Abstract
In this work, flame retarded thermoplastic polyurethane (TPU) composites were fabricated by introducing a novel flame retardant piperazine pyrophosphate. The flame retardany, thermal stability and combustion performance were systematically investigated. Flame retardant tests confirmed that thermoplastic polyurethane/piperazine pyrophosphate (TPU/PAPP) composites showed LOI of 27.5 vol% without any melt dripping when only 5 wt% PAPP was added. micro-combustion calorimetry (MCC) test implied that PAPP significantly suppressed heat release of TPU/PAPP composites. 10 wt% of PAPP endow PHRR1 and THR values TPU/PAPP10 decreased by 67.0% and 35.1% compared with TPU. Thermogravimetric-Fourier transform infrared spectroscopy (TG-FTIR) showed that PAPP significantly inhibited the release of toxic and combustible gases. The char residue investigation showed that TPU/PAPP composites formed compact char residue, which was able to effectively inhibit the heat and mass transmission in combustion. Based on the investigation, the work mechanism of PAPP in TPU/PAPP composites was proposed.
Introduction
Thermoplastic polyurethane (TPU) is widely used in many fields, including wire and cable, footwear, clothing, and national defense because of its abrasion resistance, low-temperature resistance, tear resistance, insulation, and ozone resistance.1,2 However, as a organic polymer, TPU exhibits poor flame retardancy with violent dripping phenomenon and toxic gases release in combustion, which seriously threatens people’s lives and property safety.3,4 Therefore, the development of efficient strategy to improve the fire retardation of TPU has attracted much attention in this field.
There are two methods to enhancing flame retardancy of TPU, the additive flame retardancy and inherent flame retardant.5–7 In these two ways, additive flame retardancy strategy usually incorporated flame retardant into TPU matrix by melt blending method, which was mostly used for the advantages of low cost and convenient fabrication. Currently, the commonly used additive flame retardants includes red phosphorus (RP), ammonium polyphosphate (APP), aluminum hypophosphate (AHP), intumescent flame retardants (IFR), inorganic metal hydroxides, melamine salts, et al.8–10 Among them, APP is the focus of research and development in the field of flame retardant. Chen et al. prepared microencapsulated ammonium polyphosphate (MAPP) by in-situ polymerization of hydroxyl silicone oil, and studied the effects of APP and MAPP on the flame retardancy of TPU respectively. The results showed that the LOI value of the composites containing 10% (or 20%) MAPP was 25.5 vol% (or 29.3 vol%), while the LOI values of the composites with the same amount of APP were 25.1 vol% and 28.0 vol%. 11 With the high standard of material performance required by the regulatory authorities, piperazine pyrophosphate, a phosphorus-nitrogen halogen-free flame retardant similar to APP has attract much attention by researchers. It has used in polypropylene, thermoplastic elastomer, polyamide six et al., and good fire retarding were observed. Xiao et al fabricated flame retardant glass fiber reinforced PA6 composites with addition of PAPP and aluminum hypophosphite (AHP). It was observed that PA6 composites with 16 wt% PAPP and 4wt% AHP loading exhibited LOI of 34.0 vol% and UL-94 V-0 rating. 11 Chen et al. combined PAPP with melamine phosphate (MPP) and TiO2 to form a synergistic flame retardant system, which was further applied to thermoplastic elastomer (TPE). The TPE composites exhibited LOI of 37.8 vol% and successfully passed UL-94 V-0 rating when 40 wt% PAPP/MPP/TiO2 was incorporated. 12 Bourbigot et al. combined PAPP with zinc borate (ZnB) to enhance flame retardancy of polybutylene succinate (PBS) composites. When 15 wt% PAPP and 5 wt% ZnB were added, PBS composites was endowed UL-94 V-0 rating with pHRR value decreased by 78.05% compared with unmodified TPE. 13
The above investigation confirmed that PAPP was a good candidate for enhancing flame retardancy of polymers. However, there was few report about the application of PAPP in TPU. Herein, this work tried to investigate the flame retarding effect of PAPP in TPU composites and compare the flame retardant effect of APP in TPU. Firstly, PAPP was incorporated into TPU to fabricate a series of TPU/PAPP composites by melt mixing method. Furthermore, the flame retardant properties, thermal stability, combustion properties, and microscopic morphology of TPU/PAPP composites were investigated by limiting oxygen index (LOI), vertical combustion (UL-94), micro-combustion calorimetry (MCC), scanning electron microscopy (SEM). And also, based on the investigation, the work mechanism of PAPP in TPU/PAPP composites was discussed. This work pointed a new way for preparation TPU composites with outstanding flame retardancy.
Experimental part
Experimental raw materials
Thermoplastic polyurethane (TPU, E8185) was purchased from Baoding Bangtai Polymeric New Materials Co., Ltd. Piperazine pyrophosphate (PAPP) was purchased from Hainan Zhongxin Chemicals Co., Ltd.
Preparation of TPU/PAPP composites
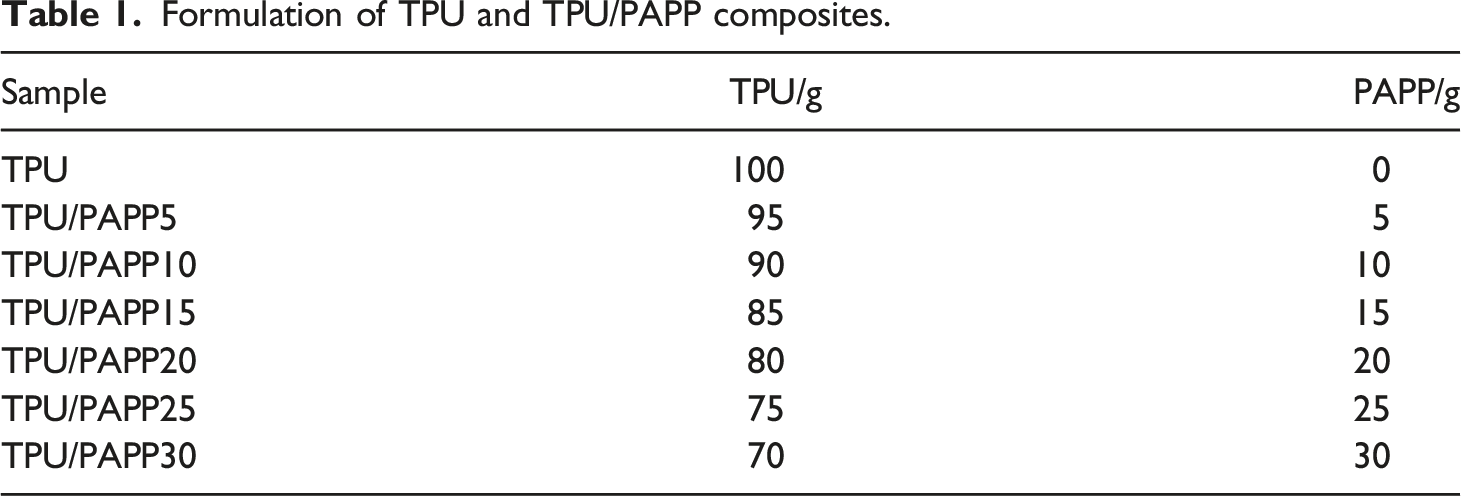
Formulation of TPU and TPU/PAPP composites.
Testing and characterization methods
LOI: The CS1-101G oxygen index meter (Shanghai Chengshi Intelligent Technology Co., Ltd.) was used to test the limit oxygen index of TPU and TPU/PAPP composites according to ASTM D2863 with sample dimension of 100 mm × 6.5 mm × 3 mm.
Vertical combustion (UL-94): The ZOT-6053 horizontal vertical combustion tester was used to test the vertical combustion level of TPU and TPU/PAPP composites according to ASTM D3801 with sample dimension of 100 mm × 10 mm × 3 mm.
Thermogravimetric (TG): The Q5000 thermal analyzer was introduced to record the thermal weight loss of samples within a nitrogen atmosphere and a rate of 20°C/min from room temperature to 800°C. sample mass, 5–10 mg.
Microcalorimetry (MCC): The GOVMARK MCC-2 MCC was characterized the combustion properties of TPU and TPU/PAPP composites according to ASTM D 730908. In the test, 4–6 mg of sample was heat from 100 to 700°C with heating rate of 1°C/s and nitrogen flowing of 80 mL/min. The combustion products in the nitrogen gas steam were mixed with 20 mL/min of oxygen before entering 900°C furnace.
Thermogravimetric-Fourier transform infrared spectroscopy (TG-FTIR): A TGA Q500 infrared thermogravimetric analyzer connected to a Nicolet 6700 FTIR spectrophotometer was used. The samples were tested the thermal weight loss and gas release from room temperature to 800°C at 20°C/min under a nitrogen atmosphere.
SEM: The JSM-6510 SEM was used to observe the microscopic morphology of char residue for TPU and TPU/PAPP composites. The sample was coated with a conductive layer before the test. The work voltage was 10 kV.
Fourier Infrared (FTIR): The char residue of TPU and TPU/PAPP composites was mixed with potassium bromide (KBr) in a mortar, and then the tablet was obtained by press process, which was then tested in Nicolet IS5 infrared spectrometer. The test range was 4000–400 cm−1.
X-ray photoelectron spectroscopy (XPS): The VG ESCALAB II spectrometer was used to analyze the elements content of char residue for TPU and TPU/PAPP composites under ultra-high vacuum with Al Kα laser radiation (hv = 1253.6 eV).
