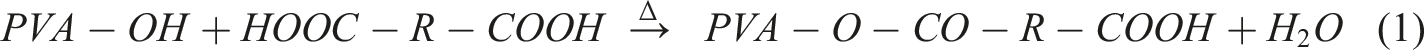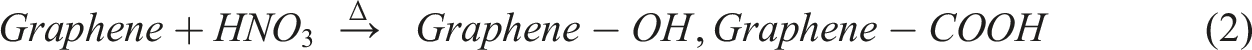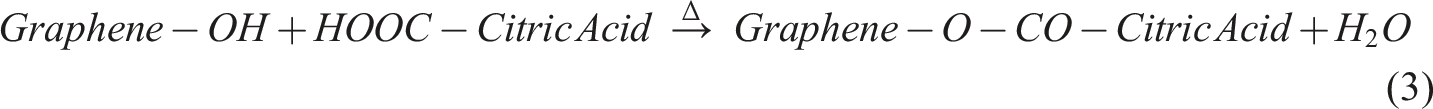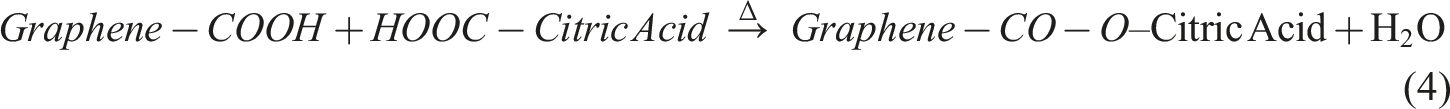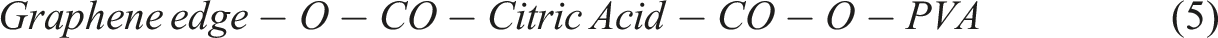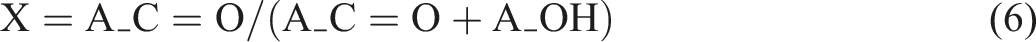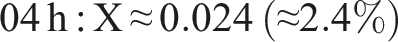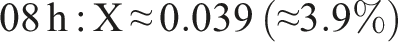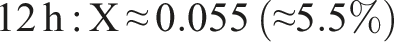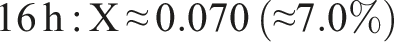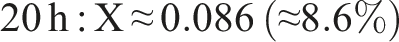Abstract
Graphene is a highly versatile and multifunctional 2-D atomic carbon nanomaterial with impressive mechanical properties, a unique combination of thermal and electrical conductivity, and a high surface area. On the other hand, polyvinyl alcohol (PVA) is a synthetic biodegradable polymer with moderate mechanical properties. The primary objective of this research work is to enhance the moisture absorption, solubility, mechanical, and thermal properties of PVA using a cross-linking agent and to optimize the parameters that influence the cross-linking process. Sustainable cross-linking enhances the solubility issue of neat PVA and reduces its hydrophilicity. To make it suitable as reinforcement with a polymer matrix, functionalization of graphene powder is required to decrease its inertness. The use of functionalized graphene as reinforcement resulted in improved mechanical performance when compared to PVA and cross-linked PVA films, and 1.5 wt% of f-graphene was discovered to be the optimal particle weight percentage, resulting in the highest value of UTS at 103.84 MPa. The strength of the composite increased by 192.7% compared to the UTS of PVA, which was 35.47 MPa. Biodegradability test results showed that the film with 0.5 wt% of f-graphene had the maximum biodegradability of 20.4%, while the film with 1.5 wt% of f-graphene had the minimum biodegradability of 5.3% due to a reduction in crystallinity caused by a dense and cross-linked microstructure. DMA analysis was performed to determine the glass transition temperature of the composite films. TGA analysis demonstrated that the thermal stability of the composite films had been enhanced, and the degradation of the matrix chain was gradual for composite films, unlike PVA.
Introduction
The escalating environmental apprehensions regarding the recyclability and safety of traditional petroleum-derived plastics have instigated substantial initiatives aimed at investigating biodegradable materials as viable alternatives.1–3 The inherent toxicity and non-biodegradability of conventional petroleum-based plastics have catalyzed significant pursuits to identify substitute materials. 4 Biodegradable polymers, which can be sourced from natural constituents such as polysaccharides, proteins, and polyesters, in addition to synthetic origins including polyamides, polyanhydrides, polyamide-enamines, and polyvinyl alcohol (PVA), have garnered notable attention as promising resolutions to these challenges.5,6 Biodegradable polymers are categorized into those derived from natural sources, such as polysaccharides, proteins, and polyesters, alongside synthetic sources, comprising polyamides, polyanhydrides, polyamide-enamines, and polyvinyl alcohol.7–10 Among these materials, PVA is distinguished as a non-toxic, biodegradable thermoplastic that, upon interaction with microorganisms, degrades into innocuous by-products such as water and carbon dioxide, which are subsequently assimilated by flora. PVA is utilized across diverse domains, including medicine, electronics, food processing, civil engineering, and drug delivery systems. 5
Polyvinyl alcohol (PVA) demonstrates utility across a multitude of domains, including but not limited to medicine, 11 electronics, 12 the paper industry, the food industry, civil engineering, and drug delivery systems. For instance, research has indicated that the cross-linking of PVA with agents such as citric acid can confer pH-responsiveness and antibacterial properties, in addition to enhancing thermal stability and minimizing swelling in comparison to unmodified PVA. Nonetheless, the direct synthesis of PVA [CH2CH(OH)]n from the vinyl alcohol monomer is not a straightforward endeavor. Rather, the production of PVA entails a biphasic process. The initial phase involves the synthesis of vinyl esters, 13 which is succeeded by a second phase of hydrolyzing the vinyl ester to yield PVA with the requisite functional group. Historically, the seminal work conducted by Herman and Haehnel in 1924 culminated in the inaugural successful synthesis of polyvinyl alcohol (PVA) from polyvinyl esters. Their methodology entailed the incorporation of alkali into an alcoholic solution of polyvinyl acetate, which serves as a precursor for PVA. 14 In 1931, Herman innovated the production of polyvinyl alcohol fibers utilizing both wet and dry spinning techniques, which are applicable as sutures for surgical interventions. Subsequently, composite films of CNF-reinforced polyvinyl alcohol (PVA) have exhibited marked enhancements in mechanical and thermal properties, albeit with a slight reduction in optical transparency. 15 Solid polymer blends comprising polyvinylpyrrolidone/polyvinyl alcohol and lithium perchlorate salt exhibit a hardness of 1.3 GPa and a tensile strength of 4.3 MPa. 16
The hydrophilic characteristics of polyvinyl alcohol (PVA) facilitate robust interfacial adhesion17–19 with reinforcing agents such as fibers, particles, or flakes. This particular attribute renders it an exemplary candidate for the fabrication of composites, culminating in materials that exhibit enhanced mechanical strength and toughness.20,21 Nevertheless, a prominent limitation of PVA is its complete solubility in aqueous environments, which is due to the presence of hydroxyl (OH-) groups. Therefore, the examination of solubility and moisture absorption emerges as critical determinants that restrict the practical applications of PVA. In an effort to augment the mechanical properties and percentage elongation of chitosan-PVA blend films, researchers have investigated various cross-linking methodologies. Jahan et al. 22 conducted an analysis on the cross-linking of chitosan-PVA blend films utilizing KNO3. Their findings revealed that the CS-PVA (50/50) blend film exhibited an extraordinary enhancement of 298% in tensile strength, alongside an increase in percentage elongation from 7% to 67%. In a separate study, Sabzi et al. (2020) employed citric acid as a cross-linking agent, which endowed the poly (vinyl alcohol) (PVA)/Ag nanoparticles (NPs) composite with pH-responsiveness and antibacterial characteristics. 23 PVA membranes developed with citric acid and succinic acid as cross-linking agents demonstrated reduced swelling compared to pure PVA, and the transition between the initial and subsequent phases of thermal degradation appeared more gradual, signifying improved thermal stability. 24
Graphene is constituted by a monolayer of carbon atoms and exhibits remarkable strength, being approximately 100 times more robust than the most resilient steel of equivalent thickness. 25 Furthermore, graphene is characterized by outstanding electrical conductivity, elevated tensile strength, and transparency, 26 thus rendering it an invaluable nanomaterial. The rigorous sonication of graphene oxide particles prior to their integration into poly (vinyl alcohol) facilitates a uniform dispersion and enhances the thermal and mechanical properties as well as the interfacial interactions. 27 The fabrication of nanocomposite scaffolds utilizing polyurethane (PU) and polycaprolactone (PCL) in conjunction with graphene oxide (GO) has been shown to augment their wettability and biocompatibility. 28 The reinforcement of polylactic acid (PLA) nanocomposites with graphene nanoplatelets (GNP) has been demonstrated to elevate the impact strength while also enhancing thermal stability and viscoelastic properties. 29
The incorporation of functionalized graphene (f-graphene) as a reinforcement agent has emerged as a promising strategy to further augment the performance characteristics of PVA composites. Graphene, consisting of a singular layer of carbon atoms, is not only extraordinarily strong but also possesses remarkable electrical conductivity, high tensile strength, and transparency. 30 Functionalized graphene, specifically carboxyl-functionalized graphene (CG), has been evidenced to significantly improve the thermal properties of polymer nanocomposites by elevating the melting and thermal decomposition temperatures, as illustrated in aromatic polyamide nanocomposites. 31 Likewise, the integration of graphene oxide (GO) into PVA matrices has been reported to enhance the physicochemical and rheological properties, indicating that graphene-based fillers can effectively bolster the mechanical and thermal stability of PVA films. 5 The employment of graphene within polymer matrices, including PVA, is substantiated by research demonstrating that even minimal quantities of graphene can induce substantial enhancements in thermal stability and mechanical strength owing to the robust interfacial interactions between the graphene and the polymer matrix.30,32 Moreover, the presence of functionalized graphene sheets within polyimide nanocomposites has been found to improve thermal expansion coefficients and gas barrier properties, further underscoring the potential of graphene to augment the thermal attributes of polymer composites. 33 The incorporation of carbon-based nanofillers, such as graphene, into polymer composites represents a promising methodology for addressing the inherent limitations associated with polymers like PVA, which are recognized for their flammability and restricted thermal stability.34,35 In summation, the deliberate integration of functionalized graphene into PVA films can culminate in enhanced mechanical and thermal properties, thus rendering these materials applicable for a broader spectrum of industrial uses.
The primary aim of this investigation is to evaluate and analyze various properties of PVA that has been cross-linked using citric acid and reinforced with functionalized graphene. Given the prospective applications of these blended films, it is imperative to acquire thorough insights into their biodegradability alongside their mechanical and thermal characteristics.
The thermal properties, particularly thermal stability, have been assessed utilizing thermogravimetric analysis (TGA) and dynamic mechanical analysis (DMA), which serve to delineate the operational temperature range and the glass transition temperature of the films. Mechanical properties were ascertained employing a universal testing machine to evaluate ultimate tensile strength, percentage elongation, and Young’s modulus.
Materials
The matrix material utilized for the composite film was polyvinyl alcohol (PVA), characterized by a cold molecular weight that varies from 850,000 to 124,000 and a degree of hydrolysis quantified at 99%. Graphene nanoplatelets procured from Reinste Nano Ventures Pvt Ltd India served as the reinforcement material. The PVA provided by HPLC, India, exhibited a purity exceeding 99% and a viscosity within the range of 23-38 cP at a concentration of 4% and a temperature of 20°C. Citric acid, possessing a molar mass of 192.12 g/mol, was employed as a cross-linking agent, containing three carboxylic groups (-COOR) and one hydroxyl group (-OH). The citric acid was sourced from the Molychem research laboratory located in Mumbai, India. For the functionalization of graphene, a nitric acid solution (HNO3) obtained from Rankem India was utilized. Additionally, distilled water and silicone gel were incorporated for the preparation of solutions and as a lubricant, respectively. The following instruments were employed in the current study for casting, analysis, and testing purposes: Magnetic stirrer, vacuum oven, centrifuge machine, oven, FT-IR spectroscopy machine, universal tensile machine, scanning electron microscope, dynamic mechanical analysis machine, and thermo gravimetric analysis machine.
