Abstract
The continuous advancement of bone restoration technologies is crucial for the creation of innovative materials that possess enhanced functional and technological capabilities. One promising area of research focuses on the development of composite materials combining polymers and ceramics for bone regeneration. These materials offer several advantages over traditional bone grafting materials, including the ability to be customized to meet the specific needs of individual patients and a reduced risk of disease transmission. In recent years, significant progress has been made in the development of polymer-ceramic biomaterials with improved capacities for bone regeneration. This progress has primarily concentrated on enhancing mechanical properties, biodegradability, and biocompatibility. The objective of this overview is to present the latest advancements in composite ceramic polymers used for bone regeneration, encompassing the corresponding manufacturing processes as well as the materials employed in these biomaterials.
Introduction
Bone, a naturally occurring complex composite material, plays a vital role in maintaining the structural integrity of the body. It is primarily composed of inorganic components (approximately 60%), a solid organic matrix (30%), and water (about 10%). 1 The intricate nature of bone arises from its composition, consisting of diverse organic and inorganic elements, which enables it to fulfill various functional requirements throughout life. Bone serves as the body's framework, facilitates muscle attachment, and regulates calcium balance, exemplifying remarkable complexity. From a biomaterial perspective, bone is characterized as a hydroxyapatite crystal matrix reinforced by a collagen fiber network, exhibiting hierarchical organization across multiple length scales. This intricate arrangement accounts for the exceptional material properties exhibited by bone. 2
In cases of abnormalities such as trauma, infection, non-union fractures, or tumor excision, the complex physiological process of bone formation necessitates bone regeneration. 3 Although bone has the potential for autoregeneration, the use of different biomaterials remains crucial to achieve optimal bone regeneration. 4
Scientific research has demonstrated that a bioactive scaffold made of ceramics and polymers is a reliable alternative to bone grafts in severe cases of bone shortage. 5 These composite biomaterials must possess suitable mechanical properties and biocompatibility to support cell attachment, development, and differentiation.
Bioactive ceramic materials are excellent candidates for developing scaffolds for bone healing, as they can maintain, repair, restore, or enhance the function of bodily tissues and organs. However, some drawbacks of using bioactive ceramic materials alone for bone healing and regeneration can be overcome by combining them with biopolymers. 6 Biopolymers are biocompatible natural or synthetic materials that, when combined with ceramics, yield composite materials with improved characteristics. Biopolymer-ceramic composites exhibit superior mechanical qualities, improved biocompatibility, and increased control over the scaffold’s breakdown rate, among other benefits for bone healing and regeneration. Advancements in the development of bioactive polymer-ceramic composites since the early 1920s have made them valuable for various clinical applications. These biomaterials form an apatite layer on their surface when implanted in the body, enabling them to adhere to living bone and promote bone regeneration. 7
In recent years, significant progress has been made in the field of polymer-ceramic composites for bone tissue regeneration. This review provides an overview of current developments in natural and synthetic polymer-ceramic composite materials for bone tissue engineering. It highlights their crucial mechanical, biological, and degradation properties, as well as their manufacturing processes. Additionally, the review explores the biopolymers and bioceramics currently applied in bone therapy.
Biopolymers for bone tissue engineering
Over the past 50 years, substantial advancements have been made in the development of biodegradable polymeric materials for biomedical applications. These materials are increasingly employed in therapeutic devices, including temporary implants and three-dimensional scaffolds for tissue engineering.8–11 Biopolymers, derived from living organisms, encompass a wide range of sources, such as plants like corn and soybeans, certain trees, and even certain bacteria.12–15
Numerous studies have demonstrated the potential of biopolymers in bone tissue engineering, primarily attributed to their biocompatibility, biodegradability, and ability to mimic the extracellular matrix of natural bone. These materials provide a three-dimensional scaffold for cell growth and differentiation, promote angiogenesis, and stimulate bone regeneration.13,16 A variety of natural biopolymers are cited in this research, including chitosan, 17 hyaluronic acid, 18 natural fibers as Flax15,19–21 and gelatin, 22 as well as synthetic polymers such as Polylactic Acid (PLA),14,23 Polyurethane (PU), Polyhydroxyalkanoates (PHA), and PHB for various biomedical applications.8,24–27
The most commonly used natural polymers for bone tissue engineering and their applications.

