Abstract
The objective of this novel research was to examine the unique effects of
Introduction
Over the last few decades, researchers and industry professionals have examined and developed various cutting-edge packaging techniques to safeguard food and address the environmental risks associated with petroleum-based packaging materials. 1 Biodegradable and edible films are a contemporary solution that has a minimal impact on the environment. They provide several advantages, including the prevention of oxidation, acting as a barrier against the transfer of substances such as moisture, fat, and gas, controlling enzymatic activation, and incorporating bioactive agents like plant extracts and essential oils that possess potent antioxidant and antimicrobial properties. Additionally, they can also include halochromic compounds.2–7
Cottonseed is a highly abundant oilseed and a significant source of plant protein. It is commonly utilized as animal feed because of its exceptional nutritional content.8,9 Studies have shown that protein from cottonseed meal produces edible films that are biodegradable, transparent, but brittle, with low tensile strength; therefore, it is advantageous to blend it with other biopolymers, as done with pectin in this research. 10
Pectin, a linear anionic polysaccharide composed of α-(1→4)-D-galacturonic acid units, is derived from citrus fruit peels and plant cell walls. The degree of esterification classifies pectin into three categories: high, medium, and low. 11 This degree of esterification influences pectin’s physical, chemical, and functional properties. A non-toxic, transparent, easily accessible, biodegradable, and environmentally friendly layer with good mechanical strength is produced when pectin is used to formulate edible films and coatings. On the other hand, it has high permeability to water vapor and lacks antimicrobial and antifungal properties. 12 Incorporating nanomaterials, antioxidants, and antimicrobial compounds into the formulation of edible films is preferred to improve the resulting films’ physicochemical and functional properties. 13
Although bioactive compounds have many health benefits, they also have several disadvantages, including the high volatility of
In nanotechnology, one dimension ranges between 1 and 100 nm; such materials have a much greater surface-to-volume ratio compared to larger particles with the same composition, resulting in improved desirable properties. Nanoparticles can be made from various materials such as proteins, polysaccharides, synthetic polymers, and metals. Recently, the use of biodegradable and biocompatible nanoparticles has gained considerable attention.19,20
In this study, chitosan nanoparticles were utilized to enhance the properties of the produced film. Chitosan is composed of N-acety-l-D-glucosamine and D-glucosamine units linked by β-(1→4) glycosidic bonds. It is derived from renewable sources, including the exoskeletons of insects, crustaceans such as shrimp and crabs, and fungal cell walls. Chitosan can be obtained through both chemical and microbiological methods.21,22 Noteworthy characteristics of chitosan include non-toxicity, barrier properties, biodegradability, biocompatibility, and antibacterial and antifungal activities. 23
In this research, a bionanocomposite film based on cottonseed protein isolate/pectin containing microencapsulated
Method and Materials
Materials
Methods
Isolation of Protein from Cottonseed Meal
The protein isolate from cottonseed meal was prepared following the method outlined by Tan et al. (2022), with minor adjustments. Initially, the purchased cottonseed meal was pulverized using a household grinder (Model SC-223FS, China) and subsequently filtered through a 0.5 mm mesh. The resulting powder was then mixed with sodium hydroxide at a volumetric ratio of 1:81 (powder to NaOH solution). The pH of this mixture was adjusted to 10.5–10.9, and the suspension was agitated for 1 hour at ambient temperature using a magnetic stirrer. Following agitation, the suspension was centrifuged at 5000 rpm for 15 min. The resulting supernatant (the liquid portion above the sediment) was carefully decanted into a new tube. To precipitate the protein, the pH of the supernatant was adjusted to 4.5 by the addition of either 0.1 or 1 M HCl. The suspension was again centrifuged at 5000 rpm for 15 min. After discarding the supernatant, the remaining precipitate was neutralized using 0.1 or 1 M NaOH and subsequently freeze-dried. Finally, the protein content was determined using the Kjeldahl method and reported to be 82%. 24
