Abstract
With the advent of the 5G era, thermal management materials for electronic packaging are evolving towards high thermal conductivity, good thermal expansion matching, low density, and integrated functional structures. Copper-carbon composite materials, leveraging the advantages of copper such as high strength, hardness, electrical and thermal conductivity, as well as the excellent mechanical, electrical, and thermal properties of carbon materials, have become one of the focal points in thermal management material research. This article provides an overview of the research progress on copper-carbon composite materials, highlighting the characteristics of different carbon materials (Diamond, Carbon nanotubes, Graphene, Graphite flakes, Graphite films, Carbon fibers) in combination with copper, as well as the preparation methods for copper-carbon composites. At present, the thermal conductivity can be increased to more than 600 W/(m·K), and the preparation method is hot pressing sintering. It discusses the main issues and control methods encountered in the preparation and application of copper-carbon composites and presents prospects for the future development of these materials, offering references for the research and preparation of copper-carbon composites.
Keywords
Introduction
With the advancement of modern electronic information technology industry, miniaturization, portability, and multifunctionality have become prominent features of electronic products. As the device assembly density continues to increase, the heat flux density has also rapidly grown. To efficiently dissipate the substantial amount of heat generated during device operation, thermal management materials for electronic packaging need to possess higher thermal conductivity. The development of thermal management materials for electronic packaging can be categorized into three stages. In the first stage, materials with relatively higher thermal conductivity and density, such as W-Cu, Mo-Cu, fusible alloys, copper, and aluminum, were utilized. 1 In the second stage, materials exhibited a noticeable enhancement in thermal conductivity, with a simultaneous reduction in thermal expansion coefficient to closely match that of semiconductor silicon. This stage mainly involved graphite-reinforced polymer matrix composites, continuous carbon fiber composites, Al/Si composites, and the like. 2 In the third stage, which has gained increasing research attention in recent years, efforts have focused on optimizing the performance of carbon material-reinforced metal matrix composites to achieve higher thermal conductivity and lower thermal expansion coefficient.
Copper-carbon composites possess outstanding properties, combining the strength, hardness, and electrical conductivity of copper with the self-lubricating and high thermal conductivity characteristics of carbon materials. Additionally, they exhibit low thermal expansion coefficient and high melting point. Moreover, they demonstrate excellent resistance to soldering and arc erosion, making them widely used in various fields such as friction materials, pantograph sliders, brushes, oil-impregnated bearings, electrical contact materials, electronic packaging materials, conductive materials, and mechanical components. Although the use of powder metallurgy enables effective bonding between copper and carbon materials, the weak interface strength between them can negatively impact their performance. To address this issue, current domestic and international research primarily focuses on improving the wettability of the copper-carbon interface through two approaches. The first approach involves alloying the matrix, wherein alloying elements are added to the matrix to enhance the wettability of copper and carbon. 3 Zhang R et al. 4 successfully prepared flake graphite/copper composites containing Zr element through vacuum hot pressing. The introduction of Zr element improved the wettability between graphite and copper. The bending strength in the X-Y direction gradually increased by 90% from 50 MPa to 95 MPa with increasing Zr content from 0 to 2.0% (mass fraction). The thermal conductivity in the X-Y plane exhibited an increasing trend followed by a decreasing trend, reaching a peak value of 640 W/(m·K) at 1.0% (mass fraction) Zr content, thereby demonstrating the feasibility of matrix alloying. The second approach involves surface treatment of carbon materials, wherein chemical plating, vapor deposition, or salt bath plating methods are employed to improve the bonding strength and dispersibility between carbon materials and copper. 5 Bai H et al. 6 improved the wettability between graphite and copper by synthesizing boron carbide on the graphite surface. High-speed mixing was used to ensure the uniform distribution of copper on the graphite surface, and reciprocating vibration was introduced to achieve perfect orientation of graphite in the composite material. The resulting composite material exhibited uniform dispersion and ordered arrangement of graphite, and when the graphite volume fraction was 70%, it demonstrated excellent thermal conductivity (676 W/(m·K)) and appropriate thermal expansion coefficient (7.1 ppm/K). The use of high-speed mixing ensured the uniform distribution of copper on the graphite surface, and the introduction of reciprocating vibration allowed for perfect orientation of graphite in the composite material. Furthermore, the type and content of appropriate additives can also improve the thermal and electrical conductivity as well as the mechanical properties of the material. The main additives currently studied are metallic elements and carbides.
The development of modification research on copper-carbon composites primarily focuses on interface control, additives, and fiber reinforcement.
Metal-based heat dissipation materials are widely used in the heat dissipation industry of electronic devices due to their excellent heat transfer performance and easy processing. 7 Among them, the thermal conductivity of Cu is about 400 W/(m⋅K), which is suitable as a heat dissipation material for high thermal conductivity electronic chips. However, Cu is limited in application due to its high density, limited thermal conductivity, high thermal expansion coefficient, and deformation and cracking caused by external forces. Carbon materials, including carbon fibers, carbon nanotubes, and graphene, have been gradually applied to the field of heat dissipation of electronic products due to their unique structure, high thermal conductivity, light weight, and high strength. 8 The composite material combines the advantages of various materials, which can effectively improve the performance of single-phase materials. Metal copper materials and carbon materials have good applications in thermal conductive materials. The combination of the two can combine the advantages of the two, greatly improve the thermal conductivity of the composite material, and meet the development trend of highly integrated and high-power electronic devices. Therefore, this paper reviews the research methods of copper-carbon composites, and provides reference for the research and preparation of copper-carbon composites, in order to better apply to the field of electronic packaging thermal management materials.
Study on copper-carbon composites
Copper, as one of the earliest metals used by human beings, is rich in mineral resources and mature in smelting technology. It has good ductility and extremely high thermal conductivity (about 400 W/(m·K)). Its thermal conductivity is the highest metal after the precious metal, and the metal copper also has excellent mechanical properties. Therefore, the use of copper as the metal matrix of the composite material can not only ensure the high thermal conductivity of the material, but also improve the mechanical properties of the composite material, thereby improving the overall performance of the composite material. At present, copper matrix composites have been widely used in daily life as high thermal conductivity and conductive materials. In order to improve their performance in some aspects or some aspects, it is usually necessary to add specific reinforcing phases to improve them. Common reinforcing phases include carbides (such as silicon carbide), carbon materials (such as diamond, graphene, carbon fiber, graphite sheet, etc.) and metals (such as tungsten, molybdenum). Among them, carbon materials have extremely high thermal conductivity and excellent mechanical properties, and have many allotropes. Therefore, carbon materials reinforced copper matrix composites have received extensive attention. It mainly includes Diamond/copper composites (D/Cu), Carbon nanotube/copper composites (CNT/Cu), Graphene/copper composites (GNS/Cu), Graphite flake/copper composites, Graphite film/copper composites, Carbon fiber/copper composites (CF/Cu), etc.
Copper-carbon composites were first reported in the early 20th century, which are metal matrix composites composed of copper matrix and carbon components. The amount of literature published has been on the rise since 1981. The main topics of research are copper matrix composites, graphene composites, graphene oxide, nanocomposites, etc. The performance mainly focuses on mechanical properties, friction coefficient, friction and wear, etc. The research disciplines include materials science, chemistry and power industry, mainly focusing on technical research and application basic research.
In recent years, researchers at home and abroad have done a lot of meaningful research work to improve the thermal conductivity of copper-carbon composites. At present, the research of copper-carbon composites is mainly focused on two aspects. On the one hand, carbon materials (flake graphite, graphene, carbon fiber, etc.) exhibit obvious anisotropy due to structural anisotropy. Therefore, this characteristic can be used to prepare anisotropic composite materials to meet the directional heat transfer requirements in some special device structures. At the same time, the morphology, size, dispersion, volume fraction and orientation of carbon materials in metal matrix will affect the properties of composites.9–11 Lu Y et al. 12 in order to study the influence of graphite angle on thermal conductivity and thermal expansion coefficient, the thermal properties of graphite reinforced Cu matrix composites were calculated by using the simulation program which can consider the cross section angle of graphite. At the same time, due to the hot pressing effect in the process of spark plasma sintering (SPS), the orientation of graphite tends to be parallel distribution, indicating that the addition of graphite with improved orientation distribution can improve the thermal conductivity of Cu matrix composites. On the other hand, it is difficult for carbon materials to react with metals and not wet, which leads to serious interface problems and affects the properties of composites. The researchers optimized the composite material through the preparation process, 13 and also modified the surface of the carbon material, that is, Ti,14,15 Cu,16,17 Ni, 5 Cr,18,19 B, 20 Wu 21 and other plating treatments on the surface of the carbon material. Improve the wettability between carbon materials and copper. Or add one or more metal elements to the metal matrix, such as Ti, 14 Cr, 22 Ta 23 and other elements. These elements react with carbon to form a carbide transition layer, which changes the chemical composition of the matrix, thereby reducing the surface tension and improving the wettability of the reinforcing phase and the matrix. The effects of material coating type and thickness, additive type and content on the mechanical properties, electrical properties, tribological properties and thermal properties of the composites were studied.
Diamond/copper composites (D/Cu)
Diamond is an allotrope of carbon, which is composed of a regular tetrahedral structure of a cubic crystal. Each carbon atom forms a covalent bond with the other four carbon atoms in a sp3 hybrid orbital. Therefore, diamond has excellent physical properties such as high density, high hardness, high melting point, high strength and excellent thermal conductivity (thermal conductivity up to 2000 W/(m·K)). With the continuous improvement of the preparation technology of synthetic diamond, diamond has gradually become a hot spot in the field of material science. In the field of thermal management materials for electronic packaging, chemical vapor deposited diamond is mainly used for cooling small-scale chips, while granular diamond is used as a reinforcing phase material for metal matrix composites.
