Abstract
Biomimetic nanoparticles (BioM NPs) are an innovative strategy for nanomedicine (NanoMed) that combines the advantages of artificial materials with the biological functioning of natural systems. Ongoing research attempts to enhance their design, and stability, and solve problems for broader applications in medicine and biotechnology. NanoMed ideas are intensively researched in preclinical research because they provide unique advantages in disease management. It has developed novel therapeutic strategies for combating viral infections and increasing treatment success rates. The creation of novel nanocarriers and drug delivery systems is critical to the evolution of NanoMed, but there is still much to learn about this quickly evolving field. To contribute to existing knowledge and cover knowledge gaps, our present review article will discuss the state-of-the-art of BioM NPs in NanoMed in three sections. First, the methods of BioM NP production and their synergistic interactions with human cells, tissues, organs, and organ systems will be discussed. Followed by a discussion of BioM NPs) for several NanoMeds (wound healing, targeted drug delivery, theranostics, cancer therapy, and bone formation and/or regeneration), and concluding with the novel application of BioM NPs, challenges associated with these novel applications, and prospects.
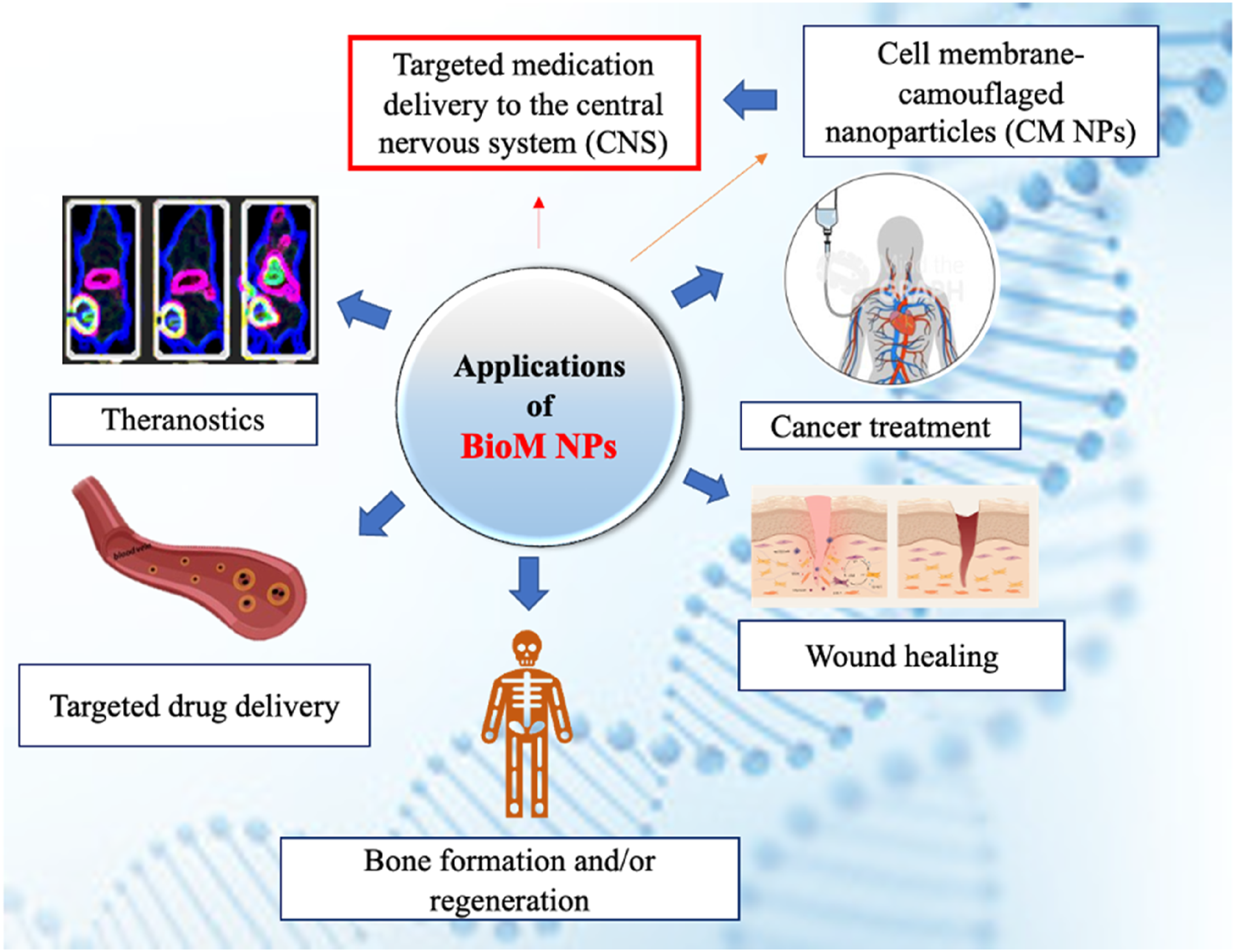
Highlights
• Magnetic BioM NPs are scalable and capable of chemical modification. • Exosome-derived vesicles as carriers for drug deliver. • Synergy between NPs and peptides/proteins leads to rapid clearance but hinders complete use in nano-based diagnostics. • BioM NPs show significant potential for wound healing, drug delivery etc. • NanoEL promotes NPs escape from the vasculature and boost therapeutic drug delivery to target tissues.
Introduction
Biomimetic nanoparticles (BioM NPs) have contributed significantly to the medical field. 1 Today’s patients require treatments that may effectively address a variety of biological issues, including drug delivery in biomedical applications. 2 This is where the concept of biomimetics has a significant role to play. BioM NPs are novel nanoscale structures that mimic and exploit the innate features of natural biological systems. These NPs are created to replicate certain properties of biological entities for diverse medicinal purposes, 3 drawing precedent from the intricate architecture and capabilities of cells, viruses, or extracellular components.4,5 These NPs are developed with functionalization that mirrors the biological system either physically or functionally, 6 providing biological functioning and structure. 5 They could have synthetic architectures and artificial antigen-presenting cells coated with materials derived biologically. 7 NPs offer an extensive array of possibilities due to their spherical form 8 and surface area-to-volume ratio. Rapid advancements in NP technology have made it possible to treat a wide range of illnesses, including neurological disorders like Parkinson’s and Alzheimer’s, 9 breast cancer, cervical cancer, 10 metastatic cancer, 11 and cholangiocarcinoma 12 among others.
The ability to protect drugs from premature degradation and interaction with the biological environment, 13 improve drug absorption into particular tumorous tissue, 14 and control the distribution profile and pharmacokinetics of the drug in tissues, 15 which minimizes systemic toxicity 16 and increases the antitumor effects of therapies are interesting benefits of BioM NPs that are worth exploring.17,18 Because of their distinctive properties and capacity to penetrate extracellular barriers and deliver therapeutic drugs directly to tumour cells, 19 BioM NP holds great promise as a method for the delivery of drugs into a variety of solid tumours.20–22 Besides, BioM NPs have been recommended for applications in tissue engineering via extracellular-matrix-based materials, 23 genomics, proteomics, diagnostics, 24 and other biomedical fields considering their potential to extend and improve life. 25 In an attempt to provide natural antioxidants with higher bioavailability, 26 improved solubility, and enhanced permeability in the stomach, with fewer side effects than dietary antioxidants, innovative therapeutic approaches based on BioM NPs are being employed. 27
BioM NPs are minuscule particles that may be altered to bind particular receptors,10,28 which makes them an ideal option for targeting the tumour microenvironment (TME). 29 They can also be created to contain drugs or other therapeutic agents.16,30,31 Compared to conventional chemotherapy and radiation therapy, BioM NPs provide several benefits and can be used with other therapeutic techniques.32,33 Despite the enhanced antitumor performance and low off-target effects, 34 BioM NPs in nanomedicine (NanoMed) have attracted, their limitations in clinical applications such as rapid clearance from the body, low biological stability, and poor targeting ability cannot be overlooked. 25 BioM NPs still pose challenges such as extracting biological materials for re-administration to patients which may present ethical concerns3,13 and also generating large amounts of BMNPs would be extremely costly. 28
Although the development of NanoMed is evident in the treatment and diagnosis of oesophagal cancer, extensive research into the diagnostic and surgical potential of NPs is needed before clinically deployed. 35 Owing to the insufficient research in the field, of cancer therapy, 19 the effective and safe administration of anticancer drugs, 32 most of which are toxic to normal cells, have low bioavailability and are not stable in vivo is lacking. 17 However, according to the research25,36,37 that can currently be accessed, BioM NPs are capable of providing numerous benefits in NanoMed. The concepts of NanoMed are extensively investigated in preclinical research as they present particular perks in the management of human diseases. It has created new therapeutic mechanisms that can be utilized to combat viral infections and raise the success rates of treatment. The development of innovative nanocarriers and drug delivery systems is essential to the advancement of NanoMed but there is still a lot to learn about this rapidly developing sector. Herein this review addresses the state-of-the-art of BioM NPs in NanoMed in three main parts: (1) Mechanisms of BioM NPs synthesis and their synergistic relationships with human cells, tissues, organs, and organ systems. (2) BioM NPs for wound healing, targeted drug delivery, theranostics, cancer therapy, and bone formation and/or regeneration (3) Novel application of BioM NPs, challenges associated with these novel applications and their prospects. This review seeks to provide researchers with an overview of the current state of research on BioM NPs for NanoMed, to serve as a guide for current and future studies.
Synthesis of biomimetic nanoparticles (BioM NPs) for nanomedical research
BioM NP synthesis entails using biological principles to create NPs with specific features for use in a variety of NanoMed applications. These methods provide benefits like as biocompatibility, targeted transport, and controlled release, making BioM NPs viable tools for improving NanoMed. While conventional methods of nanocarrier-mediated drug delivery have been mainly focused on synthetic approaches, 38 researchers 39 have exploited engineering strategies that combine synthetic NPs with natural biomaterials40,41 and have recently gained much attention. This includes a new class of BioM NPs known as cell membrane-camouflaged NPs (CMNP) which combine the unique functionalities of cellular membranes and the engineering versatility of synthetic nanomaterials (NMs) for the effective delivery of therapeutic agents. 42 This has materialized as a novel drug delivery strategy with the ability to enhance the therapeutic efficacy for the treatment of a variety of diseases. Although the CMNPs inherit the composition of cell membranes, 43 including specific receptors, antigens, and proteins, for target delivering to tumours, escaping immune from clearance and prolonging the blood circulation time.13,44 BioM NPs have a promising approach to overcoming these limitations, understanding that the cells 45 of the body have integral abilities to overcome barriers due to their cell membrane properties. 13 In cancer theranostics for instance, BioM NPs have emerged to possess advantageous bioavailability, circulation time, and lower toxicity in showing significant improvements over traditional drugs and similar non-BioM NPs and their increased tissue compatibility improves their efficacy and safety of therapies. In this rapidly accelerating field of NanoMed, the treatment of breast cancer using BioM NPs-based treatment is being studied and is currently being considered for cervical cancer treatment as well. 46 Recent research in nanotechnology has yielded a multifunctional theranostic platform for the treatment of diseases like glioblastoma by developing blood–brain tumour barrier penetrability, efficient tumour accumulation, precise tumour imaging, and significant tumour inhibition of brain cancer. 47 While providing the intricate physiological functions of normal cell membranes, biomimetic nanovessels preserve the physicochemical characteristics for effective drug delivery.