Results and discussion
Flame retardant property of TPU and TPU/PAPP composites
LOI and UL-94 test results of TPU and TPU/PAPP composites.
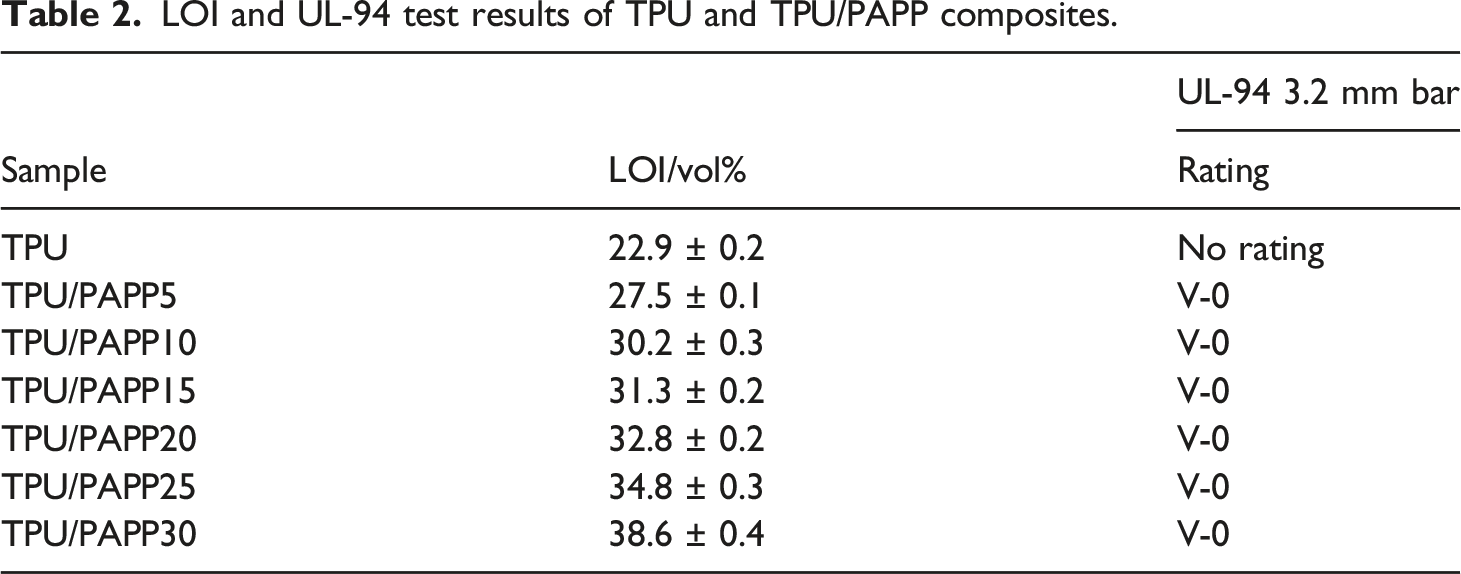
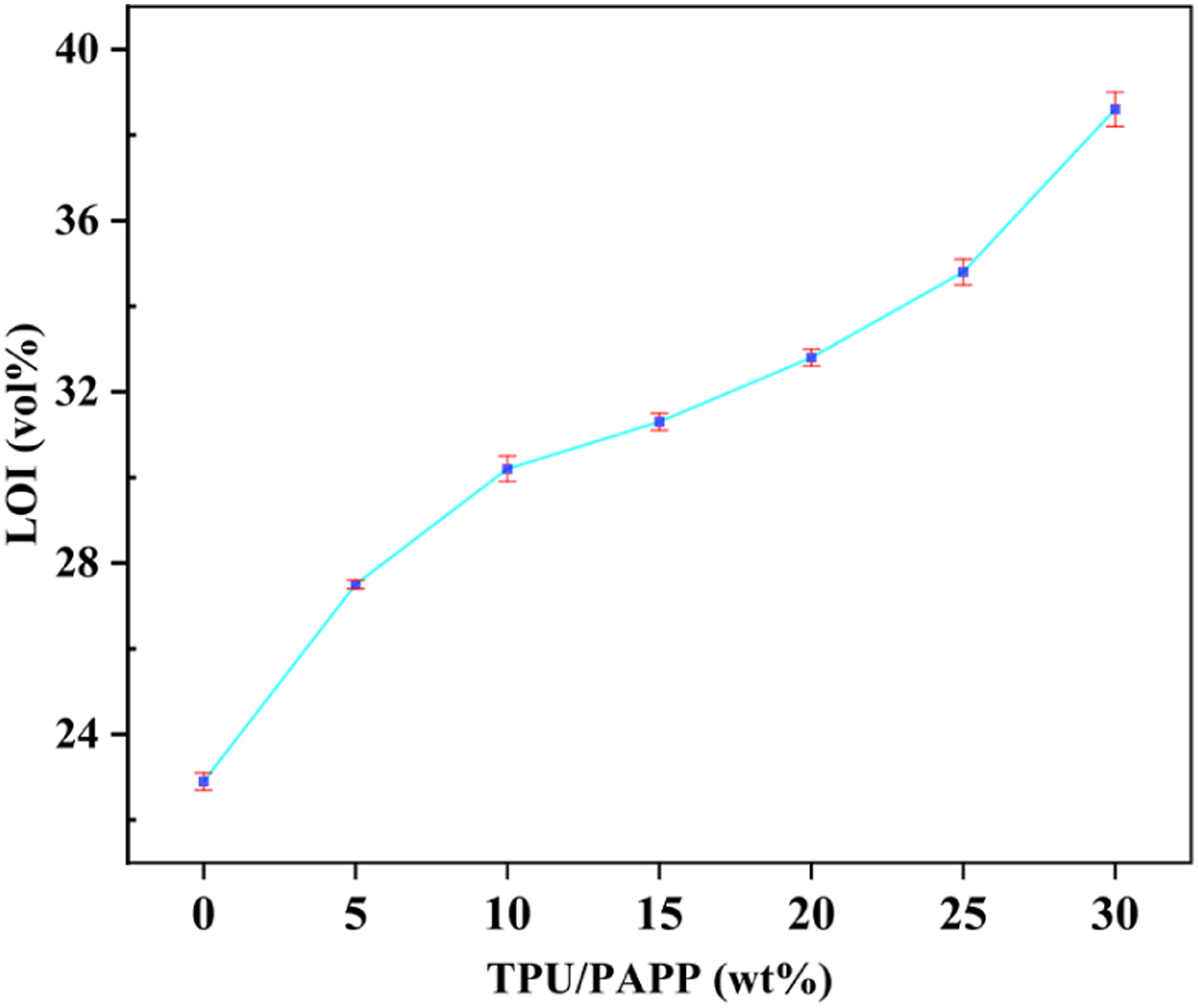
Variation of LOI value of TPU and TPU/PAPP composites with the addition of PAPP.
MCC test of TPU and TPU/PAPP composites
MCC is an important thermal analysis to investigate heat release and thermal stability of polymer composite, which only need milligrams of samples.
16
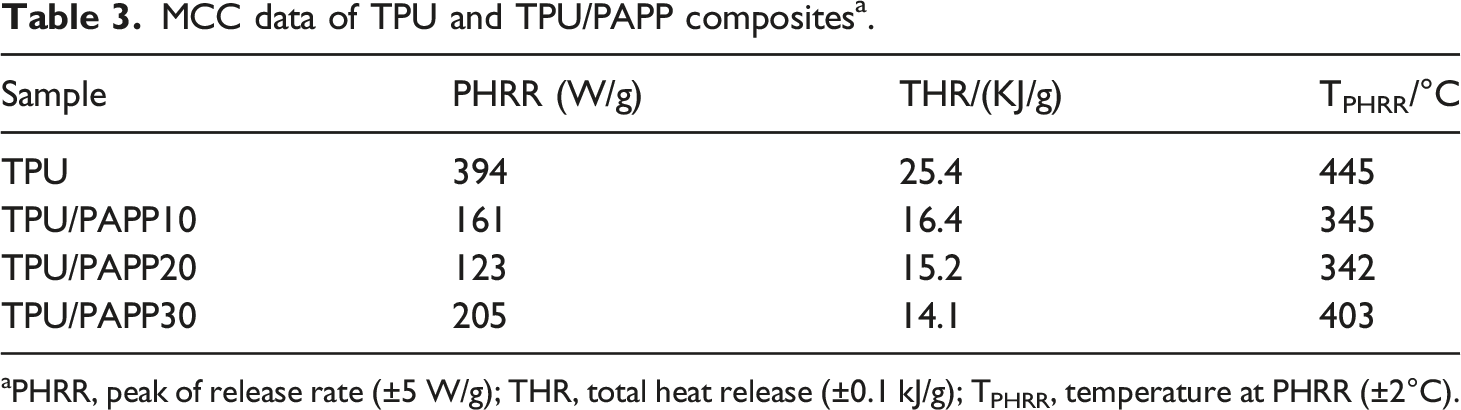
Figure 2 showed the HRR curves of TPU and TPU/PAPP composites under MCC test, the peak heat release rate (PHRR), temperature of peak heat release rate (TPHRR) and total heat release (THR) of TPU and TPU/PAPP composites were listed in Table 3. TPU showed PHRR value of 394 W/g at around 445°C, which were ascribed to hard segment and soft segment of polyurethane molecular chain, respectively.
17
TPU/PAPP composites also had two heat release peaks, and there was an obvious inflection point after the first heat release peak decreased, it was attributed to the breakage of the P-OH bond in the initial degradation of PAPP, thus reducing the initial stability of the composites.
