Abstract
Dental implants, coated with materials, are strategically implanted in the bones, serving a vital function in accelerating healing and promoting bone regeneration in the vicinty of implants. Applying a polymer matrix as a coating material emerges as a promising approach for enhancing the properties of these implants. This research investigates the impact of Zein-CaSiO3 coating on PolyEtherEtherKetone (PEEK) implants using the electrospray technique. The study includes control PEEK disks and experimental groups with different coatings: Zein-CaSiO3, Zein, and CaSiO3. Characterization involved techniques such as atomic force microscopy to evaluate the surface topography of the composite coating, scanning electron microscopy (FE-SEM) was employed to analyze the morphology of surface, water contact angle, and adhesion test. Group 3 (PEEK coated with CaSiO3) exhibited the lowest water contact angle in comparison to the control group, Also there were present an increasing in adhesion strength to the substrate with the groups 1 (PEEK coated with zein-CaSiO3) and 2 (PEEK coated with zein) compared to Group 3. This study suggests that the electrospraying of (Zein-CaSiO3) coating on PEEK dental implants is a promising option for enhanced implant performance.
Introduction
The absence of natural teeth resulting from conditions like tooth decay, periodontal disease, and injury can disrupt essential functions such as chewing, speech, and one’s physical appearance. 1
An increasing demand for dental implants has resulted in the development of numerous surgical and prosthodontic procedures, driving the rapid expansion of the market. 2
PEEK is a high-performance thermoplastic polymer that can take the place of metallic implant components in traumatology and orthopedics. These results implied that PEEK might be used as a dental endosseous implant material instead of titanium. 3
Polyether ether ketone (PEEK) belongs to the family of polyarylethrketones and represents a superior engineering plastic. Its structure comprises an aromatic structure along with ether and ketone functional groups sandwiched in between the aryl rings. PEEK possesses several great qualities, such as good X-ray penetration, heat resistance, ease of machining, frictional resistance, excellent biocompatibility, and a modulus of elasticity that is similar to human bone. 4
PEEK is now considered an alternate solution to traditional implants made of metal and orthopedics due to its superior mechanical qualities, low immunotoxicity, and strong processing performance. 5 It can address certain drawbacks associated with metal implants, including issues like stress shielding and metal allergies. 6
PEEK, an implanted biomaterial, closely resembles cortical bone in terms of its modulus of elasticity, with an unaltered value of 3–4 GPa. In contrast, PEEK’s hydrophobicity renders it bioinert and impairs cellular attachment.7,8
It is necessary to make improvements since the lack of biological reactivity of pure PEEK prevents in vivo integration with host bone tissues. Strategies for surface modification, including wet chemical, plasma treatment, and electron beam evaporation, have been suggested to enhance the osseointegration capabilities of PEEK prostheses 9 and to make a porous, hydrophilic, and rough PEEK surface, which are the desirable surface qualities of PEEK in dental applications. 10
The process of osseointegration, which is the growth and attachment of bone, has been facilitated by effective implant surface treatment. 11 Osteoblasts (bone-producing cells) react differently to implantable materials depending on the morphology and surface chemistry of those surfaces. 12 Osteointegration, described as “a direct interrelation of function and structure between healthy bone tissue and the supporting implant surface, is one of the most important variables in assessing the clinical effectiveness of implantation. 13
Plant proteins have the advantage of being cheap and renewable, which makes them appropriate for use in biomedical research. Plant proteins are considered more reputable than animal proteins due to these characteristics. 14 Zein is a corn-based, insoluble prolamin protein. It was granted FDA accreditation in 1985 as GRAS (Generally Recognized as Safe). 15
Calcium silicate (CaSiO3) has emerged as a promising candidate for bone regeneration applications owing to its verified bioactive nature, exceptional biocompatibility, superior mechanical characteristics, and inherent degradability.16–18 Because of its exceptional bioactivity, Ca-Si bioceramics is one of the most promising options for application in orthopedics and dentistry. 19
Calcium silicate (CaSiO3) has been demonstrated to elicit a specific biological response at the tissue-material interface, facilitating the formation of a connection between the materials and the tissue. The process involves the arrangement of a biological analog of the hydroxyapatite layer that contains a carbonate group (chemically and crystallographically mirroring the mineral composition of bone) through ion-exchanging reactions between the implant and the surrounding body fluids (SBFs). Such interactions foster the integration between the implanted material and the natural tissues 20
Within this research, various coatings (Zein-CaSiO3, Zein, and CaSiO3) will be applied to the surface of dental PEEK implants through the process of electrospaying.
One effective method for getting a consistent coating is electrospraying, which involves applying an electric potential to the substance used for coating. Here’s where competing forces are involved. The charge induced by the electrical field, the visco-elastic forces, and the surface tension of coating materials maintain the coating’s hemispherical form. 21
There has been no research conducted on the properties of the electrospray Zein/Calcium Silicate composite for PEEK implant coating material that can alter the rate at which the area around the implants heals. However, as PEEK is used more often in clinical applications, osseointegration has become rather poor, which is probably due to PEEK’s natural biological inertia. 22
PEEK is a hydrophobic substance, which means that it has difficulty forming bonds with liquids like water. This can make using dental cement or adhesives, which are usually hydrophilic materials, difficult to obtain excellent adherence. 10
Furthermore, there are several drawbacks to using calcium silicate ceramics as coating materials. They can impede cell growth due to their rapid dissolution rate and extreme release of Si and Ca ions. Second, the material’s capacity to sustain mechanical stability under physiological stress is diminished because calcium silicate’s mechanical strength falls short of the parameters necessary for cancellous as well as cortical bone reconstruction. Furthermore, the osteoinductivity and bone ingrowth characteristics of calcium silicate are still insufficient. Because of these drawbacks, scientists have worked to cover PEEK with other materials to enhance its mechanical and osteogenic qualities. 23 Therefore, the uniqueness of this work is to determine if calcium silicate’s performance as a coating material on PEEK is impacted by the inclusion of protein material.
