Abstract
In this work, a thermal behaviour comparison of a new bamboo-based and banana-based Green Bio-Composites (GBC) is conducted using thermogravimetric analysis (TGA) and cone-calorimeter experiments. An Intumescent Fire-Retardant (IFR) coating (a mixture of Exolit IFR36 and boric acid) has been applied on the investigated GBC materials in order to explore the flammability resistance of such GBCs. Vacuum Bag Resin Transfer Moulding (VBRTM) technique has been used to manufacture the samples. TGA test have been conducted under oxidative atmosphere with three different heating rates while cone calorimeter tests have been performed with a horizontally exposure on the top surface of the sample. The outcomes of TGA revealed that Bamboo-based (BM-GBC) and Banana-based (Bn-GBC) materials exhibited similar thermal degradation patterns. However, BM-GBC outperformed Bn-based in the cone calorimetry analysis, this is proven by the fire reaction parameters as well as the higher char residue. In addition, IFR coating improved the flame retardancy of both GBCs, reduced the Peak Heat Release Rate (PHRR) by approximately 40–50% and smoke production (SEA) by 26%. SEM and EDS analysis of char residue were performed to deeply investigate the effectiveness of the IFR as a protecting layer.
Introduction
Nowadays, the benefit of natural fibre as a replacement for synthetic composite has heightened significantly due to the increasing cost of synthetic materials and environmental problems.1–3 Several advantages of natural fibres such as the abundance of product, biodegradability, environmentally friendly applications and comparably mechanical properties.4,5 Most of the natural fibre biocomposites reinforcement have been applied in the transportation and building industrial sectors.6–9 Therefore, many researchers investigated biocomposites reinforcement with chemical or nature matrix composites, for nature-based matrix it is called Green Bio-Composites (GBCs). There is interesting to develop for better understanding of their thermal-physical characterization and spread the application of GBCs. Several studies have been conducted on the use of natural fibre-based GBCs. There are mainly related to the use of flax,10–12 sisal, 13 hemp, 14 water hyacinth, 15 jute, 16 coir, 17 abaca, 18 bamboo, 19 soybean, 20 banana21–24 and pinene 25 natural fibres as a mechanical reinforcement. To further explore the potential of natural fibres, researchers have been investigating natural fibres-reinforced biocomposite matrices, known as Green Bio-Composite (GBCs). Numerous studies have utilized different types of resin matrices in the manufacturing of biocomposites. The previous research mentioned that the Greenpoxy is a resin with high mechanical properties especially in terms of strength and tensile properties on alfa fibre 26 and flax. 11
In the basis of a literature survey, Bamboo (BMF) and Banana (BnF) Fibres are some of the natural fibre that are considered as one of the most effective alternative to synthetic fibres and to those natural fibres are extensively cultivated.25 BMF are fabricated from the starchy pulp and are reported to have outstanding properties such as high tensile strength, low cost and durability and recyclability. Among the natural fibres used as a mechanical reinforcement, Bamboo Fibres (BMF) known for their high tensile strength, low cost and durability and recyclability. Various studies have examined the mechanical properties of BMF,27–29 The findings of these studies consistently demonstrate that BMF exhibit significant mechanical strength. However, they investigated only the mechanical properties of unbiodegradable epoxy. In other hand, BnF have shown promise due to their high mechanical properties, and ease of blending. Bn-based have found high tensile strength (180–430 MPa), high density (950–750 kg/m3) and a high water absorption (around 60%). 8 Additionally, BnF can be easily hybridized with other natural fibres, as demonstrated a recent study by Abhemanyu, who investigated the hybridization of jute with BnF to manufacture biocomposites. The results showed that such hybridization increased the tensile strength by 21.5% compared to biocomposites reinforced with coconut sheath fibre. 30
Several scientific studies have demonstrated that the BM-based GBC (BM-GBC) and Bn-based GBC (Bn-GBC) are very promising as a synthetic replacement not merely for interior but also exterior transportation applications, particularly in terms of mechanical properties improvement. However, their physicochemical and thermo-physical behaviours, as well as their flammability resistance still in question and require further research to more fully investigate the use of such bio-based materials. Even though, there are many studies reporting on the biocomposite material use, none have been found on the utilization of Bn-based and BM-based in a thermos-plastic starch matrix, notably on biodegradable matrix. Therefore, this work employs a biodegradable resin matrix made from 60% carbon plant based Greenpoxy. The utilization of Greenpoxy as a resin matrix offers several environmental advantages. It is derived from renewable plant-based sources, making it a sustainable alternative to traditional petroleum-based resins. Additionally, Green-poxy is biodegradable, and its use can contribute to a reduced carbon footprint. One of the critical constraints associated with the use of Bn-GBC and BM-GBC is their flammability, which can limit their use in applications where fire resistance is essential, such as in construction, transportation and aerospace industries. Therefore, their flammability resistance requires further research to more fully investigate the use of such bio-based materials. This study examines the thermal properties of Bn-GBC and BM-GBC, which is a novel development compared to previous studies that have mainly focused on their mechanical properties. By investigating their thermal characteristics, this research aims to uncover their potential applications in fields such as thermal stability, and fire resistance, using Intumescent Fire Retardant (IFR) to protect the underlying material structural integrity. 31 One of the most extensively studied and promising synergistic IFRs is ammonium polyphosphate (APP). Which has been demonstrated to effectively protect polypropylene composites when combined with Mv-AlPi and hybrid organic-inorganic (SCTCFA-ZnO), as evidenced by previous studies.32,33 From the literature, it has been demonstrated that APP combined with another fire retardant can effectively protect polypropylene composites34,35 and epoxy resin. 36 These findings highlight the versatility and the potential of APP-based IFRs in enhancing the flame retardancy of various polymer composites.