Tissue Engineering (TE) is an interdisciplinary field that encompasses materials science in combination with biological and engineering sciences. Both cases require an excellent biomaterial excipient to achieve controlled release of therapeutic agents to provide the correct matrix for cell proliferation. Although natural polymers have demonstrated potential advantages in supporting cell function and adhesion, there are some limitations and concerns regarding their use. For example, it is challenging to control the mechanical properties and degradation rates of natural polymers, and there is a potential for a natural polymer to elicit an immune response or carry microbes or viruses. 42
Nevertheless, synthetic polymers can be modified to possess a much wider range of mechanical and chemical properties, as well as the capacity to promote cell adhesion and proliferation, than natural polymers. Furthermore, synthetic polymers can avoid problems with immunogenicity. However, biocompatibility poses a new challenge for bone regeneration.
In the context of bone regeneration, Poly(lactic-co-glycolic acid) (PLGA) stands out as one of the most commonly used synthetic polymers due to its remarkable biocompatibility, biodegradability, and excellent mechanical properties, making it ideal for bone tissue engineering. 43 PLGA can be utilized in various forms such as microspheres, nanoparticles, and scaffolds, and it has demonstrated the ability to support osteoblast growth and facilitate new bone formation. 44
Similarly, Poly(caprolactone) (PCL) is another synthetic polymer commonly employed in bone regeneration applications. It is valued for its excellent toughness, processability, and compatibility with bone tissue.45,46 The superior mechanical properties and slow degradation rate of PCL make it suitable for long-term applications in bone regeneration. 47 Taking advantage of this polymer, Kuang et al. developed biodegradable porous scaffolds with oriented interconnected pores and high mechanical strength for bone tissue engineering. The resulting oriented porous PCL scaffolds exhibited exceptional biocompatibility, induced elongated cell morphology, and possessed a highly reinforced oriented structure suitable for load-bearing bone tissue applications. 45 Polyurethane (PU) is a versatile polymer that can be formulated into various forms, including foams, films, and fibers.49,50 This adaptability makes PU a valuable material in diverse applications. Additionally, polyethylene glycol (PEG), a hydrophilic polymer, finds use as a scaffold material in bone tissue engineering. PEG offers distinct advantages over natural hydrogels, such as the ability to undergo photopolymerization, adjustable mechanical properties, and control over scaffold architecture and chemical composition.51,52 These features contribute to its appeal in creating scaffolds tailored to specific tissue engineering requirements.
Furthermore, poly(methyl methacrylate) (PMMA) serves as a thermoplastic utilized as bone cement in orthopedic surgery. PMMA exhibits favorable mechanical properties and can be easily shaped into various forms, making it a preferred choice for orthopedic applications. 53
Polymeric nanoparticles have garnered significant attention as a highly promising strategy for facilitating efficient oral delivery of bioactive compounds. These nanoparticles exhibit inherent mucoadhesive properties, allowing them to adhere to the mucosal surfaces of the gastrointestinal tract. This mucoadhesive behavior enables enhanced residence time and improved absorption of the encapsulated bioactive compounds. By capitalizing on the unique properties of polymeric nanoparticles, researchers aim to overcome the challenges associated with oral drug delivery, such as enzymatic degradation and limited absorption. 54
It seems as though the future of biopolymer application is to use various polymer combinations to develop hybrid polymers that have a better specificity profile for specific biomedical applications, be such as tissue engineering, drug delivery, or wound healing. This is in contradiction to trying to categorize specific polymers for specific biomedical applications. In fact, a large number of recent studies on polymers for biomedical use have addressed merging various biomaterials using methods including blending, grafting, and chemical cross-linking reactions, with generally beneficial outcomes.