Material and method
Preparing and cleaning PEEK implant material disks
Prepare disc shaped specimens from medical-grade PEEK material with dimensions of 2 mm thickness and a diameter of 10 mm for coating with Zein/CaSiO3
Modification of PEEK surface
Subject disc specimens to a 60-s etching process in 98% H2SO4, followed by rinsing with water and ethanol. 24
Preparation of coatings solutions
Preparations of Zein-CaSiO3 solutions (30% Zein-70% CaSiO3) involved using a magnetic stirrer to dissolve Zein protein powder in ethanol for 30 min. After that adding CaSiO3, the mixture was subjected to probing sonication for 10 min to guarantee uniform distribution of the ceramic material.
Zein solution was prepared by dissolving Zein protein powder (6 wt% to distilled water (20 wt%) as well as ethanol (74 wt%) in a 100 mL bottle and the solution was strring on a magnetic stirrer for 30 min at room temperature to ensure complete solubility of protein powder. 25
CaSiO3 solution was prepared by suspending (150 mg) in 14 mL of ethanol Then dispersing the mixture in a probe sonicator for 10 min to improve homogeneity. 26
Electrospraying process
Coating of PEEK disk was accomplished through electrospraying, employing a syringe pump (high-tension power supply, voltage 30 kV) to distribute the solution throughout a metallic needle (21 microns needle guage) at a 1.5 mL/h flow rate. The capillary-collected distance was maintained at 5 cm, powered by a voltage source 9 kV. To optimize the electrospraying parameters, the trial was initially conducted on an aluminum foil before applying the coating to a PEEK substrate.
Characterization of coating
Atomic force microscopy test (AFM)
A very popular and useful methods for identifying surface topography, roughness, and morphology is AFM. AFM is made up of a cantilever that makes contact with the material surface and has a sharp, extremely sensitive probe tip.
Scanning electron microscope (SEM)
The surface shape of PEEK disks was examined using SEM. with all different coatings Zein-CaSiO3,Zein and CaSiO3, as well as disks with simply the surface treated and control disks. Also coating thickness for all the studied groups were examined using SEM
Wettability test
To determine whether PEEK disks had better hydrophobicity, the water contact angles of the disks with all of the coatings were assessed and compared with the control disks. A distilled water drop was administered to the target surface using a tiny syringe, and then the water contact angle picture was captured by using the Si-Plasma Cam 110, Creating Nano Technologies, Taiwan. Once a droplet of room temperature liquid had remained on the intended surface for 30 seconds, a picture could be captured.
Cross-cut adhesion test
An investigation was conducted to test the adhesive connection between the different coatings and PEEK disks by using adhesion (cross-cut) testing on all coated specimens.
Results
Characterization test
AFM (atomic force microscope)
AFM was employed to evaluate the surface morphology and roughness of various groups, including the control, etched, and all studied coating groups involving (Zein–CaSiO3) composite, Zein, and CaSiO3 groups as seen in (Figure 1) to ensure the success of coating-based implants, it is crucial to measure the degree of topographical changes on their surface. AFM investigation for (a): control group, (b): The treated surface of PEEK, (c): Coated with Zein-CaSiO3,d:coated with Zein,e: coated with CaSiO3.
The modified surface of PEEK with H2SO4 acid increased PEEK surface roughnes about twice times and was examined by atomic force microscopy.
Descriptive statistic of surface roughness test (nm).
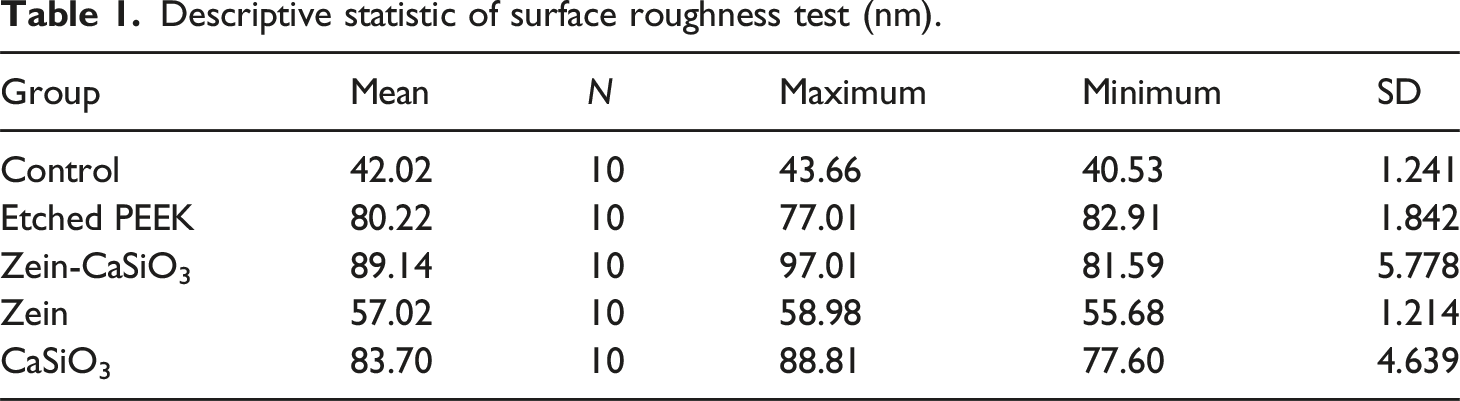
ANOVA test for surface roughness test.
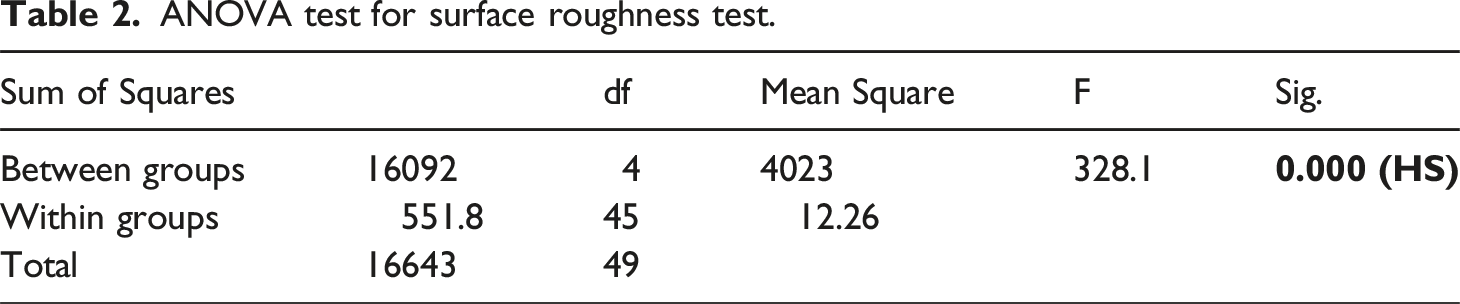
Test of Homogeneity of Variances for Surface roughness.