This study consists of proposing new bio-composite materials using biodegradable plant-based resins, combined with natural fibres. The development of GBC materials aims to promote the use of new bio-composite materials with high biodegradability in the transportation sector. Even though several works have treated their mechanical aspect, no prior research has investigated the thermal behaviour of biocomposites containing Greenpoxy reinforced with BmF and BnF fibres. After manufacturing the GBCs samples, Thermogravimetric Analysis (TGA) and cone calorimetry experiments have been performed to highlight both the natural fibre and IFR coating effects. To explore the fire resistance of such materials, IFR coating has been applied to the manufactured samples. The novelty here is to combine APP with a specific percentage of boric acid (1%). It consists also to evaluate the fire-retardant efficiency while reducing the boric acid content in order to minimize its toxicity. Results show that both materials have a great promising for several industrial applications. It has been noticed that the IFR coating delays the GBC thermal decomposition, improves the ability to withstand fire.
Materials and methods
Raw materials
Bn-GBC and BM-GBC preparation
Vacuum Bag Resin Transfer Moulding (VBRTM) method
37
were used for samples manufacturing (see Figure 1). The Bn-GBC matrix is composed of 16 banana unidirectional fibre plies [0]16, while that of BM-GBC is composed of four BMF plies [0]4. Both matrices are 200 × 200 mm2 of dimension with constant thickness (5±0.2 mm). The matrix is put on the moulding part which consists of three respective layers: (1) peel ply layer which permits to remove the biocomposite fabric without damaging the surface; (2) infusion mesh layer to ensure the infusion uniformity; (3) vacuum bag film to evacuate possible air inclusions. SR Greenpoxy 56 resin and SD 2284 hardener were mixed in a weight ratio of 100:21 based on the supplier recommendations. VBRTM experimental bench details: (a) Schematic view (b) experimental bench.
The infusion process was done under a vacuum pressure of – 0.92 bar and at room temperature after carrying a leakage check. When the resin mixture is fully infused through the fabric, the mould was left to cure for 24 hours at room temperature. Thereafter the fabric is post-cured for 48 hours in an isothermal furnace (40°C for 24 hours and then 20°C for 24 hours again) to complete the polymerization process. Finally, a visual inspection of the specimens was carried out to identify any manufacturing defect (presence of air, uneven resin distribution, non-uniformity of surfaces…).
The IFR considered in this work consists of Exolit IFR 36 (APP-THEIC based) of 29 wt.% and 1 wt.% boric acid mixture. The selected ratio is based on the work of Rashid et al.,38,39 The IFR percentage ratio in the sample is around 16%. The authors identified this ratio as the most convenient one. The IFR was first mixed with resin as binder before being applied on samples surfaces in order to explore its fire protection capacities. Due to the crystalline form of boric acid, which is challenging to break down, magnetic stirring was used for 20 minutes to speed up the homogenization process, as schematised in Figure 2. The IFR coating thickness was held constant at 1±0.1 mm. After the coating process, samples were placed in the furnace set at 40°C for 24 hours to complete the IFR coating curing cycle after being left at room temperature all day. Schematic view of the IFR preparation.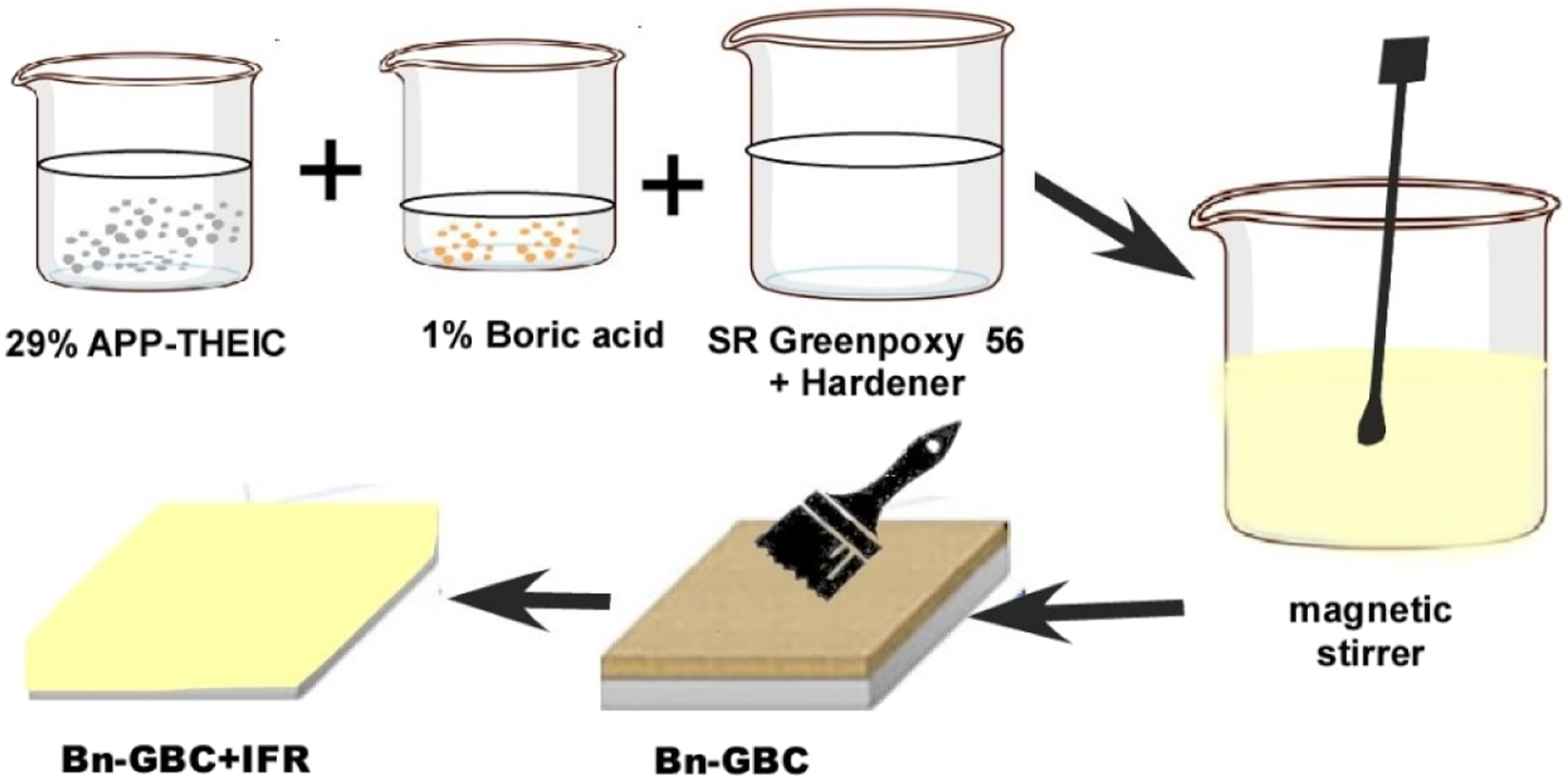
