Abstract
Recycling is a key focus in the circular economy, particularly in the sustainable utilization of materials such as Nylon 6. To enhance the properties of recycled Nylon 6 (15% RP and 20% RP) and achieve performance comparable to virgin Nylon 6, composites were prepared using a twin-screw extruder to ensure homogeneous mixing. Samples were prepared as per ASTM standards using injection moulding. The incorporation of nanomaterials such as graphene oxide (GO) and reduced graphene oxide (rGO) with varying ratios 0.5%, 1%, 1.5%, 2% and 2.5% added for improving the mechanical and electrical properties of the recycled material. This study evaluates performance of recycled Nylon 6 (15% & 20% Recycle material) with varying percentages of GO and rGO (0.5 to 2.5%) through tensile strength testing, Izod impact testing, hardness measurement and flexural testing. The results indicate that 2% addition of GO and rGO significantly enhances the mechanical properties compared to recycle material. Conductivity measure through dielectric strength test to confirm the electric properties. The analysis of dispersion of GO and rGO within the polymer matrix using scanning electron microscopy (SEM), reveals uniform distribution at the optimal 2% concentration. Fourier-transform infrared spectroscopy (FTIR) was employed to study the grafting of recycled Nylon 6 with virgin Nylon 6, confirming effective compatibility. Additionally, melt flow index (MFI) analysis was conducted to evaluate molecular weight variations. It demonstrates increased viscosity, which correlates with improved mechanical properties. These findings highlight the potential of GO and rGO in enhancing the performance of recycled Nylon 6, contributing to the development of high-performance, sustainable polymer composites.
Introduction
Convenience, durability and versatility offered by Plastics has made it an integral part of modern life. However, its improper disposal and their non-biodegradable nature has posed significant environmental concern. Polymer recycling plays a crucial role in mitigating plastic pollution, conserving natural resources, and reducing greenhouse gas emissions associated with virgin plastic production. 1 Recycling strategies, including mechanical, chemical, and biological methods, contribute to the sustainable management of plastic waste.2,3
The three R’s of circular economy; reuse, refurbish or repurpose enhances the efficiency of polymer recycling by promoting a closed loop system. The traditional linear economy follows ‘take- make-dispose, model whereas the circular economy emphasizes waste minimization and resource efficiency. By integrating effective recycling practices into the circular economy framework, industries can reduce dependency on fossil-based raw materials, lower environmental footprints, and foster sustainable economic growth.4,5
Polymeric products (plastic, rubber etc.) are ubiquitous and are an integral part of our life. Nylon, a widely known polyamide, is the most known type of polyamide due to its excellent chemical strength, resistance to abrasion and wear and low friction coefficient.6,7 Hence, nylon is extensively utilized in various applications, including fibre, molded parts for automotive components, and diverse uses such as bearings, gears, bushings, handles, wire and cable coatings, clothing, fishing nets and reinforcement materials for tires, parachutes and conveyor belts. 8
Nylons are commonly used plastics with a current global market of more than 8 million tons per annum, which is expected to grow at the rate of 2.2%, reaching up to 10.4 million tons, equivalent to 47.0 billion USD by 2027. Inevitably, the ubiquity of nylon and their nonbiodegradable nature have resulted in increased land and ocean pollution, posing a severe threat to our ecosystem. 6 Therefore, it is critical that efficient and sustainable technologies for the recycling of plastics such as nylon are developed. The current plastic recycling technologies are mostly based on mechanical recycling converting one form of plastic to another by mechanical reprocessing (melting and re-extrusion): for example, converting plastic bottles to fibres. 4
To improve the mechanical and thermal strength some Nano fillers like Graphene oxide and reduce graphene oxide are used. Graphene oxide (GO) is obtained from the oxidation and exfoliation of graphite. 5 Reduced graphene oxide (rGO) can be obtained using thermal reduction of GO. 9
RGO has been extensively explored as a promising material for multi-purpose nanocarriers owing to their large surface area, high light-adsorption ability, and excellent photothermal effect. Due to significant properties of GO/rGO, such as electrical conductivity, light absorbance and emission, and biological effects; both these materials have been widely explored for various biomedical areas.5,10
The presence of residual oxygen in rGO limits the carrier mobilities from reaching the extraordinary values of the mobilities obtained in mechanically-exfoliated or chemical vapor deposition (CVD) graphene high electron and hole mobilities in the range 100–1000 cm2V−1s−1 and sometimes exceeding 1000 cm2V−1s−1 have been measured in rGO thin films. These properties allow for superior charge transfer characteristics at rGO-based electrodes leading to larger currents and better signal-to-noise ratios in sensing applications. 11
Graphene’s low percolation threshold is related to its significant properties such as barrier resistance, electrical conductivity, electrooptic, abrasion resistance, stiffness, and fire retardancy at low filler loading content. 12 Because of its conductivity behaviour and unusual fractional quantum hall effect, it has been used in many technological fields such as nanocomposites, sensors, batteries, and super-capacitors. Graphene is rapidly gaining attention from a broad spectrum of research areas due to its remarkable properties including high mechanical strength, high carrier mobility (200,000 cm2V−1s−1), excellent electrical conductivity, and excellent thermal properties. Within ideal graphene layers, the sp2 hybridized carbon atoms are highly well-organized in shuttling and storing electrons. Hence, the graphene and graphene-based materials hold a great potential in applications like energy storage devices, field emission displays (FEDs) and photo-catalysis. 13
Generally, GO instead of graphene is preferred for combining with other materials because of its abundant oxygen-containing groups, promoting connections with other functional groups through covalent interactions. GO has attracted considerable attention as a nanofiller for polymer nanocomposites because of additional carbonyl and carboxyl groups located at the edge of the sheets, which makes graphene oxide sheets strongly hydrophilic, allowing them to readily swell and disperse in water. Covalent modifications are commonly used in GO. GO has a layered material consisting of hydrophilic oxygenated graphene sheets carrying oxygen functional groups of hydroxyl, epoxy, carbonyl and carboxyl on their basal planes and edges, which allows the attachment of other functional groups through typical organic reactions, such as amidation, silanization, esterification, substitution and cycloaddition. 14
rGO can be produced by thermal reduction of GO and electrochemical reduction of GO. A majority of graphene/polymer composites investigated are fabricated using GO, chemically reduced graphene oxide (CRGO), or thermally reduced graphene oxide (TRGO) as fillers. Several researchers have used rGO for fabrication of graphene-polymer composite. A majority of graphene/polymer composites investigated by various researchers are fabricated using GO, chemically reduced graphene oxide (CRGO), or thermally reduced graphene oxide (TRGO) as fillers. 15
Melt mixing is used to fabricate Graphene-polymer composites. The advantages of this method are its versatility, cost-effectiveness, and environmentally friendliness. In this technique, nanofiller is physically mixed with polymer melt. No solvent or any other additive/compound/chemical within the mixing unit is required in this method. 14 A thermoplastic polymer is mixed mechanically with graphene or its derivatives at elevated temperatures in molten state using conventional methods, such as extrusion and injection moulding. The process is cost-effective, and large quantities can be processed at a time.
Hou et al. (W. Hou et al., 2014), fabricated nanocomposite of functionalized graphene grafted by PA6 by in situ polycondensation of caprolactam (CPL) and connection of the PA6 to the functionalized graphene, and their continuous nanocomposite fibres were prepared by use of melt spinning and drawing process. The grafting PA6 chains on graphene sheets were confirmed by FTIR, TGA and AFM measurements. Tensile strength of the nanocomposite fibres containing the-(CH2)6 NH2 functional group with 0.1 wt% graphene loading was significantly increased, over twice as high as that of neat PA6.
Xiang et al. (Xiang et al., 2019), fabricated PA6/Cellulose-RGO-TDI nanocomposite with high electrical and thermal conductivity via reactive melt processing. Exfoliated RGO nano-sheets homogeneously dispersed between matrix and cellulose skeleton and a good interface form, which promoted formation of electrically and thermally conductive network, resulting in the remarkably improved electrically conductivity of 5.8 × 10−1 conductivity of 0.419 Wm−1 K−1 of the composite with 1.2 vol% RGO loading. 14
Graphene and its derivatives can be easily homogeneously dispersed in different polymer matrices including thermoplastics, thermosets, and conducting polymers. They have been used as a nanofiller in various polymeric matrices, such as epoxy, Polystyrene, polyurethane (PU), poly (vinylidene fluoride) (PVDF), rubber, chitosan, nafion, polycarbonate (PC) and polyaniline (PANI), PET to form graphene-polymer composites. 16
Materials
Recycled Nylon6
The recycled Nylon 6 used in this study was donated by Premium Polyalloys Pvt. Ltd, located in Daman, Gujarat. It consists of waste materials from engineering applications such as bearings, gears, filaments, and staple fibres. The granule size of the recycled Nylon 6 is approximately 10–12 µm in diameter.
Graphene oxide and reduced graphene oxide
Graphene oxide (GO) and reduced graphene oxide (rGO) were procured from AdNano Technologies Pvt. Ltd, Karnataka, India. The graphene oxide has a purity of 99%, a particle size of D50 < 10 µm, and a C:O ratio of 55:45. It is in powder form with a yellow-brown colour. The reduced graphene oxide, obtained from the same supplier, has a C:O ratio of 90:10, a thickness of 0.8–2 nm, and appears as a fluffy black powder. Graphene oxide (GO) was synthesized using a Modified Hummers Method, whereas reduced graphene oxide (rGO) was produced by the chemical reduction of graphene oxide.
Virgin Nylon 6
The virgin Nylon 6 used in this study was sourced from Hangzhou Juheshun New Material Co. Ltd. It is an injection-grade material with the series J 2700.
Experimental
Synthesis of polymer composite with graphene oxide and reduce graphene oxide
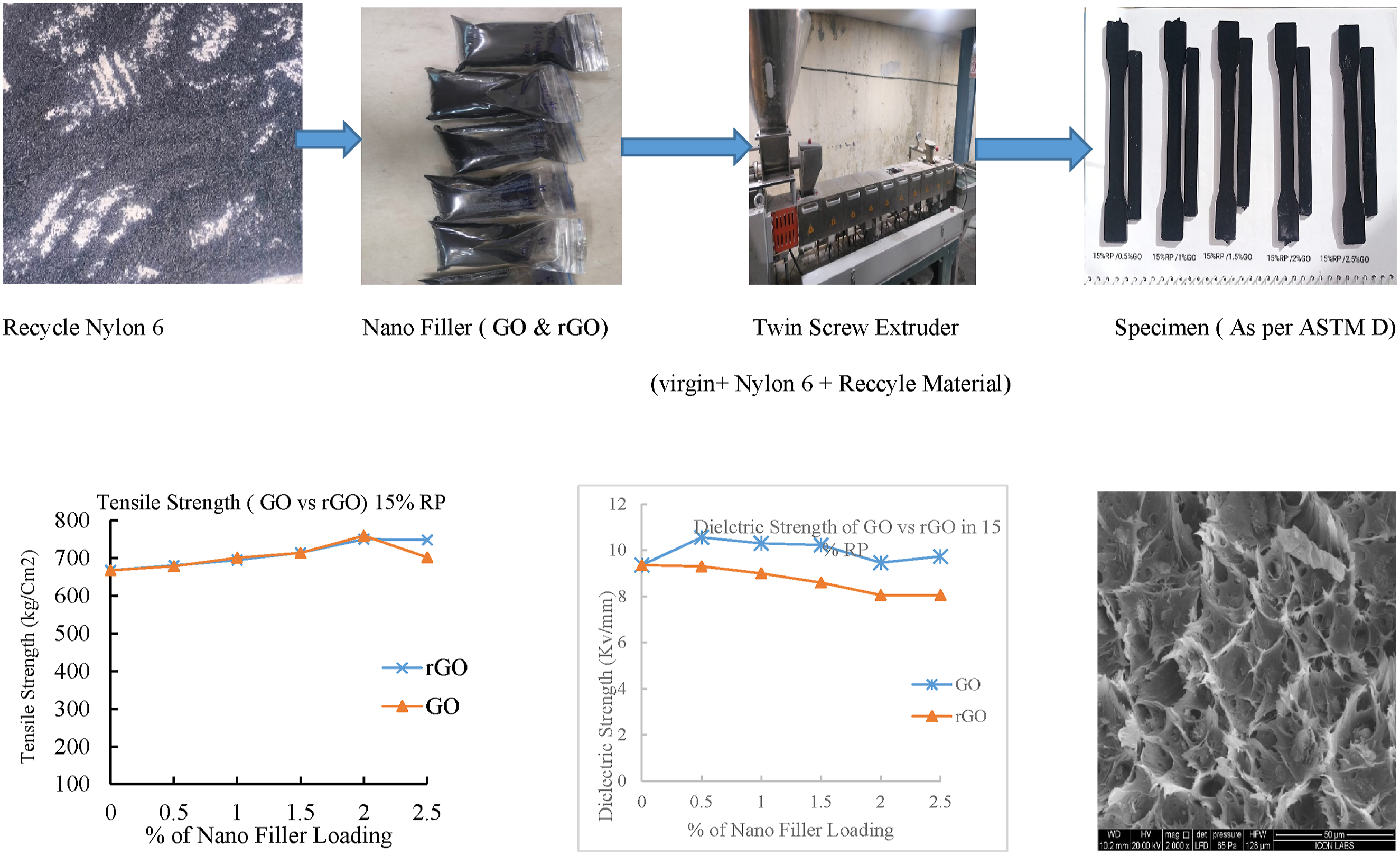
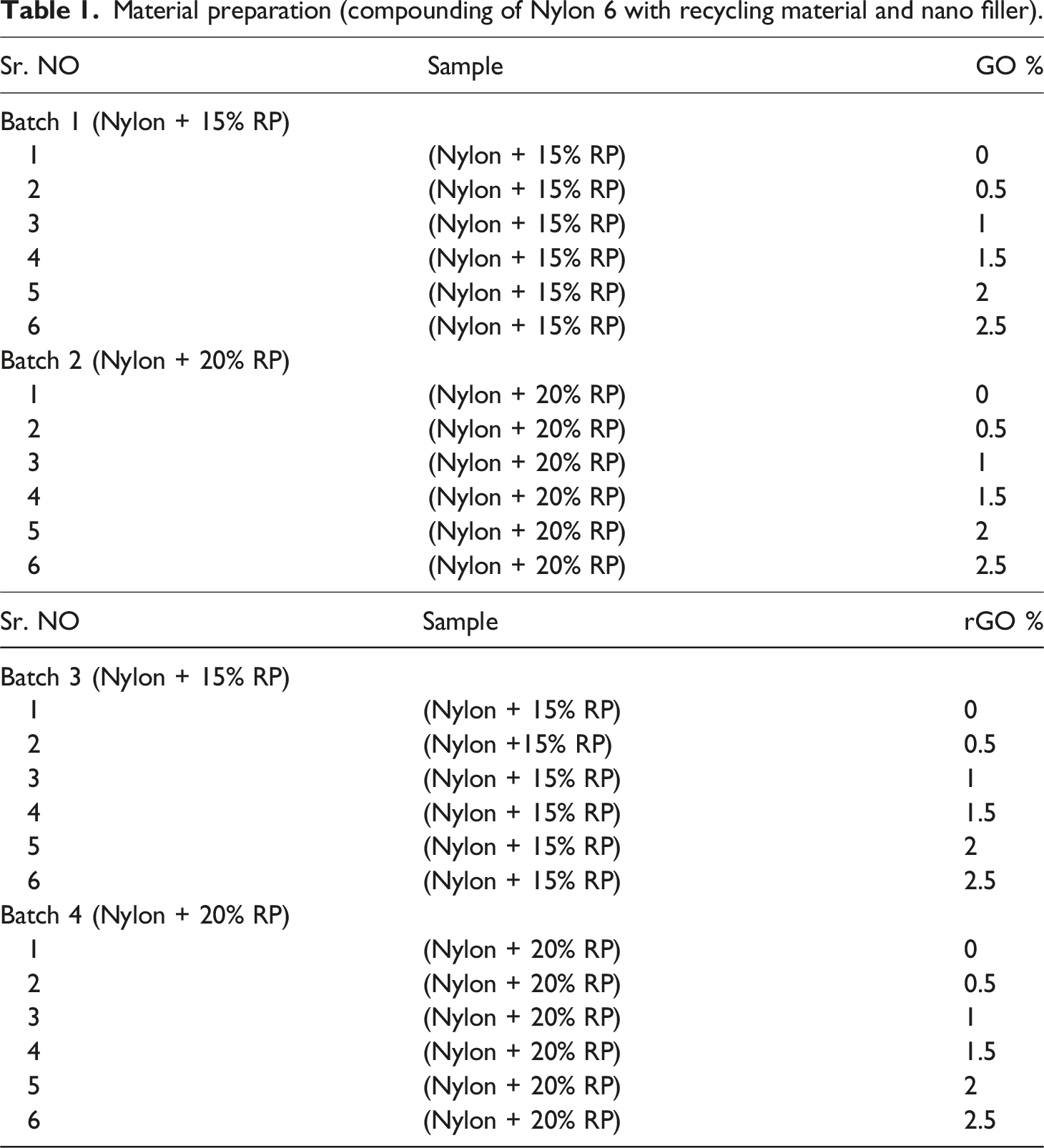
A mixture of polymere was synthesised with virgin nylon and Recycle Nylon with varying ratio (15% and 20%) of recycled nylon using a mixer. In these mixtures, varying ratios of Graphene oxide and reduced graphene oxide (0.5%, 1%, 1.5%, 2% and 2.5%) were incorporated using twin screw extruder by Nanjing Guangda International Group. The screw Diameter was 35 mm and L/D ratio 36:1. Compounding was done under vacuum of 0.84 MPa with set temperature Profile 220/220/220/225/225/226/230/230. Cutting device rpm was 16–17. Twin Screw Extruder as shown in Figure 1. Twin screw extruder.
