Abstract
This study offers a systematic review of the methodology surrounding the use of thermoplastics in three-dimensional (3D) printing for medical applications. Despite 3D printing not being extensively adopted for the creation of clinical medical devices due to safety and legal concerns, recent developments in materials, printing technology, and professional skills have broadened its clinical uses. The use of thermoplastics in 3D printing allows for the creation of economical components with diverse properties and potential applications. For example, literature consistently reports that while PLA-based structures typically exhibit tensile strengths in the range of 50–65 MPa, PEEK-based printed components achieve substantially higher strengths of 90–100 MPa, alongside superior fatigue resistance and long-term biocompatibility, making PEEK more suitable for load-bearing orthopedic applications. By employing design tactics such as thermoplastic layering, it is possible to reconcile competing demands, such as the mechanical strength and biological compatibility required for tissue structures. Consequently, this review summarizes the research efforts aimed at identifying appropriate thermoplastics, including polylactic acid (PLA), polypropylene (PP), polycarbonate (PC), polyurethane (PU), Acrylonitrile Butadiene Styrene (ABS), polyetheretherketone (PEEK), and polyvinyl alcohol (PVA), for the production of biomedical parts through 3D printing. It covers a range of produced items, including bones, high-quality prosthetics, intervertebral discs, medical devices, heart valves, and tissues containing blood vessels. Additionally, this review examines various 3D printing techniques, the challenges faced, and the future prospects for thermoplastic biomedical components.
Introduction
Major thermoplastic and their applications.

Methodology
To prepare a review titled “Developments in Thermoplastic Polymers for 3D-Printed Medical Devices: Balancing Mechanical Performance and Biocompatibility,” a search was conducted in the following databases: Sage database, Elsevier database, Web of Science - Core Collection, Clarivate Analytics, Scopus, SpringerLink Journals, and Taylor and Francis Journals. Keywords such as 3D printing, bioprinting, additive manufacturing, thermoplastic polymers, medical applications, biocompatibility, polylactic acid, polycarbonate, polypropylene, polyurethane, acrylonitrile butadiene styrene, polyether ether ketone, polyvinyl alcohol, scaffolds, prosthetics, tissue engineering, polymer blends, sterilization, biodegradation, and implants were used to retrieve relevant studies that explicitly addressed polymers used in 3D-printed medical devices. The search was limited to English-language publications only. The PRISMA checklist was used to validate the search process. The PRISMA flow chart presented graphically in Figure 1 and their screening criteria is tabulated in Table 2. PRISMA flow chart. Screening criteria.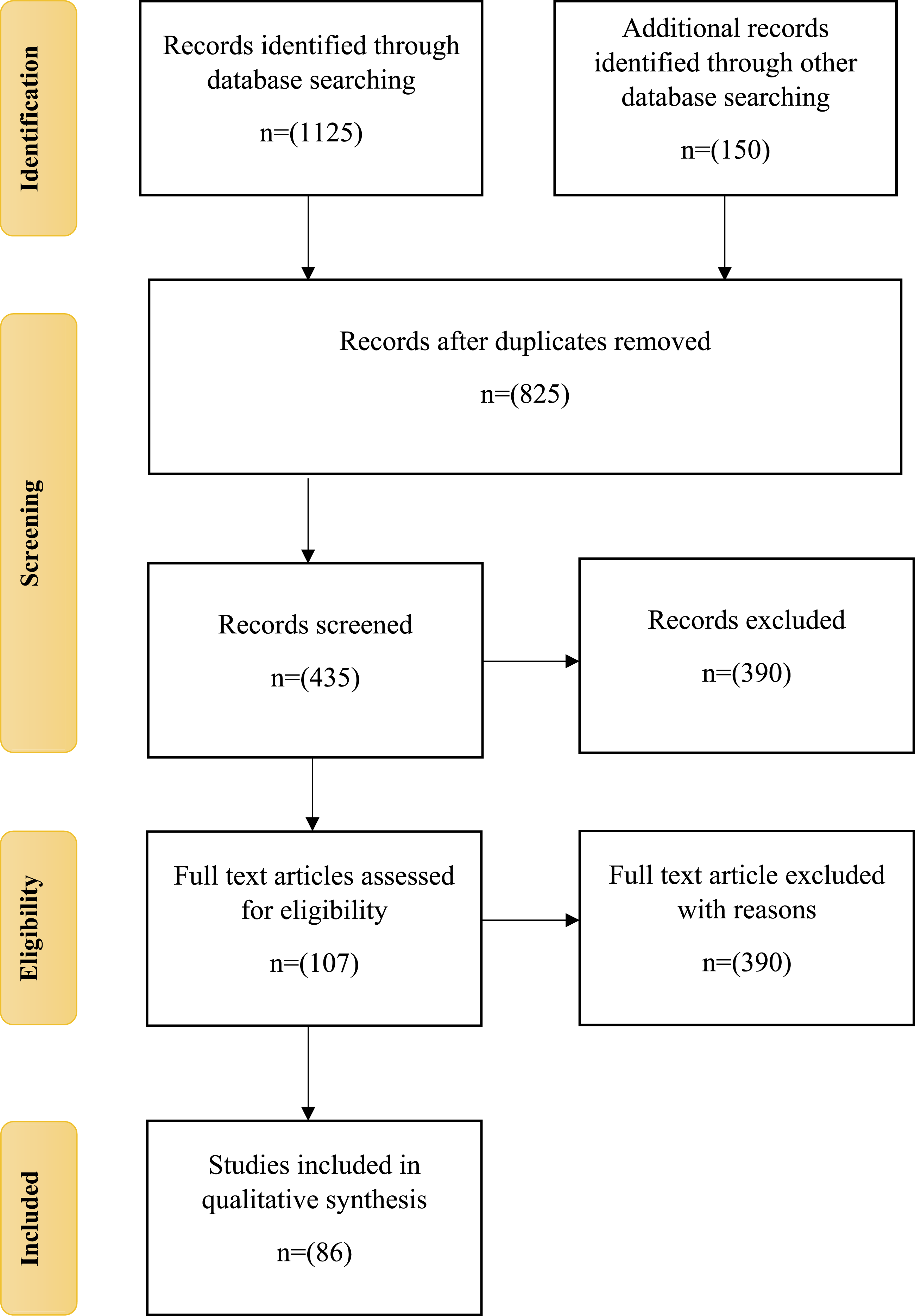
3D printing applications in prosthetics
3D printing represents a progressive and innovative technology in the manufacturing sector, facilitating the production of complex components that traditional methods cannot achieve. 47 The additive process typically commences with the design of a 3D model using CAD software, which is subsequently exported in STL format. This is followed by the adjustment of the model’s orientation, slicing it into numerous layers, generating tool paths, printing, and post-processing. 48 Various substrate-based systems are utilized, including batch polymerization and powder- or solid-based systems. 49 Stereolithography, or vat polymerization, utilizes UV light to cure layers formed from photosensitive materials.50,51 In powder-based techniques, thermal methods are used to selectively bind powder particles. Solid-state methods may encounter issues such as residual stresses, subpar surface quality, partial melting, and microholes when sheets are fused. Shot peening technology can enhance surface quality, addressing challenges like wear that can diminish product lifespan. Notably, the orientation of the printed object significantly influences material properties, build time, required support, and surface finish. 52 Increasing the number of layers can mitigate unevenness in overhang areas. Various implantation devices have advanced technologically, yet they often present operational challenges that can render them ineffective. 53 Despite the higher cost associated with prosthetic devices, many individuals continue to use them. Prosthesis fabrication generally starts with the acquisition of specific anatomical data via a 3D scanner, followed by the use of 3D CAD software to create a tailored design based on the scanned data. 54 Essential components of a 3D-printed prosthesis encompass electronics, mechanics, and software to produce a cohesive solution. Integrated sensors enable prostheses to detect motion, contract, and move various body parts. The market offers a range of prostheses equipped with Bluetooth and cellular connectivity. Numerous studies highlight the utilization of diverse thermoplastic materials for producing high-quality yet cost-effective prosthetics. 55 These affordable devices are frequently made from superior thermoplastics using 3D printing technology. The subsequent sections will delve into typical thermoplastics employed in the creation of high-quality, low-cost 3D printing applications and their related devices.
