Abstract
Poly (O-toluidine) (POT) doped with vanadium pentoxide (POT- nV2O5) nanocomposites have been synthesized by In – Situ chemical polymerization method for dielectric property and electrical conductivity. The POT has been added with V2O5 with various weight percentages, like 25, 50, and 75 wt%, respectively. These samples undergo structural, morphological, thermal, dielectric, and DC electrical conductivity analyses. From XRD data, the addition of V2O5 results in changes and crystallinity. FTIR spectrum conforms the presence of C = C stretching band of quinoid ring for poly (O - toluidine) doped with 75 wt% (PV75) at 1651 cm−1, bands observed at 1105 cm−1, 1110 cm−1, 1194 cm−1, is attributes due to benzoid ring of POT. PV75 reveals hybridization of vanadium pentoxide in POT at 540 cm−1 due to improved crystallinity and more saturated bonds, POT doped with 50 wt% of vanadium pentoxide (PV50) and PV75 exhibit good thermal stability and residual mass. Particles in PV75 SEM images exhibit globular shape in cluster groups around the POT by homogenous nucleation. The dielectric permittivity and electrical conductivity (σDC) increase with temperature and dopant weight percentage. Dielectric permittivity ranges from 1.85 F/m to 24.5 F/m and σDC starts from 0.58 × (10−5) S/cm to 2.21× (102) S/cm.
Highlights
• Higher V2O5 dopant results in optimized structural and crystallinity properties.
• PV75 exhibits a stronger thermal stability
• POT doped with 75 wt% of V2O5 found suitable for application in electrical devices.
Introduction
Electrical energy storage has prominent role in electronics due to its power density and rate competence. The growth of energy demand with echo-friendly and cost effective has become more significant for researchers. In recent years researchers focus on dielectric properties of ZnO, CuO, MoO3, V2O5, Ag, Na2O, Er2O3 and MgO1–4 with various dopant, The structural, optical, and dielectric characteristics of the Er2O3/MgO nanocomposite have been studied by A. V. Jayasrinivasan et al. and their results point out that the dielectric constant rises at higher temperatures at lower frequencies as a result of electrode polarization, resulting in space charge polarization. Metal or metal oxide based nanocomposites were developed to improve structural, optical, and electrical properties using several types of techniques, including the capacitively coupled RF discharge system, plasma enhanced chemical vapor deposition, and the melt quenching method.5,6
Dielectric based polymer materials have extensively used for sensors and optoelectronics, which have attained immense importance for researchers.7,8 Polymers have attained great interest for researchers for dielectric application due to its cost effective and flexibility, many reports suggest that polymer based nano materials has important role in electric vehicles and rails.9,10 A.A. Al-Muntaser et al. has fabricated and characterized ZnO and polyvinyl chloride/poly (N-vinyl carbazole) polymers for dielectric capacitors by solution casting method, electrical conductivity results reveals that ‘σdc’ increases from 6.46 × 10 −16 to 3.63 × 10 −13 S/cm by increasing weight perctange of ZnO nanoparticles due to orientation polarization as well as that leads to increase charges in grain boundaries. 11
Interfacial polarization is another considerable aspect for dielectric property and it occurs by applying external field, it leads to high accumulation of charges and have high energy loss. Recent research reports that polymer based nanocomposites have gained high accumulation of charges with low energy loss. Fatima Ezzahra Bouharras et al. has investigated dielectric property of BaTiO3/fluorinated polymers dielectric results reveals that by increasing the concentration of dopant dielectric permittivity enhances and similarly it was also reported that the combination of polymers and dopant is an important factor for the dielectric property. 12 It was also exposed that fluorinated polymers such as PTFEA, PMMA, PtBa and PGMA doped with BaTiO3 have various level of dielectric property; it is evident that the composites combination is an important factor for dielectric property.
Recently researchers focus on electrical conductivity of polymer composites due to cost effective, flexibility and environmental stability. 13 Organic – inorganic composite have specific properties such as mechanical, chemical and electrical stability. From this composite organic material give high thermal stability and inorganic material helps for electrical conductivity by diffusing electrons. 14 Polyaniline is one of the conjugated polymers due to its enhanced electronic properties, simplistic synthesis process and non toxic. Addition of methyl group (-CH3) (Poly (O – Toluidine)) in the orth position of polyaniline makes it faster switching instance between oxidation and reduction. Arup Choudhury 15 has synthesized and characterized Poly (O – Toluidine) with multi walled carbon nanotubes (CNT)by in Situ chemical oxidation polymerization process and from the FTIR results it was evident that quinoid rings has increased significantly by adding CNT and molecular interactions such as π – π and acid - base interaction also increases, which leads to increase the electrical conductivity. Sirirat Wacharawichanant et al. 16 has studied thermal, mechanical and electrical properties of poly (vinyl alcohol) by doped with vanadium pentoxide (V2O5) and results reveal that thermal stability and electrical conductivity of the composite enhances by increasing the weight perctange of V2O5 due to better crystallinity and uniform dispersion of dopant. From these observations it is evident that fillers nature, size, surface characteristics and ability of dispersion affects the properties of the composites.
Despite these challenges, our present investigation focuses primarily on • Analyzing the POT - V2O5 nanocomposites capacity to store electrical energy by looking into its dielectric constant; this property is crucial for capacitors and other electronic parts. • To explore on the material’s ability to carry current under direct voltage by studying its DC conductivity.
POT -V2O5 nanocomposites was synthesized by in - situ chemical oxidative polymerization method with various weight perctange of V2O5 such as 25, 50 and 75 wt% respectively. Prepared samples were designated as PV25, PV50 and PV75 were “P” represents POT and “V” represents dopant weight perctange. Crystallinity, morphological, thermal stability, dielectric property and electrical conductivity (DC) were examined to find the suitability of the prepared samples for electronics application.
Experimental
Materials
The following are the materials used to synthesize samples, without any further purification I. O - Toluidine (OT) - CH3C6H4NH2 (UNSPSC CODE: 12352100) (≥ 99% purity – Sigma Aldrich) with purity used as monomer. (With Tg 150°C, processing temperature 300°C – 400°C) II. Potassium dichromate (K2Cr2O7) – (UNSPSC CODE: 12352305) (≥ 99.5% purity – Sigma Aldrich) used as oxidant. III. Hydrochloric Acid – (HCl) - (UNSPSC CODE: 12352301) (37%, for analysis EMPARTA® ACS) used as electron donor and catalyst. IV. Vanadium pentoxide (V2O5) Nano powder - (UNSPSC CODE: 12352300) (≥ 99% purity – Sigma Aldrich) used as dopant. V. Ethanol (C2H5OH) solution (UNSPSC CODE: 41116107) (≥ 99% purity – Sigma Aldrich) used for washing precipitate. VI. Deionized water is used for synthesizing and also for washing precipitate.
In Situ chemical polymerization process of synthesizing POT – nV2O5
POT – nV2O5 nanocomposites were synthesized by using In - Situ chemical polymerization method. O – Toluidine (OT) (monomer) has polymerized with potassium dichromate as oxidizing agent and HCl as catalyst in addition to ionic conductivity where V2O5 as dopant. Initially two solutions have taken separately such as OT (0.05 mol solution) and HCl mixed with potassium dichromate (0.01 mol), both solutions are kept separately. Specific weight perctange of nano vanadium pentoxide is mixed to OT solution and kept under stirring for 2 hrs; both solutions mixed together and yellowish precipitate is formed. The obtained precipitate is washed with distilled water and ethanol for several times and dried for 8 hrs.
