Abstract
CF/PEEK composite components are often subjected to hygrothermal conditions during service, yet the microscopic mechanisms of their hygrothermal damage and failure is difficult to explain by experimental techniques alone. This study integrates experimental techniques with molecular dynamics (MD) simulations to investigate the interfacial damage and failure mechanisms of CF/PEEK bushings under such conditions. The results reveal that the primary failure mode is interfacial debonding, which leads to matrix cracking. At the atomic level, the migration of water molecules from the matrix to the interface is captured, providing insights into the water-PEEK interaction mechanisms. A systematic analysis of the effects of the failure process shows that increased moisture absorption in PEEK raises the levels of bound and free water, weakens the intermolecular forces and increases the mobility of molecular chains, which is exacerbated by the effect of elevated temperatures. The thermal expansion coefficients of CF and PEEK are calculated by MD simulation and found to be 5-8 times different, promoting interfacial debonding and matrix cracking. Both moisture and temperature exacerbate the degradation of PEEK’s tensile properties at the interface, making the matrix more susceptible to collapse and fracture under external mechanical loads. These findings provide valuable atomic-level insights into the mechanisms governing the reduced interfacial integrity and mechanical performance of CF/PEEK composites in hygrothermal environments.
Keywords
Introduction
In modern industries, carbon fibre reinforced polymer (CFRP) composites are widely used in automotive manufacturing, aerospace, biomedical fields and textile industry.1–3 This is because they have excellent mechanical properties, chemical stability and fatigue resistance. However, CFRP composites are subjected to a combination of heat, moisture and various loads during service. This exposure can lead to various modes and degrees of damage and failure in matrix, carbon fibres (CFs), and CF/matrix interface.4–6 The initiation and evolution of these types of damage significantly contribute to the performance degradation of CFRP composites and ultimately reduce the overall structural durability, shortening the service life of the materials.7–9 Therefore, enhancing the durability and extending the service life of these bushings is crucial. To achieve this, it is essential to explore the damage and failure mechanisms of CFRP composites components under hygrothermal conditions.
Experimental methods are the main approach for studying the failure mechanisms of CFRP composites in hygrothermal environments currently. For example, Tao et al. 10 used infrared spectroscopy to analyze the hygrothermal aging process of CFRP prepregs, finding that the chemical structure of the material was unaffected by moisture and heat ageing. Meng et al. 11 used scanning electron microscopy analyzed the failure mechanism of CF/epoxy composites under bending after immersion in water at elevated temperatures. They found that CF/matrix interface degraded after immersion, significantly reducing the mechanical properties of the composites. Bismarck et al. 12 study the behavior of CF/PEEK composites subjected to compressive bending loads, found that the higher the temperature, the faster the composites fails. After boiling water treatment, the tensile strength in the fibre radial direction significantly decreased. They proposed two possible causes for this result. The first cause is water absorption may plasticize PEEK, reducing the matrix’s resistance to bending on the compressive side of the CFs. The second cause is water absorption could weaken the CF/PEEK interface. These findings explain that the primary cause of hygrothermal degradation and failure in CFRP composites is the differential hygrothermal response of CF and the matrix. 13 An in-depth study of the failure mechanism is needed to support their conjecture.
Traditional experimental methods can characterize the damage patterns of CFRP composites to a certain extent; however, they are resource-intensive and time-consuming. Furthermore, CFRP composites are subjected to extreme application environments, these methods struggle to replicate the actual conditions effectively. Finite element simulation methods can availably and cost-effectively predict the hygrothermal damage and failure of CFRP composites at both macro and micro scales, including model analysis, 14 continuum damage mechanics, 15 and classical lamination theory. 16 For instance, Gholami et al. 17 proposed a novel hygrothermal mechanical coupling multiscale algorithm for accurately predicting the mechanical and destructive behavior of CFRP laminates exposed to hot and humid conditions. Xu et al. 18 established a high-fidelity explicit discrete crack macro model, which points out the impact of hygrothermal ageing on the high-velocity impact performance of CFRP composites. Yue et al. 19 reported that interface debonding is the main form of failure in CFRP thin-walled bushings under hygrothermal environment. As a key factor in determining the performance of a bushing, it is important to understand the mechanism of interface debonding and its role in the failure process to improve the overall performance of the bushing.
The mechanisms of CFRP composites damage and failure in hygrothermal environments are quite complex. Researchers are increasingly attempting to investigate these issues using novel methods at smaller microscopic scales. Molecular dynamics (MD) simulation not only allows for the modeling of extreme environmental conditions but also renders insights into the molecular interactions within the material at a molecular level.20–22 There is currently a great deal of interest in utilizing MD simulation to investigate the performance and damage behavior of CFRP composites. Vuković and Walsh 23 studied the mechanisms of moisture ingress at the CF/epoxy interface during the hygrothermal aging process. Their simulations indicated that water initially penetrates the matrix and then migrates to the interface as the polymer molecular chains move dynamically. Tam et al. 24 investigated the influence of water molecules on the CF/epoxy interface behavior at various temperatures. They found that increased temperature further diminishes the mechanical properties at interface. But MD simulation has primarily been applied to study the effects of individual factors such as moisture25–28 or temperature29–31 on the degradation of CFRP composites in environmental conditions. Researches on the combined effects of hygrothermal conditions are still limited, and the mechanisms of water molecule-PEEK matrix interactions after moisture enters the matrix are still unclear.
Thousands of CF/PEEK composite bushings in stenter heat-setting machines operate in high-temperature, humid environments, making them susceptible to damage and failure. This results in frequent maintenance and replacement, ultimately decreasing the efficiency of printing and dyeing operations. In this study, we investigate the damage and failure mechanisms of CF/PEEK composite bushings under hygrothermal conditions. Our approach includes failure experiments to observe the morphology of damaged bushings and MD simulations to investigate the internal atomic states and the evolution of interfacial damage. We systematically and quantitatively discuss the following four key aspects: the molecular interaction of water-PEEK at the interface, the expansion behavior of CFs and matrix, the interfacial interaction energy, and the mechanical properties of both the interface and the matrix. This research provides novel molecular-level insights and theoretical guidance for the application of CF/PEEK composites in extreme environments.
Methodology
Failure experiments of bushings
CF/PEEK composite bushings are prepared using an injection molding method. Primarily, solid granular raw material (VICTREX® 150CA30) is loaded into a vertical injection molding machine (SLZ-125, Changzhou Jingdiao Injection Molding Machinery Co., Ltd). Under the influence of high temperature and screw motion, the granules are melted into a molten state at 385°C. Subsequently, the molten material is injected into the mold through a gate, with the injection process controlled by the screw movement speed. The mold surface temperature is set to 190°C. Once the mold cavity is fully filled, the pressure is maintained at 80% of the filling pressure for 10 seconds. Finally, the bushing samples are ejected from the mold using ejector pins.
The actual service life of the bushings is approximately 5 months. During this period, they are subjected to temperature cycling ranging from indoor temperature to 150°C in the humid environment, while both the inner and outer surfaces experience complex and variable loads. Standard experimental equipment such as universal testing machines cannot fully replicate these real-world conditions. To better mimic the actual working environment, the bushings are installed on the chains of stenter heat-setting machines and tested during the actual production process. Faulty bushings are identified and screened for further analysis.
To observe the microscopic damage morphology of the faulty bushings, a Hitachi S-4800 scanning electron microscope (Japan, Hitachi) is utilized to examine the fracture surfaces.
Molecular dynamics simulation
Model construction
All models in this paper are constructed using Materials Studio (2019 edition). The process of constructing the molecular model of PEEK is shown in Figure 1(a) and (d). A PEEK molecular chain consisting of 10 repeating oxy-p-phenylene-oxy-p-phenylenecarbonyl-p-phenylene units (PEEK monomers) is created. Then, 30 such molecular chains are packed into a box to obtain an amorphous PEEK molecular model. The molecular weight used in the simulations (2900 g/mol) is much lower than the experimental molecular weight (100,000 g/mol), but because interfacial interactions are primarily affected by localized chain segments rather than the entire polymer chain, the use of simulated 10-repeat-unit PEEK chains is a compromise between computational feasibility and capturing the nature of interfacial chemistry. To simulate the moisture absorption effect of the PEEK matrix, water molecules is randomly added into the PEEK box. The water molecules modeled is using the TIP4P water model parameters.
32
The non-bonding calculations between the CVFF and TIP4P water models follow the Lorentz-Berthelot rules, which allows for an efficient discussion of the interactions between polymers and water molecules (hydrogen bonding interactions).