Material preparation (compounding of Nylon 6 with recycling material and nano filler).
Extrusion proves effective in manufacturing polymer composites, offering control over GO & rGO distribution, leading to materials with improved mechanical and electrical characteristics for specific industrial applications.
Preparation of specimen
Preparation of specimen was carried out by Injection moulding, specimen is prepared for Mechanical, Electrical, and thermal conductivity as per ASTM D standard. The specimen was prepared at Supermac Machinery Kathwada Gujrat with 130 Toggle Clamping. Temperature profile 255/245/235/220-degree Celsius, with 37 sec cycle time. Samples were prepared for Tensile Strength, Flexural Strength, Izod Impact, thermal conductivity, dielectric strength and Hardness strength as per ASTM D standards.
Characterization
Tensile strength
Details of specimen used for tensile strength.

Flexural strength
Flexural strength was carried out as per ASTM D 790. Standard specimen was prepared by using Injection moulding. An International equipment with load cell value 225 Kg, speed 15 mm/min was used. Figure 2 shows the samples for Tensile strength and Flexural Strength. Samples for flexural strength and tensile strength.
Izod impact strength
Izod impact strength was carried out by International Equipment as per ASTM D 256 with Specimen Dimensions. Samples for Izod Impact Strength as shown in Figure 3. • Standard: • Notch depth: Samples for Izod impact strength testing.

Hardness test
Hardness value was characterised as per ASTM D standard (ASTM D 2240) Shor D thickness of specimen used is 3 mm. The test is carried out at stetch engg, Sample and harness tester shown in Figures 4 and 5. Hardness tester. Specimen for hardness testing.

Dielectric strength
The test was conducted on instrument SCR Electronics, at Shubhada Polymers Ltd. Specimen was prepared as per ASTM D 149. A circular disc with 113 cm Diameter was used to carry out the test. Test Specimen as Shown in Figure 6. Specimen for dielectric strength testing.
Melt flow index tester
Melt flow index was calculated as per ASTM D 1238, at temp 190°C, with standard load 2.16 kg. The test was conducted at Shri Bhagubhai Maftlal Polytechnic, Vile Parle. Flow behaviour of various percentage of GO and rGO was measured along with MFI as gm/10 min.
FTIR (Fourier transform infrared spectroscopy)
FTIR spectrum analysis was performed on an instrument with an attenuated total reflectance (ATR) accessory, on a PerkinElmer Frontier FTIR spectrometer at the Central institute of Petrochemical Engg and technology Aurangabad (CIPET). Test was carried out as per ASTM E1252, in the spectral range of 4000 to 400 cm.
Scanning electron microscopy
The reduced graphene oxide (rGO) and Graphene oxide with Different Nylon and rGO and Go concentration was characterized by the FEI, model Quanta 200 netherkand with magnification range from 30X to 1xlac, Resolution :3.5 nm, High Voltage −30 Kv source – Tungsten Filament from the ICON House Navi Mumbai Sanpada, to evaluate the chemical and Morphological characteristics of the material.
Results and discussion
Tensile strength
Figure 7 illustrates the tensile strength of 15% RP with varying percentages of GO and rGO. The results indicate that increasing the filler loading enhances tensile strength in both GO and rGO.
11
The initial increase in the tensile strength can be attributed to large specific surface area of nanofillers and their excellent mechanical properties.
17
However, tensile strength reaches its peak at an intermediate filler loading level, likely around 1.5%–2.0%, before declining beyond this point. This decrease is attributed to the agglomeration of nanoparticles, which weakens the mechanical properties.
15
In the case of rGO, the tensile strength continues to increase up to 2.0%, but the overall strength remains lower than that of GO. GO exhibits superior reinforcement, achieving a maximum tensile strength improvement of 28.33% at 2.0% filler loading, whereas rGO peaks at 21.67%. The superior performance of GO can be attributed to its oxygen functional groups, which enhance interaction with the polymer matrix, leading to better load transfer and interfacial bonding. In contrast, rGO contains fewer functional groups, resulting in weaker bonding and earlier agglomeration beyond 2.0% addition of nanofillers loading. Tensile strength (Go & rGO) with 15% Loading of recycling material.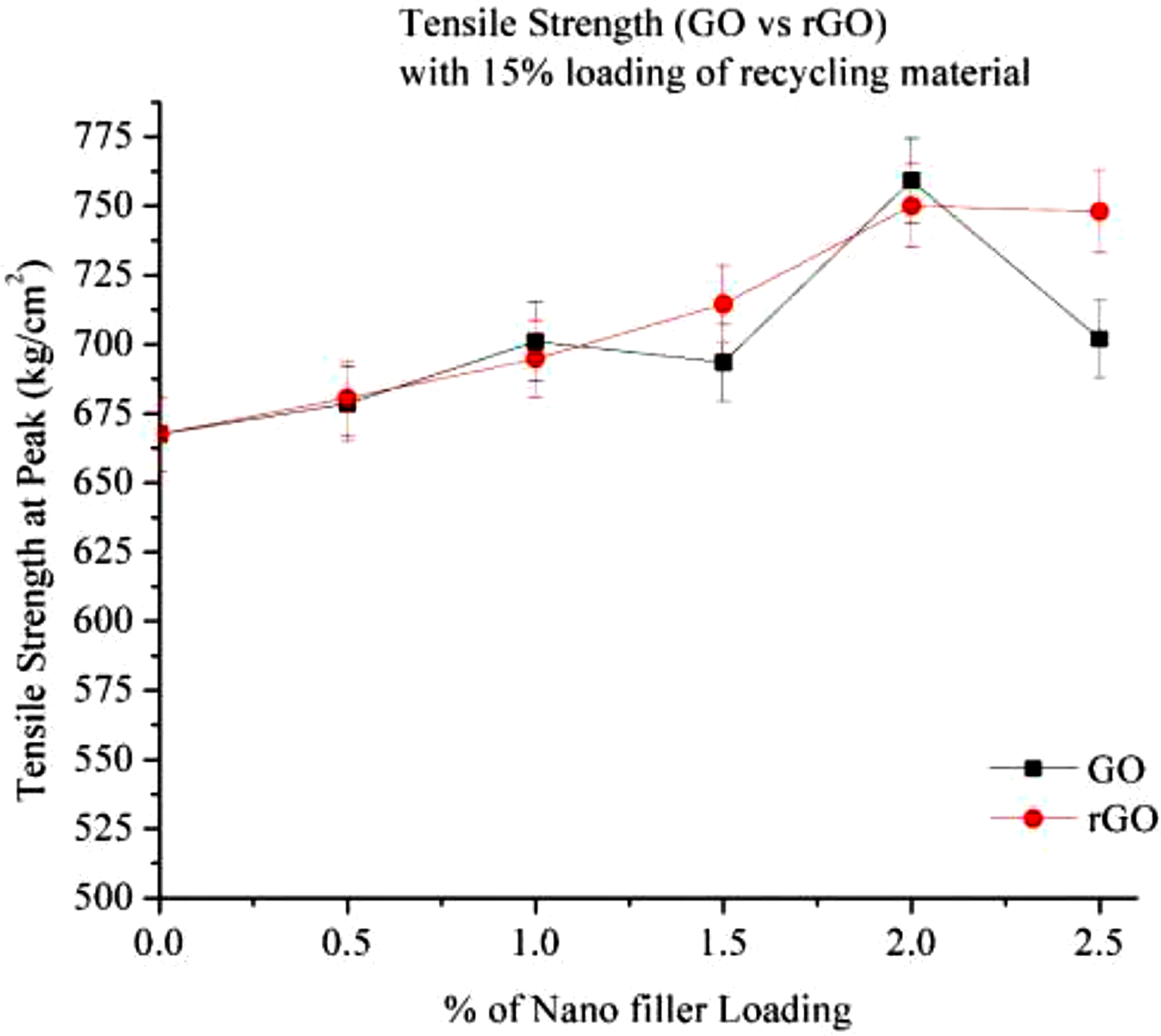
Figure 8 the provided graph compares the tensile strength of Graphene Oxide (GO) and Reduced Graphene Oxide (rGO) as a function of nano-filler loading (0% to 2.5%) in a 20% recycled Nylon 6 matrix. At 0% nano-filler loading (pure 20% recycled Nylon 6), the tensile strength is approximately 600 kg/cm2. With the addition of GO and rGO, tensile strength initially increases at filler loadings between 0.5% and 1.5%, followed by a more significant rise between 1.5% and 2.0%. Beyond 2.0%, increasing the filler loading to 2.5% results in an overall strength improvement of up to 20% compared to 0% loading in rGO. This enhancement is likely due to the homogeneous dispersion of nanofillers in the polymer matrix, resulting in improved interaction between matrix and nanofillers which improves load transfer.
