Abstract
The application of Vitrimer in carbon fiber composite materials can effectively improve the fatigue resistance of composite materials and extend their service life. Characteristics, synthesis methods, and application research of carbon fiber reinforced glass like polymer (Vitrimer) materials composites were reviewed in this paper. Different types of Vitrimers and their mechanical and self-healing properties based on different bond exchange reactions were also briefly described. Furthermore, the fatigue failure mechanism of carbon fiber composite materials was demonstrated. The potential of Vitrimer materials in suppressing fatigue crack propagation and enhancing the structural strength of composite materials was elucidated. Finally, based on the advanced properties and future development trend of Vitrimer, the extensive application prospects of this material in carbon fiber composites and other fields are prospected.
Introduction
Carbon fiber is a high-performance material composed of over 92% carbon, characterized by its anisotropic structure, high strength-to-weight ratio, and superior mechanical properties, including thermal stability, corrosion resistance, and electrical conductivity.1,2 Unlike conventional carbon materials, its woven and lightweight nature allows for reinforcement in various matrices such as polymers, 3 metals, 4 ceramics, 5 and composites. Among these, carbon fiber-reinforced polymer (CFRP) composites are widely utilized due to their exceptional mechanical strength, fatigue resistance, and oxidation stability. However, like all engineering materials, CFRPs are subject to degradation under mechanical stress, thermal exposure, and environmental corrosion, necessitating the development of advanced matrices to enhance their durability and service life.
A significant challenge in polymer science has been balancing reprocessability and structural integrity in composite materials. Traditional resin-based carbon fiber composites, relying on thermosetting matrices with rigid cross-linked networks, 6 suffer from irreversible curing that renders them non-self-healing and non-recyclable. We can only rely on traditional mechanical 7 or thermal 8 degradation methods, which cause significant environmental impacts. To overcome these challenges, dynamic covalent polymers have emerged as a promising alternative, culminating in Leibler and co-workers’ 2011 introduction of Vitrimers - a novel class of materials bridging the gap between thermosets and thermoplastics. 9 These materials undergo dynamic bond exchange through dissociative and associative mechanisms, 10 with the latter enabling temperature-dependent viscosity modulation via reversible covalent bonds in glass-like polymer networks. 11 This unique behavior, facilitated by Covalent Adaptive Networks (CAN), 12 allows Vitrimers to impart self-healing capabilities and recyclability while maintaining thermoset-like stability. The integration of such dynamic resins into carbon fiber composites not only addresses the critical issues of fatigue damage and limited service life but also establishes a foundation for sustainable cyclic utilization through their reprocessable nature.
The integration of self-healing Vitrimers into CFRPs was reviewed in this paper, focusing on their role in mitigating fatigue-induced damage and enabling sustainable reprocessing. Key synthesis strategies, bond-exchange mechanisms, and their effects on interfacial adhesion, fatigue resistance, and thermal stability are critically analyzed. As it shown in Figure 1(a),(b), the current research focuses on introducing associative exchange and dissociative exchange into the interfacial region of carbon fiber composites. Furthermore, the advantages and limitations of current healing methodologies—including extrinsic (capsule-based) and intrinsic (dynamic bond-enabled) systems—are compared. By evaluating recent research progresses, this paper aims to guide the development of next-generation CFRPs that balance durability, recyclability, and industrial scalability. (a) Associative exchange and (b) Dissociative exchange in dynamic covalent bonds.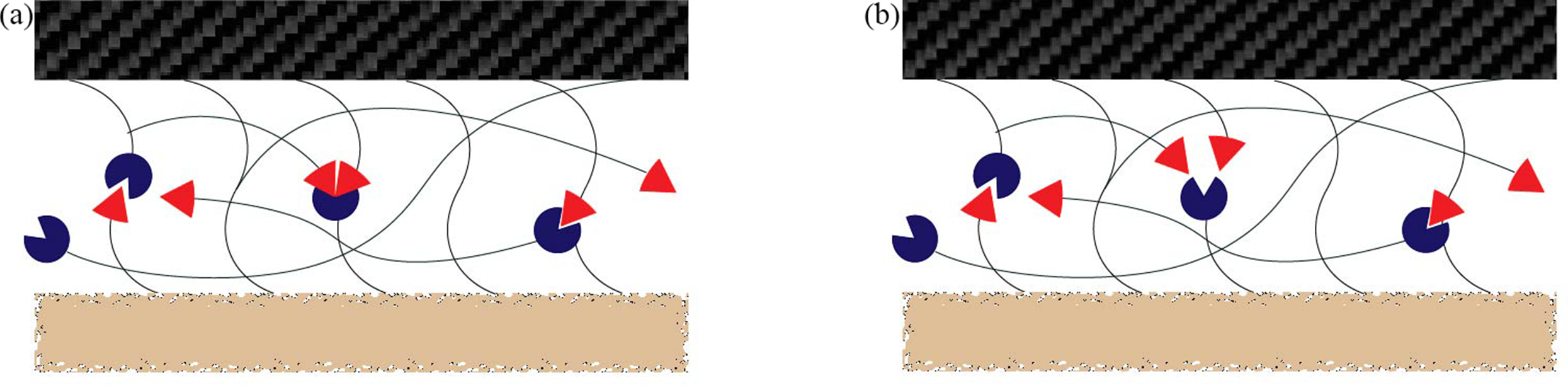
Study on the preparation and properties of Vitrimer
Comparison of properties of vitrimers with different main functional groups.
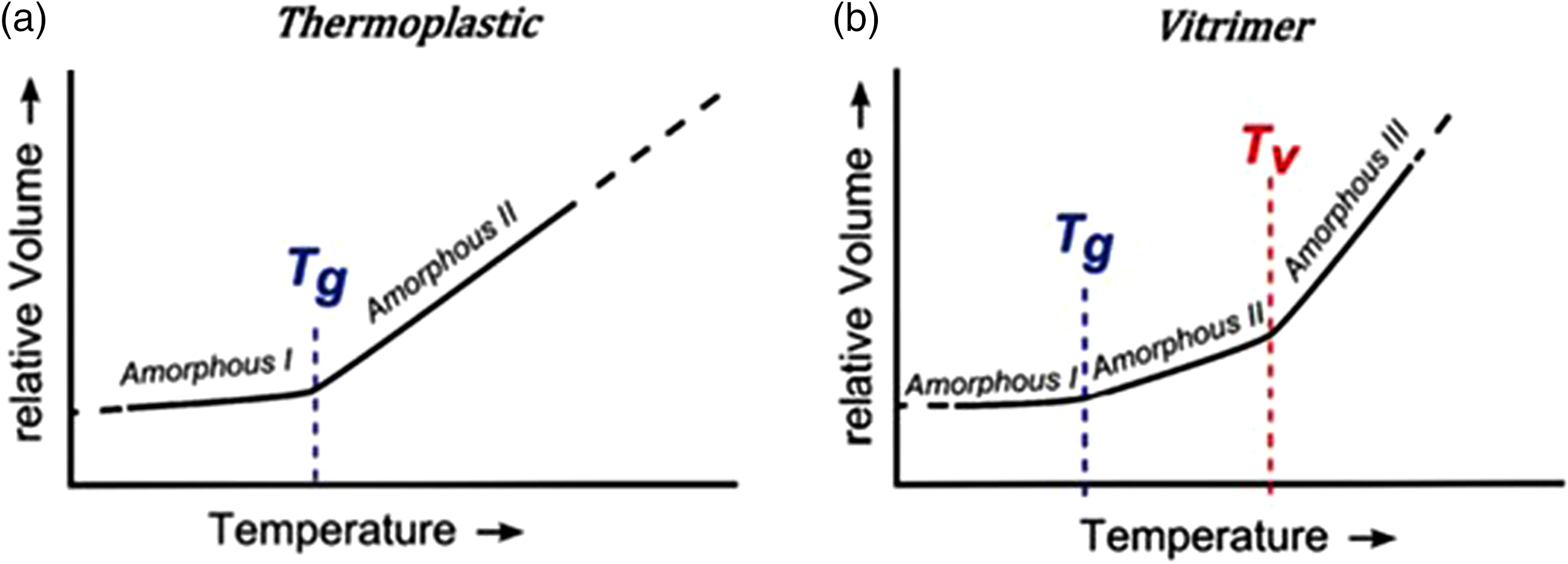
(a) Glass transition temperature of thermosetting resin and (b) special transition temperature of Vitrimer resin. 10
Vitrimer based on aliphatic ester exchange reaction
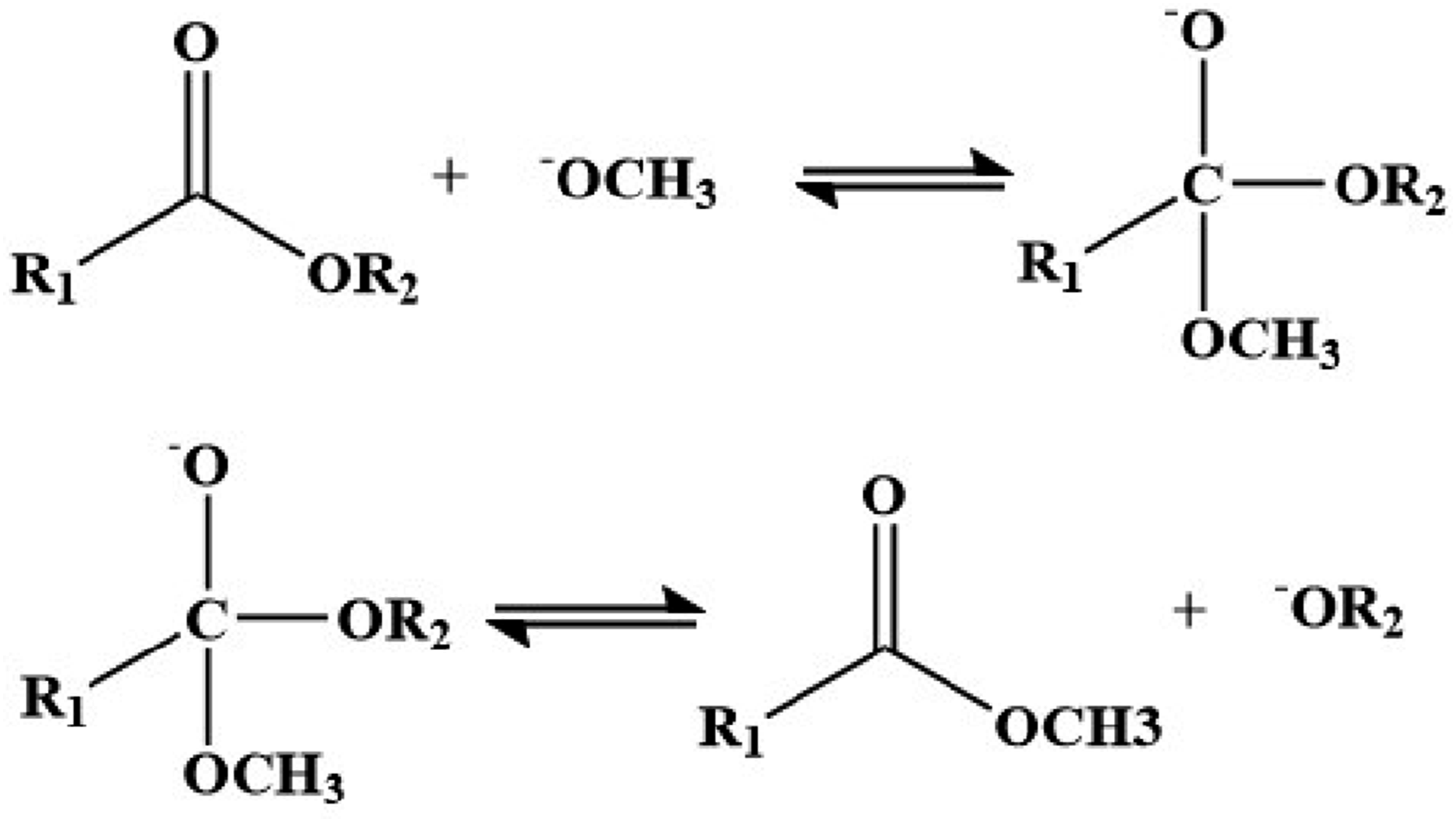
Current research predominantly involves systems where β-hydroxy esters are introduced into epoxy resins. Under the certain temperature and specific catalyst, these ester bonds can undergo exchange reactions with adjacent hydroxyl groups. These ester exchange reactions cause topological rearrangement, leading to fluid networks and achieving the material’s self-healing effect.
The ester exchange reaction presented in Figure 3 is widely used in Vitrimer’s research due to its excellent exchange rate. The source of Vitrimer raw materials based on ester exchange reaction is relatively wide, and the reaction mechanism is relatively mature, which has certain advantages in the synthesis of Vitrimer materials. Since its initial report in 2011, research on the Vitrimer esterification catalytic system has increasingly centered on three areas: low toxicity, low corrosivity, excellent compatibility with the resin matrix and superior catalytic performance for bond exchange reactions. Su et al.
24
prepared a self-catalytic transesterified Vitrimer by directly mixing miniature tertiary amine molecules with the epoxy anhydride system, thus covalently integrating the tertiary amine structure into the curing system. Moreover, the team explored the impact of tertiary amine structure and covalent integration on the various properties of Vitrimer. The study’s promoter, derived from the reaction of di (2-ethylhexyl)amine and epoxy resin (as illustrated in Figure 4), overcomes the issue of poor compatibility between tertiary amines and epoxy resin, while also introducing a highly active catalytic structure into the Vitrimer curing system. Thermally induced catalytic transesterification. (a) Reaction process and products of di-n-hexylamine with epoxy resin, (b) Storage modulus-temperature curves of various cured samples, (c) Loss tangent-temperature curves of various cured samples.
24
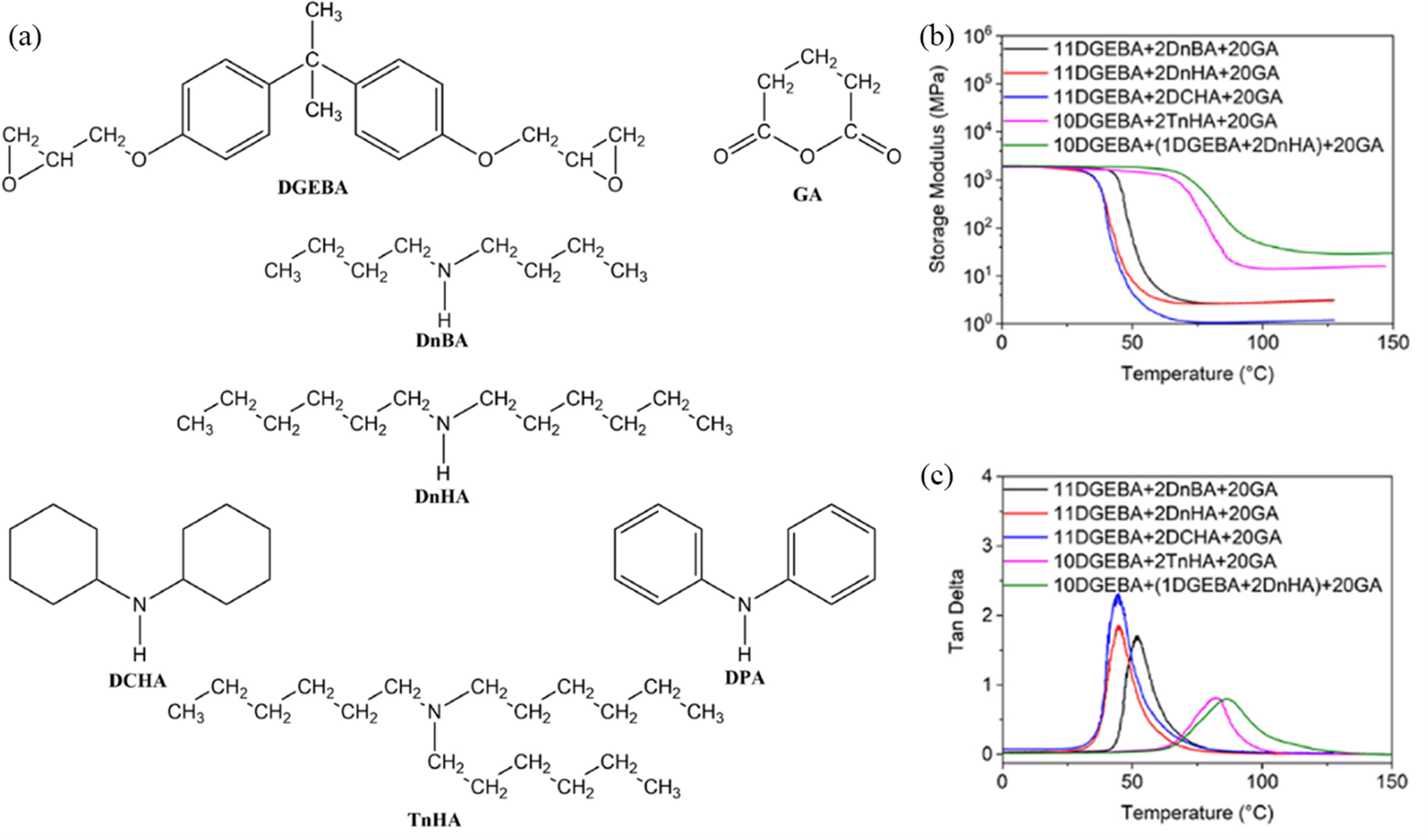
Willian et al. 15 analyzed the formation of covalently bound tertiary amines in epoxy Vitrimer, which was synthesized using DBA or BA in a mixture of DGEBA, citric acid (CA) and sebacic acid (SA). The findings revealed that the ester exchange reaction rates achieved with tertiary amines generated by DBA or BA were comparable to those achieved with other catalysts. Statistical analysis indicated that approximately 28.3% of the tertiary amines were present in the sols when DBA was used, compared to only 5.6% when BA was used. The tertiary amines in these sols likely exhibit activity similar to that in gels. This approach allows for further exploration of various primary or secondary amines at different initial concentrations.
In addition to some traditional catalysts, there are also studies using biocatalysts. Camille et al.
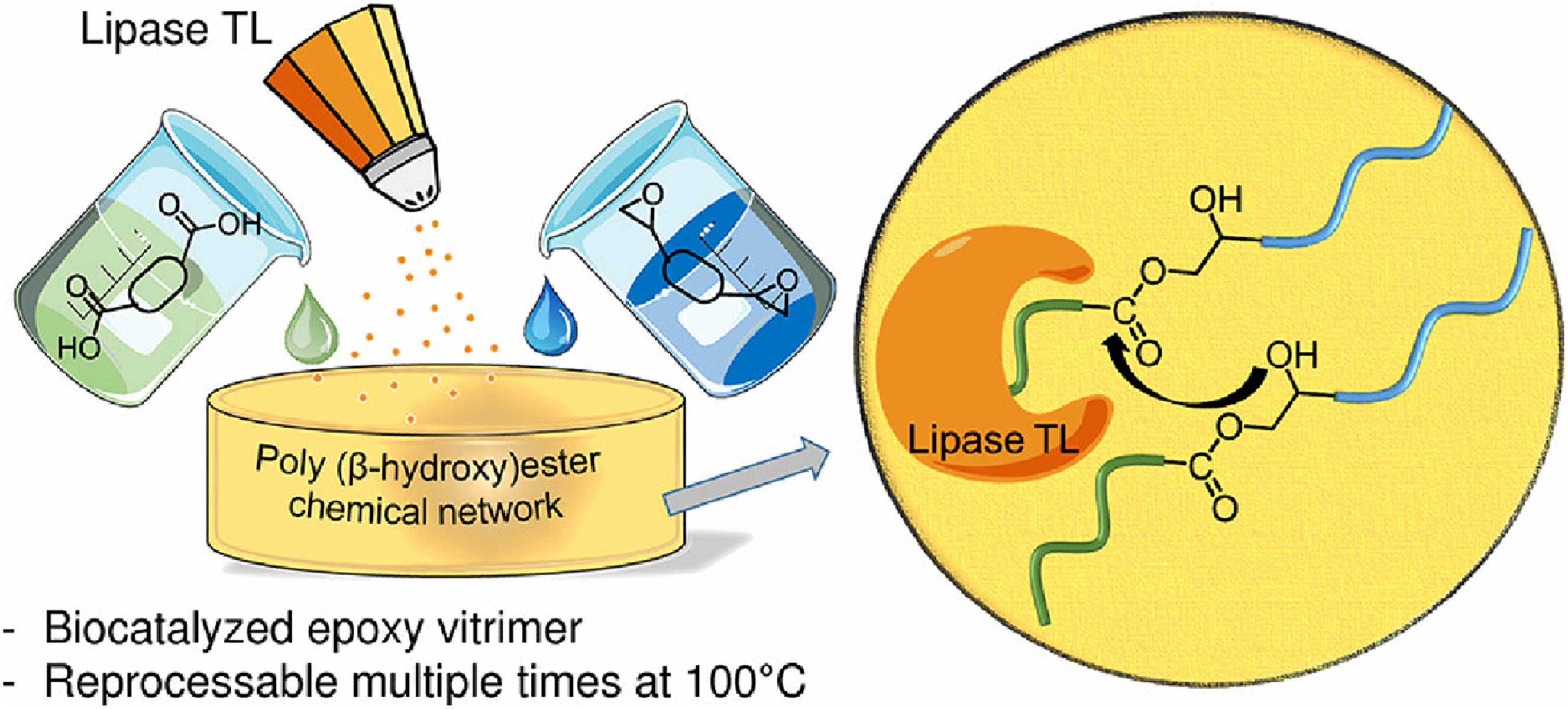
30
presented a fully biocatalytic approach for preparing and reprocessing epoxy resin trimer materials, utilizing lipase TL to facilitate network formation and exchange reactions via transesterification. Figure 5 depicts the binary phase diagram used to select optimal diacid/diepoxide monomer compositions. This approach prevents phase separation or sedimentation at curing temperatures below 100°C, thereby preserving enzyme activity. Stress relaxation experiments (70–100°C) and multiple reprocessing cycles (up to 3 times) demonstrate the lipase’s effectiveness in enabling network rearrangement and full mechanical strength recovery. However, heating to 150°C deactivates the enzyme, eliminating stress relaxation capability. This enzymatic strategy complements classical catalysts, for example TBD, which require high temperatures for complete stress relaxation, offering a sustainable alternative for dynamic covalent networks under mild conditions. Network exchange mode of epoxy Vitrimer catalyzed by lipase.