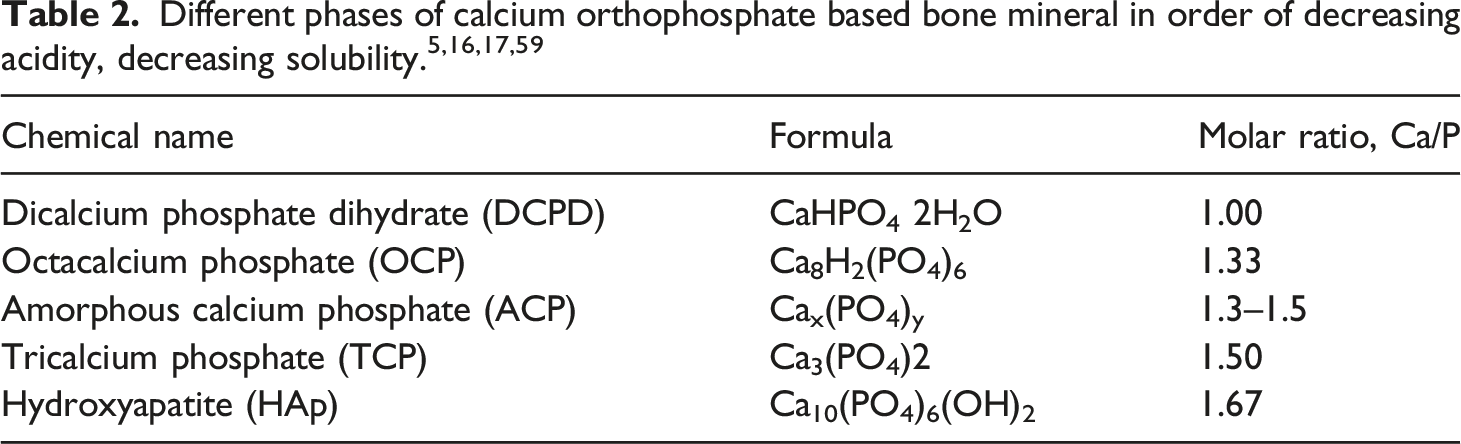
Bioceramics for bone regeneration
Comparative composition of chemical elements Ca and P in calcified tissues of adult humans and HA. 69
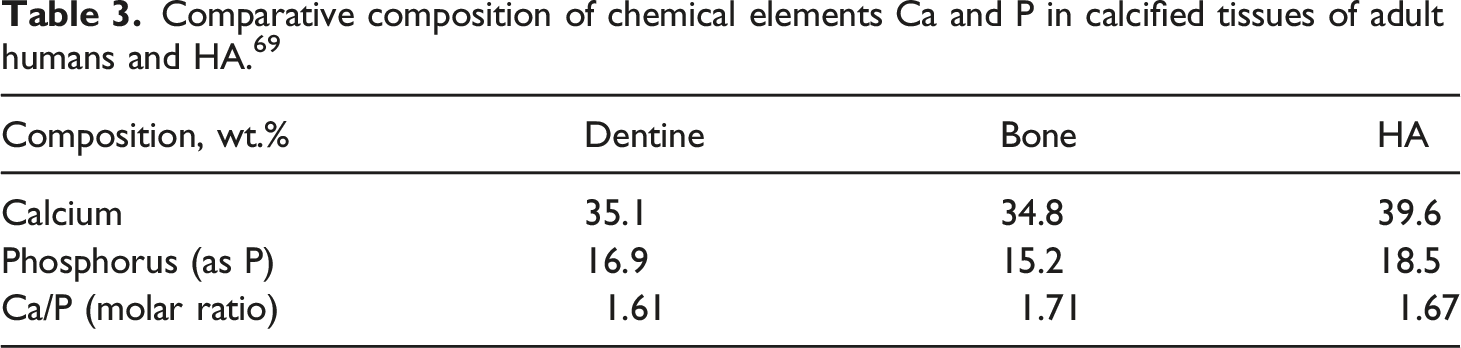
For years, bioceramics have been successfully utilized in orthopedic applications as reliable and effective materials for bone implants. However, their mechanical properties pose a significant challenge in terms of providing optimal strength and durability that can withstand the stresses and physiological demands of repaired bone.24,78 Furthermore, certain applications may require bioceramics to possess tenacity and ductility, which are not inherent properties of these materials. 26 Additionally, the rate of osseous regeneration may not align with the rates of degradation and resorption of bioceramics, resulting in complications such as implant fractures or deterioration. To overcome these limitations, several ongoing studies are exploring innovative techniques and improved materials to enhance the capabilities of bioceramics.24,79-81 These endeavors seek to overcome the challenges associated with their mechanical properties, promote enhanced strength and resilience, and ensure compatibility with the natural regeneration processes of bone. 82
Recent biocomposite polymer-ceramic applied in bones regeneration
In recent years, the field of polymer-ceramic composites for biomedical application has undergone significant advancements and developments. Researchers have discovered new types of raw materials and developed techniques using state-of-the-art procedures to fabricate, shape, and transform these materials. Moreover, new tools have been developed to study these emerging materials. These developments have led to the creation of composites with advanced capabilities for bone regeneration, providing solutions to contemporary problems. In order to cure bone pathologies and repair fractures, polymer-bioceramic composites are nowadays adapted to many uses.