18
With the increase of temperature, the char layer was broken fast and HRR reached another peak. It could be clearly observed from the figure that the addition of PAPP significantly reduced the peak HRR, and the PHRR of TPU/PAPP10, TPU/PAPP20 and TPU/PAPP30 composites decreased by 67.0%, 69.3% and 48.0% respectively compared to TPU. HRR curve of TPU and TPU/PAPP composites. MCC data of TPU and TPU/PAPP composites
a
. aPHRR, peak of release rate (±5 W/g); THR, total heat release (±0.1 kJ/g); TPHRR, temperature at PHRR (±2°C).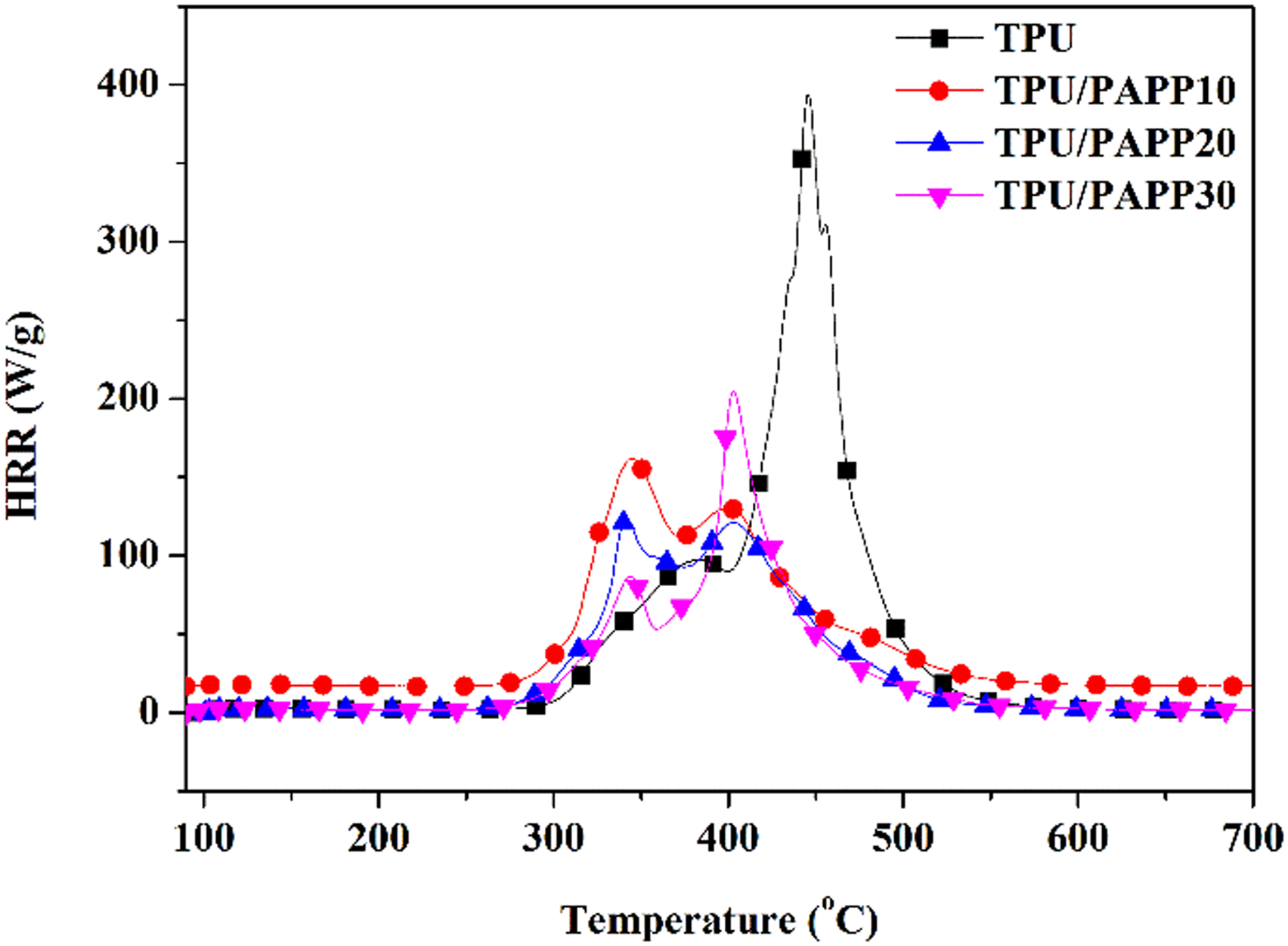
Thermal stability of TPU and TPU/PAPP composites
Thermogravimetric analysis is a common method to measure the thermal stability of polymer composites.
19
From the MCC results, it could be concluded that the TPU samples were prone to release flammable gases, and more flammable gases were released when the HRR reaches its peak. Therefore, the thermal stability of TPU samples was further investigated using TGA test. As observed from Figure 3, the thermal decomposition process of the TPU composites included two stages. The first stage from 250 to 370°C was attributed to degradation of the hard segment for polyurethane molecular chain, and the second stage from 370 to 500°C corresponded to soft segments decomposition.
20
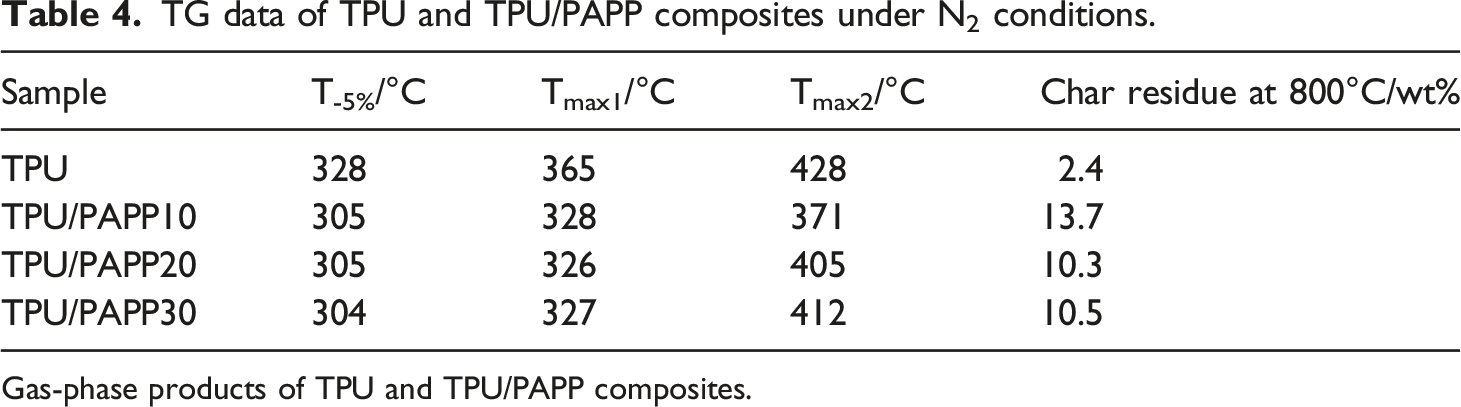
From Table 4, it was known that the initial decomposition temperature (T-5%, the temperature corresponding to 5% mass loss), Tmax1 (the temperature at maximal mass loss rate at first stage), and Tmax2 (the temperature at maximal mass loss rate at second stage) of TPU composites were 328°C, 365°C, and 428°C, respectively. When 5wt% PAPP was incorporated, TPU/PAPP5 exhibited decreased T-5%, Tmax1, and Tmax2 values of 305°C, 328°C and 371°C, respectively. It was mainly due to the acidic substances such as poly(metaphosphoric acid) generated by the combustion of PAPP promoted the decomposition of polyurethane molecular chains. With the further enhance of PAPP loading, the T-5% and Tmax1 values of TPU/PAPP composites were slightly changed, while the Tmax2 values were enhanced significantly. And also, it was observed that TPU/PAPP composites exhibited much higher char residues compared with TPU, which was attributed to carbonization effect of poly(metaphosphoric acid) come from the decomposition of PAPP.
