Abstract
This study investigates the impact of salt spray aging (per ASTM B117) on the mechanical, thermal, and morphological properties of aluminum-doped polylactic acid (PLA) composites fabricated using filament fed material extrusion-based additive manufacturing (MEX). Specimens containing 25 wt% Al were subjected to accelerated aging for 2, 4, and 8 weeks. The results indicated a dramatic degradation in mechanical properties after 8 weeks, with the ultimate tensile strength plummeting by approximately 88% (from 30.04 MPa to 3.66 MPa) and elongation at break dropping to 1.8%. Thermal analysis revealed a decrease in glass transition temperature (Tg) from 68.45°C to 67.25°C after 8 weeks, confirming polymer chain scission due to hydrolysis. The onset of thermal degradation also shifted to a lower temperature. Morphological examination via Scanning Electron Microscopy (SEM) unveiled severe interfacial debonding between the PLA matrix and aluminum particles, along with the formation of cracks and salt crystals, providing direct evidence of synergistic hydrolytic and corrosive degradation. In conclusion, the susceptible PLA/Al interface causes catastrophic failure in saline environments, necessitating strategies like coupling agents to ensure durability.
Keywords
Introduction
The lightness, durability, and low cost of plastics have paved the way for their use in a wide range of areas, from daily life to industry. 1 While the widespread use of plastics has become an indispensable part of modern life, their negative impacts on environmental pollution and the global climate crisis have become increasingly evident.2,3 The use of traditional petrochemical-derived polymers causes serious environmental problems. Due to their long degradation times and recycling challenges, these plastics contribute to global environmental pollution and increase the carbon footprint. 4 Reducing the carbon footprint and regulating sustainability concepts with some legal norms increases the demand for environmentally friendly products in material choices day by day. While plastic waste remains in nature for many years, damaging ecosystems, high carbon emissions in production processes trigger global warming. 5 In this context, the development and use of environmentally friendly polymers in line with sustainability principles has gained great importance. 6 Recyclable polymers, produced from renewable resources and having biodegradable properties, stand out for their potential to reduce carbon footprint and facilitate waste management.6–8 Therefore, the development and use of biodegradable and sustainable polymers are becoming increasingly important.
The environmental damage caused by traditional polymers and the difficulties in recycling processes have increased the demand for environmentally friendly alternatives. 5 At this point, PLA thermoplastic polymers, which are produced from renewable resources and stand out with their biodegradable properties, stand out. PLA is produced from natural raw materials such as corn starch or sugar cane and is fully biodegradable under favorable conditions. 9 PLA is used in many applications due to its biodegradability and low cost among thermoplastics. On the other hand, its biodegradability affects its thermal and mechanical properties, and PLA’s low mechanical properties limit its application.10,11 To overcome these limitations, it is possible to prepare composites by adding various fillers to the PLA polymer, including metal powders, 12 minerals, 13 and natural fibers. 14 Numerous studies have been carried out on the incorporation of suitable additives into PLA polymers, improving the chemical, thermal, and mechanical properties of PLA composites. For example, Kottasamy et al. investigated the mechanical properties of different copper composition variations in copper-reinforced PLA composites. The highest ultimate tensile strength value was observed in specimens containing 25% copper reinforcement. 15 Oksiuta et al. also investigated the mechanical and thermal properties of polylactic acid-based composites reinforced with iron and magnesium powders. Compared to pure PLA, iron powder reinforcement was found to improve tensile strength, while magnesium powder slightly improved ductility. 16 There are many studies in the literature on the reinforcement of PLA with metal powders to improve its mechanical properties and increase its functionality.17–22 The studies carried out to improve PLA’s special properties, such as thermal resistance, mechanical strength, and conductivity, to enable it to address a wider range of applications, pave the way for composites that can offer both environmentally friendly and high-performance solutions. Furthermore, studies on metal-filled PLA composites produced by additive manufacturing have demonstrated that optimizing filler type and processing parameters is critically important for achieving desired mechanical and thermal properties. 23 However, the degradation behavior of biodegradable polymers under environmental conditions plays a pivotal role in determining their long-term performance and application potential. 24 Consequently, the development of sustainable composite materials presents a promising approach to mitigate the environmental impact of traditional plastics. 25 In this sense, PLA matrix composites play a key role in leading the polymer industry toward a sustainable future. PLA material has a wide range of applications for material extrusion-based additive manufacturing (MEX), which is one of the innovative production methods, and has become widely used in the shaping of polymers. 26 PLA is a naturally insulating material with a low melting temperature. 27 PLA comes to the forefront in MEX production due to its low cost, low-temperature forming capability, and being the most widely used additive manufacturing method. In addition to various reinforcements, MEX production parameters also affect the mechanical properties of PLA, especially thermal resistance, conductivity, and surface quality.27,28 Zhang et al. investigated the mechanical properties of PLA and aluminum fiber-reinforced composite specimens with MEX. The mechanical and thermal properties of specimens produced with MEX at different raster angles were compared. It was stated that the raster angle directly affects the mechanical properties. 29 In another study, Chong et al. investigated the antibacterial properties of zinc reinforcements to PLA, as well as the mechanical properties. 21 Vidakis et al. also investigated the antibacterial properties of Silicon Dioxide (SiO2) nanoparticles reinforcement in PLA matrix. 30 Petausis et al. investigated the mechanical properties of Alumina (Al2O3) nanocomposites with a PLA matrix produced with MEX and reported that AL2O3 reinforcement positively affected the mechanical properties. 31 While the powder form of aluminum is used for different purposes in industrial applications, it provides benefits in areas such as corrosion resistance, lightness, brightness, and machinability. It is used in composite forms to produce lightweight and durable materials. It is used in additive manufacturing technologies such as sintering and melt bed fusion (SLM and DMLS). Aluminum powder has a wide range of uses with its lightness, durability, thermal, and electrical conductivity. 32 Hasanzadeh et al. also investigated the effect of manufacturing parameters on the mechanical properties of Aluminum-reinforced Composites produced with MEX and compared with Pure PLA. It was reported that aluminum reinforcement affects tensile strength, and printing speed affects elongation at break. 18
Further studies shows that Menezes et al. developed an accelerated-aging model for polylactic acid specimens immersed in 17°C artificial seawater. Their diffusion and mechanical data predicted that PLA loses just 0.5% ductility at ∼5166 h (≈30.8 weeks) and experiences a ∼10 MPa drop in yield strength after ∼2084 h (≈12.4 weeks), indicating moderate mechanical deterioration. 33 Tsuji & Suzuyoshi investigated solution-cast poly(L-lactide) films in controlled static seawater at 25°C and found that biodegradation was insignificant even after 10 weeks; the initial crystallinity had no noticeable influence, and tensile properties remained essentially unchanged. 34 Tejedor et al. examined 3D-printed PLA aged in salt water at 45°C for 45 days and showed that annealing at 100°C for 70 min increased crystallinity and reduced void content, which in turn diminished water adsorption and surface defects, thereby mitigating losses in flexural and dynamic mechanical properties. These findings demonstrate that neat PLA generally degrades slowly in saline environments and that thermal post-processing can enhance its resistance. 35 Further literature shows that neat polylactide (PLA) does degrade in saline or marine environments. Deroiné et al. accelerated PLA aging by immersing injection-molded specimens in seawater at 25°C and 40°C. After 6 months at 40°C the stress at break dropped by about 70% in seawater (compared with 85 % in distilled water), whereas at 25°C the strain at break increased from 3.4% to 6.9%; the authors also noted a slight increase in Young’s modulus due to recrystallization. 36 Duigou et al. studied unreinforced poly(l-lactide) aged for 3 months in natural seawater and reported that the tensile strength fell from 60.5 MPa to 46.6 MPa at 20°C (≈23% loss) and to 41.9 MPa at 40°C (≈31% loss), while the Young’s modulus decreased by ∼9% and the failure strain dropped by ∼48% at 40°C. Bao et al. found that the maximum tensile stress of PLA/PHB plastics decreased by ∼38% after 4 weeks in static seawater (from 21.66 MPa to 13.36 MPa), 37 and Krasowska & Heimowska reported a slight decline in tensile strength to 48.6 MPa after up to 16 months in the Baltic Sea. 38
PLA specimens produced with MEX have a young modulus close to bone and the ability to make complex shapes. Aluminum-reinforced PLA composites (PLA/Al) also have positive effects on mechanical properties.29,31 However, the brittleness and biodegradability of PLA sometimes negatively affect its application areas. In this study, the effects of degradation processes on the mechanical and thermal properties of PLA/Al specimens produced by the MEX method, since the matrix material is PLA, were investigated to examine the effects of these negativities. In this context, the specimens were kept in a controlled, conditioned environment for different periods, and then the changes in their mechanical and thermal properties were examined, and the damaged surfaces of the specimens were analyzed.
Materials and methods
Materials
The Esun brand eAL-fill filament with a density of 1.48 g/cm3, 52°C Heat Distortion Temperature (0.45 MPa), and 8 g/10 min Melt Flow Index values was used in the study. The filament with PLA matrix contains powder aluminum (Al) reinforcement. The sizes of the Al powders contained in the filament are visualized by Scanning Electron Microscope, and the amount of Al powder by weight was determined by Thermogravimetric analysis (TGA). According to the analysis, it was determined that the grain sizes were in the 3–15 µm range and contained 25 wt% powder aluminum.
Production
MEX producing parameters.

