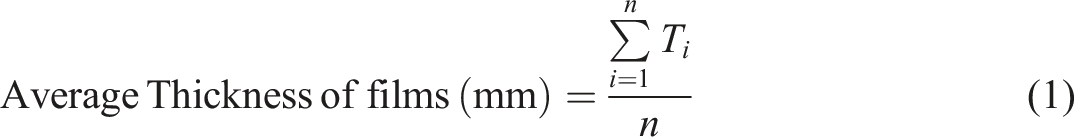Abstract
This study investigates the development of environmentally friendly thermoplastic biocomposite films based on a polyvinyl alcohol matrix, utilising underutilised Sapindus mukorossi shell particulates as bio-based fillers. SMSP was added at various loadings, and the resulting composites were thoroughly characterized. Spectroscopic analysis confirmed the presence of strong intermolecular hydrogen bonding between PVA and SMSP, even without chemical modification, while X-ray diffraction revealed enhanced molecular ordering. Thermal stability improved with an increased char yield from 0.83% for pure PVA to 17.6% with 25 wt% SMSP. Mechanical properties peaked at a tensile strength of 48.4 MPa, Young’s modulus of 69.9 MPa, and puncture strength of 54.5 MPa with 20 wt% SMSP, while higher filler contents caused minor agglomeration and reduced efficiency. It was observed that the barrier properties increased, reflected by a reduction in water uptake and water vapour permeability with increasing diffusion tortuosity. The water absorption and solubility were reduced to 31.1% and 34.6%, respectively, while the water vapour permeability demonstrated almost a 48% reduction in the case of the highest loaded SMSP content. AFM images showed increased surface roughness, enhancing mechanical interlocking. The optical transparency reduced with an increase in filler content as a result of light scattering, showing effective UV-shielding characteristics. This work hence establishes that the mechanical, thermal, and barrier properties of PVA films are improved upon reinforcement with Sapindus mukorossi shell particulates, illustrating the potential of this underused bio-waste as a low-cost, biodegradable, and high-performance reinforcement for sustainable thermoplastic packaging applications.
Highlights
• With the addition of SMSP, PVA crystallinity increased from 32.5% to 50.2%. • At 25% SMSP loading, the char yield increased from 0.83% (PVA) to 17.6%. • At 20% SMSP content, the tensile strength increased to 48.4 MPa. • When compared to pure PVA, the water-vapour permeability was reduced by around 48%. • For 25% SMSP films, UV transmittance decreased to 54% at 410 nm.
Introduction
The extensive application of traditional petroleum-based plastics in packaging has imposed a substantial environmental impact since these have been attributed to disruption in the ecosystem as well as posing human health threats, while manufacturing synthetic plastics uses non-renewable fossil fuels and produces greenhouse gases.1,2 These concerns plastics are non-biodegradable and take decades to degrade in landfills and natural environments. 3 Soil and aquatic microplastic pollution have intensified the search for packaging materials that are biodegradable in nature to be able to degrade safely after use. 4 Bio plastics made from renewable feedstocks such as starch, cellulose, and polyvinyl alcohol (PVA) offer a potential route to reduce plastic waste and carbon emissions. 5 PVA is a water-soluble and biodegradable material under microbial action, but with tensile strength and film casting capability required for packaging materials. However, neat PVA generally has excellent hydrophilicity and low barrier properties that limit its use unless supported or blended with suitable natural filler materials6,7
Crop plant agricultural residues are a readily available and not yet fully utilized source of reinforcement fillers of this type. 8 Blending such wastes into PVA, or other biopolymer films serves two technological purposes: it enhances key material characteristics such as tensile strength, water vapour barrier, and thermal stability via hydrogen bonding or covalent association of hydroxyl and carboxyl moieties.9,10 Recent studies have shown that fillers such as orange peel powder, 11 ground nut shell powder, 12 and wood apple shell powder 13 improve not only the mechanical strength of PVA composites but also their rate of biodegradation by increasing the proportion of natural elements. Further, rice hull powder, 14 coconut shell powder, 15 putranjiva powder, 16 banana peel powder, 17 and lignin-based fillers have all been utilized to reinforce PVA, resulting in improvements in tensile strength, water resistance, thermal properties, and biodegradability.
Many of the naturally available resources, such as leftovers of fruits, vegetables, roadside trees, and solid wastes, are still underutilized as natural fillers in the bioplastic formations. One such underexploited natural resource is the Sapindus mukorossi shell, commonly referred to as soapnut shell, which is a by-product of the soapnut fruit, widely known for its natural surfactant properties.18,19 Sapindus mukorossi is a deciduous tree in the Sapindaceae family that grows in South China, the Indochina Peninsula, and India, and can reach heights of over 20 m. The fruit has two covering outer layers: the fleshy pericarp (fruit pulp) and the hard shell of the seed, both of which cover a soft inner kernel.20,21 This lipid composition renders Sapindus mukorossi kernel oil a potential renewable biodiesel feedstock with a sustainable and eco-friendly energy source. Recently, many Researchers have used the Sapindus mukorossi shell for various applications. Yadav et al. (2024) 22 utilised the Sapindus mukorossi pulp along with Azadirachta indica leaves (neem) solution as a chemical agent to treat the bagasse fibers due to its antimicrobial activity. Samal et al. (2019) 23 assessed bio sorbents prepared from Sapindus mukorossi reetha pericarp waste for the removal of methyl violet dye from wastewater. Porchezhiyan et al. (2016) 24 fabricated the hydrogel-based films with chitosan and silver nanoparticles synthesized from Sapindus mukorossi extract through microwave radiation. Han et al. (2020) utilized the Sapindus mukorossi shell extract as the carbon source, which presents a scalable approach for industrial-scale commercialization and production of BC. According to Saikia et al. (2022), 25 phytochemicals from Sapindus mukorossi seed shell extract controlled the size of the nanostructures and promoted effective catalysis of C–N bond formation in a size-controllable biogenic hydrothermal synthesis of CuO nanostructures. Kasilingam et al. (2023) 26 used hydrothermal synthesis of nickel oxide nano-clusters on nano-graphitized coil sheets (NNNGCS), which are made from Sapindus mukorossi extract and then annealed at 450°C, to create an inexpensive, environmentally friendly carbon-based working electrode.
Based on the detailed literature review, the Sapindus mukorossi Shells are not utilized anywhere as a natural reinforcement filler for biofilm fabrications. This study reports this research gap by exploring, for the first time, the use of nano-sized Sapindus mukorossi shell particulates (SMSP) as a reinforcement filler in PVA-based bio-composite films. Incorporating various proportions (5%-25%) of Sapindus mukorossi shell particulates (SMSP) into the PVA matrix is a novel approach that aims to leverage the inherent strengths of both components. The objectives of the work being proposed will be to investigate and analyze the effects of SMSP incorporation on the morphological, mechanical, thermal, and biodegradability characteristics of PVA biofilms. This research will aim to explore the benefits that can be derived through using agricultural waste products as a reinforcing material within polymer technology and will aim to set the foundation for more environmentally friendly biodegradable forms of polymer packaging.
Materials and Methods
Materials
Polyvinyl alcohol with a Molecular weight (Mw = 1, 25,000) and distilled water is procured from Sakshi Chem Sciences Private Limited, Nagpur, India. The Sapindus mukorossi fruits were collected from a local garden near Sivagiri village, Tamil Nadu, India.
Preparation of Sapindus mukorossi Shell Particulates and PVA/SMSP biofilms.
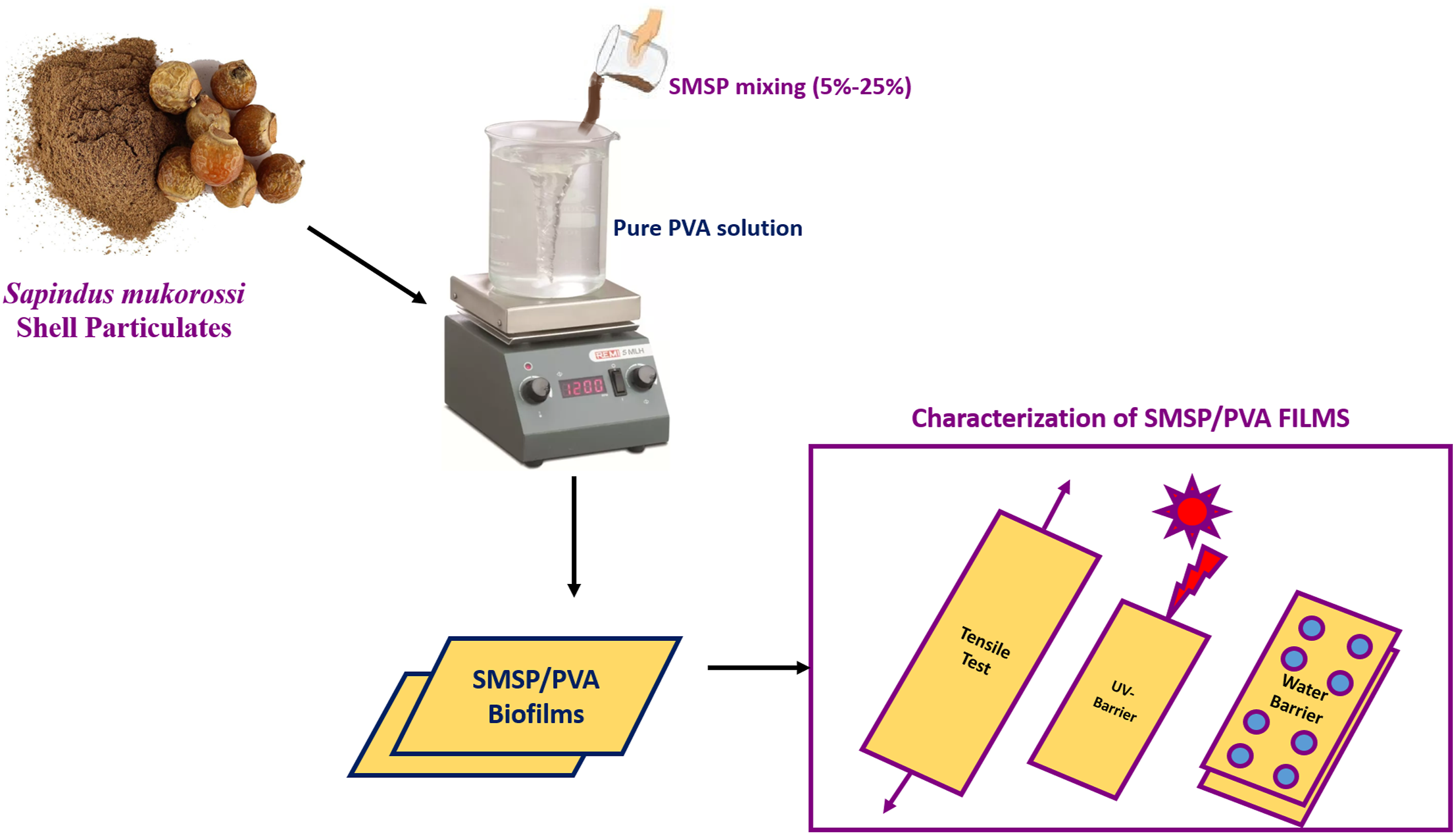
The Preparation process of Sapindus mukorossi shell particulates and SMSP/PVA films is shown in Figure 1. Preparation of Sapindus mukorossi shell particulates and SMSP/PVA films.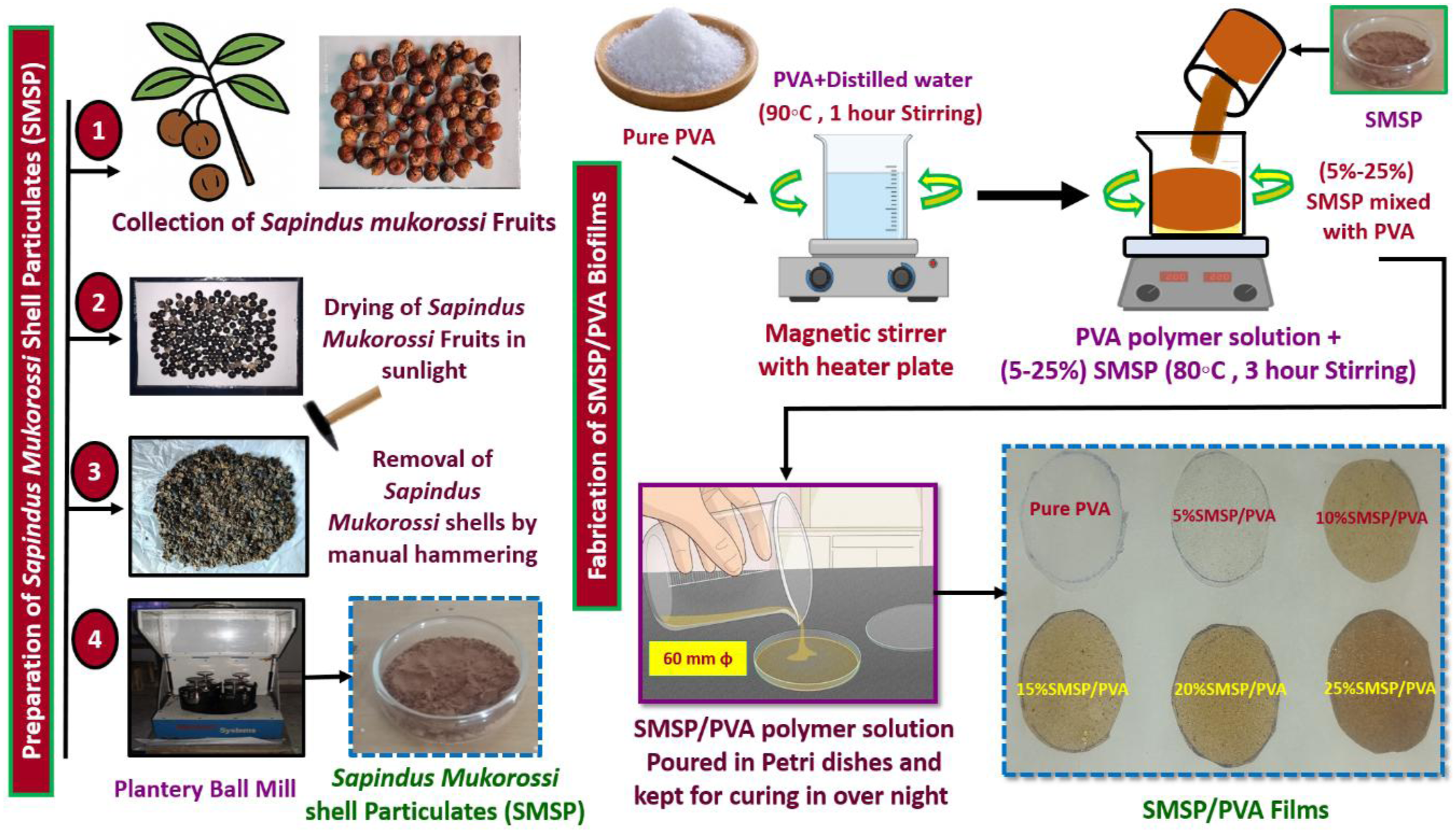
Sapindus mukorossi fruits were collected and dried in sunlight for 10 days. The shells of dried Sapindus mukorossi fruits were manually broken by using a hammer. The crunches of Sapindus mukorossi Shells are washed carefully with distilled water to remove the impurities, and oven-dried at about 60°C for 12 hours. The dried shells are then crushed and ball-milled for 6–8 hours to obtain nano-sized SMSP. For SMSP/PVA biofilms, an aqueous 20 mL of poly (vinyl alcohol) (PVA) solution is first prepared by dissolving PVA granules in distilled water with continuous magnetic stirring at 90°C for 1 hour until PVA granules are fully dissolved in hot water. The different proportions (5%, 10%, 15%, 20% and 25%) of SMSP are dispersed in distilled water and ultrasonicated for 30 minutes to ensure uniform suspension. The SMSP dispersion is then slowly added to the hot PVA solution and stirred magnetically at 80°C for another 3 hours to ensure homogeneous mixing. The resulting homogenous SMPS/PVA polymer solution is poured into circular Petri dishes for solution casting. The petri dishes were kept in an oven at 50°C for 24 hours to form a smooth SMSP/PVA biofilm. Finally, the dried SMSP/PVA films were carefully peeled off and kept at room temperature in a desiccator before use. The Interaction mechanism between SMSP and PVA is given in the Figure.