Magnetic BioM NPs (MBioM NPs) are a promising technology due to their scalability and the ability to be modified through chemical or genetic engineering. 8 Besides, Abu-Abdeen et al. 48 employed a solvent thermal approach based on a thermal procedure at critical temperatures and pressures to manufacture cobalt iron oxide magnetic NPs with a molar ratio of 2. Physicochemical methods have been used to create MBioM NPs, 49 with a small portion devoted to biological methods like protein constructions, bacteria, and fungi. 50 Bacterial magnetosomes (MTs) can be used to improve magnetic contrast in magnetic resonance imaging (MRI) as well as in the treatment of diseases like cancer. 51 Besides, metal/metal oxide NPs can be modified by altering their surface, and micro- or nano environments during manufacturing.52,53 BioM metal oxides, such as ferritin, 54 viral capsids, and bacterial cells, 55 create controlled conditions for NPs, 56 limiting shape and size distribution.57,58 These techniques are attractive due to their environmental benefits. 58 Large-scale production using organisms, 45 such as plant parts, is increasingly being explored,59,60 leading researchers to explore more advanced methods of biological BioM NP synthesis. However, simple approaches such as melt compounding by Xing et al. 61 for the production of polyphenylene sulfide (PPS)/graphite nanoplates (GNPs) nanocomposites have been studied.
Furthermore, molecular methods for protein and peptide construction can be applied to create nano-sized crystals 62 and self-generation of multi-purpose NPs.16,63 Solid-binding proteins/peptides 64 can achieve these NPs under controlled synthesis and arrangement.57,65 BioM degradation based on proteins and peptides has demonstrated efficiency in generating MBioM NPs and ceramic MBioM NPs. 66 Ceramic development NPs are used to create replicable biocompatible materials that mimic real tissues and cells, often used in bone tissue engineering.66,67 Furthermore, while the number of implants has progressively and continuously climbed over the years to roughly one million per year worldwide, there is still a lot of room for development in the field of dental implantology, which is normally increasing rapidly. 68 However, these BioM NPs can sometimes cause disorder and unsatisfactory results. 69 The development of 3D cell/tissue depends on environmental conditions, synthetic extracellular medium, and active constituents. 70 Ceramic BioM NPs can also act as carriers to immobilize multiple NM compounds, 71 enhancing their catalytic efficiency and cell/tissue engineering.71,72
Regarding nanomedical research, BioM NPs are synthesized by mimicking natural mineral formation using organic molecules as templates, often resulting in NPs with controlled properties, and by utilizing the spontaneous organization of molecules into ordered structures, creating NPs from amphiphilic molecules such as lipids or surfactants. Furthermore, biological templates such as viruses or cell membranes can be used to guide the formation of NPs, allowing for precise control over size, shape, and surface properties. Attaching biomolecules such as proteins or nucleic acids to pre-formed NPs allows for functionalization for specific biomedical applications. In other circumstances, using exosome-derived vesicles as natural carriers for drug administration or imaging, as well as inheriting biological features for targeted and controlled distribution, have all been investigated as BioM NP manufacturing approaches.
Synergistic relationship between BioM NPs and human cells, tissues, organs, and organ systems
BioM NPs are developed with functionalization that mirrors the biological system either physically or functionally as they mimic the innate features of natural biological systems. 73 BioM NPs’ unique properties enable the transportation of liquid-insoluble pharmaceuticals, 6 prevent drug discharge, and enhance bio-distribution, 15 pharmacokinetics, 19 and intracellular absorption, 20 while their small size facilitates passive tumour/cancer targeting. 43 The non-specific build-up of untargeted theranostic drugs in other tissues reduces therapeutic efficacy. 74 Two strategies for improving NP tumour selectivity are passive targeting and active targeting via a specific cell-surface receptor. 75 Integrins and heterodimeric cell bonding peptides, 75 and HCBP are involved in cell attachment, angiogenesis, and solid tumour metastasis.19,76 It is expedient to understand the engineering of physicochemical properties on materials’ surfaces for target biological functions to develop next-generation nanotechnology.77,78 Fathy et al. 79 investigated the physicochemical and antibacterial characteristics of silver-coated silica (SiO2-Ag) NPs as an active nanofiller in the polypropylene thermoplastic matrix. Upon characterization of the changed polypropylene after loading with SiO2-Ag NPs. it was discovered that the as-synthesized SiO2-Ag nanocomposite has better physicochemical properties than the other two components. 79 Besides, BioM Silicon (Si) NPs are widely used in drug distribution technologies 80 and as visual differentiating mediators due to their properties like liquid solubility, 81 biochemical and thermal stability, reduced toxicity, 82 and excellent biological compatibility. However, their immobilization is often limited by surface characteristics. 13 Mesoporous silica NPs have a highly organized architecture with consistent mesopores, despite their amorphous silica wall. 83 Bio-templated mesoporous silica NPs have a higher capacity due to various techniques for producing these NPs, 84 which have an organized interior with consistent mesopores. 85 An intricate design for synthesized Si NPs was put forth for DNA transfection and cellular/tissue absorption, among other uses. 86 Given their biocompatibility, 87 and ability to interact with different polymers show potential for use in biomedical applications such as drug distribution and NanoMed devices. 17
BioM NPs are used in diagnostics to analyze biological fluid samples and interact with complex protein mixtures (CPM). 88 Protein adsorption on nanoscale surfaces is complex, and NPs have a large surface area and similar characteristics to proteins. 89 Protein-material interactions at nano-, 90 meso-, and macro-scales are quantitatively and qualitatively distinct. 89 Additionally, BioM NPs interact with biological fluids, 76 becoming covered with peptides or proteins, altering surface charge and properties, 13 and leading to functional loss and increased hydrodynamic mass.13,28,90 The synergy between BioM NPs and CPMs involves minimizing non-specific protein interaction, limiting the immunogenicity of protein complexes, exploring BioM NPs-CPMs’ impact on personalized NanoMed, and leveraging protein binding’s beneficial properties.
Also, the synergy of NPs with peptides/proteins in biological models leads to rapid clearance but hinders their complete use in nano-based diagnostics.13,84 Novel diagnostic NPs are being developed for identifying and quantifying biomarkers in biological models, providing tools for tailored nanotechnology.35,80 When used as ultrasensitive signal transducers and protein biosensors, 91 these NPs have remarkable physicochemical features. 92 High-selectivity techniques face challenges like noise signal deconvolution, 93 crucial for biofouling, and detecting signals from limited concentration analytes with high amounts of non-specific proteins (NSP). 94 Non-specific binding is a significant issue, 95 causing false positives and reduced signal-to-noise ratios, making proteomic signature analysis challenging. 96
Therapeutic nanocomposites with similar dimensions to pathogens have been selectively chosen for evolution, making reduced immunogenicity crucial. 97 Presently, the immunological responses to NPs are poorly understood, hence further research is needed to determine which properties warrant repeated systemic delivery without adverse effects. Animal trials have found accelerated blood clearance (ABC) in nano-constructs, where sensitization results in transient immunological response and the production of IgM antibodies.98,99 The type of nanomaterial, dose, and physicochemical properties influence ABC. 98 Encapsulation of cytotoxic chemicals can impact ABC negatively, possibly affecting B cells responsible for IgM release. ABC has minimal impact in a modern context, but future research on NPs for different diseases and pharmaceuticals will need to address this problem. 100
Biomimetic nanoparticles (BioM NPs) in nanomedicine
BioM NPs provide adaptable platforms with configurable features to solve critical NanoMed concerns such as targeted drug delivery, wound healing, 88 bone formation and/or regeneration, theranostics and cancer treatment. Their BioM nature improves biocompatibility, effectiveness, and specificity, making them excellent instruments for developing healthcare innovations.
BioM NPs for wound healing
Wound healing, a complex yet dynamic vital physiological process normally involves three phases 101 : hemostasis/inflammatory, 102 proliferative, and remodelling. 103 It is essential for preserving skin integrity after trauma with abnormalities like hypertrophic scars or chronic ulcers impairing normal physical function. The process is supported by a variety of cellular events. 103 The insufficient understanding of the cellular and molecular mechanisms of dermal repair in the skin, the largest organ of the human body, results in a lack of effective wound-healing therapies. Skin repair involves the precise synchronization of various types of cells in a series of sequential steps. 103 The microenvironment, influenced by changes in mechanical forces, oxygen levels, chemokines, extracellular matrix, 13 and growth factor synthesis, directly affects cellular recruitment and activation, thereby affecting wound healing. 103
In exploring wound healing, specifically with deep wounds using growth factors such as basic fibroblast growth factor (bFGF) and vascular endothelial growth factor A (VEGFA), Wang et al.
104
inserted bFGF and VEGFA genes into plasmid (pEGFP-N1) vectors. Later, gene-loaded nanoparticle complexes were formed by loading assembled plasmids onto NPs. These complexes were then wrapped with platelet membranes, mimicking platelet characteristics, to target inflammatory damage sites. After administering PM@gene-NP complexes through rats’ tail veins, wound healing experiments were conducted. The results showed that the PM@gene-NP complexes effectively targeted burn sites, increasing blood flow and healing speed in rats. Histological results showed less inflammatory cells in burned skin tissue, and increased production of hair follicles, sebaceous glands, and other skin accessories after wound healing. Finally, it was concluded from the study that PM@gene-NP complexes can effectively deliver gene therapy to the injured area and proposed that this delivery system should be considered as a potential method for treating deep burns.
104
Besides, mineralization is a controlled process with complex physicochemical interactions at various interfaces. It efficiently synthesizes inorganic and inorganic/organic materials for intricate structures at nano-, micro-, and macro scales, but this is challenging to achieve under laboratory conditions. To shed light on this, Zhan et al.
105
presented a dynamic control strategy for nano-scale mineralization processes and an all-in-one healing agent for wound healing (Figure 1). The major point was the reversible metal-polyphenol coordination, where polyphenol stabilized the mineralized precursor solution. When combined with metal-organic frameworks (MOFs), the released mineral ions caused fast mineralization on MOF surfaces, and the mineralized frameworks were noted to have plug-and-play properties. Their results further demonstrated that the mineralized frameworks could be loaded into a gelatin-chitosan hydrogel, exhibiting antibacterial and anti-inflammatory effects, angiogenesis, collagen deposition, and hair follicle regeneration. This strategy could also be applied to thin film technology for various applications.
105
A schematic representation of polyphenol-mediated BioM mineralization of sacrificial MOF NPs for wound healing.
