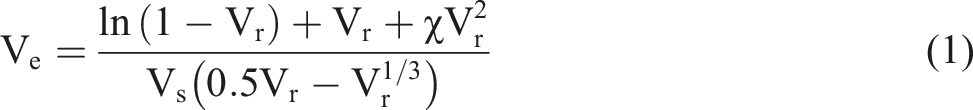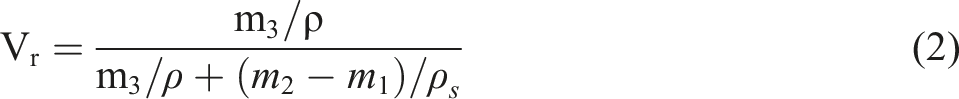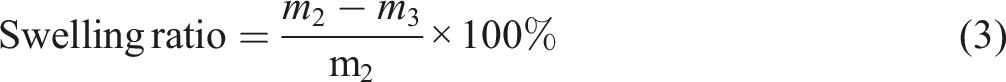Abstract
Thermally repairable elastomers combining high mechanical properties and efficient self-healing capability remain challenging due to the inherent property conflicts. In this work, an epoxidized natural rubber (ENR)-based vitrimer composite was successfully developed by constructing a hybrid dynamic network through sebacic acid crosslinking of ENR combined with interfacial β-hydroxy ester bond formation using KH560 functionalized SiO2 (KSiO2), where the functionalized silica simultaneously acts as reinforcing filler and dynamic crosslinker. Moreover, polypropylene (PP) was incorporated to enhance the mechanical performance of the elastomeric matrix. With increasing KSiO2 content, the composites achieved 34.1% higher tensile strength and 44.6% greater elongation at break, while the PP incorporated composites yielded superior tensile strength compared to the unfilled composites. Furthermore, the composites demonstrated complete incision recovery under thermal treatment, and retained more than 90% of original tensile strength after healing.
Keywords
Introduction
Rubbers are renowned for their high elasticity and indispensable applications in tires, seals, and other engineering products.1–3 These superior properties originate from their three-dimensional cross-linked networks formed during vulcanization, which provide mechanical properties, abrasion resistance and chemical resistance. However, this irreversible crosslinking fundamentally limits their recyclability and biodegradability.4,5 Currently, over 70% of discarded rubbers undergo incineration or landfill processes that lead to severe ecological challenges from accumulating rubber waste. This critical limitation underscores the necessity of developing green and effective rubber vulcanization strategies for the sustainable rubber materials.6–8
Vitrimer, pioneered by Leibler et al., 9 provides a revolutionary solution through dynamic covalent chemistry. Unlike conventional thermosets, vitrimers maintain constant crosslink density while enabling topological network rearrangement via exchange reactions of dynamic bonds, such as transesterification, 10 disulfide metathesis, 11 or imine exchange.12,13 This unique behavior combines the mechanical stability of thermosets with the reprocessability of thermoplastics. Particularly in rubber materials, the incorporation of dynamic bonds enables vitrimer based rubbers to achieve both self-healing capability and recyclability. However, a critical challenge persists across most of vitrimer based rubber systems, the reliance on individual dynamic bonds typically compromises mechanical properties.14–17
The incorporation of fillers in rubber industry is essential to overcome the inadequate mechanical properties of unfilled networks. However, these reinforcing fillers may simultaneously impede the restrict topological network rearrangements. The introduction of fillers presents a unique dilemma, achieving a balance between reinforcement and preservation of dynamic network properties.18–20 To resolve this intrinsic conflicts, Kaiser et al. 14 investigated hydrogenated carboxylated nitrile butadiene rubber (HXNBR)/epoxy group-functionalised calcium silicate (Esilicate) vitrimer-like elastomeric composites. Esilicate reacts with carboxylic acid groups in HXNBR to form dynamic β-hydroxyl ester linkages, enabling the filler to simultaneously function as crosslinker and reinforcing agent. The Esilicate-filled vitrimer-like elastomeric composites exhibits significantly improved mechanical properties, while the vitrimeric characteristics can be mostly maintained even at large quantities, as confirmed by temperature-dependent stress relaxation. Jia et al. 21 modified Ethylene-Propylene-Piene monomer (EPDM) rubber with furfurylamine to yield furan-modified EPDM (EPDM-g-FA), which was then crosslinked with 3-methacryloxypropyltrimethoxysilane modified silica (M-silica) via a Diels-Alder reaction. The M-silica served dual roles as both reinforcing agent and dynamic crosslinker. The resulting composites exhibited enhanced mechanical properties, and also showed good thermal reprocessability and self-healing behavior. Tong et al. 22 developed biobased epoxidized natural rubber (ENR) vitrimers through 57% carboxyl content oxidized starch (OST-57) as a renewable crosslinking agent. The ENR/OST vitrimer exhibited high elongation and excellent shape memory properties, and showed promising thermal-activated reprocessability via β-hydroxyl ester bonds with less than 12% mechanical properties loss after twice recycling cycles. Other research efforts have incorporation of nanoscale carbon materials such as carbon nanotubes and graphene into rubber based vitrimers.23–26 Based on the above literature, it is believed that combining dynamic covalent bonds with properly designed fillers can create vitrimer based rubbers that maintain both good mechanical properties and recyclability.