Characterization
Crstallinity and size of the prepared samples were analyzed by using nondestructive technique known as X-ray diffraction analysis (XRD) (CuKα radiation (k = 1.54 A°), Bruker D8 Advance Twin-Twin), Polymeric and organic materials of the prepared composite, identified by Fourier Transform Infrared Spectroscopy (FTIR) - Thermo Nicolet iS50 4000 cm−1 to 100 cm−1. Surface morphology is analyzed by using Scanning Electron Microscopy (SEM) (Jeol 6390 LA- Oxford XMX N), Physio –Chemical character with the influence of temperature is analyzed by Thermogravimetric analysis (TGA) (Perkin Elmer – STA6000, - Diamond) Dielectric property and Electrical conductivity is measured to identify the suitability of the sample in energy storage and other electronics application with Direct current (DC) application, (Keithley Electrometer – 6500; 6430), >200 TΏ of impedance with charge measurement of 10 fC.
Result and discussion
X- ray diffraction analysis
X- ray diffraction pattern of the prepared samples were shown in Figure 1 and it is evident that the crystalline structures of three samples are not analogous and diverse in accordance with the compositional modifications. Peaks observed at 2θ = 26.79° 27.25°, 28.46°, 30.27°, 30.79° and 31.92° is attributed due to semi crystallinity . Vasant V. Chabukswar et al.
17
has investigated enchantment of Poly (O – Toluidine) doped with organic acid and XRD results shows that the periodic distance between dopant and nitrogen atom of the benzoid ring in POT leads to semi crystalline peaks at Bragg’s angle 0 – 27°, same reason is attributed for POT – nV2O5. Sharp peaks observed for PV50 and PV75 at 2θ = 30.27°, 30.79° and 31.92° which corresponds to (301), (010), and (011), may be ascribed due to regular chain arrangement of polymer chain by increasing weight perctange of dopant.
18
The attained XRD results of PV25 show an immaterial hump
19
and confirm the persistence of amorphous nature despite the addition of various amounts of n-V2O5. Intensity of reflections is found to be increased with progressive additions of n-V2O5. This in turn ensured the improved structural orientations in the composite and particle size is calculated by Scherrer equation. X-ray Diffraction pattern of POT – nV2O5 nanocomposites.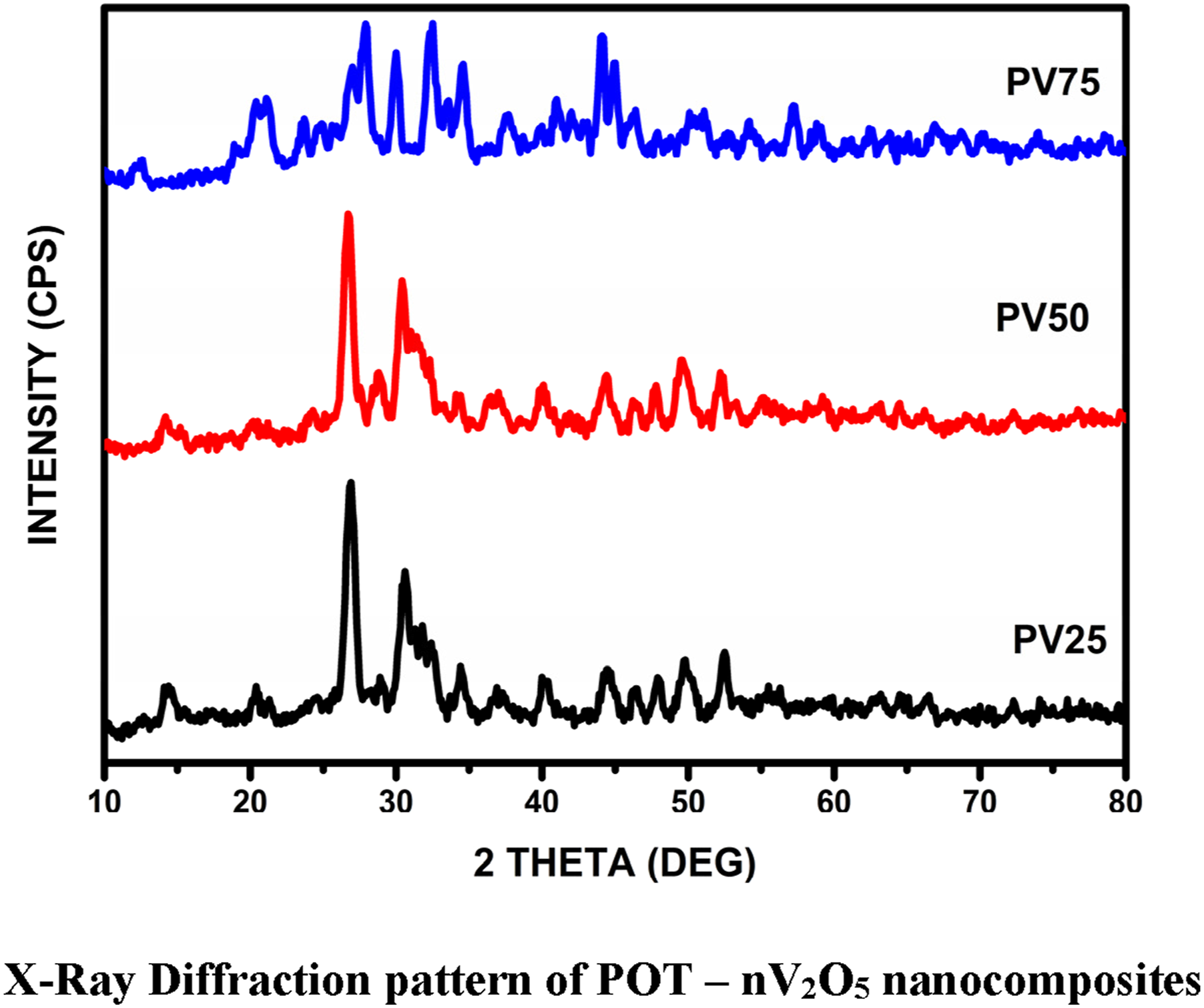
Since V2O5 is the promising intercalated material, it enlarges the van der Waals gap between the bonding and helps the V2O5 molecules to insert at regular intervals; some of the crystalline reflections are being sharper with an increased intensity while some other crystalline peaks are disappearing. Increase in sharpness of crystalline peaks in turn ensured the improvements in crystalline structure. Peaks at 34.25°, 44.1°, 48.25°, corresponds to plane (011), (002) and (411) for PV50 and PV75 are ascribed due to semi crystallinity. 20 The crystalline size of PV25, PV50 and PV75 were calculated as 40 nm, 28 nm and 12 nm respectively, by using Scherrer Equation. Presence of vanadium pentoxide in POT results in redox reaction and the same result was suggested by Magdalena Hasik et al. by doping PdCl2 with Poly (O – Toluidine). 21 Monika Jamdegni et al. 22 has investigated electrochemical performance of polyaniline doped with nickel oxide and XRD results reveals that peaks observed at 2θ = 44° - 62.8° is due to nickel oxide, same reason is attribute to PV75 for peaks observed at 2θ = 54.32°, 57.86° and 58.81° corresponds to (020), (021), and (221). Peaks observed at 58.81 are ascribed due to the better crystallinity of the composite. These observed data has good agreement with JCPDS Card No. 77-2418, 86-2248, 85-2422 and 89-2482.
