Abstract
This study presents the synthesis of zinc ferrite (ZnFe2O4) nanoparticles via a simple green route and their reinforcement in polyvinyl chloride (PVC) to fabricate nanocomposite films. The structural, optical, and thermal properties of PVC/ZnFe2O4 nanocomposites were examined at varying nanoparticle concentrations (1 wt.%, 3 wt.%, and 5 wt.%). X-ray diffraction confirmed the polycrystalline nature of the films, while FESEM analysis demonstrated a uniform dispersion of ZnFe2O4 nanoparticles within the PVC matrix. Optical studies indicated enhanced UV absorption with increasing ZnFe2O4 content, accompanied by a significant reduction in bandgap energy from 3.0 eV to 1.2 eV. Photoluminescence analysis revealed a decrease in electron-hole recombination, suggesting improved charge separation. Wettability studies showed an increase in the water contact angle, indicating enhanced hydrophilicity in the modified films. Thermal analysis demonstrated that the incorporation of ZnFe2O4 nanoparticles accelerated PVC degradation due to their catalytic activity while also increasing char residue formation. These findings highlight the potential of PVC/ZnFe2O4 nanocomposites in applications requiring tailored degradation behavior and enhanced optical performance.
Keywords
Introduction
Nanocomposites are now widely used in several industries and in everyday life. In this regard, metal nanoparticles incorporated in polymeric matrices have emerged as a viable method for customizing material characteristics in a variety of sectors. This synergistic combination takes use of the unique properties of both components, resulting in improved mechanical, electrical, thermal, and optical performance.1–3 By effectively controlling the size, shape, and dispersion of metal nanoparticles within the polymer matrix, researchers and engineers can design new materials with specific properties suitable for a variety of applications. Ongoing advancements in this field could lead to the discovery of innovative polymer-nanomaterial combinations and fabrication techniques, enhancing both electrical insulation technologies and material optical properties.4,5 The strategic selection and distribution of nanofillers throughout the polymer matrix can improve the mechanical strength, thermal stability, optical, and electrical insulating characteristics of polymer nanocomposites. This not only enhances the safety and performance of power systems but also contributes to the development of more sustainable and environmentally conscious energy infrastructure.
Spinel ferrites have recently gained significant attention as versatile inorganic nanomaterials due to their exceptional electronic, optical, electrical, magnetic, and catalytic properties, making them suitable for a wide array of advanced applications across various industries. These materials are commonly utilized in numerous technologies, such as transformer cores, inductors, microwave systems, radars, tunable signal filters, antenna rods, phase shifters, junction circulators, and isolators, underscoring their broad utility and importance in modern technological advancements.6–8 Among the spinel ferrite, Zinc ferrite is a well-studied spinel ferrite due to its unusual features, including thermal and chemical stability and magnetic features that vary with particle size and it has a usual spinel structure with Zn2+ ions at the tetrahedral site and Fe3+ ions at the octahedral site.9,10 There are several ways for synthesizing ZnFe2O4 nanoparticles and composites, including sol-gel, micro-emulsion, sono-chemical, microwave, co-precipitation, and high-energy ball milling.11–15 But the properties depend on Chemical composition and microstructural factors, such as particle size and shape, might influence these qualities during production. The objective is to obtain the required physical, thermal, and chemical properties while minimizing the dangers to human health and the nature world from product emissions. Therefore, we are emphasizing green synthesis, which is more cost-effective, generates fewer pollutants, and is safer for both the nature world and health of the humans compared to traditional chemical synthesis.16,17 To eliminate the demand of expensive substrates while also raising awareness among scientists because of its ease, rapidity, and environmental friendliness, we elected to employ an egg white fuel, which decreases the release of hazardous gases during the process of synthesis. When spinel ferrites are disseminated in a polymer matrix, they generate composite materials having the best features of both components. This synergy produces materials with improved or customized properties suited for a wide variety of applications. 18
Polyvinyl chloride (PVC) was selected as the polymer due to its excellent chemical resistance, robust mechanical strength, ease of processing, non-flammability, and cost-effectiveness, making it an ideal choice for a wide range of applications. These key attributes highlight PVC’s versatility and practicality in various industrial uses.19–22 The performance of PVC polymer requires improvement by including different additives to address its distinct limitations under diverse settings. For example, its flammability restricts its application in fire-prone areas. 23 To enhance the properties of PVC polymer, it needs to be mixed with nanoparticles. Due to their high surface-to-volume ratio, nanoparticles offer significant catalytic action or reactivity, making the material suitable for specific applications.24,25 In this research, we try to enhance the physio-chemical features of PVC such as thermal stability, structural and optical. For the intent, we made a novel endeavor to manufacture bio-synthesized zinc ferrite, and various wt.% zinc ferrite dispersed PVC (ZnFe2O4/PVC) polymer nanocomposites by simple solution casting technique. The solution casting approach is utilized to produce PVC/ZnFe2O4 films because it is simple, inexpensive, and facilitates the creation of homogeneous films with adjustable thickness26,27 and due to the risk to public health and the environment from by-product emissions of chemical methods, we are focusing on green synthesis. It is more cost-effective, generates fewer pollutants, and improves safety for both the environment and human health compared to traditional chemical synthesis. 28 When comparing to the other chemical route, bio-synthesized Zinc ferrite prepared in our work has enhanced structural, optical and thermal properties. Moreover, the different weight percentage (1, 3 and 5 wt.%) of multi-content or multi properties of ZnFe2O4 nanoparticle prepared in this work is added to the bare PVC polymer to enhance its physical and chemical properties. And especially 5 wt.% of this NP’s particle dispersion enhanced the optical and thermal characteristics of PVC when compared to 1 and 3 wt.% of NP dispersion. The novelty of this work is using a low concentration of bio synthesized nanoparticle to tune the PVC polymer properties instead of using toxic and high costly materials like carbon nanotube, graphite, silver and gold.
In the literature, it is noted that green-synthesized nanoparticles can significantly alter the properties of polymers for various applications. In our previous work, we utilized green fuel-synthesized copper ferrite and Cu-Zn ferrite nanoparticles as nanofillers in the conducting polymer polyaniline, aiming to enhance the optoelectrical properties of the polyaniline films.29,30 Similarly, Davar et al. synthesized zirconia nanoparticles using rosemary extract as a green route. These nanoparticles were then reinforced into a polyvinyl alcohol (PVA) matrix at varying concentrations. Their analysis revealed that a polymer sample with 1 wt.% nanoparticle reinforcement exhibited mechanical properties that were 5.5 times stronger than the pure polymer film. 31 Additionally, Eugine Prince et al. synthesized TiO2 nanoparticles using egg white as a green fuel. In their research, they dispersed different weight percentages (1 wt.%, 2 wt.%, 3 wt.%, and 4 wt.%) of TiO2 nanoparticles into a PVA/POM polymer composite matrix. Notably, the polymer composite film containing 3 wt.% TiO2 nanoparticles demonstrated the highest dielectric constant and the best electromagnetic shielding effectiveness. 32
According to the above literature, the significance of our work lies in the sustainable synthesis of zinc ferrite nanoparticles using a green route, specifically egg white as a natural reducing agent, which offers an eco-friendly and cost-effective alternative to traditional chemical methods. Incorporating these nanoparticles as fillers in polyvinyl chloride (PVC) polymer matrices opens up opportunities to enhance the mechanical, thermal, and functional properties of PVC, making it more versatile for advanced applications. The study examined how the concentration of ZnFe2O4 nanoparticles in the casting film affected the physicochemical parameters such as structural, optical and thermal of the resulting film.
Materials and methodology
Materials
Ferric nitrate nonahydrate, Fe(NO3)3.9H2O, Zinc nitrate hexa hydrate Zn(NO3)2.6H2O, Polyvinyl chloride (PVC) and Tetrahydrofuran (THF) were purchased from Aldrich with 99% purity and egg white solution is employed as a solvent for current preparation.
Preparation of ZnFe2O4 NP
Zinc ferrite nanoparticles were synthesized using a green solution combustion method, utilizing zinc nitrate hexahydrate (Zn(NO3)2·6H2O) and ferric nitrate nonahydrate (Fe(NO3)3·9H2O) as precursors. Egg white was employed as a natural, eco-friendly fuel and solvent due to its rich protein content, which facilitates uniform dispersion and controlled combustion. The precursors were mixed in their appropriate molar ratios and gradually added to the egg white solution under continuous stirring to ensure homogeneity. This mixture was stirred constantly for 2 h to achieve thorough blending. The resulting solution was then heated at 80°C on a hot plate until a dark brown powder was formed, indicating the development of ash. This ash was subsequently annealed at 600°C for 2 h to enhance crystallization and promote phase formation. One of the main proteins in egg whites, ovalbumin, has functional groups like carboxyl (-COOH), hydroxyl (-OH), and amine (-NH2). These groups promote uniform nanoparticle production by stabilizing precursor metal ions and avoiding agglomeration. The use of egg white as a bio-template not only supports the green synthesis approach but also aids in the controlled nucleation and growth of the nanoparticles. 33
Preparation of PVC/ZnFe2O4 NC film
The PVC/ZnFe2O4 nanocomposite (NC) film was prepared using the solution casting method. Initially, 0.8 g of PVC was dissolved in 40 mL of tetrahydrofuran (THF) and stirred with a magnetic stirrer at 50°C for 1 h to obtain a homogeneous solution. Next, varying amounts of ZnFe2O4 nanoparticles, corresponding to 1, 3, and 5 wt.%, were added to the PVC solution and stirred for an additional 2 h at the same temperature. The resulting mixture was then poured onto Petri dishes and allowed to dry at room temperature for 48 h. Finally, the film was carefully peeled off from the surface. A schematic representation of the preparation process for the PVC/ZnFe2O4 film is provided in Figure 1. Illustration of the fabrication process of PVC-ZnFe2O4 composite film.
