Abstract
The thermal decomposition process of 3D printed filament made from micro-nano rice husk (MNRH)/polylactic acid (PLA) blends was studied by dynamic thermogravimetric analysis. The characteristic temperatures and apparent activation energies of unmodified, single modified, and double modified RH/PLA composites were calculated by Friedman (FD), Flynn-Wall-Ozawa (FWO), Coats-Redfern (CR), and Kissinger (KS) kinetic models. With the modification of MNRH and PLA in the composites, the initial thermal decomposition temperature of the composite increased from 236.3°C to 244°C. At the same time, the thermal degradation degree decreased and the transition temperature interval increased. The apparent activation energy (AAE) values of different modified composites ranged from 90 to 120 kJ/mol, depending on the modification method and calculation method of the material. These four kinetic models provide methods to analyze the thermal stability of composites. It is helpful to known the thermal decomposition behavior of MNRH/PLA composites, and it will contribute to the development of MNRH/PLA filament for 3D printing in the application of automotive interior parts production.
Introduction
3D printing technology has been widely used in the molding and preparation of composite materials because of its characteristics of fast molding speed, low manufacturing cost and simple operation. In the past years, PLA composites have widely used in the preparation of 3D printing filaments.1–5 PLA, made from starch, cellulose and polysaccharide by hydrolysis fermentation, has the characteristics of excellent biocompatibility, good mechanical properties and easy processing. PLA has become the most representative biodegradable polymer to replace petroleum-based polymer. However, pure PLA also has the disadvantages of expensive cost and brittle character, which seriously limit its application scope. 6 In order to solve these problems, our previous study 7 (published in Journal of Applied Polymer Science) found that using silane coupling agents as modifier and adding micro-nano rice husk (MNRH) fiber to PLA matrix can boost the mechanical quality and thermal stability of the composites. The experiment results showed that the tensile strength, tensile modulus, flexural strength and flexural modulus of PLA composites modified by KH550 and KH570 increased by 83%, 98%, 54% and 61%, respectively. The experimental results also showed that the thermal decomposition performance of PLA composites was affected by filling PLA with rice husk fibers, and the thermal decomposition performance of PLA composites modified by KH550 and KH570 was also affected.
The thermal decomposition characteristics of composites are reflected in the activation energy (AAE) of composites. The AAE of the composite shows the minimum energy required for the thermal decomposition reaction of the activated molecules.8–10 Therefore, it is necessary to assess the activation energy of PLA composites to research the thermal decomposition performance. For example, Wang et al. 11 studied the thermal decomposition behavior of thermoplastic dry starch (DTPS)/PLA blends with maleic anhydride as compatibilizer. The experiment results showed that the AAE value of the composites reduced significantly after blending. Cai et al. 12 used the Arrhenius equation to calculate the AAE of isothermal crystallization, and found that the crystallization AAE value of pure PLA was higher than that of thermoplastic starch (TPS)/PLA composites. Professor Oza et al. 8 treated hemp fiber/polylactic acid composite by three chemical surface modifications, namely alkali, silane and acetic anhydride, and calculated the activation energy of the composite by FWO method. The results showed that the AAE of acetic anhydride modified hemp fiber composite was 10–13% higher than that of other composites. Among the three surface modifications, the AAE of acetic anhydride modified hemp fiber composite was 159–163 kJ/mol.
There are many models that from different angles to calculate the AAE value of PLA composite materials.13,14 Li et al. 15 studied the preparation of lignocellulose nanomaterials from pine bark and tobacco stem, and used four thermodynamic models of Friedman (FD), Flynn-wall-Ozawa (FWO), Coats-Redfern (CR) and Kissinger(KS) to calculated the AAE. The experiment results showed that the AAE values of pine bark and tobacco stem were 229–239 kJ/mol and 229–233 kJ/mol, respectively. Yuzay et al. 16 studied the thermal decomposition of zeolite/PLA composites, and estimated the apparent activation energy of PLA/zeolite composites by FWO and KS methods. The experiment results reported that the AAE values obtained by KS method were lower than those obtained by KWO method, and the AAE value of the zeolite/PLA composites was lower than that of PLA itself.
In addition, the application of plant fiber also can affect the thermal decomposition properties of PLA composites.17,18 For example, Yufeng Sun et al. 19 used dynamic thermogravimetric analysis to study the thermal decomposition process of 3D printed fiber with PLA/starch blend under nitrogen environment. With the increasement of starch content in starch/PLA composites, the transition temperature range increased, and the initial thermal decomposition temperature and thermal degradation degree of starch/PLA decreased. Tao et al. 20 researched the thermal decomposition of PLA composites filled with wood fiber. It was found that the initial degradation temperature of wood powder/PLA composites decreased slightly. Petinakis et al. 21 investigated the effect of hydrophilic fillers starch and wood meal on the degradation and decomposition of PLA composites. The experiment results showed that both starch and wood meal could promote the thermal decomposition of PLA, and the thermal decomposition effect of starch was stronger than that of wood meal. In summary, the addition of plant fiber affects the thermal decomposition of the PLA composites.22,23
In our previous work, (published in Journal of Applied Polymer Science), 7 MNRH fibers were compounded in PLA to prepare 3D-printed filaments, and the experimental results showed that when the MNRH fiber contents reached 6 wt%, the RH/PLA composites demonstrated the best heat stability, mechanical performance and hydrophobic property. While although researchers have done a large number of studies on plant fiber filled PLA composites, there are few studies focus on the thermal decomposition characteristics of 3D printed MNRH/PLA composites.24–26 Costa et al. investigated the thermal stability of pinecone residue toughened with acrylonitrile-butadiene-styrene (ABS) for 3D printing pens, and evaluated the thermogravimetric analysis of ABS, FR, FT, FB, and their filament composites. Results demonstrate that the first process from room temperature to 130°C is attributed to water evaporation, while the second process is related to the degradation of hemicellulose, lignin, and cellulose. Fiber degradation occurs in the first stage between 245°C and 399°C, while the second degradation event occurs above 420°C and is associated with the degradation of ABS, leading to the formation of burnt residue. 27 Hence, the objective of this study was to research the thermal decomposition characteristics of 3D printed MNRH/PLA composites. And four different thermal kinetic equations named FD, FWO, CR and KS were used to calculate the activation energy.9,15,28 The thermal stability of unmodified, KH550 modified MNRH, KH570 modified PLA and double modified MNRH/PLA composites was studied by four different thermal kinetic equations.
Materials and methods
Materials
RH was obtained from Henan Xinxiang Yanbin Rice Industry Co, LTD., harvested in mid-October 2021. PLA pellet (4032D) with a density of 1.24 g/cm3, was provided by Nature Works (USA). Two silane coupling agent of KH550 (used to modify RH) and KH570 (used to modify PLA) were provided by Nanjing Shuguang Co, LTD. Sodium hydroxide (NaOH), used for the pre-treatment of rice husk fiber, was provided by Harm Sinopharm Chemical Reagents.
Materials preparation
The rice husks were first cleaned and then dried in an oven at 80°C for a duration of 24 h. The dried rice husks were then ground for a period of 24 h, resulting in the production of rice husk fiber with micro-nano size, referred to as MNRH. Observation of the rice husk fiber using SEM revealed that it had a size range of 0.5–50 μm. Before melt blending, the MNRH and PLA were pretreated by KH550 and KH570. In the MNRH pre-treatment stage, KH550 (2 g) and MNRH (100 g) were put into 90% ethanol solution and stirred 20 min, and then dried in an oven to get dried MNRH. In the PLA pre-treatment stage, the KH570 (3 g) and PLA (100 g) were put into a beaker stirred for 30 min, and then dried in an oven to get dried PLA pellets.