30
However, due to the higher activation energy required for its exchange reaction compared to other reactions, and the fact that most ester exchange reactions require catalysts for catalysis and a certain temperature, current research is no longer limited to traditional catalyst catalyzed ester exchange Vitrimers, but instead adopts ester exchange reactions without catalyst systems, or multi dynamic exchange networks with ester exchange as the main and other exchange reactions as auxiliary.
Feng et al.
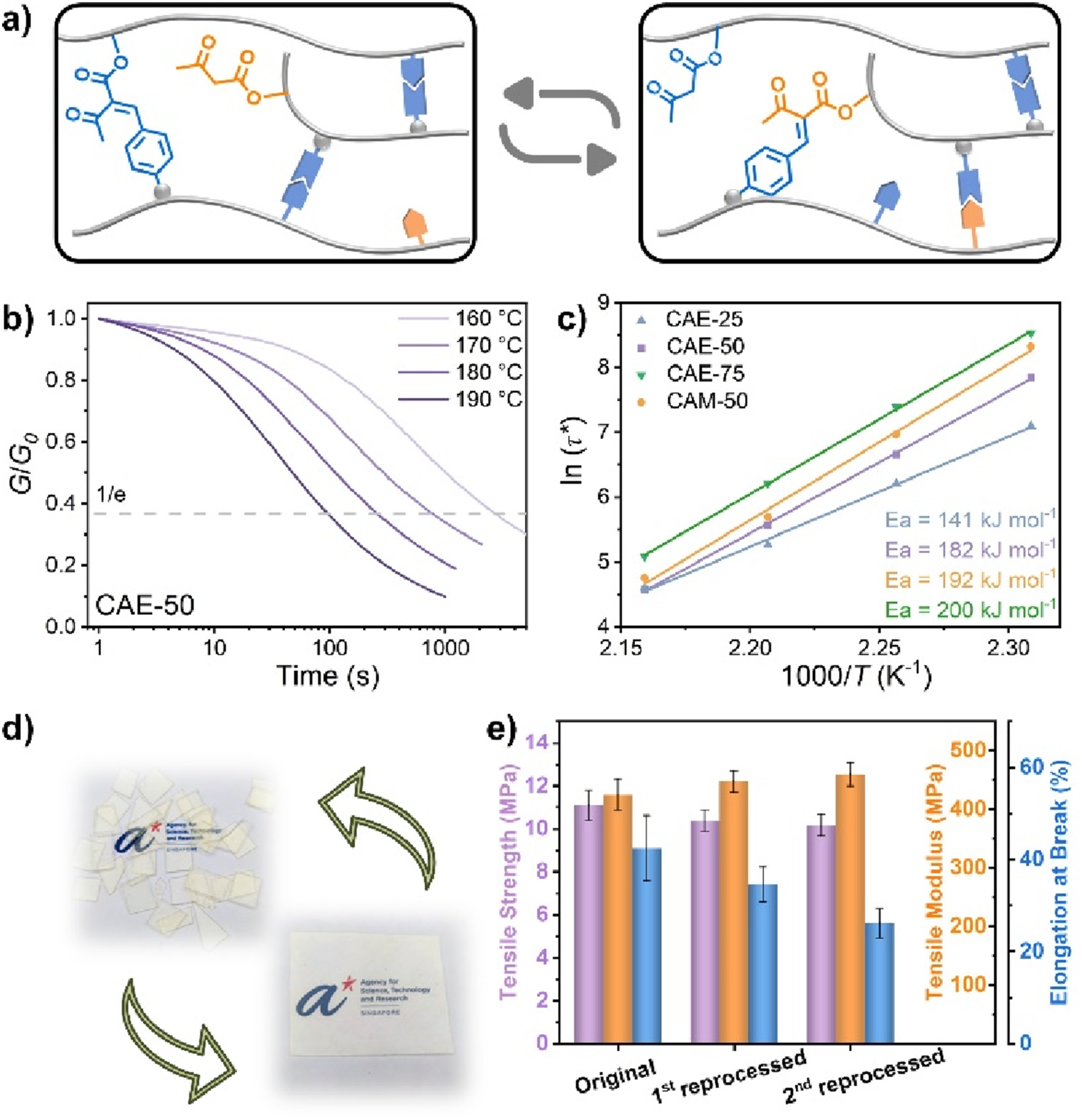
31
reported a catalyst-free α-AC/A (α-acetyl cinnamate/acetoacetate) exchange reaction, shown in Figure 6(a), to develop Vitrimers with exceptional creep resistance. Figure 6(b),(c) exhibits α-AC/A exchange occurs above 140°C but not at 120°C and their activation energy is definitely from 140 to 200 kJ.mol-1. Demonstrating this in polymers, copolymers derived from common vinyl monomers were crosslinked with terephthalaldehyde, yielding α-AC/A Vitrimers with tunable thermal and mechanical properties. These Vitrimers exhibit high stability, especially creep resistance at 120°C, while maintaining excellent reprocessability at elevated temperatures (Figure 6(d),(e)). This work introduces α-AC/A exchange as a robust dynamic chemistry for recyclable yet stable crosslinked networks. (a) Schematic of topological network rearrangement in α-AC/A Vitrimers via α-AC/A exchange with free cyanoacetate. (b) Normalized stress relaxation curves of CAE-50 at varying temperatures. (c) Arrhenius plots with linear fitting for α-AC/A Vitrimers. (d) Hot-press reprocessing of CAE-50 at 180°C for 30 min (schematic). (e) Mechanical properties of CAE-50 after two reprocessing cycles.
31
Another approach is to introduce self catalytic groups to promote ester exchange. Cuminet et al. 32 introduces a catalyst-free approach to designing ester-exchange Vitrimers by leveraging the α-difluoromethylene group as a novel activating moiety. The strong electronegativity of fluorine enables the CF2 group to activate both epoxy-acid polymerization and adjacent ester-exchange reactions, eliminating the need for metal or organic catalysts. This strategy yields highly crosslinked yet reprocessable networks under moderate heating. The resulting Vitrimer demonstrates excellent recyclability, maintaining mechanical properties over 10 reprocessing cycles. By combining durability, reprocessability, and catalyst-free synthesis, this fluorinated Vitrimer represents a sustainable advancement toward greener, recyclable polymers with enhanced performance and environmental benefits.
Vitrimer based on transamidation exchange reaction
The basis of transamidation reaction is amide ester exchange reaction, which activates the exchange reaction between amide ester (Figure 7(a)) and hydroxyl group or between two amide esters (Figure 7(b)) through certain external stimuli. The transamidation reaction is mostly used in dynamic polyurethane systems. Due to the fact that polyurethane itself has both thermosetting and thermoplastic properties, the introduction of dynamic bonds in the study of dynamic polyurethane aims to balance the thermosetting and thermoplastic properties of polyurethane, so that polyurethane has both certain mechanical properties and good repair properties. (a) Reaction between amide ester and hydroxyl group. (b) Reaction between two amide esters.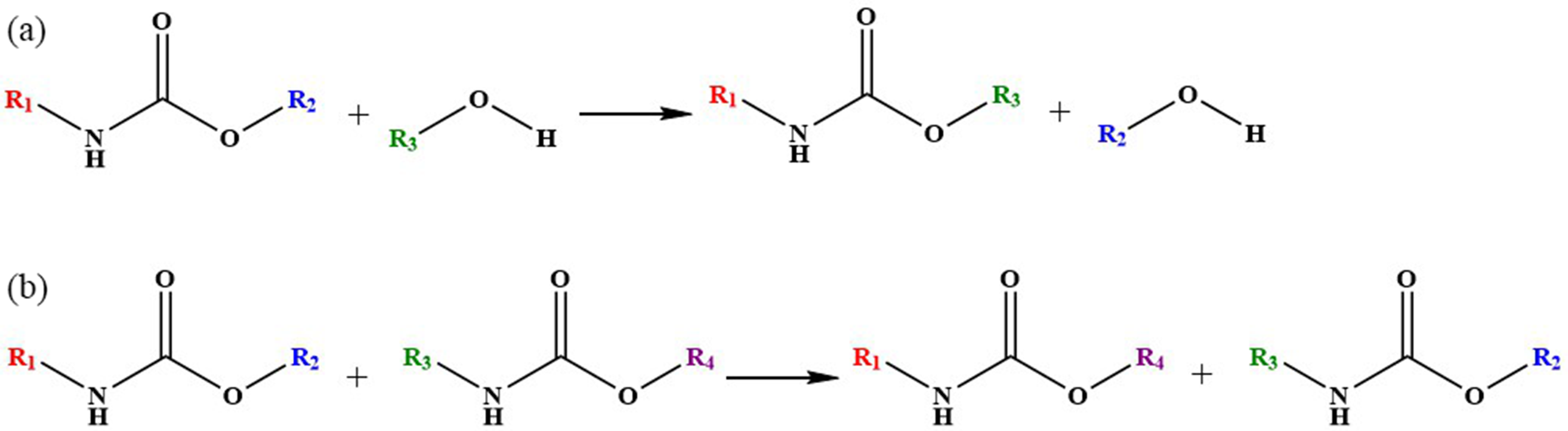
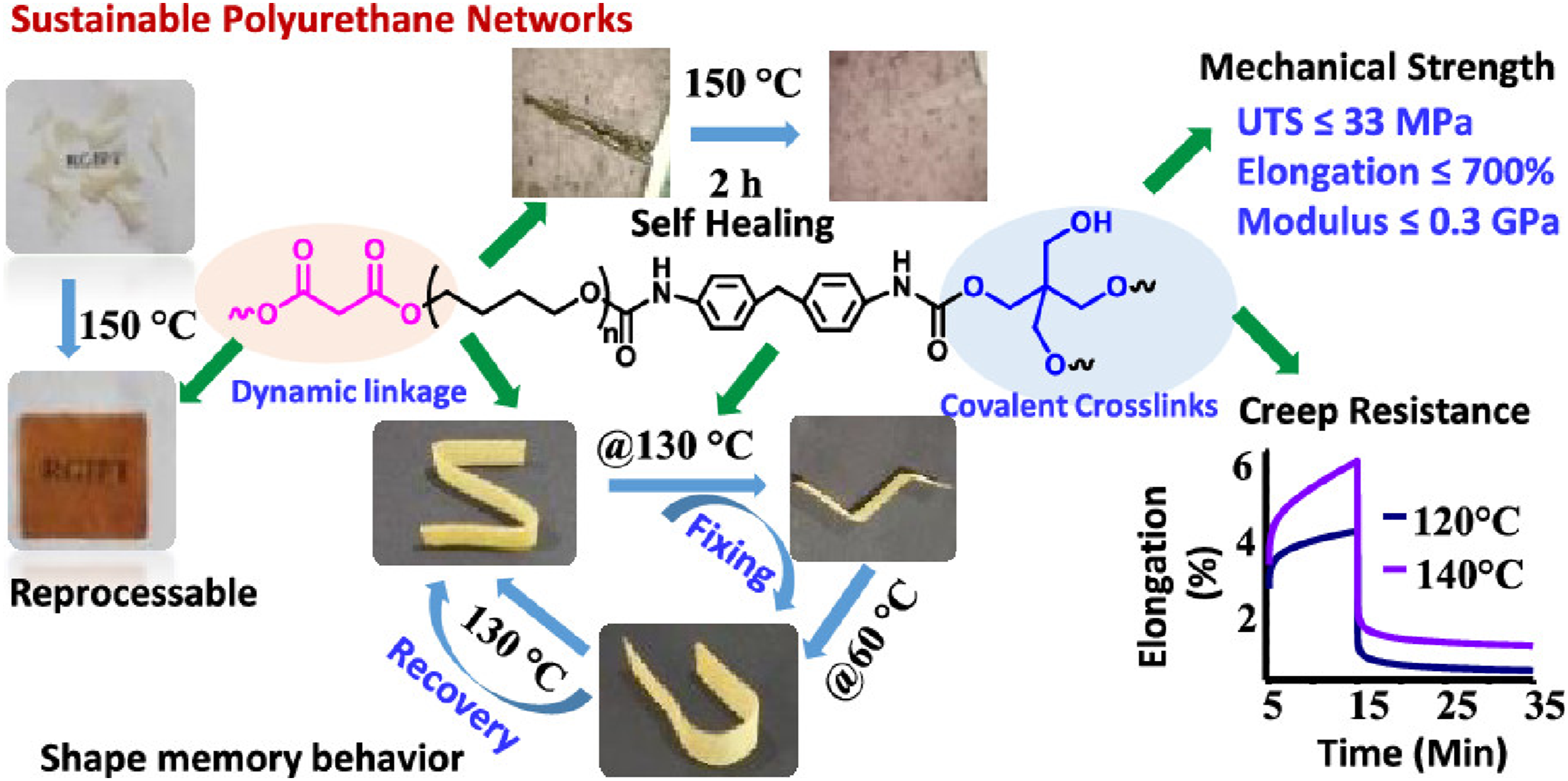
Debnath et al.
33
developed recyclable, self-healing polytetramethylene oxide (PTMO)-based polyurethane Vitrimers (PUNs) by incorporating dynamic carboxylate linkages. Figure 8 exhibits excellent mechanical properties (tensile strength: 8–33 MPa, elongation: 345–680%, modulus: 19–270 MPa) and demonstrates Vitrimeric behavior with stress relaxation at 140–180°C. Thermally reprocessed at 150°C, they recover 70–85% of original strength while showing enhanced creep resistance-a significant improvement over conventional Vitrimers. The materials also display triple-shape memory (transition at 60/130°C), autonomous self-healing, and welding capabilities. These multifunctional Vitrimeric PUNs address key limitations of traditional polyurethanes, offering sustainable alternatives with balanced reprocessability, dimensional stability, and performance for diverse applications. Regulating and controlling the proportion of soft link segments and hard link segments to chang different properties of Vitrimer.
33
Another study demonstrated by Brutman et al. 34 introduced that cross-linked polyurethanes incorporating urethane linkages can achieve reprocessability through tin-catalyzed associative exchange reactions at elevated temperatures. Using hydroxyl-terminated star-shaped poly (ethylene oxide) and poly (lactide) cross-linked with diisocyanate, researchers found that polymers with lower plateau moduli showed faster stress relaxation rates. Model reactions suggest the dynamic behavior occurs via tin-mediated urethane exchange without requiring free hydroxyl groups. While insoluble in low-polarity solvents, the networks dissolved in polar aprotic solvents like DMSO within 24–48 hours. These findings reveal that urethane linkages, easily incorporated into various polymer networks, can impart dynamic character likely through a urethane reversion mechanism, enabling the development of recyclable cross-linked materials while maintaining structural integrity.
The reaction between isocyanate and polyol is a common raw material for synthesizing polyurethane, and its high reactivity and structural diversity provide various ideas for polyurethane research (Figure 9). However, this type of isocyanate based dynamic polyurethane is mainly based on the exchange or other dynamic exchange bonds between two amide esters, so scientists have introduced another substance - cyclic carbonate.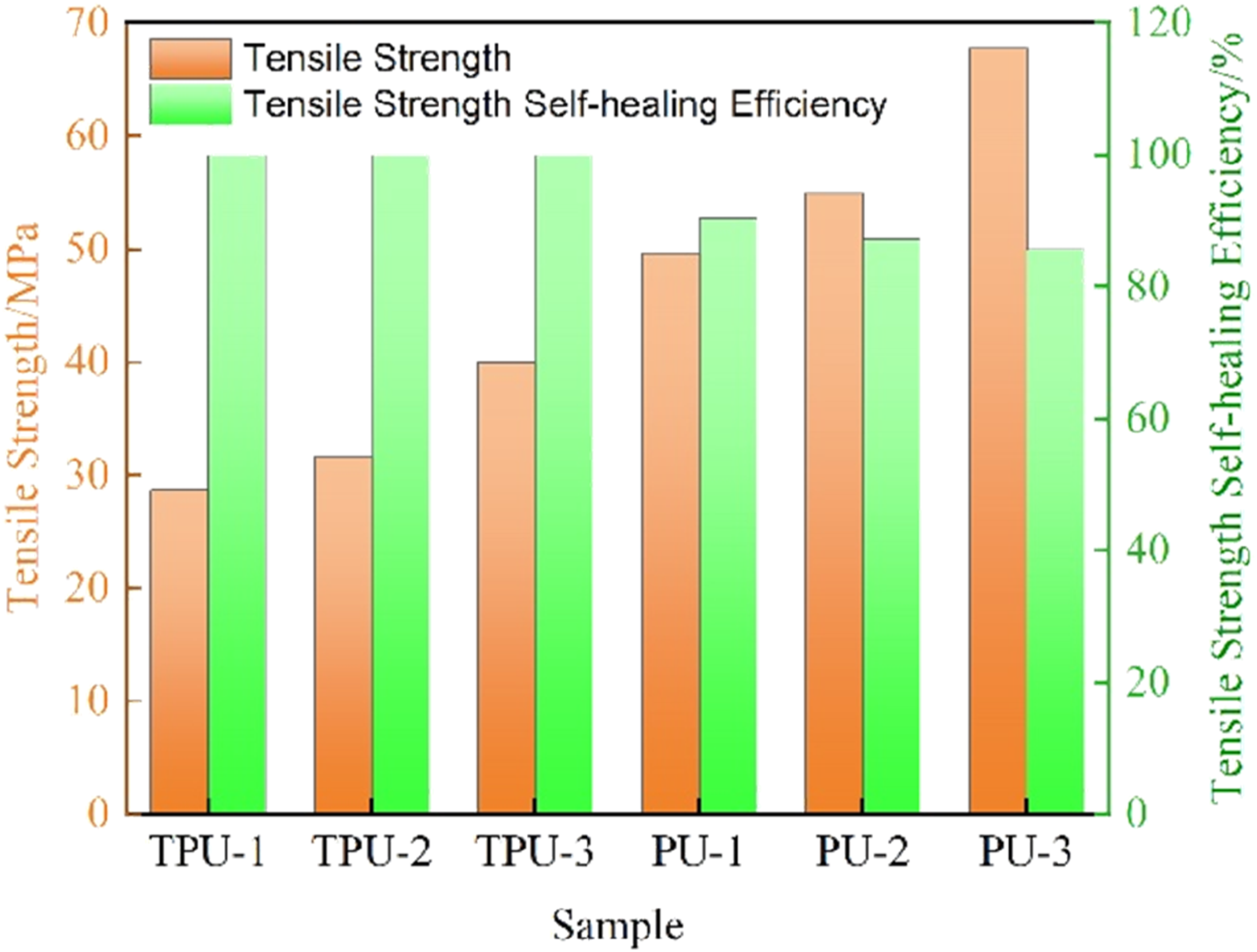
The amine group attacks the carbonyl group of the cyclic carbonate through nucleophilicity, opening the ring of the cyclic carbonate and forming an independent hydroxyl group, 40 which can further dynamically exchange with the amide ester. This reaction of cyclic carbonate synthesized polyhydroxy polyurethane (PHUs) is milder than reaction of isocyanate system synthesized polyurethane (PUs), and is gradually being applied in the research process of dynamic polyurethane.
Chen et al. 41 revealed that polyhydroxyurethane (PHU) networks synthesized from five-membered cyclic carbonates exhibit intrinsic reprocessability through dual mechanisms: transcarbamoylation exchange reactions and reversible cyclic carbonate aminolysis. These catalyst-free networks demonstrate full property recovery after multiple recycling cycles when properly designed. The discovery of concurrent reversible reactions alongside exchange mechanisms challenges the conventional understanding of dynamic covalent network reprocessing. As a sustainable alternative to traditional polyurethanes, this PHU system offers complete recyclability without requiring isocyanate chemistry, while maintaining performance characteristics through optimized monomer selection. The findings provide new insights into network polymer reprocessing while presenting an environmentally friendly platform for polyurethane-like materials.
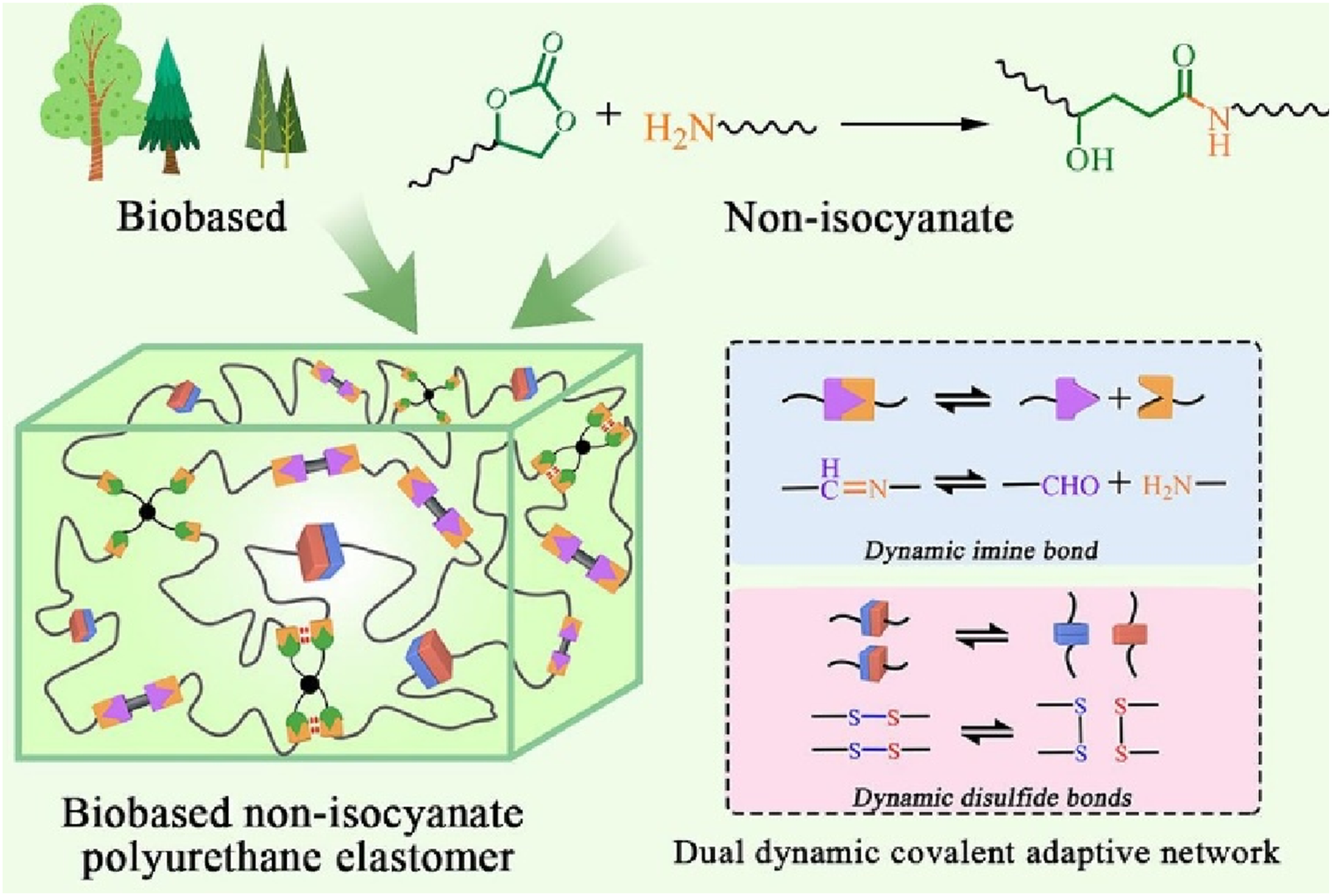
Non-isocyanate polyurethane (NIPU) elastomers have attracted significant attention, but their synthesis and functionalization remain challenging, especially based on amide exchange combined with other exchange bonds. Mou et al.
