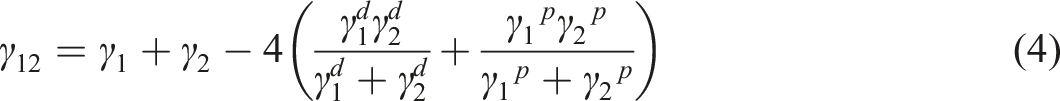Abstract
Low molecular weight oligomer polybutylene terephthalate (PBT) with carboxyl terminated groups was synthesized via dry esterification method, and then was used to esterify the surface of cellulose. The optimal esterification reaction conditions were identified as a PBT-to-cellulose mass ratio of 1:5, an esterification time of 6 h, and a catalyst dosage of 2% of the cellulose mass. The esterified cellulose was blended with PBAT to prepare composite materials, and the effects of varying amounts of esterified cellulose on the mechanical properties, thermal properties, and two-phase compatibility of the composites were explored. The performance of PBAT/esterified cellulose composites was then compared with that of PBAT/cellulose composites. The results indicated that the compatibility of cellulose with PBAT and its dispersion in PBAT were significantly improved after PBT esterification. When the ratio of PBAT/esterified cellulose was 95/5, the tensile strength, elongation at break and tensile modulus of the composite material reached 21.89 MPa, 593.82% and 86.3 MPa, respectively.
Introduction
Plastic is one of the greatest inventions of the 20th century, bringing unprecedented convenience to human work and life, and has rapidly developed into a major material category. 1 However, traditional plastic products are difficult to be decomposed in the natural environment, and the disposal methods such as burial and incineration will destroy the soil structure and produce poisonous and even carcinogenic substances. Moreover, it is difficult for the plastic products to recycle. The resulting “white pollution” is increasingly causing severe and unprecedented harm to the ecological environment.2,3 Degradable plastics refer to a category of materials which can meet all performance requirements for utilization and remain stable throughout their service period. While after disposal, they can degrade under natural conditions into harmless substances which can be consumed by the environment. 4 Research and development of degradable plastics has become an important direction of sustainable development, among which biodegradable materials are the research hotspot. Biodegradable materials are widely regarded internationally as polymers that, undergo changes in their physical and chemical properties through the action of enzymes or microorganisms under aerobic or anaerobic conditions, ultimately breaking down into H2O, CO2, CH4, or low-molecular-weight polymers. The final decomposition products must meet environmental standards for non-toxicity. 5 The development of biodegradable materials is of strategic significance under the circumstance of “carbon peaking and carbon neutrality goals”.
Poly (butylene adipate terephthalate) (PBAT) is an aromatic-aliphatic biodegradable polyester with excellent extensibility and heat resistance. 6 However, the degradation rate of PBAT is relatively slow, and the application is limited by the high cost and poor mechanical properties compared with traditional polymer materials. 7 Therefore, a series of modification methods to PBAT are used to broaden its application range, among which, blending modification is the most commonly used. 8 Choo et al. 9 used epoxidized soybean oil as a compatibilizer to prepare PBAT/PLA/PPC multiphase blends via melt processing technique. The results showed that the functional groups of PPC effectively improved the interaction between carboxyl and hydroxyl groups in the PLA/PBAT binary blends, thereby enhancing the mechanical and physical properties of the multiphase blend system. Kong et al. 10 added varying amounts of CaSO4 or CaSO4 surface-treated with stearic acid into the composite materials of PBAT and PLA. The results showed that the modified CaSO4 was uniformly dispersed within the polymer substrate, leading to the improvement of the tensile strength and elongation at break, and the tensile strength reached the maximum when 5 wt% of modified CaSO4 was added. Wang et al. 11 prepared PBAT/TPS film by using maleic anhydride (MA) and titanate coupling agent (TC) as additives. The results indicated that the addition of MA and TC improved the dispersion of TPS in PBAT and enhanced interfacial compatibility, thereby increasing the aging resistance and mechanical performance of the films.
Cellulose is the most abundant natural polymer material in nature, and is an ideal biomass filler with the advantages of low density, low cost, high toughness and biodegradability.12,13 However, cellulose is a hydrophilic polyhydroxy compound, so a phenomenon of phase separation will be caused when it is directly mixed with hydrophobic polyester. 14 Therefore, cellulose must be modified before blending with PBAT. Morelli et al. 15 modified cellulose nanocrystals (CNC) with phenylbutyl isocyanate and prepared composite materials by blending modified CNC with PBAT through melt extrusion. The results showed that after the addition of the esterified CNC, the heat-resistance temperature of the composite material was increased by 20°C, and the water vapor permeability was also significantly reduced.
Currently, the esterification of cellulose is primarily carried out in solvents, and the solvents or plasticizers are also required for the preparation of PBAT/esterified cellulose composite, to achieve good two-phase compatibility and prevent any negative impact on the composite’s performance.16,17 This process is not only complicated and difficult for industrial production, but also brings significantly environmental pollution concerns. In this work, low molecular weight oligomer, polybutylene terephthalate (PBT), with carboxyl terminated groups is synthesized and employed to esterify the surface of cellulose as an esterifying agent by a dry method. The esterified cellulose is then blended with PBAT to prepare PBAT/esterified cellulose composites. The entire process is solvent-free and environmentally friendly, which is consistent with the global “green and low-carbon” development goals. Additionally, since PBT is structurally similar to PBAT, the PBT chains grafting to the cellulose surface can entangle with PBAT chains during the preparation of composite materials, thus improving the compatibility of the two phases of PBAT and cellulose. 18 As a result, high-performance composite materials can be obtained without any solvents or additives. This work explored the optimal esterification reaction conditions for PBT-esterified cellulose and prepared PBAT/esterified cellulose composites in various ratios of PBAT and esterified cellulose, and the effects of which on the two-phase compatibility, thermal properties, rheological behavior, and mechanical properties of the composites were further investigated.
Materials and Methods
Materials
Terephthalic acid (PTA), 1,4-Butanediol (BDO), Tetrabutyl titanate, N,N'-carbonyldiimidazole: 99% purity, Shanghai Macklin Biochemical Technology Co. Ltd, Shanghai, China. Cellulose powder: 180-280 μm, Shanghai Macklin Biochemical Technology Co. Ltd, Shanghai, China. Poly (butylene adipate terephthalate) (PBAT): Liaoning Dongsheng Plastic Industry Co. Ltd, Shenyang, China.
Synthesis of Low Molecular Weight Carboxyl-Terminated PBT
1,4-Butanediol (BDO) and terephthalic acid (PTA) were added into a three-neck flask in a molar ratio of 1:1.4, and tetrabutyl titanate as a catalyst with a mass of 5‰ butanediol was added. The mixture was stirred at 200°C for 4h to obtain low molecular weight carboxyl-terminated PBT, which was then placed in a desiccator for later use.
19
The synthesis mechanism of PBT is shown in Figure 1. Diagram of the PBT synthesis mechanism.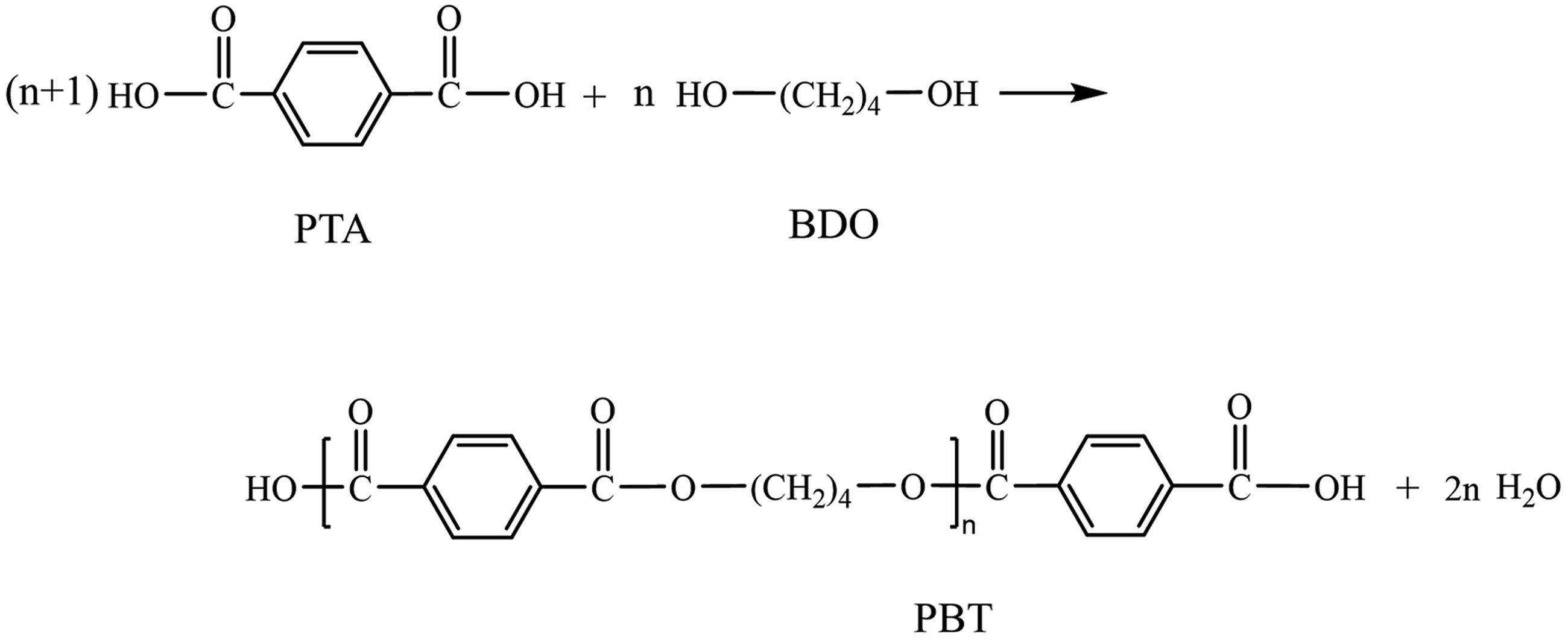
Preparation of PBT-Esterified Cellulose
The prepared low molecular weight carboxyl-terminated PBT as esterifying agent and cellulose were mixed in mass ratios of 1:3, 1:4, 1:5, 1:6, and 1:7 in a three-neck flask respectively, and stirred at 120°C. Catalyst N,N'-carbonyldiimidazole (1%, 2%, 3%, 4%, 5% of cellulose mass, respectively) was added, and the mixture was continuously stirred for 3, 4, 5, 6, and 7h. Then the mixture was naturally cooled to room temperature, filtered and washed several times with anhydrous ethanol until the filtrate was clear and neutral pH. The filter cake was vacuum dried at 60°C to yield PBT-esterified cellulose. The reaction mechanism of PBT-esterified cellulose is shown in Figure 2. Diagram of the reaction mechanism for PBT-esterified cellulose.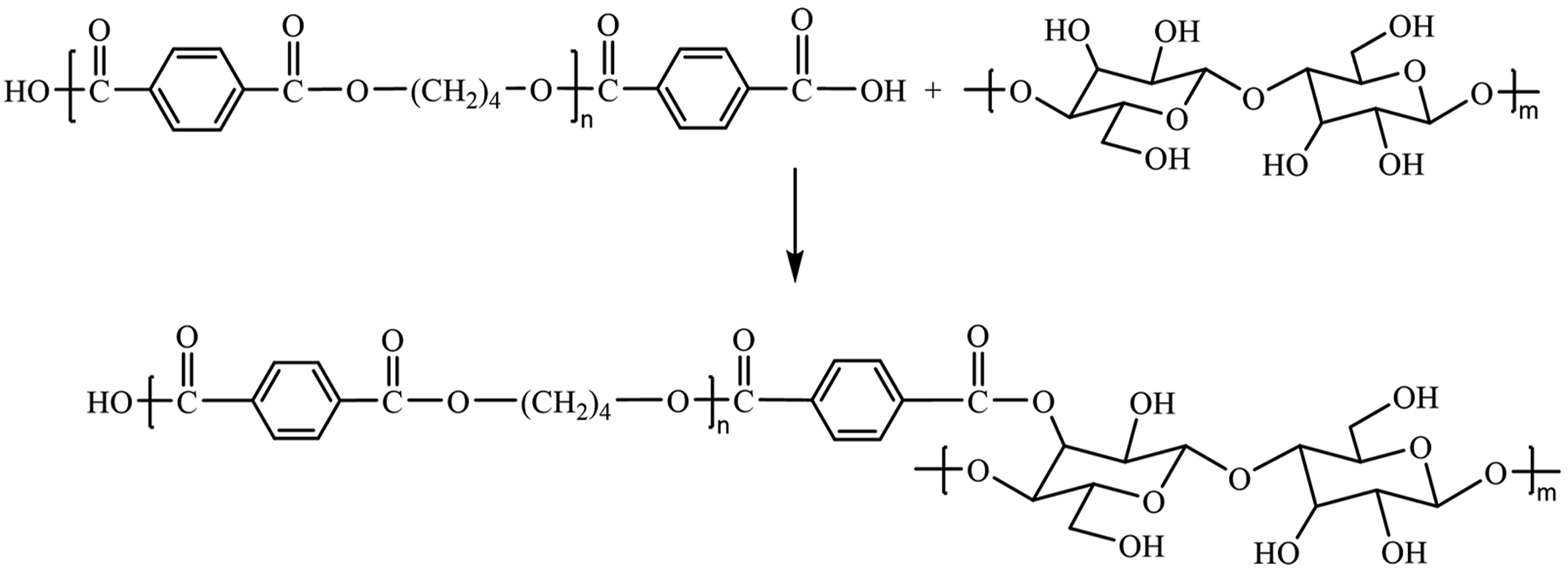
Preparation of PBAT/Cellulose Composites
PBAT, along with dried esterified cellulose or unesterified cellulose, was mixed with the ratios of 95/5, 90/10, 85/15, and 80/20, respectively, by using a torque rheometer with a temperature of 140°C and a rotational speed of 60 rad/s. The former composites were named as ECM-5, ECM-10, ECM-15 and ECM-20, and the latter were named as CM-5, CM-10, CM-15 and CM-20. The mixture was then cut into small pieces and fed into an injection molding machine with a barrel temperature of 170°C and a holding time of 12s, and then the standard test specimens were produced. 20
Compatibility Enhancement Mechanism
The compatibility enhancement mechanism of PBAT/PBT-esterified cellulose composites is illustrated in Figure 3. In the system of PBAT/unesterified cellulose composite, the compatibility between cellulose and PBAT is poor, which makes it difficult for the cellulose to be uniformly distributed within the PBAT and leads to obvious agglomeration phenomenon.