Fourier - transform infrared spectroscopy
Fourier Transform Infrared spectroscopic analysis was employed on the prepared polymer nanocomposite samples and shown in Figure 2. The spectra explore the changes in the response of functional elements against IR irradiations. The functional behaviour of constituent elements in the spectra on the other hand, reveals the structural modifications. It is evident that the incorporated additive element (n-V2O5) and its quantity made significant changes in the structure of composite samples along with it records the reduction and increment in the absorption intensity against the n-V2O5 incorporations. FTIR spectrum of POT – nV2O5 nanocomposites.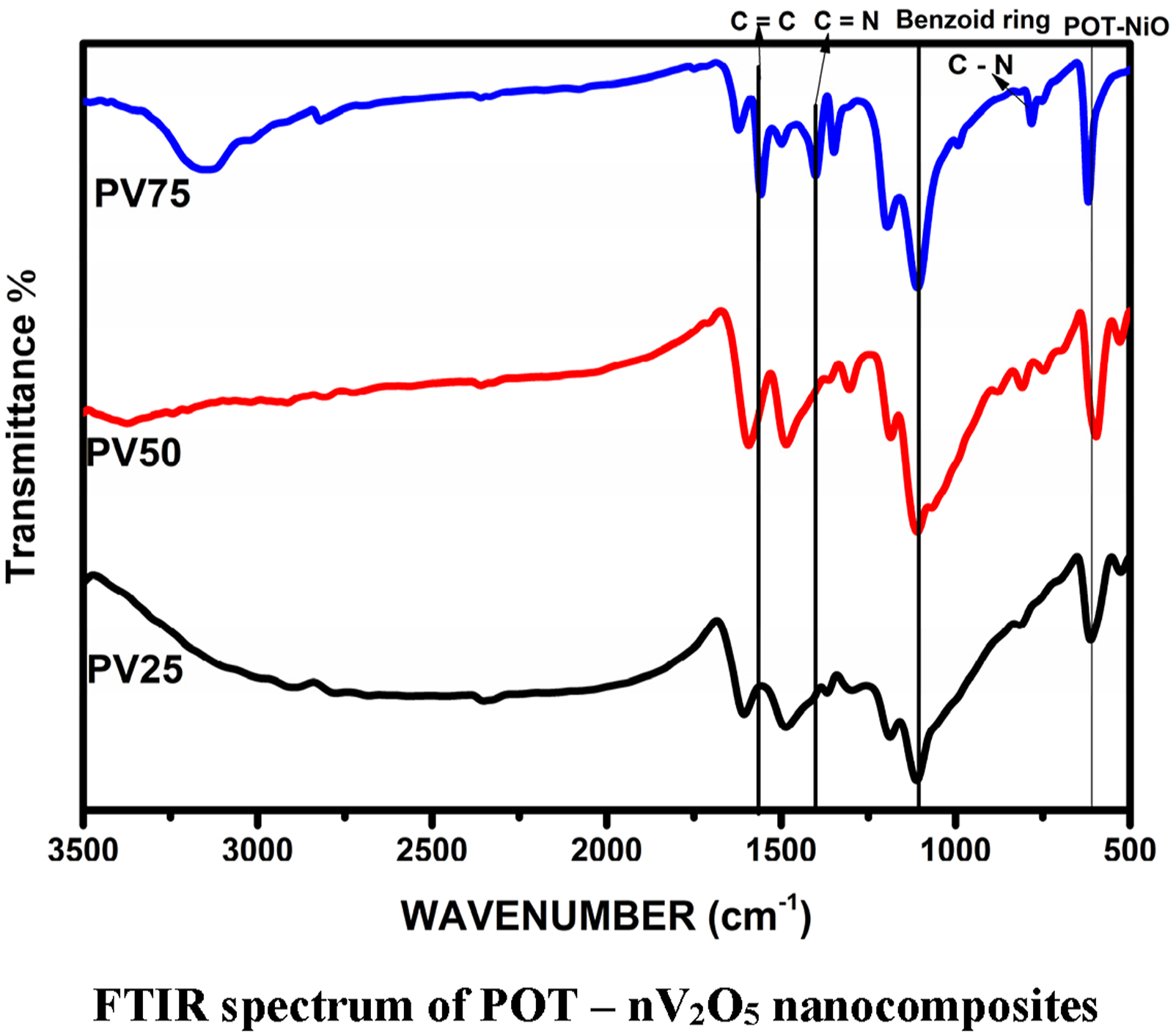
The emerged IR absorption at 593 cm−1 and 613 cm−1 in PV75 and PV50 ensured the presence of POT-V2O5 modes of symmetric and anti symmetric23,24 due to regularization of structural arrangements. Band observed below 1000 cm −1 is due to shielding of V2O5 in the polymer matrix. 25 C = C stretching band of quinoid ring is observed for PV75 at 1651 cm−1, 1600 cm−1 for PV50 and 1610 cm −1 for PV25. Sharp band is observed for PV75 comparing with other two samples.26,27 The spectrum shows C-N bonding for PV25, PV50 and PV75 at 1473 cm−1 1477 cm−1 and 1544 cm−1 respectively. 28 Bands observed at 1105 cm −1, 110 cm−1, 1194 cm−1, is attributes due to benzoid ring of POT. 29 POT- V2O5 blend is found at 609 cm−1 for PV25, 598 cm−1 for PV50 and 540 cm−1 for PV75, due to hybridization of vanadium pentoxide in POT. These results indicate the presence of POT and vanadium pentoxide in the prepared composite.