In the melt extrusion stage, PLA and MNRH were first melted by a single screw extruder (Wellzoom B, Misstar Technology LTD.). The temperature in the feeding area and the mold was set at 185 ± 3°C and 175 ± 3°C, respectively. Next, the wires were cut into particulates with a cutting into particles by granulator (Dingbo180, Henan Dingbo Machinery Manufacturing Co, Ltd, China), therefore, these particulates were put into the single screw extruder again to produce the 3D printed wire (1.75 mm ± 0.1 mm), and then printed by a 3D printer. The temperature in the left and right areas during the second extrusion was maintained at 174 ± 3°C and 184 ± 3°C, respectively. In addition, the 3D printer nozzle temperature was set at 205 ± 5°C, while the platform (glass) temperature was kept at 60°C. In this study, the MNRH/PLA composites were consisted of six precent of MNRH, and the specific MNRH/PLA composites production and PLA-based filaments were shown in Figure 1 and Table 1, respectively. Schematic framework of composites production. Designation and formula of composite material.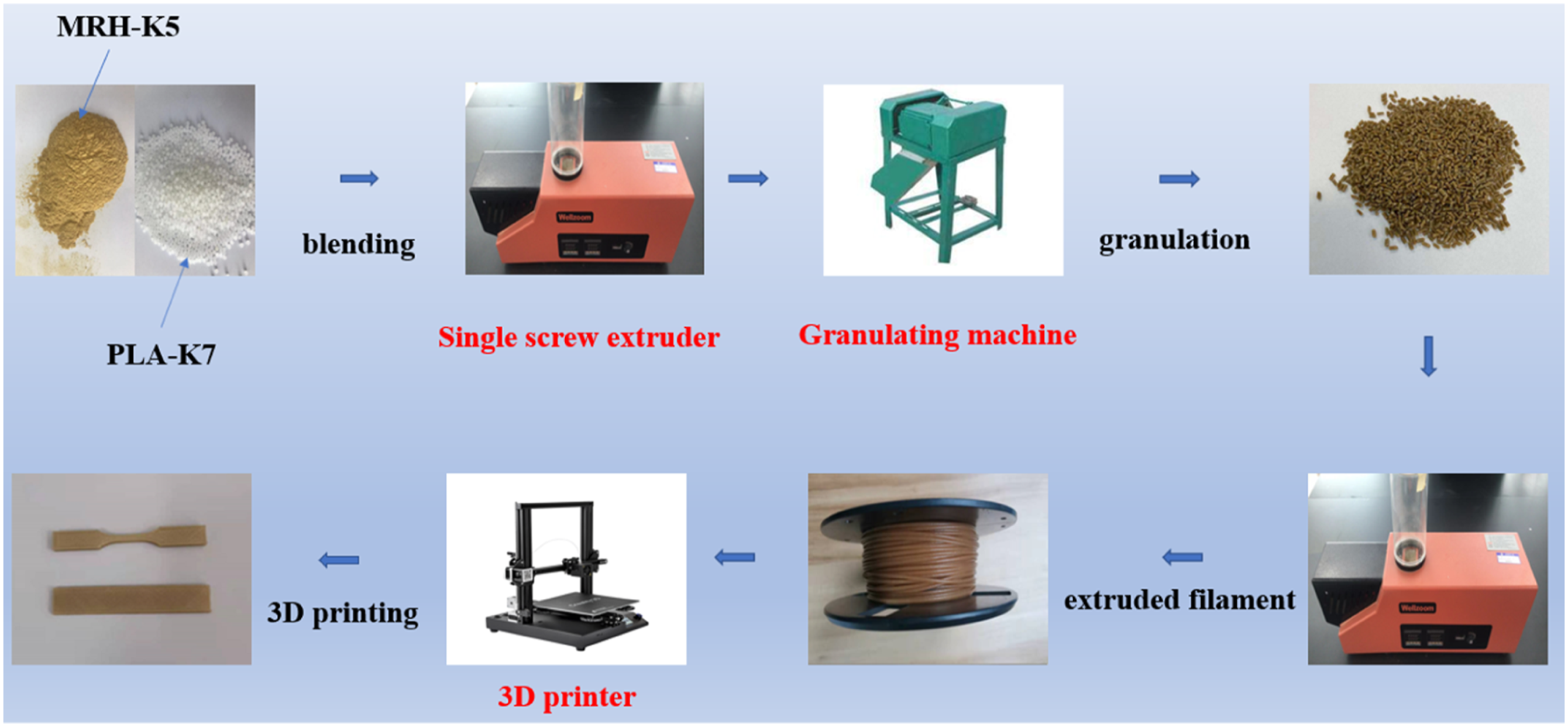

Characterization
The microstructure of four kinds of composites was observed by scanning electron microscope (Hitachi S-3400 N, Japan). Before scanning electron microscopy (SEM), the composites were immersed in liquid nitrogen, and the samples were coated with gold film about 4 nm thick on the cross-section surface by sputtering coating machine.
In order to analyze the chemical structure of the composites, The attenuated total reflection-Fourier transform infrared spectroscopy (FTIR) instrument (IR-960, Tianjin Ruian Technology Co, LTD.) to obtain the infrared spectrum. The scanning range was 3100 cm−1 ∼ 450 cm−1, and the spectral resolution was 4 cm−1.
The thermal decomposition performance of the composites was obtained with a thermogravimetric analyzer (SDT 650, TA instrument of USA) under nitrogen and at four different heating rates of 5°C/min, 10°C/min, 15°C/min and 20 °C/min in the temperature range of 30°C– 550°C. Nitrogen flow rate was 50 mL/min, and the test samples weighed at10–15 mg.
Thermal decomposition kinetics
The thermal decomposition properties of composites were evaluated according to the data of thermogravimetric analyze. The basic equation of thermal degradation kinetics was described as follows.
9

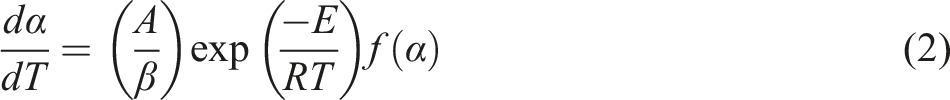
There are many thermal kinetics models to calculate AAE value, and this paper mainly uses four common models for comparison.29–31 The first method is FD, when the plot of the variable, 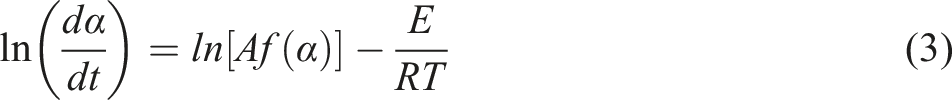
The second method is FWO. When the plot of the variable, logβ versus 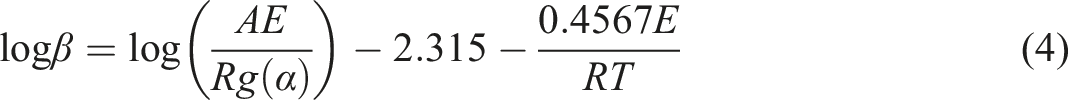
The third method is KS. When the plot of the variable, 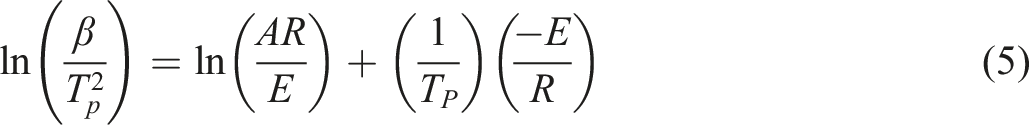
The fourth method is CR. When the plot of the variable, 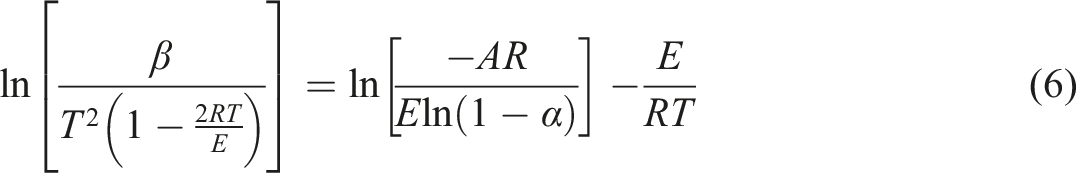
Results and discussion
Basic properties
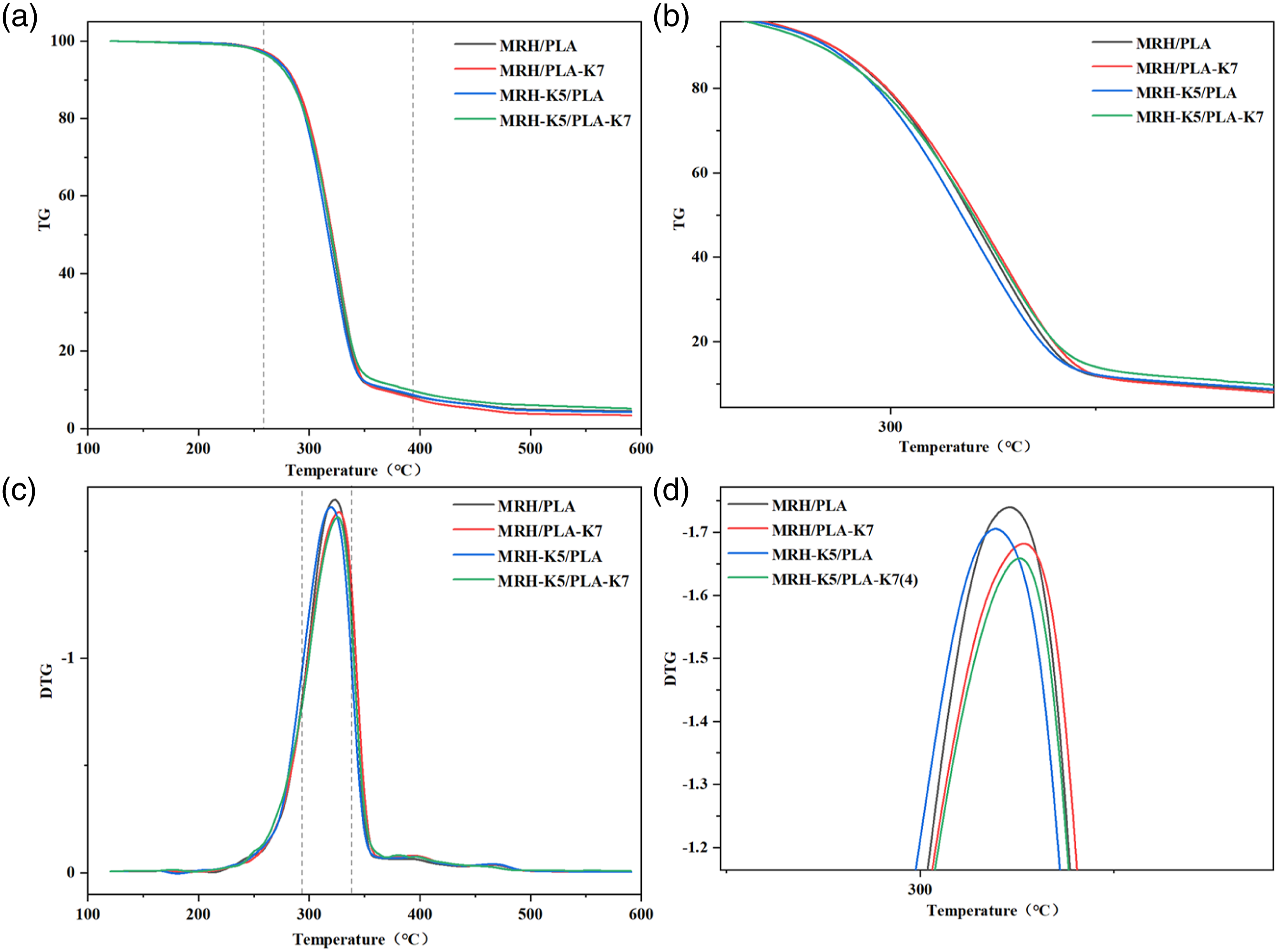
Figure 2 showed the morphology of the four composites at the same multiple level. It can be seen that the microstructure of the four MRH/PLA composites was different, and there were some holes and cracks, which affected the thermal stability of the composites. The unmodified MRH/PLA composite (Figure 2(a)) had some large cracks and holes, and the fracture was not uniform. Poor interfacial compatibility will lead to interface debonding, energy and stress concentration, which will reduce the thermal stability of composites.