Polylactide (PLA)
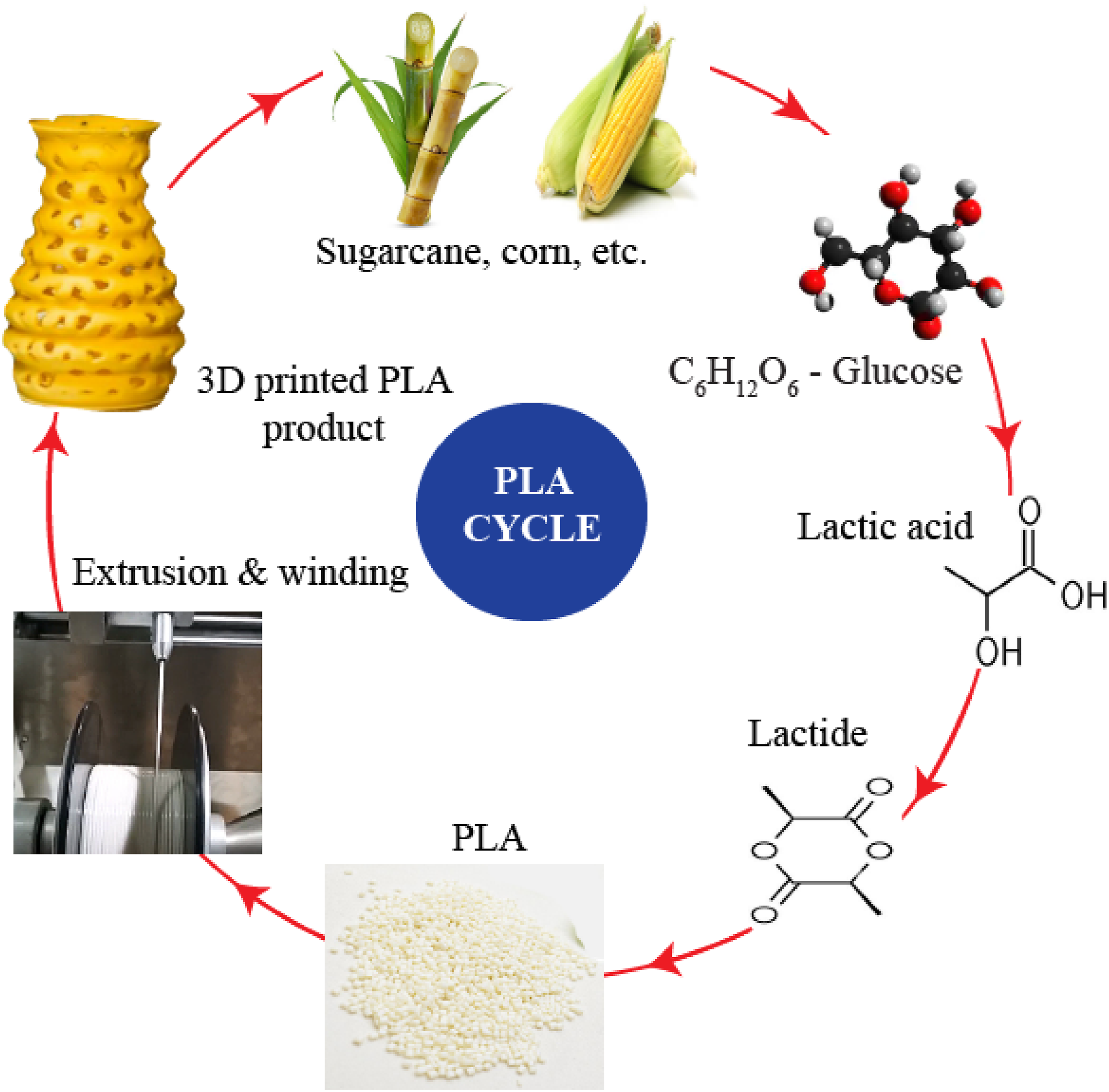
PLA is a non-aromatic hydrocarbon thermoplastic polymer derived from sustainable sources such as wheat, corn, rice, and sweet potatoes, as illustrated in Figure 2.
56
The average density of PLA is between 1.24 and 1.25 g/cm3, while its melting point varies from 147°C to 428°C. PLA offers several advantages, including low emissions and reduced energy consumption, which has led to its widespread use in medical applications.
57
Polylactic acid (PLA) is considered an attractive choice for applications interfacing with humans due to its biocompatibility, though it is regulated by the FDA through general biocompatibility guidelines (e.g., ISO 10993 testing) and product-specific pathways rather than dedicated material guidance; Class III devices require Premarket Approval (PMA), while Class II may use 510(k) clearance.
58
It can be transformed into products using various additive manufacturing techniques, although it does have limitations, such as mechanical weakness and water solubility.
10
PLA serves as an effective thermoplastic material for clinical applications at the human interface, particularly for eliminating microorganisms, viruses, and bacteria.
11
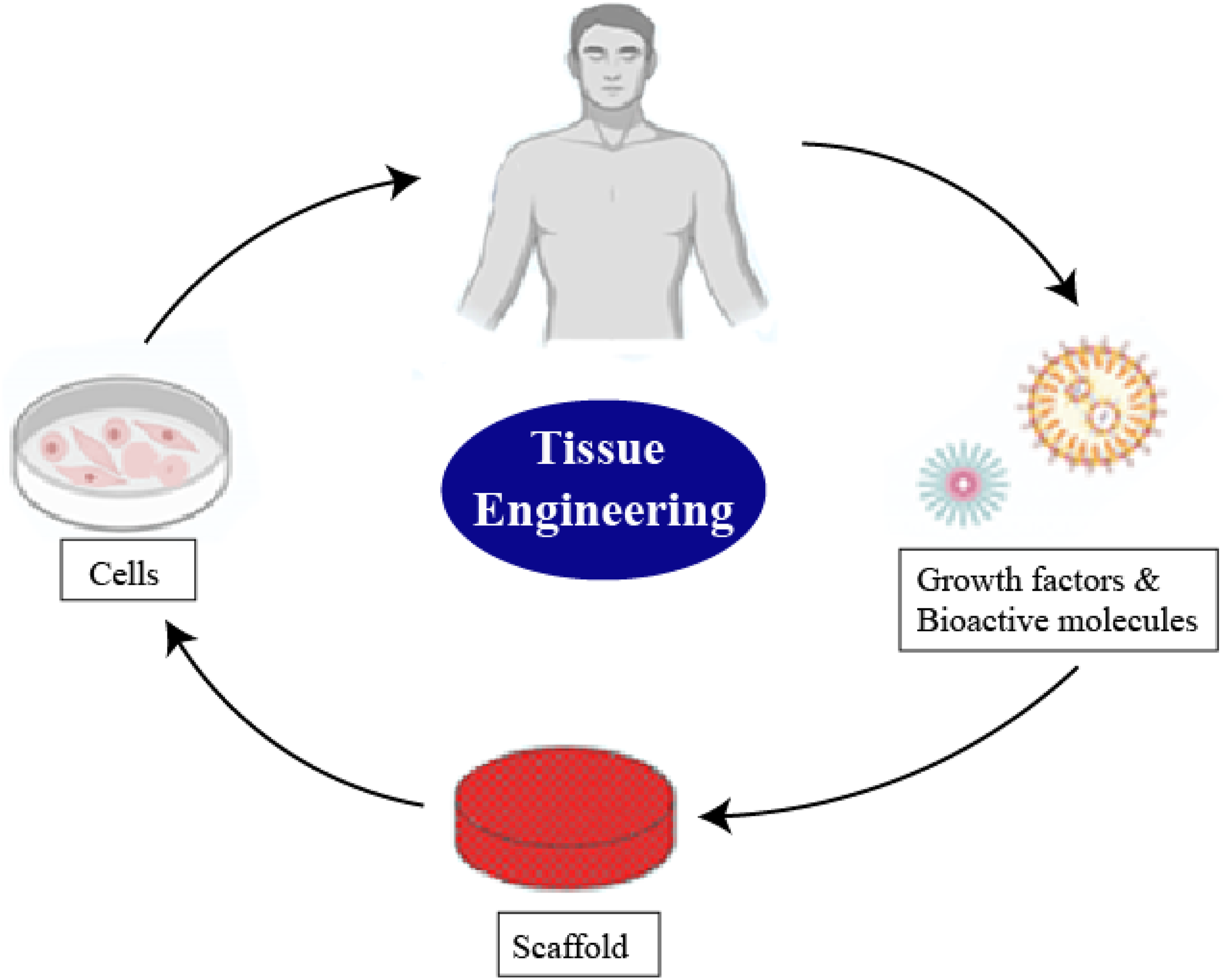
The sterilization of clinical devices involves the removal of fungi, plant cells, and spores. Effective sterilization remains a critical challenge in the clinical application of medical devices. Polylactic acid (PLA) is considered an attractive choice for applications interfacing with humans. Continual efforts by cell biologists, scientists, and engineers have fostered advancements in the use of thermoplastics as biomaterials. Scaffolds for tissue engineering, depicted in Figure 3,
12
are vital devices that facilitate the introduction of new cells and transplantation into living organisms. These scaffolds are essential because they provide a suitable environment for cells to attach, proliferate, and differentiate, enabling orderly tissue regeneration.
13
Polycaprolactone (PCL) is typically utilized as a scaffold material in tissue engineering due to its biocompatibility and ability to decompose through biological processes.14,15 To address the limitations of PLA, it is often combined with PCL, resulting in a design characterized by low hydrophobicity and improved mechanical properties.
16
Combinations of PCL and PLA, produced through soluble solutions and blending techniques, are commonly used to create scaffolds with an added mechanism for biodegradation known as bio-cell printing. In cell culture, PLA has effectively facilitated drug delivery and mechanical fixation processes over recent decades, often utilizing Fused Filament Fabrication (FFF) techniques. Thin polymeric films produced via FFF are tailored to the anatomical sizes of humans, particularly for applications involving disease treatment.
