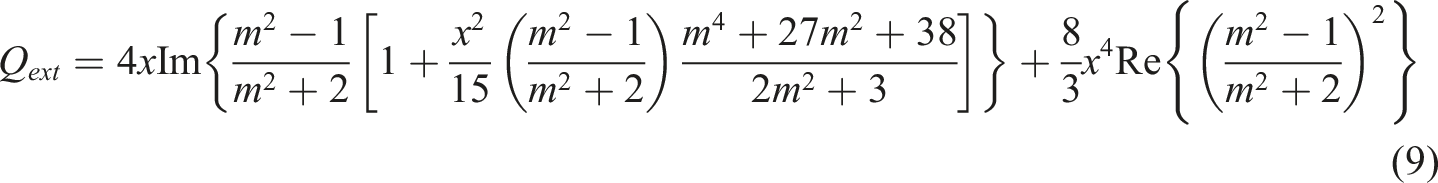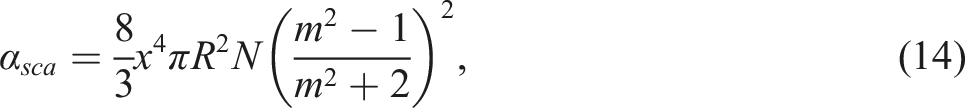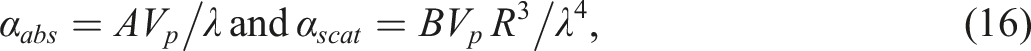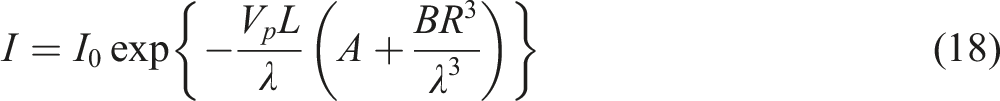Abstract
The present work studies the effect of embedded zirconia nanoparticles in polypropylene on the structure and optical absorption in the vicinity of the optical band gap. X-ray diffraction analysis, X-ray dispersion energy spectroscopy, scanning and transmission electron microscopes, IR and UV spectroscopic methods were used for investigation. It is shown that nanocomposites are comprised of two distinct phases. An increase in the filler concentration leads to a decrease in crystallinity. It has been demonstrated that the optical band gap decreases while the Urbach energy increases with increasing filler concentration. These alterations are concomitant with the establishment of novel energetic levels within the band gap, resulting in its constriction, as well as an augmentation in disorder and interaction between the nanoparticles and the matrix. Additional peaks between are also observed that due to the presence of C-C and C-H bonds in the molecular structure of polypropylene and is caused by σ→σ* type transitions. It has been observed that as the particle concentration and size increase, there is a decrease in the optical transmittance in the relatively transparent region of the composite. This phenomenon is interpreted by the theory of light absorption and scattering in the dispersed medium of nanocomposite.
Keywords
Introduction
Polymer materials are used not only in various fields of technology but also in households. One of the ways to modify and improve the physical properties of polymers is to embed various fillers into them. In this case, to the distinctive properties of polymers, such as flexibility, the ability to obtain products of different shapes, are added the properties of the filler.1–14 This process can result in the formation of a new material quality.15–18 The study of properties of polymer nanocomposites is a priority direction of modern science due to the unique properties of these materials. In order to successfully implement nanoscale systems in mass production, it is essential to undertake a comprehensive study of the properties and structure of nano-objects. This study must include a thorough investigation into the mechanisms that determine these properties.
Polymer nanocomposites with quantum dots exhibit a high degree of sensitivity to environmental changes, rendering them particularly well-suited for use in biosensors, gas analysers and temperature detectors. 19 Authors of the work 20 consider the possibility of creating active waveguides based on hybrid organic (polymer) - inorganic (semiconductor quantum dots) materials. It has been demonstrated that they can be useful for the introduction of new photonic devices, given that they combine properties with the technological capabilities of polymers. The review article 21 presents a comprehensive examination of the current research status in this area and underscores the potential of quantum dots–polymer nanocomposites in transforming food safety monitoring and sensing.
The combination of metal oxide nanoparticles of differing structures with polymers results in the formation of nanocomposites, which exhibit intriguing properties and application potential. The following applications make use of such nanocomposites: supercapacitors,22–24 sensors,25–30 biomedicine, 25 dye and nitrophenol reduction,31,32 microwave absorption, 33 absorption of heavy metals, 34 separator in Li-S batteries. 35
Polypropylene is used in many branches of technology due to its physical and chemical properties, which make it a highly versatile material. It is relatively transparent and neutrality in the presence of chemically aggressive reagents. 36 It is distinguished by its high air permeability and its notable resistance to water absorption. The material under discussion is characterized by low density and superior mechanical properties. The fabrication of the material is also a relatively straightforward process, which can be achieved through extrusion or injection moulding techniques. In comparison with other polymers, the material is relatively inexpensive. Zirconia is an excellent ceramic material that is biocompatible and commonly used in dental and orthopaedic implants. Its chemical inertness makes it useful in harsh environments. ZrO2 can be used as a catalyst support or functional filler in composite materials. Polypropylene is a hydrophobic, nonpolar polymer, so its interaction with ZrO2 is largely limited to van der Waals or dispersion forces. As a result, there is poor interphase adhesion. The combination of polypropylene, a flexible and chemically resistant polymer, and zirconia, a strong and inert ceramic, is often chosen to create composite materials that offer the advantages of both: durability, stability and versatility. Moreover, these nanocomposites have the potential to be used in various antimicrobial applications, including in the food packaging industry.
The results of differential scanning calorimetry studies before and after the effect of electrothermal polarization (ETP) of PP + ZrO2 nanocomposites are discussed in work. 37 It has been shown that the change in thermal parameters is due to the change in mobility at the interfacial boundary between the matrix and the nanoparticle depending on the field intensity value. A study of the effect of an electric field on the thermophysical properties of PP + ZrO2 nanocomposites is presented in 38. The investigation revealed that the thermograms for all concentrations of the filler exhibited a weak maximum at the temperature range of 100–130°C following electrothermal polarization. The refractive index, electrical susceptibility, dipole moment, mean polarizability, hyperpolarizability, dielectric constants, ionisation potentials, electron affinity, electronegativity, hardness and flexibility of ZrO2-doped PVDF were determined in the work. 39 It has been demonstrated that this composite exhibits good dielectric properties and is therefore a good piezoelectric material. The authors of the work 40 investigated the charge states, thermophysical and mechanical properties of polypropylene doped with zirconium oxide nanoparticles, and the subsequent changes that occur during the process of heat treatment at varying temperatures. It is shown that the magnitude of the accumulated charge in the traps and the activation energy of charge release from traps have maximal value at nanoparticle concentration of 3 vol%. The mechanical durability of the PP+3% ZrO2 nanocomposite is found to decrease with an increase in the pre-treatment temperature. Investigation of thermal stability presents in the works. 41 In particular, the present study demonstrates that elevated temperatures during heat treatment result in the partial degradation of polymer chains, thereby causing a reduction in the critical melting temperature.