During the production process, cabinet temperature was measured with Fischer Scientific brand 06-664-11 model Traceable Digital Thermometer. The minimum and maximum values are given in Table 1. Five sets of each of the specimen groups, which were kept in environmental conditioning for three different periods, were produced. Some of the specimens produced are shown in Figure 1. Produced specimens with MEX.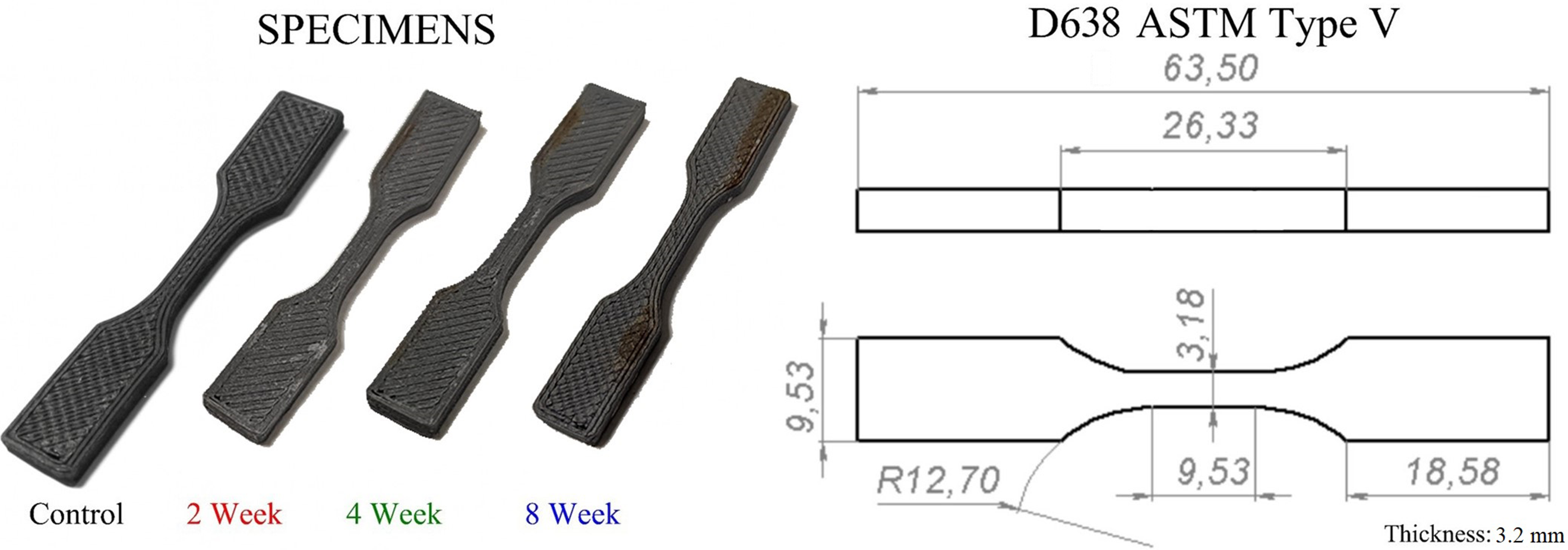
The production parameters listed in Table 1 are those recommended by the material manufacturer. Similar production parameters are also recommended for PLA in the literature. 26 The 0° raster angle was specifically chosen to maximize tensile strength along the loading axis. 29 For the salt spray aging, the conditions (5% NaCl, 2/4/8 weeks) were selected according to ASTM B117 to effectively simulate aggressive marine environments and capture progressive degradation stages.
Environmental degradation
Accelerated aging tests were performed under ASTM B117 and ISO 9227 standards in the CC1000ip model test chamber manufactured by Ascott. The primary reason for selecting this particular type of accelerated aging test device is its ability to simulate actual outdoor conditions by subjecting the specimens to repetitive cycles, thereby providing environmental conditions that closely mirror those of real-world exposure.
The test apparatus has a chamber volume of 1000 L and a saltwater reservoir capacity of 115 L. It operates at an air pressure of 4–6 bar, and the salt spray rate can be adjusted to between 0.5 and 3.0 mL per hour over an 80 cm2 area. During the accelerated aging process, the specimens were placed in the chamber in such a way that all surfaces were uniformly exposed to moisture and salt. The temperature of the test chamber was kept at 35°C ± 2°C as specified in the standard.
The saltwater solution used in the test contained 5% NaCl. To ensure that the salt water reached the specimens in the form of uniform droplets, the solution was sprayed at an angle between 150° and 300°. This approach facilitated the even distribution of salt and moisture across each specimen’s surface, thus allowing the accelerated aging test to more closely approximate realistic environmental conditions. The specimens were divided into three groups and subjected to accelerated aging for 2 weeks, 4 weeks, and 8 weeks, respectively.
Characterization
Fourier Transform Infrared (FT-IR) spectroscopy was conducted to identify various functional groups in PLA. The FT-IR spectrum was recorded within the 4000 to 375 cm−1 range. A Bruker Alpha-P FT-IR spectrometer, integrated with OPUS software, was utilized for the analysis. All measurements were carried out at room temperature.
Differential Scanning Calorimetry (DSC) analysis of the materials was conducted using a Perkin Elmer Jade DSC instrument equipped with PYRS software. The measurements were carried out with a nitrogen reference material, a low range of 320 mW, and a heating rate of 10°C/min under a nitrogen gas pressure of 3 bar. The temperature range for the analysis was set between 40°C and 200°C.
TGA measurements of the materials were performed using a Perkin Elmer TGA instrument with PYRS software installed, in a nitrogen environment, at a temperature range of 25°C to 500°C with a 10°C/min increase.
Mechanical testing
Tensile tests of the specimens produced and conditioned under environmental conditions for different periods were carried out according to the ASTM D638 standard on the MTS Systems Corp. brand Eden Prairie, MN universal testing machine at a test speed of 1 mm/min. All specimens were subjected to force until fracture, and the ultimate tensile strength and elongation at break values were recorded.
Morphological analysis
SEM analysis was carried out using a Zeiss Sigma560 Field Emission Scanning Electron Microscope (FESEM) equipped with an SE2 secondary electron detector to investigate the surface morphology of the specimens in detail. The 3D-printed specimens were cut into rectangular prisms with dimensions of 20 × 20 × 30 mm to ensure compatibility with the SEM stage and to facilitate effective imaging.
Before imaging, the specimens were affixed to aluminum stubs using conductive carbon tape to minimize charging effects. Subsequently, a 5 nm-thick gold layer was applied to the specimen surfaces via sputter coating to enhance conductivity and improve image resolution. The prepared specimens were then placed in the SEM vacuum chamber and allowed to stabilize under vacuum conditions for 1 hour to eliminate any residual moisture or contaminants.
SEM imaging was performed at varying magnifications, as indicated by the scale bars in the respective micrographs, using an accelerating voltage (EHT) in the range of 3–5 kV. Particular attention was given to identifying and analyzing microstructural features such as crack formations induced during tensile testing.
Micro-CT (computed tomography) analysis
Micro-computed tomography (µCT) was employed to non-destructively characterise the internal architecture of the PLA/Al specimens and to quantify changes in porosity and microstructural integrity following environmental ageing. The scans were performed using a high-resolution X-ray system (XT H 225 ST 2×, Nikon Metrology NV, UK) equipped with a micro-focus X-ray source and a flat-panel detector. This configuration enables the acquisition of projection images with sufficient geometric magnification to resolve micron-scale internal features.
To minimise reconstruction artefacts and achieve a voxel size of approximately 9.7 µm, each specimen was scanned under constant operating conditions of 150 kV tube voltage and 47 µA tube current. An integration time of 354 ms was applied for each projection, and eight frames were averaged to enhance the signal-to-noise ratio. A total of 4476 projections were collected over a 360° rotation. The geometric magnification factor of 15.44× resulted from a source-to-object distance (FOD) of 72.61 mm and a source-to-detector distance (FID) of 1121.20 mm. All datasets were reconstructed using CT Agent XT 6.12.1 software.