42
introduced a self-healing and reprocessable bio-based NIPU elastomer with a dual dynamic covalent adaptive network (DDCAN) was synthesized via disulfide and dynamic imine bonds (Figure 10), serving as a substrate for flexible strain sensors using MXene as the conductive material. The elastomer exhibited tensile strength and elongation at break of 3.22 MPa and 234%, respectively. Owing to DDCAN, it achieved 97.5% healing efficiency after 24 hours at room temperature. Remarkably, after three reprocessing cycles, its tensile strength increased to 136.1% of the original due to enhanced crosslinking from topological rearrangement. The resulting strain sensor demonstrated fast response (60 ms), excellent durability (1000 cycles), and successful application in motion detection and speech recognition. This work presents a novel approach to bio-based functional elastomers, advancing sustainable flexible electronics. Preparation of star shaped dynamic polymers via nucleophilic attack reaction of pentacyclic carbonates and amines.
42
Vitrimer based on disulfide bond exchange reaction
Under specific conditions, disulfide bonds can be reduced to thiol groups, which can then be oxidized back to disulfide bonds. This conversion between disulfide and thiol ester bonds is integrated into the preparation of Vitrimer, offering a higher level of compatibility compared to other synthetic approaches. It also represents a much more desirable reversible covalent system to date.
Compared to ester exchange reactions, disulfide bond exchange reactions (Figure 11) have a faster dynamic exchange rate and lower activation energy required for exchange, making them suitable for fast responsive materials. However, due to its poor thermal stability and antioxidant properties, it is not as stable as ester exchange reactions during application. Disulfide bond exchange reaction.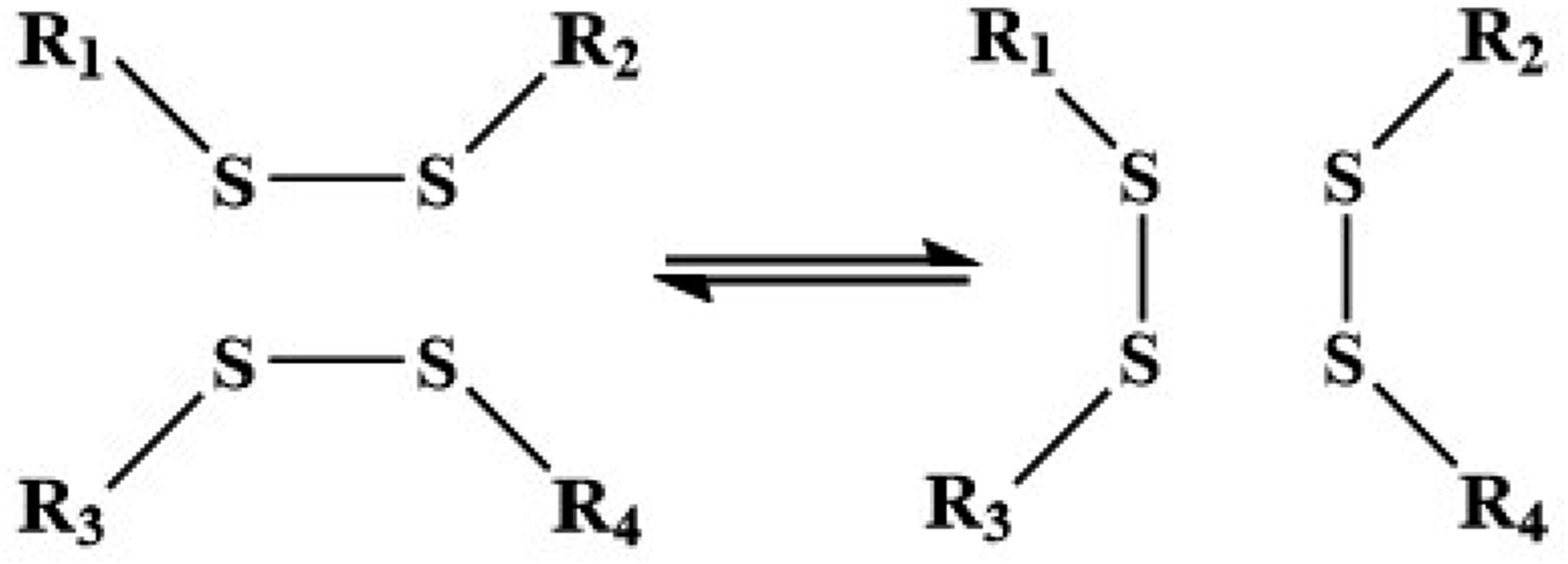
Guggari et al. 43 compared the construction unit of a dual dynamic bio-based epoxy resin with imine and disulfide bonds (DDBB) synthesized from cystamine and vanillin with its single dynamic imine-containing analog (SDBB).The results showed that the relaxation time of the resin significantly increased at 190°C, with the relaxation time decreasing to 21 s, compared to 25 min for the undoped resin. This enhancement is achieved with minimal impact on the glass transition temperature and without substantially altering the curing behavior.
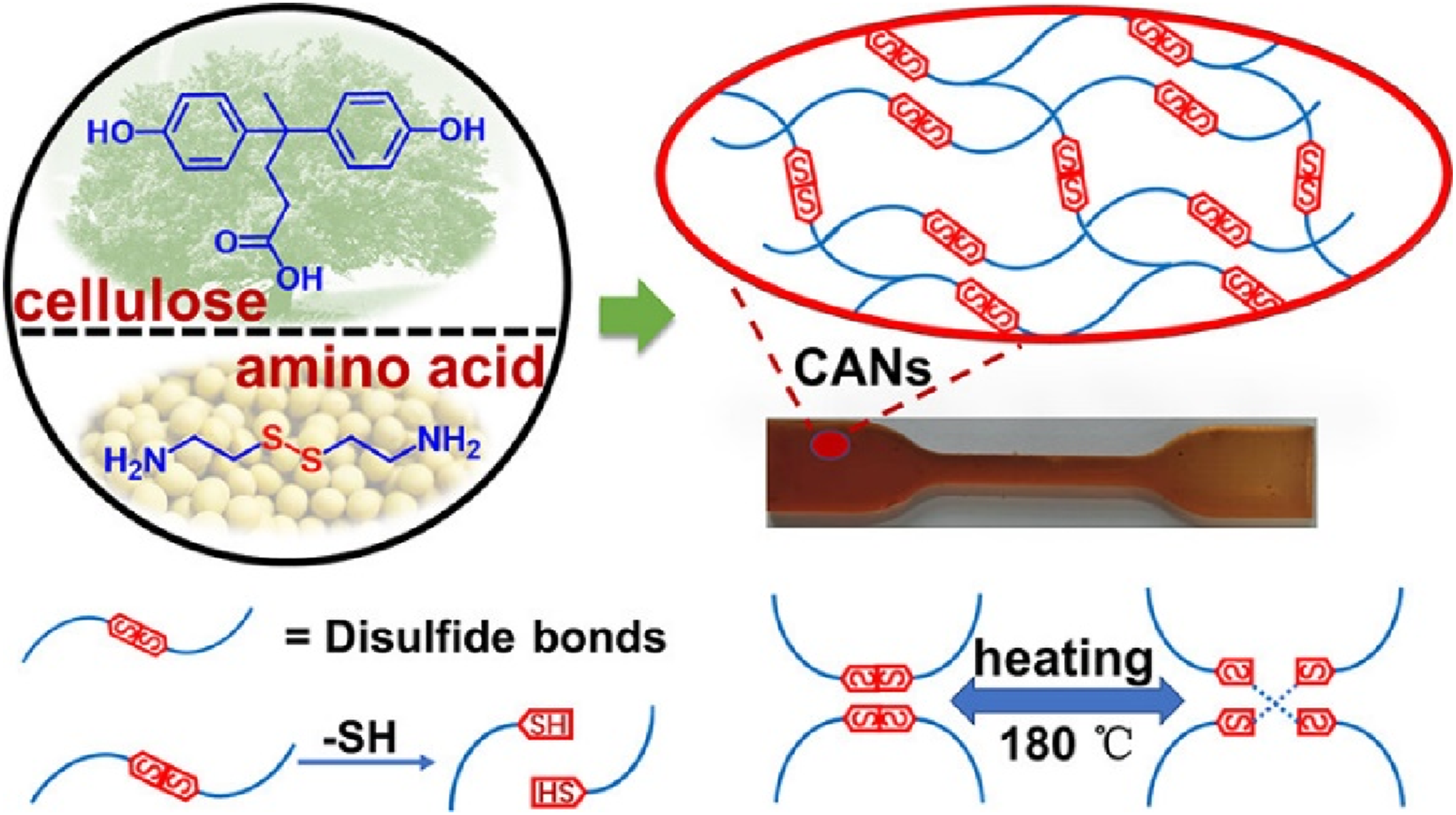
Another study researched by Chai et al.
44
developed a fully bio-based epoxy Vitrimer using diphenolic acid and cystamine. As presented in Figure 12, it creates a dynamic covalent network with disulfide bonds for balanced mechanical strength, reprocessability, and degradability. The material exhibits high cross-linking density and thermal stability, enabling self-healing and reprocessing via disulfide exchange while remaining degradable with dithiothreitol. As a matrix for carbon fiber composites, it achieves over 613 MPa tensile strength, with the resin fully degrading in 2.5 hours without damaging the fibers. This work demonstrates a sustainable approach to high-performance Vitrimers, combining robust mechanical properties with recyclability and eco-friendly degradation. Synthesis of disulfide exchange Vitrimer with certain mechanical and reparative properties using biobased epoxy and cystamine as raw materials.
44
Due to the previously mentioned issue of weak thermal stability and susceptibility to environmental influences in disulfide bonds, current research combines disulfide bonds with other dynamic bonds, which not only improves the overall stability and strength of Vitrimer, but also further enhances its self-healing properties.
Liu et al. 45 synthesized a single network structure with disulfide bonds using DGEBA and DTDPA in a certain proportion, and then added MHPPA to introduce dynamic ester bonds. Under the catalytic action of TEOA, they finally obtained Vitrimer with a dual network structure. The team established a concentration gradient of different disulfide bonds to study the self-healing properties related to potential multi-species, multi-scale physical, and electrical damage during the operation of the device. The results showed that this type of resin has excellent electrical insulation properties, and its dynamic performance enhances with the increase in the number of S-S bonds in the cross-linked network, thereby greatly improving the repair efficiency and damage repair scale.
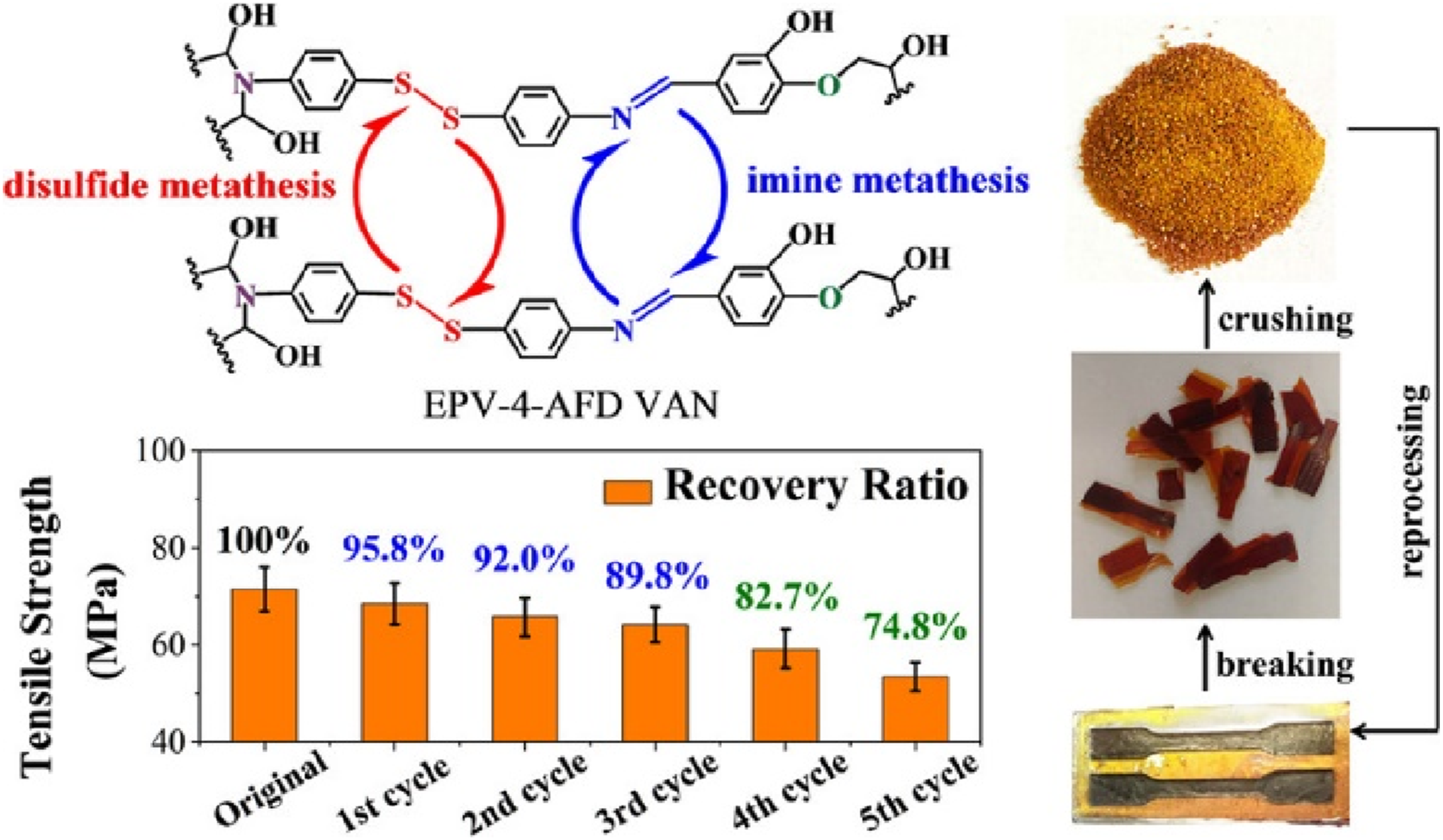
And Luo et al.
46
discovered an epoxy Vitrimer (EPV-4-AFD-VAN) using a novel curing agent (4-AFD-VAN) that incorporates dual dynamic covalent bonds—disulfide and imine linkages—via Schiff base chemistry. Compared to Vitrimers with only disulfide bonds (EVP-4-AFD), EPV-4-AFD-VAN exhibits lower activation energy, faster stress relaxation, and superior mechanical properties, achieving a tensile strength of 71.5 MPa while retaining ∼90% recovery over three reprocessing cycles (Figure 13). Like conventional thermosetting epoxy, it maintains solvent resistance, high breakdown strength, and excellent dielectric performance, yet remains chemically degradable in suitable solvents, ensuring environmental sustainability. This work advances high-performance Vitrimers by balancing robust mechanical properties, reprocessability, and eco-friendly degradation. Introducing disulfide and imine bonds into Vitrimer through Schiff base reaction using 2-Aminophenyl disulfide and bioaldehyde.
46
Vitrimer based on imine exchange reaction
The imine bond is formed through the condensation reaction of a primary amine with an aldehyde or ketone, in which the nitrogen atom in the amine replaces the oxygen atom on the carbonyl group. Generally, imine bonds are unstable, but if they are connected to aromatic groups, they become more stable. This type of imine bond is often referred to as a Schiff base.
47
Figure 14 illustrates the imine metathesis reaction which can undergo dynamic exchange under specific conditions, and are therefore used in the synthesis of various self-healing and recyclable materials. Imine metathesis reaction.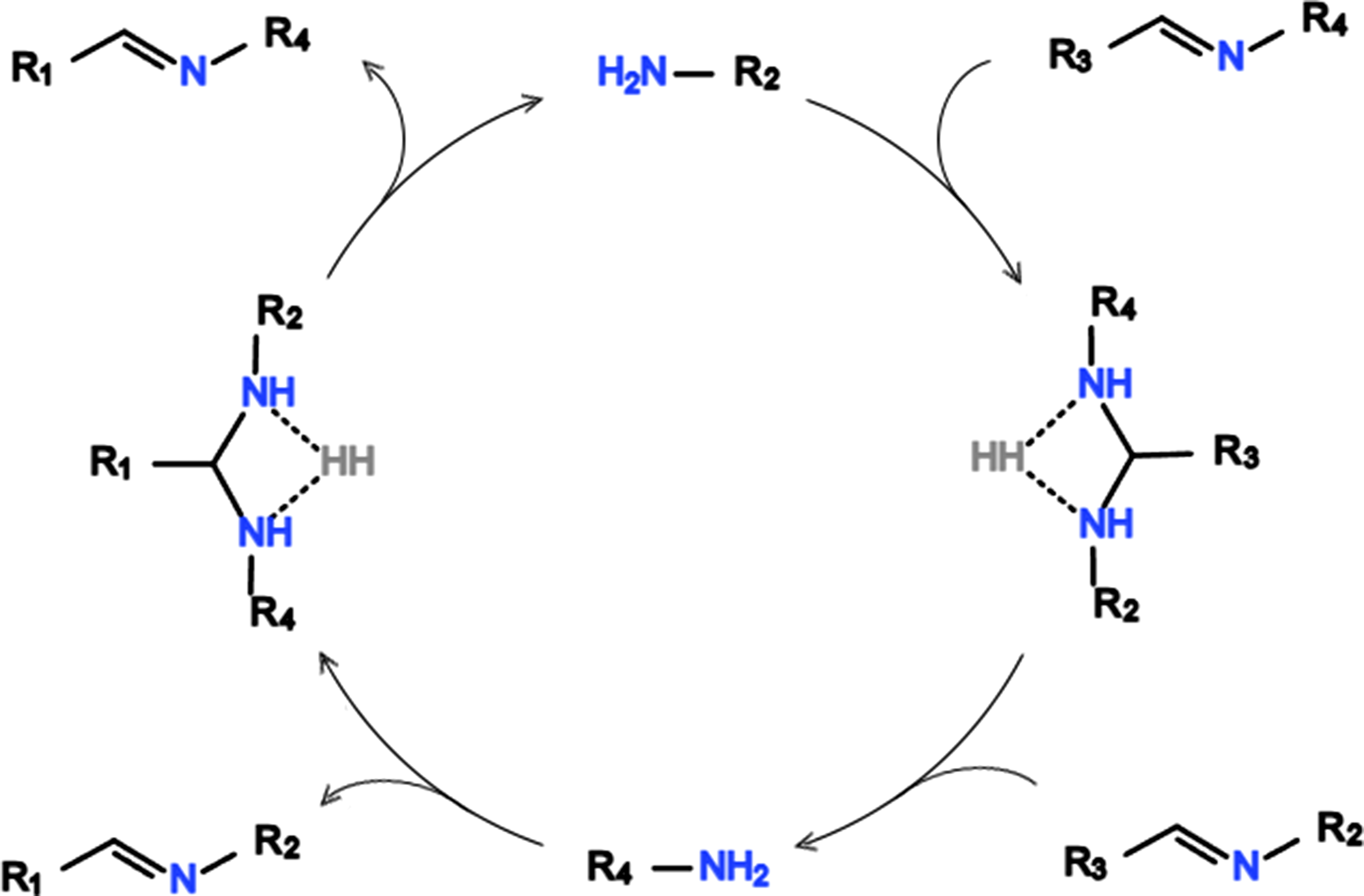
Andrea et al. 48 investigated a flame-retardant polyimide Vitrimer material formulated with ammonium polyphosphate and m-hydroxy diphenyl phosphate as additives, and tris(2-aminoethyl)phosphine triamide as a flame retardant. They characterized the Vitrimer material using DSC, TGA, LOI and other techniques. The results indicated that, relative to the pentaerythritol-based aliphatic epoxy resin system, the Vitrimer exhibited greater thermal stability, yet its fire resistance was comparatively lower. Naturally, when the phosphorus content is low, the melting temperature exceeds the transition temperature, which influences the limiting oxygen index and UL-94 test outcomes to some extent. The incorporation of flame retardants into the system leverages the synergistic effect of nitrogen and phosphorus, thereby enhancing the material’s flame retardancy to a certain degree.