ENR presents an ideal material for sustainable vitrimer development due to its renewable origin and reactive epoxy groups that readily form dynamic covalent bonds, i.e. β-hydroxy ester bonds, with carboxylic acids.27–29 This work introduces a dual-phase modification strategy incorporating polypropylene (PP) and KH560 functionalized silica (KSiO2) to enhance the mechanical properties of vitrimer based ENR composites. The rigid framework of PP can strengthen the elastomeric composites without sacrificing vitrimer behavior, while epoxy groups on KSiO2 surfaces enable covalent integration with ENR through carboxylic acid. The resulting dynamic β-hydroxy ester bonds at the ENR-KSiO2 interface not only enhance filler reinforcement efficiency but also maintain thermally activated self-healing of the composites. This work provides fundamental insights into the design of performance tunable vitrimer composites but also establishes a practical pathway for developing self-healing rubber products.
Experimental
Materials
A 50 mol% epoxidation epoxidized natural rubber (ENR) was supplied by Dongguan Zhangmutou Haobang Plastic Firm, China. Polypropylene (PP) was obtained from Shanghai Petrochemical Corporation Limited, China. (3-Glycidyloxypropyl)trimethoxysilane (KH560) was bought from Shandong Bosheng Chemical Corporation Limited, China. Fumed silica nanoparticles (HDK N20, the primary size is 5-30 nm) were purchased from Wacker Chemie, Germany. Sebacic acid (SA) and 1,5,7-triazabicyclo[4.4.0]dec-5-ene (TBD) were supplied by Sigma-Aldrich, China.
Modification of fumed silica
First, 30 mL H2O, 90 mL ethanol, 15 mL KH560, and 15 g SiO2 were added into the wide-necked bottle, magnetic stirring for 1 h, then ultrasonic dispersion for 30 min. After ultrasonic dispersion, the mixture was magnetic stirred at 60°C for 3 h. The reaction product was washed for three times with ethanol to remove unreacted coupling agent, and then centrifuged by a centrifuge for 15 min. The centrifuged solid was dried in a vacuum oven at 60°C for 48 h to obtain KH560 functionalized SiO2 (KSiO2).
Fabrication of ENR/PP/KSiO2 composites
The composites were prepared with a fixed formulation (ENR/PP/SA/TBD = 90/10/4.5/1.24 phr) and variable KSiO2 content (0, 1, 3, 5 phr). Initially, ENR was loaded into the internal mixer and mixed for 1 min at 60°C. Subsequently, KSiO2, SA and TBD were introduced, and mixing continued for an additional 8 min. Finally, before discharging the resulting ENR/KSiO2 composites were collected. For the production of ENR/PP/KSiO2 composites, PP was introduced into the preheated internal mixer at 175°C and let to melt for 5 min. After melting, the ENR/PP/KSiO2 composites was added to the mixer. The ENR/PP/KSiO2 composites were further mixed for 5 min before removed from the chamber. The mixed composites were ready to make the desired thickness using the two-roll mills for easier to vulcanization. Finally, the pre-flattened composites were compressed molding for their optimal curing times at 180°C.
Characterization
The curing parameters of the composites, including cure time (t90), minimum torque (ML), and maximum torque (MH) were analyzed using a non-rotor rheometer (YF-8017, Yangzhou Yuanfeng Test Machinery Factory, China), following ASTM D2084 at 180°C.
The crosslink density was determined using the Flory-Rehner equation via equilibrium swelling measurements.
30
Initially, the samples were extracted in boiling acetone for 12 h to remove soluble fractions. The extracted samples were dried in an oven until constant weight (m1). The dried samples were immersed in toluene at room temperature for 72 h to reach equilibrium. Afterwards, took out the samples and removed the solvent rapidly by filter paper. The swollen weight (m2) was immediately measured. Finally, the swollen samples were dried in an oven at 60°C until constant weight (m3). The crosslink density (νe) was calculated using Equation (1):
The volume fraction Vr of ENR in the swollen phase was calculated by the following equation (2):
The swelling ration
31
was calculated using the equation (3) below:
The Fourier Transform Infrared Spectra (FTIR) of the samples were characterized with a Bruker Vertex 70 spectrometer (Germany) in a range of 400-4000 cm-1 with being scanned 16 times. The samples were purified by dissolving in refluxing acetone and toluene to remove the unreacted substances.
The mechanical properties of the vulcanizates were determined by using a universal testing machine (WDL-05, Jinan Chenchi Experimental Instrument, China) at room temperature. A crosshead speed of 200 mm/min was set. At least five samples from each vulcanizate were tested. The specimens were cut into a type 3 dumb-bell, according to ISO 37:2013 standard.
Dynamic mechanical properties were evaluated using a dynamic mechanical thermal analyzer (DMA Q800, TA Instruments, USA). The rectangular specimens (20 mm × 5 mm × 2 mm) were subjected to sinusoidal deformation force in multi-frequency-strain mode at the frequency of 1 Hz. The temperature sweeps were conducted from 60°C to 100°C at a heating rate of 3°C/min under nitrogen atmosphere.