Games-Howell test between groups of surface roughness.
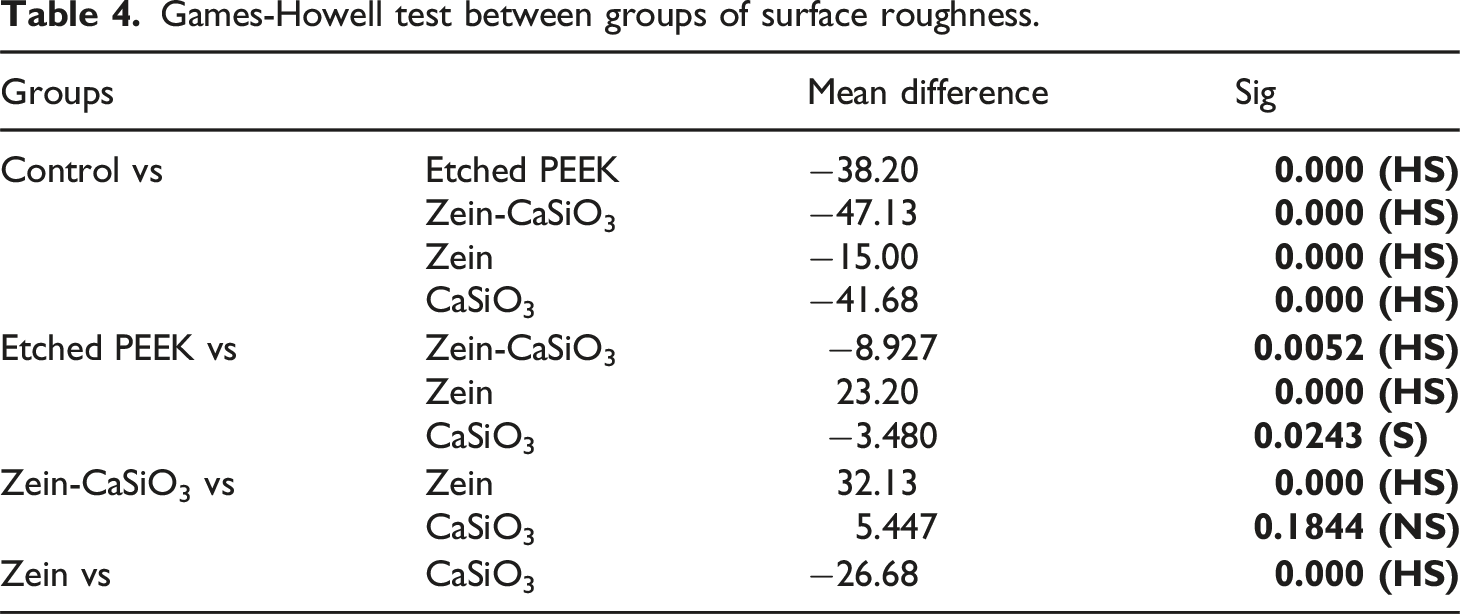
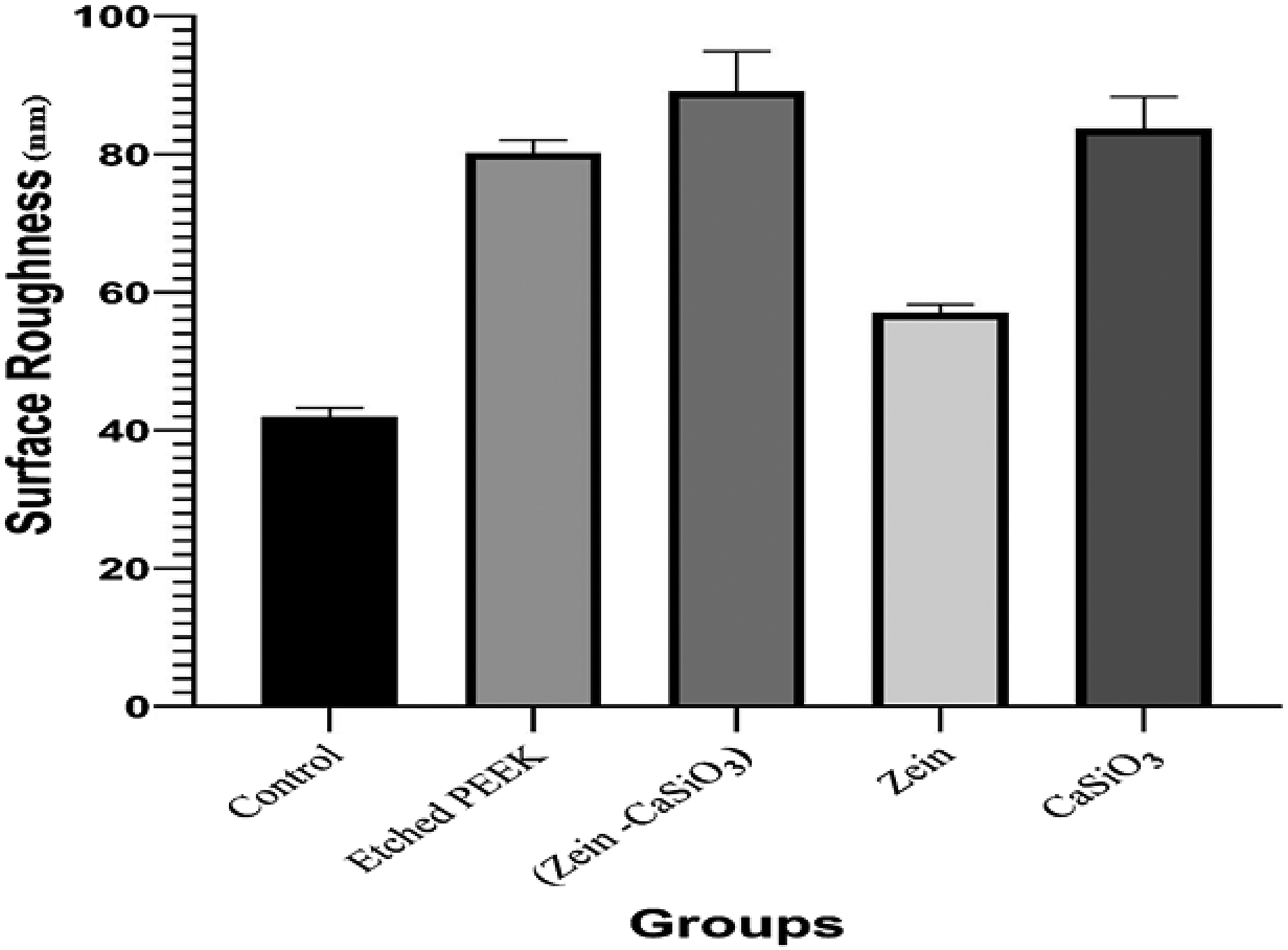
Chart Compare between different groups in Surface roughness test.
