Abstract
The field of tissue engineering has experienced transformative advancements with the emergence of smart scaffolding systems that not only provide mechanical support but also deliver dynamic biofunctionality to actively influence cellular behavior and tissue regeneration. In this review, the term “smart” specifically refers to polymeric foams that integrate stimuli-responsive properties — such as sensitivity to mechanical, chemical, or thermal cues — with the controlled and sustained release of bioactive factors, thereby enabling dynamic modulation of the cellular microenvironment. Owing to these multifunctional characteristics, polymeric foams have become especially versatile platforms, distinguished by their highly interconnected porous architecture, tunable mechanical properties, and ease of chemical and biological functionalization. This comprehensive review consolidates recent developments in the design and fabrication of biofunctional polymeric foams tailored to deliver controlled mechanical and electrical stimulation and biochemical cues critical for directing cell fate and promoting regenerative outcomes. Foam materials are systematically categorized based on their polymeric origin and structural features, while biofunctionalization strategies—such as peptide conjugation, growth factor integration, and gene delivery—are critically analyzed. Advanced fabrication techniques enabling stimuli-responsive behavior and patient-specific customization, including 3D/4D bioprinting and supercritical fluid processing, are highlighted for their capacity to generate adaptive scaffold microenvironments. Mechanobiological interactions, especially mechanotransduction mechanisms elicited by foam elasticity and dynamic deformation, are discussed in detail, along with emerging dual-responsive systems that simultaneously deliver mechanical and biochemical stimuli. In addition, this review addresses key translational challenges, including immunocompatibility, degradation kinetics, mechanical resilience, and regulatory considerations. Based on an integrative analysis of recent studies, we propose a strategic roadmap for the rational design of next-generation adaptive, multifunctional polymeric foams with enhanced clinical utility. Ultimately, this review emphasizes the critical role of smart polymeric foams in bridging the gap between laboratory innovation and patient-centered regenerative medicine.
Keywords
Introduction
In recent years, tissue engineering has witnessed significant advancements, characterized by a clear transition from passive, inert scaffolds to more dynamic and responsive biomaterials that actively engage with the cellular milieu. Modern scaffolds are now expected to offer a biofunctional microenvironment—one that not only promotes cell adhesion and proliferation but also delivers finely tuned biochemical and mechanical cues.
Among the wide range of biomaterial systems investigated, polymeric foams have garnered particular attention due to their structural and functional versatility. Their highly porous architecture facilitates efficient cell infiltration, oxygen exchange, and nutrient transport, all of which are essential for sustaining viable tissue throughout the scaffold. Furthermore, their low density reduces mechanical loading on the regenerating tissue. Importantly, the mechanical properties of polymeric foams—such as stiffness and elasticity—can be precisely adjusted to closely match native tissue, enabling proper mechanotransduction. Finally, their interconnected porous networks mimic the native extracellular matrix (ECM), providing an ideal platform for cell attachment, proliferation, and lineage-specific differentiation.1–3
Traditionally, polymeric foams were regarded as inert structural supports with minimal biological interaction, often limited to serving as temporary matrices for cell seeding. However, this view has been redefined by recent advances that establish these materials as multifunctional, bioactive platforms capable of complex interactions with the biological milieu. Modern foam scaffolds increasingly integrate stimuli-responsive components that allow them to detect and respond to diverse microenvironmental cues. These include physicochemical changes such as pH fluctuations, local temperature shifts, enzymatic activity variations, and externally applied stimuli like magnetic, electrical, or mechanical forces.
Incorporation of such “smart” components allows for precise, real-time modulation of scaffold properties, including targeted drug release, tunable mechanical properties, and regulation of cell fate decisions. These capabilities bring polymeric foams closer to mimicking the dynamic interplay between cells and the native extracellular matrix (ECM), ultimately improving the quality and functionality of regenerated tissues.4–6
The role of polymeric foams in replicating the mechanical environment of native tissues is particularly important in the regeneration of mechanically sensitive tissues such as skeletal muscle, cartilage, and vasculature. In these tissues, mechanical forces are not merely passive elements but serve as essential biophysical cues that orchestrate cellular behavior and maintain tissue homeostasis. Mechanotransduction—referring to how cells sense mechanical forces and translate them into biochemical activity—regulates key cellular functions including cytoskeletal organization, gene expression, and extracellular matrix remodeling.
Consequently, polymeric foams designed with tunable elasticity and capable of withstanding cyclic strain, compressive forces, or shear stress show significant promise in directing stem cells toward myogenic, osteogenic, or chondrogenic lineages. The ability to feature aligned pores and smart crosslinking strategies further amplifies these effects, boosting ECM production and supporting seamless integration with host tissues after implantation.7–10
Alongside mechanical cues, the spatiotemporal presentation of biochemical signals is now widely acknowledged as a key aspect of scaffold design. Delivery of growth factors, cytokines, and cell-adhesion peptides in a controlled and localized manner allows scaffolds to more closely mimic the body’s natural regenerative setting. This biochemical signaling often works in concert with mechanical cues to enhance cellular behavior and coordinate the complex interplay of multiple cell types involved in tissue repair. Achieving such finely tuned delivery profiles is especially important in tissues with heterogeneous composition and multi-phase healing processes, where gradients of signaling molecules must be carefully regulated to ensure orderly regeneration and functional restoration.11,12
Driving these advances has been the advancement of cutting-edge fabrication techniques capable of producing polymeric foams with finely controlled microarchitectures and functionalization. Techniques such as supercritical carbon dioxide (scCO2) foaming, solvent casting with particulate leaching, emulsion templating, and advanced 3D bioprinting allow for precise tuning of foam porosity, pore interconnectivity, and spatial distribution of bioactive agents at the nanoscale. These fabrication methods facilitate the direct incorporation of nanoparticles, therapeutic compounds, and stimuli-responsive moieties within the scaffold matrix, broadening the functional capabilities of scaffolds. This multifaceted approach improves not only cellular responses in vitro but also therapeutic efficacy in vivo, paving the way for future clinical translation.13–15
Despite these promising advances, several significant challenges remain before polymeric foams can fully realize their clinical potential. Achieving consistent and reproducible scaffold responsiveness across large-scale production is a major obstacle, complicated by the inherent complexity of multifunctional materials. Early degradation or loss of bioactivity, along with mechanical failure under physiological load-bearing conditions, pose serious risks to scaffold performance and durability. Additionally, navigating the stringent regulatory pathways required for clinical approval demands extensive safety, efficacy, and manufacturing reproducibility testing. Scaling up production while maintaining rigorous quality control and preserving biofunctional integrity adds further complexity. Careful optimization is necessary to balance diverse scaffold properties—including mechanical responsiveness, biochemical delivery, biocompatibility, and biodegradability—avoiding compromises that could undermine therapeutic effectiveness.16–18
Crucially, the concept of “smart” polymeric foams marks a paradigm shift beyond traditional scaffold design. These advanced materials actively engage in tissue repair by dynamically responding to biological signals, releasing therapeutic agents in a controlled, stimulus-dependent manner, and adjusting their mechanical properties in real time during tissue remodeling. Such active responsiveness is especially critical for regenerating complex, load-bearing tissues where tightly coordinated biochemical and biomechanical cues are essential for successful healing.
This comprehensive review critically examines the latest advances in biofunctional polymeric foams, with a special focus on materials that combine mechanical stimulation and biochemical delivery to produce synergistic effects. We will cover recent breakthroughs in materials science, cutting-edge fabrication methods, and comprehensive evaluations both in vitro and in vivo. The review also addresses the key translational challenges these innovative biomaterials face, aiming to provide insights that will guide future research and facilitate clinical translation of next-generation, responsive scaffolds designed to advance regenerative medicine.