The optical properties of a material, such as its refractive index, absorption and luminescence, reflect its internal structure. 42 Changes in these properties as the filler is introduced into the polymer allow the nature of the interaction between the polymer matrix and the filler to be determined. A number of studies43–51 have been dedicated to investigating the optical properties of nanocomposites. The following nanocomposites are considered: zirconium carbide (ZrC)—silicon dioxide (SiO2) nanoparticles into pure polyvinyl alcohol (PVA) 43 ; polyvinyl alcohol (PVA) and carboxymethyl cellulose (CMC) as the organic host matrix with varying concentrations of chromium trioxide (Cr2O3) and silicon dioxide (SiO2) 44 ; ternary nanocomposite comprising polyvinyl alcohol (PVA), silver (Ag), and barium titanate (BaTiO3) 45 ; (PVA–PVP) as host polymer matrix doped with ZrC nanoparticles 46 ; (PMMA-SiO2/CuO) nanocomposites(NCs) nanostructures (SiO2 and CuO) 47 ; nanostructures (SiO2 and CuO) into polyvinyl alcohol (PVA) 48 ; the silicon dioxide (SiO2), cobalt oxide (CoO), and polyvinyl alcohol (PVA) nanostructures’ structural 49 ; PVA/PVP/MnO2 50 ; mixture of two polymers, polyvinyl alcohol (PVA) and polyethylene glycol (PEG), with two nanomaterials, cobalt trioxide (Co2O3) and silicon dioxide (SiO2) nanoparticles. 51 The authors of these works showed that an increase in filler concentration leads to a decrease in transmittance and the optical energy band. Meanwhile, the absorption coefficient and refractive index increase. The review 52 gives an overview of the effect different particle materials have on the properties of transparent polymer composites with consideration of the composite’s transparency. It has been shown that it is possible to keep the polymer transparent by adding nanoparticles.
Nanocomposites with tunable optical properties have a variety of applications, including in flexible electronics, 53 displays, 54 sensors 55 organic light-emitting diode and solar cells. 56 Research in this area enables the efficiency of light absorption, transmission and emission to be optimized. Adding nanoparticles to polymers can provide UV radiation shielding without compromising transparency in the visible spectrum.
It is evident from the analysis of the references that there is an absence of a comprehensive representation of the optical properties of nanocomposites in the vicinity of the fundamental absorption edge, accompanied by a theoretical interpretation of the obtained results. The present study investigates the impact of the concentration and dimensions of zirconium oxide nanoparticles on the absorption edge and transmission near this edge, as well as the Urbach energy of polypropylene.
Methodology
The polymer matrix was fabricated using isotactic polypropylene (PP) pellets with an average diameter of 5 mm (Dema Import and Export Co. Ltd, China). Prior to preparing the composite, the polypropylene (PP) granules were dried in a vacuum oven at 60°C for 12 hours to remove any residual moisture that could affect the dispersion and structure of the composite. To dissolve the PP, the granules were added to toluene at a concentration of 5 wt % and heated to 120°C under constant stirring. The dissolution process was carried out in a three-necked flask equipped with a reflux condenser to prevent solvent evaporation, and the solution was maintained under nitrogen atmosphere to avoid oxidative degradation.
Monoclinic zirconium dioxide (ZrO2) nanoparticles (Sigma-Aldrich, USA) with particle sizes of 20 nm, 40 nm, and 100 nm were used as the inorganic filler. High-resolution transmission electron microscopy (HRTEM) images of zirconium dioxide nanoparticles were taken using JEM-1400 (JEOL company, Japan) at 80–120 kV.
The nanoparticles were weighed to achieve volume fractions of 1 vol%, 3 vol%, 5 vol%, and 7 vol% in the final composites. The specified zirconium oxide concentrations are selected to systematically analyze property changes, identify the optimal concentration range and prevent agglomeration or degradation of properties at high loadings. This aligns with standard practice in composite material research.
Prior to addition, the nanoparticles were dried at 80°C for 6 hours to remove absorbed moisture and then ultrasonicated in toluene for 30 minutes to break up any agglomerates and ensure better dispersion. Each size of nanoparticles was separately dispersed into the polymer solution under magnetic stirring for 1 hour at 120°C. To promote uniform dispersion, the mixture was also subjected to mild ultrasonic agitation in a water bath for 10 minutes. After homogenization, the mixture was thermally treated for 3 to 6 hours to improve polymer-particle interaction and promote partial evaporation of the solvent. The resulting suspension was poured into Petri dishes and dried under a fume hood at room temperature for 24 hours, followed by vacuum drying at 60°C for 12 hours to ensure complete removal of residual toluene. The dried composites were then ground into powder using a mortar and pestle.
The amount of powder used for pressing was chosen to give a final thickness of 100 μm. Additionally, the thickness of the sample was also measured using a Mitutoyo digital micrometer (Mitutoyo Corporation, Japan) with an accuracy of 3 µm.
Disc-shaped samples with diameter of 4 cm were prepared by hot pressing. The powder was placed into a steel mold and pressed at 175°C and a pressure of 15 MPa for 3 minutes, using a laboratory hydraulic press. After pressing, the mold was cooled at a controlled rate of 20°C per minute down to room temperature to minimize thermal stress and internal defects.
The analysis of the structure by X-ray dispersion energy spectroscopy (EDAX) and the presence and distribution of nanoparticles in the polymer matrix was recorded using scanning electron microscopy (SEM, JEOL JSM-7600 F, JEOL company, USA). Infrared spectra were recorded on the instrument PerkinElmer Spectrum Two FT-IR Spectrometer (PerkinElmer, Inc., USA).
The optical spectra of the composite samples in the vicinity of their band gap were characterized using a SPECORD-250 UV-Vis spectrophotometer (Analytik Jena, Germany). Spectral measurements were carried out in the range of 190–1100 nm at room temperature.
Results and Discussion
The HRTEM image of zirconium dioxide nanoparticles with particle dimension of 20 nm is presented in Figure 1. As is evident, the nanoparticles exhibit a spherical geometry, with an average diameter of approximately 18 nm as determined from the measurement of 100 particles. The HRTEM image of zirconium dioxide nanoparticles with particle dimension of 20 nm.