21
While in the system of PBAT/esterified cellulose composite, the cellulose is esterified with PBT, so the cellulose surface is coated with PBT. PBT is structurally similar and compatible with PBAT and can undergo chain entanglement with PBAT, so as to improve the dispersion uniformity of esterified cellulose in PBAT,
22
thus, the properties of composite materials can be further improved. Diagram of the compatibility enhancement mechanism.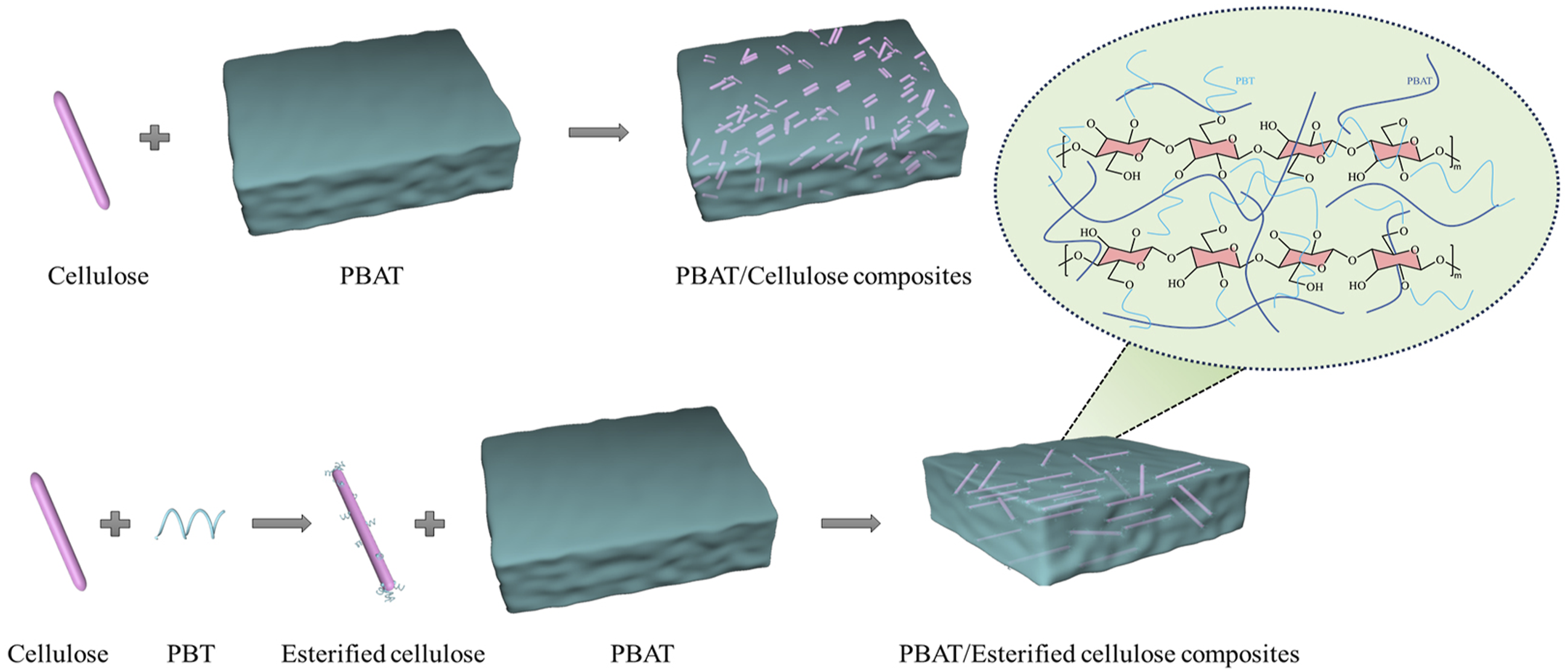
Characterization Methods
(1) The determination and calculation of molecular weight: 2g of the synthesized PBT was dissolved in a mixture of 20 mL phenol and 30 mL toluene. Meanwhile, a blank control group with 20 mL phenol and 30 mL toluene was prepared as the control. The above systems were kept at 180°C for 30min and cooled to room temperature. Five to seven drops of bromophenol blue were added as an indicator. The above system was titrated with a prepared 0.025 mol/L standard KOH solution, and the volume of KOH solution consumed at the titration endpoint was accurately recorded.
The molecular weight was calculated according to the following formula:
The molecular weight of the synthesized PBT, as determined by measurement and calculation, was 4800. (2) FT-IR: The structure of the samples was characterized by a Fourier transform infrared spectrometer (FT-IR) (Nicolet iS10, Thermo Fisher Scientific, Massachusetts, USA). 1.5 mg of the sample was ground evenly with 150 mg of potassium bromide and pressed into thin slices. The scanning range was 4000-500 cm−1. (3) DSC: The thermal performance of the samples was measured by a differential scanning calorimeter (DSC) (Q-200, TA Instruments, Delaware, USA). The measurement specimens were 5-8 mg in a crucible. The test was carried out under a nitrogen atmosphere with a temperature range of −40°C to 200°C and a heating or cooling rate of 10°C/min. (4) TGA: Thermogravimetric analysis (TGA) of the composite materials was performed using a thermal analyzer (STA 449C, Netzsch Instruments, Bayern, Germany). The sample was cut into small pieces of approximately 5 mg and heated from room temperature to 800°C at a rate of 10°C/min under a nitrogen atmosphere. (5) Vicat softening temperature (VST): The VST of the samples was tested using a vicat softening point tester (XRW-300, Gotech Testing Machines Co. Ltd, Taiwan, China). The measurement was conducted in a silicone oil bath with a heating rate of 2°C/min. A constant load of 10N was applied on the rectangular sample by a flat-ended needle with a cross-sectional area of 1 mm2, and the VST was determined when the needle penetrated the sample to a depth of 1 mm. (6) Rotational rheology: A rotational rheometer (DHR-2, TA Instruments, Delaware, USA) was used to test the rheological behavior under a nitrogen atmosphere, with a fixed strain of 0.5%, a constant temperature of 170°C, and a shear rate range of 0.1-100 rad/s. (7) Melt flow index (MI): The melt flow index of the composite materials was measured using a melt flow rate tester (MTM1000, Shenzhen Xinsansi Material Testing Co. Ltd, Shenzhen, China) with a temperature of 190°C, a load of 2.16 kg and an die inner diameter of 2.095 mm. (8) Mechanical properties: Tensile properties were tested using a universal electronic testing machine (DF-101S, Gotech Testing Machines Co. Ltd, Taiwan, China) with a testing speed of 50 mm/min. The dimensions of the dumbbell-shaped specimen are 75 mm (length) × 4 mm (width) × 2 mm (thickness). The testing speed has a significant influence on the tensile properties of the materials. Generally, as the stretching rate increases, the material’s tensile strength rises, while its elongation at break decreases. However, at lower testing speeds, the tensile strength and elongation at break exhibit greater sensitivity to the testing speed and can amplify the performance differences. Therefore, a testing speed of 20 mm/min was chosen to accentuate these performance variations when optimizing the reaction conditions. Notched impact strength was tested using an impact testing machine (GT-7045-MDL, Gotech Testing Machines Co. Ltd, Taiwan, China) with a pendulum energy of 5.5 J. (9) SEM: The surface morphology of the powder sample and the cross section morphology of the composite were observed by scanning electron microscope (SEM) (SU8010, Hitachi, Tokyo, Japan) with an accelerated voltage of 5 kV. The powder sample is glued to the surface of the conductive adhesive and sprayed with gold directly. Impact test specimens were brittle after being immersed in liquid nitrogen for 10 minutes, and then the cross section was treated with gold spraying. (10) Contact angle measurement.
The static contact angle (θ) of the samples relative to water and diiodomethane (1 μL) was measured by the contact angle tester (SINDIN SDC100, Chongqing, China), and the surface energy and interfacial tension were calculated using Wu’s equation.
Results and Discussion
In order to obtain PBT-esterified cellulose that can be well mixed with PBAT, the optimal reaction conditions for PBT-esterified cellulose were explored based on the mechanical properties of the composite material. In addition, in order to better compare the properties of different materials, a testing speed of 20 mm/min was selected to amplify the performance difference during the tensile performance test.
Determination of the Optimal Esterification Conditions for PBT-Esterified Cellulose
Ratio of esterifying agent to cellulose
A series of PBT-esterified cellulose was prepared with the esterification time fixed at 5h and the catalyst at 2% of the cellulose mass, in which, the mass ratio of PBT to cellulose was varied as 1:3, 1:4, 1:5, 1:6, and 1:7. The PBAT was then blended with the prepared esterified cellulose at a ratio of 95:5 to prepare composites, and the tensile test was carried out on the composites.