33
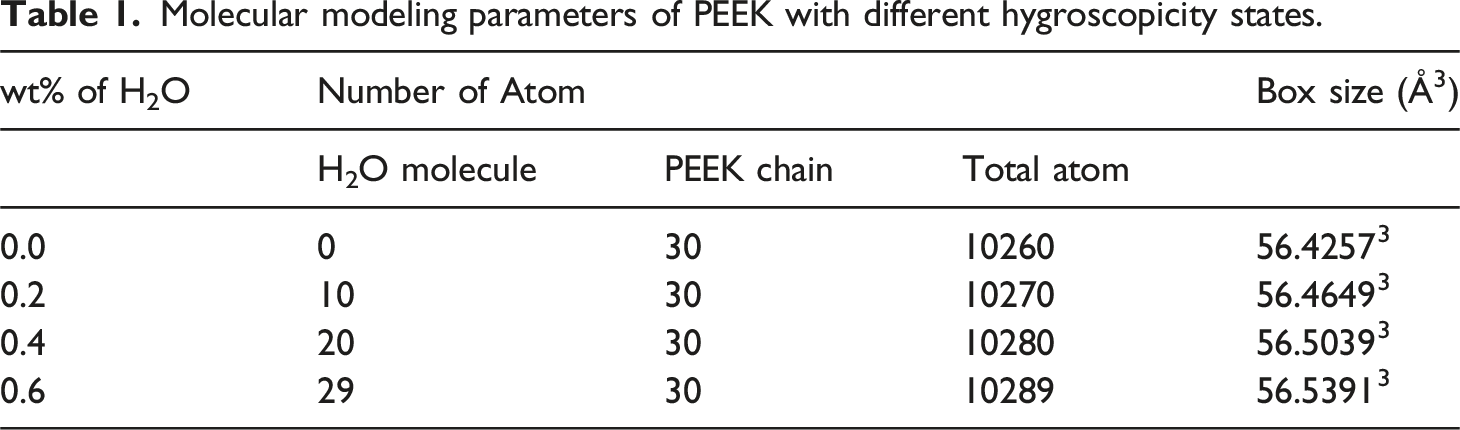
Considering that PEEK has a maximum moisture content of approximately 0.6 wt%,
34
PEEK molecular models with moisture content of 0.2 wt%, 0.4 wt%, as well as 0.6 wt% are constructed. The model parameters are shown in Table 1. Molecular modeling of (a) PEEK monomer, (b) PEEK molecular chain, (c) PEEK box without water, (d) PEEK box with water, (e) ladder structure of CF, f initial configuration of CF, (g) final configuration of CF, (h) model of CF/PEEK interface. Molecular modeling parameters of PEEK with different hygroscopicity states.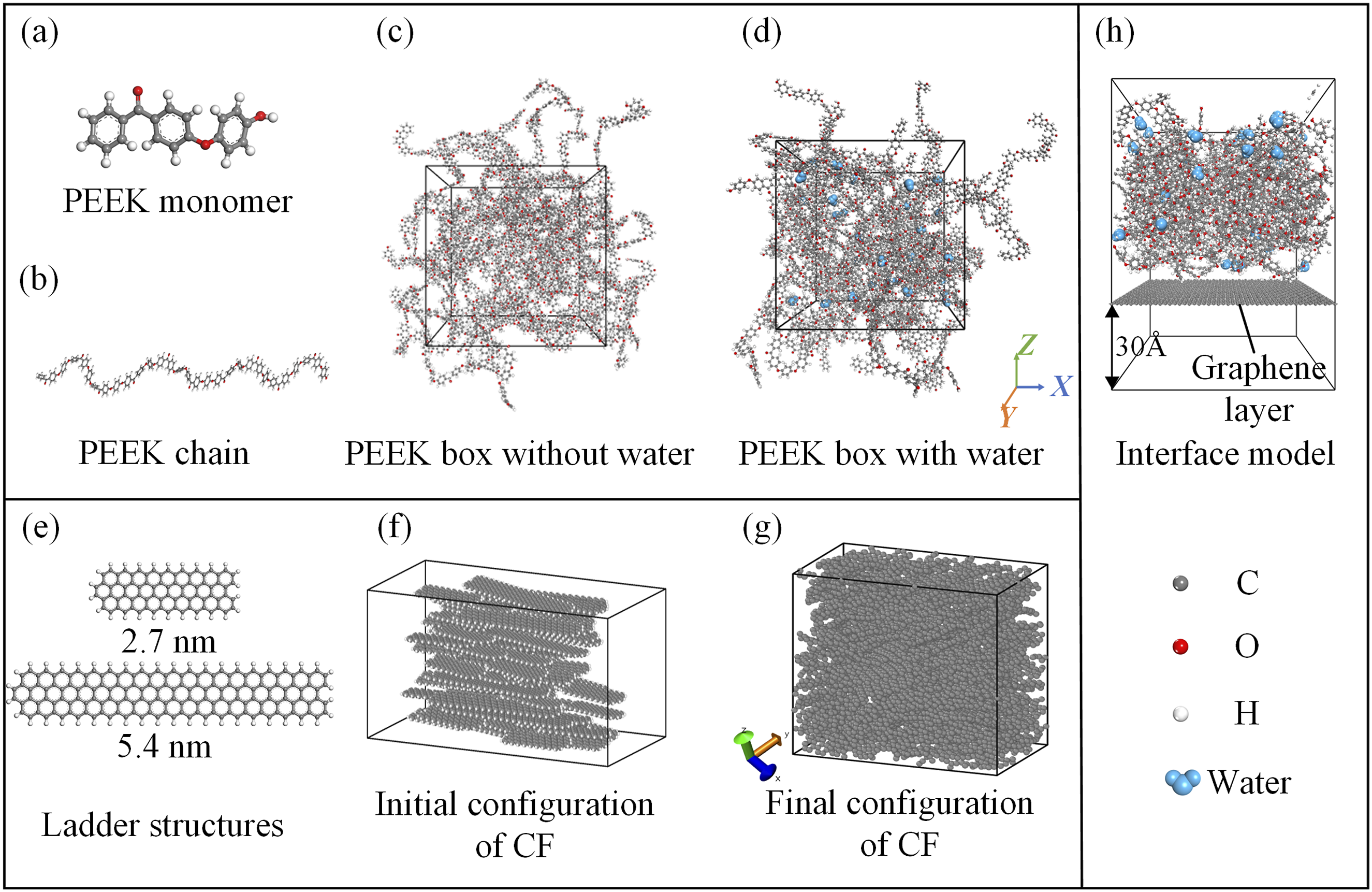
A three-dimensional model of CF is constructed to characterize the expansion of CF. It is generally believed that the microstructure of CF consists mainly of long lamellae (graphene layers) of sp2-bonded carbon atoms, 35 thus the initial configuration of the CF model is created using 50 ladder structures with a length of 2.7 nm and 25 ladder structures with a length of 5.4 nm. The size of the ladder structures can have an effect on the density, degree of graphitization, etc. of the CF model, and the size used in this study was noted to represent the CF model better. 35 The ladder structures are randomly added into the box of size 100 x 50 x 30 Å3, with an orientation range of 0-±10° along the X and Y axes, and an initial density of 0.79 g/cm3. These ladder structures consist of hexagonal carbon rings, with edges saturated by hydrogen atoms, which undergoes dehydrogenation and rearrangement to form the graphite phase. The process of CF model construction is shown in Figure 1(e)–(g).
The CF/PEEK interface model is constructed by combining the previously built PEEK molecular model with a graphene layer, as shown in Figure 1(h). To reduce computational costs, a single graphene layer with dimensions of 56.4 x 56.4 Å2 is used. Although the interfacial model is highly idealized, the effects of the surface treatment of the graphene layers and the dispersive interactions between the graphene layers are not taken into consideration, a single-layer graphene-polymer interface model can still effectively represent the interfacial adhesion properties. 36 A vacuum layer at a height of 30 Å is added below the graphene layer to eliminate any additional interactions caused by periodic boundary conditions, ensuring that the graphene layer interacts with PEEK only on one side.
It should be noted that although it has been shown that the crosslinked network of polymers affects the hygrothermal response behavior of the material to a certain extent, 37 considering the fact that the formation of crosslinked network of the PEEK matrix in industrial applications is usually more limited under regular service conditions and in order to increase the efficiency of the calculation, the hygrothermal damage to the composite bushings in the present study is investigated based on the simplified assumption of linear repeating units. This study is based on the simplified assumption of linear repeating unit to investigate the wet heat damage of composite bushings.
Model relaxation
The constructed models are imported into the Large-scale Atomic/Molecular Massively Parallel Simulator (LAMMPS). 38 The above models first underwent energy minimization using the Verlet algorithm for 1 × 106 iterations.
PEEK model and the CF/PEEK interface model use the Consistent Valence Force Field (CVFF),
39
which is suitable for thermoplastic material systems, is applied. The formulae for CVFF are composed as follows equation (1).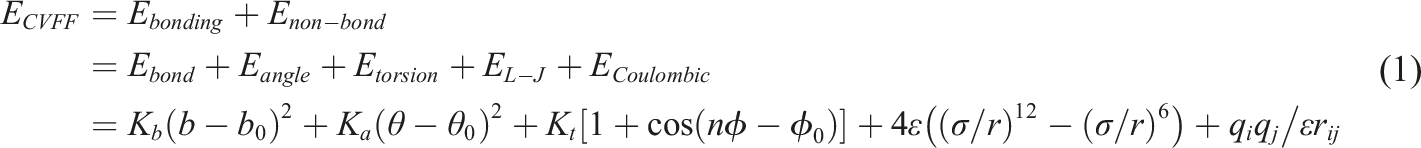
Relaxation equilibrium of models is achieved through simulations in canonical (NVT) and isothermal-isobaric (NPT) ensembles. The steps are as follows: (i) The model is maintained at a temperature of 600K for 500 ps in NVT ensemble; (ii) The temperature is first lowered from 600K to 200K and then raised back to 600K, with the pressure is maintained at 1atm in NPT ensemble. This cycle is repeated three times, for a total relaxation time of 3 ns.
The Nosé-Hoover thermostat is used to control the temperature and pressure. 40 A cut-off distance of 1.0 nm is utilized to calculate van der Waals and short-range Coulombic interactions. The particle-particle particle-mesh solver is utilized to calculate the long-range Coulombic interaction. The simulation time step in the above simulations is 1 fs.
For the initial CF model, the Adaptive Intermolecular Reactive Empirical Bond Order (AIREBO) potential,
41
which is suitable for carbon materials, is used. The AIREBO potential consists of three components as shown in equation (2).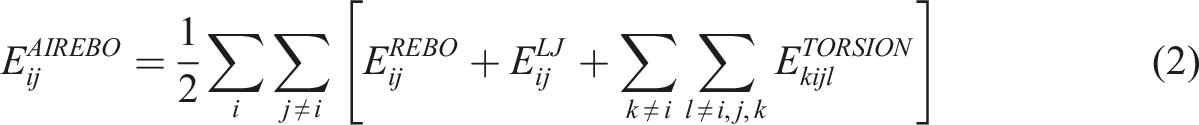
The relaxation simulation steps are as follows: (i) The model is heated to 300K within 100 ps in NPT ensemble, with the pressure is maintained at 1atm; (ii) The model is then compressed from 1atm to 0.3GPa at 300K; (iii) The temperature is raised from 300K to 900K at 0.3GPa constant pressure; (iv) At 900K and 0.3 GPa, hydrogen atoms are removed; (v) The pressure is then lowed to 1atm; (vi) An annealing cycle is conducted, in which the model is heated from 900K to 2000K over 100 ps, held at 2000K for 200 ps, and finally cooled to 300K over 100 ps.