Characterization techniques
XRD analysis
X-ray diffraction (XRD) studies handle the valuable insights into the precise structure of the prepared samples. This research employs a device {Shimadzu XRD7000 X-ray diffractometer which has an angular range of 10–80°.
Surface analysis
Atomic force microscopy (NT-MDT, Russia) was used to evaluate the surface morphology and roughness of pure and 5 wt.% ZnFe2O4 dispersed PVC films. The morphology and particle size of the ZnFe2O4 nanoparticles was examined using a CARL ZEISS Scanning Electron Microscopy (EVO18) and Carl Zeiss Gemini 300 field emission scanning electron microscopy (FESEM) device was used to analyze the size and morphological characteristics of the produced films.
Optical analysis
The optical properties like absorbance and bandgap were recorded out using Jasco V-750 UV-Vis double beam Spectrometer with wavelength of 200 – 1000 nm.
Water contact angle analysis
By measuring the water contact angle of the sample, the wetting capacity of each surface was assessed. Water contact angle was determined on the prepared samples utilizing Contact Angle Measuring System (model Phoenix 300 Plus, M/s Surface Electro Optics Co, Ltd, Korea) in Polymer Engineering Laboratory, PSG Institute of Technology and Applied Research, Coimbatore. For this, the sessile drop method was employed. Drops of 8.0 μL volume of water were deposited on the 1 × 1 cm prepared films using a micro syringe. After inserting the water drop, measurements were obtained immediately, and fluctuations were observed for 10 min.
Thermogravimetric analysis
The thermal response of nanocomposite films with different additive loading ratios was analyzed using a thermogravimetric analysis (TGA) equipment (HITACHI STA 7200) at temperatures ranging from 30 to 900°C and a heating rate of 10°C min−1 in nitrogen environment.
Results and discussion
Structural analysis
X-ray diffraction analysis was help to investigate the crystalline structure and properties of the synthesized ZnFe2O4 Nano particle with respect to pristine and 1, 3 and 5 wt.% of ZnFe2O4 PVC film. The recorded XRD pattern of prepared ZnFe2O4 nanoparticles before dispersion can be displayed in Figure 2. The produced ZnFe2O4 nanoparticles exhibit remarkable crystallinity. Sharp peaks in XRD show this clearly. There is no apparent distinctive peak linked to impurities or secondary phases in the synthesized nanoparticle’s XRD spectrum, showing their high level of purity. From the XRD, the crystallite size of the prepared ZnFe2O4 was 38.9 nm via Debye-Scherrer method. Kombaiah and his team synthesized ZnFe2O4 nanoparticles using both conventional and microwave-assisted combustion methods, achieving a crystallite size of 70 nm.
34
Yadav et al. synthesised ZnFe2O4 nanoparticles by starch-assisted sol-gel auto-combustion technique. After annealing the samples at 1000°C, the crystallite size was found to be around 72.60 nm.
35
But our green synthesis approach yields a small crystallite size and offers significant advantages over traditional chemical methods. It is evident that the preparation method using egg white as fuel in our research is superior to other chemical processes that have been revealed by just a few of researchers.34–37 According to JCPDS card no. # 22-1012, the distinctive XRD peaks of ZnFe2O4 nanoparticles correspond to the cubic spinel with face-centered cubic structure and Fd3m space group symmetry.
38
The calculated parameters values for ZnFe2O4 are summarized in Table 1. The recorded XRD pattern of prepared ZnFe2O4. Calculated parameter of the prepared ZnFe2O4 nanoparticle.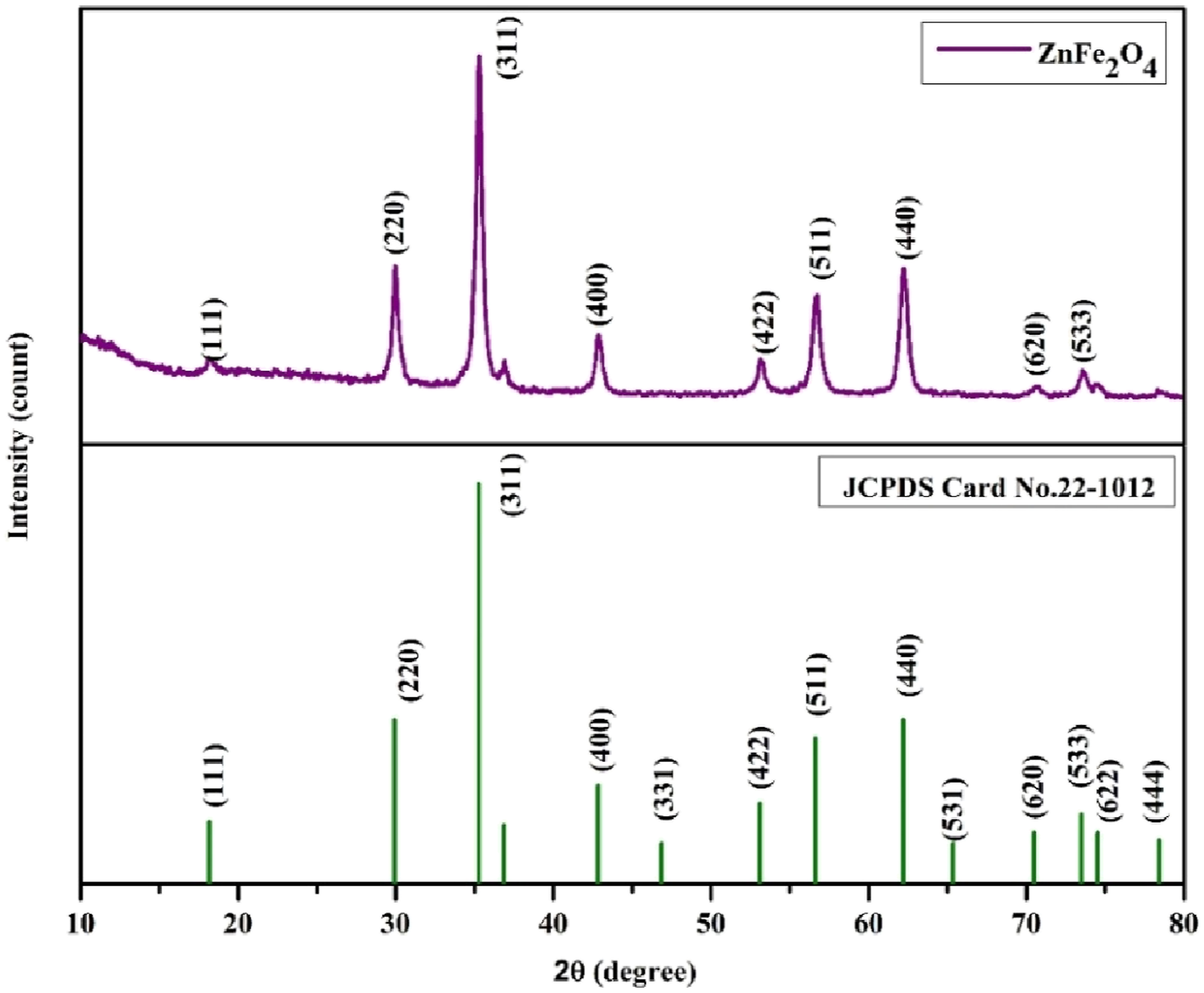
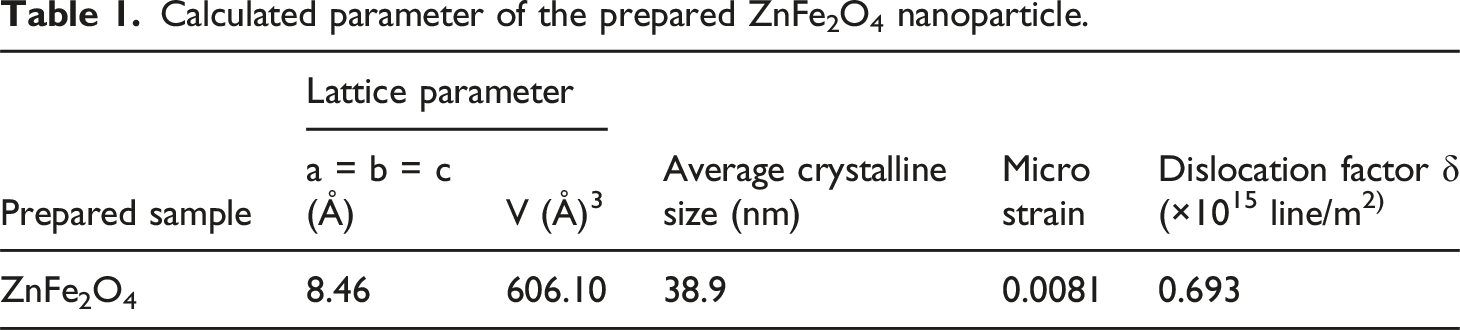
The recorded XRD pattern of prepared composite films and ZnFe2O4 nanoparticles can be displayed in Figure 3(a). PVC is an unstructured thermoplastic, having no regular crystallographic planes with high intensity peaks.