FE-SEM
Surface morphology plays a crucial role in influencing cell behavior on implants. Factors like connectivity and surface properties are key determinants affecting interactions such as cellular adhesion, migration, and proliferation.
FE-SEM investigations of the controlled, treated surface and coated PEEK with various coatings, as illustrated in Figure 3, showed that no protein particle clusters were present with bioceramic material at high resolution images of the coated composite-based samples. This suggests that the protein powder and bioceramic material were adequately mixed before electrospraying. SEM investigation (a) Control group, (b)Modified surface with H2SO4 group, (c) Coated with Zein-CaSiO3 group(d) Coated with Zein group (e) Coated with CaSiO3 group.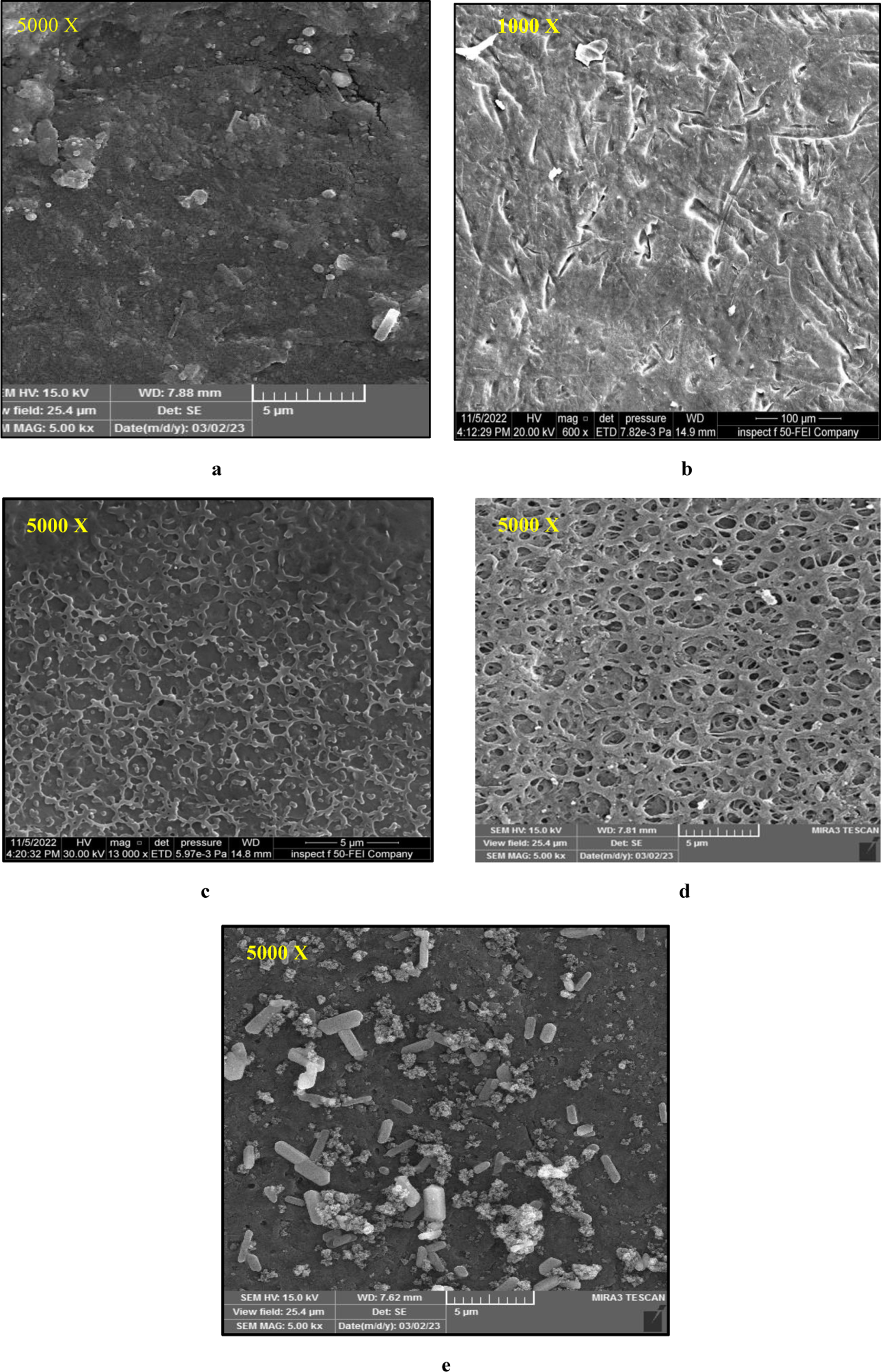
The coating thickness values for Group 1, coated with Zein-CaSiO3, ranged from 1.987 to 2.490 μm. Group 2, coated with Zein, had a thickness range of 1.025 to 2.028 μm. In contrast, the coating thickness for Group 3, coated with CaSiO3, ranged from 1.792 to 2.181 μm as shown in Figure 4 Coat thickness for a: Group 1(Coated with Zein-CaSiO3) b: Group 2(coated with Zein) c: Group 3(coated with CaSiO3).
