Abstract
The aim of this work was to investigate the physical properties of thermoplastic polycarbonate films and their potential applications in industrial technology after bombardment at fluences 0.11 × 1019, 0.22 × 1019, 0.33 × 1019, and 0.44 × 1019 ions/cm2 of oxygen ions. To evaluate the impact of ion bombardment on these samples, FTIR, UV/Vis spectroscopy, surface roughness tester, and contact angle goniometer were used. It was observed that the band positions in the collected spectra of the samples remain unchanged when subjected to oxygen ions. Surface roughness parameters are unaffected by variations in contact angles. The contact angle measurement demonstrated that enhancing the surface wettability and hydrophilicity of the tested polymer can be achieved by increasing the ion fluencies. Increasing the ion fluence caused an improvement in the adhesion work (Aw) due to decreasing the contact angles. The highest value of the solid surface free energy (γs) was detected in the bombarded sample with 0.44 × 1019 ions/cm2, whereas the lowest value was determined in the blank sample. Furthermore, the alteration in the absorption spectra of the bombarded films is noticeable in the dent region, whereas their absorption spectra at longer wavelengths are nearly indistinguishable. Increasing the ions fluences led to a reduction in the skin depth of the bombarded samples, particularly in the dent region, which indicates the reduction of their transparency. The reduction of the band gap energy values can be attributed to intermediate-level creation in the band gap of the bombarded samples. Ion bombardment induces an augmentation in the creation of covalent bonds among different chains by the action of free radicals. This leads to changes in the densities, extinction coefficient, and refractive indices of the samples under investigation.
Keywords
Introduction
The physical, structural, chemical, and optical features of polymeric materials can be drastically altered by radiation.1–3 Radiation has a significant impact on how polymeric materials develop in terms of their characteristics. 3 Polymers exposed to radiation undergo triggered modifications to their physical and chemical properties due to energy loss resulting from radiation absorbed within the intended substance. 4 For bettering the performance of polymers, a variety of radiation approaches are applied, including illumination with an electron beam, gamma-rays, neutrons, UV light, and plasma sputtering. An approach for improving the physico-chemical properties of polymers is ion beam bombardment.5,6 The possible benefits of ion beam impacts on polymers, such as radiation dosimetry, sensors, and shielding, have rendered these investigations much more important over the past years.7,8 Bond breaking, main chain scission, intermolecular cross linking, radical composition, creation of unsaturated bonds, and loss of volatile fragments are a few of the complicated events that result from ion interaction in polymers.2,8 The structure, ion beam factors (energy, fluence, mass, charge), and the objective material’s features mostly determine the degree to which such polymer conversions occur. 9 Moreover, one of the most important methods for causing significant changes in the chemical compositions, optical properties, and electrical behaviors of polymer films is to employ ion beam bombardment. 10 There are numerous mechanisms that might be responsible for the observed alterations, such as dispersal of macromolecules, cross-linking, synthesis of novel structures, carbonization, and free radical oxidation. It has been found that using ion beam energy is a practical way to alter its optical and structural characteristics. Furthermore, it has been demonstrated that ion beam bombardment is an extremely beneficial technique for modifying polymer surfaces and attributes like stiffness, wetness, and wear resistance.11–13
Exposure to ions causes a considerable change in the mechanical characteristics of irradiated polymers. 14 A lot of research has been done on broad categories of changes in mechanical properties, such as irradiation swelling, irradiation-induced embrittlement, irradiation hardening, irradiation stress/strain, and so on. These changes often indicate an increase in yield stress and ultimate stress as a function of irradiation dose.6,15,16
Two distinct outcomes are produced once ion beams engage with the polymer (nuclear stopping power (Sn) and electronic stopping power (Se)). 9 High impact strength, minimal moisture absorption, low combustibility, strong dimensional stability, and high light transmittance are among the appealing technical qualities of thermoplastic films. 17 Thermoplastic polycarbonate films is a recognized polymer utilized in solid-state nuclear track detectors. 18 Polycarbonate material is an engineering thermoplastic film with excellent strength, transparency, thermoformability, high toughness, outstanding ductility, high glass transition temperature, exceptional optical clarity, and dimensional stability. Many studies and reports on the polycarbonate thermoplastic films appeared in the literature to improve its properties by different methods, including radiation, blend composition, and doping with nanoparticles.19–23
Currently, ion bombardment is employed to modify the properties of polycarbonate films. The changed material is then utilized in a range of industrial applications. These improved properties gave it uses in many applications, including graphic design, automotive components, appliance, consumer electronics, display, signs, safety glazing, transparent armor and scratch-resistant coatings, pact discs, riot shields, safety helmets and headlamp lenses, medical equipment that needs to be sterilized beforehand, and many more. Given that polymers are frequently employed in optoelectronic devices, it is crucial for describing their optical characteristics. 24 Solid surface energy parameters such as adhesion and wettability are evaluated via the contact angle. Measuring the surface free energy, interfacial free energy, polar and dispersal energy outputs becomes simple by the utilization of contact angle studies.25,26
Ion beams have been used to study how the physical characteristics of polycarbonate films have changed at both the bulk and surface levels. We are paying close attention to how oxygen ions interact with the polycarbonate films. When an ion beam propagates at a polymer’s surface, the likelihood of a reaction will depend on both the oxygen diffusion through the material and the ion fluence rate. In addition, this study aims to enhance the molecular level structural, surface, and optical characteristics following oxygen ion exposure for usage in cutting-edge applications. Because ion beam irradiation modifies a polymer’s physical, chemical, optical, and structural properties, it is a significant factor in polymer science, which used in applications for ion beam-irradiated polymers include nanotechnology, microelectronics, agriculture, ecology, and sensing. To achieve this goal, the interaction between the oxygen ion and thermoplastic polycarbonate films was investigated using SRIM software. Fourier Transform Infrared was utilized to examine functional groups and structures. The surface examination, wettability and optical properties of thermoplastic polycarbonate films were studied after bombardment by oxygen ions with different fluences, which may be modified to include more qualities without compromising their fundamental descriptors.
Experimental details
Material
Thermoplastic polycarbonate films with a density of 1.20 g/cm3 were manufactured by Farbenfabriken Bayer A.G. (Germany). Bisphenol A with a carbonate group makes form a polycarbonate molecule. Because of its two aromatic rings, bisphenol A prevents the polycarbonate from crystallizing. The polymer gets its unique transparency from this amorphous structure. The chemical formula of the samples is C16H14O3. The film was sliced into 2 cm × 2 cm square segments. Before being bombarded, the films underwent a 10-min ultrasonic cleaning process in deionized water.
Ion beam bombardment
Thermoplastic polycarbonate films were bombarded with low energy of oxygen ions (3 keV) for 15−60 min utilizing a cathode ion emitter on a cold circuit. Four distinct ion fluences were used for the bombardment: 0.11 × 1019, 0.22 × 1019, 0.33 × 1019, and 0.44 × 1019 ions/cm2. The ion beam current density was of 150 μA/cm2. Under 2 × 10−3 mbar of pressure, the bombardment was conducted at room temperature.
Characterization studies
The present investigation employed SRIM-2013 27 to facilitate the computation of profiles of oxygen ion deposition in polycarbonate material. To guarantee accurate statistics, the input parameters for a specific projectile and target were maintained constant throughout all computations using 10,000 incident ions. We conducted irradiation calculations for polycarbonate material target using 3 keV O ions for various fluences. The ion beam was directed perpendicular to the surface and all ions were stopped within the target. 27
Utilizing a SMART OMNI-TRANSMISSION Nicolet iS10 FTIR instrument (Thermo Scientific), the Fourier Transform Infrared procedure was applied to assess the modifications in functional groups of the substances in the wavenumber range of 400-4000 cm−1.
Surface Roughness Analyzer SRT-6600 was implemented to determine the surface roughness variables. The surface roughness variables are identified by the approach of sensor touch. The readings were performed three times. DataView Software V1.2 for a higher level of accuracy was used to analyze the measurements. The test platform was adjusted at 0°.