Methodology
Cross-linking reaction
To create a PVA/graphene composite using citric acid as a cross-linking agent, the process involves several key stages. Initially, graphene powder is dispersed in water or ethanol through ultrasonication to achieve even distribution, followed by its incorporation into a heated PVA solution, where π–π stacking and hydrogen bonding occur between PVA chains and graphene sheets. Next, citric acid is introduced to the mixture, where its –COOH groups engage in initial hydrogen bonding with PVA –OH groups. The critical stage follows with thermal cross-linking at temperatures between 140 and 180°C, leading to esterification reactions that form cross-links between PVA chains through citric acid bridges, while also enabling potential covalent bonding between citric acid and any defected or oxidized edges of the graphene. This results in a robust composite structure characterized by both physical interactions and chemical bonding, enhancing the properties of the final material. Step 1: PVA Esterification with Citric Acid Step 2: Graphene Functionlisation Step 3: Graphene reaction with Citric Acid Step 4: Final Composition
This results in a thermally cross-linked PVA–Graphene composite, where citric acid acts as the molecular bridge, forming a 3D network. Subsequent sections provide detailed explanation of FTIR spectroscopy to identify the key functional groups once reaction is completed.
Optimisation
This segment delineates the methodology employed to enhance the refluxing time within the functionalisation procedure of graphene via nitric acid. Furthermore, it discusses the refinement of cross-linking parameters, which encompass the concentration of citric acid utilized as the cross-linking agent, the duration of stirring, the temperature during stirring, the temperature of the oven, and the duration of drying.
Optimisation of refluxing time
Figure 1 shows the flow chart for the functionalisation process of graphene using oxidative acidic treatment including sample extraction and centrifugation illustrated in Figure 2 and 3. Flow chart for the functionalisation process of graphene.
Subsequent to the functionalisation procedure, numerous graphene specimens were subjected to evaluation in order to determine the extent of functionalisation achieved. Fourier Transform Infrared (FT-IR) spectroscopy was utilized to examine the attachment of functional groups to the graphene surface. Setup for oxidative acidic treatment. Centrifugation process: (a) Loading of the samples and (b) Centrifuging at 3000 rpm.

Optimising cross-linking parameters
The cross-linked polyvinyl alcohol (PVA) films were synthesized via the solution casting methodology. Figure 4 delineates the procedural flowchart that elucidates the preparation of composite films employing the solution casting approach with Figure 5 illustrating the solution mixing step. Flow diagram for preparation of cross-linked films.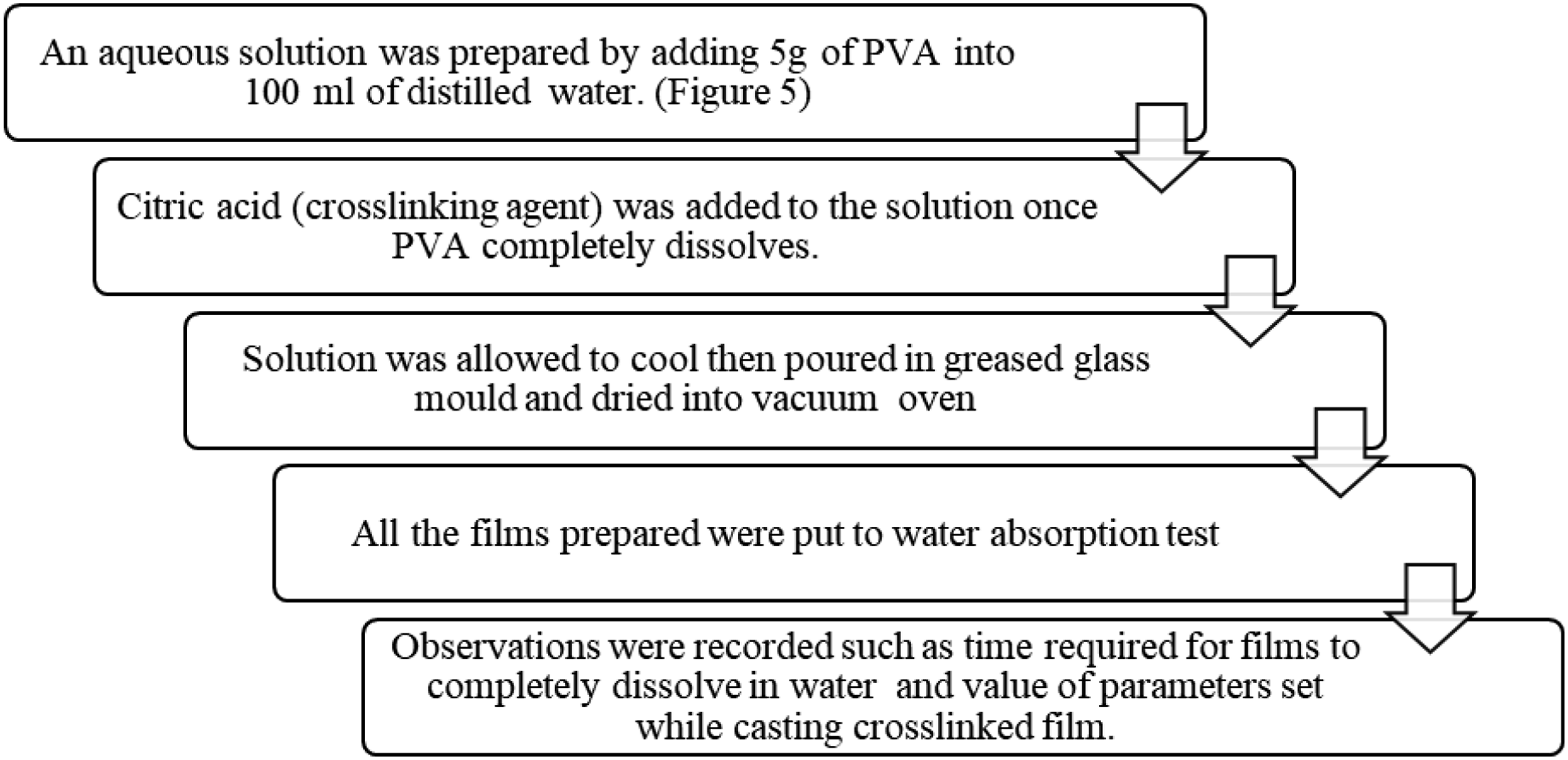
Utilizing multiple regression analysis software, the outcomes were employed to ascertain a correlation between the dependent variable (water dissolving time) and independent variables such as citric acid concentration, duration of oven exposure, oven temperature, duration of stirring, and stirring temperature. The formula derived from the analysis identified the most critical variable influencing the stability of the cross-linked film, and optimization was executed incrementally, commencing with the most impactful variable and adjusting the values of the other parameters to achieve the most stable cross-linked PVA film. Cross-linking of PVA using citric acid.
Composite film casting
The procedure initiates with the formulation of an aqueous solution by solubilizing 5 g of polyvinyl alcohol (PVA) in 100 mL of deionized water through sustained agitation utilizing a magnetic stirrer. Upon complete dissolution, the optimized concentration of citric acid, which functions as a cross-linking agent, along with other parameters adjusted to their optimal conditions, is incorporated into the solution. Various proportions of functionalized graphene (f-graphene) are subsequently introduced, and the resulting black solution is agitated to guarantee the homogenous distribution of reinforcement particles and the effective cross-linking of PVA. The resultant composite is then transferred into a glass mold and subjected to drying in a vacuum oven. The dried films are subsequently removed from the mold.
Testing
This section elucidates the methodology employed to evaluate the produced composite films through an array of testing protocols (including tensile testing, scanning electron microscopy, thermogravimetric analysis, dynamic mechanical analysis, and assessments of biodegradability).
Mechanical testing
The mechanical characteristics, encompassing ultimate tensile strength, Young’s modulus, and elongation percentage, were systematically analyzed for polyvinyl alcohol (PVA)-based films augmented with varying weight percentages of functionalized graphene (0.5%, 1.0%, 1.25%, 1.5%, 1.75%, 2.0%, 2.5%, 3.0%, 3.5%, 5.0%). The evaluations were executed utilizing a universal testing apparatus (AMT-SC-01521) in accordance with the ASTM D882-12 protocol. Specimens were fabricated with dimensions of 10 mm in width and 60 mm in length, exhibiting an average film thickness of less than 1 mm across all variations. The gauge length of the samples was established at 40 mm, and the assessments were conducted at a uniform rate of 1 mm/min for every specimen.
Scanning electron microscopy
SEM imaging represents a sophisticated high-resolution methodology that investigates the surface morphology of a specimen through the utilization of an electron beam. Comprehensive images are generated by directing focused electrons onto the material’s surface and subsequently collecting the electrons that are scattered. The JSM-6610LV SEM apparatus was utilized to examine the fractured surface of composite films, thereby yielding critical insights regarding interfacial bonding. Prior to imaging, samples underwent a dehydration process at temperatures ranging from 30 to 40°C and were coated with gold to mitigate charging complications. The samples were subsequently affixed to a stage using carbon tape and exposed to an electron beam operating at 5 kV, facilitating the capture of images across various magnification scales.
Biodegradibility test
In order to assess the biodegradability of composite films reinforced with varying weight percentages of functionalized graphene, an experimental burial test was executed. Specimens with dimensions of 1 × 2 mm2, incorporating different weight percentages of f-graphene, were strategically placed within the soil, and their initial weights were meticulously recorded. The samples were buried at a depth of approximately 15 cm in a container filled with soil, which was distinctly marked with thin dowel pins to facilitate their subsequent retrieval. Following a duration of 2 months, the samples were cautiously extracted, and their final weights were documented. The percentage of weight loss was computed by analyzing the initial and final weights of the samples in comparison.
Thermo-gravimetric analysis
The STA7300 HITACHI TGA analyzer was employed for the thermo-gravimetric analysis of composite films. Initially, the samples were meticulously wiped with tissue paper to eradicate any moisture or oil residues adhering to the film’s surface. Subsequently, the samples were sectioned into smaller fragments to facilitate enhanced analysis. These diminutive pieces were then positioned within a small crucible, which was suspended on a highly sensitive balance. The crucible was enveloped within an inert nitrogen gas atmosphere flowing at a rate of 150 mL/min. The temperature of the sample was incrementally elevated at a constant rate of 10°C/min, commencing from 30°C and culminating at 350°C. Throughout the entirety of the procedure, the balance persistently recorded the weight of the sample.
Dynamic mechanical analysis
The 7100 HITACHI Dynamic Mechanical Analyser was employed to conduct a comprehensive dynamic mechanical analysis of composite films. Specimens with dimensions of 40 mm × 10 mm were subjected to tensile testing across a spectrum of frequencies while maintaining a 5 µm amplitude. A sinusoidal stress was exerted, and the resultant strain and modulus were meticulously recorded. Throughout the analysis, the frequency of stress or temperature was systematically modified, resulting in alterations in the modulus observed. The temperature was incrementally adjusted at a rate of 2°C/min within the specified range of 25°C to 210°C. The DMA graphs generated are utilized to ascertain the storage modulus, loss modulus, and the loss tangent (tan δ). The glass transition temperature (Tg) for various composite films is derived from the tan δ curves by pinpointing the temperature at which the peak of the curve occurs.
