Abstract
Fullerene has been acknowledged as a significant nanocarbon nanofiller enhancing the imperative polymer characteristics. Since, thermoplastic polymers constitute a large group of polymeric materials, fullerene has been used as reinforcement in these matrices mostly via non-covalent means. This state-of-the-art review article summarizes thermoplastic polymer nanocomposites reinforced with fullerene nano-additives without involving any covalent interactions. Accordingly, thermoplastic polymer/fullerene nanocomposites of interest have non-covalent or physical interactions in the matrix-nanofiller such as van der Waals forces, electrostatic interactions, hydrogen bonding, and aromatic stacking interactions. Number of thermoplastic polymers including polyamide, polyurethanes, and block copolymers have been non-covalently or physically reinforced with the fullerene molecules. Ensuing high performance thermoplastic polymer/fullerene nanocomposites exhibited improved microstructure, electrical, mechanical, thermal, and other physical properties. Enhancements in the thermal, mechanical, and electrical properties of the thermoplastic/fullerene nanomaterials were found dependent upon the nanofiller contents, orientations, interfacial effects, and processing. Consequently, the polyamide/fullerene systems were found efficient to enhance the glass transition up to 260°C, in addition to optimum mechanical properties. Polyurethane/fullerene systems performed better for improved tensile strength and young’s modulus features up to 90 MPa and 48 GPa, respectively. System based on poly (methyl methacrylate) and fullerene has resulted in high thermal degradation temperature in the range of 501-633°C with fine electrical conductivity of 1.3 Scm−1. Using combination of fullerene and graphene nanofiller (due to synergistic effects) has been found to improve the electrical conductivity considerably in the range of 1.8–2.5 Scm−1 for a polystyrene and block copolymer system. However, attaining fine fullerene nanoparticle dispersion of non-covalently reinforced matrices have been found important affecting the final nanocomposite properties. Consequently, processability and essential characteristics of non-covalently fullerene filled nanocomposites can be influenced due to nanoparticle aggregation. Hence, the physical property enhancement potential of physical linking between the non-covalently linked thermoplastics-fullerene has been portrayed in this article. Research on non-covalently interacted thermoplastic polymer/fullerene nanocomposites revealed technical potential ranging from energy/electronic devices to engineering and biomedical sectors. This review article can be a useful guide for the field researchers towards the development of advanced systems using non-covalently linked polymer/fullerene nanomaterials for future technical applications.
Introduction
Continuous development of innovative polymeric nanocomposites has been the foremost focus of modern technologies. 1 In this context, various nanoparticles have been used as the nanocomposite reinforcements. Important properties of the resultant nanocomposites greatly rely on the nanofiller dispersions in the nanocomposite matrices to enhance the ultimate industrial or engineering utilizations. 2 Nanofiller addition has been found to directly influence the indispensable physical features of the nanocomposites such as electrical conduction, heat stability, mechanical robustness, flame retardance, barrier, anti-corrosion, and several other physical characters. 3 Use of nanocarbon nanofillers have been widely explored and found beneficial for developing high performance polymeric nanocomposites. 4 Among carbon nanoparticles, fullerene has gained attention owing to zero dimensional nature, unique features, and property improvement of polymeric matrices.5,6 Consequently, fullerene nanocomposites have been developed with variety of thermoplastics, thermosets, rubbers and conducting polymer matrices. As thermoplastics constitutes large important category of polymers, fullerene has been reinforced in these polymers, mostly developing the non-covalent interactions. 7 Nevertheless, some reports of covalent linking of functional fullerene molecules have been observed for thermoplastic polymers. Among non-covalent or physical interactions, fullerene molecules have been interacted with polymer matrices through various weak links. The final nanocomposite properties (microstructure, mechanical, thermal, conducting, etc.) have been found greatly dependent on interactions among thermoplastic-fullerene as well as the nanoparticle dispersal for further utilizations.8–10 As the physical interactions especially hydrogen bonding, electrostatic associations, dipole links, and aromatic stacking have been found beneficial to enhance the important physical characters of the thermoplastic polymer/fullerene nanocomposites, so, the potential of these nanocomposites has been enhanced towards practical applications electronics, engineering, transpiration, and biomedical field. 8
This state-of-the-art overview basically highpoints the importance of non-covalently linked or physically reinforced thermoplastic polymer/fullerene nanocomposites. Various fullerene reinforced thermoplastic matrices have been surveyed to explore the effect of non-covalent linking in these innovative nanocomposites. To the best of the knowledge, this cutting-edge review is novel in literature due to its innovative outline and framework including best available literature so far. This article is definitely helpful for the field scientists, engineers, and academics to understand the importance of non-covalent interactions in developing high performance polymer/fullerene nanomaterials.
Fullerene
Fullerene molecules are hollow spherical caged nanocarbons.11,12 It is a zero dimensional nanostructure. Fullerenes are five/six atom based polygons observed in 1985. 13 It can be imagined as a rounded graphene nanosheet with sp2 hybridized atoms. 14 Subsequently, alternating single and double bonds have been found responsible for π delocalization in fullerene rings. 15 Fullerene molecules may have different number of carbon atoms in the rings. 16 Smallest fullerene observed is C20 having 20 carbon atoms in spherical structure. 17 Then higher fullerene have been developed like C28, C30, C60, C70, etc. 18 Fullerene C60 is the most symmetrical molecule commonly referred as Buckminster fullerene.19,20 Technological applications of fullerene have been explored for varying engineering and energy fields. 21
Dispersion Properties of Carbon Nanoparticles in Polymer Matrices
Fullerene and quantum dots are spherical nanoparticles with zero dimensions.22–24 Nanocarbons with other dimensionalities have been observed like one dimensional carbon nanotube and two dimensional graphene nanosheets.25–27 In polymer matrices, very small amounts of nano-additives have been used to improve the physical properties of the nanomaterials. 28 The property variations have been found dependent upon the nanoparticle contents 29 as well as dispersion in the polymers. 30 Furthermore, matrix-nanofiller interactions both physical (electrostatic, aromatic stacking, van der Waals, etc.) or covalent affect the physical properties of the polymeric matrices.31–33 Variety of polymers have been filled with carbon nanoparticles to attain fine morphology and physical features.34–36 In addition to experimental studies, modeling or theoretical methods have been used to examine the matrix-nanofiller interactions in polymer/fullerene and dispersion properties of the nanocomposites.37,38 Consequently, superior electron transference, thermal stability, strength, thermal conductivity, and functional properties have been investigated for nanocarbon nanocomposites.39,40 Usually, carbon nanofillers have been identified to pointedly amend the thermal, mechanical, electrical, and other features of polymeric matrices, at low loading levels. By systematically fluctuating the nanofiller contents, functionality, dispersion, and orientation, physical properties of the nanocomposites have been altered. Actually, the physical features responsible for dispersion and morphology dependent ordering of polymer chains at matric-nanofiller-interface, thereby providing controlling and enhancing the electrical, thermal, and mechanical properties of nanocomposites.41–43 Especially, the surface modified fullerene has been found to affect the electrical and mechanical properties of polyurethanes nanocomposites. 44 Including fullerene has led to twofold upsurge in tensile strength (24-42 MPa) and Young’s modulus (33-65 MPa) of the nanocomposites. In this context, silane modified fullerene based nanocomposites have been designed to develop physical interactions with polyurethanes. These nanocomposites have finely dispersed fullerene nanoparticles. Consequently, the electrical conductivity of the nanomaterials was also enhanced. Hence, surface treatments like oxidation and silanization have been found to increase the uniform dispersion towards superior mechanical and conductivity features.