Experimental Methods
Film Thickness
To measure the thickness of SMSP Reinforced Polyvinyl Alcohol films, a digital micrometre (±0.001 mm precision) is used. The film should first be conditioned in a controlled environment (23°C and 50 % relative humidity) for 36 hours, following ASTM D882 standards.
27
Measurements should be taken at a minimum of five locations on each film, typically at the four corners and the centre, to account for thickness variations caused during casting and drying. During measurement, the film is placed on a clean, flat surface, and the gauge or micrometer is gently lowered onto the film to avoid compression, with the reading recorded once stabilized. The average thickness is calculated using equation (1).
Ti is the thickness at each point
n is the number of measurements.
Thickness of Pure PVA and SMSP/PVA biofilms.

Fourier Transform Interferometry
The chemical functional groups of SMSP/PVA biofilms were examined by using an RXI Perkin Elmer FTIR Spectrophotometer. The spectra are recorded in the range of 4000 -400 cm−1 with a resolution of 4 cm−1, averaging 32 scans to improve the signal-to-noise ratio.
X-ray Diffraction Test
XRD of Pure PVA and SMSP/PVA films is performed by cutting them into flat 10 × 10 mm pieces. The X-ray patterns of the film samples was analyzed by the X-ray Diffractometer, Bruker D8 Advance, using Cu Kα radiation (λ = 1.5406 Å) in θ–2θ (Bragg–Brentano) geometry, normally at 40 kV and 40 mA. Scans are taken over a 2θ range of 5°–80° with a step size of 0.02°–0.05°. The crystallinity index determined (CI) by using the Segal method (equation (2))
28
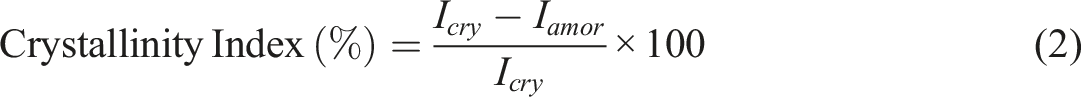
Icry = Intensity at the crystalline peak.
Iamor = Intensity at the amorphous baseline near the chosen reference.
Thermal Analysis
Thermal characteristics of pure PVA and SMSP/PVA biofilm were established with NETZSCH STA 449F3 − 1100-M thermal analyser. The analysis is carried out under a nitrogen flow at the rate of 50 mL/min to avoid oxidation, using a heating rate of 10°C/min from room temperature to 500°C.
Tensile Test
The tensile test for SMSP/PVA biofilms is performed on an INSTRON 3369 Universal Testing Machine under ASTM D882-18. 29 Specimens are obtained by cutting the biofilms into regular rectangular strips (usually 25 mm wide and 100 mm long). Testing is conducted at a constant crosshead speed of 50 mm/min until the sample breaks. The tests are carried out in triplicate, and the data are reported as mean ± standard deviation.
Puncture Test
Puncture analysis of composite films was conducted by using a texture analyzer attached to a puncture cell. Pure PVA and SMSP/PVA films were fixed into a circular cell having an internal diameter of 52.6 mm and punctured by a cylindrical stainless-steel probe having a diameter of 3 mm with a constant velocity of 1 mm/sec. Upon testing, the puncture force and the probe displacement at the point of film break-up were determined from the force displacement curves. The puncture deformation was calculated by assuming uniformly distributed stress over the film at the breaking point.
Surface Morphology
To analyze the surface morphologies of pure PVA and SMSP/PVA biofilm, a Tescan Vega (third-generation) SEM analyzer is utilized under high vacuum, with an accelerating voltage of 5–15 kV, a working distance of 10 mm, and capture images at magnifications of 500x to 10,000x. Coat the samples with a thin (5 -10 nm) layer of gold, using a sputter coater at 20–30 mA for 80 seconds.
Surface Roughness Parameters (AFM)
By using a PARK FX 40 Automatic AFM, the roughness of pure PVA and SMSP/PVA films was measured by initially cutting the biofilms into flat pieces of a small area 10 mm2. Fix the dried film samples smoothly on the AFM sample holder using double-sided adhesive tape to make it stable during scanning. Operate in tapping mode to prevent destroying the soft polymer surface, using a scan area of 5 × 5 µm to 20 × 20 µm, and a scan rate of 0.5 -1 Hz to obtain good images.
Water Absorption
The Pure PVA and (SMSP)/PVA biofilms water absorption test is performed as per ASTM D570-98.
30
Biofilm samples are reduced to a standard size of 25 mm × 25 mm and dried in an oven at 50°C for 24 hours or until the weight becomes constant, and then cooled in a desiccator, and the initial dry weight (W0) is measured. The samples are afterwards totally submerged in distilled water at 23°C for 10 hours. The specimens are then taken out after immersion, and the surface of the film is wiped clean with a lint-free cloth, and the wet weight (W1) is obtained. The percentage of water absorption is calculated using equation 4.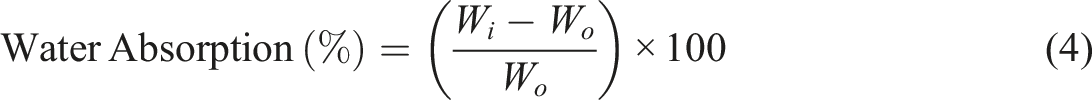
All tests are performed in triplicate, and results are reported as the mean ± standard deviation.
Solubility of Films in Water
The % solubility of PVA and SMSP/PVA films were analysed by gravimetric (dissolution–dry-weigh) technique as per ASTM D570.
31
The % solubility of films in water was calculated by equation (5).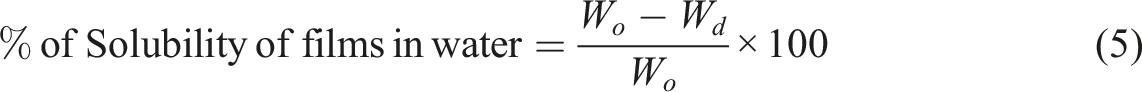
Water Vapour Permeability (WVP)
The gravimetric cup method, as specified in ASTM E96,
32
was used to measure the permeability of the PVA and SMSP/PVA films. Film samples (diameter = 5.0 cm) were placed over the mouth of a plastic test dish (diameter = 4.5 cm) that contained 15 mL of distilled water in such a way that the film closed the open face. To create a condition with almost zero relative humidity outside the film, each sealed dish was placed in a desiccator filled with about 1000 g of silica gel. Over the course of 12 hours, test dishes were weighed every 2 hours using an analytical balance. The WVP of films was calculated by equation (6).
UV-Transmittance
A multi-scan Sky High microplate spectrophotometer with a wavelength range of 200–800 nm, along with an integrating sphere, is used to study the optical characteristics of SMSP/PVA films. The total luminous transmittance (the amount of light that passes through the film) is used to assess transmissibility.
Soil burial Test
The biofilm sample soil burial test is done following ASTM D5988-18.
33
Biofilm samples are sliced into uniform pieces (40 mm × 40 mm), weighed, and taken as the initial weight (W0), and buried in natural soil to a depth of 10 cm in containers at 25°C and 50% soil moisture content to represent natural conditions. At set time intervals of 15, 30, 45, and 60 days, the samples are gently harvested, washed to dislodge soil particles, oven-dried at 50°C until a constant weight is achieved, and weighed (W1). The biodegradation is evaluated by calculating the percentage weight loss based on equation (7).
Interaction Mechanism between SMSP and PVA
The schematic representation of the interaction mechanism between the SMSP and PVA is shown in Figure 2. Poly (vinyl alcohol) (PVA) is rich in hydroxyl (–OH) group functional entities, whereas SMSP contain several polar functional groups such as cellulose/hemicellulose-derived –OH groups and saponin units. In the PVA matrix with added SMSP particulates, there will be high intermolecular forces of attraction in the form of H bonding between the PVA’s –OH groups and the SMSP’s surface tethered –OH groups. Schematic representation of the interaction between SMSP and PVA.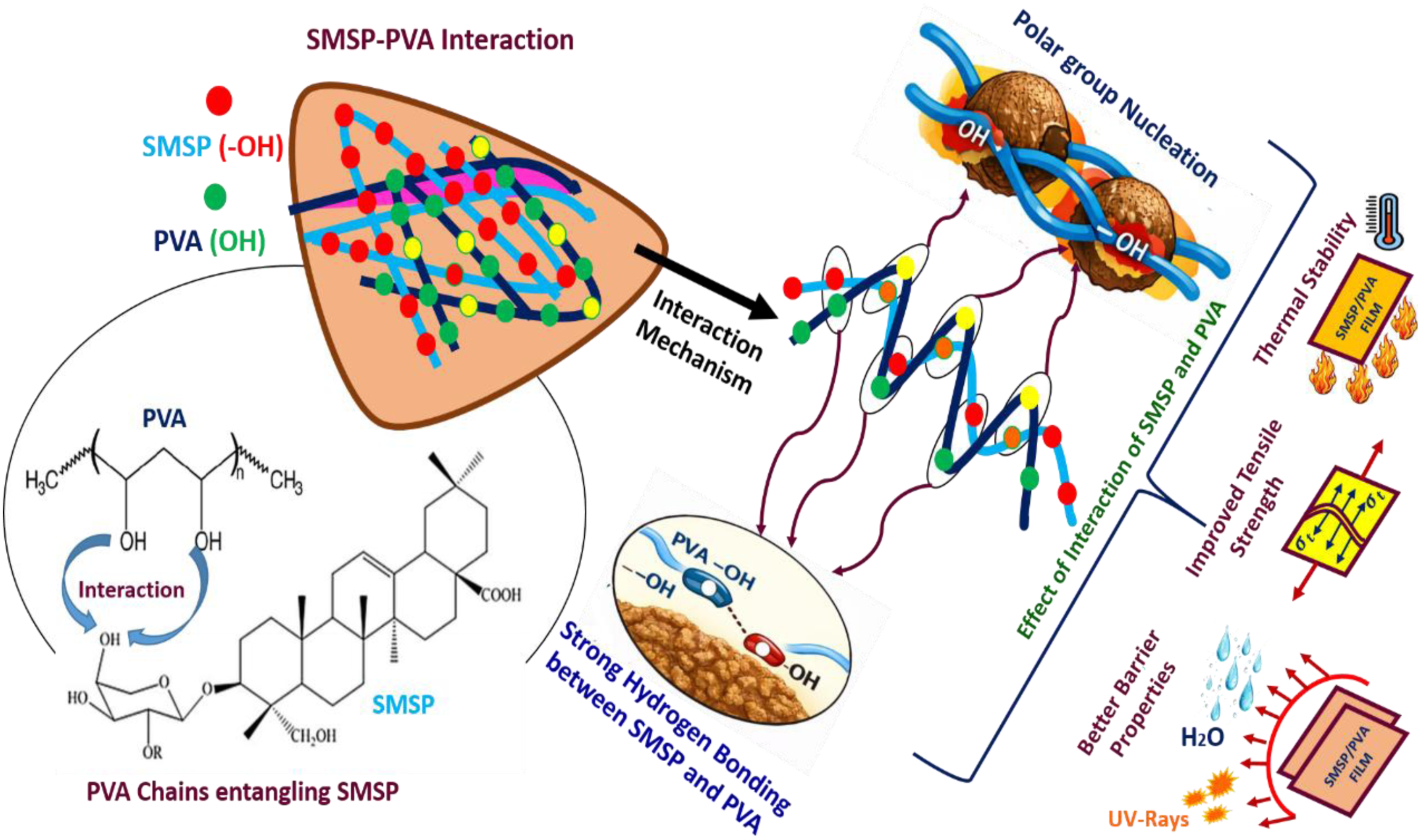
These hydrogen bonds ensure efficient interfacial bonding, leading to the physical entanglement of PVA chains on the surface of the SMSP particles, which confines the mobility of the PVA chains. Hence, the composite material shows higher crystallinity, along with improved stress transfer properties, possessing better mechanical and thermal properties than pure PVA, along with maintaining the bio-degradable and eco-friendly properties of the existing system.