Results and discussion
Optimisation of refluxing time
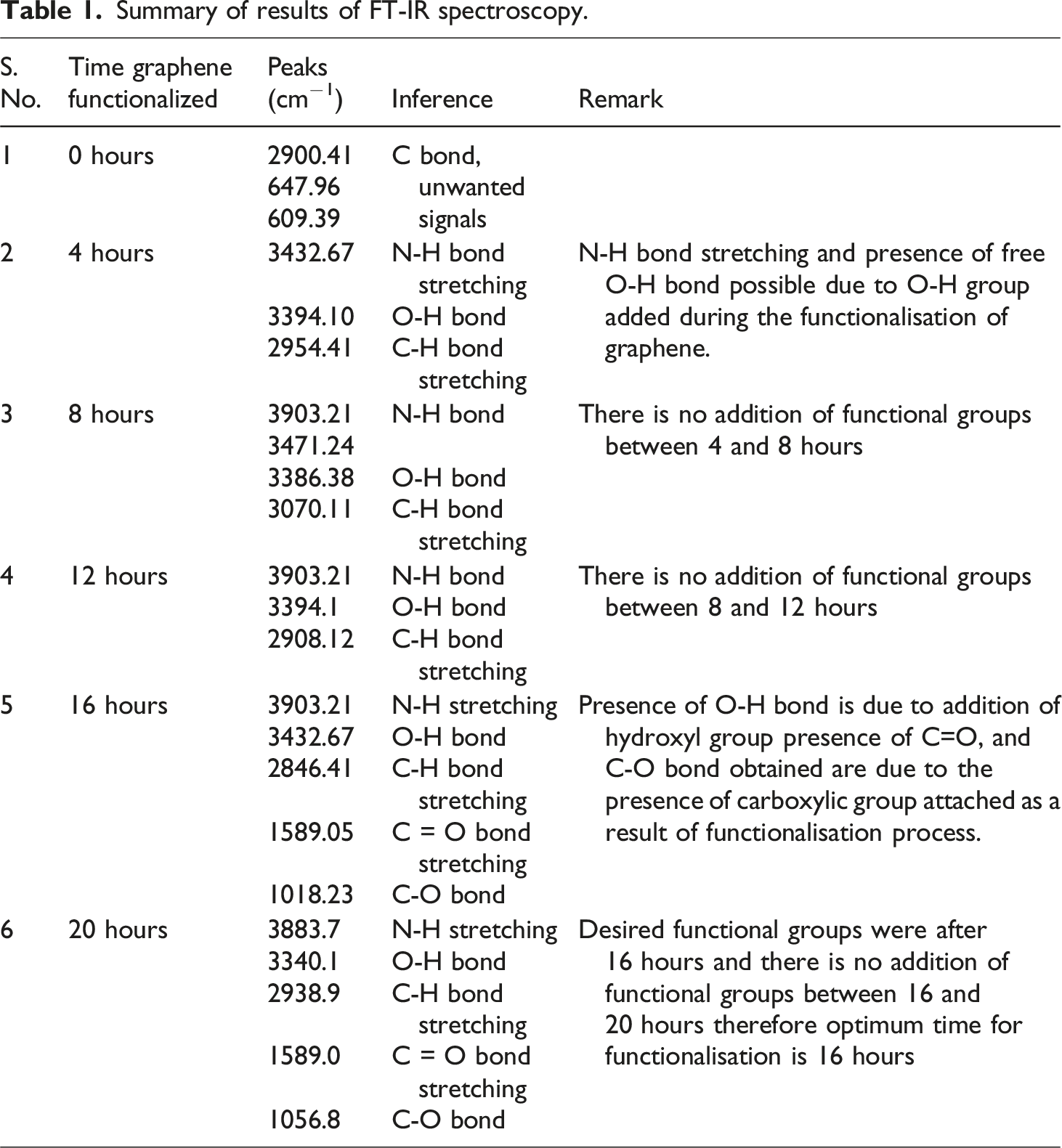
The FTIR spectra of the PVA/citric-acid/graphene composite (observed at 4, 8, 16, and 20 hours) exhibit the anticipated spectral bands corresponding to PVA and its crosslinking structures. In the unmodified PVA, one can observe a broad O–H stretching band within the ∼3200–3600 cm−1 range, attributable to the intermolecular hydrogen bonding associated with –OH groups. Distinct C–H stretching peaks manifest in proximity to 2930 and 2850 cm−1. An initial minor carbonyl band appears at approximately ∼1735 cm−1, which can be ascribed to the residual vinyl acetate present in PVA that has undergone over 99% hydrolysis. Subsequent to the cross-linking process involving citric acid, novel ester peaks become apparent: a pronounced C=O stretching band is detected around ∼1730 cm−1, alongside an ester C–O stretching band at ∼1210 cm−1. These spectral features signify the occurrence of citric-acid-mediated esterification (specifically, di-ester formation) involving the –OH groups of PVA and the –COOH groups of citric acid. Simultaneously, the broad O–H stretching band experiences a reduction in intensity (indicating hydroxyl group consumption), while the intensity ratio of C=O to O–H rises. The functionalized graphene, which has been oxidized using HNO3, introduces supplementary oxygenated functional groups (–COOH/C=O); however, these groups overlap with the existing spectral regions and primarily serve to enhance the carbonyl band intensity. Below is a summary of all key findings from FTIR spectra analysis:
3200–3600 cm−1: Broad –OH stretch (PVA hydroxyls, H-bonded)
2930/2850 cm−1: Asymmetric/symmetric CH2 stretches (PVA backbone)
∼1600 cm−1: C=O stretch (ester) from PVA–CA linkages (and any unreacted vinyl acetate)
∼1100 cm−1: C–O stretch of newly formed ester bridges (R–COO–R′)
Other bands
PVA C–O and C–C stretches near 600, 840 cm−1 remain visible
Degree of Cross-linking from FTIR:
We quantify cross-linking by comparing the ester (C=O) and hydroxyl band intensities. For example, using the integrated areas A_C=O (around 1600 cm−1) and A_OH (around 3200–3600 cm−1) as described by Castro et al.
36
gives a crosslink index as follows:
The result of the degree of cross-linking is summaried below:
This rising trend indicates progressive ester formation over time. (Using peak heights instead of areas yields larger X, e.g. ∼0.75 at 20 h, but area ratios are more robust.) In practical terms, only a modest fraction (∼5%–10%) of PVA hydroxyls have formed ester links after 20 h under these conditions. This behavior – growing C=O peak and shrinking O–H peak with time – matches literature observations for PVA–citric acid crosslinking.
The results of FTIR spectroscopy of polyvinyl alcohol-based composite films are shown in Figure 6 (a)-(f). FT-IR spectrum of (a) pure graphene powder, (b) graphene powder functionalised for 4 hours, (c) graphene powder functionalised for 8 hours, (d) graphene powder functionalised for 12 hours, (e) graphene powder functionalised for 16 hours, (f) graphene powder functionalised for 20 hours.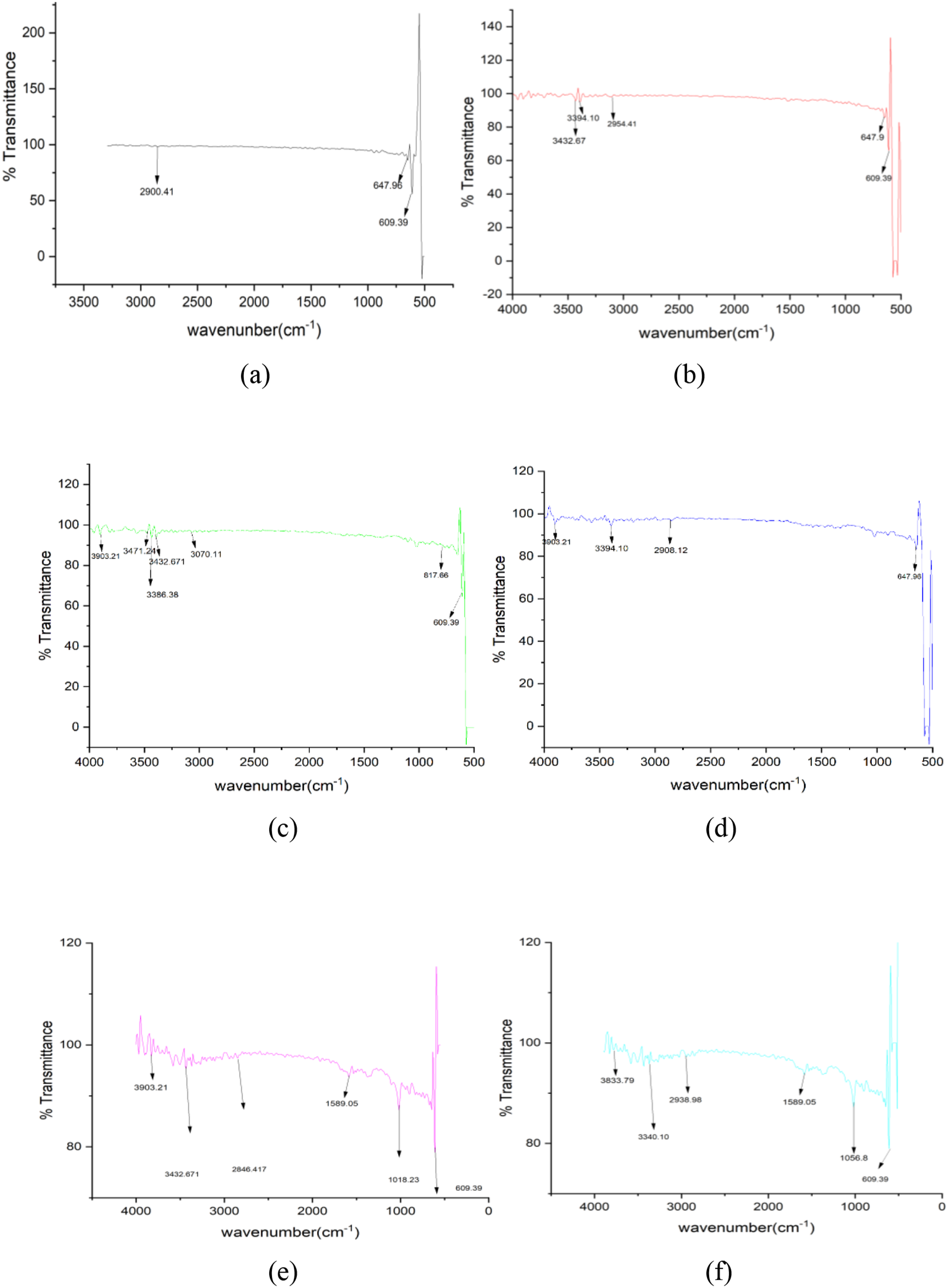
Summary of results of FT-IR spectroscopy.
Optimising cross-linking parameters
To evaluate the data derived from the water absorption test administered on all specimens, a series of multiple linear regression analyses were performed. This methodological framework sought to elucidate the correlation between the dependent variable (water solubility) and the independent variables (citric acid concentration, oven temperature, stirring temperature, oven duration, and stirring duration). This analytical procedure has the potential to ascertain the significance of the aforementioned parameters in the stability of cross-linked PVA films. The overarching relationship deduced through the multiple regression analysis is articulated by equation (1), as delineated below.
Results of regression analysis I.