Polymeric foams and their biomaterial classes
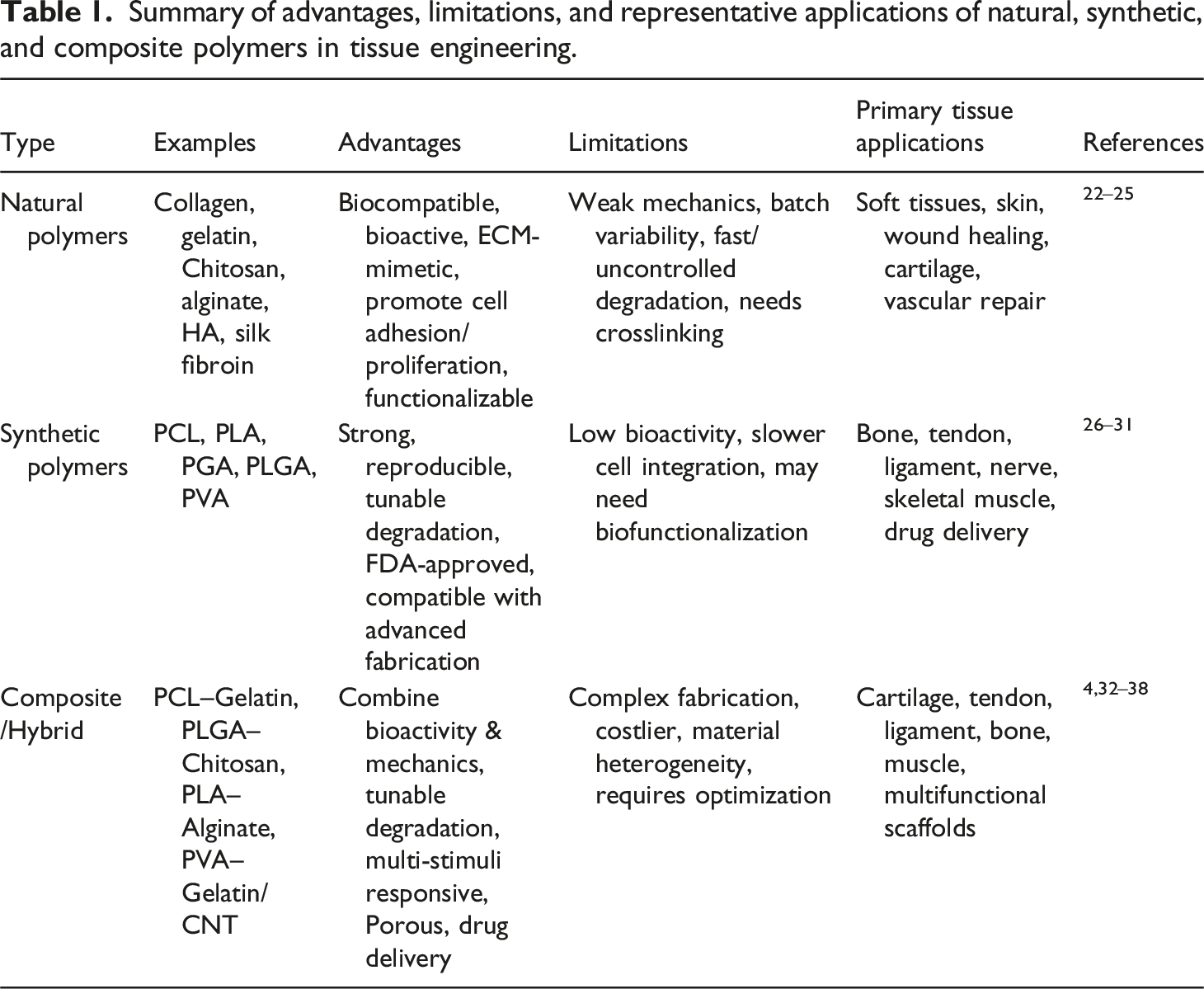
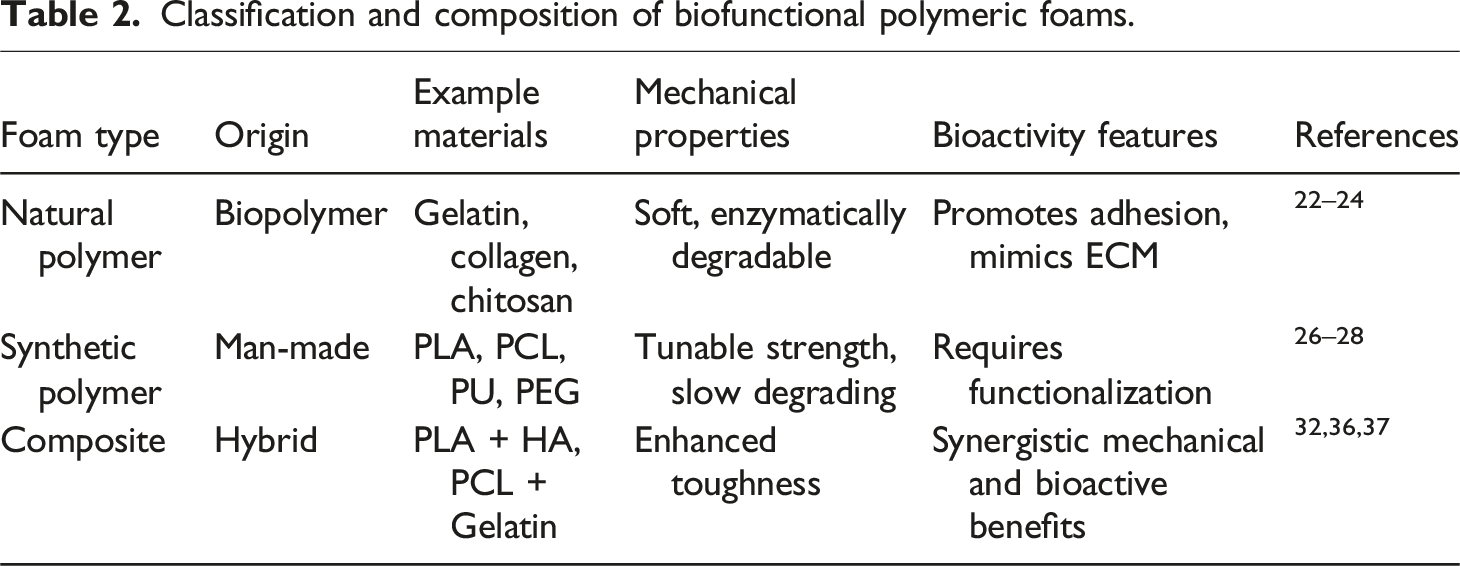
Polymeric foams used in tissue engineering form a versatile and adaptable class of biomaterials designed to replicate the structure and function of native extracellular matrices. They are mainly characterized by the chemical composition of their polymer backbones and can be broadly grouped into three main categories: natural polymers, synthetic polymers, and composite or hybrid systems. Each category possesses unique chemical, physical, and biological properties that influence its appropriateness for various clinical and research uses. This structural diversity allows polymeric foams to be custom-designed for a variety of tissues, ranging from soft connective tissues to rigid load-bearing structures. Key scaffold characteristics including porosity, mechanical strength, degradation profile, and surface bioactivity closely depend on the choice of polymer and fabrication technique.19–21
Careful and application-driven selection of polymer type is crucial for designing bioactive scaffolds that fulfill the intricate demands of tissue regeneration. An ideal polymeric foam strikes a balance between biocompatibility, mechanical stability, degradation kinetics, and responsiveness to physiological cues. Additionally, it should be compatible with fabrication methods that enable hierarchical porosity and integration of bioactive components. The following sections describe the distinctive features, benefits, and drawbacks of each class of biomaterials utilized in polymeric foams. Figure 1 provides an overview of the main classes of biofunctional polymeric foams. Classification of biofunctional polymeric foams. Polymeric foams used in tissue engineering can be categorized into four main classes: natural foams (e.g., collagen, chitosan) with inherent bioactivity; synthetic foams (e.g., PLA, PU) providing tunable mechanical strength; composite foams (e.g., PCL–gelatin hybrids) combining biofunctionality and structural integrity; and stimuli-responsive foams (e.g., magnetic PU, PNIPAAm) that dynamically respond to environmental triggers.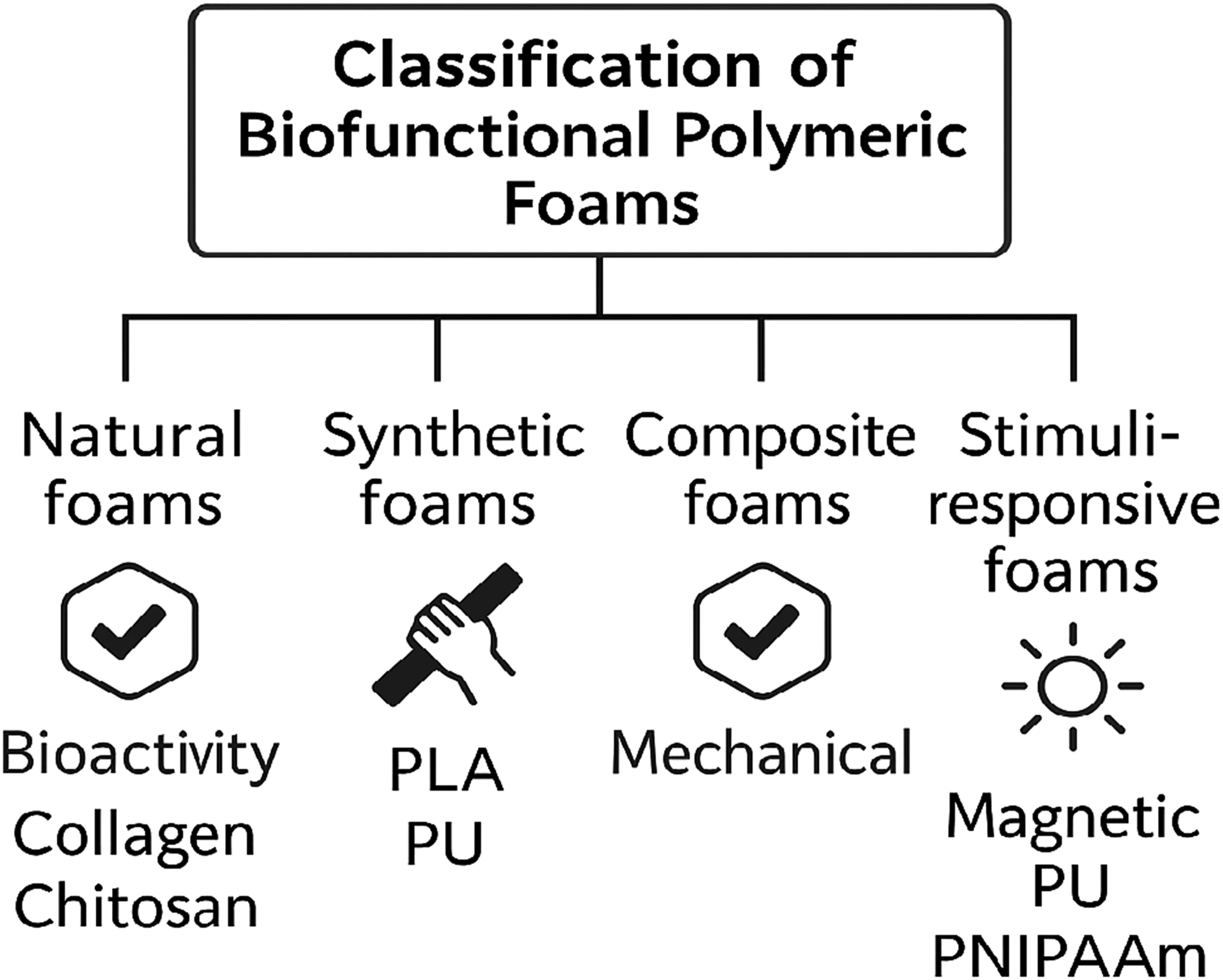
Natural polymer-based foams
Natural polymers are naturally biocompatible and bioactive, providing a biomimetic environment for cells through close imitation of native ECM components. Examples such as gelatin, collagen, chitosan, alginate, hyaluronic acid, and silk fibroin contain motifs or molecular patterns recognizable by integrins and cellular receptors, promoting cell adhesion, migration, proliferation, and ECM remodeling.22,23 Collagen and gelatin foams are extensively studied because they are abundant in human tissues and contain RGD sequences critical for integrin-mediated attachment. These foams not only sustain cell viability but also promote differentiation and the synthesis of tissue-specific matrices. Chitosan, sourced from chitin, offers added benefits including inherent antimicrobial activity, hemostatic effects, and pH responsiveness, making it suitable for wound dressings, skin substitutes, and periodontal repair. 24 Despite their biological benefits, natural polymer foams often face challenges such as weak mechanical strength, variability between batches, and rapid or uncontrolled degradation in vivo. Such limitations hinder their use in load-bearing applications unless they are reinforced. To overcome these issues, crosslinking approaches—including genipin, glutaraldehyde, and enzymatic treatments—have been employed to improve mechanical strength and regulate degradation. 25 Moreover, blending natural polymers with synthetic materials or inorganic fillers enhances mechanical durability without compromising biofunctionality, allowing their application in more challenging tissue settings.
Recent advances have also enabled functionalizing natural polymer foams with growth factors, bioactive peptides, or extracellular vesicles, boosting their therapeutic potential and shifting them from passive scaffolds to instructive biomaterials. These advancements highlight the promise of natural polymer-based foams in regenerative medicine, especially for applications demanding fast tissue integration, angiogenesis, and immune modulation.