The electron microscope image of the PP + ZrO2 nanocomposite is presented in Figure 2. The resulting substance is composed of spherical nanoparticles, as shown in the SEM images. Homogeneous distribution of particle is observed in this image. SEM images of PP dispersed by 20 nm ZrO2 nanoparticles at filler concentrations of 3 vol% (a) and 7 vol% (b).
Figure 3 shows the X-ray diffraction patterns of PP + ZrO2 samples at different volume concentrations of nanoparticles. It is notable that reflexes corresponding to pure PP are characterised by the most distinct structure which may indicate higher crystallinity. As the percentage of nanoadditives increases, the peaks become broader and less intense, indicating a decrease in crystallinity and a more amorphous structure. Broader peaks are observed at 2θ ≈ 14°, 17°, 21°, which are characteristic of matrix PP. As the percentage of zirconium oxide increases further, the corresponding peaks broaden. This indicates a fine-grained structure, a more amorphous state and the formation of additional defects and phases. The peak around 28.2° is typical for the (111) plane, while 31.5° and 4.2° correspond to the (111) and (111) planes, and 4.2° near (200) is characteristic of monoclinic ZrO2. Weak peaks at 30°–34° correspond to monoclinic ZrO2. X-ray diffraction patterns of PP + ZrO2 nanocomposites at different volume concentrations of 20 nm particles: (a) 0 vol%; (b) 3 vol%; (c) 5 vol%; (d) 7 vol%.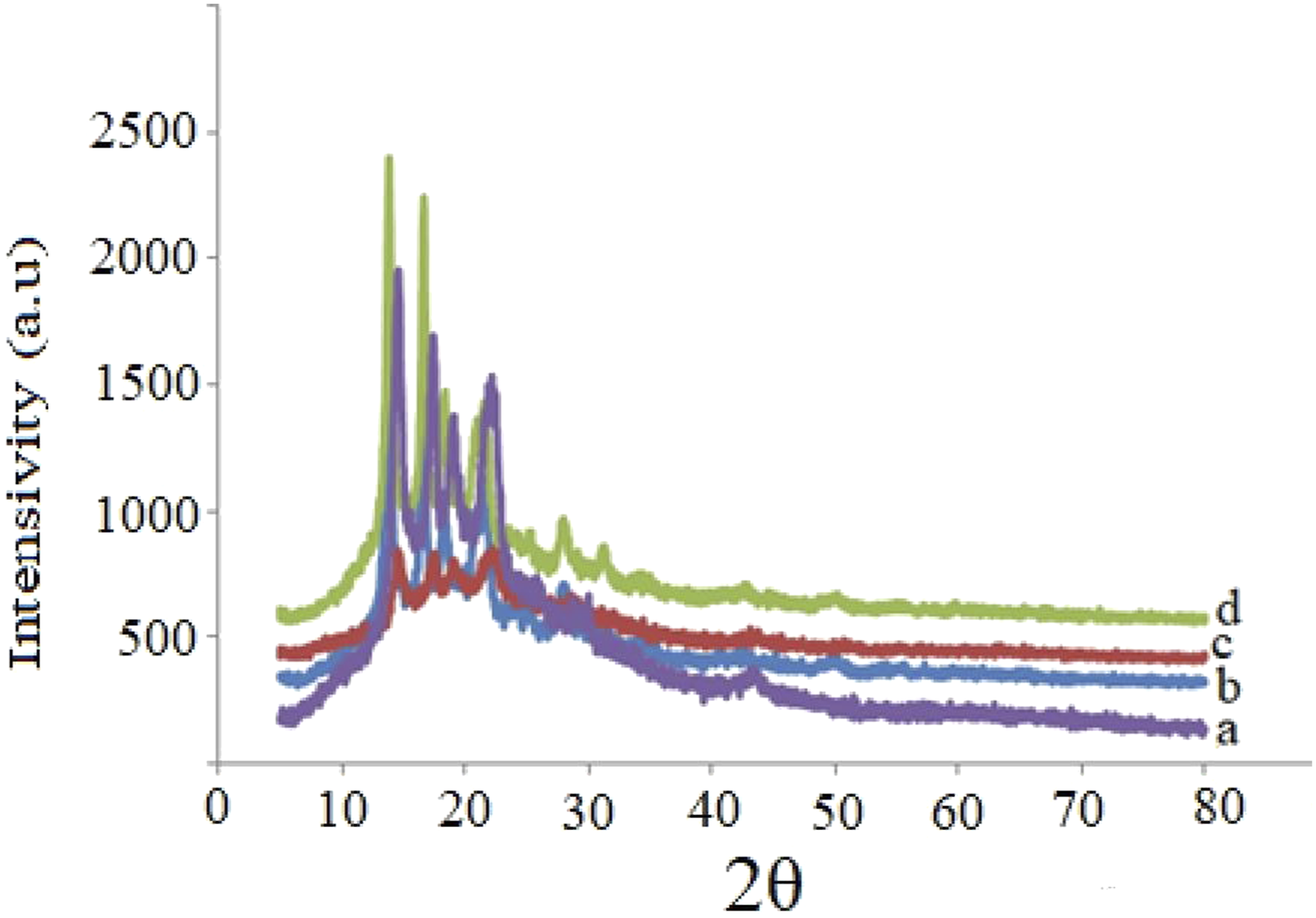
The analysis of the structure by X-ray dispersion energy spectroscopy (EDAX) is presented in Figure 4. As illustrated in the provided image, nanocomposites are comprised of two distinct phases. The incorporation of nanoparticles has been demonstrated to induce a modification in the supramolecular structure, resulting in the establishment of an ordered structure. EDAX images of the PP dispersed by ZrO2 nanoparticles with dimension of 20 nm at filler concentration of 3 vol%.