21
Meanwhile, the residual carbon rate of TPU was 2.4 wt% at 800°C, while the residual carbon rates of TPU/PAPP10, TPU/PAPP20 and TPU/PAPP30 composites were 13.7 wt%, 10.3 wt% and 10.5 wt%, respectively, indicating that the introduction of PAPP enhanced the high-temperature stability of TPU composites. TG (a) and DTG (b) curves of TPU and TPU/PAPP composites. TG data of TPU and TPU/PAPP composites under N2 conditions. Gas-phase products of TPU and TPU/PAPP composites.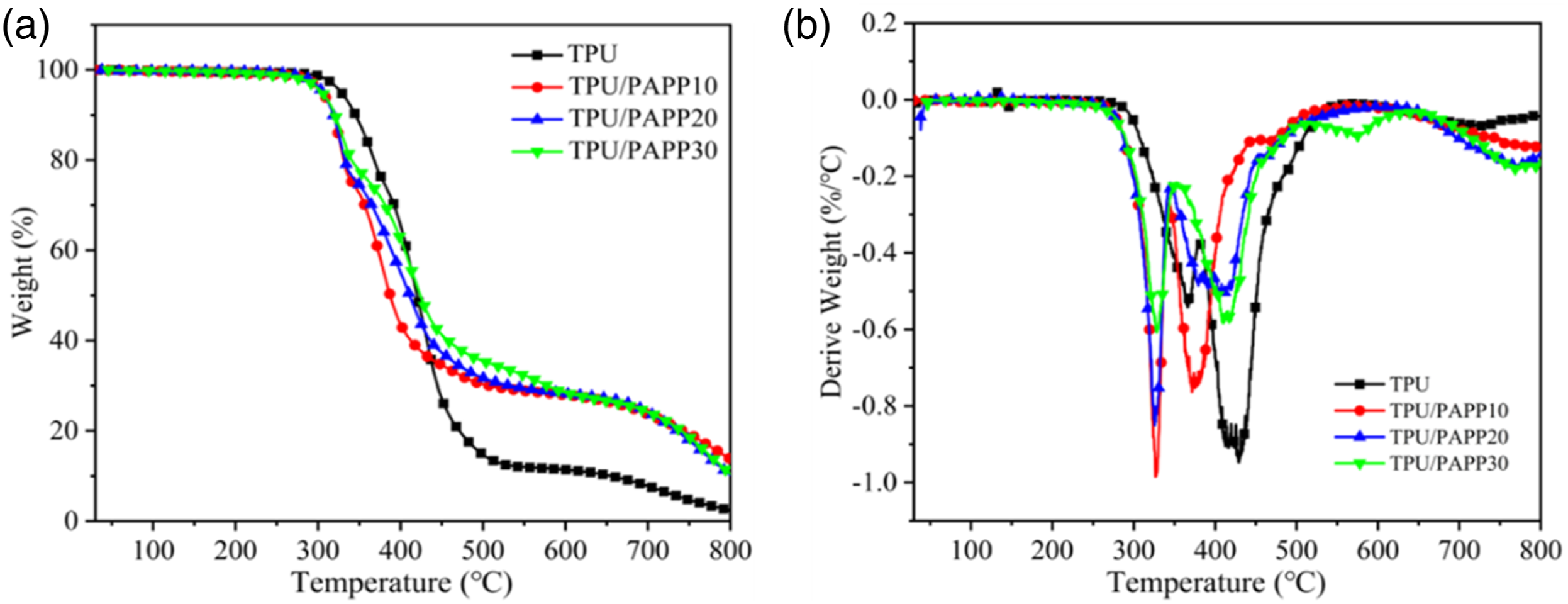
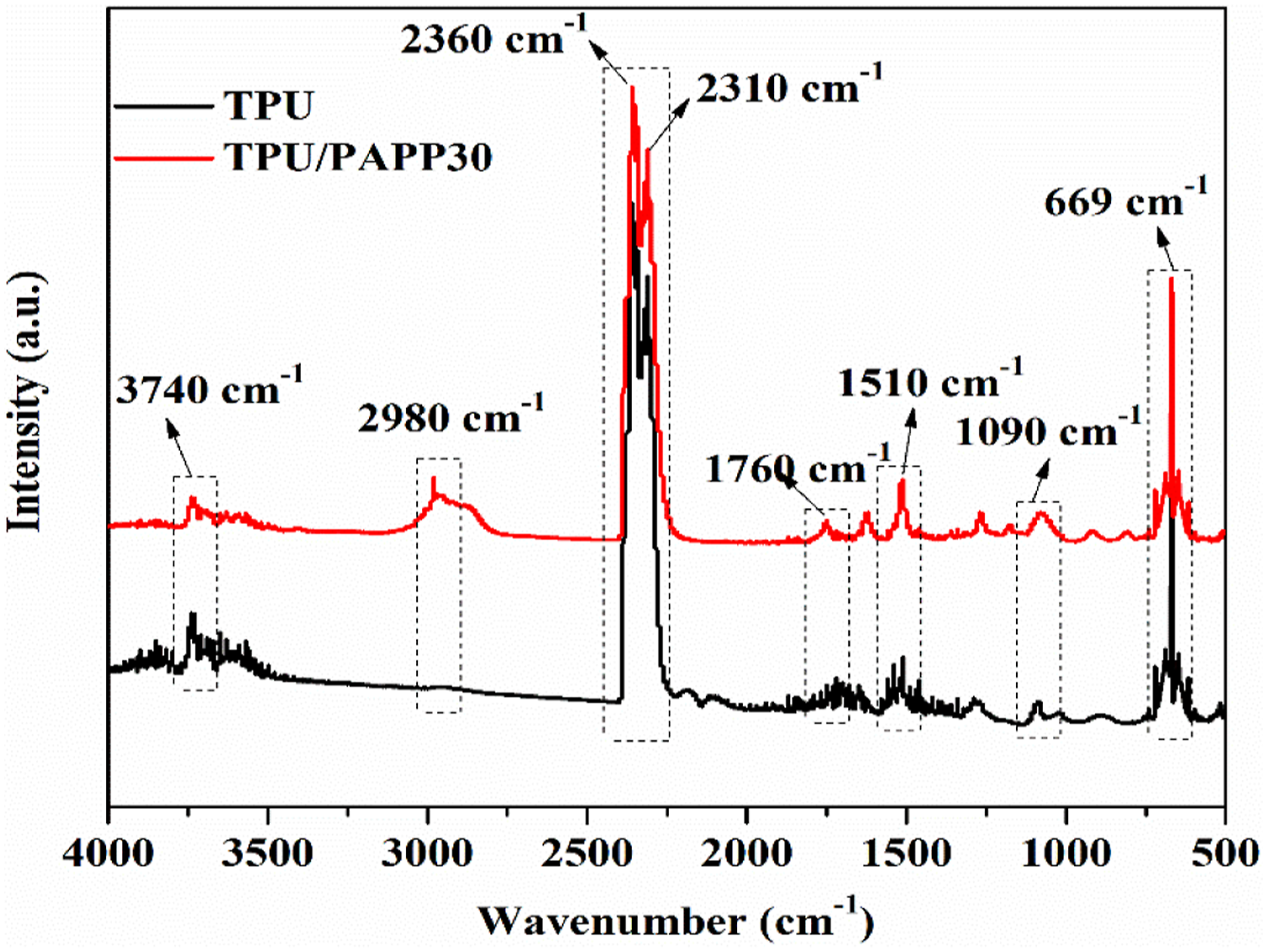
TG-FTIR was often used to characterize gas products of polymer composites for flame retardant mechanism investigation.22,23 The gas product release intensities of TPU and TPU/PAPP30 was tested using the TG-FTIR technique. Figure 4 was the 3D spectra of gas phase products for the samples. It could be seen that there was a significant change in the peak intensity of the gas phase products release of TPU/PAPP30 compared with those of TPU. The gas phase products band of the composites were mainly distributed in the range of 3500 cm−1 ∼ 4000 cm−1, 2700 cm−1 ∼ 3200 cm−1, 2000 cm−1 ∼ 2500 cm−1, 1600 cm−1 ∼ 1800 cm−1, 1400 cm−1 ∼ 1500 cm−1, 900 cm−1 ∼ 1200 cm−1, 650-750 cm−1. Figure 5 presented FTIR spectra of gas phase products released from TPU and TPU/PAPP30 at the maximum thermal mass loss rate, where significant absorption peaks were observed at 3740 cm−1, 2980 cm−1, 2360 cm−1, 2310 cm−1, 1760 cm−1, 1510 cm−1, 1090 cm−1, 669 cm−1, corresponding to carbamates, hydrocarbons, CO2, isocyanate compounds, carbonyl compounds, aromatic compounds, esters, and HCN, respectively.