However, the formation of composites between diamond and metal matrix faces the challenges of poor wettability between carbon and metal and the machinability of composites. Therefore, researchers are committed to exploring suitable process methods to better apply the excellent properties of diamond. (1) Change the particle size and content of diamond. Xie ZN et al.
24
prepared bimodal diamond particles reinforced diamond/copper composites by vacuum pressure infiltration. The effects of diamond volume fraction on the microstructure and thermal properties of the composites were studied. The results show that the thermal expansion coefficient of diamond/copper composites at 50°C is 3.8–5.8 ppm/K due to the high diamond content (63–81.8 vol%) and strong interfacial bonding strength. In the range of 50∼300°C, the change of thermal expansion coefficient of diamond/copper composites is less than 2 ppm/K. An appropriate amount of diamond addition can obtain a thermal expansion coefficient matching the semiconductor material and a high dimensional stability of the diamond/copper composite material. In order to develop diamond/copper composites with high thermal conductivity, Xu B et al.
25
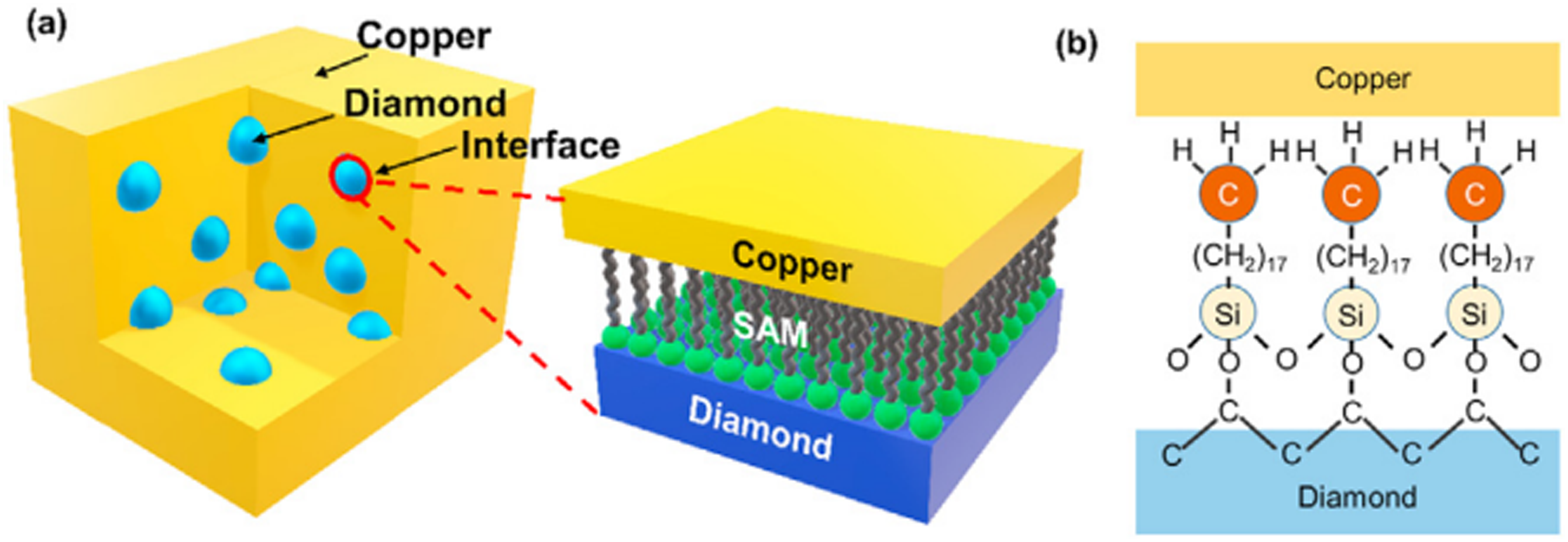
adopted an unconventional method to enhance the thermal boundary conductance (TBC) between copper and diamond by using self-assembled monolayer (SAM) before high-temperature sintering of diamond/copper composites (The schematic diagram is shown in Figure 1). It was found that the diamond particles with a size of 210 μm were functionalized by SAM at the same time. Under this condition, the TBC in the model system was the highest, and the isotropic diamond/copper composite with a volume fraction of 50% was prepared by sintering with copper. At room temperature, the measured thermal conductivity is 711 W/(m·K), which is the highest value among diamond particles with similar volume fraction and size. This work demonstrates a new strategy to enhance the thermal conductivity of composites by SAM functionalization. (1) Matrix alloying. In addition, there is also a method to prepare diamond composites with zirconium-doped copper matrix by gas pressure infiltration, and the thermal conductivity can reach up to 930 W/(m·K).
26
At present, the heat conduction effect is good. (2) Diamond surface modification. Jiao ZK et al.
27
proposed a diamond skeleton as a reinforcing element, rather than dispersed diamond particles. In order to improve the wettability of the interface between diamond and copper, vacuum evaporation technology was used to sputter metal tungsten transition layer on the surface of diamond to prepare composite materials. The experiment improves the interfacial bonding of the composites, so that the thermal conductivity of the composites reaches 575 W/(m·K). In order to reveal the intrinsic reason why the theoretical and experimental values of thermal conductivity of diamond/copper composites show the opposite trend with the increase of nano-interface layer thickness, In order to reveal the intrinsic reason why the theoretical and experimental values of thermal conductivity of diamond/copper composites show the opposite trend with the increase of nano-interface layer thickness, Sang JQ et al.
28
sputtered W layer on diamond substrate by magnetron sputtering and vacuum annealing, and prepared WC layer with a thickness of 70∼400 nm. At the same time, the corresponding diamond/copper composites were prepared by pressure-assisted infiltration method. The maximum thermal conductivity (TC) of the composites reaches 943 W/(m·K). It can be seen from the above quantitative analysis that the thermal boundary conductance of WC/Cu and diamond/WC interfaces is the main factor leading to the change of TC with the increase of WC interlayer thickness. Optimizing the high thermal boundary conductance by adjusting the thickness of the interlayer is a key factor to improve the thermal conductivity of the composite. Schematics of (a) copper/SAM/diamond composite and the zoomed up morphology of the SAM-functionalized interface; (b) Chemical structure of copper/SAM/diamond interface.
25
Carbon nanotube/copper composite material (CNT/Cu)
Carbon nanotubes (CNT) have been used to enhance the thermal conductivity of composites due to their high thermal conductivity, high strength, low density, special electronic structure and high stability. 29 The shape of CNT is similar to that of long and thin carbon fiber, which is actually a fullerene tube. The tube wall is a hexagonal plane cylindrical surface formed by sp2 hybrid carbon atoms, which is divided into single-walled carbon nanotubes or multi-walled carbon nanotubes. The mechanical properties, thermal properties and dielectric properties of the composites can be improved by embedding CNT into the matrix of the composites.
However, CNT has the problem of self-aggregation and is difficult to be uniformly dispersed in the matrix. Therefore, it is necessary to adopt a suitable dispersion method to align the orientation of CNT, and use its anisotropic characteristics to achieve the best performance of the composite material in a specific direction. Rajyashree S et al.
30
studied the coefficient of thermal expansion (CTE) of CNT/Cu macroscopic filamentary composites and its effect on the degree of CNT-Cu mixing. It was found that the CTE of the composite prepared by uniformly mixing 40 vol% nanotubes in a continuous Cu matrix was 4.42 × 10−6°C. This value is consistent with Turner ‘s model prediction, indicating that the low thermal expansion and high bulk modulus of CNTs help to offset the thermal expansion of Cu when the nanotube-Cu common contact is high. On the contrary, the non-uniform CNT-Cu mixing reduces the common interface, even if the CNT vol% is higher, it will lead to higher CTEs. In order to improve the performance of the composite material, the CNT was coated with a metal coating.
31
Some studies have modified nickel nanoparticles on CNTs to improve the interfacial bonding between carbon nanotubes and copper. Ni-modified composites form a wider interfacial transition zone between CNTs and copper, and form Ni3C interface products. At the same time, the conductivity (95.6% IACS), tensile strength (364.9 MPa) and elongation (40.6%) are improved.
32
Shin A et al.
33
analyzed the microstructure and thermal properties of CNT/Cu composites prepared by chemical mixing method, and compared them with CNT/Cu nanocomposites prepared by traditional mechanical mixing method and wet mixing method. The electroless copper plating on the surface of a single CNT makes it embedded in the copper particles, which successfully solves the problem of uneven dispersion of CNTs in the Cu matrix, which is the most important problem in the preparation of CNT/Cu nanocomposites. The CNF in the CNT/Cu nanocomposites prepared by chemical mixing method is well dispersed, and there are no pores and mesophases in the composites. The thermal conductivity (435 W/(m·K)) of the CNT/Cu nanocomposites prepared by chemical mixing method is much higher than that of the nanocomposites prepared by mechanical mixing and wet mixing. Besides, Yan F et al.