Extraction of Salix Aegyptiaca Extract
Fresh
Encapsulation of Salix Aegyptiaca Extract
The
To ensure complete dissolution and initial homogenization, the mixture was subjected to high-speed homogenization (IKA T25 digital Ultra-Turrax, Germany) at 11,000 rpm for 10 min.
Then, in the microcapsules preparation stage, the final solution was prepared using ultrasound (model UP400S, Schreck Hoelscher, Germany), with 150 W and 20 kHz and a H7 type titanium probe with a diameter of 7 mm and a length of 100 mm for 5 min. 26
Particle Size Analysis and Zeta Potential of Microcapsules
The particle size of the emulsion was determined using dynamic light scattering (DLS) and PolyDispersity Index (PDI) with a Zetasizer device (Malvern Instruments, Worcestershire, UK).
A Zetasizer (Model Nano ZS, Malvern Instruments, UK) was used at 25°C to measure the average particle size (Z-Average), the particle size distribution index (PDI), and the surface electrical charge of particles against pH. The material was diluted to a 1:100 ratio with deionized water before analysis. 27
Stability Analysis of Microcapsules
A predetermined portion of the sample was sealed to avoid evaporation and kept at room temperature for 3 weeks to evaluate the stability of samples against phase separation and coalescence. 28
Preparation of Nanocomposite Films
List of experiments in the CCD
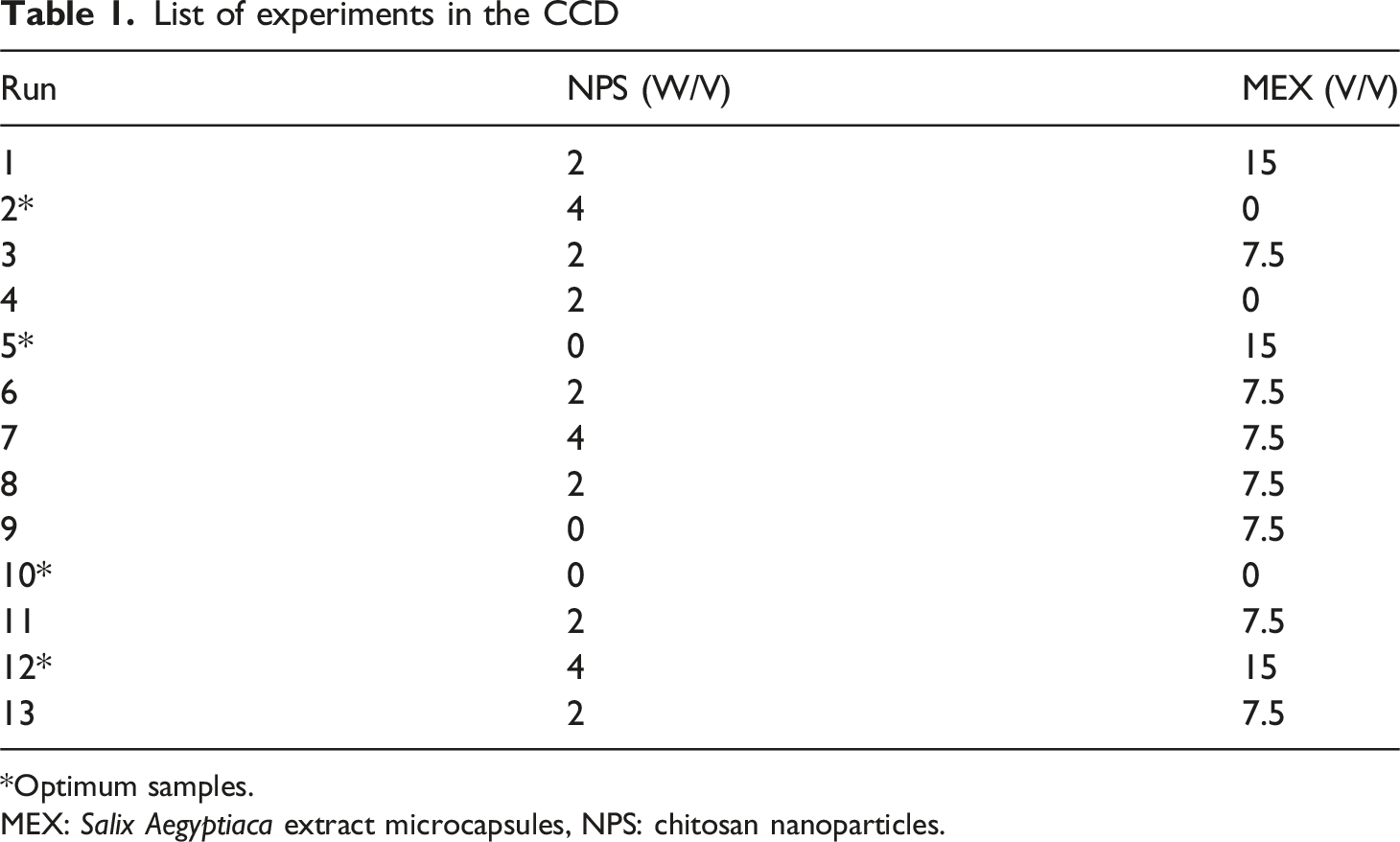
*Optimum samples.
MEX:
Exactly 25 mL of the final film solution was poured onto identical glass plates (diameter: 25 cm) and dried in a forced-air convection oven at 40°C for 24 hours. 29
Nanocomposite Film Testing
Moisture Content
The moisture content (MC) of the prepared films was determined gravimetrically. Film samples, precisely cut to 3 × 3 cm2 pieces, were initially weighed to determine the initial mass (M1). These samples were then dried in a forced-air convection oven at 130 ± 2°C until a constant weight (M2) was achieved. The moisture content was subsequently calculated using equation (1)
30
: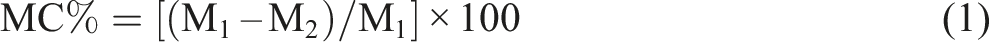
Solubility
After immersing the film samples in water for a day, solubility tests were performed to determine the percentage of dissolved dry matter. To achieve this, films were divided into 2 × 2 cm2 pieces, weighed (Wi), and left in 30 mL of distilled water for 3 hours while stirring. After removing extra water with filter paper, the samples were baked for a day at 105 ± 2°C to dry them out (Wf). The solubility was calculated using the equation (2).
31
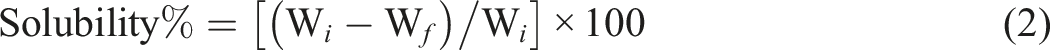
Colorimetry
A CIE colorimeter (Minolta CR300 Series, Minolta Camera Co. Ltd., Osaka, Japan) was used to measure film color parameters such as L*, a*, b*. Subsequently, Hue angle, Chroma, yellowness index, whiteness index, and total color difference (ΔE) were calculated using the equations (3)–(7) respectively.

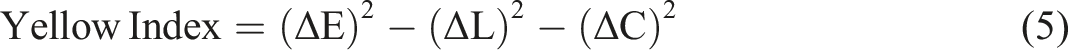
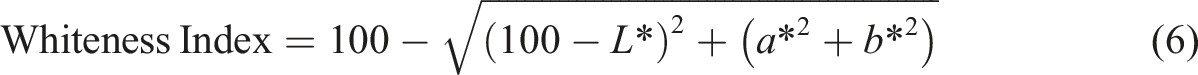
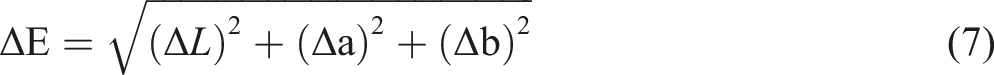
Where: the color parameters (ΔL, Δa, and Δb) were compared to a standard white plate (L = 97.39, a = −5.11, b = 7.16).
Thickness
The thickness of the films was randomly measured at several points (10 locations) using a digital micrometer (Mitutoyo-Co, Japan) with a precision of 0.001 mm. 32
Antioxidant Activity
To evaluate the antioxidant activity of the film samples, 25 mg of the samples were dissolved in 4 mL of distilled water. Then, 2 mL of the extract was mixed with 0.2 mL of a 1 mM methanolic DPPH solution and stirred well for 5 min, followed by incubation in the dark at room temperature for 1 hour. The absorbance of the samples was read at 517 nm using a spectrophotometer (Model T60 UV, USA). The percentage of DPPH free radical scavenging activity was calculated using the equation (8)33–35: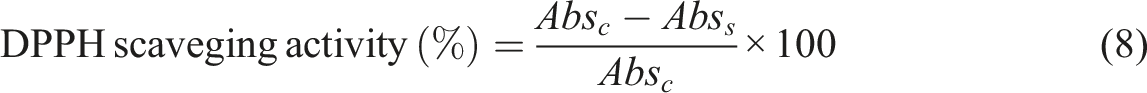
Where: Absc: Absorption of the control, Abss: Absorption of the film sample
Mechanical Properties
Using a texture analyzer (Model TA.XT plus C, UK) the mechanical characteristics of the film samples were determined. Film samples were made in 1 × 5 cm dimensions and conditioned at room temperature with 55% relative humidity (calcium nitrate) for 72 hours. The grips were initially spaced 30 mm apart, and their speed was 0.83 mm/s.