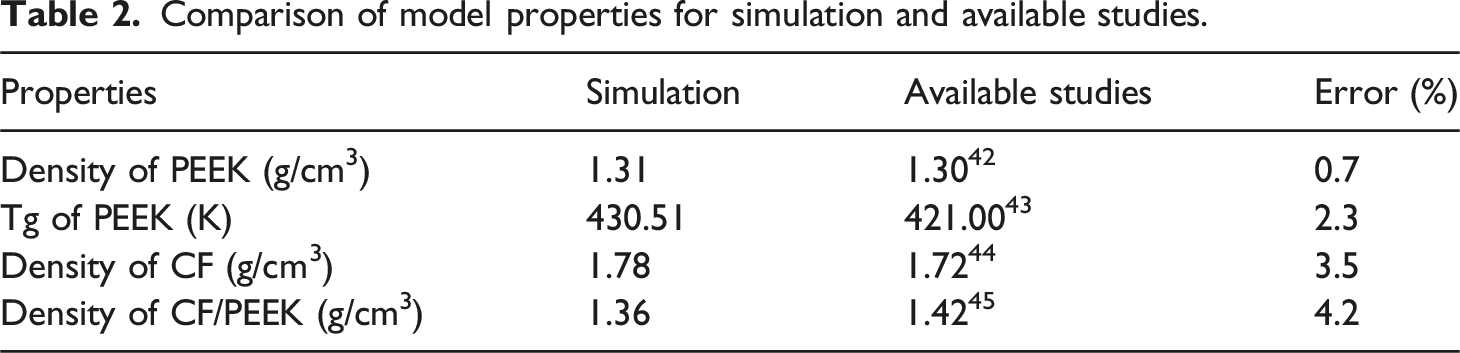
Comparison of model properties for simulation and available studies.
Interface damage evolution simulation
In order to study the damage evolution of CF/PEEK composite interfaces under hygrothermal conditions, traction-separation simulation is conducted on the CF/PEEK interface models with four different moisture contents under varying temperature conditions. The top 5 Å of the PEEK layer is fixed, while applying the Z-axis displacement of 0.015 Å to the graphene layer at every time step to induce separation between the graphene layer and the PEEK matrix, the interphase strain rate is about 0.01 fs−1 . In this simulation process, periodic boundary conditions in all directions except the Z-axis direction. When atoms moved out of one face of the periodic boundary, their coordinates are updated as if they re-entered the simulation box from the opposite face.
Characterization method
Radial distribution function and mean square displacement analysis
To investigate the interaction between water molecules and the PEEK at the interface, the radial distribution function (RDF) and the mean square displacement (MSD) are calculated. Both RDF and MSD for different moisture contents of the PEEK models are computed in NVT ensemble at the corresponding temperature conditions, with a simulation time of 100 ps.
RDF describes the probability of finding particle B around particle A within a spherical shell, which can represent the interaction between different particles, often used to study special interactions like hydrogen bonding. The expression for RDF is given as following equation (3):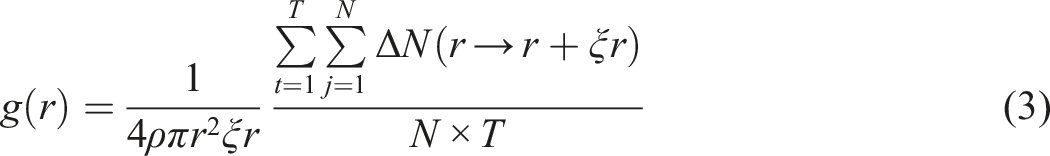
MSD represents the statistical average of the particle trajectories within the system at a given moment, characterizing the average displacement of particles over a specified time interval. The expression is given by equation (4):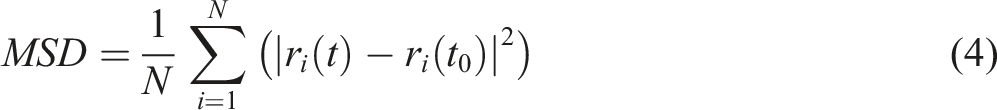
Expansion calculations
To characterize the effects of the hygrothermal environment on the expansion of interface components, the expansion coefficients are calculated. The coefficient of thermal expansion (CTE) represents the relative change in material volume for a temperature increase of 1 K. The expression is given by:


The volumes of PEEK models are calculated at four different moisture contents and CF models, with temperatures ranging from 200 K to 600 K in increments of 50 K. The simulation is conducted over a time of 500 ps, with results output every 1 ps, and the final 100 ps results are averaged.
Interfacial binding energy calculations
To investigate the interface binding properties, the interfacial bonding energy of CF/PEEK interfaces is calculated under different hygrothermal conditions using the CF/PEEK interfacial model, which indicates the interaction between CF and PEEK matrix of the interface system. The calculation process is conducted in the NVT ensemble. In the presence of moisture, the interfacial binding energy between CF and PEEK is calculated using the following formula: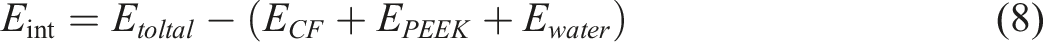
Results and discussion
Detection of damage and failure morphology
Figure 2 shows the damage and failure morphology of the faulty bushings. As seen in Figure 2(a), the damaged areas are located at both ends of the bushings. The microscopic morphology in two directions is observed: the vertical fracture surface (Figure 2(b)) and the fracture surface (Figure 2(c) and (d)). The primary forms of damage and failure include fibre pull-out, fibre breakage, matrix collapse, matrix cracking as well as fibre/matrix interface debonding. Because the secession of the matrix from the fibre surface, there are two phenomena: complete debonding and part of the matrix sticking to the fibre surface. Damage images of bushings (a) full view of the bushing damage, (b) damage micropattern parallel to the fracture cross-section, (c and d) damage micropattern perpendicular to the fracture cross-section.
The components of the interface (fibre and matrix) responded differently when subjected to hygrothermal environments, leading to debonding of the interface and initiating other forms of failure. In order to investigate the damage evolution mechanism of CF/PEEK bushings at the atomic level, interfacial damage evolution simulations are carried out using MD simulations.
Interface damage evolution process
The interface damage evolution during the separation process is analyzed by visualizing the atomic states at four key time points: the initial state (I), the onset of failure (II), the failure propagation stage (III), and the complete failure state (IV). These states correspond to Figure 11 and are visualized using the Open Visualization Tool (OVITO) software (Figure 3). Interfacial damage evolution of CF/PEEK under loads.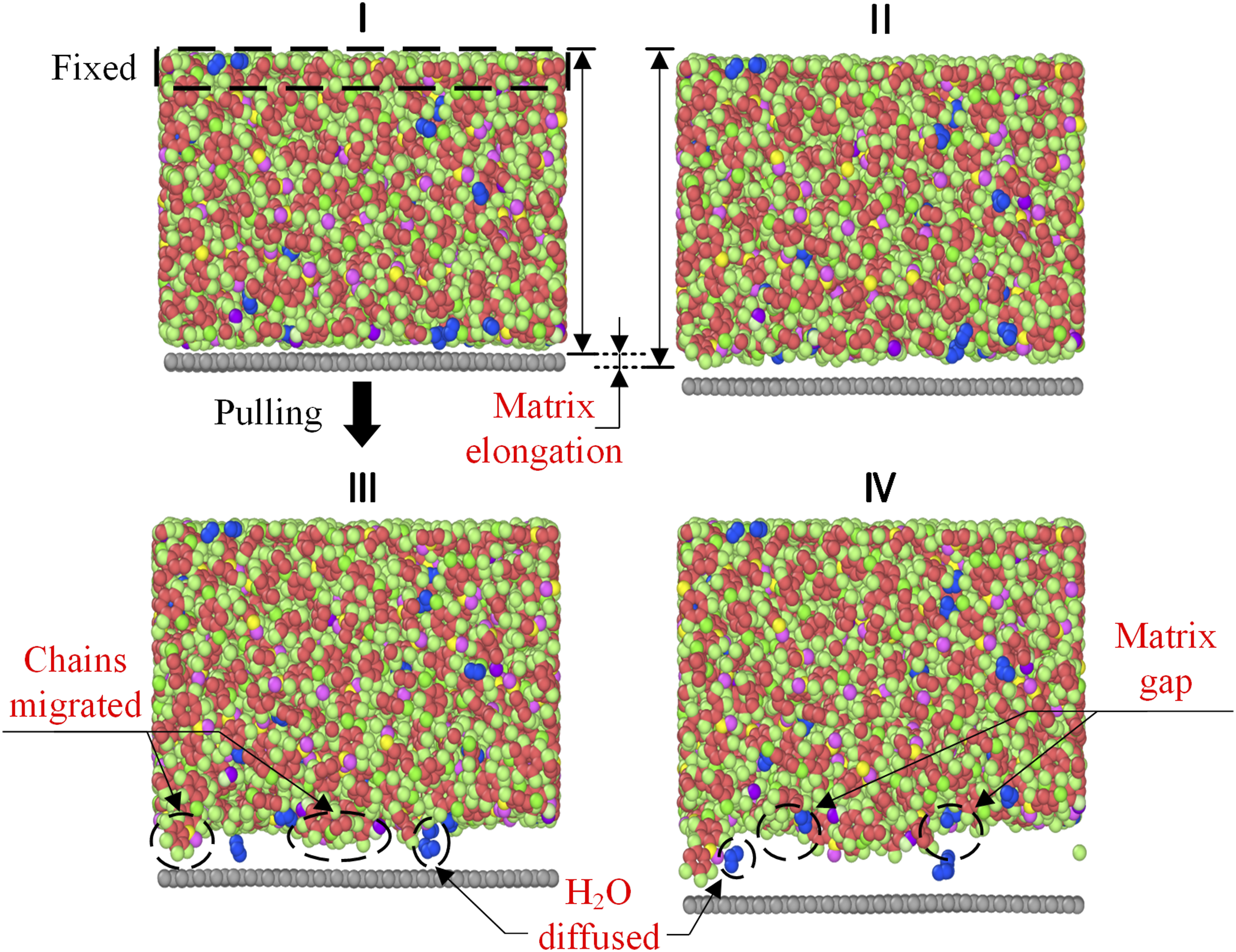
Compared to the initial state (I), because the non-bonded interactions between PEEK and graphene layer, the PEEK molecular chains are stretched as the graphene layer is pulled in state II. At this point, the interface interactions began to decrease, indicating the onset of interface failure. In state III, as the interface gap widened, the PEEK molecular chains dispersed, leading to void formation within the matrix. This phenomenon is macroscopically represented as cracking, and water molecules began to diffuse into the newly formed voids at the interface. This indicates that water molecules incline to migrate into interface voids during the separation process. In state IV, the graphene layer and PEEK matrix completely separated, with the failure of the interface characterized by adhesive failure. This observation aligns with the interface debonding phenomenon of faulty bushings shown in Figure 2(d).