Synthetic polymer-based foams
Synthetic polymers offer exceptional design versatility, chemical uniformity, and mechanical robustness, making them essential for creating durable and reproducible scaffolds. Frequently used synthetic polymers in tissue-engineered foams include polycaprolactone (PCL), polylactic acid (PLA), polyglycolic acid (PGA), and their copolymers such as poly(lactic-co-glycolic acid) (PLGA).26–28 These polymers have FDA approval and are well-characterized, enabling predictable degradation rates and scalable manufacturing processes. PCL is notable for its slow degradation, which can span several years, along with high elasticity, making it suitable for orthopedic implants, nerve conduits, and skeletal muscle regeneration where prolonged support is essential. PLA and PLGA degrade more rapidly, making them ideal for soft tissue repair, controlled drug release, and temporary scaffolds designed to resorb within weeks to months. 29
Advanced fabrication methods such as electrospinning, solvent casting/particulate leaching, and 3D printing offer the ability to fine-tune scaffold architecture and porosity. In particular, supercritical CO2 foaming has proven effective in tuning pore size and structure in biodegradable polymers such as poly(vinyl alcohol), with detailed experimental and numerical studies demonstrating bubble nucleation and growth behavior under various Thermodynamic conditions. 30 This has enabled synthetic foams to be used in complex tissue systems such as vascular grafts, tendon-to-bone interfaces, and multi-layered skin constructs, where gradients in mechanical stiffness and cellular composition are crucial. Additionally, biodegradable PVA–gelatin/CNT composite foams have shown tunable morphology and mechanical strength suitable for tissue engineering applications. 31
Composite and hybrid foams
Composite and hybrid foams embody a synergistic strategy that combines the inherent bioactivity of natural polymers with the customizable mechanical properties of synthetic materials. Functional and hybrid polymeric foams have become increasingly important in tissue engineering, serving as versatile platforms that blend the natural bioactivity of polymers like chitosan, alginate, and carboxymethyl cellulose with the mechanical support of synthetic polymers such as polyvinyl alcohol. The inclusion of functional additives—such as hydroxyapatite, graphene-modified clay, and iron oxide nanoparticles—strengthens the scaffolds, improves porosity, and enhances cell compatibility. These structures offer controlled biodegradability, interconnected pores, and the potential for targeted delivery of drugs or bioactive molecules. By closely mimicking native tissue, they promote effective regeneration and open opportunities for multifunctional applications, ranging from drug delivery and tissue repair to environmental remediation.32–35 Often engineered to incorporate diverse functionalities within one scaffold, these foams deliver dynamic responsiveness, robust mechanical performance, and enhanced cellular compatibility essential for complex tissue regeneration. Typical examples such as PCL–gelatin, PLGA–chitosan, and PLA–alginate composites synergize biodegradability, mechanical reinforcement, and biological cues within an integrated matrix. These scaffolds are particularly valuable for engineering tissues like cartilage, tendon, ligament, and intervertebral discs, which endure dynamic mechanical loads and require seamless integration with neighboring tissues. 36 Moreover, these composite foams frequently embed functional fillers that confer bioactivity and enable responsiveness to external triggers. Notable fillers encompass hydroxyapatite (HA) to enhance osteoconductivity in bone repair, graphene oxide or carbon nanotubes to improve electrical conductivity and mechanical strength, and magnetic nanoparticles (such as Fe3O4) facilitating remote actuation, magnetic targeting, or hyperthermia therapy. 37 Magnetically modified hydrogels incorporating biochar exhibit superior adsorption and enable remote manipulation, serving as prototypes for magneto-responsive biomaterials. 38
Summary of advantages, limitations, and representative applications of natural, synthetic, and composite polymers in tissue engineering.