Results and Discussions
FTIR
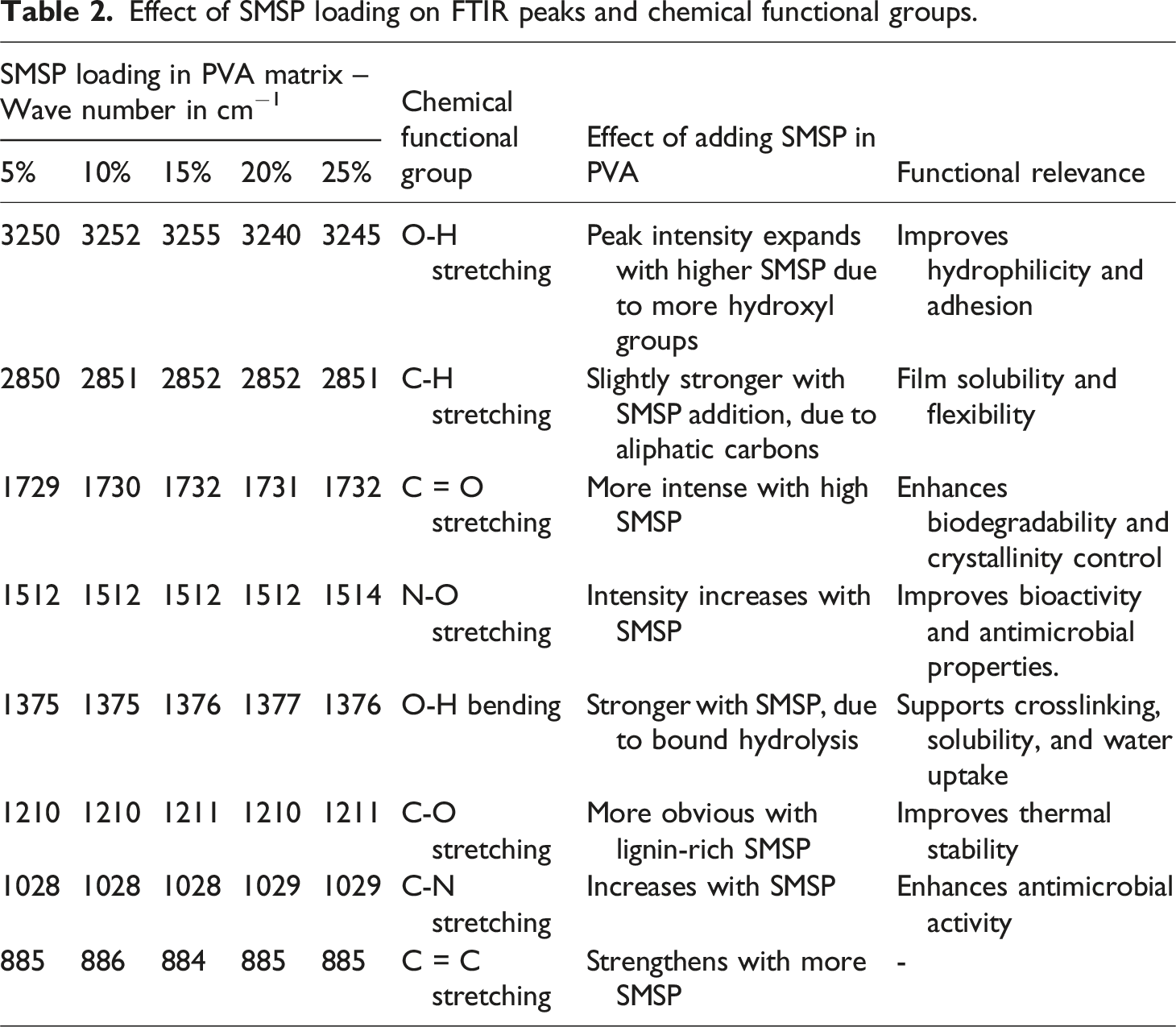
Figure 3 (a) exhibits typical FTIR peaks of polyvinyl alcohol (PVA) that reflect its primary functional groups and intermolecular interactions. The broad band at 3495 to 3192 cm−1 is due to -OH stretching arising from hydrogen bonding, accounting for the hydrophilicity and film-forming property of PVA. The peak at 2928 cm−1 is due to C-H stretching of methylene units in the polymer backbone.
34
The 1726 cm−1 band is caused by C = O stretching, associated with unreduced acetate groups resulting from incomplete hydrolysis, which impacts crystallinity and stability. The 1429 cm−1 peak corresponds to O-H bending, verifying the presence of bound hydroxyl groups. The 1234 cm−1 band is allied with C-O stretching, indicating alcohol functionalities that contribute to solubility. 841 cm−1 is related to C = C stretching from trace unsaturated groups, and the 594 cm−1 band to aromatic or skeletal vibrations associated with minor impurities. (a) FTIR Spectra of PVA and SMSP (b) FTIR Spectra of SMSP/PVA films.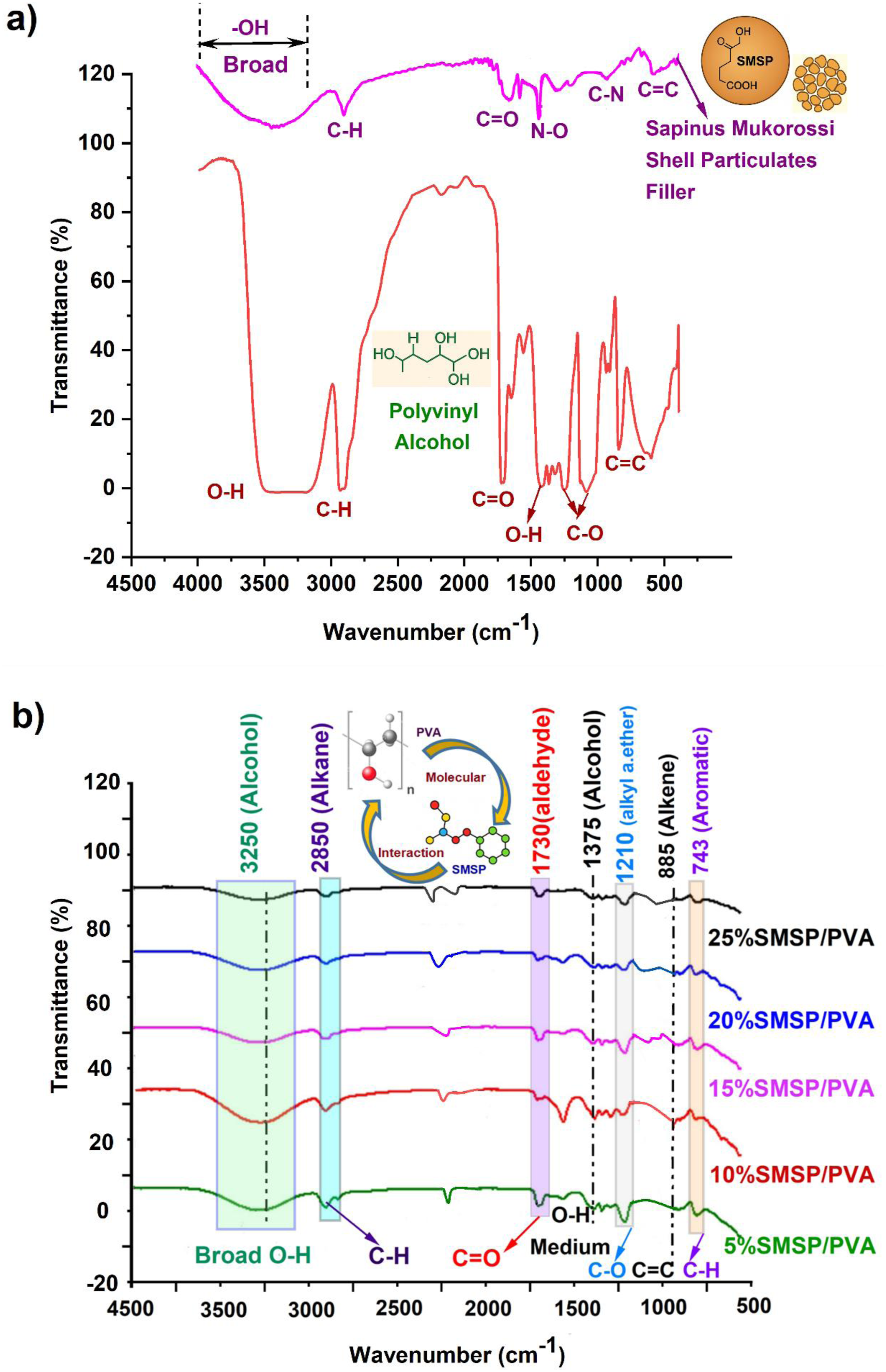
The FTIR spectrum of SMSP shows sharp peaks for its natural lignocellulosic and bioactive contents (Figure 3(a)). The broad band at 3545 cm−1 is due to O-H stretching vibrations, suggesting hydroxyl moieties from cellulose, hemicellulose, and phenolic groups. The peak at 2916 cm−1 is due to the C-H stretching of aliphatic groups, reflecting the long carbon chains in lignin and fatty constituents. The band at 1738 cm−1 is due to C = O stretching vibrations of ester or carboxyl groups in lignin, hemicellulose, and residual fatty acids. The absorption band at 1506 cm−1 is attributed to N-O stretching vibrations, commonly associated with nitro compounds or protein derivatives found in plant shells, indicating potential nitrogen-containing biomolecules. 35 The 1026 cm−1 band is due to C-N stretching, typical of alkaloids or amine-containing compounds. Lastly, the 663 cm−1 band is attributed to C-C skeletal vibrations, indicative of the aromatic skeleton of lignin and other carbons with a stable structure. Collectively, these peaks confirm the existence of hydroxyl, carbonyl, aromatic, and nitrogenous functional groups in SMSP, the presence of which can be the reason for its possible uses in biodegradable composites, adsorbents, and antimicrobial materials.
The FTIR spectra of PVA/SMSP biofilms shown in Figure 3 (b) disclose the existence of typical functional groups from both the PVA matrix and SMSP. The broad band at 3250 cm−1 represents –OH stretching vibrations of alcohol groups, reflecting intense hydrogen bonding due to hydroxyl-containing PVA and SMSP. The band at 2850 cm−1 is due to C-H stretching of alkanes, showing the aliphatic backbone of PVA and lignocellulosic SMSP. The band at 1730 cm−1 corresponds to C = O stretching of aldehydes or ester groups, primarily residual acetate in PVA and carbonyl compounds in SMSP.
Effect of SMSP loading on FTIR peaks and chemical functional groups.
X-ray Diffraction Analysis
The XRD spectrum of pure PVA, as displayed in Figure 4, shows a narrow crystalline peak at 2θ = 19.57° corresponding to the (101) reflection plane of its semi-crystalline structure based on inter- and intermolecular hydrogen bonding among hydroxyl groups that favour ordered chain packing. Its 32.5% crystallinity index is strong evidence that PVA is a partially crystalline polymer whose crystalline portions provide mechanical strength and thermal stability.
