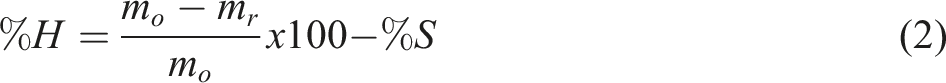Abstract
For their use in polymer and composite materials engineering, and with a view to sustainable development, a great deal of work has been carried out with the aim of describing the properties of plant fibers. This work presents the effects of the extraction method on the chemical, physical and mechanical properties of oil palm petiole fibers (OPPF). To this end, the FPPH to be analyzed were obtained by water retting and chemical retting with soda (5% solution). Their physico-chemical and mechanical characteristics were then studied and compared. Fourier transform infrared spectroscopy (FTIR) analysis confirmed the existence of the main components of lignocellulosic fiber (lignin, cellulose and hemicellulose). Lignin was reduced by 22.74%, hemicellulose by 27.33% and cellulose by 16.87% compared to water retting. XRD revealed a crystallinity index of 58.66% for fibers extracted with a 5% sodium solution and 58.51% for fibers extracted with water retting. Soda retting reduces density by 3%. Tensile tests revealed an average tensile strength of 156.082 MPa for water-rubbed fibers and 205.875 MPa for soda-rubbed fibers, Young’s modulus of 3.432 GPa for water-rubbed fibers and 5.346 GPa for soda-rubbed fibers. Elongation at break for FPPH extracted by water retting was 5.178% and 4.41% respectively. This shows that the extraction method affects mechanical properties (flexibility and stiffness) and should be chosen according to the desired characteristic.
Introduction
For several decades, the use of lignocellulosic fibers extracted from seeds, fruits, leaves, stems, pseudotrunks, lianas and roots as reinforcement in polymer engineering and composite materials has risen significantly.1–3 In addition, plant fiber-reinforced composite materials are increasingly employed in several domains such as transportation, construction, sports and entertainment industries, but also in art.4,5 Plant fibers stems are more and more used due to their low cost, abundance, but also because of their physical and mechanical characteristics which offer a diversity of uses and an alternative to synthetic fibers. Environmental protection and sustainable development through the reduction of carbon impact are elements that favor the use of plant fibers in industries because, unlike synthetic fibers, they are biodegradable.3,6–8 Agriculture being a field that contributes to atmospheric pollution, the valorization of agricultural waste as a reinforcement in polymer engineering and composite materials is of significant benefit to the environment because in most cases, agricultural waste is incinerated. This incineration contributes up to 18% of greenhouse gas emissions.9–14 Agricultural residues, in the case of plants such as okra, wheat and barley straws, maize breaks, banana pseudostems, bagasse, pineapple leaves and oil palm petiole fibers (OPPF), have already been the subject of several studies.15–19 Oil palm is one of the most widespread crops in agriculture, and the least environmentally friendly (as it requires the felling of trees). Recovering the residues from this plant would help reduce the environmental impact of this crop. There are several varieties of palm and several studies have been carried out to characterize palm fibers. Among the varieties studied are the Washington variety, date palm and doum palm.20–25 Due to their natural character, the physical, mechanical and chemical properties of plant fibers vary from one fiber to another. This dispersion is linked to several factors, including the type of plant, the growing area, the growing conditions, the part of the plant from which the fiber is taken (root, stem, leaf, seed), and the extraction method. The extraction process has an impact on fiber properties.26–30 To achieve this, two extraction methods were used, namely water retting and cold soda chemical retting. Trials were carried out to determine fiber characteristics for each extraction method.
Materials and methods
Petiole fiber extraction protocol
The plant fibers used in our work are OPPFs. These petioles were collected in the central region from palms that had produced once (i.e., an age of around 5 years).
Extraction by retting
The main disadvantage of the traditional extraction method (water retting) is the time it takes for the cellulose to decompose.
31
To achieve this, we followed the following steps: 1. Cutting the oil palms; 2. Cut the palms (petioles) into 25 cm long pieces; 3. Separate the bamboo from the pith of each element; 4. Immerse each 25 cm-long pith element in tap water without additives for 3 weeks; 5. Manually remove and rinse the fibers with water.
Figure 1 summarizes the steps involved in extracting OPPF by water retting. Water retting steps: (a) Palm cut; (b) Petiole removed; (c) Petiole removed from bamboo; (d) Immersion in water.
Chemical retting
This process uses only NaOH soda to dissolve non-cellulosic subsistence such as lignin, pectin and hemicellulose, as well as the various constituents forming the reserve and outer wall of the plant stem.31,32 The temperature, pressure, concentration and duration of the treatment must be defined according to the batch, age and type of plant, so as not to degrade the cellulose fibers. Figure 2 shows the immersion of petioles in the soda solution; Figure 3 shows the physiognomy of the fibers obtained by each extraction method. Oil palm petiole immersed in a soda solution (NaOH à 1N). Fiber physical appearance: (a) Petiole fibers obtained by chemical retting with soda; (b) Petiole fibers obtained by water retting. Petiole fibers obtained by water retting.