Porosity and Microarchitecture
The pore structure of polymeric foams plays a crucial role in their regenerative performance. Factors such as pore size, shape, interconnectivity, and spatial distribution directly impact cell migration, angiogenesis, nutrient transport, and waste removal. Optimizing these parameters is essential to mimic the native extracellular matrix environment and achieve uniform tissue formation. Research indicates that pore diameters ranging from 100 to 300 µm are ideal for promoting osteogenesis and myogenesis, as they support both vascular infiltration and myoblast alignment. Conversely, smaller pores (<100 µm) are better suited for neural tissue, skin, and endothelial networks, where high cell density and complex signaling are necessary. 39
Classification and composition of biofunctional polymeric foams.
Fabrication techniques and structural features
The method used to fabricate polymeric foams is crucial in shaping their final structural stability, mechanical behavior, and biological performance. These attributes fundamentally influence the scaffold’s capacity to support cell growth, direct tissue regeneration, and seamlessly integrate with surrounding tissues. Over the past decade, remarkable progress in fabrication technologies has expanded the possibilities for designing polymeric foams with high porosity, interconnected networks, and stimuli-responsive features tailored to meet the specific mechanical and biological demands of different tissues such as bone, cartilage, skin, and neural tissues.41–43
Striking the right balance between porosity, pore size distribution, interconnectivity, and mechanical strength continues to be a core challenge. Porosity plays a key role in facilitating nutrient and oxygen transport as well as waste elimination, while mechanical properties must align with or support those of the native tissue to prevent early scaffold failure or hindered regeneration. Additionally, incorporating dynamic responsiveness to environmental cues within scaffold architecture is gaining growing importance in the development of next-generation biomaterials.
The following sections provide an overview of essential fabrication techniques for polymeric foams, discussing their underlying principles, benefits, drawbacks, and recent advancements.
Solvent casting and particulate leaching
Solvent casting combined with particulate leaching (SC/PL) is among the oldest and most commonly used methods to fabricate porous scaffolds. The process starts by dissolving a polymer in a suitable solvent, then uniformly mixing this solution with porogens like sodium chloride (NaCl), sugar crystals, or other soluble particles. The mixture is cast into molds, and later, the porogens are leached out by soaking the scaffold in water or another solvent. This creates a porous network where the porogens previously resided, forming interconnected voids. 44 SC/PL offers several benefits, including simplicity, cost-effectiveness, and fairly good control over pore size and overall porosity by adjusting the porogen’s size and concentration. However, the method has some drawbacks limiting its wider use. For example, pore interconnectivity can be less than ideal, which restricts cell movement and blood vessel infiltration. Residual solvent traces may remain trapped in the scaffold, posing potential cytotoxicity risks. Moreover, batch-to-batch reproducibility can be inconsistent, resulting in variability in scaffold characteristics and performance. 45
Recent advancements have targeted these challenges through smarter selection and design of porogen materials. Optimizing porogen size and shape has enhanced pore interconnectivity and mechanical strength. Adding freeze-drying steps after leaching improves scaffold uniformity and helps remove leftover solvents. The use of dual-solvent systems, involving two solvents with different volatilities or solubilities, has been tested to better control pore structure and shorten processing times, thereby boosting mechanical robustness and consistency. 46 Despite these improvements, SC/PL remains most appropriate for simpler foam structures and less mechanically demanding applications.
Supercritical CO2 foaming
Supercritical carbon dioxide (scCO2) foaming has gained considerable attention as a solvent-free fabrication technique, especially for biomedical applications that demand high purity and excellent biocompatibility. In this method, polymers are saturated with CO2 under supercritical conditions—characterized by high pressure and moderate temperature—allowing CO2 molecules to evenly diffuse throughout the polymer matrix. A sudden drop in pressure triggers phase separation, creating micro-to nanoscale pores as the CO2 escapes, resulting in a highly porous foam structure. 47 This technique offers several key advantages, including the complete elimination of harmful organic solvents, the ability to finely tune pore size and distribution by adjusting temperature, pressure, and depressurization speed, and the production of scaffolds free from residual toxic chemicals. These benefits make scCO2 foaming especially well-suited for fabricating biocompatible scaffolds intended for clinical use. 48 Process parameters can be carefully controlled to customize the foam’s morphology and mechanical properties. For example, higher saturation pressures generally increase CO2 solubility and nucleation density, leading to smaller pores and greater porosity. 49 Furthermore, scCO2 foaming can be combined with other techniques, such as particulate leaching, to produce composite scaffolds with hierarchical porosity.
Despite these advantages, scCO2 foaming still faces challenges, including limited solubility of some polymers in CO2 and difficulties scaling up production for large or complex shapes. Nevertheless, ongoing research is actively working to broaden the range of compatible polymers and optimize processing conditions for industrial-scale applications.