37
The crystallite size of 0.91 nm, calculated by Scherrer’s equation, provides evidence of nanoscale crystalline domains as a sign of limited chain folding characteristics of PVA. When compared to the cellulosic structures, the 19.57° diffraction angle in PVA is matched with the (101) physical structure, which reflects its semi-crystalline nature. The XRD pattern of SMSP demonstrates a sharp crystalline peak at 2θ = 20.07° that indicates the existence of semi-crystalline cellulose-based structures due to the ordered chain arrangement of polysaccharides in the biomass. The degree of crystallinity of 56.5% reflects a relatively high crystallinity, which implies that SMSP has well-ordered crystalline domains responsible for its rigidity, thermal stability, and structural integrity, while the amorphous fraction provides flexibility and chemical modification ability.
38
The crystallite size of 1.98 nm for SMPS indicates the nanoscale crystalline domains that increase surface reactivity and facilitate interfacial bonding. According to the diffraction angle, the peak at 20.07° indicates the cellulosic (002) physical structure typical of native cellulose I and represents the ordered stacking of cellulose chains, thus proving that SMSP maintains the crystalline cellulose structure. XRD plot of PVA, SMSP, and SMSP/PVA films.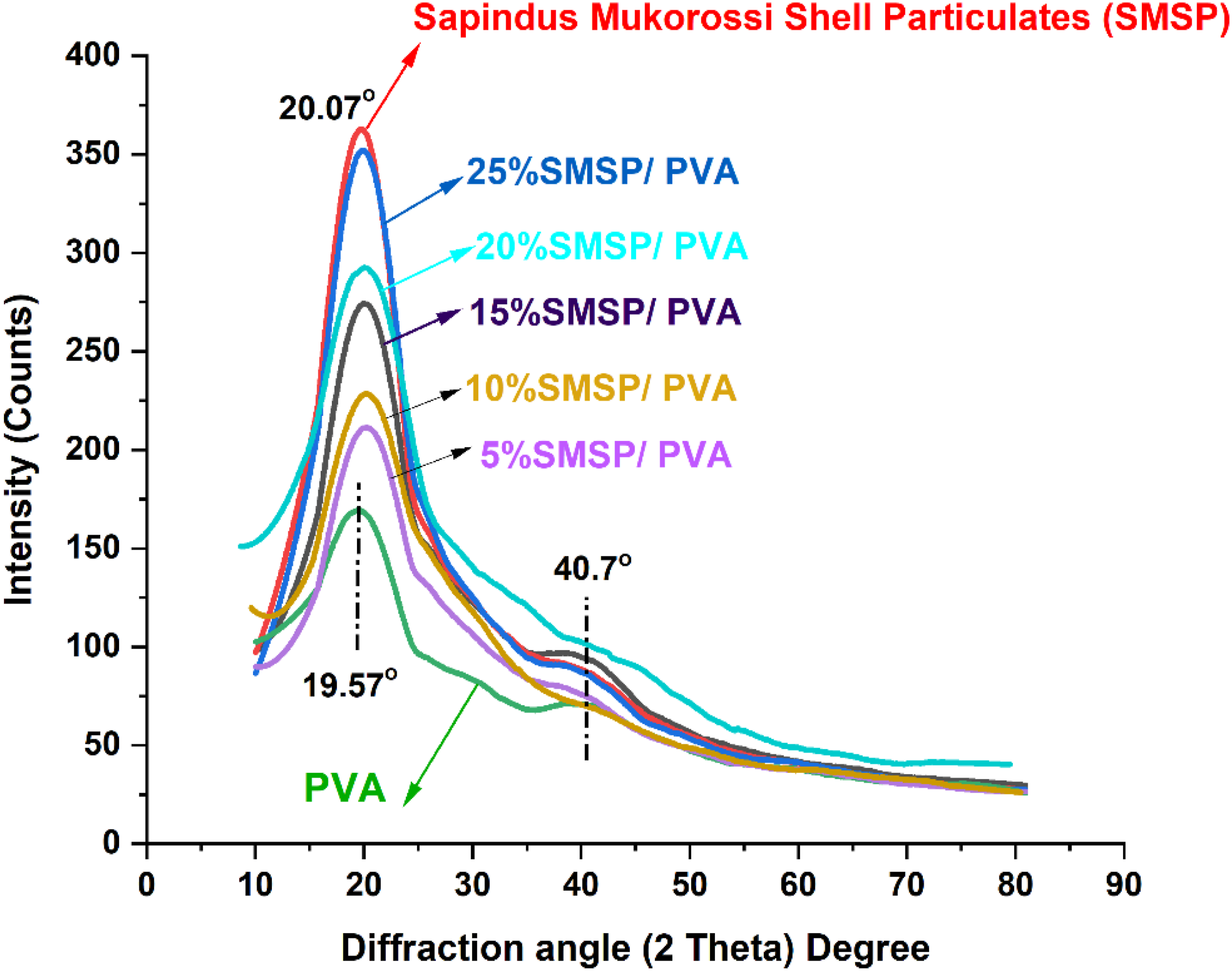
SMSP/PVA biofilms exhibit crystalline peaks at 2θ = 19.97°, 20.10°, 20.07°, 20.16°, and 20.19° for 5%, 10%, 15%, 20%, and 25% loadings of SMSP, respectively, indicating that addition of SMSP to the PVA matrix preserves the semi crystalline characteristic of the films with minor adjustments in peak positions due to polymer - filler interaction and restructuring of hydrogen bonding networks. Progressively increasing crystallinity index, like 42.5% (5% SMSP), 43.7% (10% SMSP), 46.8% (15% SMSP), 48.3% (20% SMSP), and 50.23% (25% SMSP), signifies that increased SMSP content develops crystalline ordering in the PVA matrix as nucleating sites and facilitates chain alignment and packing efficiency.
This crystallinity increase provides for enhanced thermal stability, barrier, and mechanical strength of the biofilms, while retaining some of the amorphous zones for flexibility and processability. 39 The diffraction angles of all combinations of SMSP/PVA films range from approximately 20° is typical of the cellulosic (002) physical structure, which confirms that the incorporated SMSP is enhancing the network structural integrity of PVA and functional performance with cellulose-derived crystalline domains.
TGA & DTG
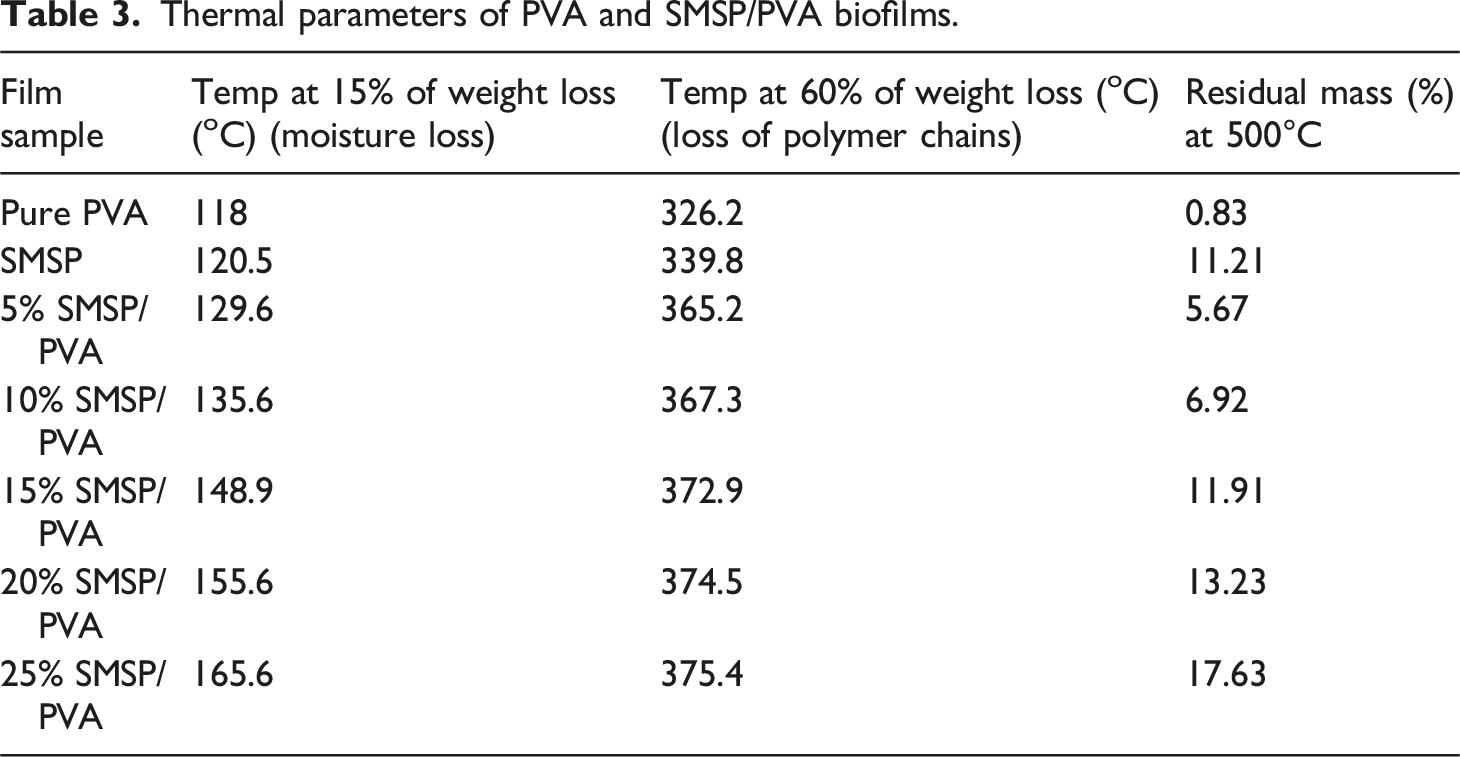
The thermogravimetric degradation behaviour of PVA, SMSP, and SMSP-reinforced PVA biofilms is shown in Figures 5(a) and 5(b). The initial weight loss at 70°C–190°C is attributed to the removal of physically absorbed water and volatile materials. As the content of SMSP increases, the onset temperature of the second stage slightly moves towards higher values from 15% weight loss at 118°C for neat PVA, 120.5°C for 5% SMSP/PVA, and 155.6°C for 25% SMSP/PVA films, which shows that SMSP limits the mobility of water by establishing strong hydrogen bonds with the hydroxyl groups of PVA. The second stage of thermal degradation occurs at 200°C - 360°C, which is due to the breakdown of polymeric chains like cellulose, hemicellulose, and PVA backbones. In this, the 60% weight loss takes place mostly, and degradation temperatures rise with loading of SMSP (339.8°C for 5% SMSP/PVA and 374.5°C for 25% SMSP/PVA), showing that SMSP provides greater thermal resistance due to its lignocellulosic composition and robust interfacial interactions with the PVA matrix.
40
The third phase, 365°C - 500°C, relates to the degradation of lignin and other impurities, which decompose in a wide temperature range because of their intricate aromatic rings. The low char residue of pure PVA (0.83 % at 500°C) indicates that it decomposes almost completely into volatile products on heating, reflecting poor thermal stability. In contrast, SMSP show a much higher char residue of 11.21 %, which is attributed to their lignin, cellulose, and mineral content that promote the formation of a carbon-rich protective layer during pyrolysis. This difference highlights the inherent thermal resistance of SMSP when incorporated into PVA. Notably, char residue is considerably raised with the increase in SMSP content from 0.83% for pure PVA to 17.63% for 25% SMSP/PVA films, indicating a high carbonaceous yield of SMSP that supports char formation and hence thermal stability. Differential thermogravimetric (DTG) analysis (Figure 5(b)) also supports this trend, with Tmax changing from 257.5°C (PVA) to 325.5°C (25% SMSP/PVA), indicating that films become increasingly thermally stable with increasing SMSP content. These thermal enhancements are due to hydrogen bonding, interfacial adhesion, and barrier effect of SMSP, which limit polymer chain mobility, postpone thermal scission, and enhance char formation.