As shown in Figure 4, when the mass ratio of PBT to cellulose was 1:5, the toughness of the composite material was significantly improved, with the maximum elongation at break of 1026.54%, the maximum tensile strength of 24.02 MPa and the minimum tensile modulus of 37.05 MPa. Tensile properties of PBAT/esterified cellulose composites (Esterified cellulose was prepared in different mass ratios of PBT to cellulose).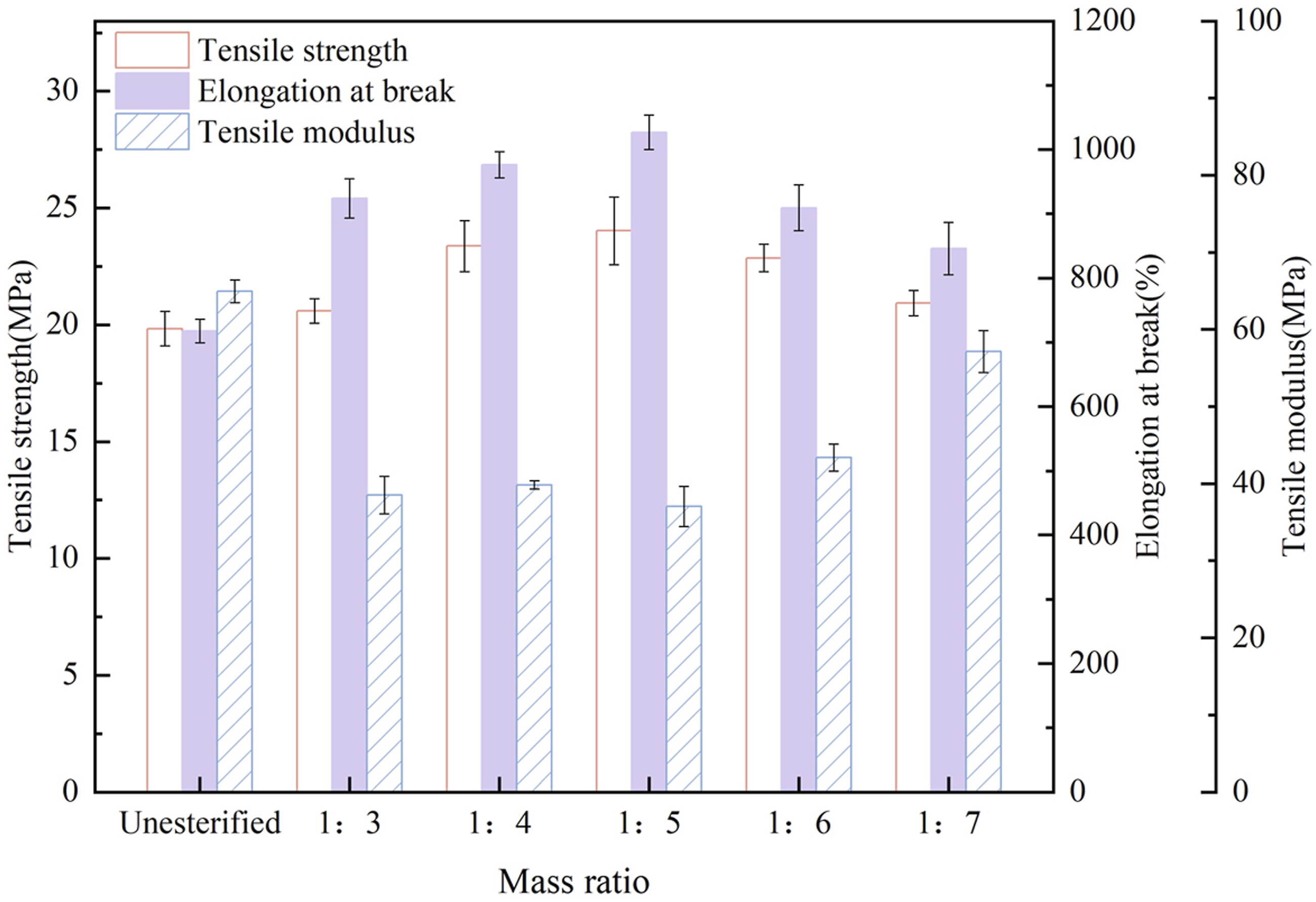
Reaction Time
A series of PBT-esterified cellulose was prepared with the mass ratio of PBT to cellulose fixed at 1:5 and the catalyst at 2% of the cellulose mass, in which, the reaction time was varied to 3h, 4h, 5h, 6h, and 7h. The composites of PBAT and esterified cellulose were prepared in the same proportion and tensile test was performed. As shown in Figure 5, (a) longer reaction time allowed the reaction to proceed more thoroughly, thus increasing the degree of esterification. When the esterification time was 6h, the composite material exhibited the best toughness, with an elongation at break of 1084.17%. The elongation at break of the PBAT/esterified cellulose composite was higher than that of the PBAT/cellulose composite. The tensile modulus reached 33.95 MPa, which was lower compared to the PBAT/cellulose composites. Moreover, the mechanical properties of the composite were also improved as the esterification time increased, with the maximum tensile strength of 24.64 MPa at 6h. Tensile properties of PBAT/esterified cellulose composites (Esterified cellulose was prepared with different reaction times).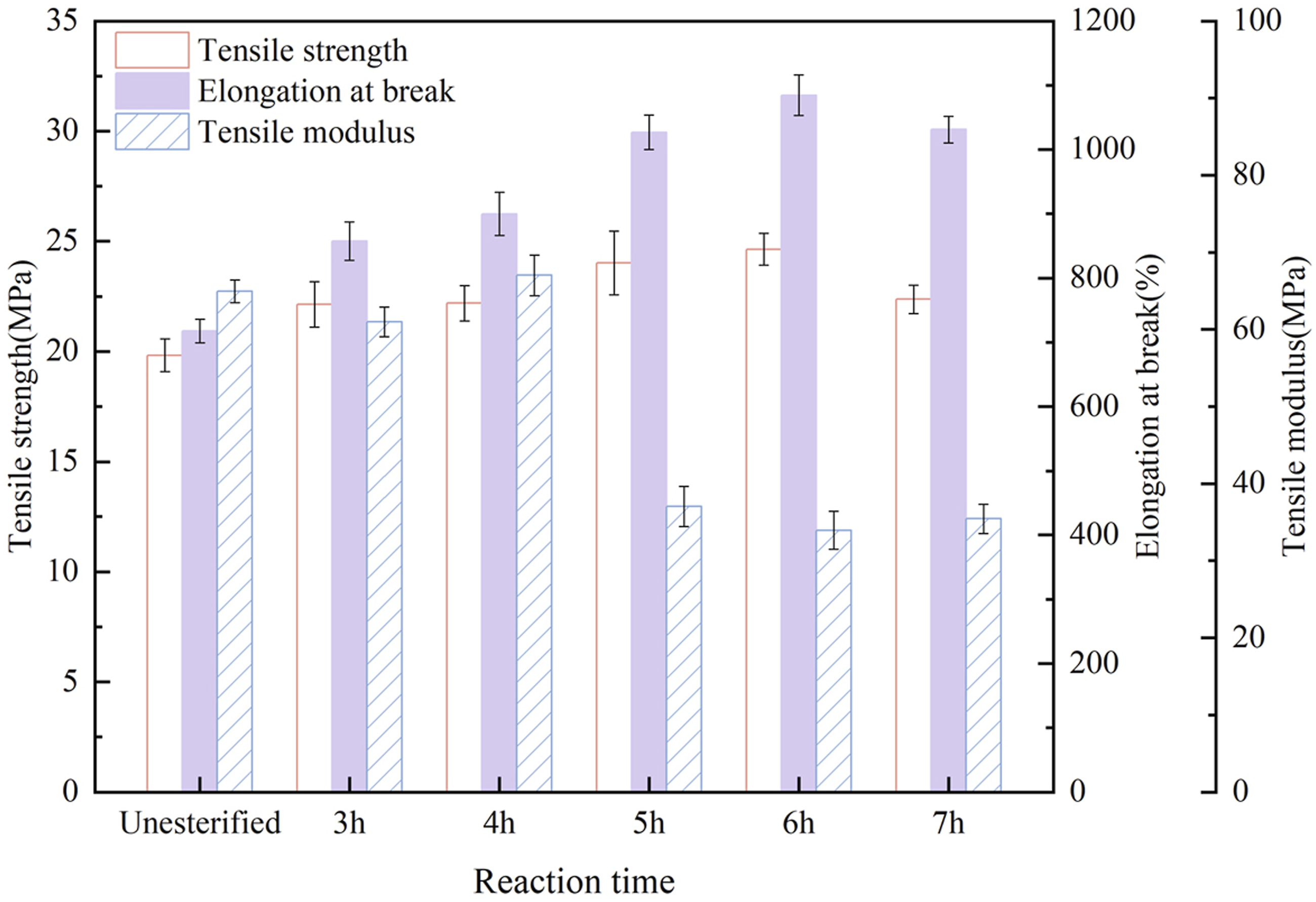
Catalyst Dosage
A series of PBT-esterified cellulose was prepared with the mass ratio of PBT to cellulose fixed at 1:5 and a reaction time of 6h, in which, the catalyst dosage was varied at 1%, 2%, 3%, 4%, and 5% of the cellulose mass. The composites of PBAT and esterified cellulose were prepared in the same proportion and tensile test was performed. As shown in Figure 6, when the catalyst dosage was 2%, the composite material exhibited the best toughness, with a tensile modulus of 33.95 MPa, an elongation at break of 1084.17% and the maximum tensile strength of 24.64 MPa. An appropriate catalyst dosage provided sufficient active sites, allowing the reaction between the terminal carboxyl groups of PBT and the hydroxyl groups of cellulose to proceed more thoroughly. Meanwhile, it also elevated the reaction rate and promoted the esterification equilibrium toward the direction of ester formation. Tensile properties of PBAT/esterified cellulose composites (Esterified cellulose was prepared with different catalyst dosages).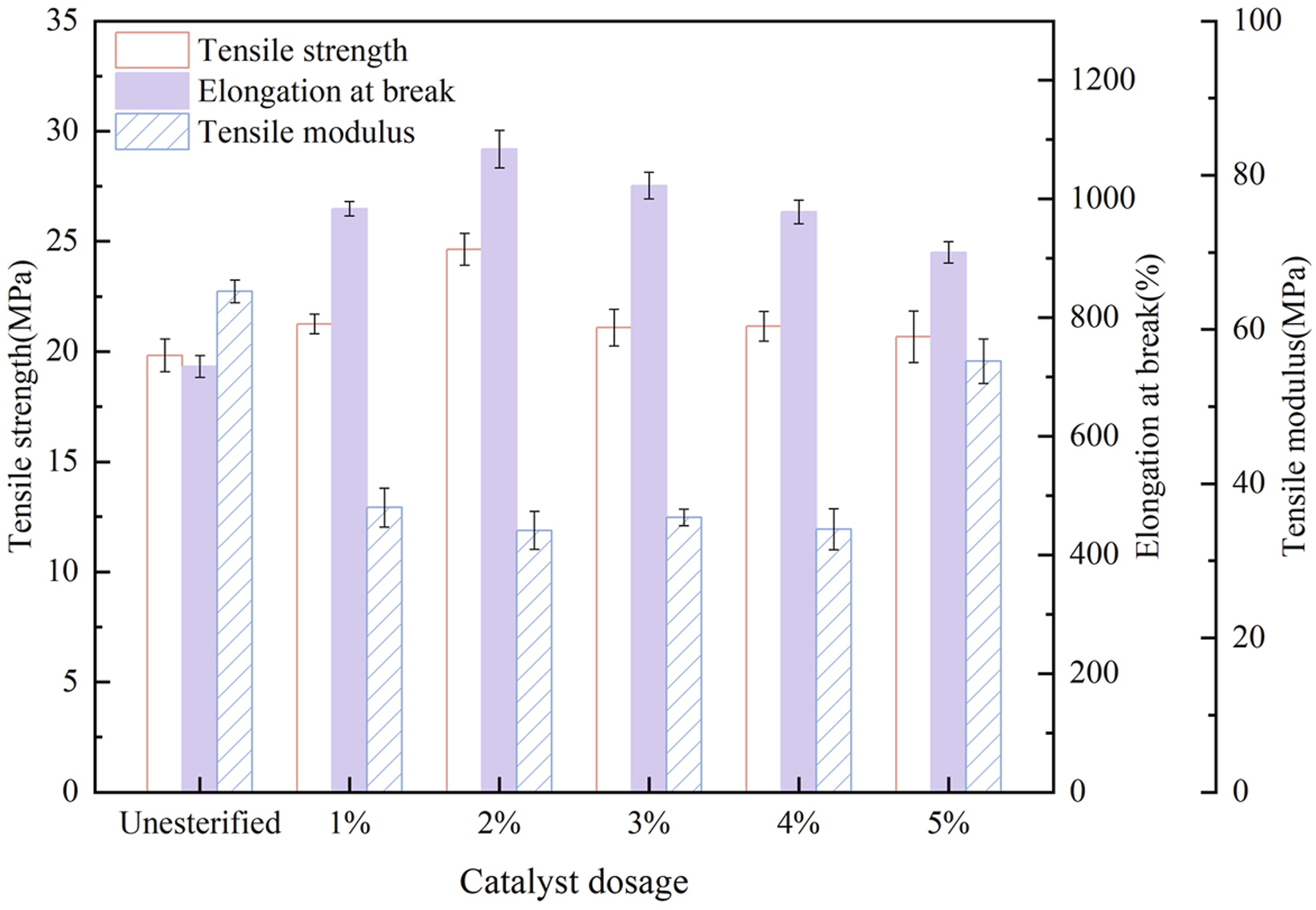
In summary, the optimal conditions for the esterification of cellulose with PBT were as follows: the mass ratio of PBT to cellulose was 1:5, esterification time was 6h, and catalyst dosage was 2% of the cellulose mass.