Fiber characterization protocol
Chemical characterization of fibers
Fourier transform infrared spectroscopy
The principle of infrared (IR) spectroscopy is based on the absorption of light by most molecules in the IR region of the electromagnetic spectrum, and the conversion of this absorption into molecular vibration. Fourier transform infrared spectroscopy (FTIR) was used to characterize FPPH. Analyses were performed on the ALPHAP ”BRUKER” Fourier transform spectrophotometer. Infrared spectra were recorded by accumulating 32 scans with a wave number of 400 up to 4000 cm−1. A small quantity of the mixture in the micrometer range was deposited on a previously cleaned diamond crystalline surface. These analyses were carried out at the Analytical Chemistry Laboratory of the University of Yaoundé 1.
Determination of fiber chemical component percentages
To determine the percentages of chemical elements in petiole fibers, we used the following methods and equipment:
Lignin content
Fiber samples were dried in a ventilated oven at 105°C for 24 h; then a mass of (2 ± 0.01) g was introduced into an Erlenmeyer flask, then 30 mL of a 72% 𝐻2𝑆𝑂4 solution was gradually added using a burette, while macerating the fibers with a glass rod; the Erlenmeyer being kept in a thermostatic bath at (20 ± 1) °C during material dispersion, then covered for 2 h. The substrate is then transferred to a flask containing approximately 600 mL of water. To achieve a 𝐻2𝑆𝑂4 concentration of 3%. The flask was then placed on a reflux unit and heated without magnetic stirring; after settling, the lignin was transferred to the filtration unit and washed with hot water. Finally, the residue was dried in a ventilated oven at 105°C for 12 h. The lignin content (%Li) was obtained from formula (1).
Hemicellulose content
Samples are first dried in a ventilated oven, then weighed on a laboratory balance. Sample mass 𝑚0 is introduced into a 500 mL flask, to which 150 mL of sulfuric acid solution is added. The assembly is mounted on a reflux system, so that the acid hydrolyzes the hemicelluloses contained in the sawdust. At the end of the process, the solution is poured into a Büchner assembly and washed with lukewarm water. The residue obtained is dried for 24 h at 105°C, then weighed on a 0.001 mg precision balance. The hemicellulose content (%H) is obtained using formula (2).
The cellulose content (%C) in the fibers is obtained using formula (3).
Water-soluble sugar content
The fibers are first washed with a toluene/Ethanol mixture in a soxhlet, then dried in an oven. The massed 𝑚𝑂 is taken and inserted into a 250 mL flask, then mounted on a reflux bench. After dissolving the sugars, the solution is filtered and washed hot on a Büchner assembly fitted with a pump to recover all particles. The entire residue is removed, placed on a watch glass and dried in a ventilated oven. The residue is weighed and the mass 𝑚s obtained. The water-soluble sugar content (%S) is determined from formula (4).
Physical characterization of fibers
Determination of external fiber diameters
This test involves determining the average diameter of fibers extracted by retting with water and a 1N sodium hydroxide solution, by measuring the diameter at three regularly spaced points using a dial thickness gauge. The fibers are previously dried and then conditioned at 65% relative humidity in a hygroscopic tank, for a period of 24 h. The average diameter is calculated using formula (5).
Determination of bulk density
The aim of the test is to determine the absolute density of OPPFs. For this purpose, three weighings are carried out: the mass (m0 = 1 ± 0.01 g of the anhydrous sample); the mass m1 of the pycnometer filled with ethanol; the mass m2 of the pycnometer containing the fiber + ethanol mixture. The density is given by formula (6) shows the experimental setup.
ρeth (density of ethanol) at 25°C is 0.789 g/cm3)
Fiber morphology
Scanning electron microscopy
Scanning electron microscopy (SEM) is a technique for visualizing the surface of massive structures, with the sample appearing in volume. Observations of the fibers were carried out on a FEG-ESEM XL3 at the chemistry laboratory of the University of Liège. Images were obtained with a secondary electron detector after gold metallization of the powders by plasma spraying (distance 5 cm, 30 mA, 0.05 atm argon, and 50 s). The accelerating voltage is indicated on the images (10.0 kV and 15.0 kV). Images were taken at different magnifications (3, 5 and 10 μm) according to the texture. Local chemical analysis was determined by combining the SEM with a dispersion spectrometer (EDS).
X-ray diffraction
X-ray diffractometric is a chemical analysis method. It can be used to identify products with the same gross chemical composition, but different crystallization forms. The analyzed sample is placed in a rectangular steel mold (15 × 10 mm) in powder form. The principle of this analysis is that of the AGEs (Argile Géochimie et Environnements Sédimentaires) laboratory in the Geology Department of the University of Liège. It uses a set of two diffractometers that record the data. One is of the ™ Bruker D8-Advance type and the other is of the ™Bruker Advance Eco type. These diffractometers operate using the Kα1 radiation of copper (λ = 1.5418 Å) and the accelerating voltage is 40 KV, the current intensity 30 mA and the scanning angle is between 2° and 70°.
Mechanical characterization: Petiole fiber tensile test
The aim of the test is to determine the Young’s modulus, mechanical strength and maximum deformation of OPPFs in unidirectional tension. Samples are first dried in a ventilated oven (ISO 3344), then fixed to tensile supports in accordance with ASTM D3822. On leaving the oven, 25 mm long fibers are taken and the diameters (in accordance with ASTM D 3822) are measured on three (03) equidistant points of the fiber using a micrometer. Each sample is then mounted on a tensile support, numbered and conditioned for 24 h in a hygroscopic tray (in accordance with ASTM D1776). After 24 h, the fibers are successively installed on the jaws of the universal testing machine (force: 5 KN and speed: 2 mm/min). During the test, the Test Master software plots the curves and calculates all the test parameters formulas (7) and (8) enable us to determine the mechanical characteristics of the fibers for each extraction method.