32
The starting length of the film sample (L0), the maximum force at the breaking point (Fmax), the cross-sectional area of the film (A), and the maximum elongation of the film at the breaking point (Lmax) were all noted. Then the tensile strength and elongation at break was calculated using equations (9) and (10) respectively.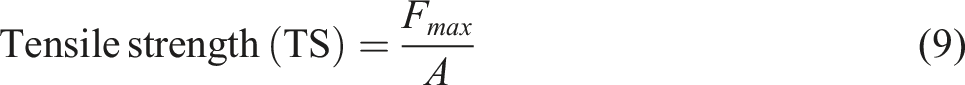
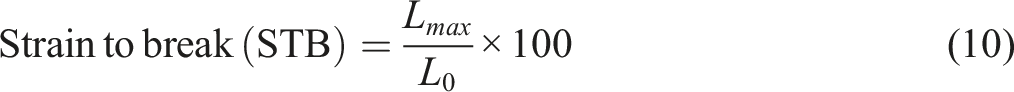
FTIR
FTIR spectroscopy (Spectrum Two, Perkin-Elemer, USA) investigated the structure and interactions within the optimized film samples. Tablets of the film (thickness ≤1 mm) were prepared by mixing with water, grinding the samples, coating them with potassium bromide at a 20:1 ratio, and applying a pressure of about 60 kPa for 10 min. The prepared samples were then analyzed with a resolution of 0.5 cm−1 in the wavenumber range of 4000-400 cm−1. 36
SEM
The microstructure of the produced bionanocomposite films was analyzed using a scanning electron microscope (Philip, Netherlands). For this purpose, samples fractured in liquid nitrogen were mounted on a metal base with carbon adhesive. During preparation for analysis, the samples were coated with gold particles, and imaging was performed at various magnifications. 36
XRD
X-ray diffraction (PHILIPS_PW1730, Netherlands) characterized the crystalline structure of the optimized bionanocomposite films. During this examination, the X-ray generator operated at an energy of 40 kV and a current of 30 mA. The diffracted rays were measured throughout a 2°θ angle range of 2 to 70°θ, using a scan rate of one degree and a step size of 0.02°θ. 37
DSC