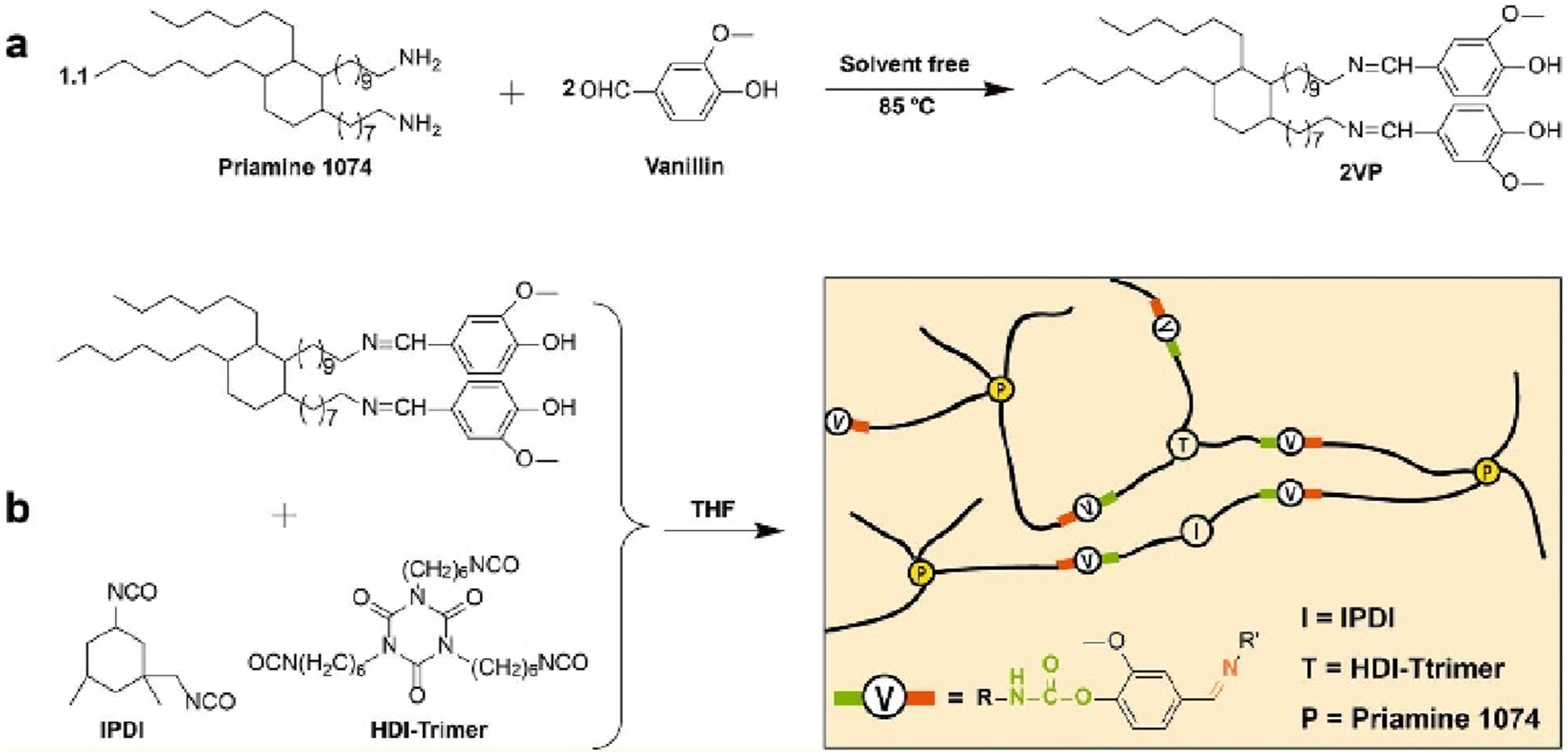
Wang et al.
49
studied another method of using vanillin and branched diamine to perform a Schiff base reaction to prepare a completely biobased bifunctional imine modifier (2VP) under solvent-free conditions (Figure 15(a)). 2VP reacts with isocyanates (Figure 15(b)) (hexamethylene diisocyanate trimer-HDI-T is used as a crosslinking agent, and isophorone diisocyanate-IPDI is used as a chain extender) at moderate temperatures (80°C) to produce imine micro-aggregates with a high biomass content (about 70%).The rigid aromatic vanillin and flexible branched diamine impart medium strength and excellent ductility to the imine Vitrimer; The ester bond showed rapid stress relaxation at 80°C to 100°C, indicating that the internal network structure rearranged under the effect of imine segregation. Constructing Vitrimer resin with dual dynamic network through (a) Schiff base reaction and (b) isocyanate reaction.
49
Regarding the convenience of Schiff base reaction and the traditional amine based epoxy curing method, Wang et al. 50 proposed a solvent-free, one-pot synthesis of Schiff-base Vitrimers using commercial diamines, dialdehydes, and bisphenol epoxy resin, yielding crosslinked polymers with high tensile strength (59.6 MPa) and elongation (7.1%). Unlike conventional acid-sensitive Schiff-base Vitrimers, these exhibited superior chemical resistance. High-temperature (>150°C) imine bond exchange enabled rapid stress relaxation, facilitating welding, reshaping, and recycling. Carbon fiber-reinforced polymers (CFRPs) made from this Vitrimer matrix could be thermally deformed at 180°C, with welded strips achieving 8.33 MPa lap shear strength and 2.99% elongation.
Due to the high activity and easy synthesis of transamination reactions, this imine group with dynamic exchange ability is often introduced into Vitrimers with single exchange mechanisms such as ester exchange, disulfide exchange, and carbamoyl exchange. Zhao et al.
51
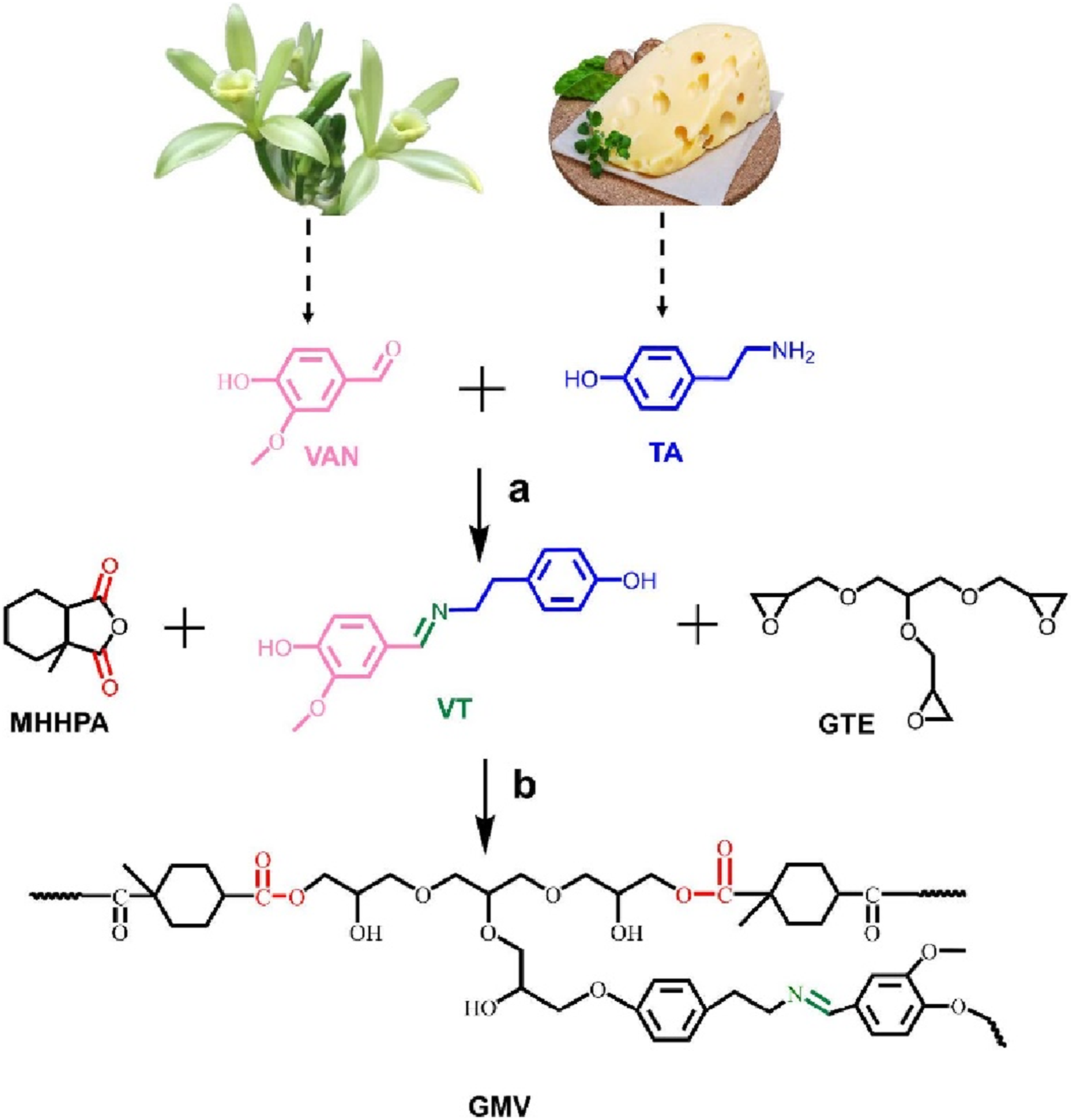
have developed a bio-based epoxy Vitrimer (GMV) (Figure 16(a)) with dual dynamic covalent bonds by co-curing glycerol triglycidyl ether (GTE), vanillin-tyramine-derived diol (VT), and methylhexahydrophthalic anhydride (MHHPA) (Figure 16(b)). Unlike traditional epoxy-anhydride Vitrimers requiring heavy catalysts, GMV utilized free hydroxyl groups in the network to accelerate ester exchange, achieving rapid stress relaxation (τ = 12.15 s at 140°C) and high thermal stability (Td5% = 281.27°C). The Vitrimer demonstrated excellent self-healing (near-complete scratch recovery at 120°C in 60 min), reprocessability, and shape-memory properties without external catalysts. By optimizing the anhydride/hydroxyl ratio, GMV combined robust mechanical performance with dynamic functionality, advancing catalyst-free epoxy Vitrimers for sustainable applications. This work expands solvent-free, bio-derived solutions for recyclable thermosets. (a) Schematic overviews of the synthetic route of VT, and (b) curing reaction of GMV vitrimer.
51
Zhao et al. 52 also studied another dual dynamic Vitrimer system based on imine groups, a sustainable poly (imide-imine) Vitrimer (ADTA) by integrating multiple dynamic covalent bonds (imide, imine, and disulfide) through the condensation of vanillin-derived dialdehyde (DVA), imide-containing diamine (ATFA), and tris(2-aminoethyl)amine (TAEA). The ADTA material exhibited excellent thermal stability (Td5% = 278.87°C), high storage modulus (2421.26 MPa), and rapid stress relaxation (36.39 s), attributed to its stable alicyclic imide structure. Additionally, it demonstrated superior solvent resistance and self-healing capability. By leveraging dynamic bond exchange, this work provides a new approach to enhancing the recyclability and reparability of high-performance polyimides without compromising their mechanical or thermal properties, offering a promising pathway for sustainable polyimide applications in demanding environments.
Vitrimer based on transamination exchange reaction
Compared to the previous Vitrimer preparation methods, the process based on transamination reaction shown in Figure 17 is easier to synthesize and exhibits higher thermal stability. The transamination reaction also has an advantage in that it does not require the use of a catalyst during the reaction process, while the transesterification reaction requires the addition of a catalyst to enhance the exchange efficiency of the network structure, which not only reduces the mechanical properties of Vitrimer to a certain extent but also complicates the synthesis process. Transamination between amino acids and α-ketoacids.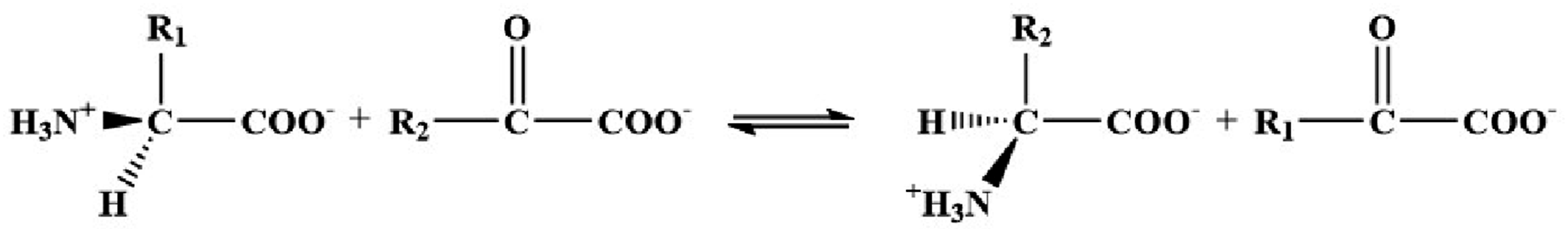
Prez et al.
53
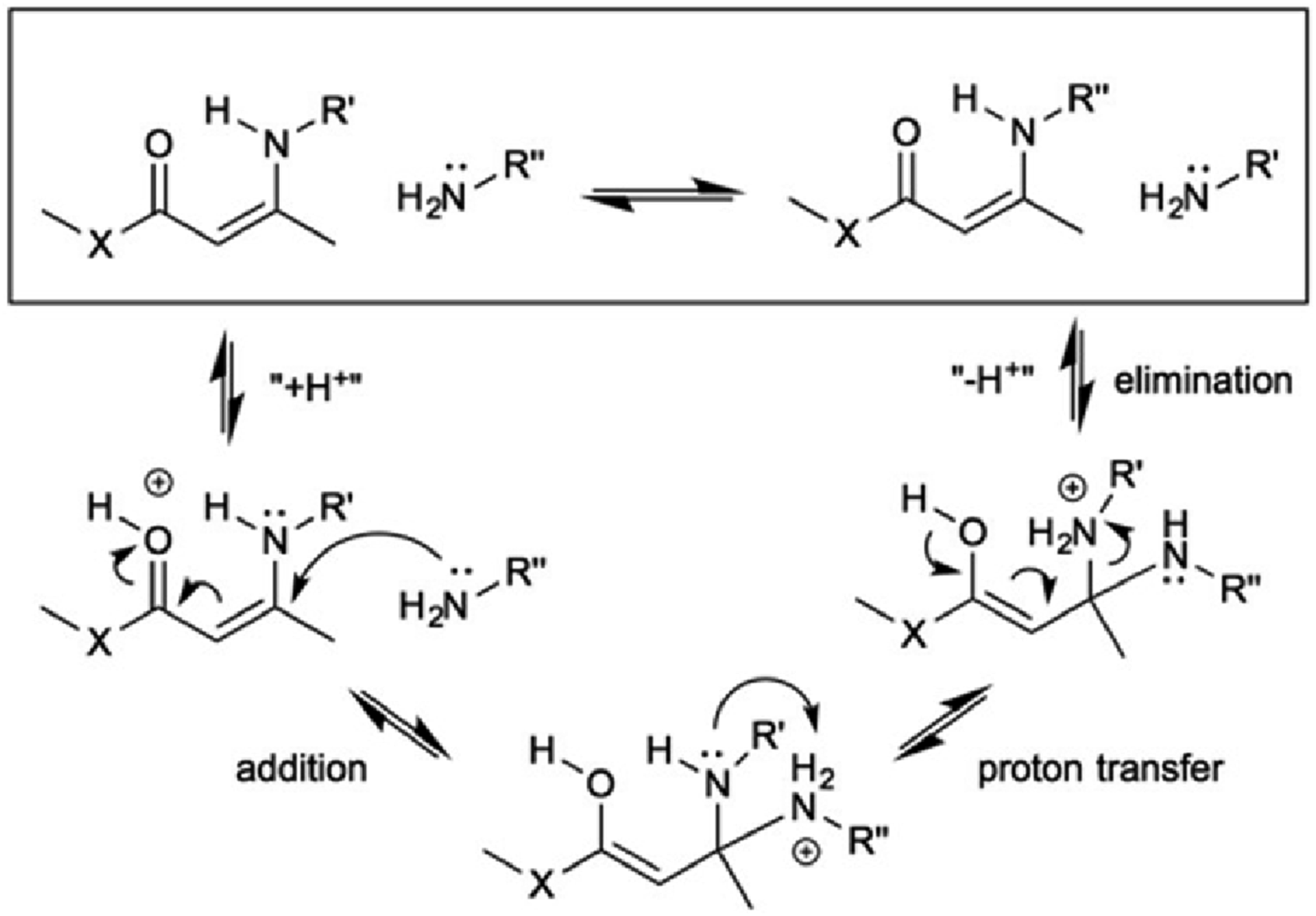
used cyclohexane diethanol diacetate, m-toluene diamine, and tris(2-aminoethyl)amine (Figure 18) in their study. They successfully synthesized a glass-like polymer material with thermosetting and thermoplastic properties, as well as reversible crosslinking characteristics. Without a catalyst, the relaxation time of such materials at high temperatures above 150°C is only about 80 s. In addition, Prez et al. also found that its network structure can be repeatedly recycled multiple times, and its performance does not significantly change after continuous molding and grinding. Interestingly, the glass transition temperature Tv (29°C) of this material is lower than the glass transition temperature Tg (87°C), which is rare in similar Vitrimer materials, and this is also the first time that a catalyst-free similar glass polymer material has been reported. Proposed mechanism for protonation-mediated exchange.
53
Hajj et al. 54 synthesized a series of bio-based polyimide glassy polymers through the reaction of bifunctional and trifunctional amines with furan-based dialdehydes. By adjusting the ratio of triamines to diamines, they created a set of crosslinked networks with similar chemical structures but different crosslinking densities. The results indicated that the crosslinking density significantly affects the relaxation characteristics of these glassy polymers. As the crosslinking density decreases, both the relaxation time and activation energy decrease. This means that the relaxation properties of the glassy polymers can be tuned by controlling the crosslinking density without the need to introduce catalysts, chemical groups, or modify the chemical structure.
Furthermore, current research is gradually moving towards green and environmentally friendly directions, so the use of photocatalysis has also become a hot topic in current research. Among them, relatively typical and mature photocatalytic reactions, such as thiol-alkene click reactions.
55
Figure 19 presents addition reactions between thiol groups and double bonds under photocatalytic action. Dugas et al.
56
presented a photopolymerized thiol-alkene Vitrimer based on dynamic covalent transamination of cyclic β-triketone-derived enamines, enabling uncatalyzed bond exchange with low energy barriers for reprocessing and rapid depolymerization. A designed olefin-functionalized β-triketone, 5,5-dimethyl-2-(pent-4-en-1-yl)cyclohexane-1,3-dione, was reacted with 1,6-hexanediamine in an off-stoichiometric ratio (≈1:0.85 amine:triketone), yielding a network with a subambient Tg (5.66°C) and a topological freezing temperature (Tv) of −32°C. Rheological analysis identified an effective reprocessing threshold (Tv, eff). Excess amine facilitated network degradation into monomers, which were repolymerized into reprocessable networks. Photopolymerization enabled spatiotemporal control, expanding the toolbox for Vitrimer upcycling and broader applications. Thiol-ene click chemistry.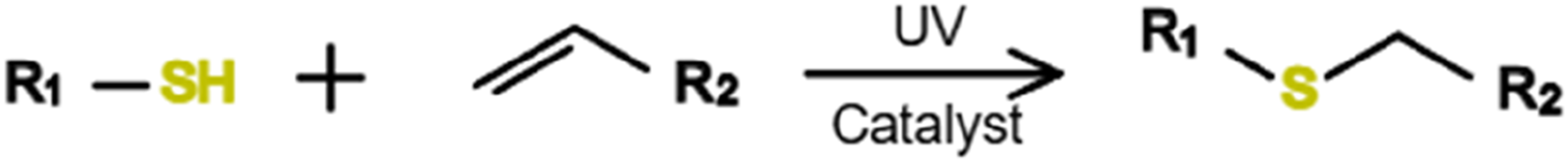
Vitrimer based on siloxane exchange reaction
Compared to linear polymeric precursors, silicone-based Vitrimers exhibit significantly enhanced creep resistance while maintaining excellent flowability at high shear rates and elevated temperatures. Consequently, silicone-based Vitrimers can be reprocessed and recycled multiple times without degradation of mechanical properties. Additionally, silicone-based Vitrimers demonstrate improved thermal resistance, oxidation resistance, and hydrolysis resistance. Furthermore, during mechanical blending, siloxane exchange (Figure 20) between different Vitrimers leads to self-compatibilized blends without the need for any compatibilizers. Mechanism of siloxane exchange reaction.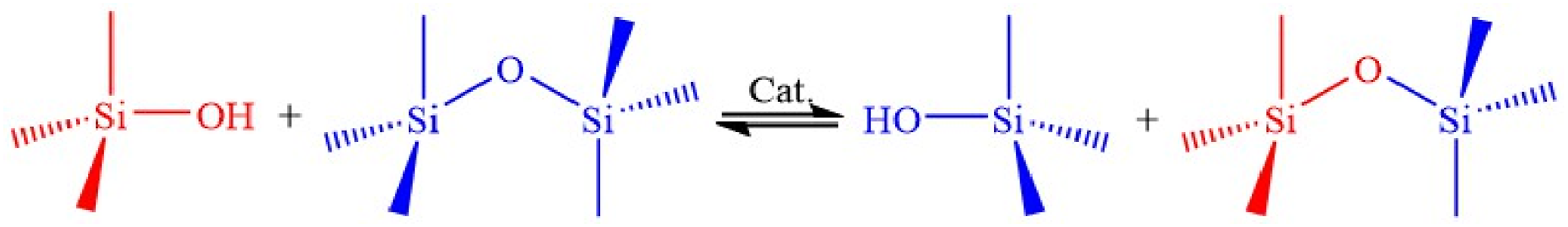
Inspired by classical elasticity theory, Luo et al. 57 presented a design strategy for ultra-tough and recyclable elastomeric Vitrimers by constructing a well-defined network through chemically crosslinking pre-synthesized disulfide-containing polydimethylsiloxane (PDMS) chains with tetra-arm polyethylene glycol (PEG). The defined network, achieved by minimizing dangling short chains and ensuring uniform molecular weight distribution, endows PDMS-disulfide-D Vitrimers with superior mechanical properties compared to random-network analogs and commercial silicone thermosets. Unlike conventional Vitrimers, which degrade upon recycling, these designed Vitrimers exhibit excellent thermal recyclability without significant mechanical loss, even when reused in electromyography sensors.