Calculating the number of water molecules along the Z-axis direction (perpendicular to the interface) provides insight into the migration behavior of water during interfacial debonding. As shown in Figure 4, the number of water molecules along the Z-axis direction is compared for the 0.6 wt% interfacial model in the pre-relaxation state, the initial state, and the complete failure state, with 0 Å corresponding to the top position of the interfacial model. From the figure, it can be seen that the water molecules are randomly distributed in the system in the pre-relaxation state, and therefore are more uniformly distributed in the Z-axis direction. After the system has been relaxed under the NPT system, the model size decreases, so the Z-axis size of the initial state is smaller than that of the pre-relaxation state. The higher number of water molecules near 30 Å indicates that the water molecules accumulate at the interface location. The same phenomenon was observed in the simulations of fiber-epoxy and quartz-epoxy interfaces.23,46 Due to the downward movement of the graphene layer along the Z-axis, the Z-axis dimension is larger than that of the initial state in the fully failed state, and the excess is the interfacial void, to which the water molecules migrate with the movement of the graphene layer. Number of water molecules along the Z-axis (perpendicular to the interface).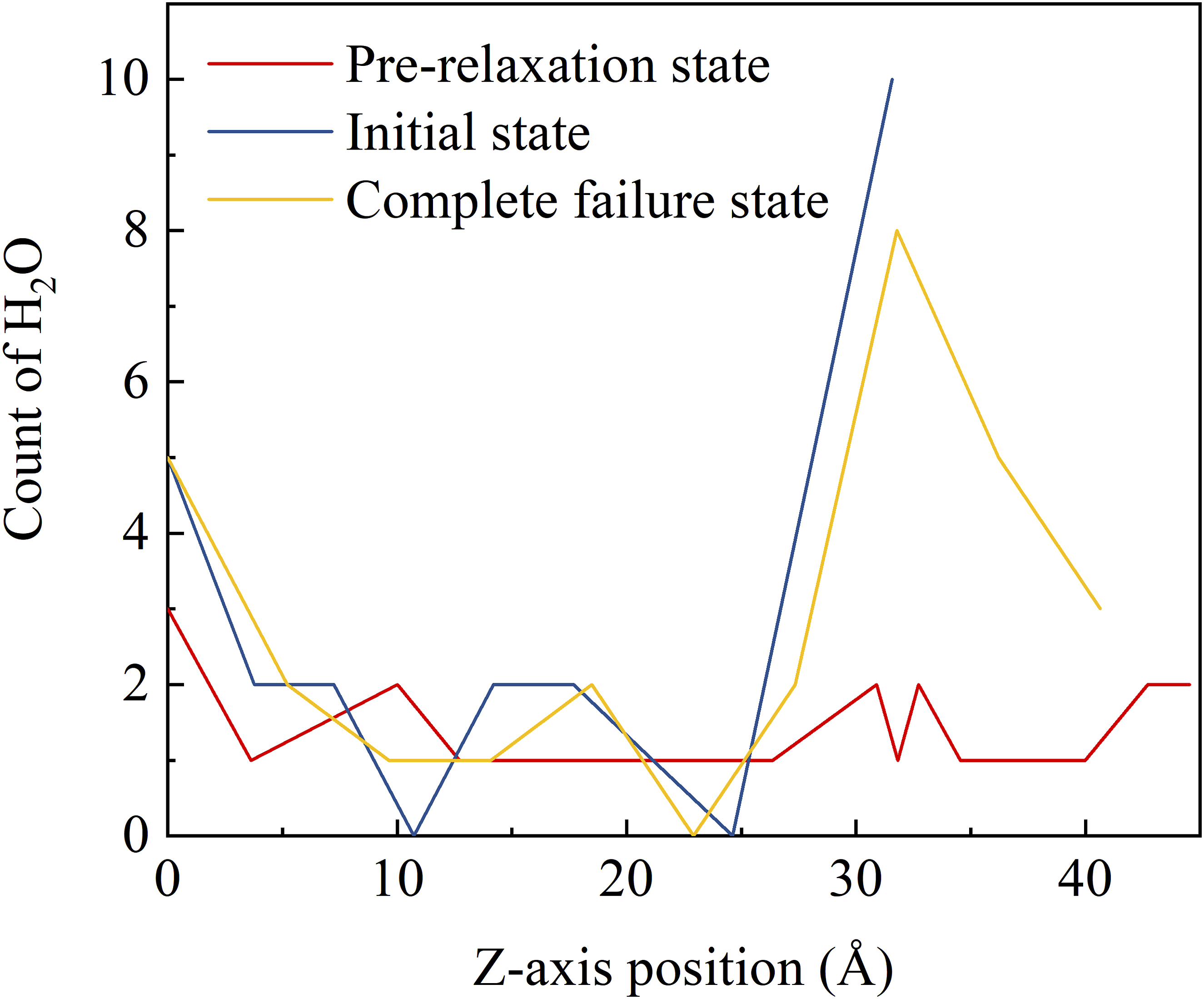
Discussion of interface failure mechanisms
Interaction mechanism between water molecules and PEEK in hygro-thermal environments
(1) Radial Distribution Function Analysis
Figure 5(a) shows the RDF for the electronegative atoms of PEEK (Figure 1(d)) and oxygen atoms of water molecules at 300K under various moisture contents. All curves show the first peak at 0.27 nm, indicating the hydrogen bonds between the electronegative atoms on the PEEK molecular chains and the water molecules are formed. The peak gradually increases with increasing water content, suggesting that as more water enters the PEEK matrix, the greater the number of hydrogen bonds formed between water molecules and PEEK molecules. The water molecules that form hydrogen bonds with the polymer chains are referred to as bound water, while the water molecules that reside in the free volume of polymer without forming hydrogen bonds are known as free water.
47
The RDF between the polar atoms of PEEK and the oxygen atoms of the water molecule (a) influence of moisture effect, (b) influence of thermal effect.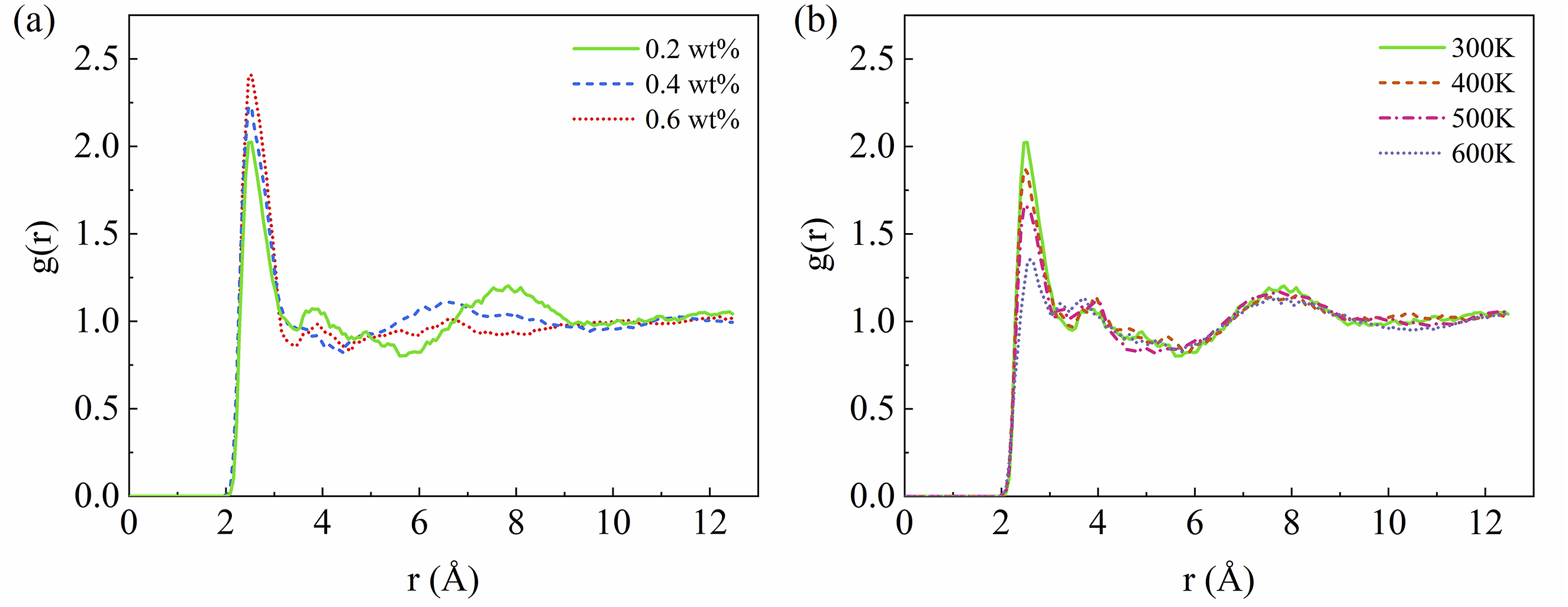
The RDF for the electronegative atoms of PEEK and the oxygen atoms of water molecules at various temperatures under a constant moisture content of 0.2 wt% is shown in Figure 5(b). When the moisture content is constant, the peak decreases with increasing temperature. This occurs because, as the temperature rises, the thermal motion of molecules increases, leading to unstable intermolecular distances. As a result of this fluctuation, the number of hydrogen bonds formed between the polar atoms of PEEK and water molecules is greatly reduced.