24
3D-FTIR spectra of TPU (a) and TPU/PAPP30 (b). TG-FTIR spectra of gas products for TPU and TPU/PAPP30 at maximal decomposition rate.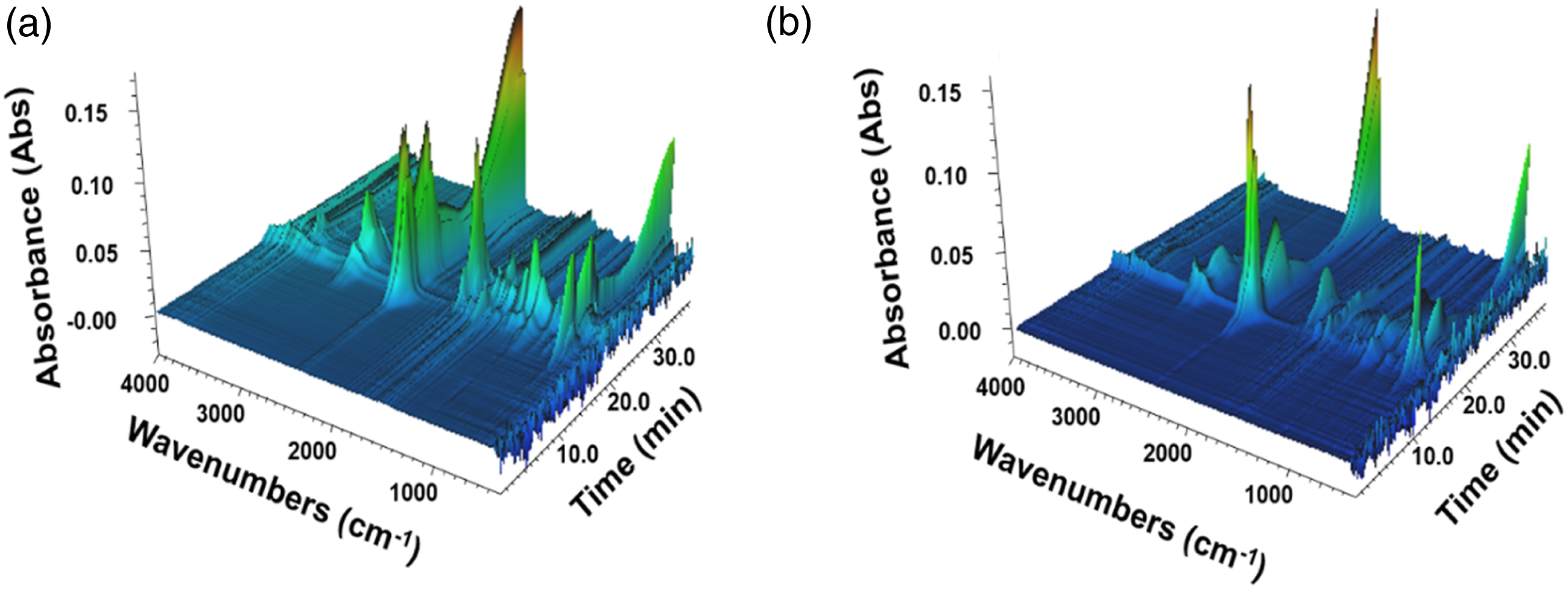
Further more, quantitative analysis of the release intensity of typical gas phase products of TPU and TPU/PAPP30 composites per unit mass as a function of time was shown in Figure 6. It could be seen that the gas products of TPU and TPU/PAPP30 composites were released around 750 s, but the release rate of TPU/PAPP30 was stronger than that of TPU, indicating that the addition of PAPP promotes the early decomposition of the composites. Absorbance of pyrolysis products of TPU and TPU/PAPP versus time: (a) urethane; (b) CO2; (c) carbonyl compound; (d) aromatic compound; (e) esters; (f) HCN.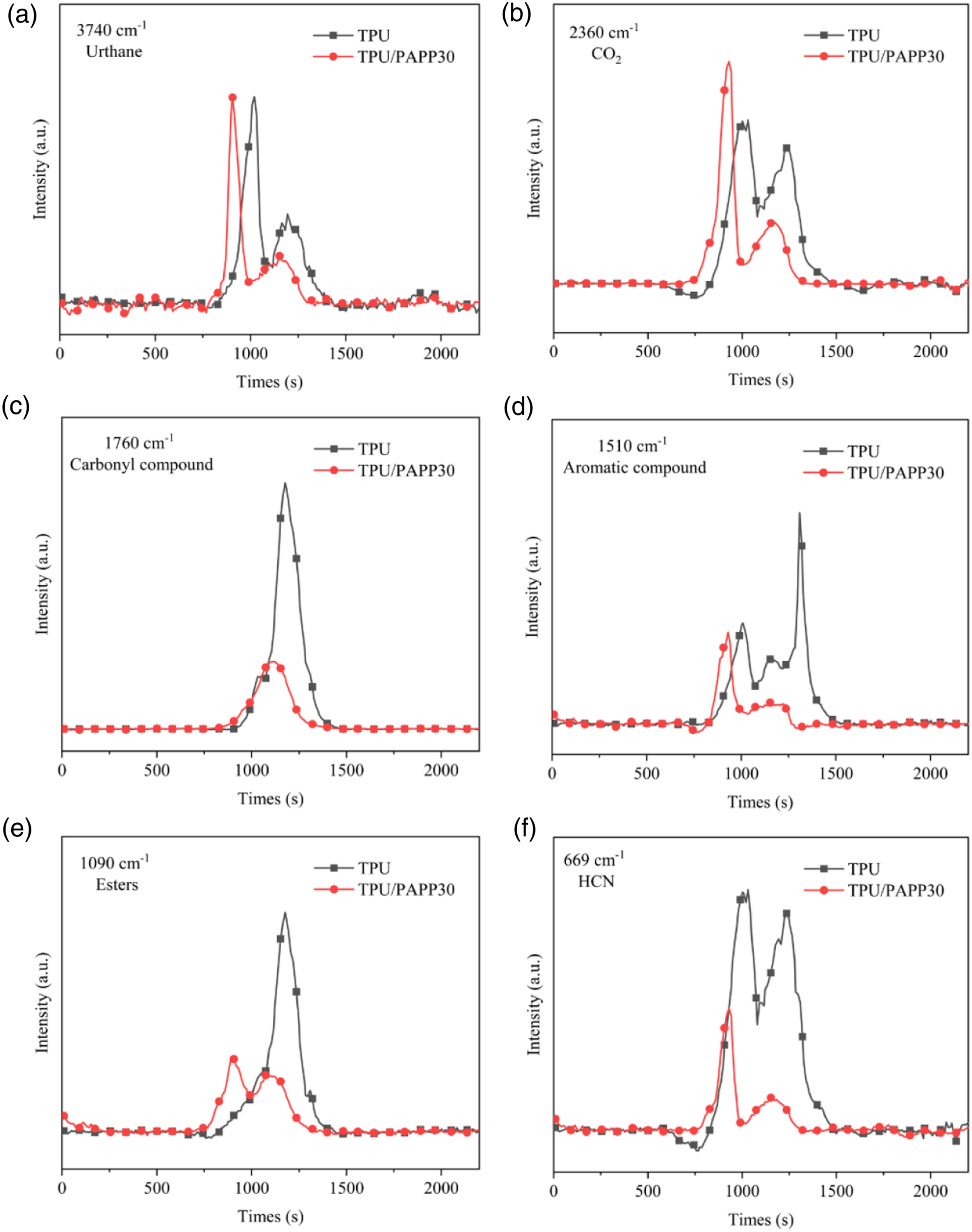
Char residue investigation of TPU and TPU/PAPP composites
Char residue analysis is an invaluable method to evaluate the flame retardant mechanism of composites.
25
Which was obtained by calcining in a muffle furnace at 600°C for 10 min. As observed in Figure 7(a), the char layer of TPU had a large number of honeycomb-like pore structures. When 10 wt% PAPP was added, Macro-continuity of char layer for the TPU/PAPP10 was significantly improved, but there were still many pore structures on the surface of the char layer (Figure 7(b)). Further increasing the content of PAPP, the pore structure on the surface of the char layer for TPU/PAPP20 was further reduced compared to TPU/PAPP10 (Figure 7(c)). When 30 wt% PAPP was loaded, TPU/PAPP30 exhibited the thickest char layer (encircled part) without any pore structure, and almost no pores with large size were observed on the surface of the char layer of TPU/PAPP30 composite (Figure 7(d)). From the above investigation, PAPP could effectively improve the char layer structure of composite materials. SEM images of char residues for TPU and TPU/PAPP composites: (a) TPU; (b) TPU/PAPP10; (c) TPU/PAPP20; (d) TPU/PAPP30.
Figure 8 exhibited the FTIR spectra of char residue for TPU and TPU/PAPP composites. It was observed that there were obvious changes of char residue for TPU/PAPP composites compared with TPU. The intensity of absorption peaks at 3430 cm−1 (N-H bond stretching vibration) and 1640 cm−1 (C = C bond stretching vibration) was enhanced with the increase of PAPP content. The C = C bond was benefit to form aromatic ring and compact char residue. And also, some new bands were observed in char residue of TPU/PAPP composites. The peaks at 2400 cm−1, 1150 cm−1, and 1008 cm−1corresponded to the stretching vibrations of P-H, P-O-P, and P-O-N, respectively.26,27 P-O-P and P-O-N structures further enhanced the dehydration and charring ability of TPU composites during combustion, resulting in better flame retardancy. FTIR spectra of char residues for TPU and TPU/PAPP composites.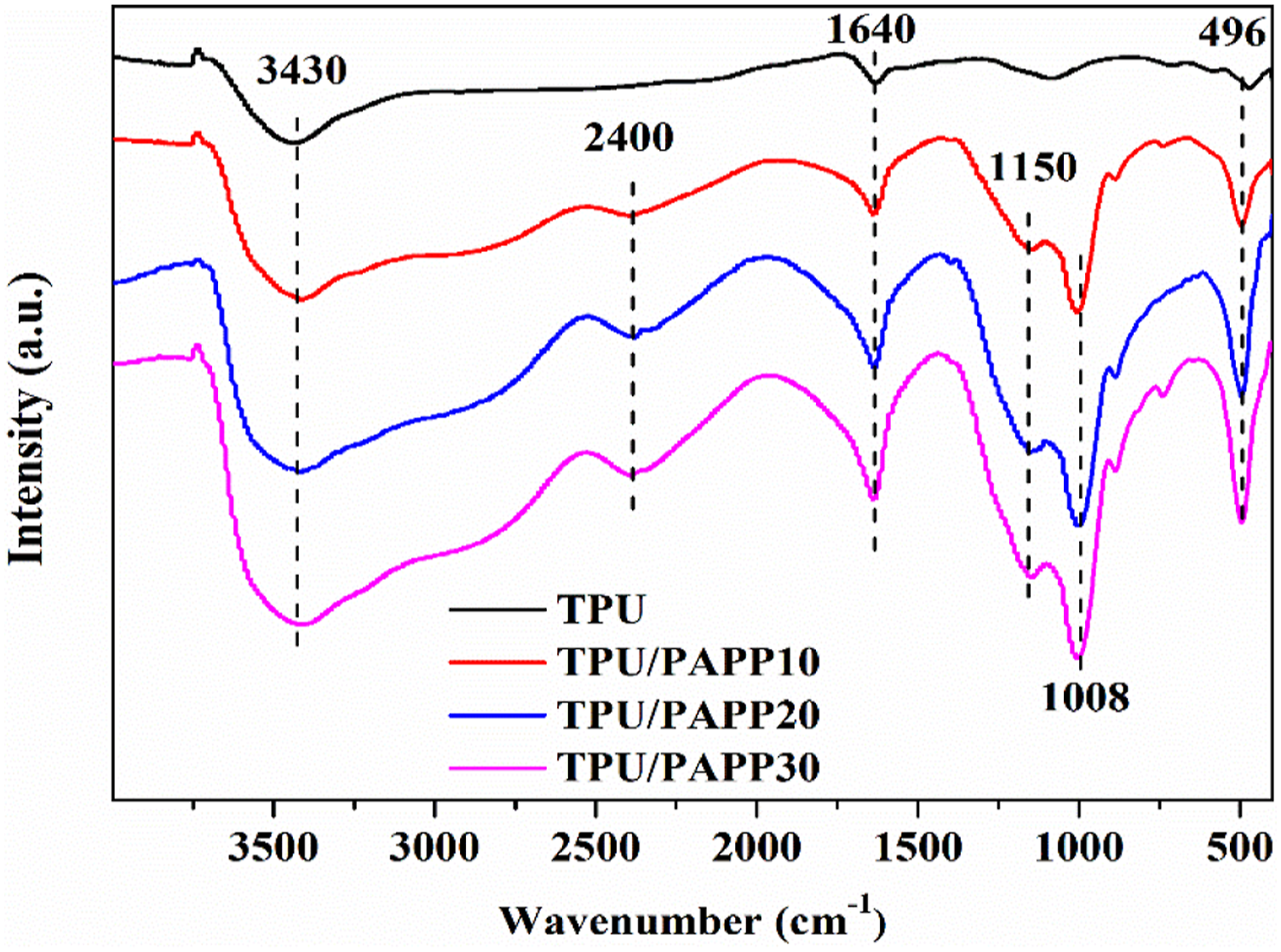
Figure 9 illustrated the XPS spectra of the char residue for TPU and TPU/PAPP30 composite, the corresponding data were listed in Table 5. The char of TPU mainly contained C, N, and O elements with contents of 88.2%, 8.07%, and 3.73%, respectively. Compared with TPU, the P element was observed in the char residue of TPU/PAPP30 composites. It was also found that the O element content increased to 45.88%, which was ascribed to the P-O or P = O structures in phosphates resulted from the decomposition of PAPP.