39
The inclusion of ZnFe2O4 nanoparticles in polymeric matrix resulted in the production of polycrystalline structures in cubic lattices with varying degrees of crystallinity. The common diffraction peaks from all the prepared samples are (220) (311) (400) (422) (511) (440) and 5 wt.% ZnFe2O4 dispersed PVC has highest number of peaks which were well matched with the diffraction peaks of the ZnFe2O4 NPs and it evident the formation of polymer nano composite. Figure 3(b) shows the peak broadening of the high intensity peak, in the 2θ range of 34 to 37°. From this figure we clearly observed that the increase in filler concentration increases the peak broadening. This is due the stronger interaction between the between the polymer chains and the ZnFe2O4 nanoparticles. (a) XRD pattern for prepared PVC/ZnFe2O4 film, (b) high intensity peak 2θ ranges from 34 to 37.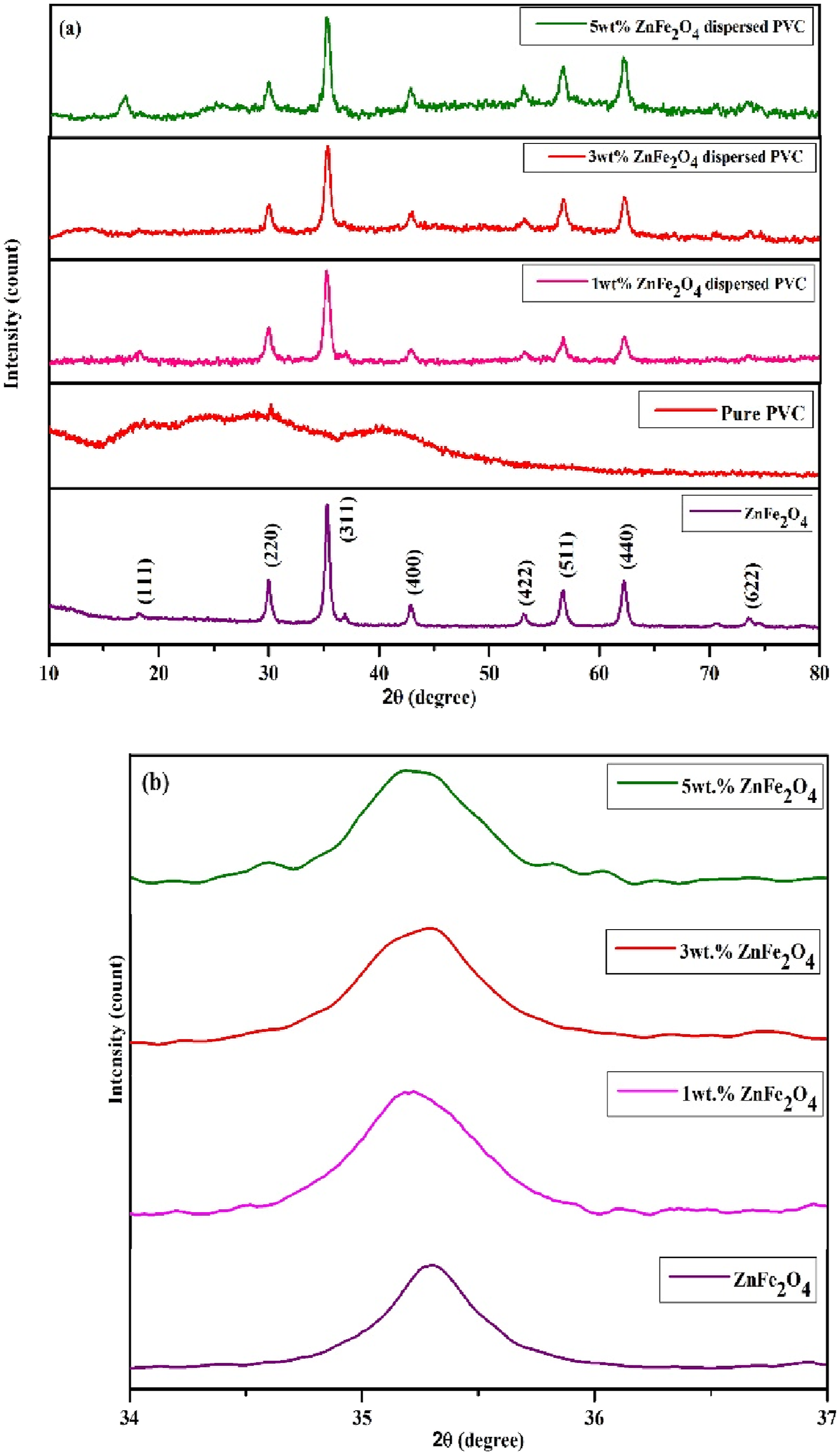
From Table 1, Using egg white as a fuel simplifies the process because it acts as a natural reducing agent and stabilizer, eliminating the need for further chemicals. Furthermore, its abundance, sustainability, and protein content enable efficient and ecologically friendly nanoparticle manufacturing, making it superior to alternative chemical approaches.
The overall XRD pattern in our study revealed no secondary peaks indicating the formation of impurity phases. The weight % of manufactured ferrite nanoparticles and the peak position are clearly correlated, as the weight percentage of dispersed nanoparticles in the PVC polymer matrix grows in tandem with the stipulated peak position attributed to the ferrite nanoparticles. This finding implies that adding the produced zinc ferrite nanoparticles in a higher weight% allows them to be more visible inside the polymer matrix, potentially improving their interaction with the PVC. This better connection could have a substantial influence on magnetic and electrical devices, among other applications.
Morphological analysis
AFM analysis
An AFM analysis was performed to determine the surface topography and roughness of manufactured PVC and 5 wt.% ZnFe2O4 dispersed PVC film. Figure 4(a) and (b) show the 2D and 3D topographic pictures of PVC and a 5 wt.% ZnFe2O4/PVC nanocomposite film. The root mean square (RMS) surface roughness values of pure PVC are 4.97 nm and 16.03 nm for a 5 wt.% ZnFe2O4/PVC film. The roughness is increased more when the additive concentration increases. The distribution of ZnFe2O4 nanoparticles within the PVC matrix plays a crucial role in modifying the surface characteristics. The AFM images suggest that the nanofillers are homogeneously distributed at the surface, forming a network that alters the surface morphology. The fine dispersion of ZnFe2O4 particles leads to an increase in surface irregularities, which is reflected in the enhanced roughness values.