Polyamide Matrices with Fullerene Reinforcement
Polyamide is a class of thermoplastic polymer made up of amide monomers.45,46 Polyamide may exist naturally or can be synthesized. Important properties of polyamides like microstructural, crystallinity, mechanical, thermal, and chemical properties rely on the hydrogen bonding and interactions among the amide groups of the polyamide chains.47–49 Properties of polyamides have been enhanced using various nanocarbon (graphene, carbon nanotube) and inorganic (nanoclays) nanofillers.
50
Among nanocarbon nanofillers, fullerene molecules have been found important nanofiller for polyamide matrices such as polyamide 6, polyamide 66, and polyamide 12 to attain enhanced physical features.51,52 Consequently, polyamide and fullerene derived nanocomposites have been designed and studied for matrix-nanoparticle interactions and physical properties.53–55 Faykov and co-workers
56
investigated the nanocomposites of a polyamide, poly (m-phenylene isophthalamide), with heteroarm star fullerene C60 core macromolecule to form nanocomposite and ionic liquid. The resulting nanomaterials were studied for morphology and gas separation studies for He/N2, CO2/N2, and O2/N2 gases. Figure 1 shows the scanning electron microscopy images of pristine polyamide, poly (m-phenylene isophthalamide)/heteroarm star fullerene C60 nanocomposite, and poly (m-phenylene isophthalamide)/heteroarm star fullerene C60 ionic liquid through solvent casting method. The unfilled polyamide had typical uniform polyamide morphology. On the other hand, the polyamide matrix filled with fullerene revealed spherical domains. In the ionic liquid form, the spherical domains of the fullerene molecules were more homogeneously distributed throughout the matrix with sizes of 0.3-2 µm. Such distinct sphere-shaped structures were suggested to be developed due to polar armed heteroarm fullerene nanostructures in the matrix. Scanning electron microscopy images of cross-sections for (a) PA; (b) PA/HSM; and (c)&(d) PA/(HSM:IL) membranes.
56
PA = polyamide; HSM = heteroarm star macromolecular fullerene C60; PA/HMS:IL = heteroarm star macromolecular fullerene C60 ionic liquid. Reproduced with permission from MDPI.
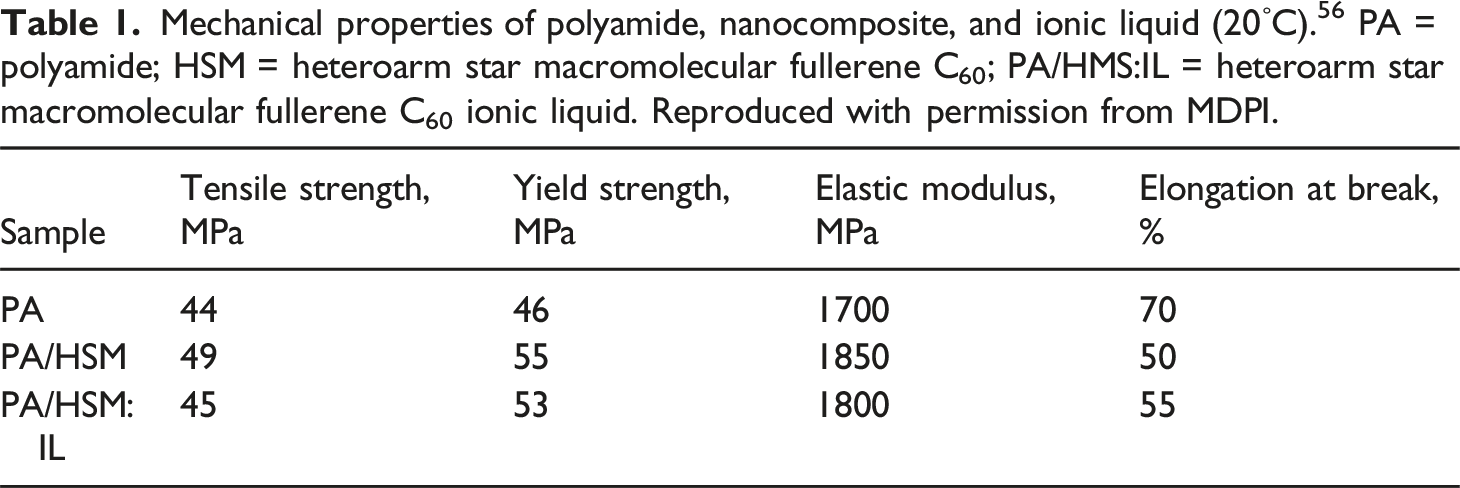
Mechanical properties of polyamide, nanocomposite, and ionic liquid (20 ◦ C). 56 PA = polyamide; HSM = heteroarm star macromolecular fullerene C60; PA/HMS:IL = heteroarm star macromolecular fullerene C60 ionic liquid. Reproduced with permission from MDPI.