17
Additionally, products manufactured from PLA, including those for cell culture and drug delivery, are both cost-effective and enhance the reliability and performance of biomedical devices when produced through 3D printing. Recent years have seen significant advancements in research pertaining to thin films, especially those made from PLA, focusing on the optimization of process parameters, the effects of stacking and orientation, and variations in film thickness.
18
Though the thermoplastic properties of PLA are often modified through various blending techniques, its hydrophobic characteristics have not been fully addressed. Nevertheless, this issue has been mitigated by incorporating an adequate amount of porous gel, as the polymer’s high hydrophobicity is thought to be a major contributor to skin irritation and itching associated with PLA use. Moreover, PLA can be effectively combined with natural fibers to enhance thermal insulation, water resistance, antibacterial properties, and degradation resistance.
25
Consequently, designers and technologists in the field of 3D printing regard PLA as a highly promising biodegradable polymer. However, factors such as texture morphology, porosity, and surface chemistry are critical to the clinical application of PLA in specific natural fiber contexts. Polylactic acid (PLA) cycle. Scaffolding process in tissue engineering.
42
In the realm of modern materials, PLA emerges as a promising contender, cherished for its ability to serve as a sustainable and customizable biomaterial. It holds the potential to meet the demands of both traditional polymers and complex biomedical applications. The material’s adaptability shines particularly in additive manufacturing, where it integrates seamlessly with natural fiber reinforcements, allowing for creative customization through blending. This versatility positions PLA as a vital player in advanced clinical applications. As researchers continue to explore ways to enhance its mechanical, biological, and functional properties, the prospects for PLA grow even brighter, hinting at its influential role in personalized medicine, regenerative therapies, and the crafting of eco-friendly biomedical devices.
Polypropylene (PP)
Polypropylene (PP) is a versatile and rapidly expanding material due to its inherent properties. Its cost-effectiveness and exceptional characteristics make it attractive for applications across medicine, consumer electronics, packaging, and the automotive industry.
26
PP has greater strength than other polyolefins, with a high density of 0.9 g/cm3, and is recognized as one of the softest thermoplastics. Furthermore, polypropylene has a higher melting point (ranging from 160°C to 170°C) and superior heat resistance when compared to cheaper thermoplastics.
59
PP is resistant to most organic solvents and exhibits high resilience to alkalis and acids at certain temperatures. While typically opaque, polypropylene can also be dyed in a variety of colors. Polyolefins, including polyethylene and polypropylene, are susceptible to photooxidation when exposed to light, heat, and oxygen. However, extensive research has been engaged in the study of polymer degradation, particularly regarding polypropylene-based plastics, due to their extensive use across various industries, including medical and marine sectors.
60
For example, PP is used to manufacture numerous medical products, such as vials, capsules, and syrup containers filled with polylactic acid. PP medical devices are regulated by the FDA through biocompatibility testing per ISO 10993 standards and ECRI safety summaries, rather than material-specific guidance; Class II applications (e.g., surgical meshes) typically pursue 510(k) clearance, while Class III requires Premarket Approval (PMA).27,28 Additionally, polypropylene can be integrated into numerous medical devices, as shown in Figure 4. The use of nanomaterial-enhanced polypropylene has also enabled the design of several medical products with composite structures. Nevertheless, many of these medical devices fall under FDA hazard levels 1 or 2. This classification has allowed researchers and students, such as Khan et al.,
29
to understand not only the functionality of polyethylene and polypropylene but also their effectiveness in clinical applications. In this context, polypropylene optimized with multilayer carbon nanotubes and hydroxyapatite nanorods helps reinforce the creation of lightweight nanocomposites suitable for load-bearing applications, like bone implants. Liao et al.
30
examined the production and development of polypropylene composites reinforced with carbon nanorods and nanofibers. Their cell viability tests indicated that the biocompatibility of the PP/CNF–HANR hybrid composite was sufficient for osteoblasts. Specifically, the survival rate of osteoblasts with the PP/2% CNF–20 HANR hybrid material after 10 days of development was recorded at 78%, the highest observed rate. Such advancements allow bone implants to achieve their intended functions. A study by Ploypetchara et al.
61
compared the weight of PLA films with those of PP incorporating PP-g-MA (polypropylene grafted with maleic anhydride). The results demonstrated that blending PLA with PP improved vapor permeability while decreasing oxygen permeability. Isotactic polypropylene (IPP) is the second most widely used thermoplastic, and ongoing research is investigating its various potential applications.
31
This material is particularly prone to supercooling and can crystallize into multiple glassy structures, including monoclinic (α), hexagonal (β), orthorhombic (γ), and smectic mesophases. In industrial contexts, multiple inorganic fillers, additives, and modifiers are applied during rapid cooling to establish the monoclinic polypropylene base phase. High-performance polypropylene composites, featuring a central hexagonal phase, can be obtained by combining several nucleating agents with a limited number of inorganic fillers, requiring precise cooling conditions to achieve successful results. Overall, the versatility of polypropylene, together with advancements in hybridization technology and composite development, continues to enhance its importance in modern materials science. Various medical applications of polypropylene.
Propylene (PP) stands out as an essential material in 3D printing due to its beneficial properties, such as low density, remarkable durability, and excellent chemical resistance. These traits open up a wide range of applications, particularly in biomedical fields, packaging solutions, and industrial settings. Although the material faces challenges like warping and shrinkage during the printing process, recent innovations in surface treatments, adhesion promoters, and the incorporation of nanofillers have markedly enhanced its printability. Importantly, PP’s capability to create structures that are both lightweight and strong is crucial for developing functional prototypes, orthopedic devices, and housings for medical equipment. Furthermore, its semi-crystalline nature allows for precise adjustments to its crystallinity, facilitating the design of components with customized mechanical properties and enhanced barrier functions. Continuous research into hybrid composites and process improvements suggests a bright future for polypropylene in the additive manufacturing landscape. As it effectively bridges the gap between high-performance engineering polymers and cost-effective, biodegradable options, PP is poised to play a pivotal role in the advancement of 3D printing technologies.
Polycarbonate (PC)
Polycarbonate (PC) is a type of plastic utilized in medical devices due to its nonlinear mechanical behaviour, which is dependent on the strain rate.
62
The melting temperature ranges from 288°C to 316°C, and the density of polycarbonate is 1200 kg/m3. Applications of polycarbonate include catheters, vascular prostheses, artificial heart valves, hemodialyzers, bulletproof windows, food packaging, mineral water containers, and self-cleaning wires, among others. PC medical devices are regulated by the FDA via ISO 10993-1 biocompatibility evaluations and standards like ASTM F997, with most following Class II 510(k) clearance (e.g., catheters/hemodialyzers) rather than material-specific guidance; higher-risk devices may need PMA.32,33 Figure 5 illustrates these applications of polycarbonate in medical devices. This material is soluble in organic solvents such as dichloromethane (DCM), dimethylformamide (DMF), and tetrahydrofuran (THF), and it has a low critical angle (CA) content (∼84°C) in granulation water.
34
The use of polycarbonate in various products began to increase significantly in the 1960s; however, demand has declined recently due to the lack of adequate assembly equipment.
35
It is primarily used in mining vehicles as a substitute for hard rubber, the optimal material for battery casings. Its application in clinical devices commenced in the 1980s and continues today.
63
One major limitation of polycarbonates is their susceptibility to structural changes caused by gamma radiation or environmental stress cracking, both of which are affected by the physicochemical properties related to bacterial adhesion. Consequently, numerous studies have focused on the physicochemical properties of polycarbonate materials, with amphiphilic polycarbonates emerging as popular polymers used against gram-positive bacteria. These polymers possess two different binding sites, enabling effective interaction with both types of bacterial cell membranes. The antimicrobial activity of amphiphilic polycarbonate polymers stems from their ability to disrupt the membranes of gram-positive bacteria. This disruption occurs because of the amphiphilic nature of the polymers, allowing interaction with both the hydrophobic and hydrophilic layers of the bacterial cell membrane. Cheng, Chin, Dong, Xu, Zhong, Huang, Li, Xu, Wu, and Hedrick
22
explored the efficacy of biodegradable antimicrobial polycarbonates in combating multidrug-resistant Staphylococcus aureus (MRSA) infections, underscoring their potential as alternatives to traditional antibiotics. The study found that the polycarbonate pButyl 0.5 Benzyl 0.5 exhibited notable antibacterial activity against MRSA, along with lower toxicity than vancomycin in a mouse model. Akelshya et al.
23
investigated amphiphilic polycarbonates with primary amino groups, demonstrating their strong antimicrobial efficacy against multidrug-resistant gram-positive bacteria. This research highlighted their low toxicity to human cells, bolstered by high selectivity ratios and effective mechanisms disrupting bacterial membranes. Studies conducted by Amanda C. Engler et al.