40
The presence of nanoparticles creates protrusions and valleys on the surface, contributing to the overall higher surface roughness. AFM images of (a) pure PVC (b) PVC/5%wt.% ZnFe2O4.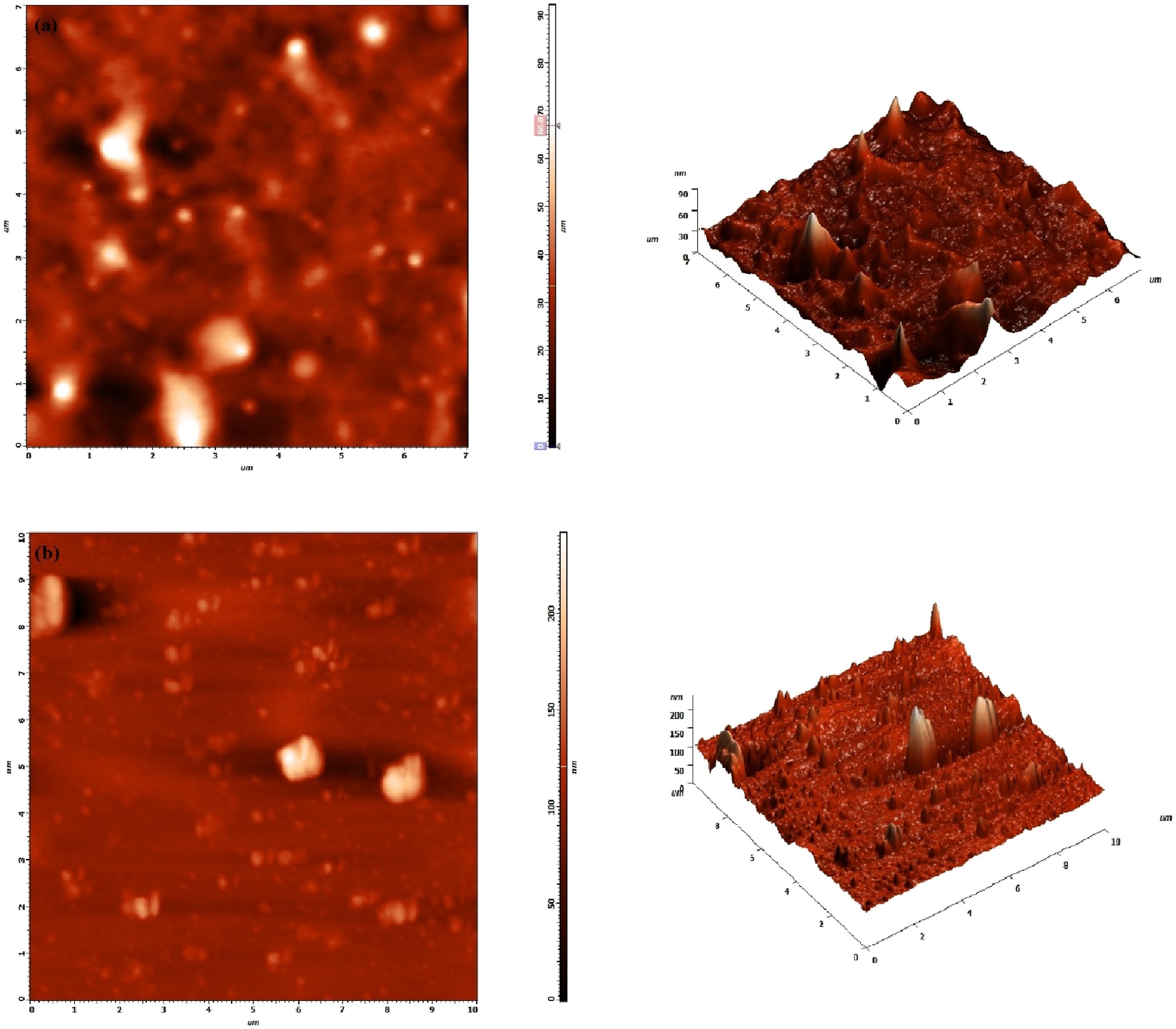
Additionally, the increased surface roughness can be attributed to the strong interfacial interactions between ZnFe2O4 and PVC. 26 The nanoparticles provide nucleation sites that influence the crystallization and phase morphology of the polymer. The interfacial contact between ZnFe2O4 and PVC promotes microstructural modifications, resulting in higher adhesion and better filler-matrix bonding. 40 This interfacial interaction can also affect other macroscopic properties such as wettability (contact angle), mechanical strength, and dielectric behavior.
SEM analysis of zinc ferrite (ZnFe2O4) nanoparticle
Figure 5 presents the standard morphology of the synthesized ZnFe2O4 NPs before dispersion. The SEM analysis reveals a narrow size distribution among the nanoparticles and it exhibit minimal agglomeration, predominantly forming regular-shaped grains with spherical morphology. Using log normal fit, histogram analysis confirms that the majority of particles fall within the size range of 20–40 nm. Sharon et al.
41
explored zinc-doped cobalt ferrite nanoparticles prepared via a modified sol-gel method. They discovered that the precursor plays a critical role in determining the size of the nanoparticles. From the particle size histogram and SEM micrographs, it is evident that the synthesized ZnFe2O4 exhibit minimal agglomeration and a homogeneous particle distribution. The narrow size range of particles in the histogram emphasizes the effectiveness of egg white as a precursor, demonstrating its capability to produce size-controlled ZnFe2O4 nanoparticles.
33
A controlled particle size distribution ensures consistency in key properties such as optical band gap, dielectric permittivity, and surface roughness, which directly impact the performance of the polymer nanocomposites. SEM of prepared ZnFe2O4 nanoparticle along with histogram.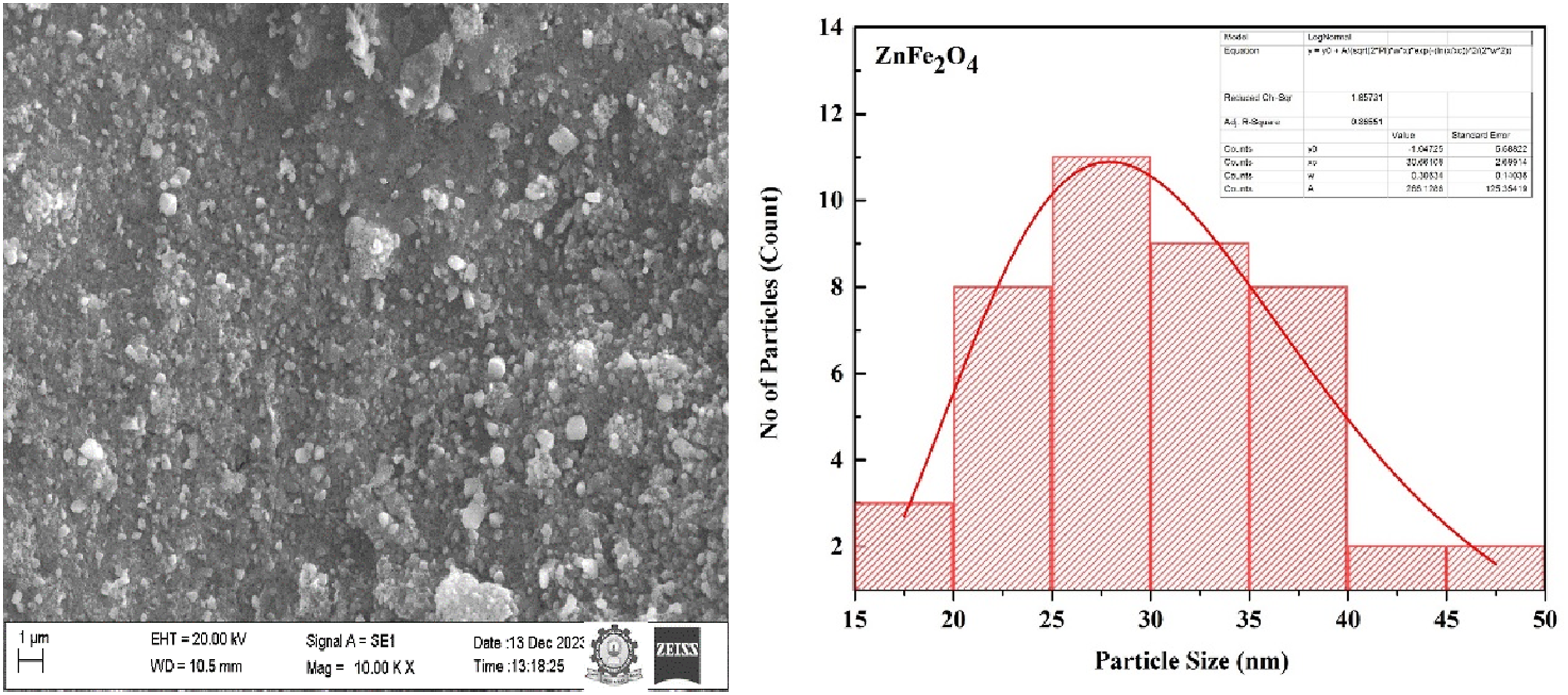
FESEM Analysis of bare and different wt.% ZnFe2O4 dispersed PVC
The morphology of pure PVC and 1, 3, 5 wt% ZnFe2O4 dispersed PVC film is shown in Figure 6(a)–(d), at varying magnifications. As per the obtained SEM figures pure PVC is entirely amorphous, it has no distinguishing features. Its surface is uniform, translucent, homogeneous, and smooth. This experimental result is completely consistent with Alhulw et al.’s findings.
42
Increasing the low concentration (1 wt.% and 3 wt.%) of ZnFe2O4 NPs in films leads to increasingly visible surface imperfections, as shown in the images. Surface imperfection in ferrite dispersed polymer films arise from ferrite particle agglomeration and polymer ferrite interfacial incompatibility.43,44 Especially, incorporating 5.0 wt.% ZnFe2O4 into the PVC film shows less agglomeration than the other two wt.%, which resulted in a continues network formation and considerable morphological change. This affects adhesion strength, particle dispersion, and ultimately the film’s characteristics. Similarly, in a study by ipsita and shrabanee
45
incorporated ZnFe2O4 nanoparticles into polyvinylidene fluoride (PVDF) films. The incorporation of these nanoparticles led to the formation of a continuous network structure, resulting in significant changes to the film’s morphology and characteristics. The interfacial interactions between ZnFe2O4 and PVC, including adhesion strength and particle dispersion, play a crucial role in determining the film’s final properties. Here the interfacial interaction influences key measured properties such as optical, thermal and wettability.
46
So, the dispersion of the green route prepared ZnFe2O4 nanofillers onto the surface of the polymeric matrix is improve their properties and the surface of filler-dispersed films is rougher than that of pure film. SEM photographs of (a) pure PVC; (b) PVC/1 wt.% ZnFe2O4; (c) PVC/3 wt.% ZnFe2O4; (d) PVC/5%wt.% ZnFe2O4.