Once all Bn-GBC and BM-GBC fabrics are fully polymerized, samples for TGA and cone calorimeter are prepared. For TGA tests, a total of 18 samples (10 × 10 × 5 mm3 of dimension) were cut from each GBC. Nine samples are coated with IFR, while the rest is kept uncoated. The purpose here is to investigate the IFR coating effect under three heating rates while ensuring tests repeatability. Concerning the cone calorimeter experiments, six samples are prepared from each GBC fabric (100 × 100 × 5 mm3 of dimension). As for TGA samples, three samples are coated with IFR at their surface (for repeatability leads), while the remaining samples are kept without coating.
Testing and characterization
The thermal decomposition behaviour of each sample was analysed using a thermogravimetric analyser (TGA/DSC, THEMYS ONE, SETARAM – France). Bn-GBC and BM-GBC samples with around 10±0.5 mg of mass were placed in a platinum crucible under O2 environment (airflow rate of 100 ml/min). TGA tests were conducted under a temperature range of 30–900°C for three heating rates (5°C/min, 10°C/min, and 15°C/min). All TGA experiments were repeated three times to assess test’s reliability, and standard deviations were summarized accordingly.
At larger scale, the combustion properties of the investigated materials were measured using a cone calorimeter in conformance with ISO 5660-1 standard. 40 The bottom of each sample was wrapped in tinfoil and the top surface was exposed horizontally to 50 kW/m2 external heat flux. Spark ignition was used to ignite the pyrolysis products at atmospheric temperature – pressure conditions. The standard uncertainty of the measured HRR was ±10%. The combustion gases were directed to an exhaust hood located above the cone heater. CO2, CO and O2 gases were simultaneously sampled and analysed by a probe located along the exhaust line. The residue after tests was photographed using a digital camera.
The morphology and crystallography of the char residue of Bn-GBC, Bn-GBC + IFR, BM-GBC, and BM-GBC + IFR were analysed using a Scanning Electron Microscope (SEM) with magnification ranging from ×50 to 10,000×. SEM micrographs were obtained from JSM-IT300 instrument from JEOL, Japan and the acceleration voltage used was 20 kV. All specimens were coated with Au prior to observation due to the non-conductive material nature. The SEM analyses were complemented with Energy Dispersive X-Ray Spectroscopy (EDS) using the Xmax20 instrument (Oxford Instruments, United Kingdom) to determine percentage amount of the elemental compounds present in the samples.
Result and discussion
TGA analysis
Natural fibre effect
The first subsection highlights the effect of reinforcement i.e. BMF and BnF on the thermal degradation of the BM-GBC and Bn-GBC, respectively. Figure 3 displays TGA result for BM-GBC and Bn-GBC, it is clearly seen that BM-GBC and Bn-GBC have two reaction steps that happened, and the onset degradation temperature exhibited similar values at 210°C. The BM-GBC material lost half of its mass because of the first reaction degradation process, which also affected the Bn-GBC. It might be due to their similar lignocellulosic BMF component consisting of lignin 18.36 wt. % and hemi cellulose of 23.79 wt.%, while for banana leaf have lignin of 10 wt.% and 6–19 wt.% of hemicellulose.41,42 Additionally, the previous research mentioned that the onset decomposition of raw BnF at 135°C and BMF at 125°C.