64
have shown that the antibacterial activity of synthetic polycarbonates is significantly affected by their stability and hydrophobicity. Specifically, the OctCl homopolymer performed exceptionally well against various bacteria, while hemolytic activity increased with the polymer’s molecular weight. Ng, Tan, Leong, Voo, Hedrick, and Yang
65
researched the synthesis of biodegradable cationic polycarbonates quaternized with nitrogen-containing heterocycles, which exhibit broad-spectrum antibacterial activity against diverse pathogens. Additionally, these materials show high selectivity against animal pathogens, presenting a promising strategy for addressing multidrug-resistant infections and serving as a viable alternative to conventional antibiotics. These studies collectively suggest that synthetic polycarbonates, with their unique structural properties that demonstrate effective and non-toxic activity, offer significant potential as antibacterial agents against a wide range of microorganisms. Optimizing these materials could provide an alternative to traditional antibiotics while ensuring human safety. Difference polycarbonate devices used in medical field.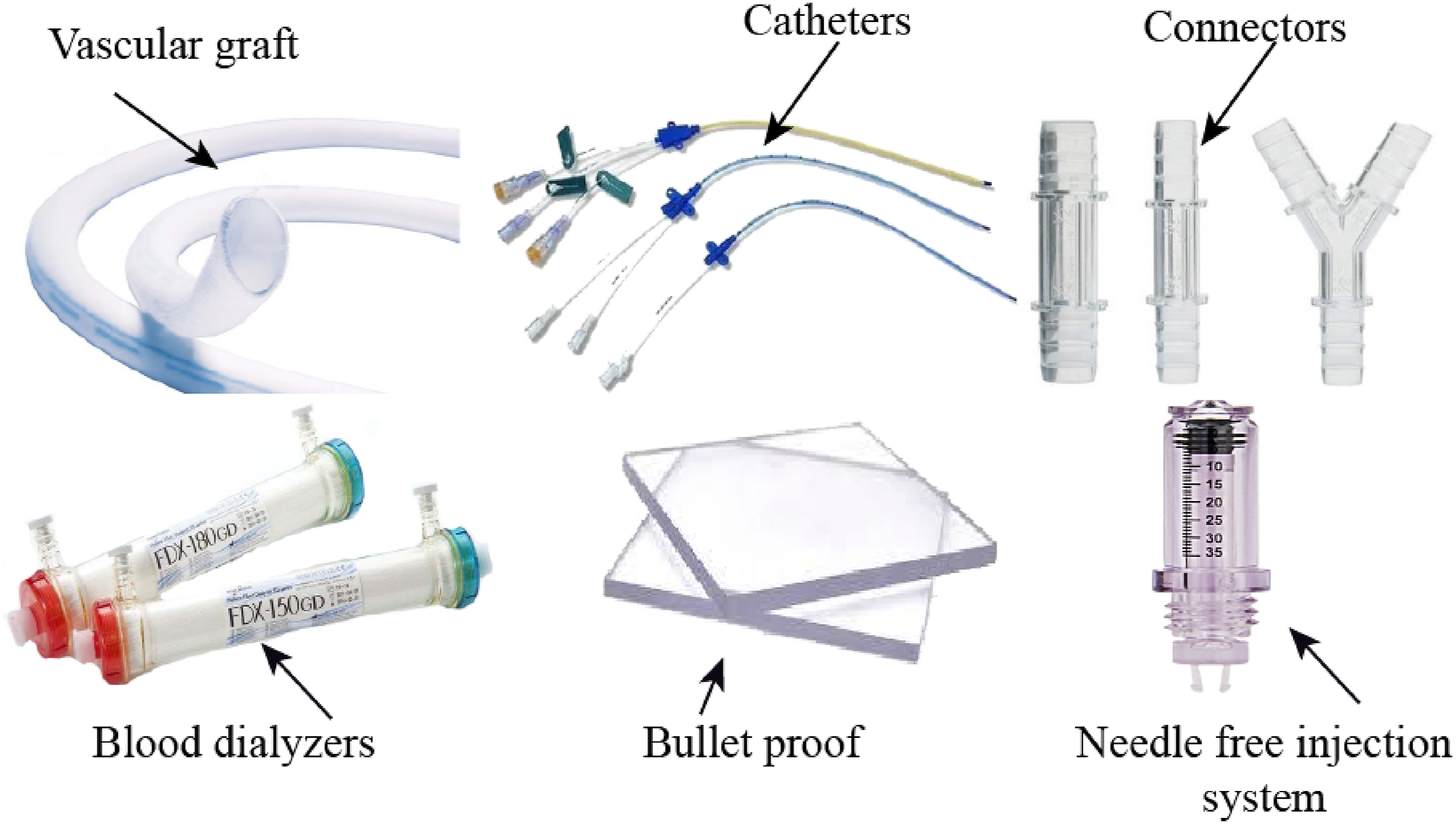
The unique characteristics of polycarbonate (PC) in 3D printing position it as a strong candidate for sophisticated biomedical and engineering applications due to its combination of strength, toughness, and transparency. In contrast to materials like PLA or PP, PC exhibits enhanced impact resistance and thermal stability, which are essential for producing robust clinical devices, prosthetics, and high-performance components. Although its widespread adoption has been hindered by challenges such as warping, high processing temperatures, and a vulnerability to radiation-induced degradation, significant progress in print-bed adhesion techniques, extrusion optimization, and the introduction of composite reinforcement with nanofillers has markedly improved the printability of PC. Additionally, the capacity to integrate amphiphilic and antimicrobial modifications into PC matrices expands its applicability for creating infection-resistant implants and medical housings. Given these ongoing advancements in additive manufacturing, polycarbonate is poised to emerge as a premier material for the next generation of 3D-printed biomedical devices, particularly in applications demanding high mechanical reliability and antibacterial capabilities.
Polyurethane (PU)
World War II began in 1939, but 2 years prior, Otto Bayer and his team made a groundbreaking contribution to material science by developing polyurethanes.
66
This innovation paved the way for significant advancements in various applications, primarily due to their favorable properties and ease of processing. The polyurethanes industry originated in 1937 with the formulation of diisocyanate-polyether diols, showcasing that polyurethane could be synthesized through a complex series of 37 reactions involving diisocyanate- and hydroxyl-terminated primers.
24
By 1938, this led to the creation of the first polymer coatings using polyurethane systems for textiles and paper, effectively rendering them resistant to tear gas. Polyurethane coatings are now widely regarded for chemically treating a range of materials, including stone, wood, and metals, as they provide a protective layer on high-quality metals. The versatility of polyurethane makes it applicable across multiple sectors, from footwear to aircraft components. Polyurethanes are recognized for their resilience, strength, and chemical resistance, offering mechanical properties that often exceed those of many metals, plastics, and rubbers. Their unique flexibility positions them as excellent choices for a broad spectrum of applications, including biomedical devices, automotive parts as presented in Figure 6. PU medical devices are regulated by the FDA via ISO 10993-1 biocompatibility evaluations and standards like ASTM F624, with most pursuing Class II 510(k) clearance (e.g., catheters/dressings) or PMA for Class III (e.g., pacemaker leads) rather than material-specific guidance.
67
The various marketed forms of polyurethane—such as polycarbonate polyurethane, acrylate polyurethane, polyurethane polyol, and polyester urethane—serve distinct functions. Specifically in healthcare, synthetic polyurethanes demonstrate greater stability compared to other medical-grade alternatives, with polycarbonate formulations exhibiting admirable biocompatibility and biostability. Their use extends to packaging for medical instruments such as pacemakers, scaffolds, breast implants, gastric balloons, catheters, nerve devices, dressings, and ophthalmic tools.
68
The production of PU scaffolds employs several manufacturing techniques, including solvent casting, pellet extrusion, and gas expansion to create a porous structure. However, these methods might limit the production of thin scaffolds or intricate networks. In contrast, advancements in 3D printing technology allow precise control over scaffold dimensions and pore architecture, which can significantly enhance tissue responses, including vascularization and nutrient supply.
69
Various 3D printing techniques like stereolithography (SLA), Powder Bed and Inkjet Head printing (PIP), selective laser sintering (SLS), fused deposition modeling (FDM), and liquid freeze-drying (LFDM) exist, each with specific benefits and limitations regarding material compatibility and residue concerns. While some methods can retain toxic photoinitiators, others might compromise biodegradable substrates due to high processing temperatures. Low-temperature printing of biodegradable thermoplastic polyurethane (TPU) presents options for creating scaffolds with remarkable cell compatibility and the ability to encapsulate bioactive factors. TPU materials are both biocompatible and hemocompatible, making them strong candidates for medical applications. Despite their advantages, the use of thermoplastic polyurethane in medical devices can have serious drawbacks, such as pathogen adhesion and biofilm formation.70,71 With millions of healthcare-associated infections reported annually in Europe and the U.S., addressing growing bacterial resistance and immunocompromised populations is paramount. To combat these issues, PU-based polycarbonate has been engineered to withstand hydrolysis and oxidation better than polyether polyurethane. Additionally, incorporating biodegradable components such as cellulose, alginate, and starch into polymer matrices can create biodegradable PU. This combination facilitates hydrolysis of ester bonds, allowing for degradation at a controlled rate. Although biodegradable PU has promising applications, its production processes must be refined to mitigate the release of acidic byproducts and harmful residues. Factors influencing biodegradation within the body include enzymatic degradation, oxidation, and hydrolysis, which can impact the mechanical properties and safety of PU in medical applications, highlighting the need for careful material selection and innovative processing methods.