41
Finally, the thermal analysis results point out that SMSP not only mechanically strengthens PVA but also serves as a natural thermal stabilizer, such that SMSP/PVA biofilms are potential candidates for biodegradable packaging. The thermal parameters of PVA and SMSP/PVA films are listed in Table 3. (a) TGA, (b) DTG curves of PVA, SMSP, and SMSP/PVA biofilms. Thermal parameters of PVA and SMSP/PVA biofilms.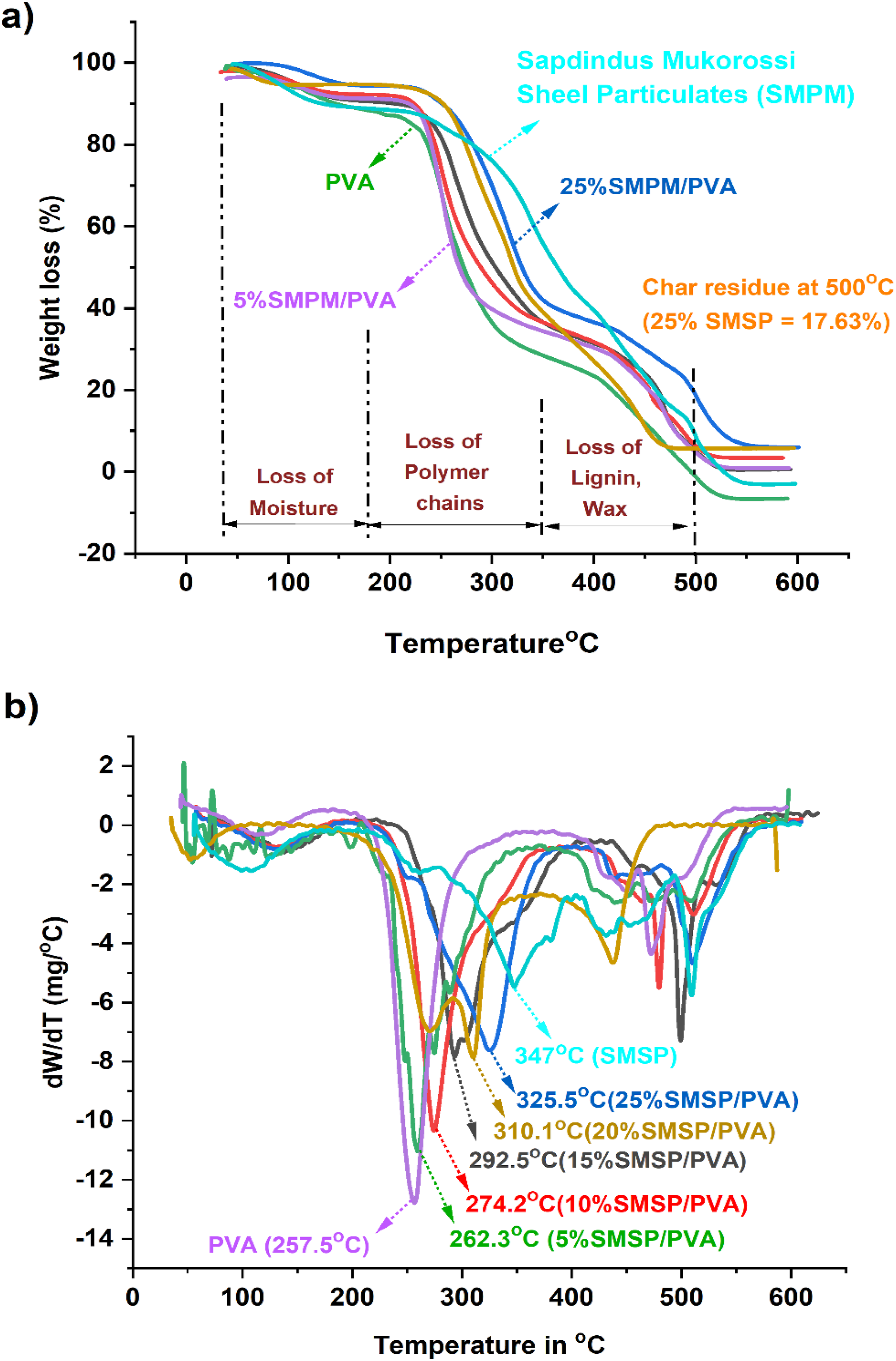
Tensile Test
The tensile testing reveals that SMSP reinforcement of polyvinyl alcohol (PVA) significantly improves the mechanical properties of the biofilms to an optimal filler content, it is depicted in Figure 6 (a). Untreated PVA film has a tensile strength of 23.1 MPa, whereas SMSP/PVA biofilms register stepwise improvements to 28.5 MPa (5% SMSP), 36.8 MPa (10 %), 43.9 MPa (15%), and up to a maximum of 48.4 MPa at 20% loading with SMSP. This consistent improvement is likely due to the efficient transfer of stress from the ductile PVA matrix to rigid, lignocellulosic SMSP particles that serve as natural reinforcements.
42
The hydroxyl-functionalized surface of SMSP favours effective hydrogen bonding and interfacial adhesion to the PVA chains, limiting polymer chain mobility and enhancing stiffness and load-bearing strength. However, beyond a 20% loading, the tensile strength reduces to 34.6 MPa at 25 % SMSP. At increased filler content, particle agglomeration and poor dispersion form micro voids and stress-concentration sites that compromise the composite.
43
(a) Variations of Tensile stress, Young’s Modulus, and % of elongation, (b) Stress Vs % elongation curve for pure PVA and SMSP/PVA biofilms.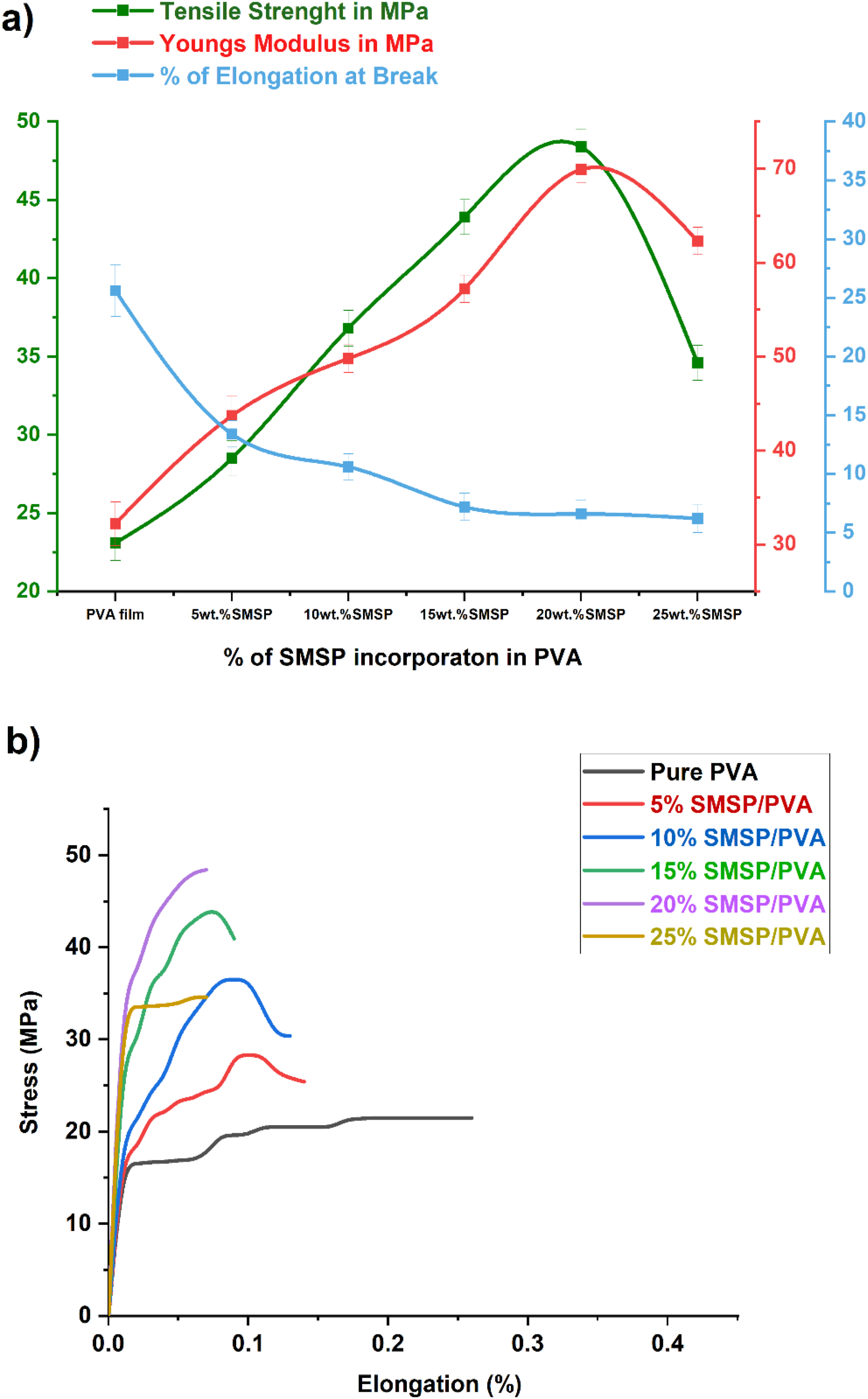
The variations of Young’s modulus and % elongation of PVA and SMSP/PVA biofilms are also presented in Figure 6(a). The Young’s modulus of pure PVA is 32.2 MPa, which increases consistently to 43.7 MPa (5%), 49.8 MPa (10%), 57.2 MPa (15%), and reaches a maximum of 69.9 MPa for 20% SMSP, after which it decreases slightly to 62.3 MPa at 25% loading. This trend shows that the rigid, lignocellulosic SMSP particles strongly limit the molecular mobility of PVA chains and facilitate effective stress transfer, leading to increased stiffness and elastic modulus. SMSP’s high surface hydroxyl content facilitates hydrogen bonding with PVA, leading to strong interfacial adhesion and enhanced load-carrying capability up to an optimum 20% filler loading. Conversely, the percentage elongation at break declines significantly from 34.5% for neat PVA to 13.4%, 10.6%, 7.2%, 6.6%, and 6.2% for 5–25% SMSP, indicative of loss in ductility. The filler particles of hard nature restrict chain mobility and are physical barriers to deformation, resulting in a harder structure. 44 This is characteristic of particulate filler reinforced polymers, where enhanced stiffness is brought about by increasing filler content at the expense of toughness and elongation. The 20%SMSP/PVA biofilms show optimal stiffness as required in structural or packaging applications.
The Stress strain curves for SMSP/PVA biofilms is shown in Figure 6(b). Pure PVA exhibits a linear proportional stress-strain curve; the stress values become constant after threshold strain values. The 5% and 10% SMSP-loaded films showed the same stress-strain behaviour, and the elongation of the films is restricted due to filler content. Further, the stress-strain curves of highly SMSP loaded films show reduced elongation and the stiffness of the films is increased gradually with the filler content up to 25%. This trend indicates that the rigid, lignocellulosic SMSP particles effectively restrict the molecular mobility of the PVA chains, enhancing stiffness and the elongation of 25% SMSP/PVA films is reduced by 82% to pure PVA films. 45 This phenomenon is distinctive for bio waste fillers reinforced polymers, the stress-strain curves indicate that the material underwent a ductile to brittle transition stage corresponding to SMSP loading. 43
Surface Morphology
Tensile‐fractured SEM micrograph of neat PVA and SMSP/PVA biofilms clearly demonstrates (Figure 7 (a)-(f)) the systematic microstructural evolution caused by increasing SMSP loading, in turn controlling the mechanical behavior of the composites. A smooth and featureless fracture surface typical of a homogeneous polymer network with limited energy dissipation is exhibited by pure PVA, reflecting a largely brittle failure it as shown in Figure 7 (a). Figure 7 (b)-(d) represents the incorporation of 5–15 wt% SMSP results in brittle surfaces with complete dispersion of fillers into continuous PVA layers without any observed agglomeration, reflecting strong hydrogen bonding between dominant hydroxyl groups in PVA and functional polar groups in SMSP. The strong interfacial bonding facilitates the stress transfer from matrix to filler effectively, which contributes to enhanced tensile strength while maintaining a ductile fracture mode. At 20 wt% loading (Figure 7 (e)), the occurrence of small voids and incipient clusters shows that the matrix is at its saturation point for uniform wetting of particles; debonding or incomplete impregnation leads to stress concentrations in localized areas, producing initiation of micro-cracks, responsible for the beginning of loss of strength. At 25 wt% of SMSP content (Figure 7(f)), there are well-defined filler agglomerates and pull-out cavities that occlude the fracture surface with a clear indication of poor interfacial compatibility brought about by excessive particulate crowding and restricted polymer chain mobility near fillers. These agglomerations decrease the effective surface area for hydrogen bonding and produce weak interfaces susceptible to easy crack propagation, causing embrittlement and reduced tensile properties.
46
Such observations are consistent with composite theory: a best filler fraction strengthens the matrix by interfacial bonding and crack-deflection mechanisms, while overloading exceeds the percolation threshold, breaks polymer continuity, and creates stress raisers that weaken mechanical integrity. SEM micrographs of (a) Pure PVA, (b) 5%SMSP/PVA, (c). 10% SMSP/PVA, (d). 15%SMSP/PVA, (e). 20% SMSP/PVA, (f). 25% SMSP/PVA biofilms.