Figure 5 shows the IR spectra of both pure PP and nanocomposites. IR spectra of PP + ZrO2 films at different volume concentrations of 20 nm particles: (a) 0 vol%; (b) 1 vol%; (c) 3 vol%; (d) 5 vol%; (e) 7 vol%.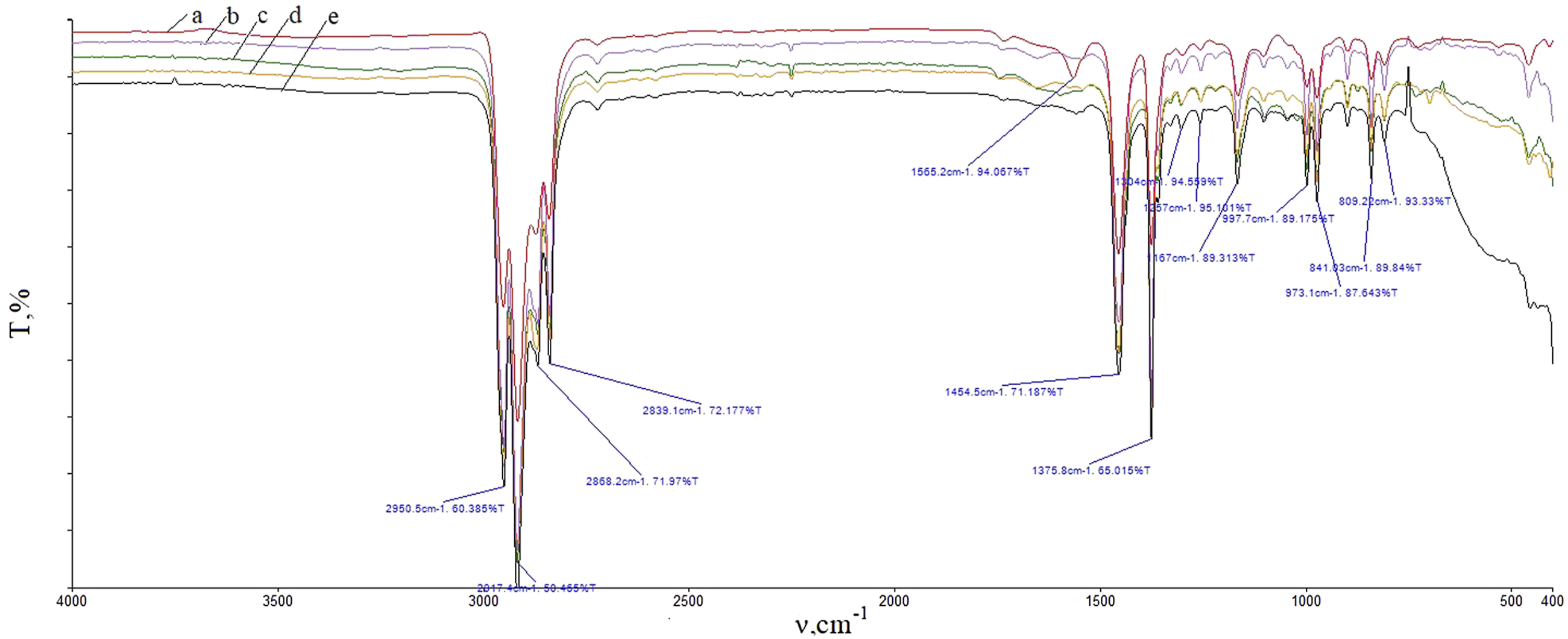
The spectra exhibit a number of bands. In order to assess the crystallinity of the samples, the most informative bands are those at 2839 cm−1, 2808 cm−1 and 841 cm−1. The first two bands indicate vibrations associated with CH. 57 The 841 cm− 1 band is assigned to the vibration coupling of C–C stretching and CH2 and CH3 rocking of 12 monomer units. This band is related to crystallization, 58 and generally employed as a probe to monitor the crystallization process of PP. As can be seen, as the concentration of nanoparticles increases, the intensity of these bands decreases. This finding is indicative of a decrease in the density of chain order and a disruption in the order of macromolecules. As a result, the crystallinity decreases.
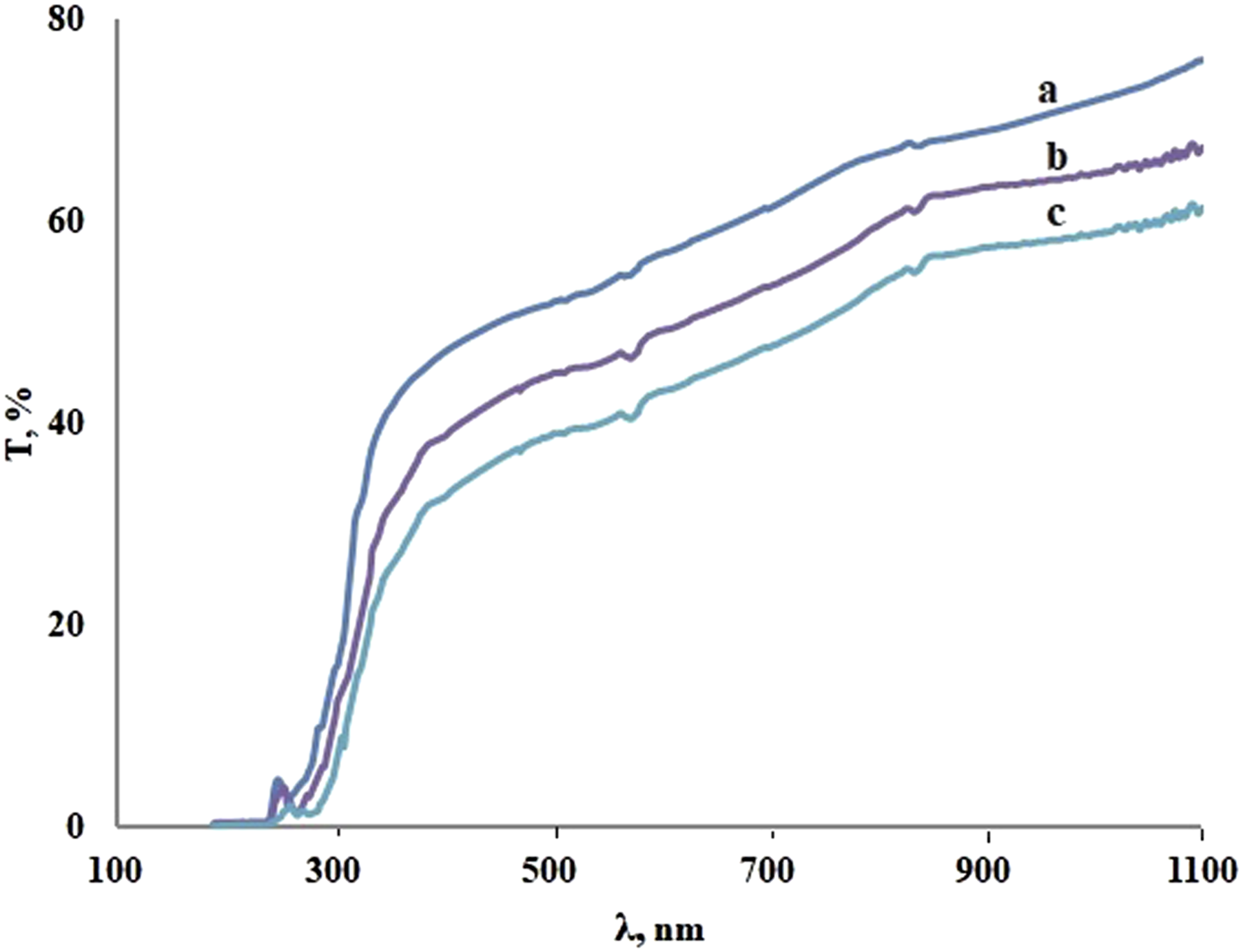
Figure 6 shows the optical transmission spectra of PP + ZrO2 films at different volume concentrations of filler with particle size of 20 nm at room temperature. The transmission spectra of PP + ZrO2 at concentration of 3 vol% for different particle sizes are presented in Figure 7. The given spectra can be divided into three frequency regions, as demonstrated in the following analysis. The first region is located at wavelengths below 300 nm and corresponds to the strong absorption of the composite with absorption coefficient greater than 104 cm−1. The second region is an intermediate one where absorption coefficient varies between 1 cm−1 and 104 cm−1. Finally, the third region is relatively transparent. As can be seen in the transparent region, transmittance