Thermogravimetric analysis (TGA)
Thermogravimetric analysis (TGA) was used to determine the thermal stability of the prepared nanocomposites. Thermal degradation and effect of temperature were analyzed under controlled temperature in N2 atmosphere. TGA analysis curves of the POT – nV2O5 undergoes weight loss in three steps as shown in Figure 3. The employed thermal analysis represents the response of each sample for the applied thermal energy from the ambient temperature (≈150°C) to ≥ 900°C. Thermogravimetric analysis (TGA) of prepared samples.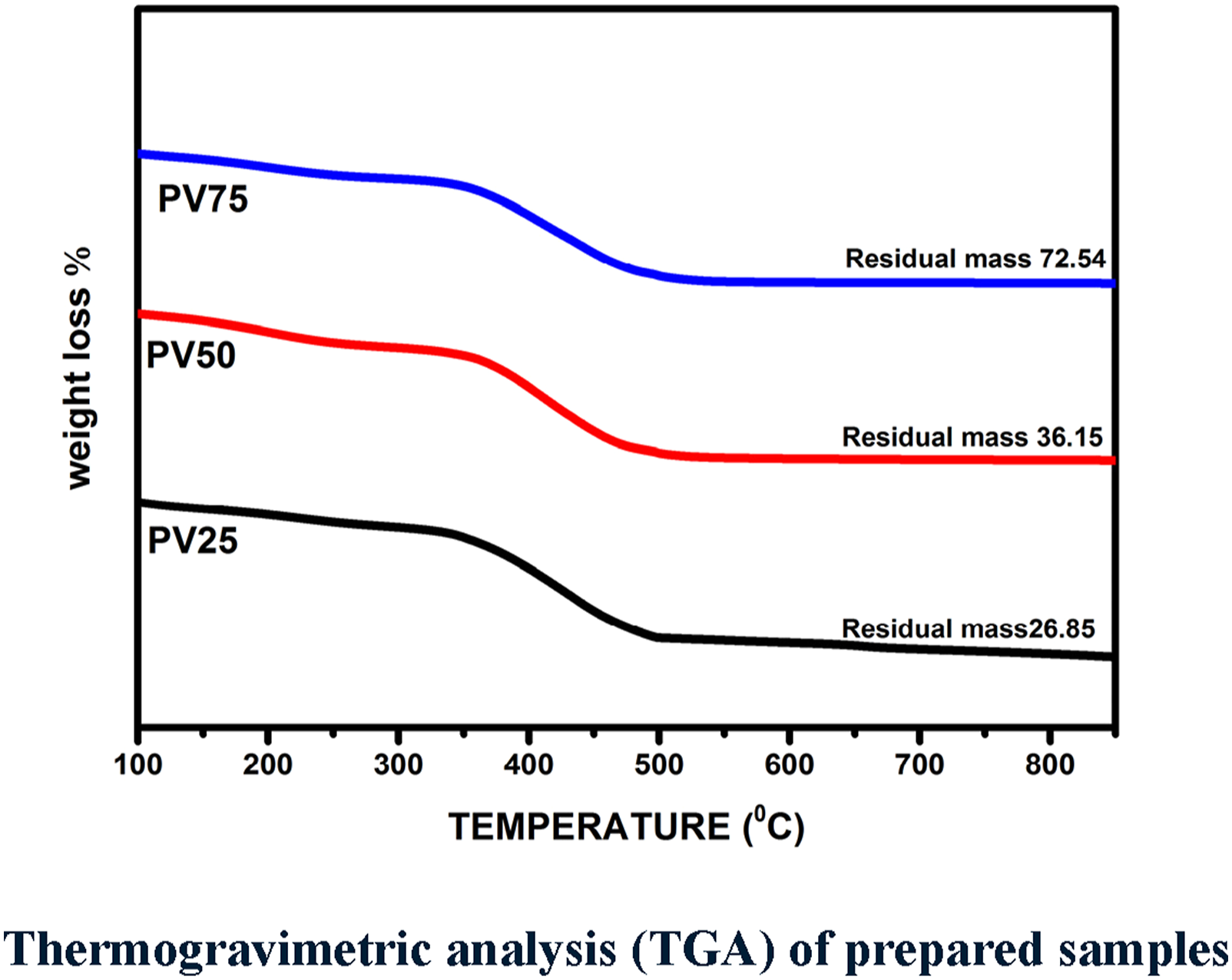
Initial weight loss of the samples starts from 150°C for PV25°C, 250°C for PV50 and 270°C for PV75 (80% higher than PV25) respectively due to dehydration of water molecules in POT structure. 30 R. Anbarasan et al. 31 has studied the effect of substituent’s and dopant on polyaniline structure and thermogravimetric results reveals that weight loss step at 400°C is due to the degradation of HCl and polyaniline (PANI), furthermore presence of – CH3 at meta position of PANI leads to low thermal stability, same reason is suggested for POT – nV2O5. Second stage of weight loss for PV25 ranges from 325°C to 485°C due to dopant and POT.
From the XRD and FTIR results it was evident that PV25 has low crystallinity and weak bonding, thus it is another factor for high degradation and less residual mass at low temperature (second stage). PV50 and PV75 has better crystallinity and more saturated bonds due to the presence of high weight perctange of vanadium pentoxide and it leads to strong thermal stability with better residual mass. Second stage of degradation was slow for PV50 (400°C – 507°C) and PV75 (475°C – 580°C) with broad temperature range due to removal of dopant and POT structure. 32 Third stage of degradation starts from 550°C for PV25 and 600°C for PV50, although PV75 have less degradation (670°C) due to high compatibility between POT and V2O5. 33 These result suggested that thermal stability of POT – nV2O5 enhances by increasing the weight perctange of V2O5.
SEM
Surface morphology of the prepared sample was investigated by SEM analysis and shown in Figure 4(a)–(c). Figure 4(a) shows surface morphological of PV25, with irregular scattering of vanadium pentoxide on the surface of POT. SEM images of PV50, Figure 4(b) reveals that V2O5 has well dispersed into the monomer (ortho toluidine) and forms POT – n V2O5 composites with better crystallinity by comparing with PV25, same results was suggested by Chuanbo Hu et al.
34
Crystalline aggregates are crowded with thickly packed arrangements of dopant in PV50. SEM micrographs of PV75 (Figure 4(c)) shows globular shaped particles through a group configuration of POT and dopant, it proves that dopant has inserted to the POT composition and form homogeneous nucleation.
35
V. B. Aaditya et al.
36
has studied dielectric properties of polypyrrole/titanium dioxide and polypyrrole/titanium dioxide-MWCNT nano composites and SEM photographs revels that by increasing dopant weight perctange cluster formation in polypyrrole enhances, same reason is attributed for PV75 nanocomposite. (a–c) SEM images of (a) PV25, (b) PV50 and (c) PV75.