Wettability test
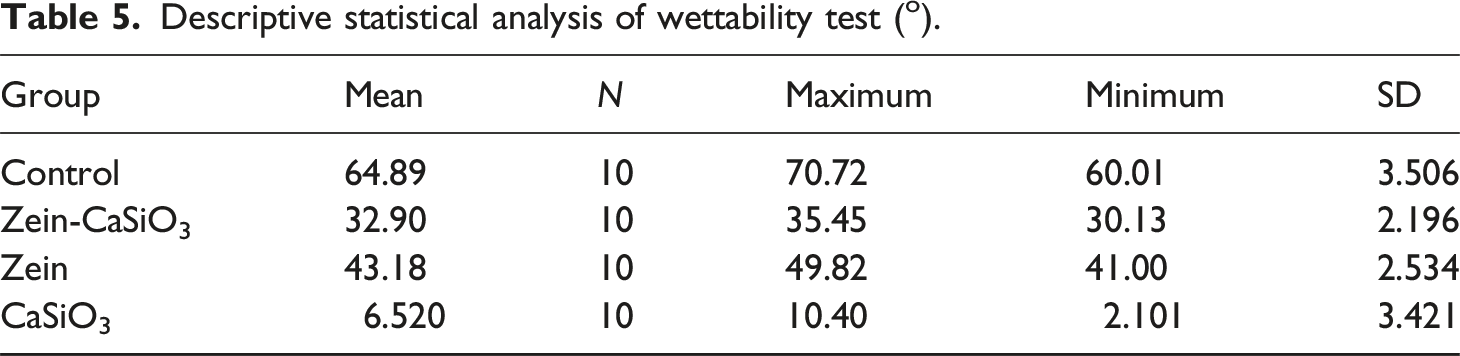
Descriptive statistical analysis of wettability test (o).
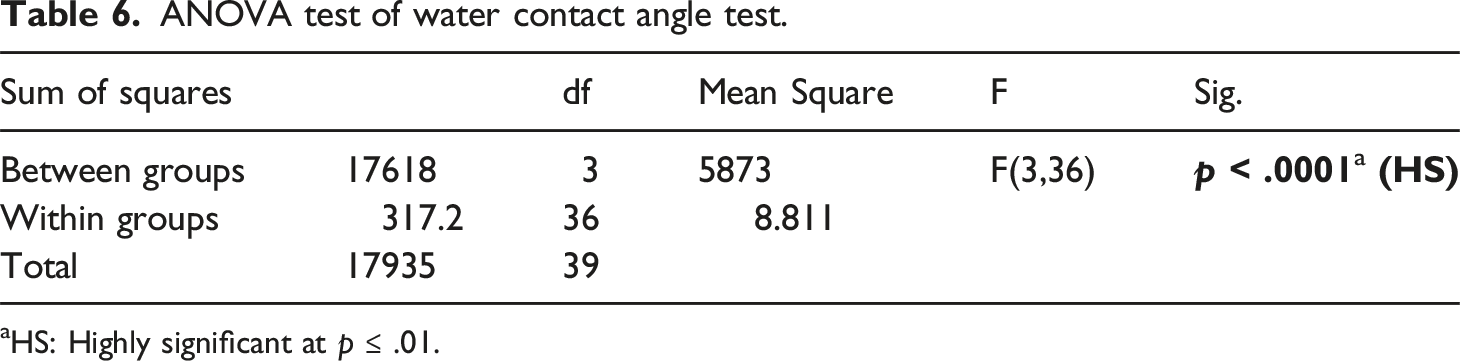
ANOVA test of water contact angle test.
aHS: Highly significant at p ≤ .01.
Test of Homogeneity of Variances of water contact angle.

aNS: Non significant at p > .05.
Bonferroni test between groups of water contact angle test.
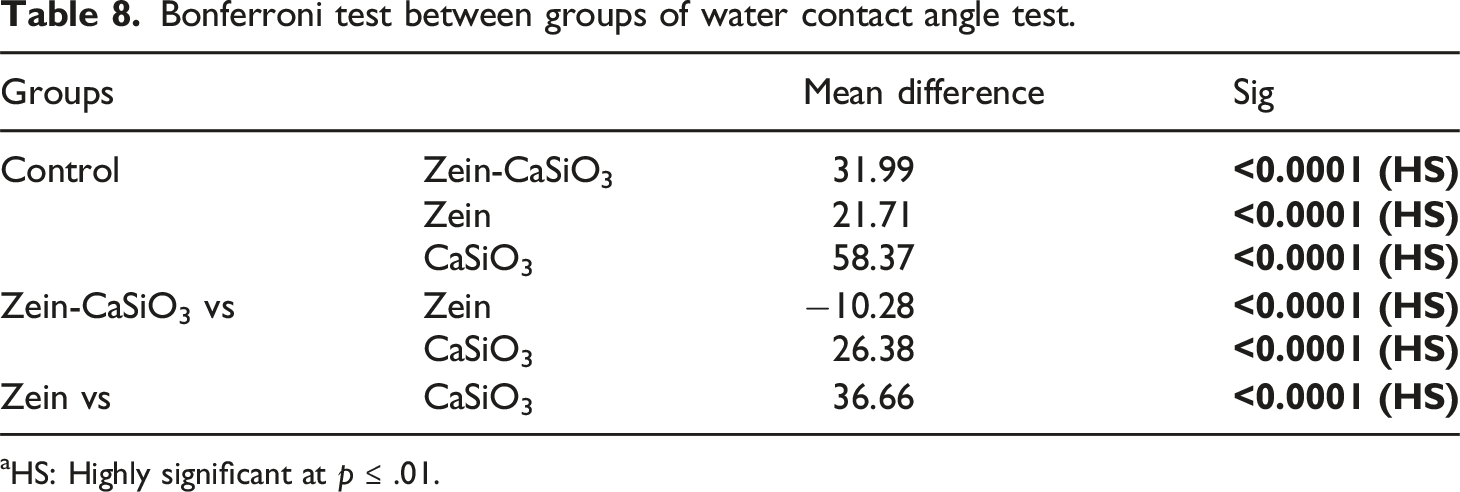
aHS: Highly significant at p ≤ .01.