19
Polyurethane medical applications.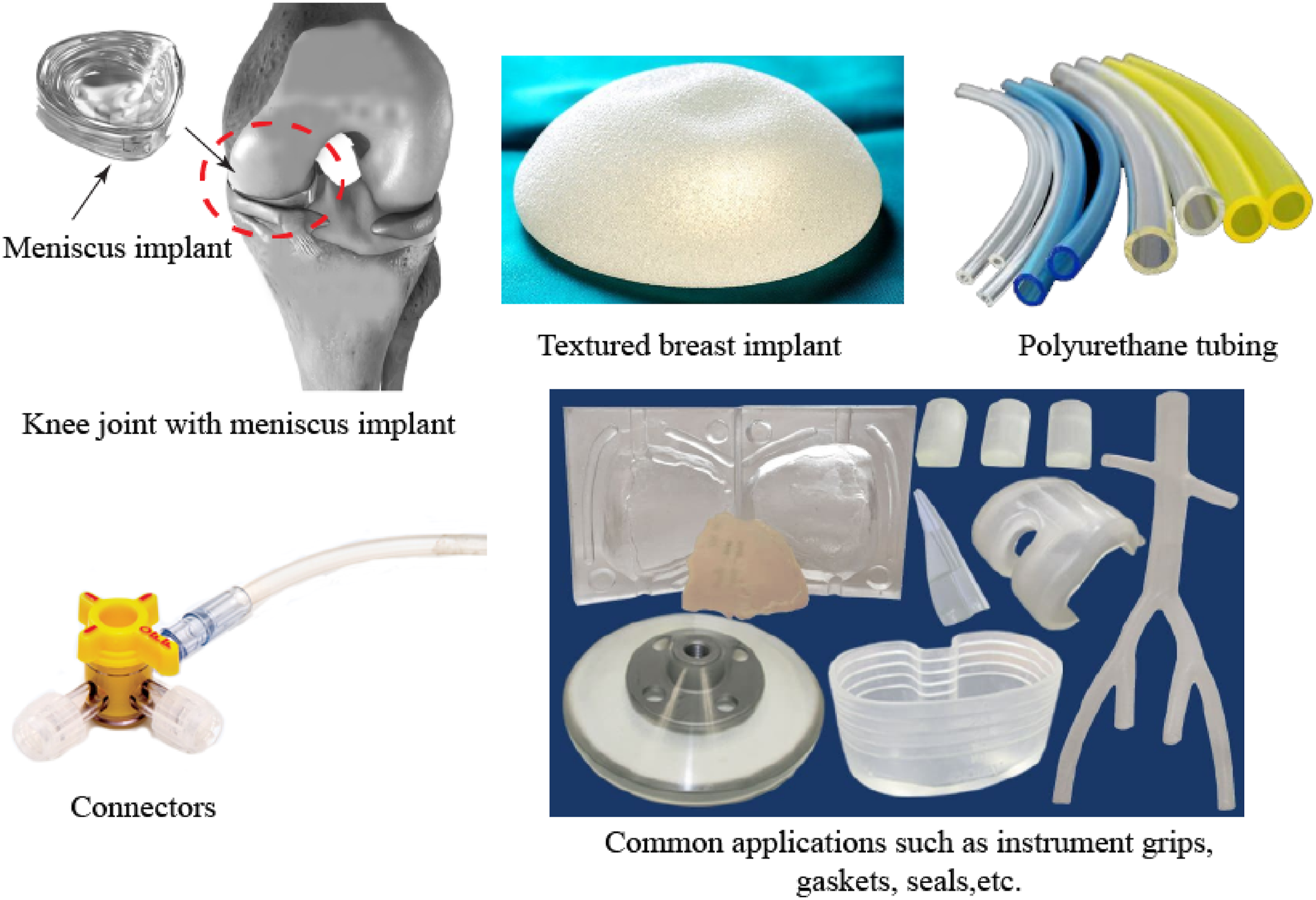
In conclusion, polyurethane (PU) exemplifies a cutting-edge material in the realm of 3D printing, primarily due to its remarkable flexibility, biocompatibility, and customizable mechanical properties, which align seamlessly with the demands of biomedical applications that require both elasticity and durability. The unique characteristics of thermoplastic polyurethane (TPU) facilitate the development of patient-specific scaffolds, implants, and soft-tissue devices, enabling precise manipulation of pore structures and mechanical gradients. Unlike the more brittle polylactic acid (PLA), PU maintains its elasticity under repetitive stress, rendering it highly suitable for cardiovascular, orthopedic, and soft-tissue engineering. Recent advancements in low-temperature printing techniques and hybrid composite materials further enhance PU’s capabilities, allowing for the incorporation of bioactive agents that promote tissue regeneration and enable controlled drug delivery. Despite challenges such as biofilm formation and issues related to degradation byproducts, ongoing research into antimicrobial treatments and biodegradable PU formulations underscores its potential as a next-generation additive manufacturing material. With its versatile compatibility across various 3D printing methods and its ability to replicate the mechanical properties of native tissues, polyurethane emerges as a leading contender for future innovations in the biomedical field.
Acrylonitrile butadiene styrene (ABS)
ABS, or acrylonitrile-butadiene-styrene, is a prominent amorphous polymer known for its excellent impact resistance, which makes it a popular choice in 3D printing.
72
A schematic representation of ABS is presented in Figure 7. This polymer combines three critical monomers: acrylonitrile, butadiene, and styrene, resulting in a material that boasts rigidity, good impact resistance even in low temperatures, and high abrasion resistance. ABS typically melts between 190°C and 220°C but should not be heated beyond 246°C.
20
It can be molded into various shapes following heat treatment, contributing to its versatility in numerous applications, particularly in the automobile, electronics, and transportation industries. Recent studies emphasize the medical applications of ABS and are presented in Figure 8. ABS medical devices are regulated by the FDA through ISO 10993 biocompatibility evaluations, with favorable reduced testing for intact skin contact (e.g., boluses, external guides) via documented safe use—no full cytotoxicity required for ≤30 days contact; most pursue Class I/II 510(k) or exemption rather than material-specific guidance.21,40 Clinical devices such as breathing instruments, miniature implants, and autoinjectors are produced using this material. However, ABS presents limitations in terms of weight reduction for specific applications. Research by Aung, Churei, Tanabe, Kinjo, Togawa, Li, Tsuchida, Tun, Hlaing, Takahashi and Ueno
36
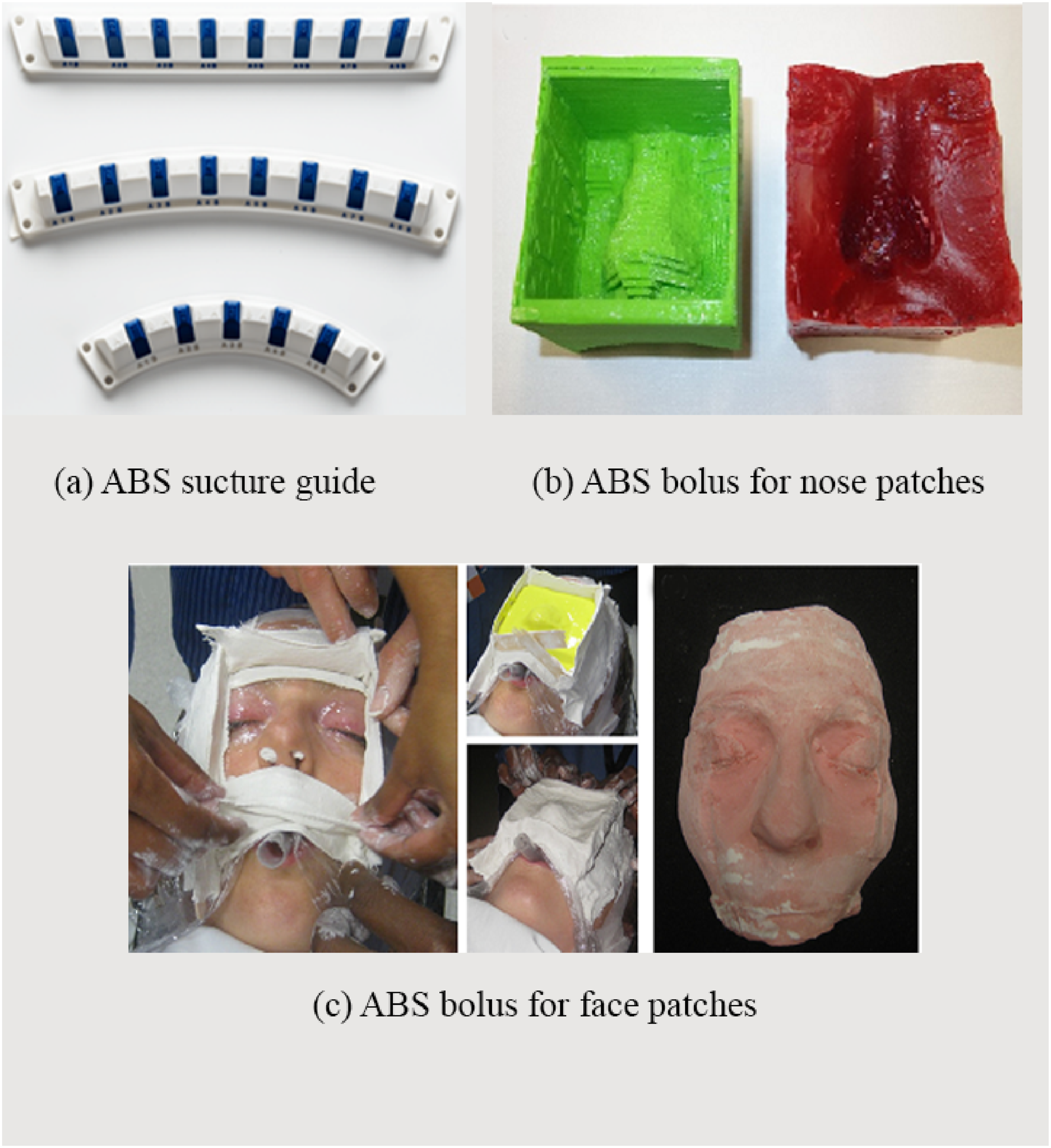
focused on enhancing both weight reduction and ventilation in sports faceguards. Also, in cardiology, ABS sutures are utilized in devices implanted within patients’ anatomies. Custom-designed suture guides made from ABS are employed during crucial heart valve surgeries, chosen for their stiffness and rigidity. Unfortunately, these guides faced rejection from the ANSM due to design flaws, prompting ongoing research for more effective designs. In radiation therapy, tissue-equivalent boluses made from ABS are reshaped and applied to a patient’s skin, aiding in a uniform radiation dose distribution while treating neoplastic lesions.