Emulsion templating and PolyHIPEs
Emulsion templating, especially using high internal phase emulsions (HIPEs), presents a novel and effective approach to creating ultra-porous polymeric foams with exceptionally high porosity—often above 80%. This technique involves a concentrated emulsion where the internal phase volume fraction exceeds 74%, serving as a template. Within this system, droplets of the internal phase are dispersed throughout a continuous polymerizable phase. Once polymerization solidifies the continuous phase, the scaffold’s droplet network is preserved, producing an open-cell foam with interconnected pores. 50 PolyHIPEs (polymerized HIPEs) have garnered significant attention in tissue engineering because their highly interconnected pore structures facilitate efficient cell infiltration and nutrient exchange. Materials such as methacrylated gelatin and poly(ethylene glycol) diacrylate (PEGDA) have been successfully utilized to fabricate PolyHIPE foams tailored for regenerative therapies targeting liver, vascular, and cartilage tissues, where both porosity and mechanical flexibility are essential.51,52
One of the major strengths of this method is its versatility—pore size, interconnectivity, and mechanical properties can be precisely adjusted by modifying emulsion composition, surfactant type, and polymerization conditions. Nonetheless, some challenges remain, including difficulties in completely removing surfactants, maintaining foam uniformity at larger scales, and ensuring thorough polymerization without leftover monomers. Ongoing progress in surfactant chemistry and polymerization techniques continues to tackle these issues, enhancing the biocompatibility and reproducibility of PolyHIPE scaffolds. Figure 2 illustrates the main fabrication methods and how they influence scaffold architecture. Fabrication techniques and structure–property relationships of porous polymeric foams. This schematic illustrates major methods used in the fabrication of porous polymeric foams, including particulate leaching, gas foaming, and emulsion templating. These techniques govern the resulting pore morphology, which in turn influences mechanical performance and bioactivity of the scaffold. The ability to modulate pore size, interconnectivity, and structural integrity is critical for tailoring cell infiltration, nutrient transport, and tissue integration in regenerative applications.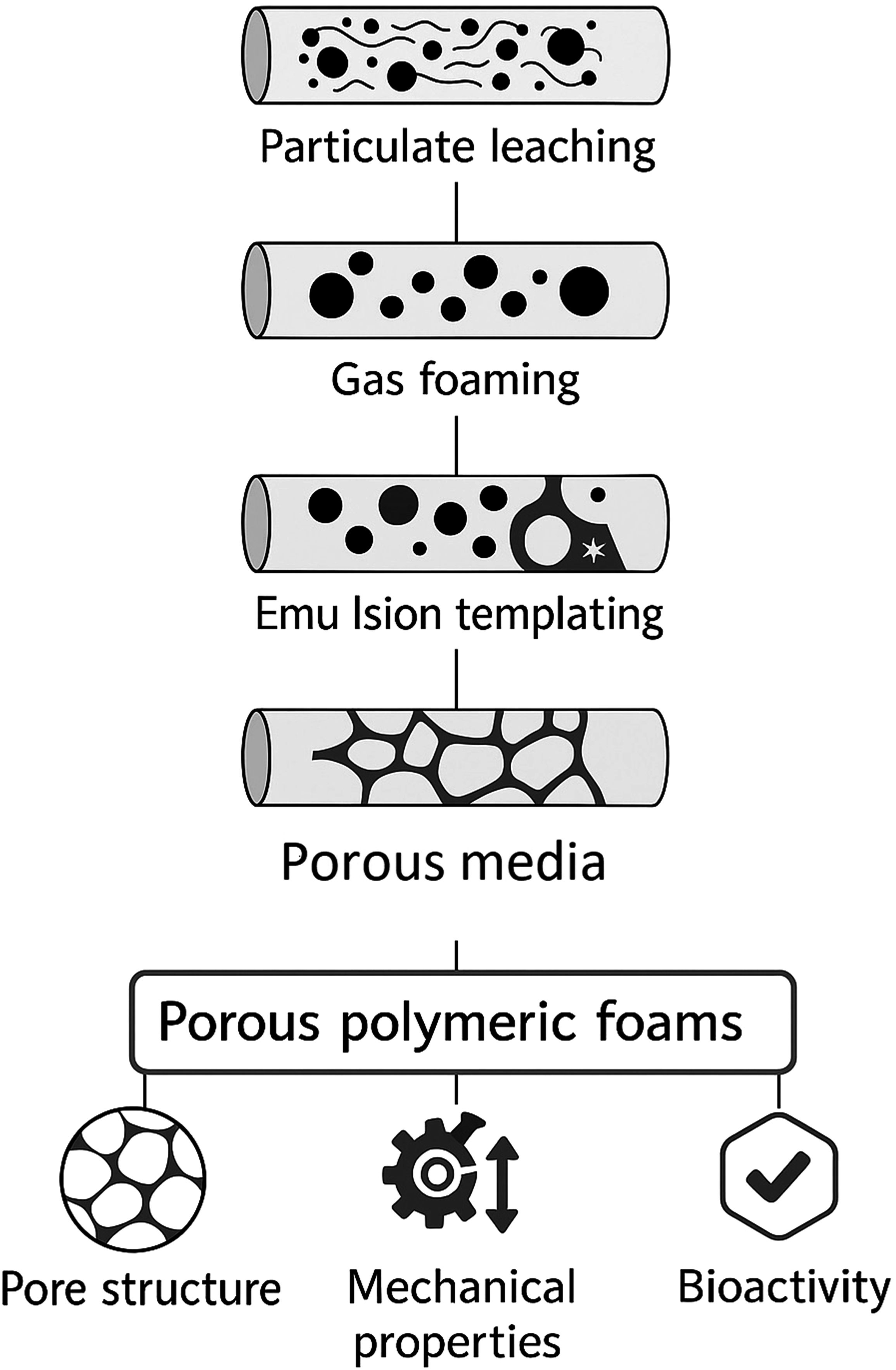
3D printing and additive manufacturing
3D bioprinting has truly transformed scaffold fabrication by offering unparalleled precision in designing scaffold architecture, geometry, porosity, and even the spatial placement of cells or bioactive molecules. Additive manufacturing methods—like extrusion-based printing, stereolithography (SLA), and digital light processing (DLP)—enable the creation of highly tailored foam-like constructs that can finely control pore size gradients, anisotropy, and complex shapes. This allows these scaffolds to closely mimic the hierarchical structure of natural tissues. 53 Building on this, 4D printing introduces the exciting possibility of scaffolds that change shape or properties over time in response to environmental cues such as pH, temperature, or light. This dynamic behavior means “smart” foams can adapt after printing—for example, by increasing pore size during early healing phases or stiffening as new tissue forms.54,55
While these technologies hold great promise, they also face challenges such as optimizing bioink formulations, overcoming resolution limits, and scaling up production. Striking the right balance between mechanical stability, porosity, and cell viability requires careful tuning of printing conditions and materials.
Freeze-drying and cryostructuring
Freeze-drying, also known as lyophilization, is a long-established technique for creating porous foams. It works by freezing a polymer solution and then sublimating the ice crystals under reduced pressure. The size, shape, and orientation of the ice crystals formed during freezing directly determine the foam’s pore structure. By carefully adjusting the freezing rate, polymer concentration, and degree of crosslinking, researchers can precisely control the porosity and mechanical traits of the scaffold to match specific tissue needs. 56 Scaffolds produced through freeze-drying are typically soft and hydrated, making them excellent environments for cell attachment and growth—especially in delicate tissues like nerves, skin, and eyes. These scaffolds closely replicate the hydrated, porous nature of the natural extracellular matrix, providing an optimal setting for tissue repair and regeneration.57,58
Biofunctionalization strategies for polymeric foams.
Mechanical activation and stimuli-responsive behavior
In recent years, scaffold engineering has seen a major breakthrough with the shift from passive biomaterials to advanced, smart polymeric foams that can dynamically respond to a variety of external stimuli. These innovative materials are designed to detect and react to environmental signals—such as mechanical forces, magnetic fields, temperature changes, electrical impulses, and chemical variations—by adjusting their physical properties like shape, stiffness, or biochemical activity in real time. This ability to respond is essential because it allows the scaffolds to better mimic the complex and ever-changing environment of native tissues, greatly improving their effectiveness in supporting and directing tissue regeneration and healing processes.59–61 Such smart materials pave the way for adaptive tissue engineering platforms that actively contribute to regeneration, rather than just acting as passive structural frameworks.
Mechanotransduction in cell regulation
Mechanotransduction is a fundamental biological process through which cells sense and transform mechanical forces—like stretching, compression, or shear stress—into biochemical signals inside the cell that regulate vital functions. This process is pivotal in tissue development, homeostasis, and repair, orchestrating vital events such as stem cell differentiation, ECM production, and new blood vessel formation.62–64 Designing polymeric foams with adjustable mechanical properties—like elasticity, cyclic deformation, and direction-dependent stiffness—allows precise control of the mechanical signals given to the cells. Studies have shown that scaffolds exposed to controlled mechanical forces can influence stem cell fate, promoting differentiation toward bone, muscle, and cartilage lineages in both in vitro and in vivo settings. Furthermore, designing foam structures with aligned pores and adding stimuli-responsive crosslinkers enhances these mechanobiological effects, improving tissue organization, increasing ECM deposition, and ultimately resulting in better tissue regeneration.65,66 These results highlight the key role of mechanically active scaffolds as dynamic controllers of cell behavior and tissue formation.