Ar (mm) is the measured elongation at break of the fiber; εr (%) is the maximum strain of the specimen; Fm (N) is the maximum tensile strength of the fiber; S0 (mm2) is the cross-section of the specimen.
Results and discussion
Chemical characterization of fibers
Fourier transform infrared spectroscopy
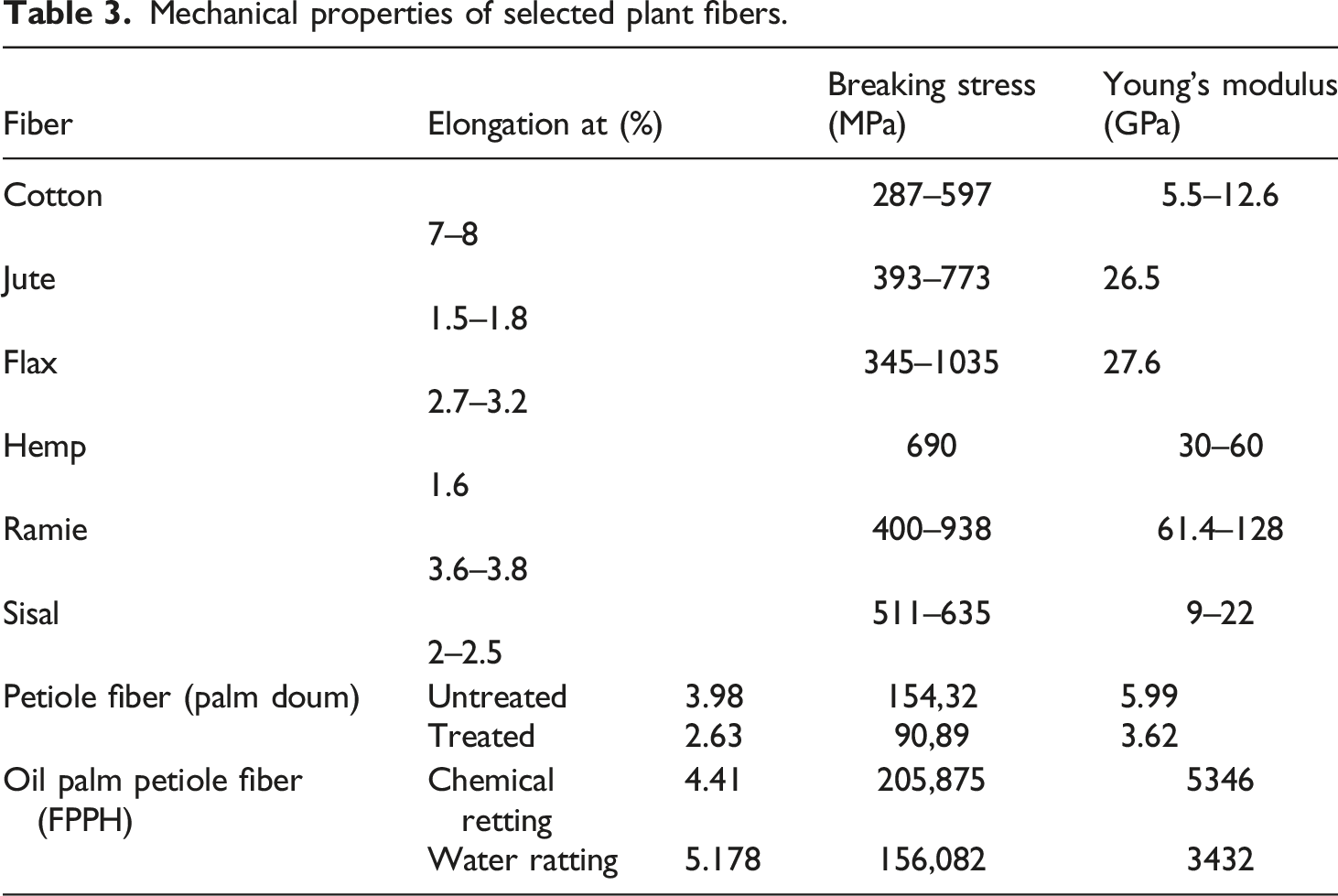
Figure 4 shows the Fourier-transformed infrared spectrum of FPPH in the wave number of 400 cm−1 up to 4000 cm−1. The bands show several peaks, namely; 1. Peaks at 897.13 cm−1 for fibers extracted by water retting (Figure 4(a)) and 896.60 cm−1 for fibers extracted by soda retting (Figure 4(b)). These peaks are attributed to the presence of β glycosidic bonds between the monosaccharides; 2. The strong peaks at 1026.58 cm−1 for fibers extracted by water retting (Figure 4(a)) and 1027.64 cm−1 for fibers extracted by soda retting (Figure 4(b)) attributed to the CO and OH stretching vibration belonging to the polysaccharide in cellulose; 3. Peaks [1232.36; 17 31.18] cm−1 for fibers extracted by water retting (Figure 4(a)) and [1236.06; 1590.97] cm−1 for fibers extracted by soda retting (Figure 4(b)). These peaks are due to the antisymmetric deformation of the C-O-C band in cellulose and hemicellulose and the aromatic skeletal vibration present in lignin; 4. Peaks around [1971.78; 2351.32] cm−1 for fibers extracted by water retting (Figure 4(a)) and [1960.16; 2322.97] cm−1 for fibers extracted by soda retting (Figure 4(b)) are linked to stretching vibrations characteristic of the carbonyl (C = O) band in hemicelluloses; 5. The peak at 2896.57 cm−1 for fibers extracted by water retting and 2920.60 cm−1 for fibers extracted by soda retting corresponds to CH stretching in lignin and waxes (Figure 4(b)); 6. The hydrophilic character of these fibers is reflected by broadband absorbance peaks appearing at around 3032.60 cm−1 for fibers extracted by water retting (Figure 4(a)) and 3037.53 cm−1 for fibers extracted by soda retting (Figure 4(b)), attributed to axial deformation of OH groups. This broad band is commonly observed in plant fibers (28; 29; 30; 31; 48] cm−1. Infrared spectra of FPPH according to extraction method: (a) Extraction by water retting; (b) Soda chemical retting extraction.