105
Reproduced with permission. License number 5760021503246.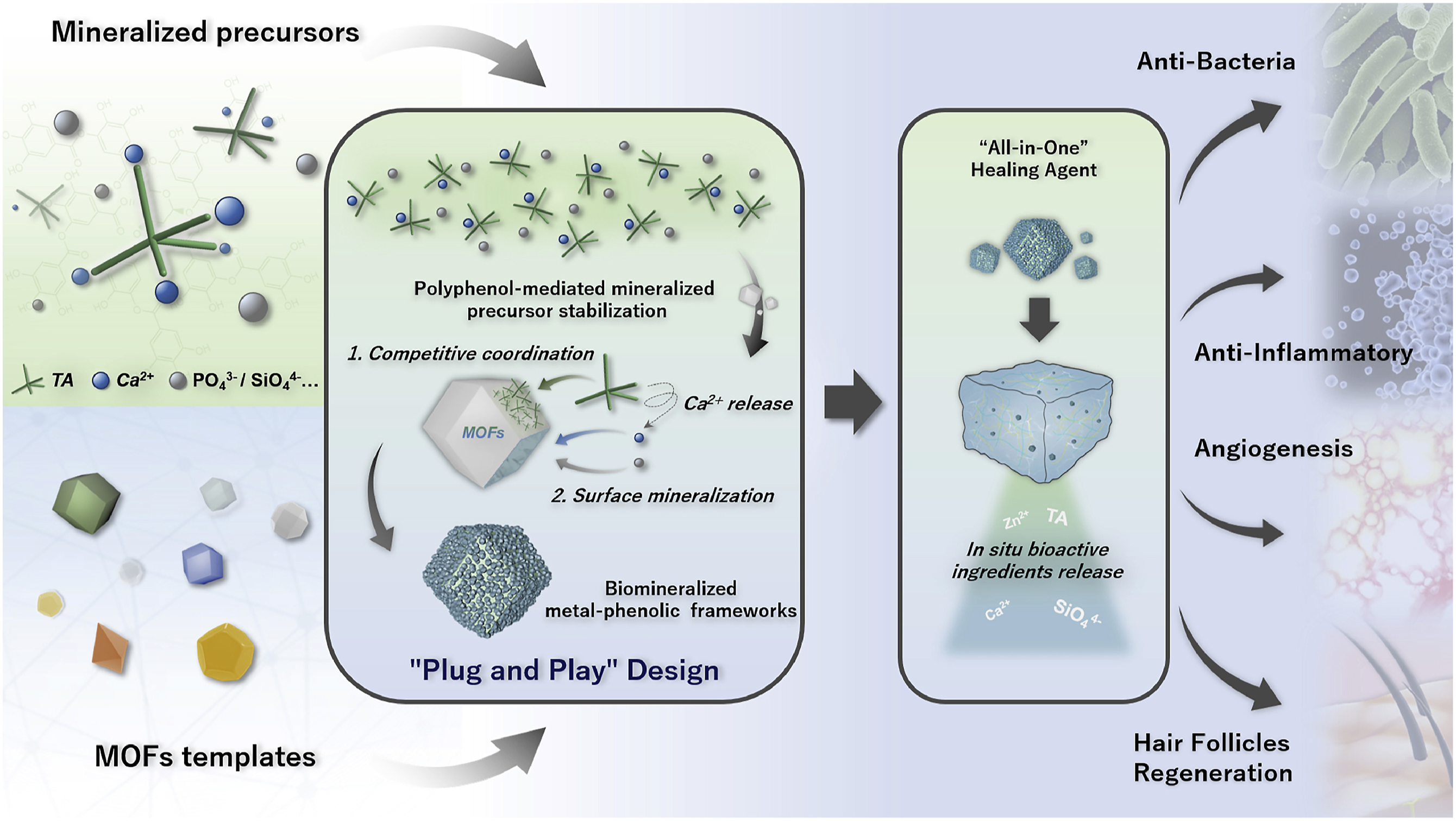
Infectious wound healing is a significant global healthcare concern due to its high prevalence, disability, and mortality rates. Recent investigations have revealed that Selenium NPs (SeNPs) are multifunctional drugs found to inherit all physiological functions of selenium but with less toxicity and more biological activity. Fang et al. 106 in essence, suggested a strategy to use SeNPs’ anti-inflammatory ability to stimulate wound healing by destroying bacterial membranes and allowing bacterial contents to outflow, achieving an antibacterial effect. Overall, because of their exceptional antibiotic-free effects and biomimetic immune evasion capabilities, SeNPs could be potentially useful in clinical settings for wound healing. 106 Moreover, electrical stimulation (ES) can be used to produce techniques that increase healing results in the wound bed microenvironment, which is critical for wound repair. 107 , According 108 to a study by Li et al. 14 sought to employ electrical stimulation (ES) to enhance the paracrine activity of rat adipose-derived stem cells (rASC). We discovered that ES, particularly 5 μA ES, encouraged rASCs to create more paracrine factors (5 μA-PFs) by analyzing the quantities of two major growth factors, VEGF and PDGF-AA, in the cell culture supernatant. Further research revealed that ES may influence the paracrine characteristics of rASCs by increasing the levels of TRPV2 and TRPV3, causing intracellular Ca2+ influx. To transfer the PFs to the wound and enhance the wound microenvironment, we created a heparinized PGA host-guest hydrogel (PGA-Hp hydrogel). In rats, PGA-Hp hydrogel with 5 μA-PFs significantly accelerated full-thickness wound healing. Our findings demonstrated the impacts of ES on the paracrine capabilities of rASCs, highlighting the potential use of heparinized PGA host-guest hydrogels loaded with PFs produced from electrically stimulated rASCs in wound healing. 14
Additionally, Lactobacilli, probiotic bacteria, have potential benefits in tissue regeneration and wound healing due to their biocompatibility and immunomodulatory effects, 109 but their formulation as living bacteria is challenging and their therapeutic use is limited in immunocompromised patients. Here, Kuhn et al. 110 suggest a new therapeutic approach to address these problems by creating a bacteriomimetic hydrogel based on membrane vesicles (MVs) generated by Lactobacilli. The group attached MVs from Lactobacillus plantarum and Lactobacillus casei to the surfaces of synthetic microparticles and implanted such bacteriomimetics in a pharmaceutically acceptable hydrogel matrix. The wound microenvironment changes while the wound heals, including pH adaptations and changes in oxygen delivery. The next performed proteome characterisation of the MVs extracted under various growth settings, identifying distinctive proteins associated with the biological action of the probiotics in each culture state. Furthermore, they highlight several distinct proteins expressed and sorted into the MVs for each culture condition. Using several in vitro models, they revealed that the enhanced cell migration and anti-inflammatory effects of bacteriomimetic microparticles were dependent on the secreting bacteria’s growth conditions. Finally, they showed that the bacteriomimetic hydrogel improved healing in an in vivo mouse full-thickness wound model. The findings provide a strong foundation for the future use of probiotic-derived vesicles in the treatment of inflammatory conditions and encourage the start of additional preclinical studies. 110 Also, diabetic wound infections, usually complicated by reduced cytokine responses, infections, poor vascularization, and delayed healing processes 111 raise the risk of amputation and the worldwide medical burden. 112 Although endothelial cell-derived nanovesicles promote angiogenesis, their effectiveness in treating infected diabetic wounds is restricted because of intricate niches since unhealed wounds are mostly caused by impaired endothelium. To synthesize biomimetic hybrid nanovesicles (HNVs), Jiang et al. 88 in this study, generated extrusion-derived endothelial nanovesicles (NVs) and hybridized them with rhamnolipid liposomes. HNVs then demonstrated superior penetration and antibacterial activity, were biocompatible, and were delivered with endothelium-targeted efficiency. Finally, the research developed a novel bioactive delivery system, HNV, with enhanced targeting, penetration, and antibacterial activity, which could be an effective strategy for treating infected diabetic wounds. 88
BioM NPs show significant potential for wound healing applications because of their unique ability to stimulate tissue regeneration, decrease inflammation, prevent infection, and expedite wound closure. Continued research and development efforts in this sector are likely to result in the translation of BioM NP-based therapeutics into clinical practice, improving wound care and patient outcomes.
BioM NPs for targeted drug delivery
BioM NPs are excellent carriers for delivering therapeutic substances such as medicines, genes, and proteins to specific areas in the body.
113
Their BioM characteristics allow them to avoid immune detection, increase circulation time, and improve targeting efficiency, resulting in optimal therapeutic results while avoiding off-target consequences. BioM techniques can be used to enhance biopharmaceutical properties. As a result, BioM reconstitutable pegylated amphiphilic lipid nanocarriers offer a high translational potential for systemic controlled drug administration; however, no such makeshift system for systemic aspirin delivery based on nanotechnology is now available. Systemic aspirin dosing and regulated distribution can greatly reduce blood clotting events that cause stroke, which has urgent implications for cardiovascular disease and COVID-19. According to Figure 2, Rajeev et al.
114
employed aspirin sustained release pegylated amphiphilic self-assembling NPs to construct reconstitutable aspirin injections using a solvent-based co-precipitation process with phase inversion methodology that leads to innovative “biomimetic liposomal NPs (BNNs).” DOE-led optimization is performed to provide a design of space for optimized particles. Reconstituted solid powder with a particle size of 144.8 ± 12.90 nm and a surface charge of −29.2 ± 2.24 mV. The entrapment efficiency was 49 ± 0.15%, and 96.99 ± 1.57% of the drug was released after 24 h, indicating a super case II transport-based mechanism. The formulation causes the least hemolysis while significantly suppressing platelet aggregation. The MTT experiment did not demonstrate any substantial cytotoxicity. This is a possible nanoparticle that might be used to manufacture aspirin injections, which are currently unavailable.
114
Illustrates preparation and formulation of CR- NanoMed using ANNs. (a) Scheme for preparing FP-ANNs. (b) FP-ANNs lyophilized using mannitol to create BANNs as powder and then reconstituted with PBS (pH 7.4) (c) SEM pictures of BANNs without mannitol and (d) with mannitol; (e) TEM images of BANNs.