18
Both GO and rGO function as stress distributors, preventing crack propagation. GO contains oxygen functional groups (-OH, -COOH, -C = O), which increase hydrophilicity but also lead to agglomeration at higher loadings. At 2.5% filler loading, tensile strength reaches saturation. Compared to GO, rGO provides superior tensile strength at all loadings. This can be attributed to rGO’s better dispersion, fewer oxygen functional groups, and stronger interfacial interactions with Nylon 6. When comparing GO and rGO in a 20% recycled polymer (RP) matrix, rGO exhibits a 25% increase in tensile strength at 2.0% loading. Additionally, Figures 7 and 8 indicate that increasing the percentage of recycled material results in a decline in mechanical strength. Tensile strength (Go & rGO) with 20% Loading of recycling material.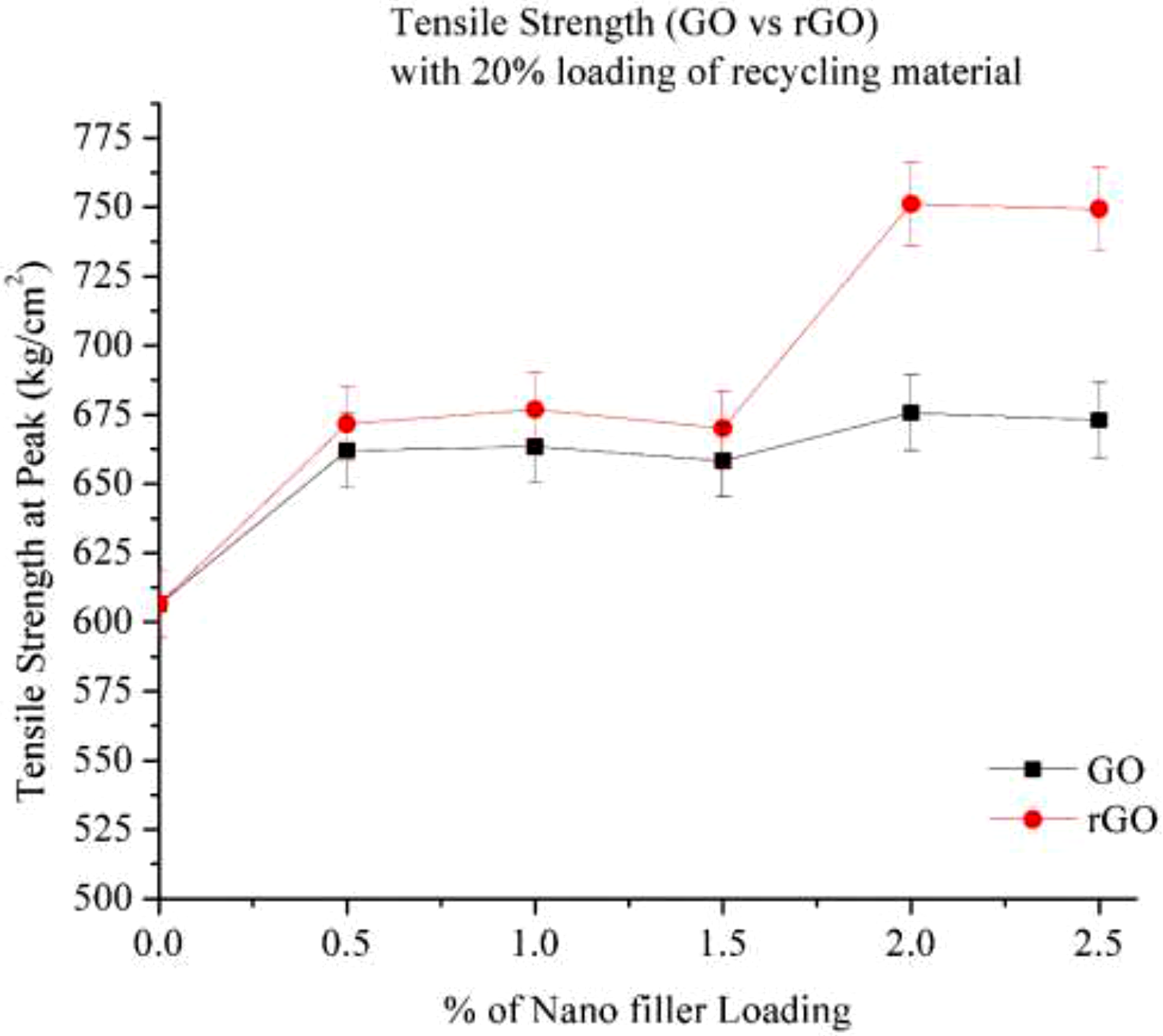
Figure 9 the provided figure illustrates the tensile modulus of 15% recycled Nylon 6 (RP) with varying percentages of nano-fillers such as Graphene Oxide (GO) and Reduced Graphene Oxide (rGO). The results indicate that as the filler loading increases in 15% recycled Nylon 6, the composite’s modulus also increases, leading to improved material stiffness. When comparing GO and rGO, rGO exhibits a higher modulus than GO. This may be due to its better dispersion and lower oxygen content, which contribute to increased stiffness.15,19 The highest tensile modulus is observed at 2.0% filler loading for both GO and rGO, suggesting that uniform dispersion is achieved up to this level. Beyond 2.0% loading, agglomeration of the fillers likely occurs, leading to a reduction in modulus. Tensile modulus (Go & rGO) with 15% loading of recycling material.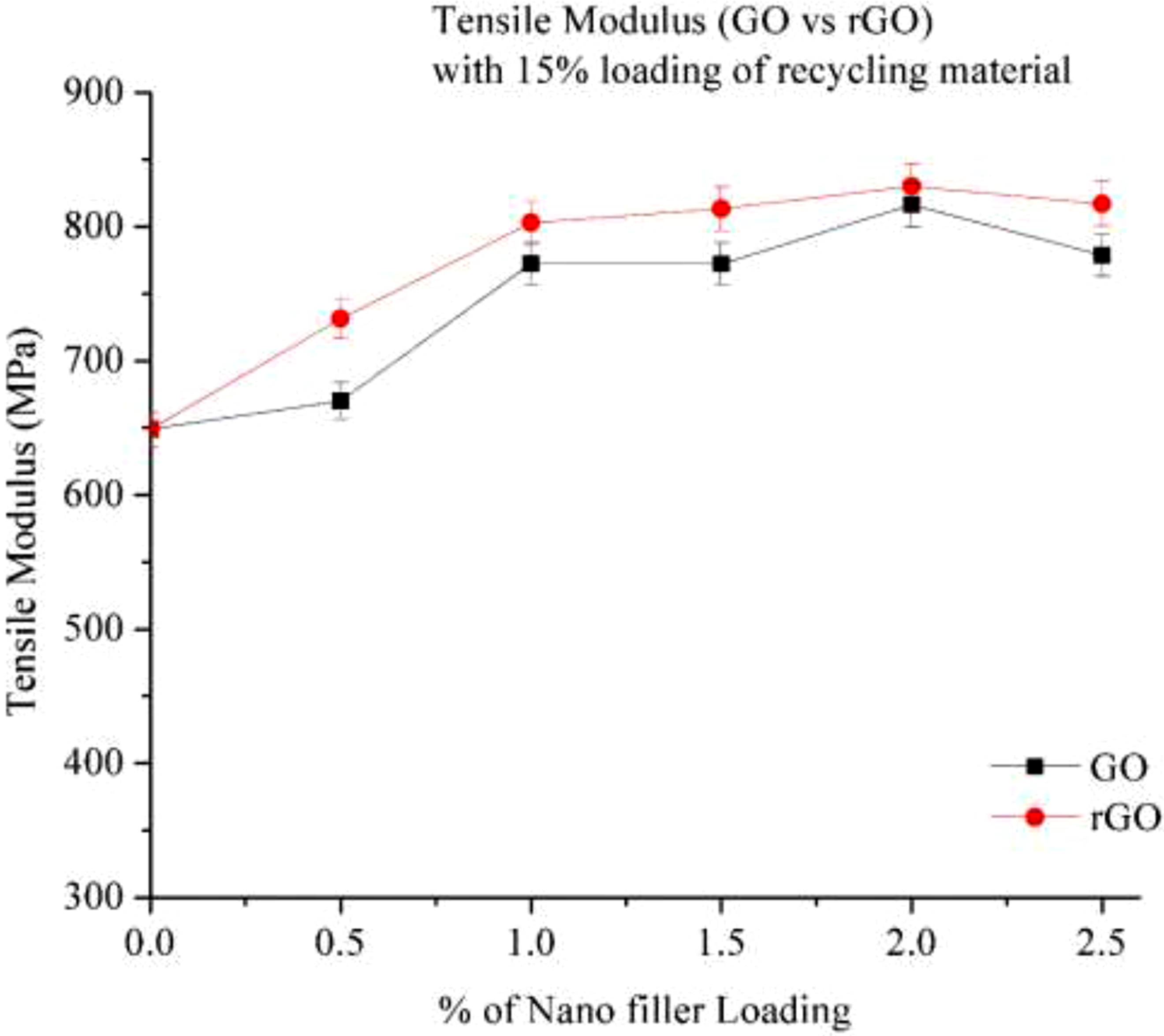
Figure 10 the figure illustrates the variation in tensile modulus with increasing nano-filler content in a 20% recycled Nylon 6 (RP) matrix. The results show that tensile modulus increases as the percentage of nano-fillers (GO and rGO) increases. At 0% filler loading (20% RP), the tensile modulus is relatively low. However, it rises at 0.5% filler loading and continues to increase with the addition of GO. At 2.0% GO loading, the modulus improves by approximately 11%, which can be attributed to the effective dispersion of GO within the recycled Nylon 6 matrix. The increase in tensile modulus at 2.0% loading is due to good adhesion and uniform dispersion of graphene oxide, which benefits from its high colloidal stability and large surface area.
5
Beyond 2.0% loading, tensile modulus begins to decline, with a reduction of nearly 7% at 2.5% loading, likely due to agglomeration of GO. In the case of rGO, the tensile modulus initially increases with rising filler content but starts to decline beyond 1.5% loading, possibly due to improper dispersion within the Nylon 6 matrix. When comparing GO and rGO, rGO provides a higher tensile modulus than GO, which can be attributed to its lower oxygen content and better interfacial interactions with the polymer matrix.
5
Tensile modulus (Go & rGO) with 20% loading of recycling material.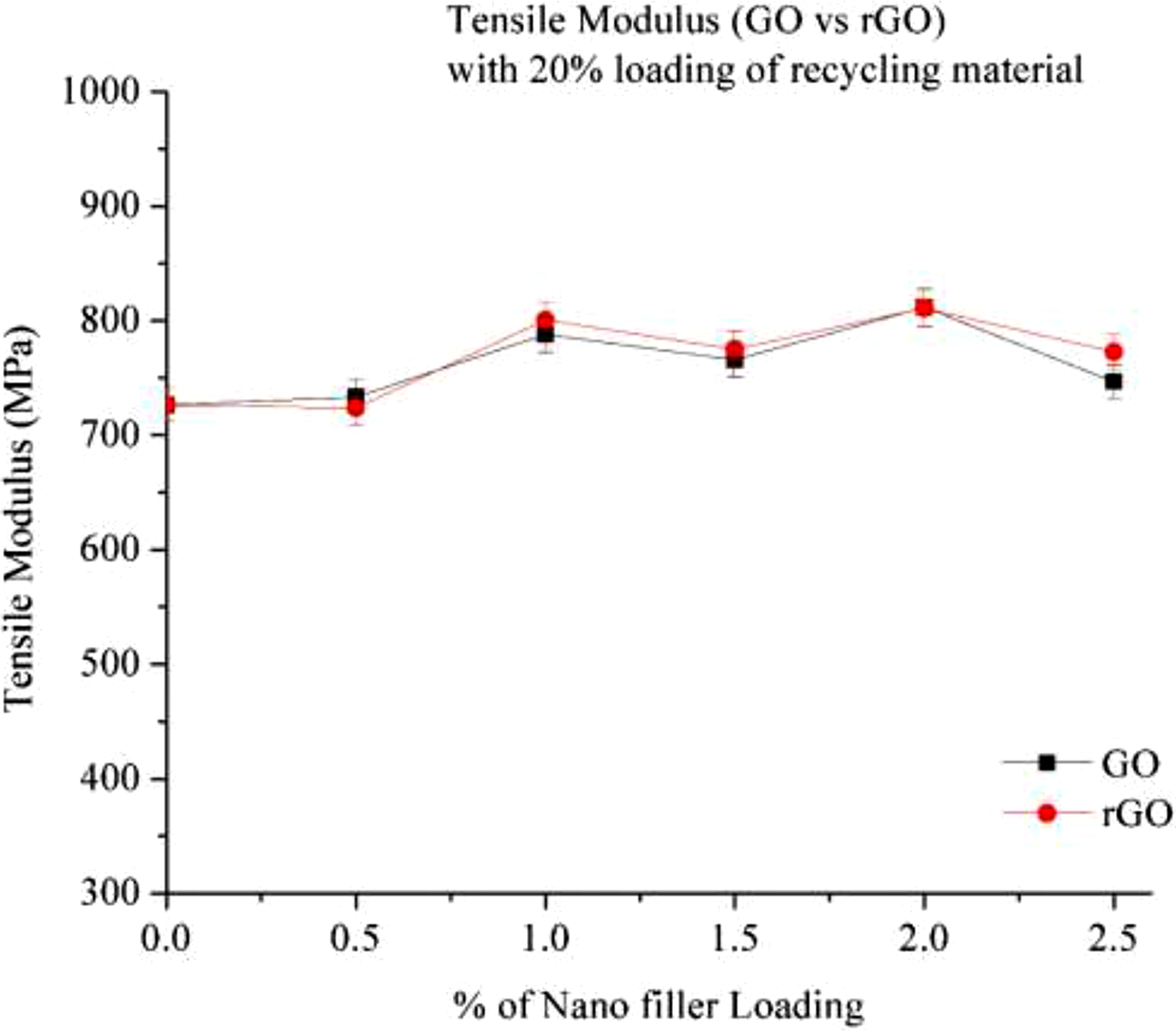
Figure 11 illustrates the percentage elongation of Graphene Oxide (GO) and Reduced Graphene Oxide (rGO) at varying nano-filler loadings (0% to 2.5%) in a recycled Nylon 6 matrix with 15% recycled content. The results indicate a decrease in elongation with increasing filler content, with the best performance observed at 2.0% loading for both GO and rGO. The addition of GO and rGO reinforces the polymer matrix by restricting polymer chain mobility, reducing flexibility and leading to a lower elongation percentage. At 0% filler loading, elongation is at its highest (∼10.5%–11%). Initially, at 0% loading, both GO and rGO exhibit similar elongation values. However, at 1.0%–2.0% filler loading, elongation stabilizes at approximately 8.5%–9.0%, with rGO consistently maintaining a slightly higher value than GO. Although GO and rGO enhance mechanical strength, their interaction with the polymer matrix creates localized stress points, further contributing to the reduction in elongation.
15
% Elongation of Go and rGO in 15% RP.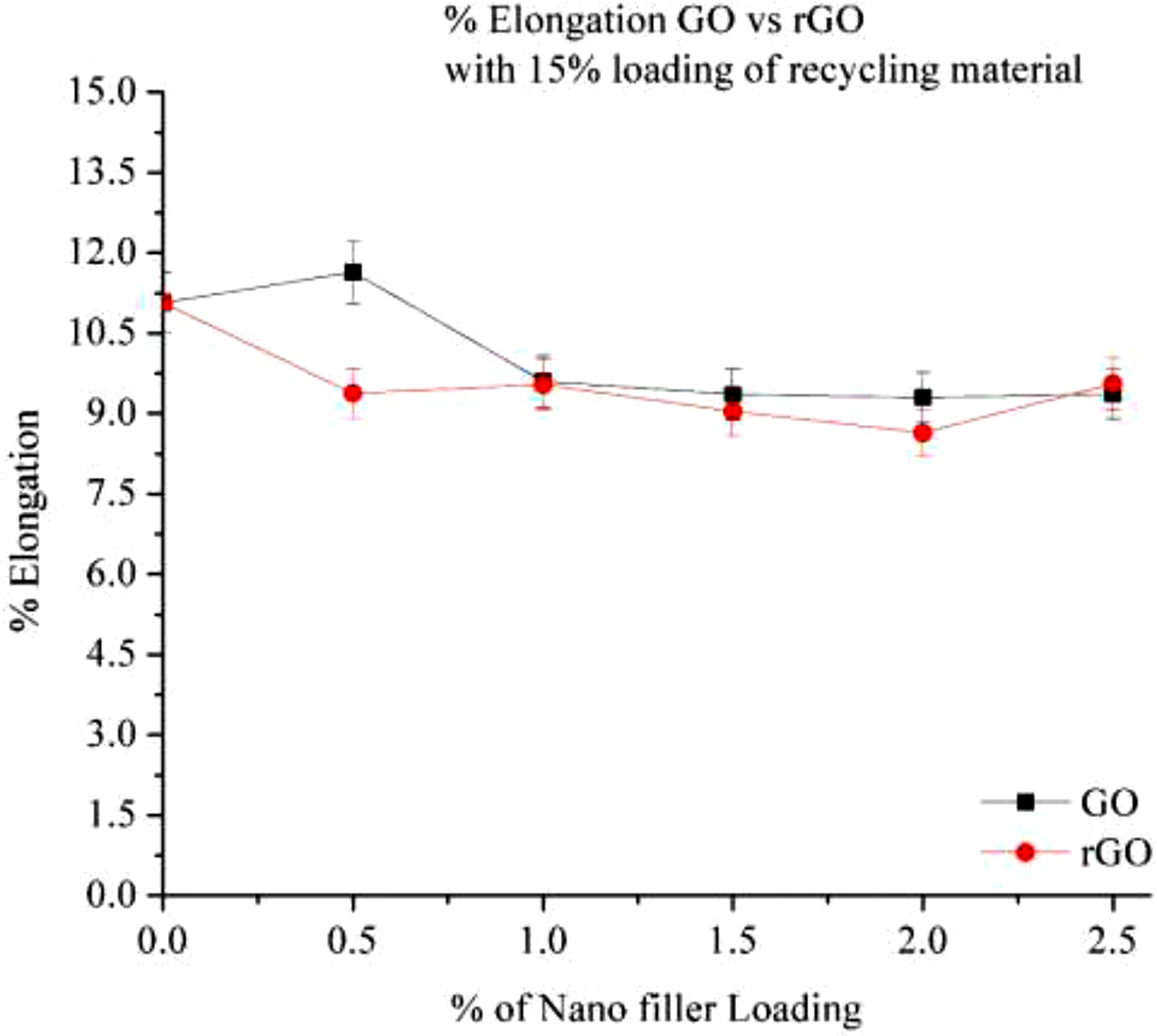
Figure 12 depict the % change in elongation of 20% RP after addition of GO and rGO, at 0% Loading elongation is high, due to containing recycle material the flexibility increases at 0 % loading resulting increasing the elongation. After addition of 0.5% filler elongation drastically decreases it’s because of ductility of polymer reduces significantly with increasing the filler loading.