The thermal behavior of the optimized bionanocomposite films was investigated using differential scanning calorimetry (DSC- Model DSC-R, Japan). A specific amount of the samples (ranging from 0.02 to 0.03 g) was analyzed in an aluminum pan at a heating rate of approximately 10°C/min, over a temperature range of −20 to 180°C, under a constant flow of nitrogen atmosphere. The melting temperature (TM), crystallization temperature (Tc), and glass transition temperature (Tg) were determined based on the obtained thermograms. 38
Antimicrobial Properties
The antimicrobial properties of the optimized bionanocomposite film samples were assessed using the agar diffusion method. For this purpose, the bacteria used in this method were sub cultured twice consecutively in Mueller-Hinton broth. This was followed by injection with 0.1 mL of broth culture containing bacteria (105 - 106 CFU/mL) prepared the night before. Cut to 15 mm film samples were placed on plates containing Gram-positive bacteria (
Statistical Analysis
In this study, a central composite design was employed at three levels for two variables: microencapsulation of
Results and Discussion
Particle Size, Particle Size Distribution, Zeta Potential, and Microcapsule Stability
Zeta potential and particle size of microcapsules act as pivotal indexes of their physical properties, including electrical charge and dimensions properties, and provide insights into their potential to interact with surrounding environments and biological systems. The zeta potential, particle size, and particle size distribution of the produced Particle size, particle size distribution and zeta potential of 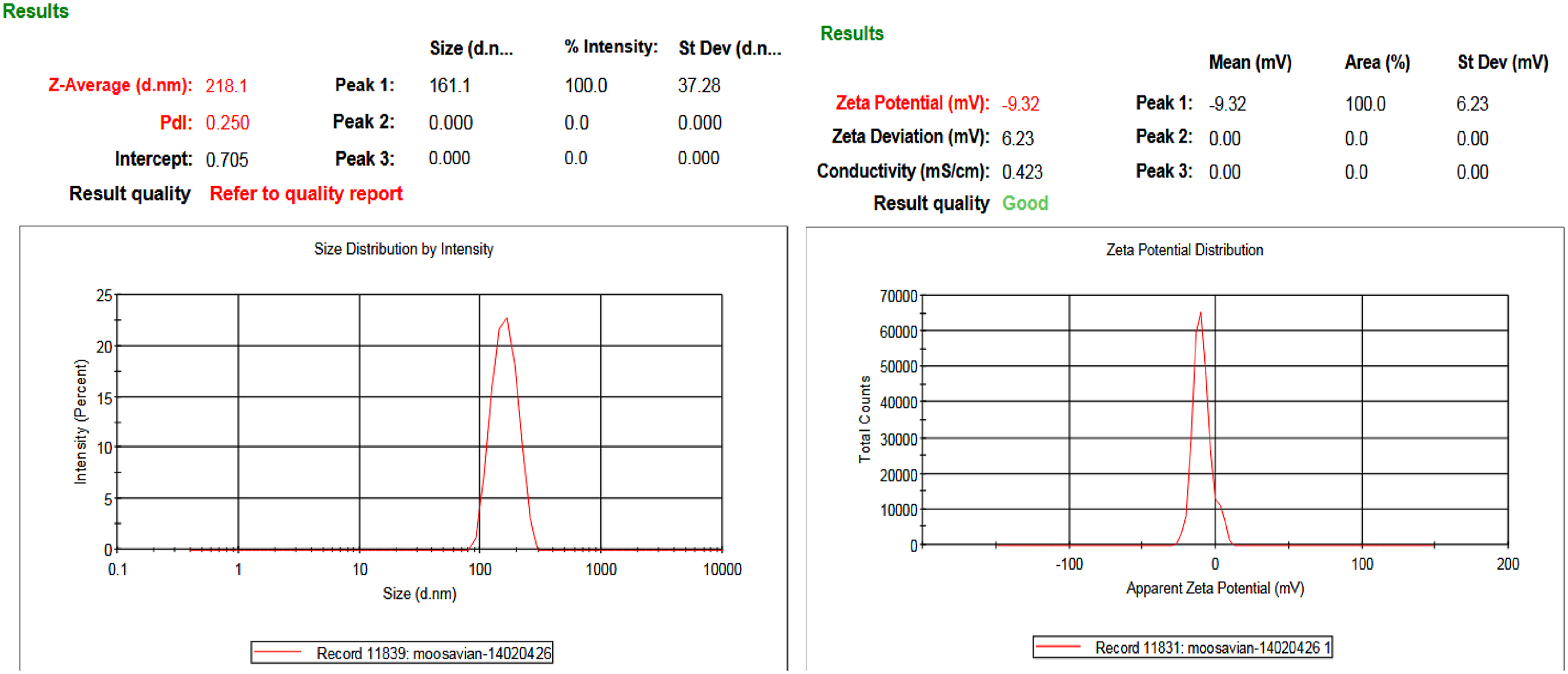
Particle size has a significant impact on numerous characters of microcapsules, particularly including permeability, specific surface area and solubility.43,44
Physicochemical and Instrumental Tests
Moisture Content
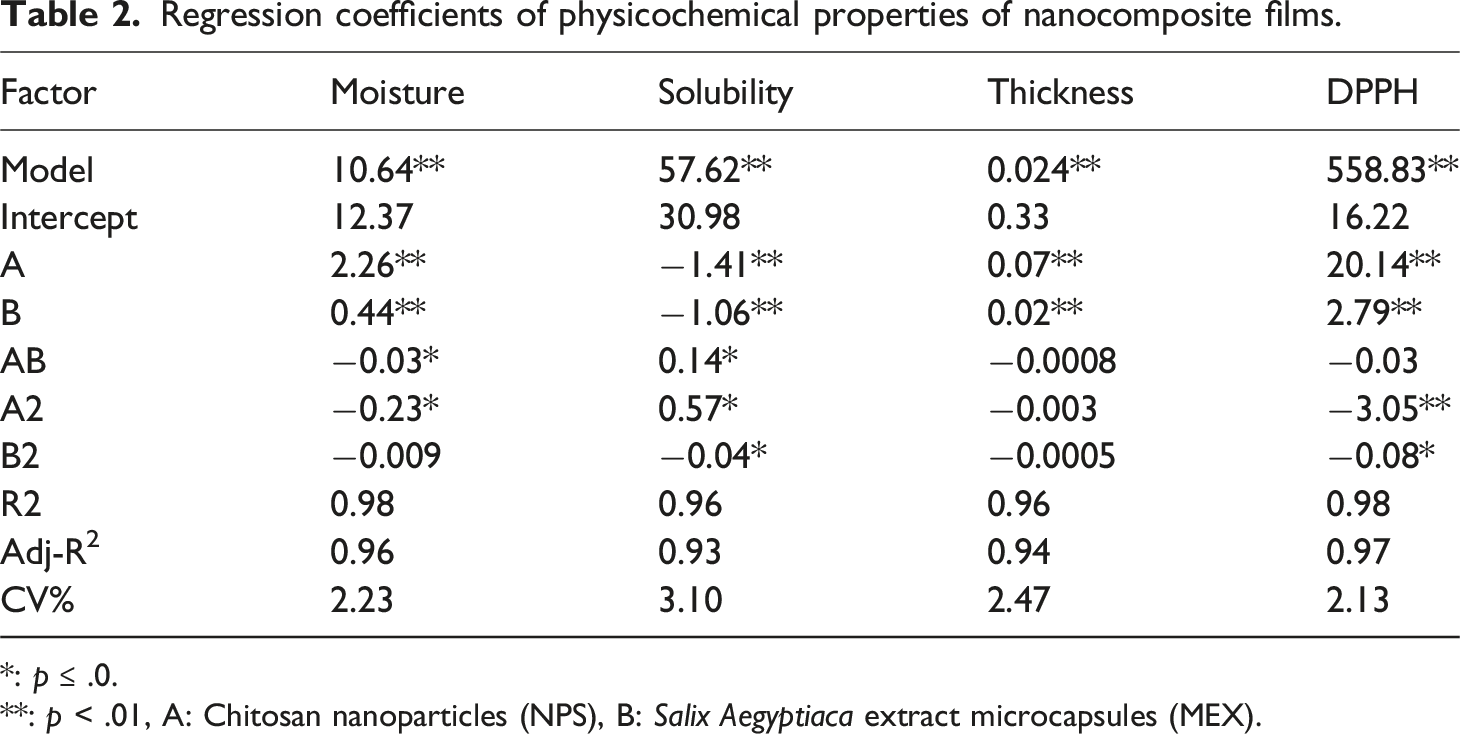
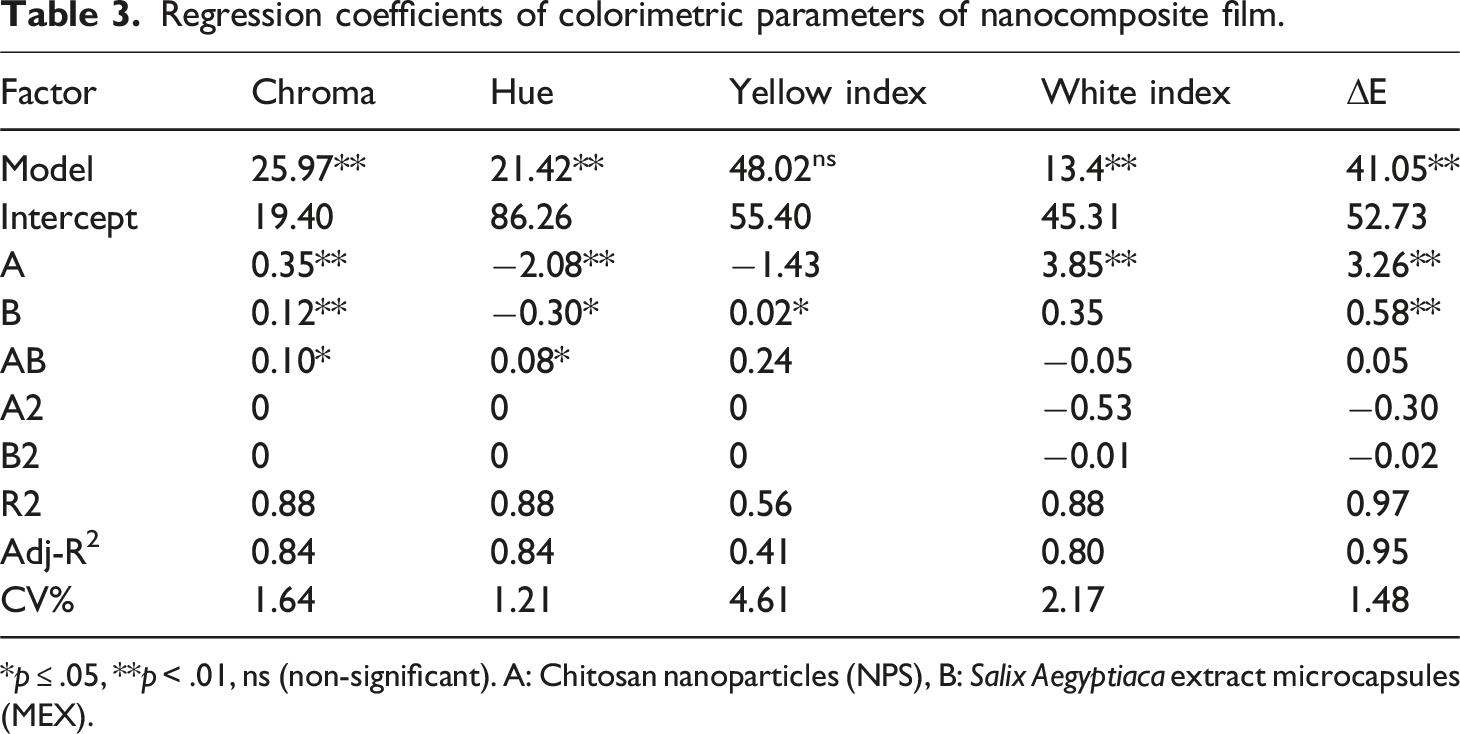
Regression coefficients of physicochemical properties of nanocomposite films.