34
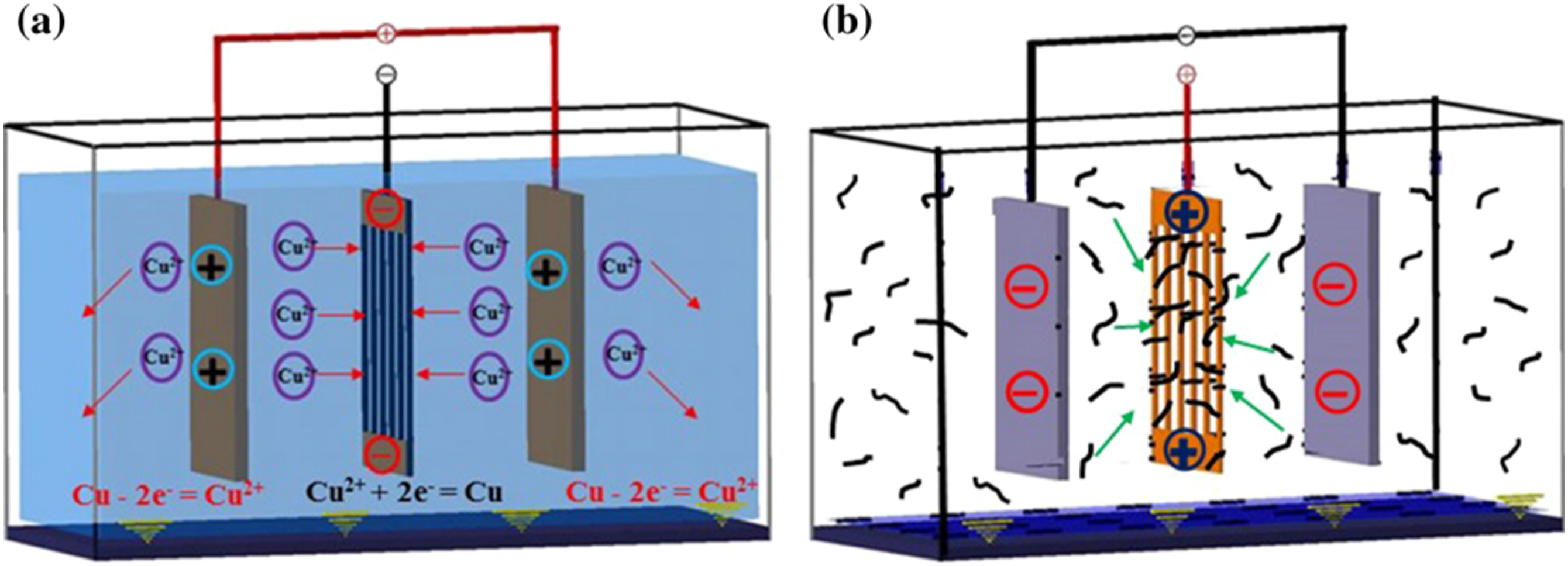
proposed a simple electrophoretic deposition (EPD) method (as shown in Figure 2) to deposit Cu and CNTs on the surface of carbon fiber (CF) to improve the thermal conductivity and interfacial properties of carbon fiber reinforced polymer (CFRP) composites. In the device, the conductive copper strip is attached to both ends of the CF, so that uniform deposition can be achieved. In the two-electrode system, CF and Cu sheets act as cathode and anode, respectively, and react on them: Schematic illustration of the EPD process for a Cu-CF and b CNTs-Cu-CF.
34
Graphene/copper composites (GNS/Cu)
Graphene (GNS) is a one-or multi-layer planar densely packed carbon nanomaterial with a hexagonal honeycomb lattice connected by sp2 hybrid orbitals. It was prepared by mechanical exfoliation in 2004 by Professor Andre Geim and Kostya Novoselo researchers at the University of Manchester in the United Kingdom. It exhibits extremely high thermal conductivity (3080–5300 W/(m·K)), high electron mobility and low resistivity. 35 Monolayer or multilayer graphene nanosheets (GNS) exhibit excellent electrical and thermal properties, which makes it have great potential in the field of batteries and thermal management materials.
Recently, many studies have emerged on the use of GNS reinforced metal-based or polymer-based heat dissipation composites,
36
and have shown an increasingly broad application prospect. Liu B et al.
37
first proposed and constructed a new electronic thermal conduction path for graphene metal matrix composites. Conjugated 4-ethynylaniline functionalized graphene (FGr) was used to prepare FGr/Cu composites by pulsed co-electrodeposition of FGr onto copper surface. The prepared FGr/Cu composites can maintain excellent and stable thermal conductivity at high temperature, and the thermal conductivity at 100°C and 150°C is 1.61 and 1.31 times that of Cu, respectively. The delocalized conjugated p-bond in graphene is bridged by graphene and metal with conjugated organic molecules with p-orbitals, and the Cu interface is successfully established, thus constructing a new electronic heat conduction pathway. This innovation provides a new direction for the development of graphene-based metal matrix composites with efficient heat dissipation. In order to further improve the thermal conductivity of GNS/Cu composites, nitrogen doping or surface modification of graphene nanosheets has also been studied.
38
Obtaining a close and strong interface is crucial to improve the load transfer capacity and strength of graphene/metal composites. Chu K et al.
39
talloyed Ti matrix. The results show that Ti8C5 nanolayers and Ti8C5 nanoparticles are in-situ formed at the local interface of rGO/Cu-Ti composites, which play a role of “rivet” and enhance the interface bonding between rGO and Cu-Ti matrix. The formed interfacial Ti8C5 nanolayer and Ti8C5 nanoparticles are derived from the amorphous carbon nanolayer and amorphous carbon nanoparticles on the rGO surface, respectively. The excellent orientation of graphene is the key to improve the properties of composite materials, Nazeer F et al.
40
prepared copper-reduced graphene oxide composites by a new method of layer-by-layer self-assembly combined with hot pressing sintering. At a very low concentration of reduced graphene oxide (0.8 vol%), the reduced graphene oxide/copper composite obtained an abnormally high anisotropic thermal conductivity, and the difference between the in-plane and inter-plane thermal conductivity was 8.82 times. The laser test results confirm the highly anisotropic behavior of the reduced graphene oxide/copper composite and its remarkable heat dissipation performance. Chu K et al.
41
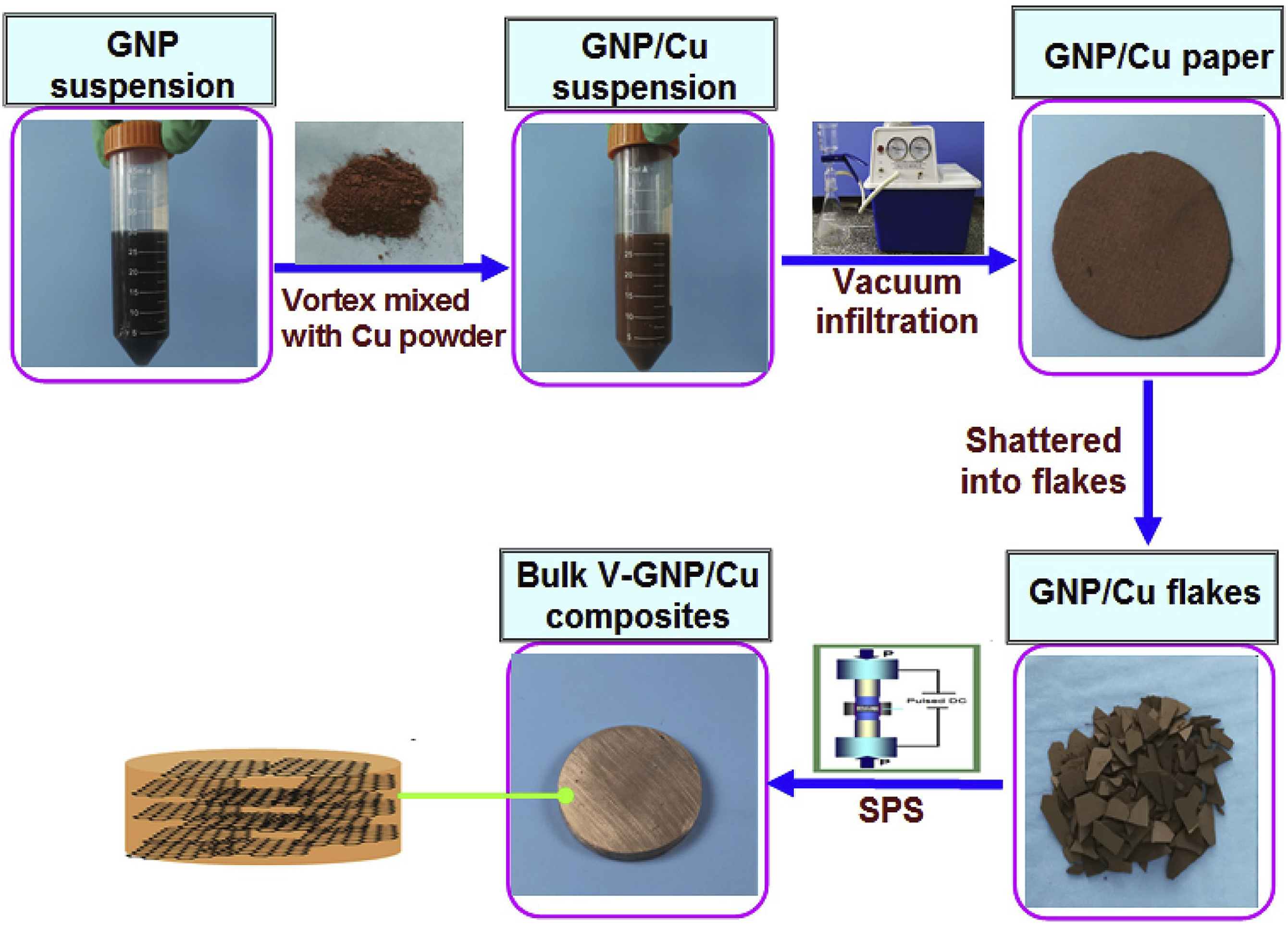
uses vacuum filtration technology to achieve good dispersion and pre-orientation of GNS in the mixed powder, which helps to generate highly oriented GNS in the bulk composite material and effectively improves the thermal conductivity of the material. The preparation process is shown in Figure 3. The results show that the thermal conductivity of the composite material with 35% volume fraction of GNS reaches 535 W/(m·K). Preparation process of GNS/Cu composites.