20
Both GO and r Go shows the decreases in % of elongation it means after addition of filler material get brittle it can be attributed to the presence of rigid and brittle 2-D phase Graphene material in it, it leads to restriction in the polymer chain movement. GO provide reduction in % elongation compare to rGO, rGO consistently maintains slightly higher elongation compared to GO, especially at higher loadings. At 2.5% rGO, elongation is 12% compared to 10% for GO, meaning rGO retains more ductility. High filler loading creates rigid networks, reducing polymer chain mobility, leading to lower elongation. % Elongation of Go and rGO in 20% RP.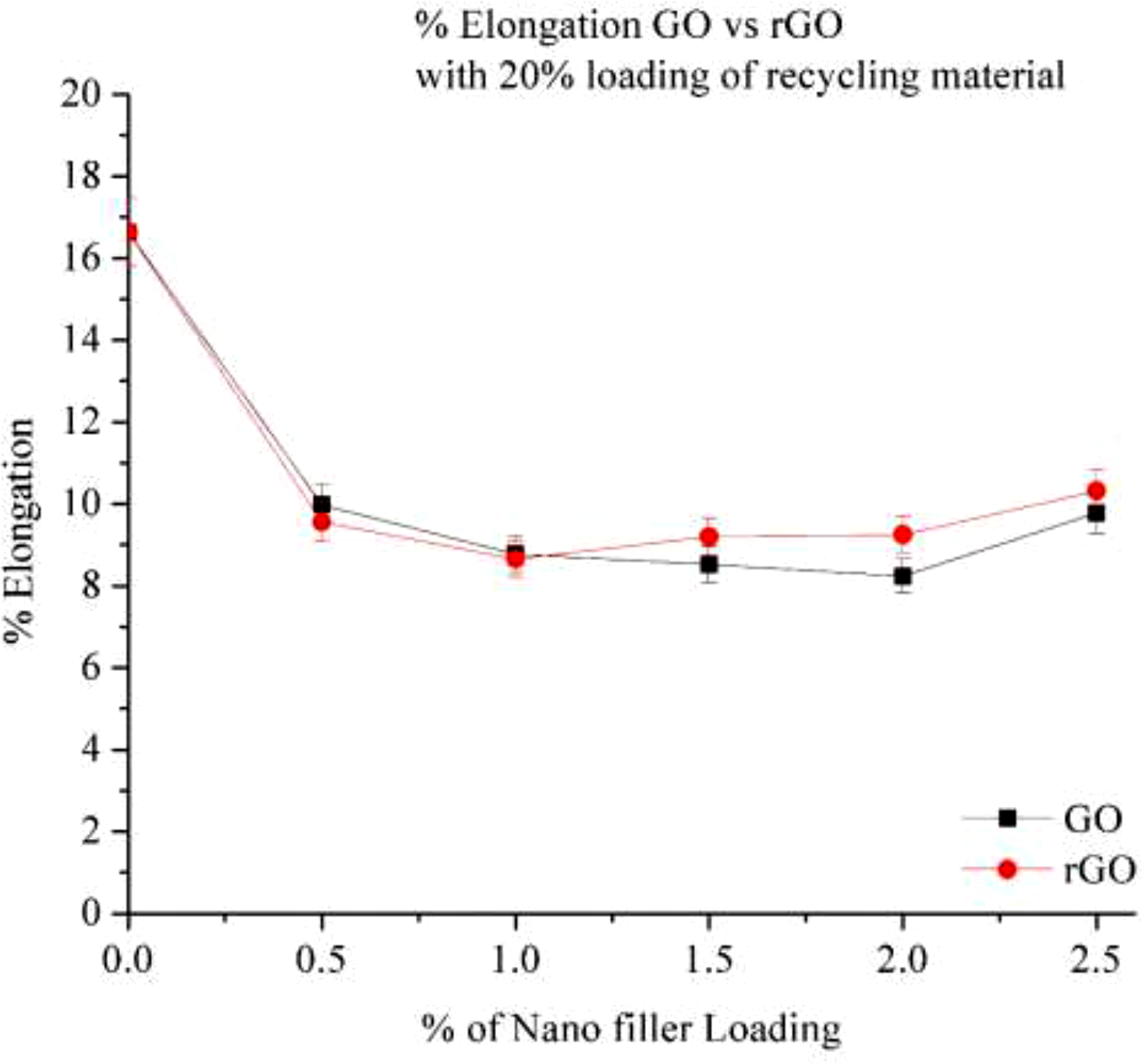
Flexural strength
The result of Flexural strength of GO and rGO in 15% RP material are presented in Figure 13, Nylon with 15% RP (0% Loading) shows the lower flexural strength reported as 751 kg/cm2, rise in filler loading leads to increase in Flexural strength, at 0.5 % loading flexural modulus increased nearly 8% , further increment in % of GO flexural strength increase with increasing the loading , which is nearly 12% at 2% GO loading. The initial increase in the flexural strength can be attributed to high aspect ratio and large surface area of nanofillers, resulting in improved load transfer and stress distribution.21,22 After that there is no much increment is observed , it attributed because of improper dispersion of filler or agglomeration at higher percentage, similarly in rGO, effect of strength is increases while increasing the loading of rGO in comparison with Go over rGO, rGO shows better performance than GO, because absence of oxygen help improved the flexural strength at different loading rGO performed well compare to GO, at 0.5% loading Flexural strength increases nearly 10% improved slightly in 1 to 1.5% rGO, but at 2% loading the rise in flexural strength compare to 0% loading reported as 13%. Flexural strength of Go and rGO with 15% RP.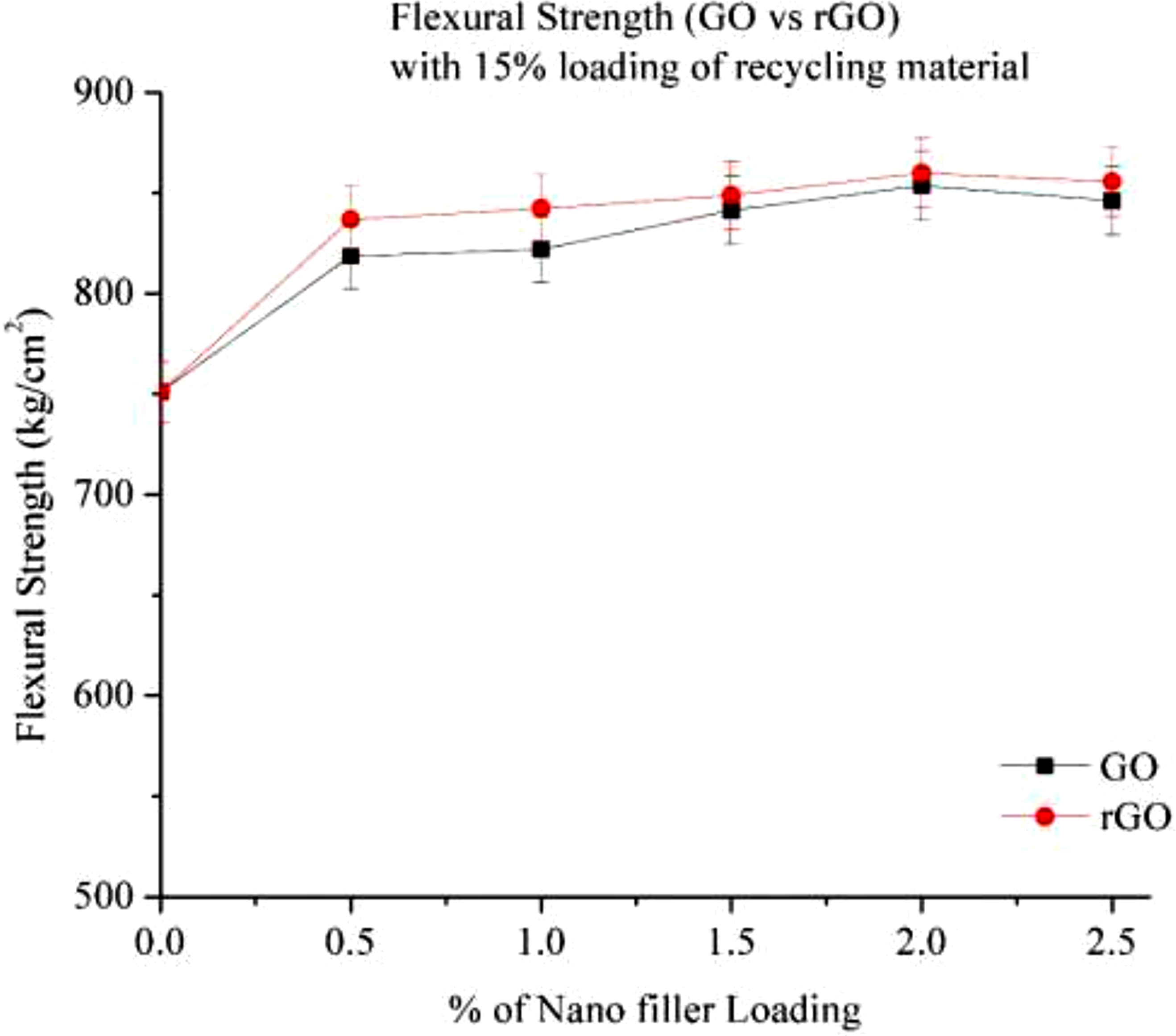
Figure 14 Shows the flexural strength of 20% RP with GO and rGO, as rise in % of filler will increase the flexural strength. It can be attributed due to GO and rGO have abundant functional groups (hydroxyl, carboxyl, epoxide) that improve adhesion with the polymer matrix. In both GO and rGO flexural strength increase nearly 8% and 13% respectively at 2% Loading. GO shows a slight increase (∼7%–8%) at 2.0%, but stabilizes or slightly drops at 2.5%, possibly due to agglomeration effects. Whereas it consistently increases nearly 10–12% at 2% Loading, rGO shows better performance in Flexural Strength compare to GO due to rGO Has fewer oxygen groups after the reduction process, making it structurally closer to pure graphene, which has higher mechanical strength and better load transfer capability.7,11 Flexural strength of Go and rGO with 20% RP.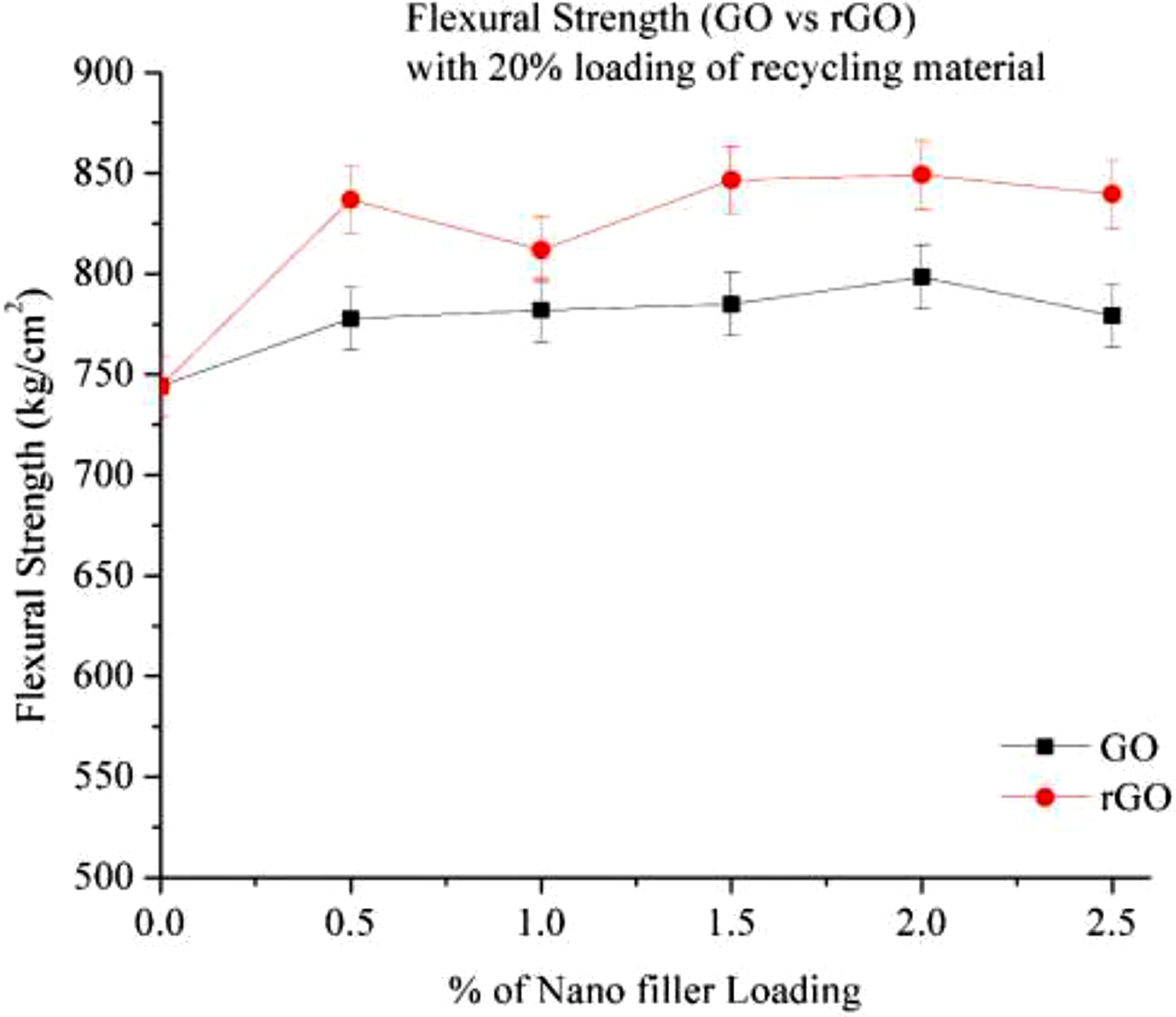
Figure 15 illustrates the flexural modulus of GO and rGO in a 15% RP composite. The modulus increases with higher GO loading; however, between 0.5% and 1% loading, only a slight improvement is observed, likely due to the non-uniform dispersion of GO. With further loading at 1.5% and 2%, a significant increase in flexural modulus is evident. Flexural modulus of Go and rGO with 15% RP.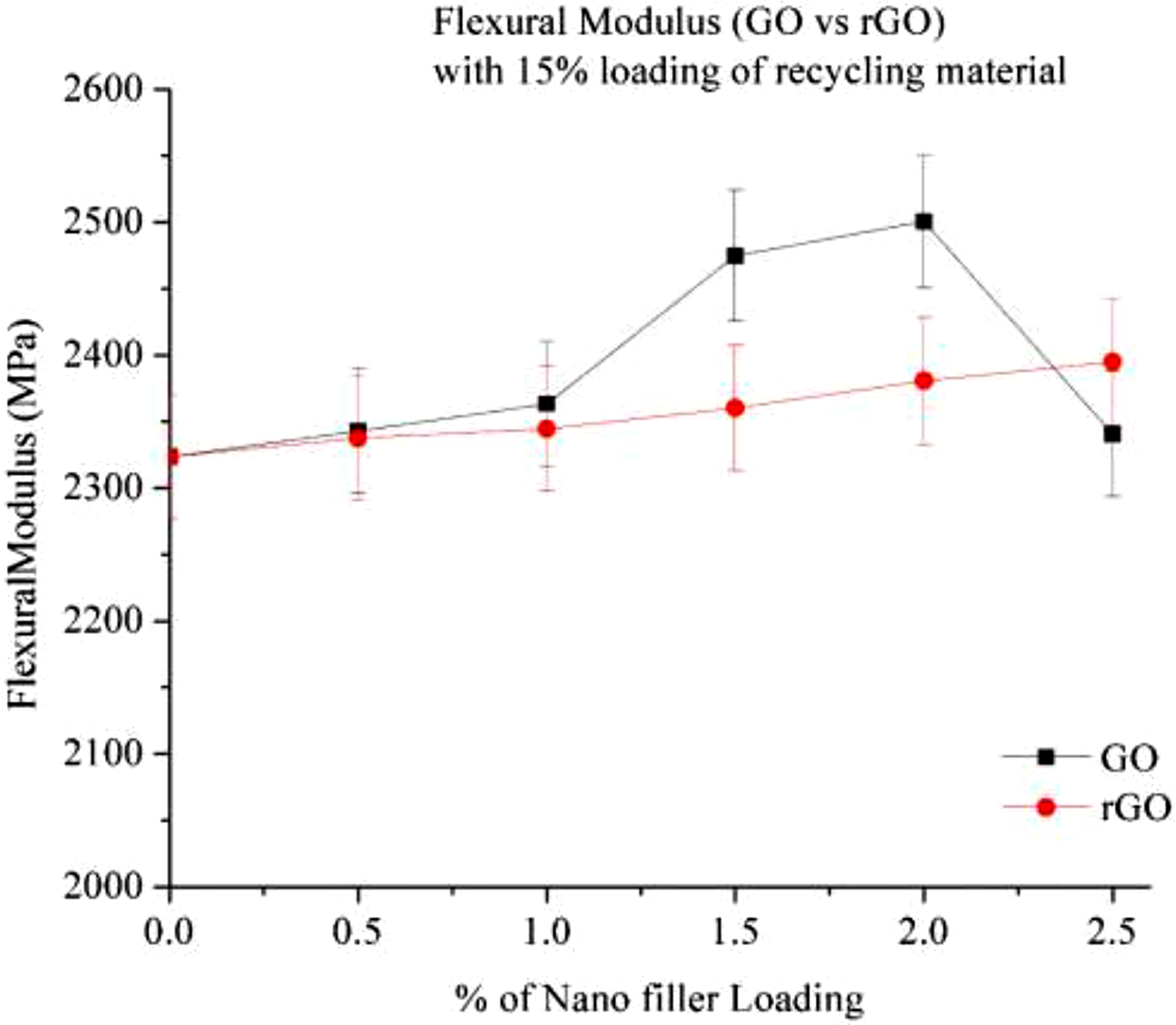
Initially, the surface functionality and dispersion of GO are inadequate, but as the loading increases, the composite’s properties improve. The interfacial adhesion also enhances due to the better compatibility between GO and the Nylon matrix. A similar trend is observed with rGO.