*:
**:
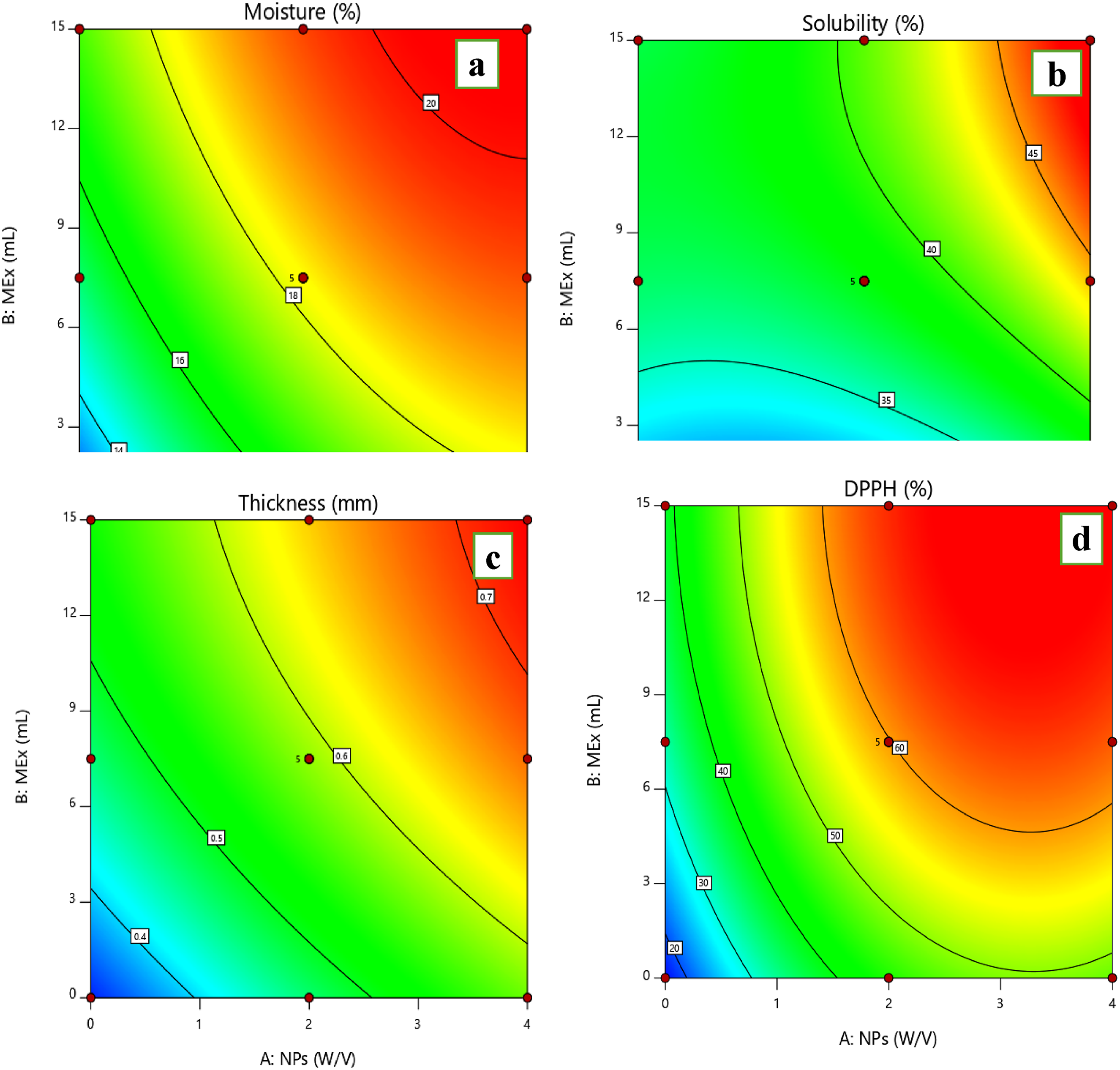
Effect of Chitosan nanoparticles (NPX) and
Solubility
According to the results of the analysis of variance (Table 2) and Figure 2(b), the individual, interactive, and quadratic effects of the studied factors were significant (
The findings demonstrated that when the amounts of
Thickness
Based on the regression coefficients shown in Table 2 and Figure 2(c), the individual effects of
Antioxidant Activity
The antioxidant capacity of the created bionanocomposite film samples was significantly affected (
Colorimetriy
Regression coefficients of colorimetric parameters of nanocomposite film.