Puncture Test
Figure 8 represents the result of the puncture test, which clearly demonstrates that SMSP strengthen the PVA biofilms. Pure PVA film exhibits a puncture load of 13.6 N and a puncture strength of 28.9 N/mm with a puncture deformation of 14.34%. When SMSP is incorporated at 5%, 10%, 15%, 20%, and 25% in the PVA matrix, the puncture load gradually increases to 14.6 N, 14.9 N, 15.2 N, 15.9 N, and 16.2 N, respectively. The puncture strength rises sharply to 39.4 N/mm at 5% SMSP, 43.5 N/mm at 10%, 48.5 N/mm at 15%, and shows a maximum value of 54.5 N/mm at 20% SMSP. Puncture force, Puncture strength, and Puncture deformation of pure PVA and SMSP/PVA biofilms.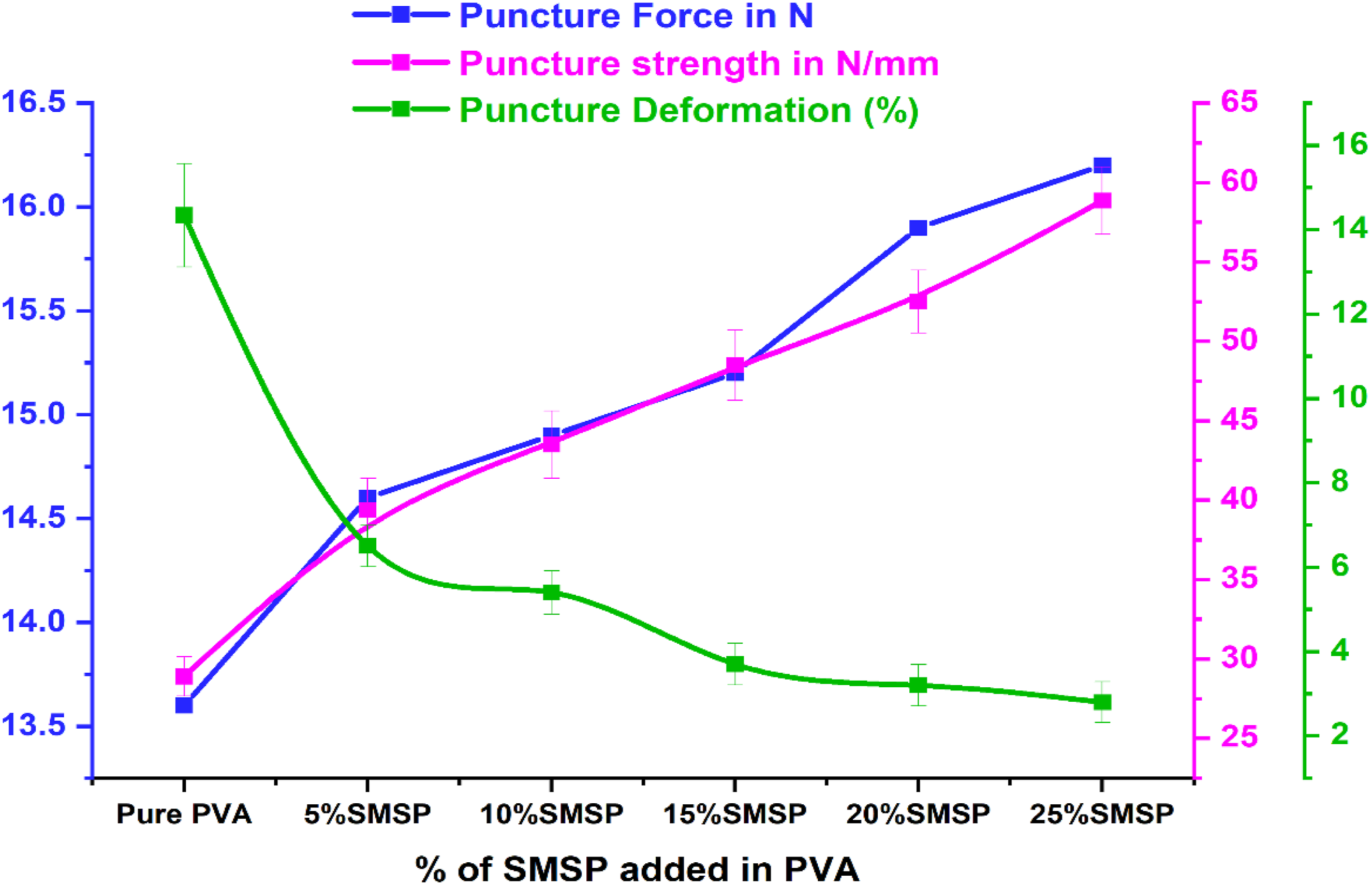
At the highest loading (25%) of SMSP in the PVA matrix, the puncture strength was decreased by 3.04% (52.85 N/mm). The progressive rise in puncture load and strength up to 20 % is mainly attributed to the rigid, well-dispersed SMSP particles, which provide effective stress transfer, restrict polymer chain mobility, and hinder crack propagation. 47 The slight declination in puncture strength at 25% likely results from particle agglomeration or weak interfacial bonding at higher filler content, which can create micro-defects. Conversely, the punching deformation gradually decreases from 6.5% at 5% SMSP to 2.8% at 25% of SMSP, indicating increased stiffness and reduced ductility. The consistent decrease in puncture deformation reflects the reduced flexibility of the matrix as SMSP occupies free volume, thereby limiting molecular rearrangements. 48 Overall, the addition of SMSP enhances puncture resistance by increasing the rigidity of the SMSP/PVA biofilms and load-bearing capacity. The gradual increase in puncture load with filler content highlights the reinforcing capability of SMSP within the PVA matrix.
UV-Barrier Property
The UV-transmittance of Pure PVA and SMSP/PVA biofilms is indicated in Figure 9 (a). The UV transmittance of the SMSP/PVA biofilms is clearly decreased by increased SMSP loading compared with pure PVA. Untreated PVA allows through approximately 87 % of incident UV radiation, while for the 5%, 10%, 15%, 20%, and 25% SMSP/PVA films at 410 nm transmission figures are only 78%, 75 %, 69%, 62%, and 54%, respectively corresponding to UV-transmittance decreases of approximately 10 %, 14 %, 21 %, 29 %, and 38 % compared with pure PVA. The same trend is noticed at 450 nm, where the same films at 70 %, 64 %, 58 %, 53 %, and 48 % transmittance in pure PVA result in 19 %, 26 %, 33 %, 39 %, and 45 % lower transmittance. The 410 nm and 450 nm wavelengths are in the transition zone between the near-UV and visible region, where violet-blue high-energy light is responsible for polymer photo degradation, colour, and loss of mechanical stability.
49
The declining transmittance indicates lignocellulosic SMSP absorbs the UV light via cellulose and phenolics, whereas its particles enhance the scattering of light in the film. (a) UV-Transmittance (b) Transparency and Opacity of Pure PVA and SMSP/PVA biofilms.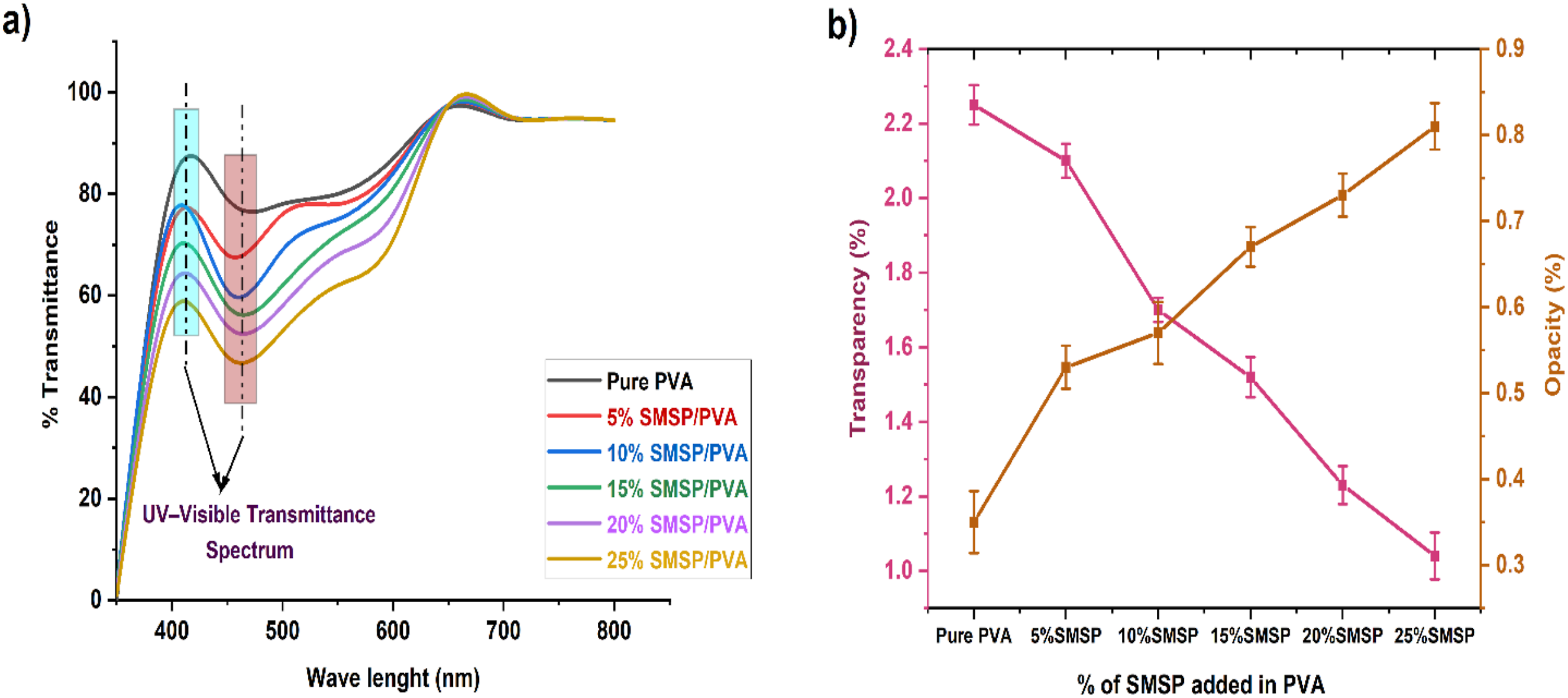
The incorporation of SMSP into the PVA matrix markedly alters the optical properties of the biofilms. Figure 9 (b) shows that the pure PVA film exhibits a high transparency of 2.25 % and a low opacity of 0.35%, reflecting its relatively smooth, homogeneous polymer network that allows light to pass through with minimal scattering. As SMSP is added at 5–25 wt% %, transparency drops dramatically to 2.1%, 1.7%, 1.52%, 1.23%, and 1.04%, signifying that SMSP particles efficiently scatter and block light from coming in. Compared to pure PVA, these figures reflect reductions in transparency of about 7%, 24%, 32%, 45%, and 54%, respectively, which show an increasing blocking light effect with increasing filler content. Simultaneously, the clarity varied from 0.35% for neat PVA to 0.53%, 0.57%, 0.67 %, 0.73%, and 0.81% for 5–25% SMSP, representing approximate gains in clarity of 51%, 63%, 91%, 109%, and 131%. This trend can be attributed to the presence of SMSP’s lignocellulosic particles, which create heterogeneous interfaces and micro-voids that scatter and absorb light, thereby enhancing UV shielding and reducing visual clarity. 50 Incorporating SMSP reduces transparency and increases opacity, converting PVA into a UV-blocking biofilm ideal for packaging or coatings that protect against near-UV and visible photo-oxidative damage.
Water Absorption (%)
The % water absorption of SMSP/PVA biofilms and Pure PVA is illustrated in Figure 10. The Figure illustrates a systematic and pronounced decrease in the absorption of moisture when SMSP are added to the PVA matrix. Pure PVA absorbs 46.7% after 10 hours of soaking, whereas 5% - 25% SMSP-loaded biofilms absorb 40.6%, 37.4%, 35.4%, 33.5% and 31.1% of water, respectively, which shows that the increase in SMSP content progressively enhances the water resistance of films. Systematically, this phenomenon can be understood from FTIR and XRD analysis of SMSP/PVA biofilms. FTIR spectra reveal a high cellulose composition of the SMSP and, most importantly, intensive -OH interactions between PVA and SMSP, those interfacial hydrogen bonds that efficiently use free PVA hydroxyls that otherwise would be coordinating water, transforming available hydrophilic sites into PVA/SMSP bonds and thus reducing sorption capacity. Concurrently, SMSP particles serve as physical barriers that enhance the tortuosity of the diffusion paths and can facilitate enhanced packing of chains at the interface.