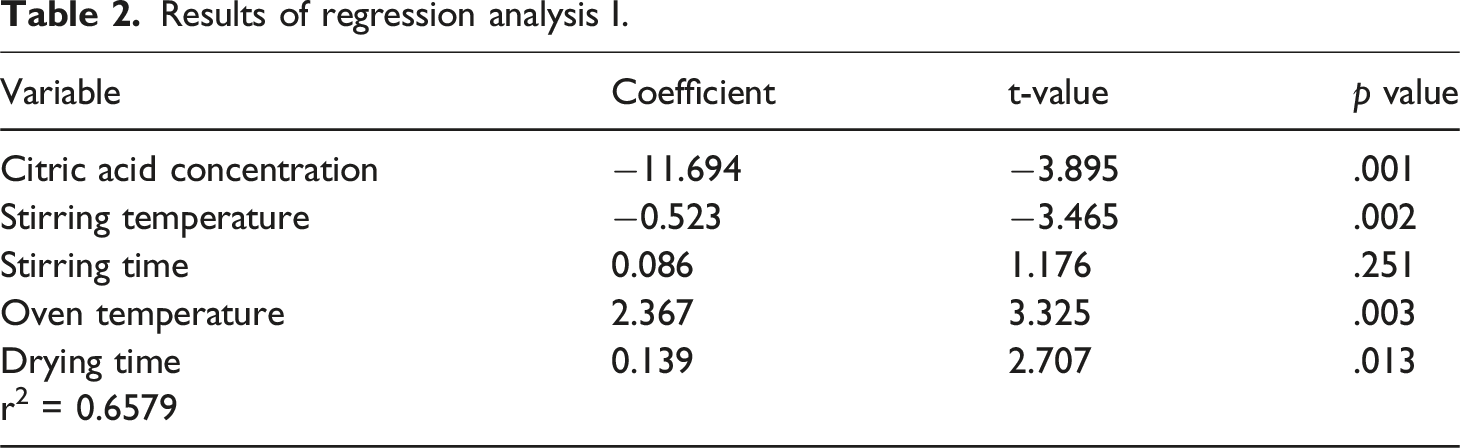
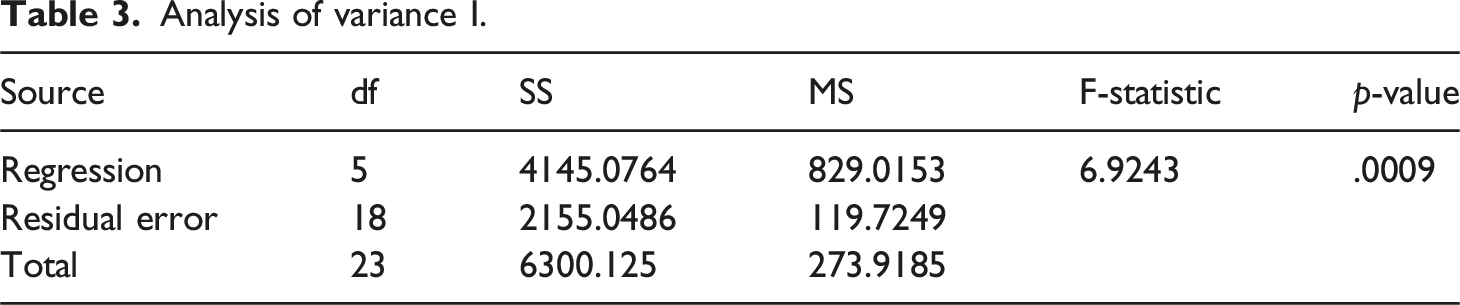
The following tables show the results obtained after application of regression analysis
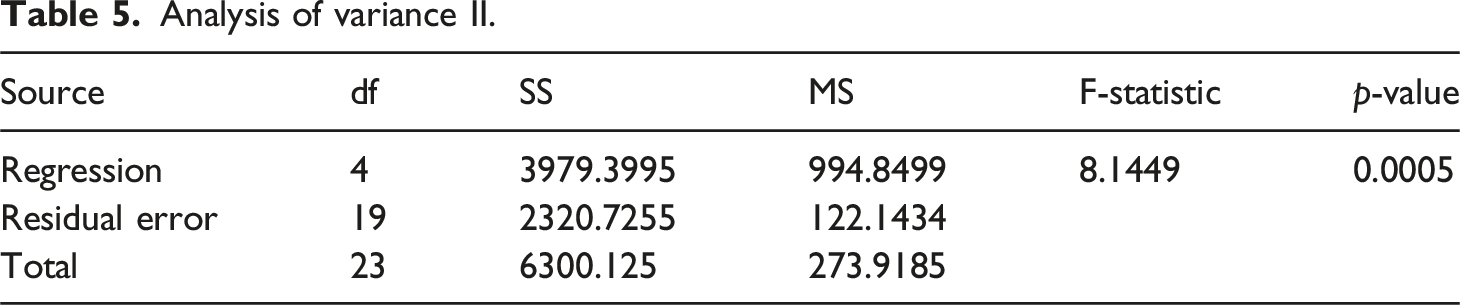
Analysis of variance I.
Results of regression analysis II.
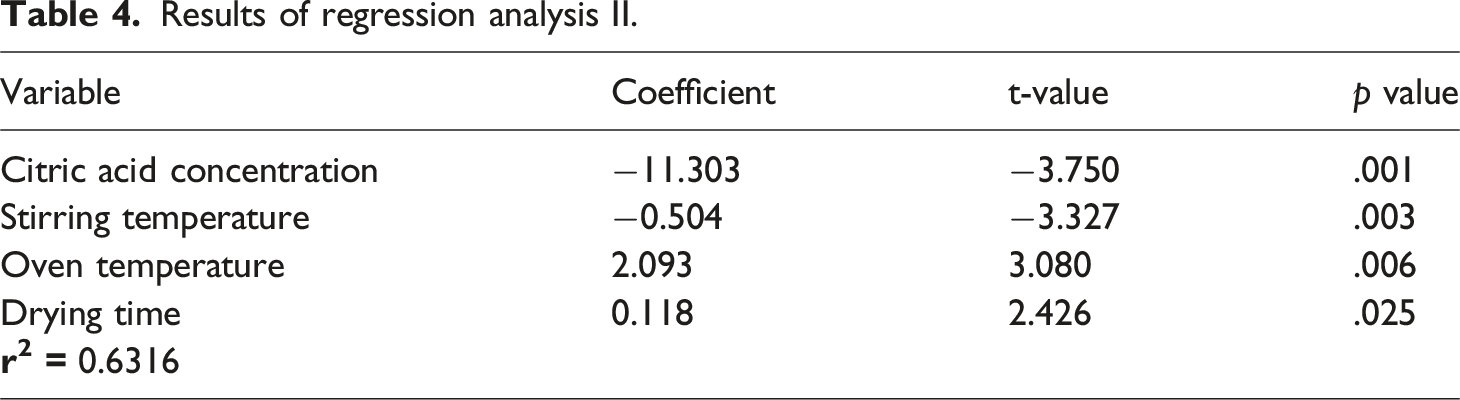
Analysis of variance II.
Optimum value of parameters.

Mechanical testing
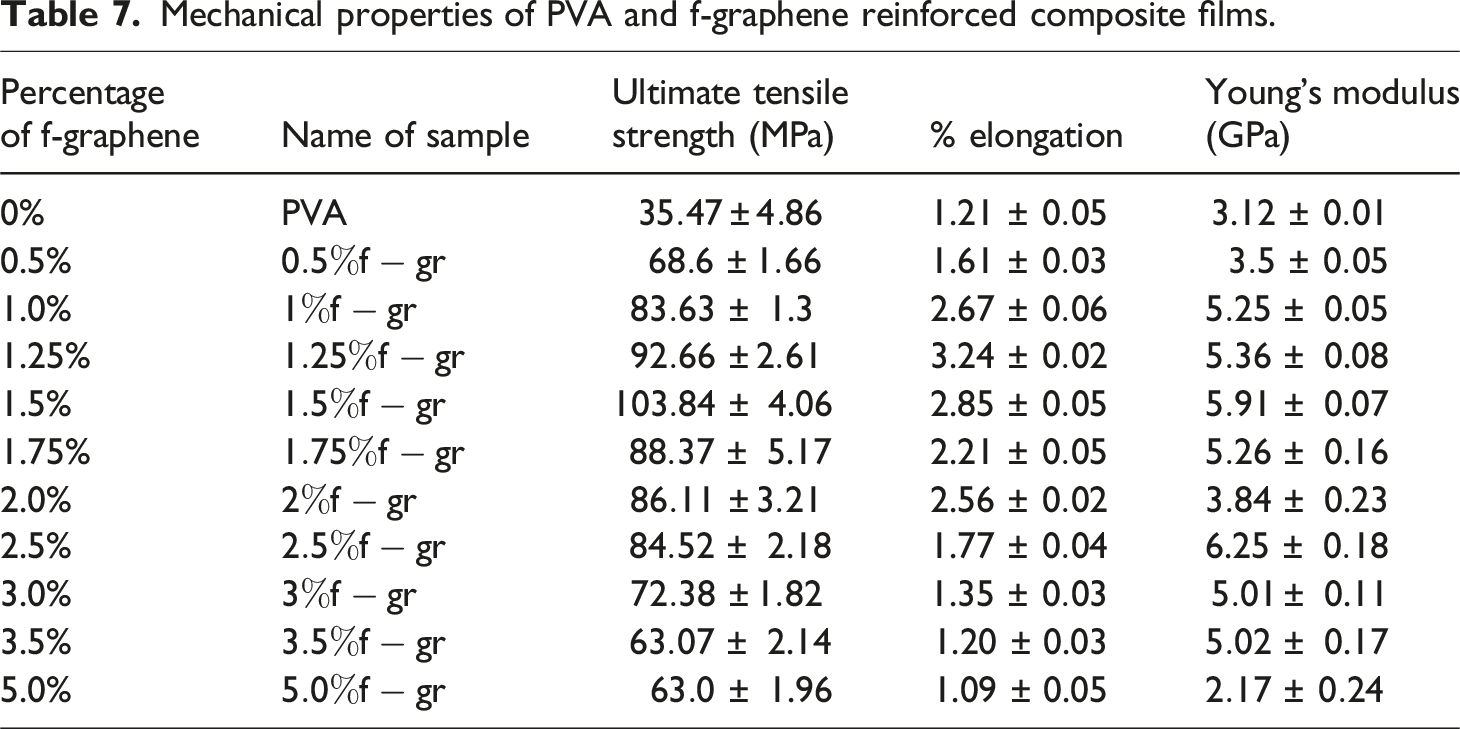
Mechanical properties of PVA and f-graphene reinforced composite films.
The stress-strain curve (Figure 7) elucidates the mechanical characteristics of composite films comprising varying weight percentages of functionalised graphene subjected to the influence of external forces or stresses. The unadulterated PVA film exhibited the lowest ultimate tensile strength (UTS) value, quantified at 35.47 MPa. Conversely, the composite film containing 1.5 wt% of functionalised graphene displayed the highest UTS value, attaining a measurement of 103.84 MPa. This graph distinctly demonstrates that the incorporation of functionalised graphene into the PVA matrix markedly improved the UTS of the composite film. Stress-strain curve for different compositions of f-graphene in composite films.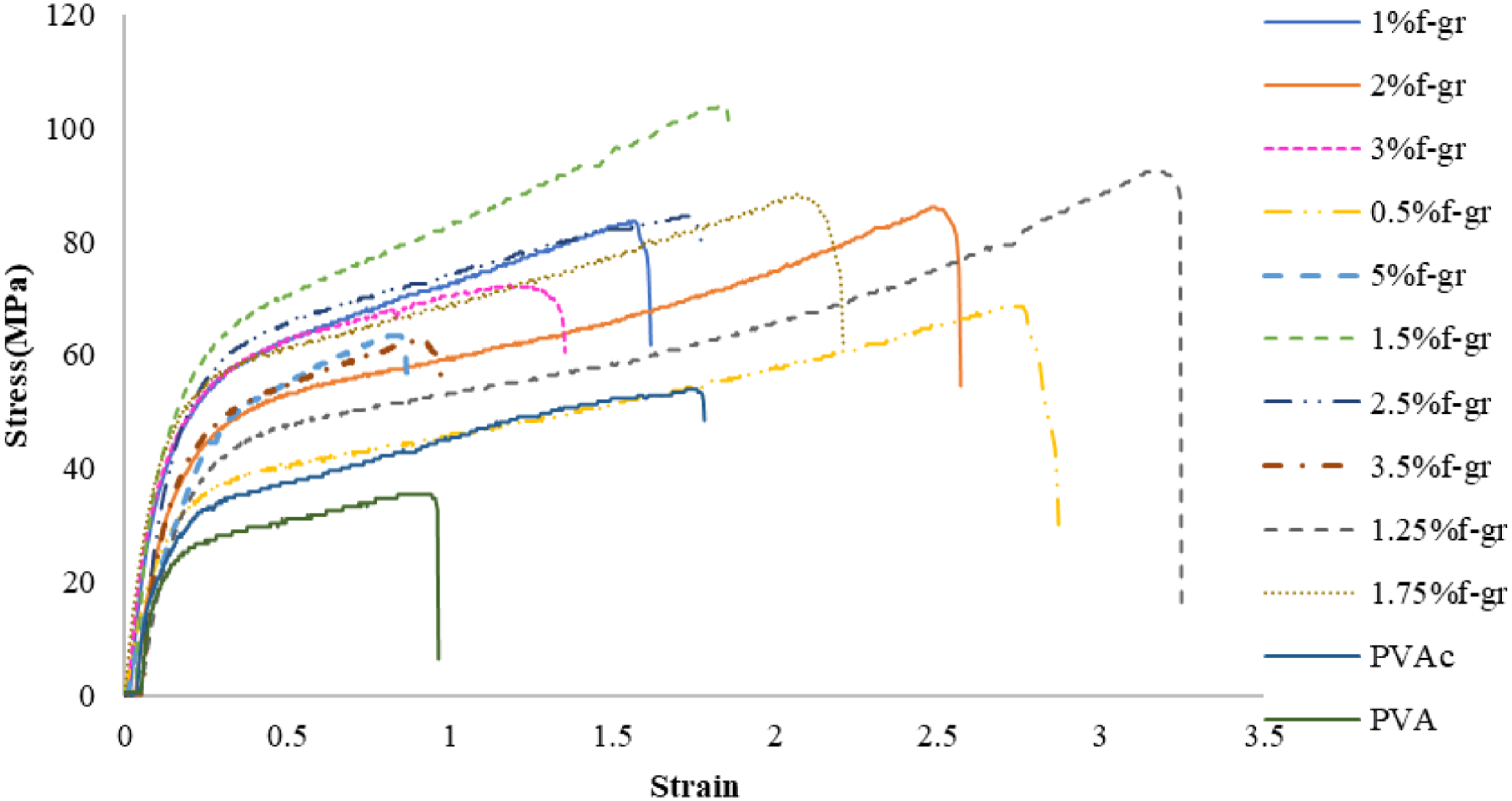
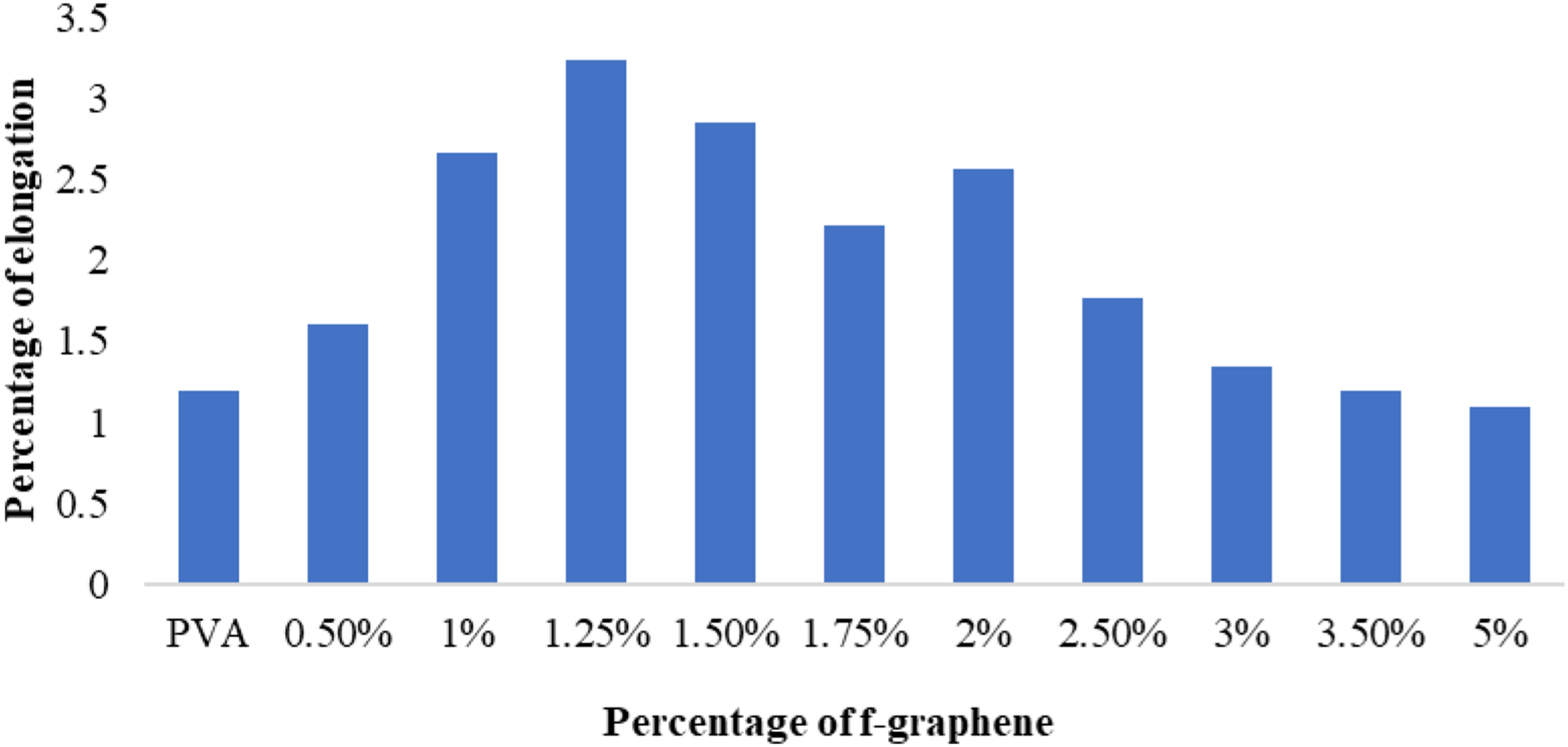
Figure 8 illustrates the relationship between the weight percentage of functionalised graphene and the tensile strength, while Figure 9 presents the variation in the maximum percentage of elongation corresponding to different weight percentages of functionalised graphene. Variation of ultimate tensile strength with f-graphene percentage. Variation of the maximum percentage of elongation with f-graphene percentage.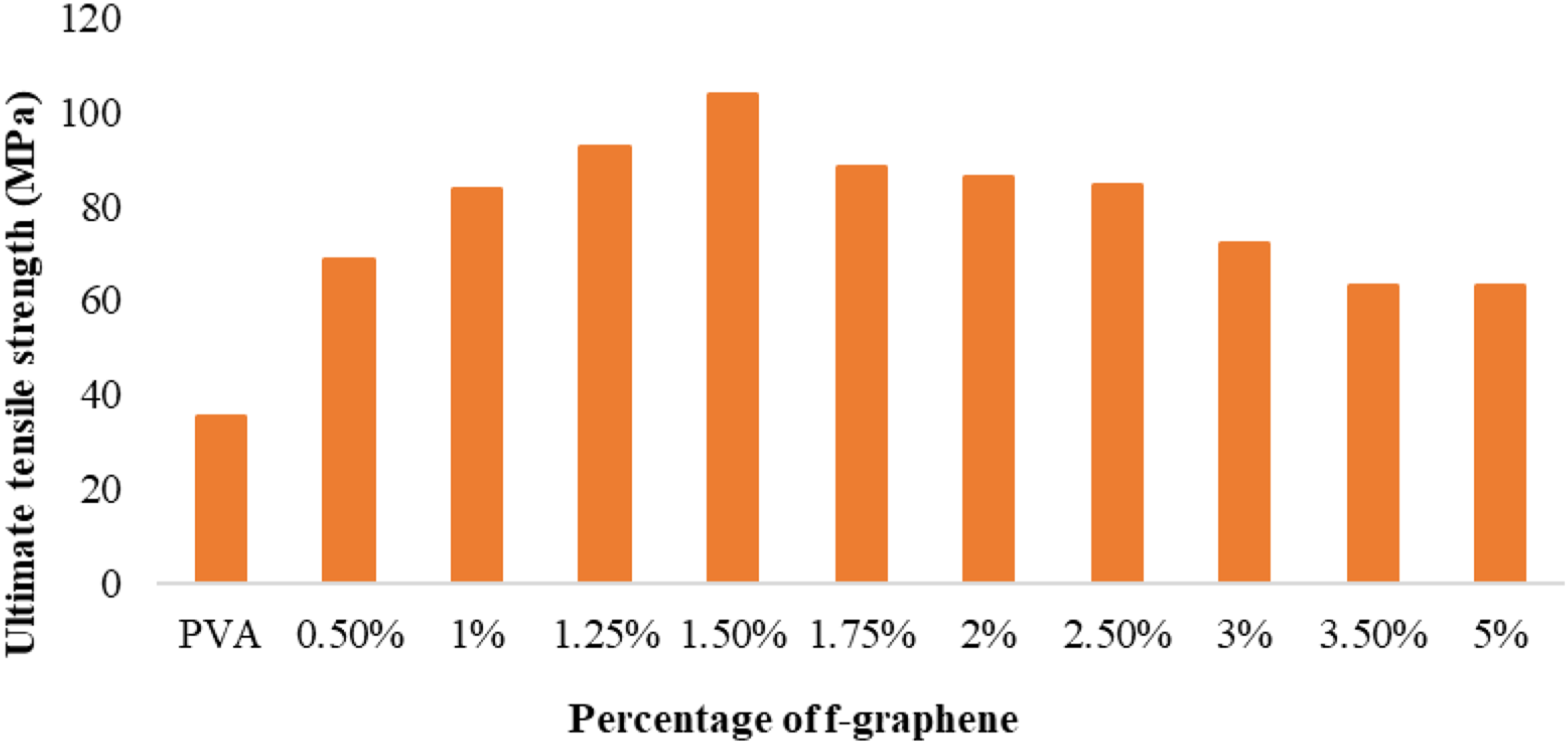