Stress relaxation tests were performed on the DMA Q800 apparatus (TA Instruments, USA). Rectangular specimen (10 mm × 4 mm × 0.5 mm) was were mounted between fixtures and subjected to an initial preloaded force of 1 × 10-3 N to maintain straightness. The system was then equilibrated for 15 min at 180°C. The sample was then deformed to a constant strain of 1%, and the stress decay was monitored over time. 32
The healing efficiency (η) of was calculated using equation (4). The vulcanizates were subjected to a complete incision using a razor blade. Then, the damaged sample were placed in a mold and compressed molding for self-healing at 180°C under 10 MPa pressure for vary durations, i.e. 1 h, 2 h and 3 h.
Results and discussion
Preparation of ENR based vitrimers with hybrid dynamic covalent networks
The construction of a hybrid dynamic networks is illustrated in Scheme 1(a). Sebacic acid as a dicarboxylic acid crosslinker, reacts with epoxy groups on both ENR and KSiO2 through ring-opening esterification. This dual reaction creates a hybrid dynamic covalent network, ENR crosslinked through dynamic β-hydroxy ester bonds, KSiO2 anchored in the ENR matrix, transforming the filler into active crosslinking points. The dynamic covalent bonds provide the hybrid networks with thermally reversible characteristics through transesterification reactions at elevated temperatures, as is shown in Scheme 1(b).34,35 The process of ENR based vitrimers (a) formation and (b) transesterification.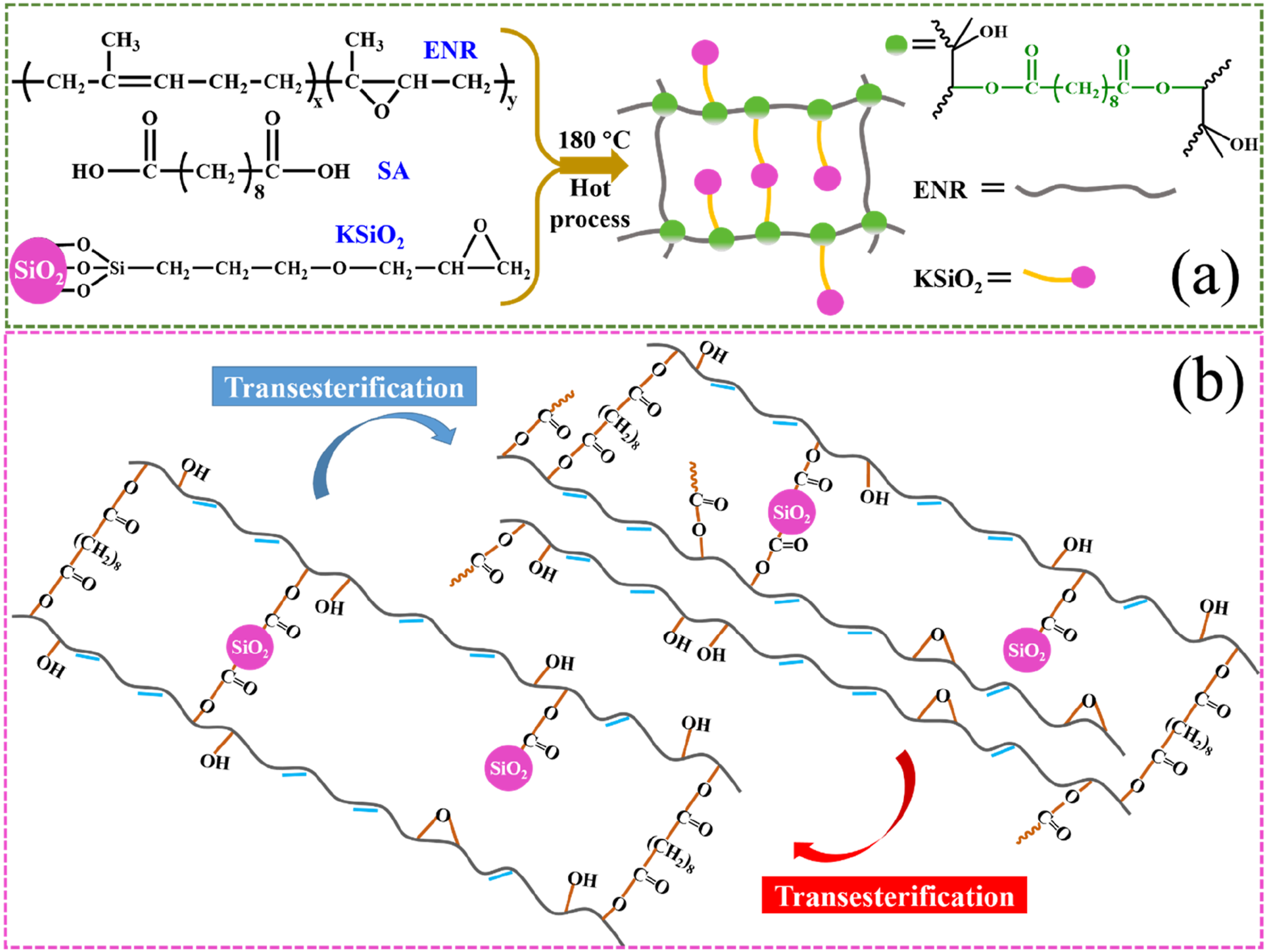
The successful functionalization of SiO2 by KH560 was confirmed using FTIR is shown in Figure 1(a). It is evidenced by the appearance of characteristic epoxy group absorption at 920 cm-1, the absorption peaks of Si-CH2-R at 1201 cm-1. The stretching vibration of Si-O-Si bonds can also be observed at 1112 cm-1 and 798 cm-1.36–38 This modification enabled subsequent chemical interactions between the epoxy functionalized SiO2 and the ENR/PP matrix. FTIR spectrum of (a) SiO2 and KSiO2; (b) t90 of ENR/PP/KSiO2 composites; (c) MH-ML of ENR/PP/KSiO2 composites; (d) crosslink density of ENR/PP/KSiO2 composites; (e) sol content of ENR/PP/KSiO2 composites; (f) FTIR spectrum of ENR/PP/KSiO2 composites with varying KSiO2 content.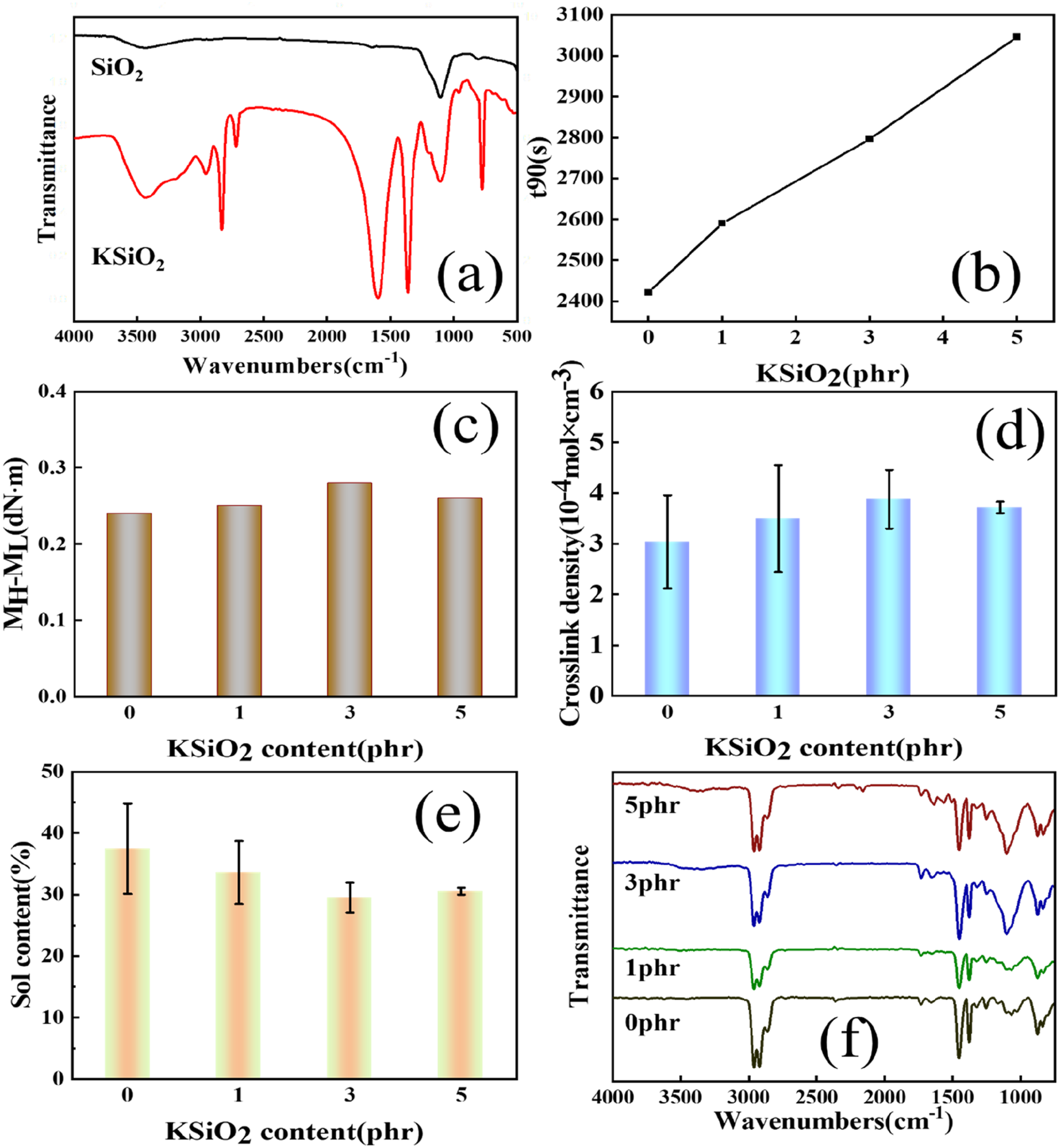