32
As shown in Figures 2(b) and (c), the surface of single-modified MRH-K5/PLA and MRH/PLA-K7 composites were also found to have aggregates and gaps, which were caused by poor adhesion and debonding of the interface region.33,34 As shown in Figure 2(d), the binding conditions of MRH-K5/PLA-K7 composite with double modification were better than those of MRH/PLA, MRH-K5/PLA and MRH/PLA-K7 composites. The MNRH fibers of composites were well mixed into PLA and distributed more evenly, which indicated that the adhesion between MNRH and PLA were improved by K550 and K570.35,36 Electron fiber mirror images of different composite samples at the same magnification. (a) MRH/PLA; (b) MRH-K5/PLA; (c) MRH/PLA-K7; (d) MRH-K5/PLA-K7.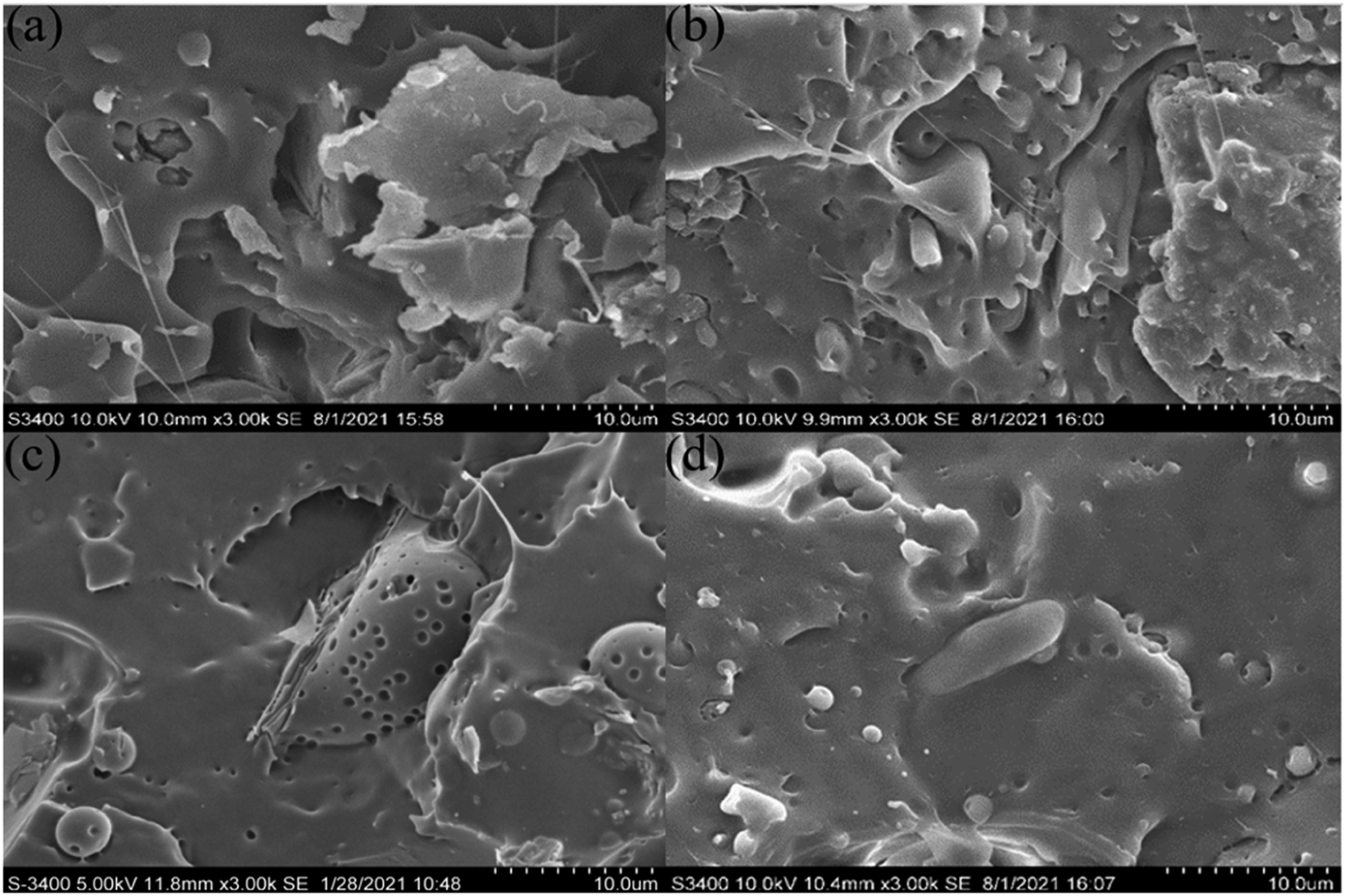
Figure 3 showed a contrast of the FTIR spectra curves of the four composite specimens. The peaks at 2995 cm−1 and 2945 cm−1 were due to the stretching of the methylene stretching vibrations,
37
The absorption peak at 1750 cm−1 was the stretching vibration of the C = O functional group.11,38 The absorbance intensity at 1650 cm−1 of MRH/PLA decreased obviously due to the loss of water molecules.
39
The weak peaks 1455 cm−1 were caused Si-O-PLA stretching, indicating that the silane coupling agent treated with MNRH fiber and PLA.39,40 The absorption peaks at 1330 cm−1 and 1365 cm−1 correspond to the symmetric and asymmetric flexing vibrations of -CH, respectively.
41
The regions of 1245 cm−1 and 1182 cm−1 represented the scaling of Si-CH2 in alkanes.35,42 Due to the asymmetric and symmetric stretching of Si-O-C and Si-O-Si, the fluctuation appeared in the 1140-900 cm−1 region.
39
The FTIR curves of MRH-K5/PLA, MRH/PLA-K7 and MRH-K5/PLA-K7 showed superior peaks at 680 cm−1, 760 cm−1 and 812 cm−1 than that of MRH/PLA, indicating that KH550 and KH570 were smoothly grafted onto PLA and RH fibers.39,42,43 The MRH6/PLA composite showed a low peak at about 550 cm−1, it also indicated that the silane coupling agent treated with MNRH and PLA.