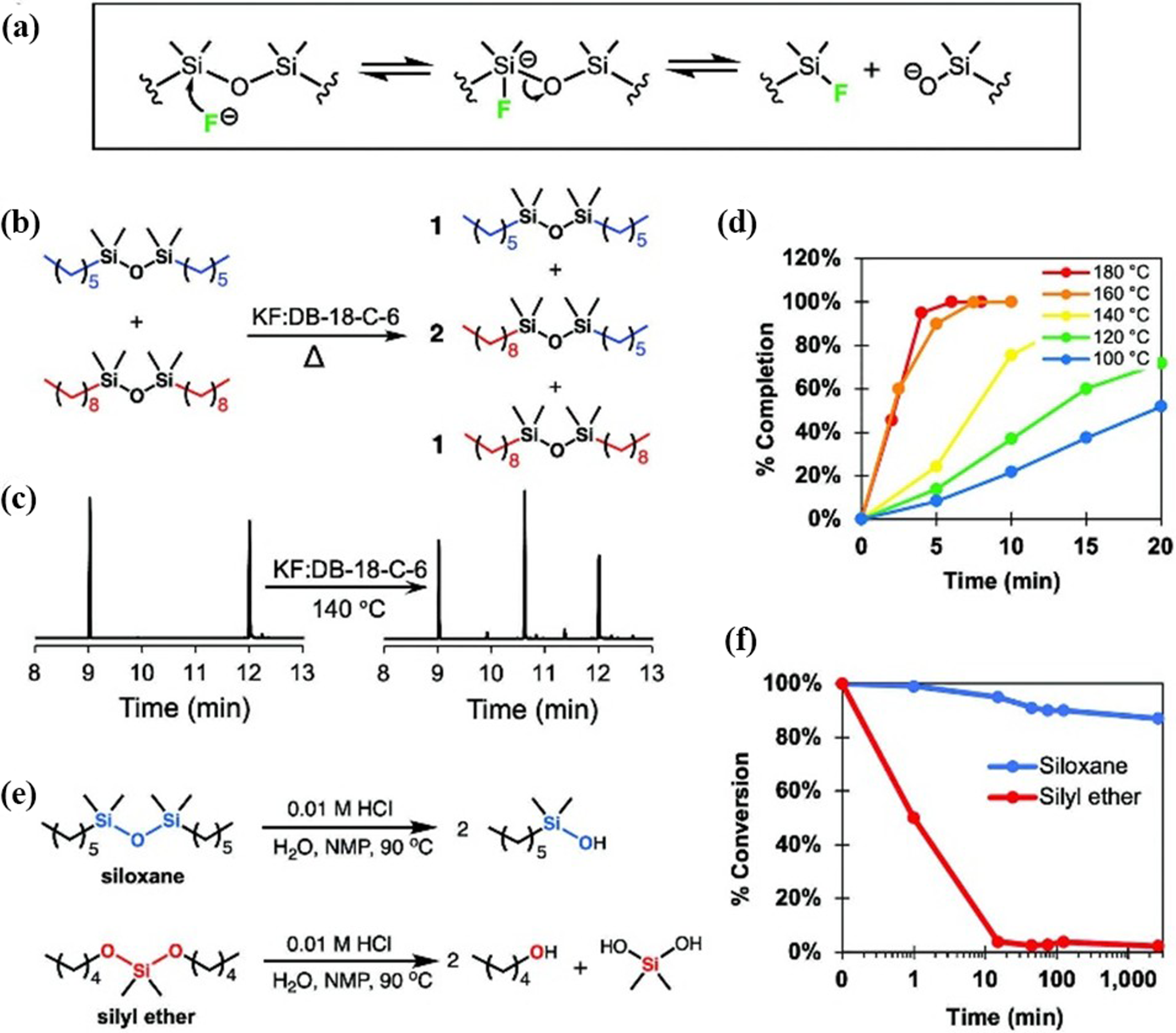
In order to improve the exchange rate of siloxane bonds, Tretbar et al.
58
have introduced fluoride (F−)-catalyzed siloxane exchange (Figure 21(a)) as a robust dynamic chemistry for producing high-performance Vitrimers from commodity thermoplastics like PMMA, polyethylene, and polypropylene via reactive extrusion. The F− catalyst enables rapid, selective siloxane bond exchange, enhancing thermal, oxidative, and hydrolytic stability while maintaining reprocessability (Figure 21(b)–(d)). The resulting Vitrimers exhibit superior creep resistance, shear-thinning behavior for easy processing after multiple recycling cycles, and hydrolytic stable properties compared to silyl ether (Figure 21(e),(f)). Notably, blending incompatible Vitrimers (e.g., HDPE-v and PMMA-v) induces self-compatibilization through dynamic siloxane exchange, offering a novel strategy for upcycling mixed plastic waste. This scalable approach combines industrial feasibility with sustainability, addressing key challenges in polymer recycling and high-performance material design. (a) Proposed mechanism for F-catalyzed siloxane exchange. (b–d) Reaction scheme, GC traces of starting materials and products, and reaction kinetics at different temperatures for small molecule siloxane exchange catalyzed by fluoride. (e,f) Comparison of hydrolytic stability of siloxane and silyl ether compounds.
58
Despite the advantages of dynamic polymer networks with multiple dynamic bonds, identifying compatible combinations of dynamic chemistries that synergistically achieve desired properties remains a major challenge. Fadlallah et al. 59 focuses on leveraging both siloxane and ester dynamic bonds in epoxy-acid cured Vitrimers to fine-tune chemical exchange reactions. We investigate how the co-catalytic effect of TBD simultaneously activates siloxane and ester exchange in epoxy-based Vitrimers. Results show that TBD not only promotes network formation but also enhances dynamic behavior, with stress relaxation revealing faster overall exchange rates than the sum of individual chemistries, indicating synergistic cooperation between siloxane and ester bonds. By adjusting dynamic bond and catalyst concentrations, we studied how adjacent chain mobility near specific bonds is modulated. This strategy enables dual-dynamic networks with significantly shorter relaxation times than single-dynamic-bond Vitrimers, improving (re)processability.
Study on the failure mechanisms and properties of carbon fiber/Vitrimer composite materials
In the last decades fiber-reinforced polymer composites (FRPC) have seen an enormous growth however, at the end of their lifetime, FRPC made of epoxy resins cannot be easily recycled because of the permanently crosslinked structure of the resins. Moreover, during long-term service under cyclic loading, conventional carbon fiber–reinforced thermoset composites are susceptible to fatigue damage, such as matrix cracking, fiber/matrix interfacial debonding, and delamination, which leads to a progressive degradation of mechanical performance and limits their service lifetime. Since the fibrous reinforcements usually have high costs, the possibility of developing recyclable FRPC is very advantageous both from environmental and economical point of view. Carbon fiber/Vitrimer composites represent a groundbreaking advancement in sustainable high-performance materials, combining the exceptional mechanical properties of carbon fibers with the unique dynamic characteristics of Vitrimer matrices. Unlike conventional thermoset-based composites, 3 these materials maintain their structural integrity while offering self-healing capabilities and recyclability through thermally activated bond exchange reactions.
The Vitrimer matrix, typically incorporating dynamic covalent bonds such as transesterification linkages or boronic esters, enables network rearrangement at elevated temperatures without losing crosslink density. This allows for multiple reprocessing cycles while preserving mechanical performance, addressing critical limitations of traditional composites.
Comparison of mechanical and self-healing properties of carbon fiber composites based on vitrimers with different main functional groups.
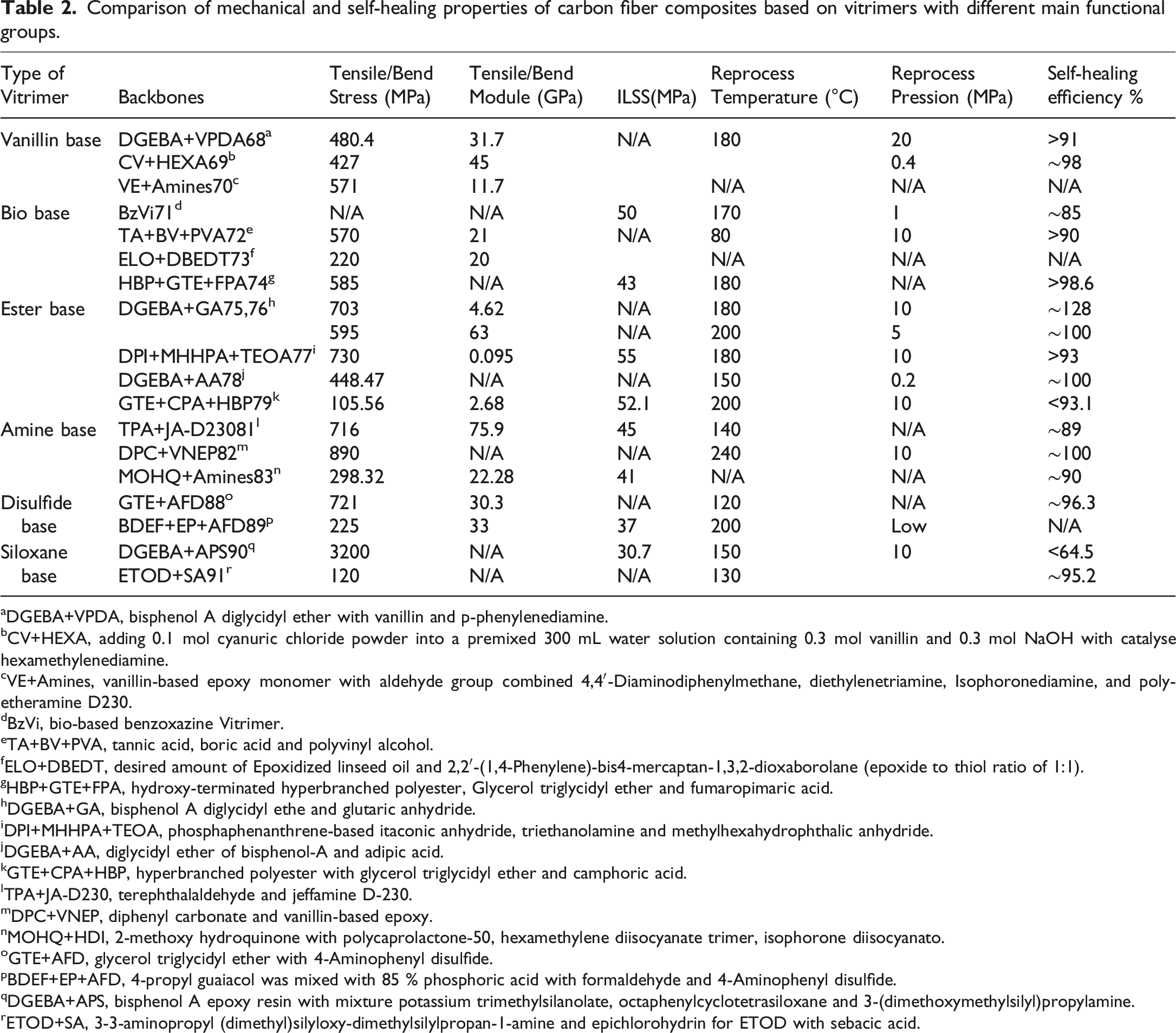
aDGEBA+VPDA, bisphenol A diglycidyl ether with vanillin and p-phenylenediamine.
bCV+HEXA, adding 0.1 mol cyanuric chloride powder into a premixed 300 mL water solution containing 0.3 mol vanillin and 0.3 mol NaOH with catalyse hexamethylenediamine.
cVE+Amines, vanillin-based epoxy monomer with aldehyde group combined 4,4′-Diaminodiphenylmethane, diethylenetriamine, Isophoronediamine, and polyetheramine D230.
dBzVi, bio-based benzoxazine Vitrimer.
eTA+BV+PVA, tannic acid, boric acid and polyvinyl alcohol.
fELO+DBEDT, desired amount of Epoxidized linseed oil and 2,2′-(1,4-Phenylene)-bis4-mercaptan-1,3,2-dioxaborolane (epoxide to thiol ratio of 1:1).
gHBP+GTE+FPA, hydroxy-terminated hyperbranched polyester, Glycerol triglycidyl ether and fumaropimaric acid.
hDGEBA+GA, bisphenol A diglycidyl ethe and glutaric anhydride.
iDPI+MHHPA+TEOA, phosphaphenanthrene-based itaconic anhydride, triethanolamine and methylhexahydrophthalic anhydride.
jDGEBA+AA, diglycidyl ether of bisphenol-A and adipic acid.
kGTE+CPA+HBP, hyperbranched polyester with glycerol triglycidyl ether and camphoric acid.
lTPA+JA-D230, terephthalaldehyde and jeffamine D-230.
mDPC+VNEP, diphenyl carbonate and vanillin-based epoxy.
nMOHQ+HDI, 2-methoxy hydroquinone with polycaprolactone-50, hexamethylene diisocyanate trimer, isophorone diisocyanato.
oGTE+AFD, glycerol triglycidyl ether with 4-Aminophenyl disulfide.
pBDEF+EP+AFD, 4-propyl guaiacol was mixed with 85 % phosphoric acid with formaldehyde and 4-Aminophenyl disulfide.
qDGEBA+APS, bisphenol A epoxy resin with mixture potassium trimethylsilanolate, octaphenylcyclotetrasiloxane and 3-(dimethoxymethylsilyl)propylamine.
rETOD+SA, 3-3-aminopropyl (dimethyl)silyloxy-dimethylsilylpropan-1-amine and epichlorohydrin for ETOD with sebacic acid.
Failure mechanisms of carbon fiber composites and improvement through Vitrimer
Despite their high strength, carbon fiber composites are susceptible to various forms of failure. Common failure mechanisms include corrosion, mechanical stress, high temperature, high pressure, radiation, and fatigue. The focus of this review primarily concerns fatigue damage repair in carbon fiber composites and find one way to enhance their fatigue resistance.
Fatigue damage in carbon fiber–reinforced composites is typically a multiscale and progressively evolving process and are able to be visualized fatigue damage in fibers using thermal imaging
65
and photo-sensing technologies.
66
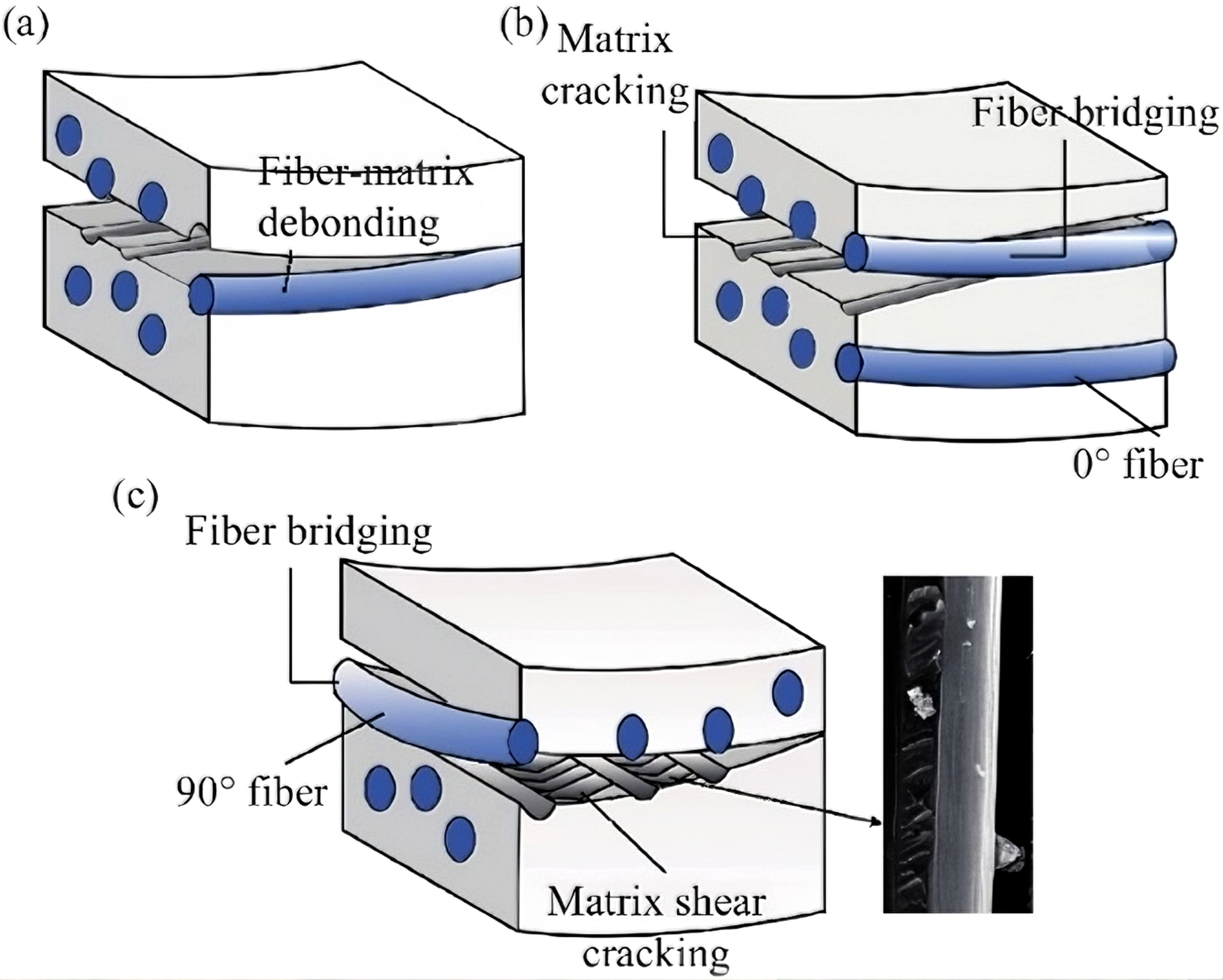
In Figure 22(a)–(c), fatigue damage initiates in the polymer matrix in the form of microcracks, which subsequently propagate toward the fiber/matrix interface, leading to interfacial debonding and a reduction in load transfer efficiency. With continued damage accumulation, matrix cracking and interfacial degradation promote interlaminar delamination, resulting in a gradual loss of structural stiffness and load-bearing capacity. Under high stress amplitudes or prolonged cyclic loading, local stress concentrations eventually cause carbon fiber fracture, leading to macroscopic fatigue failure of the composite. This matrix–interface–interlaminar–fiber-dominated progressive damage mechanism governs the fatigue degradation behavior of carbon fiber composites. Schematic diagram of damage mechanism in crack tip damagezone: (a) 0//0 ply angle matrix cracking and matrix/fiber separation, (b) Fiber bridging and dislocation cracks caused by 0//0 ply angle matrix/fiber separation, (c) 0//90 ply angle matrix shear cracking and fiber bridging.
67
Valter 68 utilized the concept of “damage units” to describe the evolution of tensile fatigue damage in plain woven carbon-epoxy composites. Through measurements from thermal dynamic analysis (TDA) and observations using scanning electron microscopy (SEM), it was found that once the damaged unit proportion (p) exceeds 0.527 (referred to as the “percolation threshold” (pc)), the size of the largest damaged cell cluster (C{max}) increases rapidly. Once (p) surpasses (p_c), rapid accumulation of transverse cracks, local delamination expansion, and fiber fracture occur, leading to final fatigue failure.
Lei et al.
69
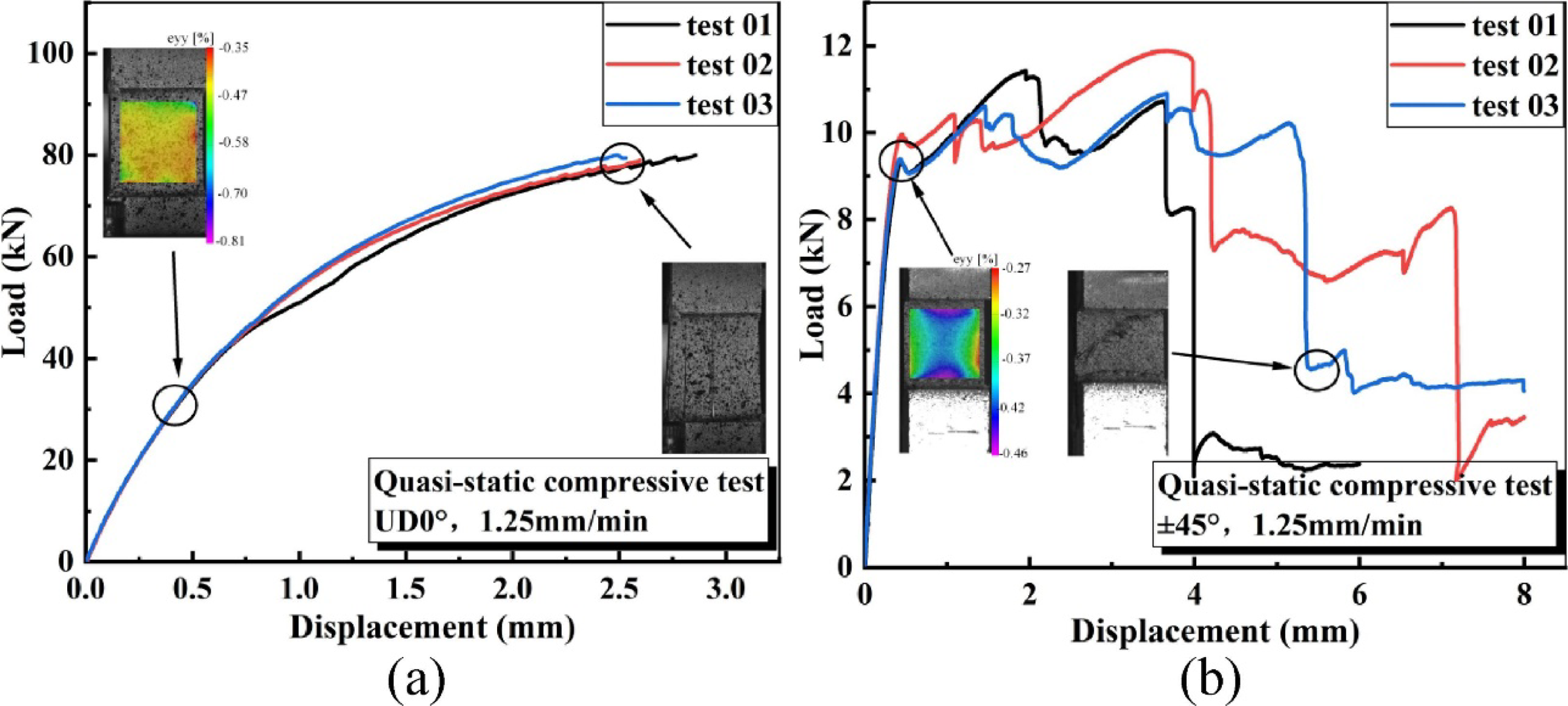
studied the influence of fiber orientation on the fatigue resistance of carbon fiber reinforced polymer composite laminates. Using digital image correlation and scanning electron microscopy techniques, it was observed that laminates with different fiber orientations exhibited significantly different fatigue lifetimes. Samples with 0° fiber (Figure 23(a)) orientation experienced only a 30% stiffness reduction before final fatigue failure, whereas those with ±45° fiber (Figure 23(b)) orientation experienced a 93% stiffness reduction. Under compressive loading, laminates with ±45° fiber orientation primarily exhibited fiber bending and delamination failure modes. Furthermore, after 15% total cycles, the residual tensile strength at 30% stress increased by 6.3% for laminates with ±45° fiber orientation. These findings provide crucial insights for optimizing the structural design of composite laminates and enhancing their fatigue properties. (a) Force-displacement curves of 0° laminates in quasi-static compression tests, (b) Force-displacement curves of ±45° laminates in quasi-static compression tests.