18
The P elements in char residue of TPU/PAPP30 was enhanced to 9.79%, suggesting phosphates were formed in decomposition of PAPP, which effectively enhanced compactness of the char residue. XPS spectra of char residues for TPU and TPU/PAPP30. Aromatic content of C, O, N and P in char residue of TPU and TPU/PAPP30.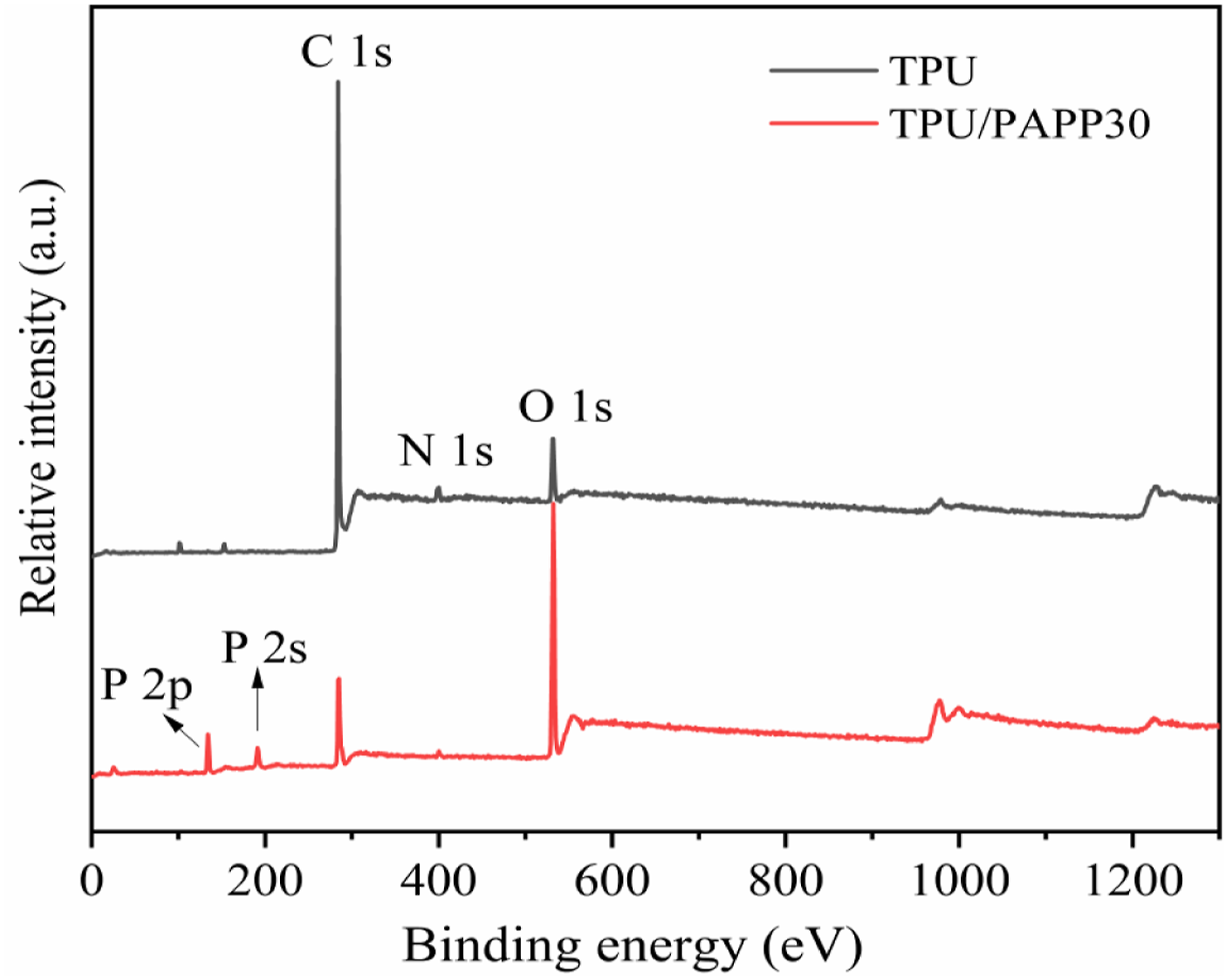
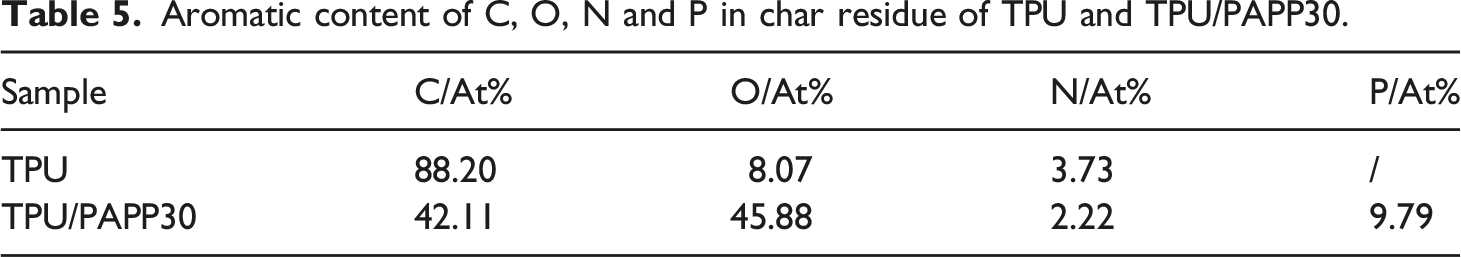
The XPS PEAK41 software was used to fit the peaks of C, O and N elements in the char residue to further investigate of the main components of the char residue for TPU and TPU/PAPP30. The fitted spectrum of C1s was shown in Figure 10 and the relevant data were listed in Table 6. The C element at 284.7 eV corresponded to C-C/C-H bond of the graphitic carbon and aliphatic species in the char residue, the C element at 285.7 eV was attributed to C-O/C-N structure in the char residue, and the C element at 287.4 eV corresponded to C = O/C = N bond of carbonyl, carboxyl, ester in the char residue. As observed from Table 6, the contents of the C-C/C-H bond, C-O/C-N bond and C = O/C = N bond of the TPU composite were 61.05%, 35.65% and 3.3%, respectively. And the contents of the C-C/C-H bond, C-O/C-N bond and C = O/C = N bond of the TPU/PAPP30 composite were 46.13%, 42.60% and 11.27%. By compare with TPU, the addition of PAPP promoted the formation C-O/C-N and C = O/C = N, which was benefit to form aromatic heterocyclic structure and thus enhance compactness of char residue. C1s spectra of char residue for TPU (a) and TPU/PAPP30 (b). Bonding state data of C in char residue of TPU and TPU/PAPP30.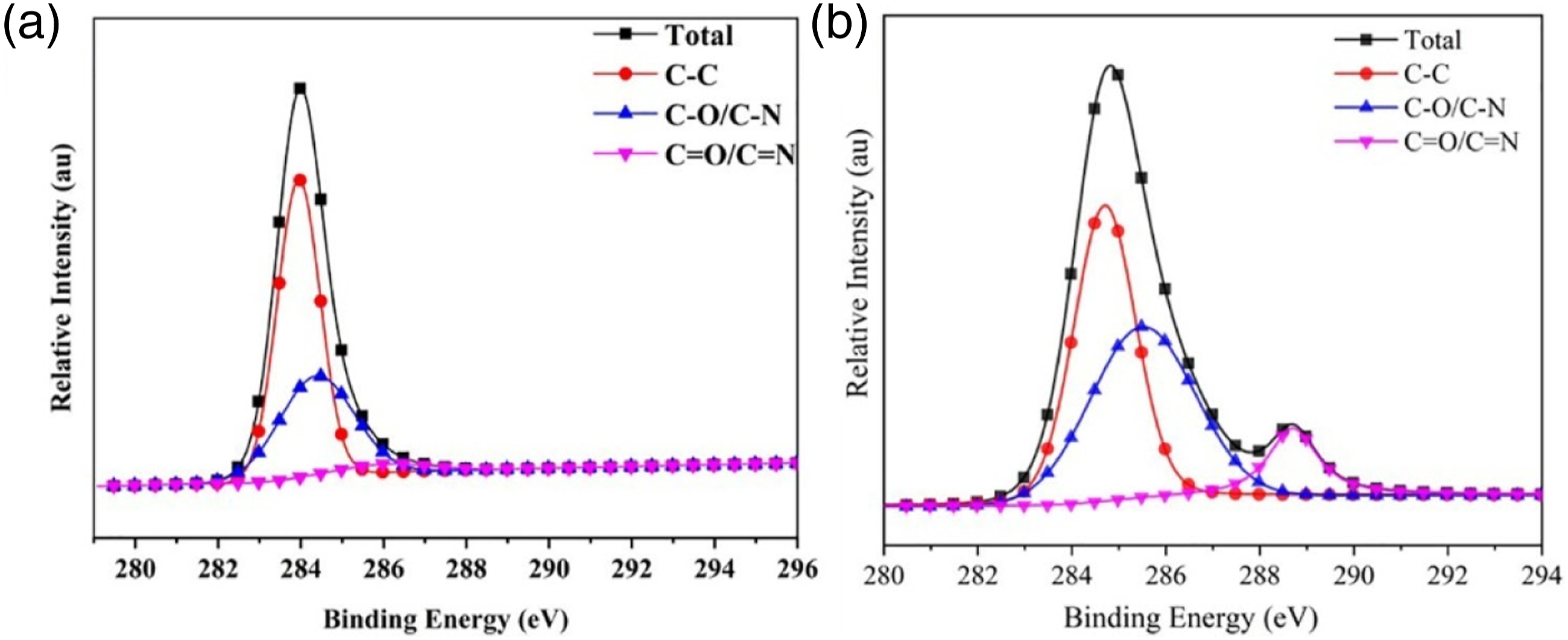
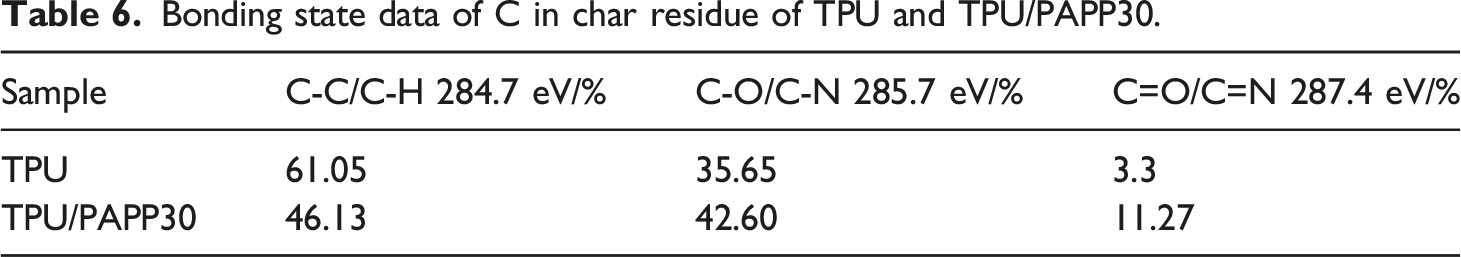
