Abstract
Ulexite and colemanite filled acrylonitrile-butadiene-styrene parts are manufactured by injection molding method. Tensile and wear tests are applied to reveal specific properties of composite parts. Various characterization methods are used to confirm the filler-matrix interactions, polymer melt flow, friction mechanisms, and fracture modes. This study investigated the use of ulexite and colemanite as fillers in acrylonitrile-butadiene-styrene composite parts, focusing on their impact on mechanical and frictional properties. Results showed that the addition of ulexite and colemanite fillers significantly improved mechanical properties such as compared to pure ABS, however ulexite filler showed much better performance compared to colemanite. In terms of wear test, ulexite filled ABS specimen showed a smooth wear while pure and colemanite filled ABS provided severe wear characteristics. These findings have implications for the development of high-performance composite materials for use in industries such as automotive and aerospace.

Introduction
Acrylonitrile butadiene styrene (ABS) is a versatile polymer that is utilized in varios industries due to its preferable properties.1–6 Its toughness, rigidity, and heat and chemical resistancy make it an ideal material for a variety of applications such as automotive parts, electronic housings, drainage pipes, and construction materials.7–10 The automotive industry uses ABS to manufacture various interior and exterior car components, while the construction industry uses it to make lightweight, corrosion-resistant pipes and fittings.11–13 In the electronics industry, ABS is used to create housings for electronic devices such as computers, printers, and televisions.14–16 Additionally, ABS is used in the production of toys, luggage, and sports equipment due to its durability and impact resistance.17,18 Other than traditional production methods, ABS is suitable for additive manufacturing for a variety of applications.5,19–26 Overall, the use of ABS in industry is widespread and continues to grow due to its unique combination of mechanical and thermal properties.
Despite the fact that the ABS has many desirable properties, there are also some disadvantages about its usage in the industry. One of the main drawbacks is that ABS is not biodegradable and can take a long time to decompose in landfills, leading to environmental concerns. 27 Additionally, ABS is prone to warping and shrinking during manufacturing processes, which can result in the deformation of final products. 28 ABS is also not suitable for high-temperature applications because it can degrade and release toxic fumes29,30. Its limitations should be carefully considered when selecting a material for a specific application.
Besides pure ABS, it is reinforced with numerous filler materials such as carbon nanotube, 31 glass, 32 carbon black, 33 and bamboo fiber. 34 Maleki and Zolfaghari 31 investigated electrical and mechanical properties of conductive polymer composites (CPCs), utilizing 3D printing with multi-wall carbon nanotubes (MWCNTs) and ABS. The resulting nanocomposites exhibit impressive electrical conductivity (26 times greater than ABS), heightened electromagnetic interference shielding, and improved tensile strength and modulus. Microwave heat treatment refines structural properties by minimizing void spaces. Despite nozzle wear from CNT abrasion, this research underscores the potential of 3D-printed CPCs for applications demanding lightweight structures with both electrical and heat conductivity. Liang 32 studied the effects of hollo glass particles in the ABS matrix and the article investigated the impact of three types of hollow glass beads (HGB1, HGB2, and HGB3) with different sizes and distributions on the mechanical properties of acrylonitrile ABS composites. Young's modulus showed a linear increase with filler volume fraction, while tensile yield stress and stress at break decreased slightly with increasing filler volume. Impact strength decreased rapidly at low filler volume fractions, while flexural strength increased initially and then declined. Generally, filler size and distribution have limited influence on these mechanical properties. Shenavar et al. 33 investigated to enhance the thermal stability of ABS to preserve color and gloss by adding carbon black (CB). The melt flow index (MFI) indicated that smaller CB particles have a more significant impact on decreasing MFI compared to larger particles. Scanning electron microscopy revealed a semi-brittle fracture surface in impact tests with added CB, while the Young's modulus of ABS is increased, and the yield stress decreased with higher CB loading, suggesting potential for improving ABS mechanical properties. Singh et al. 34 studied that the rising interest in environmentally friendly natural fiber-reinforced polymer composites. It comprehensively examined their applications, discussing various surface treatment methods and their effects on properties. Bamboo fibers blended with recycled ABS at 190°C exhibit higher tensile and flexural properties than recycled ABS alone. While good adhesion enhanced tensile properties, the presence of bamboo fibers acts as stress concentrators, reducing impact strength. SEM, FTIR, and XRD analyses confirmed improved adhesion, crystallinity, and composite properties. Besides coventional fillers, borate compound fillers are utilized for various goals such as flame retardancy,35,36 mechanical properties and combustion resistance, 37 flammability, 38 tensile and fire residue properties. 39
Borate minerals are a group of minerals that contain boron and oxygen, often along with other elements such as sodium, calcium, and magnesium.40–42 These minerals are widely distributed and can be found in many geological environments, including desert salt flats, volcanic deposits, and marine evaporites. 43 Some of the most common borate minerals include borax, ulexite, and colemanite.44,45 Borate minerals have a range of industrial uses, including as a source of boron for the manufacture of fiberglass, ceramics, and detergents.46,47 They are also utilized as a flame retardant in polymers and as a fertilizer in agriculture. 48 Borate minerals are primarily important and versatile group of minerals with a wide range of applications in the industry.