43
TG and DTG results of (a) BM-GBC and (b) Bn-GBC for 5, 10 and 15°C/min.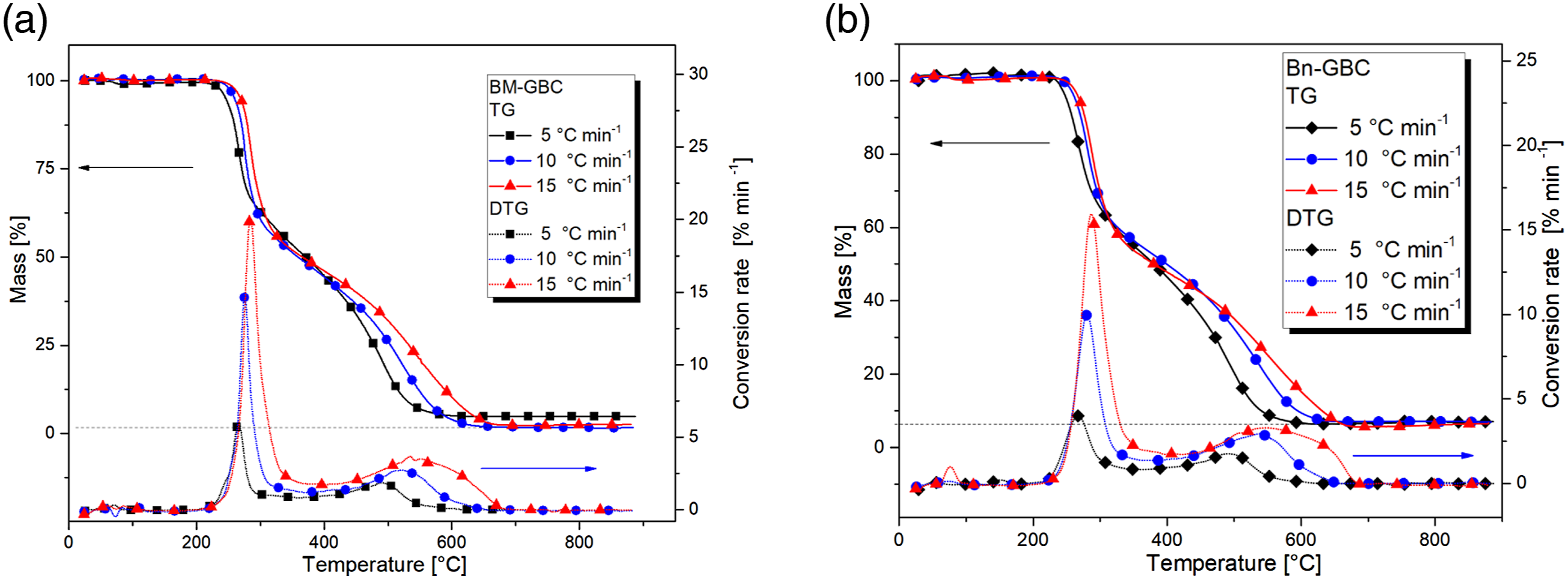
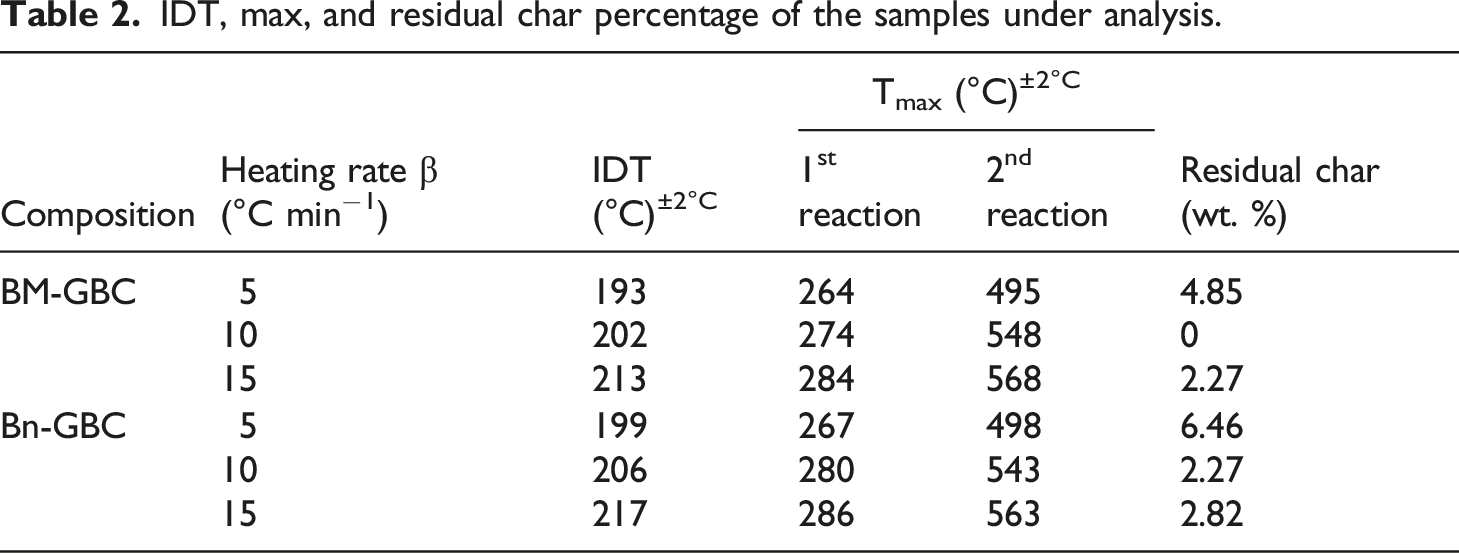
IDT, max, and residual char percentage of the samples under analysis.