Dielectric analysis
Dielectric permittivity
Dielectric permittivity Dielectric constant of PV25, PV50 and PV75 at 60°C and 120°C.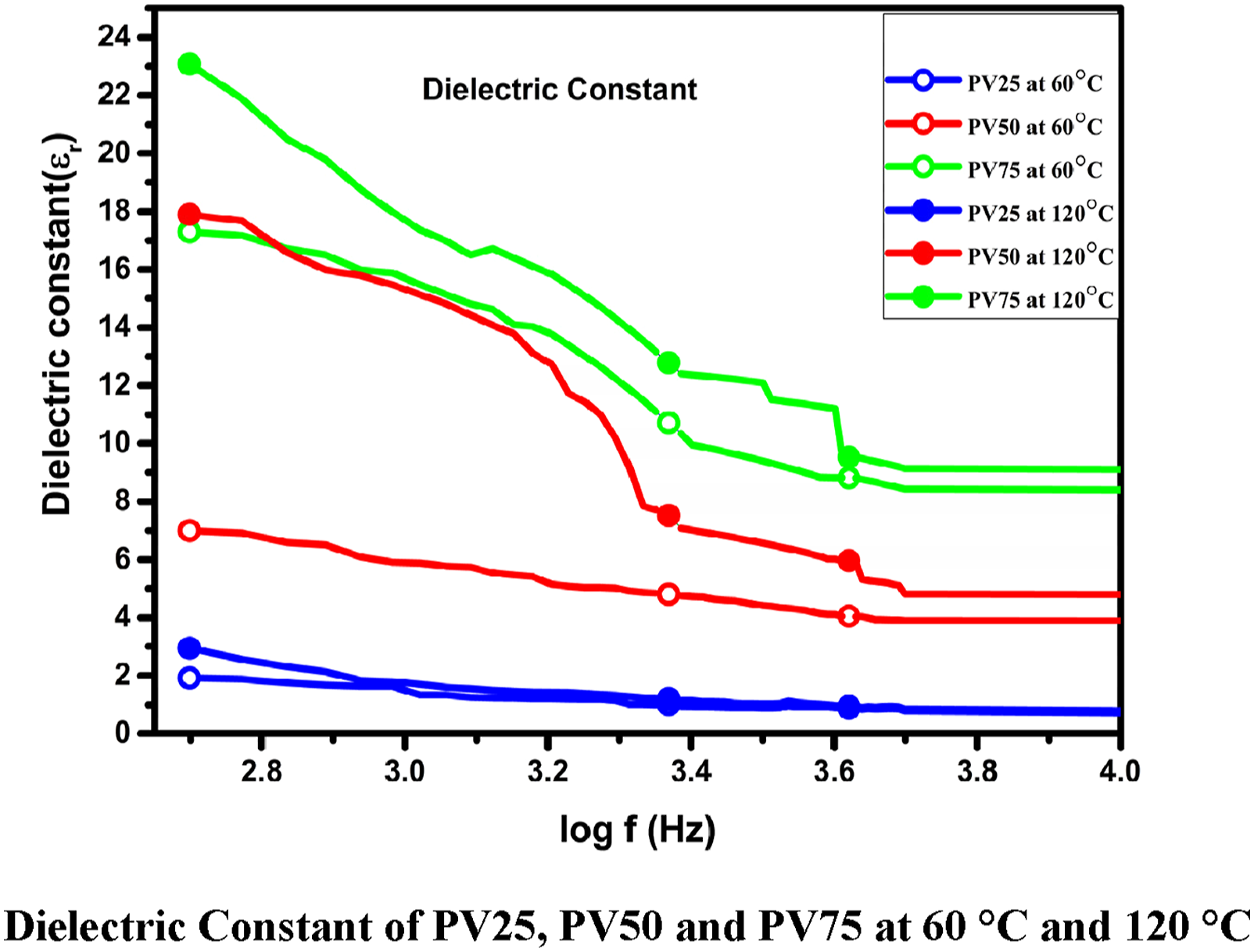
Adel M. El Sayedand Gomaa Khabir has investigated dielectric property of Polyvinyl pyrrolidone by doping Mg, Cu, Ni, or Cd) Acetates and dielectric results reveals that prepared samples increases due to charge transfer,
39
same results is attributed for PV75. Moreover from the XRD and SEM result it was evident that PV75 has better crystalline structure and improved homogenous distribution of dopant comparing with other two samples, which leads to develop polarization in the composite. Dielectric permittivity of the prepared sample is calculated by using equation
From the above equation, ‘C’ represents capacitance and ‘D’,
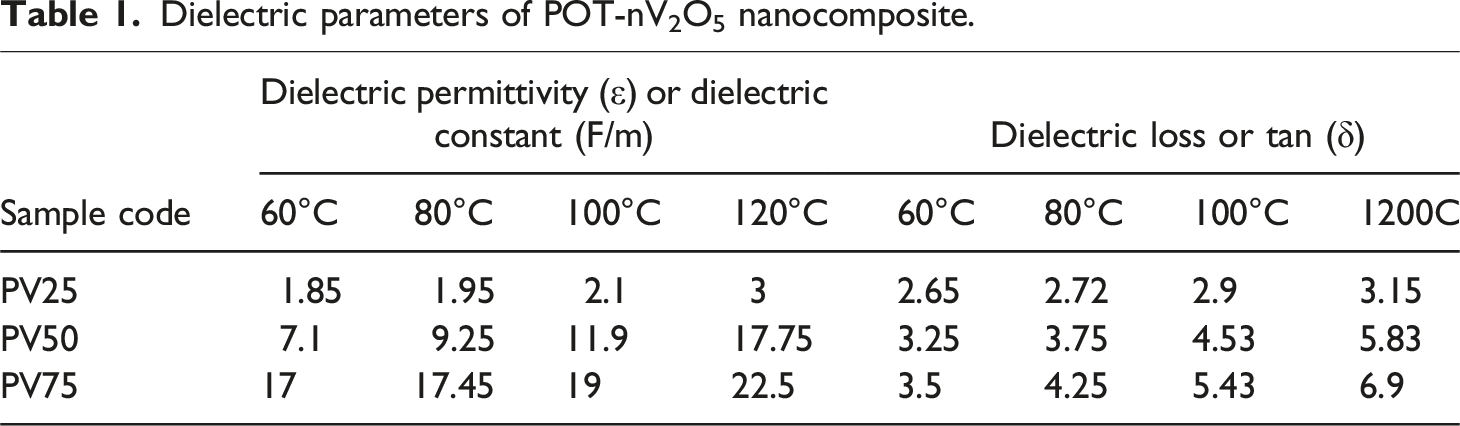
The permittivity of the prepared sample decreases at higher frequencies is due to decrease in polarization when polarons and bipolarons show lagging to the applied field at higher frequencies, Hemshankar Saha Roy et al. has suggested same reason. 40 Charge carriers (space charge) needs minimum time to form parallel with applied field. These charges starts to move randomly before the applied field changes its phases and does not form polarization, which leads to decrease the permittivity of the composite at higher frequencies. Dielectric permittivity of the prepared samples is tabulated in Table 2.