Borate minerals can have beneficial effects when added to acrylonitrile butadiene styrene (ABS) as a flame retardant. 49 Borate minerals such as borax and colemanite can react with the polymer matrix of ABS during combustion, releasing water molecules that cool the material and prevent the spread of flames. 50 Borate minerals have gained significant attention as effective mechanical reinforcement agents in polymer composites.51–54 The incorporation of borate minerals in polymer matrices improves their mechanical properties such as stiffness, strength, and toughness due to their unique crystal structure and chemical composition.55,56 Borates can form strong interactions with the polymer chains, resulting in improved interfacial adhesion and enhanced mechanical properties of the composite material. 57 Several studies have reported the beneficial effect of borate minerals on the mechanical properties of polymer composites. Guzel et al. 54 studied the separate involvement of ulexite and colemanite in the epoxy resin in terms of thermal and mechanical properties. Effect of filler amount, hardener type, and plasticizer on mechanical properties of composite resin is investigated and it is reported that an effective distribution of ulexite and colemanite is achieved. The composites showed enhanced tensile properties up to a 5 wt% filler content, while water sorption was significantly reduced with increasing filler content as compared to the neat epoxy. Furthermore, all composites demonstrated exceptional corrosion resistance and strong adhesion properties. Sugozu et al. 53 investigated the tribological properties of ulexite filled break lining materials for testing of newly formulated brake pads. The application of heat treatment resulted in a more uniform structure, leading to a reduction in micro-structural variations during brake action. The inclusion of ulexite as an additive had a positive impact on the braking performance. This led to a stable and increased coefficient of friction in the samples containing ulexite. The experimental results indicate that the overall performance was significantly improved using ulexite in the friction layer. Yanen et al. 52 studied the production of a composite material by adding colemanite, ulexite, and tincal ore to unsaturated polyester. The influence of these fillers in various mass ratios is investigated on density, hardness, thermal conductivity, and thermal stability. The findings showed that as the mass ratio of boron components in the mixture increased, density, hardness, activation energy, and thermal conductivity coefficient of the composite material also increased. The activation energies of the polyester composite are ranked in the order of colemanite, ulexite, and tincal ore from highest to lowest. Besides mechanical and structural properties, flame retardancy of boron minerals are thoroughly studied by Dogan et al. 58 and it is reported that boron compounds are highly potential flame-retardant additives in polymer-based materials because of their low toxicity, and molecular diversity.
Borate minerals are mostly utilized for fire resistance and a comprehensive investigation is not made in terms of mechanical and tribological properties for borate mineral filled ABS matrix. Various characterization methods are used to evaluate the test results and effect of ulexite and colemanite fillers on mechanical and tribological properties of ABS composites are investigated. The aim of this study is to reveal the relationship of mechanical and frictional properties of ulexite and colemanite filled ABS parts. This study's findings could pave the way for the development of advanced materials with improved mechanical and frictional properties, which could have significant practical applications.
Materials and method
Borate minerals
Classification of various borate minerals.

Extrusion and injection molding of matrix and reinforcement
ABS is selected as matrix material and reinforcement is separately provided by borate minerals of colemanite and ulexite. Borate mineral powders and ABS granules are brought to similar size for a better composite bonding process. Colemanite and ulexite powders are separately mixed with ABS granules in a shaker, and the powder mixture is extruded through the double and single screw extruder to form chemical bonding between borate mineral and ABS. Extruded composite filaments are used for the production of test specimens. At last, test specimens are fabricated and the composition of specimens are 5 wt% ulexite filled ABS (U5), 5 wt% colemanite filled ABS (C5), and pure ABS.
Test methods and characterization
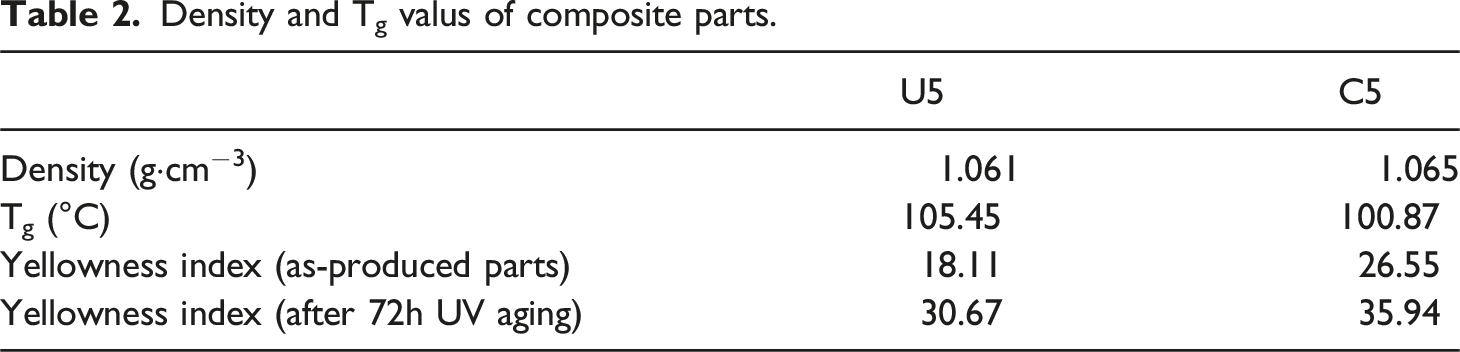
Density and Tg valus of composite parts.
Results and discussion
Density measurement is made and results clearly showed that density is almost the same for ulexite and colemanite filled ABS parts. This can be attributed to the similar chemical structure and homogenous distribution of filler material. Yellowness is another important parameter for polymers and it is revealed that colemanite filled ABS parts are weaker to UV light compared to ulexite filled ABS. Ulexite seems to be better filler in terms of UV resistance. In terms of Tg, ulexite filled ABS resists against heat more in comparison with colemanite. Dominance of ulexite over colemanite can be associated with that colemanite is a secondary product after a reaction of borax and ulexite.