41
Graphite flake/copper composite material
Natural flake graphite is composed of up to 98 % crystalline graphite, which has good softness and excellent electrical and thermal conductivity. However, its obvious layered structure also leads to significant anisotropic physical properties, such as ultra-high thermal conductivity along the graphite layer direction (about 1000 W/(m·K)) and low thermal conductivity in the vertical direction (about 20 W/(m·K)). Due to its excellent thermal conductivity and thermal insulation properties, flake graphite can be widely used in electronic components. It can be used as a heat dissipation material for heat dissipation of local hot spots or as a thermal insulation material.
In recent years, great progress has been made in the thermal, electrical, mechanical properties, interface control and related theoretical research of graphite reinforced composites involving copper matrix. The content and orientation of graphite determine the thermal conductivity of graphite/copper composites. Wang ZM et al.
42
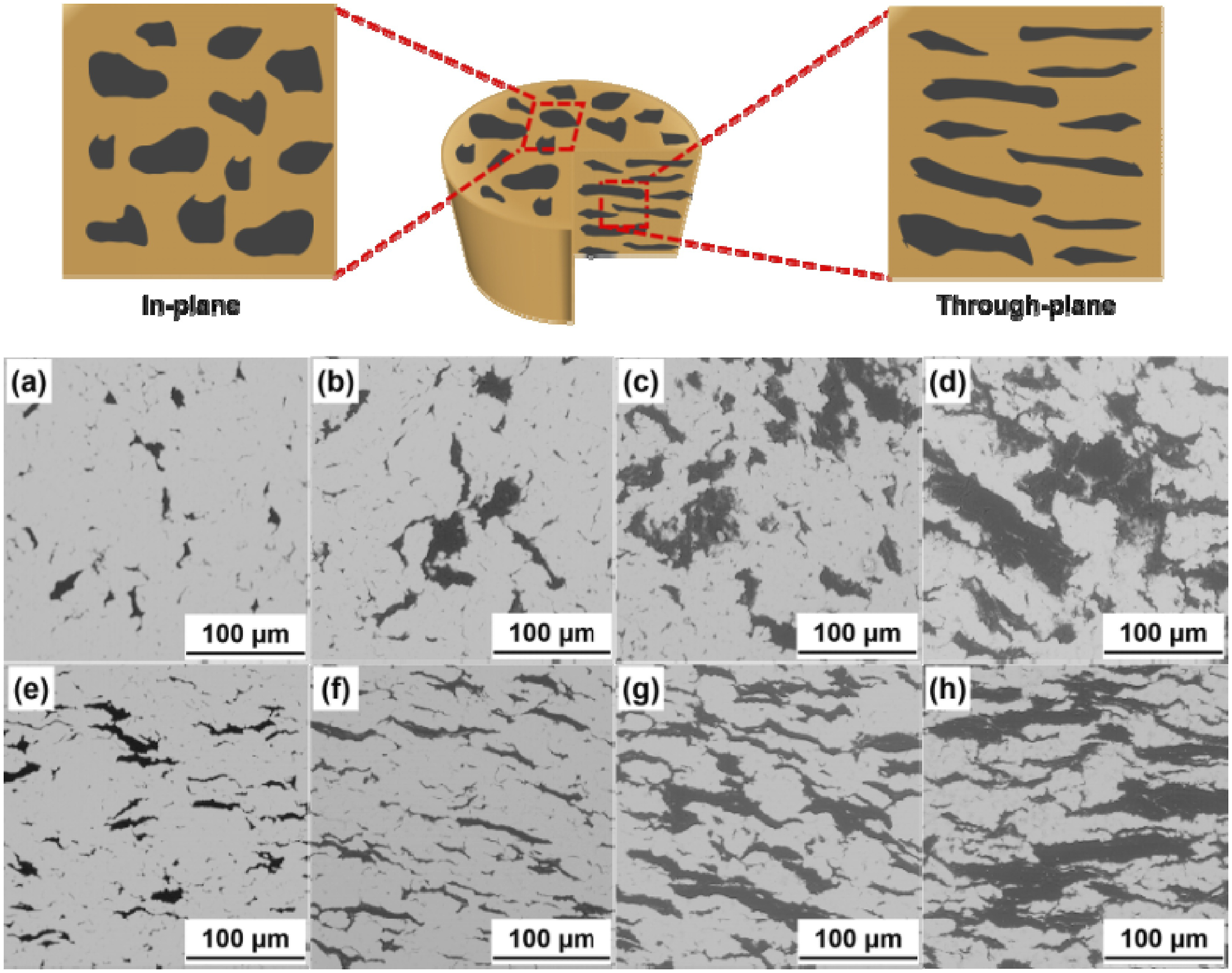
prepared graphite/copper composites by mechanical mixing and microwave sintering, and studied the effect of graphite volume fraction on the thermal conductivity and thermal expansion coefficient of the composites. The results show that the graphite volume fraction significantly affects the TC and CTE of graphite/copper matrix composites. The lower volume fraction leads to higher thermal performance. The effect of thermal cycling on the thermal properties of composites was studied. The thermal cycle leads to a periodic decrease in TC and a gradual increase in CTE. This is mainly attributed to the CTE mismatch between the Cu matrix and the graphite phase. Through scanning electron microscopy (SEM) observation, it was found that thermal cycling produced micro-cracks and micro-pores in the composite, as well as the sliding and detachment of the graphite-copper interface. The SEM image is shown in Figure 4. Liu B et al.
37
prepared flake graphite/copper composites with high volume fraction of flake graphite (72.08%−93.34%) by vacuum hot pressing method. The results show that the composites are anisotropic because the graphite sheets are arranged perpendicular to the pressing direction. The density of the composites decreased from 4.07 g/cm3 to 2.63 g/cm3 with the increase of the volume fraction of flake graphite due to the incomplete infiltration of Cu into the pores between flake graphite. When the volume fraction of flake graphite was greater than 82.6%, the in-plane thermal conductivity reached a maximum of 663.73 W/(m·K). Liu Q et al.
43
prepared flake graphite/Cu composites coated with Cu or TiC by powder metallurgy. The effects of flake graphite surface coating on the microstructure and thermal conductivity of flake graphite/Cu composites were studied. The results show that a good contact interface structure is established when TiC or Cu coating is introduced. When the graphite content is 60 vol%, the thermal conductivity of TiC and Cu coated composites reaches 668 W/(m·K) and 612 W/(m·K), respectively. In addition, Zhang R et al.
14
successfully prepared flake graphite/Cu-Ti composites by vacuum hot pressing. The effects of Ti addition on the microstructure, flexural strength and TC of the alloy were studied. The results show that the TiC interface layer is formed between the matrix and the flake graphite in the composites with Ti addition. With the increase of Ti content, the thickness of the interface layer first increases significantly, and then increases slightly. The thermal conductivity increases first and then decreases with the increase of Ti content, and reaches a maximum value of 597 W/(m·K) when the Ti content is 1.0 wt%. The graphite sheet/Cu-Ti composites with appropriate amount of Ti addition have excellent properties and can be used as candidate materials for electronic packaging materials. SEM images of graphite-copper composites at different graphite volume fractions: In-plane direction: (a) 10 vol.%; (b)20 vol.%; (c) 30 vol.%; (d) 40 vol.%; Through-plane direction: (e) 10 vol.%; (f) 20 vol.%; (g) 30 vol.%; (h) 40 vol.%.
42
Graphite film/copper composite material
High thermal conductivity graphite film is a new type of heat dissipation material, which is based on carbon material, and is made by unique sintering and heat treatment process. It has a layered superposition structure, and its crystal structure is similar to natural graphite sheet. This material has extremely high thermal conductivity, up to 800–1900 W/(m·K). The excellent thermal conductivity is affected by the grain orientation of the graphite film, showing obvious anisotropy. The material is soft and easy to cut, and the surface is easy to bond with metals, plastics and other materials. It should be noted that the thermal conductivity of the material is uniform along the plane direction of the graphite film, and the thermal conductivity in the vertical plane direction is low, generally only 10–20 W/(m·K). The thermal conductivity of the composites can be improved by using the macro structure design of the reinforced phase. 44 High thermal conductivity graphite film has achieved large-scale applications in the field of electronic equipment, such as mobile phones, computers and other heat dissipation applications. This graphite film is usually used as a thermal interface material, which can effectively conduct and emit heat, improve the heat dissipation performance of the equipment, and ensure the stability and reliability of the equipment under high load operation. In addition, some studies have also focused on the application of graphite film as a thermal conductivity enhancement in bulk metal matrix composites. The research of this composite material aims to make full use of the high thermal conductivity of the graphite film and combine it with the metal matrix to enhance the overall thermal conductivity of the composite material.
By introducing a graphite film as a reinforcing phase, the composite material can simultaneously possess the excellent mechanical properties of the metal and the excellent thermal conductivity of the graphite film, thereby achieving more efficient heat conduction and heat dissipation effects. Jiang B et al.
45
prepared copper-graphite-copper sandwich 2D thin film thermal interface materials. The thermal conductivity can reach 626 W/(m·K). Graphite film is often added to metal matrix composites as a continuous thermal conductive reinforcing phase due to its higher thermal conductivity and lower density. However, due to the non-wetting interface between graphite film and carbon materials and metal matrix materials, the preparation of graphite film reinforced metal matrix composites with excellent and stable properties needs to solve this technical problem. Huang Y et al.
46
used physical vapor deposition method to uniformly plate copper on graphite film (the coating mechanism is shown in Figure 5) to promote interface wetting. A new type of graphite film/copper/aluminum composite with good interface bonding was prepared by optimized vacuum hot pressing process. The results show that the Cu coating can effectively improve the interfacial thermal conductivity of 17.4%–53.2% (volume fraction) graphite film reinforced aluminum matrix composites, and the in-plane and out-of-plane thermal conductivity are increased by more than 20 times, respectively. Graphite film/Cu/Al composites with graphite volume fraction of 17.4%–53.2% have excellent in-plane thermal conductivity of 397–805 W/(m·K). Liu TL et al.