42
In summary, the absorption band of the composites modified by silane coupling agent changed obviously, which indicated the increased C = O and Si-O-PLA content of the composites. Fourier transform infrared spectra of MRH/PLA, MRH-K5/PLA, MRH/PLA-K7 and MRH-K5/PLA-K7 wires.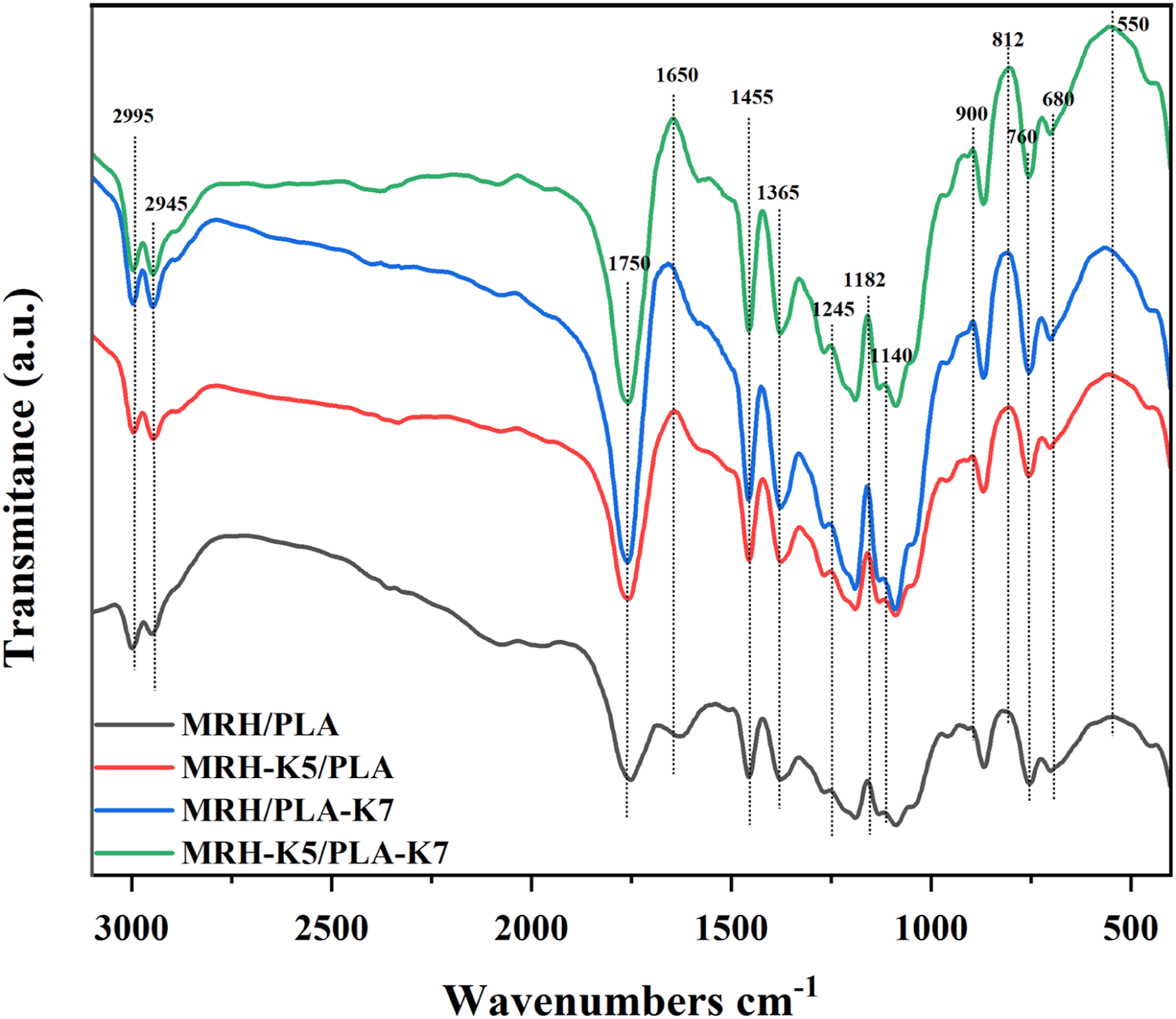
Thermal degradation characteristics
TGA data
The TG and DTG curves of MRH-K5/PLA-K7 composites at four heating rates of 5°C/min, 10°C/min, 15°C/min and 20°C/min were illustrated as an example (Figure 4). It can be seen that the TG and DTG winding moved to the right and the peak value of DTG curve became weak with the increase of heating rate. The MRH/PLA, MRH-K5/PLA, MRH/PLA-K7 and MRH-K5/PLA-K7 composites at a heating rate of 15°C/min were used as examples (Figure 5). The results showed similar phenomenon that with the growth of heating rate, the TG curve moved to the right, and the peak value of the DTG winding became weaker. In the final stage, the thermogravimetric curve flattened out, which was the residue left over from the decomposition of the composite, such as coke. The results showed that the TG of MRH-K5/PLA-K7 tended to flatline earlier with the same heating rate, and the peak value of DTG curve bacame weaked, which may be due to the higher thermal stability of the dual-modified composite. Chen et al.
44
has covered that the silane coupling agents improved the interfacial compatibility between wheat straw fiber and PLA, thus, the thermal stability is enhanced. TG (a) and DTG (b) curves of MRH-K5/PLA-K7. TG (a) and DTG (c) curves of MRH/PLA, MRH-K5/PLA, MRH/PLA-K7 and MRH-K5/PLA-K7 at the heating rate of 15°C/min,(b) is a locally enlarged view of (a),(d) is a locally enlarged view of (c).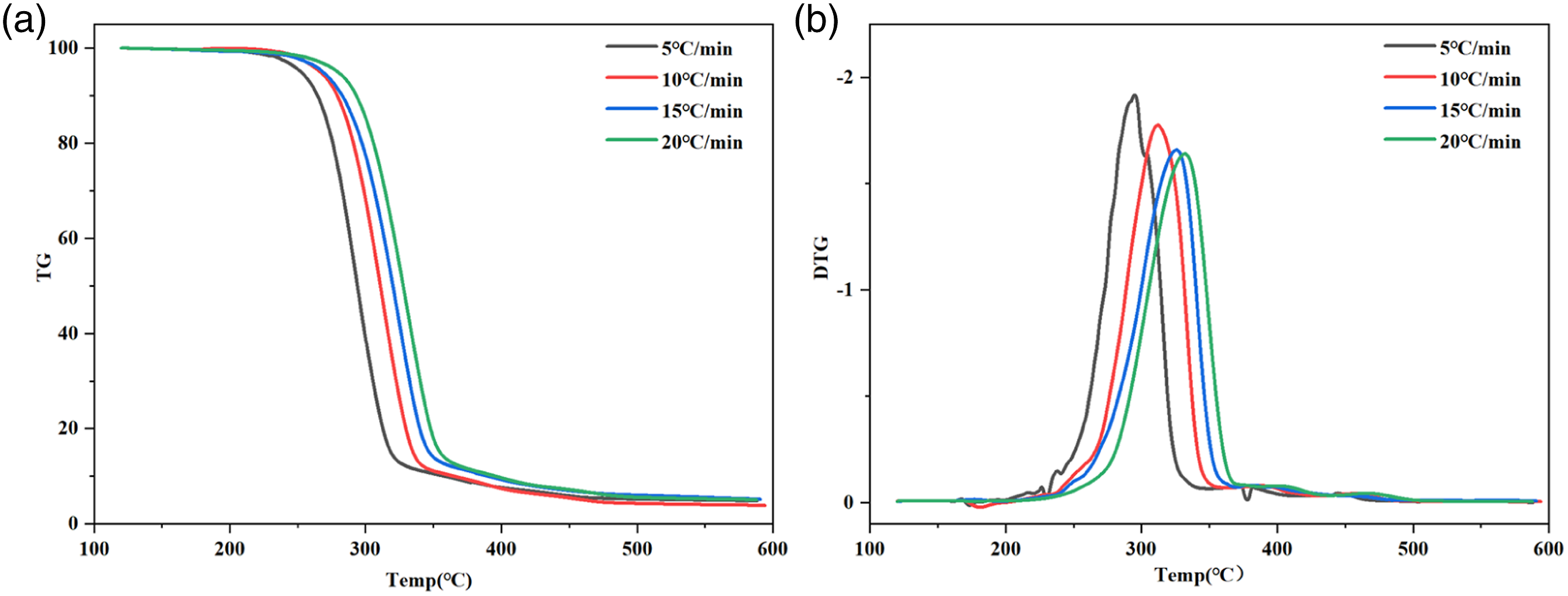
According to the TG, DTG and D2TG curves, the thermal decomposition process of the fiaments was determined by kinetic parameters. The TG and DTG of RH-K5/PLA-K7 at 15°C/min were calculated as an example (Figure 6). The initial temperature, T0, was calculated by taking the first peak value of the second derivative of TG, D2TG. The tangent line of the first derivative of TG corresponding to the first peak value, DTG, forms an intersection with the horizontal tangent line of DTG.