UV-visible spectroscopy
The Ultraviolet-Visible-Near Infrared spectra of pure PVC and the different wt.% of ZnFe2O4 dispersed PVC film versus wavelength has been recorded and demonstrated in the Figure 7(a) and (b). PVC exhibits great transparency in visible and near-infrared regions. It is also obvious that PVC is highly transparent in the ultraviolet band (200–400 nm). Furthermore, the finding in Figure 7(a) showed that increasing the quantity of ZnFe2O4 nanoparticles in the composite films significantly decreased transmittance in this area. The decline is due to enhanced photon scattering from denser nanoparticles in the polymer mix. This behavior can be explained in terms of ZnFe2O4’s UV spectrum filtering property, which is supported by the absorption graphs of produced films. As seen in Figure 7(b), increasing the additive amount of nanoparticle ratio (PVC-ZnFe2O4) in the casting solution improves film absorption in the ultraviolet range. Higher ZnFe2O4 concentrations in PVC films improve UV absorbance by enhancing light scattering and interactions between the nanofiller and the polymer matrix. As the concentration of ZnFe2O4 increases, more aggregates form, leading to enhanced light absorption at longer wavelengths. (a) UV transmittance spectrum, (b) absorption spectrum of pure PVC and different wt.% ZnFe2O4 dispersed PVC.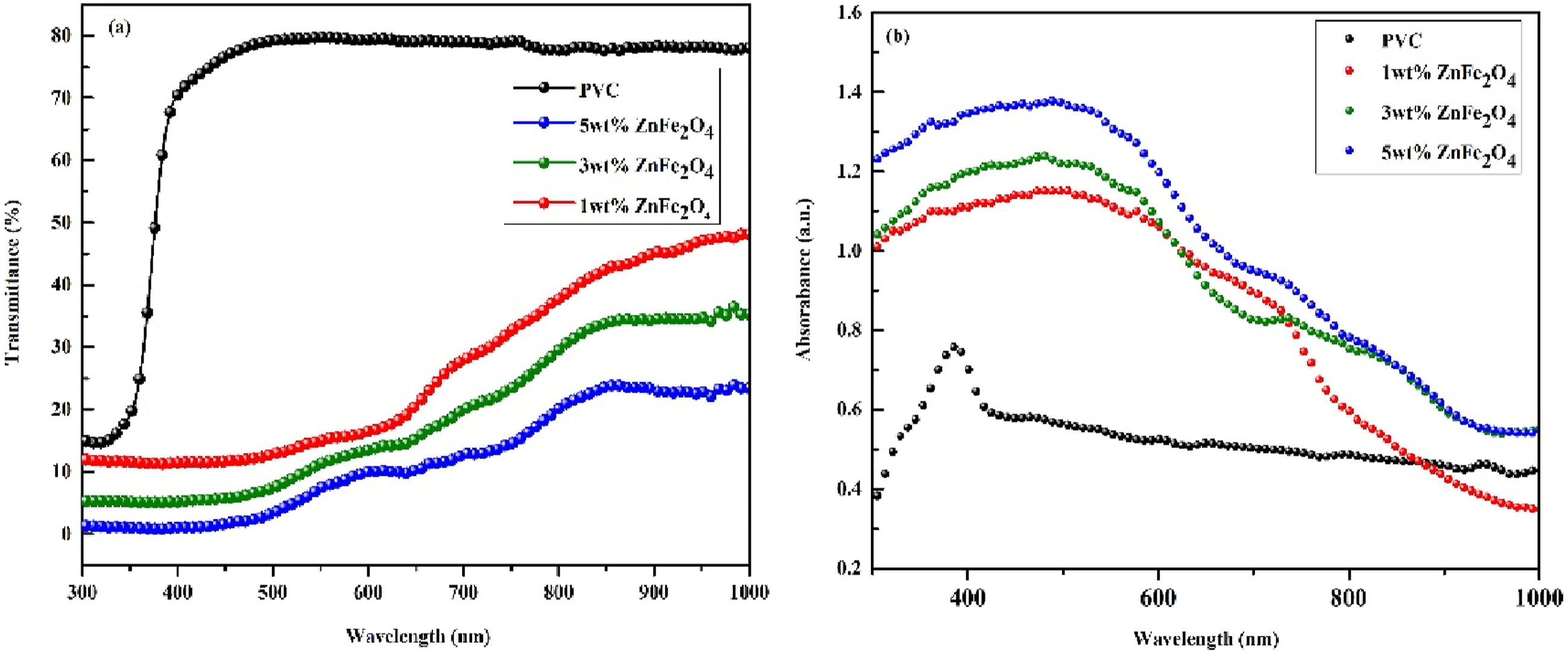
Figure 8(a) shows a graph of absorption coefficient (a) versus various wavelength, using Beer-lamberts Law,
47
(a) Absorption coefficient (b) Extinction coefficient of Pure PVC and different wt.% ZnFe2O4 dispersed PVC.
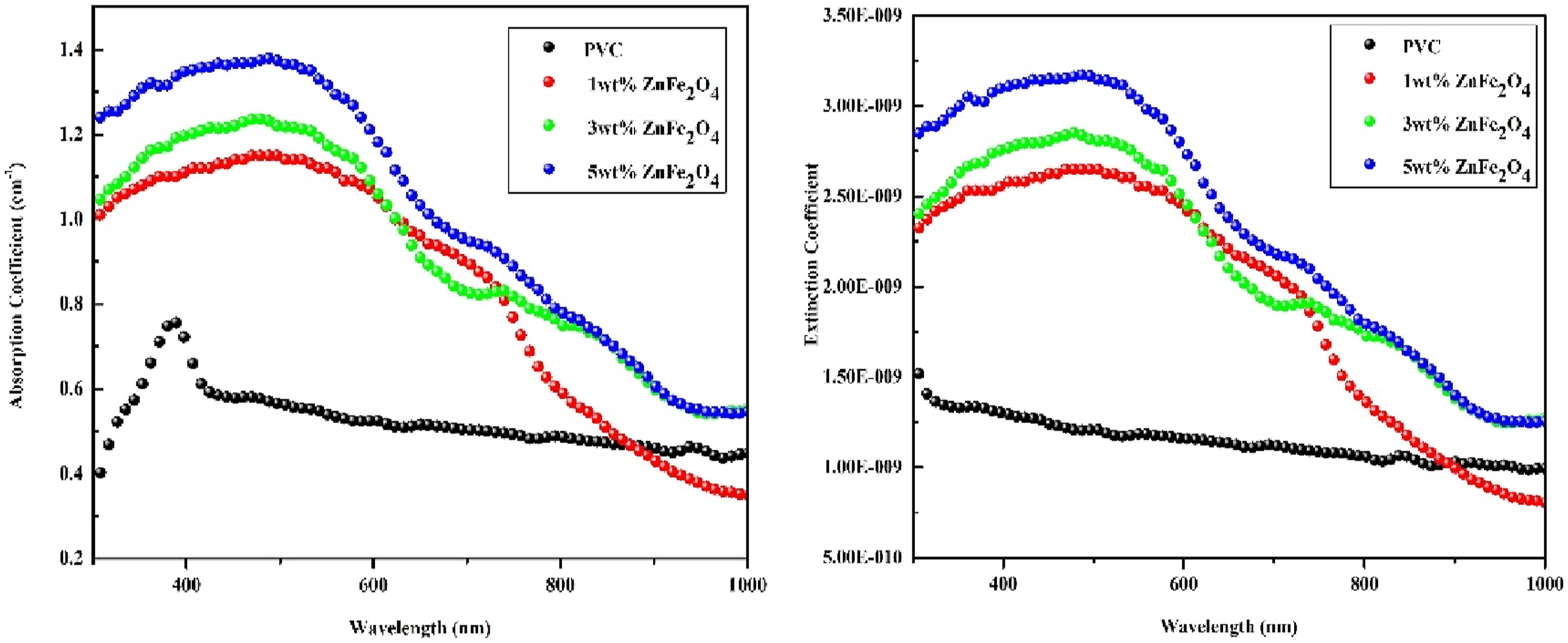
Figure 8(a) shows that utilizing ZnFe2O4 nanoparticles increases absorption, which is further enhanced by increasing the filler additive content. The contact between the PVC matrix and zinc ferrite nanoparticles can generate localized electric fields, altering the material’s absorption properties. These interfacial effects can also introduce defect states or localized states in the band structure, contributing to increased absorption and rendering the material more efficient in applications requiring enhanced light absorption, such as sensors, photodetectors, or photocatalytic systems. The absorption spectrum is connected to the extinction coefficient (k) and it is used to measure of how strongly a chemical substance absorb light at particular wavelength. Figure 8(b) shows the Extinction coefficient plot, which may be computed using the formula
48

Figure 8(b) depicts the steady increase in K as 1%, 3%, and 5% ZnFe2O4 NPs are introduced to the PVC polymer thin films. Scattering and absorption events in the PVC/ZnFe2O4 nanocomposite lose a large quantity of light energy, and the high weight % of ZnFe2O4 NPs increases the extinction coefficient. High extinction coefficients in polymer nanocomposite films are critical for optimizing the functioning of optoelectronic devices such as photographic detectors and LEDs because they allow for effective light absorption and emission. 49
The optical energy band gap of pure PVC and varying wt.% of ZnFe2O4 distributed PVC was analyzed using Davis and Mott’s equation, which provides a connection with incoming photon energy (hʋ) and absorption coefficient (α) Using the Tauc plot equation.