Ossila Contact Angle Goniometer (Ossila Digital Goniometer, Model L2004A1, Sheffield, UK) was used to measure the contact angle. The contact angle of the pristine and bombarded films at room temperature was measured with an accuracy of ± 1o. The assessment platform has a zero-degree level and setting. For every sample, a 5 μL drop of liquids was placed on the assessment platform. The goniometer’s software, Ossila Contact Angle v.3.1.2.2, was used to examine the taken pictures.
Electronic transition data from the samples were evaluated using UV-visible spectroscopy. The UV/Visible double-beam spectrophotometer, JASCO Model V-630, was applied to measure the samples’ UV–visible spectra. A scan of wavelengths was conducted in the range of 200–1100 nm.
Results and discussion
Theoretical aspect (SRIM/TRIM simulation)
The SRIM software code (Ziegler) was used to assess the interaction variables of oxygen ion bombarded thermoplastic polycarbonate films.

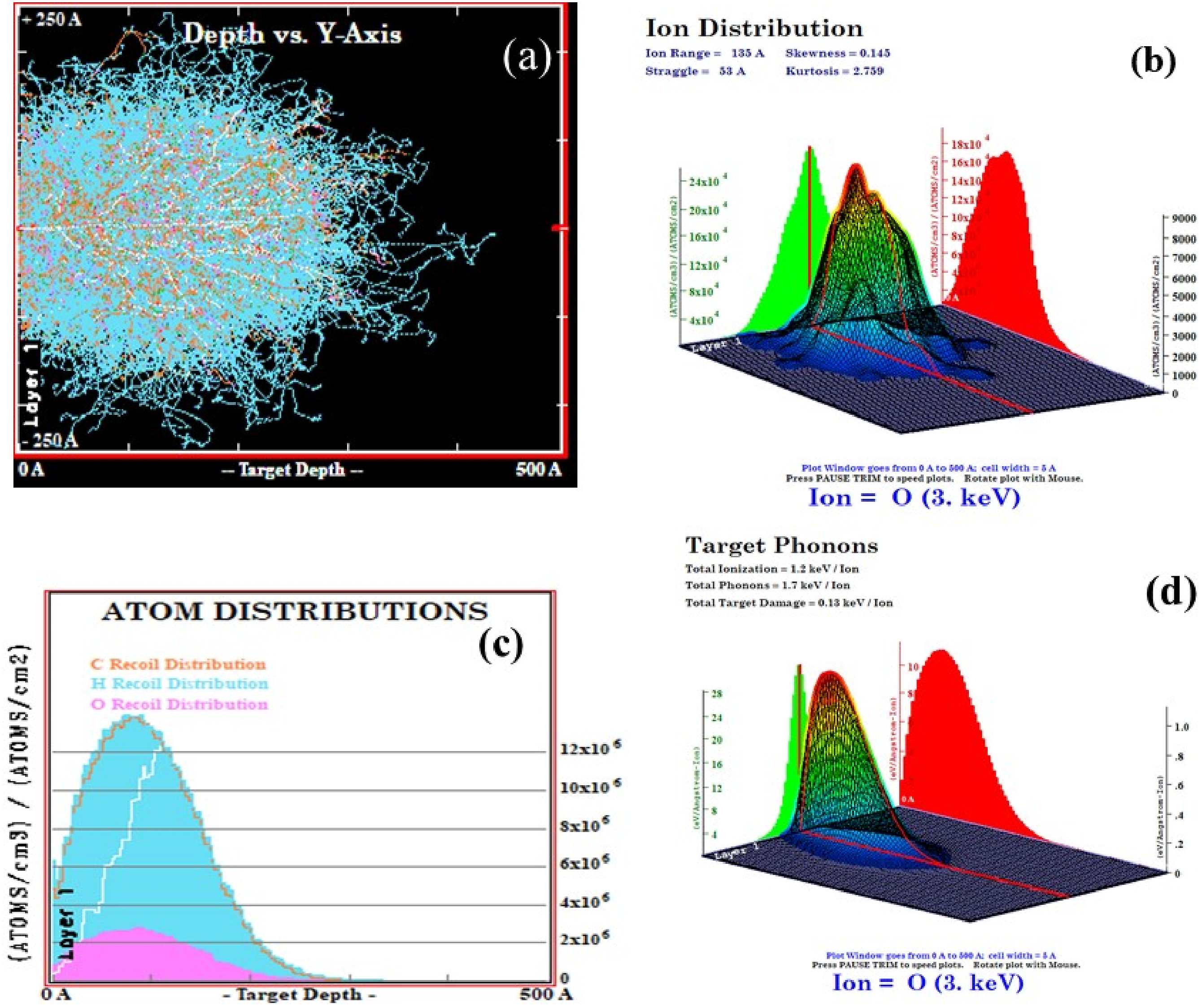
Displays (a) the ion tracks of oxygen ions versus objective range, (b) the oxygen ion distribution, (c) atom distributions, and (d) target distribution of phonons generated by recoil atoms and oxygen ions.
FTIR spectroscopy
The chemical composition of polymers can be favored by analyzing their vibrational modes with infrared spectroscopy.17,29 The structural alterations triggered in the thermoplastic polycarbonate films polymer by undergoing oxygen ion beam bombardment were examined employing FTIR spectral analysis techniques. The variances in peak magnitude were exploited to distinguish proportional modifications in functional groups within the polymers. Aromatic group is the substance employed in this investigation. It has methyl, phenyl ring, carbonyl, ether, and hydroxyl functional groups. The absorbance of distinct bands from the same function group follows the same pattern as oxygen ion beam bombardment. At 886, 1798, 2331, 2879, 3049, and 3528 cm−1, distinct absorption bands of thermoplastic polycarbonate films film were reported. These bands correlate to the C-O-C bond, the C = O bond, the CO2 bond, the CH3 bond, the C-H stretching bond, and the OH bond, in that order.2,4 FTIR spectra in Figure 2 show absorption bands with fluence outcomes in bombarded specimens. Figure 2 demonstrates that at oxygen fluence 0.11 × 1019 ions/cm2, the rate of absorption at low frequencies increases. The absorption intensity grows owing to crosslinking, and then diminishes as the oxygen ion fluences rise, reflecting the deterioration of the C-O and C-O-C bonds. This drop is due to a split in the carbonate bond as well as (-H) removal from the polymer chain’s backbone. Such a trend may be related to the likelihood of the formation of OH and CO2 groups of varying strengths.8,29 The polymer’s bonding features have shifted dramatically. It is possible to highlight the presence of the two benzene