Reconstructed volumes were segmented to extract both the specimen bounding-box volume and the total µCT-derived solid volume. Porosity was calculated as: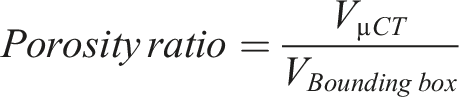
Results and discussion
Chemical characterization
FT-IR Spectra
The FT-IR spectra presented in Figure 2 provide an in-depth analysis of the chemical structure and stability of Al-doped PLA filament specimens over different exposure periods: control, 2 weeks (2W), 4 weeks (4W), and 8 weeks (8W). Each spectrum exhibits characteristic absorption bands associated with functional groups integral to the PLA polymer matrix, notably Al-O, C=O, C-O-C, C-H, and -CH2. These bands are essential for assessing structural evolution or degradation within the doped PLA material over time. FT-IR spectra of Al-doped C, 2W, 4W, and 8W PLA filament specimens.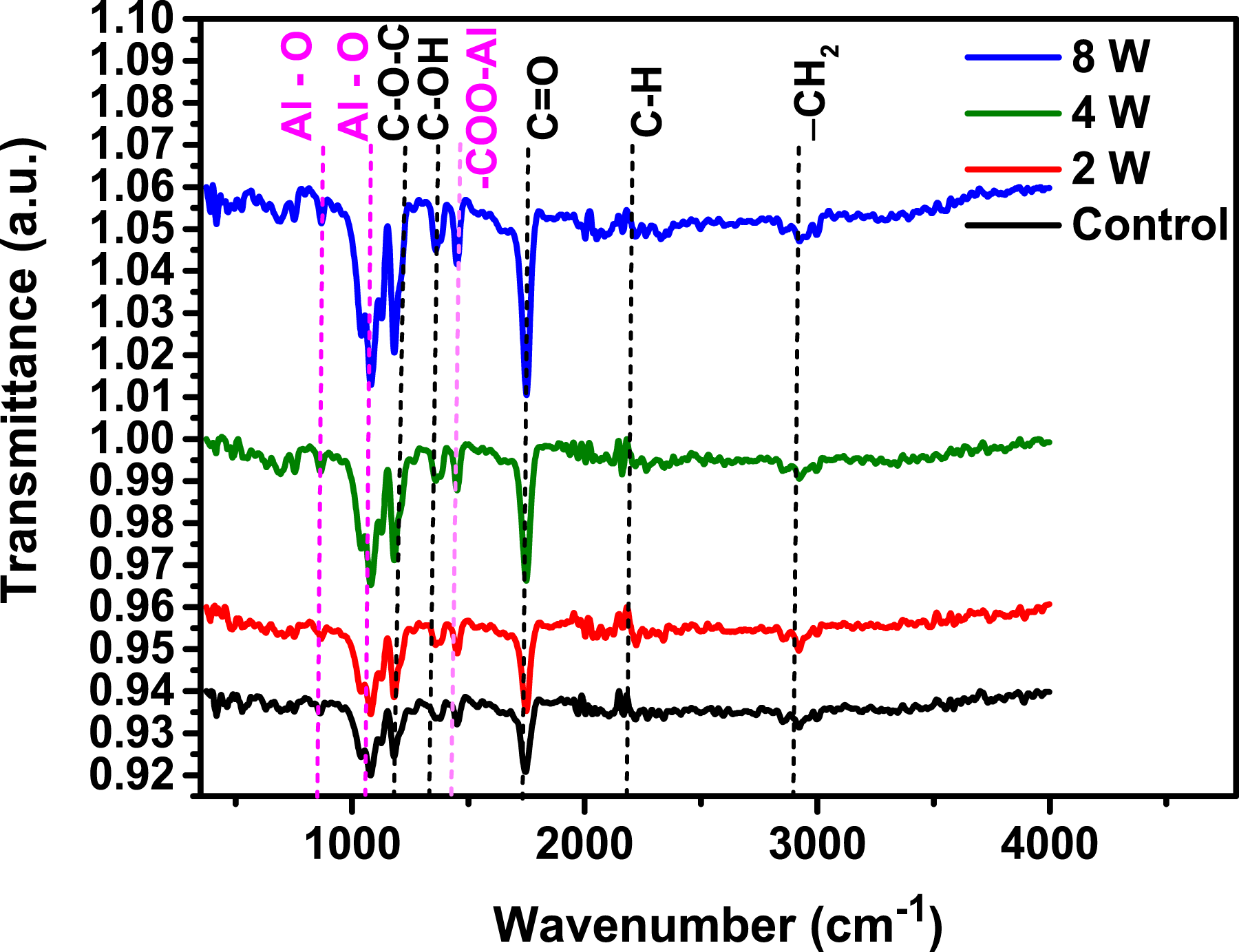
Asymmetric and symmetric vibrations of -CH2 are seen at 2851 cm−1 and 2927 cm−1.39–41 The peak appearing at 1450 cm−1 is attributed to the absorption of the ester group in –COO–Al. 41 The Al-O peaks at 740 cm−1 and 1063 cm−1 are attributed to the peak values where -O is grafted onto the Al doping in PLA. 42 Bands of PLA bond vibrations were observed in all spectra, including C-O-C stretching at 1182 cm−1, C-O-H at 1323 cm−1, and C=O asymmetric bending at 1725 cm−1.42,43 The intensity of these bonds increased with the week’s number.
Significant observations can be drawn from the transmittance variations across the different specimens. The control specimen exhibits a baseline spectral profile, reflecting the unaltered chemical structure of the Al-doped PLA before any temporal exposure. As we progress to the 2W and 4W specimens, slight shifts in the intensity of the C=O42,43 and C-O-C 42 absorption bands emerge, suggesting early-stage interactions within the polymer matrix. This indicates that Al doping may initiate subtle chemical modifications or interactions that could enhance or destabilize specific molecular bonds in the PLA.
By the 8-week mark (8W), these spectral changes become more pronounced. The increase in transmittance in key regions, particularly near the Al-O 40 and C=O bands suggest potential degradation or rearrangement of the polymer structure. This phenomenon may imply oxidative degradation, hydrolysis, or other forms of polymer breakdown that Al doping may influence over time. The stability or evolution of the C-H39,41 and -CH2 39 bands further support insights into the resilience of the PLA backbone in prolonged conditions.
In conclusion, this time-dependent FT-IR analysis provides valuable insights into the durability and chemical stability of Al-doped PLA filaments. The observed spectral shifts indicate that while Al doping may initially stabilize certain functional groups, extended exposure leads to structural changes that could impact the polymer’s mechanical and chemical properties. These findings are critical for applications that rely on the longevity and stability of PLA materials under environmental or operational stresses, highlighting the need for further optimization in doping and stabilization strategies for sustainable, long-term use of polymers.
Thermal characterization
DSC spectra
Tg given in Figure 3(a) are as follows: control specimen 68.45°C, 2W aged specimen 69.98°C, 4W aged specimen 70.85°C, and 8W aged specimen 67.25°C. The Tg values indicate an initial increase with 2W and 4W aging durations, followed by a decline at 8W. DSC of aged PLA doped with Al nano powder, (a) glass transition temperature (Tg) value of the specimens, and (b) fusion enthalpy (ΔH) of the specimens.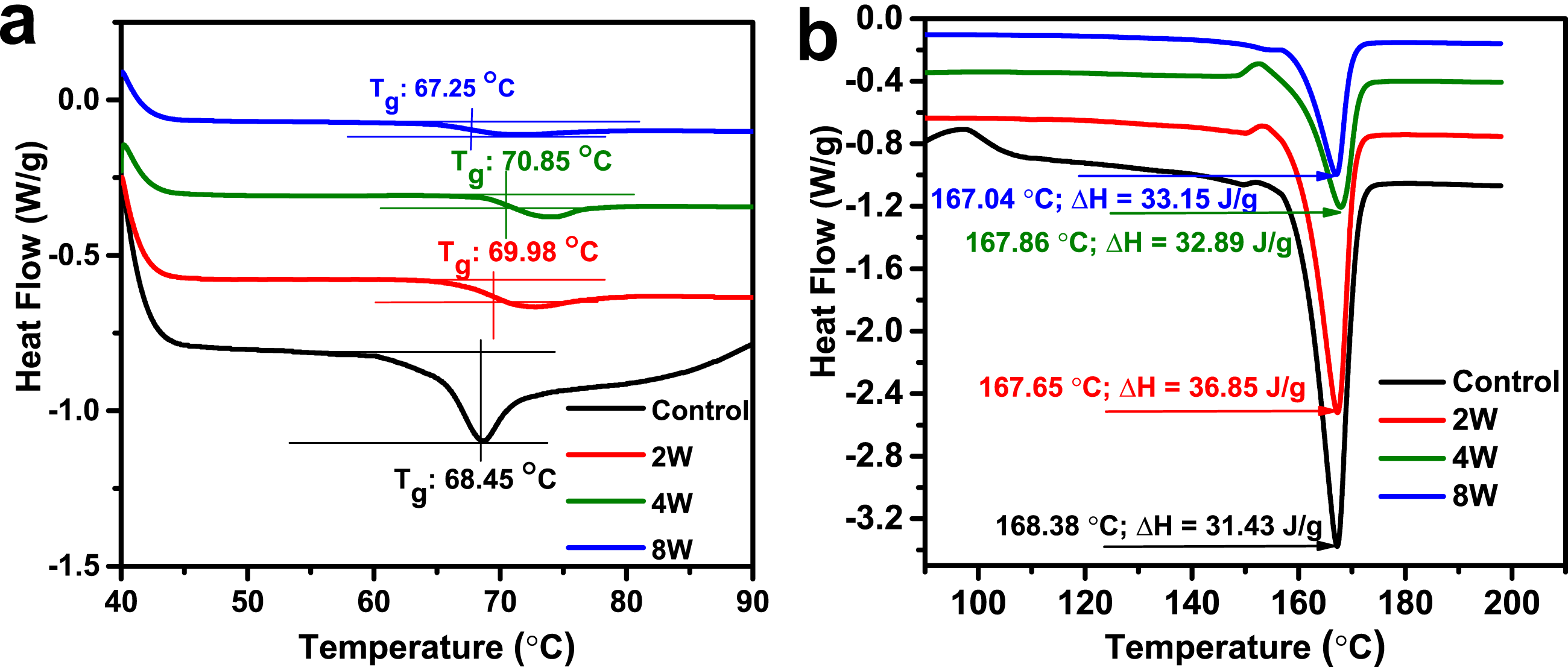
The increase in Tg for the 2W and 4W aged specimens is attributed to structural relaxation in the amorphous regions of PLA during the aging process. This phenomenon, known as physical aging, reduces free volume as polymer chains rearrange into a more stable configuration. This stabilization restricts chain mobility, thereby increasing Tg. Similar trends have been documented in aging studies of PLA, where thermal stability improves with short-term aging. The decrease in Tg for the 8W specimen is attributed to hydrolytic degradation. The prolonged aging process, especially in a humid or controlled environment, likely facilitated the hydrolysis of ester bonds in PLA, leading to chain scission and a reduction in molecular weight. This degradation disrupts the compact arrangement of polymer chains, lowering the glass transition temperature. This behavior is consistent with reports in the literature, where hydrolytic degradation of PLA composites 44 reduces Tg due to the breakdown of polymer chains.