Determining the percentage of chemical components in fibers
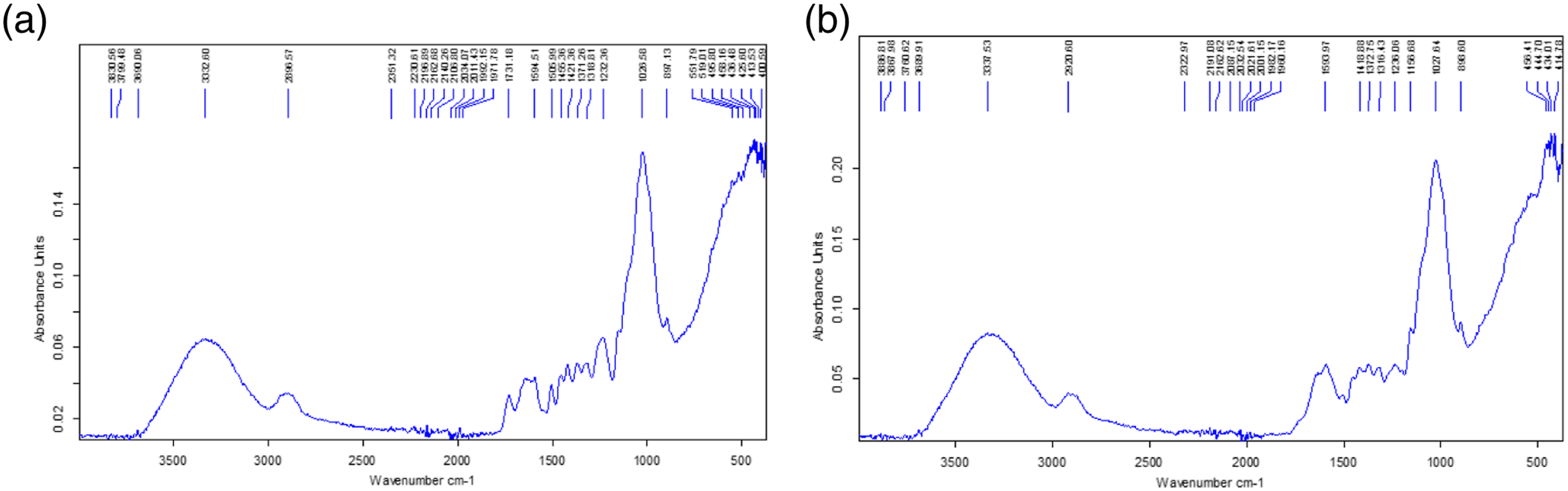
Figure 8 shows the chemical element content of FPPH according to extraction method
33
. For water retting, FPPH contains 56% cellulose (%C), 27% hemicellulose (%H) and 17% lignin (%L) (Figure 5a). For soda retting, FPPH contains 67% cellulose (%C), 19% hemicellulose (%H) and 13% lignin (%L) (Figure 5b). These values are close to those of kenaf, jute, flax, hemp and coir fibers in the literature for cellulose content.34–40 Chemical composition of FPPH: (a) Extraction by water retting; (b) Extraction by chemical retting with soda.