47
prepared a sandwich-like composite film with good thermal conductivity by combining graphite film (upper and lower layers) and copper with diamond (middle layer) using vacuum hot pressing technology. Because diamond is embedded in the copper film and in contact with the graphite film, and in order to enhance the interface bonding between graphite and copper, a complete and dense WC coating is deposited on the surface of graphite film and diamond, so that the interconnection structure between graphite film and copper film is established. It is found that the in-plane thermal conductivity of the composite film reaches a peak of 780 W/(m·K), and the out-of-plane thermal conductivity reaches a maximum of 211 W/(m·K). Graphite film/copper-based materials with excellent properties have a very broad prospect. Graphite film/copper-based materials with excellent properties have a very broad prospect. Schematic diagram of PVD coating mechanism.
46
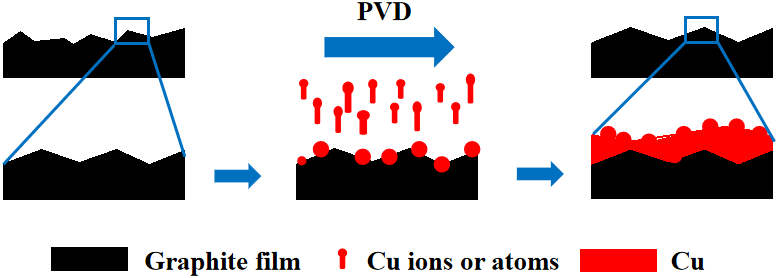
Carbon fiber/copper composite
Carbon fiber is a kind of microcrystalline graphite material which is stacked along the axial direction of the fiber by organic fibers such as flake graphite crystallites, and then carbonized and graphitized. It has excellent mechanical properties, chemical stability, tensile strength, thermal expansion, electrical and thermal conductivity (800 W/(m·K)). Carbon fiber mainly plays the role of bearing load in copper carbon composites, which can eliminate the stress in copper matrix and enhance the strength and wear resistance of copper matrix composites. 48 Carbon fiber also has excellent self-lubricating and corrosion resistance. Therefore, carbon fiber/copper composites are widely used as sliding materials, electronic components, contact materials and self-lubricating materials.49–51 In addition, as early as 1982, a new copper-carbon composite material with copper as the substrate and carbon fiber composited on it has been successfully developed in Japan. However, the domestic research is still late, and there is still a certain gap between the research level and the overall strength compared with foreign countries. The research mainly focuses on the friction and wear mechanical properties of carbon fiber reinforced composites.
However, there are few studies on the thermal conductivity of carbon fiber reinforced copper-carbon composites. Cui LH et al. 52 modified carbon fibers and copper alloys by CVD or copper alloy infiltration technology. Yi LF et al. 53 prepared a new type of copper/graphite/carbon fiber hybrid composite by spark plasma sintering (SPS) process by replacing part of graphite with copper-coated graphite. The incorporation of graphite leads to an increase in interface density, a decrease in twin boundaries, and a significant grain refinement of the Cu matrix. The thermal expansion coefficient of the composite material is reduced.
Kong B et al.
54
enhanced the wettability of carbon/carbon-copper composites with Cr3C2 interface by solution impregnation method, which made them have high wear resistance and thermodynamic properties. However, in this case, copper is isolated in the matrix in the form of one-dimensional (e.g., copper powder or copper alloy) or two-dimensional (e.g., copper mesh) structures, which limits the improvement of electrical conductivity. In order to overcome this problem, copper foam with a three-dimensional (3D) structure is widely used to prepare supports and electrodes due to its high conductivity, low density and specific mechanical properties. Wang P et al.
55
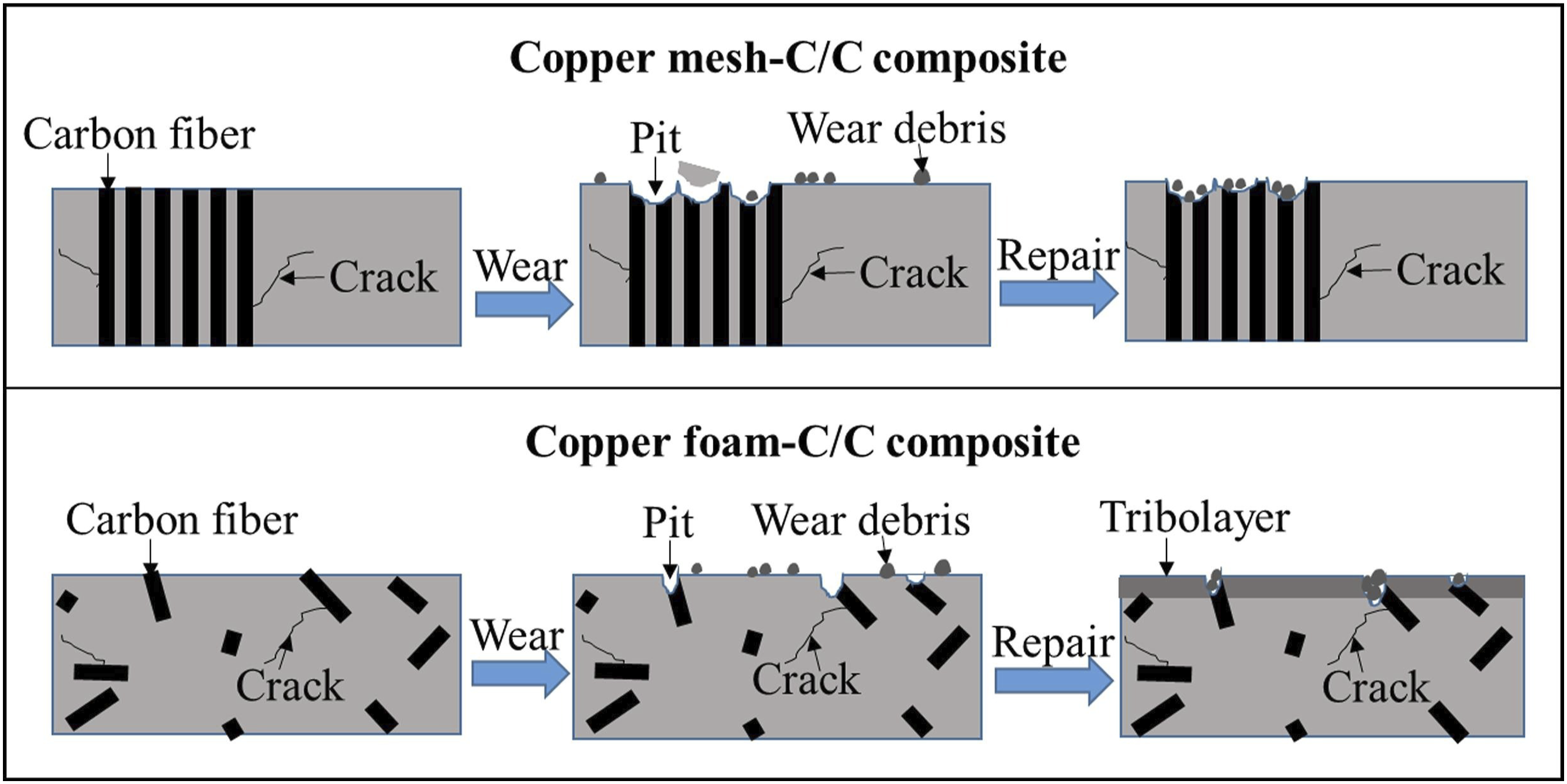
successfully prepared two new carbon fiber reinforced carbon/copper composites for pantograph slides by CVD and impregnation/carbonization (PIC) methods. The failure surface was detected by SEM and 3D profile, and the fracture and wear mechanisms were studied. Figure 6 shows the physical model of the current-carrying friction and wear mechanism of carbon fiber reinforced copper/carbon composites. The experimental results show that copper mesh-carbon/carbon (C/C) composites and copper foam-carbon/carbon composites have better electrical conductivity, density and wear resistance than pure carbon ribbons. Physical models of wear mechanisms of carbon fiber reinforced copper/carbon composites during current-carrying friction tests.
55
At present, the research on the preparation and thermal conductivity of diamond/copper composites has been relatively mature, but because of its high hardness and difficulty in processing, it leads to high production costs, which limits its application. For carbon fiber/copper composites, most of them are focused on the study of mechanical properties and friction properties, and there are few studies on thermal conductivity. The research on copper-carbon composites mainly focuses on the problem of non-wetting of the interface between carbon materials and copper. Chemical plating, adding additives, hot pressing sintering, chemical deposition and other methods are used to improve the interface between graphite and copper. Copper matrix is often in powder state, often compounded with graphite powder, flake graphite and graphene. The research focuses on the morphology, content and orientation of carbon materials, the type and thickness of coatings, the type and content of additives and the preparation methods. The effects of carbon materials on the mechanical, electrical and tribological properties of composites are analyzed microscopically. The thermal conductivity research started late, and the current research can increase the thermal conductivity to about 600 W/(m·K), which is not as good as the in-plane thermal conductivity of graphite.
Preparation method of copper-carbon composites
Preparation process of graphite/copper composites.