9
The first peak value of DTG curve represented the peak temperature, TP, in which the maximum decomposition rate occurred. Degradation termination temperature TS was determined by taking the point of the maximum peak value corresponding to D2TG. The maximum peak value corresponded to the horizontal coordinate of the intersection of the tangent line of the point on the first derivative of TG and the horizontal tangent line of DTG after decomposition.9,15 As shown in Figure 6, there were two decomposition peaks of DTG curve. The first peak occurred during the major pyrolysis stage, where most organics such as PLA, MNRH and silane coupling agent were broken down, leading to a rapid decline in quality of the material. The second peak was very weak, which corresponded to the carbonization stage of the recomposite material residue occurred. When the temperature exceeded 450°C, the residue of composites almost completely burned and the mass tended to remain unchanged. Calculation of decomposition kinetic parameters of MRH-K5/PLA-K7 at the 15°C/min heating rate.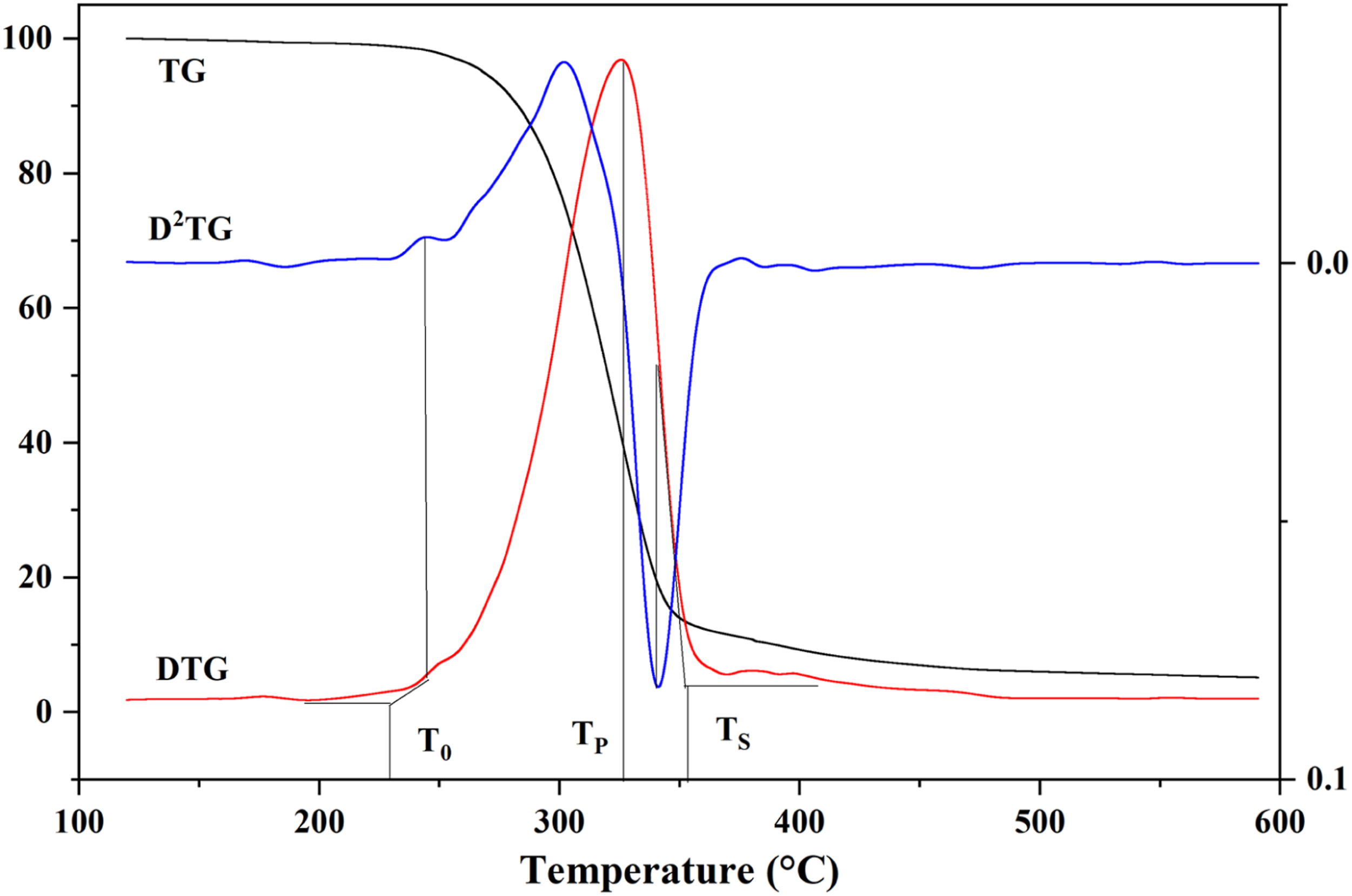
Thermodynamic parameters of four materials.
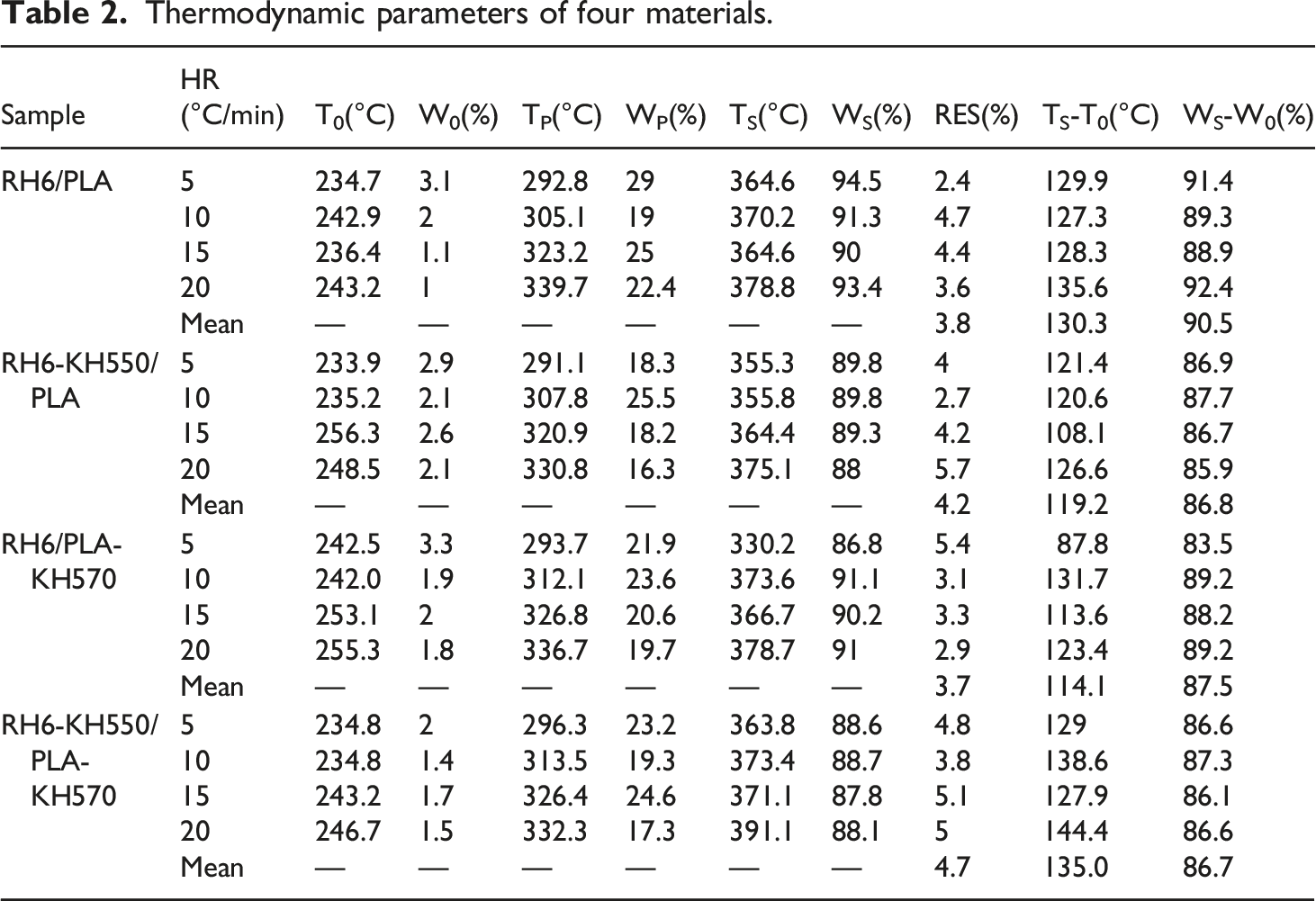
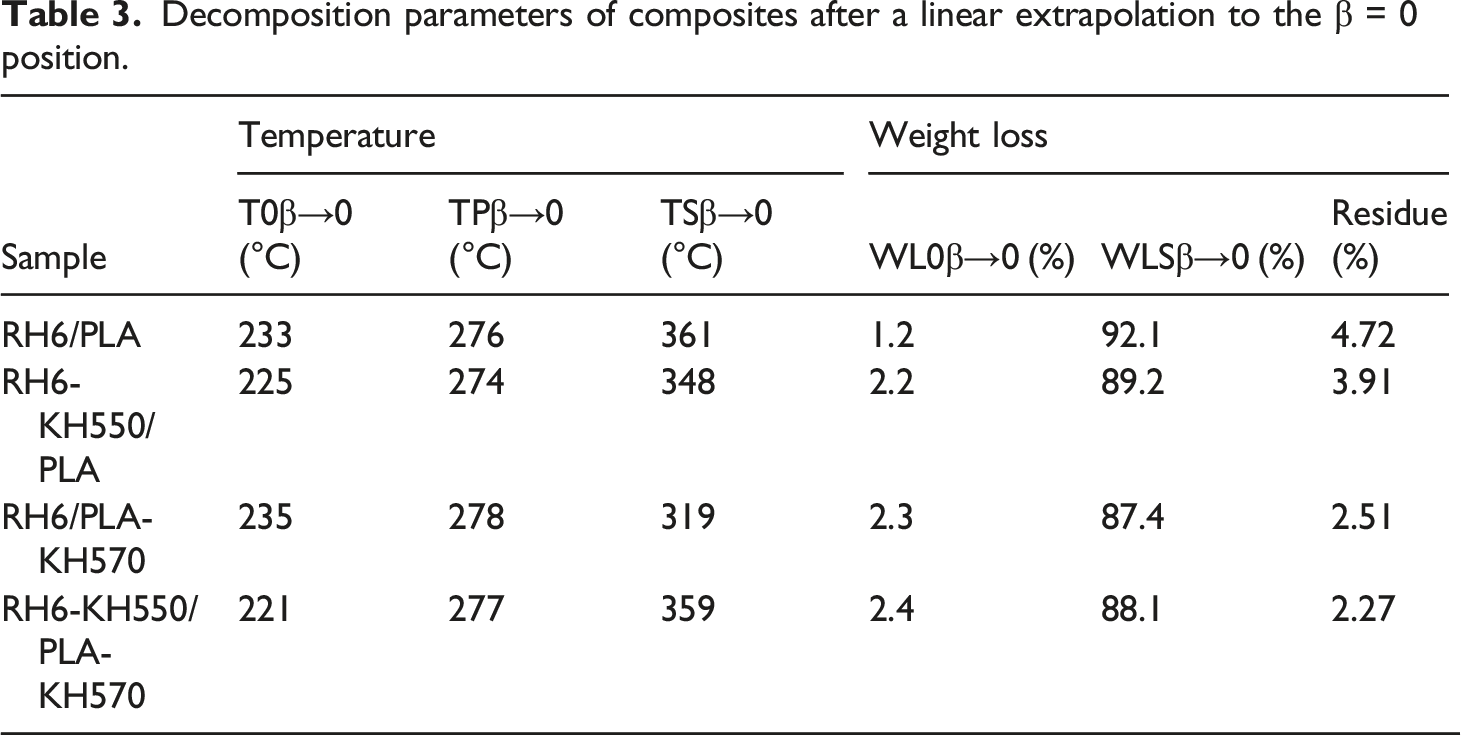