rings through the C-H stretching band, which is located around 3050 cm−1 (CH aromatic) and 2974 cm−1 (C-H3 aliphatic). Understanding the behavior of C-H bonds is crucial, and findings show that their intensity changes in response to ion fluences. Specifically, low ion fluences lead to an increase in intensity, followed by a decrease as oxygen ion fluences rise. FTIR spectra provide valuable insight into the behavior of these bonds under varying conditions. Upon bombardment with low-energy oxygen ions, the band positions were found to remain unchanged across the wavenumber 4000-400 cm−1 in the collected spectrum for the thermoplastic polycarbonate films. FTIR spectra of thermoplastic polycarbonate films at various oxygen ion fluences of (a) blank, (b) 0.11 × 1019 ions/cm2, (c) 0.22 × 1019 ions/cm2 (d) 0.33 × 1019 ions/cm2, and (e) 0.44 × 1019 ions/cm2.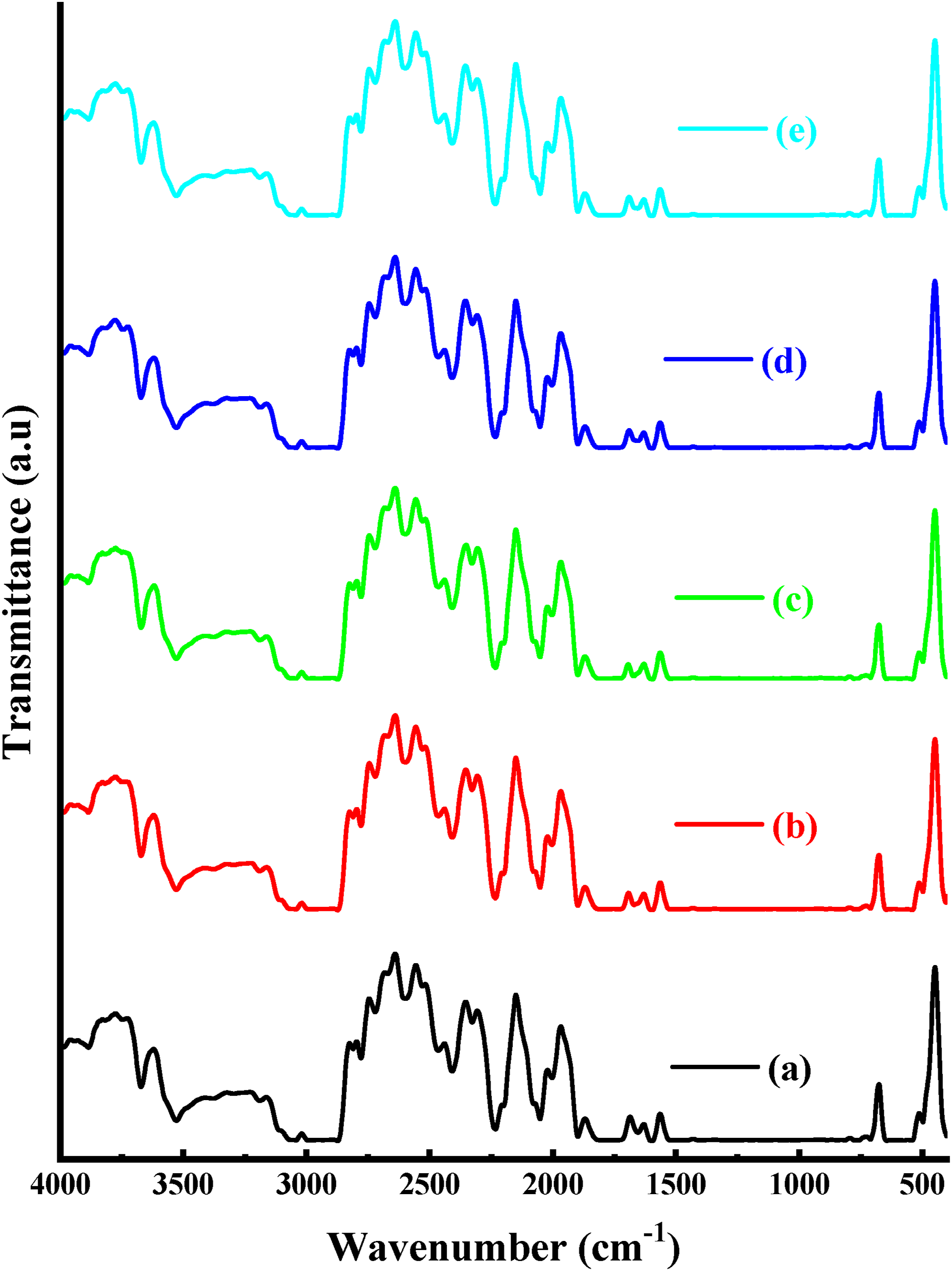
Surface examinations
Surface roughness
The roughness of the surface assessment is critical for several essential concerns, including friction, contact distortion, heat, and electric current conduction, touch joint tension, and placement precision. 30 The morphology of the surface is so intricate that a finite set of variables cannot offer an in-depth characterization. Accurate characterization can be achieved by boosting the assortment of variables. The most significant feature for characterizing surface topography is amplitude parameters. The surface roughness of blank and ion beam bombardment films was assessed with the surface roughness procedure. The approach relies on a sensor that goes over the outermost layer of the polymer film and monitors surface roughness indicators.
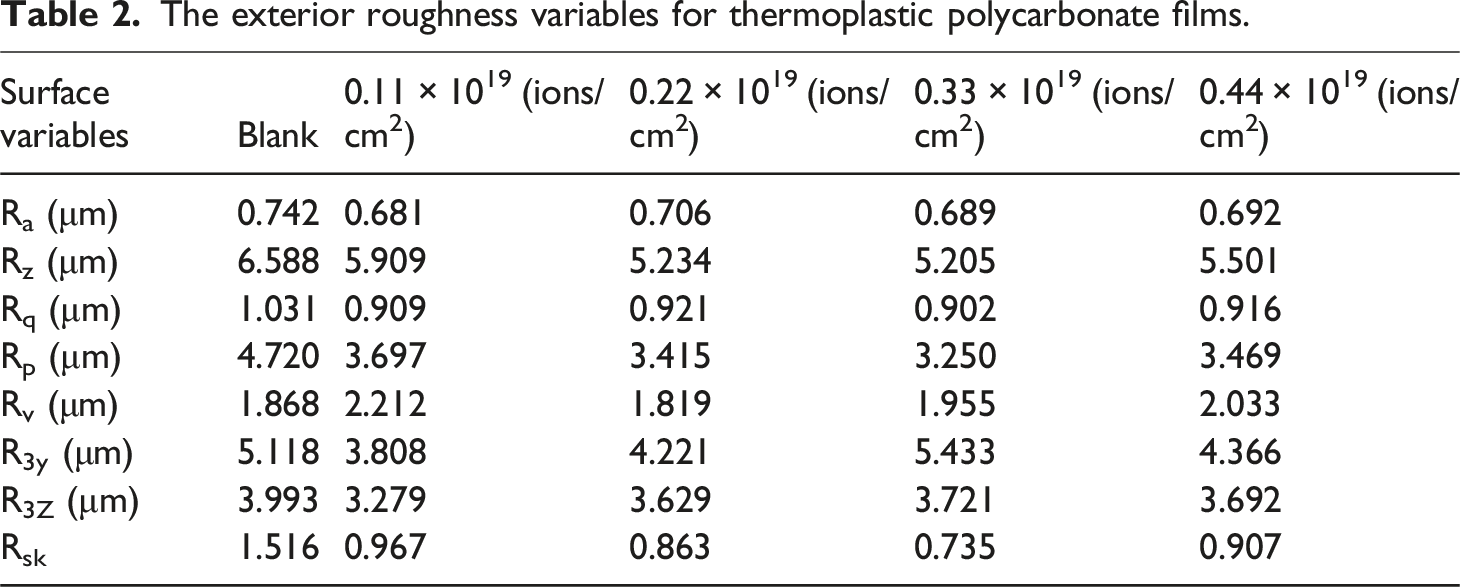
The exterior roughness variables for thermoplastic polycarbonate films.
Ra is described as the average absolute departure of the roughness anomalies from the mean line throughout the sample length.
28
The mathematical description and the numerical realization of the arithmetic average height variable may be studied by the subsequent equations.
Root means square roughness (Rq) is a vital statistic for describing surface roughness since it reflects the typical dispersion of the distribution of surface heights can be computed by subsequent formula.
32

(Rq) the root means square average of the profile height over the evaluation length, L.
The typically employed roughness parameter for basic quality assurance is the arithmetic average height variable, referred to as the center line average (CLA).
(Rz) (the profile’s Centre top height over the evaluation length, L). The Rz has the following algebraic meanings
Maximum altitude of peaks (Rp) is the greatest height of the pattern above the stander line within the examination length. The complete profundity of the profile below the purpose line within the evaluation length is depicted as the greatest deepness of valleys (Rv).
