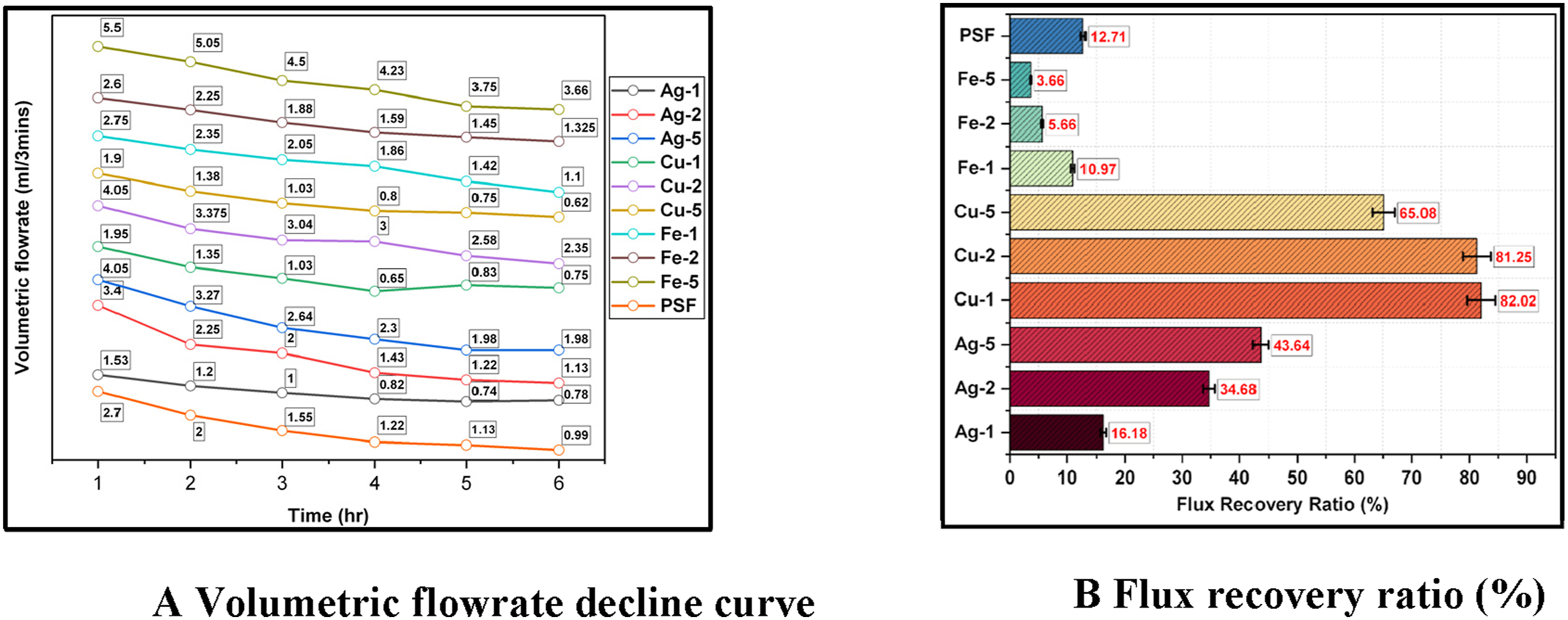
Cu-1 demonstrated superior FRR compared to others because of its inherent antifouling properties, which inhibit the protein deposition and adhesion. 14 This superior FRR was attributed to its high hydrophilicity, no significant leaching, and oxide stability revealed by XPS. Silver, despite its high antifouling property, showed lower FRR due to its leaching and instability in the polysulfone matrix. Also, relatively higher contact angle of silver-embedded membranes showed a more hydrophobic nature as compared to the other nanocomposite membranes.
Trade-off between contact angle, roughness and flux
Trade-off between contact angle, roughness and flux.
Conclusion
This research successfully integrated silver, copper, and iron nanoparticles into polysulfone membranes through in situ reduction and interfacial polymerization. The characterization of the membranes revealed that the incorporation of nanoparticles improved hydrophilicity, surface roughness, and pore formation, which resulted in enhanced water flux. The iron-embedded membrane exhibited the highest flux rate (276.46 LMH/Bar) with no detectable leaching, though it demonstrated poor antifouling performance. Silver-embedded membranes faced challenges due to significant leaching and lower stability against fouling. Copper-embedded membranes exhibited a well-balanced profile, achieving high antifouling efficiency (represented by high FRR of 82.02%) with minimal leaching, making them the most promising candidate for developing stable and high-permeance ultrafiltration membranes. In summary, copper-embedded PSF membranes offer a feasible approach for creating durable, environmentally friendly, and high-performance membranes, with potential for future research to enhance their operational efficiency, longevity, and applicability to various water treatment situations.
Footnotes
Acknowledgements
CSIR-CSMCRI PRIS NO 159/2025. Authors acknowledge the Marine Elements and Marine Environment Division for analytical support. Authors acknowledge the open-source image processing software ImageJ for pore size determination from SEM images. Authors acknowledge the funding support of Council of Scientific and Industrial Research Mission Mode Project on specialty chemicals.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Central Salt and Marine Chemicals Research Institute, Council of Scientific and Industrial Research; MMP035201, Mission Mode Project on specialty chemicals.