69
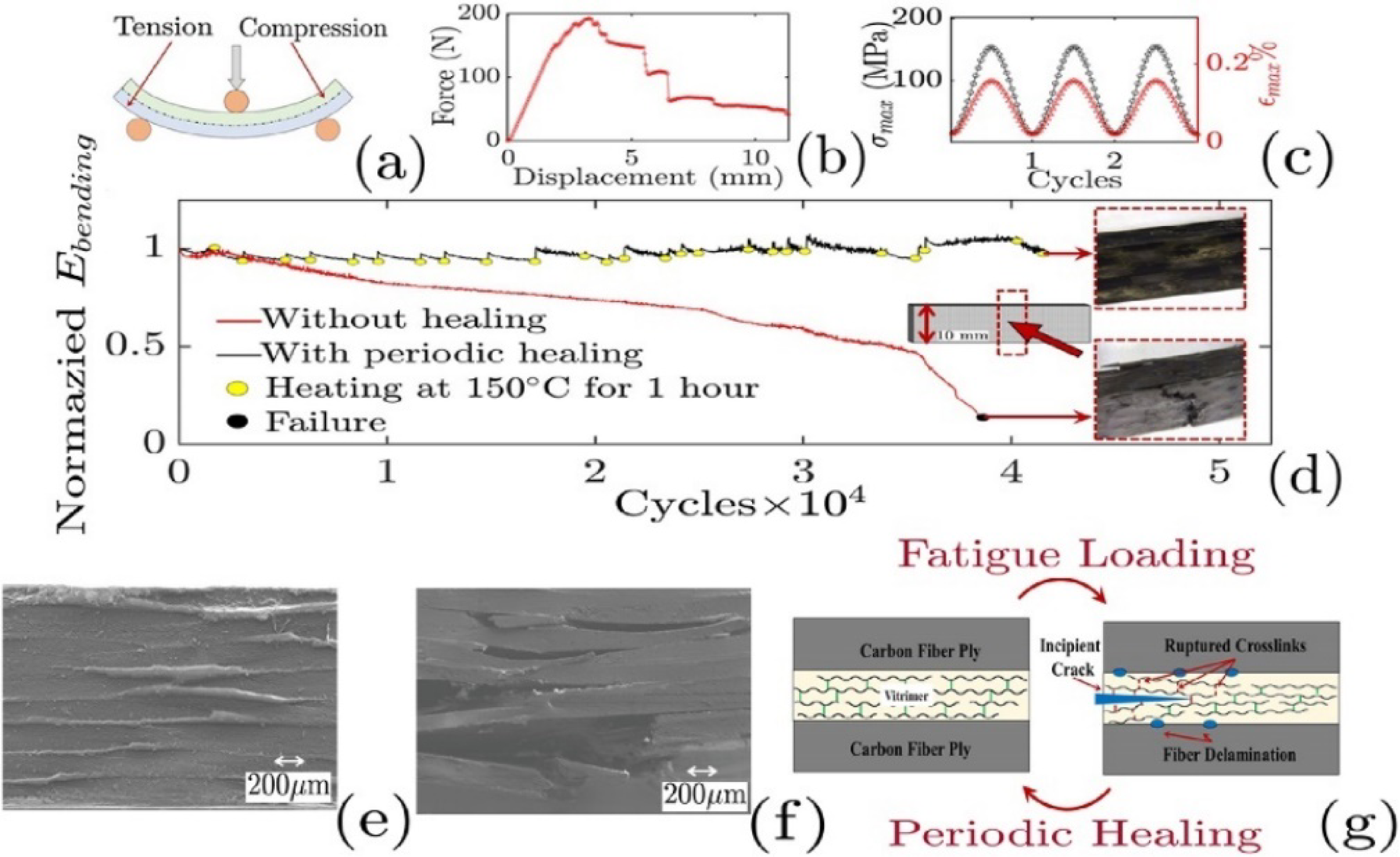
The topological network in Vitrimer matrices plays a key role in improving the fatigue performance of carbon fiber composites. As it shown in Figure 24(a)–(c), during cyclic loading, the dynamic covalent bonds can break and reform, allowing the network to locally rearrange and relieve stress concentrations around crack tips and fiber/matrix interfaces. This rearrangement helps slow down crack growth, reduce interfacial debonding, and limit interlaminar delamination. Under appropriate thermal conditions (Figure 24(d)), partial network reorganization can further decrease the accumulation of micro-scale fatigue damage. Manufacturing strategies, such as controlling temperature gradients during hot pressing and ensuring uniform distribution of dynamic bonds, can enhance network exchange at the interface and promote a more even stress distribution, moreover, the changes from Figure 24(e) to (f) can explain the topological transformation of the dynamic network in Vitrimer resin at the interface between the reinforcement and the matrix under the dual conditions of temperature and pressure (Figure 24(g)). Together, these factors allow Vitrimer-based composites to maintain better mechanical performance over long-term cyclic loading, offering a practical approach for designing durable structural materials. Reversal of fatigue degradation in vCFRP: (a) Schematic of 3-point bend tests used for vCFRP testing, (b) Monotonic bend test for a typical vCFRP specimen, (c) Typical bending stress and strain cycle used in the fatigue experiment, (d) Bending fatigue test for vCFRP indicates that as the load is cycled, (e) Scanning electron microscopy (SEM) image of the vCFRP sample which was subjected to periodic healing, (f) SEM image of vCFRP sample subjected to the same loading conditions and same number of cycles (without healing), (g) Schematic explains the process of healing reversal.
70
Mandal 71 examined the self-healing behavior of a high–thermal-resistance Vitrimer based on an aromatic thermosetting polyester (ATSP) using repeated Mode I fracture–healing cycles. Complete healing was achieved in the first two cycles, whereas healing efficiency gradually decreased with further cycling. Microscopic observations revealed progressive local mechanical damage near the crack region, while infrared spectroscopy confirmed the absence of chemical degradation. The decline in performance was therefore attributed to accumulated mechanical damage and limited chain diffusion.
Philip
72
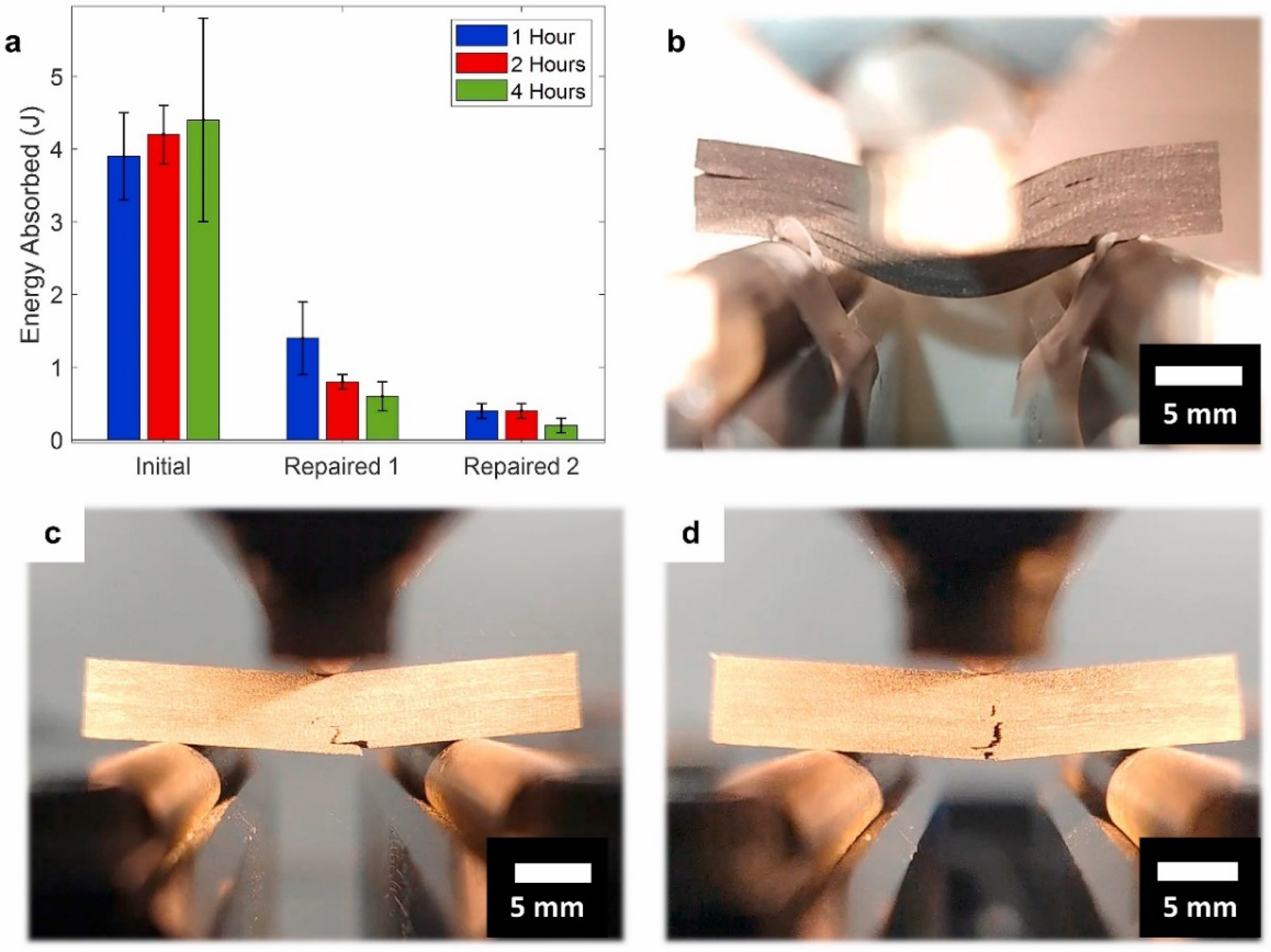
demonstrated a carbon fiber/Vitrimer composite material production method that can optimize the curing process (Figure 25(a)). This method uses the Vitrimer film method, which utilizes expandable compression molding technology (Figure 25(b)–(d)) to inject Vitrimer into fiber preforms to form fully cured and room temperature storable organic sheets, which are then reinforced into laminates. At the same time, team members tested the mechanical properties, interlayer properties, and repairability of the prepared carbon fiber composite material. The results showed that additional processing time can make the material harder and improve the lifespan of the carbon fiber/Vitrimer composite material. (a) Energy absorption of initial and repaired samples, (b)The failure behavior of composite materials exhibits interlayer debonding in the initial sample, (c) bending failure after one repair cycle, (d) and a significant decrease in the initial load of bending failure after two repair cycles.
72
Li et al.
73
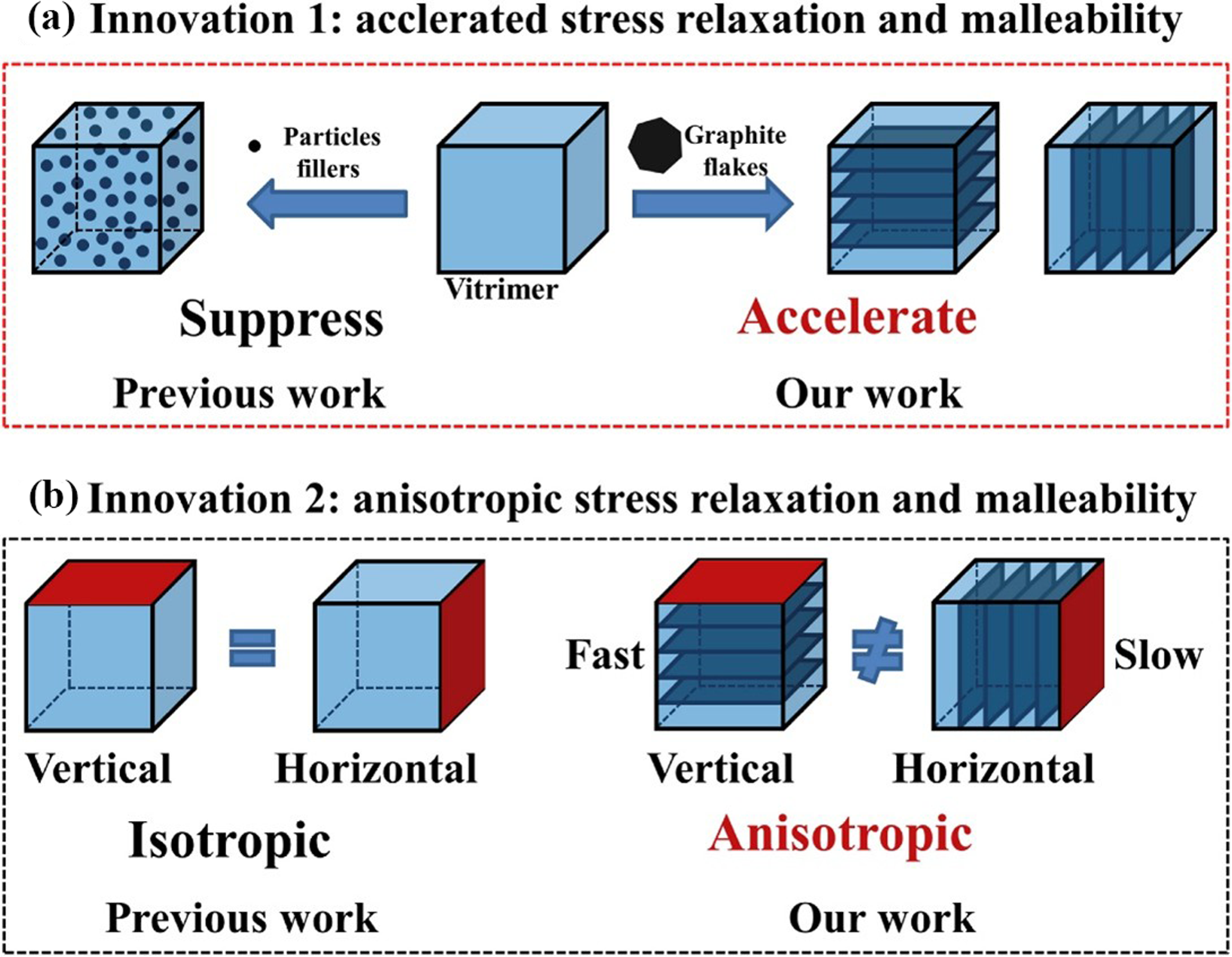
improved the dynamic property of Vitrimer resin by introducing neatly arranged graphite flakes. Figure 26(a) presents carbon fiber/single-layer Vitrimer composite materials and carbon fiber/multi-layer Vitrimer composite materials. In contrast to the consistent effect of particle fillers, the neatly arranged graphite flakes enhanced the dynamic characteristics of carbon fiber/Vitrimer composite materials, accelerating stress relaxation behavior and optimizing ductility (Figure 26(b)). The innovations of Vitrimer composites based on aligned graphite flakes: (a) accelerated stress relaxation and optimized malleability, (b) anisotropic stress relaxation and malleability.
73
And Liu 74 develops a self-regulating heating unit for welding stainless steel-carbon fiber hybrids using a sandwich-structured Vitrimer film. The design combines negative/positive temperature coefficient carbon nanotubes and fibers for rapid heating and autonomous temperature control, boosting joint strength by up to 253.5% over conventional methods. Optimized layer thickness enhances fracture resistance through nanofiller pull-out while preventing thermal degradation. The system enables efficient electrothermal repair (21.5% better than hot pressing) via Vitrimer network reorganization, achieving temperature regulation, multi-cycle repairability, and mechanical robustness for sustainable hybrid structure welding.
Carbon fiber/bio-based Vitrimer
At present, research on biobased carbon fiber composites mainly focuses on optimizing precursor selection, improving preparation processes, and enhancing performance stability. Lignin and cellulose have become research hotspots due to their renewability and low cost, but issues such as purification, spinning, and carbonization efficiency need to be addressed. New processes such as plasma assisted carbonization 75 and catalytic pyrolysis 76 can reduce energy consumption and improve fiber performance. Interface modification techniques, such as nano coatings, are used to enhance the adhesion between fibers and resins.
Vanillin accounts for a large proportion in the preparation process of Vitrimer, and ester compounds, amine compounds, and acid compounds derived from vanillin have been mentioned in many studies of carbon fiber composite materials. An innovative chemical recycling method allows for efficient delamination of carbon fiber layers, supporting cost-effective remanufacturing. Green chemistry and simplified life cycle assessments were used to evaluate the environmental impact of the manufacturing process. Tian 77 cured bisphenol A diglycidyl ether with an imine hardener derived from vanillin and included DOPO-HQ as a reactive flame retardant. The results indicated that the recyclable carbon fiber reinforced composite material has a flame retardant temperature above 135°C, bending strength of 480.4 MPa, and a bending modulus of 31.7 GPa. The flexural strength and flexural modulus of the recycled EVHQ2.0/CF composite material are 440.7 MPa and 25.5 GPa with 91% recycling efficiency.
Above-mentioned carbon fiber composite holding recycling potential principally focus on effective flame retardancy, which is crucial for developing recyclable composites with fire resistance and enhanced mechanical properties. As a refractory composite material Zamani 78 focuses on sustainable manufacturing of carbon fiber reinforced polymer (CFRP) composites using low-cost, rapid techniques and renewable materials. A recyclable vanillin-based Schiff base polyimide network was developed, cured without organic solvents, and utilized in CFRP fabrication via compression molding. The resulting composite exhibited excellent fire retardancy and impressive mechanical properties, with tensile strength of 427 MPa and modulus of 45 GPa. It achieved excellent UL-94 V0 rating in vertical combustion testing, surpassing traditional epoxy resin CFRP. And Wang 79 explores another recyclable and reprocessable vanillin-based epoxy Vitrimers, highlighting their potential for carbon fiber (CF) recycling. Notably, composites cured with Isophoronediamine achieved superior tensile strength of 571 MPa and Young’s Modulus of 11.7 GPa compared to Bisphenol A epoxy/CF and other Vitrimers. The imine exchange reaction enables complete recyclability, though mechanical and thermal properties are slightly reduced, underscoring their potential as lignin-based materials.
The two types of carbon fiber composite materials prepared by Vitrimer derived from vanillin mentioned above have only undergone explanatory experiments, without exploring the optimal conditions for repair. Perrin
80
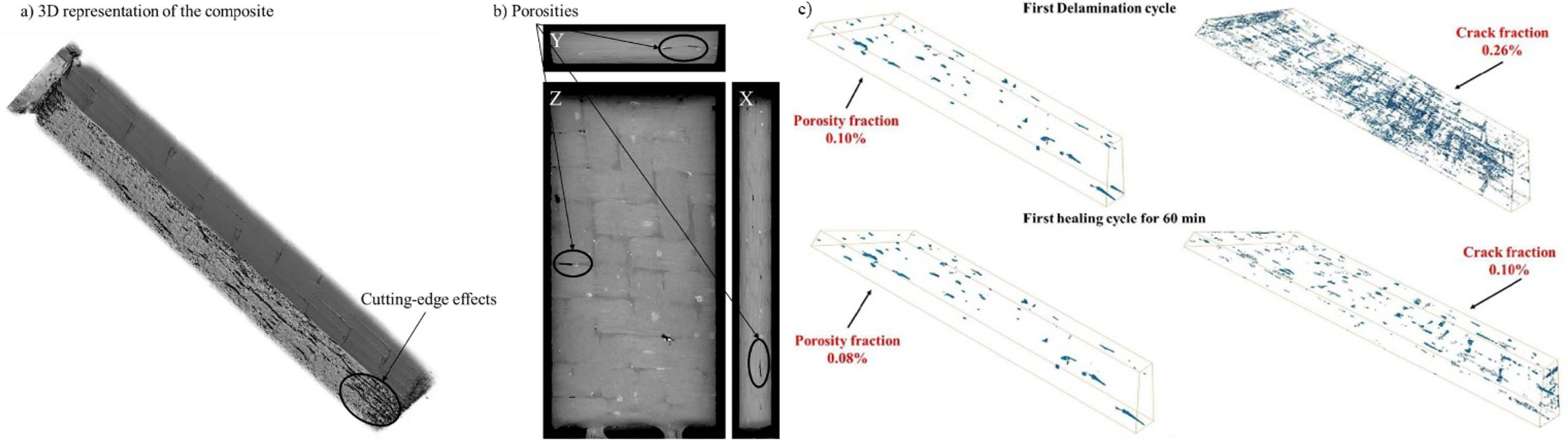
aimed to explore the self-healing ability of a new type of composite laminate (Figure 27(a) and (b)) composed of bio-based benzoxazine cross-linkers in the peeling-self-healing cycle. To evaluate the interlaminar shear strength (ILSS) and degree of damage, the study used three-point bending test short beam shear specimens. The self-healing process is achieved by applying a pressure of 1 MPa at 170°C. Three different sets of self-healing experiments were conducted under three different thermal cycles (1, 10, and 60 min) (Figure 27(c)). The degree of repair of interface damage was evaluated through four cycles of peeling-self-healing. Although the ILSS value gradually decreased in each cycle, the sample showed significant recovery ability after 60 min of self-healing treatment. After three cycles, ILSS recovered 80%, highlighting the efficient self-healing properties of composite materials based on block cross-linking. X-ray micro CT scans of 20 × 10 mm composite laminates before ILSS testing revealed: (a) a three-dimensional reconstruction, (b) a two-dimensional cross-section displaying rod-shaped voids situated among clusters, (c) 3D observation of porosities and cracks in blue under first delamination -healing cycle.