Figure 16 depicted Flexural modulus of 20% RP Govs rGO, when 20% recycled Nylon 6 (RP) is blended with varying percentages of graphene oxide (GO) and reduced graphene oxide (rGO), the flexural modulus of the composite increases as the nano-filler content rises. This improvement is attributed to graphene’s high specific surface area and aspect ratio.
23
The addition of GO to the 20% RP composite initially shows a slight increase in modulus at 0.5% loading. However, at 1% and 1.5% loadings, the modulus remains relatively unchanged. A significant enhancement of approximately 7% is observed at 2% loading. Beyond this point, the modulus declines due to excessive GO content, which leads to the formation of defects and weak interfacial bonding.
19
In contrast, the incorporation of rGO into the 20% RP composite results in a steady increase in modulus with increasing dosage. A slight decline is noticed at 1% loading, but from 1.5% to 2%, a remarkable improvement in flexural modulus is observed. This enhancement is attributed to better dispersion and stronger adhesion with Nylon 6, owing to the reduced oxygen functional groups in rGO. Compared to GO, rGO demonstrates superior performance due to its improved surface properties, achieving nearly a 16% increase in modulus at 2% loading. Flexural modulus of Go and rGO with 20% RP.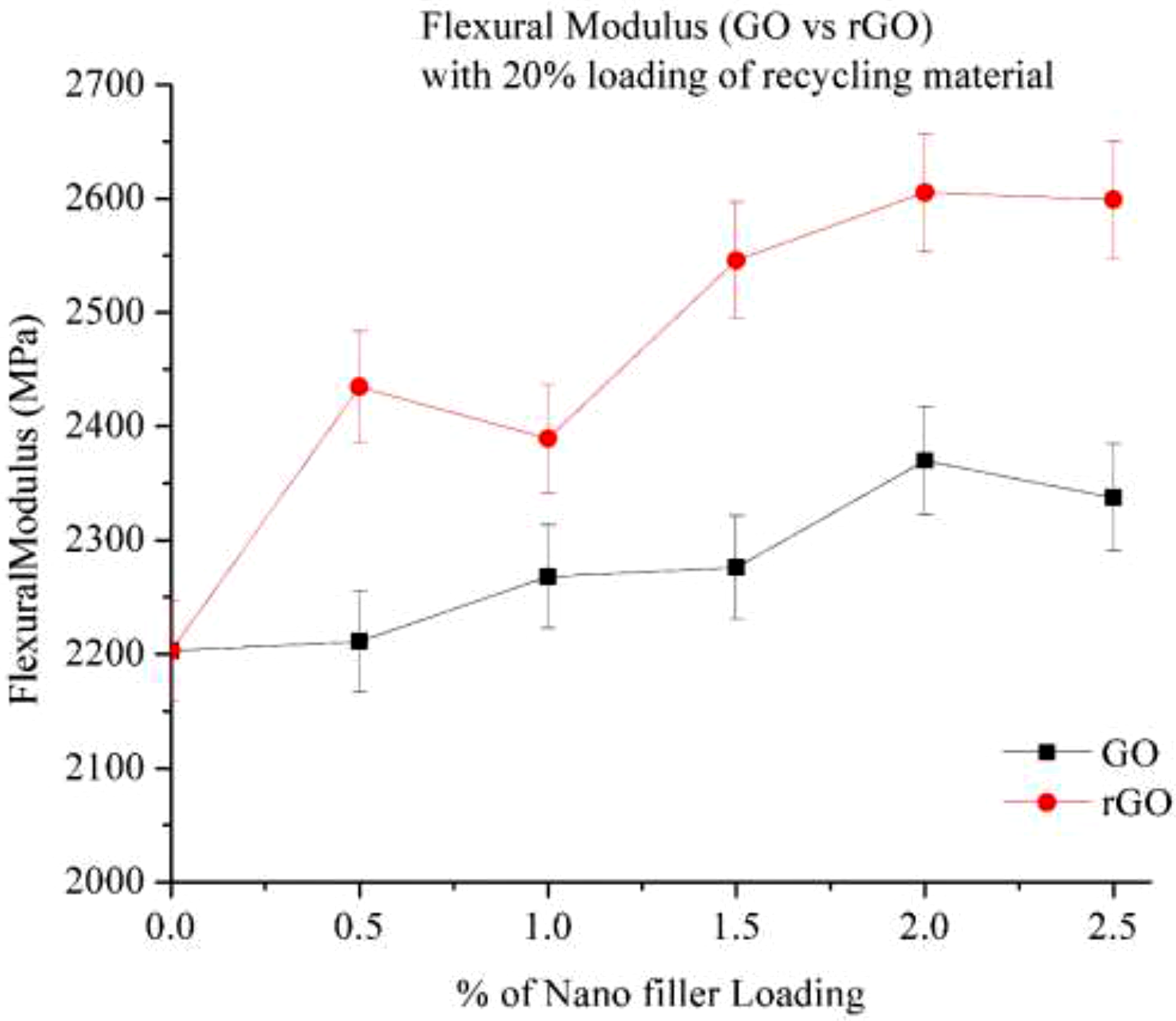
Hardness
Figure 17 illustrates the hardness variation of Graphene Oxide (GO) and Reduced Graphene Oxide (rGO) in a 15% recycled Nylon 6 composite. The graph indicates that hardness increases with increasing nano-filler loading. For GO, hardness improves with increasing filler content, reaching its peak at 2.0% loading. Beyond this point, a decline is observed, resulting in hardness values lower than those at 2.0% loading. An overall ∼5% increase in hardness is noted. This enhancement is attributed to the presence of oxygen functional groups (-OH, -COOH, -C = O), which enhance adhesion between GO and the polymer matrix. The rigid planar structure of nanofillers improves the load bearing capacity resulting in an increase in the hardness.
24
In contrast, rGO exhibits a continuous increase in hardness with increasing filler content, achieving the highest value at 2.5% loading. The superior performance of rGO is due to its lower oxygen content, which reduces the hydrophilicity of the composite. This hydrophobic nature enhances compatibility with Nylon 6, promoting better interfacial bonding and efficient stress transfer, ultimately increasing the modulus and hardness. The hardness of the composite increases from 69 Shore D to 74 Shore D. Hardness of Go and rGO with 15% RP.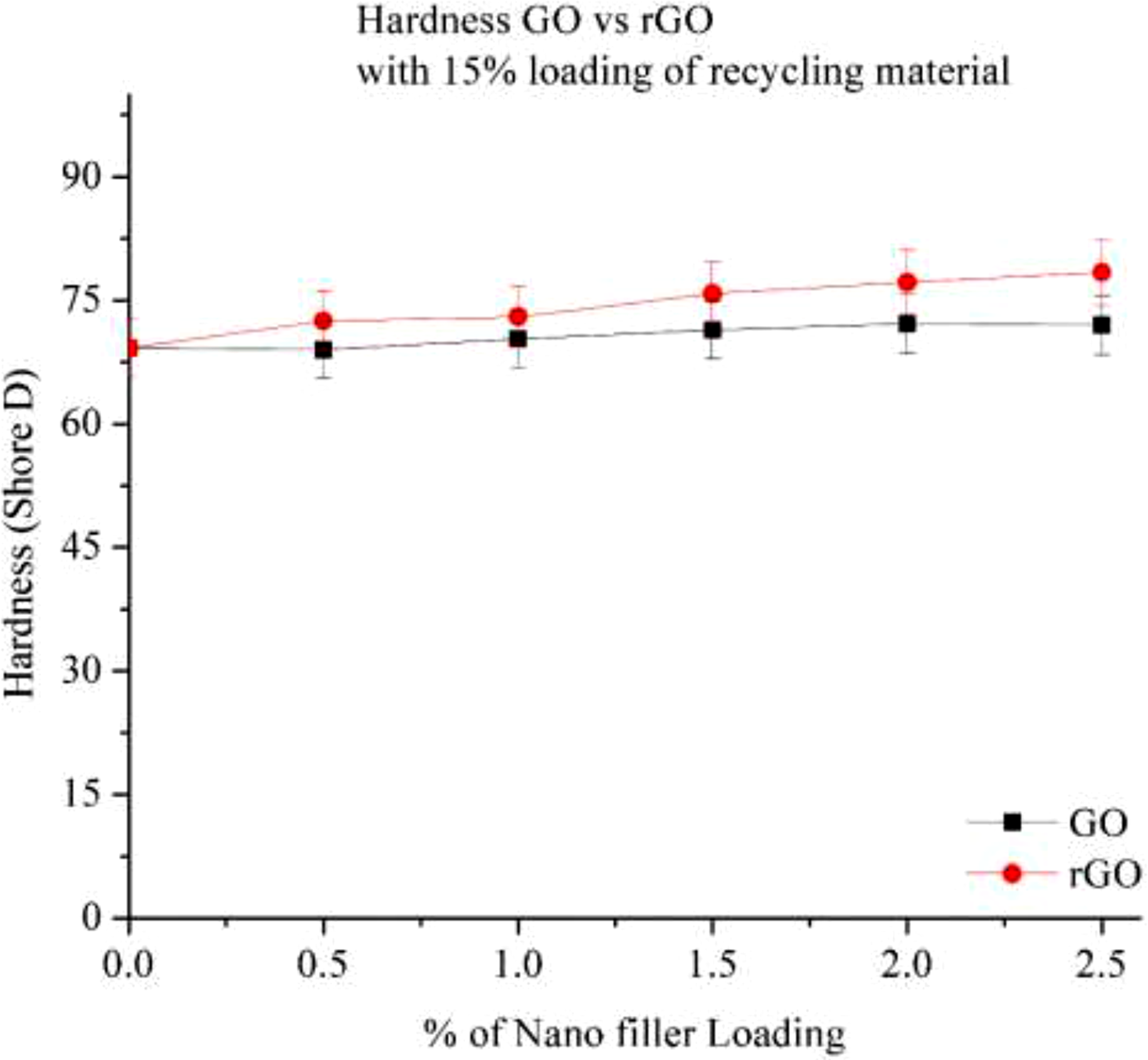
Figure 18 illustrates the hardness of a 20% recycled Nylon 6 composite with GO and rGO. As the percentage of recycled material increases, the hardness decreases compared to the 15% RP composite. However, the addition of nano-fillers like GO and rGO helps enhance hardness. For GO, hardness increases with rising filler content, reaching its peak at 2.0% loading, with an 18% improvement. Beyond this point, hardness begins to decline, likely due to improper dispersion or agglomeration of GO particles. Despite this reduction, GO exhibits better strength than rGO in the 20% RP system, as GO shows better compatibility with the recycled material, leading to stronger interfacial adhesion. For rGO, hardness continues to increase with increasing filler content, achieving a 13% rise at 2.5% loading. The superior performance of rGO at higher loadings is attributed to its better dispersion and reduced oxygen content, which enhances its interaction with the polymer matrix. Hardness of Go and rGO with 20% RP.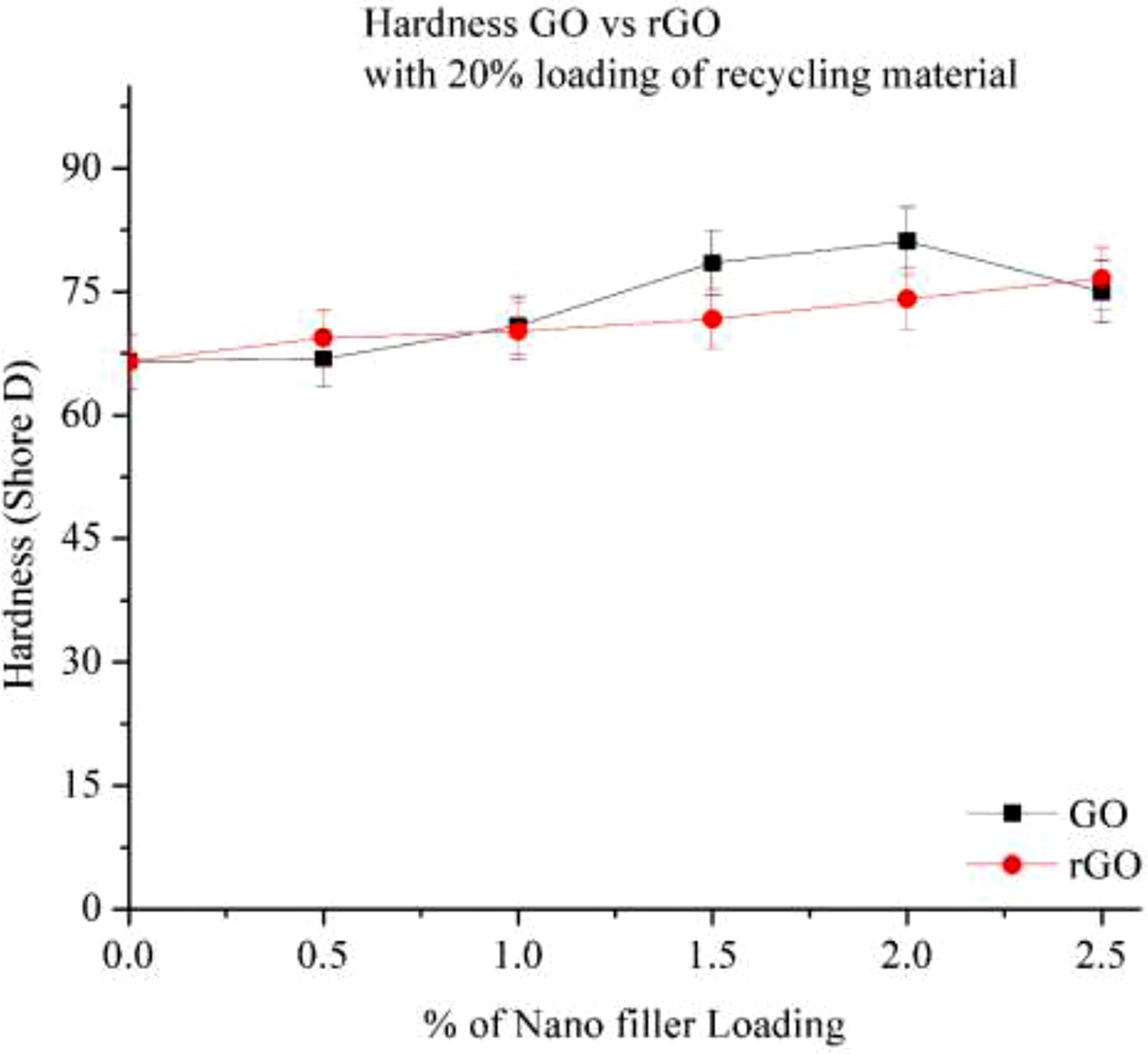
Impact strength