The RDF of intermolecular oxygen atoms of water at different contents in the matrix at 300 K is shown in Figure 6(a). The peak appears around 0.32 nm, indicating the presence of hydrogen bonding. Compared to the peak values in Figure 5, the hydrogen bonding interaction between water molecules is stronger than the hydrogen bonding between the water molecules and electronegative atoms of PEEK. As the moisture contents increase, the probability of hydrogen bond formation between water molecules decreases monotonically. Weakened interactions between water molecules can cause water molecules to tend to aggregate at higher water contents, forming larger water clusters and existing in a free state, thereby increasing the amount of free water inside the PEEK matrix.
48
The RDF between the oxygen atoms of the water molecule, (a) influence of moisture effect, (b) influence of thermal effect.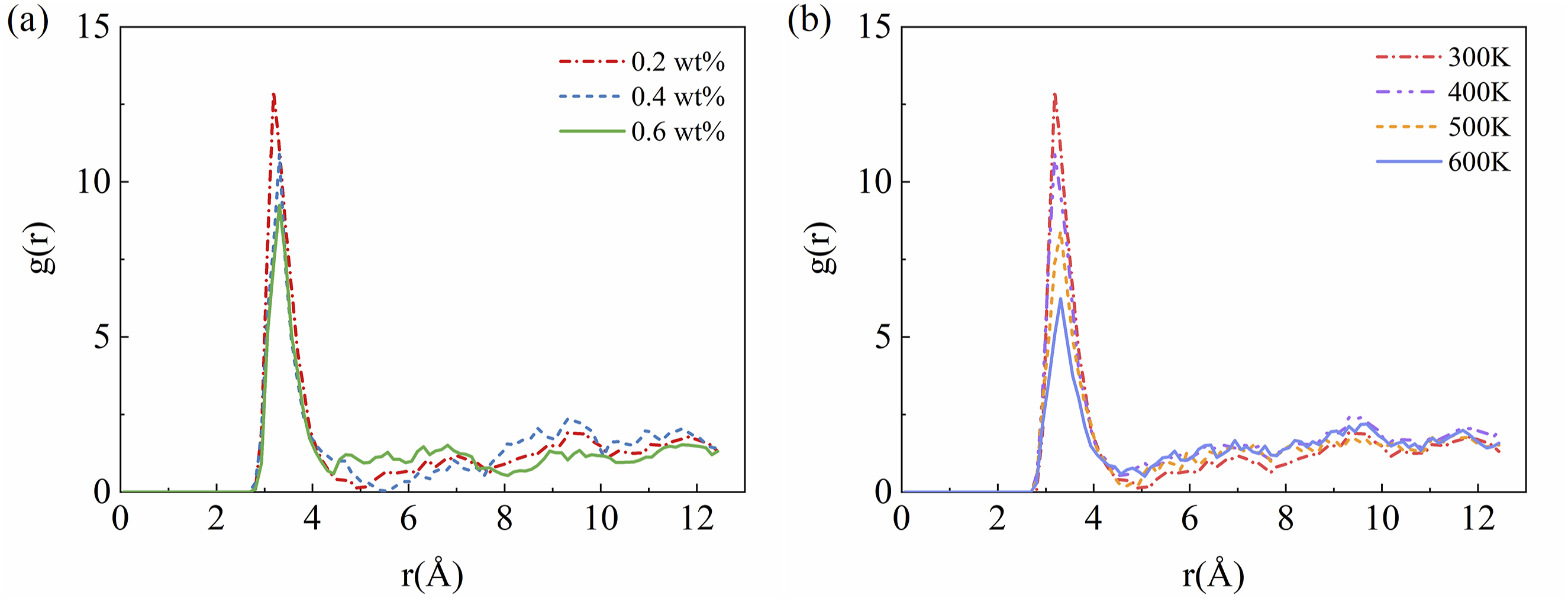
The RDF results for oxygen atoms between water molecules at different temperatures under a constant moisture content of 0.2 wt% are shown in Figure 6(b). The probability of hydrogen bond formation between water molecules decreases with increasing temperature. This is because higher temperatures increase the thermal motion of molecules, reducing the number of hydrogen bonds between water molecules. (2) Mean Square Displacement Analysis
The relationship between MSD of PEEK molecules and moisture contents is shown in Figure 7(a). As time progresses, the MSD value gradually increases and eventually levels off, indicating that the diffusion behavior of PEEK molecules reaches a state of equilibrium after a certain period. By comparing the difference in MSD between different moisture contents at the same temperature, it is found that the higher the moisture contents the larger the MSD in the smoothing region. This suggests that an increased number of water molecules within the PEEK allows for a greater range of motion of the PEEK molecular chains. MSD results for PEEK molecular chains (a) influence of moisture effect, (b) influence of thermal effect.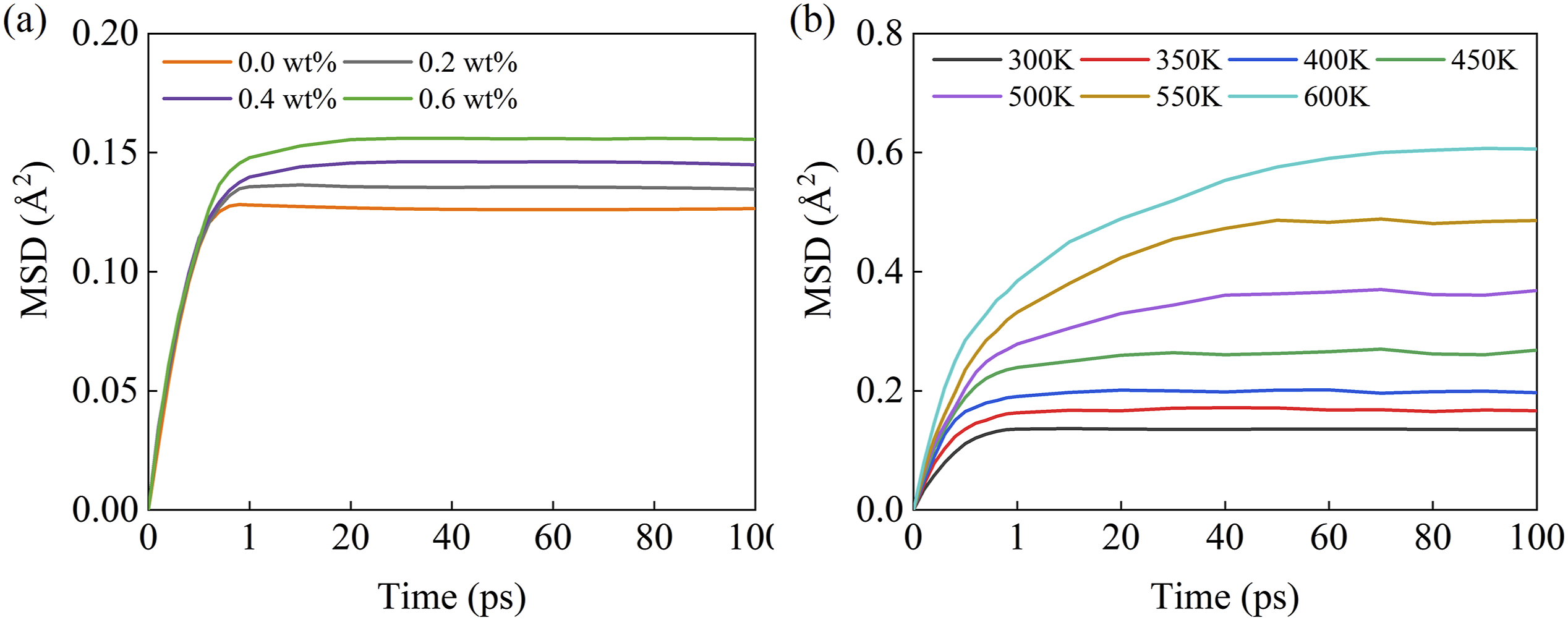
The relationship between MSD of PEEK molecules and temperature is illustrated in Figure 7(b). The MSD of PEEK molecules increases with increasing temperature, indicating more vigorous molecular motion and greater degrees of freedom for the molecular chains at higher temperatures. The figure also shows a change in the spacing of the steady region of MSD between adjacent temperature values around PEEK’s Tg (approximately 427K). This change is associated to the glass transition behavior of polymer, with smaller spacings before Tg corresponding to the rubbery state of PEEK and larger spacings after Tg corresponding to the viscous flow state.
Combining the RDF and MSD analyses, the results showed that with an increase in moisture contents, both bound water and free water within PEEK rise. The presence of bound water diminishes the interactions between PEEK molecular chains, expands the mobility of the chains and induces swelling in PEEK. Although free water does not directly form hydrogen bonds with the molecular chains, it further increases the intermolecular spacing and enhances the mobility of the chains. Additionally, higher temperatures enhance the thermal motion of the molecules. This reduces the number of hydrogen bonds between water molecules and PEEK molecular chains, as well as between the water molecules themselves. As a result, intermolecular forces decrease, leading to increased mobility of the PEEK molecular chains.