The superposition of TG and DTG curves of Bn-GBC and BM-GBC at 15°C/min shown in Figure 4 clearly reveals that both GBCs exhibit similar thermal degradation profiles except for the slight difference in the first reaction peak. The lowest heating rate gives a higher char residue in both materials, although Bn-GBC materials have a higher char residue overall than BM-GBC. By observing the maximum thermal decomposition rate of both samples, it can be noticed that the maximum rate of BM-GBC sample is four-time higher than that of Bn-GBC. By comparing both curves, the gap in the mass loss begins at temperatures around 300°C. This temperature is within the range of lignin decomposition (from 160°C to 400°C). This gap is mainly due to the lignin content of the two samples. The BM based sample has around 32.2% wt. of lignin while the Bn based sample has 5% wt. to 10% wt.
45
In addition, it is well known that the lignin increases the char production. Thus, the MTDR of BM-GBC is higher than that of Bn-GBC sample. The acquired results are in good coherence with previous research on Bn-based composite with epoxy.
46
However, the results of the BMF-based composite with epoxy are rather different with this reference
47
. It might be due to the amount of the fibres and the chemical composition of the resin. The comparison of TG-DTG curves of the BM-GBC and Bn-GBC samples.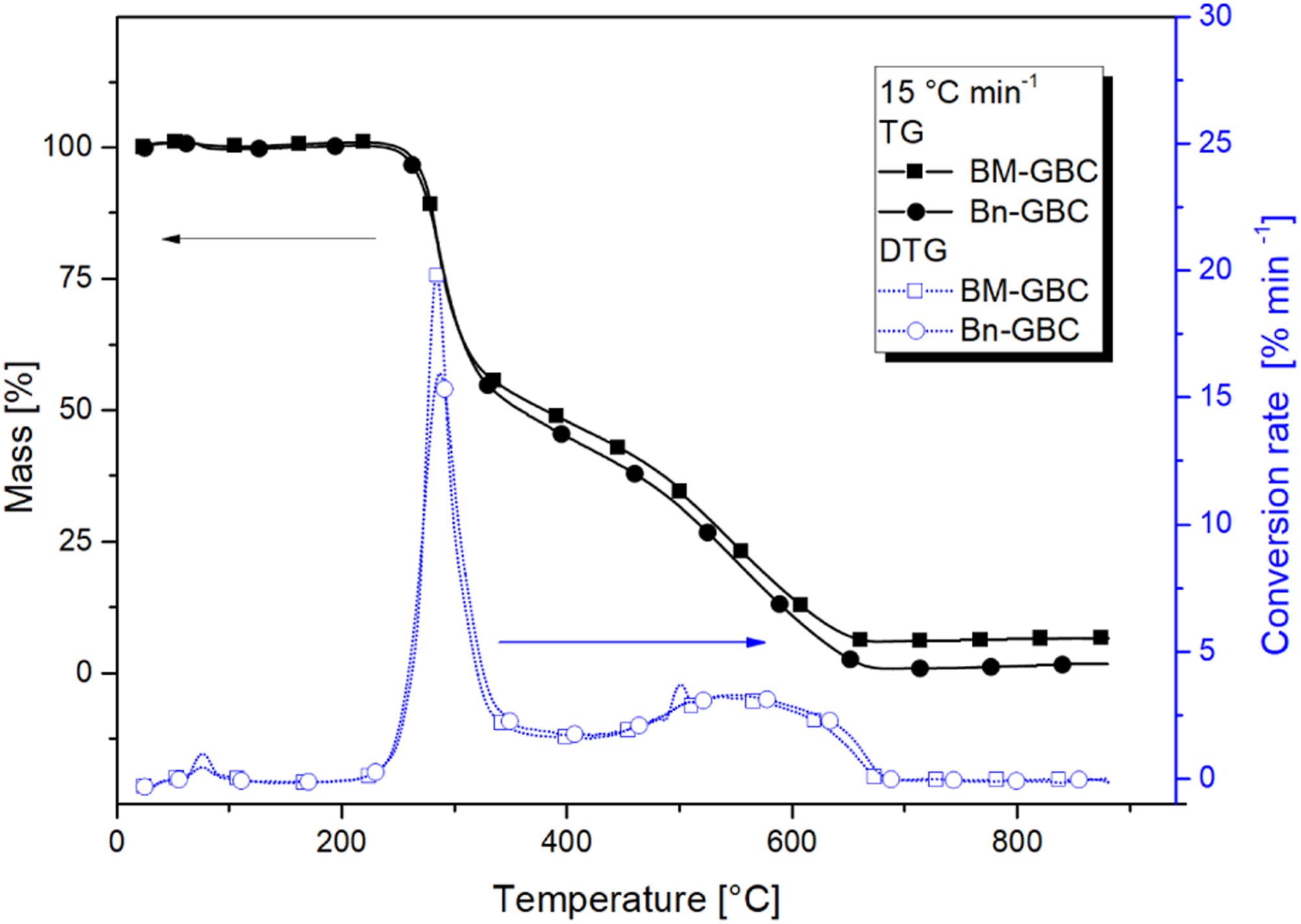
Fire retardant coating effect