41
Customized ABS boluses produced through 3D printing reportedly outperform traditional paraffin wax by soothing the skin prior to treatment. These boluses can also be filled with water and paraffin, mimicking the properties of biological tissues.
42
The 3D printed ABS boluses are presented in Figure 9. However, potential issues like dimensional contraction or mechanical property alterations can arise due to irradiation. Suture guides made from ABS play a crucial role in this field. Moving forward, techniques such as magnetic resonance cholangiopancreatography (MRCP) are utilized to prevent biliary injuries before laparoscopic cholecystectomy. Nevertheless, high costs and limited availability lead to the exploration of alternative imaging techniques. Silicone materials are used to create biliary tree models that assist surgeons in surgical planning and improving their training. The rise in global surgical simulation highlights the importance of these training methodologies for enhancing the skills of novice surgeons while ensuring patient safety. While ABS has numerous medical applications, it also exhibits certain limitations. At temperatures above 250°C, ABS can decompose into its original monomers, some of which are potentially carcinogenic.
37
Moreover, 3D-printed ABS components often lack sufficient fatigue resistance, failing under repeated stress, which necessitates material enhancements before its extensive use in additive manufacturing. To address these challenges, modifications using additional compounds like polycarbonate, mica, clay, and zinc oxide are being explored, alongside fillers like carbon nanotubes or graphite powders. Blending ABS with polycarbonate has shown potential for improved processability and thermal stability, fostering the development of polymer blends within the Fused Deposition Modeling (FDM) domain.38,39 These advancements aim to enhance the capabilities of 3D printing materials while broadening their functional properties. Enhancements in ABS’s piezoelectric properties through graphene blending also represent a promising area of research, with studies indicating improved thermal stability and reduced melt flow index in ABS composites.
73
Schematic representation of ABS constituents & its properties. Various ABS medical applications. 3D printed ABS boluses.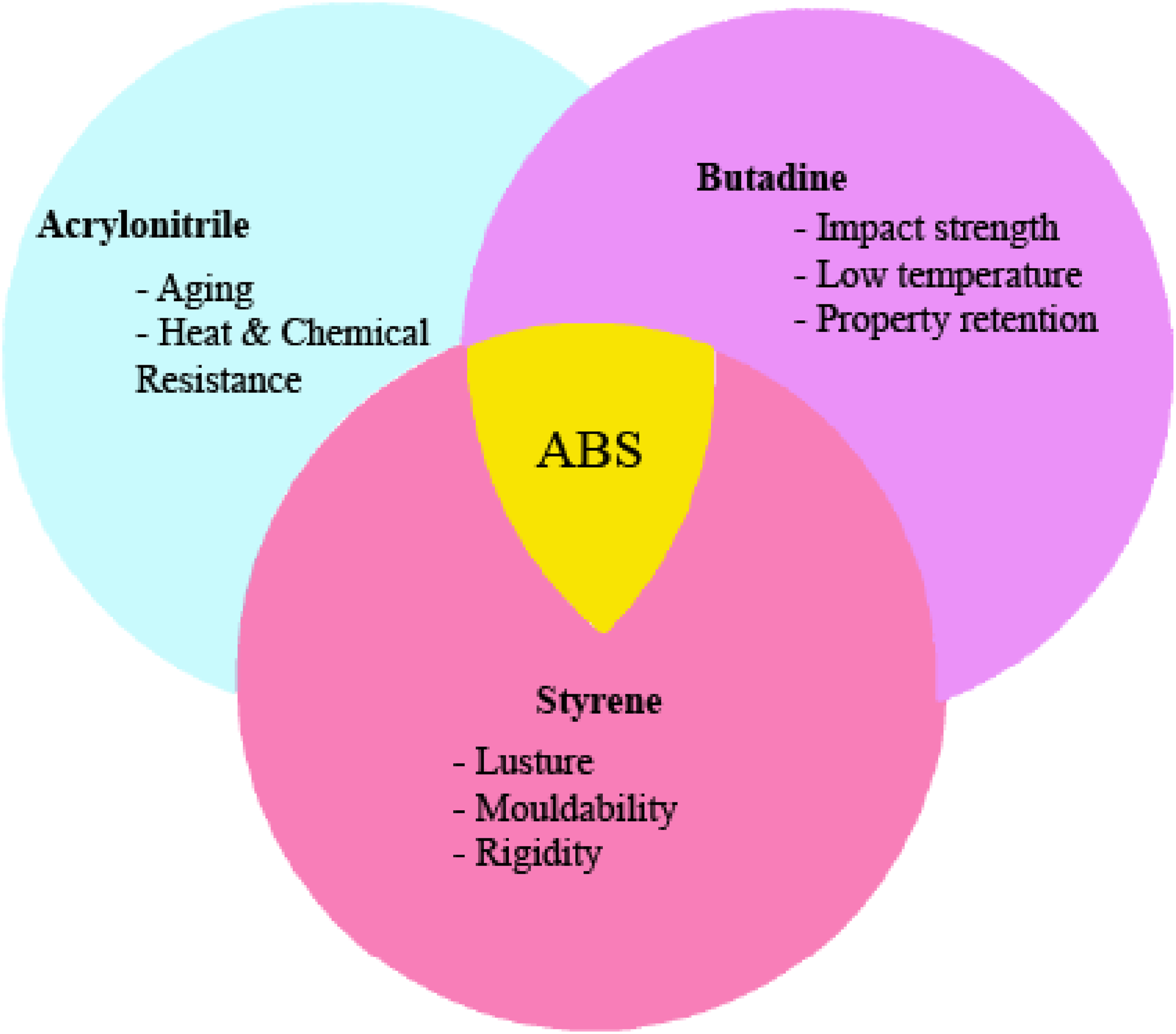

In summary, ABS is a highly versatile and durable engineering polymer that outperforms biopolymers like PLA in terms of toughness and thermal resistance, making it ideal for long-lasting applications. Despite challenges such as fatigue resistance and hazardous emissions during processing, ongoing innovations—like blending with other materials and incorporating nanofillers—are enhancing its mechanical and thermal properties. As composite development and 3D printing techniques advance, ABS is poised to become increasingly significant in biomedical engineering, particularly for applications in surgical planning, customized devices, and multifunctional therapeutic solutions.
Polyetheretherketone (PEEK)
Polyetheretherketone (PEEK) is a semicrystalline thermoplastic polymer characterized by its polycyclic aromatic structure (–C6H4–O–C6H4–O–C6H4–CO–), making it highly suitable for biomedical applications due to its mechanical properties.
43
The polymerization of PEEK occurs via a stepwise dialkylation reaction involving bis-phenolates, allowing for various formulations to be created. Modifications to enhance its properties can be achieved through the addition of functionalized monomers during pre-polymerization or post-polymerization chemical processes like sulfonation, amination, and nitration. PEEK medical devices are regulated by the FDA via ISO 10993 evaluations and ASTM F2026 standards, with spinal/orthopedic implants typically following Class II 510(k) clearance via predicates or PMA for novel designs rather than material-specific guidance. PEEK has been employed in multiple surgical fields, including spine surgery, orthopaedics, and maxillofacial surgery, as shown in Figure 10.