Hygrothermal expansion behavior of interface components
(1) Expansion of PEEK
As a thermoplastic polymer, PEEK exhibits a distinct glass transition region, resulting in two different linear relationships for volume change before and after the glass transition, as illustrated in Figure 8(a). By fitting the slopes of the two segments of the curve according to equations (5) and (6), we obtain the CTE. The slope of the first segment is denoted as CTE1, while the slope of the second segment is denoted as CTE2. The relationship between these coefficients and the moisture contents of PEEK is shown in Figure 8(b). CTE1 and CTE2 showed a similar trend: with increasing moisture content, CTE also gradually increased. This indicates that the absorption of water molecules enhances the thermal expansion capabilities of PEEK. The underlying reason is that the ingress of water molecules weakens the interactions within PEEK matrix, thereby increasing the mobility of PEEK molecules and expanding the movement range of the molecular chains. Consequently, this results in an enhanced capacity for expansion deformation, reflecting the effect of hygroscopicity on the thermal behavior of PEEK. Thermal expansion data of PEEK (a) fitted curve of CTE, (b) curve of CTE versus moisture content.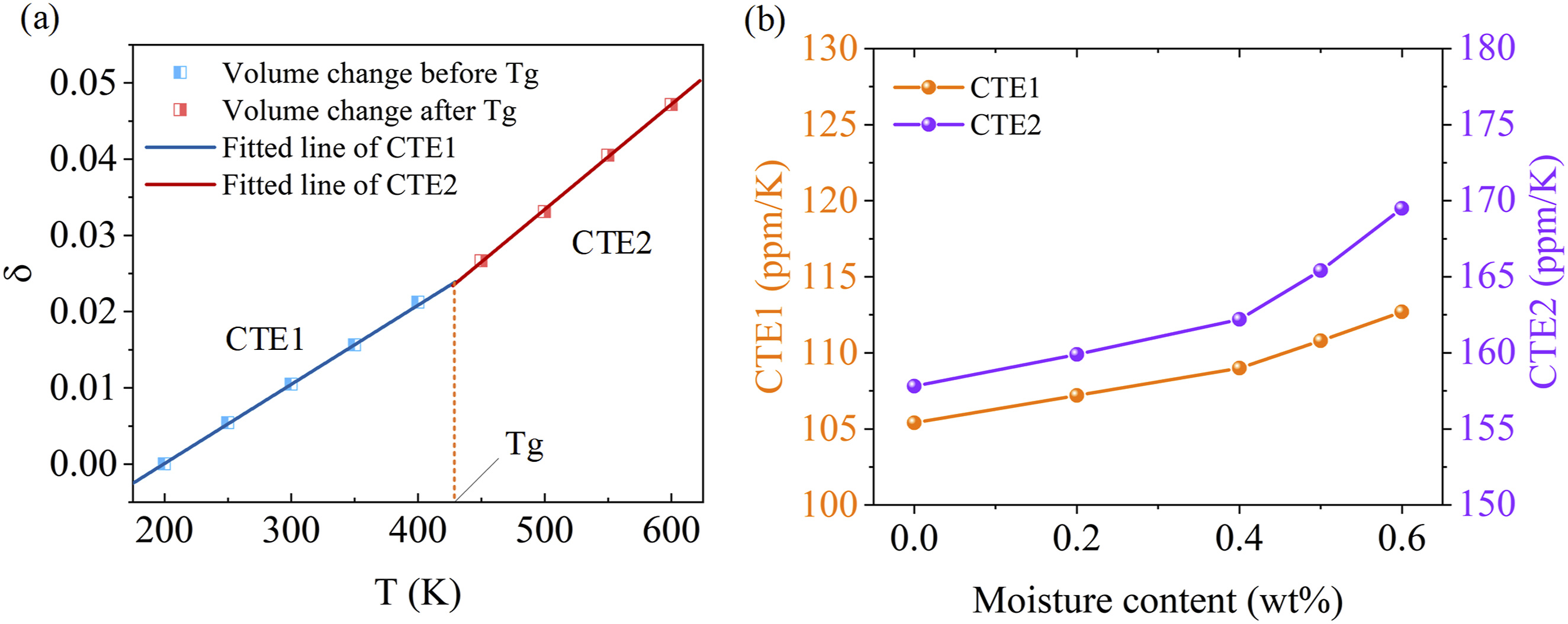
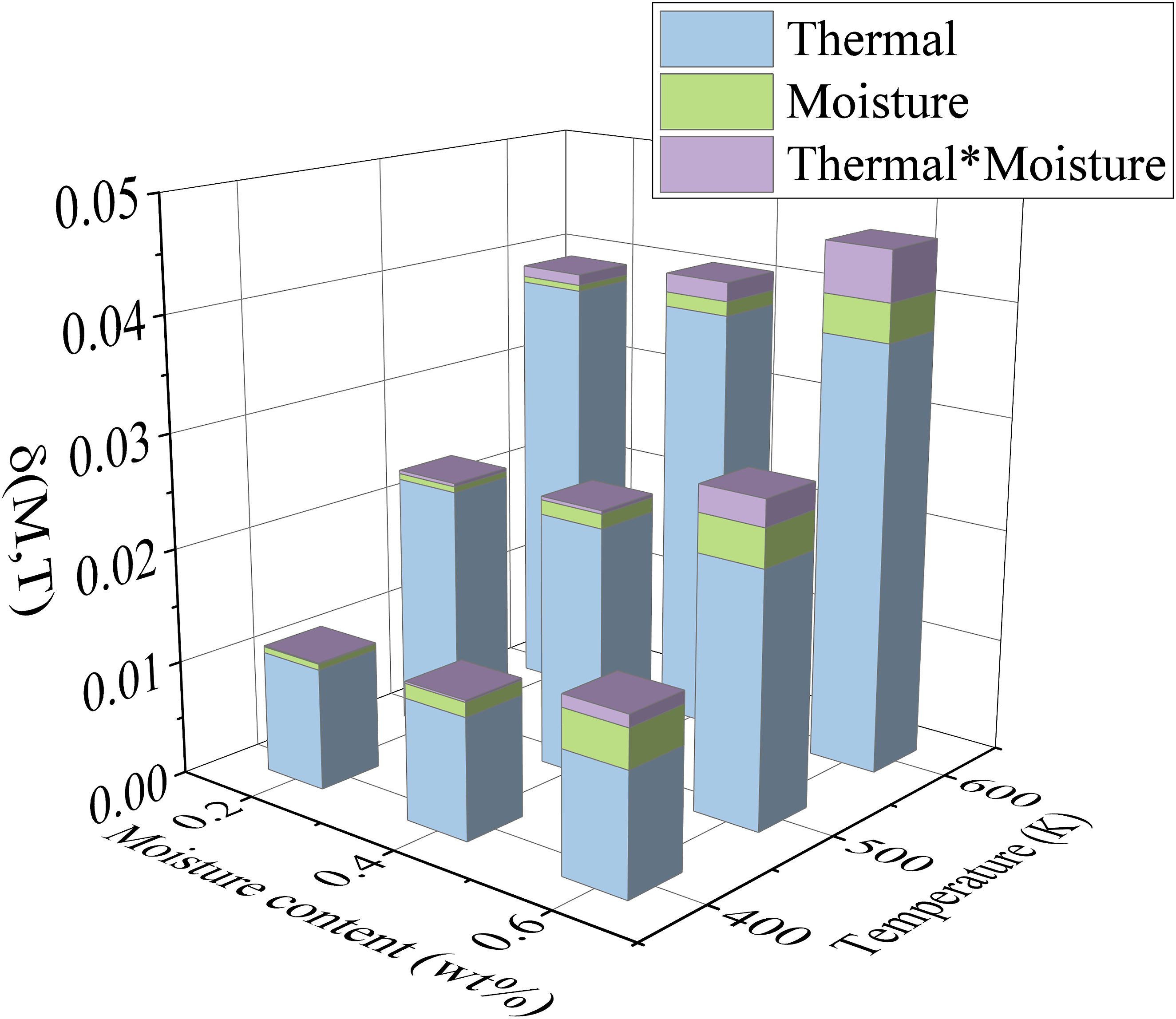
To investigate the specific effects of moisture content and temperature, as well as their interaction, on the expansion of PEEK, we use the volume of the PEEK model at 300K and 0.0 wt% moisture contents as the baseline. Using equations (6) and (7), we calculate the total expansion ( (2) Expansion of CF Degree of influence of moisture and temperature and interaction on PEEK expansion.