The fire-retardant coating effect on the BM-GBC and Bn-GBC are depicted in Figures 5 and 6, respectively. Figure 5 shows the effect of the IFR coating on the thermal degradation of BM-GBC materials by comparing their TG and DTG curves. Figure 5(a), it is clearly seen that BM-GBC + IFR shows three reactions mechanism that occur during flaming. The lower heating rates gives smaller char residue than other, however for 10°C/min and15°C/min have the same char residue. The first reaction mechanism of the BM-GBC IFR happens at 200°C–400°C and the reaction continues until 450°C for second reaction. Then, it undergoes third reaction where it is completely degraded at 900°C. IFR is able to maintain the mass loss especially in the higher temperature (more than 300°C). (a) TG-DTG curves of BM-GBC + IFR (b) comparison on TG-DTG results of BM-GBC against BM-GBC + IFR. (a) TG-DTG curves of Bn-GBC + IFR, (b) comparison of TG-DTG curves of Bn-GBC + IFR against Bn-GBC at 15°C/min of heating rate.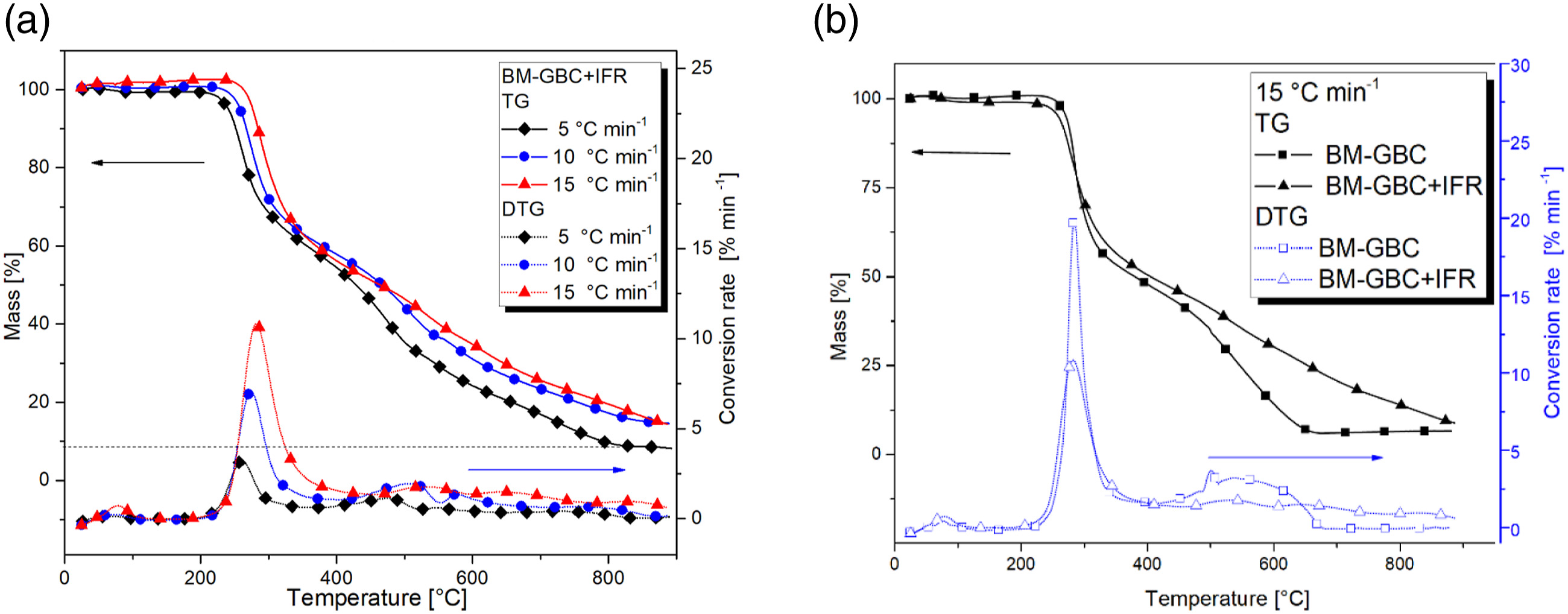
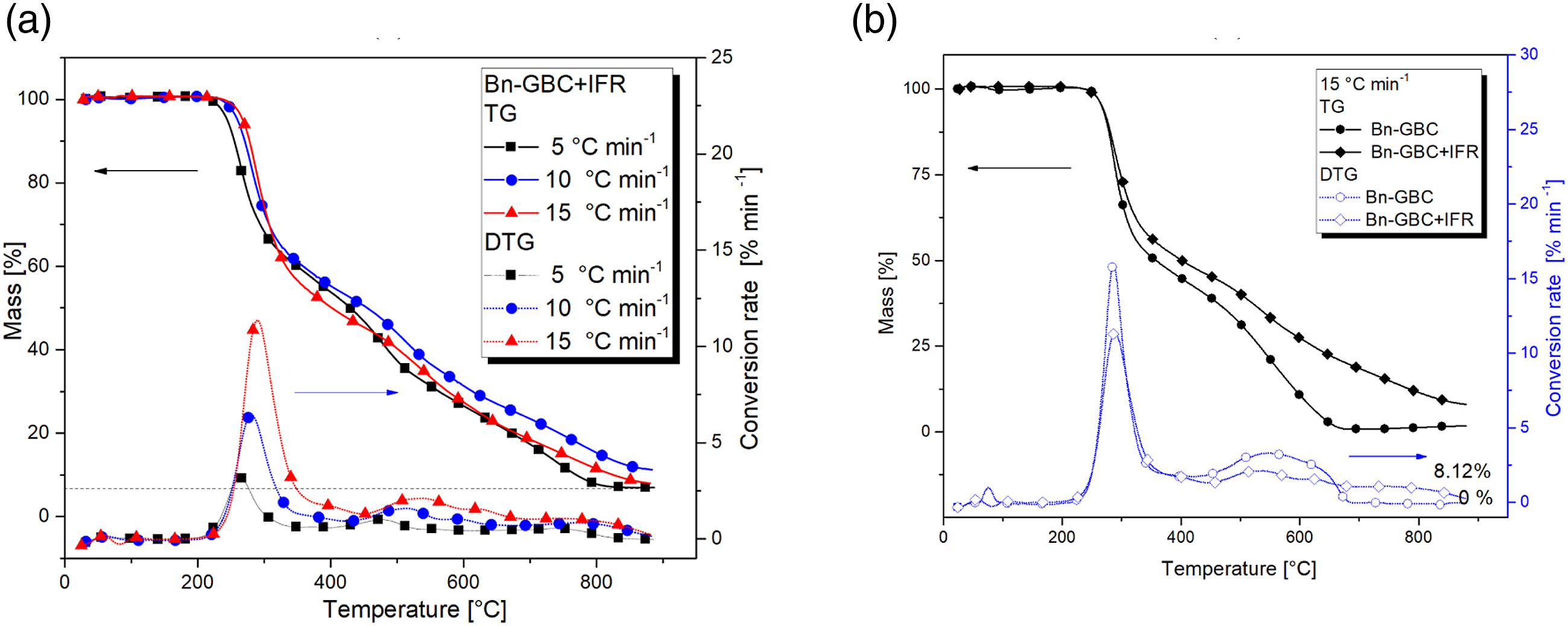
In addition, Figure 5(b) demonstrates a comparison uncoated and coated BM-GBC which shows. In a point remark shows the presence of fire-retardant increasing char residue of 8% and delaying decomposition process by adding reaction mechanism. As described in the above section, uncoated BM-GBC just only has two reaction mechanism however for coated BM-GBC has three reaction mechanism. Nevertheless, the onset temperature (IDT) and the temperature peak of first reaction uncoated BM-GBC is quite like the coated BM-GBC. The fire-retardant works effectively on the second reaction until the highest temperature treatment.
Figure 6 displays the first reaction of Bn-GBC + IFR undergoes 235°C–406°C and the second reaction undergoes from 406°C–555°C. It continues to the third reaction to complete degradation. As mentioned above, the TGA result on the fibre based is nearly similar due