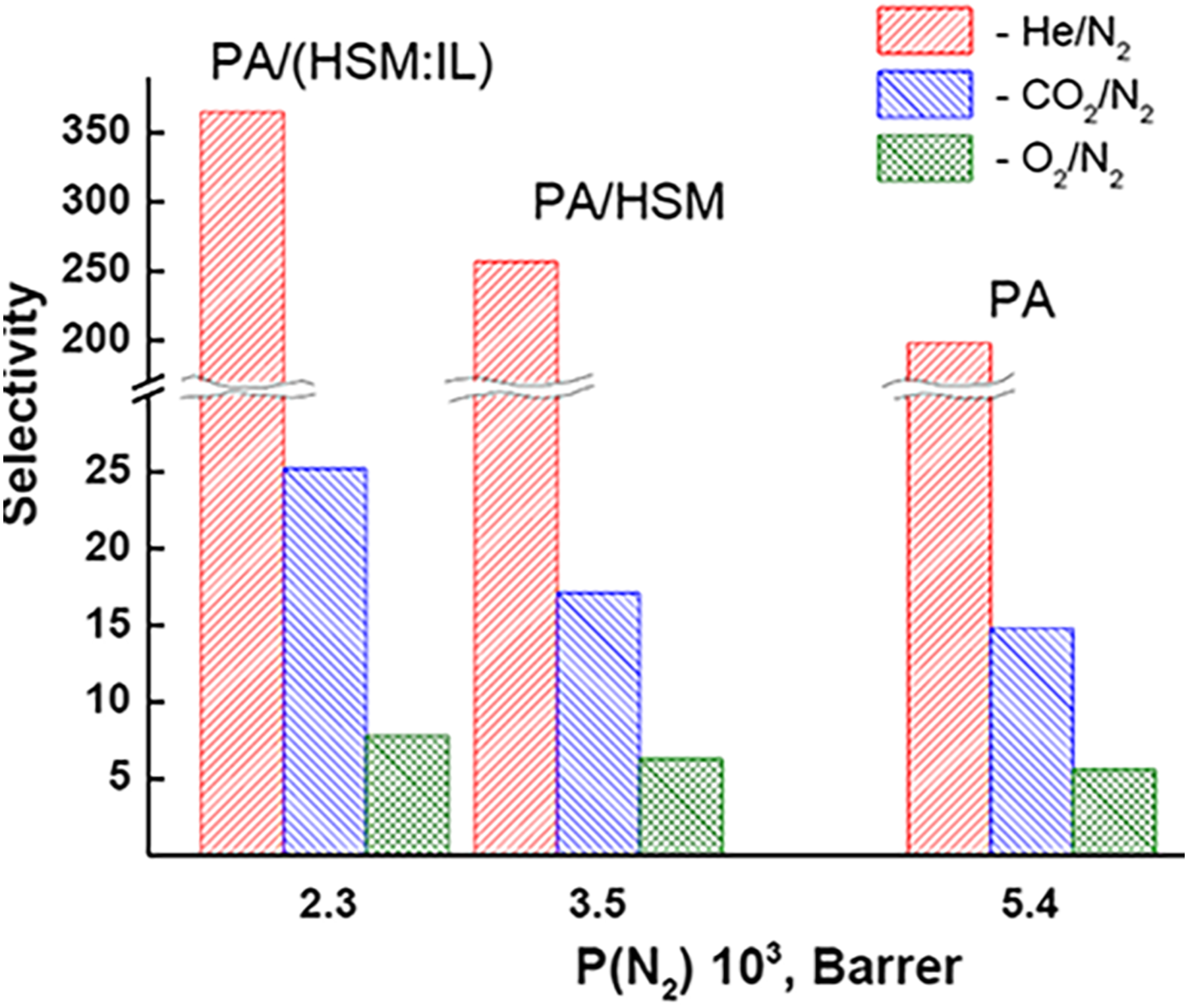
Dependence of ideal selectivity in separation of He/N2, CO2/N2, and O2/N2 gas pairs on nitrogen permeability using PA (3), PA/HSM (2), and PA/(HMS:IL) (1) ionic liquid membranes. 56 PA = polyamide; HSM = heteroarm star macromolecular fullerene C60; PA/HMS:IL = heteroarm star macromolecular fullerene C60 ionic liquid. Reproduced with permission from MDPI.
Mechanical properties of few reported polyamide/fullerene nanocomposites.

Polyurethane Nanocomposite with Fullerene Nanofiller
Polyurethane belongs to important category of thermoplastic polymers.64–66 Polyurethane main chain is made up of recurring urethane monomers. These are simply the condensation polymers formed by reacting together the isocyanate (R-(N = C = O)n and hydroxyl (R-(OH)n groups. 67 Polyurethanes have been processed by the in situ approaches and solvent routes.68–70 Among polyurethanes, segmented polymers have been formed through the reaction of a polyurethane prepolymer with the polyols or polyamines compounds. 71 Polyurethanes, especially segmented polyurethanes, have fine heat, mechanical, and chemical stability properties. The urethane functionalities in polyurethane have tendency to develop hydrogen bonding between the chains and also the nanofiller particles.72,73 Due to segmented structure having hard and soft units in the backbone, polyurethanes have superior structural and stability properties. 74 However, due to random chain growth of these polymeric chains, crosslinking effects have been observed. 75 Nanofillers like nanocarbon nanoparticles have been filled in the polyurethane matrix to improve the matrix characteristics.76–78 Fullerene C60 and functional fullerene molecules have also been filled in the polyurethane matrix to form the advanced nanocomposites. 79 Kausar 80 designed polyurethane using poly (ethylene adipate) tolylene 2,4-diisocyanate and polycaprolactone diol. The acid treated fullerene nanoparticles were studied for the formation of hydrogen bonding link with the segmented polymer. Inclusion of nanoparticles through physical route affected the physical features of the nanomaterials. The resulting polyurethane/fullerene nanocomposites revealed enhancements in the tensile strength and tensile modulus from 86.9 to 90.3 MPa and 33.3–47.6 GPa, respectively with the nanofiller addition. In addition, the non-covalent linking also enhanced the thermal stability profiles of the nanomaterials and maximum decomposition temperature and char yield were observed at 521°C and 45%, respectively. In another attempt, Kausar 81 formed a system based on the polyurethane/poly (2-chloro-5-methoxyaniline)/fullerene С60 nanocomposite using solution method. The nanocomposite revealed maximum temperature in the range of 501–633°C. The electrical conductivity enhancement was observed in the range of 10−2 to 1.3 Scm−1 due to interactions between the matrix and nanofiller. Shen et al. 82 reported on the thermoplastic polyurethane and fullerene С60 based system. Fullerene nanoparticles formed electrostatic attractions and π-π stacking interactions with isocyanate groups of polyurethane soft segments. The strong decoupling effects in translational-rotational diffusion and low ratio of translational-rotational diffusion coefficient (DT/DR) were observed with C60 addition. The technical applications of polyurethane and composite materials have been investigated for aerospace, automobiles, electronics, construction, packaging, and other important fields.83–85
Thermoplastic Copolymers and Block Copolymers Reinforced with Fullerene
Poly (methyl methacrylate) belongs to the category of commodity thermoplastic polymers due to facile processing and wide ranging application scope.86–88 This interesting polymer is comprised of repeating methyl methacrylate monomer in the main chain.
89
Owing to colorless and transparent texture, it has been used as an alternative to glass in to attain light weight and durability properties.
90
Generally, poly (methyl methacrylate) own high heat resistance, scratch resilience, and weather or corrosion consistency.91,92 Due to indispensable features, poly (methyl methacrylate) had applications in coatings, optical fibers, and biomedical fields.93–95 To further improve the features and applications of poly (methyl methacrylate), nanocarbon nanoparticles have been added to form the nanocomposites.96–98 Subsequently, fullerene has been identified as an important nanofiller for poly (methyl methacrylate) matrices.
99
Qi and co-workers
100
prepared the syndiotactic poly (methyl methacrylate) nanocomposites filled with fullerene C60. Fullerene was used in the form of small one-dimensional nano-wires having interlinking molecules. The syndiotactic poly (methyl methacrylate) chains may yield helical structures encapsulating small fullerene chains. These structures are often referred as peapods of fullerene. This encapsulation effect was observed due to the electrostatic interactions of fullerene molecules with the helical polymer chains (Figure 3). Like fullerene C60, C70, and C84 molecules have also be encapsulated in the helical polymer cavity.