decreases as the concentration and size of particles increases. Additional peaks between the first and second regions are also observed. Transmission spectra of PP + ZrO2 films at different volume concentrations of 20 nm particles: (a) 0 vol%; (b) 1 vol%; (c) 3 vol%; (d) 5 vol%; (e) 7 vol%. Transmission spectra of PP + ZrO2 at concentration of 3 vol% for different particle sizes: (a) 20 nm, (b) 40 nm, (c) 100 nm.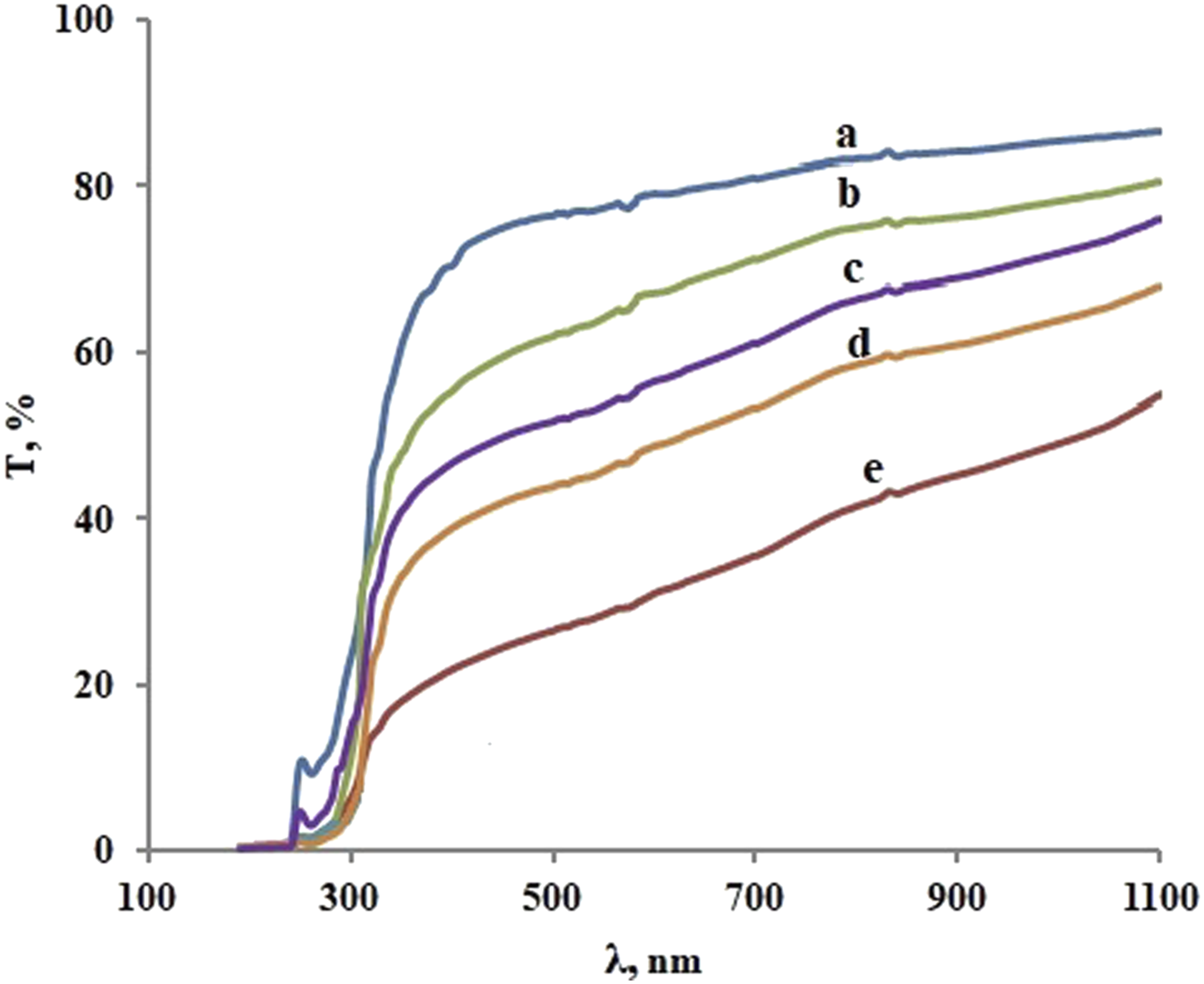
It is known that the optical behavior of polymer nanocomposites is attributable to a combination of electronic transitions and light scattering phenomena. Consider the transmittance in the samples. The light that passes through a layer of matrix with embedded particles of thickness dL undergoes extinction, the value of which can be represented in the form:
The extinction coefficient α
ext
is an additive quantity. It consists of the absorption coefficient α
abs
and the scattering coefficient α
sca
:
There are transitions between the highest and lowest molecular orbitals, known as the HOMO (highest occupied molecular orbital) and the LUMO (lowest unoccupied molecular orbital). HOMO indicates the presence of molecules with a high ability to donate electrons, while LUMO is characterized by the ability to accept electrons.59,60 In this case, HOMO and LUMO correspond to the valence band and the conduction band, respectively. The presence of ZrO2 nanoparticles can influence these transitions by modifying the local electronic environment of the polymer chains or by introducing defect states and surface energy levels at the polymer-nanoparticle interface. These modifications can lead to changes in the absorption edge position and shape, including the Urbach tail, which reflects localized states near the band edge due to structural disorder.