114
Reproduced with permission. License number 5760051486857.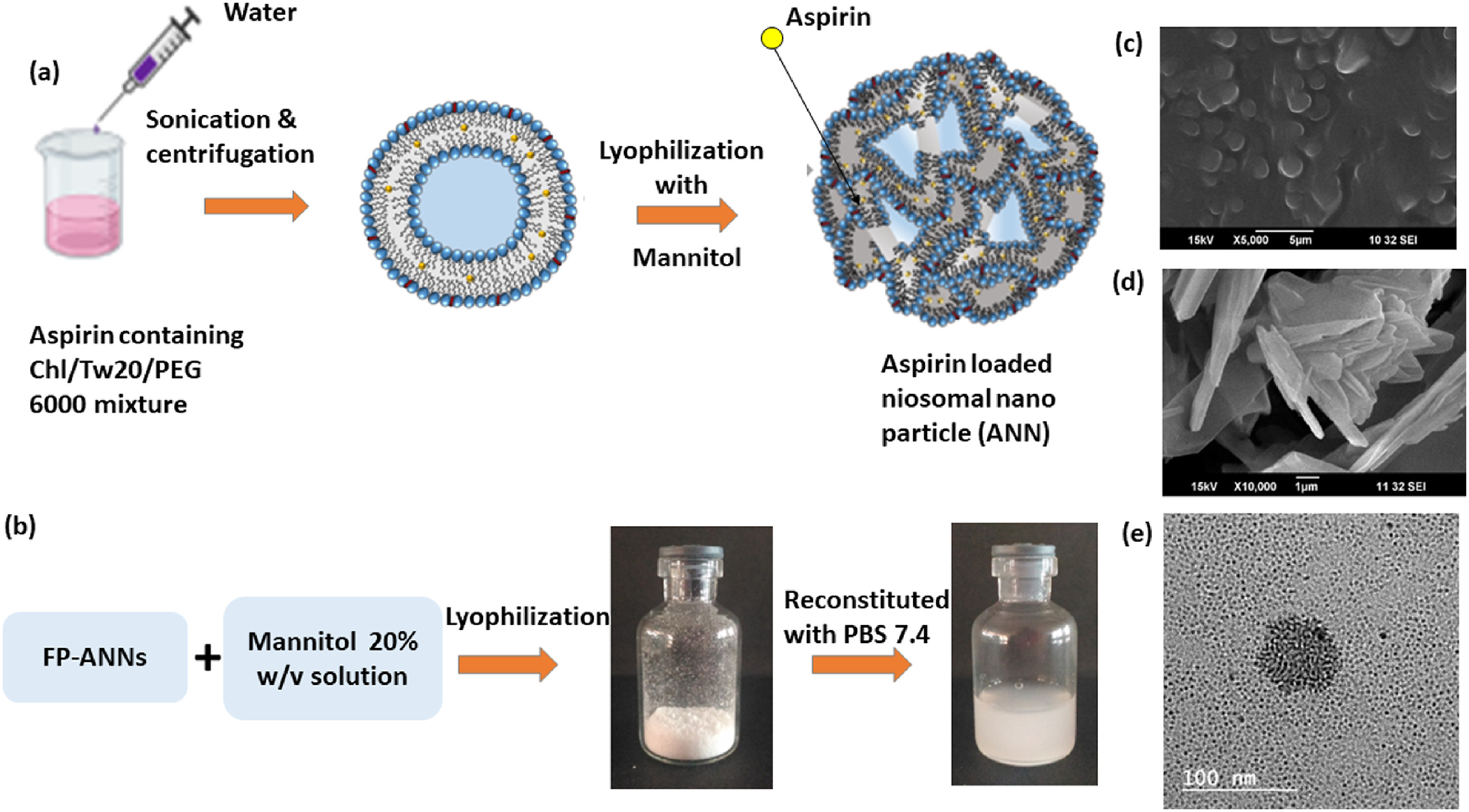
Stainless steel (SSL) miniscrew implants are now being employed as temporary anchoring devices in orthodontic clinics. Despite their great physical qualities, their biocompatibility is quite low. Previously, Li et al. 115 ’s team created a two-phase biomimetic calcium phosphate (BioCaP) coating that dramatically improves the biocompatibility of medical equipment. The goal of this work was to increase SSL’s biocompatibility by covering the surface with BioCaP. In their investigation, they employed titanium (Ti) discs and SSL discs. To create an amorphous layer, the Ti discs were submerged in a BioM-modified Tyrode solution (BMT) for 24 h. SSL discs were submerged in the same solution for 0, 12, 24, 36, and 48 h. To produce a crystalline layer, the discs were submerged in a supersaturated calcium phosphate solution (CPS) for 48 h. The surface characteristics of BioCaP coatings were analyzed. As a model protein, bovine serum albumin (BSA) was also introduced into the crystalline layer during biomimetic mineralization. The morphology, chemical content, and drug-loading capability of the BioCaP coating on smooth SSL were all validated. This coating enhanced the roughness and wettability of the SSL surface. In vitro, extending the BMT coating duration improved cell seeding efficiency, spreading area, and proliferation on the BioCaP coating. These in vitro results demonstrate that the BioCaP coating may enhance the surface qualities of smooth medical-grade SSL and act as a carrier system for bioactive substances. 73 Also, owing to the varied hybrid frameworks and customizable pore diameters, as well as their biodegradability and intelligent performance, hollow mesoporous organosilica NPs (HMOSNPs) have presented new prospects to increase the therapeutic efficacy of encapsulated pharmaceuticals. In the current work, Zahiri et al. 116 created HMOSNPs capped with red blood cell (RBC) membrane to produce BioM RBC-coated HMOS platform with regulated glutathione (GSH)-responsive doxorubicin (DOX)-releasing performance. A DNA aptamer against the MUC-1 receptor was employed to create the targeted system (Apt-RBC-HMOS@DOX) for the controlled delivery of DOX to colorectal carcinoma cells. Experimental results showed that the MUC1 aptamer could significantly improve drug absorption of the developed biomimetic NPs into HT29 and C26 cells. Furthermore, the targeted Apt-RBC-HMOS@DOX system significantly improved tumour-targeting capabilities in C26 tumour-bearing mice as compared to non-targeted RBC-HMOS@DOX and free drugs. The GSH-responsive Apt-RBC-HMOS@DOX system also decreased DOX-induced toxicity in terms of tumour growth suppression and survival rate, indicating its potential application in cancer treatment. 116
BioM NPs can penetrate target cells and deliver therapeutic payloads via cellular internalization mechanisms such as endocytosis and receptor-mediated endocytosis. 23 BioM NPs’ interaction with cellular receptors and uptake processes may be modified by altering their surface features 117 and composition, hence improving cellular internalization and intracellular drug delivery.23,117 BioM NPs can include a variety of therapeutic substances, including small molecules, proteins, nucleic acids, 118 and NPs, within their core or lipid bilayer.118,119 Encapsulating therapeutic cargo within BioM NPs protects from enzymatic destruction, extended circulation duration, and controlled release kinetics, 120 resulting in increased therapeutic efficacy and lower systemic toxicity. 13 BioM NPs can be created with stealth coatings or surface changes to avoid immune surveillance and clearance processes such as opsonization and phagocytosis. 59 BioM NPs can improve targeted drug delivery efficiency by reducing interactions with immune cells and serum proteins.46,121 BioM NPs provide a flexible and adjustable platform for targeted drug administration, with precise control over drug release, biodistribution, and therapeutic effectiveness. Using BioM NPs’ unique features, personalized techniques for targeted drug administration may be devised to treat specific disease indications and enhance patient outcomes.
BioM NPs for Bone Formation And/or Regeneration
BioM NPs have sparked significant interest in bone formation and regeneration applications due to their ability to replicate the natural extracellular matrix (ECM) and enhance interactions with bone cells. Bone tissue engineering is an ideal method for repairing bone defects.
36
Recent research122,123 has looked into the possibilities of functional modification of scaffolds in bone tissue engineering.
124
Zhou et al.
125
created an SF-CS-nHA (SCN) BioM bone scaffold and functionalized it by adding a polydopamine (PDA) covering containing marrow mesenchymal stem cell (BMSC) exosomes (exo) using a vacuum freeze-drying/chemical crosslinking process (Figure 3). The effect of the functional composite scaffold (SCN/PDA-Exo) on BMSC proliferation and osteogenic differentiation was studied. Furthermore, the SCN/PDA-Exo scaffolds were implanted in rats to assess their impact on bone regeneration. In comparison to the SCN scaffold, the SCN/PDA-Exo-functionalized composite scaffold displayed higher average surface roughness and hydrophilicity. In vitro, BMSCs absorbed Exos mounted on SCN/PDA-Exo scaffolds. The SCN/PDA-Exo group showed considerably improved BMSC shape, proliferation capacity, and osteogenic differentiation impact compared to the other control groups. The SCN/PDA-Exo functional composite scaffold significantly improved bone defect healing and new bone production compared to other control groups. The study discovered that the SCN/PDA-Exo-functionalized composite scaffold increased BMSC proliferation and osteogenic differentiation in vitro while improving bone regeneration efficiency in vivo. Combining Exos with biomimetic bone scaffolds via functional PDA coatings may thus be a useful technique for functionally altering biological scaffolds.
125
Illustration of PDA-coated BioM bone scaffolds functional composite scaffold (SCN/PDA-Exo) with exos stimulate osteogenic differentiation of marrow mesenchymal stem cell (BMSC) exosomes and bone repair.
125
Reproduced with permission. License number 5760110932344.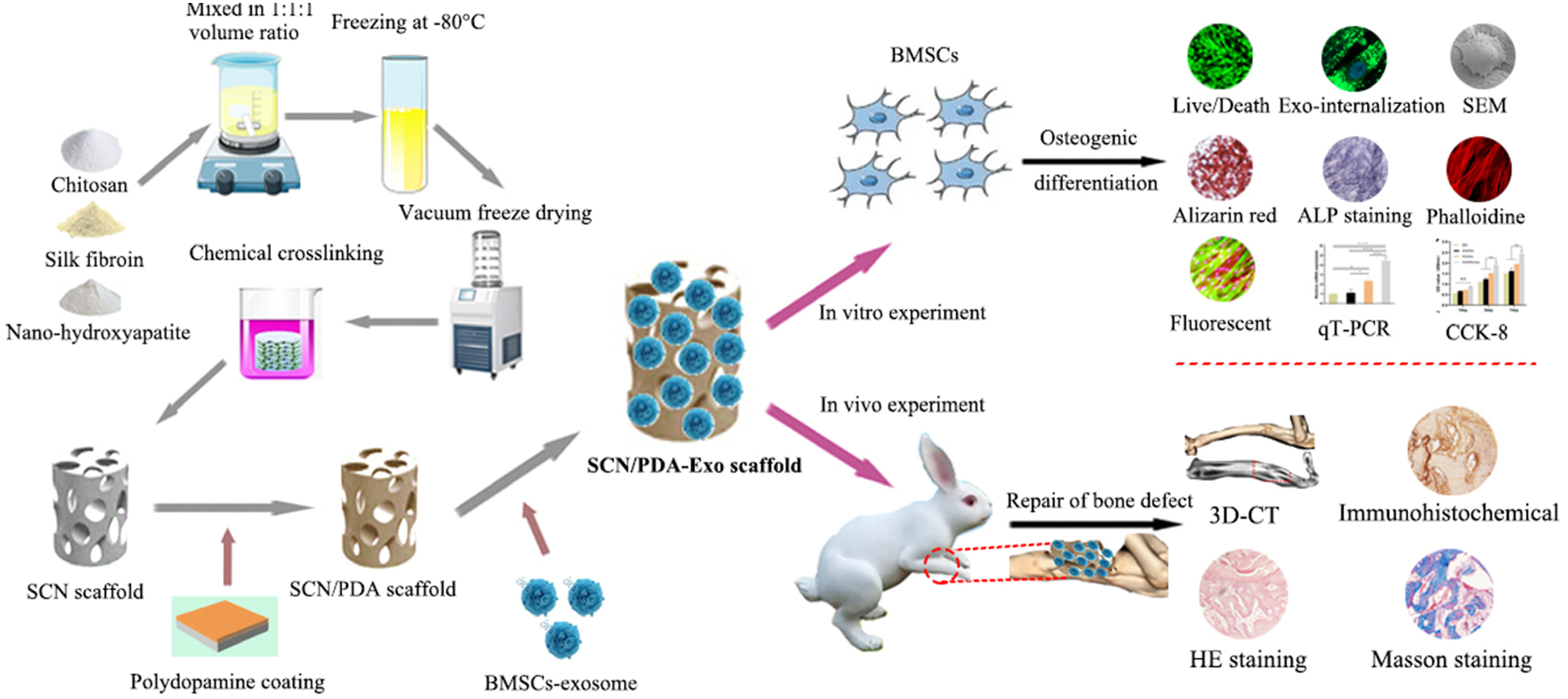
Coating techniques can improve the biocompatibility and osteoconductivity of porous scaffolds by forming micro- and nanostructures on their surfaces. A BioM method was employed by Gu et al. 126 to create a bone morphogenetic protein-2 (BMP-2) integrated calcium phosphate (CaP) coating on the surface of porous Ti scaffolds. In vitro characterizations revealed that the scaffolds have mechanical characteristics similar to those of human cortical bone. The BioM coating features a nanoscaled surface structure that releases BMP-2 consistently and efficiently. Bicortical bone defects were created in the bilateral mandibles of beagle dogs, and the coated and uncoated scaffolds were fixed for 8 weeks. According to micro-CT and histological examination results, the BioM covering dramatically increased bone growth in the Ti scaffold. The coated scaffold group exhibited considerably greater bone volume fraction, bone area fraction, and bone-to-implant contact rates than the uncoated scaffold group. Furthermore, haematological indices and histopathological data from visceral organs verified the scaffold and coating’s biocompatibility. As a result, the current work shows that BMP-2 integrated biomimetic CaP coating may considerably enhance bone formation in porous titanium scaffolds and aid in the healing process of a bicortical mandibular lesion in beagle dogs. 126
Besides, hybrid organic-inorganic nanofibrous membranes show promise for guided bone regeneration (GBR) in the treatment of clinical bone abnormalities because they imitate the in vivo bionic milieu and promote osteogenic differentiation. Li et al. 127 created a degradable and osteogenic membrane for GBR by combining the natural biomacromolecules silk fibroin (SF) and gelatine with the bioactive nano-hydroxyapatite (nHA), anhydride-modified gelatine-nano hydroxyapatite (GelMA-nHA) composites in situ and introduced into silk fibroin to prepare nanofibrous membranes with different ratios using electrospinning and photocrosslinking. The nanofibrous membranes, particularly those with a mass ratio of 7:2:1, were found to exhibit satisfactory elongation at break of up to 110 %, maintain the nanofibrous structure for up to 28 days, and rapidly form bone-like apatite within 3 days, providing advantages for guided bone regeneration. In vitro cell studies revealed that the SF/GelMA/nHA membranes were highly biocompatible and promoted the osteogenic development of hBMSCs. In vivo, research showed that hybrid composite membranes can promote bone healing in critical-sized calvarial lesions in rats. As a result, the unique hybrid nanofibrous membrane is presented as an alternate choice for generating a bionic milieu that promotes bone regeneration, implying that it can cure bone injuries. 127
Furthermore, in bone tissue engineering investigations, creating vascular networks is critical for improving bone transplant healing procedures. 124 While most synthetic bones rely on host vascular invasion upon implantation, this process is frequently delayed. As a result, investigating highly vascularized techniques may help to improve the effectiveness of tissue restoration. Cui et al. 128 used dual-model composite printing to create bone scaffolds with biomimetic vascular networks, which they hoped would promote angiogenic integration and improve osteogenic healing. First, the polycaprolactone composite nano-tricalcium phosphate scaffold had acceptable tensile characteristics, porosity, and sufficient space for the bionic vascular network. Meanwhile, researchers employed concentrated growth-factorized gelatin methacryloyl hydrogel-loaded endothelial cells to create a 3D interoperable cavity structure with multi-level vascular connection into a network that spread across the scaffold. The gradual release of biological signals from hydrogel improved the biological activities of scaffold acetogenesis and osteogenesis. In vivo and in vitro investigations revealed that the composite bionic scaffold increased angiogenesis, facilitated host vascular integration, and synergized nano-tricalcium phosphate osteogenesis induction, hence enabling bone defect repair and regeneration. This vascularized tissue technique proved adaptable and offered a solid foundation and favourable insight for creating large-scale vascularized bone transplants. 128
BioM NPs may be designed to have mechanical characteristics similar to those of real bone tissue, 126 resulting in structural support and stability at the defect location. 36 These NPs, which resemble the mechanical characteristics of bone, can sustain mechanical stresses and allow integration with surrounding tissues, enhancing the long-term stability and usefulness of regenerated bone. BioM NPs can induce angiogenesis, 127 or the development of new blood vessels, which improves vascularization and nutrition delivery to rebuild bone tissue. 28 Surface modification of BioM NPs with angiogenic factors or peptides can stimulate endothelial cell proliferation and capillary development, allowing for the construction of a functioning vascular network within the newly produced bone. Ultimately, BioM NPs provide a diverse platform for boosting bone formation and regeneration by replicating the natural ECM and delivering personalized signals to encourage cellular activity and tissue development. Continued research and development in this subject hold promise for the creation of sophisticated biomaterials and treatments for bone repair and regeneration in a variety of clinical settings, including orthopaedics, dentistry, and tissue engineering.