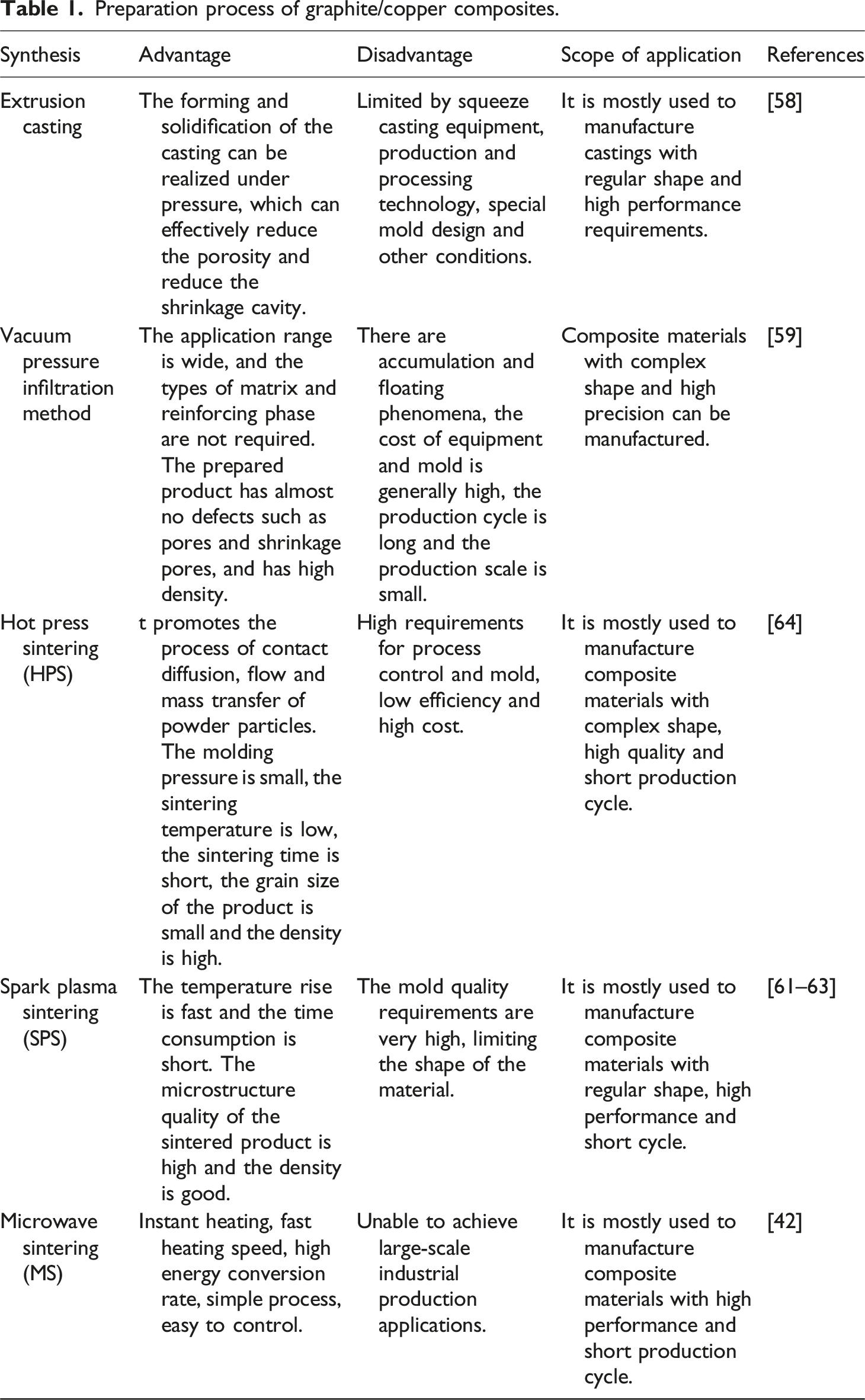
The liquid phase method is the process of adding dispersed reinforcing phases to the molten matrix material to solidify it to obtain the required material. It can be divided into two methods: squeeze casting method and vacuum pressure infiltration method. In the liquid phase treatment, at least one component is in the liquid phase state, usually the metal matrix is in the liquid state, while the reinforcement is in the solid state. 56 The key is the interfacial tension between the reinforcing phase and the liquid matrix. Liquid phase treatment is one of the cheapest methods for manufacturing metal matrix composites. The solid phase method can be divided into hot pressing sintering, spark plasma sintering and microwave sintering. In the solid phase method, powder metallurgy technology is the main process method for preparing composite materials. It is an advanced process technology for manufacturing metal materials, alloy materials, composite materials and other products by mixing, forming and high temperature treatment with metal powder (or mixture of metal powder and non-metal powder) as the original material. 57 For copper-carbon composites, powder metallurgy can accurately control the proportion of each phase, thereby regulating the performance of the composites.
Interface control of copper-carbon composites
The wettability of copper and carbon is poor, and the two are not easy to dissolve each other or react chemically. Therefore, in copper-carbon composites, the interface is not chemical bonding but physical bonding, mainly mechanical bonding. However, the surface of carbon material is relatively smooth, and it is difficult to form strong mechanical bonding. Even if the copper is not wetted after melting, the two will not react, so the interface bonding often shows unsatisfactory conditions.64,65 It is found that for carbon material reinforced copper matrix composites, the bonding state of the interface has a great influence on the thermal and mechanical properties of the composites. When the interface bonding is poor, the interface thermal resistance increases. Although the thermal conductivity of copper and reinforced phase is high, the overall thermal conductivity of the composites is still very low, even lower than that of copper. After optimizing the interface of the composite material, the interface bonding state between copper and carbon is improved, and the thermal and mechanical properties of the composite material are improved. In order to obtain high-performance copper-carbon composites, the bonding strength of the copper-carbon interface must be improved. At present, there are two main methods to improve the wettability of carbon materials and copper, one is to modify the surface of carbon materials, and the other is to alloy the copper matrix.
Surface modification of carbon materials
Surface modification of carbon materials is an effective means to improve the wettability of carbon materials and copper to improve the interfacial bonding strength, and this method has almost no negative impact on the composites. Common treatment methods for surface modification of carbon materials include electroless plating, vapor deposition and salt bath plating.
Electroless plating
Electroless plating refers to the surface treatment method of forming metal or alloy coating on the surface of substrate material by using metal salt and reducing agent to carry out redox reaction in the same solution on the surface of substrate material with catalytic activity. It has the advantages of many kinds of substrate materials and simple plating process. 66 In the process of electroless plating, the key is to accurately select the appropriate activator and electroless plating solution, and adjust the appropriate plating parameters. If the improper activator, electroless plating solution or inappropriate concentration is selected, it will be difficult to obtain the ideal coating effect. 4
For copper-carbon composites, on the one hand, copper is regarded as the matrix. Researchers often chemically plate Cu, Ti, Ni and other layers on the surface of carbon materials, and then mix the surface-modified carbon materials with copper powder to prepare copper-carbon composites by powder metallurgy technology. Wang YR et al.
67
modified the graphite surface by depositing an electroless nickel plating layer on the graphite surface. Figure 7(a) is the cross-section morphology of nickel-coated graphite powder, and figure (b) is the interface morphology of nickel coating and EDS test at the interface. The interface characteristics were studied and the modification mechanism of nickel coating in composites was analyzed. The interface characteristics were studied and the modification mechanism of nickel coating in composites was analyzed. The results show that the addition of nickel plating layer improves the interface problem between graphite and copper, so that its mechanical properties are significantly improved. Microstructure details of nickel coated graphite powder (a) Cross section morphology of Ni-Gr. (b) Interface morphology of nickel coating and EDS test at the interface.
67

On the other hand, the graphite powder can also be directly sintered after copper plating, and the copper in the coating is regarded as the matrix. 23 Lee J et al. 68 deposited Cu on the surface of graphite sheet (GF) by electroless plating to increase the interfacial bonding between Cu and graphite, and then consolidated the composite by spark plasma sintering. The Cu content varied from 20% to 60% to study the effect of graphite fraction and microstructure on the thermal conductivity of the composites. The thermal conductivity of 40 wt% graphite/Cu composites is as high as 692 W/(m·K), and the thermal expansion coefficient is 4.12 ppm/K. The results show that electroless copper plating can improve the distribution of expanded graphite in copper matrix, avoid the aggregation between graphite sheets, and maintain the continuity of matrix metal. Such an improvement helps to promote the sintering process and increase the density of the composite material, thereby improving the overall performance; Liu B et al. 60 successfully prepared a copper-based composite material composed of a mixture of modified copper-coated graphite flakes and copper powder by vacuum hot pressing and electroless plating. When the equivalent particle size of graphite sheet is 292 μm, the composite has excellent thermal conductivity (612 W/(m·K)), ideal flexural strength (425 MPa), suitable electrical conductivity (14.7% of the international annealed copper standard) and suitable thermal expansion coefficient (6.6 ppm/K).
Vapor deposition
Vapor deposition is a surface film forming technology, including physical vapor deposition (PVD) and chemical vapor deposition (CVD). In physical vapor deposition, the substrate surface is vaporized into gaseous atoms, molecules or partially ionized ions by solid or liquid material sources under vacuum conditions, and deposited by low-pressure gas (or plasma) to prepare films with special functions. At present, physical vapor deposition technology includes vacuum evaporation, sputtering coating, arc plasma plating, ion plating and other methods. The basic process includes gasification of the plating material, migration of atoms, molecules or ions, and deposition on the surface of the substrate.
Li JW et al.
69
prepared copper/diamond (Ti) composites by a combination of Ti plating on the surface of diamond particles and physical vapor deposition. The composites obtained a high thermal conductivity of 716 W/(m·K) and a low thermal expansion coefficient of 5.8 ppm/K at 323 K. Auger electron spectroscopy (AES) characterization shows that a TiC layer is formed between the Cu matrix and the diamond reinforcement, which is the reason for the enhancement of thermal conductivity. The results show that Ti coating can significantly promote the interfacial bonding between Cu and diamond, and physical vapor deposition is an effective method for preparing copper/diamond composites. Chen YK et al.