FTIR Analysis of PBT-Esterified Cellulose
The FTIR spectra of the PBT-esterified cellulose were basically the same, so the PBT-esterified cellulose prepared with the mass ratio of PBT to cellulose of 1:5, the esterification time of 6h, and the catalyst dosage of 2% the cellulose mass was taken as an example, and the FTIR spectra of that PBT-esterified cellulose and unesterified cellulose are shown in Figure 7. In the spectrum of cellulose, the peak at 3347 cm−1 was corresponded to -OH stretching vibration; the O-H bending vibration at 1600 cm−1 demonstrated that the sample combined micro amount of water; and the peaks at 2906 cm−1 and 1432 cm−1 belonged to the -CH2 stretching and bending vibrations. In the spectrum of esterified cellulose, a new absorption peak corresponding to C = O stretching vibration appeared at 1716 cm−1, indicating the formation of ester groups.
23
And the absorption peak at 743 cm−1 indicated the presence of benzene rings. All of these indicated that part of the hydroxyl groups (-OH) of cellulose had reacted with the terminal carboxyl groups (-COOH) of PBT, and the cellulose was successfully esterified. Infrared spectra of esterified cellulose and unesterified cellulose.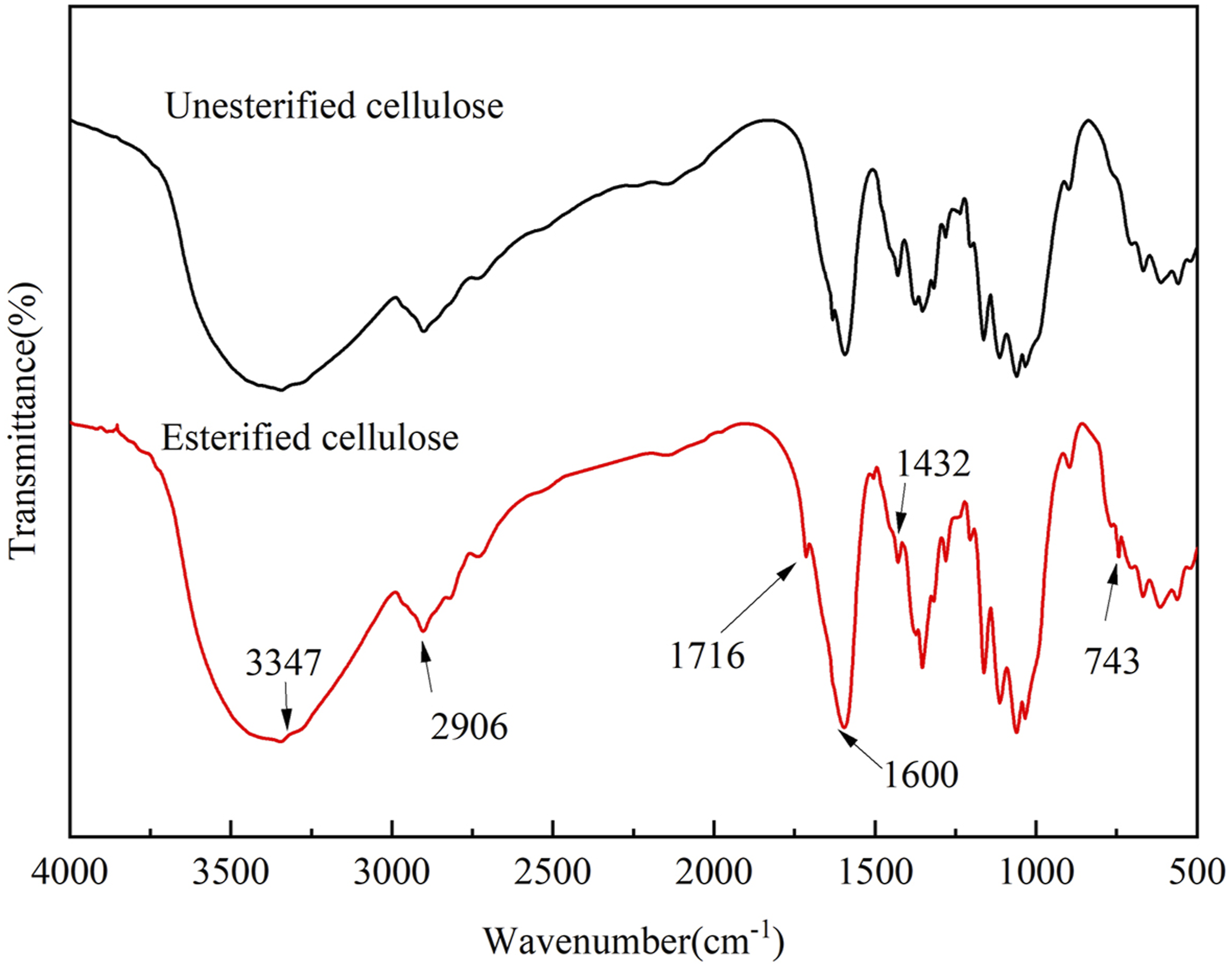
Thermal Properties of the Composites
Figure 8 shows the DSC curves of PBAT/esterified cellulose composites (ECM), and the related thermodynamic data are summarized in Table 1. DSC curves of PBAT/esterified cellulose composites: (a) cooling curve (b) second heating curve. DSC thermodynamic data of PBAT/esterified cellulose composites. ECM represents the PBAT/esterified cellulose composites; the suffix number refers to the amount of esterified cellulose.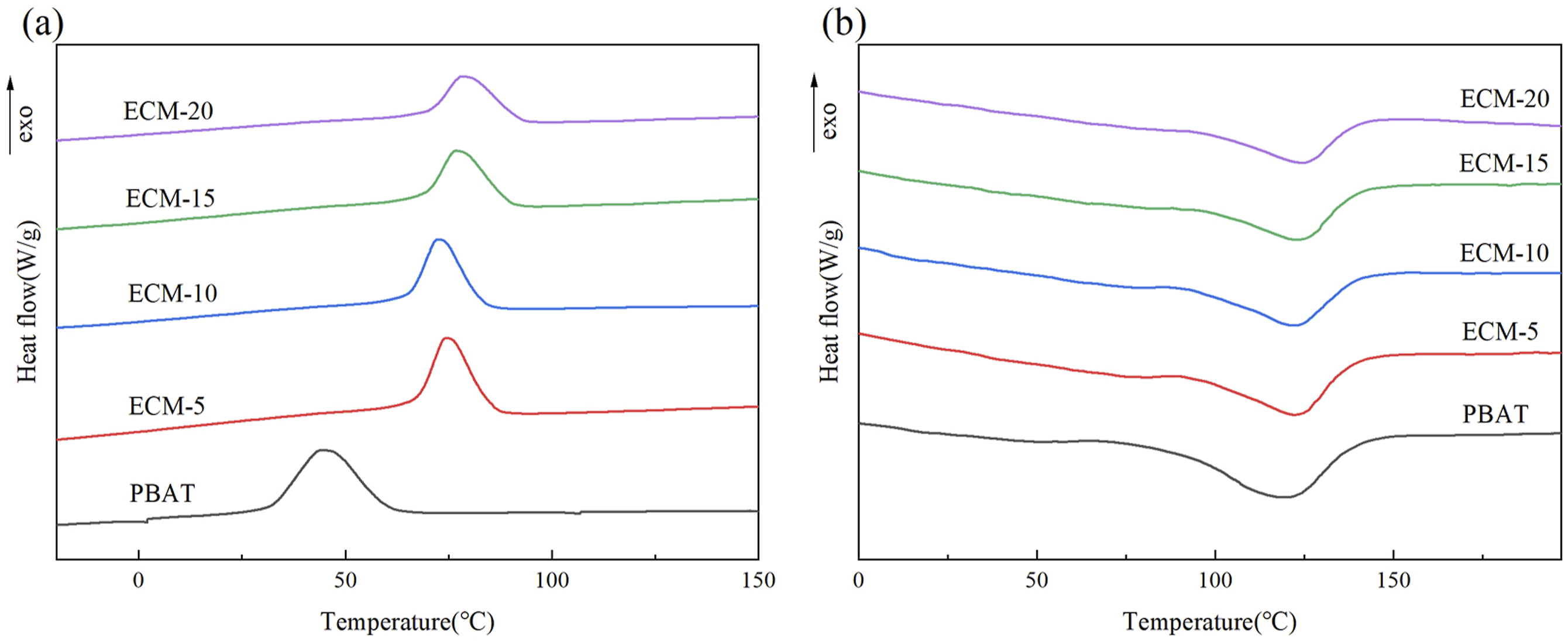
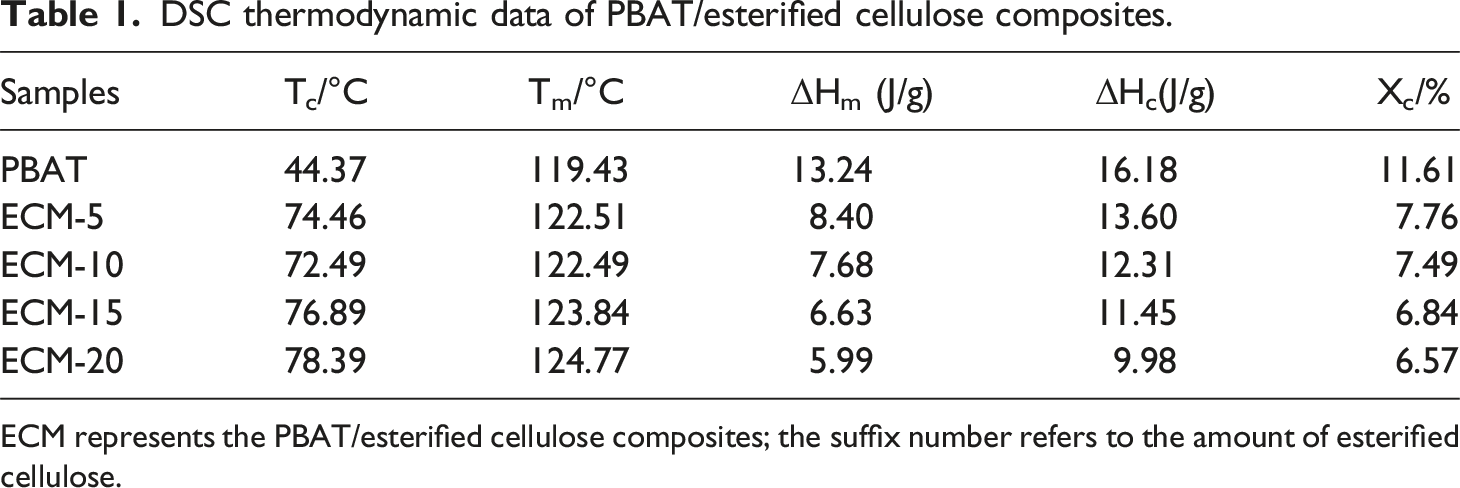
As clearly shown in Figure 8(a), the addition of PBT-esterified cellulose had a significant impact on the crystallization temperature (Tc) of PBAT. Compared with pure PBAT, the crystallization temperature (Tc) of the ECM increased significantly, and the Tc decreased first and then increased with the increase of the esterified cellulose amount. This trend was attributed to the fact that the added PBT-esterified cellulose played a role of heterogeneous nucleation in the PBAT matrix. 24 Furthermore, the high thermal conductivity of esterified cellulose compared to the polymer made it more prone to facilitate the formation of crystallization nuclei on its surface. As shown in Figure 8(b), the addition of PBT-esterified cellulose also caused a slight increase in the melting temperature (Tm) of the composites, along with a slight decrease in the melting enthalpy and the crystallinity. This was because the heterogeneous nucleation of PBT-esterified cellulose led to an increase in the number of newly formed crystals, while the size of the crystals decreased,25,26 resulting in a reduction in the crystallinity of the composite material.