The melting temperatures (Tm) and fusion enthalpy (ΔH) values given in Figure 3(b) are Control Tm = 167.04°C, ΔH = 33.15 J/g; 2W aged specimen Tm = 167.65°C, ΔH = 36.85 J/g; 4W aged specimen: Tm = 167.86°C, ΔH = 32.89 J/g; 8W aged specimen: Tm = 168.38°C, ΔH = 31.43 J/g.
The Tm shows a slight increase across the specimens as the aging period increases. This rise in Tm is attributed to secondary crystallization during the aging process, where the polymer chains rearrange to form more thermodynamically stable crystalline regions. The increase in crystalline order enhances the thermal stability of the material.
The ΔH value represents the heat required to melt the crystalline regions and reflects the degree of crystallinity. The ΔH value for the 2W aged specimen is higher than the control, indicating an increase in the degree of crystallinity due to secondary crystallization. For the 4W and 8W aged specimens, the ΔH values decrease progressively. This reduction is attributed to the competing effects of crystallization and hydrolytic degradation. 45 The hydrolysis of ester bonds at longer aging durations reduces the ability of PLA chains to form well-ordered crystalline regions, thereby lowering ΔH.
Promotes physical aging and secondary crystallization, resulting in higher Tg, Tm, and ΔH values. 46 These effects are associated with the stabilization and ordering of polymer chains. Hydrolytic degradation becomes dominant, leading to chain scission and a decline in both Tg and ΔH.45,47 While Tm continues to increase due to the stability of the remaining crystalline regions, the overall crystallinity decreases.
The findings of this study are consistent with research in the literature regarding the thermal aging behavior of PLA composites. Studies by Elsawy et al. have highlighted PLA’s susceptibility to hydrolytic degradation in humid environments, leading to reduced molecular weight and thermal properties. 45 The decrease in Tg and ΔH for the 8W specimen aligns with these observations.45,46,48 Other research has demonstrated that physical aging can induce secondary crystallization in PLA, resulting in higher melting temperatures. This phenomenon is reflected in the increased Tm values observed in this study.49,50
The aging process significantly influences the thermal properties of PLA/aluminum powder composites. Short-term aging enhances thermal stability through physical aging and secondary crystallization. However, prolonged aging introduces hydrolytic degradation, which compromises the thermal and crystalline properties of the material. These results provide valuable insights into designing and applying PLA-based composites for environments where aging effects are a critical consideration.
TGA spectra
TGA experiments were carried out under a nitrogen atmosphere, between room temperature and 500°C, and at 10°C/min conditions. The TGA plot shown in Figure 4 (a) provides a comparative assessment of thermal stability for aluminum-doped PLA filament specimens at different exposure durations: control (black), 2 W (green), 4 W (red), and 8 W (blue). The x-axis represents temperature in °C, while the y-axis shows the percentage weight loss, a direct indicator of material decomposition. TGA spectra of Al-doped C, 2W, 4W, and 8W PLA filament specimens (a) weight loss and (b) DTG.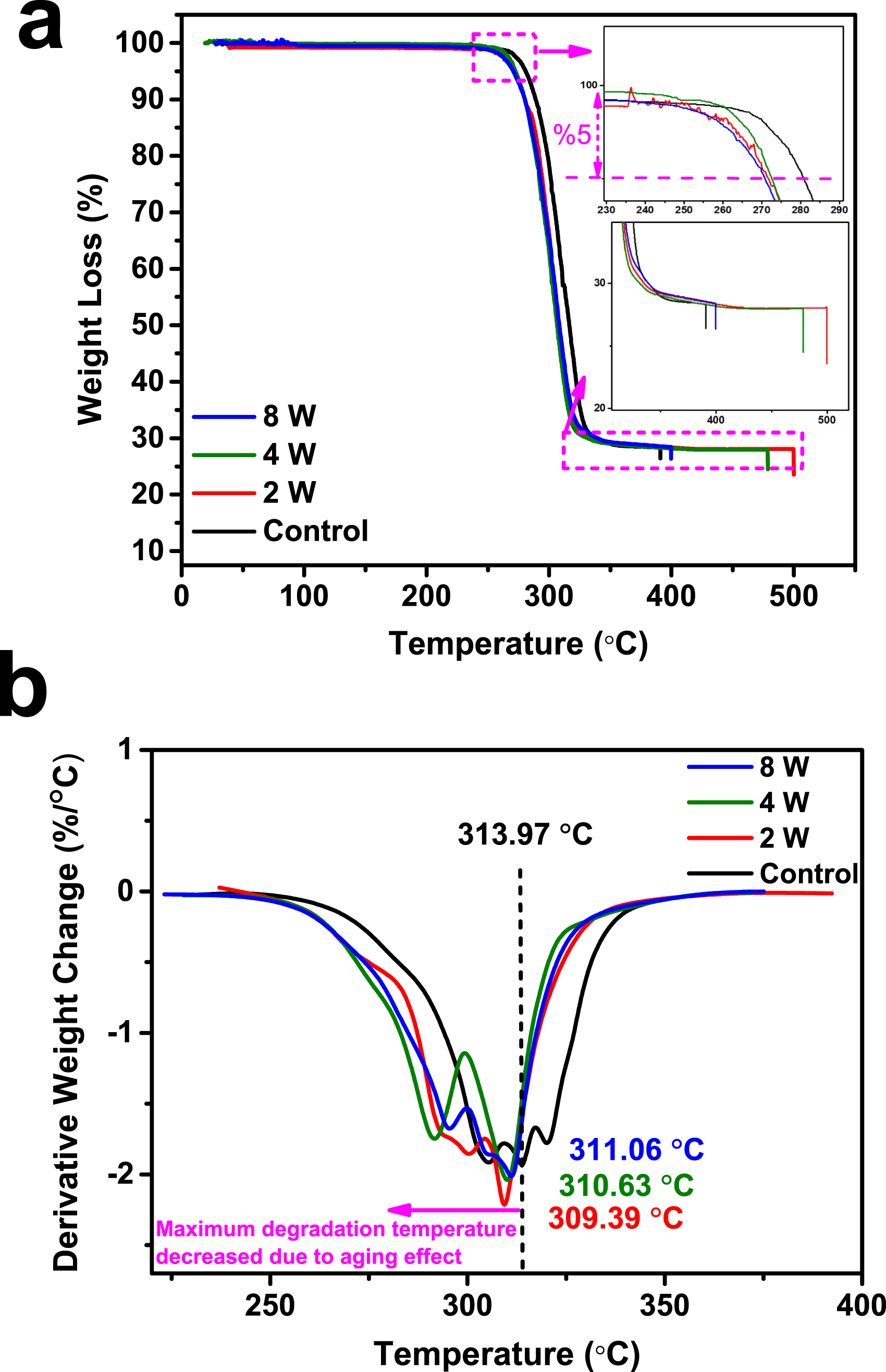
The TGA curves reveal a characteristic single-step degradation profile, typical for PLA, which suggests that the primary weight loss corresponds to the breakdown of the polymer matrix. All specimens show minimal weight loss up to approximately 250°C, indicating thermal stability up to this temperature. However, slight differences in the decomposition onset temperature are noticeable between the control and the exposed specimens, as highlighted in the inset magnified regions of the graph.
The onset of decomposition, often marked by a 5% weight loss threshold, appears to vary slightly among the specimens. The control specimen begins to degrade at a slightly higher temperature compared to the 2W, 4W, and 8W specimens, implying that prolonged exposure, even with aluminum doping, may gradually reduce the thermal stability of PLA. This minor shift suggests that the aluminum-doped PLA undergoes subtle structural changes over time, potentially due to environmental interactions or degradation pathways 31 that reduce the onset temperature as exposure lengthens. In the major decomposition region (between 300°C and 400°C), there are subtle differences in the weight loss profiles among the specimens.
In particular, specimen 8W shows a slightly faster degradation rate than the control, which has been attributed to cumulative structural degradation or catalytic effects of aluminum 51 that becomes more pronounced with prolonged exposure. The final residual weight remaining beyond 450°C is similar across all specimens, suggesting that the primary decomposition products remain largely consistent regardless of the exposure period.
In summary, this TGA analysis reveals that while aluminum doping offers initial thermal stability to PLA, extended exposure slightly lowers the onset temperature of degradation, indicating a reduction in thermal stability over time. This time-dependent decline is critical to consider for applications requiring prolonged thermal resistance, as even minor degradative effects can accumulate and impact the performance and longevity of aluminum-doped PLA materials in real-world environments. Further optimization of the aluminum content 52 and protective strategies against environmental factors 53 may help mitigate these long-term effects, enhancing the durability of PLA for industrial and sustainable applications.
In Figure 4(b), the maximum degradation temperature, indicated by the peak of each curve, decreases as aging time increases. For the control specimen, this peak degradation temperature is at 313.97°C. After aging for 2 weeks, it decreases to 311.06°C, then to 310.63°C after 4 weeks, and further down to 309.39°C after 8 weeks. The progressive decrease in the maximum degradation temperature with aging suggests that thermal stability decreases over time. This indicates that as the Al-doped PLA filament ages, it becomes more susceptible to thermal degradation, 53 likely due to changes in the filament’s molecular structure or polymer chain scission.