In hygrothermal environment, the expansion of CF is usually mainly governed by thermal expansion. The relationship between the volume expansion coefficient of CF and temperature is illustrated in Figure 10, which shows that the volume expansion coefficient of CF gradually increases with rising temperature. By applying equations (5) and (6), we perform a linear fit on the data points presented in the figure; the slope obtained represents the predicted CTE for CF, which is measured at 21.7 ppm/K. In contrast, the CTE of dry PEEK ranges from 105.4 to 157.8 ppm/K, indicating a difference of approximately 1/8 to 1/5. Comparing the experimental values of both CTEs reveals a discrepancy of 1/8 to 1/4,49,50 suggesting that the simulation results align well with the experimental findings. Volume change of CF as a function of tem-perature.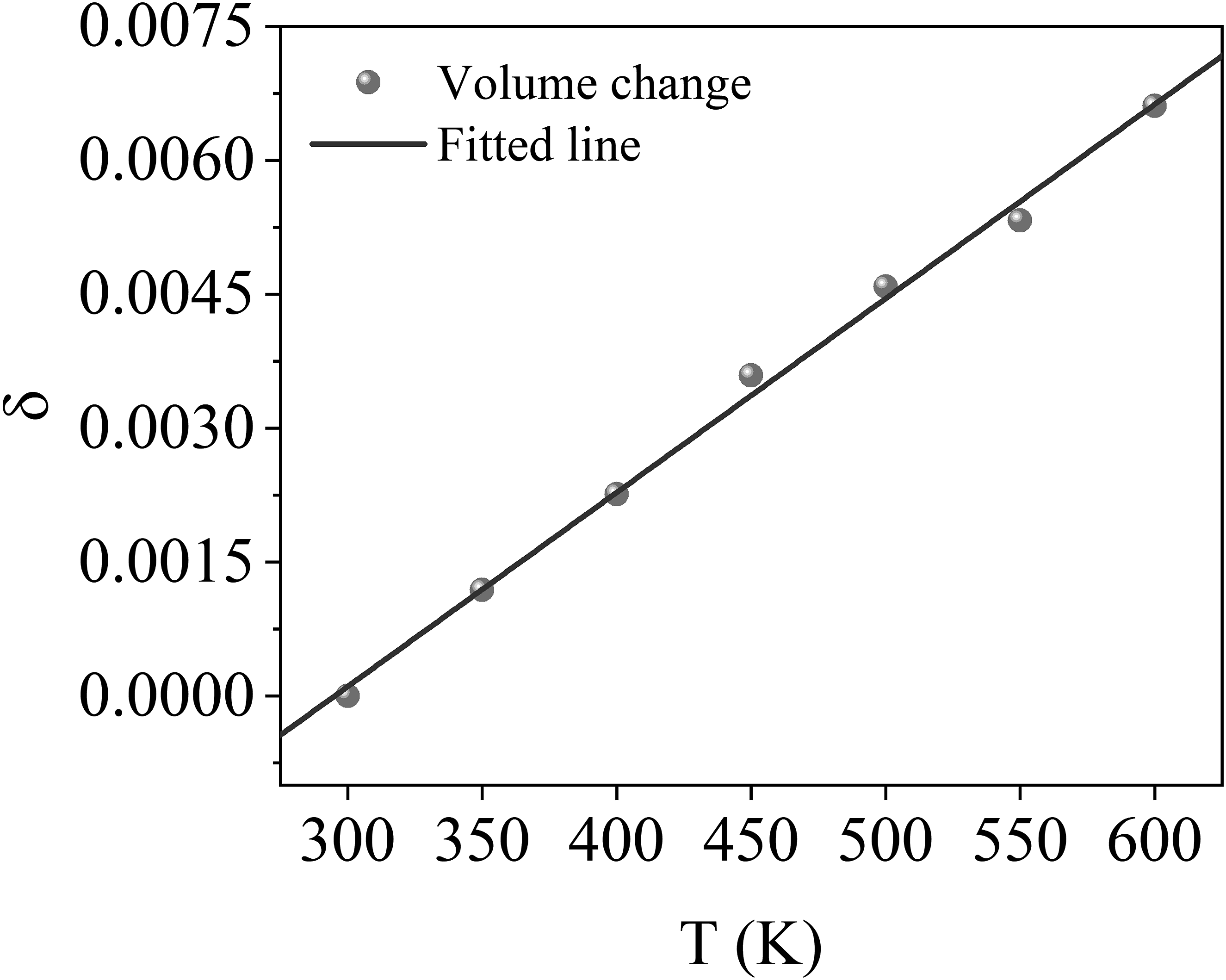
Both the matrix and CF exhibit varying degrees of expansion in a hygrothermal environment, primarily driven by thermal expansion. Calculating their CTE reveals a difference of 5 to 8 times between them. This discrepancy leads to differing expansion amounts in CF/PEEK composite bushings under hygrothermal conditions, resulting in hygrothermal stresses at interface locations. Such stress concentrations can trigger interfacial delamination, ultimately causing matrix cracking. This phenomenon aligns with the faulty bushings shown in Figure 2(c) and (d).
Hygrothermal degradation of CF/PEEK interfacial binding energy
The results of the interfacial binding energy under various hygrothermal conditions are shown in Figure 11(a) From the perspective of moisture effects, as the moisture contents increase, the interfacial binding energy gradually decreases under all four temperature conditions. Notably, when comparing dry and water-containing states, the interfacial binding energy significantly decreases when CF/PEEK contains water, with an average reduction of 25.3%. This phenomenon is more pronounced at higher temperatures. In terms of thermal effects, as the temperature increases, the interfacial binding energy also decreases under the same moisture conditions. For dry states, the binding energy decreases from −1815.32 Kcal/mol at 300K to −1122.51 Kcal/mol at 600K, a reduction of 38.16%. For a 0.6 wt% moisture content, the energy decreases from −1603.31 Kcal/mol at 300K to −244.41 Kcal/mol at 600K, a reduction of 84.76%. This indicates that water molecules amplify the decline in interfacial binding energy caused by temperature increases. Interfacial properties of CF/PEEK (a) interfacial binding energy at different hygrothermal states, (b) COM distances under the influence of moisture effect versus thermal effect.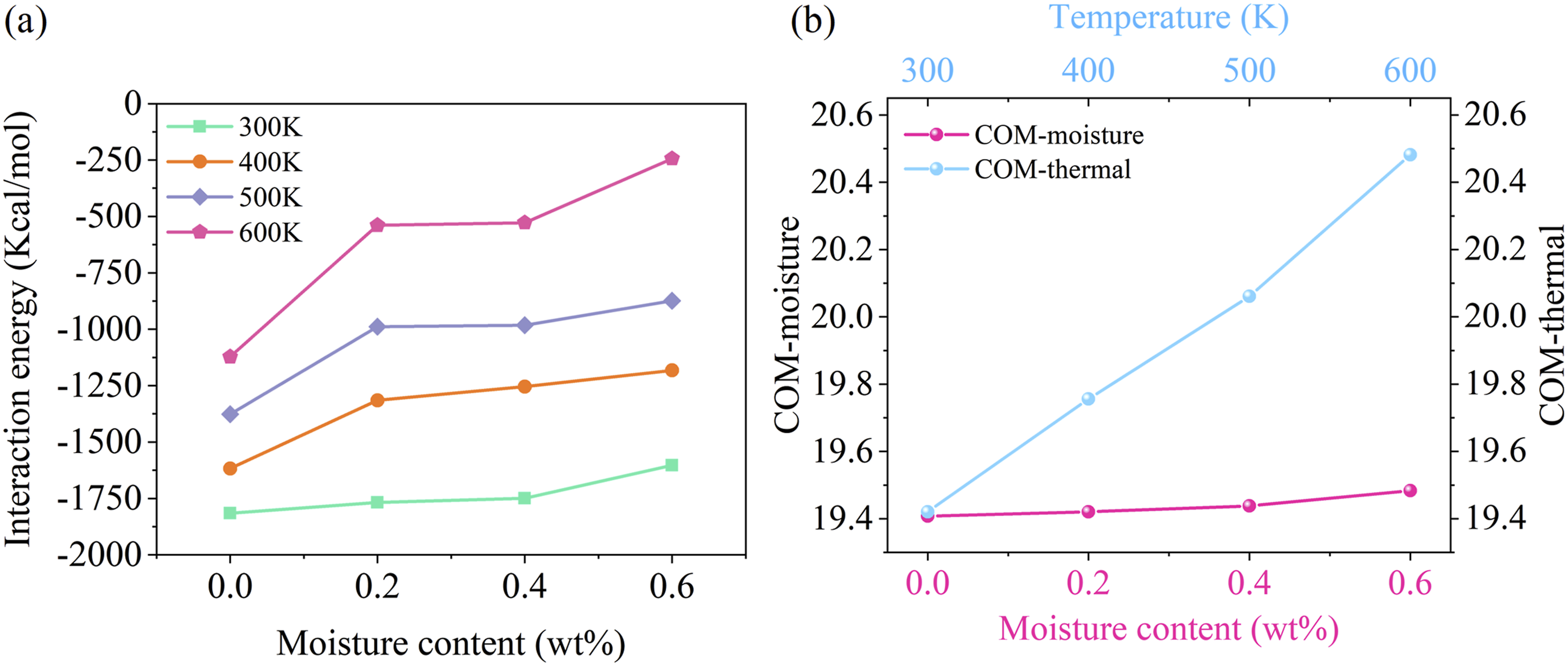
The center of mass (COM) distance of PEEK and the graphene layer under different hygrothermal conditions is calculated. Figure 11(b) shows that as the moisture contents and temperature increase, the COM distance between the PEEK and the graphene layer gradually increases. The magnitude of this increase corresponds to the decrease in interfacial binding energy. The influence of water molecules and temperature causes expansion of the interface components, slightly increasing the distance between them and weakening their interaction. This phenomenon structurally explains the degradation of the interfacial binding energy of the CF/PEEK interface under hygrothermal conditions.