Dielectric loss (tan δ)
Dissipation factor or tan δ is measured for the prepared samples, to determine the amount of energy loss through applied voltage. Dependence of dielectric loss with function of frequency at 60°C and 120°C is shown in Figure 6. From the dielectric loss graph it was observed that tan δ varies with two reasons such as by dopant and other by temperature. Dielectric loss (tanδ) of PV25, PV50 and PV75 at 60°C and 120°C.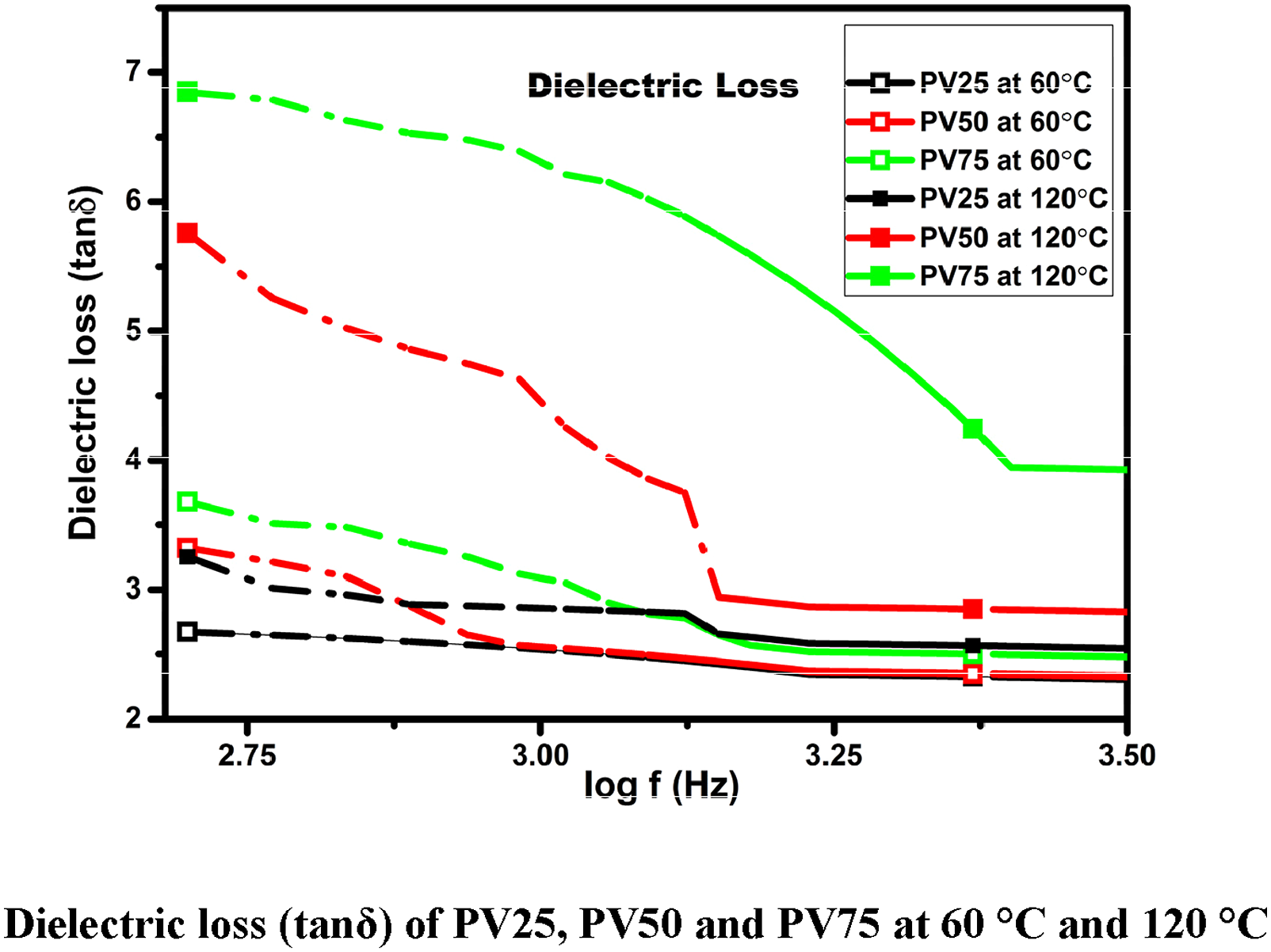
PV25 dielectric loss rises from 1.85 to 3 Hz by increasing temperature from 60°C to 120°C, PV50 records its dielectric loss from 7.1 to 17.75 Hz with same frequency and temperature range. Dielectric loss is calculated by using equation
Where ‘
PV75 has maximum loss of energy which ranges from 16 to 22.5 Hz with same frequency and temperature range of other two samples. It was observed that dielectric loss was decreasing at higher frequencies and it was attributed due to time lagging for the dipolar orientation of the prepared samples. Dopant addition optimizes dielectric loss and consequently improves surface area, polarization, and electrical conductivity. 41
Dielectric parameters of POT-nV2O5 nanocomposite.
Electrical conductivity (σdc)
The electrical conductivity (σdc) of the prepared samples were measured under various temperatures which starts from 60°C −120°C. POT synthesized with various weight perctange of vanadium pentoxide in different temperature results in increasing the electric conductivity. From the Figure 7 (shows electrical conductivity at 60°C and 120°C) it was evident that the prepared samples have increased its electrical conductivity by factors such as by dopant weight perctange and temperature. Ag2S, Ge-Te-Se, and other metal composites decrease the scattering cross-section as the dislocation density raises that result in increasing DC conductivity.43,44 Electrical conductivity for PV25 and PV50 at 60°C and 120°C.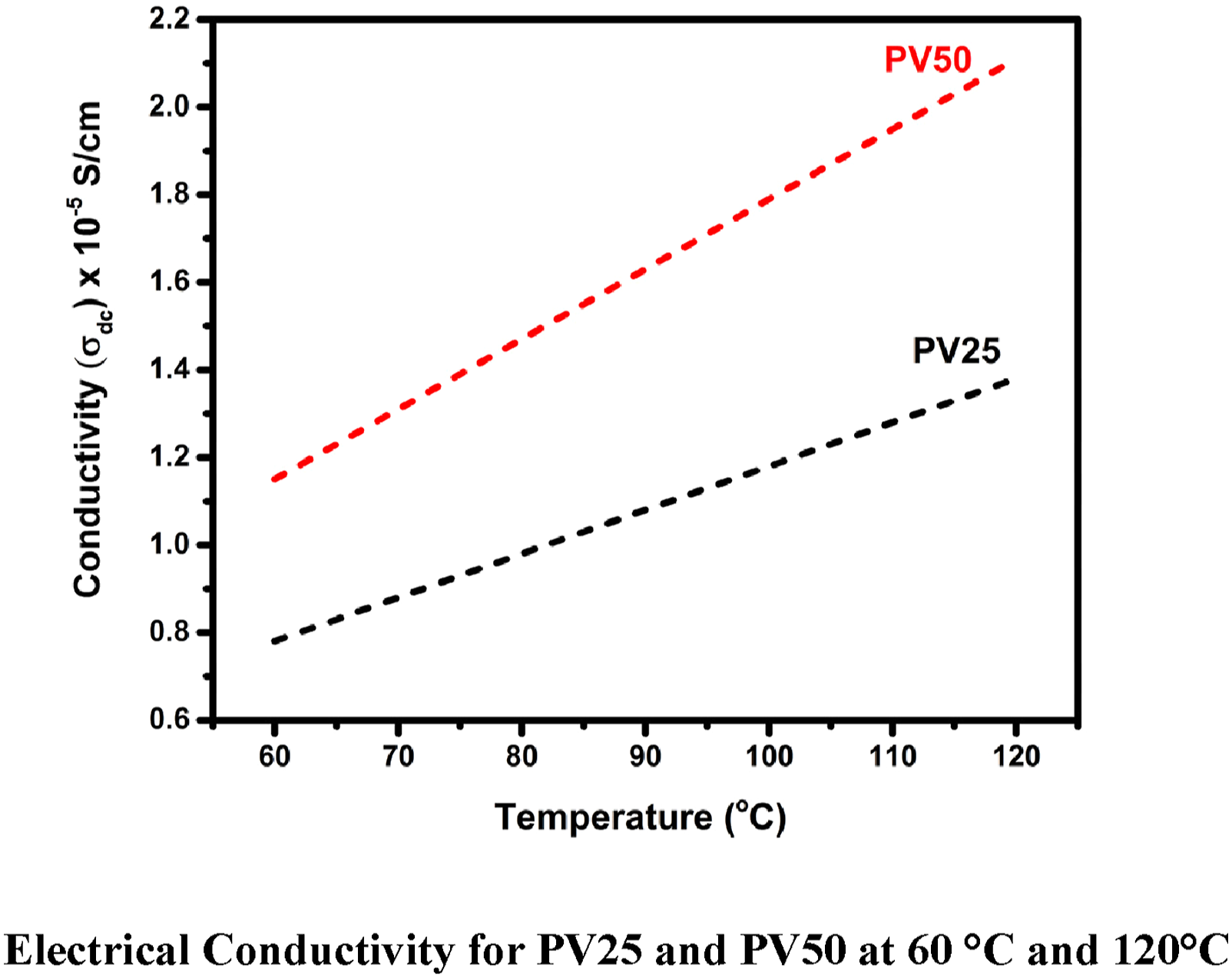

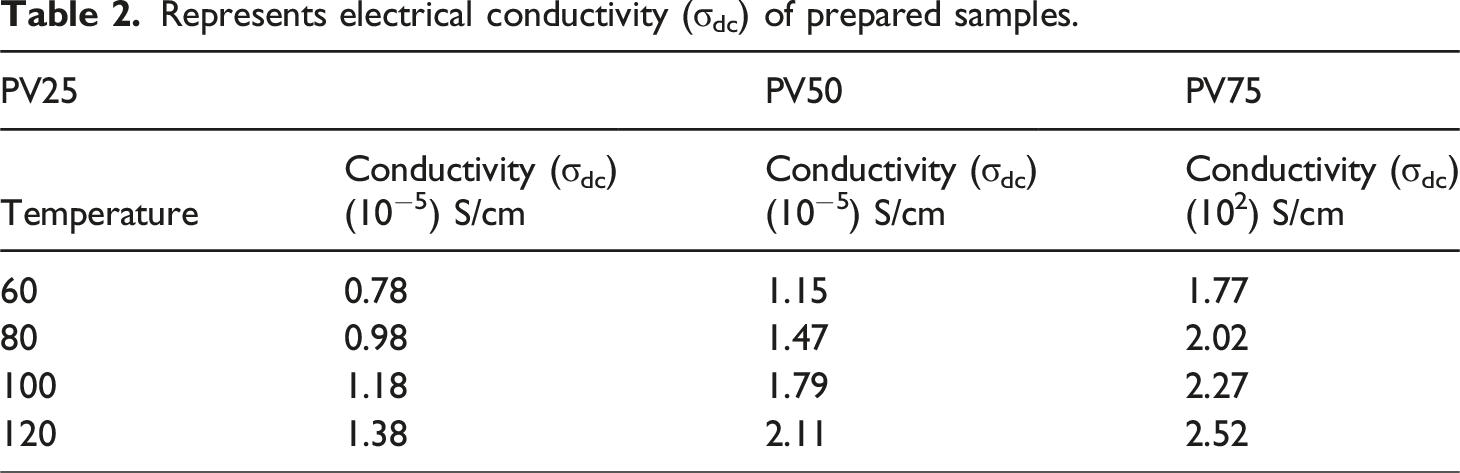
Represents electrical conductivity (σdc) of prepared samples.
From the above equation′
Doping of dopant creates polarons and bipolarons in the POT – nV2O5 nanocomposites by the influence of temperature applied to it. Electrostatic interaction occurs in the counter ions of the prepared composite and electrons are free to move along the chain of the POT structure, which leads to better conductivity. 