BioM NPs for cancer treatment
BioM NPs show significant potential for cancer therapy because of their capacity to imitate natural biological structures and processes,
129
allowing for targeted medication administration, increased therapeutic efficacy, and fewer side effects.
92
BioM NPs can alter the tumour immune milieu and boost anticancer immune responses by delivering immunomodulatory drugs,
129
tumour antigens, or immune checkpoint inhibitors directly to the tumour site.
130
BioM NPs can improve cancer immunotherapy effectiveness and produce long-lasting antitumor immune responses by increasing immune cell infiltration, activation, and cytotoxicity inside the tumour microenvironment.
131
Furthermore, colorectal cancer (CRC) regimens have therapeutic limitations because of a lack of understanding of tumor-supportive microenvironments. To combine the therapeutic effects on tumour cell growth and immunosuppressive tumour microenvironments (TME), Peng et al.
132
proposed the artesunate (AS) and chloroquine (CQ) combination and created a poly (d,l-lactide-co-glycolide) (PLGA)-based BioM NPs for CRC treatment (Figure 4). Hydroxymethyl phenylboronic acid linked PLGA (HPA) is produced to provide a BioM NPs core that is sensitive to reactive oxygen species (ROS). To get a BioM NPs-HPA/AS/CQ@Man-EM, a unique surface modification approach produces a mannose-modified erythrocyte membrane (Man-EM) that is cloaked on the AS and CQ-loaded HPA core. It shows great potential in suppressing CRC tumour cell growth and correcting TAM phenotypes by targeting both tumour cells and M2-like tumour-associated macrophages (TAMs). In an orthotopic CRC animal model, BioM NPs demonstrated better accumulation at tumour tissues and significantly decreased tumour development by both tumour cell growth suppression and TAM repolarization. Notably, uneven distribution to tumour cells and TAMs is critical for achieving outstanding anti-tumor effects. This study presented an effective biomimetic nanocarrier for treating CRC.
132
Dual-targeting of artesunate and chloroquine to tumour cells and tumour-associated macrophages by BioM PLGA NPs for colorectal cancer treatment.
132
Reproduced with permission. License number 5760151235795.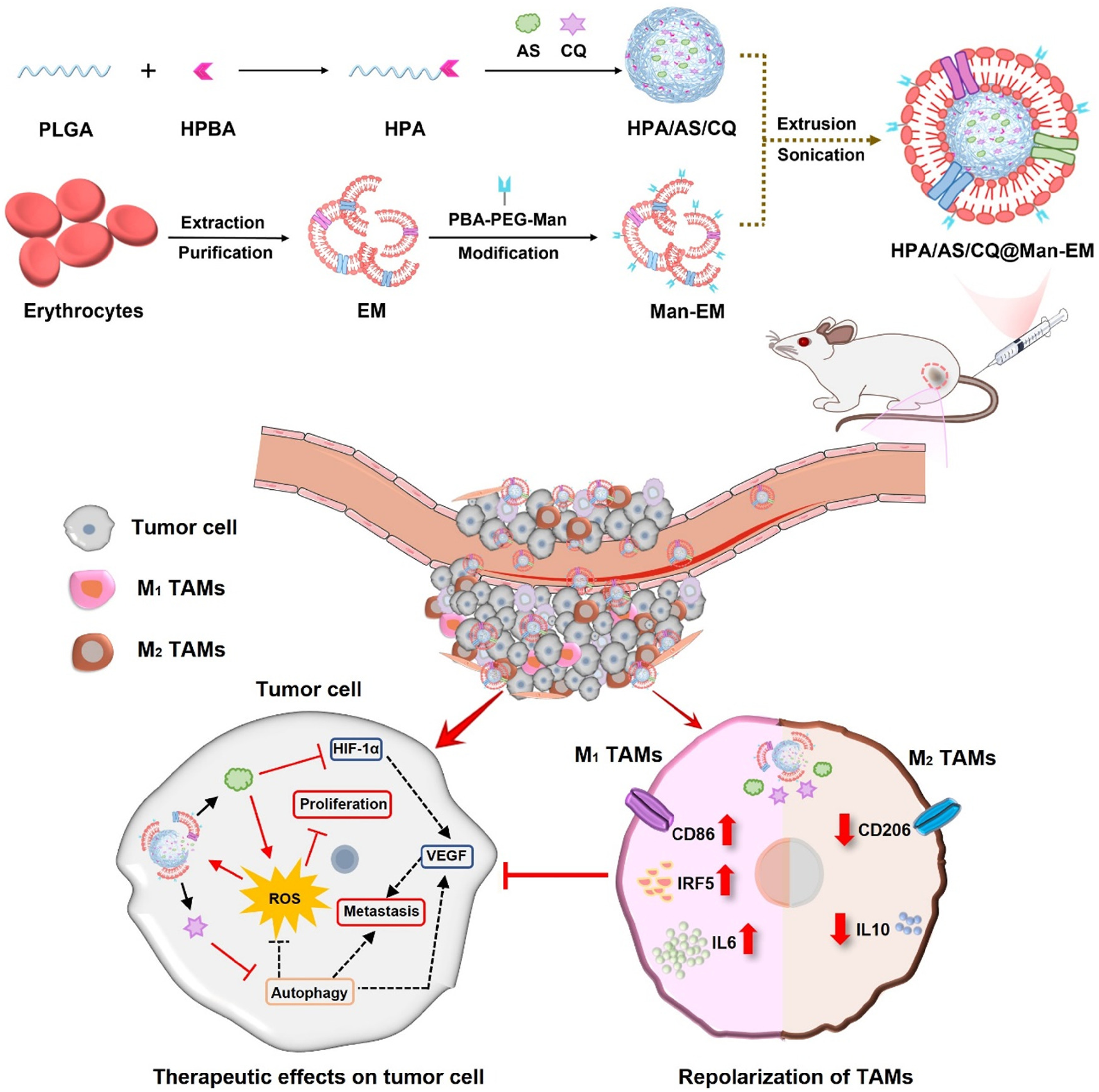
The most successful treatment for pancreatic cancer (PC) is the mFOLFIRINOX plan. 133 However, this regimen has very significant toxic effects and is often confined to patients with a sound physique, which is weak in drug selectivity and non-specific, resulting in high toxicity, and severely limiting clinical applicability. In the future, enhanced pancreatic cancer therapies will require a safer and more effective technique that operates flawlessly and synchronously. Wang et al. 134 used a BioM NP-based platform to optimize the mFOLFIRINOX regimen by creating PCCM-cloaked NPs, CNP@folfirinox, by covering the surface of polymeric cores loaded with mFOLFIRINOX with the pancreatic cancer cell membrane (PCCM). These NPs inherit the antigenic exterior structure and related functions of PC cells. The resultant CNP@folfirinox has a core-shell nanostructure composed of a polymeric core and a cancer cell membrane shell of appropriate size and surface charge. CNP@folfirinox, in particular, benefits from inheriting the natural membrane protein expression profile, resulting in homologous homing to tumour tissue and deep penetration. It also has fewer adverse effects than intensive mFOLFIRINOX i. v. dosing, with no significant damage in organs beyond the tumour in vivo. Meanwhile, CNP@folfirinox has the “Don’t eat me” privilege, making it difficult for the immune system to identify and reduce reticuloendothelial system absorption, resulting in longer blood circulation and improved drug delivery efficiency following intravenous injection. As a result, our study reshapes a viable paradigm for a safer and more successful mFOLFIRINOX method for future clinical pancreatic cancer treatment. 134
Zhang et al. 20 showed that patients with triple-negative breast cancer (TNBC) exhibited greater shear-wave elasticity (SWE) stiffness and α-SMA expression in their tissues compared to non-TNBC patients. Furthermore, SWE stiffness has a significant association with neoadjuvant response efficiency. To further explain these phenomena, TNBC cell membrane-modified polylactide acid-glycolic acid (PLGA) NPs were created to precisely transport artesunate to modulate SWE stiffness by suppressing the CAF functional state. As tested in MDA-MB-231 and E0771 orthotopic tumour models, 231M-ARS@PLGA NPs (231M-AP NPs) inhibited CAF functional status, reduced SWE stiffness, and attenuated tumour hypoxia as a tumour soil loosening agent, amplifying the antitumor effects of paclitaxel and PD1 inhibitor. Single-cell sequencing revealed that extracellular matrix and wound healing CAFs create extracellular matrix, which can impact tumour SWE stiffness and medication anticancer activity. Furthermore, BioM NPs suppressed CAF activity, which might reduce tumour hypoxia by increasing the fraction of inflammatory blood vessels and oxygen transport capability. As a result, our findings are critical for understanding the role of CAFs in influencing SWE stiffness and drug anticancer effects, which may be extended to possible clinical theranostic prediction in neoadjuvant treatment effectiveness using non-invasive SWE imaging analysis. 20
BioM NPs can lessen the systemic toxicity and off-target effects associated with traditional cancer therapy by allowing therapeutic chemicals to be delivered to tumour tissues. 135 BioM NPs assist in retaining normal organ function and enhance the safety profile of cancer therapies by reducing exposure of healthy tissues to cytotoxic medications, 136 hence increasing patient tolerance and compliance.130,131,136 In summary, BioM NPs provide a diverse and effective platform for cancer therapy by leveraging their distinct features to precisely transport therapeutic molecules to tumour cells, overcome biological obstacles, and improve therapeutic results while reducing adverse effects. Continued research and development in this sector show great potential for enhancing customized cancer therapy and improving patient outcomes.