*
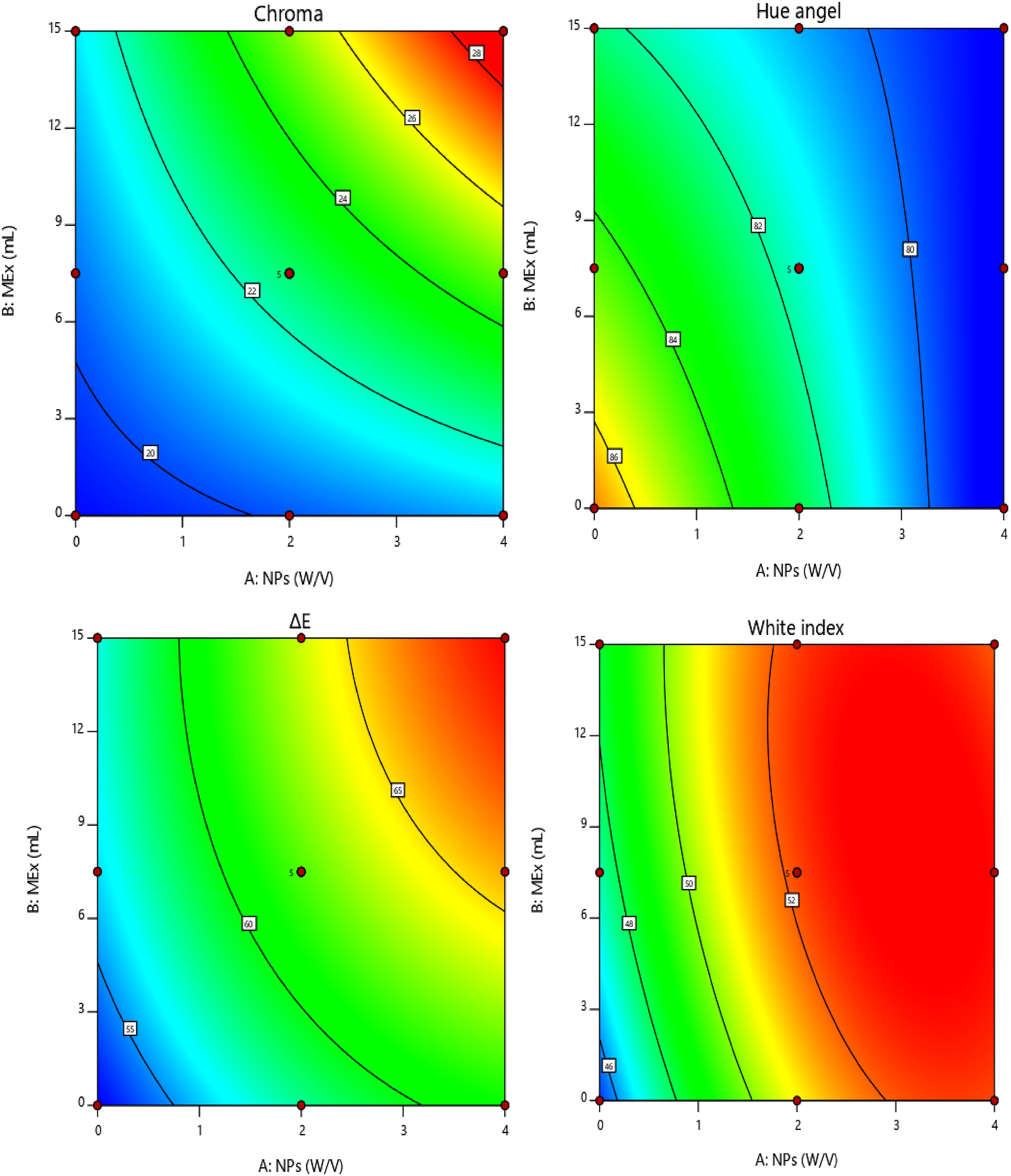
Effect of Chitosan nanoparticles(NPS) and
Regarding the yellowness index, its lack of statistical significance suggests that while the overall color profile shifted markedly (ΔE increased), the yellow component itself was either present at a very low concentration or its variation across factor levels was too consistent to yield a significant statistical result. These results underscore that the concentration of active components dictates the final color quality of the film. This dependence of film color on formulation type and concentration aligns with established literature. 23
SEM
Scanning electron microscopy images of the optimized bionanocomposite films are shown in Figure 4. As seen in Figures 4(a), the cottonseed protein isolate-pectin bionanocomposite film exhibited a uniform microstructure with no pores, surface cracks, or irregularities, contributing to mechanical properties and low water vapor permeability.
56
The addition of 15% SEM of active films: (a) (pure film), (b) (film containing 15% FTIR of active films: (a) (pure film), (b) (film containing 15% 
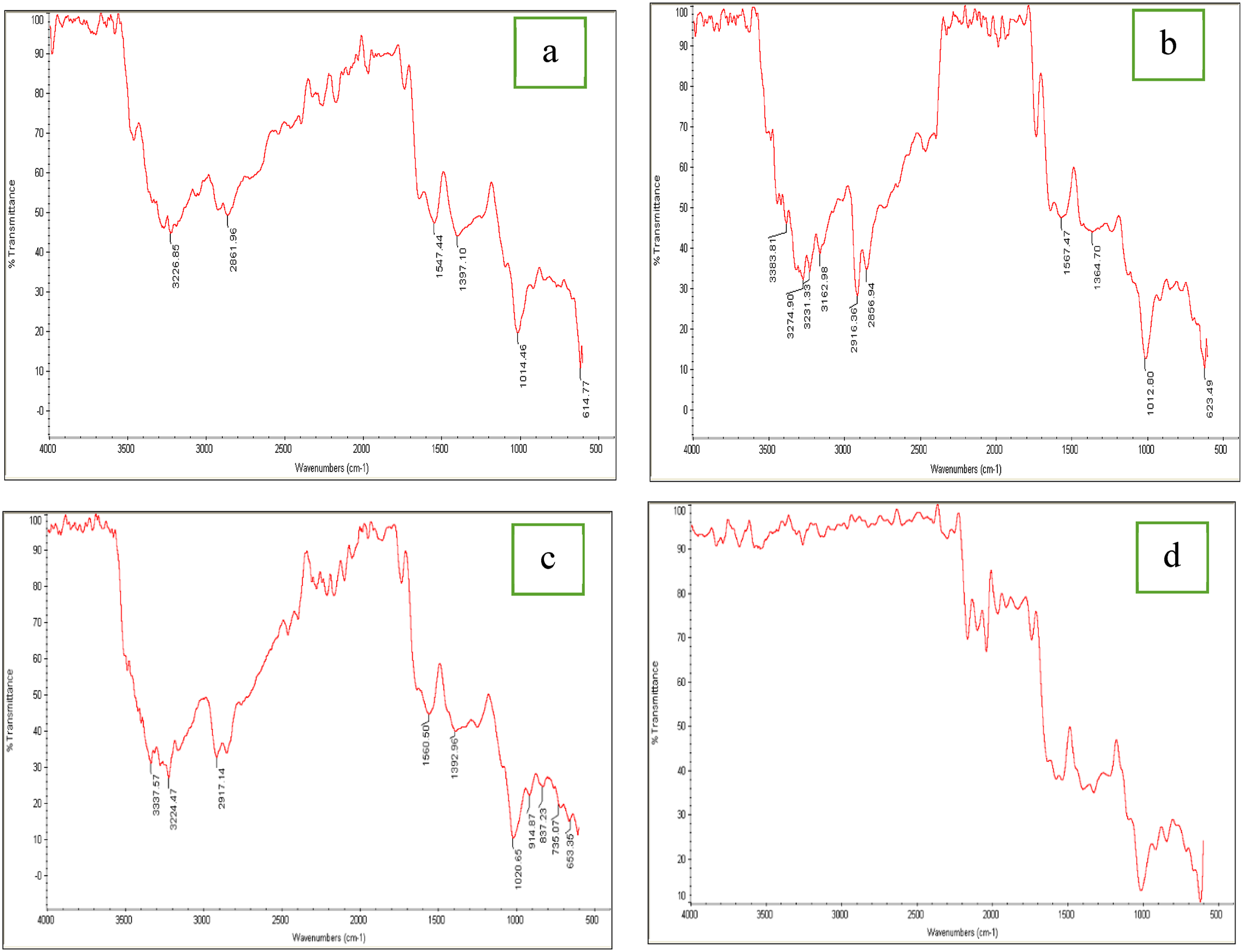
FTIR
FTIR spectra of the optimized bionanocomposite film samples are presented in Figure 5. The absorption pattern of the samples is reported in the range of 4000 to 500 cm−1. The FTIR spectrum of the control film sample shown in Figure 5(a) indicates the presence of asymmetric stretching vibrations of the NH2 group in the protein structure at approximately 3226.85 cm−1.