The gap between the third top peak and the 3rd deepest ravine is determined, and the greatest range is taken as the third location’s height (R3y). Third-point elevation median (R3z) is the standard deviation of the five third point height parameters (R3y1, R3y2, R3y3, R3y4, and R3y5) are utilized to determine this variable. This parameter’s mathematical explanation is as a result
The skewness (Rsk) of a profile is determined across the assessment length as the 3rd core instant of the profile intensity chance density curve. It is employed for figuring out the regularity of the profile around the intent line. This metric is prudent for deep dips and high peaks. A uniform elevation dispersion with the same number of peaks and ravines exhibits no skewness. Negative skewness is present in profiles with deleted peaks or extensive scratches. Positive skewness is found in profiles with full valleys or peaks of great height (Figure 3). Thermoplastic polycarbonate films both blank and bombarded, examined forms at various oxygen ion fluences.
There is no correlation between the fluence values and the roughness parameter values. When compared to the blank sample, the bombarded surface may have more surface roughness because of the creation of ion trails. This is because the oxygen ions ionize the polymer surface directly, depositing their energy on the target sample and creating species defects and clusters. Moreover, these surface material defects and clusters are reactive chemically. This refers to the development of polar components on the surface subjected to bombardment, which interact with liquid droplets to improve the wettability behavior and support various high-tech applications. 33
On the other hand, the creation of distinct surface parameters resulting from variations in the absorbed energy amounts could be the cause of the decrease in roughness parameters. Furthermore, it is possible that these differences in surface roughness parameters result from the oxidation products that break down the backbone chains as a result of the degradation process. 4
Ultimately, it can be concluded that, in our study, there is no correlation between the variations in contact angle measurements and surface roughness changes. Consequently, rather than surface roughness changes, the decrease in contact angle values may be caused by the creation of hydrophilic groups such polar species that were influenced by oxygen. 25
Surface wettability
When the liquid and solid surfaces meet, the angle created between the wetted solid surface and the tangent to the wet liquid surface is referred to as the contact angle (θ), which gives the measure of wettability. When the contact angle is zero, the liquid is evenly dispersed across the surface. This also indicates that the liquid completely covers the solid surface, a phenomenon known as complete wetting. However, if the contact angle equals 180°, the outcome is entirely non-wetting. On the other hand, if the contact angle value is less than 90o, the surface of a solid becomes wet. 34 The properties of surface energy, such as adhesion and wettability, can be determined by measuring the contact angle.
Three probe liquids were used in our study, including deionized water, glycerol, and N-dimethylformamide (DMF). Figure 4(a) illustrates the variation of the contact angles of the three probing liquids with the oxygen ion fluence (ions/cm2). It is known that the humidity, surface roughness, the kind of probing liquid, surface pollutants, surface rigidity, temperature, and drop size are several parameters that substantially impact the contact angle value.
35