Figure 7 showed the method for calculating the characteristic parameters T0β, TPβ, TSβ. These values were obtained by extrapolating the temperature curve to the point where the heating rate β = 0. The effect of heating rate on appeal characteristic temperature can be eliminated by this method. Parameter values for the above temperature and associated weight loss were given in Table 3. Compared with MRH/PLA, MRH-K5/PLA and MRH6/PLA-K7, the residue of MRH-K5/PLA-K7 was the highest due to modification of both MNRH and PLA. The high residual amount of RH-K5/PLA-K7 indicated that RH-K5/PLA-K7 had better thermostability than other materials. Calculation of thermal decomposition kinetic characteristic parameters using MRH-K5/PLA-K7 as an example. Decomposition parameters of composites after a linear extrapolation to the β = 0 position.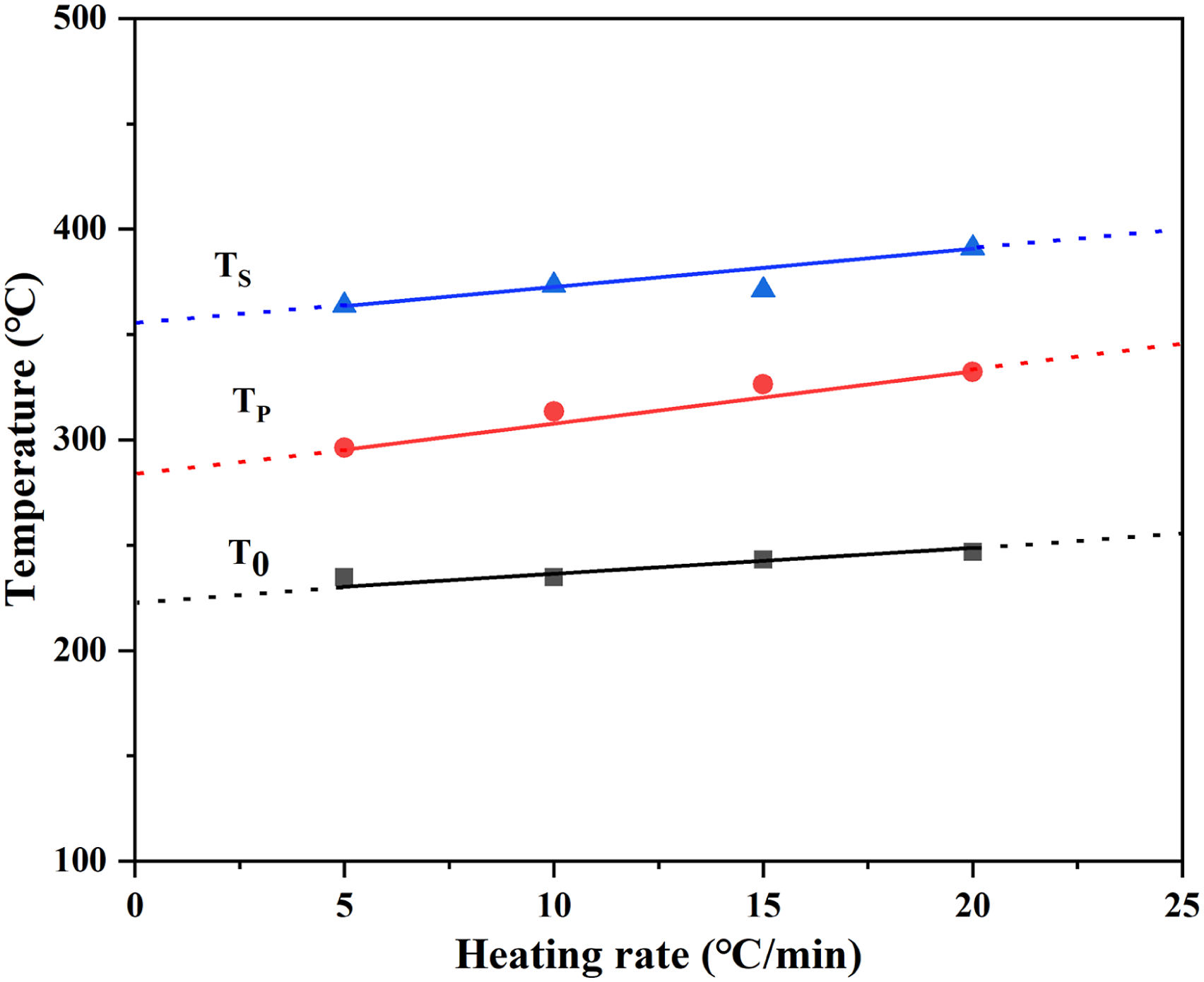
Determination of apparent activation energy
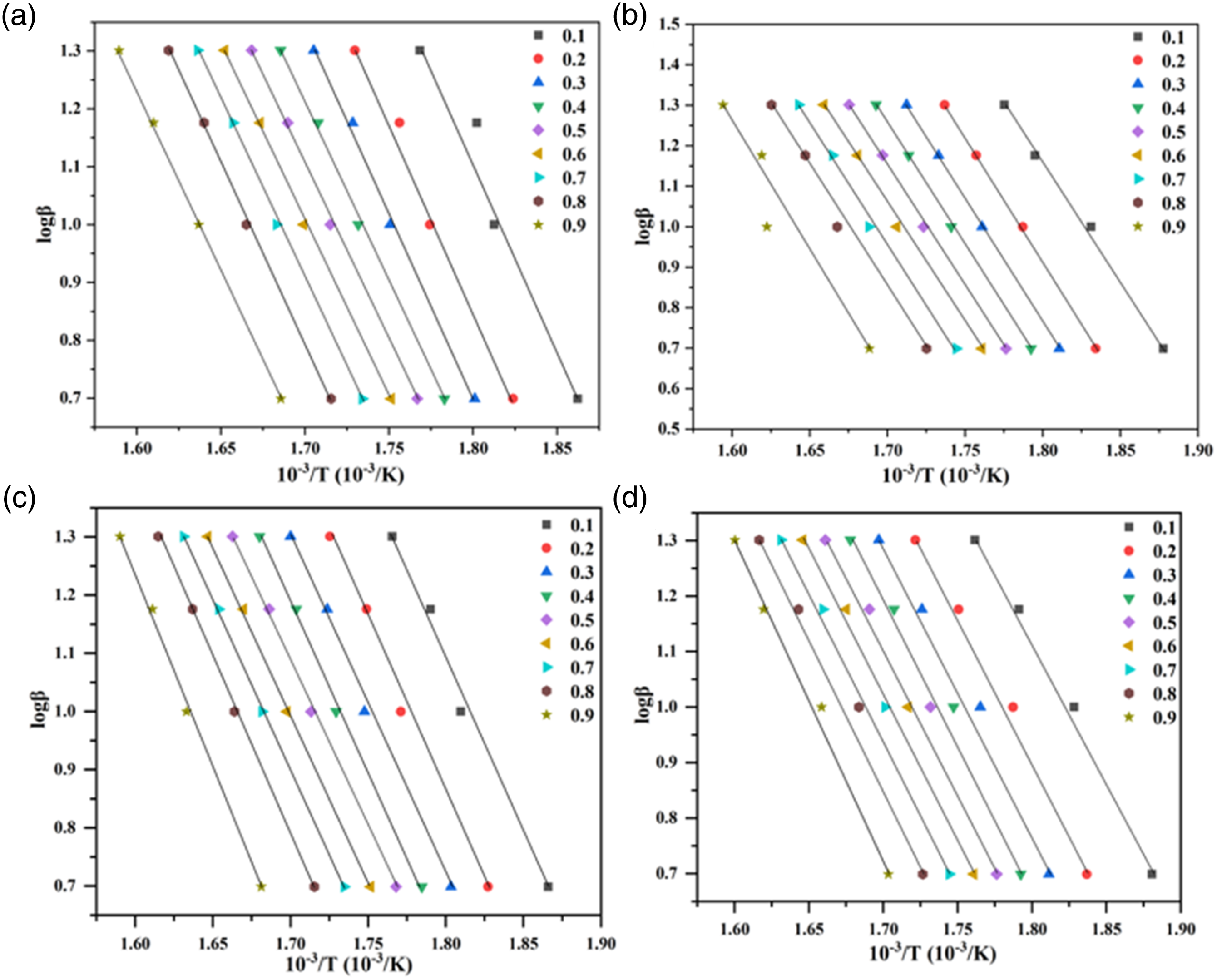
Typical dynamics curves of FD, FWO and CR methods were used to investigated the primary decomposition trend of the AAE of MRH/PLA, MRH-K5/PLA, MRH/PLA-K7 and MRH-K5/PLA-K7 composites (Figure 8–10). At conversion rate of 0.1–0.5, MRH/PLA sample curves obtained by FD method almost coincided in parallel, as shown in Figure 8(a), which indicated that AAE values at these locations were similar. After the conversion rate reached 0.6, the sample curves began to stop parallel coincidence, indicating that the thermal reaction mechanism may have varied after this stage.