Figure 1(b) shows that the curing time t90 increases with higher KSiO2 content, from 43 min (0 phr KSiO2) to 51 min (5 phr KSiO2), indicating the delayed vulcanization behavior of the composites. This retardation effect can be attributed to the carboxyl groups of SA reacts competitively with epoxy groups on both ENR chains and KSiO2 surfaces. Increased KSiO2 content increases the composite viscosity, hindering the molecular collision between reactive species. Nevertheless, the torque difference (MH-ML) increases with KSiO2 content, as is shown in Figure 1(c), demonstrated the enhanced crosslinking density at higher KSiO2 content. Surface epoxy groups of KSiO2 participate in the vulcanization reaction, forming covalent ENR-SA-KSiO2 bridges that act as additional crosslinking points, and thus the torque difference (MH-ML) increases. Quantitative evidence of network formation is provided in Figure 1(d) and (e). With the incorporation of 3 phr KSiO2, the crosslinking density increased by 27.5% (from 3.04 ± 0.92 to 3.88 ± 0.57 mol/cm3), accompanied by a 21.2% decrease in swelling ratio (from 37.46 ± 7.33% to 29.51 ± 2.41%). The increase in crosslinking density and decrease in swelling ratio reflect the efficient interfacial bridging within the composites. At 5 phr KSiO2 content, both parameters showed slight reversals, due to the aggregation of KSiO2 reduces accessible epoxy sites. These results confirm KSiO2 as a multifunctional filler that enhances the matrix through covalent integration. 39
The chemical evolution of the crosslinked network was further investigated by FTIR spectroscopy, as is shown in Figure 1(f). The characteristic absorption peak of epoxy group absorption at 870 cm-1 gradually diminished with increasing KSiO2 content, while the intensity of the Si-O-C vibration band at 1100 cm-1 intensified.40,41 These spectral changes confirm the vulcanization mechanism of the composites, SA reacts with the epoxy groups of ENR to form dynamic β-hydroxy ester linkages, and also esterification with the epoxy groups of KSiO2. This synergistic process constructs a hybrid dynamic covalent network, which consistent with the increased crosslink density (Figure 1(d)) and reduced swelling ratio (Figure 1(e)).