The tensile strength exhibits an initial augmentation correlating with an increase in the weight percentage of f-graphene, reaching a zenith of 103.84 MPa for the composite film incorporating 1.5 wt% f-graphene (Figure 8). Nevertheless, a subsequent increment in the reinforcement results in a decline in tensile strength, as evidenced by the composite film comprising 5 wt% f-graphene, which demonstrates a tensile strength of 63.0 MPa. The peak tensile strength of the composite film with 1.5 wt% f-graphene is 192.75% superior to that of the pristine PVA film, suggesting a considerable enhancement in the intermolecular interactions between f-graphene and PVA. The processes of cross-linking and reinforcement of PVA through the dehydration of hydroxyl groups and the formation of hydrogen bonds play a pivotal role in the augmentation of tensile strength. The incorporation of f-graphene further amplifies the strength by diminishing the hydroxyl group content of PVA up to a defined concentration (1.5 wt%). Beyond this threshold concentration, the carbon present in f-graphene interferes with the hydroxyl groups of citric acid, thereby reducing cross-linking efficacy and leading to a decline in tensile strength. This reduction past a specific weight percentage of f-graphene may be ascribed to the suboptimal interfacial bonding between f-graphene and PVA.
The percentage elongation serves as an empirical indicator of a material’s ductility, reflecting its capacity to sustain plastic deformation prior to fracture. As illustrated in Figure 9, the maximum percentage of elongation exhibits an upward trend concomitant with an increase in the weight percentage of f-graphene. This phenomenon may be ascribed to the augmented interaction between the particulate reinforcement and the polyvinyl alcohol matrix, facilitating a greater relative mobility of molecular chains prior to the onset of material failure. Nevertheless, when the reinforcement content exceeds 1.5 wt%, a deterioration in interfacial bonding between the reinforcement and the matrix becomes evident, resulting in a diminished rate of stress transfer and premature failure of the composite materials.