50
Tauc plot for pure PVC and different wt.% ZnFe2O4 dispersed PVC as a function of photon energy. Optical band gap of bare and nanoparticles dispersed PVC films.
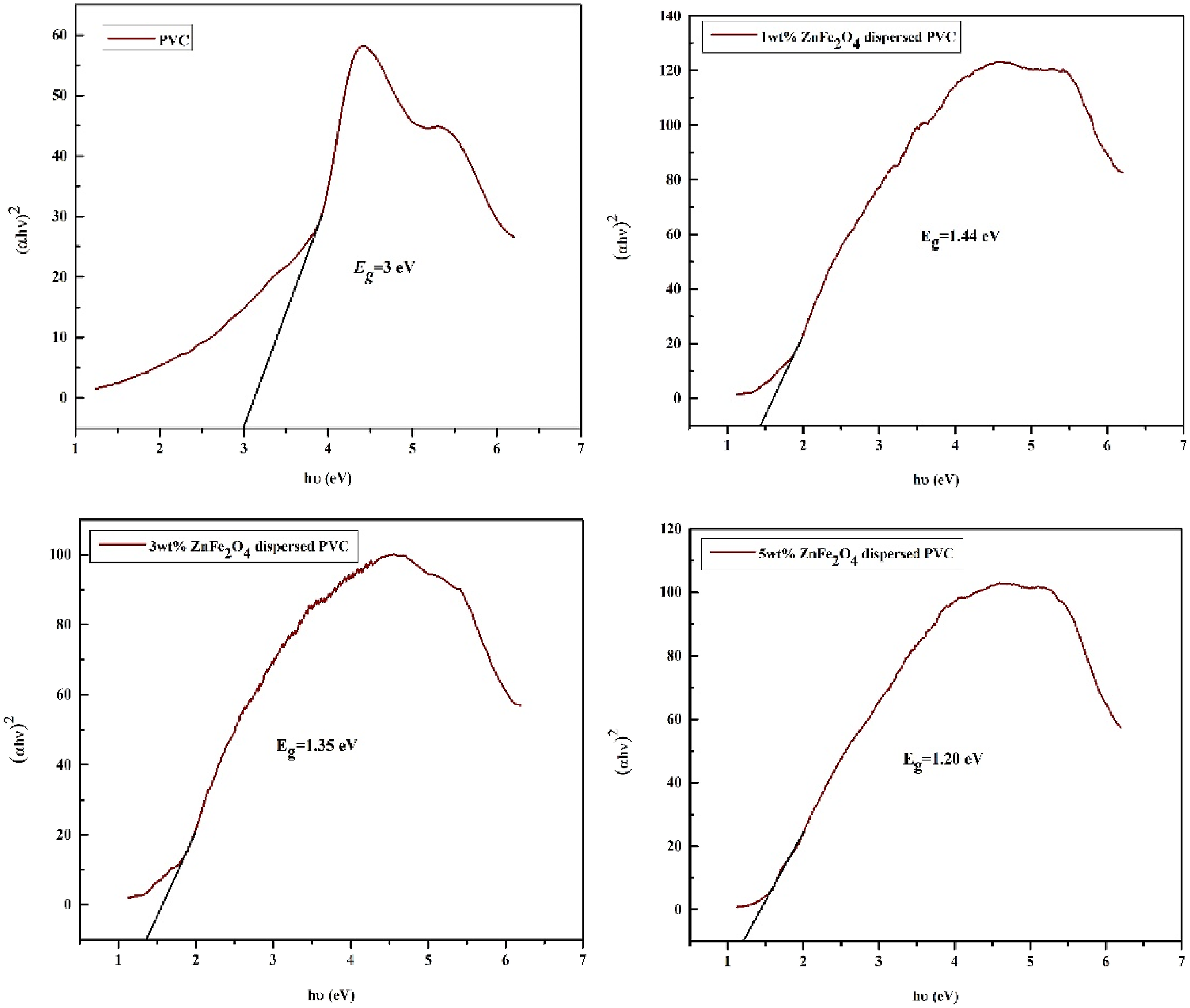

From this, we find that altering the nanoparticle proportions in the PVC matrix allows us to modify the band gap of dispersed PVC films. In recent times, customizable small band gap nanomaterials with well-suited band edges have become more prevalent, notably in the field of optoelectronics applications.55,56
Photoluminescence
Photoluminescence (PL) spectroscopy is a valuable tool for analyzing charge carrier recombination and defect states in semiconductor materials.
57
Figure 10 presents the PL spectra of pure and ZnFe2O4 dispersed PVC composites at room temperature. A noticeable decrease in PL intensity is observed with increasing ZnFe2O4 content, indicating enhanced charge carrier separation. In pure PVC, the higher PL intensity suggests fewer recombination centers, allowing for significant radiative recombination. However, when ZnFe2O4 nanoparticles are incorporated, they act as charge trapping sites, reducing radiative recombination and promoting charge carrier separation.
58
This behavior is consistent with the role of ZnFe2O4 as a semiconducting filler, enhancing electrical conductivity by minimizing electron-hole recombination.
59
The reduction in PL intensity with increasing ZnFe2O4 content suggests enhanced charge carrier trapping and reduced radiative recombination. This behavior is indicative of the formation of new defect states or energy levels within the band structure. These defect states contribute to the observed reduction in optical band gap, as confirmed by the UV-Vis Tauc plot analysis.
60
The synergy between PL quenching and band gap narrowing reinforces the role of ZnFe2O4 nanoparticles in modifying the electronic structure of the PVC matrix, making the nanocomposite more suitable for optoelectronic applications. Photoluminescence spectra of prepared samples.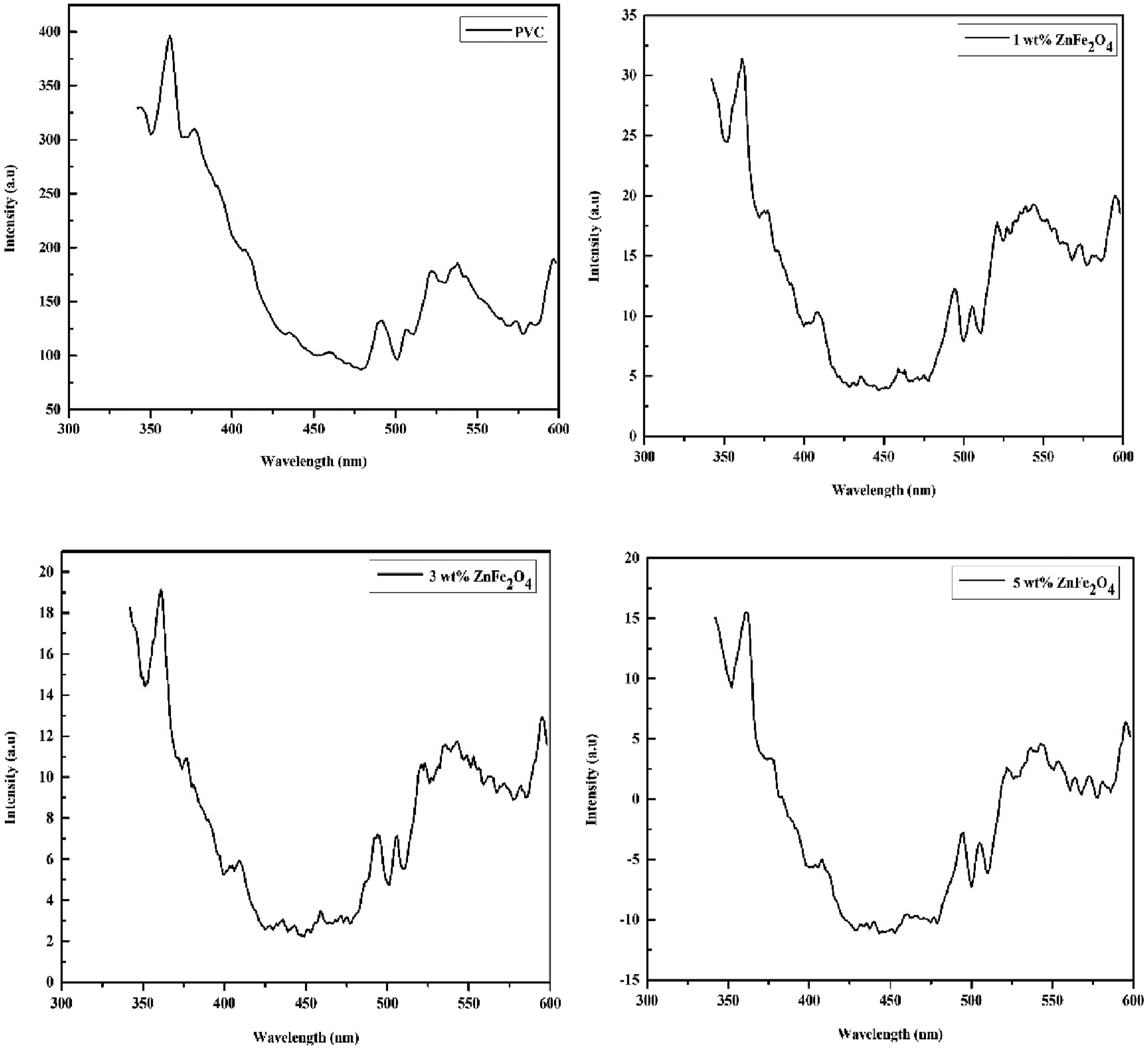
Contact angle measurement
The hydrophobic property of PVC and its composites is examined utilizing the water contact angle method, which measures the number of water droplets that may be dispersed over a surface.