Thermal Stability of the Composites
Cellulose and PBAT present distinct thermal decomposition characteristics and are thermodynamically incompatible, so the esterification modification and blending ratio will affect the interdependence of the two phases.
27
Therefore, it is necessary to conduct thermogravimetric characterization and analysis on ECM with different blending ratios. Figure 9 shows the TGA and DTG curves of PBAT and composites with different blending ratios, and the related TGA thermodynamic parameters are summarized in Table 2. Thermogravimetric curves of PBAT/esterified cellulose composites: (a) TGA curves (b) DTG curves. TGA thermogravimetric data of PBAT/esterified cellulose composites.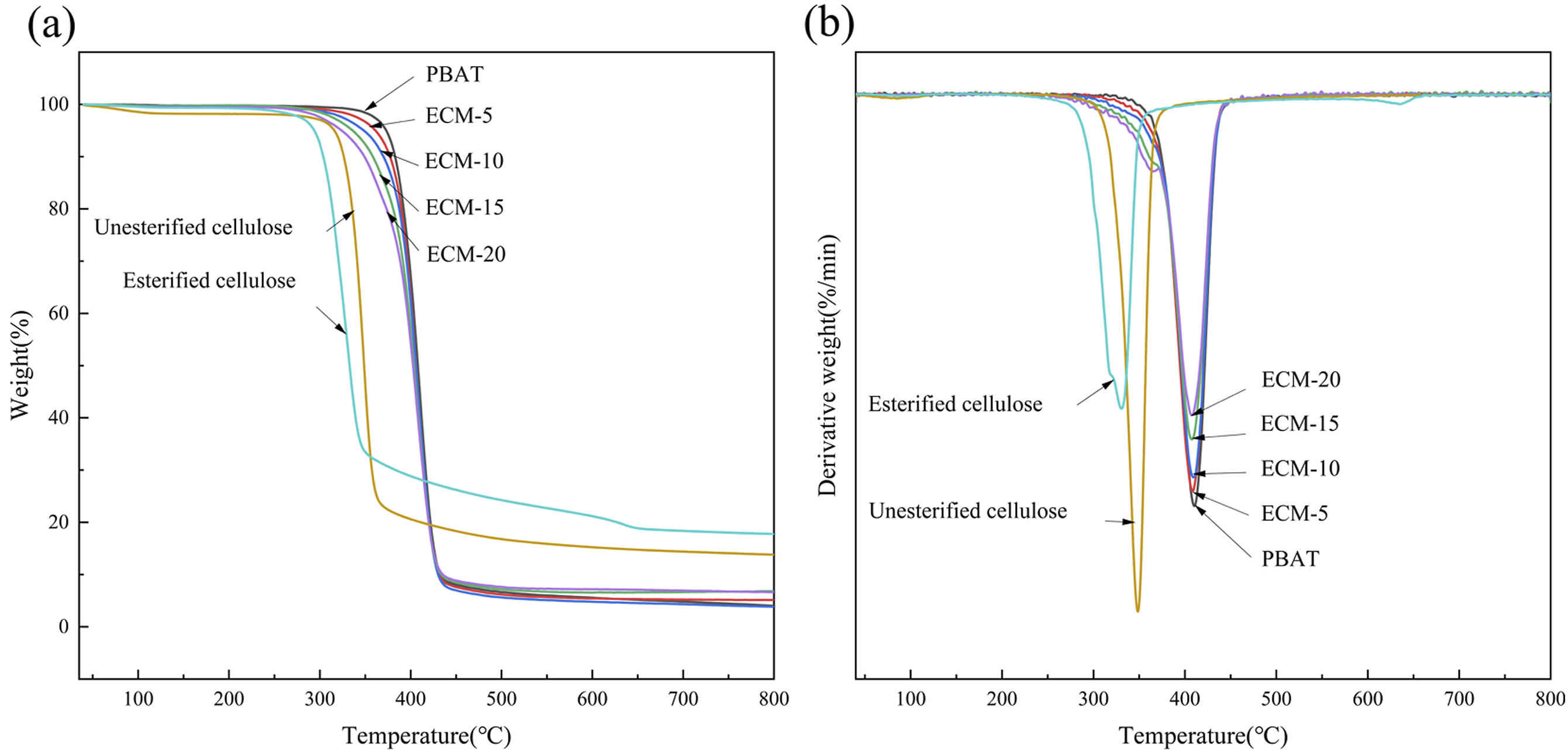
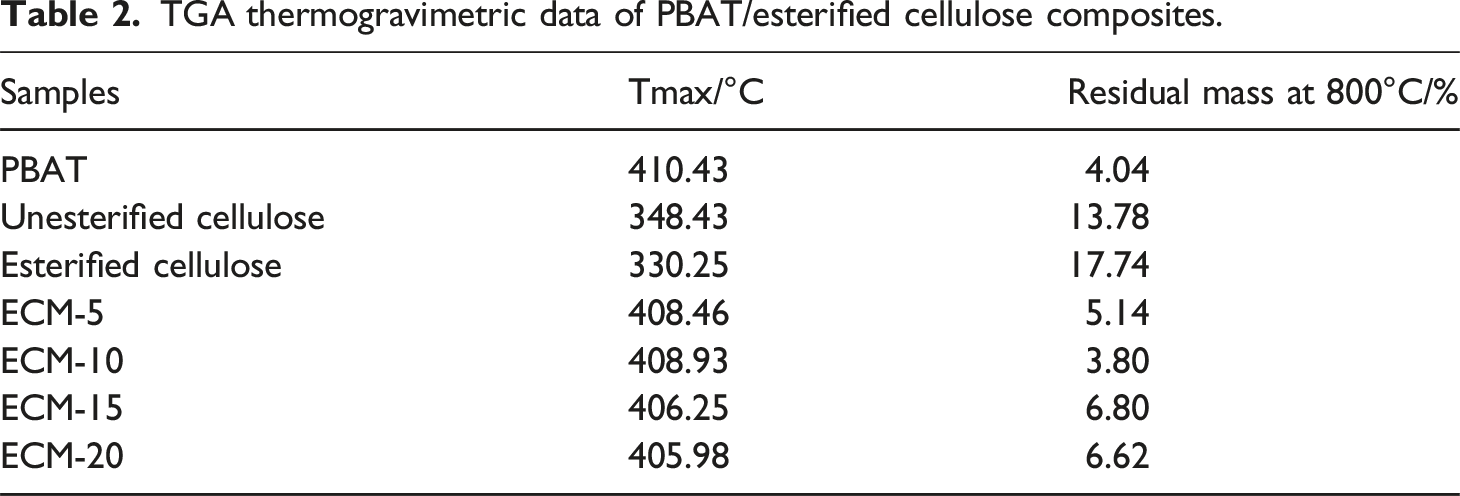
The Figure 9 showed that the thermal decomposition process of the composites all contained only a single decomposition stage, which indicated that the esterification modification of cellulose enhanced its interdependence and compatibility with PBAT.
As shown in Figure 9(a), it could be observed that cellulose exhibited relatively higher thermal stability than esterified cellulose. This was attributed to the low molecular weight PBT components introduced through surface esterification, which underwent thermal degradation prior to cellulose. The initial thermal degradation temperature of the composites was lower than that of PBAT, and it gradually decreased with the increase of the esterified cellulose amount, which was caused by the lower thermal stability of the esterified cellulose. 28 The primary thermal decomposition temperature of PBAT and the composite materials ranged between 300 and 400°C, and the temperatures for complete decomposition were similar, higher than 400°C. When the temperature was rose to 800°C, there was residue for PBAT and the composite materials, 29 and the composite ECM-10 showed the least residual mass of 3.8%.