For this remarkable generation of biomaterial, ceramics play a crucial role by imparting bioactivity and osteoconductivity to the scaffold. The incorporation of ceramic components within the composite provides a robust framework that can support the growth of new bone tissue. On the other hand, polymers within these composites contribute valuable properties such as flexibility and resorbability. The polymer constituents ensure that the composite remains pliable, allowing it to adapt to the surrounding bone tissue and minimize the risk of mechanical failure. Furthermore, the polymer’s resorbable nature facilitates the gradual breakdown of the composite over time, enabling the natural integration and remodeling of the regenerated bone tissue.
The composites polymer-ceramic are generaly composed of ceramic particles like hydroxyapatite (HA)83,84 or tricalcium phosphate (TCP) with a polymer matrix such as poly(lactic acid), poly(glycolic acid), or polycaprolactone (PCL).85,86 The key benefits of using these biodegradable biocomposites are that they can be naturally expelled from the body, eliminating the need for a second surgery to remove them after use. Second, due to the biocompatibility, bioactivity, and non-toxicity of degradable implant materials, osteoblasts will regenerate bone as the implant material gradually wears away. Further technical benefit of using this biodegradable biomaterial is its ability to replace stainless steel bone plates, screws, etc… used for bone stabilization. 87 Their potential is considerable, whether it is bone augmentation, controlled delivery mechanisms or a revolution in hard tissue engineering.
Hydroxyapatite (HA) is the most used ceramic biomaterial since it mimics the mineral composition of the bone in vertebrates. However, this biomimetic material has poor mechanical properties, such as low tensile and compressive strength, which make it not suitable for bone tissue engineering (BTE). For this reason, HA is often used in combination with different polymers and crosslinkers in the form of composites to improve their mechanical properties and the overall performance of the implantable biomaterials developed for orthopedic applications.83,88
The following section will describe a number of recent biodegradable polymer-ceramic composites designed for bone regeneration and highlight several of their unique characteristics.