22 PV75 has better crystallinity due to high weight perctange of vanadium pentoxide and it leads to more number of free electrons comparing with other samples, which results for high electrical conductivity. This observation agrees with XRD data, in which doping of 75 wt % of V2O5 increases the crystalline nature of the POT. According to the results of Ziadan KM et al. 47 and Khan AA 48 investigation into the DC electrical characteristic of POT, electrical conductivity increases with the addition of different dopants, such as Si and Sn(IV) tungstate, and reaches a maximum of 6 × 10−6 S/cm 3.29 × 10−2 S/cm. In the current report, it was found that adding 75 wt percent of nano vanadium pentoxide results in a maximum of 2.52 × 102 S/cm comparing these findings, the prepared composites exhibit a high capacity to carry current under direct voltage.
Comparative review of electrical conductivity.
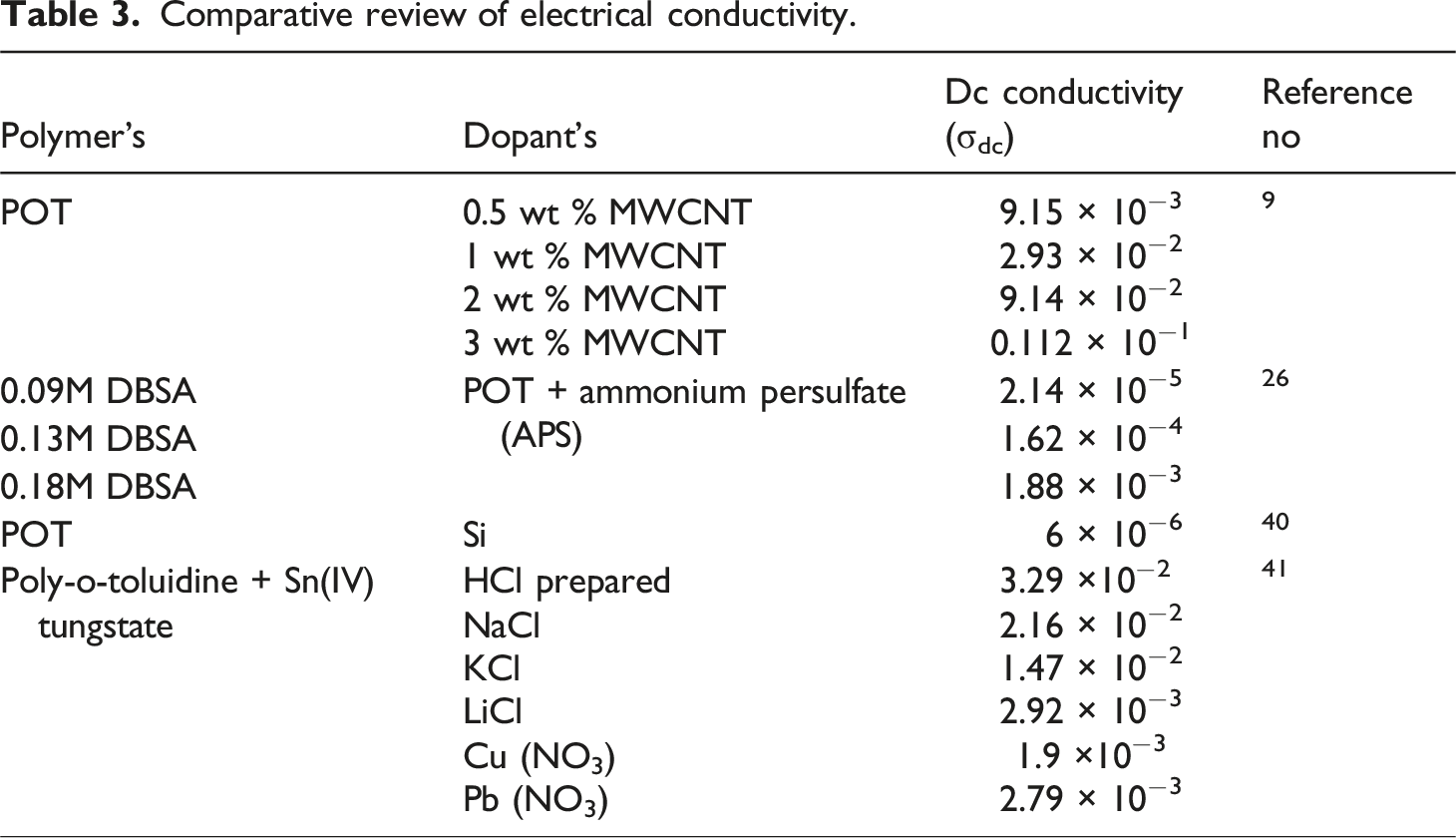
Dc conductivity (σdc) of the POT – nV2O5 nanocomposites increases significantly by increasing the weight perctange of dopant to the POT and PV75 has better conductivity, almost five orders of magnitude. Higher concentration of dopant in the POT creates well-built π - π* interaction between dopant and functional groups of POT. This strong interaction helps to delocalize electrons into the interfacial region and creates better conductivity to the prepared composites, as conformed from the FTIR analysis π - π* interaction between the functional groups was in better for PV75 comparing with other two samples. Arup Choudhury 9 has studied electrical property of POT doped with MWCNT and its results reveal that increasing the weight perctange of MWCNT’s gives better conductivity due to creation of energy transitions by polaron and bipolaron and acted as a conducting bridge for the motion of free charges along the chain of POT, same suggestion is attribute in our present study. Aljaafari et al. 49 has explored the mechanical and electrical characteristics of poly (vinyl chloride) loaded with carbon nanotubes and carbon nanopowder. Their outcomes suggested that though the weight percentage of carbon nanopowder increases, it enhances electrical conductivity by activating charge carriers and moving across filler materials. In accordance, Elham Aram et al. 50 study on the DC conductivity of a combination of poly (methyl methacrylate) and poly (ethylene oxide) by incorporating graphene nanosheets, charge transportation arise from a percolation threshold above the conducting networks. These aspects also influence our present work to improve DC electrical conductivity.