A higher peak implies a more rapid decomposition at a specific temperature. The gradual decline in peak temperature with aging implies that aging accelerates degradation, leading to lower thermal stability. Aging might lead to changes in the PLA matrix, such as oxidation or hydrolysis, which can reduce the molecular weight 54 and make the material more susceptible to thermal degradation at lower temperatures. This aligns with findings in polymer degradation studies, where aging (especially in the presence of oxygen or moisture) lowers thermal stability.55–57
In conclusion, this graph suggests that the Al-doped PLA filament undergoes a notable reduction in thermal stability with increased aging, as seen in the decline of the maximum degradation temperature. This is consistent with observations in polymer science, where aging often leads to reduced structural integrity and an earlier onset of degradation under thermal stress.
Mechanical characterization
P-values obtained from the Shapiro–Wilk normality test.

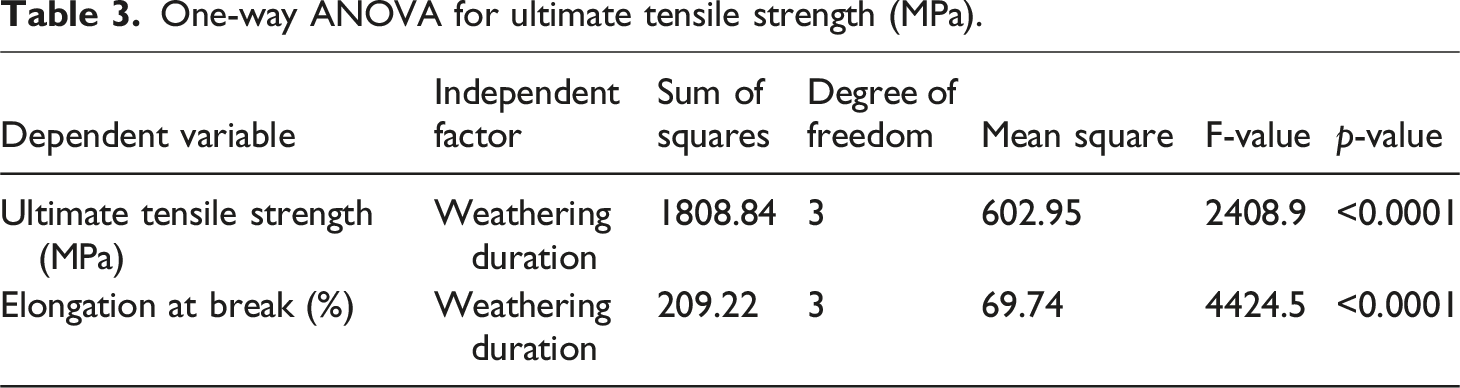
Next, Levene’s homoscedasticity test was applied. Levene’s test assesses the equality of variances (homoscedasticity) among groups, a crucial assumption for ANOVA. A non-significant p-value (p > 0.05) indicates equal variances, supporting the assumption, whereas a significant p-value (p < 0.05) indicates unequal variances, violating the assumption. 59 The p-values were found to be 0.1764 and 0.1641 for ultimate tensile strength and elongation at break, respectively. Since both p-values are greater than 0.05, Levene’s test indicates that the variances among the groups (Control, 2-week, 4-week, and 8-week) are not significantly different, confirming that the homoscedasticity assumption holds. Therefore, there is no violation of the equal variance assumption.
One-way ANOVA for ultimate tensile strength (MPa).
Tukey’s HSD test for dependent variables.
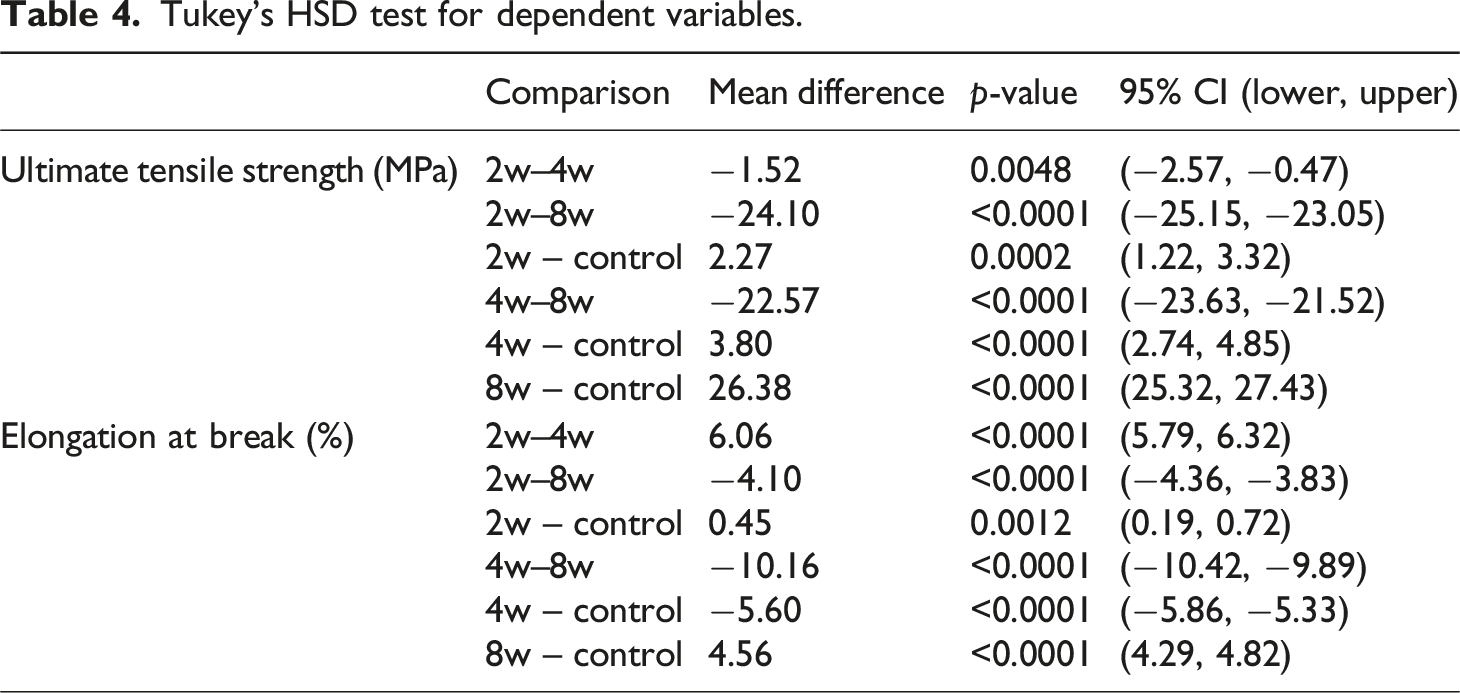
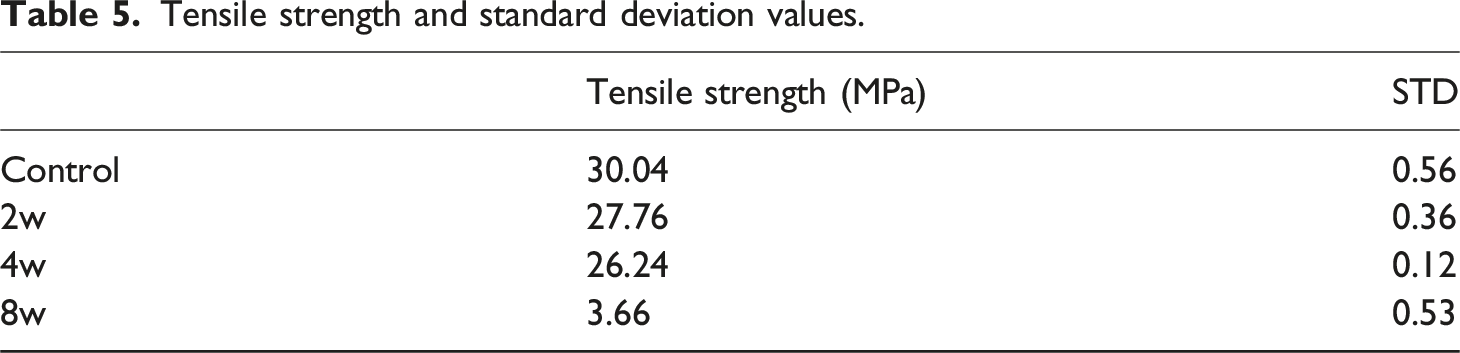
The results of the Tukey HSD post-hoc test indicate that all pairwise differences are statistically significant, demonstrating that the mechanical properties at each weathering interval differ significantly from one another. The tensile test results of the specimens are presented in Figure 5. Ultimate tensile strength and elongation at break values are given for each specimen group. The average ultimate tensile strength value of 30.04 MPa was measured for the control group specimens, 27.77 MPa for the 2-week specimens, 26.24 MPa for the 4-week specimens, and 3.66 MPa for the 8-week specimens. Notably, the average tensile strength of the 8-week group is significantly lower than all other groups, with a reduction of about 24–26 MPa. Up to the 2- and 4-week processes, a small degradation gradient was observed, while the 8-week specimens showed a dramatic decrease. This indicates that the specimens have degraded and lost a bond structure that would maintain their structural integrity. Tensile test results.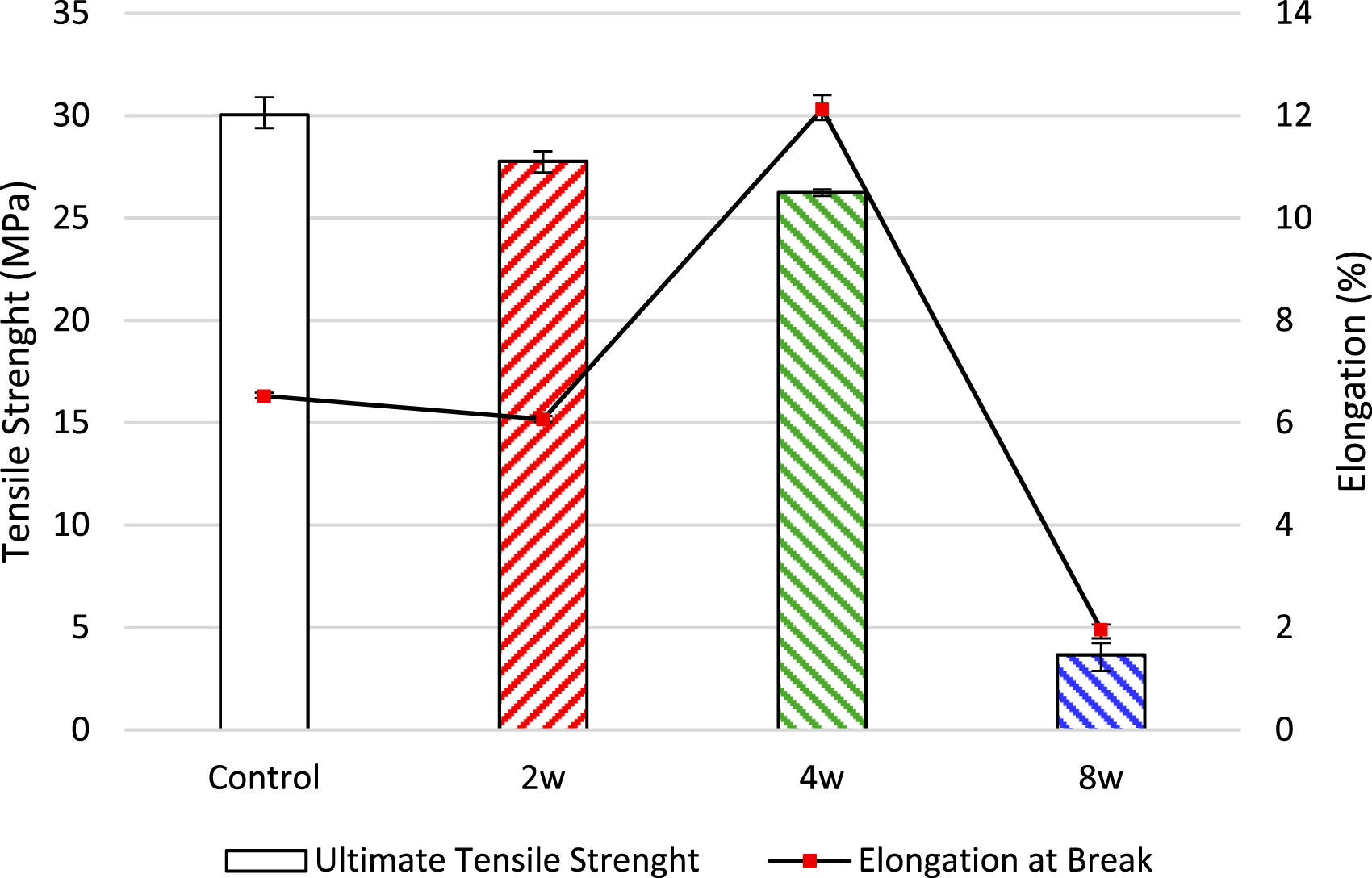
Tensile strength and standard deviation values.
The stress-strain chart of one of the specimens tested in each specimen group is given in Figure 6. Stress-strain curves.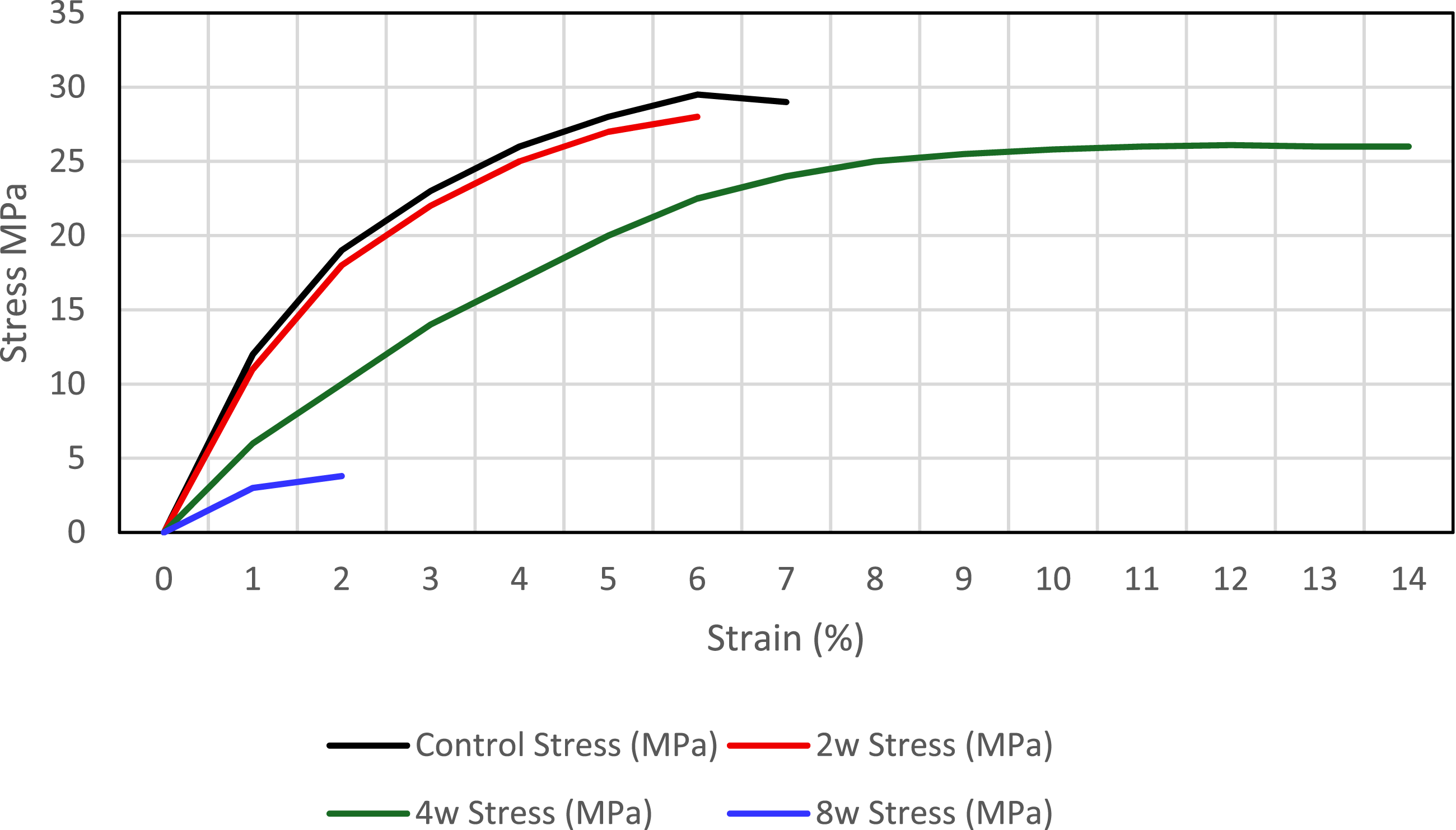