63
Elemental analysis of composite parts are made with EDS detector and results are revealed in Figure 1. Elements related in borate and boric acid compounds such as boron, oxygen, calcium, and sodium are detected in the filler material of ABS matrix. Due to polymer chains, concentration of carbon and oxygen is higher compared to other borate related elements. EDS spots of (a) C5 and (b) U5 specimens and (c) elemental composition of spots.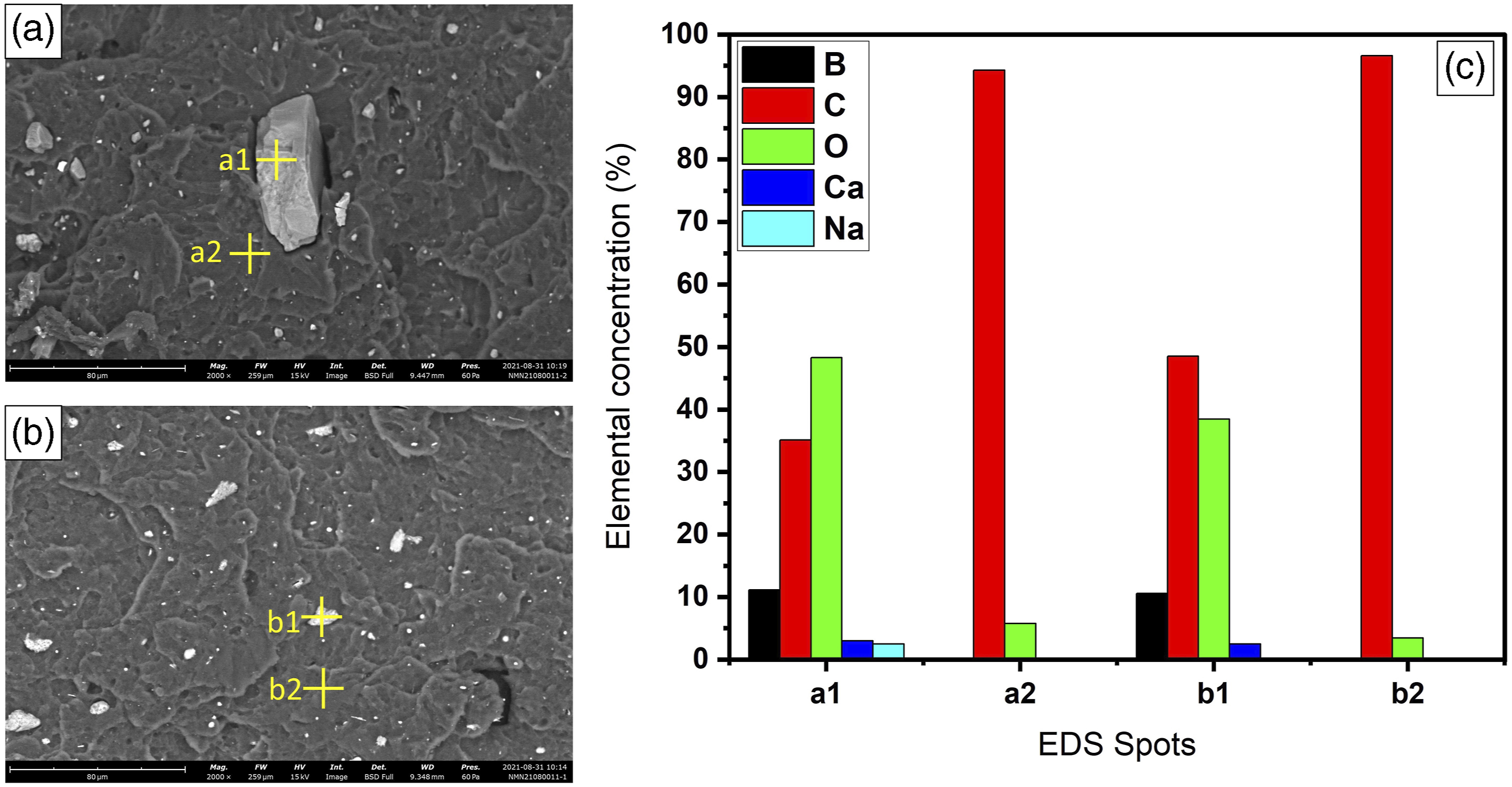
Melt flow analysis
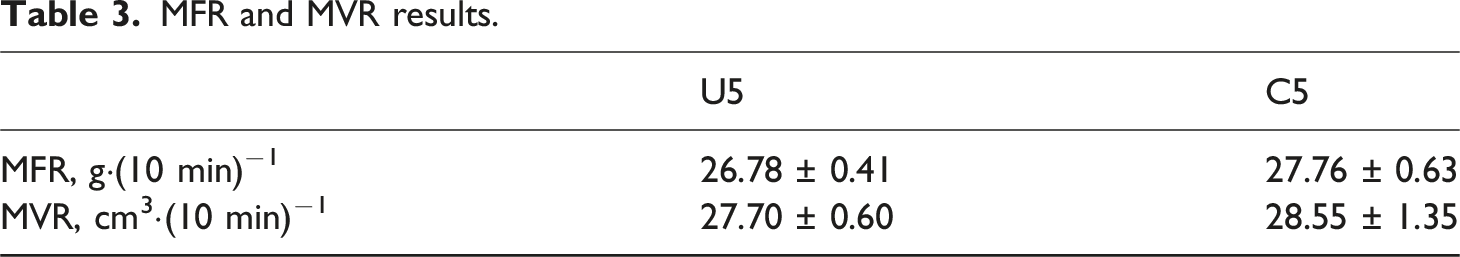
MFR and MVR results.
SEM micrographs for tensile tests
Fracture surfaces of pure and composite ABS parts exhibit significant differences in their morphology and appearance as in Figure 2. In pure ABS (Figure 2(a)), the fracture surface typically showed a relatively smooth and uniform appearance, with very little evidence of distinct features or heterogeneity. This is because the fracture occurs primarily through the breaking of covalent bonds within the polymer chains, resulting in a cohesive failure that tends to produce a relatively clean fracture surface. One significant difference in the fracture surface of these two materials is the presence of ulexite and colemanite particles in the composite polymer. Figure 2(b) and 2(c) exhibited the fracture surfaces of U5 and C5 composite polymers which is observed to be rough and more irregular surface compared to a pure ABS (Figure 2(a)). The boron particles are partly visible on the fracture surface as pulled-out or broken, depending on the mode of failure. Another difference is the presence of large voids or inclusions are visible on the fracture surface. Smaller filler particles seems to have better interfacial adhesion compared to larger particles. Surface morphology and roughness of smaller particles enabled better adhesion as a result of mechanical interlocking. Smooth surface of larger particles and brittle structure of colemanite and ulexite worsened the interlocking mechanism and a poor endurance is observed.65,66 Larger particles are assumed that they are involved in the crack initiating elements in the matrix.