Figure 11 showed the O1s peak fitting spectra of char residues for TPU and TPU/PAPP30. The O element at 533.2 eV existed in the form of adsorbed oxygen (O2) and free water (H2O). The O element at 532.5 eV corresponded to the -O- bond, which was existed in the form of alcohols and ethers in the char residue. The O element at 531.3 eV was ascribed to = O bond, which was existed in the state of carboxyl, carbonyl and quinone in the char residue. As shown in Table 7, the O2/H2O, -O-, = O bond contents of the char residue for TPU were 26.90%, 44.35% and 28.75%. The O2/H2O content in char residue of TPU/PAPP30 was slightly increased by 1.62% compared with TPU. And also, -O- content in char reidue of TPU/PAPP30 was significantly increased to 51.43% with =O content decreased to 21.25%. O1s spectra of char residue for TPU (a) and TPU/PAPP30 (b). Bonding state data of O in char residue of TPU and TPU/PAPP30.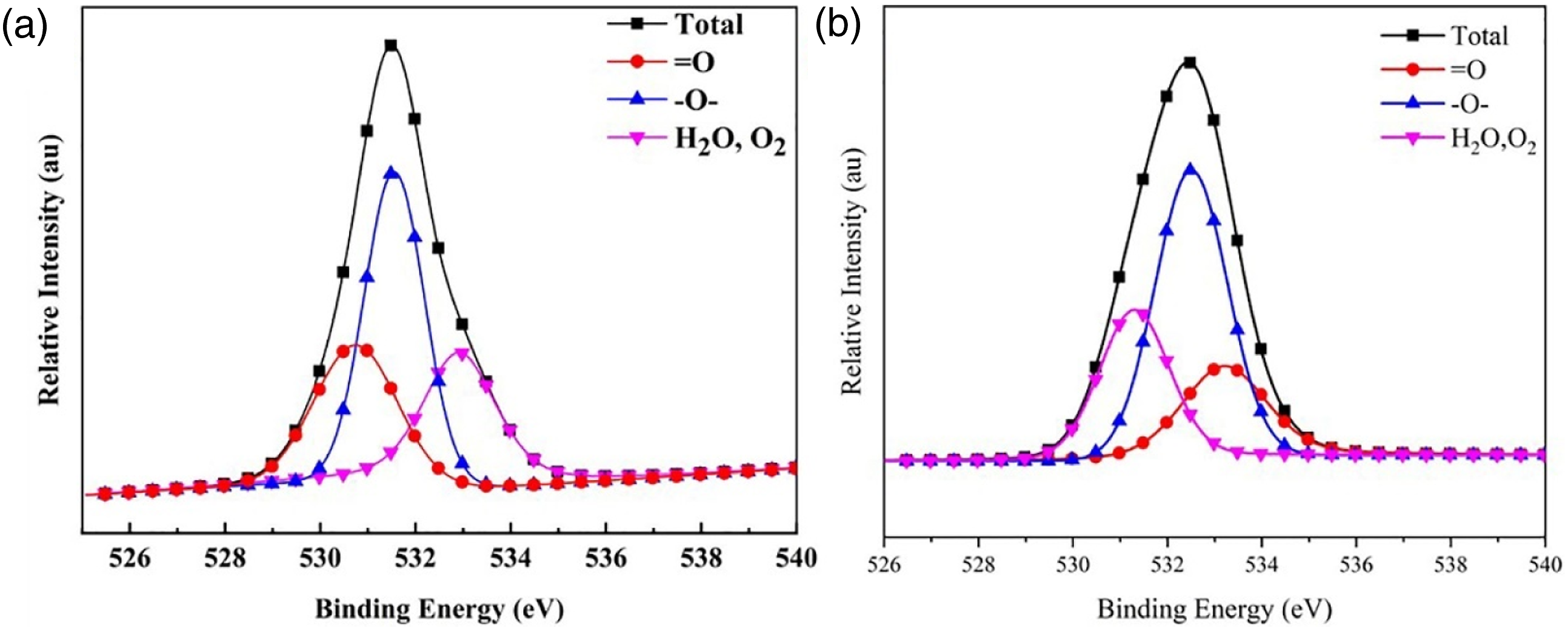

Figure 12 presented the N1s peak fitting spectra of char residues for TPU and TPU/PAPP30. The N element at 398.7 eV corresponded to -NH bond, which was existed in the state of amides and amines in the char residue. The N element at 400.4 eV corresponded to = N bond, which was existed in the form of aromatic heterocyclic compounds in the char residue.
21
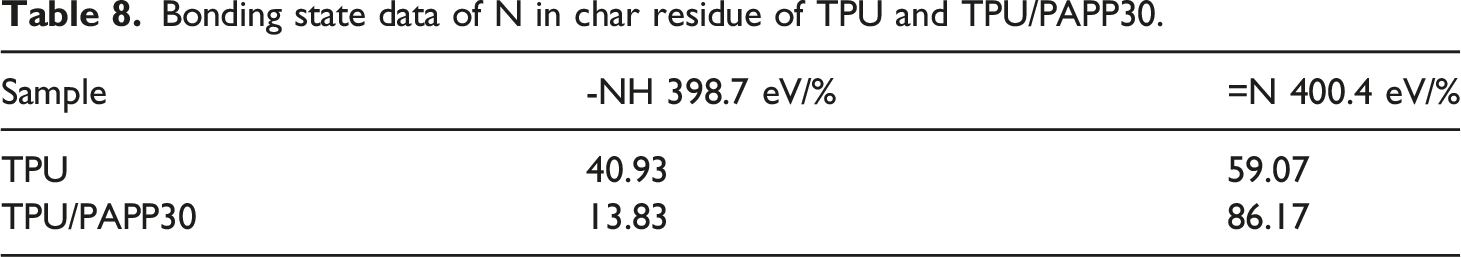
As shown in Table 8, the -NH and =N bond contents of TPU were 40.93% and 59.07%, respectively, and the -NH and =N bond contents of TPU/PAPP30 were 13.83% and 86.17%, indicating that the addition of PAPP promoted the formation of aromatic heterocyclic structures in TPU/PAPP30 composites. The aromatic heterocyclic structures was benefit to the enhancement of fire resistance for TPU/PAPP composites, which was high consistent with SEM test results.