It can be observed from the DTG curves in Figure 9(b) that, when the ECM with the blending ratios of 80/20 and 85/15, the width of the peak corresponding to the maximum decomposition rate was broader than that of the ratios of 90/10 and 95/5. This indicated that when the amount of esterified cellulose was up to a certain amount, the two phases of PBAT and esterified cellulose showed weaker interdependence and poorer compatibility, while when the blending ratios was 90/10 and 95/5, the two phases exhibited better compatibility. 30
VST of the Composites
As shown in Figure 10, the VST of the composites gradually increased with the increase of the esterified cellulose amount, and when the ratio of PBAT to esterified cellulose was 80/20, the VST reached the maximum of 98.3°C. This indicated that the addition of esterified cellulose improved the thermal performance of PBAT. Although the well-dispersed esterified cellulose destroyed the continuity of the PBAT matrix, it significantly restricted the mobility of PBAT molecular chains. As a result, the VST of the composite was increased, which allowed the composite material could be used in a broader range of conditions. VST of PBAT/esterified cellulose composites.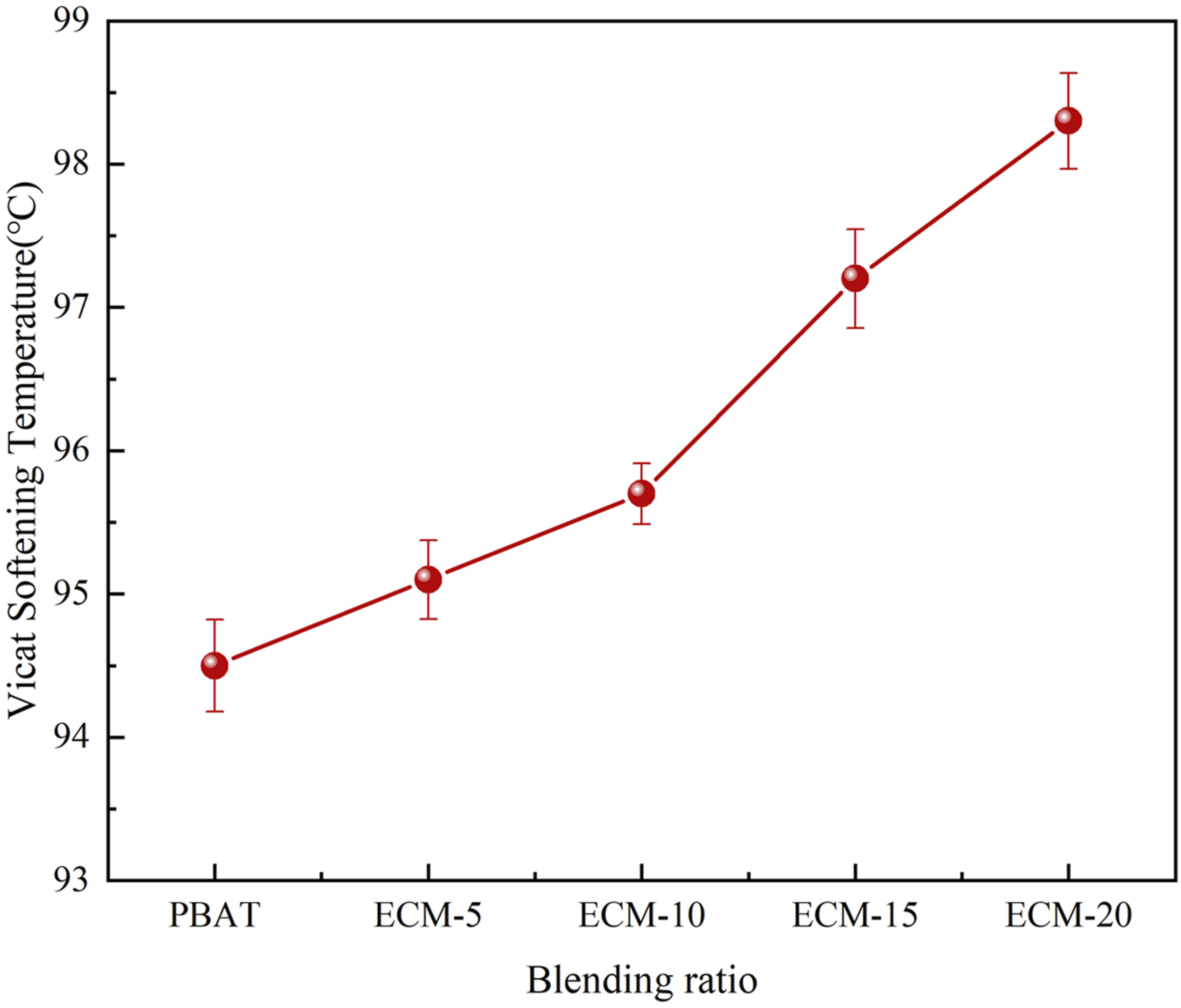
Rheological Properties of the Composites
Figure 11 shows the relationship between the storage modulus and loss modulus with angular frequency for the composites of PBAT and cellulose before or after esterification. The storage modulus and loss modulus of PBAT/esterified cellulose composites (ECM) both increased with the increase of angular frequency, and the storage modulus was slightly higher than the loss modulus, indicating that the elastic properties dominated over the viscous properties. From Figure 11(a) and (c), it can be observed that the storage modulus of PBAT/unesterified cellulose composites (CM) was higher than that of pure PBAT in the low-frequency region, while the change trend and final value of the loss modulus tended to be similar to that of pure PBAT. In contrast, Figure 11(b) and (d) showed that the storage modulus of ECM was lower than that of pure PBAT in the high-frequency region, and the loss modulus was also lower than that of pure PBAT. The reason might be that esterified cellulose in the PBAT matrix could act as a lubricant or diluent, so as to reduce the friction between PBAT molecular chains.31,32 On the other hand, the addition of esterified cellulose destroyed the ordered structure of PBAT molecular chains, so that the relative movement between the molecular chains was more prone to occur under shear forces. Storage modulus curves of the composites: (a) PBAT/unesterified cellulose composites (CM); (b) PBAT/esterified cellulose composites (ECM); and loss modulus curves: (c) CM; (d) ECM.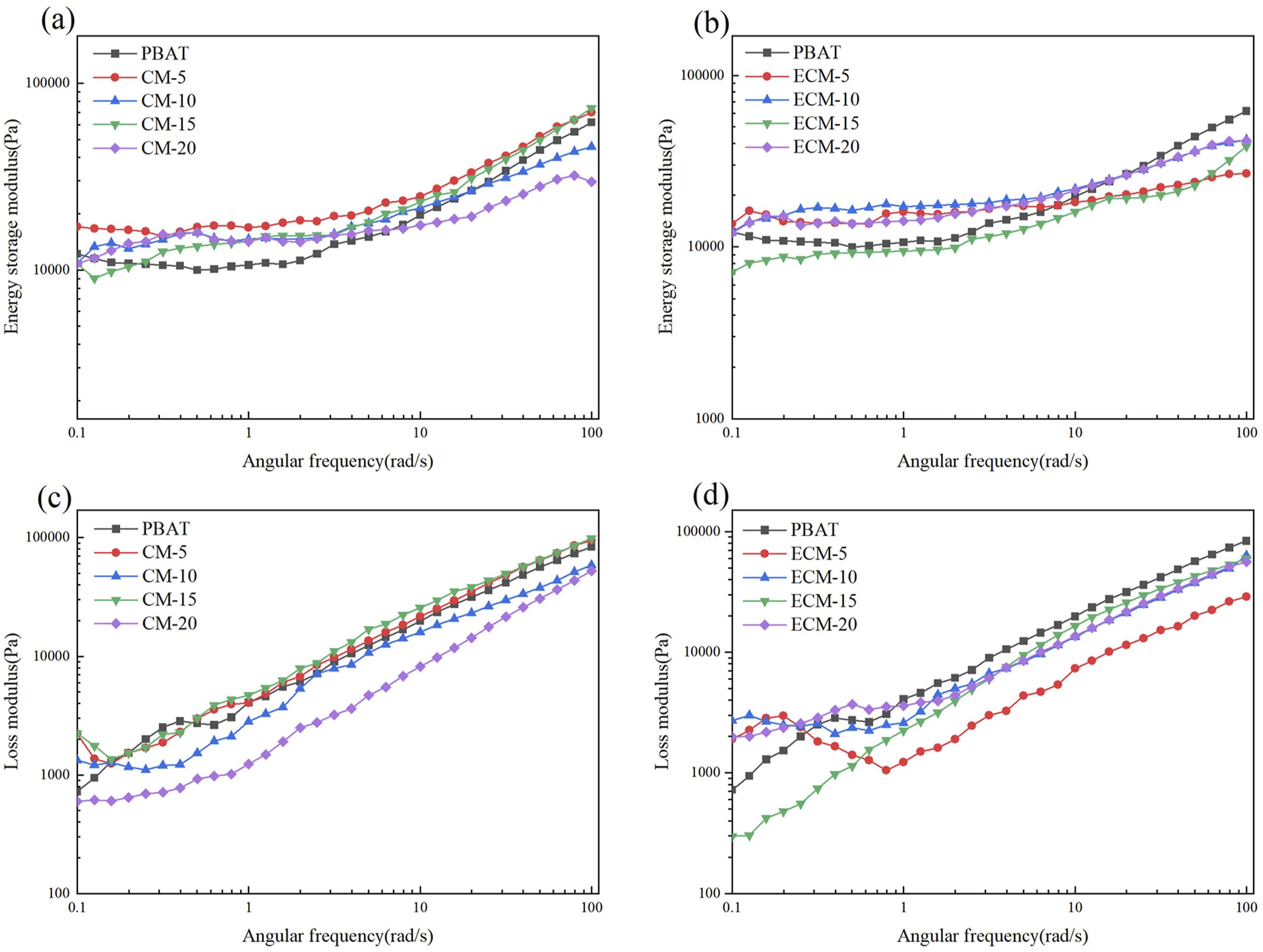
Figure 12 illustrates the variation curves of complex viscosity with angular frequency for the composites. Overall, the complex viscosity decreased with the increase of angular frequency. As shown in Figure 12(a), the complex viscosity of CM was higher than that of pure PBAT. In Figure 12(b), the complex viscosity of ECM was also higher than that of pure PBAT in the low-frequency region, but lower in the high-frequency region. When angular frequency increased, the stress applied to the material also increased correspondingly, which led to the entanglement between esterified cellulose and PBAT, PBAT and PBAT macromolecules being more easily opened, so the flow resistance of the melt was reduced. As observed in Figure 12(b), when the ratio of PBAT to esterified cellulose reached 80/20, the complex viscosity of the composite nearly coincided with that of pure PBAT, which meant that when the esterified cellulose amount in PBAT exceeded a certain value, the viscosity and rigidity of the composite melt would be reduced further.33,34 Complex viscosity curves of the composites: (a) CM (b) ECM.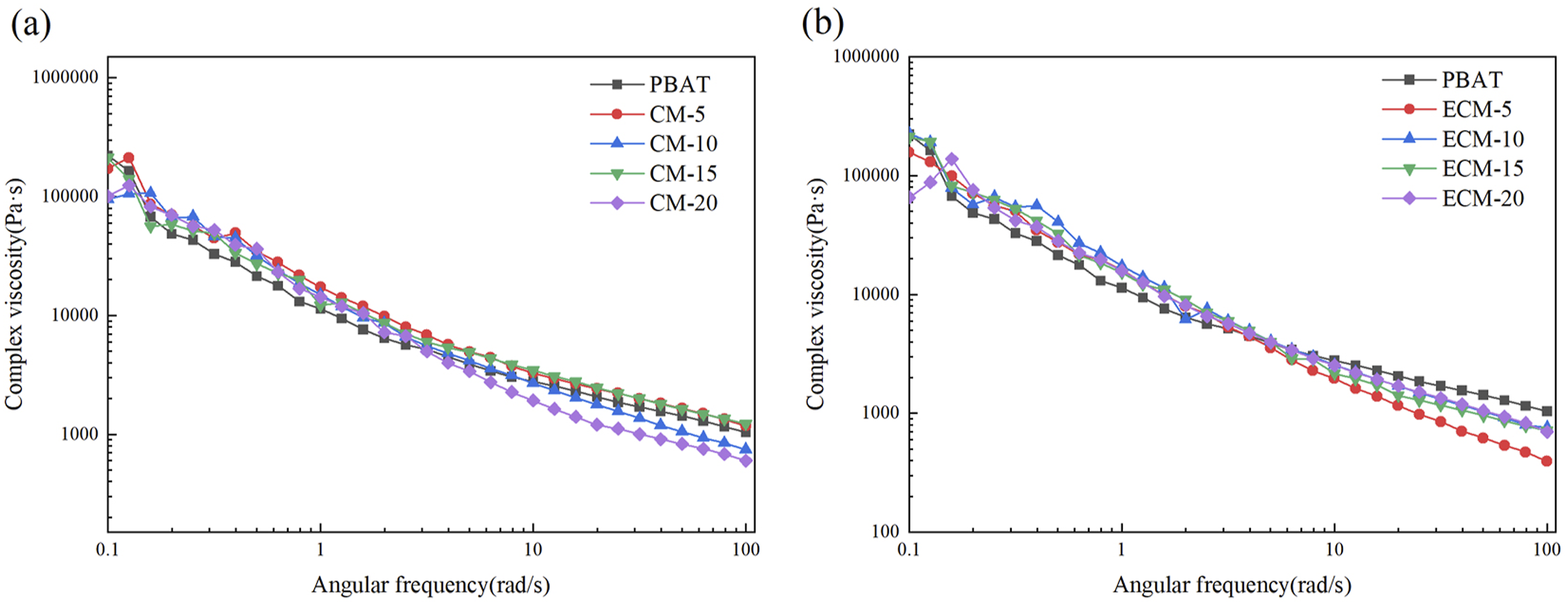
Figure 13 shows the Han curves of the composites. The Han curves of pure PBAT were temperature independent since it was a homogeneous system. The curves of all composite systems were in good consistency with the PBAT curve and there was no shift at the end,