60
The peak at approximately 2861.96 cm−1 corresponds to CH2 stretching vibrations in the pectin chain,
61
and the peaks at approximately 1547.44 cm−1 and 1397.10 cm−1 represent the bending vibration of N-H and the stretching vibration of C-N in the amide groups of the cottonseed protein isolate structure.
62
The measured peak in the range of 1014.46 cm−163 is attributed to the stretching vibration of C-O, whereas the peak at 614.77 cm−164 is attributed to the stretching vibrations of the polysaccharides C-O-C. Spectrum 5-b shows that adding 15%
XRD Analysis
The XRD spectra of the optimized bionanocomposite film samples are presented in Figure 6. According to the results, Figure 6(a) shows a broad peak at a 2θ angle of approximately 20.10° in the control sample, indicative of the semi-crystalline structure of the film based on cottonseed protein isolate and pectin; And also the crystallinity index for the pure film sample was equal to 15.2%. Upon the addition of 15% XRD of active films: (a) (pure film), (b) (film containing 15% 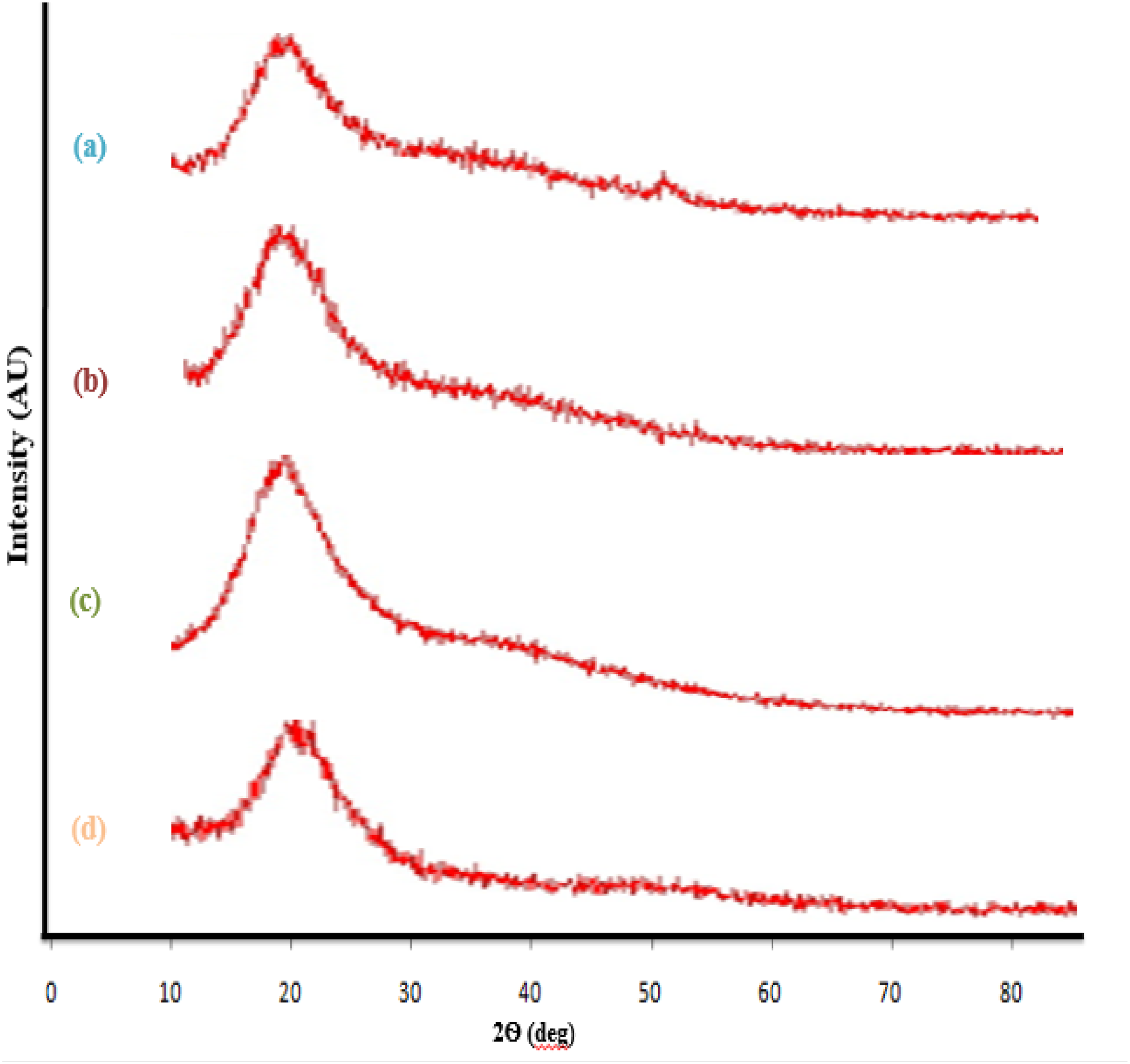
DSC Thermograms
Figure 7 shows the DSC thermograms of the optimized bionanocomposite film samples. The control sample (Figure 7(a)) showed two endothermic peaks: the glass transition temperature (Tg) is represented by the peak at 42.14°C, and the melting temperature (Tm) of the cottonseed protein isolate/pectin is defined by the minor peak at 73.74°C. The spectrums of the pectin, DSC of active films: (a) (pure film), (b) (film containing 15% 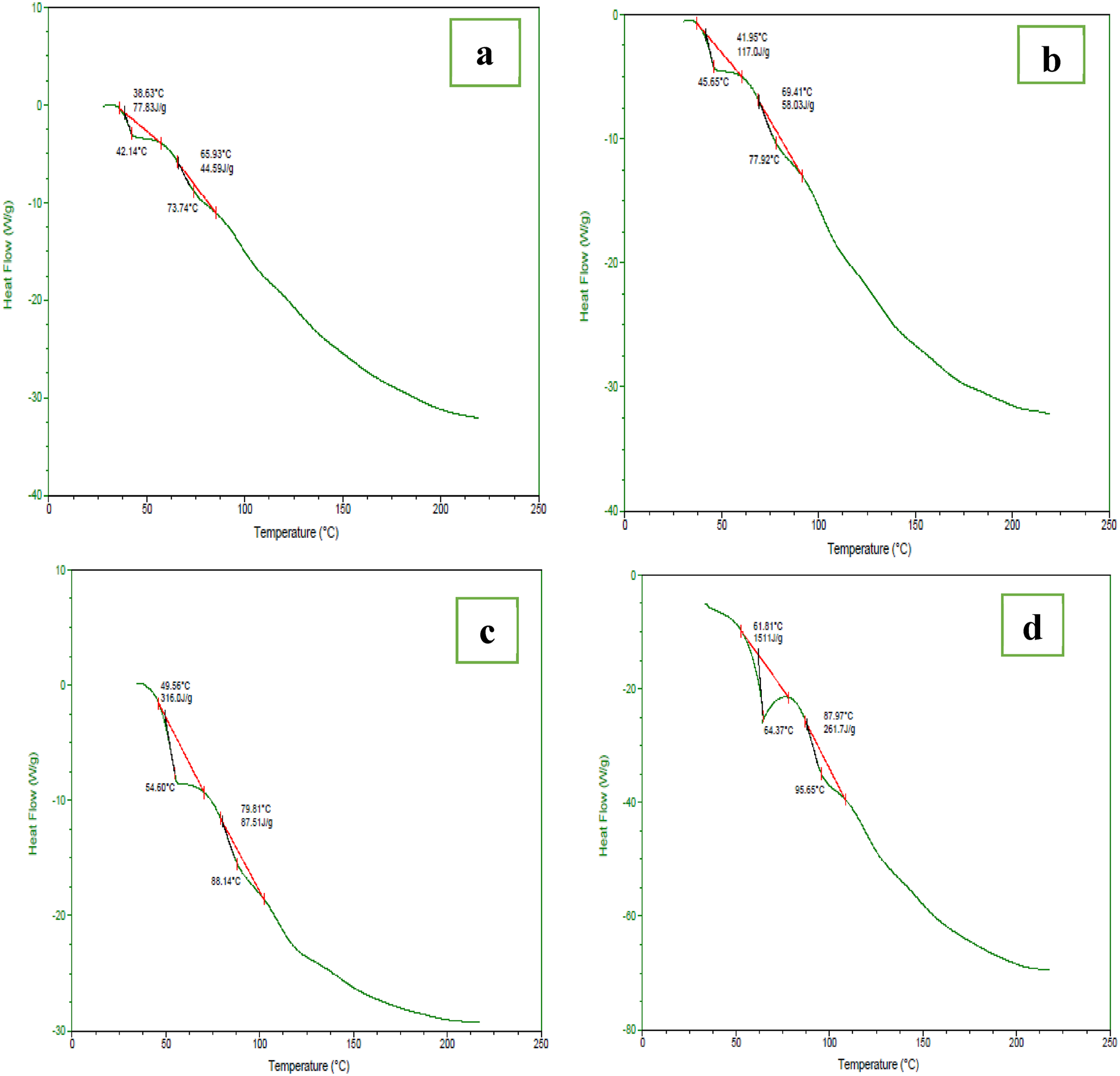