67
In contrast, voids or inclusions are not observed in the pure ABS as in Figure 2(a) which also supports the fact that a successful plastic injection process occurred. Furthermore, the fracture surface of composite parts show evidence of delamination or debonding between boron particles and the polymer matrix due to poor adhesion strength at the boundaries. SEM micrographs of fracture surfaces after tensile test; (a) pure ABS, (b) U5, (c) C5.
Tensile and impact Strength Test results
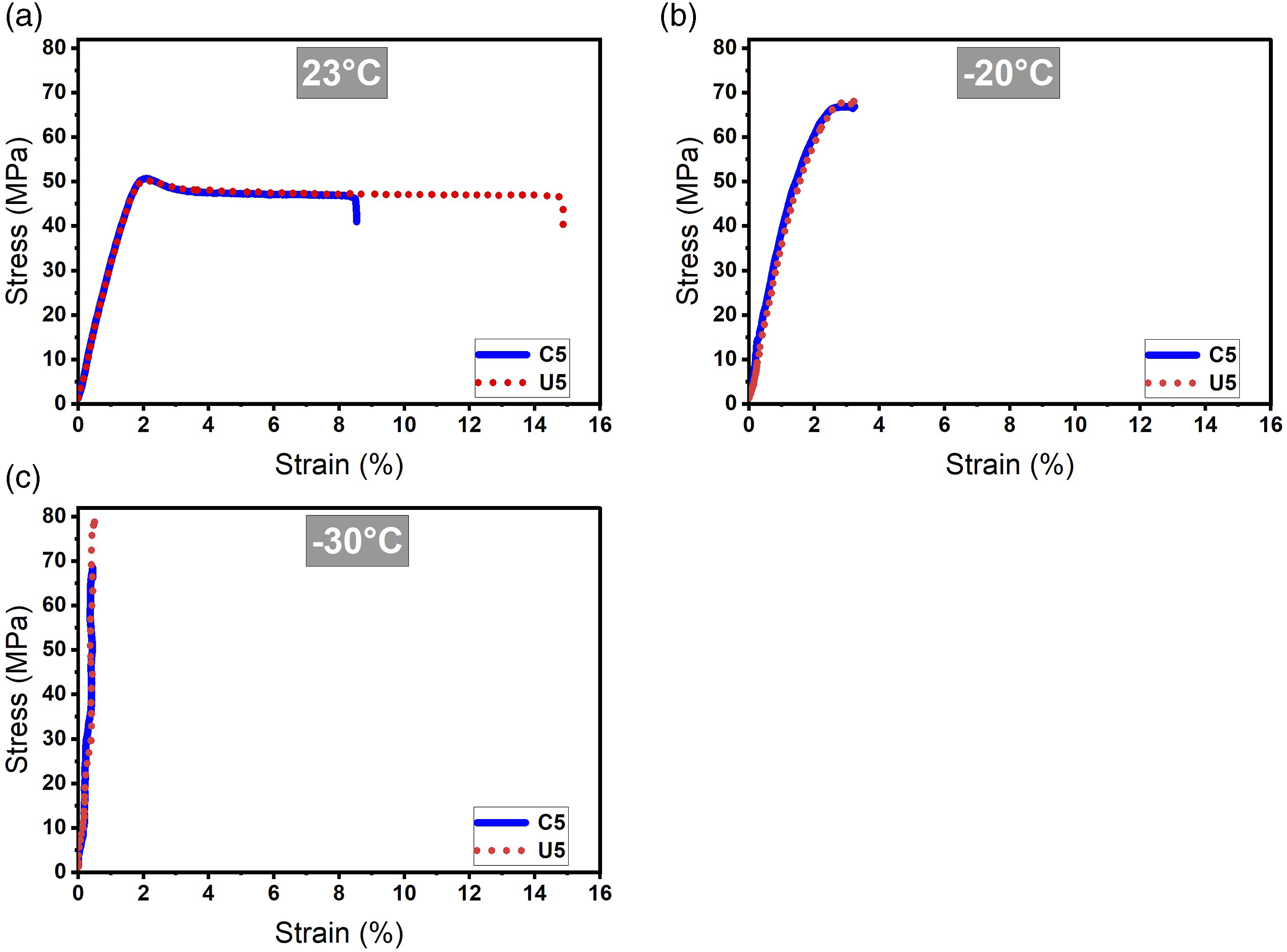
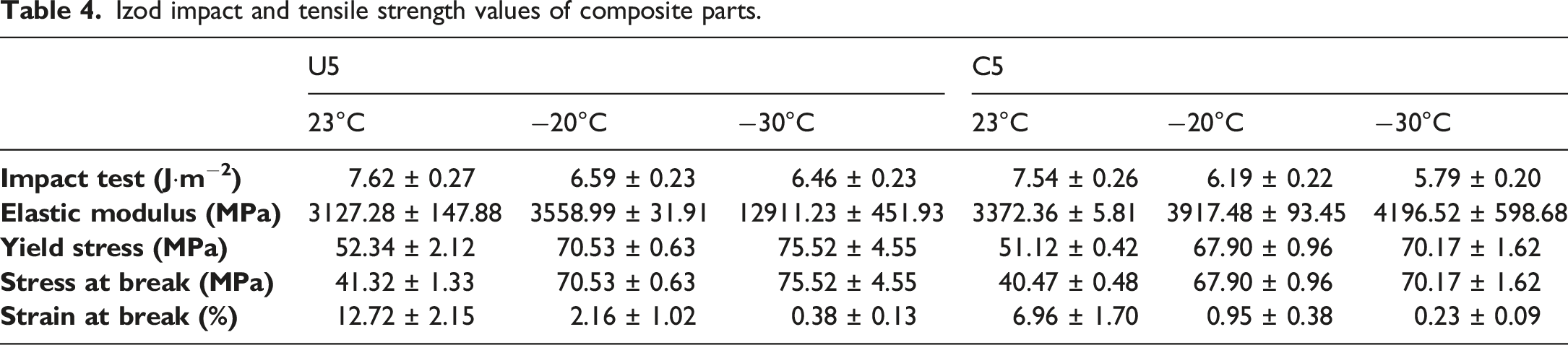
Impact and tensile strength tests are applied to reveal mechanical properties of composite parts and results are illustrated in Figures 3 and 4 and Table 4. In terms of impact strength energy values, ulexite filler provided slightly better impact energy absorption for all test temperatures compared to colemanite filler. As the test temperature decreses, energy absorption capacity of both fillers proportionally reduced which can be related to increasing of brittleness at lower temperatures. Besides, long chains of ABS matrix are damaged in the cross-linking process in the extruder by the effect of temperature, pressure, and hard boron particles. Ulexite has superiority over colemanite filler by 1.06%, 6.46%, and 11.57% in terms of impact absorption energy for test temperatures of 23°C, −20°C, −30°C, respectively. Additionaly, impact absorption energy decreases when the test temperature is lowered. It is calculated that impact absorption energy is lower by 13.52% and 17.60% for test temperatures of −20°C and −30°C, respectively, in comparison with impact absorption energy at 23°C for ulexite filler; and for colemanite filler, reduction of impact absorption energy is by 21.81% and 30.22% for test temperatures of −20°C and −30°C compared to test temperature of 23°C. In terms of tensile test results, ulexite has higher tensile strength in terms of yield stress, stress at break, and strain at break which can be associated with the flexibility of ulexite
54
compared to colemanite. The flexible structure of ulexite enables higher tensile stress values by increasing resilience of polymer chains. Ulexite filled ABS parts have superiority over colemanite filled ABS parts by 2.39%, 3.87%, and 7.62% for 20°C, −20°C, and −30°C test temperatures in terms of yield stress, respectively. Stress at break values of ulexite filled ABS are also higher than colemanite filler by 2.10%, 3.87%, 7.62% for 20°C, −20°C, and −30°C test temperatures, respectively. In addition, strain rates of ulexite filled ABS parts are higher than colemanite filler by 82.76%, 127.37%, and 65.22% for 20°C, −20°C, and −30°C test temperatures, respectively. The superiority of ulexite in strain at break values can be related to water molecules in the boron-oxygen crystal structure which enables remarkable flexibility. In terms of colemanite, it also includes water molecules in the crystal structure, not as much ulexite, and its flexibility relies on sliding of the layers due to sheets of borate groups.54,68 Lowering test temperature has a developing effect on tensile strength, except strain at break, which can be attributed to improved hardness and toughness due to test temperatures below zero.