70
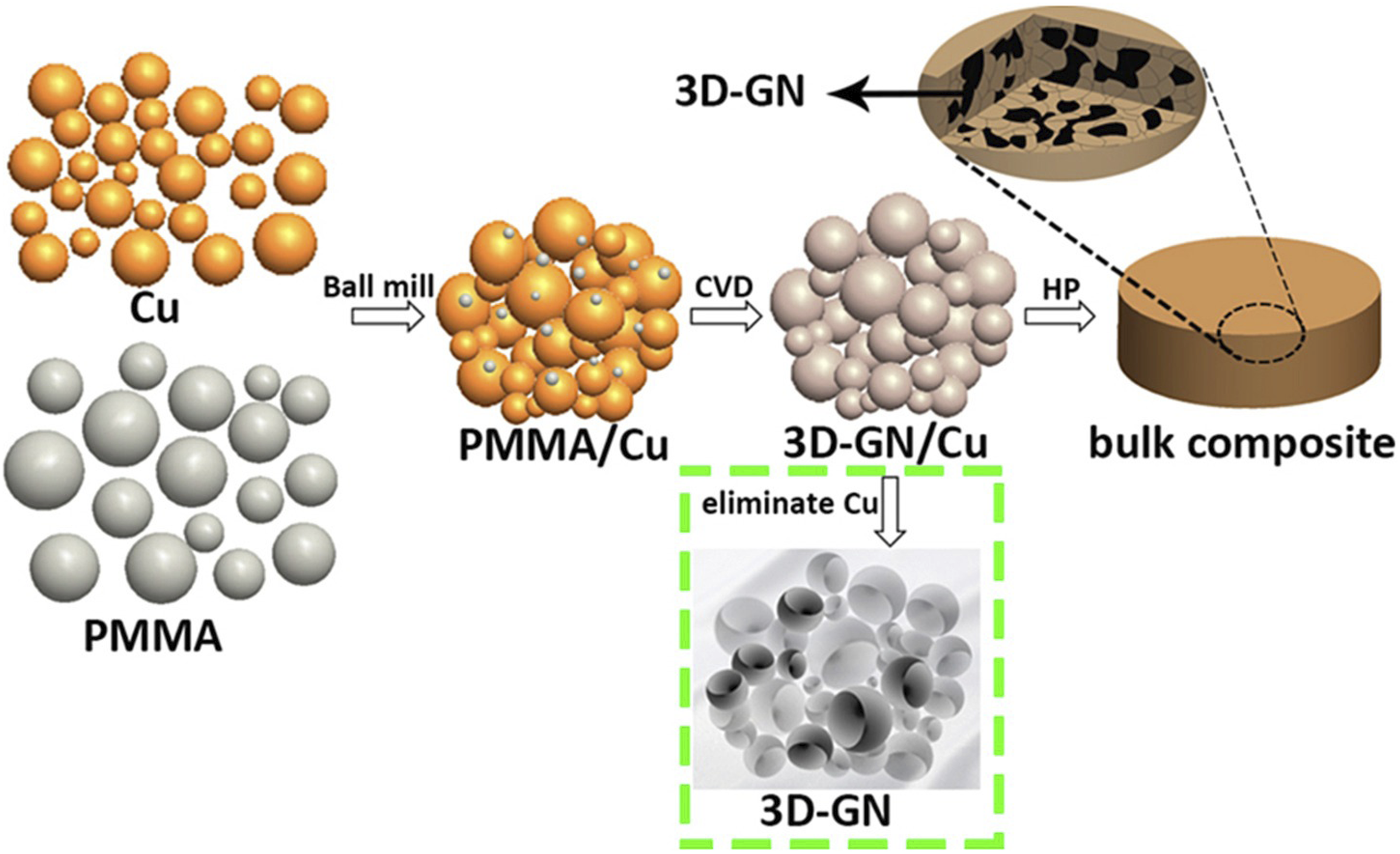
prepared in-situ grown three-dimensional graphene reinforced copper matrix composites by CVD method. Figure 8 shows the formation process of in-situ grown 3D-GN. In the CVD process, the carbon atoms in the pyrolytic polymethyl methacrylate (PMMA) diffuse and precipitate on the copper powder matrix. By inheriting the morphology of the copper powder, three-dimensional graphene is formed in situ on the copper powder. Finally, three-dimensional graphene/copper composites were prepared by vacuum hot pressing sintering. The experiment shows that the interface bonding strength of the composites is quite excellent, and the three-dimensional graphene is well dispersed in the Cu matrix, which effectively hinders the dislocation movement. Schematic diagram of the formation process of in-situ grown 3D-GN.
70
Salt bath plating
Salt bath plating is a kind of hot infiltration plating technology. Its principle is to melt the metal salt into a liquid, and add metal powder containing coating elements, such as titanium, chromium, molybdenum, etc., and then the carbon material to be plated is immersed in the salt solution for treatment. The unique feature of salt bath plating is that its salt system has the characteristics of fast ion migration and diffusion, and uniform mixing of reactants. The corresponding compound coating layer can be obtained on the graphite surface by treating at a temperature range of 800°C–1250 °C for 0.5–10 h. The salt bath plating technology has several advantages: 1. It can achieve strong plating ability in a single time and provide the required reinforcement layer for carbon materials. 2. The cost of salt bath plating is relatively low, making it an economical and practical plating method. 3. In the process of salt bath plating, the metal salt melting liquid provides the elements needed for plating, and the high temperature environment is conducive to the reaction and diffusion between the elements and the carbon material, thus forming a uniform compound coating on the surface of graphite. This coating can not only improve the performance of carbon materials, but also enhance their heat resistance and corrosion resistance.
However, it should be noted that although salt bath plating has certain advantages, there are also some challenges in practical applications, such as the control of coating thickness and the maintenance of uniformity. Kong B et al.
71
synthesized Cr3C2 layer on the surface and internal pore wall of graphite foam by ammonium dichromate solution, and prepared Cr3C2 coated graphite/copper composite with porosity of 2.9% and density of 7 g/cm3 by pressureless infiltration method. The thermal conductivity of graphite/copper composites increases from 209.1 W/(m·K) to 323.0 W/(m·K) due to the enhanced infiltration and good interfacial bonding of copper by the synthesized Cr3C2 layer. Bai H et al.
6
prepared graphite/copper composite material by vacuum hot pressing process, and synthesized boron carbide-boron coating on the surface of graphite by salt bath plating method. The experimental process is shown in Figure 9. The obtained composites have excellent thermal conductivity (676 W/(m·K), 180% of copper) and suitable thermal expansion coefficient (7.1 ppm/K), which is attributed to the uniform dispersion and good orientation control of graphite in the composites. Schematic of preparation process of Gr/Cu composites.
59
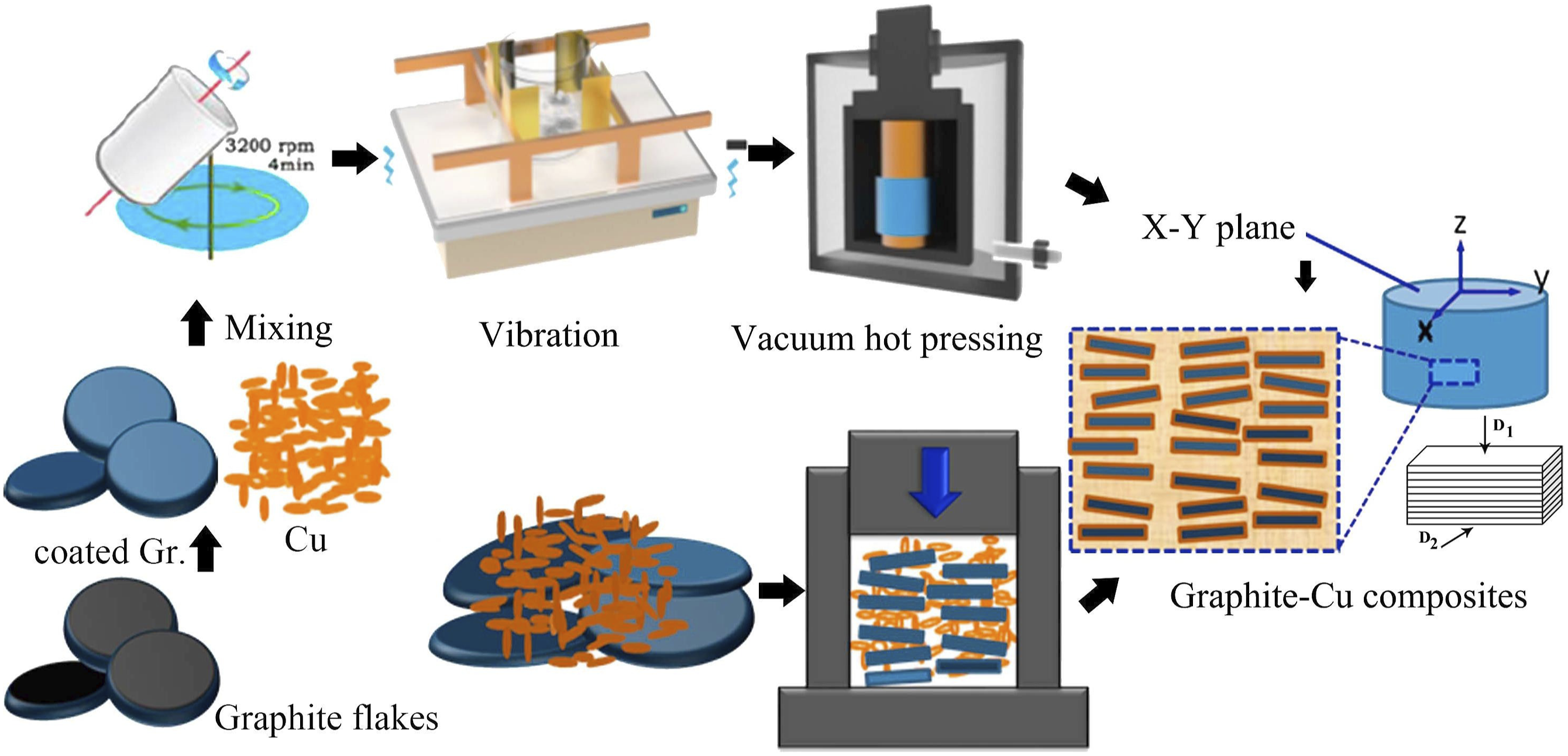
Matrix alloying
Matrix alloying refers to changing the chemical composition of the matrix by adding one or more metal elements to the metal matrix to reduce the surface tension and improve the wettability of the reinforcing phase and the matrix. This method aims to improve the performance of metal matrix composites, so that they have better interface bonding and interaction, thereby improving the overall performance of the overall material. Through matrix alloying, the chemical composition and microstructure of the material can be adjusted to achieve more effective energy transfer and enhanced phase dispersion.