Transitions between LUMO and HOMO correspond to the absorption of electrons. In this case, the scattering effects are negligibly small. The energy difference between HOMO and LUMO indicates band gap. It can be expressed as follows
Assuming that the electronic state density distributions have root dependence in the energy band corresponding to HOMO and LUMO, the Tauc’s method can be used to determine the optical band gap
64
: The determination of optical indirect band gap from the plots of (αhν)0.5 versus photon energy hν at different volume concentrations of 20 nm particles: (a) 0 vol%; (b) 1 vol%; (c) 3 vol%; (d) 5 vol%; (e) 7 vol%.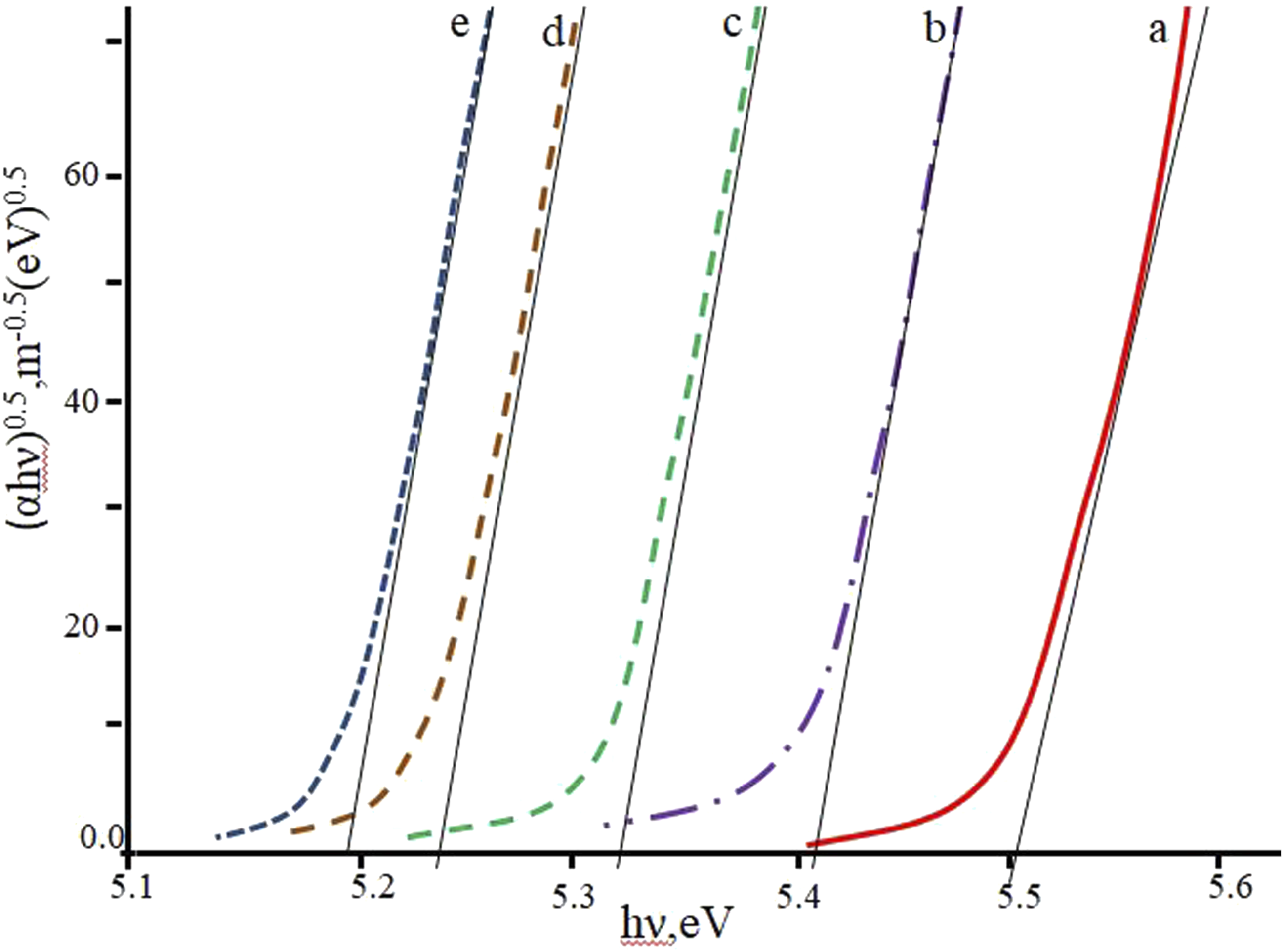
Figure 9 shows the obtained values of Eg for indirect transition depending on the filler concentration. The optical band gap of PP + ZrO2 films at different volume concentrations of 20 nm particles.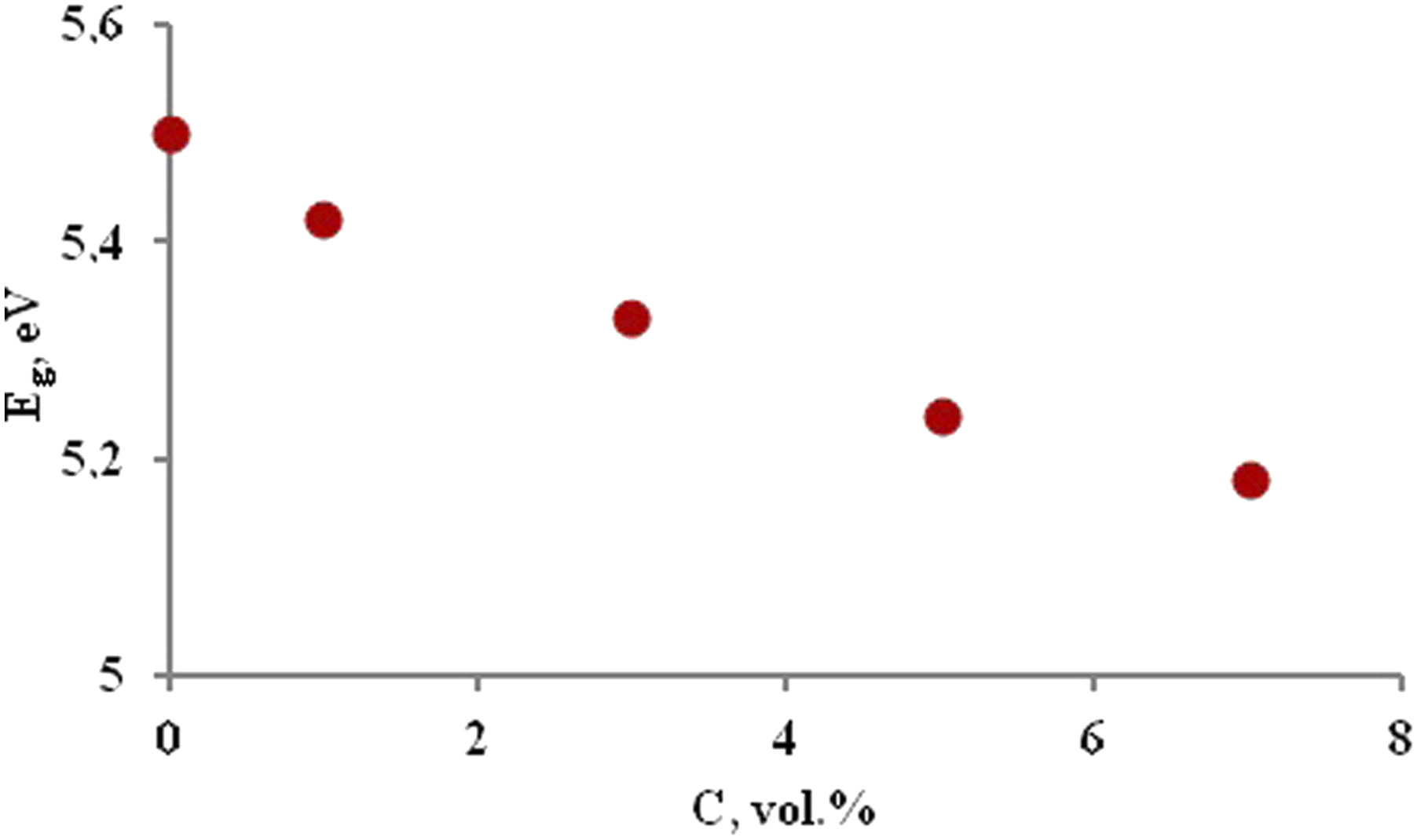
As can be seen, the optical band gap decreases with increasing filler concentration. This decrease is due to the creation of new levels in the band gap, which makes it easier for electrons to transition from the valence band to the current levels in the conduction band. Moreover, the enhancement of carrier-carrier interaction, attributable to the elevated carrier concentration within the valence and conduction bands, results in a reduction in the band gap. 65 This decline can be ascribed to the development of complexes resulting from the interaction between nanoparticles and the polymer matrix. 66 An increase in the density of defects leads to an expansion of the valence band within the band gap of the composite.67–69
As noted above, a fine absorption structure is observed between the first and second spectral region. This is due to the presence of C-C and C-H bonds in the molecular structure of polypropylene and is caused by σ→σ* type transitions. 70 Moreover, both pure polypropylene and composites demonstrate additional peaks within the same wavelength range. These peaks are indicative of electronic transitions associated with defects and phase boundary transitions. 71