Stoia et al. measured the tensile strength value of bronze particle-reinforced PLA composites produced with MEX as 12 MPa. It was also stated that a tensile strength value of 40 MPa was measured for pure PLA. 61 Upadhyay et al. evaluated the mechanical degradation of pure PLA specimens stored in seawater for 30 days. In the study, the tensile strength value under high loading was measured as 52.96 MPa. Although the study did not examine the effect of the marine environment on tensile strength, it was noted that the partial crystallinity of the PLA material degraded under simulated seawater conditions. 62 The results obtained for bronze-reinforced PLA are lower than the values obtained in the control group of AL doped PLA composite specimens in our study. The values for pure PLA are significantly higher than the tensile strength observed in the control group and aged specimens. This difference may be due to the additive material and production parameters, as well as the layered structure and possible interfacial weaknesses resulting from production by the MEX method. Furthermore, the dramatic decrease in strength under aging conditions is consistent with the environmental sensitivities of pure PLA reported in the literature. It can be said that aluminum-reinforced PLA composites are disadvantageous in terms of mechanical performance compared to pure PLA but exhibit similar degradation tendencies under environmental conditions.
The environmental conditions applied in the conditioned cabin showed a decrease in the ultimate tensile strength values of the specimens after two and 4-week. The hydrolysis reactions during the first two and 4-week showed a slight progression, and the effect on the mechanical properties was therefore low. PLA is a polymer containing ester bonds and is susceptible to hydrolysis reactions under high temperature and humidity conditions. Hydrolysis causes PLA chains to break down into low molecular weight compounds by interacting with water.63,64 This process led to a decrease in mechanical strength. At the end of 8 weeks, the molecular weight decreased significantly, and structural integrity was disrupted. In addition, salt ions penetrated the PLA matrix, increasing the hydrophilic structure and accelerating the entry of water into the polymer.65,66 Al reinforcements in the PLA matrix may also have caused microcracks and contributed to the rapid decrease in ultimate tensile strength. 16 In addition, the interaction of Al reinforcements with salt water caused corrosion and local stresses in the composite material,67,68 resulting in a decrease in tensile properties with time. Regarding the changing temperature values in environmental conditioning, high temperatures may have increased hydrolysis, 69 and low temperatures may have increased brittleness and accelerated crack formation.70,71
When the elongation at break of the specimens was analyzed, it was observed that the elongation at break of the 2-week specimens decreased compared to the control group. However, the 4-week group exhibited a significantly higher elongation at break with an increase of approximately 5%-6% compared to the control and 2-week groups. Under the influence of the water and saline environment, the PLA matrix was subjected to the hydrolysis process. Although in the first 2 weeks the molecular chains were not yet largely broken down, the elongation at break may have decreased due to microcrack formation in the crystalline regions and reduced plastic deformation ability. Subsequently, as hydrolysis progressed, the decrease in the molecular weight of PLA and the shortening of the polymer chains may have caused some softening of the polymer matrix. Pillin et al. reported that the elongation at break of PLA decreased from 4.4% to 2.9% after three extrusion cycles. This is attributed to a reduction in the chain length of the biopolymer. 72 Hernandez et al., in their study, examined the degradation of PLA due to reprocessing (twin-screw extrusion) and exposure to environmental conditions (UV light, temperature, and humidity), and noted that mechanical recycling of PLA is compromised when exposed to environmental conditions. 73 Similar decreases in elongation have also been observed in studies in the literature.