Similar results were reported by Jridi et al. (2019) with the addition of blood orange peel extract in a gelatin-based edible film. 67 Additionally, Oleyaei et al. (2016) also noted that the addition of TiO2 nanoparticles in a starch-based edible film increased the glass transition and melting temperatures. 68
Mechanical Properties
Regression coefficients of colorimetric parameters of nanocomposite film.
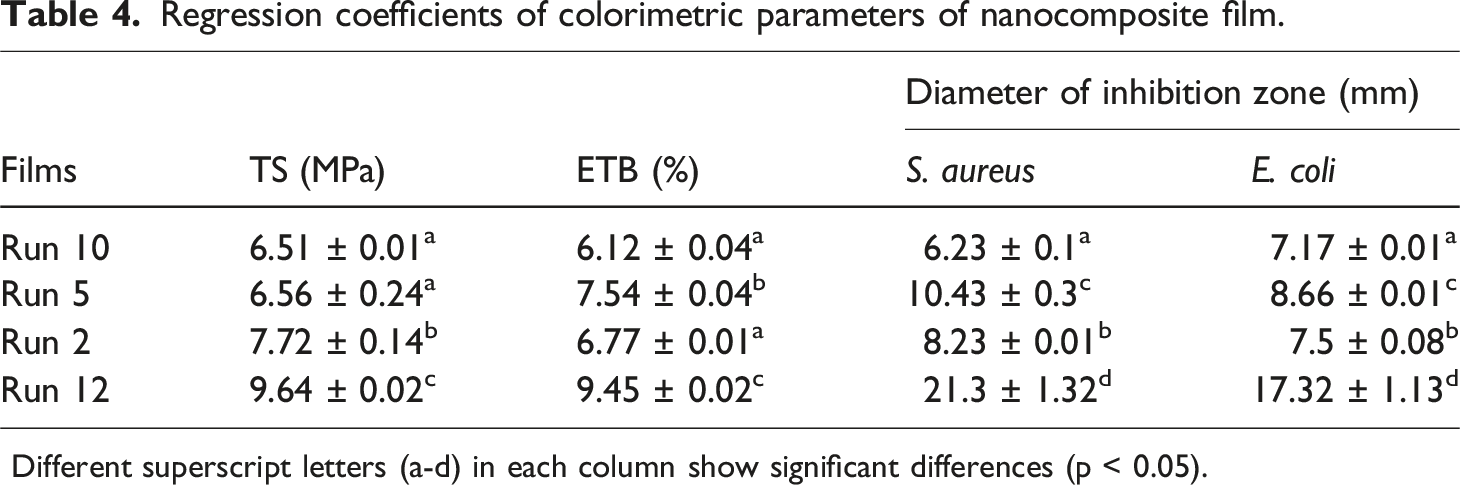
Different superscript letters (a-d) in each column show significant differences (p < 0.05).
Antimicrobial Properties
Table 4 shows the antibacterial efficiency of the optimized bionanocomposite film samples against
Conclusion
Improving food products’ quality and shelf life while being stored has become a significant concern in recent years. Researchers and business experts use various techniques to increase the quality and durability of food products. Using edible films and coatings for packaging that include bioactive substances like antioxidants and antimicrobials is a creative method. In light of this, this work investigated the physicochemical and structural characteristics of bionanocomposite films based on pectin and cottonseed protein isolate combined with chitosan nanoparticles (0, 2, 4% w/v) and
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
CRediT Authorship Contribution Statement
Data Availability Statement
Data will be made available on request.