Figure 8 shows the electrical conductivity of PV75 and it turned out that the conductivity had increased substantially by 75%. This was ascribed due to electron transitions that were recently reported by Arup Choudhury et al. These findings reveal POT doped with 75 wt% vanadium pentoxide exhibits enhanced electrical conductivity than prior work, making it preferable for semiconductor applications. Electrical conductivity for PV75 at 60°C and 120°C.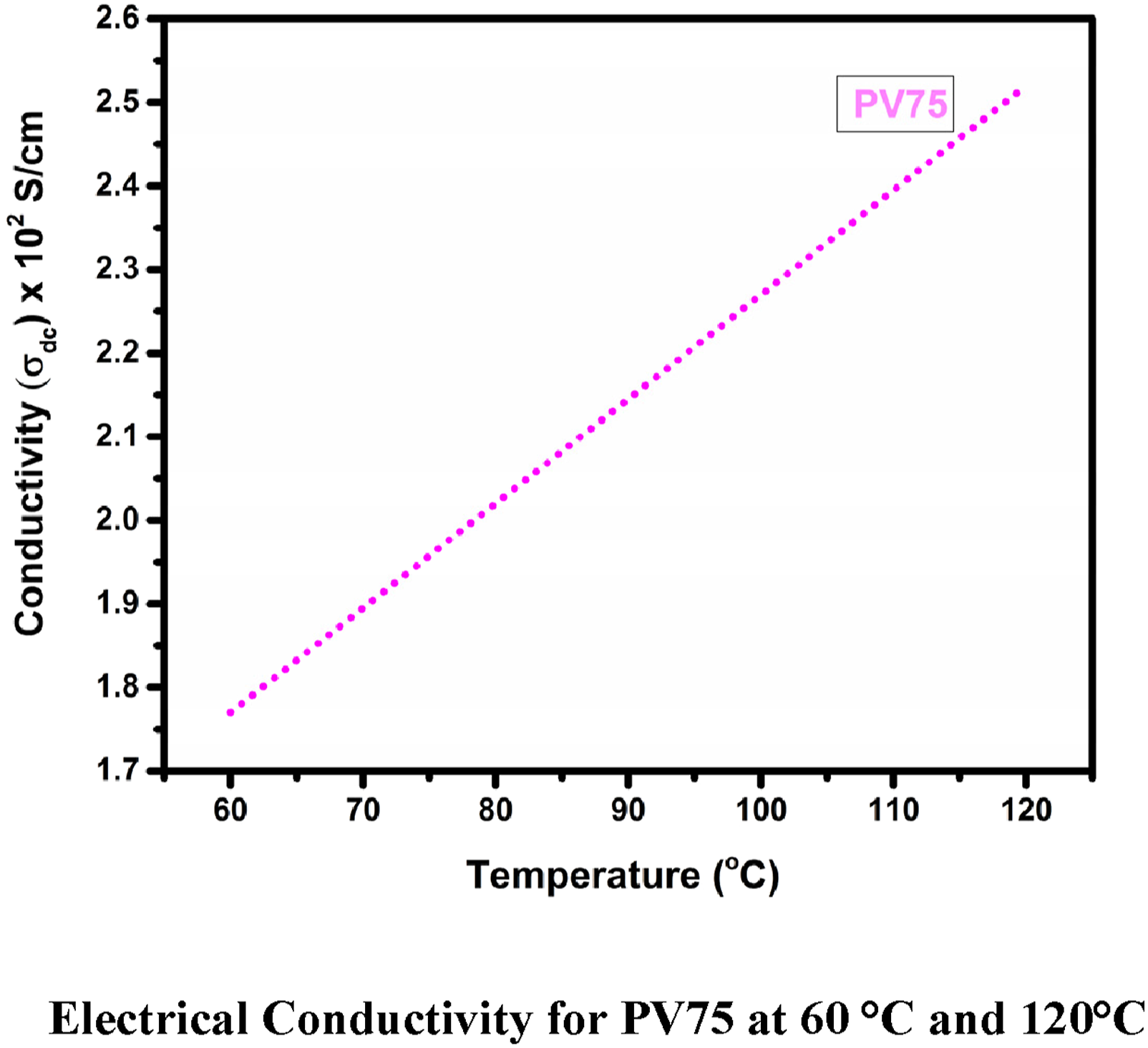
Conclusion
POT-nV2O5 was developed by using In - Situ chemical polymerization technique. XRD analysis reveals that the crystalline size decreased by 55% (40 nm to 12 nm), consequently furthermore PV75 exhibits better crystallinity with a strong peak at 2θ = 58.81°. It was observed from FTIR study that functional groups of the prepared composite, benzoid rings, quinoid rings and V2O5 were confirmed by observing peaks at 1105 cm−1, 1651 cm−1 and 540 cm−1. The nanoscale level scattering of V2O5 has been examined by SEM images, whereas PV25 exhibits an irregular particle structure with fewer clusters. TGA analysis shows that thermal stability was enhanced by 80% due to strong covalent bonds. Dielectric permittivity data show that increasing the weight percentage of V2O5 increases dielectric permittivity by 7 times, PV25 shows 3 F/m, while PV75 shows 22.5 F/m it was due to electronic and ionic polarization. Dielectric loss doubles from 3.15 to 6.9 as temperature and dopant weight percentage increases. This is the result as multiple free charges travel to the interfacial area. The creation of polarons and bipolarons as a result of temperature and dopant weight perctange causes electrical conductivity to increase by 75%, from 2.08× (10−5) S/cm to 2.21× (102) S/cm. The raise in dielectric constant and electrical conductivity shows sample has suitability for energy storage (capacitors) and other electronic devices.
Footnotes
Acknowledgements
We express our gratitude to the management of the KGISL Institute of Technology, Coimbatore - Tamilnadu - India for delivering us with the research lab. resources.
Author contributions
The idea and methodology was developed by Praveen.H and also contributed to draft the manuscript. V. Girish chandran has contributed and supervised, both authors have reviewed the final version of the manuscript and approved for the further process
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