80
In addition to vanillin, there are also many studies on the preparation of carbon fiber/Vitrimer composites using other bio based materials. Hong 81 reports robust Vitrimer films prepared from an aqueous ternary solution of tannic acid (TA), boric acid (BA), and polyvinyl alcohol (PVA), featuring dynamic B-O-C covalent bonds. Carbon fiber-reinforced Vitrimers (CFRVs) show exceptional strength (570 MPa) and modulus (21 GPa) while retaining recyclability. Recycled Vitrimer solutions can reproduce films and CFRVs with properties akin to originals, highlighting their potential for sustainable carbon fiber composites. The tensile strength of the reproduced CFRV (568 MPa) recovered to almost 100% of that of the original CFRV (569 MPa), whereas the recovery efficiency of the Young’s modulus was recorded as 67%
And Sangaletti
82
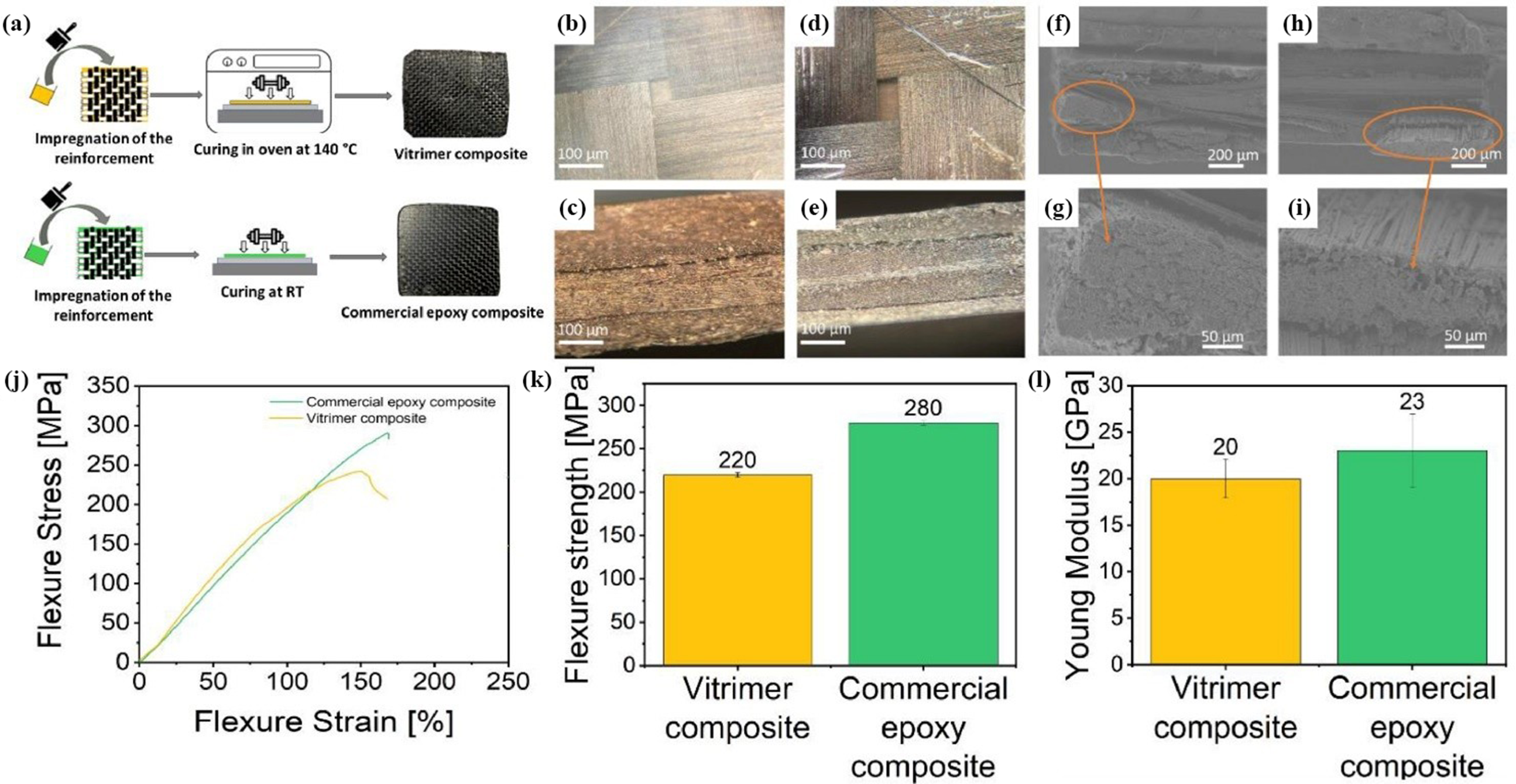
developed a recyclable, environmentally friendly Vitrimer resin from epoxidized linseed oil and a boronic ester cross-linker (Figure 28(a)–(i)). The average elastic modulus of this Vitrimer composite material is 20 GPa, and the bending strength is 220 MPa, as shown in Figure 28(j)–(l). Moreover, the composites could be conveniently recycled under mild conditions by reversible hydrolysis of the boronic ester crosslinks using aqueous EtOH, recovering both the carbon fiber reinforcement and the Vitrimer matrix. (a) Fabrication of the carbon fiber-reinforced composites, (b–c) Optical microscope images of the top surface and the cross-section of the Vitrimer composite and (d–e) commercial epoxy composite, (f–g) SEM images of the cross-section of the Vitrimer composite and (h–i) the commercial epoxy composite, (j) Typical flexural curve, (k) flexural strength and (l) Young Modulus of the Vitrimer composite and the commercial epoxy composite.
82
Xu 83 addresses the challenge of closed-loop recycling in carbon fiber-reinforced polymers (CFRPs) by developing a Vitrimer matrix from hydroxy-terminated hyperbranched polyesters (HBPs), rosin-derived fumaropimaric acid (FPA), and glycerol triglycidyl ether (GTE). The resulting FPA/GTE/HBP Vitrimer exhibit excellent mechanical properties due to the reactive HBP and rigid FPA, enabling self-healing and reprocessing. Composites reinforced with CFs show high mechanical strength and interlayer shear strength. The matrix can be degraded and closed-loop recycled using ethylene glycol without compromising the recycled CFs’ properties. This approach presents an eco-friendly solution for recyclable CFRPs.
Carbon fiber/ester-based Vitrimer
For ester based carbon fiber/Nitrimer composites, the main source is traditional fatty esters, derived from compounds extracted from petrochemicals, and is the most common type of carbon fiber/Nitrimer composite material. Adding Vitrimers containing fatty chains can endow carbon fiber composite materials with higher strength, like A Vitrimer composite (CF/Vx) was synthesized by Fang 84 from DGEBA and GA using Zn (acac)2 catalyst, enabling stress relaxation and reprocessability. Optimized CF/V0.05 exhibited a low relaxation time (1.0 × 10−3 s) and high activation energy (60 kJ/mol). Its flexural modulus dropped from 87 GPa (RT) to 4.62 GPa (220°C), allowing thermoplastic forming. A lid-shaped part was successfully fabricated via thermoforming. Additionally, CF/V0.05 achieved 128% repair efficiency for interlaminar fractures under heat and pressure. These reprocessable and repairable composites show great potential for engineering applications.
Fang 85 prepared another new type of carbon fiber reinforced Vitrimer epoxy resin composite material and studied its interlayer repair property and thermoplastic forming mechanism. The Vitrimer exhibits a Tg of 92.8°C and significant stress relaxation above Tg. Three-point bending tests confirmed excellent repair efficiency (nearly 100%) at 180–220°C under 5 MPa for 1.5–2 h. At 180°C for 1 hour of hot pressing, the bending strength of the sample is 98.66% of the original sample (598 MPa), and the bending modulus is 90.32% of the original sample (62 GPa); After hot pressing at 200°C for 1 hour, the bending strength and bending modulus of the sample were 95.99% and 75.81% of the original sample, respectively; After hot pressing at a higher temperature of 220°C for 1 hour, the bending strength and bending modulus of the sample reached 96.15% and 91.93% of the original sample, respectively. A 3D structural part was successfully fabricated via hot-pressing (200°C, 5 MPa, 2 h), demonstrating V-CFRP’s reprocessability.
This high-strength and high modulus carbon fiber/Nitrimer composite material requires a certain amount of catalyst in the process of matrix preparation, and the purification of the product cannot achieve purity. It is inevitable that there will be a certain amount of catalyst residue, and the residual catalyst also has a direct impact on the performance of the composite material. So at present, research is gradually developing towards the direction of catalyst free and green environmental protection. Zhang
86
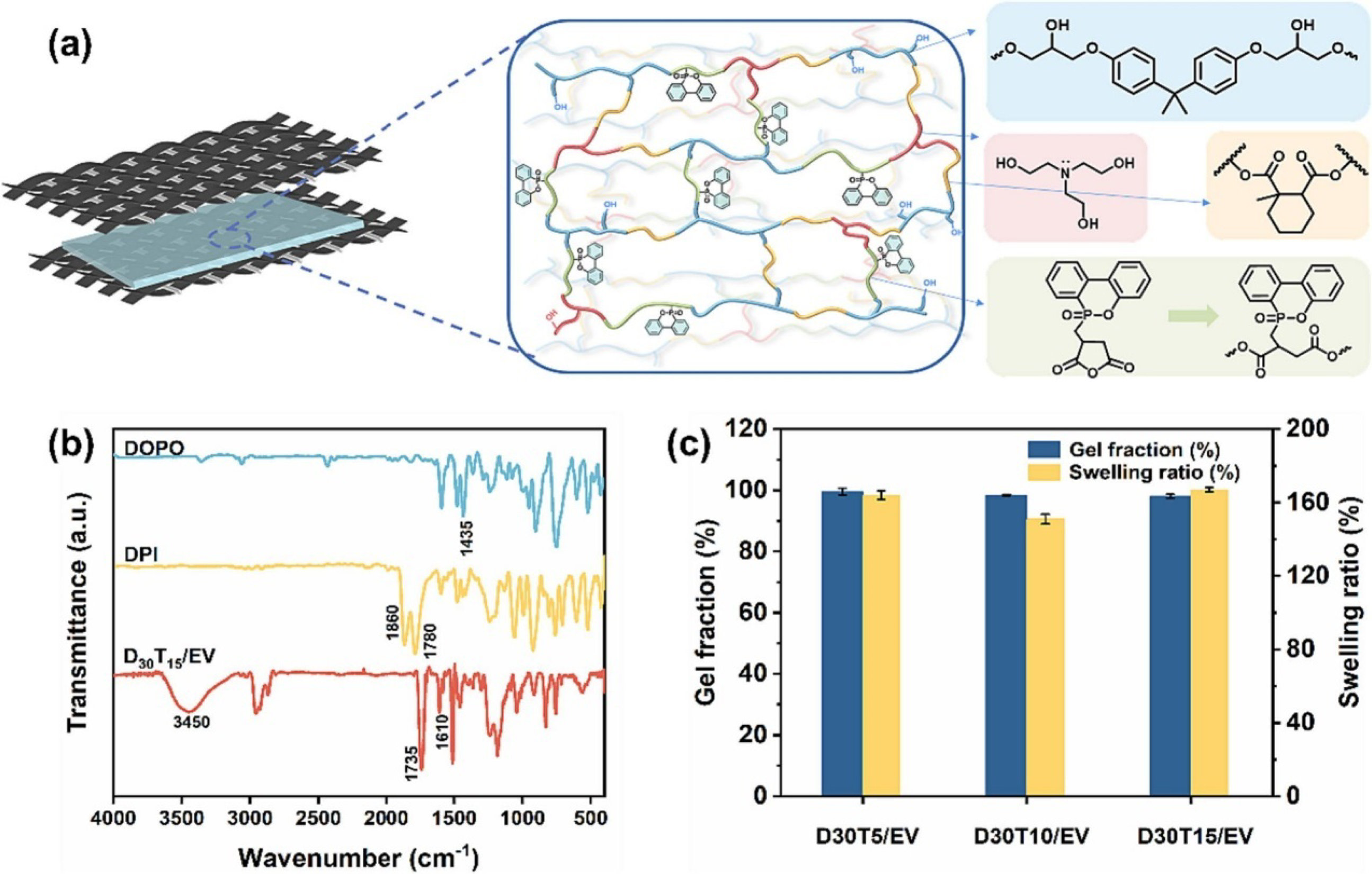
developed a catalyst-free epoxy glass transition material (Vitrimer) featuring flame retardancy and multiple reprocessing capabilities, applied to carbon fiber reinforced composite materials, and advancing the development of cost-effective and sustainable fire protection applications. Figure 29(a)–(c) displays the use of phosphoric anhydride (DPI) which derived from phenanthrene and triethanolamine (TEOA) as multifunctional ester exchange modifiers addresses both flame retardancy and recyclability issues. The incorporation of DOPO-derived curing agents as partial substitutes for anhydride-based ones significantly enhances the mechanical performance of the composites. Specifically, D30T15/EV/CF demonstrates superior flexural strength (730.3 MPa), a 15.9% improvement in interlaminar shear strength (ILSS), and a 17.8% increase in impact strength compared to D0T15/EV/CF, highlighting the effectiveness of this modification strategy. (a) Schematic representation of the dynamic covalent network, (b) FTIR spectra of DOPO, DPI and D30T15/EV, (c) Gel fraction and swelling ratio of the DT/EVs.
86
Mithil. 87 reported a trimer system that can repeatedly reverse fatigue damage by heating the material above its topological freezing transition temperature. As fatigue-induced damage accumulates in the trimer matrix, this method can achieve intermittent damage healing. It was found that the fatigue failure of vitreous and carbon fiber reinforced vitreous (vCFRP) can be postponed indefinitely. The maximum tensile stress in the bending specimen of vCFRP is 448.47 ± 52.8 MPa. The vCFRP was cured by heating at ∼ 150°C for ∼ 1 hour under a pressure of ∼ 200 kPa, and exceeded the baseline lifespan of the vCFRP after 25 healing cycles.
Fang
88
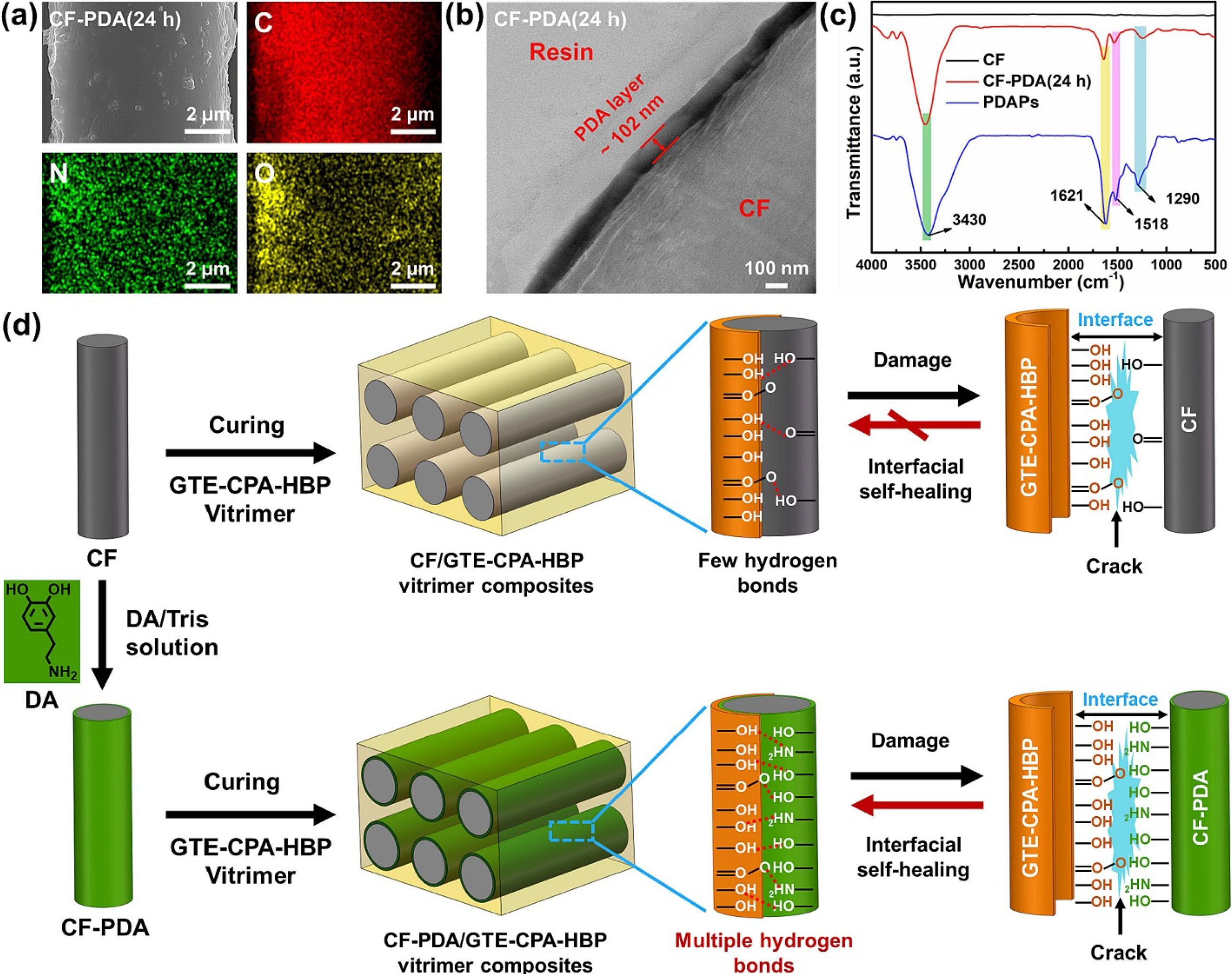
found traditional carbon fiber-reinforced epoxy composites (CFRECs) pose sustainability challenges due to their non-self-healing and non-recyclable nature. To address this, we designed a carbon fiber/Vitrimer (CF/Vitrimer) composite with dual self-healing (resin and interface) and dual recyclability (Vitrimer resin and CF) (Figure 30). The Vitrimer resin rapidly self-heals scratches (>95% recovery in 5 min). A self-healing interface between functionalized CF and Vitrimer maintains 77.4% efficiency after 5 healing cycles. Additionally, CF/Vitrimer composites degrade efficiently in ethylene glycol, enabling resin and CF recovery. This work highlights the potential of self-healing CF/Vitrimer composites for sustainable development. Preparation and interface self-healing mechanism of CF/GTE-CPA-HBP and CF-PDA/GTE-CPA-HBP trimer composites: (a) SEM images and corresponding EDS elemental maps of C, N, and O in the same area and (b) Cross-sectional TEM images, (c) FTIR of desized CF, CF-PDA (24 h), and PDAPs, (d) Preparation of CF/GTE-CPA-HBP and CF-PDA/GTE-CPA-HBP Vitrimer composites and its mechanism of interfacial self-healing.
88
Carbon fiber/amine-based Vitrimer
Amino-based carbon fiber/dendrimer composites have garnered significant attention due to their unique properties and potential applications. These materials primarily fall into two categories: those with imine-based structures and those utilizing polyurethane as the matrix. Their key advantages include low activation energy requirements for reactions and rapid responsiveness to fatigue damage in carbon fiber composites. However, challenges remain, such as the high-temperature demands during preparation and room for improvement in mechanical performance. Addressing these limitations has spurred innovative research into alternative composite designs, including self-healing and recyclable systems.
In this context, Li 89 made notable strides by developing a carbon fiber/Vitrimer composite with dual self-healing and recyclable capabilities. Their work focused on evaluating the material’s self-healing efficiency by creating controlled damage and measuring recovery times at varying levels. Additionally, the team explored the composite’s degradability, successfully reclaiming reusable carbon fibers—a critical step toward sustainable material design. This research highlights the potential of Vitrimer-based composites to overcome durability and environmental challenges, paving the way for further advancements in the field.
The versatility of Vitrimer matrices is further exemplified by Kim, 90 who engineered an antibacterial carbon fiber-reinforced plastic (CFRP) incorporating imine-functionalized Vitrimers. By synthesizing a liquid curing agent without purification, they streamlined the integration of the Vitrimer matrix with commercial epoxy. The study demonstrates that Vitrimer-based carbon fiber composites achieve competitive mechanical properties compared to traditional composites, with a tensile strength of 716 MPa (vs 745 MPa), tensile modulus of 75.9 GPa (vs 75.2 GPa), and interlaminar shear strength (ILSS) of 45 MPa (vs 51 MPa). While showing a slight reduction in strength metrics, the Vitrimer composites offer unique advantages including dynamic bond exchange for recyclability, reshaping capability, and intrinsic antibacterial properties (84.9%). This combination of structural performance and functional benefits positions Vitrimer composites as promising sustainable alternatives for advanced applications.
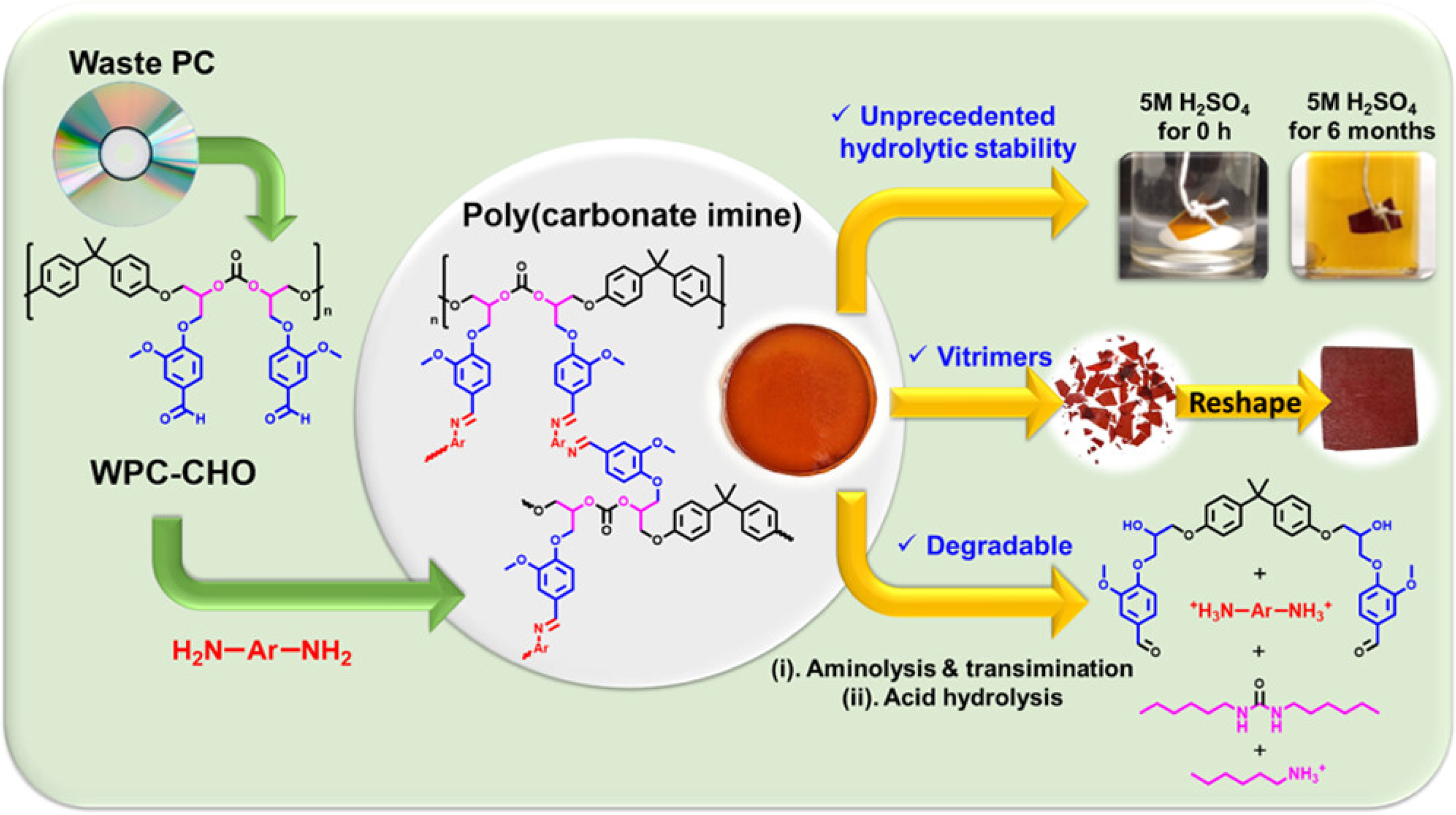
Expanding the scope of Vitrimer applications, Chen
91
presents a sustainable upcycling of waste polycarbonate (WPC) into WPC-derived aldehyde (WPC-CHO), which is then transformed into poly (carbonate imine) Vitrimers (PCIs). Figure 31 presents that this Vitrimer exhibits exceptional acid resistance (withstanding 5 M H2SO4 for 6 months), robust thermal/mechanical properties, and Vitrimer-like reprocessability at 240°C. The tensile stress of composite materials is 890 MPa, and after repair, it can achieve almost 100% repair. They enable catalyst-free degradation via hexylamine, allowing carbon fiber recovery from PCI-based CFRPs. The work offers a green solution for circular materials with extended acidic-environment applications. Upcycling of waste polycarbonate to poly(carbonate imine) Vitrimers (PCIs), aminolysis degradation, and unprecedented acid resistance of PCIs in 5 M H2SO4.