Magnetically responsive foams
Magnetically responsive polymeric foams are created by embedding magnetic nanoparticles, typically superparamagnetic iron oxide, into the polymer network, allowing remote and non-invasive activation via external alternating magnetic fields. When exposed to these fields, scaffolds deform or vibrate in a controlled way, delivering precisely adjustable mechanical forces directly to encapsulated or infiltrating cells without direct contact.67,68 This method has great potential especially in skeletal muscle engineering, where cyclic forces from magnetic activation mimic natural muscle contractions, boosting muscle cell differentiation, fiber growth, and healing. Similarly, magnetic compression stimulates cartilage cells and promotes matrix production, helping to restore functional cartilage.69,70 Besides mechanical stimulation, magnetic foams can be combined with hyperthermia therapy or serve as vehicles for targeted drug delivery, expanding their use in regenerative medicine and cancer therapy.
Electroactive and piezoelectric systems
Electroactive foams incorporate conductive polymers such as polypyrrole or PEDOT:PSS, or piezoelectric materials like polyvinylidene fluoride (PVDF) and zinc oxide, which generate localized electrical potentials in response to mechanical deformation. These bioelectric cues closely mimic the native electrical microenvironment found in excitable tissues like nerves, cardiac muscle, and skeletal muscle, providing novel strategies to enhance tissue regeneration through electrical stimulation.71,72 Some electroactive foams can be injected in a minimally invasive way and solidify in place, forming self-assembled networks that deliver simultaneous mechanical and electrical stimuli. This combined stimulation has been shown to synergistically promote cell proliferation, lineage-specific differentiation, and better functional integration of engineered tissues.
73
The ability to precisely adjust electrical conductivity and piezoelectric response further allows customization for specific tissue types and therapeutic purposes. Figure 3 illustrates key cellular responses to mechanical, electrical, and magnetic stimuli in biofunctional foams. Cellular responses to different physical stimuli in biofunctional polymeric foams. (a) cyclic mechanical strain aligns myotubes and enhances muscle cell fusion. (b) electrical stimulation promotes neural differentiation and axonal outgrowth. (c) magnetic actuation induces cytoskeletal organization and directional elongation. These stimuli-responsive mechanisms synergistically improve scaffold–cell interactions in engineered soft tissues.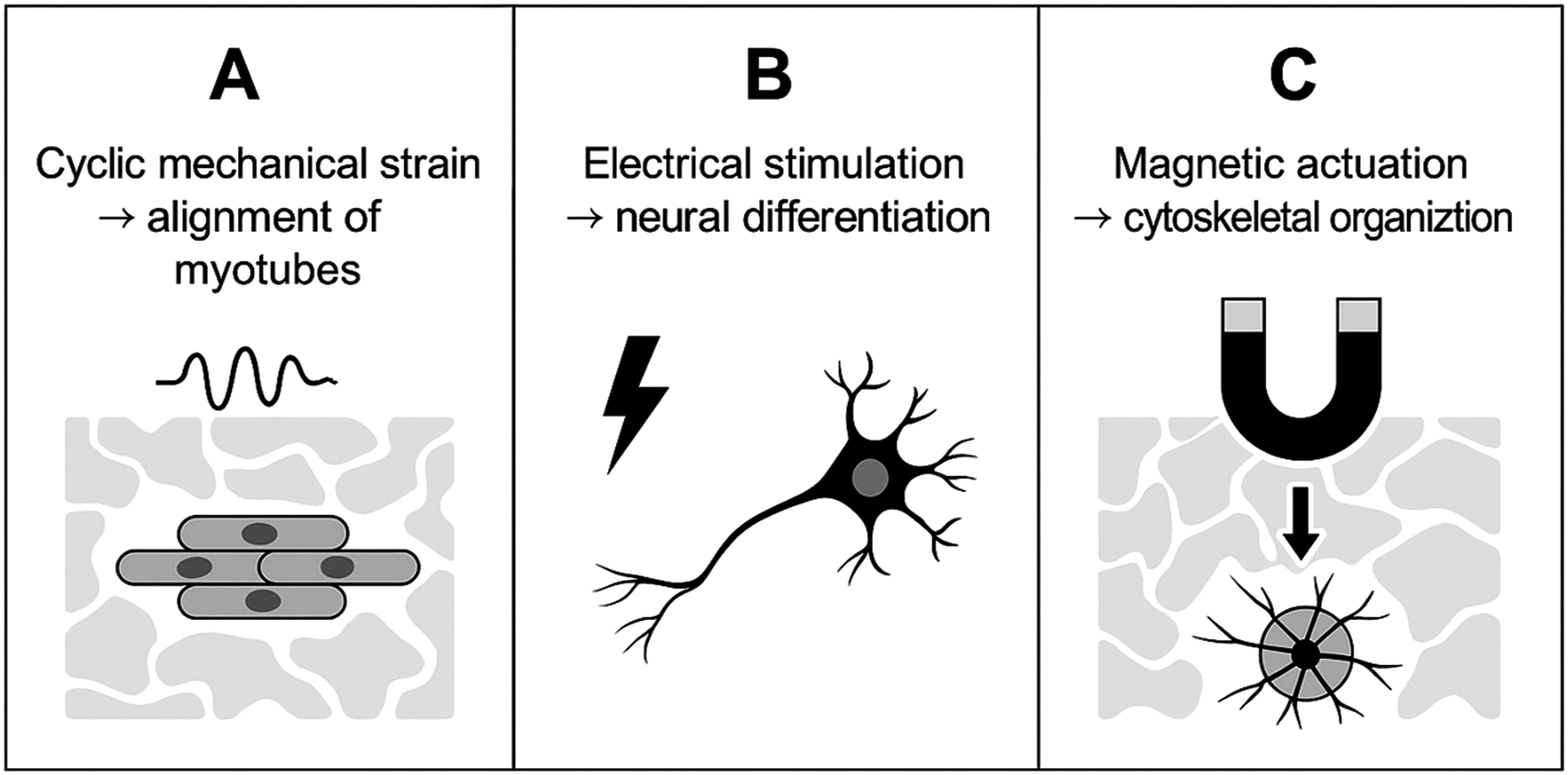
Thermo-, pH-, and enzyme-responsive foams
Thermo-responsive polymeric foams, such as those based on poly(N-isopropylacrylamide) (PNIPAAm), exhibit reversible volumetric changes near physiological temperatures. This property can be leveraged for minimally invasive implantation, where foams are delivered in a compact form and expand once reaching body temperature, ensuring a precise fit in irregular tissue defects. Additionally, these foams enable controlled drug release triggered by temperature changes, improving therapeutic effectiveness while reducing systemic side effects. 74
pH-responsive foams utilize the acidic microenvironments typical of inflamed or tumor tissues to trigger scaffold degradation or controlled payload release selectively at target sites, facilitating localized therapy with minimized off-target toxicity. 75 Enzyme-responsive foams are designed to degrade specifically in the presence of enzymes like matrix metalloproteinases (MMPs), which are secreted by cells during tissue remodeling and regeneration. This targeted degradation synchronizes scaffold resorption with new tissue formation, promoting seamless integration and natural healing. 76 Together, these stimuli-responsive platforms provide a sophisticated combination of mechanical and biochemical signals finely tuned to meet the dynamic needs of complex biological environments, thereby optimizing tissue repair and regeneration.
Scaffold types and biological responses to mechanical and physical cues.