101
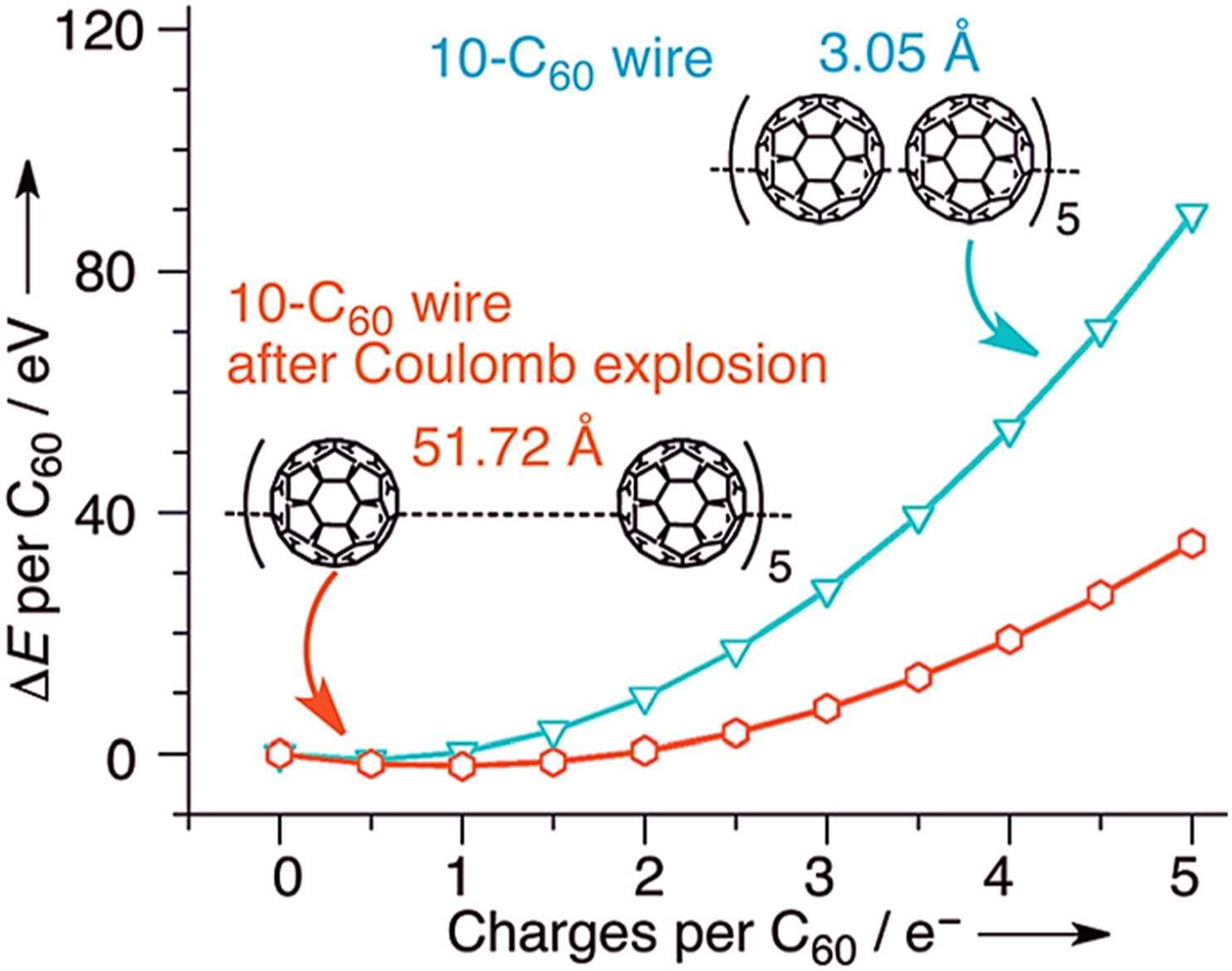
Figure 4 exhibits average energy changes before/after coulomb explosion versus average charge on fullerene molecules. The changes in the fullerene molecule orientations in small chains have been observed due to coulomb explosions. Izadi et al.
102
reported on poly (methyl methacrylate)/fullerene nanocomposites through molecular dynamic simulation method. Inclusion of 4 wt% fullerene contents enhanced the Young’s modulus to 3.77 GPa, that is, 24% higher than the unfilled polymer. Moreover, Glass transition temperature was observed around 358-364 K. The representative volume elements or RVE was found as 500K. Nasr et al.
103
prepared the poly (methyl methacrylate)/fullerene nanocomposites. They studied the dielectric and electrical properties of the matrix with varying fullerene loadings. The dielectric and electrical properties were reported in frequency range 1 kHz to 5 MHz. The electrical modulus was increased considerably in the range of 743–1,386,755 kHz. The increase in properties were due increasing fullerene contents and secondary interactions in matrix-nanofiller. Illustration of syndiotactic poly (methyl methacrylate) (st-PMMA) and C60 based helical nanostructure.
100
Reproduced with permission from Wiley. The average energy changes (DE) per C60 molecule as a function of average charge loaded on each C60 molecule in the 10-C60 molecular wire before and after Coulomb explosion at 27 e_, here only single-point energy calculation was done here.
100
Reproduced with permission from Wiley.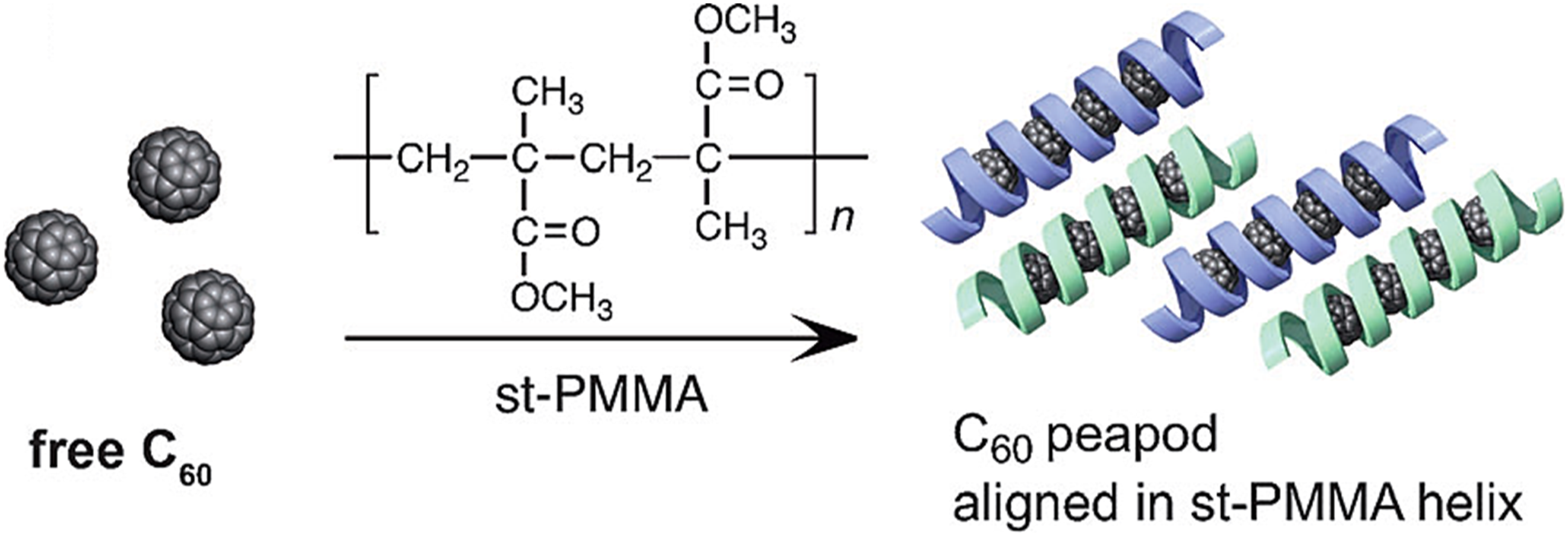
Among block copolymers, poly (n-alkyl methacrylate)s have side alkyl chains and are categorized as semicrystalline thermoplastic polymers.