Zimina et al. developed a composite material by combining polylactide acid (PLA) and hydroxyapatite (HA) particles, and as a result of physicochemical analysis, the presence of essential functional groups and uniform distribution of HA particles within the PLA/HA composite were confirmed. The composites exhibited macropores with a total porosity of 79%, leading to an improved water contact angle compared to pure PLA. Moreover, in vitro and in vivo evaluations demonstrated 3.2 times higher adhesion of multipotent mesenchymal stromal cells to PLA/HA scaffolds compared to pure PLA. Additionally, subcutaneous implantation of the porous scaffolds in mice showed excellent tolerance and widespread tissue ingrowth, while the HA-containing scaffolds exhibited a reduced inflammatory response after 2 weeks compared to pure PLA. 85
Similarly, Si et al. (2019) fabricated scaffolds using PLA and PLA/HA composites with a porosity exceeding 85%. The increase in the concentration of hydroxyapatite (HA) resulted in a decrease in sample porosity, while simultaneously enhancing hydrophilicity, mechanical stability, and cytocompatibility. 89 Additionally, 3D-printed PLA/PCL/HA composite scaffolds with a PLA/PCL ratio of 70/30%–35% showcased a porosity of 77%, an average pore size of 160 μm, and a Young’s modulus of 1.35 MPa. In vitro studies investigating MG63 cells examined cell adhesion, viability, and mineral deposition in PLA/PCL/HA scaffolds with varying PLA/PCL ratios (70/30, 50/50, and 30/70) and a fixed HA content of 35%. 90
Furthermore, Wu et al. (2020) attempted to prepare PLA/HA composites mimicking the structure of trabecular bone, leading to enhanced mechanical behavior and elastic modulus with increasing HA content in the PLA matrix. 91 In another study, Masoumeh et al. (2018) fabricated a GBR membrane consisting of polycaprolactone (PCL)/gelatin/chitosan, which was modified with different percentages of β-tricalcium phosphate (β-TCP) to improve biocompatibility, mechanical properties, and antibacterial activity. 92
Wang et al. (2019) investigated the applicability, cellular efficacy, and osteogenic activities of porous nano-hydroxyapatite/Poly (glycerol sebacate)-grafted maleic anhydride (n-HA/PGS-g-M) composite scaffolds. The study revealed increased apatite deposits on the surface of n-HA/PGS-g-M scaffolds compared to PGS-g-M scaffolds. These porous n-HA/PGS-g-M composite scaffolds demonstrated acceptable biocompatibility and mechanical properties, while also stimulating hADSC cell proliferation and differentiation. 93
Additionally, Najah et al. conducted research on a calcium phosphate reinforced polyhydroxyalkanoate (PHA) biocomposite, where the addition of CaP to PHA resulted in fluctuations in tensile strength and impact strength. The composite exhibited a drop in these properties at a level approximately three times lower than that of neat PHA. 79
In another study by Monia (2022), a bone implant (β-TCP/DCPD-PHBV) based on Poly(hydroxybutyrate-co-hydroxyvalerate) (PHBV) and bioceramic β-TCP/DCPD demonstrated efficacy in healing bone defects. Mechanical investigations revealed that this biomaterial possessed satisfactory mechanical strength (195.21 MPa), which closely resembled that of natural bone. Furthermore, the combination of the two biocompatible materials in this study provided an additional advantage, as the ductility of PHBV mitigated the brittleness typically associated with pure β-TCP/DCPD. 26
Orthopedic implants’ functionality may be hampered by inadequate host tissue integration, inflammatory reactions, and infection. These bone biomaterials' surface characteristics have an impact on how immunological and osteogenic cells function biologically. We may now make nanostructured surfaces with controlled physicochemical properties that affect osteogenesis-related and immune cell activity, affecting bone integration and local immune response thanks to modern nanofabrication techniques.
The manufacturing process significantly influences the properties of a biocomposite. The selection of manufacturing techniques, such as extrusion, compression molding, 3D printing, or electrospinning, can have a profound impact on the final characteristics of the biocomposite material. The following section describes the most commonly employed manufacturing processes used to create polymer-ceramic composites.