61
Figure 11 shows that the contact angle increased from 79.4° (hydrophilic) to 107.6°(hydrophobic) and 78.1° as the concentration of ZnFe2O4 increased from 0 to 1 wt.%. The reduction in contact angle is caused by the communication of ZnFe2O4 nanoparticles with water molecules. The number of nanoparticles on the surface of the PVC disc rises in proportion to the quantity of ZnFe2O4, resulting in enhanced hydrophobicity. Increasing ZnFe2O4 concentration to 3 and 5 wt.% results in a modest rise in contact angle from 78.1° to 88.9° and 107.6°. The distribution of zinc ferrite nanoparticles is mostly due to the interaction of surface roughness and hydrophilicity changes, which usually promotes the Cassie-Baxter wetting state. This state happens when water droplets rest on the peaks of rough surfaces, limiting the soaking area and increasing the contact angle.
62
The suggested surface modified films were employed in waterproof coatings in electronics, construction and medical application. Photographs of contact angle for bare PVC and different wt.% ZnFe2O4 dispersed PVC.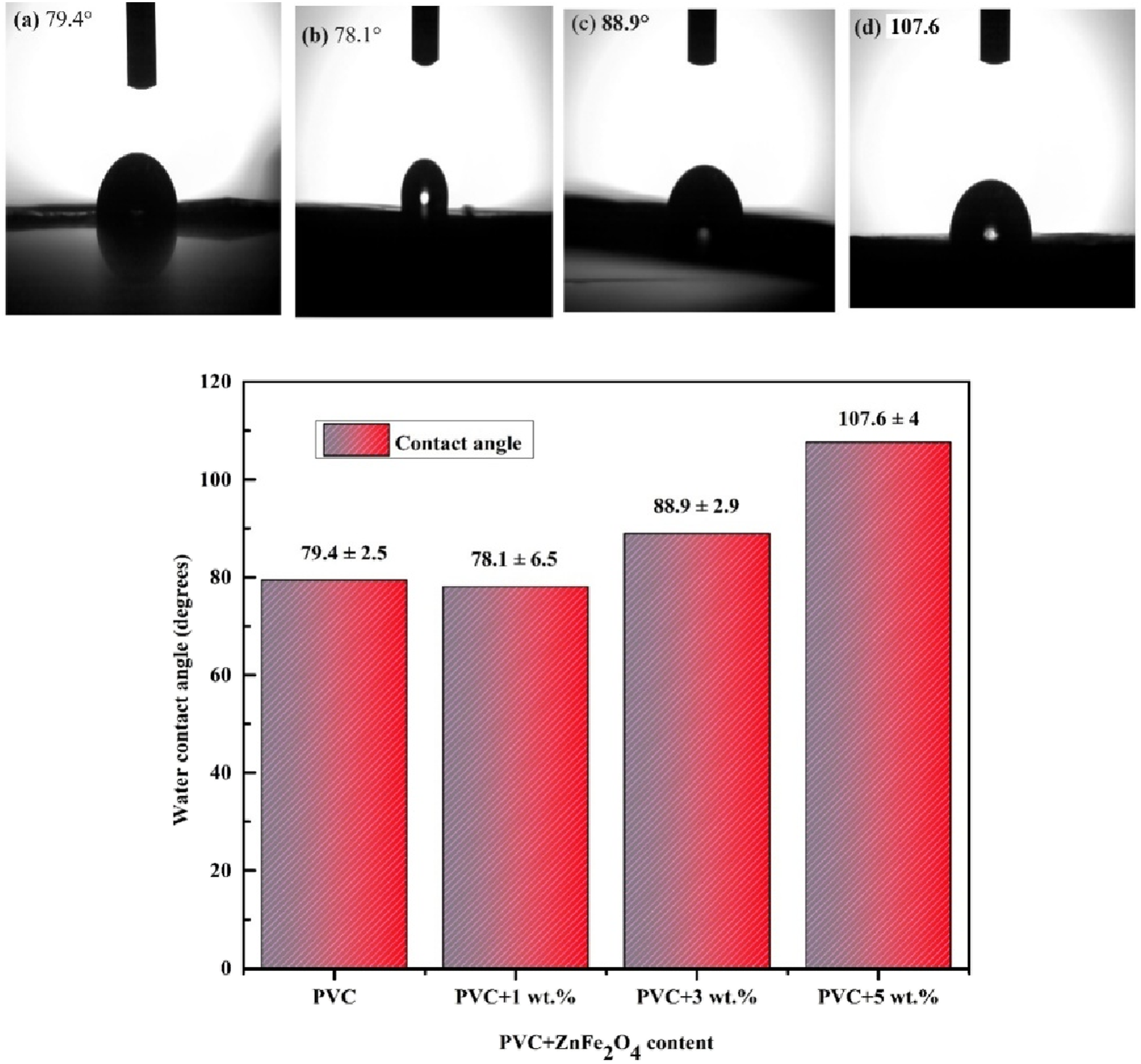
Thermogravimetric analysis
Thermogravimetric analysis (TGA) was conducted on pure PVC and PVC/ZnFe2O4 nanocomposites containing 5 wt.% zinc ferrite NPs in a nitrogen atmosphere to examine their thermal behavior is shown in the Figure 12. PVC film represent two thermal degradation stages which was well consistent to the one by Hasan and Lee
52
and Sadek et al.
63
The first stage began at 140°C and gradually decreased to 355°C, resulting in a net weight loss of roughly 77.9%. This pattern shows the removal of hydrogen chloride (HCl) from the PVC structure (dehydrohalogenation).
64
The second degradation stage observed between 406°C and 509°C, resulting in a weight loss of around 17.8%. This is due to the condensation of unsaturation (C=C) and breakage of C-C, C-H, and C-Cl caused by the elimination of HCl during the first step. On the other hand, 5 wt.% ZnFe2O4 nanocomposites show lower initial degradation temperatures than pure PVC. Weight loss in composites below 100°C was caused by the evaporation of THF solvent and contaminants absorbed by zinc ferrite nanoparticles. The diminution may be related to the catalytic properties of ZnFe2O4 nanoparticles,52,65 which expedite the dehydrochlorination of PVC chains. From 310 to 410°C, no weight loss was recorded, showing that the nanocomposite films are stable at this temperature. The PVC/ZnFe2O4 nanocomposite demonstrated greater stability compared to pure PVC, likely due to improved interfacial relations between the dispersed ZnFe2O4 and PVC. The second significant loss occurred between 410 and 490°C, which was significantly lesser than the preceding step. This is because of, thermal breakdown of the polyene backbone caused the synthesis of volatile aromatic chemicals and an inert carbonaceous residue.
66
Additionally, Figure 12 shows that nanocomposite films have higher residuals than pure PVC. Adding zinc ferrite nanoparticles to PVC improves char yield and carbonization of the polymer. Primary thermograms of PVC and 5 wt.% PVC/ZnFe2O4 film.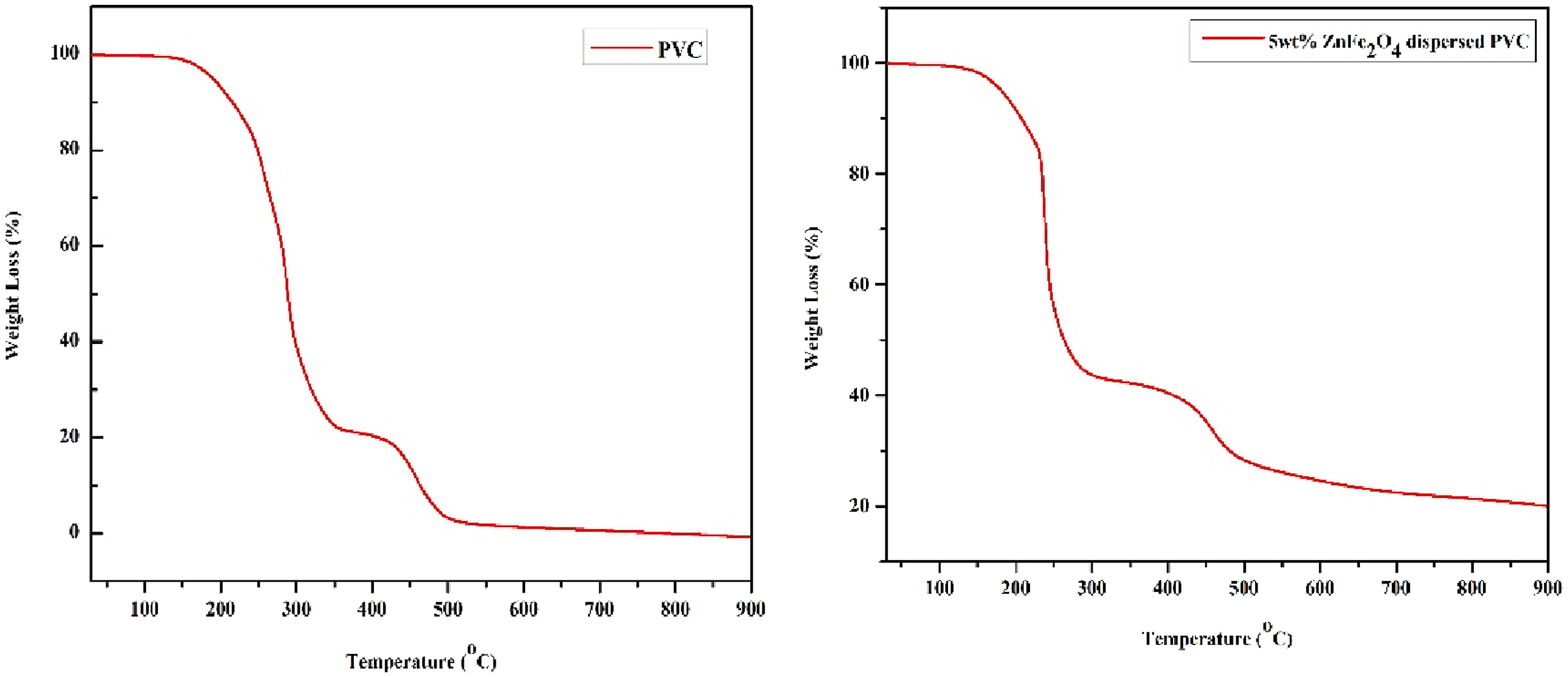

Thermogravimetric data for bare PVC 5 wt.% ZnFe2O4 dispersed films.