to their chemical component. Thermal stability of this material reaches an approximately at 800°C which a char residue almost 8% in all heating rates. In addition, the highly difference located on the third reaction mechanism has not happened in the neat GBC. Therefore, the IFR retards thermal decomposition by adding a reaction mechanism and increasing the peak temperature especially in the second reaction (elevated temperature).
The IFR effect on the thermal decomposition.
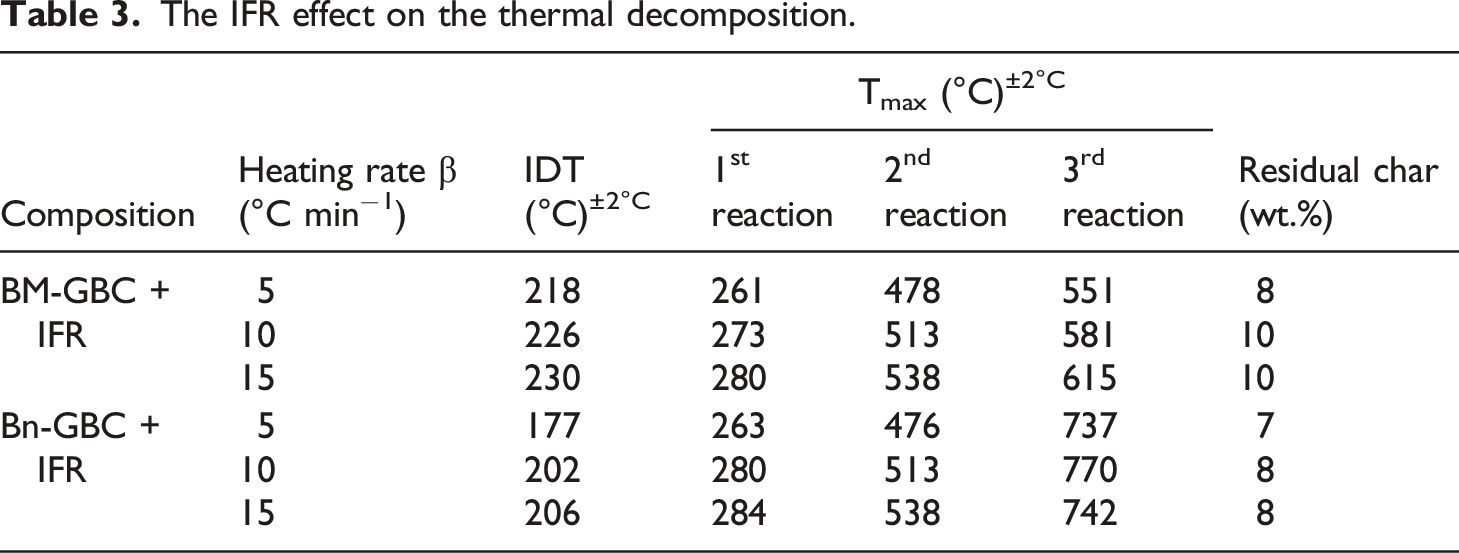
On the other hand, the result demonstrates that degradation area of BM-GBC + IFR and Bn-GBC + IFR are reached, it can prove that IFR can maintain the decomposition process gradually until both materials reach the thermal stability. The effect of IFR delay degradation at the higher temperature (second reaction) is more evident than in the lower temperature for both materials (first reaction). This behaviour is due to the Exolit IFR36 and boric acid effectiveness which is highly visible for the temperature above 300°C
41
and 500°C,
48
respectively. In addition, the Bn-GBC + IFR and BM-GBC + IFR thermal stability is investigated. Results on the thermal stability are compared to those of uncoated GBC at T50–T90 mass loss (See Figures 5 and 6). It can be clearly observed that the IFR coating is increasing the thermal stability of both coated samples, e.i. Bnf and BmF based biocomposite, particularly at temperatures ranged between 400–900°C. This improvement in terms of thermal stability could be attributed to the formation of boron phosphate, which is a highly stable material. The boron phosphate is formed around 400°C due to the reaction between APP-THEIC and boric acid.