9
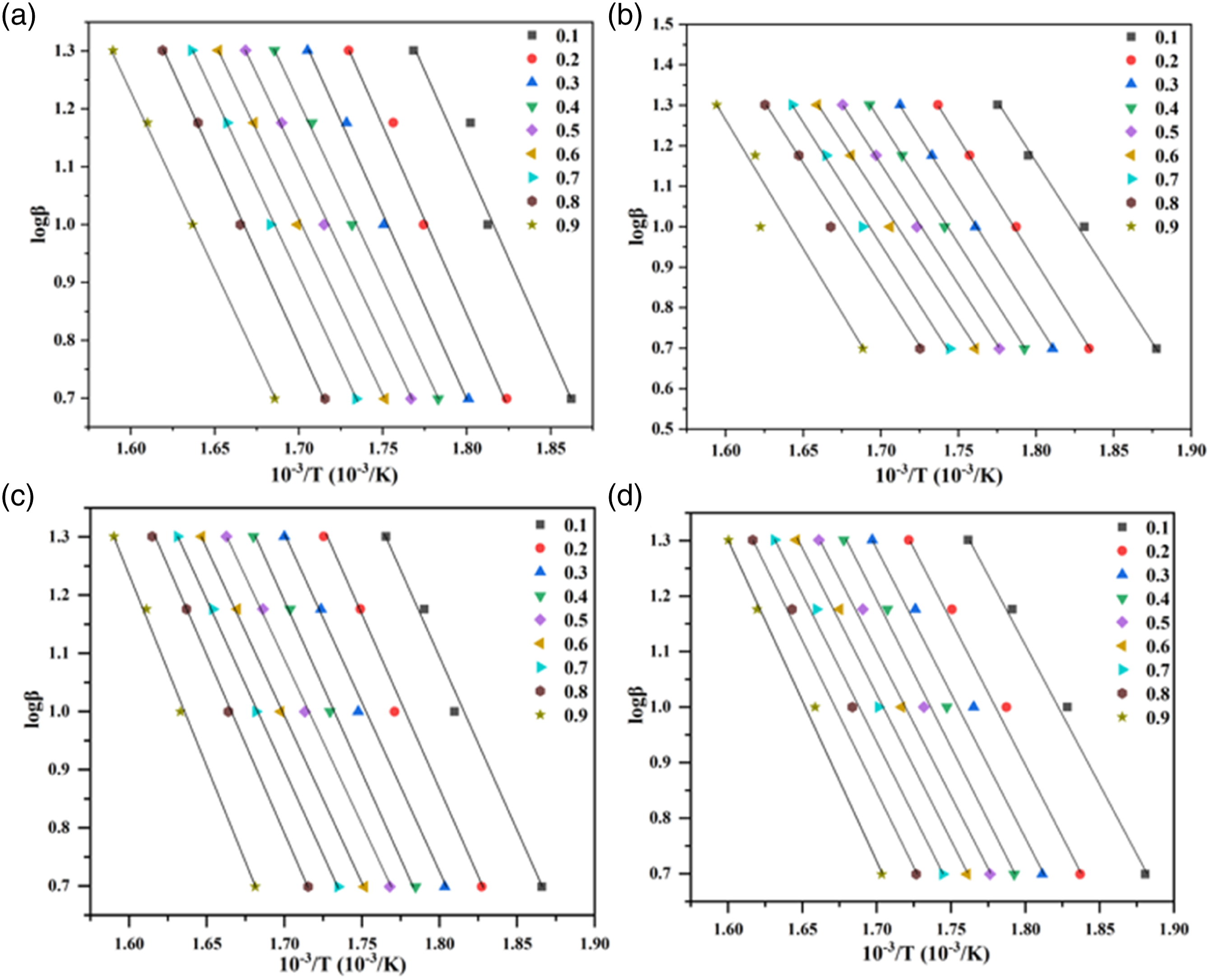
In FWO and CR models (Figures 9 and 10), the curves became parallel until a higher conversion rate (α = 0.9), indicating that there was no change in thermal reaction mechanism within the conversion rate. The curves of MRH-K5/PLA, MRH/PLA-K7 and MRH-K5/PLA-K7 composites drawn by FD, FWO and CR models were analogous to those of MRH/PLA. These results suggested the four composites had similar spectra by FD, FWO and CR methods and had similar thermal decomposition mechanisms. Iso-conversion plot for MRH/PLA, MRH-K5/PLA, MRH/PLA-K7 and MRH-K5/PLA-K7 using Friedman (FD) method (a), (b), (c), (d). Iso-conversion plot for MRH/PLA, MRH-K5/PLA, MRH/PLA-K7 and MRH-K5/PLA-K7 using Flynn–Wall–Ozawa (FWO) method (a), (b), (c), (d). Iso-conversion plot for MRH/PLA, MRH-K5/PLA, MRH/PLA-K7 and MRH-K5/PLA-K7 using Coats–Redfern (CR) method (a), (b), (c), (d).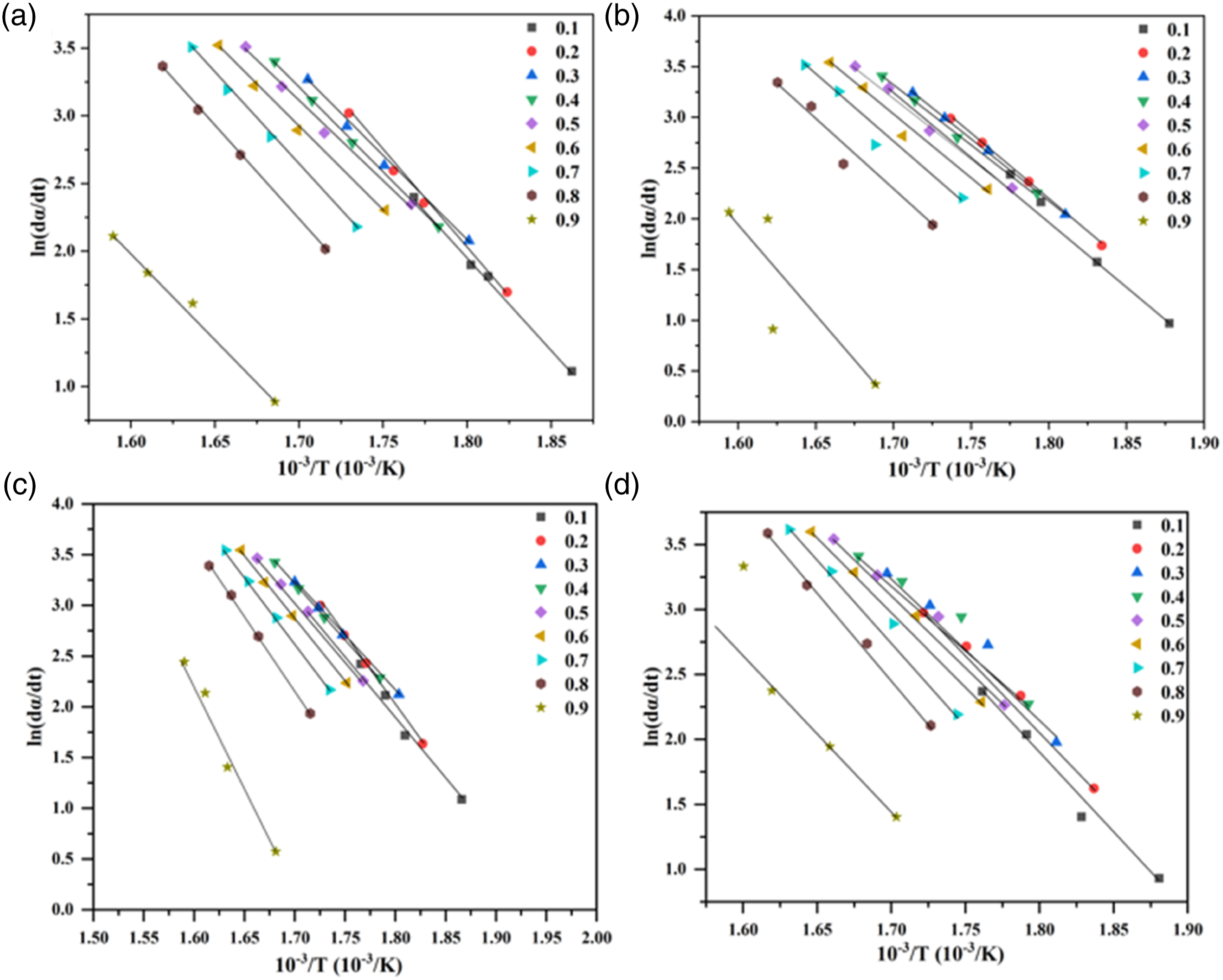
The AAE values calculated by the slopes of the three model methods at different conversion rates of MRH/PLA, MRH-K5/PLA, MRH/PLA-K7 and MRH-K5/PLA-K7 were shown in Figure 11. For MRH/PLA composite, the average AAE value measured by FD model was 103.8 kJ/mol, slightly higher than 96.8 KJ/mol measured by FWO model and 92.2 KJ/mol measured by CR model. For MRH-K5/PLA composite, the average AAE value measured by FD method was 110.9 KJ/mol, slightly higher than 110.1 KJ/mol measured by FWO model and measured 106.2 KJ/mol by CR method. For MRH/PLA-K7 composite, the average AAE value measured by FD method was 111.3 KJ/mol, slightly higher than 107.9 KJ/mol measured by FWO method and 103.8 KJ/mol measured by CR model. For MRH-K5PLA-K7 composite, the average AAE value measured by FD model was 108.1 KJ/mol, which was lower than that of FWO method (113.4 KJ/mol) and lower than that by CR method (109.6 KJ/mol). In general, the AAE values measured by a given models (FD, FWO and CR) were in a similar range for the four composites, considering the actual mixing variability and relatively low MNRH load (6% content) in the composite. As shown in Figure 11, the observed AAE values of modified composites were mainly between 90 and 120 KJ/mol, and the silane coupling agents improved the blending property and the AAE values of the PLA-based composites, indicating that the thermostability of the composite was enhanced. A comparison of AAE values at different conversion rates for MRH/PLA (1), MRH-K5/PLA (2), MRH/PLA-K7 (3), and MRH-K5/PLA-K7 (4) from FD, FWO, and CR models.