From the histogram in Figure 6, it can be seen that FPPH extracted by water retting contained 16.41% less cellulose, which proves that the water dissolved part of the cellulose, 29.62% more hemicellulose and 23.52% more lignin than FPPH extracted by soda retting. Chemical composition comparison.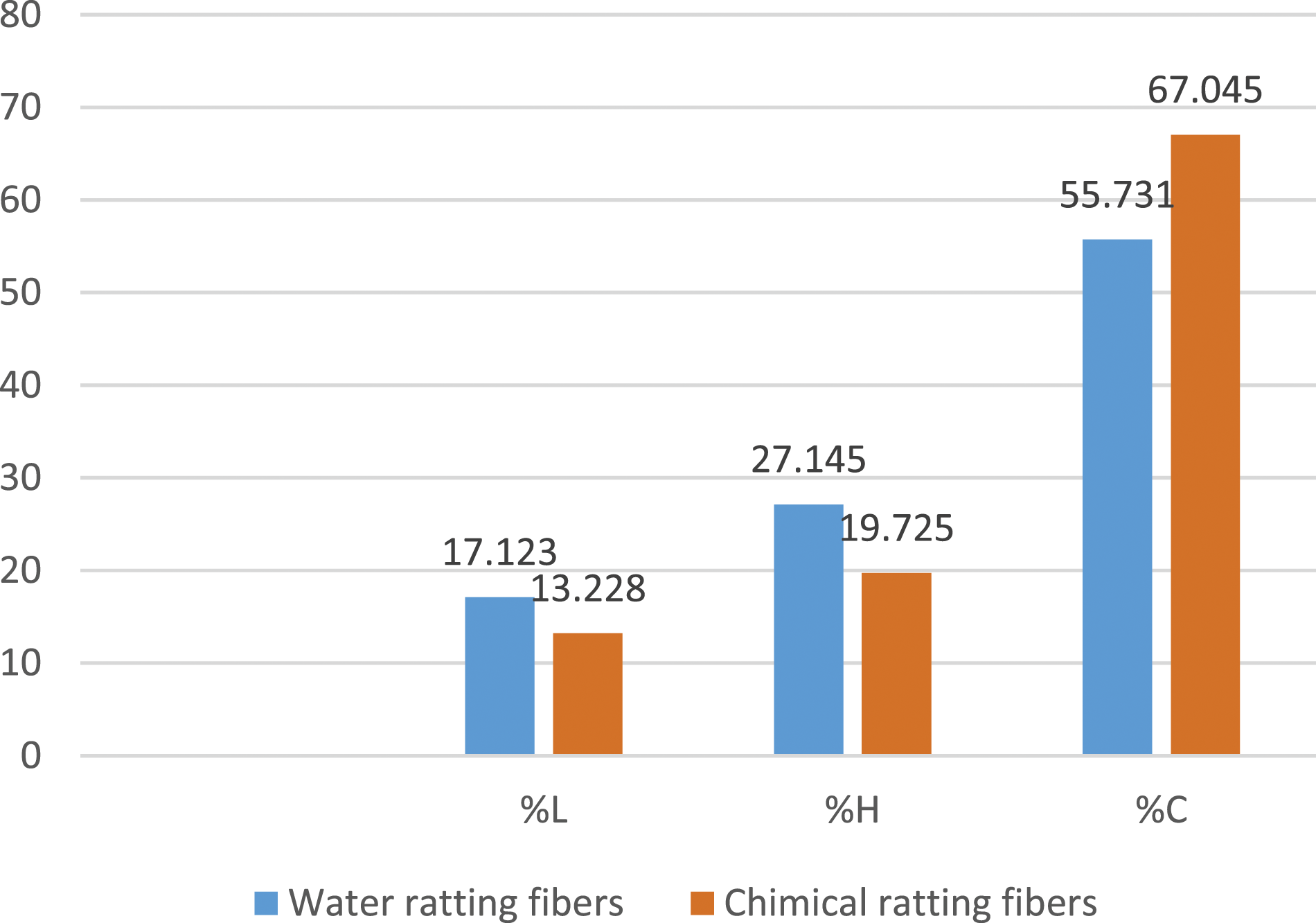
Physical characterization of fibers
Fiber outside diameters
As the histograms in Figure 7 shows, the average diameter of FPPH extracted by water retting is 6.86% greater than that of FPPH extracted by soda retting. This difference can be explained by the fact that extraction by soda retting produces fibers with an almost smooth surface, free from impurities and waxes. In addition, the diameters of FPPH are larger than those of certain plant fibers such as jute, kenaf, banana trunk, pineapple leaf and hemp.41–44 Fiber diameters by extraction method.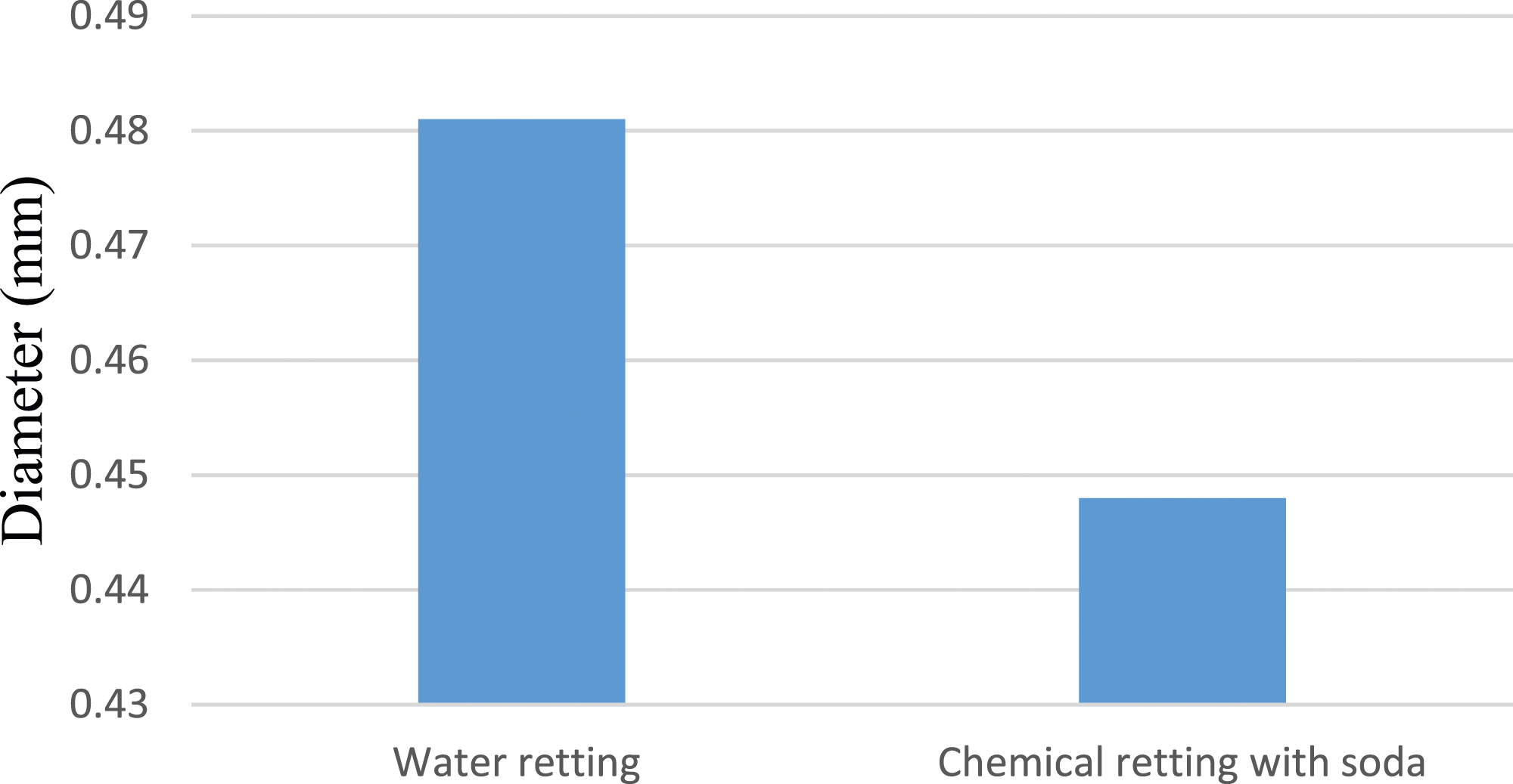
Apparent densities
Fiber density by extraction method.

Scanning electron microscopy
Scanning electron microscopy images are shown in Figures 8 and 9. In this images, a, b, c and d represent the different zooms used. From these images it can be seen that the surface of fibers extracted by water retting is smooth and have particles inclusions (Figure 8(d)). Certain particles may be wax, lignin or impurities. In contrast, the surface of fibers extracted with a 5% soda solution is very rough and does not show particles inclusions (Figure 