104
Katiyar and co-workers
105
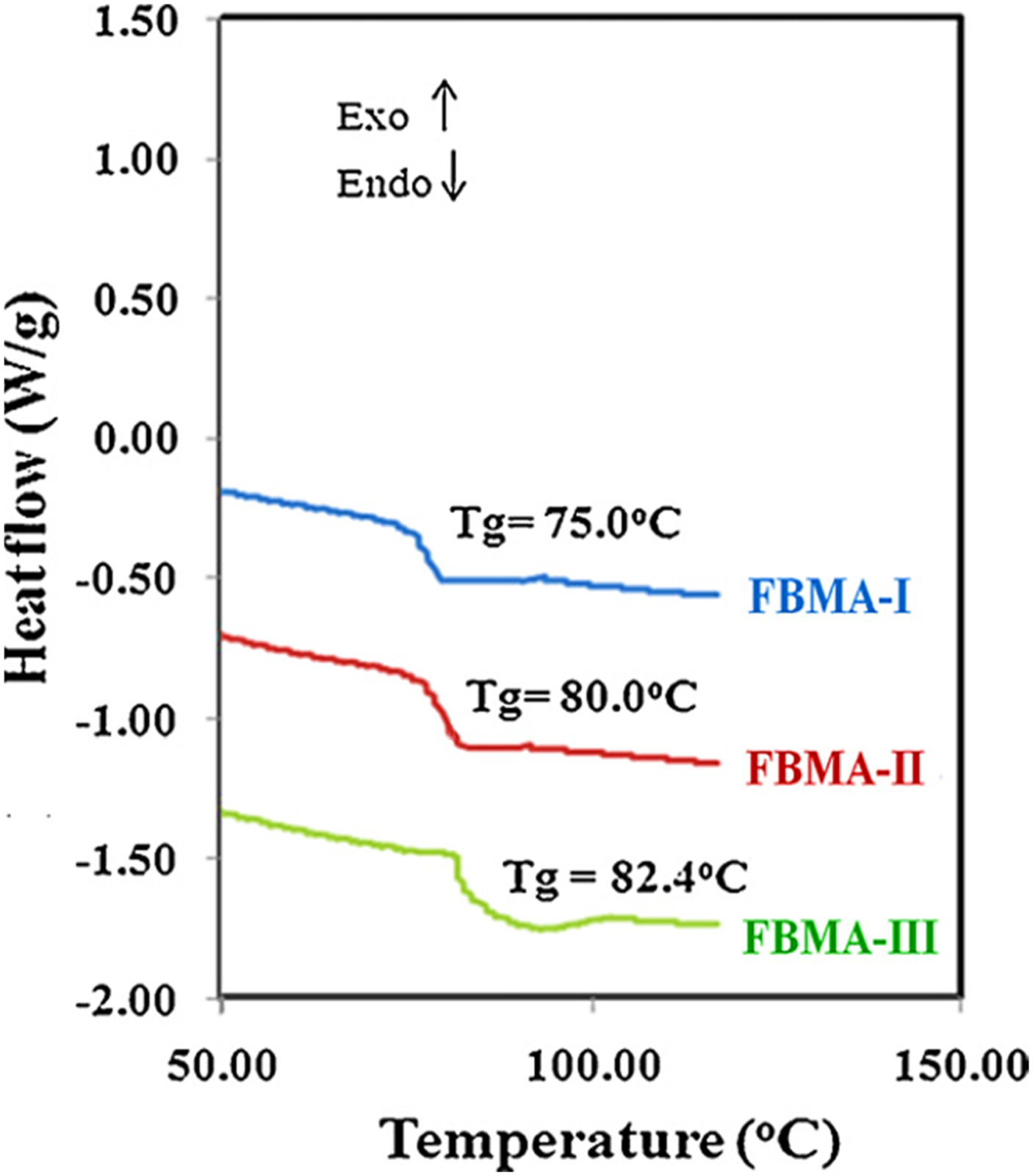
reported on the fullerene C60 filled poly (n-alkyl methacrylate)s nanocomposites prepared using in situ method. Figure 5 demonstrates the synthesis of poly (alkyl methacrylate)s/fullerene nanomaterial through the electrostatic interactions. Inclusion of nanoparticles improved the thermal profiles of the nanocomposites, compared with the unfilled matrix. Figure 6 shows the differential scanning calorimetry scans of these nanocomposites. Unfilled neat polymer had glass transition temperature of 75°C. Loadings of 0.015 wt% fullerene molecules raised the glass transition values to 82°C. The results revealed restricted polymer chain movements due to the presence of rigid fullerene molecules. However, neat polymer had loosely bound chains resulting in lower glass transition values. Schematic representation of synthesis of fullerene containing poly (n-alkyl methacrylate)s.
105
Reproduced with permission from Elsevier. DSC thermograms of FBMA polymer samples.