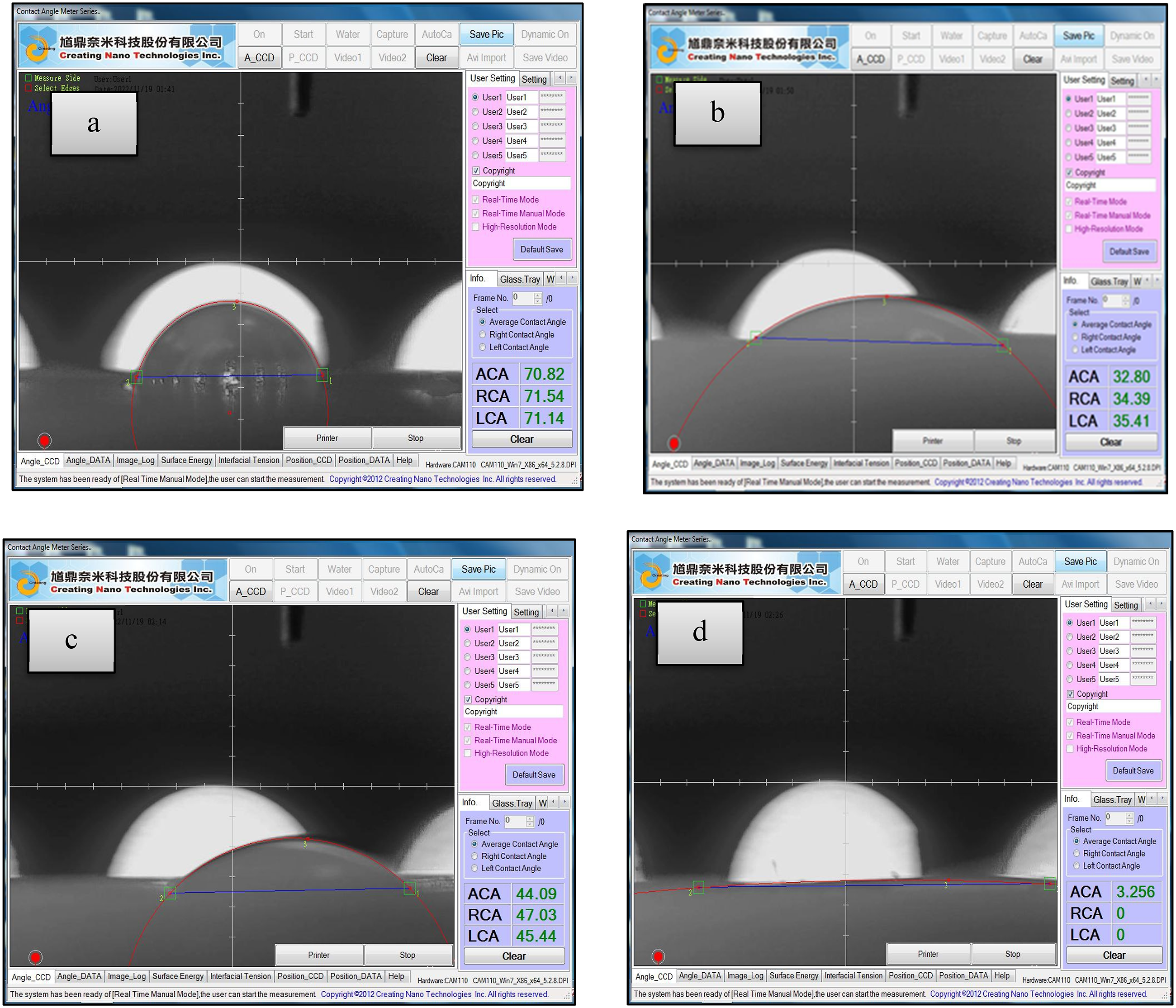
Water contact angle images of a: Control group, b: Zein-CaSiO3 coated group, c: Zein coated group, d: CaSiO3 coated group.
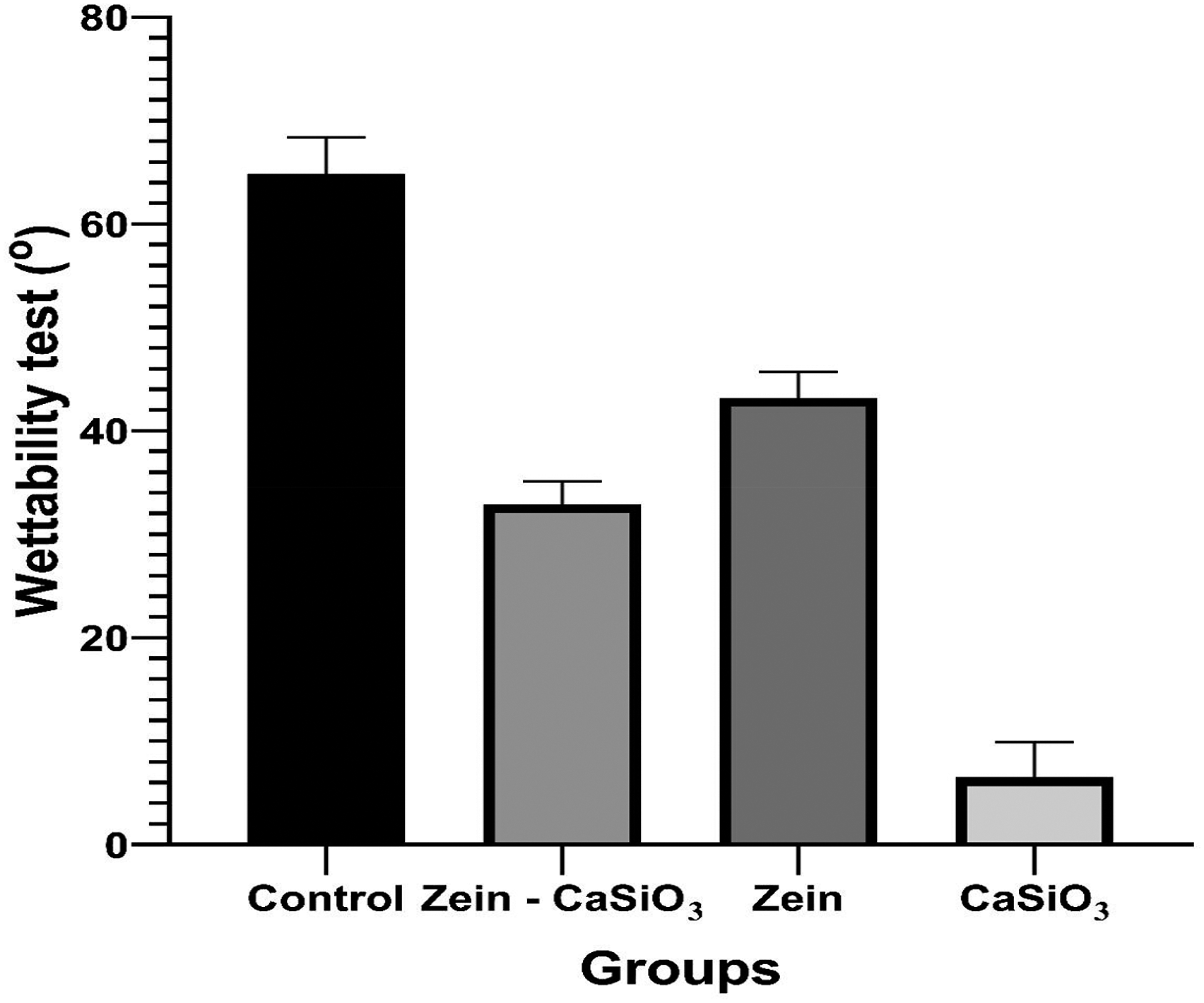
Chart Compare between different groups in water contact angle test (o).
Cross-cut adhesion test
A cross-cut, scratch adhesion test uses a tool to cut a rectangular grid design within the coating and pass through the substrate in order to assess a coating’s resistance to delamination from a substrate. This process does a fast pass/fail test. It is possible to assess how resistant certain layers are to separating from one another while evaluating multi-layer systems. ASTM D 3359 27 provides standards for this procedure. Cross-cut test advice provides a six-step categorization. When a pass/fail evaluation is necessary, the first three phases will be employed because they are enough in most cases. There may be exceptional situations when the whole six-step categorization is required.
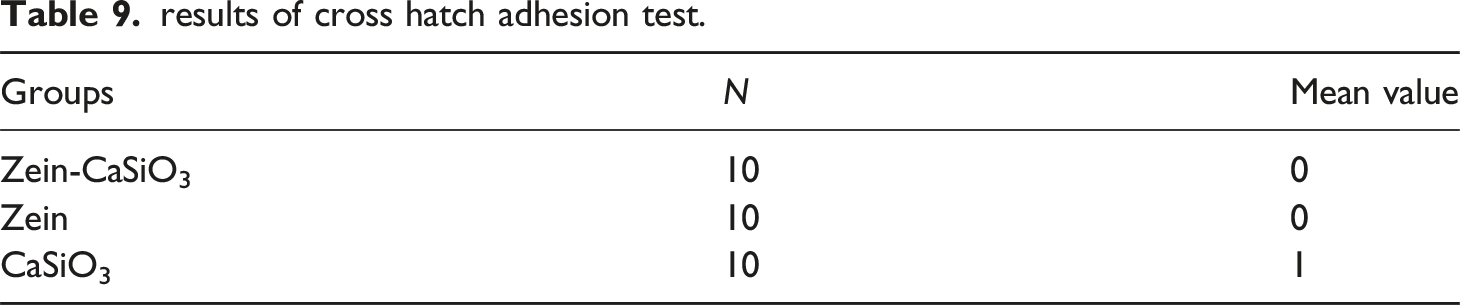
results of cross hatch adhesion test.
Discussion
In biomedical applications, electrospraying is often used to coat implants with bioactive materials that promote tissue growth and improve implant integration. 28
The study’s findings demonstrated that the surface modification of PEEK with H2SO4 acid significantly increased the surface roughness of PEEK, approximately twice fold. This finding is consistent with previous research that has demonstrated the ability of acid etching to increase surface roughness and improve the bonding properties of PEEK substrates.10,29 The increased roughness of the surface can be explained by the removal of the PEEK’s outer layer, exposing a more irregular and textured surface by sulfonation reaction producing nanopores 24
Surface topography plays a crucial role in the success of implants with coatings, as it affects the adhesion and integration of the coating with the implant surface. 30