69
The effect of temperature decrease on the toughness and hardness of composites depends on the specific composite material and its composition. A decrease in temperature led to a reduction in the toughness of composites. This is because colder temperatures cause the polymer matrix to become more brittle, which can lead to more rapid crack propagation and failure. Ulexite and colemanite fillers in the polymer matrix become more brittle at lower temperatures, further reducing the toughness of the material. On the other hand, a decrease in temperature improved the hardness of composites. This is because colder temperatures cause the polymer matrix to become more rigid, which results in a harder and more resistant structure. However, this increase in hardness is accompanied by a decrease in ductility and toughness. Impact energy results of specimens after Izod impact strength tests. Tensile results of specimens. Izod impact and tensile strength values of composite parts.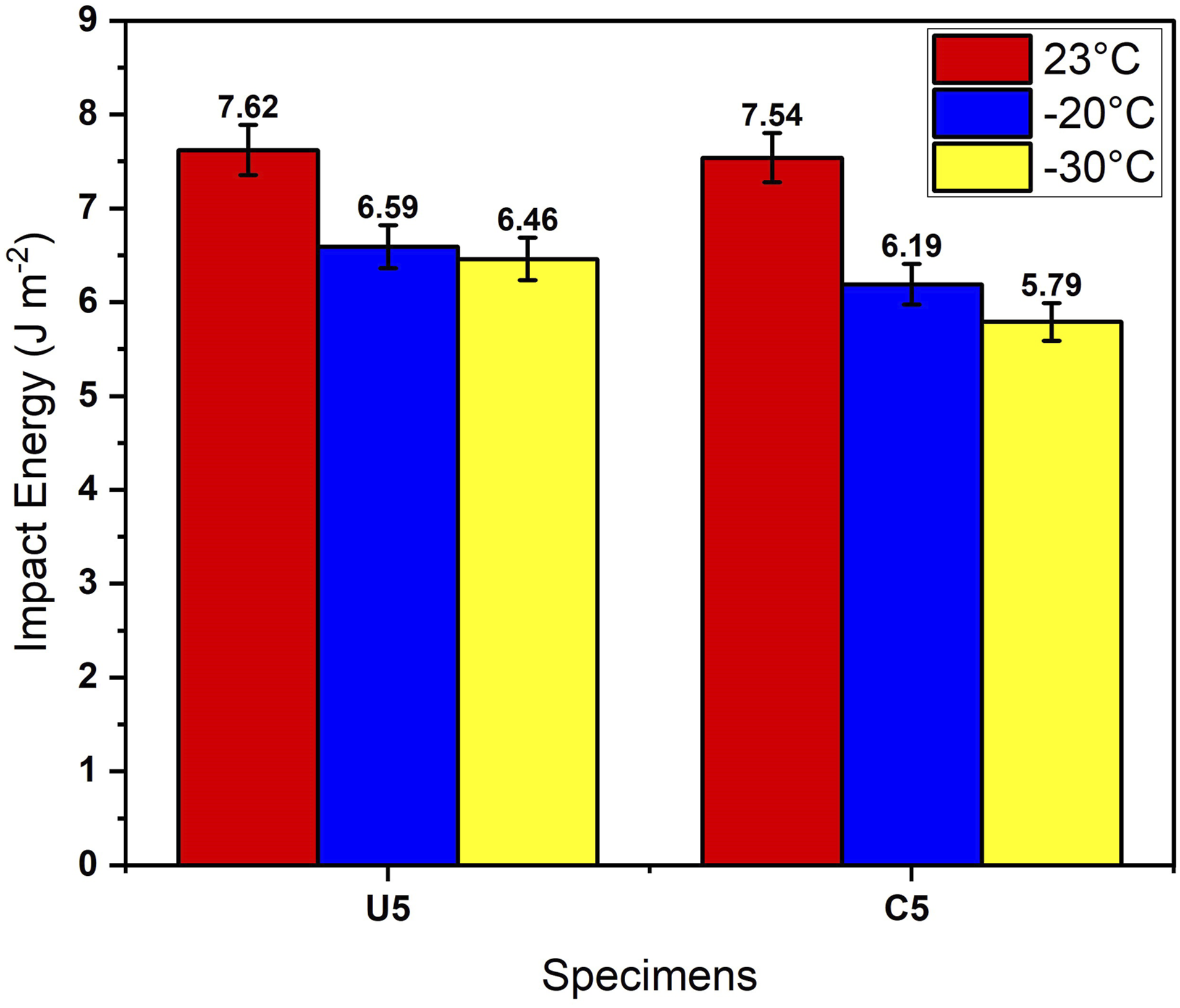
SEM micrographs for wear tests
Specimens are subjected to wear test under an applied load of 5 N and the worn surfaces of specimens are illustrated in Figure 5. As it is seen in Figure 5(a), a smootly abraded surface is observed for pure ABS specimen which is expected under the mild load of 5 N. Plastic deformation occurred in the pathway of abrasive ball due to the polymer debris which clinged to the abrasive ball. As a result of repetitive movement of abrasive ball, debris is distributed and cold-welded over the surface. In terms of colemanite filled ABS specimen (Figure 5(b)), brittle structure of colemanite particles are broken or pulled out due to frictional force of abrasive ball. Micro voids are developed owing to wear mechanisms of pull-out and particle breaking. It can be considered that low strength bonding between borate particles and polymer caused aforementioned mechanisms, however brittle structure of colemanite and frictional forces is responsible for voids and broken particles. Figure 5(c) illustrated the worn surface of ulexite filled ABS part in which smooth groove marks are observed. This can be attributed to the flexible structure of ulexite, compared to colemanite, resisted against frictional forces. Furthermore, abrasive ball related plastic deformation and broken ulexite particles which adhered to the abrasive ball performed groove marks. SEM micrographs of wear tracks after wear tests for (a) pure ABS, (b) C5, and (c) U5 specimens
Composite parts are worn under an applied load of 10 N and microscopic surface examinations are depicted in Figure 6. As it is seen in Figure 6(a), the wear surface of pure ABS deformed more compared to wear test under 5 N which can related to increased damage capacity and frictional heat of abrasive ball. Delaminations around the edges of plastic deformation zones are observed which are probably occurred owing to stick slip phenomenon70,71 which can be classified as partial-slip.