Mechanical properties: Static and dynamic behavior
Representative stress-strain curves and the corresponding tensile modulus of the ENR/PP/KSiO2 composites are presented in Figure 2(a) and (b), respectively. The mechanical performance of ENR/PP composites exhibited significant dependence on KSiO2 content. As shown in Figure 2(b), the tensile modulus increased significantly from 2.33 ± 0.10 MPa (0 phr) to 3.55 ± 0.31 MPa (5 phr), representing a 52.4% enhancement. Similarly, both tensile strength and elongation at break improved considerably with higher KSiO2 loadings, as demonstrated in Figure 2(c) and (d). The tensile strength rose from 3.26 ± 0.20 MPa (0 phr) to 4.37 ± 0.54 MPa (5 phr), representing a 34.1% increase, while elongation at break rose from 215.9 ± 19.43% to 312.2 ± 73.93%, an increase of 44.6%. This simultaneous improvement in modulus, strength and ductility originates from the dual functionality of KSiO2, which acts as both reinforcing filler and dynamic crosslinker. The covalent ENR-SA-KSiO2 bridges enhanced the crosslink density of the matrix, while the efficient coupling between silica nanoparticles and matrix enabled the stress transfer within the matrix. These mechanical property trends correlate well with the increase in crosslink density of the interconnected hybrid network. Additionally, the mechanical properties of the ENR/KSiO2 composites without PP was also compared. It is revealed that ENR/PP/KSiO2 composites exhibited superior tensile strength at all filler KSiO2 loadings, attributable to the rigid framework of PP. While the ENR/KSiO2 system demonstrated greater elongation at break due to enhanced chain mobility in the absence of PP crystalline domains.
42
The dynamic mechanical analysis (Figure 2(e)) reveals that the loss factor (tanδ) peak temperature, corresponding to the glass transition temperature (Tg), decreases systematically from 0.6°C (0 phr) to -3.2°C (5 phr) as KSiO2 content increased. This depression in Tg is accompanied by a concurrent decrease in the storage modulus in the glassy state (Figure 2(f)), suggesting an enhancement in segmental mobility. This phenomenon may be attributed to the competing effects between network constraints and interfacial plasticity. The formation of ENR-SA-KSiO2 covalent bridges increased the crosslink density, which would restrict the polymer segmental mobility and raise Tg. However, the interfacial plasticity, originating from dynamic β-hydroxy ester bonds and propyl ether spacers in KH560, enhanced polymer chain mobility through free volume effects, which decreased Tg.