9(d)).25,32,45 The surface irregularities of fibers extracted by water-retting have a great advantage in composite systems, offering better adhesion between the fiber and the matrix. Microstructure of petiole fibers extracted by water retting. Microstructure of petiole fibers extracted by chemical retting.

X-ray diffraction
X-ray diffraction was used to study changes in the physical structures of fibers depending on the extraction method. To achieve this, we determined the crystallinity index (Icr) of the fibers for each extraction method following the formula (9);
With: I2ɵ: the capital intensity of peak cellulose around; Iam: the intensity attributed to amorphous cellulose around 2θ = 18.9°. I2ɵ = 26.825° for fibers extracted by soda retting (Figure 10(a)). I2ɵ = 26.656° for fibers extracted by water retting (Figure 10(b)). X-ray diffraction of FPPH: (a) Chemical soda retting; (b) Water retting. Water retting.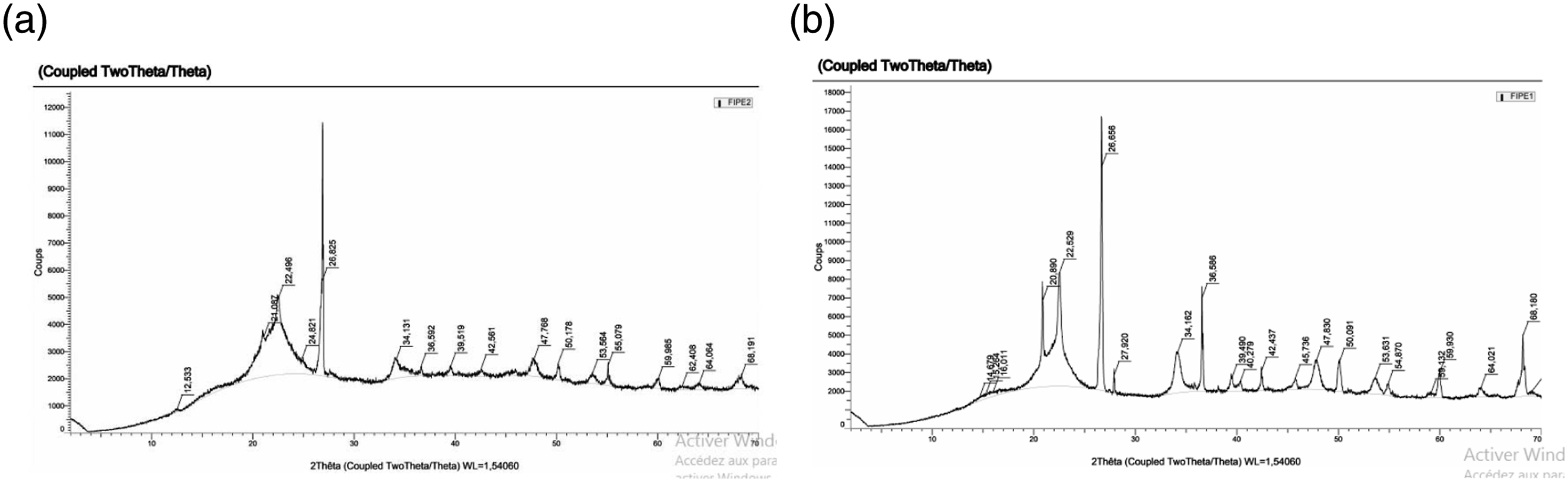
Using the formula, we obtain a crystallinity index of 58.66% for fibers extracted with a 5% sodium solution and 58.51% for fibers extracted by water retting. This is in line with the results obtained by 31 on date palm fibers, which show similar spectra to those found in this work with a crystallinity rate equal to 50 and 55% respectively. The values of the crystallinity index obtained differ little, therefore we can say that the extraction method has no significant impact on the suppleness of petiole fibers.