72
Wear behavior of colemanite filled ABS part can be examined in Figure 6(b). Dominant wear mechanism seems to be abrasive wear and increased applied load abraded polymer and colemanite partices. Further, colamanite particles are pulled out due to loosened interaction of polymer and colemanite particle which is the result of severe periodic movement of abrasive ball. Spalling mechanism also occurred over the wear surface which can be related with micro-cracks while pulling out of colemanite particles. Figure 6(c) depicted microscopic examination illustration of the wear track of ulexite filled ABS part. As in 5 N test, ulexite as filler resisted against wear much more compared to pure ABS and colemanite filled ABS under the applied load of 10 N. In other studies, lubricant effect of sodium-related chemical compounds is investigated for lowering the influence of frictional forces,73,74 accordingly, partial and smooth plastic deformations can be related with lubricating effect of sodium in the chemical composition of ulexite.
75
Besides smooth plastic deformation, debris formed smooth grooves in the wear surface. SEM micrographs of wear tracks after wear tests for (a) pure ABS, (b) C5, and (c) U5 specimens under 10 N applied load.
Wear tests are accomplished under an applied load of 15 N for composite specimens and results are depicted in Figure 7. Pure ABS and colemanite filled ABS specimens are both showed nearly similar wear behavior when the applied load is increased to 15 N. In both specimens, deformation zones occurred, however deformation is more severe for colemanite filled ABS compared to pure ABS. It can be affiliated with broken and grinded colemanite particles which developed a harsh abrasion between the wear surface and abrasive ball, that is why deformation zones are formed over the surface. Ulexite filled ABS specimen resisted against wear in a strong way that lubrication, owing to sodium compund in the chemical composition of ulexite, decreased the severity of frictional forces regarding the applied load of 15 N. SEM micrographs of wear tracks after wear tests for (a) pure ABS, (b) C5, and c) U5 specimens under 15 N applied load.
After examining the wear surfaces of pure, colemanite filled, and ulexite filled ABS specimens that as the applied load increases, colemanite filled ABS specimens tends to show a wear characteristics similar to pure ABS as a result of hard and brittle structure of colemanite. Besides this, flexible structure of ulexite and sodium compound in its chemical composition revealed a much smoother wear behavior compared to colemanite filled ABS. Increased work pace impaired the filler materials property for colemanite by showing a similar wear mechanism as pure ABS. In harsher working conditions, ulexite seems to be a preferred filler in terms wear mechanisms and characteristics.