BioM NPs for theranostics
BioM NPs integrate therapeutic and diagnostic characteristics in a single platform, allowing for disease imaging and therapy at the same time. 137 This method, known as theranostics, provides for real-time monitoring of treatment response and disease progression while administering targeted therapy, enabling customized medical approaches. 5 BioM NPs may be created with imaging probes that allow the NPs to be seen using a variety of imaging modalities. 138 For example, combining fluorescent dyes with MBioM NPs or radionuclides enables dual-modal imaging with fluorescence, 22 magnetic resonance imaging (MRI), 139 or positron emission tomography (PET), providing complementary information for improved tumour visualization and treatment response monitoring. 140 Imaging modalities such as MRI, PET, and optical imaging may be utilized to monitor the in vivo accumulation and pharmacokinetics of BioM NPs, guiding treatment parameter modification and guaranteeing optimal therapeutic payload distribution to target areas. 139 Because of monkeypox’s (Mpox) fast spread and a lack of effective treatments and preventative techniques, finding new ways to treat and disrupt transmission remains an essential issue. Wang et al. 141 presented a BioM nano-drug-based targeted photothermal and photodynamic dual-modality therapy method for Mpox treatment. Coating vaccinia virus (Mpox replacement virus)-activated macrophage membranes onto polymeric NPs loaded with a versatile photosensitizer with aggregation-induced emission features, the nano-macrophages (PN-AIE MØ) with virus-targeting receptors enable precise binding within infected pustules. PN-AIE MØ exhibits near-infrared-II fluorescence, photothermal, and oxygen-independent type I photodynamic capabilities after adding the photosensitizer agent. In vivo investigations reveal that when PN-AIE MØ is injected intravenously, it stays in viral lesions for lengthy durations, allowing imaging and efficient virus eradication under 808-nm laser therapy. More critically, this technique halts viral transmission and avoids future epidemics. This NM-based dual-mode therapy provides a road forward in Mpox care, which might inform future therapeutic methods. 141
Furthermore, cancer is a primary cause of death globally, prompting the development of novel treatment techniques. NP-based drug delivery systems have sparked widespread attention because of their multifunctionality, which has the potential to increase cancer therapy effectiveness and patient tolerance. Membrane-coated drug delivery systems have enormous promise for improving the therapeutic efficacy of nanoparticle-based anticancer therapeutics. The research described the production of multifunctional iron-functionalized mesoporous polydopamine NPs (MPDAFe NPs).
142
These nanoformulations show great promise for combining effective drug administration and magnetic resonance imaging (MRI) and highlight the benefits of BioM coating with tumour cell-derived membranes (Figure 5). This coating improves the NPs’ targeting capabilities and prolongs their circulation. Furthermore, rigorous biosafety assessments demonstrate low harm to normal cells, but the combination of chemo- and phototherapy showed considerable cytotoxicity to cancer cells. Furthermore, the photothermal impact evaluation emphasizes the increased cytotoxicity produced by laser irradiation, demonstrating the NMs' synergistic effects with photothermal treatment. Importantly, the chemotherapeutic effect evaluation shows that doxorubicin-loaded MPDAFe@Mem NPs (cancer cell membrane-coated MPDAFe NPs) are more effective than free doxorubicin at suppressing cancer cell viability and proliferation. This study thoroughly explores theranostic, membrane-coated drug delivery methods, emphasizing their potential to improve the efficacy of cancer treatments. The multifunctionality of the iron-functionalized polydopamine NPs enables effective drug transport and imaging, while the biomimetic coating improves their biocompatibility and targeting ability. These discoveries provide important insights into the development of enhanced nanomedicine for better cancer treatments.
142
Scheme of BioM theranostic NPs for efficient anticancer treatment and MRI imaging.
142
Reproduced with permission under the terms of the Creative Commons CC-BY license.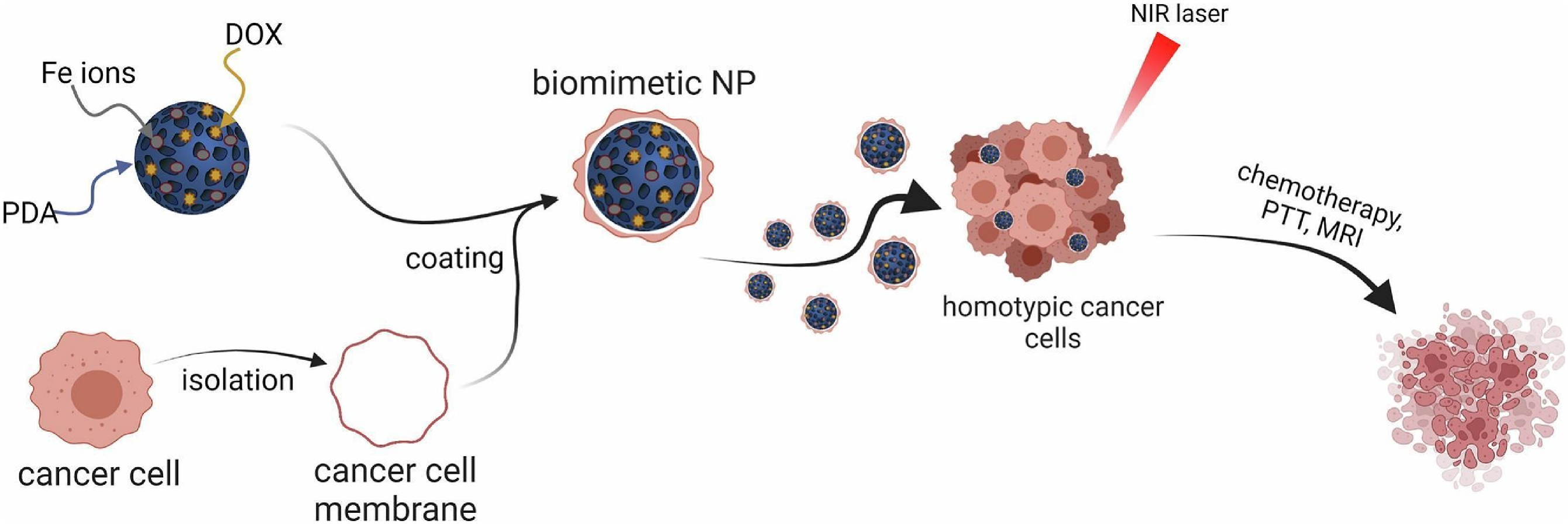
NP formulations that combine optical imaging contrast agents and medicines have been a key component of preclinical theranostic applications. However, NP-based theranostics clinical translation presents obstacles in terms of repeatability, brightness, photostability, biocompatibility, and selective tumour targeting and penetration. Prasad et al. 143 combined multimodal imaging and treatments in cancer cell-derived nanovesicles, resulting in BioM brilliant optotheranostics for cancer metastasis detection. Upon NIR light irradiation, the tailored optotheranostics enable deep vision and exact localization of metastatic lung, liver, and solid breast malignancies, as well as solid tumour ablation. Metastatic cell-derived nanovesicles are designed to include emissive organic dye, gold NPs, the anticancer medication doxorubicin, and photothermally active organic indocyanine green dye. BioM brilliant optotheranostic NPs administered systemically exhibit escape from mononuclear phagocytic clearance, fast tumour accumulation and retention, real-time monitoring of metastatic lung, liver, and solid breast tumours, and threefold image-guided solid tumour decrease. These findings are backed by improvements in X-ray, fluorescence, and photoacoustic signals, as well as tumour reductions when compared to single treatments such as chemotherapy, photodynamic therapy, and photothermal therapy (PTT). The suggested revolutionary platform offers new pathways for improving cancer diagnostic and treatment results by monitoring cancer spread, enabling accurate cancer imaging, and delivering synergistic therapeutic drugs to the solid tumour site. 143 Besides, the logical design of theranostic systems is crucial for resolving complex cancer-related concerns. Liang et al. 144 created the multifunctional BioM superparticle DOX-QDs-Lip@M to achieve this goal, which can transport drugs to tumours while also monitoring their therapeutic effects synergistically. Initially, the anticancer medication doxorubicin hydrochloride (DOX) and imaging agent quaternary quantum dots (QDs) were loaded into the liposome’s hydrophilic core area and hydrophobic chamber by self-assembly, respectively. The integrated nanostructure may dramatically boost the fluorescence intensity of the signal unit, thus improving diagnostic sensitivity. Subsequently, the BioM DOX-QDs-Lip@M was created by fusing and coating the isolated macrophage membranes on the surface of a liposome, extending the circulation of the whole blood and successfully targeting tumour locations. Furthermore, the naturally generated biofilm can help to maintain the artificial liposome structure, preventing the loaded contents from leaking out. These combined features enable the biomimetic DOX-QDs-Lip@M to enhance tumour imaging and anti-metastasis therapy in live systems. 144
BioM NP-based theranostic drugs allow for customized medical methods by giving real-time data on tumour features, drug accumulation, and therapy response in specific patients. BioM NP-based theranostics, which adapt treatment regimens based on patient-specific imaging and biomarker data, have the potential to enhance therapeutic results, reduce side effects, and increase patient survival rates. Ultimately, BioM NP-based theranostics provides a flexible and comprehensive approach to cancer diagnosis, treatment, and monitoring, with potential applications in precision medicine, image-guided therapy, and individualized cancer care. Continued research and development in this sector have the potential to further the clinical translation of BioM NP-based theranostic medicines and improve patient outcomes in cancer care.