43
As a viable alternative to traditional metal or ceramic implants, PEEK composites can incorporate bioactive materials such as hydroxyapatite (HA) and tricalcium phosphate, expanding its use across all surgical specialties. Notable properties of PEEK include biocompatibility, durability, low moisture absorption, temperature resistance, flexibility, and elasticity. These qualities make it ideal for crafting medical frames for implants and craniofacial reconstructions using selective laser sintering (SLS).
74
For effective reconstruction techniques, it is essential to achieve complete tumor removal while minimizing complications. Integrating PEEK with carbon from fused deposition modeling (FDM) has shown improved mechanical properties and biocompatibility compared to pure PEEK, with enhancements in tensile and bending strengths and cell viability exceeding 96%.
44
Efforts to utilize 3D printing technology in developing patient-specific surgical implants focus on layer-by-layer fabrication, allowing intricate designs that traditional methods cannot achieve. However, creating bioactive PEEK composites via conventional methods poses challenges in bioactive phase distribution and microstructure control, potentially limiting osseointegration and mechanical property enhancement. Effective dispersion of HA within the PEEK matrix is vital for connectivity, with an optimal calcium hydroxyapatite (cHAp) content of approximately 15 wt% ensuring mechanical integrity while promoting biological performance suitable for load-bearing in orthopaedic implants. Additionally, incorporating reduced graphene oxide (rGO) and cHAp has been found to further improve mechanical properties and support cell growth.
45
Alterations in FDM parameters, including nozzle temperatures and layer thickness, can mitigate structural flaws like gaps and tension pipes, ultimately enhancing the performance of PEEK composites. Furthermore, research indicates that specific additive manufacturing defects can adversely affect mechanical properties, leading to localized damage and crack formation. The design of personalized cranial implants for unique skull anatomies presents significant challenges. Implants must fulfill aesthetic and functional criteria, particularly for asymmetrical defects, while demonstrating bone bonding capacity and strength. PEEK’s high fitting accuracy (with deviations as low as 0.5919 mm) and low biomechanical stress provide a promising alternative to titanium in cranial applications.
46
Crystallinity is crucial, as elevated processing temperatures during 3D printing can increase it significantly, enhancing mechanical properties. Studies revealed that raising temperatures from 25°C to 200°C could shift crystallinity from 16.937% to 32%. Consequently, developing porous PEEK structures is essential for improving osseointegration, mimicking the features of cancellous bone and enhancing cell anchorage. Despite its biologically inert surface, which may lead to poor cellular response, PEEK is favored in clinical dentistry for its aesthetic benefits and excellent biomechanical characteristics. It is also gaining popularity in the manufacturing of facial devices, although creating components involves complexities due to the need for precise thermal control. To further enhance PEEK properties, it is often reinforced with short and long-woven fibers to minimize wear.
75
Investigations have indicated that optimal processing conditions contribute to better mechanical performance and reduced porosity, particularly for carbon-fiber-reinforced composites produced through 3D printing, which can suffer from crack formation.
76
In conclusion, PEEK is a highly promising biomaterial in orthopaedics, celebrated for its superior thermal strength, toughness, and ease of shaping through various processing techniques, including injection molding and additive manufacturing. PEEK medical applications.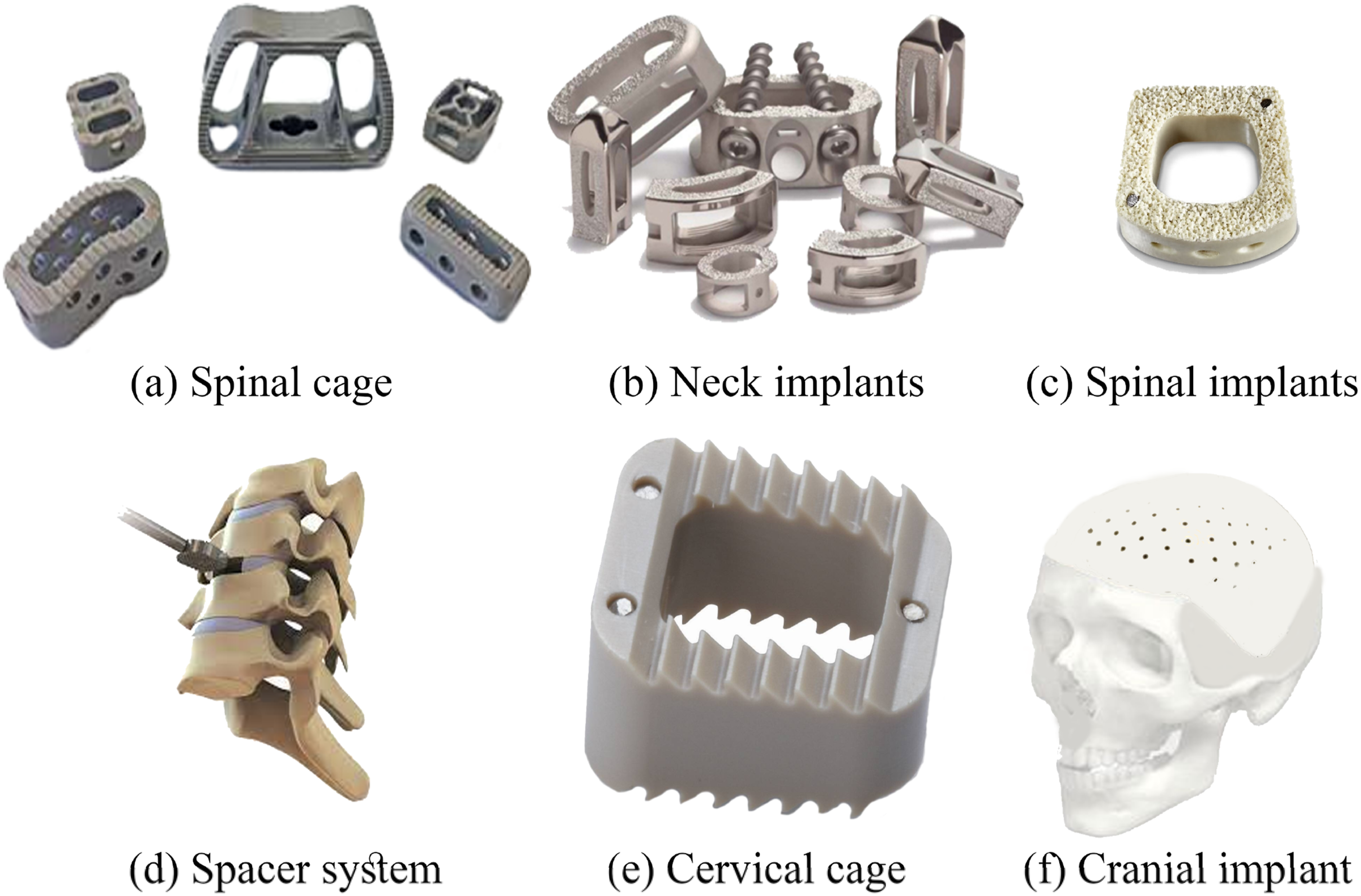
PEEK’s unique combination of metal-like strength and polymer-like processability positions it as a leading candidate for next-generation biomedical implants. It surpasses traditional polymers such as ABS and PLA in thermal stability, biocompatibility, and durability in physiological environments. Enhancing PEEK with fillers like HA, rGO, or carbon fibers improves its bioactivity and osseointegration, making it suitable for applications in load-bearing orthopedic devices and complex craniofacial reconstructions. As advancements in additive manufacturing continue, PEEK presents the opportunity to replace conventional implant materials, providing customized, lightweight, and durable options for individual patient needs.
Poly vinyl alcohol (PVA)
Polyvinyl alcohol (PVA) is a water-soluble polymer recognized for its excellent biocompatibility, making it a suitable drug carrier in numerous medical applications, as illustrated in Figure 11. PVA medical devices are regulated by the FDA via ISO 10993 biocompatibility standards and Class II 510(k) clearance (e.g., embolic microspheres, scaffolds), leveraging GRAS status for pharma apps rather than material-specific guidance; successful clinical uses include vascular embolization particles for AVMs/uterine fibroids (e.g., K032619 MicroStat™, K250209 Canyon Microspheres) and wound dressings/sponges.77,78 In modern pharmaceutical practices, when patients request customized drug regimens, PVA is often used in combination with paracetamol to form caplets that deliver single or multiple drugs with a targeted release profile. Typically, these drug-loaded profiles are fabricated using 3D printing techniques. A significant challenge in this process is maintaining the quality of the medication due to the high temperatures involved in 3D printing. Many studies have investigated this concern for various drug formulations.
79
To mitigate this issue, most researchers have employed insoluble drug containers to shield the drug from the elevated temperatures during printing. However, these studies also indicated that preparing tablets through 3D printing could result in chemical transformations.