28
N1s spectra of char residue for TPU (a) and TPU/PAPP30 (b). Bonding state data of N in char residue of TPU and TPU/PAPP30.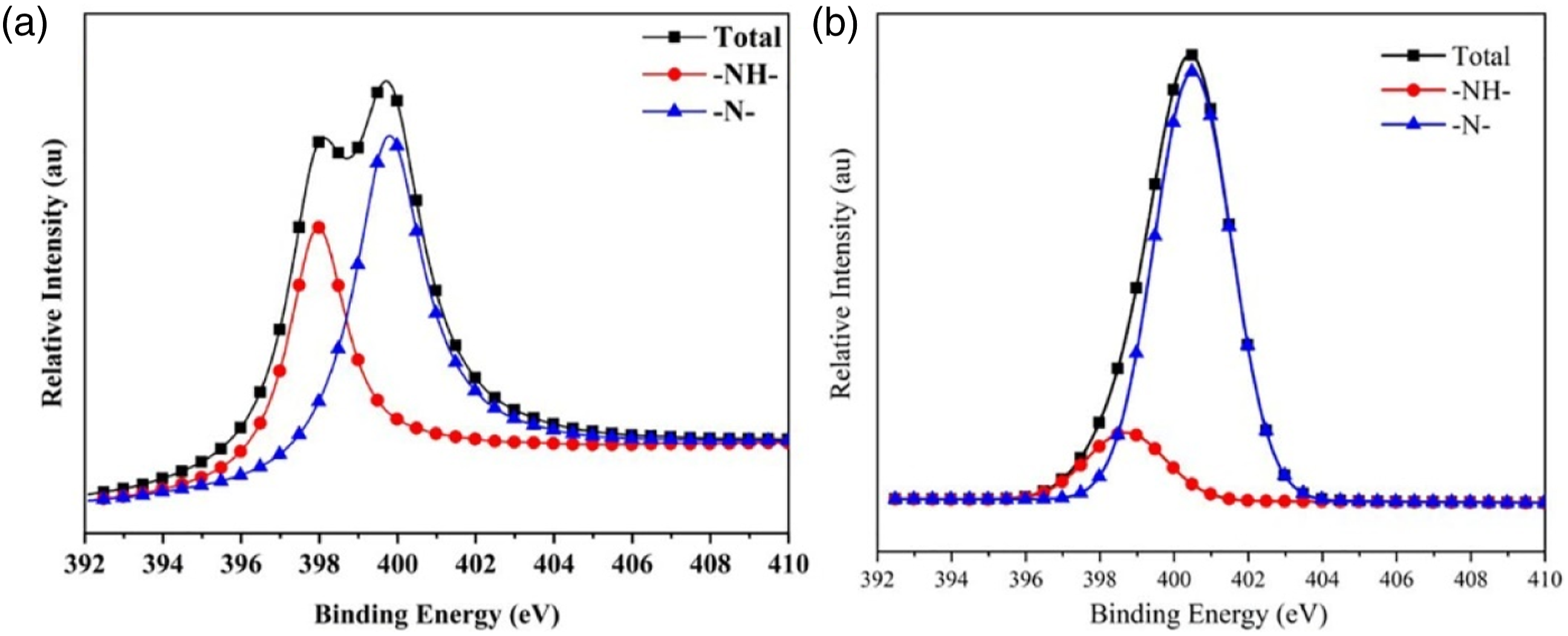
Mechanism consideration
In summary, the flame retardant mechanism of TPU/PAPP composites in this work was proposed, which was shown in Figure 13. The PAPP decomposed into viscous of polyphosphate, NH3, H2O, and P based radicals. The polyphosphate promoted the carbonization process of TPU molecular chain in the condensed phase to improve the quality of char layer. And also, the NH3 and H2O produced by PAPP decomposition diluted the combustible gases, inhibiting the combustion of the composites. Furthermore, P based radicals such as PO and PO2 captured the OH and H to play a quenching effect and retarded the combustion process in gas phase. Thus, TPU/PAPP composites was endowed flame retardancy by condensed-gas phase flame retardant mechanism. Schematic illustration for flame retardant mechanism of TPU/PAPP composites.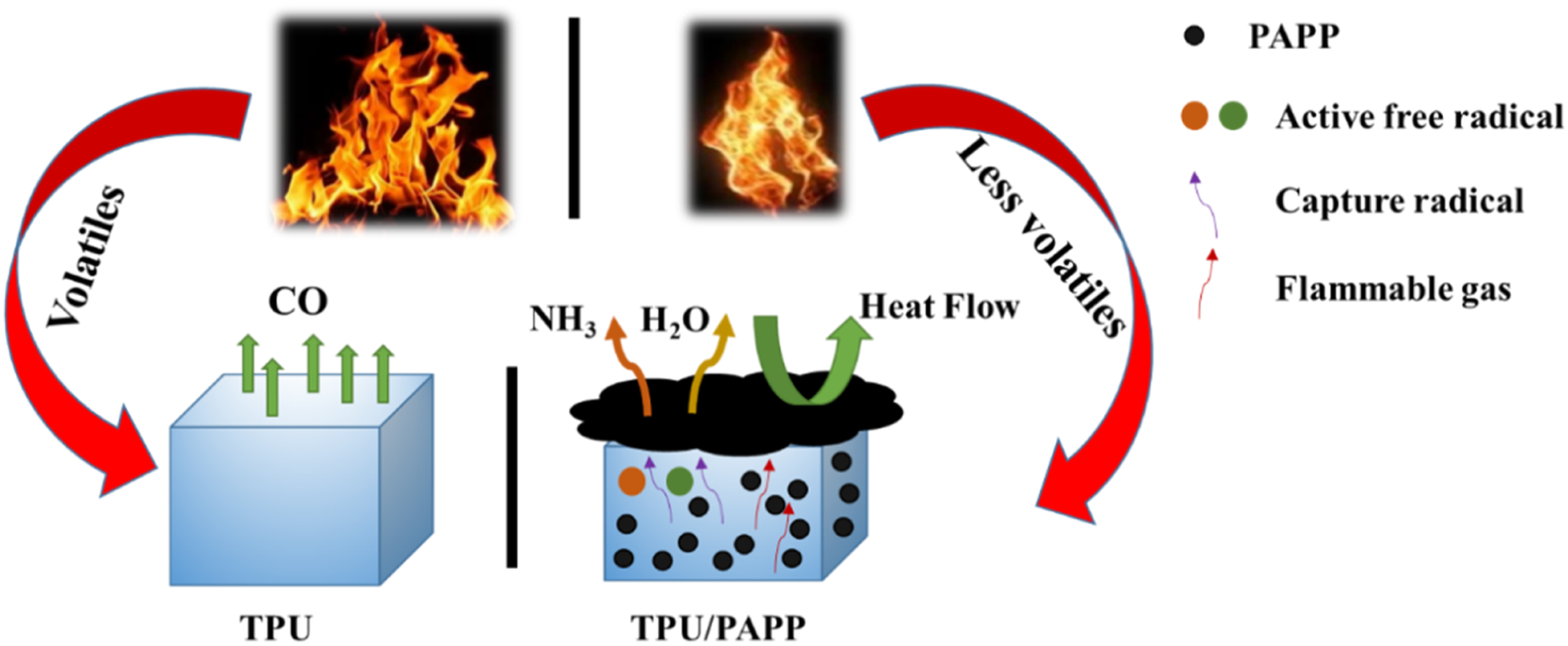
Conclusion
In this work, TPU/PAPP composites were prepared using PAPP as a flame retardant, and the flame retardancy, thermal stability, combustion behaviors, et al. of TPU/PAPP composites were investigated. SEM confirmed that PAPP promoted the formation of compact char residue. TG test showed that PAPP promoted the decomposition and carbonization process of TPU/PAPP composites, endowing them with decreased T-5% value and significantly enhanced char residue at high temperature. Flame retardant tests confirmed that PAPP was suitable for enhancing flame retardancy of TPU/PAPP composites. The TPU/PAPP composites with only 5 wt% PAPP loading exhibited LOI value of 27.5 vol% with V-0 rating in UL-94 test. TG-FTIR test implied that PAPP significantly inhibited the release of combustible gases and toxic gases. Char residue investigation implied that TPU/PAPP composites exhibited significantly increased O content with decreased C content in char residue compared with those of TPU. The PAPP loading promoted the formation of aromatic and aromatic heterocycle structure. Based on the investigation, a condensed-gas phase flame retardant mechanism of PAPP in TPU composites was proposed. This work provide a novel strategy for fabricating high performance TPU composites with outstanding flame retardancy.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Grants from Anhui Province postdoctoral research funding project (Grant No. 2019B336), Anhui Province Key Laboratory of Metallurgical Engineering & Resources Recycling (Anhui University of Technology) (Grant No. SKF21-03), the Open Project Program of Key Laboratory of Metallurgical Emission Reduction & Resources Recycling, Ministry of Education (Anhui University of Technology) (Grant No. JKF21-06), and Ma’anshan postdoctoral research funding project (Grant No. 2020 A11).