Wear rate values
Specimens are subjected to wear test and wear tracks are measured in the SEM and results are illustrated in Figure 8. As it is examined in the SEM micrographs in Figures 5–7, the least to the most wear resistant specimens as it follows: pure ABS, colemanite filled ABS, and ulexite filled ABS. As the applied load is increased, wear rate of pure ABS specimen is also increased by 81.33% and 162.56% for applied loads of 10 N and 15 N compared to 5 N. As the wear resistance drops by increasing applied load, wear chracteristics stayed same for the pure ABS specimens as it is seen in Figures 5–7. Among test specimens, colemanite filler showed a moderate wear resistance in the wear tests in which as the appliead load increased, wear rate is also increased by 28.94% and 80.05% for applied loads of 10 N and 15 N compared to 5 N. The wear rate values of pure ABS at 5 N and colemanite filled ABS at 15 N applied load showed almost similar wear resistance in the tests. This can be affiliated with that brittle structure of colemanite becomes weaker as the severity of wear test increases. Ulexite filled ABS parts seem to be the most wear resistant specimen among others. As the applied load increases, wear rate dropped by 28.56% and 41.79% for applied loads of 10 N and 15 N compared to 5 N. In contrast to other specimens, wear resistance of ulexite filled ABS parts increased as the applied load increased which can be related with flexible structure of ulexite and lubrication effect of sodium compounds in the chemical structure of ulexite. Ulexite seems to be a proper candidate for harsh and severe wear environments. Wear rate values of pure ABS, C5, and U5 specimens.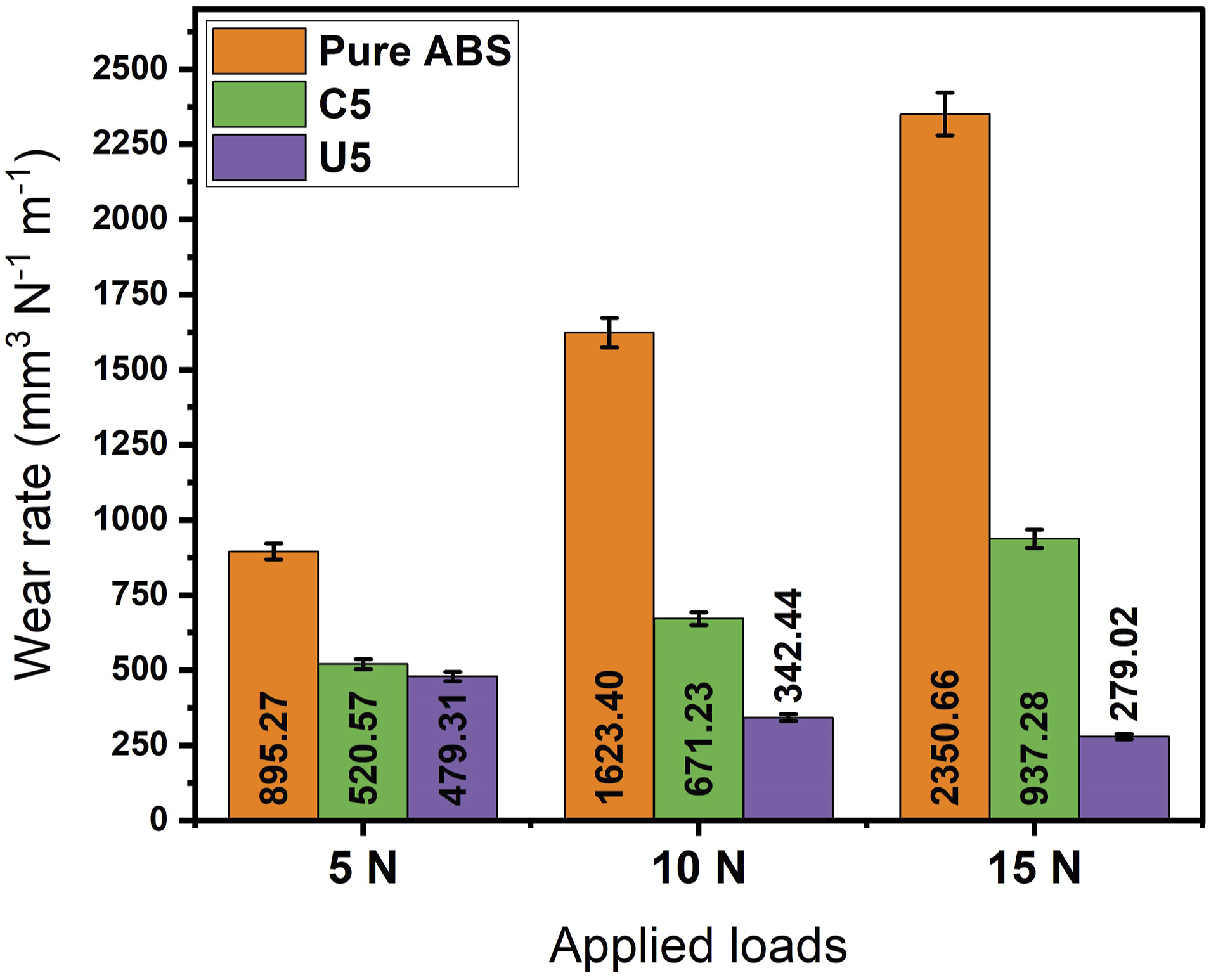
Coefficient of friction values
COF values are recorded in the wear tests and results are illustrated in Figure 9. As it is seen, tests reached a stable regime after a sliding path of 30 m. In the steady friction condition, colemanite filled ABS specimens had higher COF values due to its hard and brittle structure. It is expected that pure ABS might have the lowest COF values in accordance with absence of filler material. However, debris adheres to the abrasive ball and it eagers to stick to the wear surface which increases severity during wear test. Besides, ulexite filled ABS specimen yielded the smoothest COF regime in the wear test which can be based upon flexible structure of ulexite and lubrication effect of sodium in the chemical composition of ulexite. Considering the fact that ulexite filled ABS had the lowest wear rate value, it also provides the smoothest working condition among other specimens which makes it a proper composite material to operate in severe working conditions. COF values of pure ABS, U5, and C5 specimens under applied loads of (a) 5 N, (b) 10 N, (c) 15 N.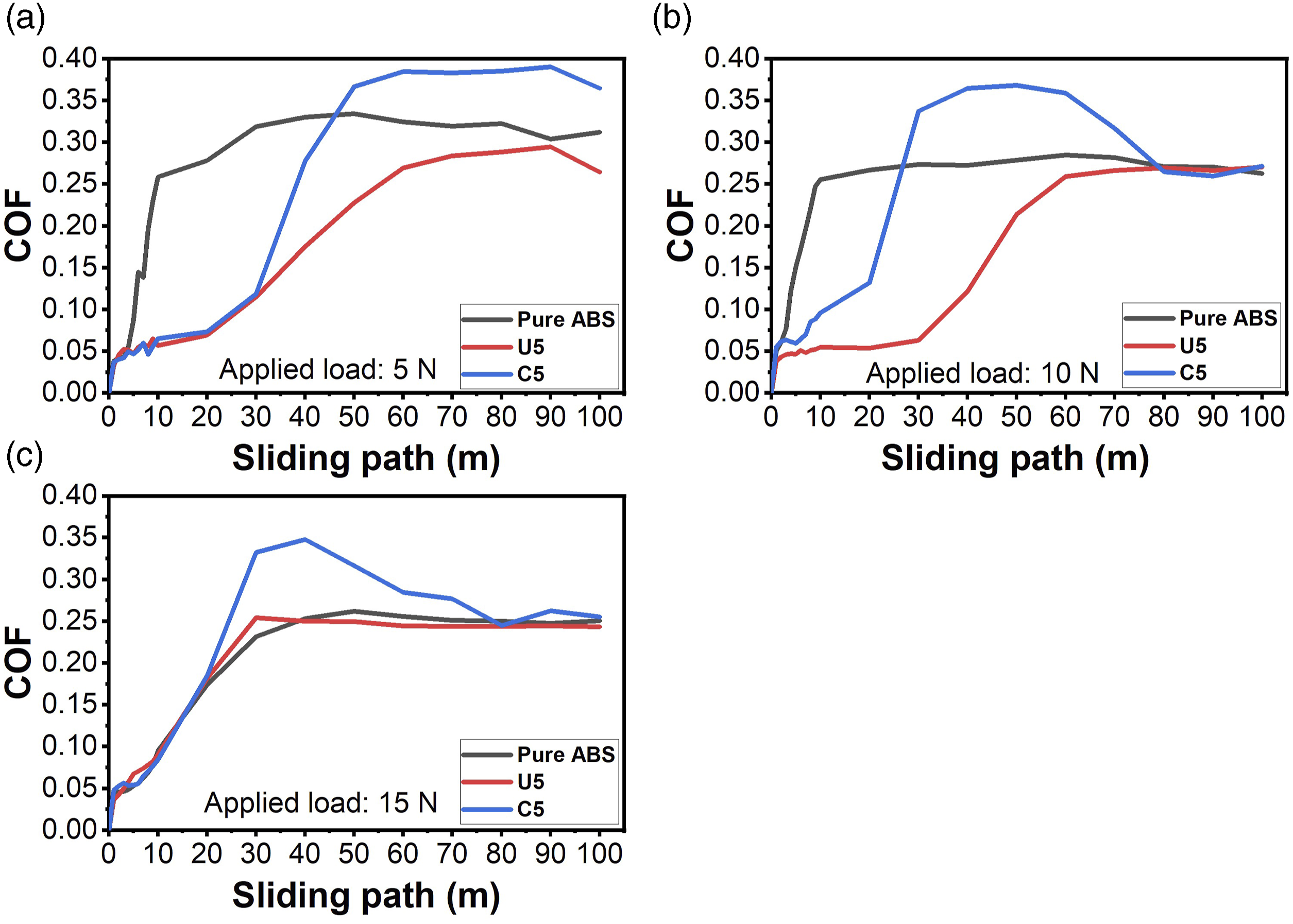