Scanning electron microscopy
The cross-sectional views of the fractured surface of the neat polyvinyl alcohol film and films with different wt. % of functionalised graphene are presented in Figure 10. Comparison of SEM micrographs for composite films with different weight percentages of f-graphene.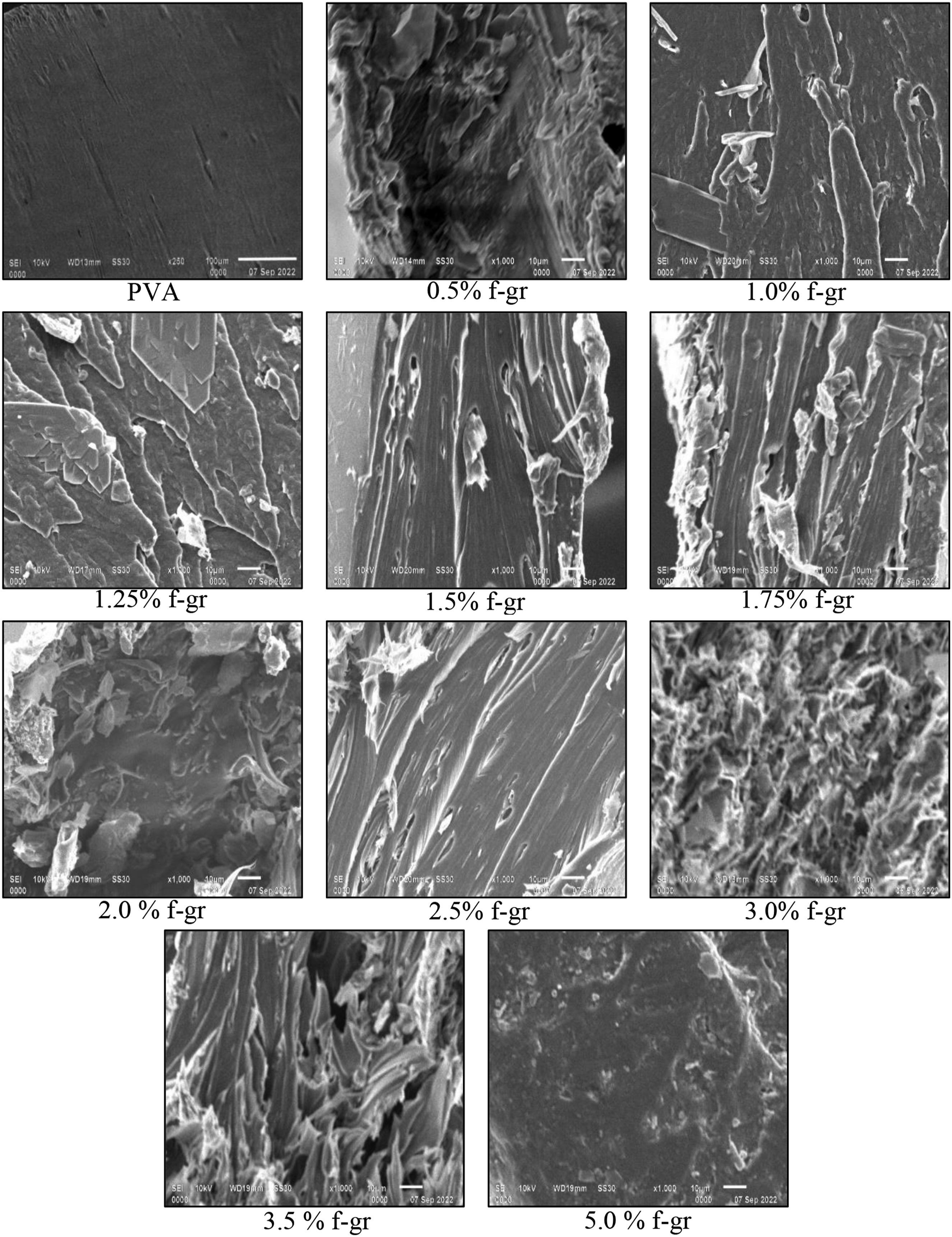
The micrograph of the PVA film exhibited a homogenous surface devoid of distortion, thereby signifying the absence of extraneous or reinforcing particulates. The fracture assessment of composite films containing reinforcement levels of up to 1.5% w/w of PVA indicated that the fracture mechanism was attributable to a tearing process, which suggests ductile behavior. A uniform dispersion of the reinforcement within the matrix was noted, culminating in enhanced mechanical properties and consistent load transfer. This phenomenon was ascribed to effective blending and mixing during the preparation phase. The lack of internal voids, fissures, or interface disruption corroborated the composite film’s homogeneity and the robust adhesion between the reinforcement and the matrix. Nevertheless, at elevated particle concentrations (exceeding 1.5%), the f-graphene particulates exhibited a propensity to aggregate rather than form bonds with the PVA matrix. This aggregation adversely influenced the interaction, resulting in diminished strength and heterogeneous distribution, thereby engendering localized zones of weakness. The deficiency in adequate bonding and distribution impeded load transfer, thereby compromising the mechanical characteristics of the composite film.
Biodegradability test
Figure 11 illustrates the biodegradability % or weight loss % of composite reinforced with different weight percentages of functionalised graphene. Biodegradability % of films reinforced with different wt.% of functionalised graphene.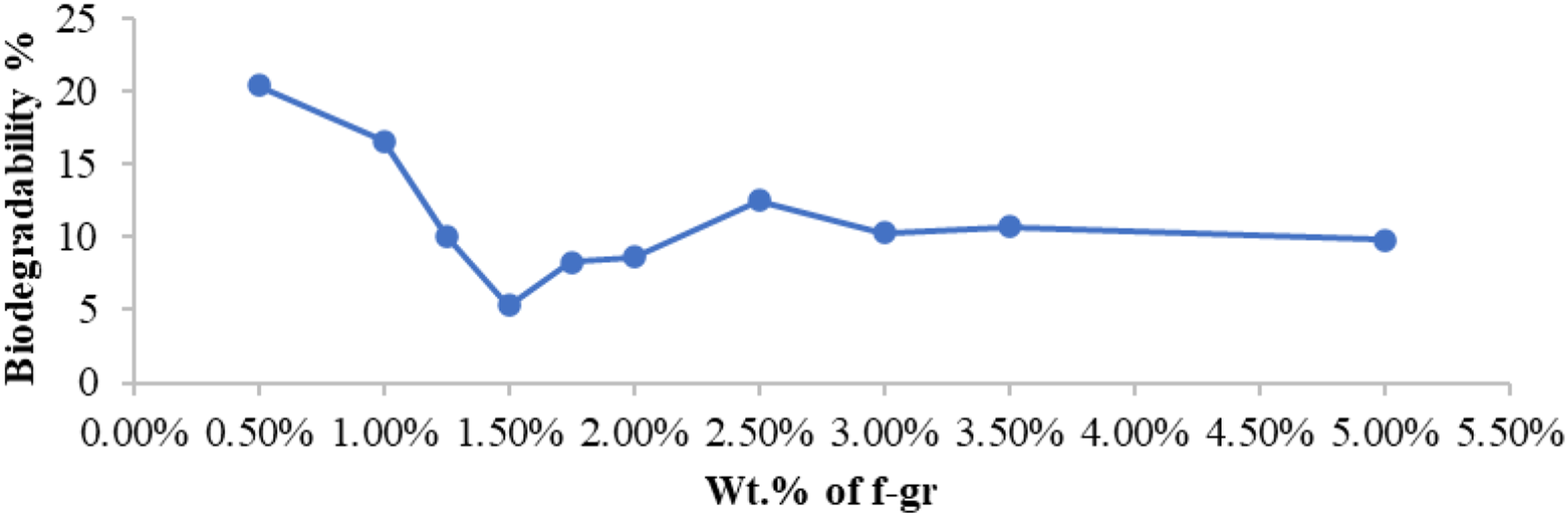
The test aimed to assess the weight loss of composite films when exposed to soil containing degrading microorganisms. The biodegradation of plastics depends on crystallinity. Figure 11 shows biodegradability percentages, highest at 0.5 wt% f-graphene (20.4%) and decreasing up to 1.5 wt% (5.3%), attributed to improved interfacial interaction causing reduced crystallinity. After 1.5 wt%, reduced interfacial interaction led to increased biodegradability.
Thermo gravimetric analysis
The films undergo considerable mass reduction across three separate temperature intervals. The initial interval is characterized by moisture evaporation or dehydration, while the subsequent interval pertains to the thermal degradation of the polymer backbone. The final interval is associated with bond dissociation, resulting in the formation of carbonaceous materials and hydrocarbons. In the case of pure polyvinyl alcohol (PVA), the thermal degradation occurring within the second interval generates significant gaseous products, including carbon monoxide, carbon dioxide, as well as aldehydes, acetone, ethanol, and various other compounds, as documented by Tsuchiya and Sumi. 37
The thermogravimetric percentage (TG%) of composite films with differing weight percentages of functionalized graphene was evaluated over a specified temperature range, as illustrated in Figure 12. TG % curves for films with various weight% of functionalised graphene.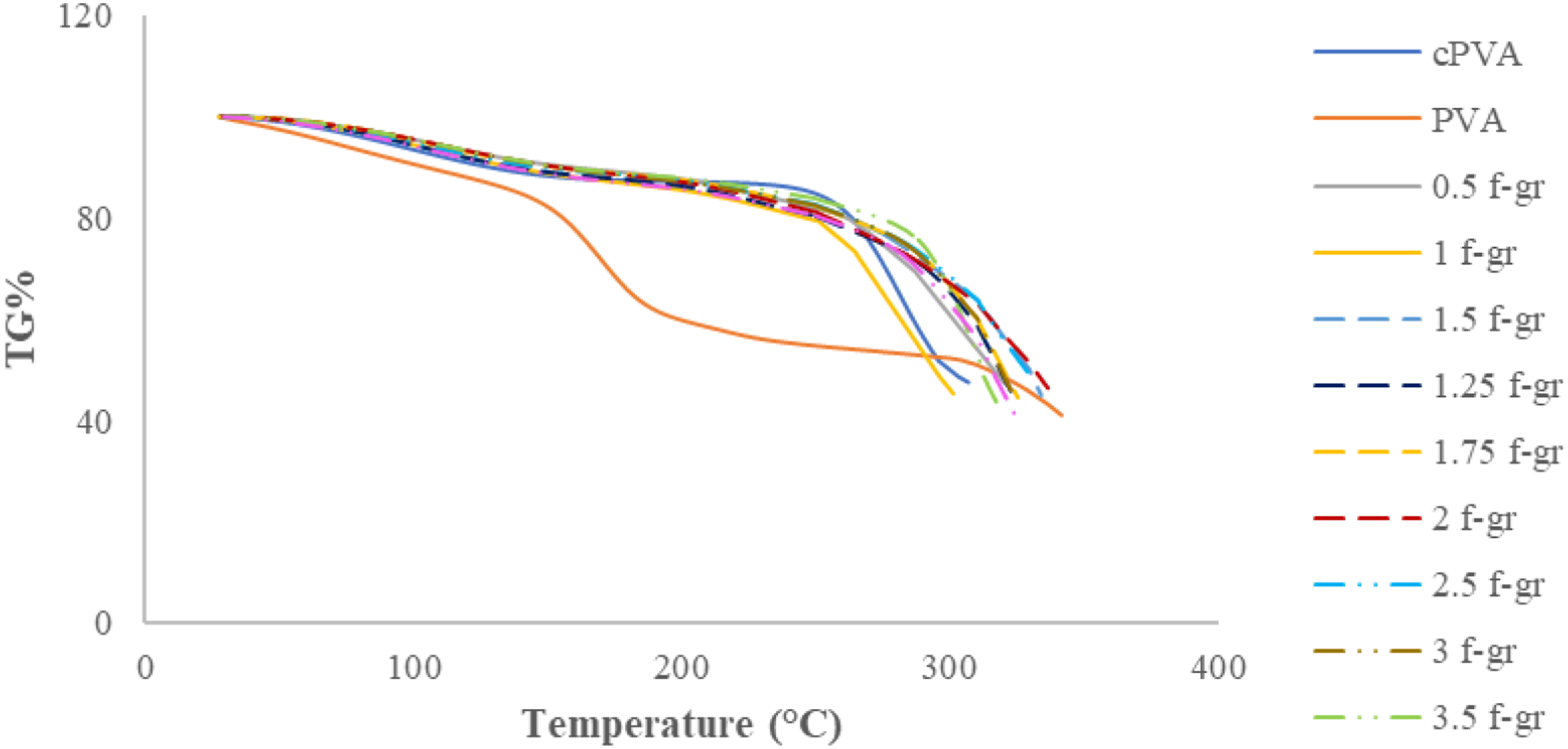
The results indicated that the residue weight % after degradation stages for composite films was higher than for PVA film, demonstrating better thermal stability (Figure 12). The weight loss during the second decomposition stage was gradual for composites, unlike the sudden loss for PVA, further confirming their enhanced thermal stability. Composite films also showed a higher second-stage decomposition temperature than PVA, supported by peaks at higher temperatures in their DTG curves. The functionalised graphene’s better thermal stability and improved interfacial bonding contributed to the enhanced thermal stability of the composites.
Dynamic mechanical analysis
DMA curves typically exhibit three phases: the β-relaxation, indicating bond stretching and functional group movement; the α-relaxation, representing parent chain slipping due to increased molecular motion and mean free volume in the polymer matrix; and the plateau region Menard.