The contact angles of the deionized water, glycerol, and DMF before bombardment to the oxygen ion fluence (blank sample) are 88.34o, 83.65o, and 81.22o, respectively, see Table 3. However, after bombardment to 0.44 × 1019 ions/cm2 of low-energy oxygen ions, the measured contact angles of the deionized water, glycerol, and DMF are 54.55o, 45.43o, and 38.56o, respectively, see Table 3. Consequently, the contact angle of the probing liquids was reduced as the oxygen ion fluencies were bombarded to the thermoplastic polycarbonate to a greater extent. This finding indicates that enhancing the surface wettability and the hydrophilicity of the tested polymer can be achieved by increasing the fluencies of oxygen ions. According to the data shown in Figure 4(a), the root mean square roughness (Rq) of the blank sample is 1.031 μm, while it has values of 0.909 μm, 0.921 μm, 0.902 μm, and 0.916 μm for the bombarded samples with 0.11 × 1019, 0.22 × 1019, 0.33 × 1019, and 0.44 × 1019 ions/cm2, respectively. Conversely, the values of Rq are not influenced by the fluencies of oxygen ions. This suggests that the decrease in the contact angle by increasing the oxygen ion fluence cannot be attributable to alteration in the surface roughness of thermoplastic polycarbonate. The enhanced hydrophilicity of the tested polymer by increasing the oxygen ion fluence can be attributed to the presence of a hydrophilic group as C = O,
36
which is in agreement with the investigated results of the FTIR analysis. Also, FTIR results illustrate that the modification of the tested polymer surface can result from C-O bond and C-O-C bond scission induced by the bombardment of oxygen ions.
37
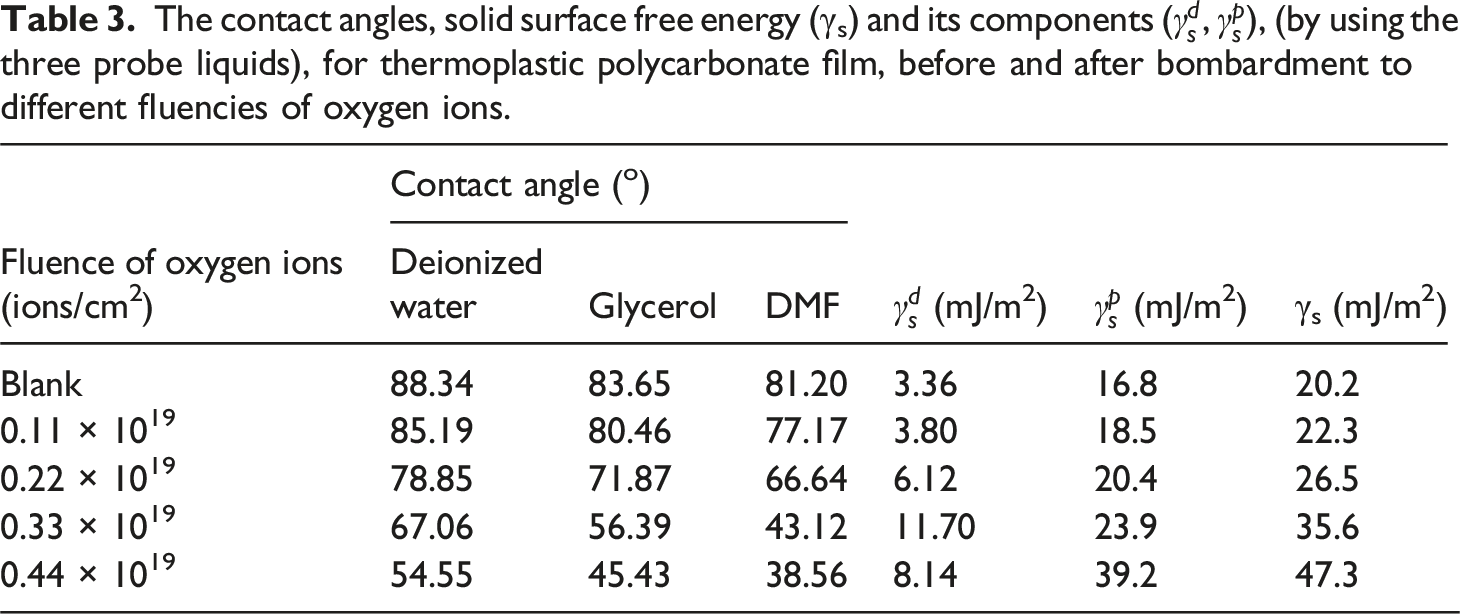
The variation of (a) the contact angle and the root mean square roughness (Rq), (b) the adhesion work (Aw), and (c) the spreading coefficient (Sc) with the oxygen ion fluence (ions/cm2). The contact angles, solid surface free energy (γs) and its components (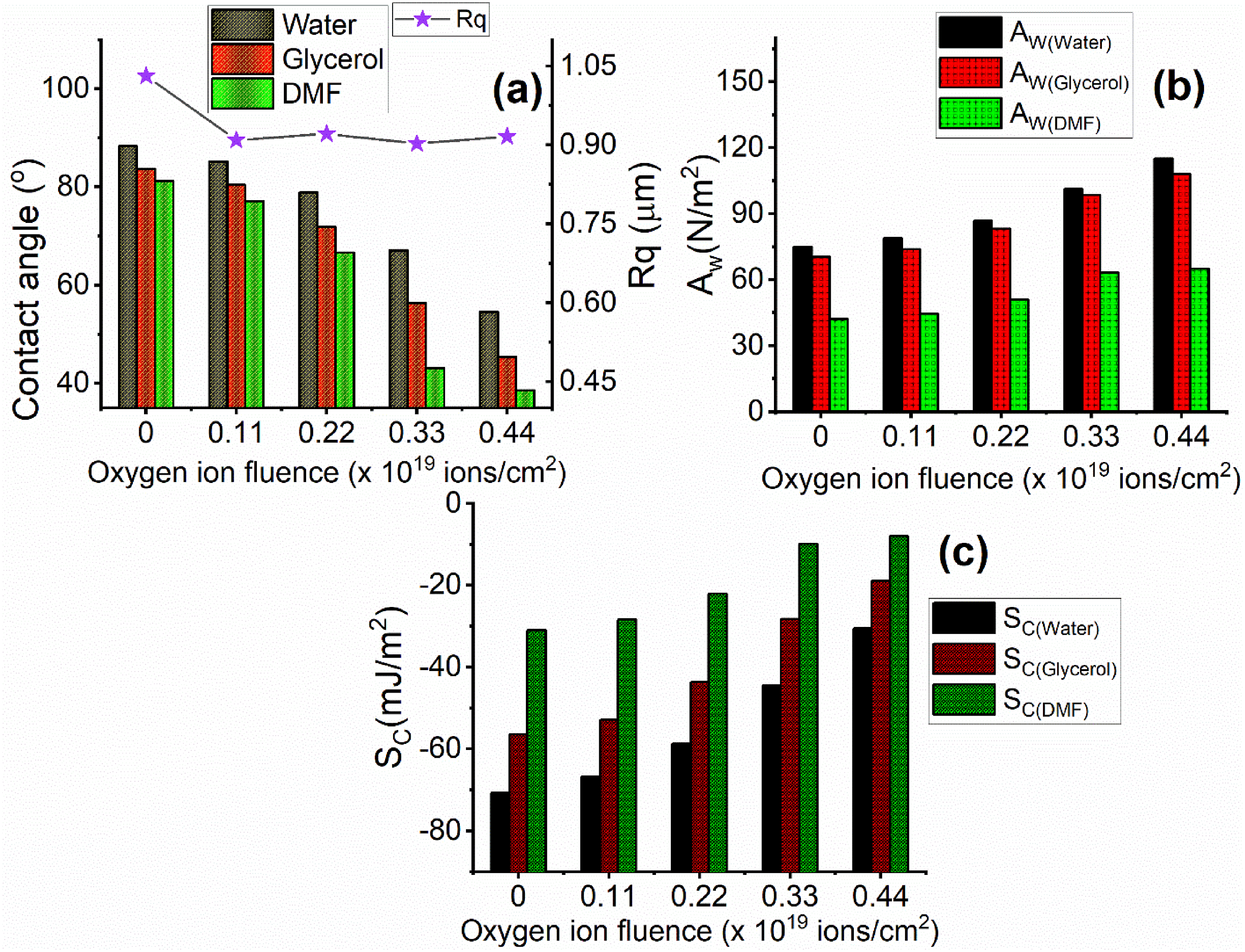
Calculating the thermodynamic adhesion work (Aw) can provide insights into the wetting behavior of a liquid on a solid surface. Specifically, it refers to the work necessary to separate the solid from the liquid. Furthermore, liquid wettability can be comprehended using the concept of liquid cohesion (Cw), which refers to the energy needed to separate a unit area of two contacting phases. The cohesive work (Cw) exhibits resistance, resulting in a contraction of the liquid. The standard Young–Dupre’ formula was used to compute the thermodynamic work of adhesion as follows38,39

The equilibrium spreading coefficient (Sc) refers to the disparity in surface free energy per unit area between the solid surface when it is dry and wet. A liquid will naturally wet and spread on a solid surface when the spreading coefficient (Sc) has a positive value. Thus, it is necessary to have negative Sc values to indicate a lack of wetting, resulting in contact angles with values greater than zero.
42
The equilibrium spreading coefficient (Sc) is also known as the quantitative measure of the difference between (Aw) and (Cw) as expressed in the following equation26,43
An increase in the values of the equilibrium spreading coefficient (Sc) was observed by increasing the oxygen ion fluence (ion/cm2) for the three probe liquids, as illustrated in Figure 4(c). Deionized water showed the lowest value of Sc (−70.69 mJ/m2) before bombardment to the oxygen ion fluence (blank sample). However, after bombardment to the highest oxygen ion fluence (0.44 ×1019 ions/cm2), the DMF probe liquid showed the highest value of Sc (−7.95 mJ/m2). As a result, DMF has a higher wetting and spreading ability on the surface of the thermoplastic polycarbonate film compared to the other liquids being tested.
The solid surface free energy (γs) and its components (