The surface modification and coating of PEEK with different coatings, as well as the controlled observations using SEM showed uniform distribution of the composite coating, indicating that the electrospraying technique resulted in a well-crafted surface.
The absence of clusters of protein particles with bioceramic material in the electrosprayed coating can be attributed to the high electric field used during the electrospraying process. This high electric field causes the materials to break up into fine droplets, which then solidify in mid-air before landing on the surface to form a uniform coating. 31 The absence of clusters of protein particles with bioceramic material in the electrosprayed coating on PEEK implants indicates that the electrospraying technique resulted in a well-designed surface with uniform distribution of the composite coating.
Moreover, the adequate mixing of the materials before electrospraying can be attributed to the use of high-shear mixing techniques, such as sonication and high-speed homogenization, which ensure adequate mixing and dispersion of the materials in the solvent before electrospraying. 32
The coating thickness in this investigation ranged from 1 to 3 μm. According to the research conducted by Marchiori et al., 33 the coating that was 4 μm thick achieved the most favorable outcome, whereas the coating that was 1 μm thick performed the least well. Regarding sample stress, the coating with a thickness of 1 μm exhibited the highest level of stress, while the coating with a thickness of 4 μm had inferior results compared to the coating with a thickness of 0.5 μm. The latter option allowed for the most accurate adherence to the specified loads. The danger of debonding from the implant and potential partial breakdown increases when the coating thickness of A exceeds 50 µm. 34
Sopcak et al. 35 proved that implants with a thickness of 200 µm had better adhesion strength compared to thinner coatings with an average thickness of less than 50 µm. Even though ultra-thin coatings fall short of protective standards, adhesion does improve with decreasing thickness.