For copper-carbon composites, appropriate addition of alloying elements can react with carbon materials to form carbide layers, such as Ni, Ti, Cr and other elements. At the same time, it can also reduce the surface tension of copper and effectively improve the interface bonding between graphite and copper. Zhang R et al. 14 successfully fabricated graphite/copper-titanium composites with titanium (Ti) elements by vacuum hot pressing. The effects of Ti addition on microstructure, flexural strength and thermal conductivity were studied. The results show that the TiC interface layer is formed between the matrix and the graphite sheet in the composites with Ti addition. The growth rate of the interface layer thickness increases first and then slows down with the increase of Ti content. The bending strength of the material is proportional to the Ti content. After adding 2.0% (mass fraction) Ti, the bending strength is increased by 60% compared with the unadded one. The thermal conductivity increases first and then decreases with the increase of Ti content, and the maximum value is 597 W/(m·K) when 10% (mass fraction) Ti is added. Ren SB et al. 22 prepared graphite flake reinforced copper matrix composites (GFs/Cu) by powder hot pressing method, and directly added alloying chromium (Cr) elements to the Cu matrix powder to enhance the interface bonding between GFs and Cu matrix. The results show that the TC of 50vol% GFs/Cu composites increases from 560 to 628 W/(m·K) after adding 2.0 wt% Cr in the X-Y direction. The improvement of thermal conductivity is attributed to the formation of carbide transition layer at the interface between Cu and graphite, which improves the interfacial wettability between copper matrix and graphite. Therefore, it is feasible to improve the interfacial wettability between carbon materials and copper by matrix alloying. In order to achieve this goal, it is necessary to select and optimize the additives, addition amount and process to ensure the ideal interface interaction and bonding effect.
Summary and prospect
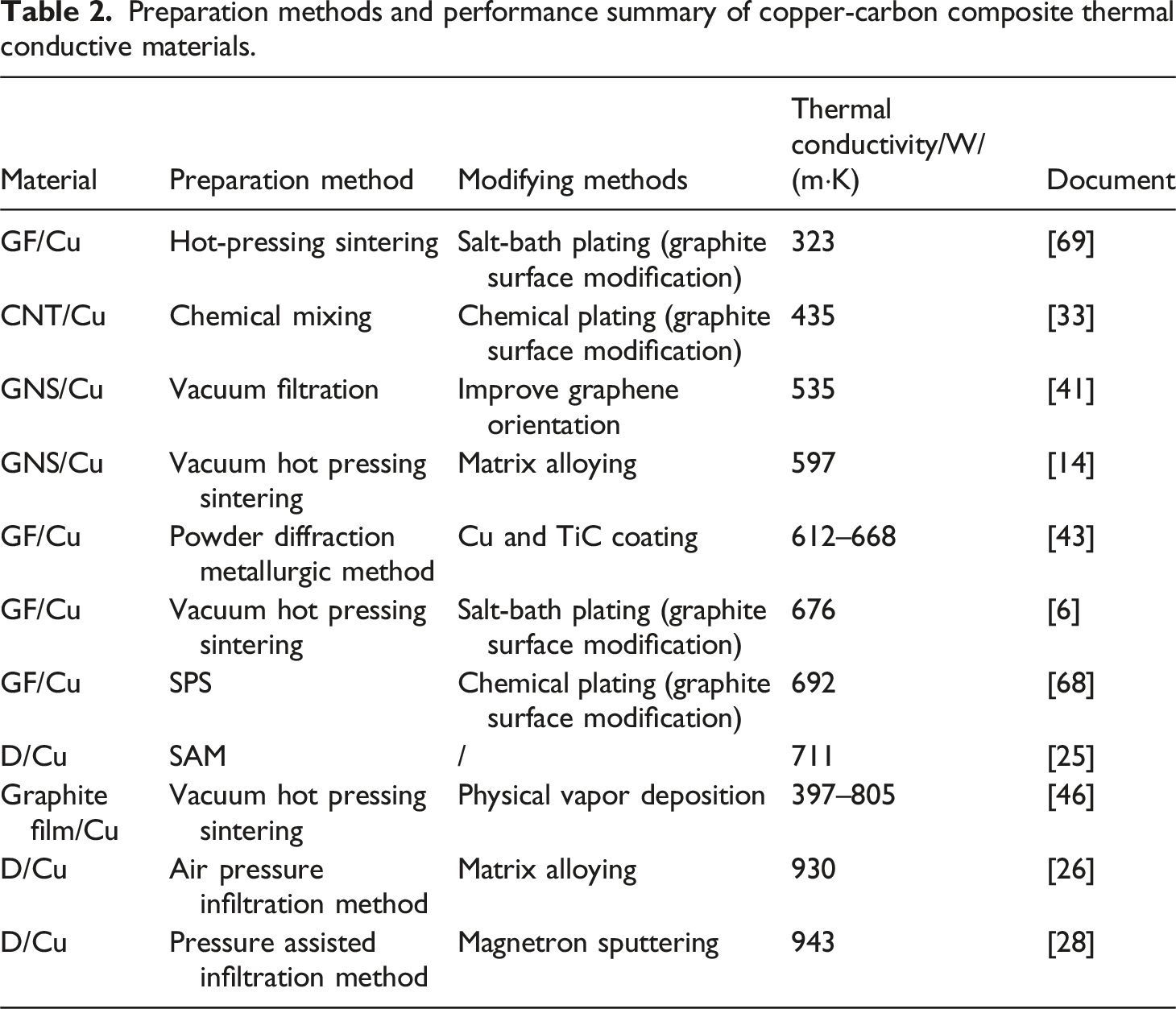
Preparation methods and performance summary of copper-carbon composite thermal conductive materials.
The main research progress is summarized as follows: (1) The research on the preparation and thermal conductivity of diamond/copper composites has been relatively mature. However, due to its high hardness and difficulty in processing, it leads to high production costs, which limits its application. The mechanical properties and friction properties of carbon fiber/copper composites are mostly studied, and the thermal conductivity is still less studied. Graphite materials have good research and application prospects due to their excellent properties such as ultra-high thermal conductivity, low bulk density, thermal expansion coefficient and corrosion resistance. (2) The research of copper-carbon composites mainly focuses on the problem of non-wetting of the interface between carbon materials and copper. Chemical plating, adding additives, hot pressing sintering, chemical deposition and other methods are used to improve the interface between carbon materials and copper. Copper matrix is often in powder state, often compounded with graphite powder, flake graphite and graphene. The research focuses on the morphology, content and orientation of graphite, the thickness of the coating type, the content of the additive type and the preparation method. Through the microscopic analysis of its influence on the mechanical, electrical and tribological properties of the composite material. The thermal conductivity research started late, and the current research can increase the thermal conductivity to about 600 W/(m·K), which is not as good as the in-plane thermal conductivity of graphite. (3) The preparation method of copper-carbon composites restricts its performance and production cost. At present, the preparation process of copper-carbon composites is mainly liquid phase method and solid phase method. (4) The wettability of copper and carbon is poor, the interface bonding is mostly mechanical interlocking, and the bonding strength is poor. At present, there are two main methods to improve the interfacial bonding strength between carbon materials and copper, namely, surface modification of carbon materials and matrix alloying.
At present, copper-carbon composite thermal conductive materials have made great progress in types, preparation processes, interface control and other aspects. However, in order to obtain higher thermal conductivity and good comprehensive performance and reduce costs, there is still much room for research on the production process, the type and content of additives. In this regard, the following prospects are proposed: (1) Make full use of the excellent properties of carbon materials and copper to explore more application prospects of copper-carbon composites. (2) The anisotropy of thermal conductivity of highly oriented carbon materials is caused by the anisotropy of their structure. Therefore, the high orientation and uniform dispersion of carbon materials in the collective can greatly improve the thermal conductivity of composites. At the same time, the arrangement of carbon materials in the matrix can be effectively controlled by improving the preparation process, such as vacuum filtration and external field induction, so as to obtain highly oriented composites. To explore an efficient directional arrangement method of carbon materials. (3) The poor wettability between carbon materials and copper matrix leads to poor thermal conductivity. At present, the methods to improve wettability mainly include collective alloying and surface modification of carbon materials. However, most of the current research focuses on improving the macroscopic properties of materials by improving the process. The microscopic interface bonding mechanism between carbon materials and copper matrix needs to be further studied. Carbon materials and copper matrix are prone to react during the preparation process. The formation of carbides on the one hand inhibits the exertion of their own thermal conductivity, and on the other hand improves the bonding degree of the copper-carbon interface. Therefore, exploring the reaction of copper-carbon interface and making full use of carbides to design materials will become a research hotspot in this field in the future.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was financially supported by Inner Mongolia First Machinery Group Lutong Spring Co., LTD., Inner Mongolia Natural Science Foundation (2024LHMS05002), Young Leading Talent of “Grassland Talents” Project of Inner Mongolia Autonomous Region (no. QNLJ012010), Innovative Research Team in Universities of Inner Mongolia Autonomous Region (no. NMGIRT2211), Inner Mongolia University of Technology Key Discipline Team Project of Materials Science (no. ZD202012).