51
Plots of XRD indicate that film crystallinity increases with increased SMSP loading, which further minimizes water uptake since crystalline parts are densely packed and impermeable compared to amorphous regions, which are the main sites for water sorption and transport. Together, while cellulose in SMSP is inherently hydrophilic, its hydroxyl groups seem to be largely involved in PVA filler hydrogen bonding (FTIR) and in the creation of nucleation sites that enhance overall crystallinity (XRD); therefore, the net result is reduced free volume and reduced accessible hydrophilic sites, reduced diffusion, and lower equilibrium water uptake as SMSP content rises This dual chemical (hydrogen bonding) and structural (enhanced crystallinity and tortuous channels) explanation is responsible for the progressively enhanced moisture resistance of SMSP/PVA films to 25 wt%. % of Water Absorption for pure PVA and SMSP/PVA films.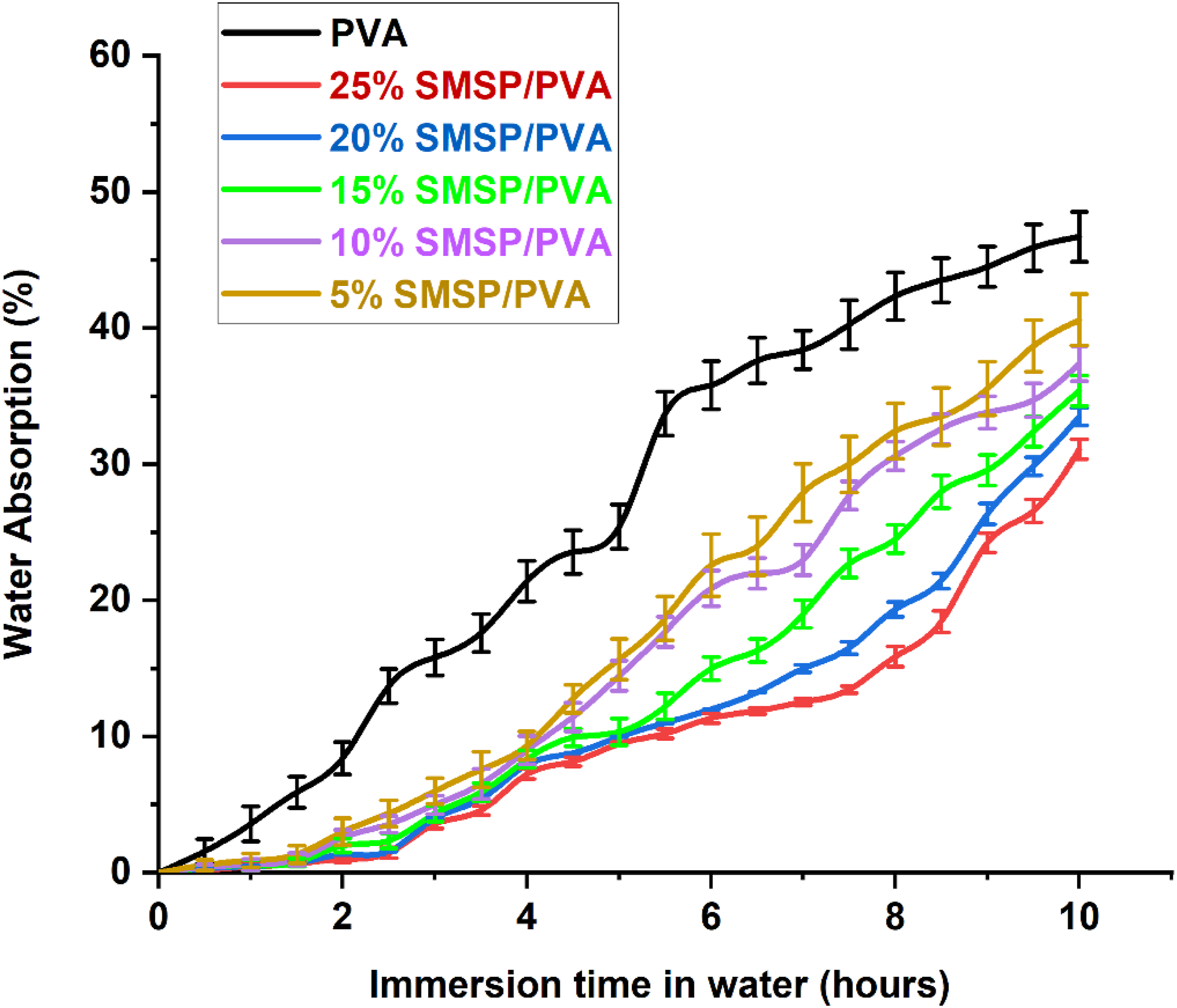
Film Solubility in Water and WVP
The % solubility of pure PVA and SMSP/PVA biofilms is displayed in Figure 11. The addition of SMSP to PVA greatly lowers the water solubility of the films as compared to neat PVA (57.5%). The % Solubility of SMSP/PVA films in water decreased by 9.73%, 15.13%, 19.6 %, 20.1% and 22.9% to pure PVA films as the SMSP fraction is raised from 5% to 25%. The hydrophobic and lignocellulosic nature of SMSP, which limits the penetration and diffusion of water molecules inside the polymer matrix, is proven by this decrease in film solubility in water. The water absorption shows the same pattern, decreasing as the filler concentration increases. The diminished absorption reveals that SMSP not only fills the free volume inside the PVA network but also improves hydrogen bonding and interfacial interactions so as to reduce the number of accessible hydroxyl sites for water binding. % Solubility in water and Water vapour permeability of Pure PVA and SMSP/PVA biofilms.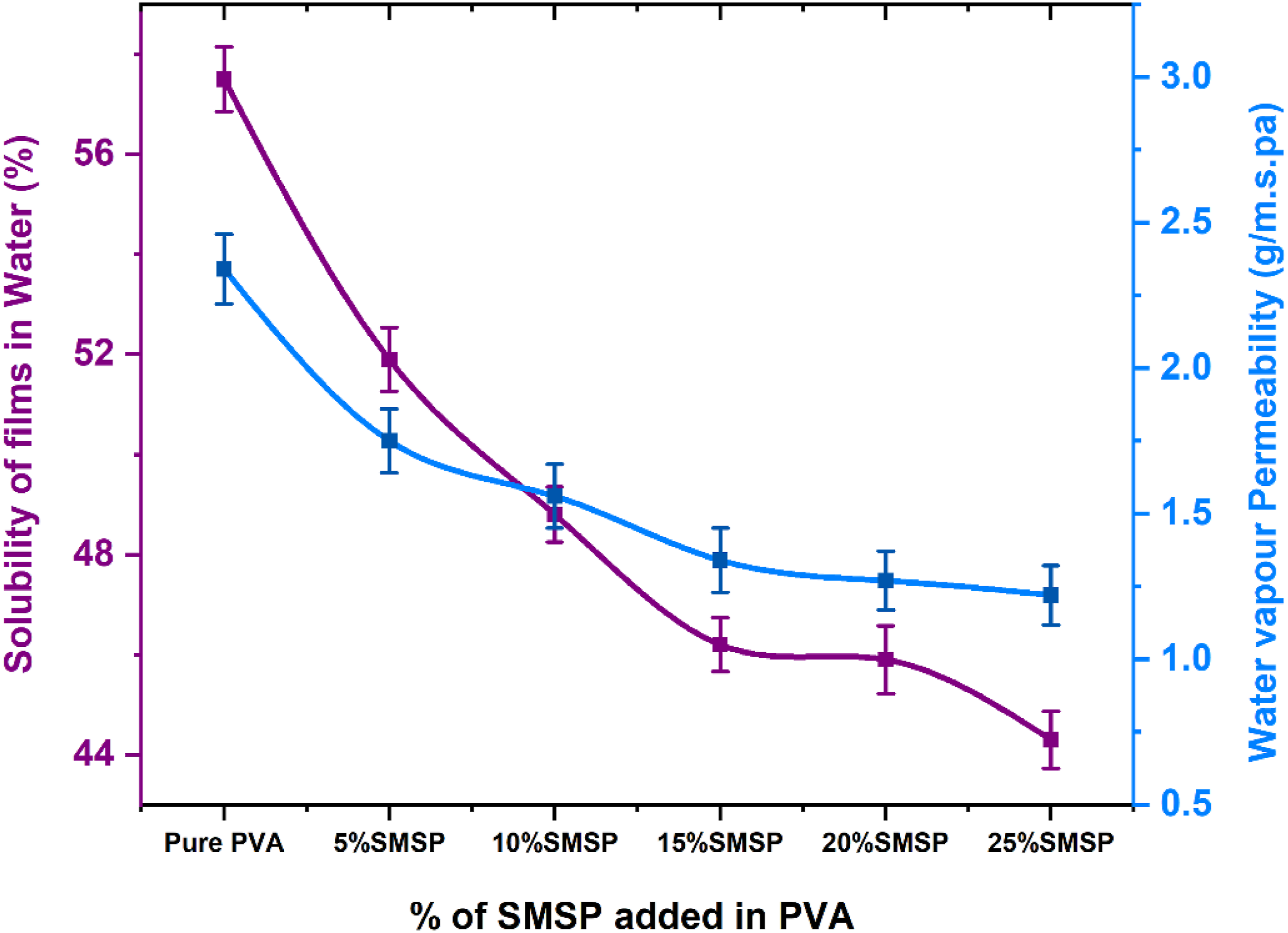
As a result, the simultaneous decline in water absorption and water solubility further ascertains that SMSP is an effective water uptake and dissolution barrier, enhancing the water resistance of the composite films.
The water vapour permeability of SMSP/PVA biofilms decreases progressively with rising concentrations of SMSP in the PVA matrix, as illustrated in Figure 11 The WVP of a pure PVA is 2.34 g/m.s. Pa, whereas the WVP values for films with 5, 10, 15, 20, and 25% of SMSP are 1.75, 1.56, 1.34, 1.27, and 1.22 g/m.s. Pa, with decreases of about 25.2, 33.3, 42.7, 45.7, and 47.9% relative to neat PVA. Concurrently, the thickness of the films rises from that of pure PVA at 0.254 mm to that of 25% SMSP/PVA films at 0.334 mm, a 31.5% increase in thickness. If the decrease in permeability were caused entirely by thickness, with WVP being inversely related to thickness, a change from 0.254 mm to 0.334 mm would lead to a WVP reduction of approximately (0.334 - 0.254)/0.334 = 23.95%, or approximately 24%. Since the real WVP reduction with the incorporation of 25% SMSP is approximately 47.9%, half of the improvement can be related to the thickness increase, and the rest is due to microstructural and chemical consequences induced by the SMSP. The behavior is consistent with the well-dispersed SMSP particles related to hindering the water flow in the PVA matrix. These straight particles increase the path of water diffusion, decrease open hydrophilic channels, and create hydrogen bonds with PVA, restricting chain mobility and decreasing free volume for water transport. 52 Cellulose and lignin in SMSP are less polar compared to PVA, decreasing water sorption further. The fact that no increase in WVP is found at high filler loadings indicates negligible particle agglomeration or interfacial void creation. Combined, the two effects of greater thickness and the barrier property of the filler result in almost a doubling of moisture resistance, reducing WVP from 2.34 to 1.22 g/m.s. Pa.
The SMSP/PVA films having the lowest WVP (20,25 % SMSP, 1.27 &1.22 g/m.s. Pa) are best suited for packaging requiring high moisture protection like dry food, powdered food, and single-use bags. Intermediate levels of SMSP (10 &15 %, WVP 1.56 &1.34 g/m.s. Pa) provide an optimum blend of barrier and breathability for use in bakery products, snack foods, and semi-fresh vegetables where restricted moisture exchange prevents condensation. Pure PVA, with its 2.34 g/m s.Pa higher WVP and thinner films are preferable to applications requiring airflow, i.e., wound dressings, breathable coatings, compostable films. The addition of biomass-derived SMSP filler enhances the moisture barrier of PVA films and maintains it eco-friendly, making the films ideal for sustainable packaging requiring moderate to high moisture resistance.
Surface Roughness Parameters
The surface roughness parameters of Pure PVA and SMSP/PVA biofilms are shown in Figure 12. The increase in maximum peak height (from 132 nm to 245 nm) and in peak to valley (Rpv, from 205 nm to 327 nm) indicates that the surface forms deeper troughs and taller peaks, and thus the topography becomes more heterogeneous and has more potential for mechanical interlocking and scattering of light. The Root-Mean-Square roughness (Rq) increases from 27 nm to 76 nm, along with the increase of mean roughness (Ra) from 19 nm to 54 nm and Rz (sampled mean peak-to-valley) from 127 nm to 265 nm, which quantitatively validates that the overall amplitude of SMSP/PVA film micro-scale roughness increases significantly with increased filler content. Skewness (Rsk) is most enlightening regarding the asymmetry of the height distribution: a negative Rsk indicates dominance by valleys, while a positive Rsk indicates dominance by sharp peaks. The Rsk is positive, the surface is dominated by sharp peaks, which enhance localized stress concentration and abrasion susceptibility.
53
Rsk is positive (peak-dominated), the surface is covered with sharper peaks and fewer deep valleys. This decreases the true contact area for water and decreases the ability to hold surface water pooling, so the film will take up less water through surface retention.
54
However, the polymer itself continues to be able to sorb moisture. The kurtosis (Rku) rises from 3.18 nm to 8.57 nm, indicating a transition from a nearly-Gaussian height distribution to one of more extreme asperities (leptokurtic), i.e., greater outlier peaks or very deep pits, so contact mechanics will be dominated by fewer high asperities as opposed to a wide population of moderate ones. Collectively, these alterations suggest that increasing SMSP content creates a more rugged, topographically inhomogeneous surface with higher peak/trough amplitudes, which will affect mechanical interfacial behavior, liquid interactions (wetting, barrier function, swelling sites), optical properties (increased scattering and diffuse reflectance), and most probably localized biodegradation or microbial adhesion patterns. For SMSP loaded PVA in particular, SMSP is a plant based natural filler with many hydroxyl groups. The sites are inherently hydrophilic; therefore, the filler will not release or replenish water to the environment; it will mostly absorb and retain water in the composite.