Conversely, it is well-known that dense ceramic coating deposition processes can result in breakage or cracking. 36 When the thickness of coatings is increased, they become more prone to cracking or delamination, as stress accumulates with it. In the case of thicker coatings, the outer layer of the implant may separate from it, while thinner coatings may cause the covering to resorb too quickly during bone regeneration1,37
Water contact angle is a measure of the wettability of a surface, with a smaller contact angle indicating higher surface hydrophilicity. Evaluating water contact angles can provide insights into the surface properties and coating effectiveness of different materials.
According to the results presented, the control group had the highest mean water contact angle of 64.89°, indicating a relatively hydrophobic surface. However, the group coated with Zein (a protein-based material) showed a decreased water contact angle of 43.18°, indicating improved surface hydrophilicity in comparsion with the control group. Additionally, the group coated with Zein-CaSiO3 (a composite material) exhibited a further decrease in the water contact angle (32.90°). The group coated with CaSiO3 (ceramic material) showed the lowest water contact angle of 6.520°, indicating the highest surface hydrophilicity among the studied groups.
The decrease in water contact angle observed with the application of these coatings can be explained by the chemical composition and surface morphology of the coating materials. Zein, as a protein-based material, contains hydrophilic functional groups like hydroxyl and peptide such as glutamine; thus, it enhances the surface hydrophilicity. 38 The addition of calcium silicate to Zein enhances its affinity for water, resulting in a more hydrophilic coating. Furthermore, the presence of glutamine chains on the coating’s surface may further contribute to its water-attracting properties. 25
The lower water contact angle observed with the CaSiO3 coating indicates the highest surface hydrophilicity. This can be attributed to the hydrophilic nature of CaSiO3, which increases the affinity to water molecule, promoting wetting and reducing the contact angle. 39
The results demonstrate that coating the surface with Zein, Zein-CaSiO3, and CaSiO3 materials progressively reduces the water contact angle, resulting in enhanced surface hydrophilicity. The improved hydrophilicity of the coated surfaces can have implications for various biomedical applications, including improved cell adhesion, protein adsorption, and potentially enhanced biocompatibility.
Stable attachment between implants and tissues takes a long time to develop since bioinert materials do not encourage interaction with live tissues. 40
Calcium silicate (CaSiO3) has been suggested as a promising substance for the regeneration of bone tissue due to its shown bioactivity and degradability. 16
The rate at which apatite forms on the CaSiO3 surface is quicker than that of other glass-ceramics and bio-glasses in simulated bodily fluid (SBF). Eventually, a negative charged surface containing silanol (Si-O) groups is produced when the Ca2+ ions in CaSiO3 exchange with H+ in the SBF. This causes the surface layer to create Si-OH and raises the pH value at the coating-SBF interface, which explains the bioactivity of CaSiO3. Apatite will precipitate onto the coating when the ionic activity product of the interfacial area is high enough, following the formation of the negatively charged surface. This occurs because the Ca2+ ions in the SBF are attracted to the coating/SBF interface. 16
A cross-cut adhesion test is a widely used method to determine the adhesion characteristics of coatings. It involves creating a rectangular grid pattern on a coated surface with a cutting tool and then applying a pressure-sensitive tape to the surface and removing it. The extent of coating removal from the substrate is evaluated by a visual inspection. This test is systematized by ASTM D 3359 (Standard Test Methods for Measuring Adhesion by Tape Test) and is also described in a recent publication by Magdaleno-López and de Jesús Pérez-Bueno. 27
The six-step categorization included in the guidelines of adhesion tests facilitates the interpretation of the test results. Although the complete six-step grade may not be necessary for general purposes, the first three steps are usually satisfactory and are used to determine a pass or fail result when assessing coating adhesion. In exceptional circumstances where a more detailed assessment is necessary, the complete six-step classification may be required. 27
This study found that the composite material-coated group and the Zein-coated group demonstrated perfect adhesion, with a score of zero and smooth edges. This result can be attributed to the excellent binding properties of both materials.
In contrast, the group coated with CaSiO3 exhibited partial detachment, as indicated by a score of 1. This outcome suggests that the adhesion of CaSiO3 to the substrate was not as strong as that of the composite material and Zein coatings. According to previous research, the adhesion of coatings to substrates is influenced by various factors, including the surface energy of the substrate and the chemical bonding between the coating and the substrate. 41 It is possible that the substrate used in this study had a low surface energy, which could have affected the adhesion of the CaSiO3 coating.
The critical factors influencing the efficacy of ceramics-substrate adhesion encompass interfacial chemistry, interfacial morphology, and mechanical stress dynamics. 42 It is feasible to utilize a rough PEEK substrates for improving the bond strength by means of mechanical interlocking.
Extensive research has shown that coating delamination rates are significantly reduced when surface roughness is gradually increased from 11 to 305 nm. This effect of surface roughness is as a result of the higher surface area of the interface and the correspondingly greater number of contacts that are present across the interface. 43
The adhesion of the coating to the substrate is the most critical mechanical property that determines the operation and performance of any biomedical implant that has been coated. Adhesion failure or separation of the coating from the substrate are the causes of implantation failure and unstable long-term performance. Both of these issues might occur simultaneously. 44
Overall, the results suggest that the composite material and Zein coatings demonstrate superior adhesion properties compared to CaSiO3. Understanding the factors that influence coating-substrate adhesion can help in the development of more effective coatings in various applications.
Conclusion
By keeping the study limitation in mind. It can be said that the group coated with CaSiO3 exhibited the lowest water contact angle in comparsion with the control group. Additionally, there was an increase in adhesion strength to the substrate with group coated with Zein and group coated with Zein-CaSiO3 compared to group coated with CaSiO3. Therefore, the study suggests that electrospraying a coating of Zein-CaSiO3 on PEEK implants shows promise for enhancing implant performance.
Footnotes
Acknowledgements
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Data availability statement
All the data analyzed in study contained within the main manuscript.