38
The glass transition temperature is when the α-relaxation starts, corresponding to the peak tan δ value. The storage modulus reflects elastic behaviour and stiffness, with higher values indicating greater stiffness. The loss modulus measures viscous behaviour and heat dissipation, while tan δ represents the loss-to-storage modulus ratio. Figure 13, 14 and 15 displays the variation of storage modulus, loss modulus and tan δ, respectively, with temperature for composite films with different weight percentages of functionalised graphene. Storage modulus of PVA and composite films with different wt.% of functionalised graphene. Loss modulus of PVA and films with different wt.% of functionalised graphene. Tan δ curve for PVA and films with different wt.% of functionalised graphene.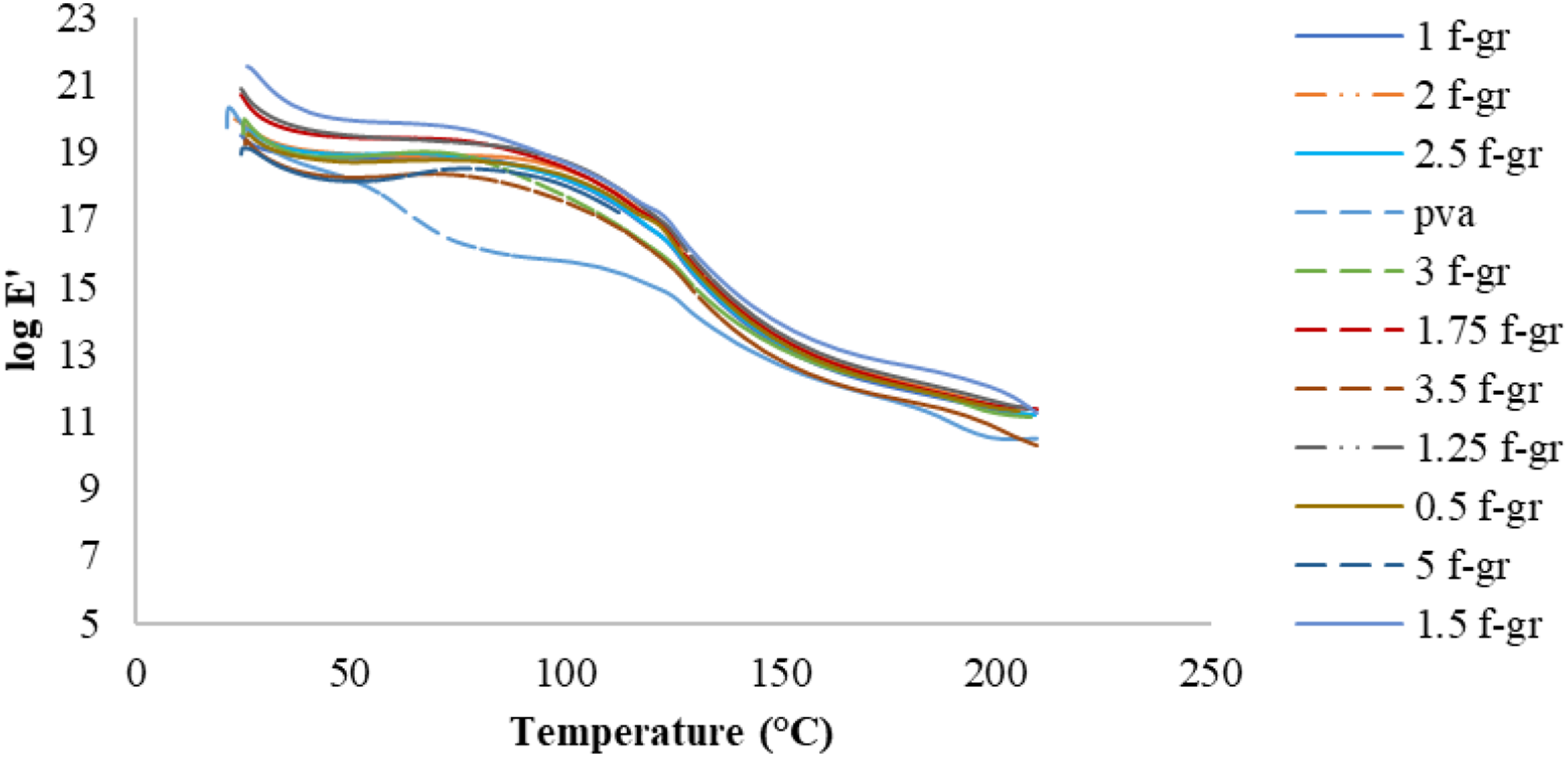
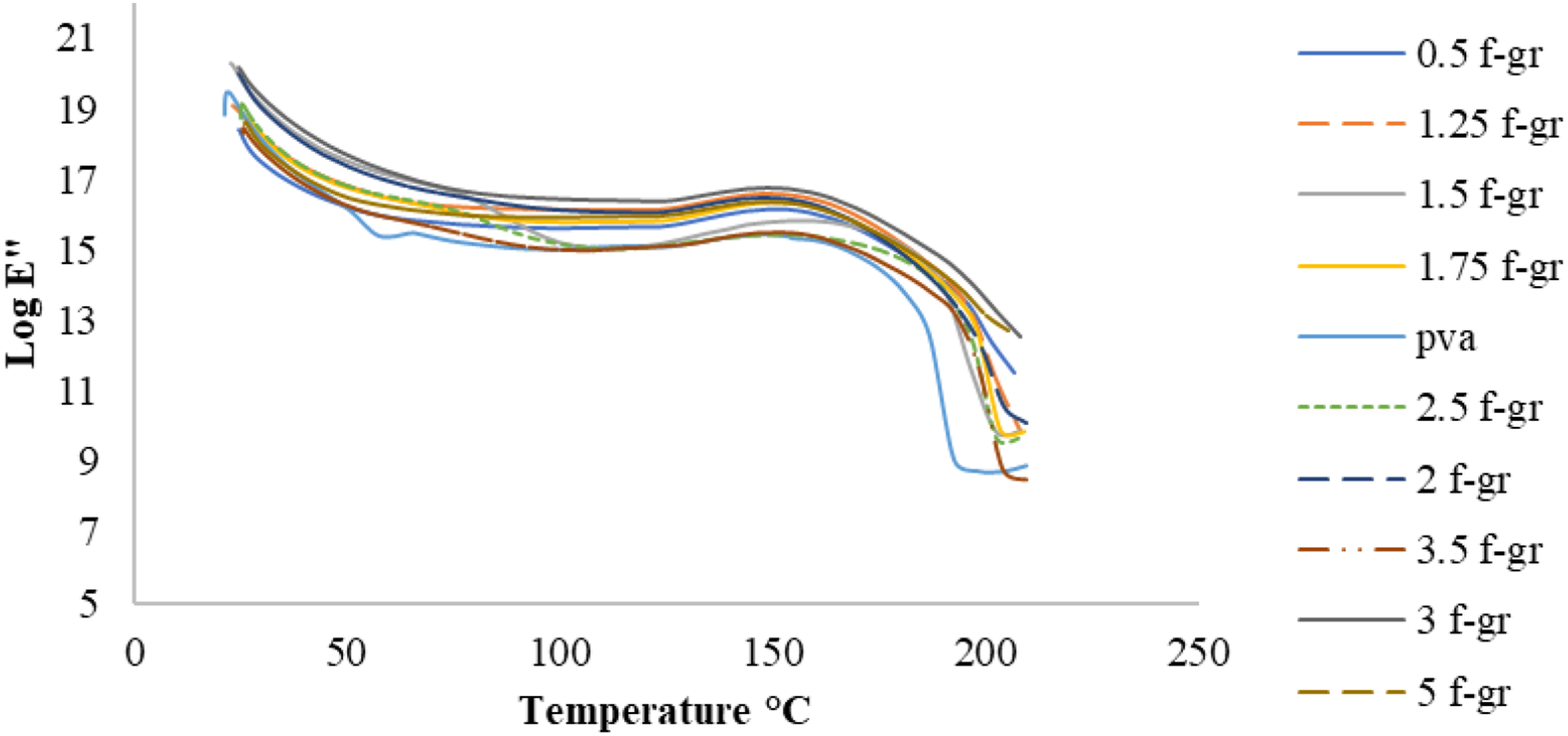
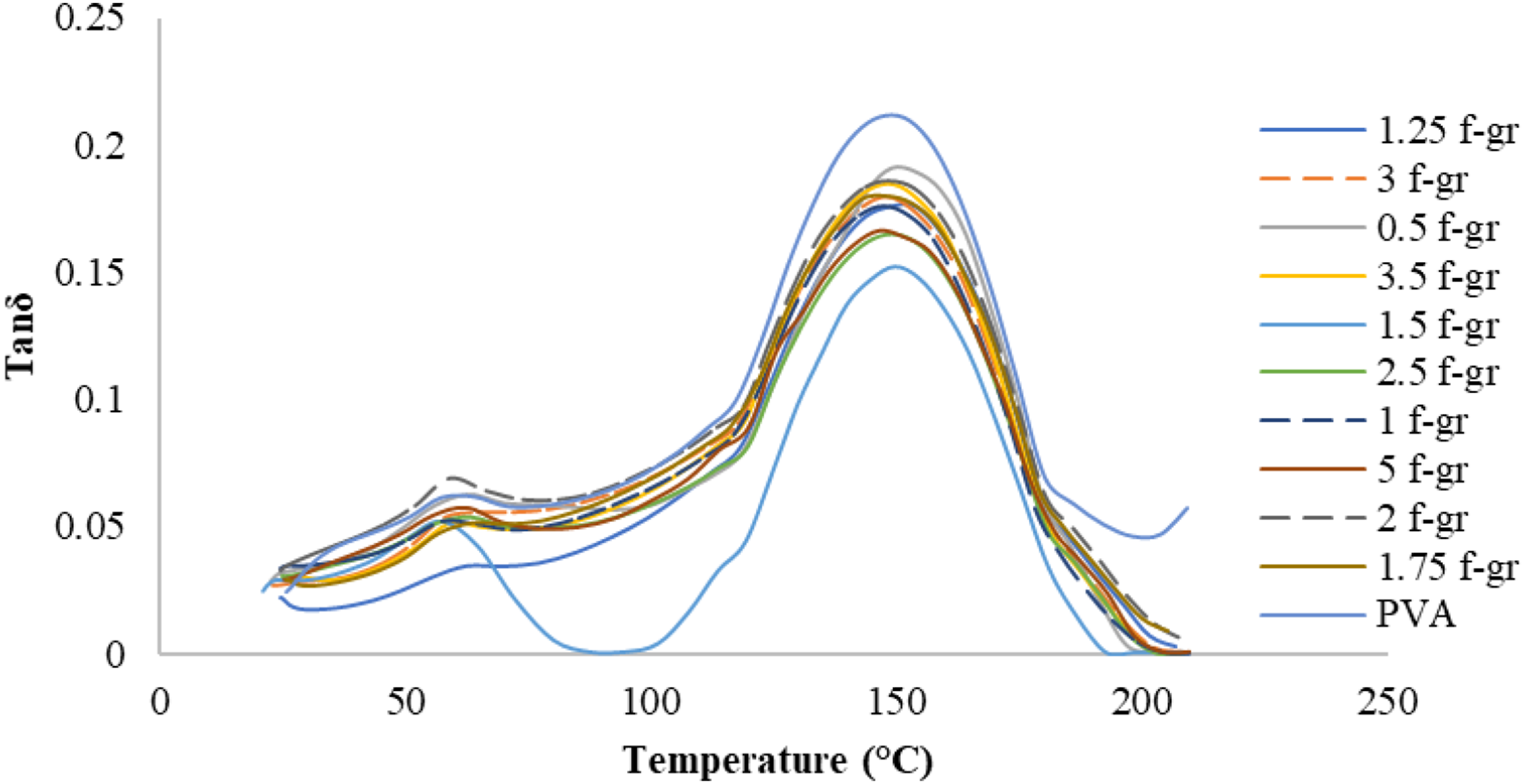
Figure 13 illustrates that the composite films exhibited higher storage modulus than PVA, indicating increased stiffness due to interfacial bonding between the matrix and reinforcement. Among different weight percentages of reinforcement, 1.5 wt% f-graphene had the highest storage modulus, reflecting the strongest interfacial bonding, while 1.75 wt% f-graphene showed the lowest values. Figure 14 indicated a slight increase in loss modulus values around 150°C, the glass transition region. The composite film peaks were flat due to reinforced bonding, inhibiting matrix chain relaxation. PVA showed a lower loss modulus than composite films.
Tan δ is vital for understanding the damping behaviour of composite films, with its maximum value corresponding to the glass transition and reflecting molecular mobility. The viscoelastic behaviour of functionalised graphene-reinforced composite films can be assessed by the tan δ value, where lower values indicate higher elasticity and energy storage. The damping factor peaks for PVA were slightly higher than those for composite films, with a slightly lower glass transition temperature (Figure 15).
Dynamic mechanical properties of PVA and composite films reinforced with different wt. Percentages of f-graphene.