80
Additionally, the layer-by-layer creation of such tablets is both complex and time-consuming. Recently, numerous researchers have explored the use of PVA cross-linked with cellulose nanofibers to develop highly biodegradable scaffolds, especially for the creation of multidrug formulations. Hydroxyapatite is frequently utilized in the production of 3D-printed scaffolds for the repair and replacement of intricate tissues and bones.81–83 Common materials for scaffold manufacturing in 3D printing include collagen, alginate, and gelatin, which are sometimes combined with cellulose nanofibers to enhance their biocompatibility and biodegradability. Scaffolds typically feature 3D porous structures that facilitate biological tissue regeneration. Various 3D printing techniques have been examined for advancements in modern medical practices, including stereolithography and selective laser sintering. Among these, fused deposition modeling (FDM) has emerged as a promising method, particularly for fabricating PVA-based 3D printed scaffolds, due to its ability to accommodate amorphous solid dispersions and melt granulation. Goyanes, Chang, Sedough, Hatton, Wang, Buanz, Gaisford and Basit
84
developed a PVA-based drug carrier utilizing fluorescein with different infill patterns and weights through the FDM technique. They found that the loading efficiency of the PVA-based drug carrier was low, indicating that the FDM method may not be suitable for producing fluorescein-based PVA carriers. Similarly, Liu, Li, Liu, Yuan, Yang, Zhou, Hu and Yang
85
created a chitosan/polyvinyl alcohol porous structure via 3D printing to enhance the mechanical properties and minimize the swelling behavior of hydrogel scaffolds. Their findings suggested that increasing the percentage of PVA in the 3D printing process elevated the viscosity, while still allowing the porous structure to exhibit favorable characteristics for sustained drug release. Additionally, Luo, Le Fer, Dean and Becker
86
fabricated 3D-printed alginate/polyvinyl alcohol scaffolds by adding a CaCl2 solution, which created numerous micropores to improve drug delivery. They demonstrated that the incorporation of 3D-printed micropores significantly enhanced water absorption. Hence, PVA showcases good biodegradability and chemical stability, making it suitable for blending with solutions or hydrogels to strengthen mechanical properties or accelerate drug delivery. PVA drug carrier used in medical applications.
PVA shows considerable potential in 3D printing as a drug delivery matrix and biodegradable scaffold. By combining PVA with bioactive fillers and natural polymers, it is possible to enhance characteristics like degradation rate, mechanical strength, and drug release tailored to individual patient requirements. Its properties of water solubility and biocompatibility render it suitable for temporary support structures in tissue engineering, facilitating gradual resorption. Given the progress in low-temperature printing techniques and composite formulations, PVA is expected to be integral in advancing personalized medicine, bioresorbable implants, and novel solutions for tissue regeneration.
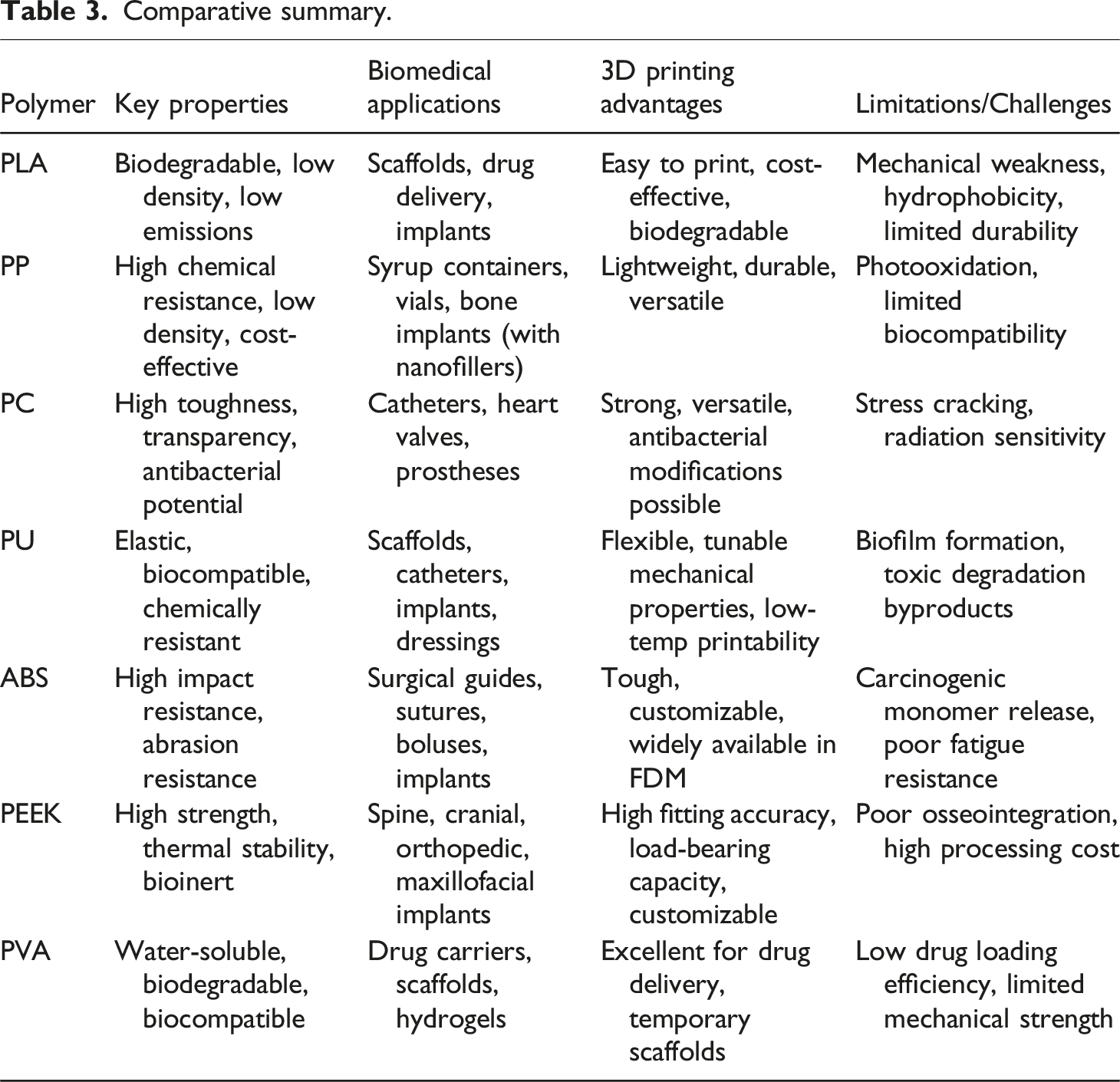
Comparative summary
Comparative summary.
Common biocompatibility assays and their grade level.

Future scope
Current advanced manufacturing technologies, inspired by computed tomography, enable the simultaneous printing of large-volume objects within a reduced production timeframe. Additionally, the surface quality and mechanical properties of these objects have seen significant improvements. Thorough research and exploration of volumetric printing can elevate 3D printing to unprecedented levels, allowing designers to bring their visions to life in just a few minutes. This capability enables the fabrication of incredibly complex, custom-made products, thereby broadening their potential applications. Promising future uses include commercial buildings, agricultural machinery, and wastewater filtration systems. The production of human organs, bones, and tissues through 3D printing eliminates the dependency on donors. Likewise, astronauts and space explorers are striving to enhance their longevity on other planets, and 3D printing presents a remarkable opportunity; lunar materials could be utilized to construct habitats and various devices on the Moon. Research into 3D printing is vital for discovering new technological advancements and exploring potential future uses. Essential challenges, including large-volume production, precise material specifications, and optimal process parameters, must be addressed to successfully adopt this innovative technology. To broaden applications, new materials with desirable properties need to be developed. Ultimately, 3D printing stands as a pivotal technology for a nation’s economic growth and plays a crucial role in both current and future industrial revolutions.
Conclusion
This review primarily focuses on biomedical production using 3D printing technology. The literature review highlights challenges related to the fabrication and design of biomedical components utilizing various polymeric materials. It aims to explore the materials, methods, conflicts, and potential of different types of 3D bioprinted components. This study contributes the following insights: - An end-user may seek a custom-fitted prosthesis that can be produced through 3D printing technology without the manufacturing delays often associated with traditional prostheses. - The 3D printed templates that support bone, dental, and orthopedic structures can be adjusted to simulate bone-like characteristics, prioritizing internal bone structure over a hollow design. - 3D printing is transforming the healthcare sector by enabling the creation of intricate and innovative features, such as personalized medical devices that can cut down on time and enhance accuracy.
However, the technology faces limitations related to the cost of devices and the time needed for manufacturing, particularly in urgent situations. Another constraint arises from the use of individually designed implants and specialized implant guides, especially in assistive surgeries, where planned templates often restrict motion—something that is preferred in modified scenarios. The findings from the review indicate that nearly any type of polymer form could find applications in the medical, aviation, energy, and chemical industries. Nevertheless, despite these possibilities, many sectors are still in the conceptual stage, with a continual need for the development of polymers tailored to meet a wide array of demands, including entirely new types.
Footnotes
Consent for publication
The authors affirm that there are no external data or images incorporated in the present project for publication.
Author contributions
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this study.