Figure 19 illustrates the impact strength of a 15% recycled Nylon 6 composite with GO and rGO. As the nano-filler percentage increases, the impact strength decreases, indicating that the material becomes more brittle due to the presence of Nano-fillers. In the case of GO, its amorphous nature with randomly attached oxygen functional groups (-OH, -COOH, -C = O) on the graphene plane makes the Nylon composite more brittle, leading to a reduction in impact strength while improving stiffness. The impact strength declines from 76.4 J/m (15% RP) to 29.9 J/m (2.5% GO), reflecting an almost 60% reduction. This suggests that while GO improves the mechanical strength and stiffness, it negatively affects impact resistance. For rGO, the impact strength also decreases with increasing filler content, but not as consistently as in GO. A drastic reduction is observed at 0.5% loading, followed by a ∼35% increase at 1.0% loading, likely due to non-uniform dispersion of rGO within the Nylon composite. The impact strength then declines at 1.5% loading before increasing again at 2.0% loading, suggesting variations in filler dispersion and interfacial interactions. Overall, the best impact strength for GO is observed at 2.0% loading, while rGO shows fluctuations in impact resistance due to its dispersion behaviour in the polymer matrix. Impact strength of Go and rGO with 15% RP.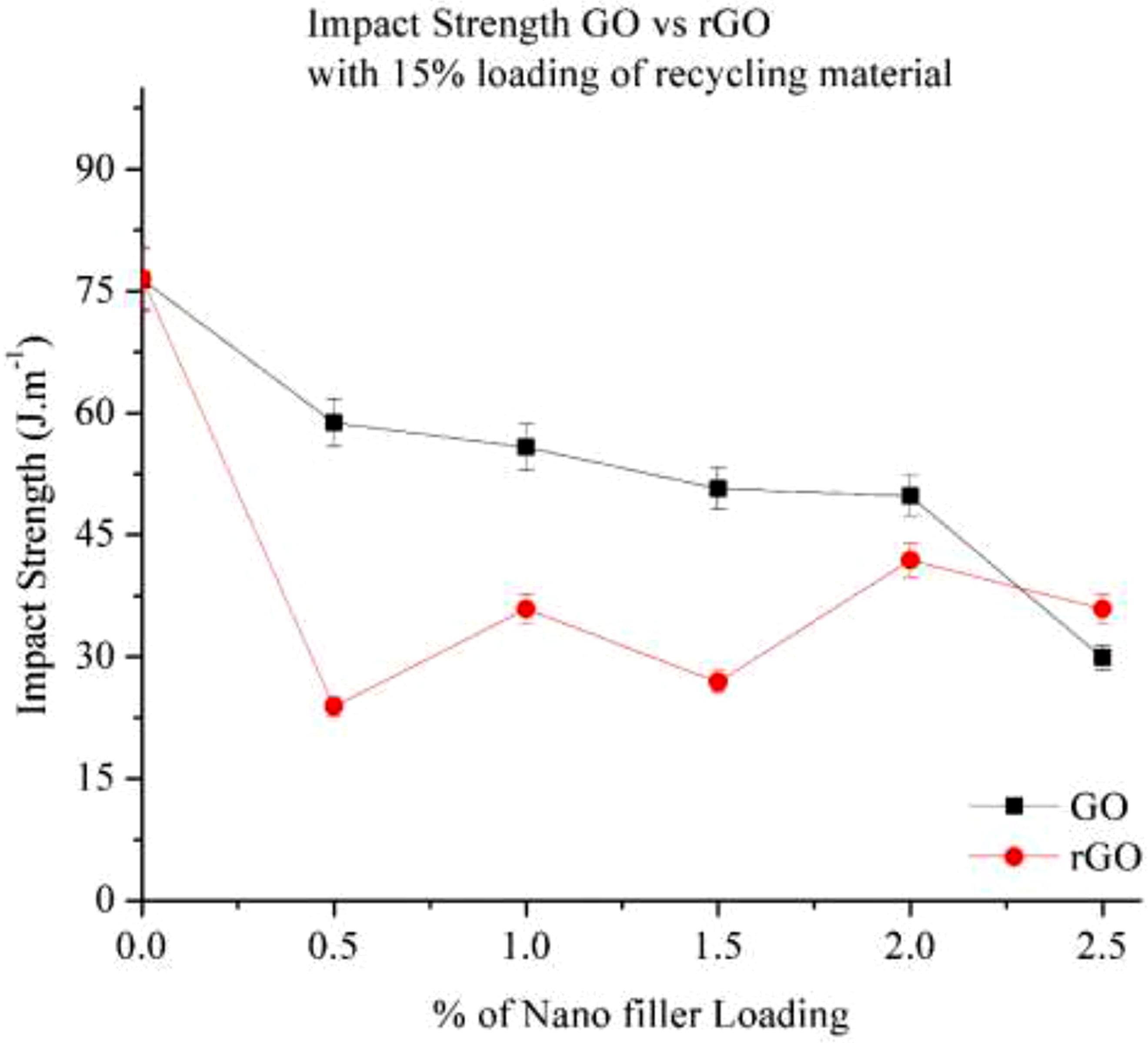
Dielectric strength
Dielectric strength of 15% RP with GO vs rGO depicted in Figure 20, as shown in figure the dielectric strength is increase compare to o % Loading, at this loading it content 15% Recycle material it contains some impurities. Recycling introduces contaminants, oxidation, and polymer chain scission, which create defects in the material. These defects increase free charge carriers, leading to higher conductivity and lower dielectric strength. After adding GO in recycling material dilectric strength not much changes as it contents many oxygen-containing functional groups (-OH, -COOH, -O-), disrupting the conductive sp2 carbon network and increasing resistivity. After adding the GO in 15% RP its dielectric strength increase and then it is stable after 2%, not much change in Dielectric strength were observed . in rGO the dielectric strength get reduces with increasing the % of rGO loading resulting good Electric conductivity.
25
GO has many oxygen functional groups (-OH, -COOH, -O-), which create polar domains that increase dielectric strength. When GO is reduced to rGO, these groups are removed, reducing polarizability and dielectric breakdown resistance. so compare to GO and rGO Go is good insulator but rGO has good conductor. Dielectric strength of Go and rGO with 15% RP.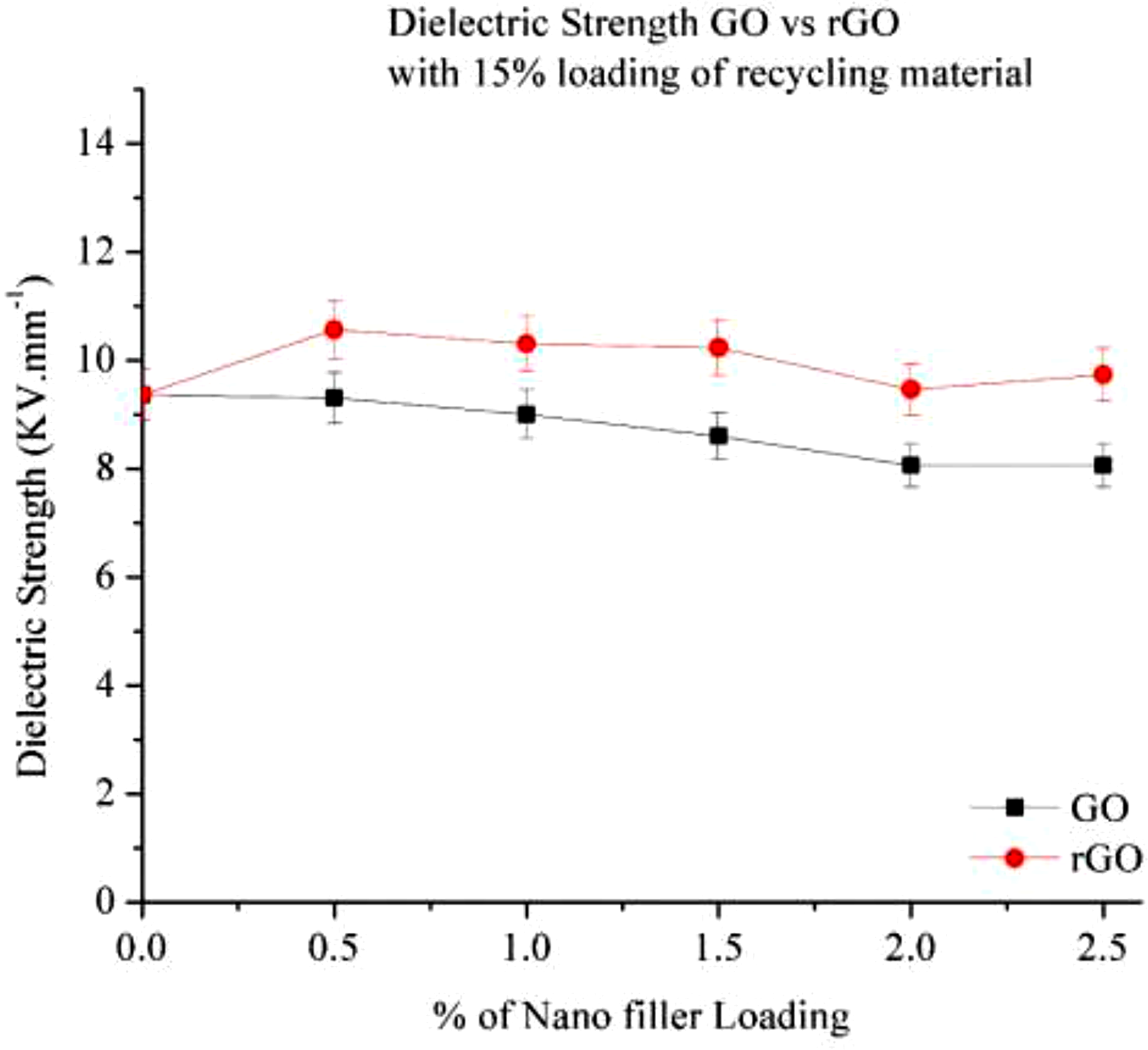
Figure 21 illustrates the dielectric strength of GO and rGO in a 20% RP composite. The data reveals that dielectric strength decreases with increasing GO and rGO content, indicating an improvement in electrical conductivity, making the material more conductive. Dielectric strength of Go and rGO with 20% RP.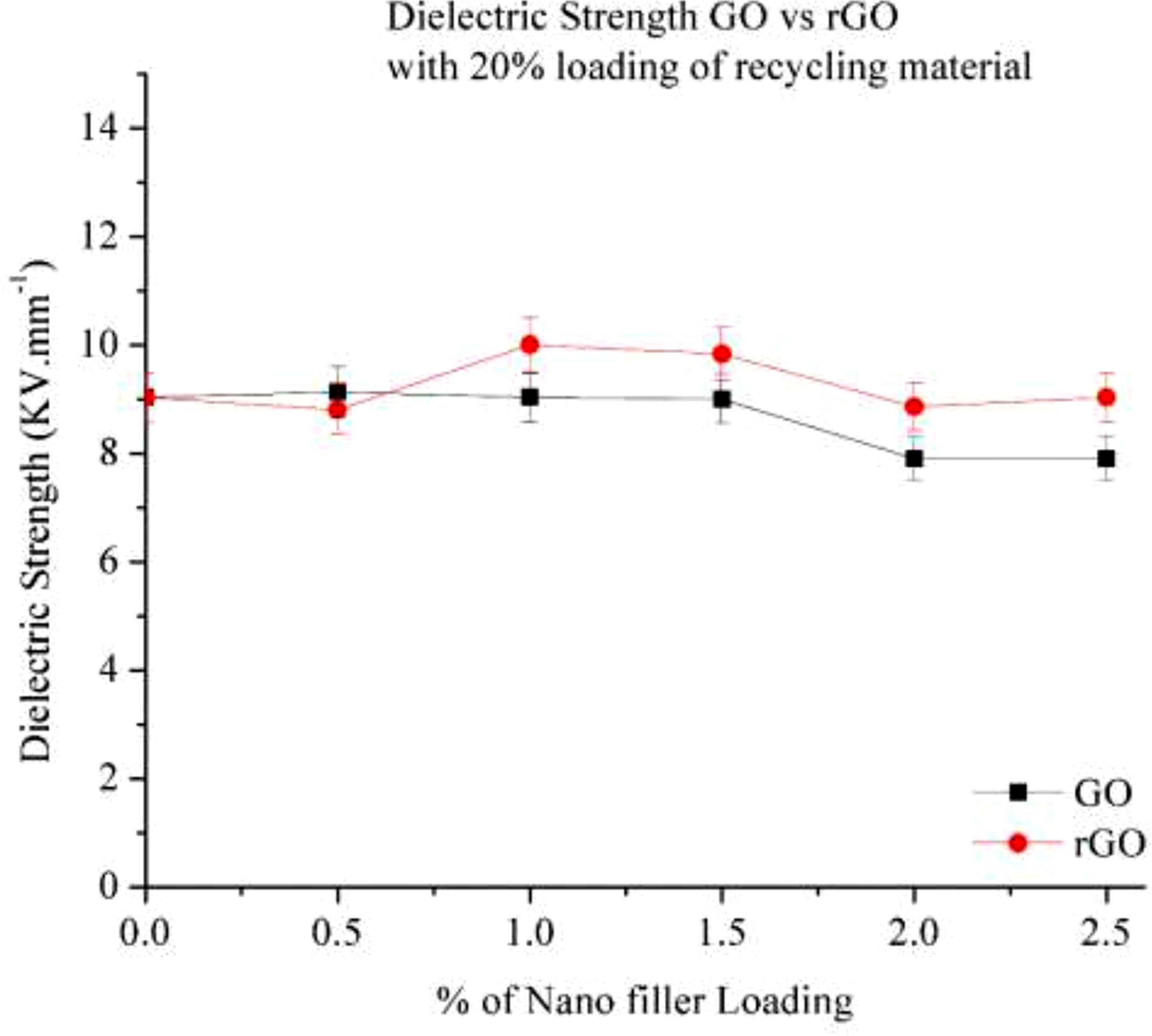
At 0.5% GO loading, dielectric strength increases, suggesting that GO initially acts as an insulator. However, with the addition of 1% and 1.5% GO, a slight decrease in dielectric strength is observed. The best conductivity is achieved at 2% GO loading, after which dielectric strength increases again, possibly due to the agglomeration of GO particles.
In contrast, when rGO is added to the 20% RP composite, dielectric strength continuously decreases with increasing rGO content, indicating that rGO is more electrically conductive than GO. This is due to its lower oxygen content, which enhances its conductivity. At 2% and 2.5% rGO loading, a significant improvement in electrical conductivity is observed, with a nearly 12% reduction in dielectric strength.