43
The hybrid ENR-SA-KSiO2 dynamic covalent networks enhanced static mechanical properties and dynamic chain mobility. Typical stress-strain curves (a) and tensile modulus (b) of ENR/PP/KSiO2 composites; tensile strength (c), elongation break (d) of ENR/KSiO2 and ENR/PP/KSiO2 composites; tanδ (e) and storage modulus (f) of ENR/PP/KSiO2 composites; stress relaxation curves (g) for ENR/PP/KSiO2 composites with 1% strain.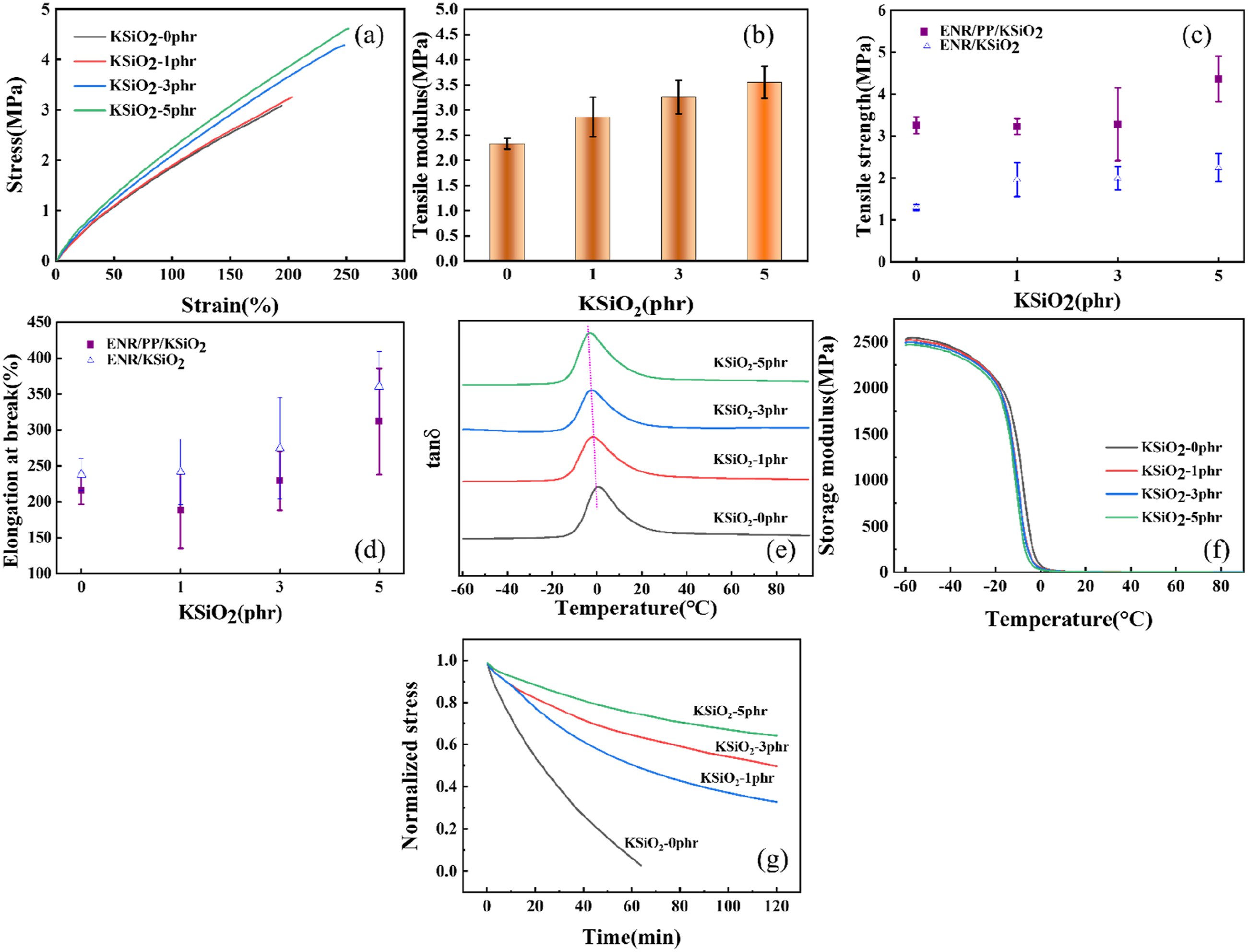
Stress relaxation experiments were performed to investigate the dynamic behavior of the hybrid ENR-SA-KSiO2 dynamic covalent networks. All composites exhibited progressive stress relaxation at 180°C, indicating that the composites can undergo ester exchange reaction at elevated temperatures. The stress relaxation curves (Figure 2(g)) of the ENR-SA-KSiO2 covalent networks show a high silica dependence of stress relaxation. Higher loadings of KSiO2 not only slowed down the relaxation rate but also elevated the residual stress after relaxation. It is attributed to the increased crosslink density and enhanced initial modulus with increased KSiO2. The crosslink points restricted molecular chain mobility and reduced the ester exchange efficiency at higher filler loadings.