Translational challenges and clinical perspectives
Despite significant advancements and encouraging preclinical results for polymeric foams in tissue engineering and regenerative medicine, moving these technologies from the lab. bench to clinical practice remains a complicated journey filled with many obstacles. The process of translating these innovative scaffolds into standard clinical treatments faces numerous technical, biological, regulatory, and manufacturing challenges. These range from ensuring consistent biocompatibility and managing immune responses in patients to overcoming issues of large-scale manufacturing, maintaining long-term functionality inside the body, and meeting rigorous regulatory standards required for clinical use.77,78
Biocompatibility and immunological response
Many of the base polymers commonly used in foam fabrication—like polycaprolactone (PCL), polylactic acid (PLA), and gelatin—have well-established safety records and are approved by agencies such as the FDA or classified as Generally Regarded as Safe (GRAS). However, adding functional elements like nanoparticles, synthetic crosslinkers, or chemical initiators can significantly change how these materials interact biologically. While these additives improve mechanical strength or enable responsiveness to stimuli, they may also unintentionally provoke immune reactions, including cell toxicity, activation of inflammatory pathways, or chronic inflammation. Such immune responses can interfere with scaffold integration and delay or even prevent effective tissue healing and regeneration.79,80 To overcome these immunological challenges, advanced surface modification techniques have been developed. For example, PEGylation can provide “stealth” properties to evade immune detection, coating scaffolds with proteins derived from the extracellular matrix (ECM) improves compatibility with body tissues, and embedding bioinert hydrogels helps reduce macrophage activation and fibrosis around the implant. These strategies have shown promising results in minimizing adverse immune effects. Nonetheless, the complex and dynamic interactions between the host immune system and these multifunctional, degradable biomaterials are still not fully understood. As scaffold designs grow more sophisticated, further detailed immunological studies and long-term in vivo evaluations are critical to fully understand these processes and ensure the safety and effectiveness of these scaffolds in clinical settings. 81
Mechanical stability and functional longevity
A key challenge in translating polymeric foams for use in mechanically active tissues lies in ensuring their mechanical integrity and long-term functionality. These scaffolds must endure complex physiological loads, repetitive stresses, and subtle micro-movements over extended durations without failing prematurely. Common mechanical issues such as fatigue, creep deformation, and gradual loss of elasticity can severely compromise scaffold performance, leading to diminished structural support and hindered tissue regeneration. 82 To address these challenges, researchers have explored innovative solutions involving reinforced composite structures and advanced material systems. Notably, shape-memory polymers and interpenetrating polymer networks (IPNs) have emerged as promising candidates, as they offer a balance between preserving elasticity and enabling controlled, predictable degradation. These smart materials are designed to sustain mechanical function throughout critical tissue remodeling phases, progressively transferring the mechanical load to the newly formed tissue. Such advancements are essential to guarantee that scaffolds continuously provide mechanical stimuli while facilitating effective native tissue regeneration until full functional recovery is achieved.83,84
Degradation control and drug release kinetics
Another significant challenge in translation is achieving precise control over scaffold degradation alongside the kinetics of drug release. Many polymeric foams are designed to perform dual roles—not only providing structural support but also acting as carriers for the controlled delivery of therapeutic agents, such as growth factors, anti-inflammatory drugs, or genetic materials—adding complexity to their design. Successful tissue regeneration heavily depends on the careful timing of biochemical signals with cellular responses; therefore, sustained and predictable drug release profiles are critical. An initial burst release or prolonged retention of bioactive molecules can result in suboptimal therapeutic outcomes or unwanted side effects. 85 In this study, porous silk protein-based memory foams were investigated as a novel platform for the sustained and localized release of ciprofloxacin. The incorporation of protease XIV enabled regulation of scaffold degradation, while drug release remained governed by solubility and diffusion characteristics. Results demonstrated that ciprofloxacin release could be tuned from one day to over 200 days, while scaffold degradation could be modulated by up to 80% within two weeks. These findings highlight the strong potential of this system for the development of advanced drug delivery devices and other implantable medical applications. 86
The degradation behavior of scaffolds is strongly influenced by local microenvironmental factors, including variations in pH, enzymatic activity, and fluid shear stress, which can differ substantially between tissue types or pathological conditions. These variables introduce complexity and unpredictability in controlling drug release kinetics. To overcome these issues, computational modeling combined with advanced real-time in situ imaging techniques have become powerful tools. Such approaches enable the prediction of scaffold behavior within living tissues, allowing for rational design and precise adjustment of degradation rates and drug delivery profiles tailored to specific clinical needs.87,88
Manufacturing, sterilization, and storage
Scaling up the production of polymeric foams from laboratory prototypes to clinically viable devices that comply with Good Manufacturing Practices (GMP) poses significant manufacturing challenges. Ensuring batch-to-batch consistency, strict material traceability, and uniform physicochemical properties across large-scale production is essential to meet clinical quality standards.
Many stimuli-responsive components incorporated into these foams—such as enzymes, growth factors, or thermolabile bioactive fillers—are sensitive to conventional sterilization methods. For example, gamma irradiation, a commonly used sterilization technique, can degrade polymeric scaffolds or damage their functional groups, negatively impacting scaffold performance and safety.
89
As a result, alternative sterilization methods like ethylene oxide treatment, supercritical carbon dioxide sterilization, and low-temperature plasma sterilization are actively being explored to preserve scaffold integrity while ensuring sterility. Moreover, the stability of polymeric foams during storage and transport is critical. Scaffolds containing sensitive biological agents or designed for stimuli-responsive behavior require carefully optimized packaging and preservation protocols. These precautions are crucial to prevent degradation, loss of bioactivity, or changes in physical properties before implantation, thereby ensuring clinical efficacy and reliability.
90
A roadmap summarizing the translational challenges for clinical application is illustrated in Figure 4. Translational challenges and clinical roadmap for smart polymeric foams in tissue engineering. This roadmap illustrates the sequential stages required for the clinical translation of biofunctional polymeric foams, starting from laboratory research and scaffold design through preclinical validation, manufacturing scalability, and regulatory approval, culminating in clinical integration. Key challenges at each step include ensuring reproducibility, sterilizability, cost-efficiency, and compliance with regulatory frameworks. Addressing these issues is vital for achieving real-world application of smart scaffolds in regenerative medicine.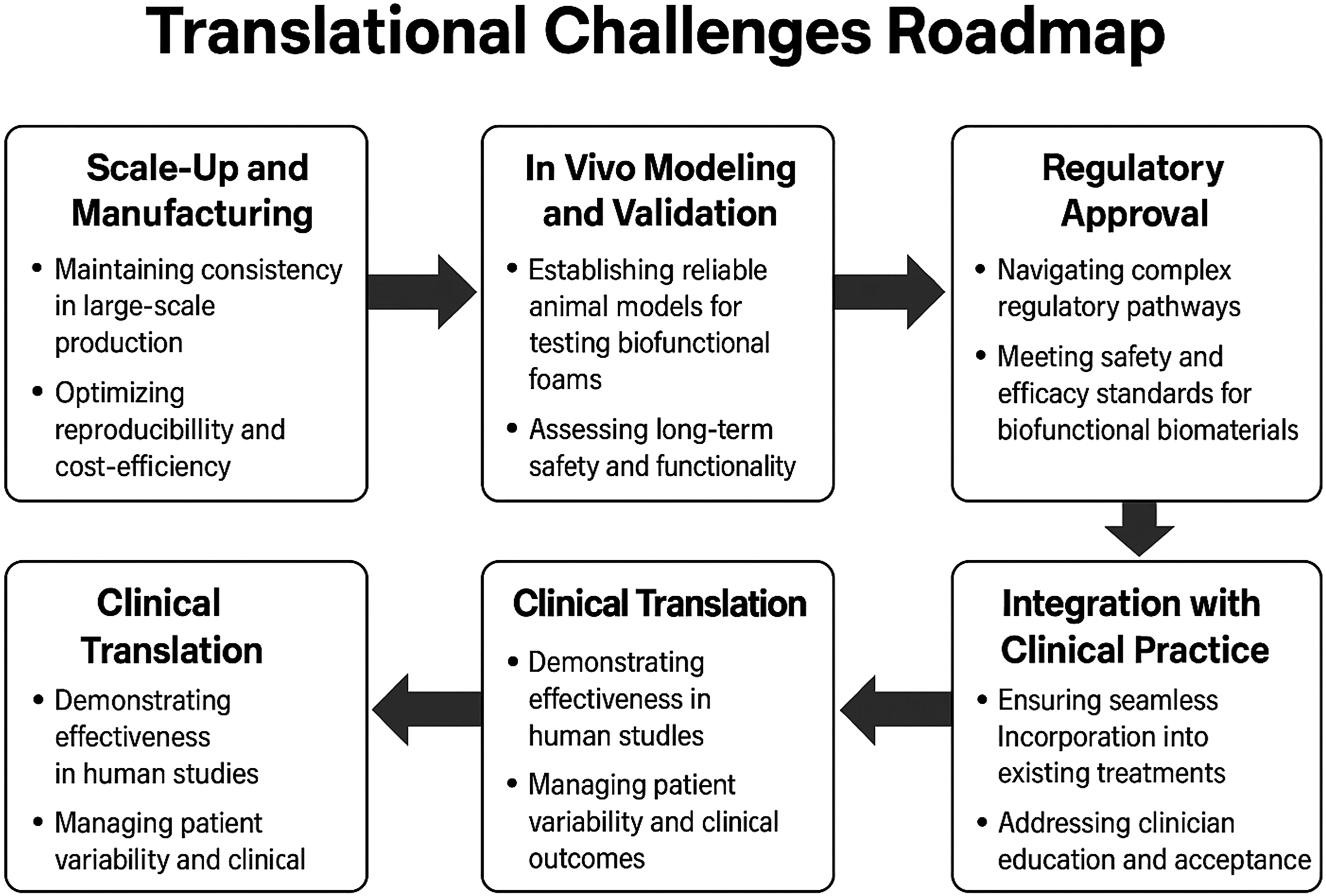
Regulatory and ethical landscape
The multifunctional and complex nature of polymeric foams—often combining device, biologic, and drug components—poses unique regulatory challenges. The classification, regulatory pathways, and approval processes set by agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) are still evolving and can be quite complex. This is especially true for scaffolds that include smart feedback mechanisms, biosensors, or live cell components, which tend to blur the traditional lines between medical devices, biologics, and combination products. 91
Translational barriers and roadmap for clinical adoption of biofunctional polymeric foams.