Melt flow index
Figure 22 illustrates the Melt Flow Index (MFI) behaviour of GO and rGO in a 15% RP composite. The results indicate that as the GO content increases, the MFI decreases. At 0.5% GO loading, no significant change in MFI is observed. However, beyond 1% GO loading, a notable reduction in MFI occurs, with a 13% decrease at 1% GO and a 14% decrease at 1.5% GO. At 2% GO loading, the MFI drops significantly by approximately 34%, indicating a substantial increase in material viscosity and molecular weight. Interestingly, beyond 2% GO loading, a slight increase in MFI is observed, possibly due to the uneven distribution of GO within the Nylon composite. Melt flow index of Go and rGO with 15% RP.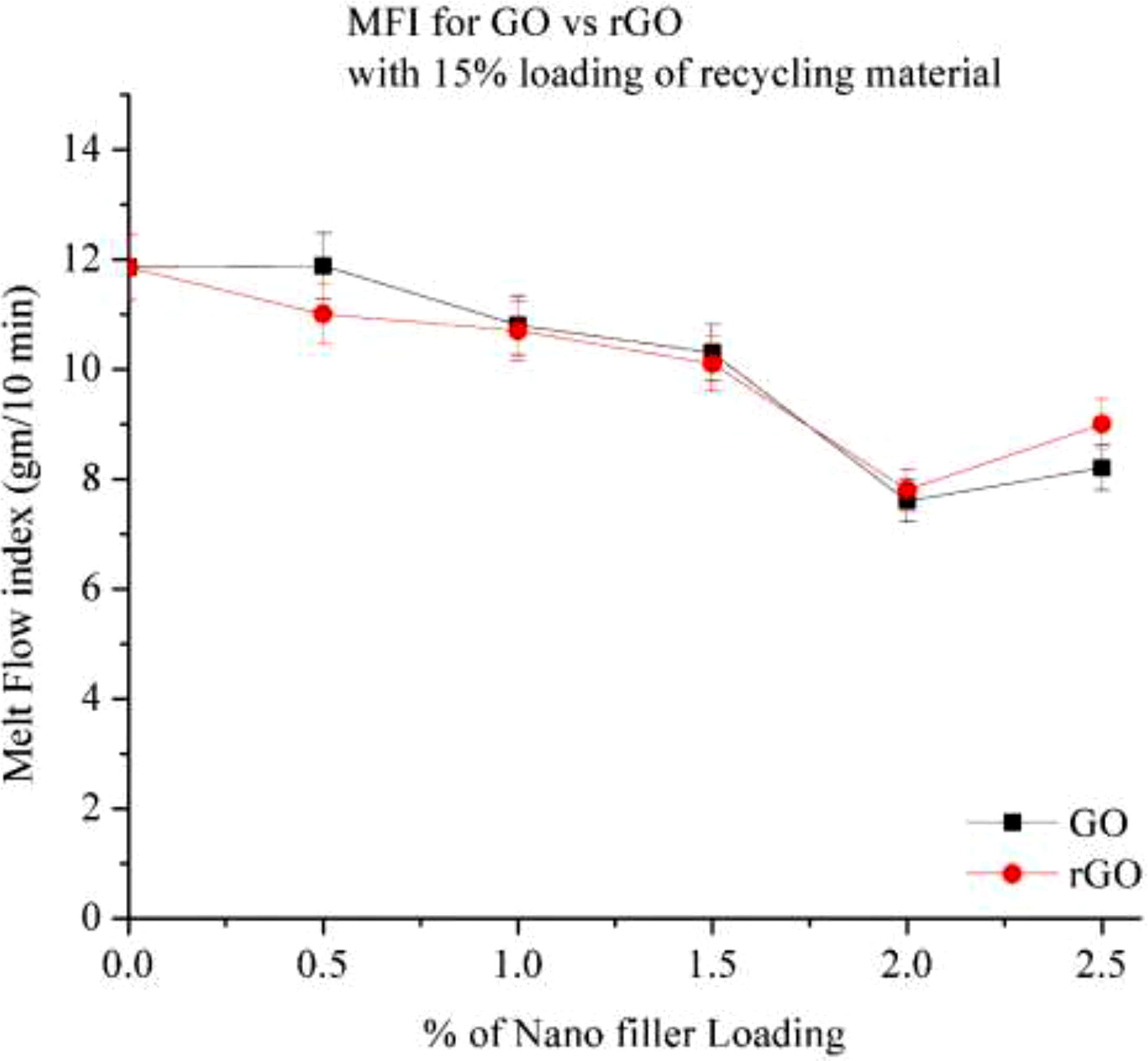
In contrast, for rGO, the MFI consistently decreases with increasing rGO content. However, at 1.5% rGO loading, minimal changes in MFI are observed when compared to the 15% RP composite. Beyond this point, the MFI continues to decrease. When comparing GO and rGO, GO demonstrates a more pronounced effect on MFI reduction. This is attributed to the presence of oxygen-containing functional groups (such as hydroxyl and carboxyl), which enhance interfacial bonding and result in a stiffer material. Conversely, rGO, with fewer oxygen-containing groups, does not significantly impact MFI to the same extent as GO.
Similar to the 15% RP composition, the melt flow index (MFI) of the 20% RP composition is illustrated here in Figure 23. Increasing the filler percentage leads to a decrease in MFI for both GO and rGO compounded with 20% RP. At 15% RP, the MFI is 11.85 g/10 min, whereas at 20% RP, the MFI increases to 12.25 g/10 min. This indicates that mechanical recycling deteriorates the material properties, resulting in weaker mechanical strength. After the addition of 0.5% filler, the MFI increases, possibly due to impurities in the recycled material, which weaken the bonding and lead to poor results. Melt flow index of Go and rGO with 20% RP.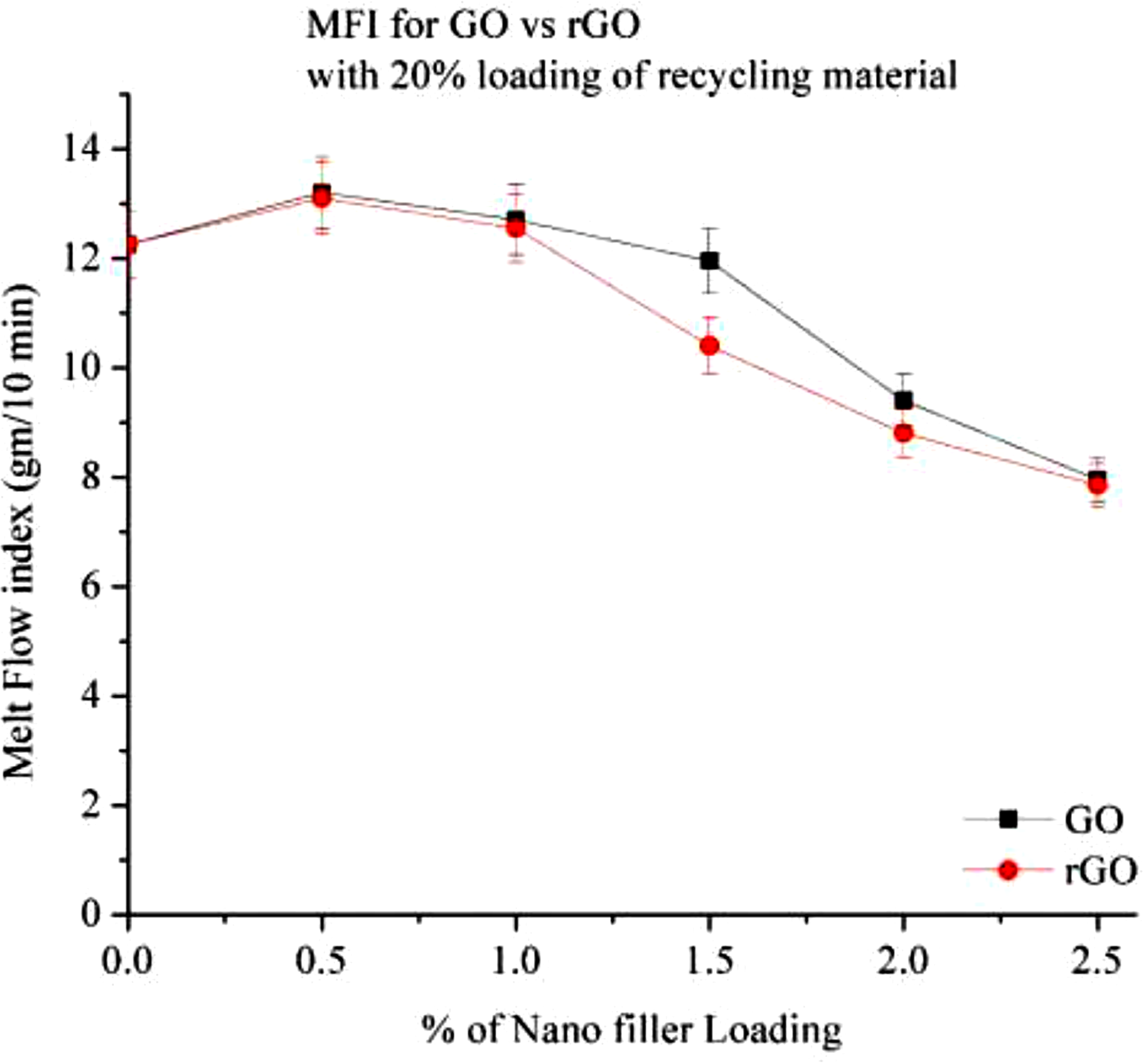
The initial increase in MFI at low concentration of nanofiller cab be attributed to lubricating effect of the nanofillers. However, higher loading leads to increase in viscosity and thereby reduction in MFI. 26 A noticeable change in MFI is observed at 1% filler loading, while at 1.5% loading, significant differences in MFI values are recorded. Both GO and rGO exhibit similar MFI trends.
FTIR: (Fourier transform infrared)
FTIR of virgin Nylon 6.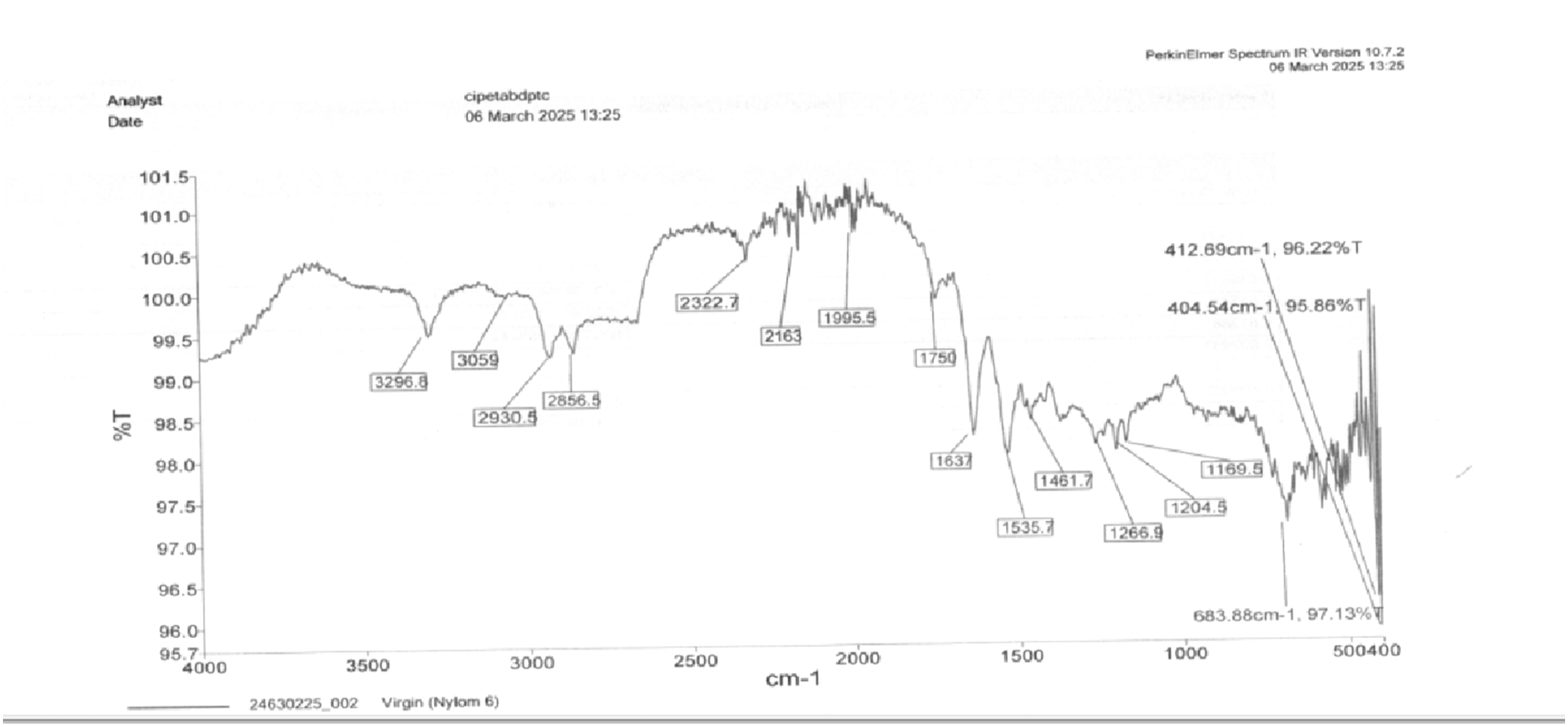
Figure 25 shows the FTIR image of 15% RP and 2.5 % GO as per the spectrum. 3306.1 cm−1 (N-H Stretching - Amide Group). Slight shift from 3296.8 cm−1 in pure Nylon 6, indicating interaction between N-H groups and rGO. 2948.9 cm−1, 2871 cm−1 (C-H Stretching - Aliphatic Groups). These peaks confirm the presence of CH2 groups in the polymer backbone. 2676.93 cm−1 (Possible O-H Stretching). This might be due to moisture absorption or residual hydroxyl (-OH) groups from rGO. 2330.5 cm−1 (Possible C ≡ C or C ≡ N Stretching). This may be related to the sp2 hybridized carbon domains in rGO. 2165.6 cm−1 (Overtones or Carbonaceous Groups). Could indicate π-π interactions between Nylon 6 and rGO. 1987 cm−1 (Combination Bands). Not typically observed in pure Nylon 6, might be due to rGO-induced changes. 1629.8 cm−1 (Amide I Band - C = O Stretching). Shifted from 1637 cm−1 in pure Nylon 6, suggesting interaction between amide and rGO. 1533.6 cm−1 (Amide II Band - N-H Bending & C-N Stretching). This remains similar to pure Nylon 6, confirming the polymer’s structure. 1373.3 cm−1 (C-H Bending or C-OH Stretching).