Surface free energy is a thermodynamic quantity that indicates the degree of imbalance in intermolecular interaction at the interface between two mediums. Furthermore, it explicitly influences the actual magnitude of the contact angle. It is known that the intermolecular forces of attraction within the liquid are greater than those between the liquid surface and the solid surface in case of putting a high surface tension liquid droplet on a solid surface with a low surface energy. Therefore, a specific measurement exists for the angle at which it contacts the surface. Nevertheless, when the liquid’s surface tension is reduced or the solid’s surface free energy is elevated, the attractive force between the liquid molecules and the solid atoms surpasses the force between the liquid molecules. Consequently, the liquid begins to spread across the solid surface. The total surface free energy (γi) of each phase consists of two main parts as proposed by Fowkes44,45
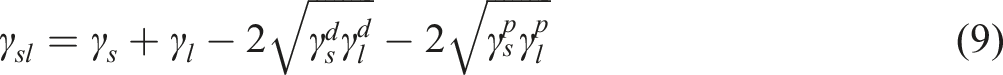

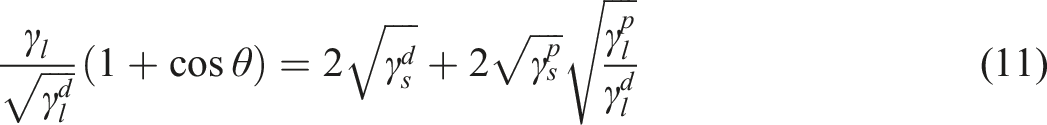
The variation of the quantity of ( The variation of the quantity of (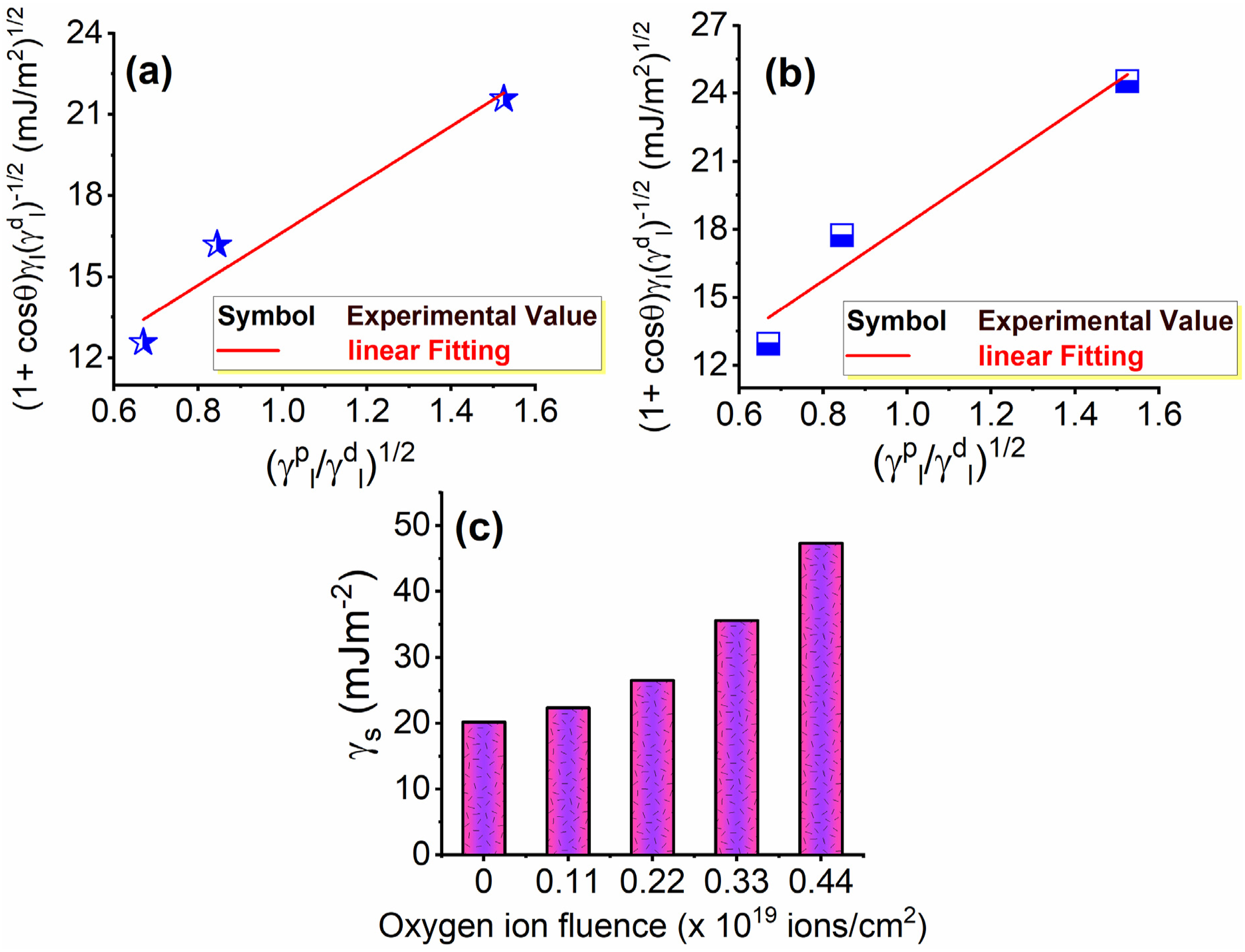
If the total surface tension of the probe liquid (γ
l
) is less than or equals to the solid surface free energy (γ
s
), the liquid drop becomes flat immediately and the solid will suffer from wetting which is not suitable for contact angle measurement.
26
Except for the probe liquid of DMF at the maximum influence of oxygen ions, Table 3 shows that the values of γ
s
are smaller than the total surface tension of the three probe liquids (γ
l
) before and after exposure to varying fluencies of oxygen ions. Consequently, the deionized water and glycerol succeeded in investigating the condition of
Optical properties
Figure 6(a) depicts the changes in the absorption spectra of the thermoplastic polycarbonate film when bombarded with different fluencies of oxygen ions. A dent was found in the wavelength range of 300 nm to 490 nm for the blank sample as a characteristic behavior of the thermoplastic polycarbonate film consistent with a previously published study.
49
An increase in the absorption spectra was observed by increasing the fluence of the oxygen ions. Nevertheless, the observed dent in the pristine sample suffered from a reduction in its shape by improving the fluence of the oxygen ions, see the inset in Figure 6(a). Also, the position of the lowest point in the dent showed a red shift by increasing the influence of oxygen ions as it changed from 343 nm for the blank sample to 373 nm for the bombarded sample with 0.44 × 1019 ions/cm2. This indicates an alteration in the bottom of the conduction band and the upper of the valence band.
50
The change in the absorption spectra of the blank sample compared to the bombarded samples can be attributed to band-to-band transitions caused by the free radicals induced by oxygen ions bombardment. Furthermore, the alteration in the absorption spectra of the bombarded samples is noticeable in the dent region, whereas their absorption spectra at longer wavelengths are nearly indistinguishable. (a) The variation of the absorption with the wavelength. (b) The variation of the skin depth with the photon energy (E). (c) The dependence of the quantity of (αE)2 on the photon energy (E), and (d) The dependence of the values of Eg, N, and M on the oxygen ion fluence (ions/cm2).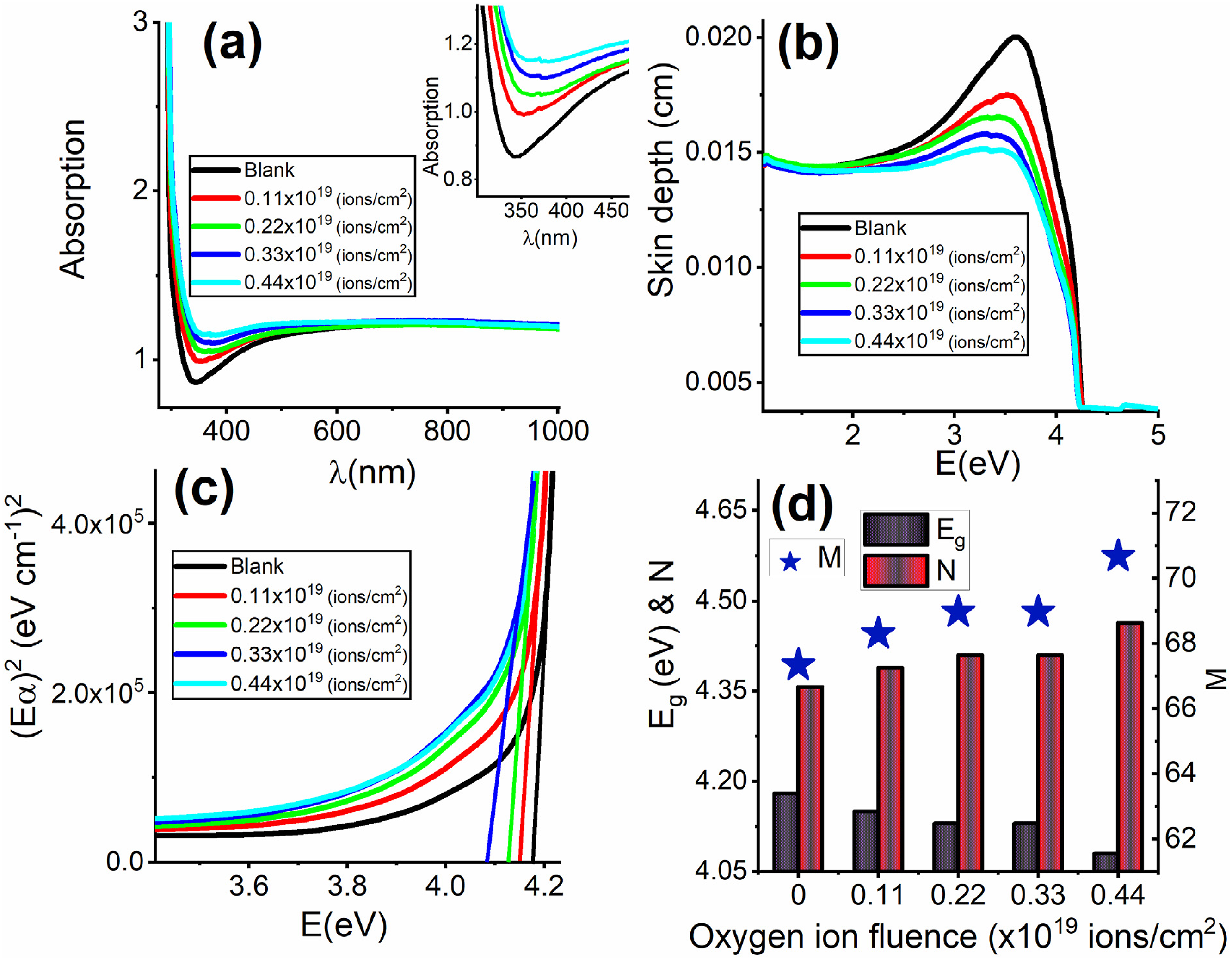
Skin depth refers to the depth at which an electromagnetic wave can effectively permeate a material. The skin depth can be determined by dividing the sample’s thickness by its absorbance. 51 Except for the dent region, which has a range of 2 eV to 4.18 eV, Figure 6(b) indicates that the skin depth lacks any dependence on the oxygen ion fluence (ions/cm2) in any respect. Increasing the fluence of oxygen ions (ions/cm2) resulted in a decrease in the skin depth of the bombarded samples, particularly in the dent region, which indicates the reduction of their transparency. This finding is consistent with several published studies.52,53