The values from the Table 3 indicates that utilizing ZnFe2O4 nanoparticle into the matrix of PVC enhance the char yield and LOI of the polymer. Normally the material which have high LOI has low flammability, in this case the value of LOI for 5 wt.% ZnFe2O4 dispersed PVC was high when compared with pure PVC. The obtained LOI value is higher than the PVC/ZnO nanocomposites reported by the author Zarrinkhameh et al. 68 This improvement is attributed to the ZnFe2O4 nanoparticles forming a protective barrier within the polymer matrix, which effectively impedes the permeation of oxygen and volatile combustible gases that are essential for the combustion process. By disrupting the pathways for thermal degradation and gas diffusion, the nanocomposite film exhibits enhanced flame-retardant properties. 69
The derivative of thermogravimetric analysis of pristine and 5 wt.% ZnFe2O4 dispersed PVC was displayed in Figure 13. At specific temperatures, two notable peaks indicate the highest rate of weight loss deterioration and the maximum temperature for weight loss in PVC and 5 wt.% ZnFe2O4 was 285 and 240°C, respectively. The availability of ZnFe2O4 nanoparticles in the produced film resulting in a lower initial step breakdown temperature for the 5 wt.% sample than for bare PVC. In the second stage of degradation, both samples show nearly identical values. Overall, ZnFe2O4 nanoparticles accelerate the disintegration of PVC while slowing its weight loss. This suggests that the produced film could be beneficial in applications requiring controlled degradation. This study investigates the role of ZnFe2O4 as an environmentally safe and cost-effective alternative to existing flame-retardant additives such as halogen-based chemicals, which pose environmental risks. Derivative thermograms of PVC and 5 wt.% PVC/ZnFe2O4 film.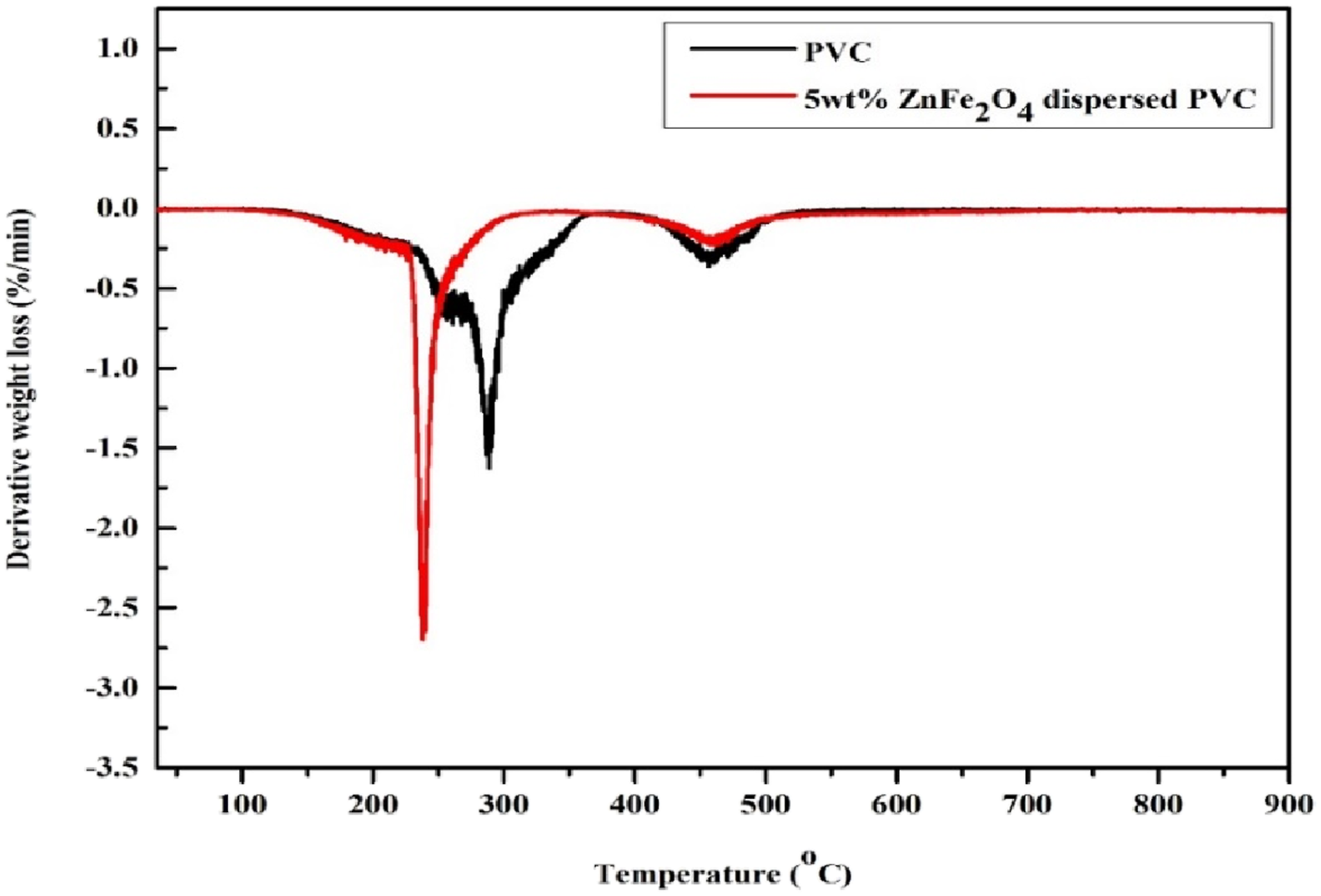
Conclusions
The successful integration of ZnFe2O4 nanofillers into the PVC matrix was achieved using a single-step green route strategy, employing the solution casting technique to fabricate polymeric composite thin films. The structural analysis via XRD confirmed that the virgin PVC film exhibited an amorphous nature, while the incorporation of ZnFe2O4 nanoparticles induced polycrystallinity in the composite films. FESEM analysis further revealed that the nanoparticles were uniformly distributed on the surface of the PVC film. Optical studies demonstrated that virgin PVC films exhibited high transparency in the UV-VIS region. However, the addition of ZnFe2O4 nanoparticles enhanced UV absorption and reduced the optical band gap, with a more pronounced effect at higher filler concentrations. This tunable optical property suggests the potential application of the composite films in optoelectronic devices, particularly in flexible display technologies. Photoluminescence analysis indicated a reduction in electron-hole recombination with increasing nanoparticle concentration, which could enhance the semiconducting efficiency of the nanocomposite films. Moreover, contact angle measurements revealed a transition from hydrophilic to hydrophobic behavior with higher ZnFe2O4 content, suggesting improved surface wettability for specific applications. The different weight percentages (1, 3, and 5 wt.%) of ZnFe2O4 nanoparticles incorporated in this study effectively enhanced the physical and chemical properties of the PVC polymer, contributing to its improved performance. Thermal analysis (TGA) confirmed that the incorporation of ZnFe2O4 nanoparticles influenced the thermal stability of PVC composites by modifying the degradation behavior and reducing weight loss. This enhancement in thermal properties, along with the observed optical and structural improvements, suggests that the prepared PVC/ZnFe2O4 nanocomposites hold promise for applications in optoelectronic devices and flame-retardant materials.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is funded by the ongoing Research funding program (ORF-2025-117), King Saud University, Riyadh, Saudi Arabia.
Correction (June 2025):
The Funding section in the article has been updated since its original publication.