28
Could be related to oxygenated groups in rGO. 1208.4 cm−1 (C-N Stretching - Amide III Band). Consistent with Nylon 6. 625.64 cm−1, 429.94 cm−1, 413.96 cm−1 (Fingerprint Region). These shifts suggest modification due to rGO incorporation. FTIR of 15% RP and 2.5% rGO.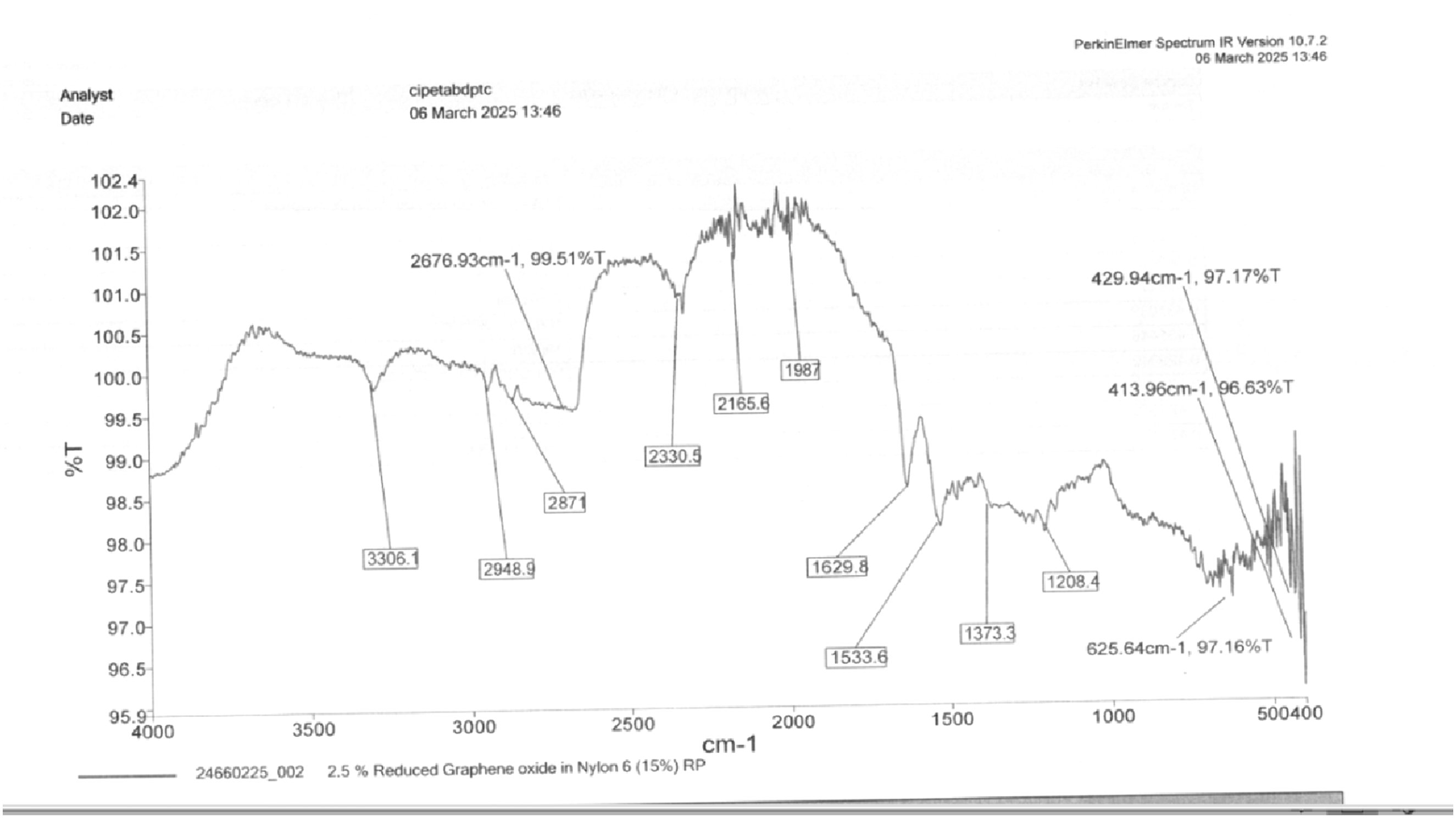
The amide I (C = O) and amide II (N-H, C-N) bands shifted, indicating interactions with rGO. The new peaks at 2676.93 cm−1, 2330.5 cm−1, and 1987 cm−1 suggest functional groups from rGO. The broadening and shifting in the 3306 cm−1 (N-H) and 1629.8 cm−1 (C = O) bands indicate hydrogen bonding and π-π stacking between Nylon 6 and rGO. The spectrum confirms successful incorporation of rGO into Nylon 6. The peak shifts and new absorption bands suggest interactions between the polymer matrix and rGO. 16
Figure 26 Shows that (N-H Stretching - Amide Group) Characteristic peak for amide groups in Nylon 6. This peak slightly shifts compared to pure Nylon 6, indicating interactions with graphene oxide. 2933 cm−1, 2863.8 cm−1 (C-H Stretching - Aliphatic Groups). These confirm the presence of CH2 groups in Nylon 6.(Possible O-H or C-H Stretching) Could be due to moisture absorption or hydroxyl (-OH) groups from GO. 2329 cm−1 (C ≡ C or C ≡ N Stretching). Indicates graphene oxide’s carbonaceous structures. 2167.7 cm−1, 2057.1 cm−1 (Possible Carbonyl or Overtones). These peaks might be related to oxygen functionalities in GO. 1633 cm−127 (Amide I Band - C = O Stretching). Slightly shifted from 1637 cm−1 in pure Nylon 6, suggesting hydrogen bonding with GO. 1536.2 cm−127,28 (Amide II Band - N-H Bending & C-N Stretching) indicates the presence of amide bonds. 1462.5 cm−1 (C-H Bending). Related to the polymer backbone. 1370.3 cm−1 (C-H Deformation or C-OH Stretching). The observed FTIR spectrum confirms the presence of both Nylon 6 and graphene oxide in the composite. The characteristic peaks of Nylon 6 remain intact, indicating that the polymer backbone has not undergone significant degradation upon GO incorporation. The presence of additional peaks at 2167 cm−1 and 1079 cm−1 confirms the successful dispersion of GO within the polymer matrix. FTIR spectrum of 2.5% graphene oxide (GO).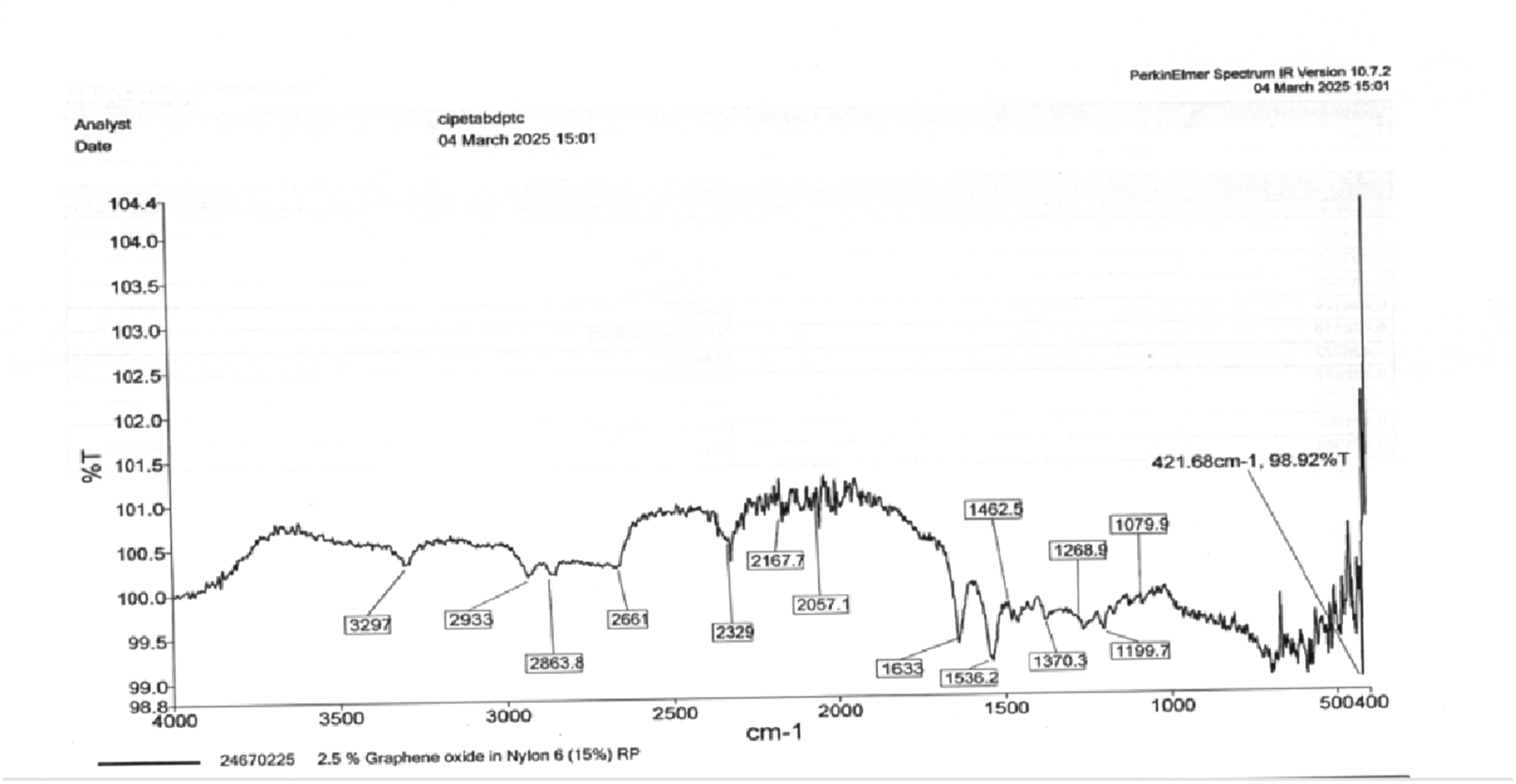
The interaction between GO and Nylon 6 is evident from the slight shifts in the amide peaks (3207 cm−1, 1643 cm−1, and 1536 cm−1), suggesting hydrogen bonding or π-π interactions between GO’s oxygen functional groups and the amide functionalities of Nylon 6. These interactions contribute to enhanced interfacial adhesion, which could lead to improvements in mechanical, thermal, and electrical properties.
Scanning electron microscope (SEM)
Scanning Electron Microscopy carried out using gold coating to get details dispersion, Figures 27–29 shows the SEM analysis of fracture surface of 15% RP with GO, rGO and virgin nylon 6, good mechanical strength, Dielectric strength observed in 2% Loading of GO and rGO is because of Good dispersion of reduce graphene oxide and graphene oxide in Nylon 6 with 15% RP material. In Figure 29 we can see that there is no such bonding between the molecule at void area resulting poor mechanical strength. Viscosity and conductivity. SEM of 15% RP + 2% rGO. SEM of 15% RP + 2% Go. SEM of 15% RP + nylon.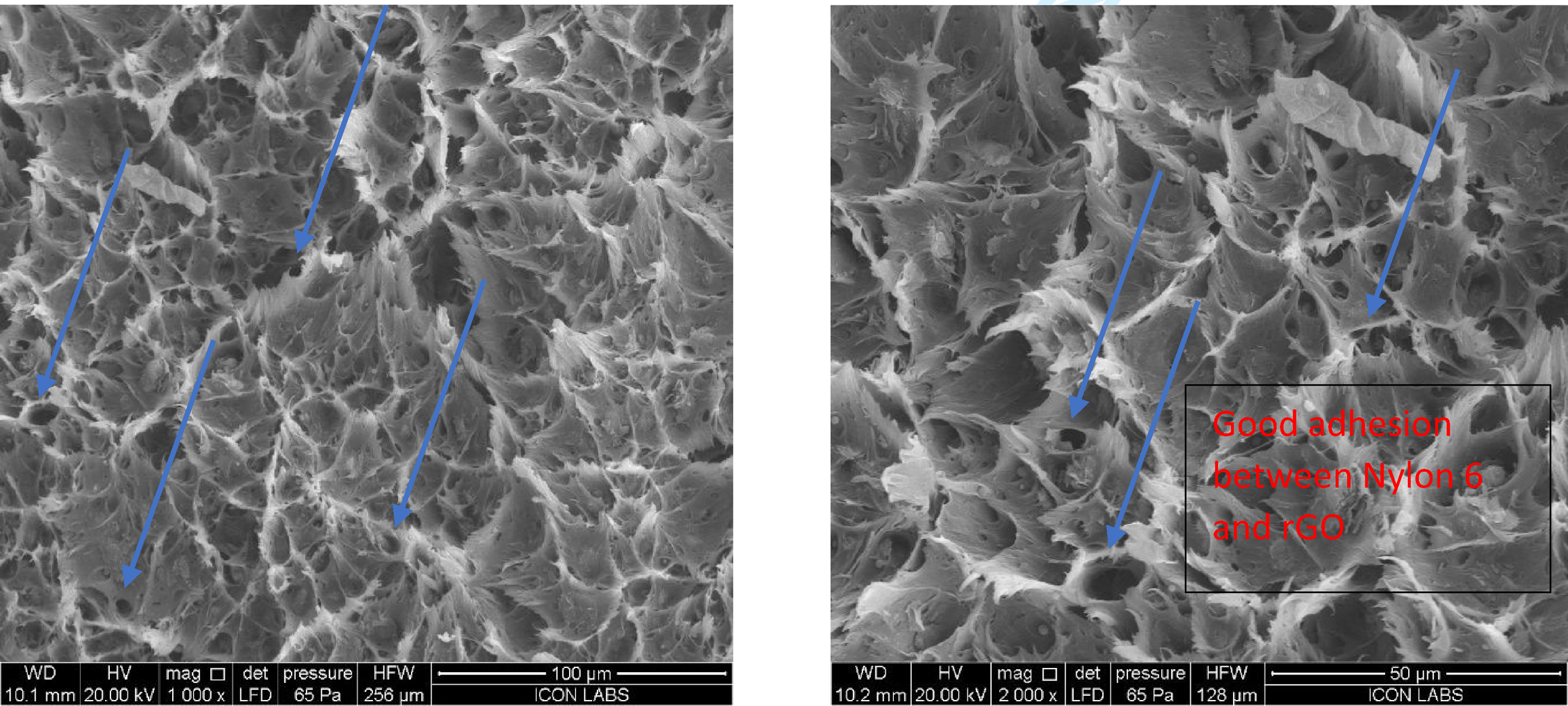


Conclusion
This study investigates the effect of Graphene Oxide (GO) and Reduced Graphene Oxide (rGO) (0.5%–2.5%) on the mechanical, electrical, and flow properties of recycled Nylon 6 with 15% and 20% recycled polymer (RP). The key findings are: • Tensile properties (strength and modulus) improved with increasing GO and rGO content, reaching an optimum at 2% loading. In 15% RP, GO exhibited a 28.33% increase in tensile strength, while rGO showed a 21.67% increase. In 20% RP, both fillers enhanced strength at 2% loading, though GO in 15% RP provided superior results. However, beyond 2% loading, tensile properties declined due to agglomeration effects. • Material stiffness increased with GO and rGO addition, as indicated by a reduction in elongation at break. The highest stiffness enhancement was observed at 2% loading in both 15% and 20% RP, while higher filler concentrations led to embrittlement. • Flexural properties (strength and modulus) improved with increasing GO and rGO content, with rGO performing better than GO. In 15% RP, rGO resulted in a 13% increase in flexural strength, while GO showed an 8% increase at 2% loading. Similarly, flexural modulus increased by 7% with GO and 16% with rGO, attributed to the reduced oxygen functional groups in rGO, which enhanced bonding. • Hardness increased with nano-filler loading, with GO exhibiting a 5% improvement at 2% loading, while rGO provided better performance at 2.5% loading. In 20% RP, GO showed superior hardness, possibly due to impurity content improving adhesion. • Impact strength decreased with increasing GO and rGO concentration, leading to material embrittlement. A consistent decline was observed with GO, while rGO showed some variation, with the best performance at 2.5% loading. • Electrical conductivity increased with filler loading, enhancing the electrical performance of both 15% and 20% RP composites. However, dielectric strength decreased with increased nano-filler content, with a maximum reduction of 14% at 2% rGO loading in 15% RP, while GO had a lesser effect. At 2% loading in 20% RP, dielectric strength reduced by 5% for GO and 12.5% for rGO. • Melt Flow Index (MFI) decreased with increasing GO and rGO content, indicating higher viscosity and improved mechanical properties. Optimal MFI performance was observed at 2% loading in 15% RP, while 20% RP exhibited a further decline at 2.5% GO loading. • 2% loading of GO and rGO provided the best balance of mechanical, electrical, and flow properties in both 15% and 20% RP composites. • Excessive loading (>2%) led to agglomeration, embrittlement, and reduced flowability, negatively affecting overall performance. • rGO generally outperformed GO in flexural properties, modulus, and electrical conductivity, while GO showed superior hardness and adhesion in 20% RP.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