Self-healing properties of ENR/PP/KSiO2 composites
The ENR/PP/KSiO2 composites exhibited self-healing capabilities attributable to their dynamic reversible bond, i.e. β-hydroxy ester bonds and hydrogen bonds. The self-healing performance was demonstrated by the cut recovery test, where a complete incision was made through the rubber sheet. The sample was placed in a preheated compression mold (180°C) and subjected to 10 MPa pressure for varying durations. Changes of the incision were observed and presented in Figure 3(a), the incision complete disappearance after thermal treatment, indicating the effective self-healing ability of the composites. Meanwhile, the self-healing mechanism of the ENR/PP/KSiO2 composites is proposed, as shown in Figure 3(b). During healing, the ENR/PP chains first diffuse across damaged interfaces facilitated by thermally enhanced mobility. Simultaneously, the β-hydroxy ester bonds undergo thermally activated exchange to rearrange the crosslinked network. Finally, the covalent networks fully reconnect at the interface, completing the healing process. (a) Visual demonstration of self-healing behavior; (b) self-healing mechanism of ENR/PP/KSiO2 composites.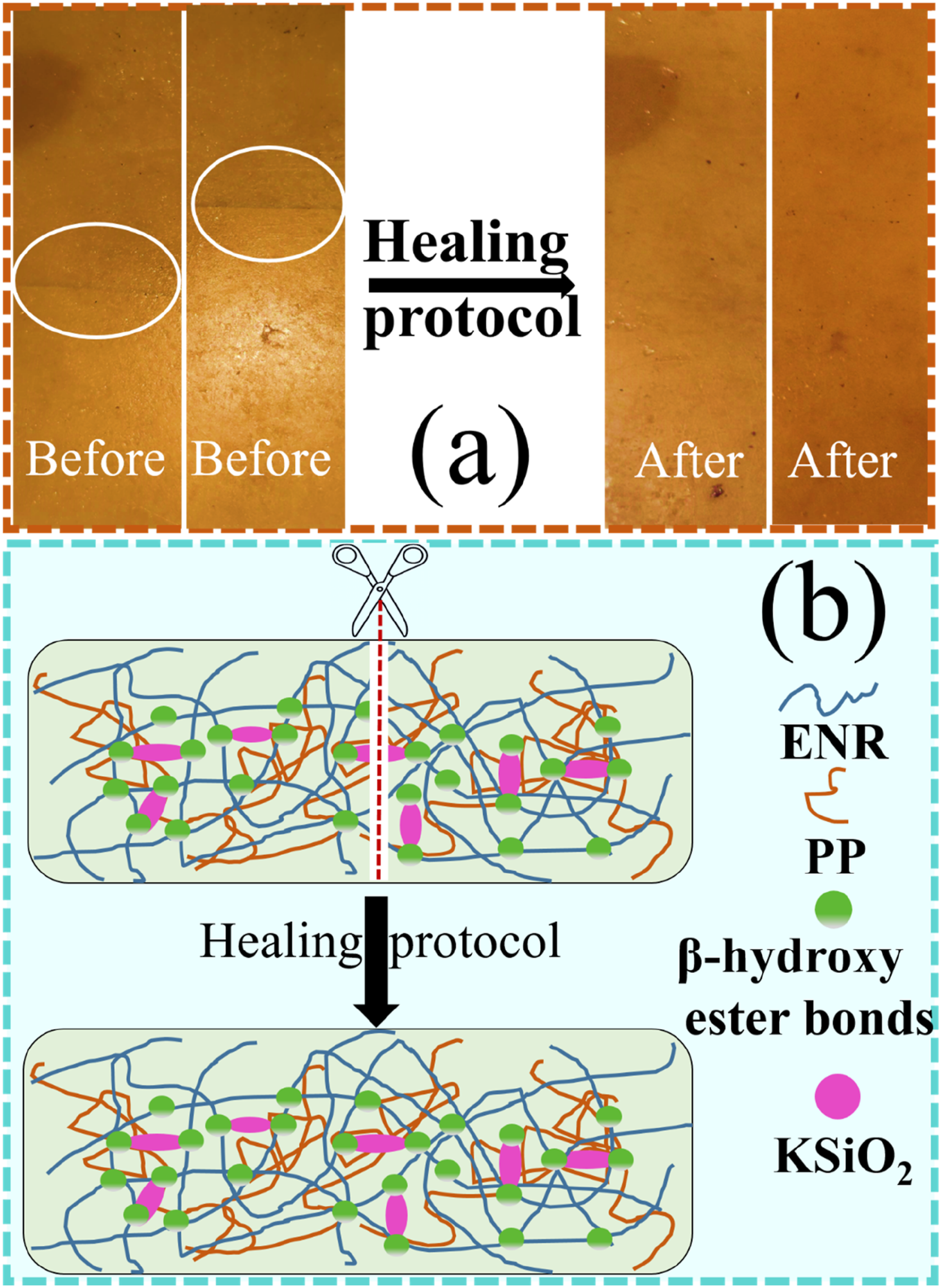
To quantify the self-healing properties, the mechanical properties of the composites after thermal treatment were conducted, as is illustrated in Figure 4(a) and (c). And the self-healing efficiency was calculated by comparing the tensile properties before and after healing, as is shown in Figure 4(b) and (d). Compared to the virgin samples, all the healed ENR/PP/KSiO2 composites exhibited reduced tensile strength and elongation at break. The observed mechanical properties reduction arises from the interfacial microvoids and the side reactions, including oxidation and over crosslinking reaction, during the thermal treatment process. As evident from the results, the self-healing performance exhibits dependences on both KSiO2 content and treated durations. The specimens healed for 2 h demonstrate higher tensile strength and elongation at break than those healed for 1 h, consequently yielded higher healing efficiency. However, after 3 h of healing, the mechanical performance fluctuations were observed, which may stem from the side reaction within the systems. Furthermore, the tensile strength of healed specimens presented a non-monotonic dependence on KSiO2 content, the tensile strength first decreased and then increased, reaching a minimum at 3 phr, while the elongation at break showed a continuous increase with