The second intermediate absorption range in which the absorption depends exponentially on the photon energy, which is described by the following Urbach’s formula
72
: The determination of the Urbach energy of PP + ZrO2 from the plots of ln (α) versus photon energy hν at different volume concentrations of 20 nm particles: (a) 0 vol%; (b) 1 vol%; (c) 3 vol%; (d) 5 vol%; (e) 7 vol%.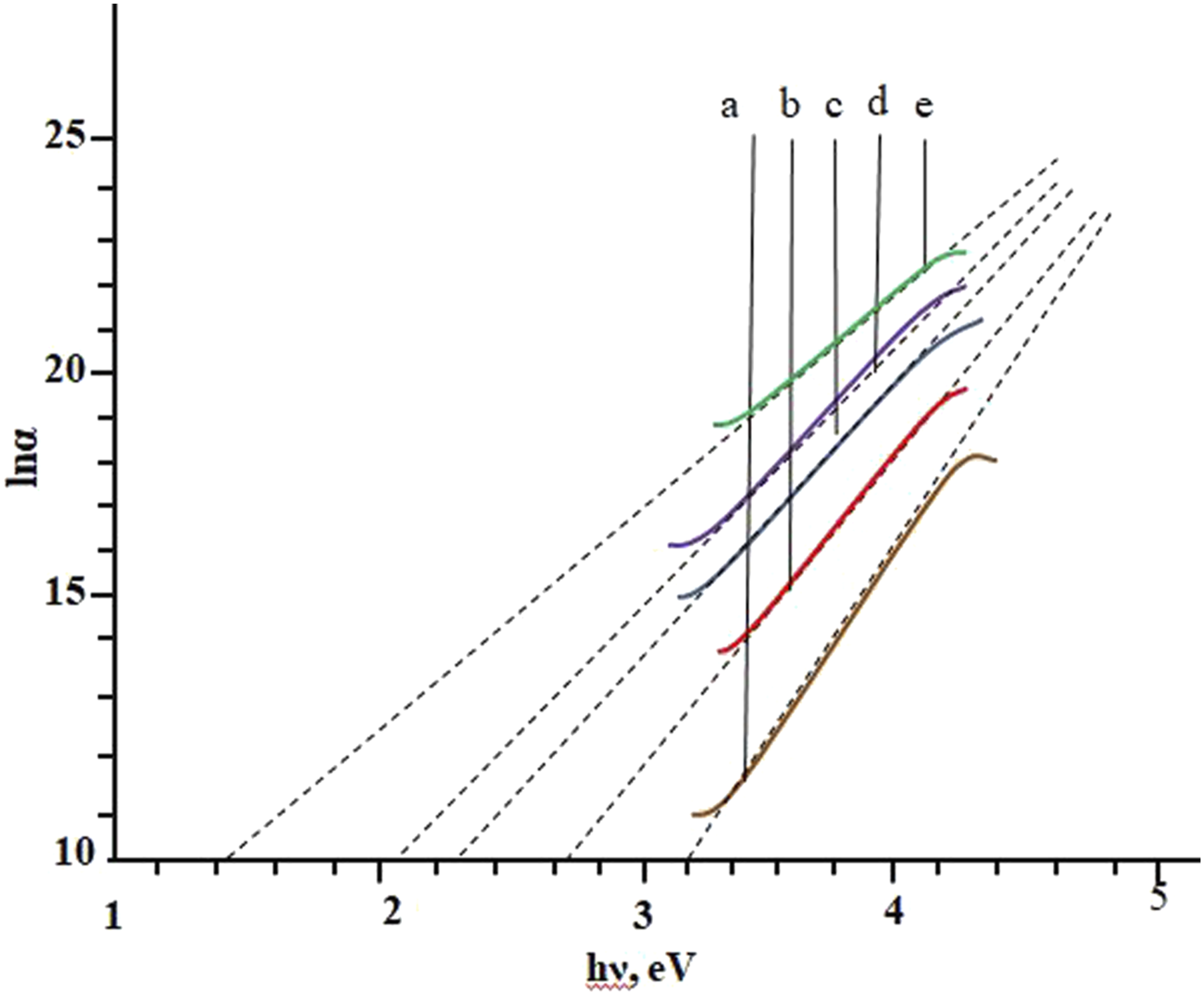
Figure 11 shows the estimated E
U
values of the nanocomposite samples. The Urbach energy of PP + ZrO2 films at different volume concentrations of 20 nm particles.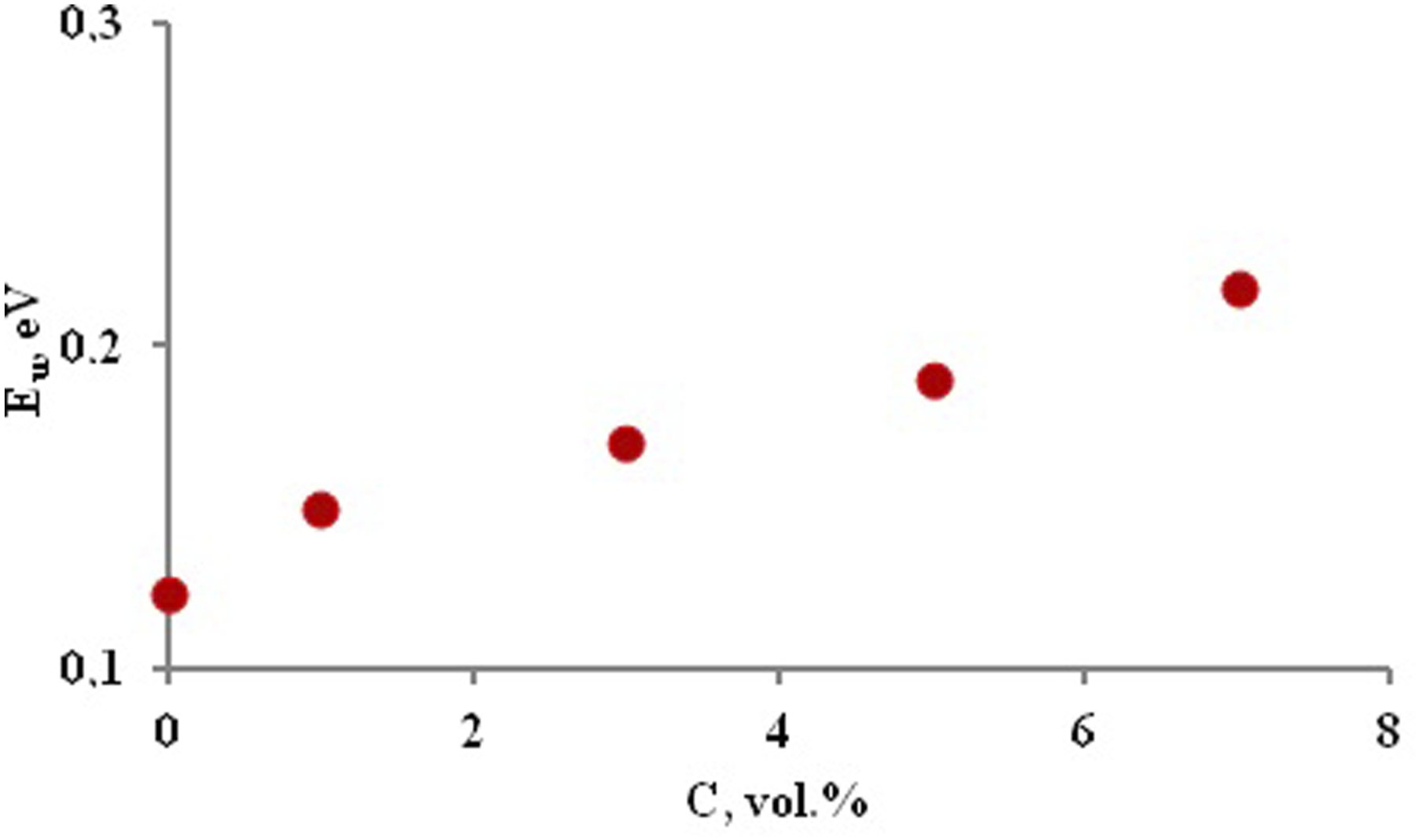
It has been observed that the value of the Urbach energy has increased from 0.123 eV to 0.218 eV with increasing filler concentration. This suggests that disorder and surface interactions increase when nanoparticles are embedded into the polymer.
Let us consider in more detail the optical absorption in a relatively transparent region of the composite. The authors of the work52,69,75–78 explain the decrease in optical transmittance of composites by the difference in the reflective index of the polymer matrix and filler. However, they do not take into account that with small nanoparticle dimensions, the Christiansen effect does not occur, in which scattering increases sharply and, accordingly, transmission decreases.79–82
Since the particle fraction is monodisperse, then:
The extinction efficiency Qext of a particle is defined as the extinction cross section Cext normalized to the particle cross section. In the case of spherical particles, it is defined as
Efficiencies of extinction Q
ext
and scattering Q
sca
of a single particle are determined by the expressions
83
:
The number of particles N per unit volume is equal to the ratio of the total volume of particles V
p
per unit volume (volume fraction) to the volume of one particle:
Taking into consideration (10-12) we can write the following relations:
The following conclusions can be drawn from the expressions (18). Firstly, as the refractive index of polypropylene varies little with wavelength, light transmission increases with increasing wavelength. Secondly, the total particle volume Vp increases with increasing volume concentration. Consequently, as the concentration rises, light transmission reduces. Thirdly, an increase in particle size leads to an increase in the scattering coefficient and consequently a decrease in transmittance. The above considerations agree well with the experimental results.
Conclusion
It is shown that nanocomposites are comprised of two distinct phases. An increase in the filler concentration leads to a decrease in crystallinity. It can be attributed to a decline in the density of chain order and a concomitant disruption of order in macromolecules.
It has been observed that as the filler concentration increases, the optical band gap decreases and the Urbach energy increases. These changes occur due to the emergence of new energy levels in the band gap, which leads to its narrowing, as well as to in disorder and interaction between nanoparticles and the matrix.
It has been demonstrated that with an increase in particle concentration and size, there is a decrease in optical transmittance in the relatively transparent region of the composite. The transmittance phenomenon is well described by the theory of light absorption and scattering in the dispersed medium of the nanocomposite.
Footnotes
Author contribution
Hijran S. Ibrahimova: investigation, formal analysis, writing original draft (equal). Tahir D.Ibragimov: methodology, writing original draft (equal).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.