Novel application of biomimetic nanoparticles (BioM NPs) in NanoMed
One unique use of BioM NPs is their potential for targeted medication delivery to the central nervous system (CNS), 145 especially for the treatment of neurological diseases.37,145,146 BioM NPs can be designed to replicate the surface features and interactions of biological entities that naturally pass through the Blood–Brain Barrier (BBB), 147 such as viruses or cell-penetrating peptides. By making use of these natural mechanisms, BioM NPs may efficiently cross the BBB and transport therapeutic drugs directly to the brain parenchyma, bypassing the restrictive barrier and increasing drug accumulation at the target location.93,147
BioM NPs can be loaded with targeted ligands that preferentially bind to receptors or antigens found on the surfaces of neurons or glial cells. 148 BioM NPs can transport therapeutic compounds to damaged areas of the brain with great accuracy, reducing off-target effects and boosting therapeutic efficacy for neurological illnesses such as Alzheimer’s disease, Parkinson’s disease, and brain cancers.148,149 BioM NPs can enhance intracellular drug delivery to neurons and glial cells in the CNS, 150 allowing for specific manipulation of cellular pathways involved in neurodegenerative illnesses or neurological disorders. Encapsulating therapeutic drugs into BioM NPs and stimulating their internalization via endocytosis or receptor-mediated uptake allows for precise regulation of drug release and pharmacological effects in the intracellular milieu.150,151
BioM NPs can be engineered to have longer circulation durations in the bloodstream, allowing for continuous release and exposure of therapeutic compounds to the CNS. 152 BioM NPs can improve medication bioavailability and distribution to the brain by reducing reticuloendothelial system (RES) clearance and preventing fast removal from circulation, resulting in better treatment results for CNS illnesses.152,153 BioM NPs can combine imaging and therapeutic functions into a single platform, 4 allowing for real-time monitoring of drug distribution, pharmacokinetics, and treatment response in the CNS. 154 Simultaneous imaging and therapy can be performed by adding imaging probes such as fluorescent dyes, MRI contrast agents, or radionuclides into BioM NPs, allowing for more tailored treatment techniques and improved patient outcomes.4,154
As a whole, the use of BioM NPs for targeted medication delivery to the CNS is a promising strategy for addressing the hurdles of treating neurological illnesses. Novel therapeutics to address unmet medical needs in the brain can be developed by using BioM NPs’ unique capabilities, such as their capacity to cross the BBB, target specific cell types, and transport therapeutic chemicals intracellularly. Continued research and innovation in this field have the potential to transform the treatment of CNS illnesses and improve the lives of millions of people worldwide.
Nanomaterials induced endothelial leakiness (NanoEL)
Nanomaterials-induced Endothelial leakiness (NanoEL) is a phenomenon in which NPs interact with endothelial cells, resulting in brief and reversible holes or gaps between the cells.
155
Endothelial cells line the inside of blood arteries, and their major purpose is to act as a selective barrier between blood and tissues, regulating the movement of chemicals and cells.
156
The NanoEL effect occurs when NPs come into touch with the endothelial cell membrane initiating a signaling cascade, resulting in a temporary and reversible increase in the permeability of endothelial monolayers.155–157 Owing to this knowledge, Setyawati et al.
158
demonstrated that gold (Au) NPs ranging in size from 10 to 30 nm are effective NanoEL inducers. Because not all endothelial cells have the same permeability, they discovered that human breast and skin endothelial cells are sensitive to Au-induced NanoEL, but human umbilical vein endothelial cells are insensitive, reflecting their intrinsic endothelial permeability.
158
Besides, delivering cancer therapies to tumours needs their escape from the blood arteries. Tumor vasculatures are not necessarily very leaky. Setyawati et al.
159
again generated therapeutically competent leakage of medicines from tumour vasculature using Au NPs capable of triggering NanoEL (Figure 6). These NanoEL Au NPs induced the loss of endothelial adherens junctions without causing any detectable damage to endothelial cells. Microscopically, using real-time live animal intravital imaging, we show that NanoEL particles caused leakiness in tumour artery walls and enhanced penetration into the tumour’s interstitial region. In both primary tumour and subsequent micrometastase animal models, they demonstrated that pretreatment of tumour vasculature with NanoEL particles before therapeutic delivery can entirely regress the malignancy.
159
Au NPs induced endothelial cell leakiness (Au NanoEL) in size and surface roughness dependent manner: (a) Representative transmission electron microscope (TEM) images depict different surface roughness of Au NPs with primary size 30 nm (Au30); (b) Immunofluorescence images show the formation of intercellular gaps (white arrowheads) on the monolayer endothelial cell barrier treated with Au NPs (1 × 109 particle/mL, 1 h); (c) Number of gaps (n = 12; 4 IF images/group pooled from three independent biological replicates/group) and (d) gap size (n = 90; 30 gaps/group pooled from three independent biological replicates/group) observed within a certain area; (c, d) Data presented in are mean ± SEM. One-way ANOVA, Tukey HSD post-hoc test, *significant against control, p < .05; (e) Increase in measured NanoEL index was detected with the increase of Au NPs surface roughness (1 × 109 particles/mL, 1 h).
159
Reproduced with permission under a Creative Commons Attribution 4.0 International License.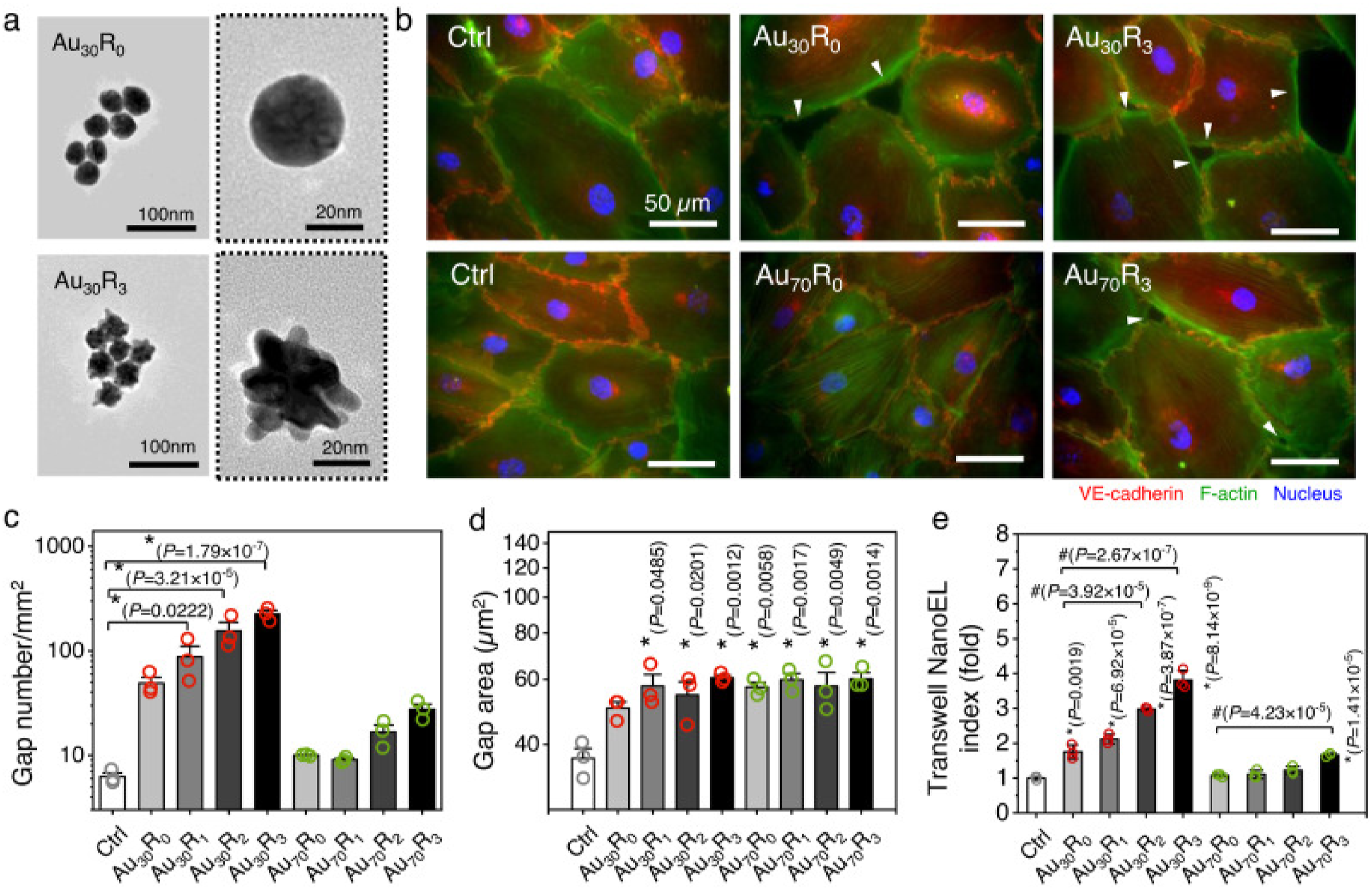
Furthermore, NanoEL can boost the capacity of NPs to reach the tumour environment 157 ; however, it can also have detrimental effects such as tumour spreading. To address these challenges, it is critical to develop an NP design strategy that capitalizes on the NanoEL effect while avoiding undesired side effects throughout the medication delivery process. Huang et al. 157 developed the PLGA-ICG-PEI-Ang1@M NP, which consists of a core of poly (lactic-co-glycolic acid) (PLGA) and an inner shell composed of positively charged polyethyleneimine (PEI) and the anti-permeability growth factor Angiopoietin 1 (Ang1), while the outer shell is disguised with a Jurkat cell membrane. During medication delivery, the NPs demonstrate their potential to specifically target and penetrate endothelial cell layers. Once the NPs have penetrated the endothelium layer, the proton sponge effect caused by PEI in the acidic environment around the tumour location might tear the cell membrane on the NPs’ surface. This rupture, in turn, allows the positively charged Ang1 to be released due to electrostatic repulsion from PEI, allowing the ruptured endothelium layer to be repaired. As a result, the proposed NPs may enter endothelial layers, enhance cell layer healing, inhibit tumour spread, and support effective cancer therapy. 157 Furthermore, a series of ultralow protein corona NP was reported by Huang et al. can penetrate the endothelial cell junction without obviously interacting with the vascular endothelial cadherin and phosphorylating the tyrosine 658 and tyrosine 731 residues on vascular endothelial cadherin, thus preventing the vascular endothelial cadherin from being activated by Src kinase, and this avoids inducing the NanoEL effect and cancer cell migration, regardless of particle material, density, and surface These findings gave a fresh approach for designing innovative NPs that have no side effects and increase their cancer-killing efficacy.
The NanoEL effect offers a promising way for NPs to escape the vasculature and boost therapeutic drug delivery to target tissues. Researchers can increase the efficacy of NP-based therapeutics by adjusting NP features like as size and surface qualities.
Cell membrane-camouflaged nanoparticles (CM NPs)
BioM NPs can be coated with membrane fragments from certain cell types, such as red blood cells, platelets, and cancer cells. 45 This BioM coating gives the NPs biological features such as surface receptors, adhesion molecules, and antigenic profiles that are similar to those found in the donor cells. Biologically modified BioM NPs can preserve the cell membrane’s intrinsic targeting capabilities and immunomodulatory activities, allowing for selective recognition and binding to target cells in both treatment and imaging.33,45 Cell membrane-mimetic surface engineering and cell membrane camouflaging technologies have been extensively studied in the context of natural biology. 3 Notably, cell membrane-camouflaged NPs (CM NPs), which are formed by fusing synthetic NPs with cellular membranes, are receiving unprecedented interest in biomedical sectors. Compared to typical nanosystems, these BioM NPs have the advantage of extended circulation duration, immune evasion, 45 and active targeting, all of which have significant promise in a variety of biomedical applications and are expected to transform traditional nanomedicine.