Understanding the absorption process is crucial for comprehending the band structure of polymeric materials. Quantum mechanics dictates that the electronic transitions between states can be controlled by selection rules, which are determined by the band structure. The fundamental absorption influences the band-to-band transitions. To ascertain the energy of the band gap (Eg), Tauc’s model was used by plotting the quantity of (αE)1/n against the photon energy (E) according to the following relation54–56
The absorption process occurs when the electron is elevated from the ground to the excited state. The conjugation system generates one p orbital adjacent to a double bond, potentially allowing compounds with unsaturated centers to undergo π – π* transitions. This conjugation reduces the energy absorption in the UV and visible regions, thanks to the necessary energy for π – π* transition. Studying the optical characteristics of polymers can yield valuable insights into the release of carbon atoms derived from their chains during irradiation. Subsequently, the liberated carbon atoms are joined together to form clusters. The band gap energy (Eg) efficiently conveys information about the number of carbon atoms in a conjugated chain structure (N) and a carbon cluster (M) by using the following relations49,57,58

The refractive index (n) is regarded as one of the most essential optical factors for characterizing polymers. It is closely associated with electronic polarizability and the local electric field. Moreover, the refractive index is crucial in identifying the chemical bonding and density of the substance of the investigated samples. Fresnel’s formula was used for calculating the refractive index
59
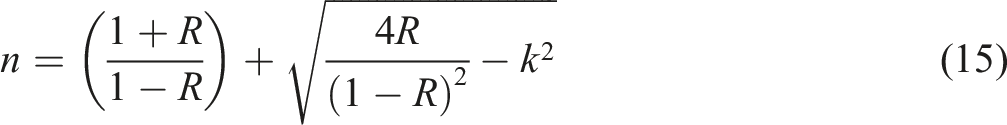

While k is the extinction coefficient, which is based on the incident wavelength (λ) and the absorption coefficient (α)
61

The extinction coefficient (k) is a crucial metric in optical applications that quantifies the energy loss resulting from molecules’ absorption and scattering processes in the tested samples. Figure 7(a) and 7(b), respectively, illustrate the alteration of the extinction coefficient (k) and the refractive index (n) by increasing the incident wavelength and the oxygen ions' fluence. At lower wavelengths (prior to the dent region), the extinction coefficient (k) diminishes due to the sufficient energy to cause an excitation of the electron to a higher energy level and vice versa at higher incident wavelengths (after the dent region). The variation of (a) the extinction coefficient (k) and (b) the refractive index (n), with the wavelength. The dependence of (c) the extinction coefficient (k) and (d) the refractive index (n), at different constant wavelengths (340 nm and 650 nm), on the oxygen ion fluence (ions/cm2).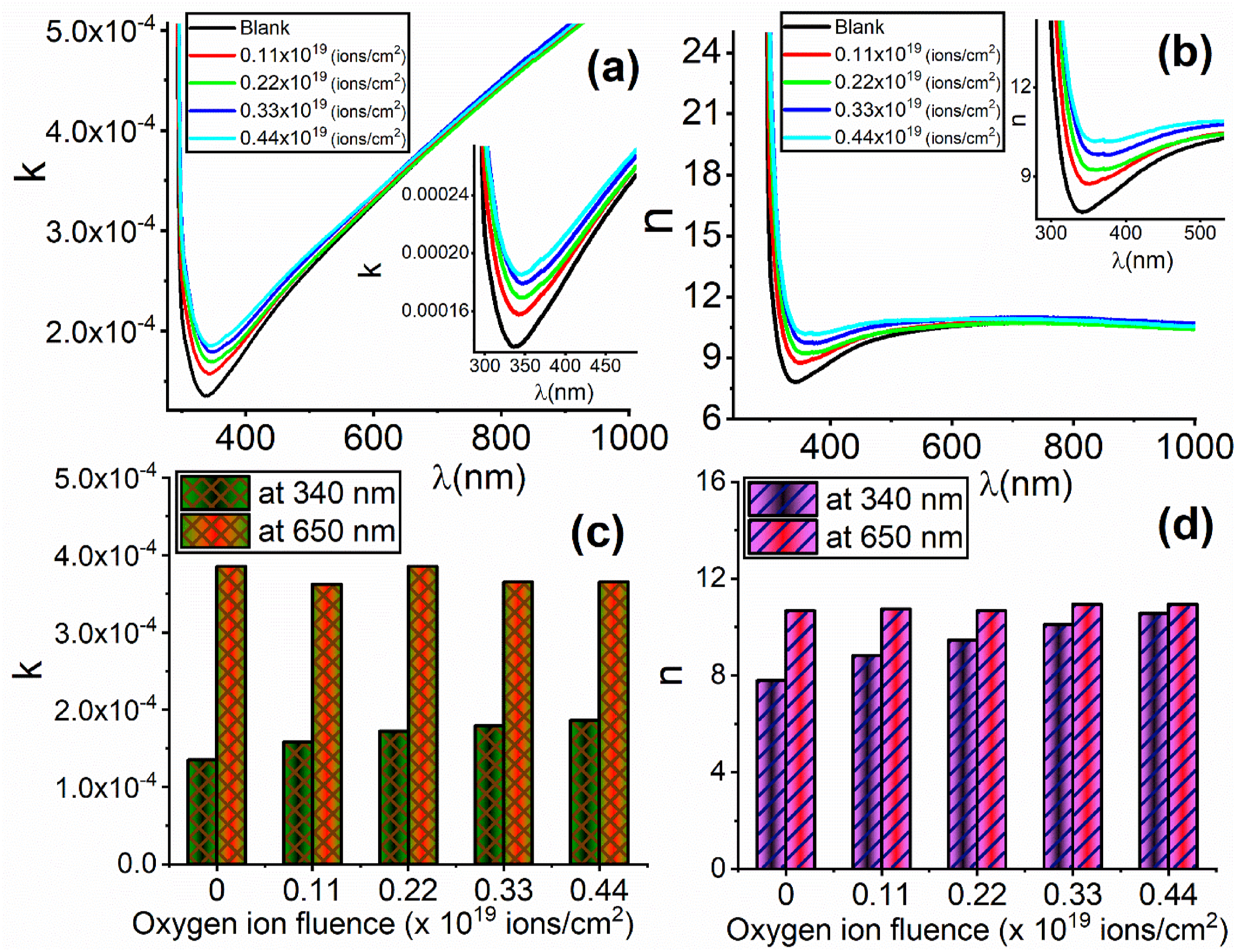
A noticeable enhancement of the values of k and n was detected in the dent region (see the insets in Figure 7(a) and 7(b), respectively) by increasing the oxygen ion fluence (ions/cm2). However, the other regions of the incident wavelengths display an independence of the values of n and k on the fluence of the oxygen ion. This can be understood from Figure 7(c) and 7(d), respectively, which illustrate the increasing of the extinction coefficient (k) and the refractive index (n) by increasing the oxygen ions fluence at a constant wavelength of 340 nm, which is in the dent region. A higher refractive index is primarily advantageous for enhancing the visual properties of electronic displays, including televisions with quantum dot QDLED, OLED, and LCD panels.