higher KSiO2 content. This may be attributed to the crosslinked network structure, as is demonstrated in Figure 1(d), the crosslink density peaks at 3 phr. At this critical KSiO2 loading, the crosslinked network restricts the molecular chain mobility, thereby, hindering molecular migration for self-healing. The optimal self-healing efficiency was achieved at 2 phr KSiO2 loading with 3 h thermal treatment, demonstrating 92% recovery of tensile strength and 78% recovery of elongation at break. The mechanical properties of self-healing materials exhibit relatively large standard deviations, as the healing process is stochastic and highly sensitive to local interfacial contact and molecular dynamics. (a)Tensile strength of self-healed samples and (b) healing efficiency of tensile strength; (c) elongation at break of self-healed samples and (d) healing efficiency of elongation at break.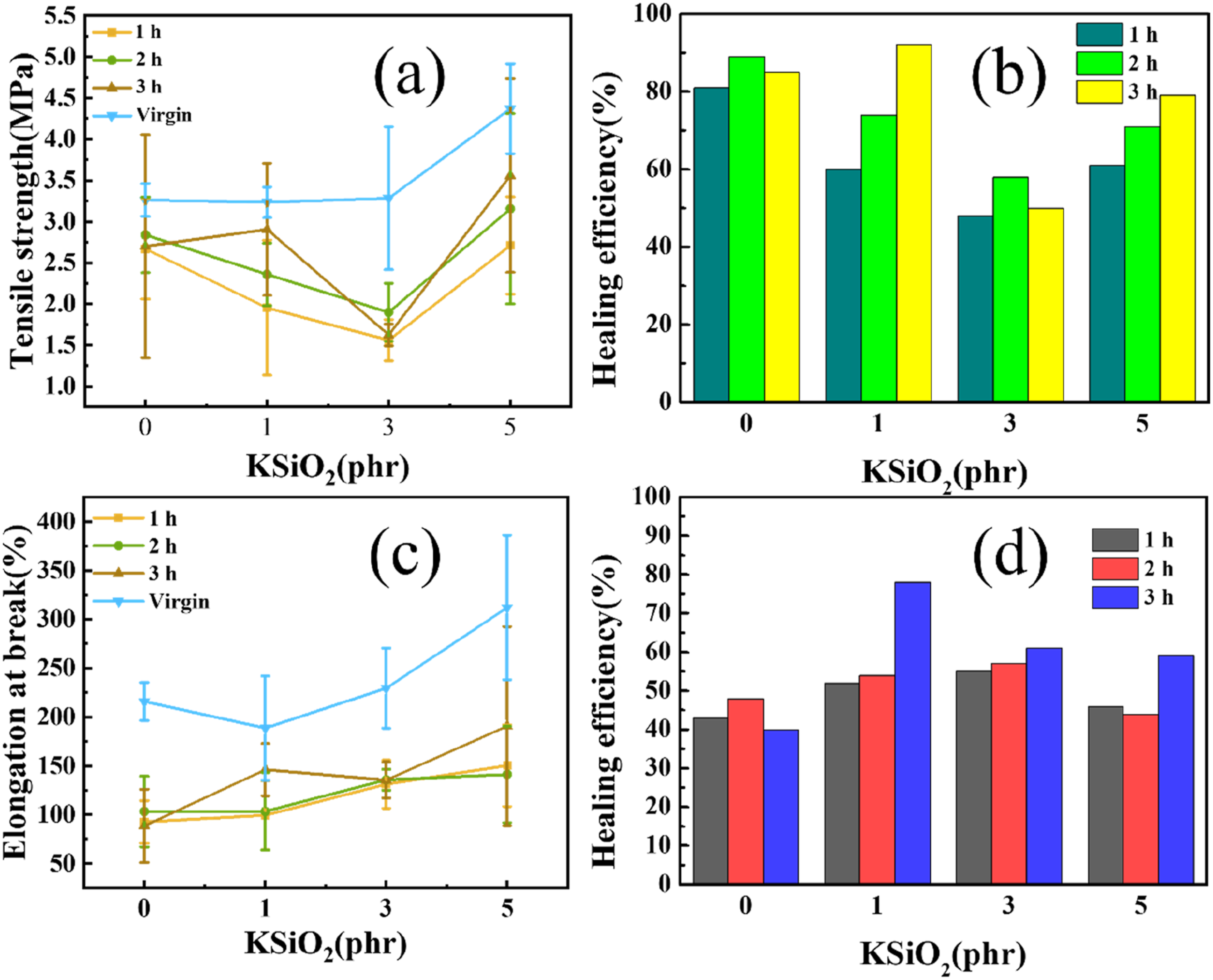
Conclusion
In this study, a sustainable ENR-based vitrimer composite was successfully developed by integrating PP and KSiO2 into a dynamically crosslinked network. The covalent bonding between ENR and KSiO2 through β-hydroxy ester linkages not only enhanced mechanical properties but also maintained the vitrimeric characteristics to a certain extent, enabling self-healing capabilities. The composites exhibited a significant improvement in tensile strength (up to 4.37 MPa) and elongation at break (312.2%) with increasing KSiO2 content, attributed to effective stress transfer and crosslink density enhancement. Dynamic mechanical analysis indicated that the hybrid network balances rigidity and chain mobility, while stress relaxation confirmed the dynamic nature of the covalent bonds at elevated temperatures. Furthermore, the composites demonstrated excellent self-healing capability, with complete incision recovery under thermal treatment, and retained high mechanical properties after healing. This work provides a viable strategy to balance mechanical performance and dynamic reparability in sustainable elastomers. We believe that this new vitrimer composite system, which combines strength, self-healing, and tunable dynamics, can provide a valuable reference for the design of tunable sustainable materials.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the PhD Research Startup Foundation of Nanchang Hangkong University (grant no. EA202001381), the National Natural Science Foundation of China (grant no. 52403096), Youth Innovation Team of Shandong Province Higher Education Institutions (grant no. 2023KJ308).