So far, many cell membranes, as well as a wide range of core materials, have been intensively studied to create a BioM system with distinct functions. Erythrocytes are frequently employed for the production of cell membrane camouflaged NPs because of their diverse origins. However, it is unclear if the distinction between autologous and allogeneic sources for erythrocyte membranes affects the performance of disguised NPs. Dai et al. created two aggregation-induced emission (AIE) photosensitizers camouflaged with erythrocyte membranes (E-M), termed E-Mauto@P and E-Mallo@P, which were synthesized using autologous and allogeneic erythrocytes, respectively. In vivo, E-M@P-mediated photodynamic therapy (PDT) substantially suppressed tumour development, and the therapeutic effect was the same for E-Mauto@P and E-Mallo@P. Significantly, there were no variations in general condition, organ function, or immune system between the mice administered with E-Mauto@P and E-Mallo@P. Both E-Mauto@P and E-Mallo@P have been demonstrated to not pass the placental barrier and have no effect on embryo development, suggesting that they might be a promising platform for treating pregnancy-related diseases. These findings offered additional support for erythrocyte membrane disguised NPs as a viable therapeutic platform, as there is no difference in effectiveness or biosafety between autologous and allogeneic erythrocyte-derived NPs.
Breast cancer has emerged as the most often diagnosed cancer, accounting for around 11.7 % of all cases worldwide, and poses a significant danger to human health. The development of a nano-drug delivery system that is extremely relevant to the clinical real world is an ongoing and pressing issue. Sui et al. described an intelligent bioinspired nano-platform that uses mesoporous bioactive glass NPs (MBG NPs) in conjunction with cell membrane camouflage and enzyme-catalyzed death to treat in situ breast cancer. M0 phenotype macrophages were found to have a particularly high infiltration proportion in breast cancer patients utilizing TCGA analysis and were therefore chosen as cell membrane-derived donors. As a result, macrophage-coated MBG NPs outperformed cell membrane-free and breast cancer cell membrane-coated NPs in terms of evasion of clearance by myeloid lineage cells and good targeting qualities to tumours (Figure 7). The macrophage membrane-camouflaged MBG NPs were then used to load the glucose oxidase (GOx), which catalyzed glucose to cause high oxidative stress and the death of breast cancer cells, ultimately preventing tumour development. The schematic design for comparing the immune escape and tumour-targeting capabilities of MBG, 4T1@MBG, and RAW M@MBG. (b) Flow cytometry gating technique for myeloid and lymphoid lineage cells to analyze the fluorescence intensity of labelled MBG, 4T1 M@MBG, and RAW M@MBG in the six immune cells. (c) Fluorescence intensities of tagged MBG, 4T1 M@MBG, and RAW M@MBG in monocytes, neutrophils, DC, NK cells, B cells, and T cells. (d) Fluorescence images showing intensities of MBG, 4T1 M@MBG, and RAW M@MBG on the surface of 4T1 cell spheroids. (e) In vivo and ex vivo tissue distribution fluorescence imaging of orthotopic breast tumour-bearing mice following intravenous injections of MBG, 4T1 M@MBG, and RAW M@MBG. (f) The average CPS of the lungs, livers, and tumours from the animals listed. Reproduced with permission un. License number 5765431117053.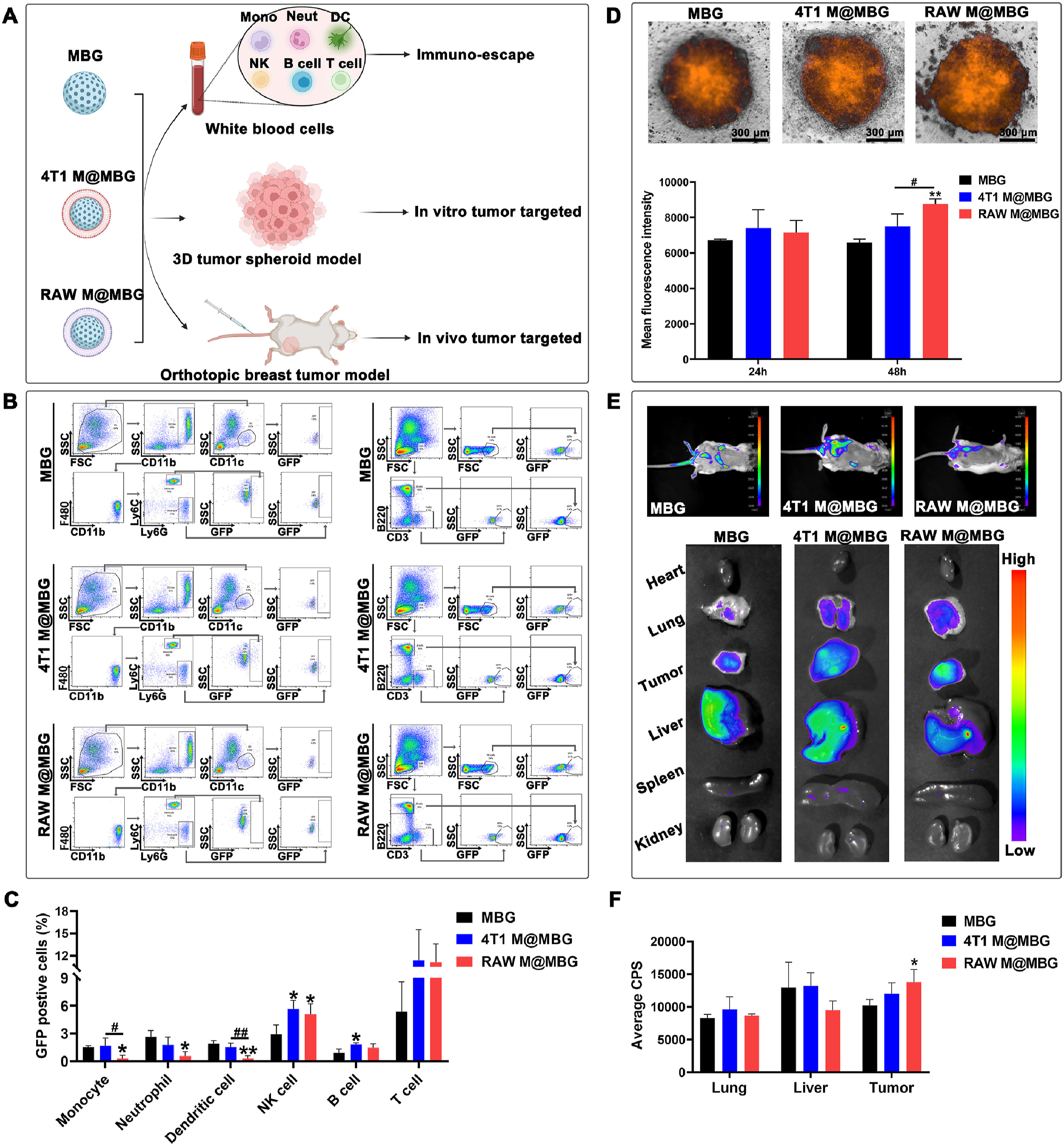
Furthermore, the macrophage membrane-camouflaged MBG embedding glucose oxidase delivery method efficiently overcomes the substantial alterations in haematological and biochemical parameters caused by non-specific glucose catalysis, potentially reducing the risk of toxicity. Taken together, the findings open the path for the development of an intelligent biomimetic MBG nano-loading enzyme platform appropriate for targeted breast cancer treatment. The natural chemical cinobufagin (CS-1) has demonstrated potential for colorectal cancer treatment. However, the limitations of hydrophobicity and cardiotoxicity prompted an upgrade in chemical composition. Meanwhile, single chemotherapy for colorectal cancer (CRC) has problems such as limited drug accumulation in tumour locations and nonspecific toxicity. To circumvent these restrictions, cinobufagin-loaded Prussian Blue NPs (PC NPs) were created by Luo et al. for CRC treatment by combining them with PTT. The hybrid cell membrane coating was used to enhance the immune escape and tumour-homing potential of PC@M NPs. Both in vitro and in vivo studies showed that chemo/photothermal treatment not only lowered CRC growth but also prevented primary metastasis by downregulating the expression of Vimentin, matrix metalloproteinase-9, and hypoxia-inducible factor1-alpha. Furthermore, our method restored some of the natural intestinal microbiota in CRC-bearing animals. When combined, these suggested nanoplatforms are a possible method for CRC treatment.
Overall, cancer cell membranes include a plethora of highly unique homotypic moieties that can be exploited to identify their type. These cell membranes are frequently employed to encapsulate spherical NPs to improve nanomedicines’ targeting specificity and uptake. At any one time, a sphere makes just one point of contact with a surface. Zhu et al. demonstrated that by stiffening the cracked cell membrane with in situ synthesized Au nanomaterials, an increased area of interaction could be maintained, thereby improving the in vitro and in vivo homotypic targeting capabilities of cancer cell types. This improvement was particularly relevant in vivo, as any nanomedicine containing targeting moieties is likely to interact with the target cell once before being cleared by the system. Possible future clinical applications may involve the use of a patient’s autologous tumour biopsy tissues, which are in short supply, so ensuring maximization of the entire collective surface area of the cancer cell membrane available becomes an important consideration in the design and delivery of cell membrane-derived nanomedicines. Besides, CM NPs have been shown to retain their physicochemical properties while still possessing natural cell membrane components, presenting a novel strategic notion for overcoming the problems that nano-drug-delivery systems face. Amid many membrane modifications, erythrocyte membrane coating has gained attention for improving stability, immune evasion ability, and blood retention duration thanks to a self-recognition protein produced on the surface. Meanwhile, cancer cell membrane coating was embraced due to its superior homotypic targeting power and potential to increase tumour formation. Thus, the hybrid membrane camouflage technique has been widely investigated and used to offer NPs with full benefits of erythrocyte and cancer cell membranes for tumour treatment.
Concluding remarks
BioM NP has significant promise for increasing the functioning of nanoscale structures by duplicating and utilizing the inherent qualities of natural biological systems; recreating specific attributes of biological entities for a variety of medical objectives; and drawing inspiration from the complicated design and capabilities of cells, viruses, or extracellular components. Magnetic BioM NPs (MBioM NPs) are a promising technology because of their scalability and potential to be manipulated via chemical or genetic engineering. MBioM NPs have been created via physicochemical methods, with a minor part dedicated to biological processes such as protein synthesis, bacteria, and fungus. Furthermore, molecular approaches for peptide assembly may be employed to generate nano-sized crystals and self-generation of multi-purpose NPs. Solid-binding proteins/peptides can achieve these BioM NPs under-regulated synthesis and organization. BioM NPs offer flexible systems with customizable characteristics for addressing essential NanoMed issues such as targeted drug delivery, wound healing, bone formation and/or regeneration, cancer therapy, and theranostics. Their biomimetic nature enhances biocompatibility, efficacy, and specificity, making them ideal tools for generating healthcare advances. Besides, cell membrane-mimetic surface engineering and cell membrane camouflaging technologies have been extensively studied, drawing inspiration from natural biology. Notably, CM-NPs, which are formed by fusing synthetic NPs with cellular membranes, have received exceptional attention in biological sectors. Owing to these advancements, this review article is divided into three sections covering BioM NP production methods and their synergistic interactions with human cells, tissues, organs, and organ systems, BioM NPs for wound healing, targeted drug delivery, theranostics, cancer therapy, and bone formation and/or regeneration; and a discussion of BioM NPs’ novel applications. We aim to give researchers an overview of the present status of research on BioM NPs for NanoMed, which will act as a guide for current and future investigations.
Footnotes
Author contributions
Eliasu Issaka: Conceptualization, Methodology, Investigation, Resource, Supervision, Writing original draft, Review and editing; Blessing Tornyeava: Resource, Investigation, Writing original draft, Enock Adjei Agyekum: Investigation, Writing original draft; Michael Enyan: Methodology, Resource, Investigation; Jesse Nii Okai Amu-Darko: Investigation, Resource, Writing original draft; Hither Tariro Chimuza: Resource, Writing original draft.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
Most of the data generated or analyzed during this study are included in this published article.