34
indicating that for the composite at this temperature, PBAT and PBT-esterified cellulose exhibited good interfacial compatibility. Han curves of the composites: (a) CM (b) ECM.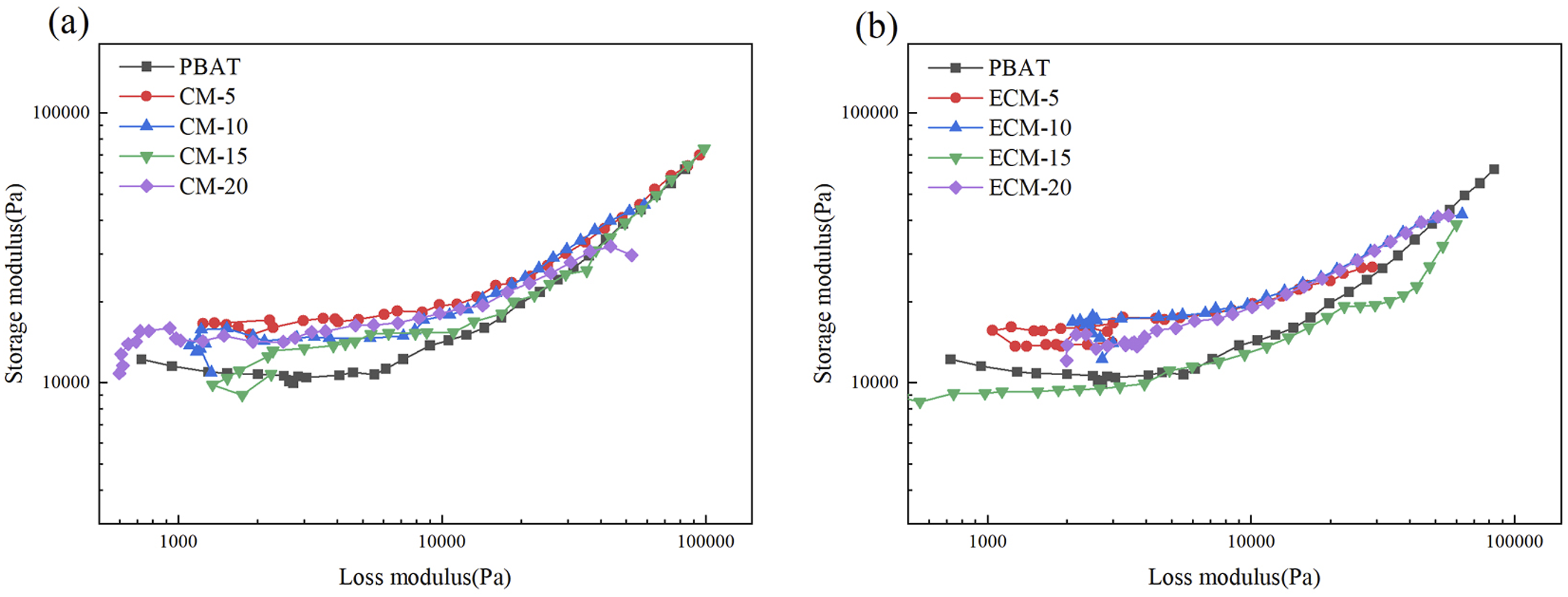
Melt Flow Index of the Composites
Melt flow index of the composites.

The sample name was the combination of the first column and the first row.
As shown in Table 3, the addition of unesterified cellulose had a minimal impact on the melt flow index (MI) of the composites. However, for the ECM, the MI of the composites initially decreased and then increased with the increase of the amount of PBT-esterified cellulose, but it remained higher than that of pure PBAT. The MI reached its maximum value of 12.9 g/10min when the ratio of PBAT to esterified cellulose was 95/5. PBAT molecular chains showed high flexibility and formed a highly dense entanglement structure. In the high-temperature molten state, the PBT-esterified cellulose disrupted the entanglement points between PBAT molecular chains, which resulted in a decrease of the entangled structure density and the intermolecular forces among PBAT chains, thereby enhancing the mobility of the PBAT chain segments. The MI results were consistent with the results of the storage modulus and loss modulus in the rheological performance.
Mechanical Properties of the Composites
Figure 14 shows the stress-strain curves of the composites. For PBAT and all the composite materials, the stress of the material increased linearly with the strain in the elastic stage, indicating that all the material underwent elastic deformation. In Figure 14(a), the yield stress of only CM-10 in the PBAT/cellulose composites (CM) was higher than that of PBAT at the yield stage. In Figure 14(b), the yield stress of the PBAT/esterified cellulose composites (ECM) was all lower than that of PBAT at the yield stage, showing the highest yield stress of 9.07 MPa with a PBAT/esterified cellulose blending ratio of 85/15. After the yield stage, the stress and strain became a linear relationship again, indicating that the material underwent irreversible permanent deformation. The material fracture occurred when the stress reached the highest value. Stress-strain curves of the composites: (a) CM (b) ECM.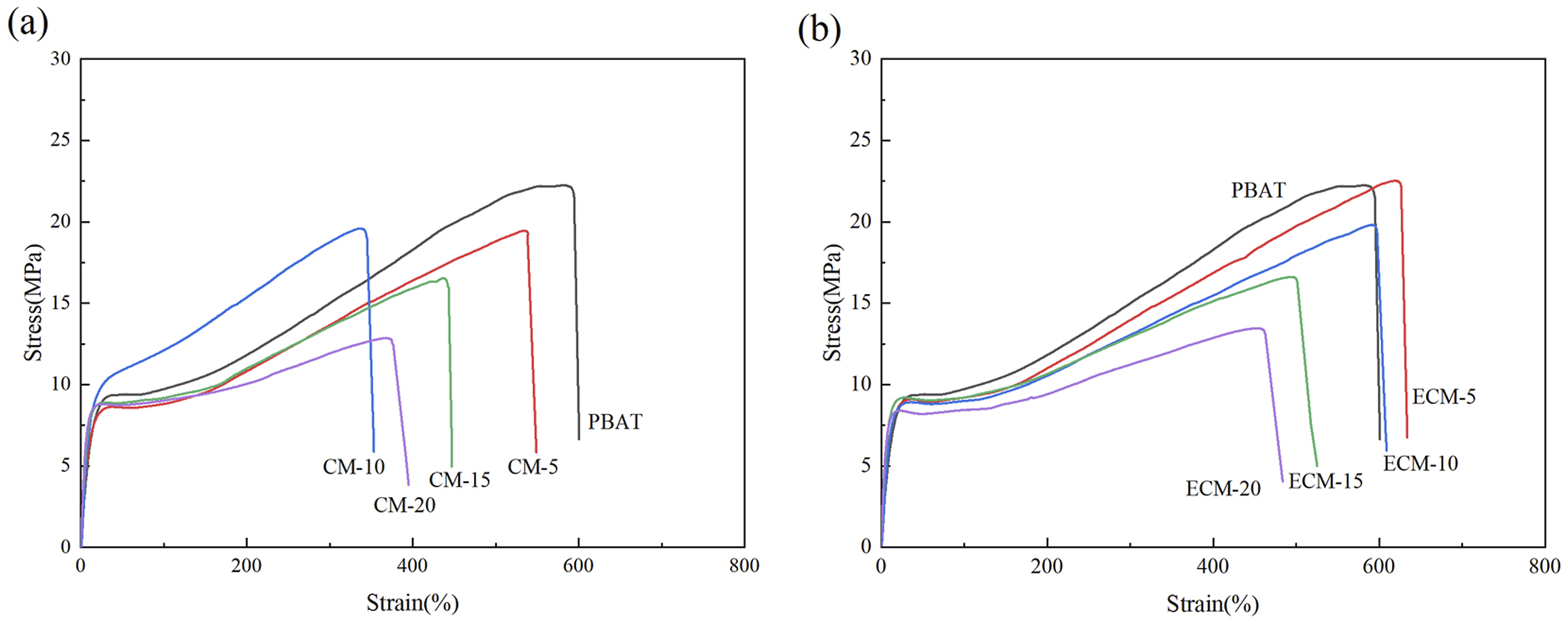
Figure 15 shows the tensile strength, elongation at break and tensile modulus of the composites, and the related data was summarized in Table 4, Table 5 and Table 6. It is evident that the addition amount of cellulose, either before or after esterification, had a significant impact on the mechanical properties of the composites. And with the same blending ratio, the tensile strength of ECM was higher than that of CM. This was due to that the compatibility between cellulose and PBAT matrix was improved after the cellulose esterification, resulting in the mutual entanglement between the both, and the increase of the internal friction of the composite material. Figure 15(a) and Table 4 showed that the tensile strength of the composites increased first and then decreased with the increase of the cellulose amount, either before or after esterification. When the amount of esterified cellulose was lower, the PBT grafted on the surface of cellulose improved its compatibility with PBAT, which resulted in better dispersion. Moreover, molecular chain entanglement was generated between PBT extending from the cellulose surface and PBAT, which enhanced the interfacial force and internal friction force between PBAT matrix and cellulose filler, thus the tensile strength was increased. When the blending ratio of PBAT and esterified cellulose was 95/5, the tensile strength of the composite reached the maximum value of 21.89 MPa. However, when the esterified cellulose amount continued to increase, the defects caused by the agglomeration of esterified cellulose increased the phase interface,
35
leading to worse compatibility, which in turn resulted in a greater loss of composite strength.