55
At increased loadings of SMSP, the PVA matrix tends to be less continuous and more tortuous and may reduce the total bulk water uptake compared with pure PVA (as your previous measurements demonstrated), but still controls how rapidly water first wets and penetrates. Surface Roughness Parameters of Pure PVA and SMSP/PVA biofilms.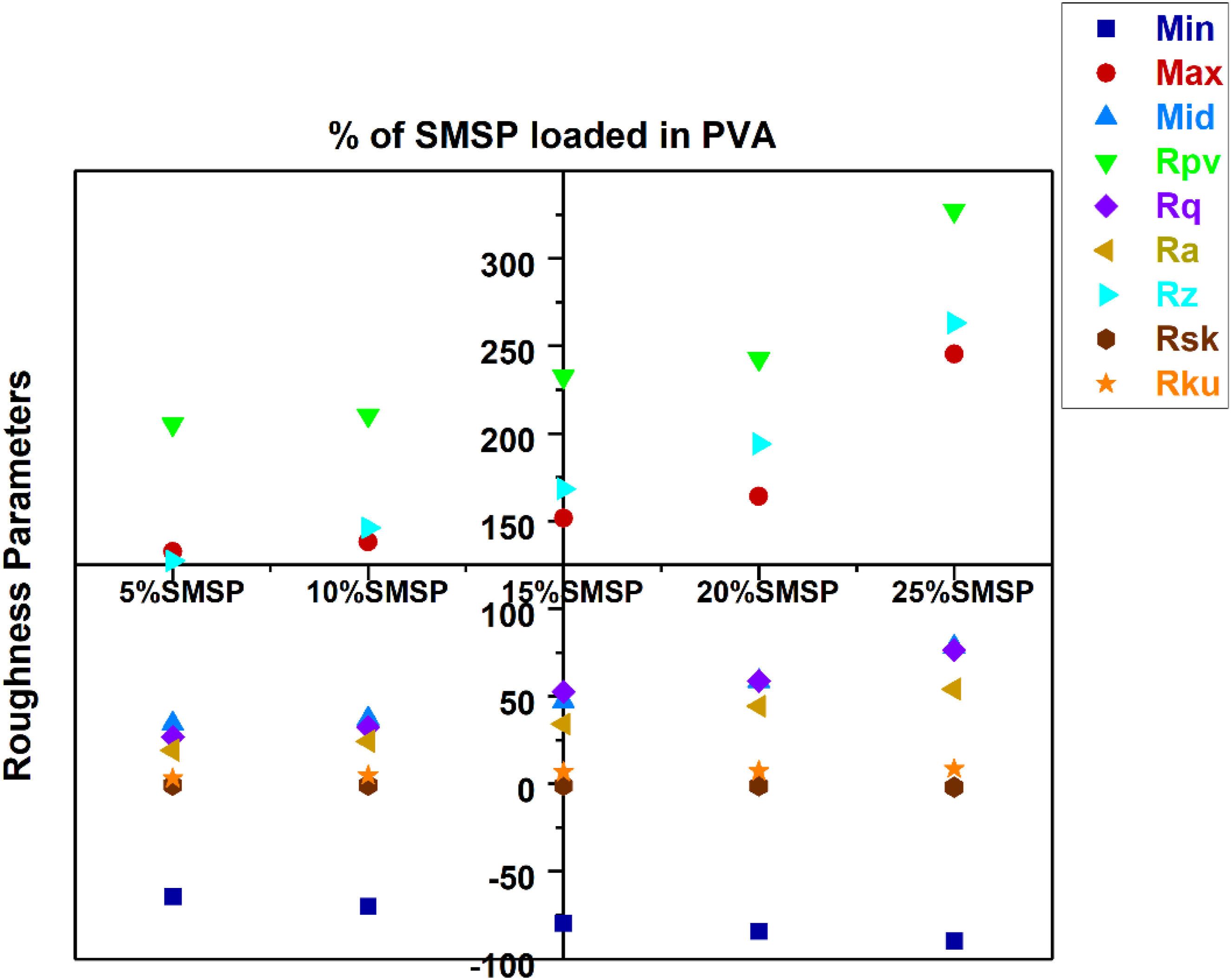
Soil Burial Test
Figure 13 represented the pure PVA films degraded at a faster rate, with 48.5% weight loss after 60 days, while SMSP loaded PVA films degraded at slower rates, decreasing consistently from 46.2% for 5% loading of SMSP up to 27.6% for 25% loading. This lesser biodegradation coincides with the lower water absorption, water solubility, and water vapour permeability (WVP) of the films with increasing content of SMSP. Given that soil biodegradation is mostly catalysed by microbial attack and hydrolytic degradation, decreased hydrophilicity and negligible water imbibition in the SMSP/PVA films hinder the diffusion of water and microbial enzymes into the polymer matrix, thus retarding chain scission and microbial assimilation. Mechanistically, PVA film degradation proceeds through hydrolysis of the polymer backbone, microbial enzymatic cleavage, and ultimate mineralization into CO2 and H2O.
56
In the SMSP/PVA films, the presence of lignocellulosic SMSP functions as a reinforcing filler that lowers the mobility of the polymer chains and forms a denser structure, thereby inhibiting water ingress and microbial action. As a result, the addition of SMSP not only enhances the barrier property of the films but also increases resistance against quick biodegradation, which makes the films more sustainable in soil environments while maintaining eco-friendly degradability with long-term passage of time. % of weight loss of pure PVA and SMSP/PVA films in Soil.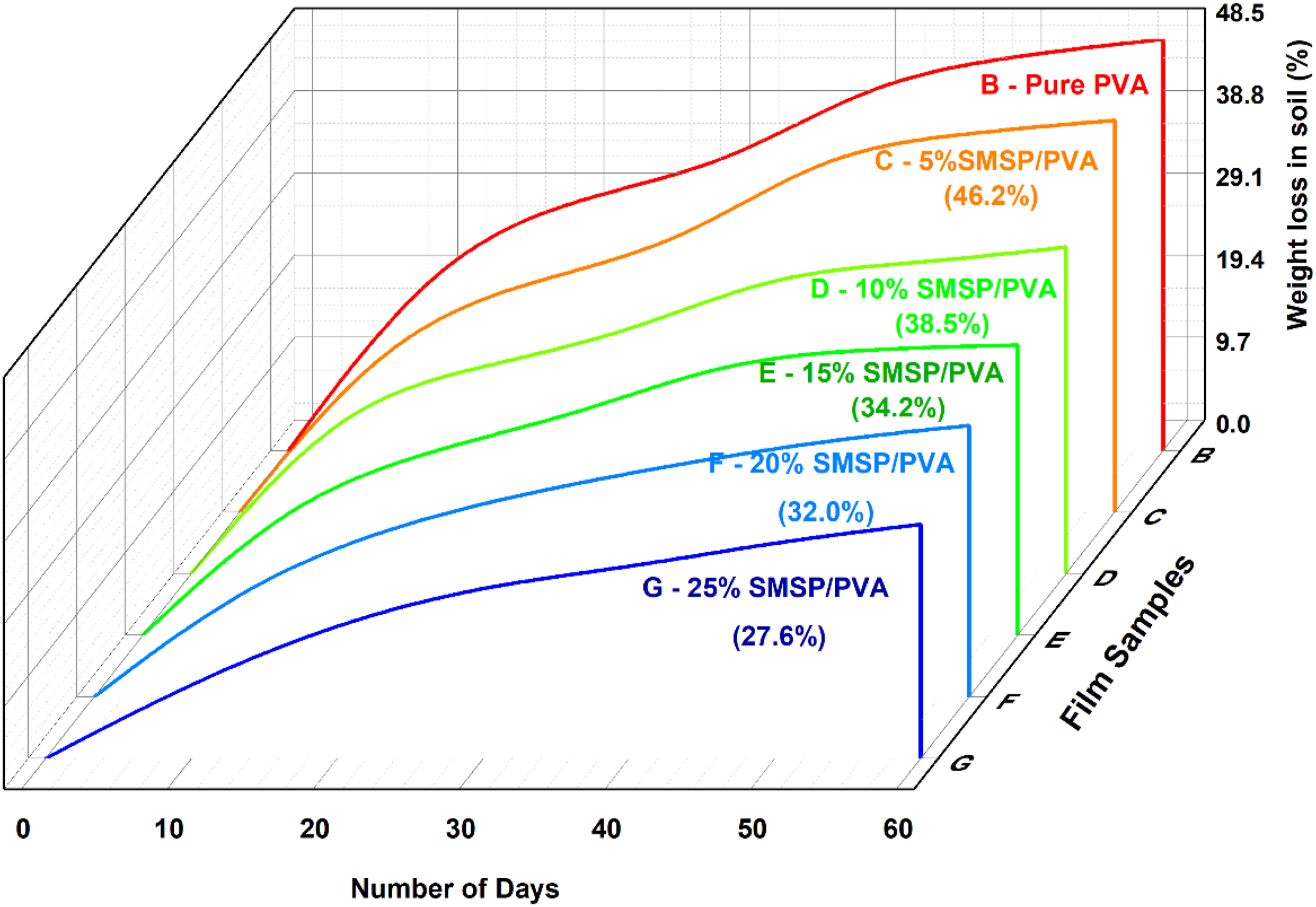
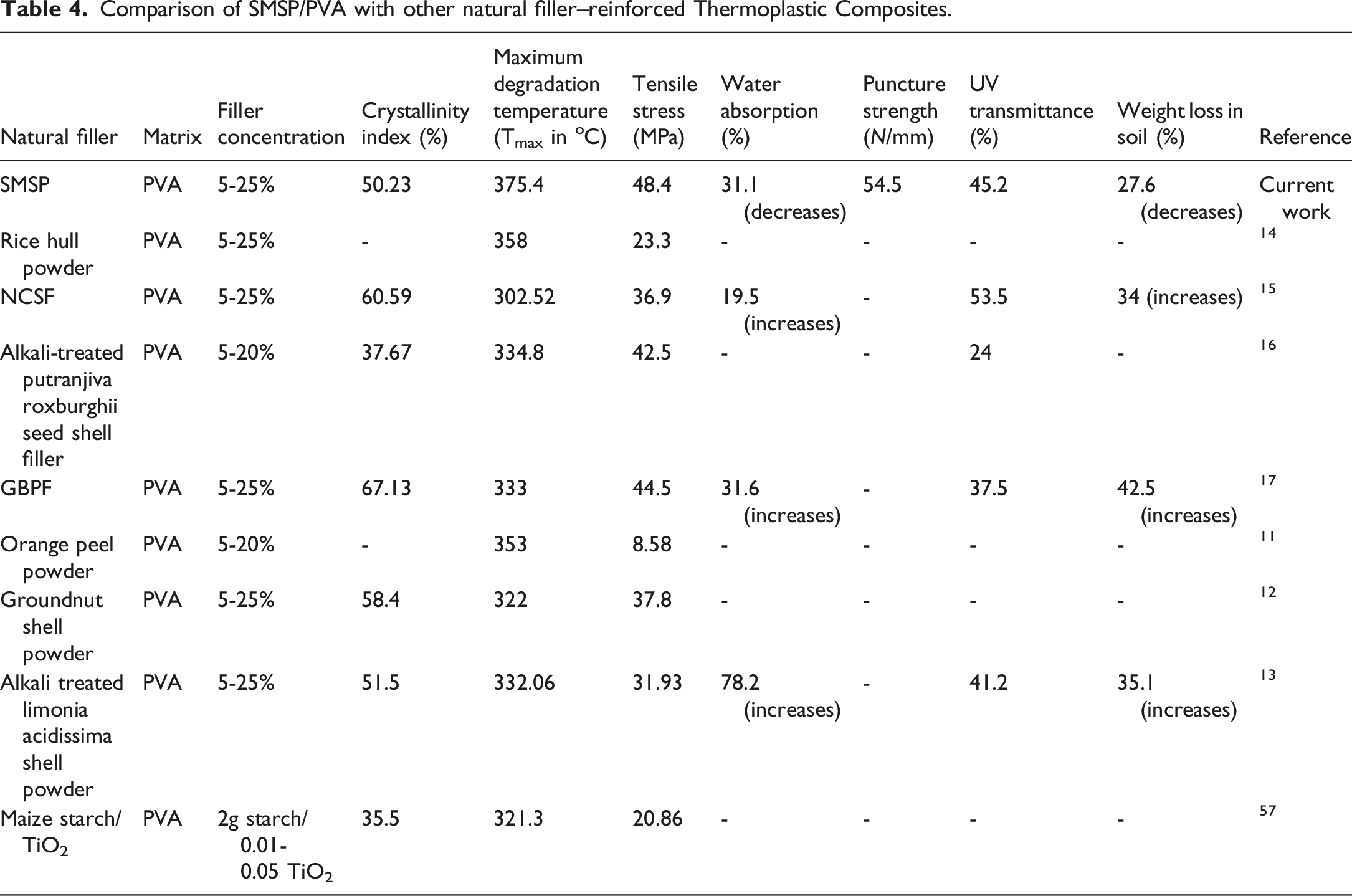
Comparison of SMSP/PVA with other natural filler–reinforced Thermoplastic Composites.
Conclusion
This study not only proves SMSP to be an effective lignocellulosic reinforcing agent in polyvinyl alcohol biofilms but also confirms that substantial improvements in mechanical strength, thermal durability, UV protective effects, and moisture barrier properties can be realized in biofilms without modifying them chemically using SMSP-induced crystallization and hydrogen bonding effects. The key to this innovation is utilizing an underexplored agro-bio waste source to create a biofilm that simultaneously remediates the natural hydrophilicity and thermal susceptibility of pure PVA films, while preserving their beneficial biological degradability to meet the demand for green packaging materials with extended shelf life and sufficient industrial durability for effective real-world packaging applications. Perhaps one of the most important aspects of moving forward with future studies is to focus on longer-term environmental ageing processes that need to be addressed alongside adequate strategies to optimize flexibility and barrier properties to fulfil real-world industrial uses without additional modifications.
Consent to participate
Patient consent does not apply to this study as it did not involve human participants.
Footnotes
Acknowledgements
The authors have nothing to report.
Authors Contribution
B.Devi – Conceptualisation, Formal analysis, Methodology, Investigation, Writing original draft. P.Udhayakumar - Investigation, Supervision, review & editing, Balavairavan Balasubramanian - Validation & Visualisation, Resources & Drafting
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical considerations
The authors have nothing to report.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.