105
DSC = differential scanning calorimetry; FBMA = poly (n-butyl methacrylate) polymers. I = 30 mg/L; II = = 100 mg/L; III = 150 mg/L. Reproduced with permission from Elsevier.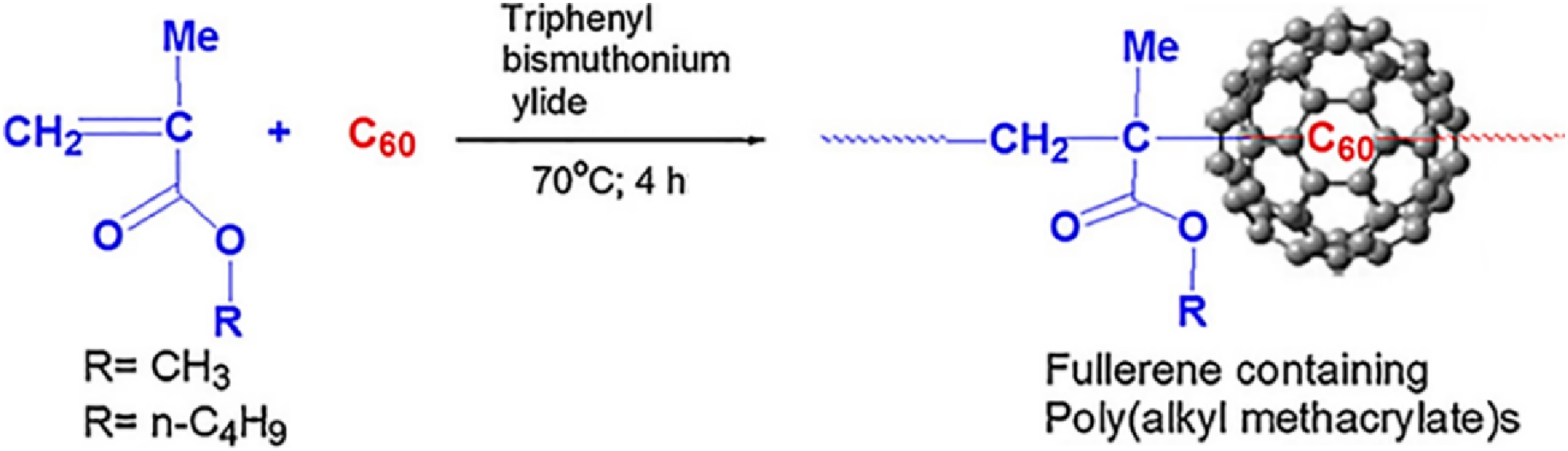
Teh et al.
106
used in situ technique for the formation of polyacrylonitrile/fullerene nanocomposites. The design of non-covalent interactions through aromatic stacking in the polyacrylonitrile-fullerene was studied. Figure 7 depicts an electron donor-acceptor linking model. The π-π non-covalent interactions in the donor-acceptor polymer/fullerene nanostructure is given in Figure 8. Figure 9 reveals a density functional theory and Gaussian basis set derived potential energy curve of the polyacrylonitrile/fullerene nanocomposite versus distance between fullerene center and acrylonitrile monomer. The change in potential energy profile was observed due to the non-covalent interactions between the aromatic groups of C60 and the monomer. Non-covalent electron donor-acceptor interactions of fullerene: (a) binding of acrylonitrile with fullerene; and (b) Binding of styrene acrylonitrile monomer with fullerene.
106
Reproduced with permission from ACS. Illustration of the formation of an electron donor-acceptor interactions.
106
Reproduced with permission from ACS. Potential energy curve of acrylonitrile/fullerene (AN-C60) with the C-C bond of AN aligned to the center of the hexagon on C60.