Future perspectives and conclusion
The evolving landscape of tissue engineering is rapidly shifting from the use of static scaffolds toward highly dynamic and interactive biomaterials capable of sensing, responding, and adapting to the intricacies of the cellular microenvironment. Among the diverse scaffold systems under investigation, polymeric foams have emerged as particularly promising due to their structural adaptability, customizable mechanical performance, and broad potential for chemical and biological functionalization. These features uniquely position polymeric foams as leading candidates in the development of intelligent regenerative platforms that move beyond passive support to actively engage with the healing process.94,95
Smart and transformative strategies
A particularly exciting direction lies in the development of four-dimensional (4D) polymeric foams—materials designed to undergo temporally programmed changes in form, stiffness, or biochemical cues in response to environmental stimuli such as pH fluctuations, thermal changes, or mechanical loading. This time-resolved adaptability enables the construction of biointerfaces that more accurately emulate the dynamic nature of tissue development and repair. For instance, a foam scaffold may exhibit initial compliance to promote cell infiltration and extracellular matrix deposition during early healing stages, followed by gradual stiffening to support tissue maturation—thereby recapitulating native morphogenetic transitions. 96
Simultaneously, significant strides have been made in integrating vascularized networks into foam constructs via advanced bioprinting techniques. This breakthrough directly addresses the critical limitation of nutrient diffusion and waste removal in large or complex tissue architectures, substantially improving the viability and functionality of embedded cells. The synergy between engineered vasculature and porous foam matrices expands the scope of clinical applications, particularly for thick or metabolically demanding tissues.
Moreover, the incorporation of stimuli-responsive nanocomponents and bioelectrical interfaces within foam scaffolds has opened promising avenues for applications in neural tissue engineering, musculoskeletal repair, and cardiac regeneration. The integration of embedded biosensors further elevates scaffold functionality by enabling real-time, in situ monitoring of cellular activity, scaffold degradation, and mechanical loading. These innovations collectively represent a leap toward the development of closed-loop, smart regenerative systems capable of autonomously modulating therapeutic responses, ultimately enhancing clinical precision and patient outcomes.97–99
Personalized and precision scaffolds
As precision medicine continues to advance, the demand for patient-specific and highly customizable scaffolds is rapidly increasing. Polymeric foams are especially well-suited to meet these demands due to their compatibility with advanced fabrication technologies, including 3D and 4D bioprinting. These techniques combine patient-derived imaging data with AI-based computational modeling to design scaffolds that closely match individual anatomical, physiological, and pathological parameters. This level of customization enhances fit, mechanical performance, and biological integration, thereby improving therapeutic outcomes.100,101
Moreover, polymeric foams can be personalized further by incorporating autologous cells, bioactive extracellular vesicles (such as exosomes), or customized growth factor cocktails. These components provide localized and potent stimulation, supporting the repair of complex defects in bone, cartilage, skin, and other tissues, while minimizing the risk of immune rejection or adverse effects. Such personalized scaffolds represent a significant advancement toward safe, effective, and patient-centered regenerative solutions. 102
Sustainability and biofabrication
Alongside clinical and technological advancements, environmental sustainability has emerged as a critical consideration in scaffold development. The biomedical field is increasingly emphasizing the adoption of green biomaterials that are renewable, biodegradable, and inherently bioactive to support tissue regeneration. Notable examples include marine-derived polysaccharides, bacterial cellulose, and other naturally sourced polymers, which offer reduced ecological impact without compromising functional performance. 103 Additionally, eco-conscious fabrication techniques such as supercritical carbon dioxide (CO2) processing and solvent-free manufacturing are gaining attention. These methods minimize the use of hazardous chemicals, reduce energy consumption, and align scaffold production with broader environmental and sustainability objectives. Effectively integrating these ecological priorities with the rigorous performance and safety standards of biomedical devices will be essential for ensuring the long-term clinical and commercial viability of polymeric foams. 104
Conclusion
In summary, polymeric foams represent a highly versatile and promising class of biomaterials for regenerative medicine. By integrating favorable structural characteristics with advanced stimuli-responsive functionalities and intrinsic biocompatibility, these scaffolds have evolved beyond static frameworks. They now actively engage with biological systems through mechanical, electrical, and biochemical cues, thereby enhancing the natural processes of tissue regeneration.
Despite these advances, significant challenges remain—particularly in the areas of scalable, reproducible manufacturing; navigating complex rfegulatory landscapes; and conducting rigorous long-term in vivo studies to validate safety and efficacy. Nevertheless, the convergence of smart biomaterials and advanced additive manufacturing techniques provides a clear roadmap for the development of next-generation regenerative therapies. Ultimately, the successful clinical translation of polymeric foam scaffolds will depend on sustained interdisciplinary collaboration among materials scientists, biomedical engineers, clinicians, regulatory bodies, and bioethicists. With a foundation built on strong scientific evidence, ethical responsibility, and technological innovation, these intelligent, adaptive biomaterials have the potential to transform regenerative medicine and significantly improve patient outcomes worldwide.
Footnotes
Acknowledgements
The authors declare that no specific funding or external support was received for the preparation of this work.
Authors’ contributions
The authors confirms sole responsibility for all aspects of the manuscript, including conceptualization, literature review, writing, editing, and submission.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of AI and AI-assisted technologies in the writing process
During the preparation of this manuscript, the authors used ChatGPT (OpenAI, 2025 version) to assist with grammatical refinement and improvement of scientific language clarity. The tool was used solely to enhance the readability and linguistic quality of the text. All content was carefully reviewed and edited by the authors, who take full responsibility for the accuracy and integrity of the final manuscript.