Hygrothermal degradation of mechanical properties of interface and matrix
The mechanical properties of CF/PEEK interfaces significantly and directly influence interfacial failure. Figure 12(a) and (b) show the cohesive force-displacement curves from the interfacial damage evolution simulations. Comparing the curves under different hygrothermal conditions, it can be seen that with increasing moisture content and temperature, the maximum tensile force of the interface gradually decreases. At 300 K, the maximum cohesive forces for moisture contents of 0 wt% and 0.6 wt% are 64.20 nN and 62.45 nN, respectively, representing a difference of 2.73%. For a 0.2 wt% moisture content, the maximum cohesive forces at 300K and 600K are 63.79 nN and 48.89 nN, respectively, a difference of 23.36%. This demonstrates that the mechanical properties of the CF/PEEK interface degrade in hygrothermal conditions. The position corresponding to the maximum cohesive force indicates the point at which interfacial failure begins, this further suggests that the interface is more prone to failure in a hygrothermal environment. Cohesive force-displacement curves during damage evolution at the CF/PEEK interface under loads a influence of moisture effect b influence of thermal effect.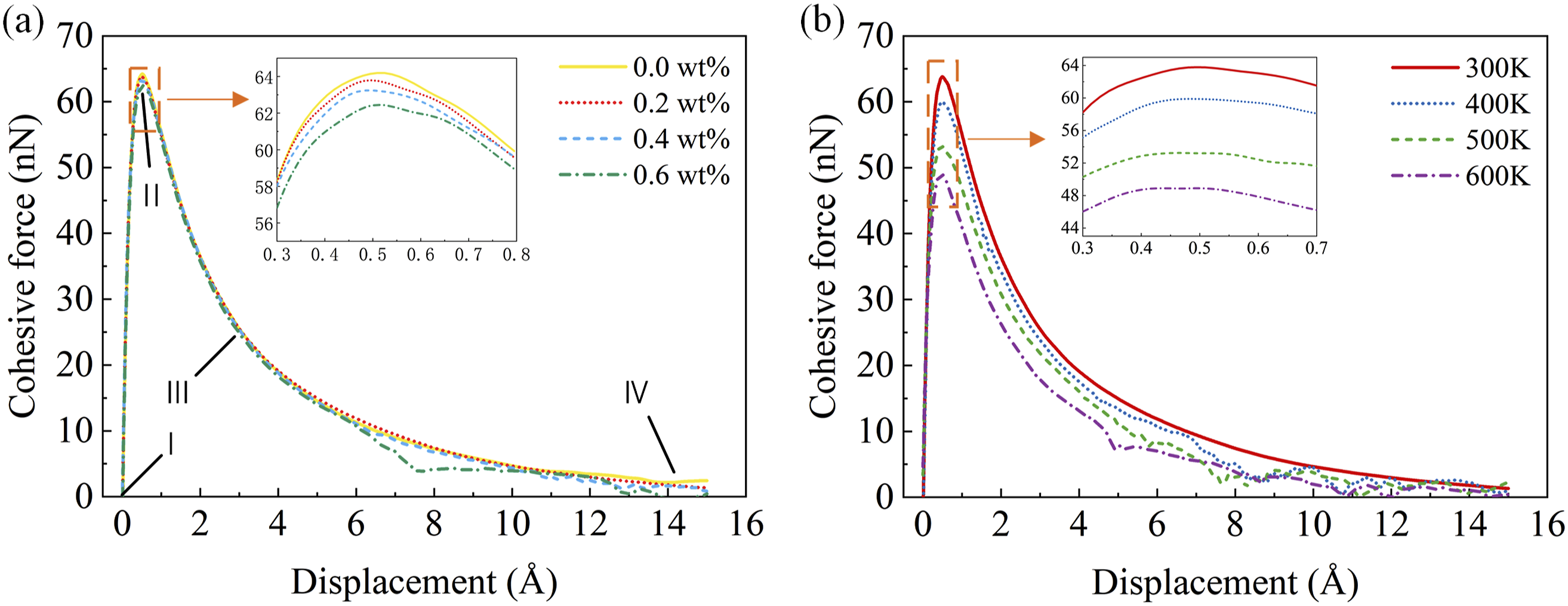
During the evolution of interfacial damage, the matrix is subjected to tensile stress, indicating a strong correlation between CF/PEEK interfacial failure and the degradation of the mechanical properties of the matrix. Uniaxial tensile simulation is performed on PEEK models with different moisture contents at various temperatures. These simulations are conducted in NPT ensemble, applying the constant strain rate of 1.0 × 108 sec−1 along the X-axis while using periodic boundary conditions in all directions. Figure 13(a) shows the tensile stress-strain curves. The tensile modulus of dry PEEK at room temperature, obtained through fitting, is 4.33 GPa, which is close to the experimental result,
51
validating the rationality of the tensile simulation method. Figure 13(b) and (e) shows the simulated values of the tensile modulus of PEEK under various hygrothermal conditions. As the moisture content and temperature increase, the tensile modulus of PEEK gradually decreases. At 300 K with a moisture content of 0.6 wt%, the tensile modulus of PEEK is 3.81 GPa, a decrease of 12.01% compared to dry state at room temperature. For a 0.0 wt% moisture content, the tensile modulus of PEEK at 300K and 600K is 4.11 GPa and 1.65 GPa, respectively, a difference of 59.85%. Although there is a certain degree of error between the simulated values and experimental values, the trend presented indicates that the matrix is more prone to tensile deformation in hygrothermal conditions compared to dry, ambient conditions, which weakens the interfacial bonding performance, further exacerbating interfacial failure. PEEK tensile stress-strain curves a dry state at room temperature, b influence of moisture effect, c influence of thermal effect, d tensile modulus at different moisture content at 300K, e tensile modulus at different temperature at 0.0 wt%.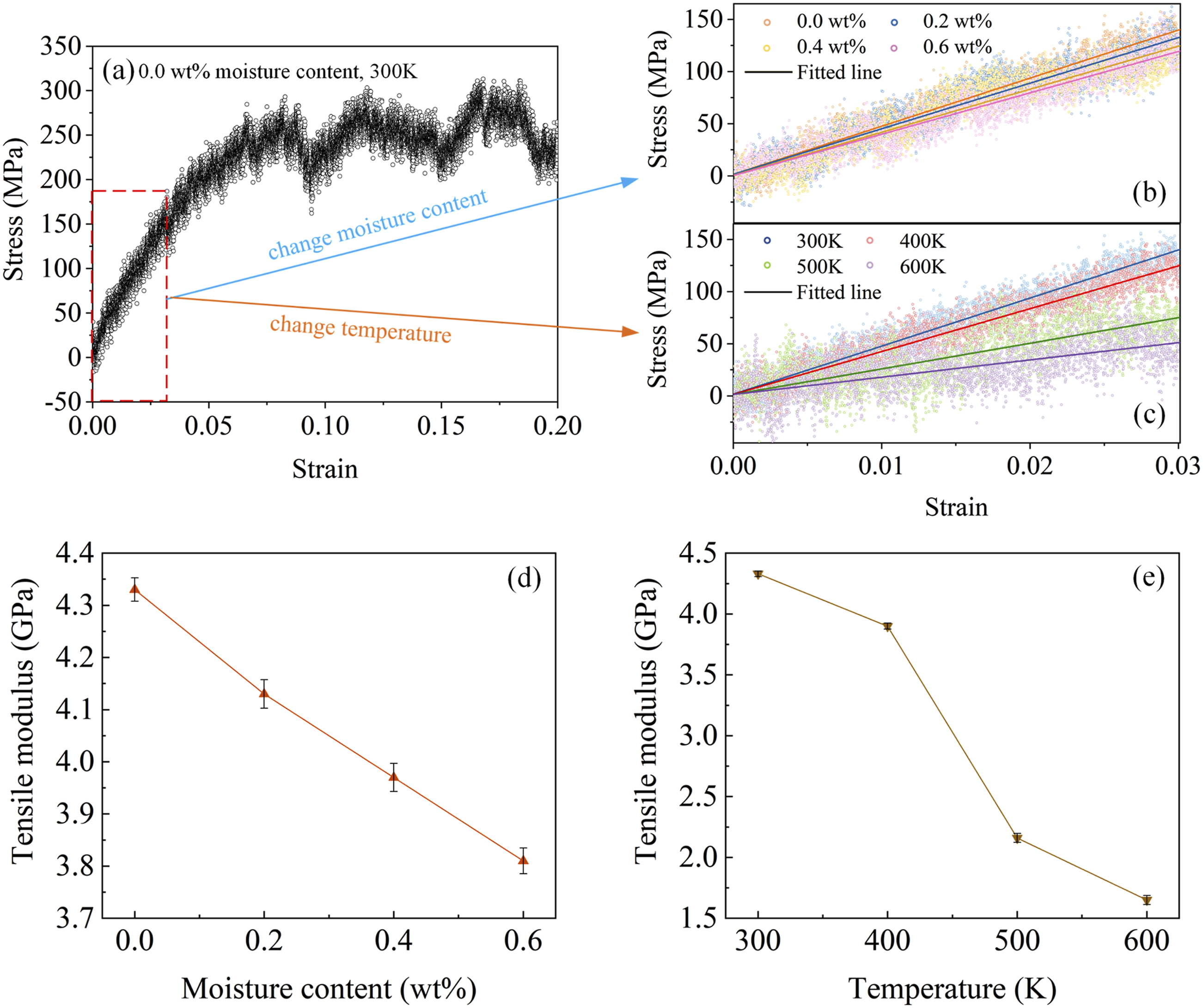
Conclusions
In this paper, experiments and simulations of interfacial damage evolution revealed that CF/PEEK composite bushings in service under the hygrothermal environments led to damage failure mainly in the form of interfacial debonding. Moreover, adhesive failure at the interface often leads to matrix cracking near the interface. By using MD simulations, the mechanism of CF/PEEK interfacial damage failure has been systematically analyzed, the following conclusions are drawn:
RDF and MSD analyses showed that after moisture absorption, both bound water and free water within the PEEK matrix increase with the moisture contents. This leads to an increase in the spacing between PEEK molecular chains, weakening intermolecular interactions and enhancing chain mobility, which subsequently causes PEEK to swell. Additionally, higher temperatures increase molecular thermal motion. This reduces the number of hydrogen bonds both between PEEK molecular chains and water molecules and among the water molecules themselves. As a result, intermolecular forces weaken, accelerating the diffusion of PEEK molecular chains. These findings elucidate the mechanism of interaction between PEEK matrix and water molecules from the perspective of intermolecular interactions.
Based on the understanding of PEEK’s swelling behavior, further analysis of the expansion deformation of PEEK and CF in a hygrothermal environment revealed that the CTE of CF is only 1/8 to 1/5 that of the PEEK matrix. This disparity leads to different expansion rates of CF/PEEK composite bushings under such conditions, resulting in hygrothermal stresses at the interface. These stress concentrations can cause interfacial debonding, often accompanied by matrix cracking. Furthermore, the expansion of both the matrix and CF increases the center-of-mass distance between them, weakening the interfacial bonding energy decreases by an average of 25.3% after moisture absorption. And degrading the mechanical properties at the interface, which is one of the key reasons for interface damage and failure of CF/PEEK composite bushings.
Both moisture and thermal effects also lead to the degradation of the tensile mechanical properties of PEEK at the interface. At 300 K with a moisture content of 0.6 wt%, the tensile modulus of PEEK has a decrease of 12.01% compared to dry state at room temperature. For dry state, the difference between the tensile modulus of PEEK at 300K and 600K is 59.37%. Consequently, the matrix is more prone to failure modes such as collapse and tearing in the hygrothermal environments.
Footnotes
Author contributions
Xiaoli Yue contributed to Writing – Original Draft, Writing – Review & Editing, Supervision, Funding Acquisition. Feiyou Liang contributed to Data Curation, Visualization, Validation, Methodology. Huimin Chen contributed to Data Curation
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key R&D Program of China (No. 2017YFB0309700) and the Foundation of Key Laboratory of Vibration and Control of Aero-Propulsion System, Ministry of Education, Northeastern University under Grant VCAME202104.
Data Availability Statement
Some or all data, models, or code generated or used during the study are available from the corresponding author by request.