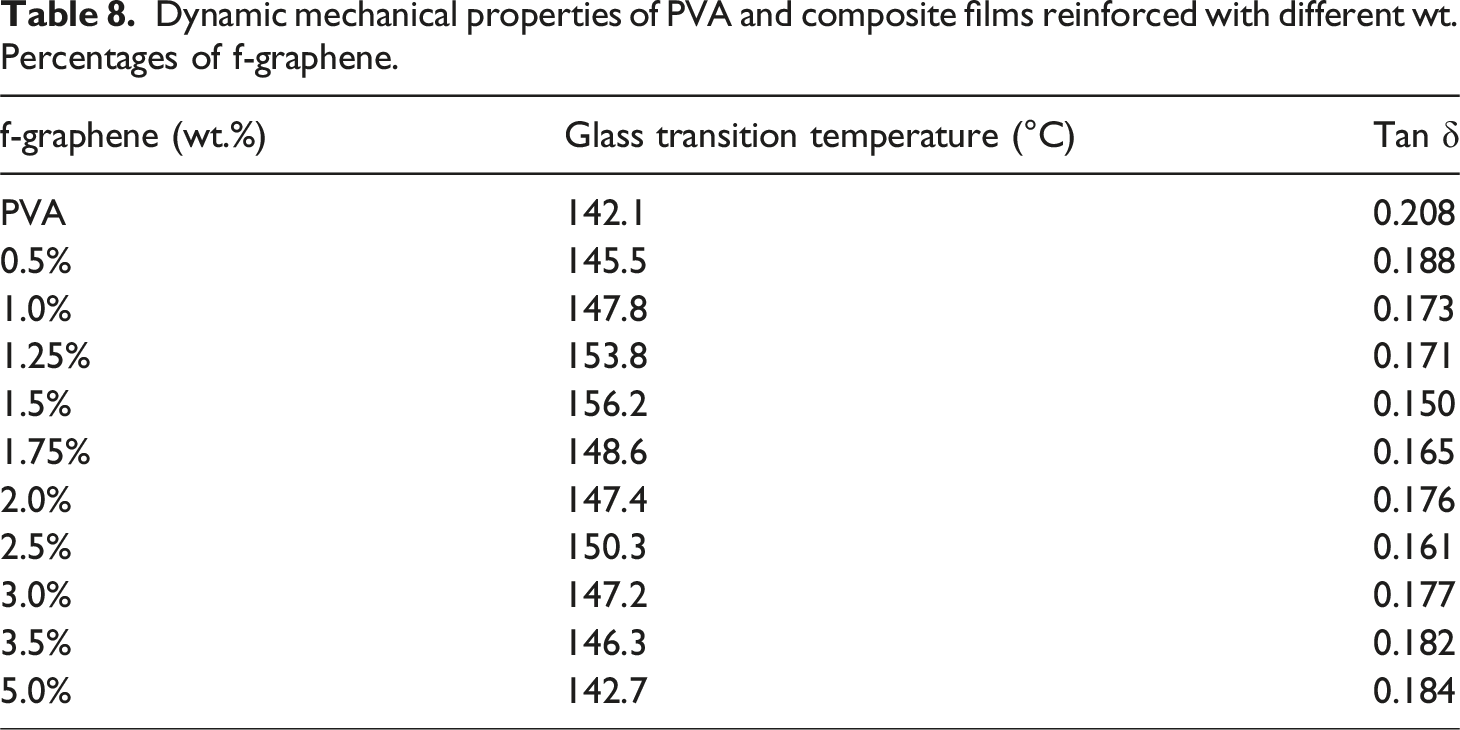
The highest glass transition temperature (156.2°C) for the composite film with 1.5 wt% functionalised graphene was observed compared to PVA (142.1°C) (Figure 16). This increase is attributed to strong interfacial bonding. However, overloading with graphene particles beyond 1.5 wt% reduces the bonding and lowers the glass transition temperature. The composite films exhibit viscoelastic behaviour with a high storage modulus, suitable for noise reduction, vibration isolation, and shock absorption. Variation of Tg with different wt.% of functionalised graphene.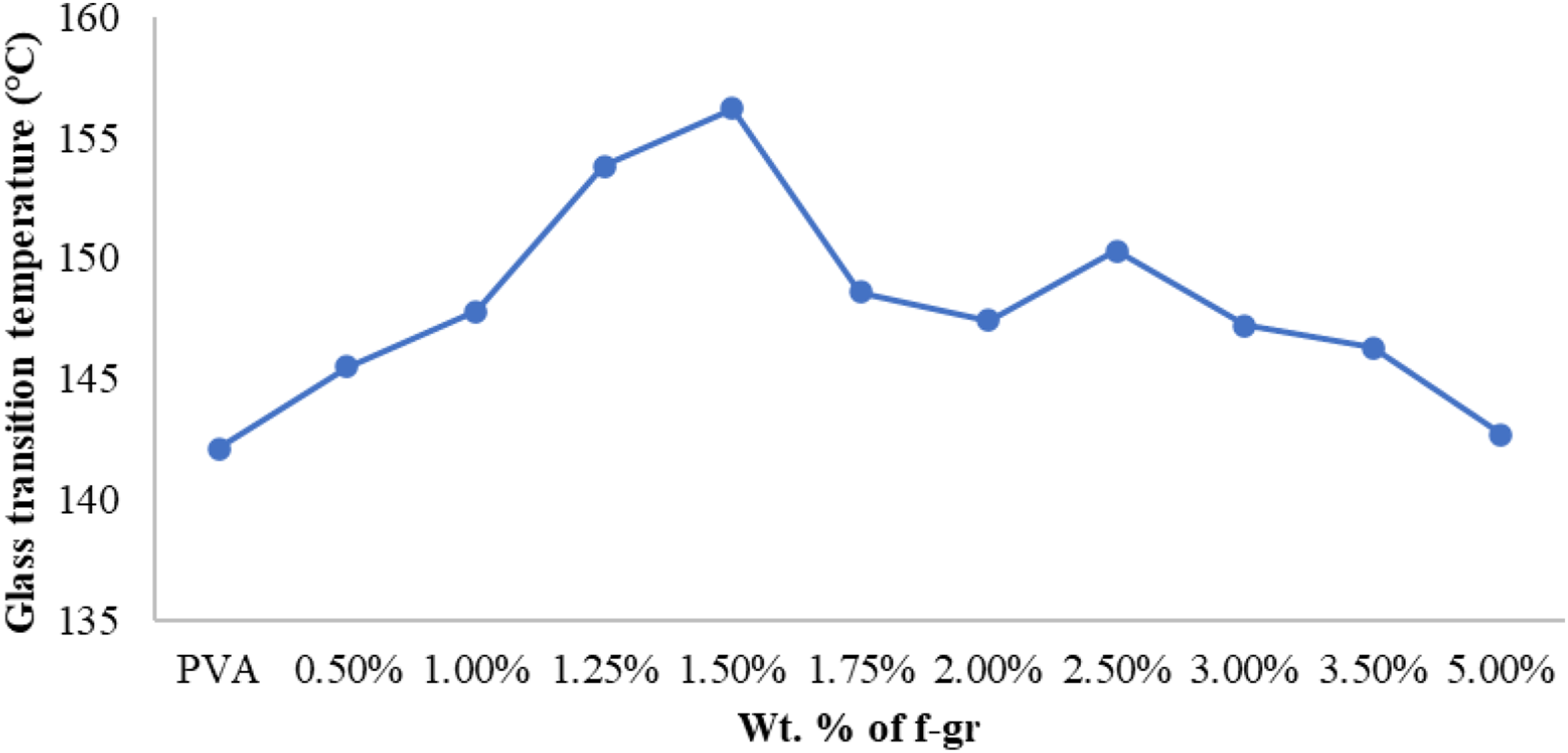
Composite properties’ summary
The next part outlines the key characteristics of pure PVA, modified graphene, and the composite material created in this research.
Solubility
Pure PVA: Polyvinyl alcohol dissolves well in water because of its plentiful –OH groups. It absorbs water and dissolves easily, which limits some uses.
Pure Graphene (nitric-acid functionalized): Graphene powder, even after nitric-acid treatment (which adds –COOH/–OH groups), doesn’t dissolve well in water. The oxygenated edges may allow for stable mixtures, but graphene still acts as a non-absorbing, water-repellent filler.
PVA/CA–Graphene Composite: This mixture is notably water-insoluble. Citric-crosslinked PVA creates a non-soluble 3D structure, and dispersed graphene sheets further obstruct water flow. In practice, including 1.5% wt% of graphene in the PVA/CA composite significantly lowers water absorption and vapor permeability.
Biodegradability
Pure PVA: PVA is viewed as biodegradable compared to many synthetic polymers, although it often needs specific enzymes or microbes. Research indicates that PVA is “more biodegradable than other biopolymers”, but actual degradation can be slow without appropriate conditions.
Pure Graphene (nitric-acid functionalized): Graphene is usually very stable in the environment. Most graphene substances do not easily biodegrade. In practice, nitric-acid treated graphene filler will remain in soil or water environments indefinitely.
PVA/CA–Graphene Composite: The biodegradation of the composite is mainly influenced by the PVA framework. In water, PVA can hydrolyze or degrade slowly, but the CA crosslinks significantly slow this process. Meanwhile, the graphene filler stays intact. Overall, the composite breaks down much slower than pure PVA.
Tensile strength
Pure PVA: Pure PVA films are moderately strong and flexible (tensile strength around 20–40 MPa, elongation >100%). Standard PVA can have a strength of about 20–50 MPa.
Pure Graphene (nitric-acid functionalized): Graphene has very high inherent strength (single-layer graphene ∼100 GPa). Functionalization (with nitric acid) creates defects, making powder sheets less than perfect graphene, but still significantly stronger than polymers.
PVA/CA–Graphene Composite: In the citric-crosslinked composite, small amounts of graphene (≈1–5 wt%) strongly enhance the PVA structure. Graphene sheets bond effectively through hydrogen bonding and entanglement, so 1%–2% often achieves peak strength. However, because both crosslinking and graphene add stiffness to the matrix, high filler levels (>5%) can lead to brittleness. Overall, the PVA/CA + graphene composite exhibits significantly higher tensile modulus and yield strength than pure PVA.
Thermal properties
Pure PVA: PVA is semi-crystalline; it generally melts or becomes soft around 180 °C–220°C and breaks down (through dehydration and chain scission) roughly between 200 °C and 350°C. Its glass transition (Tg) is approximately 80 °C–85°C for high-M_w PVA.
Pure Graphene (nitric-acid functionalized): Graphene itself is thermally very stable (begins to decompose above ∼500–600°C in air). As a filler, graphene serves as a heat sink and barrier to mass transport. It transfers heat well and delays polymer breakdown.
PVA/CA–Graphene Composite: Composites demonstrate considerably better thermal stability compared to pure PVA. The even distribution of graphene creates complex paths that slow the escape of volatile substances, while the CA structure prevents early melting. The overall result is a higher glass transition and a greater residual mass.
Conclusions
Based on the outcomes and discoveries of the investigation, the following deductions may be formulated: FTIR analysis substantiated the successful implementation of oxidative acid treatment on graphene subsequent to a reflux duration of 16 hours. The process of cross-linking significantly enhanced the solubility and diminished the hydrophilicity of polyvinyl alcohol (PVA). The incorporation of functionalised graphene as a reinforcing agent markedly improved the mechanical characteristics, with a loading of 1.5 wt% demonstrating the optimal ultimate tensile strength (UTS) of 103.84 MPa, which is threefold greater than that of PVA. Scanning Electron Microscopy (SEM) elucidated a tear fracture morphology and a uniform distribution at a concentration of 1.5 wt% functionalised graphene, thereby signifying the ideal extent of interfacial interactions. Thermogravimetric analysis revealed an enhancement in thermal stability and a gradual degradation of the matrix in composite materials when juxtaposed with pure PVA. Dynamic Mechanical Analysis (DMA) illustrated an increased storage modulus for the composite films, thereby rendering them stiffer. The glass transition temperature exhibited an increase up to the 1.5 wt% functionalised graphene threshold, followed by a subsequent decline. Biodegradability was observed to be highest at 0.5 wt% functionalised graphene (20.4%) and lowest at 1.5 wt% (5.3%).
Footnotes
Author contributions
Priya: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Software, Validation, Writing (original draft). V.K. Singh: Conceptualization, Investigation, Funding acquisition, Supervision, Validation, Writing (review and editing). Rakesh Saxena: Conceptualization, Investigation, Funding acquisition, Supervision, Validation, Writing (review and editing).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
Data will be made available at request.