Conversely, the 8-week group had the lowest elongation at break (1.79%–2.06%), which was significantly lower than the control, 2-week, and especially the 4-week group. The brittle structure at the end of 8 weeks may be due to the advanced hydrolysis and a significant decrease in the molecular chain length. The load-bearing bonds within the PLA matrix are largely broken, which causes the structure to lose its ductility. The growth of microcracks and voids caused by the salt-heat effect of aluminum particles may have caused the specimens to become much more brittle, and a sudden rupture may have occurred. This is evidenced by the imaging performed in SEM analysis.
The severe degradation observed in our PLA/Al composites contrasts strongly with the modest property changes reported for neat PLA. For example, after only 8 weeks in salt-spray conditions our composites’ ultimate tensile strength plummeted by ∼88% (from 30.04 MPa to 3.66 MPa). By comparison, Deroiné et al. noted that unreinforced PLA aged 6 months in seawater at 40°C experienced about a 70% drop in stress at break, 37 and Duigou et al. recorded ∼23–31% reductions in tensile strength after 3 months at 20–40°C. 36 Bao et al. found that the maximum tensile stress of PLA/PHB plastics decreased by ∼38% after 4 weeks in static seawater (from 21.66 MPa to 13.36 MPa), 37 and Krasowska & Heimowska reported a slight decline in tensile strength to 48.6 MPa after up to 16 months in the Baltic Sea. Even long-term SRPLA specimens showed only gradual losses, requiring around 2 years to reach a ∼20% reduction. These comparisons indicate that the combined effects of aluminium corrosion, galvanic coupling and hydrolytic chain scission accelerate degradation far beyond the levels observed for neat PLA or PLA/PHB blends. 38 In particular, the rapid decline in tensile strength and elongation in our composites suggests that interfacial debonding and pitting corrosion of aluminium fillers create internal stresses and microcracks that propagate quickly under salt-spray exposure. Such synergies are absent in neat PLA systems, where degradation proceeds mainly via surface hydrolysis and recrystallization. Future work could explore coupling agents or protective coatings to mitigate these interfacial effects and bring the composite’s durability closer to that of neat PLA.
Considerations on mass change, dimensional stability, and Tg evolution
A pertinent point raised during the review process concerns the direct measurement of mass change due to moisture absorption, potential mass loss from surface degradation, dimensional stability, and the evolution of the Tg throughout the aging period. While the present study did not explicitly track gravimetric changes or dimensional variations over time, the significant alterations observed in the mechanical, thermal, and morphological properties provide compelling indirect evidence of these underlying processes. The drastic reduction in ultimate tensile strength (from ∼30 MPa to ∼3.6 MPa) and the complex shift in elongation at break after 8 weeks of exposure strongly suggest substantial hydrolytic degradation. This hydrolysis inherently involves water absorption (potentially leading to an initial mass increase) and the chain scission of ester bonds, generating low molecular weight oligomers that could be leached out from the polymer matrix, ultimately leading to mass loss.55–57 The extensive debonding of aluminum particles, severe cracking, and the formation of voids and salt crystals (NaCl) observed in SEM analysis of the 8-week specimens (Section 3.4) visually confirm the material’s removal from the specimen surface and support the hypothesis of mass loss and likely dimensional changes due to erosive degradation. Furthermore, the observed decrease in Tg for the 8-week specimen (67.25°C compared to 68.45°C for the control), as determined by DSC analysis (Section 3.2.1), is a direct thermodynamic consequence of this hydrolytic chain scission, which reduces the average molecular weight and disrupts the chain entanglement, thereby lowering the energy required for the glass transition. Therefore, while gravimetric and dimensional measurements were not the focus of this experimental setup, the comprehensive dataset presented herein consistently points towards a degradation mechanism dominated by hydrolysis, leading to mass transfer, structural disintegration, and a consequent reduction in Tg. Quantifying these mass and dimensional changes represents a valuable direction for future research to fully elucidate the degradation kinetics of PLA/Al composites in harsh environments.
Degradation mechanisms at the PLA/aluminum interface
The degradation mechanisms of the PLA/Al composites in saltwater are multifaceted, involving the individual degradation of PLA, the corrosion of aluminum, and the failure of their interface. The primary adhesion mechanism between the PLA matrix and aluminum particles is widely accepted to be weak physical adhesion (van der Waals forces) and mechanical interlocking, as the polar PLA chains have limited chemical affinity for the metallic filler surface. 59 In a saline environment, the degradation is severely accelerated. The PLA matrix undergoes hydrolysis, where ester bonds are cleaved by water molecules, leading to polymer chain scission, a reduction in molecular weight, and a loss of mechanical integrity.55–57 Concurrently, the saltwater electrolyte penetrates the interface, initiating galvanic corrosion of the aluminum particles. This corrosion generates aluminum oxides/hydroxides, which can create localized stresses and further disrupt the weak interfacial bonding.65,66 The presence of chloride ions (Cl−) from NaCl is particularly aggressive, catalyzing both the hydrolysis of PLA and the pitting corrosion of aluminum. This synergistic effect between hydrolytic degradation and metal corrosion leads to rapid interfacial debonding, visible in SEM micrographs as voids and particle pull-out (Figure 6). The difference between pure water and salt solution lies in this catalytic effect of ions; salt solution not only facilitates water ingress due to osmotic pressure but also actively accelerates the degradation processes through ionic corrosion and catalysis, leading to a more severe and rapid loss of properties compared to pure water-based degradation. 57 The focus was on the effects of degradation mechanism on the relationship between Al and PLA. The effect on the interface between this reinforcement particulars and the matrix was investigated in tensile test specimens, and it was found that this effect was consistent across all regions of the specimens during the all-test periods.
Morphological analysis
SEM analyses were conducted on the fracture surfaces of PLA/Al composites, following tensile tests conducted according to ASTM D638. The specimens were subjected to accelerated aging under salt spray conditions for 0, 2, 4, and 8 weeks, and the morphological changes at the matrix–filler interface are shown in Figure 7. SEM images.
In the control group (0W), SEM images revealed weak interfacial adhesion between the PLA matrix and aluminum reinforcements. This poor bonding likely stems from insufficient wettability and limited chemical affinity between the polar PLA chains and metallic filler particles. 74 Similar interfacial weaknesses have been reported in polymer–metal composites, where inadequate bonding leads to compromised mechanical performance.
After 2 weeks (2W) of salt spray exposure, noticeable interfacial debonding and voids formed between the matrix and Al particles. These gaps can be attributed to the diffusion of water and salt ions into the polymer matrix, leading to hydrolytic degradation and polymer chain scission. The morphological damage supports the hypothesis that moisture ingress disrupts the filler–matrix adhesion, increasing the risk of microcrack formation and premature failure. This phenomenon aligns with findings from studies on aluminum matrix composites, where corrosion susceptibility increased due to environmental exposure.
Interestingly, 4-week-aged specimens (4W) exhibited comparatively better bonding between the matrix and aluminum particles. The fracture surfaces appeared more cohesive, which may explain the slight increase observed in elongation at break. This improvement could be due to the plasticization of the PLA matrix by absorbed moisture, reducing its glass transition temperature and temporarily increasing ductility. 62 Similar behavior has been observed in polymer composites subjected to environmental aging, where moisture absorption leads to transient enhancements in flexibility. 75
By 8 weeks (8W), extensive structural deterioration was observed. SEM images showed that aluminum particles had completely debonded from the matrix, accompanied by severe cracking and voids. Distinct cubic salt crystals (NaCl) were detected, particularly near the outer edges of fracture surfaces, indicating salt ingress and crystallization within the composite structure. 76 This suggests that prolonged salt spray exposure accelerated hydrolytic degradation through ion diffusion, weakening the polymer network and disrupting matrix continuity. 77 Similar degradation mechanisms have been documented in studies on the corrosion behavior of aluminum alloys under salt spray conditions. The SEM results confirm that environmental aging under salt spray conditions leads to progressive degradation of PLA/Al composites. The weakening of interfacial bonding and emergence of structural defects over time are closely linked to water and salt ion diffusion, as supported by findings in existing literature.
The salt molecule seen in Figure 8 was observed in the 8W specimens. This molecule was imaged in the surface region of the specimen, near the edge of the fractured surface. Salt molecule.