Various polymer-ceramic manufacturing processes
Polymer-ceramic materials can be synthesized using various techniques. One commonly used method is Sol-Gel Processing, where ceramic particles are synthesized in a sol solution and incorporated into the polymer matrix.94,95 This process allows for precise control over the size, shape, and content of the ceramic particles, resulting in composites with exceptional mechanical and thermal properties.96,97 In addition, the study conducted by Dorozhkin provides further evidence of the successful application of the sol-gel technique, as demonstrated in the creation of a Titanium-HA composite with outstanding mechanical qualities and good biocompatibility. 66
Another technique, in situ polymerization, involves polymerizing the polymer matrix while the ceramic particles are formed in situ. This technique ensures a homogeneous distribution of ceramic particles within the polymer matrix. 98 The PCL-CaP composite synthesized using this method exhibits excellent mechanical properties and good biocompatibility, supporting the growth and proliferation of human mesenchymal stem cells. 99 Similarly, the synthesis of Poly(methyl methacrylate)-Zirconia Composites utilizing this technique has found applications in dental implants and bone fillers. 100
On the other hand, melt mixing is a technique that entails mixing ceramic particles with a polymer matrix using an extruder or mixer.88,101 By employing this process, carbon nanotube (CNT)-reinforced polymer composites are synthesized, leading to improved mechanical, thermal, and electrical properties compared to the pure polymer matrix. 102
Three-dimensional (3D) printing is an additive manufacturing method that enables the fabrication of complex structures using a wide range of materials.103,104 Numerous biocomposites for bone repair, including poly(lactic acid) (PLA)/Hydroxyapatite (HAP) composites, zinc oxide (ZnO)-grafted/polylactic acid (PLA) composites, Polylactide/Poly(butylene adipate-coterephthalate) (PLA/PBAT) composites, abalone/ polycaprolactone (aba/ PCL), have been produced through this technique have been produced through this technique.105–108
Additionally, ultrasonic processing is an innovative method that allows for the fabrication of CNT nanopaper nanocomposites with a high CNT loading at a fast infiltration rate. 109 By employing ultrasonic waves, the resin is impregnated into the CNT nanopaper, resulting in the formation of nanocomposites. 110
Furthermore, the electrospinning technique integrates ceramic particles into a polymer solution, utilizing an electric field to produce polymer-ceramic nanofibers.111–114 These nanofibers exhibit improved mechanical and thermal properties due to their increased surface area-to-volume ratio.46,54,115–118
Lastly, compression molding is a method in which ceramic particles are mixed with a polymer matrix and subjected to high pressure and temperature, resulting in a composite with excellent mechanical and thermal properties.119,41
In summary, the selection of a processing method for polymer-ceramic composites should consider factors such as the desired properties, uniformity of particle distribution, cost-effectiveness, and application requirements. Each method has its own advantages and limitations, and choosing the most suitable technique will contribute to the development of composites with improved performance and functionality.
Conclusion and prospects
In conclusion, polymer-ceramic composites have emerged as a promising solution for bone repair and regeneration by combining the advantageous properties of ceramics, such as bioactivity and osteoconductivity, with the flexibility and resorbability of polymers. This combination allows for the development of composites with enhanced mechanical strength, biocompatibility, and controlled degradation kinetics. By incorporating ceramic particles into a polymer matrix, these biomaterials offer alternatives to traditional approaches and materials, opening up new possibilities in the field of bone repair.
Looking ahead, the future of polymer-ceramic composites in bone repair will be influenced by intelligent manufacturing techniques. The rapid development of intelligent manufacturing, including 3D, 4D, and 5D printing, will revolutionize the production of these composites. These advanced manufacturing techniques, aided by intelligent systems and robotics, will contribute to improved processing and quality control, leading to more efficient and cost-effective production of polymer-ceramic composite parts. This integration of intelligent manufacturing will help address labor costs and reduce production defects.
Another perspective for advancing polymer-ceramic composites in bone repair lies in the incorporation of bioactive molecules, growth factors, and drug delivery systems. By integrating these factors into the composites, the therapeutic potential of these biomaterials can be further enhanced. The inclusion of bioactive molecules facilitates active promotion of bone regeneration, angiogenesis, and antibacterial activity. Moreover, the controlled release of these bioactive molecules allows for modulation of the healing process and targeted tissue regeneration in specific clinical scenarios.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.