36
Tensile properties of the composites: (a) tensile strength (b) elongation at break (c) tensile modulus. Tensile strength of the composites. The sample name was the combination of the first column and the first row. Elongation at break of the composites. The sample name was the combination of the first column and the first row. Tensile modulus of the composites. The sample name was the combination of the first column and the first row.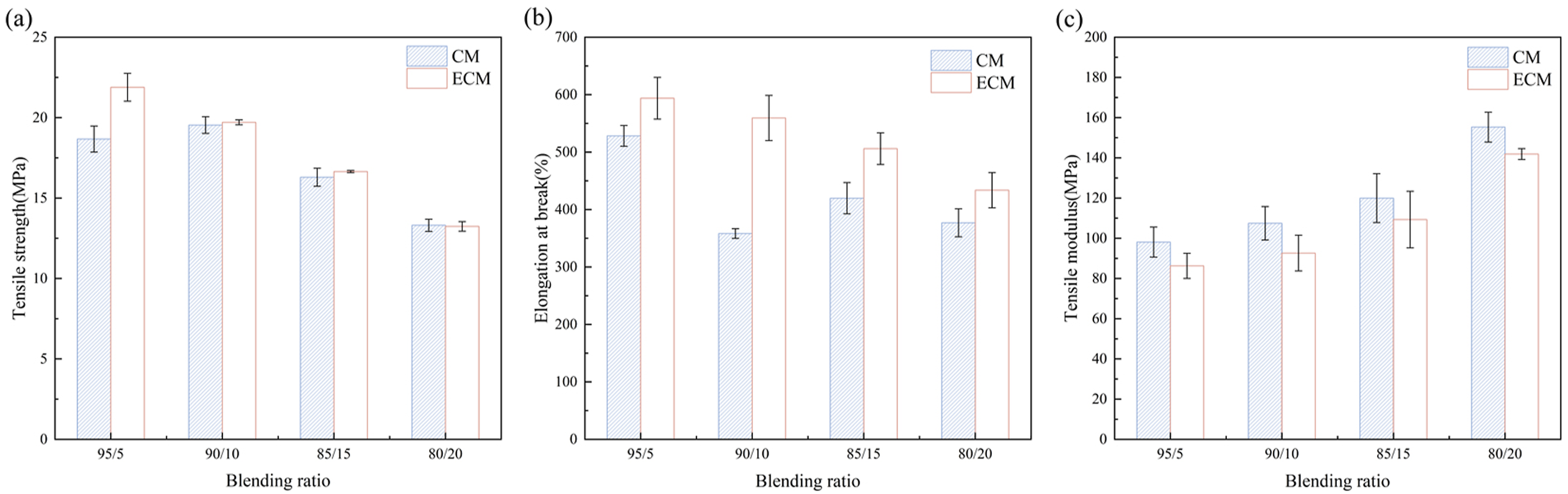



As shown in Figure 15(b) and Table 5, the elongation at break of ECM increased initially and then decreased with the increase of esterified cellulose amount. However, it was all higher than that of CM, indicating that PBT esterification of cellulose could enhance the toughness of the composite. As the blending ratio of the composite was larger, the elongation at break decreased. This may be because that when a higher amount of esterified cellulose was added, the agglomerated esterified cellulose was prone to produce defects in the PBAT matrix, the weak links of the material damage, which easily became the fracture source during the stretching process, so the material was more susceptible to break under tensile force. 37 When the blending ratio of PBAT and esterified cellulose was 95/5, the elongation at break of the composite reached the maximum value of 593.82%. This was owing to the fact that the grafted PBT chain on the surface of cellulose could tangle well with the PBAT matrix, and the intermolecular force increased. During the tensile process, the PBT-esterified cellulose was moved together with the PBAT molecular chains, which could result in large plastic deformation during the tensile process. And the deformation ability was improved, which proved that the toughness of the composite material was increased while maintaining the strength.
As shown in Figure 15(c) and Table 6, the tensile modulus of PBAT/esterified cellulose was higher than that of PBAT. This was attributed to the strong molecular polarity and intermolecular forces in cellulose, as well as the six-membered pyranose rings in the structure conferred rigidity to cellulose. In the PBAT matrix, cellulose acted as a rigid particle, thereby the tensile modulus of the composite material was increased. As the amount of esterified cellulose increased, the tensile modulus of PBAT/esterified cellulose exhibited an upward trend, although it remained lower than that of PBAT/unesterified cellulose. When the blending ratio of PBAT to esterified cellulose was 95/5, the composite material exhibited the lowest tensile modulus of 86.3 MPa.
Figure 16 shows the impact notch diagram of the composites. During the impact process, the composites exhibited only larger notches without fracture, demonstrating good toughness. The notches of the ECM were significantly smaller than those of CM with the same ratio, which indicated that esterification modification of cellulose could effectively enhance the toughness of the composites. This finding was aligned with the results of the elongation at break. Impact notch diagram of the composites.
Microstructure
Figure 17(a) and (b) depict the surface morphology of cellulose before and after esterification. As shown in Figure 17(a), unesterified cellulose exhibited a compact fibrous bundle-like structure with large particles, and the surface was relatively rough with holes and gullies.
38
In contrast, Figure 17(b) shows that of cellulose after esterification, in which, the aggregated structure was broken into smaller fragments, and the surface was damaged with the pores in the cellulose surface disappeared. The magnification diagram also revealed that there were layered structures of the esterification products on the surface. The PBT-esterified cellulose was easier to be infiltrated and coated by polymers, so that it could be well compatible and combined with PBAT to form composite with good performance. SEM images of cellulose before (a) and after esterification (b) and brittle fracture surface of composites: (c) CM (d) ECM.
Figure 17(c) and (d) show the morphology of the brittle fracture surfaces of the composites. As shown in Figure 17(c), the CM exhibited partial phase separation with visible cellulose particles. This phenomenon was caused by the poor compatibility of hydrophilic cellulose and oleophilic PBAT, and the cellulose could not be effectively coated by PBAT and exposed outside, so the interdependence between the two phases was relatively weak. As shown in Figure 17(d), after cellulose was esterified by PBT, the fracture surface of the composite ECM became smooth and uniform, indicating that the esterified cellulose was be effectively coated by PBAT. Moreover, there was “crazing” wrinkles appeared, demonstrating that the esterified cellulose had improved the interfacial compatibility and toughness of the composite, which was consistent with the mechanical performance results. at the same ratio, it was evident that esterified cellulose exhibited better dispersion in PBAT than unesterified cellulose. On the one hand, PBT esterification disrupted the ordered structure of cellulose, resulting in reduced crystallinity. On the other hand, since PBT and PBAT had similar structures, the compatibility between the PBT-esterified cellulose and PBAT was elevated according to the principle of the dissolution in the similar material structure. Moreover, the strongly hydrophilic hydroxyl groups on cellulose were partially replaced by ester groups through the esterification process, and the hydrophobicity of cellulose was increased, which would further improve the interfacial compatibility between the two phases of PBAT and esterified cellulose. 28
Surface Energy and Interfacial Tension
Surface energy of PBAT, unesterified cellulose and esterified cellulose.
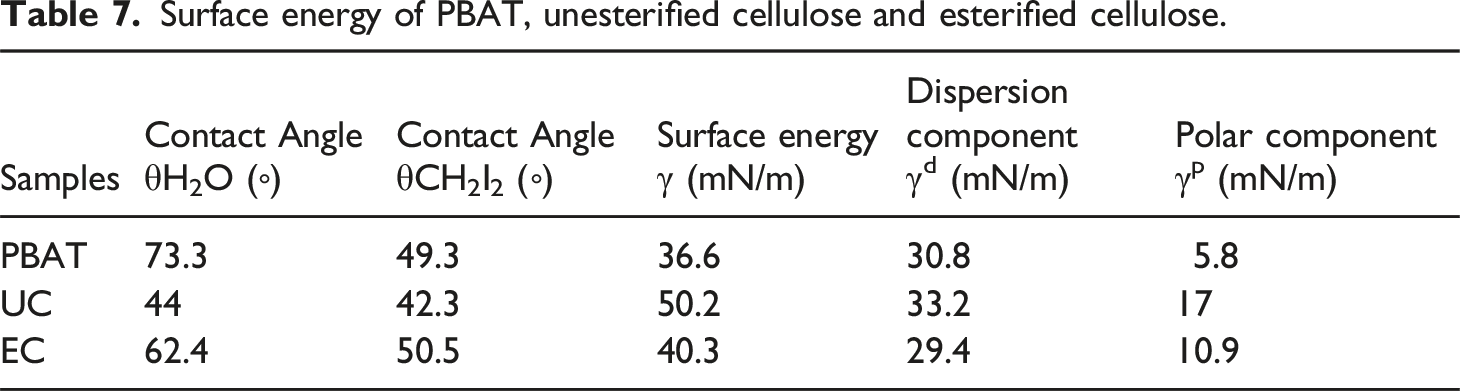
As shown in Table 7, the water contact angle of esterified cellulose was greater than that of unesterified cellulose, and the surface energy was lower than that of unesterified cellulose. After esterification by PBT, part of the hydroxyl groups on the surface of cellulose were replaced by PBT polyester with low molecular weight, so the number of hydroxyl groups on the surface was reduced, resulting in reduced surface energy and improved hydrophobicity. After esterification, the water contact angle and surface energy of cellulose became closer to PBAT, indicating that PBAT and esterified cellulose were more similar and compatible in hydrophilicity.
The interfacial tension can be determined by equation (4).39,40 The interfacial tension between PBAT and unesterified cellulose was 5.7 mN/m, and that between PBAT and esterified cellulose was 1.6 mN/m, which indicated that the interfacial tension between PBAT and esterified cellulose was smaller and the interface compatibility was better.41,42 After the surface of cellulose was esterified with low molecular weight PBT, the surface of cellulose was coated with PBT, and the PBT was evenly distributed at the interface of the composite ECM. The PBT on the surface of cellulose exhibited good compatibility with PBAT, which improved the dispersibility of cellulose in PBAT and reduced the interfacial tension between PBAT and cellulose, which was conducive to the stress transfer.
Conclusion
In this work, carboxyl-terminated PBT with molecular weight of 4800 was synthesized and used as an esterifying agent to modify cellulose and prepare PBT-esterified cellulose through a dry esterification process. The optimal formulation for the preparation of esterified cellulose was determined as follows: the mass ratio of esterifying agent to cellulose was 1:5, the esterification time was 6h, and the catalyst dosage was 2% of the cellulose mass. PBAT/esterified cellulose composites (ECM) were prepared using PBAT and the PBT-esterified cellulose prepared based on the above optimal formulation, and it was found that the crystallization temperature, thermal stability, and Vicat softening temperature of the composites were improved. The storage modulus of ECM was slightly higher than the loss modulus, and both the storage and loss moduli were lower than those of pure PBAT in the high-frequency range. Additionally, the flowability of ECM was superior to that of PBAT/unesterified cellulose composite (CM). When the blending ratio of PBAT/esterified cellulose was 95/5, the mechanical properties of ECM was the best with an elongation at break of 593.82%, a tensile strength of 21.89 MPa and a tensile modulus of 86.3 MPa. The impact notch diagrams, SEM images of the brittle fracture surfaces, the surface energy and interfacial tension further demonstrated that the esterification of cellulose with PBT significantly enhanced its compatibility with PBAT and improved the strength and toughness of the composite materials. This work offers a new direction for the development and production of degradable plastics with advantages of greenness, low-cost and good overall performance, and it is expected to broaden the application scope of PBAT.
Footnotes
Author contributions
Conceptualization and Methodology: Tiejun Ge and Xiaofeng Liu; Data collection, analysis and writing original draft preparation: Mingzi Chen; Visualization and Investigation: Yang Yu; Supervision: Xiaofeng He and Qi Yue; Reviewing and Editing: Xiaofeng Liu; Funding: Tiejun Ge, Xiaofeng Liu, Xiaofeng He and Yang Yu. All the authors approved the final articie.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the 2022 Liaoning Province Science and Technology Innovation Platform Construction Plan project (Liao Kefa (2022) No.36), Key technology research project of Yingkou City in 2022 (YingKefa (2022) No.38), Key Technology Research Project of Yingkou City in 2023 (YingKefa (2023) No.32), 2023 Basic Scientific Research Project of the Education Department of Liaoning Province (JYTMS20231504), the 2021 Scientific Research Foundation of Education Department of Liaoning Province (LJK Z0463), and 2023 Shenyang University of Chemical Technology Discipline Construction Standard Project (HuaDafa (2023) No.20).