Mechanical characterization: Tensile test
The oil palm petiole fibers (FPPH) tested were grouped into two categories of eight fibers each (extracted by water retting and extracted by soda solution). Figures 11 and 12 show typical stress-strain curves for tests carried out on water-rubbed and soda-extracted fibers. The results of all the tests carried out are summarized in the table below. Behavior curve for FPPH extracted by chemical retting. Behavior curve for FPPH extracted by water retting.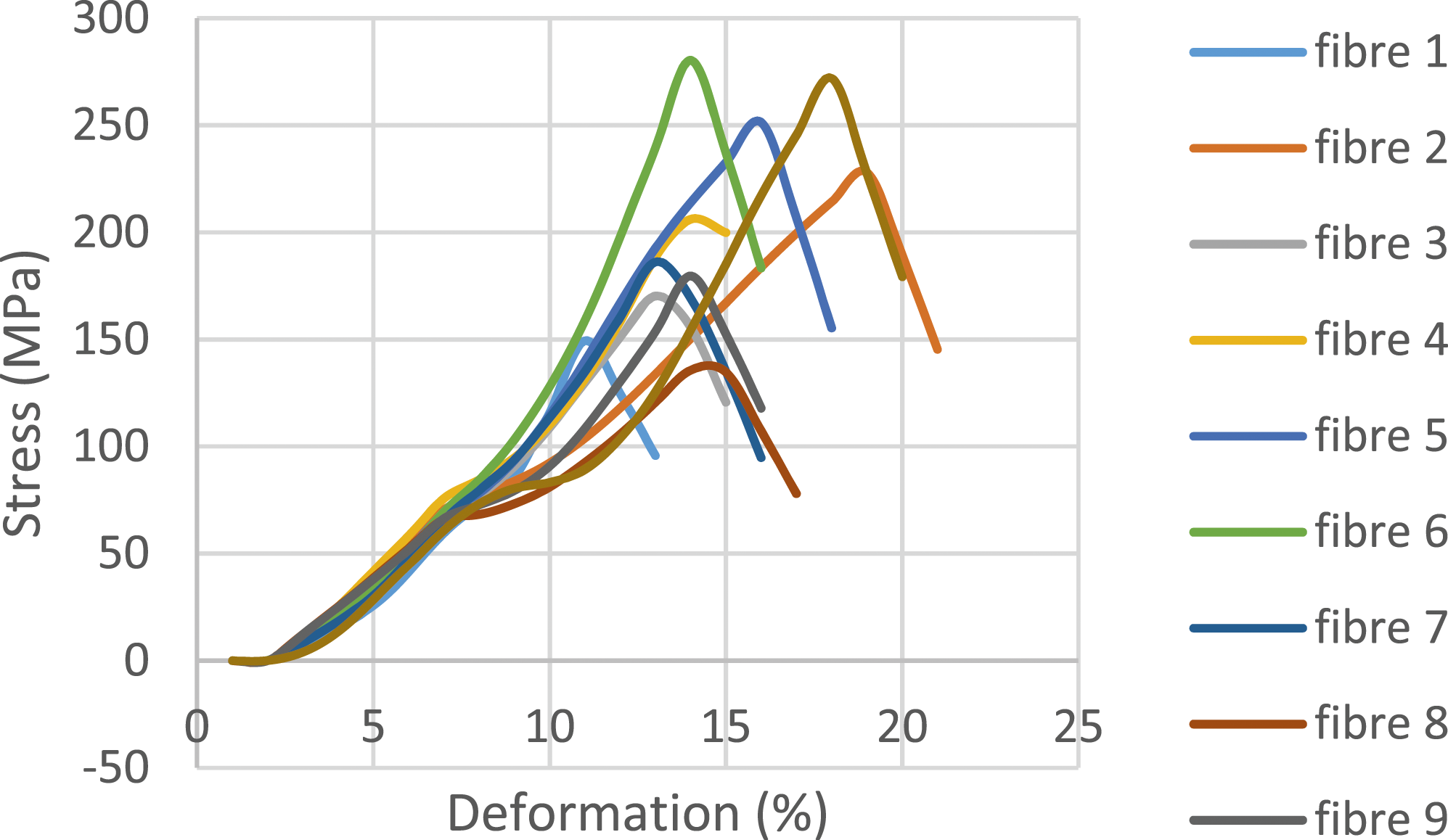
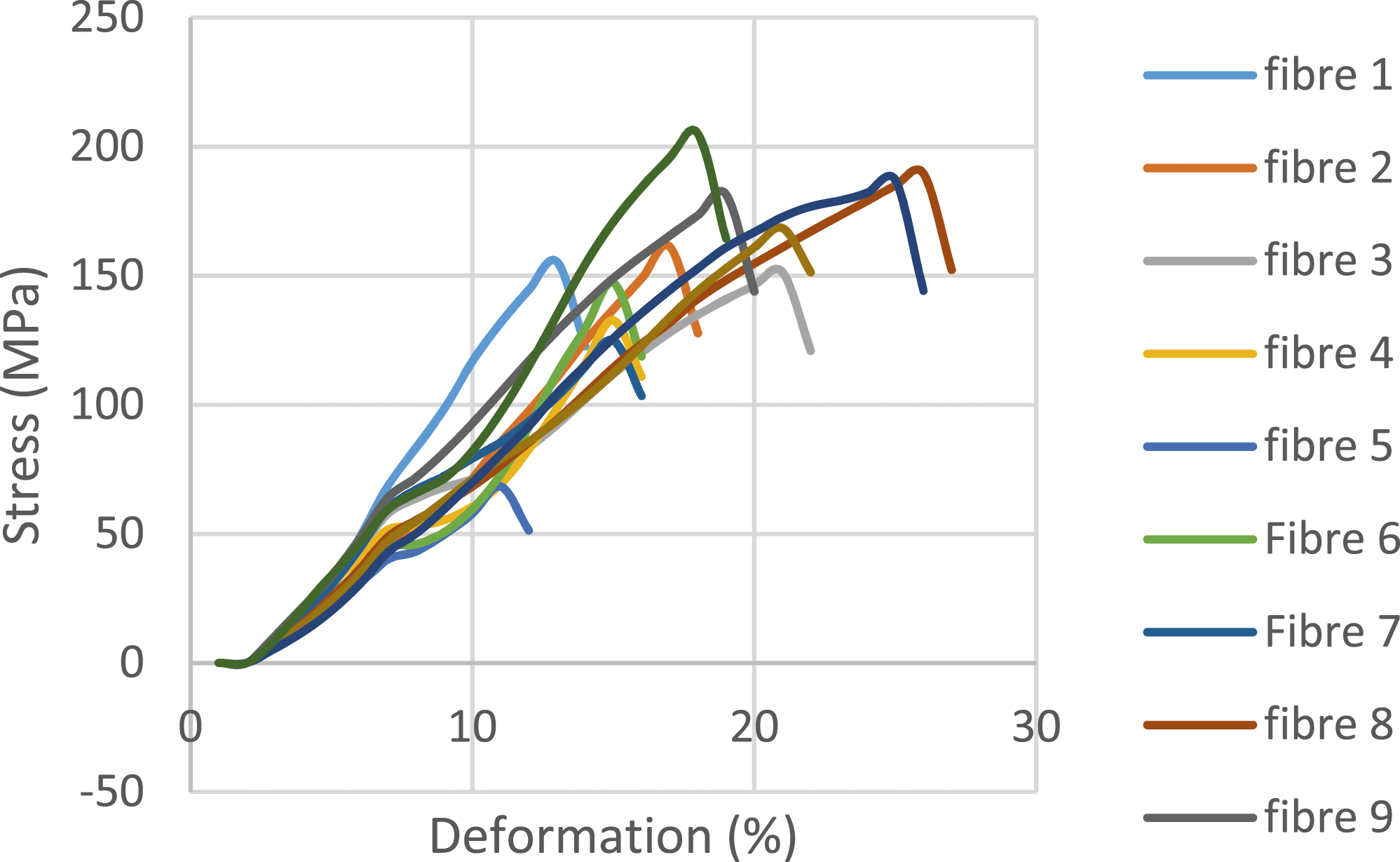
Mechanical properties of FPPH according to extraction method.

Conclusion
In this work, we studied the effect of the extraction method on OPPFs. The petioles were taken from oil palm grown in the Central Cameroon region. We extracted FPPH using two extraction methods: water retting and chemical retting with soda. Fourier transform infrared spectroscopy, XRD, SEM and tensile tests were carried out on the extracted fibers. The results of these tests showed that chemical soda retting increased the percentage of cellulose content, while reducing the percentage of hemicellulose and lignin content of FPPH. As for XRD and SEM, extraction by soda chemical retting slightly improved the crystallinity index and considerably improved the surface condition of the fibers by removing impurities and waxes. The tensile test showed that water retting produced softer fibers than soda retting. We can conclude that the choice of extraction method depends on the use and characteristics desired for the fibers (softness or stiffness).
Footnotes
Acknowledgements
The authors would like to acknowledge Analytical Chemistry Laboratory of the University of Yaoundé 1; the chemistry laboratory and the AGEs laboratory of Geology Department of the University of Liège and PYCNOLAB for the technical support in the physico-chemical and mechanical characterization of the studied fibers.
Author contribution statement
Eric Parfait Ze: wrote the paper, Performed the experiments, Analyzed and interpreted the data; Jean Calvin Bidoung; Wolfgang Nzié;Théodore Tchotang; Bienvenu Kenmeugne: Performed the experiments, Analyzed and interpreted the data, and Wrote the paper
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.