91
To address CFRP waste challenges, a catalyst-free bio-based polyurethane (V-PU) was synthesized using vanillin derivatives and PCL, featuring dynamic phenol-carbamate bonds. Through using V-PU, Jian 92 addresses the recycling challenge of carbon fiber-reinforced polymer (CFRP) waste by developing a catalyst-free, self-healing, and recyclable vanillin-based polyurethane (V-PU) resin derived from biodegradable PCL and bio-based vanillin. When used as a CFRP matrix, the composites achieve an interlaminar shear strength (ILSS) of 41 MPa and 85.34% healing efficiency. The tensile strength and Young’s modulus of the composite material prepared with recycled resin and carbon fiber are 298.32 MPa and 22.28 GPa, respectively. These values are 92.23% and 88.28% of the original composite material, respectively, strongly demonstrating the excellent closed-loop recovery ability of V-PU15/CF composite material. Leveraging dynamic phenol-carbamate bonds, the CFRP enables closed-loop recycling of carbon fibers and resin via solvent dissolution, offering an eco-friendly solution for sustainable materials and circular economies.
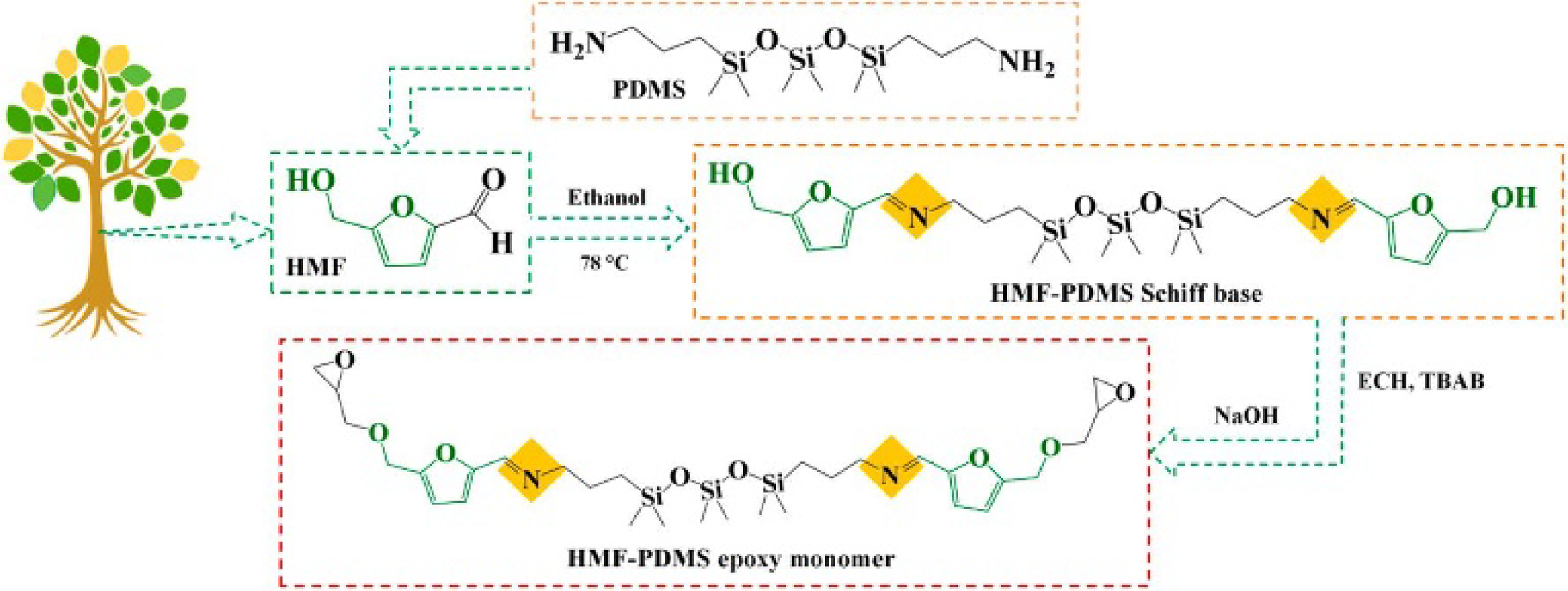
Nabipour et al.
93
A novel bio-based Schiff base epoxy thermoset was synthesized from furan derivatives, demonstrating exceptional recyclability, mechanical properties, and flame retardancy (Figure 32). When cured with 5,5-methylenedifurfurylamine, it achieved a 54% char yield, Tg of 207°C, storage modulus of 2695 MPa (30°C), and UL-94 V-0 rating—outperforming bisphenol A epoxy. Its dynamic Schiff base network enabled nondestructive recycling of carbon fiber composites under mild acid, offering a sustainable high-performance alternative for recyclable CFRPs. Synthetic scheme for bio derived Schiff base-containing compounds.
93
Carbon fiber/disoulfide-based Vitrimer
The application of disulfide bond based Vitrimer in carbon fiber composite materials is relatively limited, mainly due to its poor chemical stability 94 (prone to fracture in humid/oxidizing environments), weak interface bonding with carbon fiber 95 (requiring high temperature/catalyst triggered dynamic exchange), difficulty in balancing mechanical properties and recyclability (dynamic properties leading to significant decrease in modulus), and potential environmental risks (sulfur-containing catalyst by-products). The current research is optimized through hybrid dynamic bonding or fiber surface modification, but industrialization still faces challenges.
Tang 96 presents a novel guaiacol-based epoxy resin (BGF-EP) synthesized from renewable resources, replacing petroleum-based bisphenol A. Cured with 4-aminophenyl disulfide, BGF-EP-AFD exhibits excellent thermal stability (Tg: 143°C, Td5%: 295°C) and mechanical properties (tensile strength: 55.54 MPa). It demonstrates stress relaxation and can be reprocessed into a complete film. Additionally, an efficient method for dissolving the cured resin allows for recycling of carbon fiber reinforced composites in a closed-loop system using a 50% DMF/50% β-ME solution at room temperature, maintaining fiber integrity for future use.
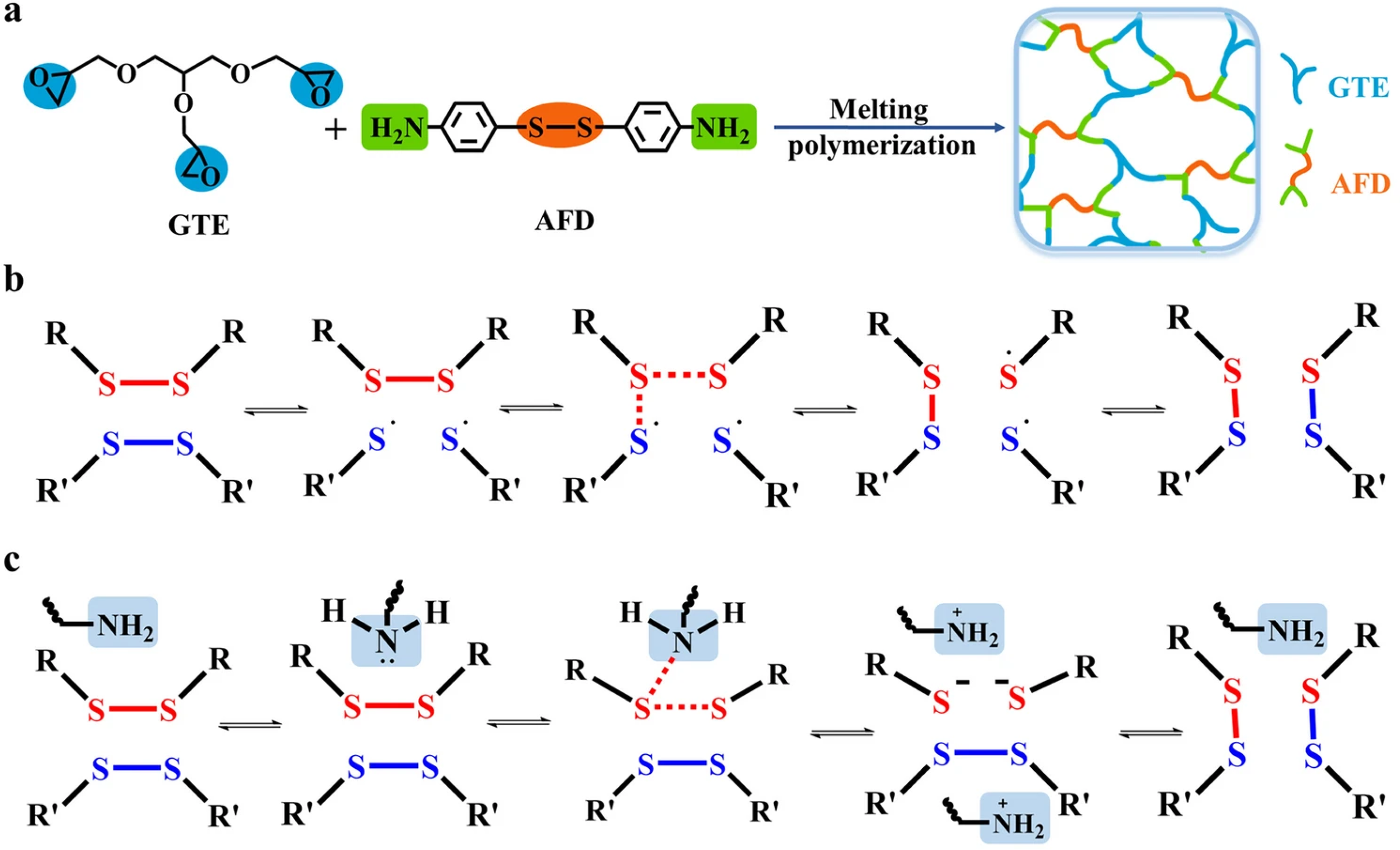
Zhang
97
developed aromatic disulfide Vitrimers by incorporating AFD as both curing agent and catalyst (Figure 33(a)-(c)). The optimized GTE-AFD1.25 Vitrimer demonstrated outstanding properties: a 99.27°C glass transition temperature, high tensile/flexural strength (94.62/157.16 MPa) and modulus (3151/2970 MPa), along with ultrafast dynamic exchange (14 s relaxation at 180°C) enabling 96.25% self-healing efficiency. The material exhibited triple-shape memory and excellent reprocessability, while achieving complete degradation within 5 hours in DTT/DMF solution. The GTE-AFD1.25-based CFRP composites showed superior mechanical performance (721 MPa flexural strength, 30,334 MPa modulus) while maintaining recyclability and shape-memory functionality, enabling efficient carbon fiber recovery. This work presents a high-performance, multifunctional Vitrimer system for sustainable CFRP applications. (a) Curing reaction, (b) 2 + 1 radical-mediated mechanism, (c) amino-catalyzed dynamic disulfide bond exchange mechanism.
97
Dong 98 developed an amine-catalyzed disulfide-based Vitrimer enabling rapid stress relaxation (14 s at 180°C) and efficient carbon fiber composite recycling. The dynamic network demonstrates fast self-healing, shape memory, and 5-h resin degradation, while maintaining high mechanical performance. Applied as a matrix, it facilitates carbon fiber recovery, offering a sustainable solution for automotive/aerospace composites through accelerated reprocessing and reduced energy-intensive recycling steps. The tensile strength of BDEF-EP-AFD/CF composite material is about 225 MPa, the tensile modulus is about 33 GPa, and the interlaminar shear strength (ILSS) is about 37 MPa, reaching 86%, 75%, and 65% of DGEBA-AFD/CF composite material, respectively. The small difference in tensile modulus indicates that this bio based Vitrimer has high mechanical strength.
Carbon fiber/siloxane-based Vitrimer
Silicon based Vitrimer carbon fiber composite materials utilize dynamic Si-O-Si bonds to achieve reshaping and fiber recovery under mild conditions (80–150°C), with both heat resistance (Tg > 180°C) and weather resistance. The current challenges include insufficient mechanical performance (ILSS 30-50 MPa) and slow dynamic response (relaxation >300 s), and efforts are being made to optimize the interface and exchange rate through nanohybridization/side chain modification, promoting lightweight applications in aviation.
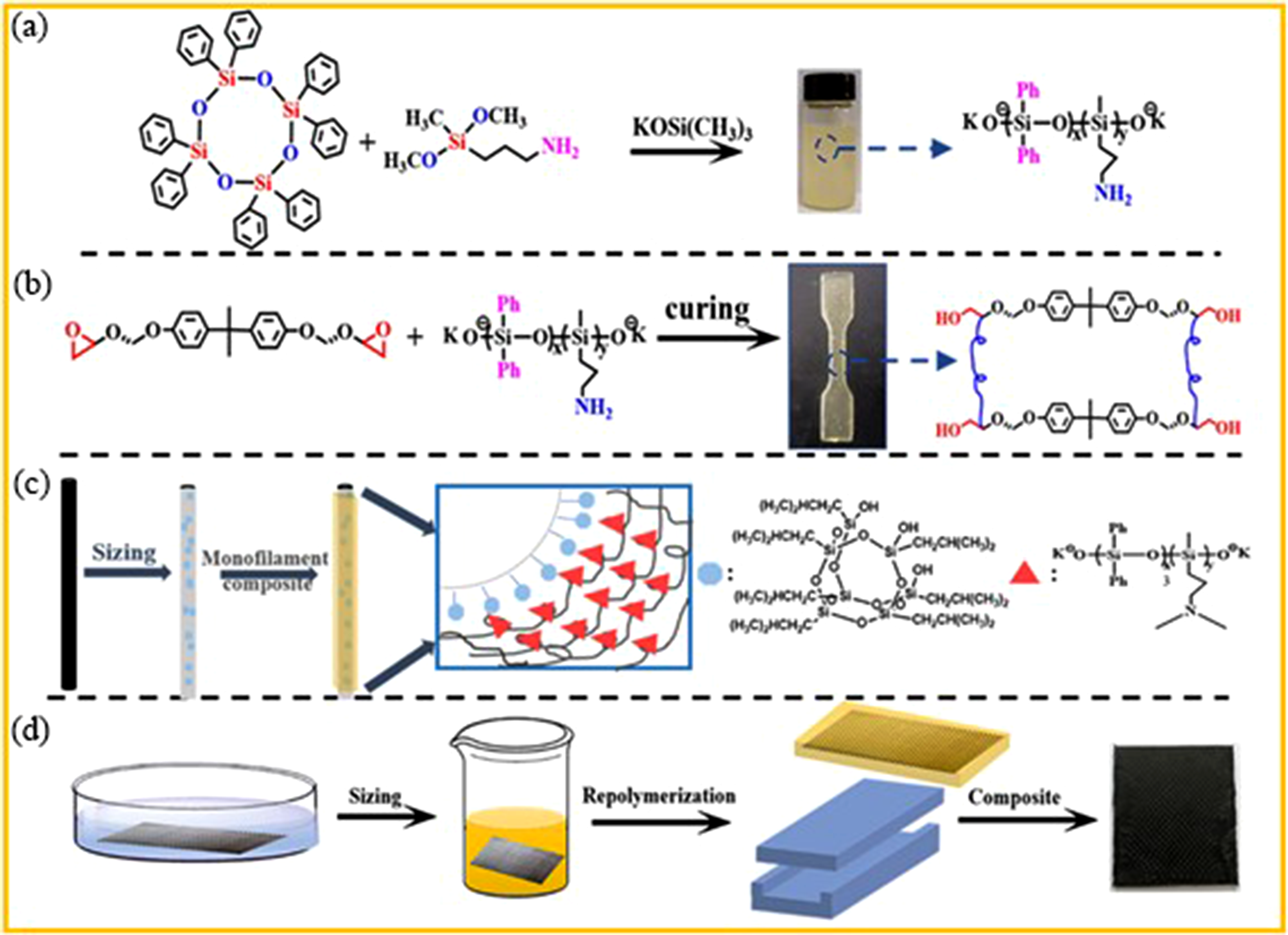
Xu
99
developed an integrated self-healing CFRP composite with simultaneous matrix and interface damage repair by incorporating siloxane dynamic bonds into both the resin matrix and interface through a simple method. Figure 34(a) and (b) shows the process for making the silicon resin and it exhibites excellent thermal and mechanical properties, achieving a healing efficiency of 65.4%, balancing robustness and reparability. The preparation method for CFRP is illustrated in Figure 34(c) and (d) and the carbon fiber surface activation enhanced interfacial adhesion without compromising strength, with initial and healed interfacial shear strengths (IFSS) of 68.11 and 43.92 MPa, respectively. The composite showed interlaminar shear strength (ILSS) and flexural strength (FS) of 30.73 and 351.46 MPa, retaining 96.4% and 89.1% post-healing. This work provides an effective strategy for durable, high-performance CFRP composites. (a) Synthesis and chemical structure of crosslinking agents. (b) The synthesis and structure of epoxy resin. (c) The treatment and surface structure of carbon fibers (CFs). (d) Preparation of Carbon Fiber Reinforced Polymer (CFRP) Composite Materials.
99
Chen 100 developed a multifunctional epoxy Vitrimer with excellent shape memory, reprocessability, and low dielectric constant (2.75). The flexible siloxane chains and internal tertiary amines in ETOD-SA enhanced stretchability (135%), reversible deformation (30 cycles), and rapid self-healing (36 μm scratch in 10 min). ETOD-SA-CF composites also exhibited self-repair, shape memory, and degradability, demonstrating potential for recyclable carbon fiber composites. This work provides an effective strategy for sustainable, high-performance Vitrimers, offering new possibilities for eco-friendly applications.
And Debsharm 101 have developed siloxane-containing Vitrimers with ultrafast stress-relaxation properties (relaxation times <10 s) by leveraging a catalyzed siloxane exchange mechanism. Using 1,5,7-triazabicyclo4.4.0dec-5-ene (TBD) as an organic catalyst in the presence of hydroxyl groups, these Vitrimers exhibit rapid dynamic behavior at practical temperatures (180–220°C). The system, based on an epoxy resin and siloxane-amine hardener, enabled the fabrication of glass fiber-reinforced composites via vacuum-assisted resin infusion. The resulting composites were thermoformable, allowing for shape reconfiguration and potential reuse, demonstrating promise for sustainable high-performance materials. This approach combines fast reprocessability with industrial scalability, offering a pathway for recyclable thermoset composites.
Conclusion and prospect
Vitrimer materials, due to their unique dynamic exchange ability, achieve self-healing, reprocessing, and recyclability through bond exchange reactions such as ester exchange, transamidation, disulfide exchange, and imine metathesis, and are used in the preparation of various composite materials to cope with different degrees of fatigue or damage in different environments. As a special resin material between thermosetting resin and thermoplastic resin, Vitrimer material deserves our attention to whether it can integrate the advantages of thermosetting resin and thermoplastic resin. However, from the current research results, scientists cannot fully balance the mechanical properties and dynamic repair performance of Vitrimer resin. Whether it is the dynamic ester exchange reaction mentioned in the article or the low activation energy dynamic imine reaction, its dynamic performance may be lacking when mechanical properties are the dominant direction.
Carbon fiber reinforced Vitrimer composites represent a significant advancement in sustainable high-performance materials, combining the mechanical strength of carbon fiber with the dynamic performance of Vitrimer. At present, there is relatively little research on carbon fiber Vitrimer composite materials. Most of them simply combine carbon fibers with Vitrimer resins containing different bond exchange systems to explore their fatigue resistance, tensile resistance, and impact resistance. Reprocessability is currently the main direction of research on carbon fiber Vitrimer composites. However, studying from the perspective of resin alone is not enough to improve the reprocessing ability of carbon fiber Vitrimer composites. Research should also take into account the carbon fiber itself and its connection with resin. Starting from the interface bonding ability, combined with the modification research of carbon fiber itself, the surface roughness can be improved, or chemical active groups can be introduced on the surface of carbon fiber to gradually enhance the survival ability of composite materials in different environments.
Therefore, the research direction of carbon fiber Vitrimer composite materials in the future is bilateral development. It is necessary to pay attention to the strength and modulus of carbon fiber itself, modify the surface of carbon fiber, and select suitable Vitrimer resin to adapt to the modified carbon fiber material. The two complement each other, jointly improving the environmental adaptability of carbon fiber composite materials and slowing down their fatigue failure in key fields such as aerospace.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
No new data were generated during this study. All analyzed datasets are publicly available and cited appropriately.