The linear curves calculated by KS model of the four materials were shown in Figure 12, and The AAE values calculated by KS model of MRH/PLA, MRH-K5/PLA, MRH/PLA-K7 and MRH-K5/PLA-K7 were 85.9 kJ/mol, 241.6 kJ/mol, 173.2 kJ/mol and 149.3 KJ/mol, respectively. In contrast with the other three methods, the AAE values of MRH/PLA composites calculated by KS method were slightly smaller, and the AAE values of MRH-K5/PLA, MRH/PLA-K7 and MRH-K5/PLA-K7 composites calculated by KS method are slightly larger. This diversity was thought to be due to the diverse modeling methods used to get AAE values. In summary, four kinetic models provided complementary technical means for the analysis of thermal stability of composites material. The AAE value was beneficial to understand the thermal decomposition process of composites under different thermodynamic models.
45
Relationship of ln(β/Tp2) versus 1/TP from the Kissinger model for MRH/PLA, MRH-K5/PLA, MRH/PLA-K7 and MRH-K5/PLA-K7 samples.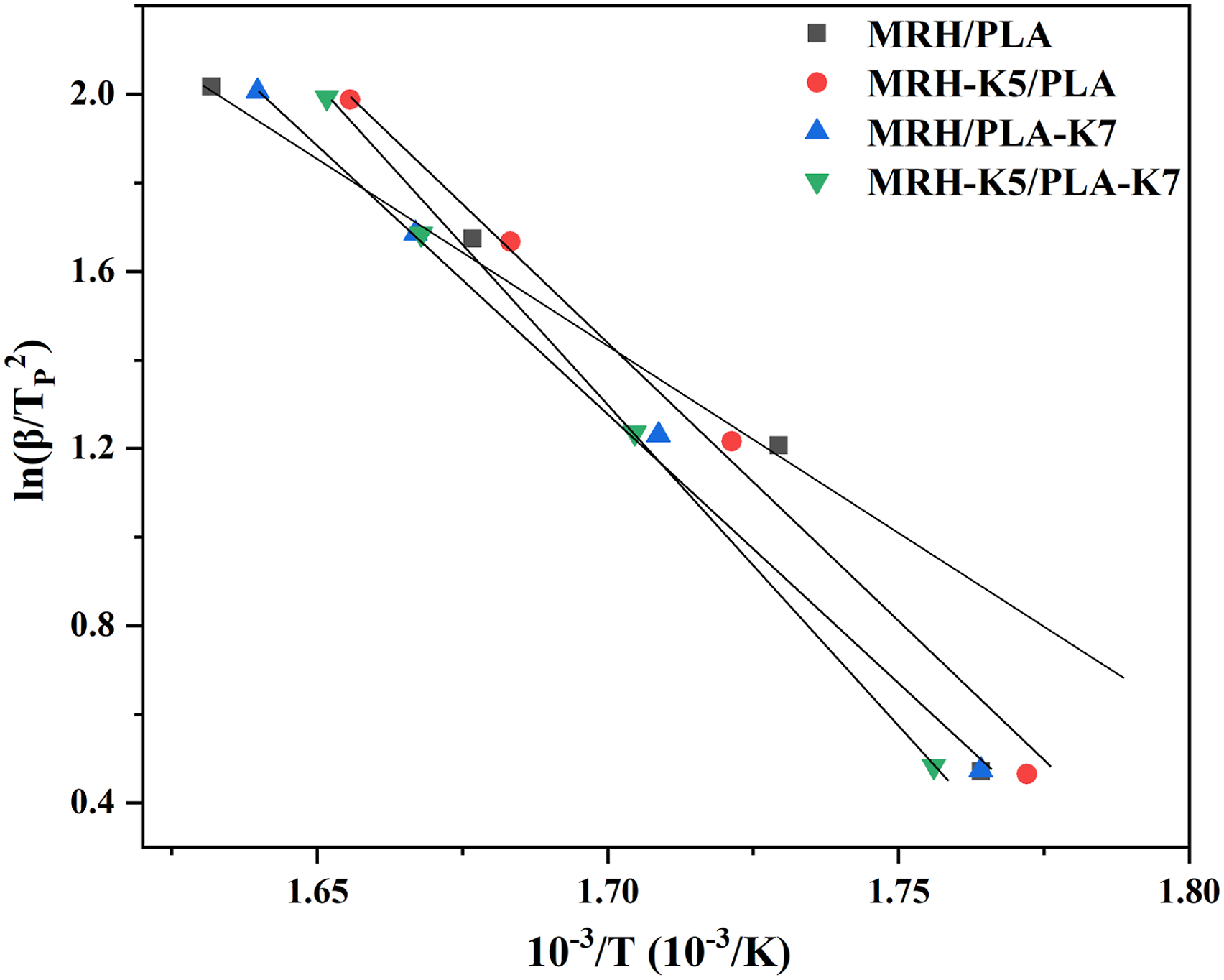
Conclusions
This study demonstrated the morphology, chemical structure and thermal decomposition performances of four 3D printed PLA-based composites. Morphological analysis showed that MNRH fibers of the MRH-K5/PLA-K7 samples were well mixed in the PLA and dispersed more homogeneous than other composites. The chemical functional group analyzed from FTIR explained that the silane coupling agents of KH550 and KH570 were successfully connected between the MNRH and the PLA. The TGA data illustrasted similar phenomenon that the TG curve moves to the right with the increase of heating rate, and the peak value of the DTG curve becomes weaker. According to the different modification methods and calculation methods of composite materials the AAE value of composites varied from 90 to 120 KJ/mol. Compared with the AAE values calculated by FD, FWO and CR models, the AAE values calculated by KS method were slightly smaller for MRH-K5/PLA, MRH/PLA-K7 and MRH-K5/PLA-K7 composites, and slightly larger for MRH-K5/PLA composites. In general, these four thermal kinetic models (FD, FWO, CR and SK) can provide a complementary technique for analyzing the thermal stability trait of PLA-based composites. The kinetic temperature and AAE values calculated by different models are helpful to better understand the thermal decomposition trait of PLA-based filaments used for 3D printing, and it will contribute to the development of MRH/PLA filaments for 3D printing in the application of automotive interior parts production. Although this study has characterized the thermal stability of PLA/MNRH, additional research is needed to fully determine its feasibility for commercial use. Specifically, future work should focus on identifying other qualifying characteristics, such as UV aging degradation, water absorption, and soiling degradation, and defining the criteria necessary to meet market demands. Such efforts will facilitate the development and commercialization of PLA/MNRH products, and create new opportunities for the use of green and sustainable materials in industry.
Footnotes
Author contributions
All authors contributed to the paper. The first draft of the manuscript was written by Yufeng Sun and Jinwei Wang, data collection, material preparation by Jinwei Wang, Yapeng Wang and Zipeng Zheng, data analysis by Bin Yang and Ziheng Zhang, Resources supported and reviewed by Wenlong Mu, Jilai Ying, Defeng Li and Guangyin Xu.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported from China Scholarship Council (Grant No. 201808410102), Henan Scientific and Technological Research Program (Grant No. 182102110027).