However, at 650 nm, the values of n and k are independent of the ion fluence. Ion bombardment induces an augmentation in the creation of covalent bonds among different chains by the action of free radicals. This leads to changes in the densities and refractive indices of the samples under investigation.62,63 In addition, the bombarded films exhibit an elevated density of defects, leading to a rise in the absorption coefficient and, thus, an increase in the value of the extinction coefficient (k).
The optical conductivity (σoptical) offers valuable information about the electronic states of a material. It is a representation of the electrical conductivity that is formed as a consequence of the transfer of charge carriers owing to the fluctuation in the electric field of the electromagnetic waves that have fallen. The optical conductivity (σoptical) was calculated using the formula that takes into account the refractive index (n), the velocity of light in a vacuum (c), and the absorption coefficient (α)
64

Figure 8(a) displays the variation of the optical conductivity (σoptical) with the incident wavelengths for the different bombarded samples. Consistent with the absorption and refractive index patterns (see Figures 6(a) and 7(b), respectively), the optical conductivity spectra are also present. (a) The variation of optical conductivity (σoptical) with the wavelength. (b) The variation of optical conductivity (σoptical) with the oxygen ion fluence at different constant wavelengths (340 nm, 650 nm, and 900 nm).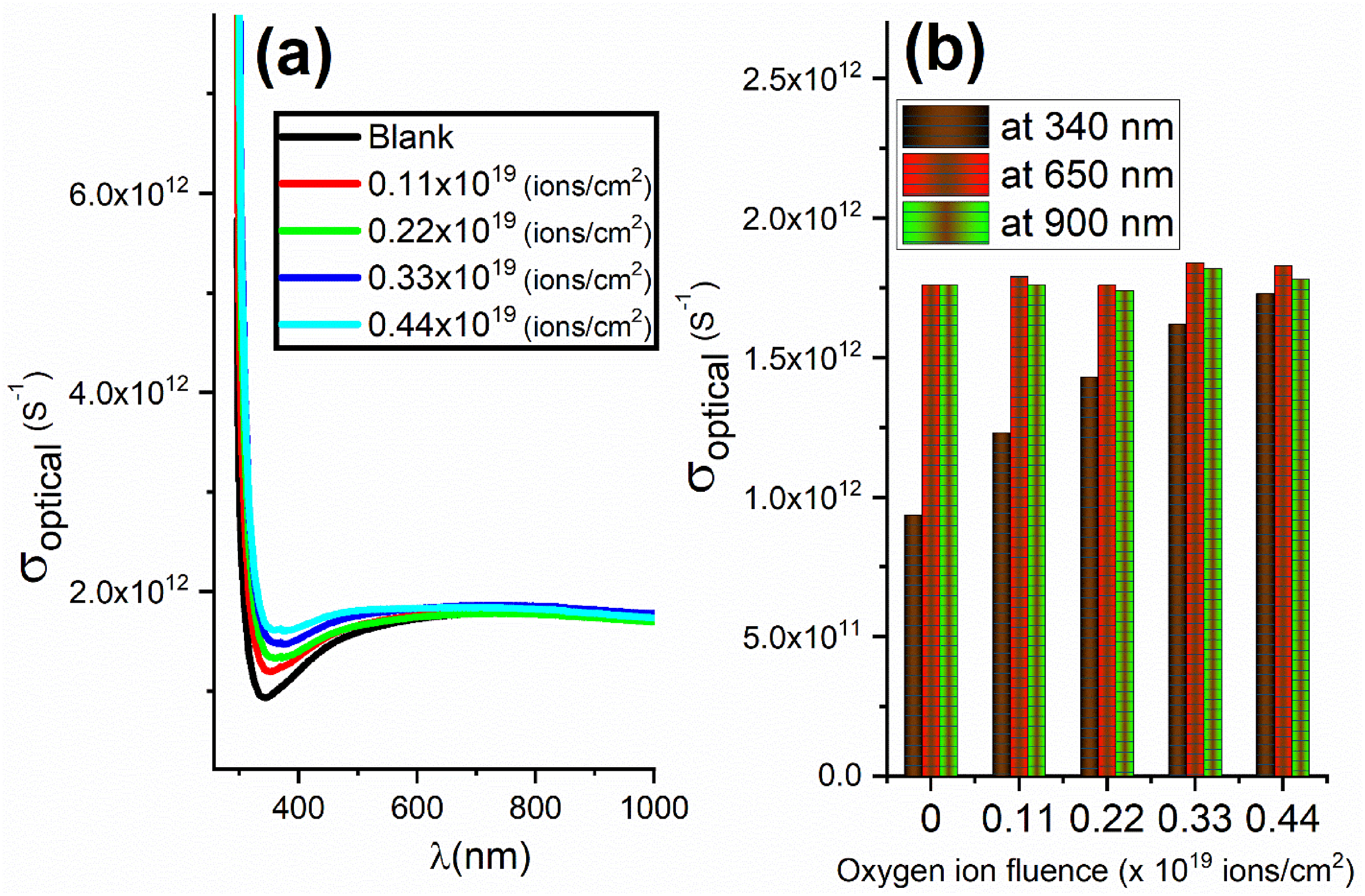
In the dent region, the values of σoptical noticeably rose as the fluence of oxygen ions was raised. Nevertheless, at longer wavelengths, the optical conductivity remains unaffected by the fluence of the bombardment. This finding can be confirmed by Figure 8(b), which shows the variation of σoptical with fluence of oxygen ions at some constant wavelength. The lack of reliance of the σoptical on the fluence of the bombardment is evident at wavelengths of 650 nm and 900 nm. In contrast, the apparent change of the optical conductivity was evaluated at a wavelength of 340 nm (the dent region).
Conclusion
It has been investigated the effect of oxygen ion beam bombardment on the thermoplastic polycarbonate films to enhance their physical characteristics. The results of this work showed that, following an oxygen ion beam bombardment of the films, modifications were significantly induced in the wetting behaviors, surface roughness, and optical characteristics. The enhanced hydrophilicity of the tested polymer by increasing the oxygen ion fluence can be attributed to the presence of a hydrophilic group as C=O, which agrees with the investigated results of the FTIR analysis. DMF has a higher wetting and spreading ability on the surface of the polymer films compared to the other liquids being tested. Therefore, the oxygen irradiation significantly enhanced the biocompatibility parameters, where the free radical produced during irradiation reacts with oxygen to produce polar groups on the irradiated surface, increasing surface polarity and contributing to these improvements in biocompatibility properties. The change in the absorption spectra of the blank sample compared to the bombarded samples can be attributed to band-to-band transitions due to the free radicals induced by oxygen ions bombardment. Except for the dent region, the skin depth lacks any dependence on the oxygen ion fluence (ions/cm2) in any respect. The values of N and M increase as the ion beam fluences increase. This is because the polymer chain suffers a breakdown of C-H bonds, leading to the release of hydrogen. At lower wavelengths (prior to the dent region), the extinction coefficient (k) diminishes due to the sufficient energy to cause an excitation of the electron to a higher energy level and vice versa at higher incident wavelengths (after the dent region). The bombarded films exhibit an elevated density of defects, leading to a rise in the absorption coefficient and, thus, an increase in the value of the extinction coefficient (k). In the dent region, the values of σoptical noticeably rose as the fluence of oxygen ions was raised. Nevertheless, at longer wavelengths, the optical conductivity remains unaffected by the fluence of the bombardment. The characterization analyses clearly highlighted that the ion bombardment technique is a valuable tool for improving the hydrophilic, structural, and surface properties of polymers by altering their physico-chemical properties. These findings are applicable to applications such as coating, cleaning, printing, painting, hydrophilic coating, dispersion, syringes, catheters, joint implants, medical tubing, and bonding.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