106
Reproduced with permission from ACS.
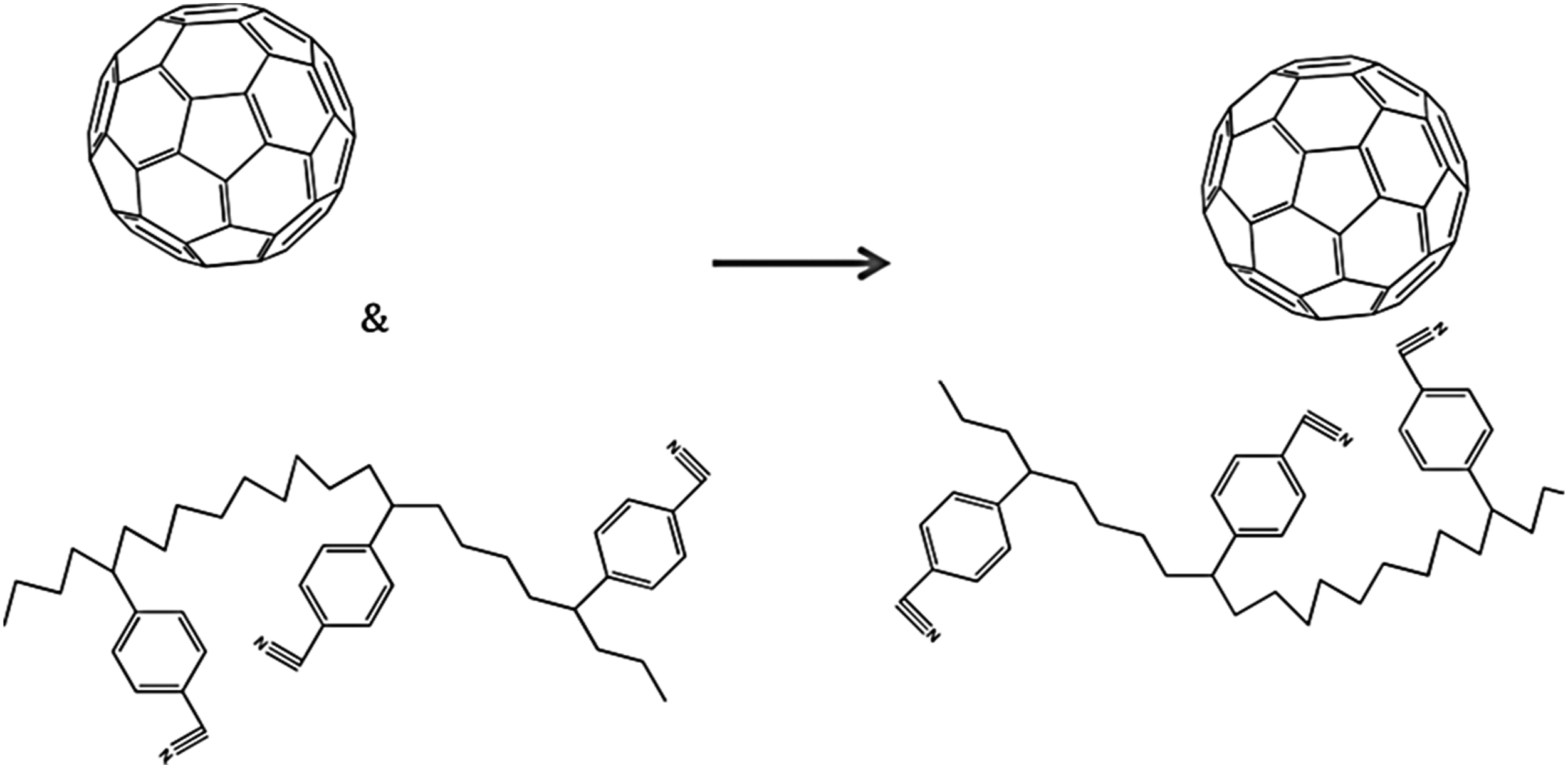
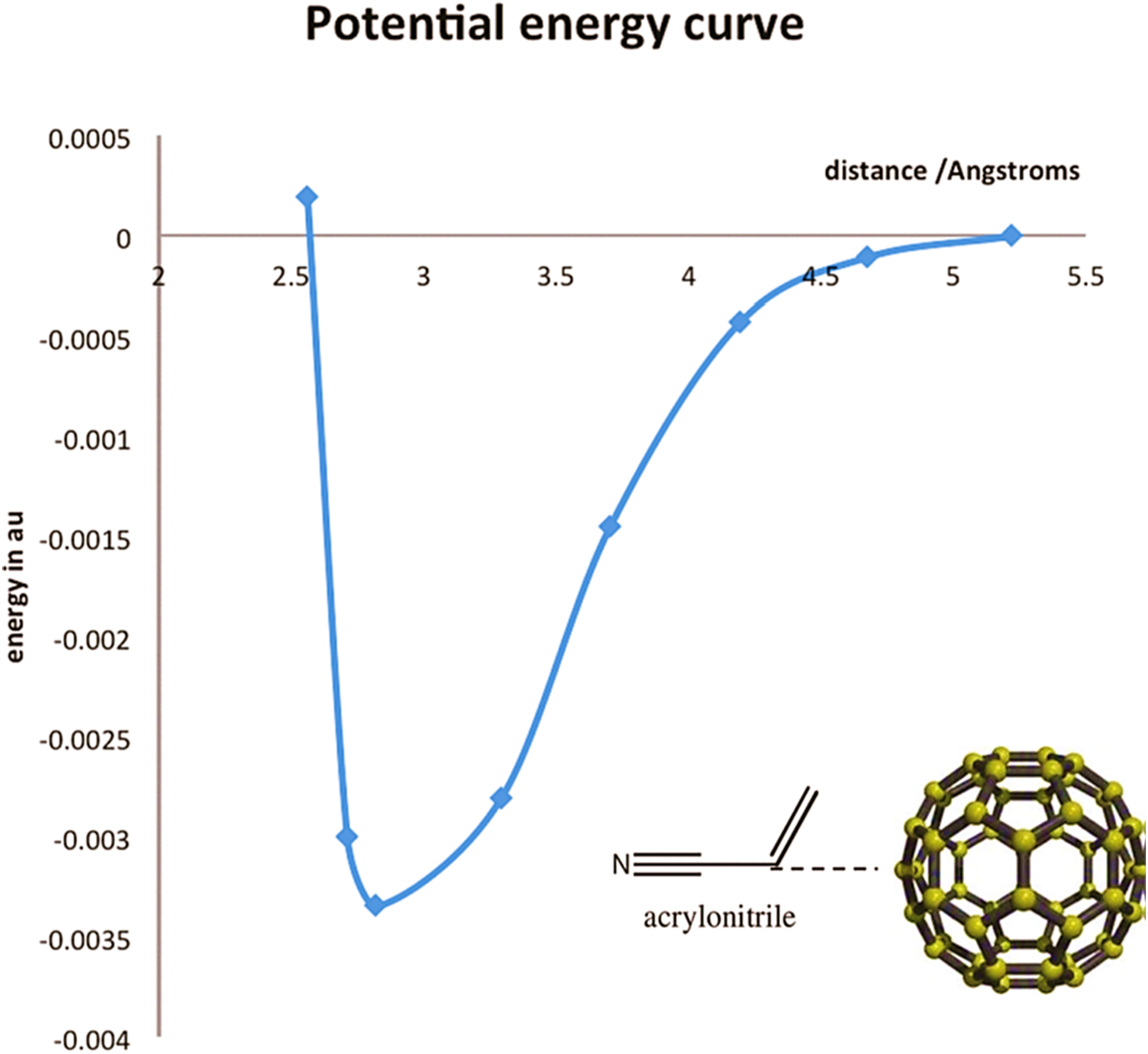
Figure 10 predicts the physical linkings probabilities of the poly (cyanostyrene-ran-styrene), poly (styrene-ranacrylonitrile), and poly (dimethyl amino ethyl methacrylate-ran-methyl methacrylate) matrices towards the fullerene molecules.
106
The electron donor or withdrawing behavior of block copolymers were dependent upon the nature of main chain functionality. Pereira and co-workers
107
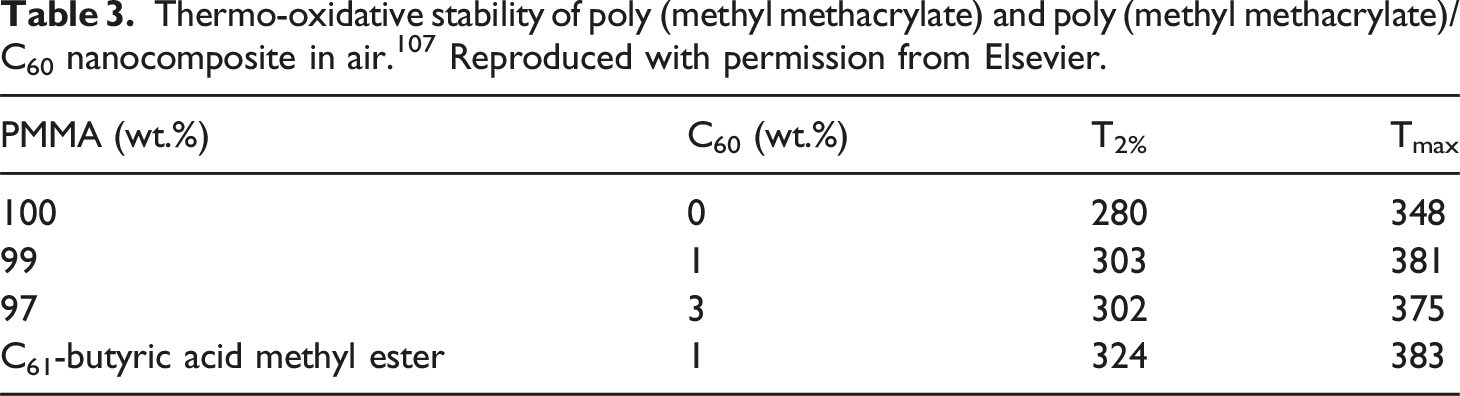
reported on the non-covalently reinforced fullerene C60 and C61-butyric acid methyl ester additives in the poly (methyl methacrylate) matrix. Fullerene nanoparticles were loaded up to 3 wt%. Inclusion of 1 wt% nanoparticles enhanced the thermal properties, compared with the neat matrix and 3 wt% nanofiller addition (Table 3). The thermo-oxidative stability of the nanomaterials was decreased at higher nanofiller addition due to nanoparticle aggregation effects. Structures of the copolymers used in this study.
106
(a) poly (styrene-ranacrylonitrile); (b) poly (cyanostyrene-ran-styrene); and (c) poly (dimethyl amino ethyl methacrylate-ran-methyl methacrylate). Reproduced with permission from ACS. Thermo-oxidative stability of poly (methyl methacrylate) and poly (methyl methacrylate)/C60 nanocomposite in air.