Micro-CT (computed tomography) analysis
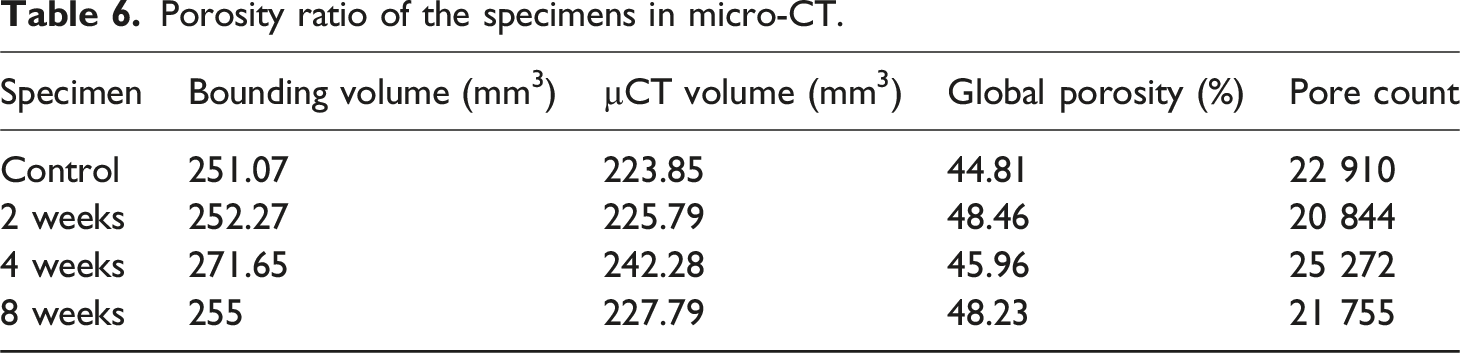
Porosity ratio of the specimens in micro-CT.
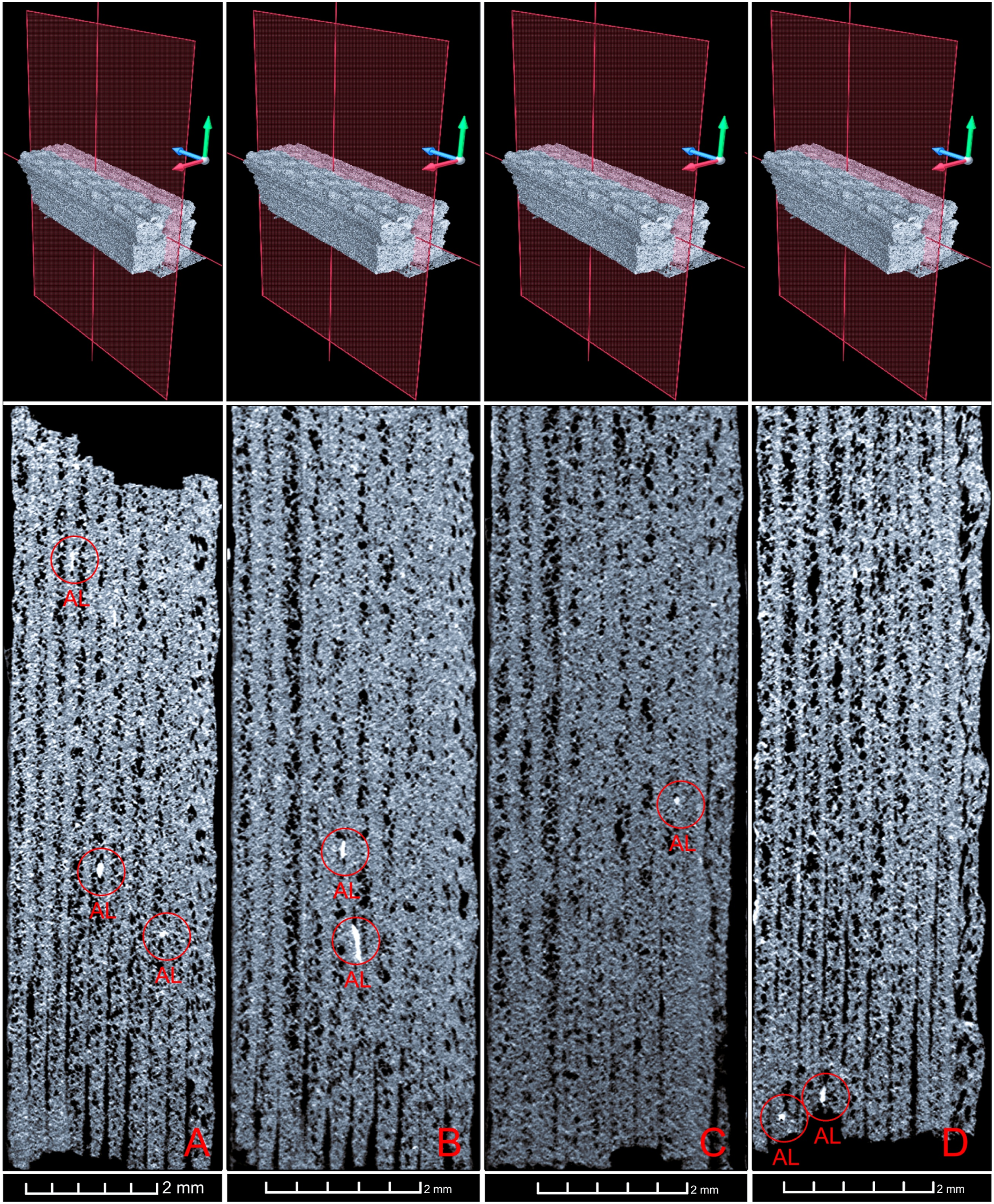
Micro-CT analyses and cross-sections of specimens, (a) control, (b) 2 weeks, (c) 4 weeks, (d) 8 weeks.

Micro-CT analyses and longitudinal section of specimens, (a) control, (b) 2 weeks, (c) 4 weeks, (d) 8 weeks.
At 4 weeks, global porosity decreased to ∼46.0%. µCT images revealed fewer open voids, and the maximum pore diameter fell slightly. This reduction is consistent with the pore‐clogging phenomenon: as salt crystals precipitate inside pores, they increase pore size locally but also obstruct some voids. In an ignimbrite salt-mist study, the authors observed that open porosity increased initially because salt crystallisation enlarged certain pores and fractures, but water absorption declined because salt crystals blocked access to other voids. 80 After about 40 cycles, accumulation of salt crystals in voids reduced the variation in physical properties and even decreased open porosity. A comparable clogging mechanism in PLA/Al composites would cause some pores to fill with crystallised salts, temporarily lowering global porosity.
By 8 weeks, global porosity rose again to ∼48.2%, despite many pores being partially filled. µCT reconstructions showed microcracks and coalesced voids, especially near aluminium particles. SEM observations of aged epoxy joints have reported that chemical degradation can increase porosity inside entangled polymer chains, promoting chain expansion and the formation of microcracks within the polymer network. 81 These microcracks and interfacial debonding propagate under environmental cycling, forming new voids and reversing the earlier pore-clogging trend. In the flax/glass composite study, irreversible defects such as delamination, debonding and matrix microcracks persisted at the end of the salt-fog/dry cycle, reducing mechanical stiffness. The dramatic reduction in ultimate tensile strength observed in the current PLA/Al specimens (from 30 MPa to ∼3.7 MPa) therefore correlates with microcrack evolution rather than global porosity changes. Aluminium particle detachment was confined to surface layers in µCT images, indicating that internal damage is dominated by microcracking and void coalescence rather than particle migration.
The porosity trend indicates that environmental ageing involves competing processes: initial swelling and microcrack formation increase porosity; subsequent salt crystallisation fills some voids and reduces porosity; and long-term chemical degradation and interfacial failure generate new microvoids and raise porosity again. These findings echo observations in natural stone, where salt crystallisation both enlarges pores and blocks water pathways. For PLA/Al composites, durability is controlled more by interfacial adhesion and microcrack suppression than by global porosity. Future research should explore surface coatings or coupling agents that limit salt ingress and strengthen the PLA–aluminium interface. Strategies to mitigate cracking, such as toughening the PLA matrix or employing graded particle sizes, may also enhance long-term performance.
Conclusion
This study systematically investigated the effects of salt spray aging on the properties of aluminum-doped PLA composites produced by MEX. The results demonstrate that environmental exposure leads to progressive degradation, culminating in a severe loss of mechanical performance after 8 weeks. FT-IR and DSC analyses confirmed hydrolytic degradation of the PLA matrix and a consequent reduction in glass transition temperature. TGA revealed a slight decrease in thermal stability with increased aging time. Most significantly, SEM micrographs provided direct evidence of the dominant failure mechanism. Interfacial debonding between the PLA matrix and aluminum particles, exacerbated by salt corrosion and water ingress. The synergistic effect of PLA hydrolysis and aluminum corrosion ultimately compromises the composite’s integrity. These findings underscore that while aluminum reinforcement can enhance PLA’s initial properties, its long-term performance in humid, saline environments is limited without improved interfacial adhesion or protective strategies. Future work will focus on surface treatments of aluminum filler to enhance compatibility and inhibit corrosion, thereby improving the durability of sustainable PLA-based composites for demanding applications.
Footnotes
Acknowledgements
The preliminary findings of this study were presented at the VI. International Ege Composite Materials Symposium 2024. The authors would like to Haşim Fırat Karasu for his contributions to the aging tests. We would like to express our sincere appreciation to Karfo Endüstriyel for their valuable support in performing the detailed and high-precision micro-CT analyses that significantly contributed to this study.
Author contributions
Conceptualization, A.D., C.B, A.S., and F.M.; methodology A.D., C.B, A.S., and F.M.; validation, A.D, and F.M. Analysis, A.D., C.B, A.S., and F.M.; writing—review and editing, A.D., C.B, A.S. and F.M.; All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