Conclusion
ABS material is reinforced with ulexite and colemanite particles and test specimens are fabricated by injection molding method. Filler ratio is determined as 5 wt% and a comparison is made with pure ABS specimens. After tests and microscopic characterization, the following outcomes are obtained: • Impact and tensile tests are applied at different temperatures to reveal filler materials behavior. It is showed that ulexite is stronger and better in terms of yield stress, stress at break, and strain at break for all test temperatures which is attributed to flexible structure of ulexite. Brittle and hard structure of colemanite dropped tensile properties of colemanite filled ABS for all test temperatures. In terms of impact test, ulexite also had much better impact absorption energy capacity for all test temperatures compared to colemanite. • Fracture surface of all test specimens are observed in SEM to determine crack behavior. It is showed that pure ABS specimen yielded a smooth fracture surface due to breaking of covalent bonds within the polymer chains while ulexite and colemanite filled specimens provided rough and irregular fracture surfaces as expected. It is related to poor adhesion strength of boundaries and acted as defect inititation zone. Applied force in the tensile test damaged bonds of borate mineral and polymer so that borate particles are broken or pulled out. • Wear surfaces are characterized using SEM and following results are revealed that as the applied load increases pure ABS and colemanite filled ABS specimens tend to show poor wear resistance while ulexite filled ABS specimens’ wear resistance is increased proportional with applied load. It is considered that providing smooth wear characteristics is a result of lubrication effect of sodium compounds in the chemical composition of ulexite. • COF values showed that the smoothest regime is obtained with ulexite filled ABS specimen which has the capacity to provide smooth operation condition even in severe working environments. Colemanite filled ABS had a much more fluctuant compared to pure and ulexite filled ABS specimens which can be related with hard and brittle colemanite particles are broken and pulled out from the surface due to friction. Colemanite debris provoked stick slip phenomenon which also occurred slightly for pure ABS specimens as a result of ABS debris. It is important to state that flexible ulexite particles restrained stick slip phenomenon by getting contact with ABS debris and got away from the wear surface.
Footnotes
Acknowledgements
Authors thank to Eurotec Engineering Plastics for the support made in the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