107
Reproduced with permission from Elsevier.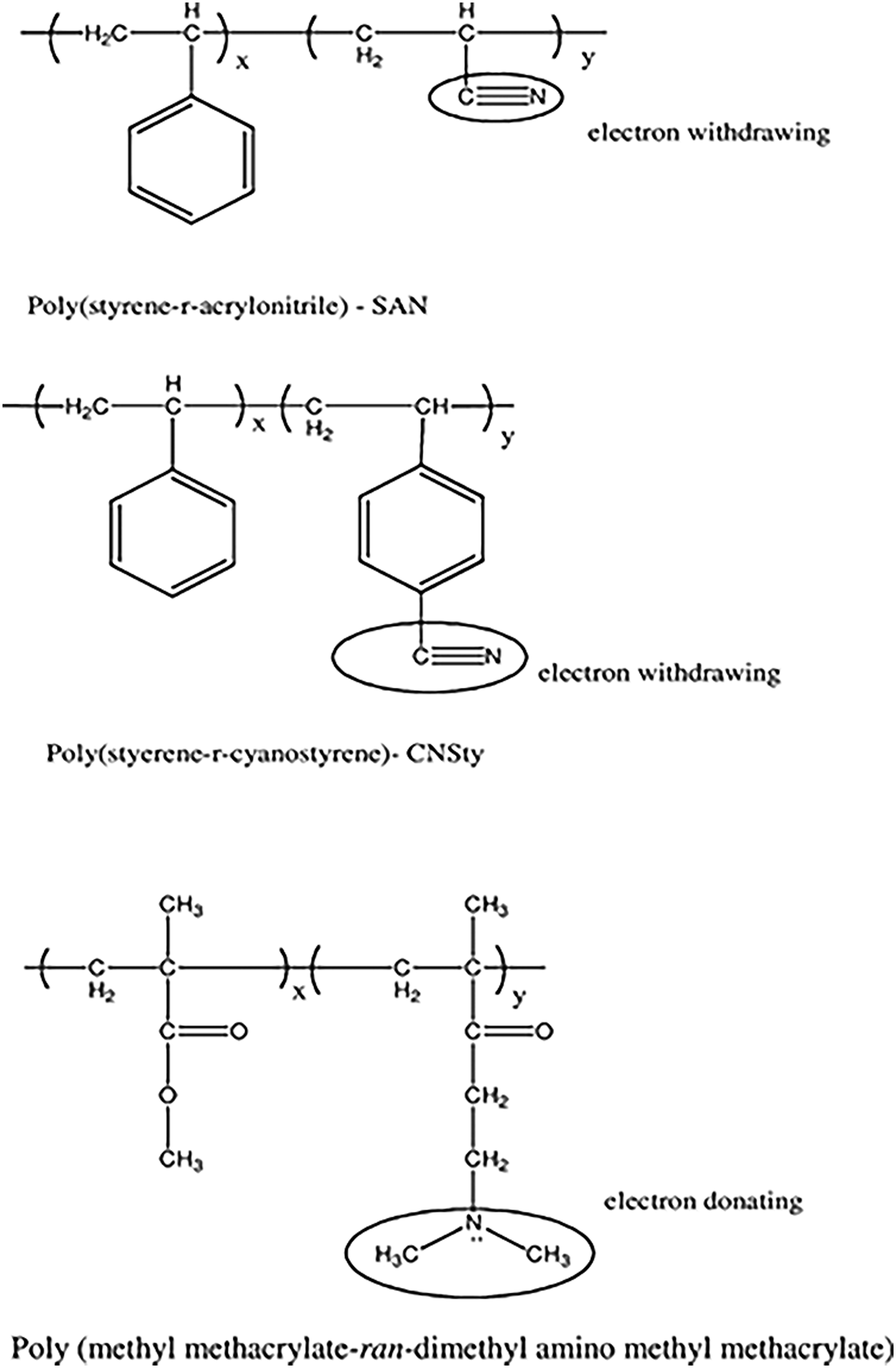
Kausar et al. 108 formed the solution blends of poly (vinyl butyral-co-vinyl alcohol-co-vinyl acetate)/polystyrene and filled with fullerene and graphene oxide based nanofiller. The resulting nanocomposites increased the tensile strength from 22.1 to 28.3 MPa with increasing loading level of 0.1–5 wt%. Furthermore, fullerene based nanoparticle addition caused positive effects on the electrical conductivity of the nanomaterials to enhance in the range of 1.8–2.5 Scm−1, showing the reinforcement effects, formation of matrix-nanofiller interactions, and percolation effects.
Viewpoints
Thermal, mechanical, electrical, physical properties of thermoplastic polymer/fullerene nanocomposites.

Controlled grafting of chains on the fullerene surface has been found to enhance the heat stability of the thermoplastic polymer/fullerene nanomaterials. As compared to physically interacted polystyrene/fullerene nanocomposite with maximum degradation temperature in the rage of 400 °C–420 °C, 107 the reported system on covalently grafted polystyrene/fullerene revealed higher maximum degradation temperature of about 550°C.116–118 Similarly, compared with the physically interacted polyurethane/fullerene or polyamide/fullerene nanocomposites,56,57,80,81 enhanced degradation temperature of 598°C, strength of 98 MPa, and Young’s modulus of 58 GPa have been observed for a reported covalently linked system. 62 Similarly, covalently interacted polymer/fullerene nanocomposites have been found effective for electrical conductivity enhancement of these system. 119 Despite of these facts, the physically interacted polymer/fullerene nanocomposites have been frequently developed and studied for potential applications.
Literature reports on the potential of these nanomaterials in wide ranging fields of energy (supercapacitors, solar cells, fuel cell, etc.), electronics (batteries, sensors, microelectronics), membranes, packages, textiles, space/automotives, and biomedical fields.120,121 To avoid the aggregation problems of the non-covalent filled nanocomposites, functional fullerene nanoparticles have been used.122–124 Consequently, the physical interactions govern the interfacial aspects, dispersion, and resulting features of the polymer-fullerene nanomaterials.125–127
Conclusions
This front-line review is undoubtedly a crucial effort focusing the polymer/fullerene nanocomposites having non-covalently linked nanostructure. Particularly, most of the polymer/fullerene nanocomposite research has been detected regarding the physically reinforced fullerene thermoplastics matrices, which are covered in this article. Accordingly, the non-covalently linked polymer/fullerene nanocomposites revealed exclusive microstructural and physical property profiles leading to the high performance nanocomposites. Briefly, this cutting-edge article summarizes critical characteristics of the fullerene reinforced thermoplastic matrix nanocomposites. In this concern, most widely used thermoplasts include polyamide, polyurethane, polystyrene, poly (methyl methacrylate), and block copolymers. Facile techniques have been used to incorporate the fullerene molecules in the matrices. The resulting properties of the nanomaterials depend upon the type of polymer used, nanofiller form, nanofiller dispersion, matrix-nanofiller interactions, synthesis technique, and other aspects. Synergistic effects between the matrix and nanoparticles have been found essential for better scattering and percolation properties of the nanomaterials. Consequently, electrical conductivity of such systems may be significantly high even >2 Scm−1. The glass transition temperature and degradation temperatures have also been detected high >200°C and >600°C, respectively. Non-covalent reinforcement of fullerene nanoparticles in thermoplastic matrices have also resulted in high tensile strength and young’s modulus values close to 100 MPa and >50 GPa, respectively. Subsequently, the high-tech physically linked thermoplastic polymer/fullerene nanocomposites have found important advantages for vital industrial sectors like aerospace, automotive, energy storage and production, and electronics devices.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