49
Furthermore, the IFR coating increases the char residue formation. For instance, both coated samples have 8% to 10% of char residue beyond 900°C, while it is around 2% for uncoated samples (see Table 2). To confirm this result, TGA tests have been performed on the Greenpoxy + IFR sample. The purpose here is to deeply investigate the thermal stability of the IFR. TGA curves and their derivatives are depicted on Figure 7. Same behaviour can be noticed by comparing the TG curve of Greenpoxy + IFR to those of Bn-GBC + IFR and BM-GBC + IFR. In addition, the char residue beyond 900°C is equal to 8% for Greenpoxy + IFR sample, while it is equal to 2% for Greenpoxy sample, which is in accordance with the TGA data of Bn-GBC + IFR and BM-GBC + IFR samples. TGA curves (a) and conversion rate evolution (b) of basic components used for BLF-based BC samples.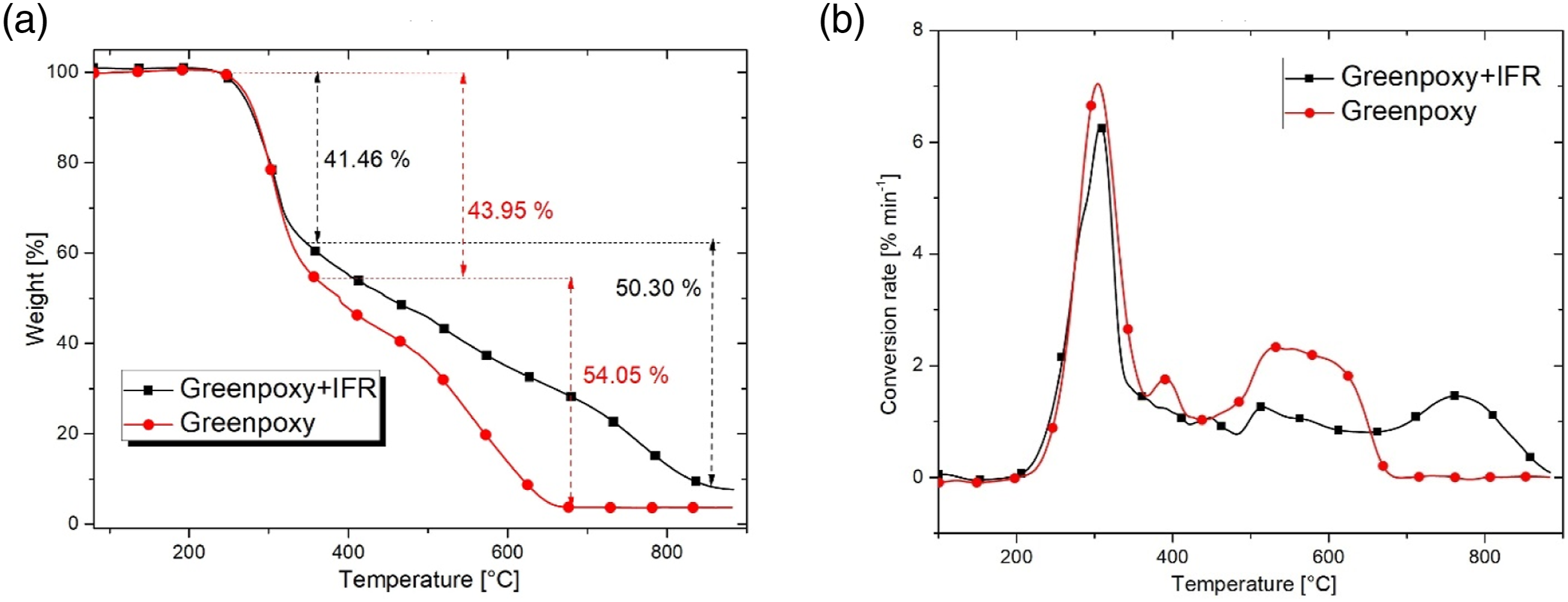
Due to the role of Ammonium Polyphosphate as carbonic agent and boric acid as char builder, therefore the synergism of those IFR gives the effectiveness for delaying degradation process of BM-GBC and Bn-GBC materials. The contribution of boric acid supports the appearance of a hard glassy fire protective coating of borophosphates. 50 Due to the lignin chain that is highly crosslinked and the C–C bond that is highly unbreakable which appear in the IFR during the flaming material.
That is proven that IFR plays a role in protecting the layer for both materials. Comparing the neat (virgin GBC), IFR works effectively on material Bn-GBC because it more delaying thermal decomposition (almost 150°C–200°C) in the third reaction comparing with BM-GBC even though BM-GBC had 2% of higher char residue. Generally, due to the capability of Exolit IFR and boric acid synergism able to prevent collaborating with highly lignin content in BMF. Due to their function, Exolit IFR 36 which is biodegradable non halogenated as blowing agent and boric acid which its glassy layer build as carbonisation agent.
Cone calorimetry
Cone calorimetry tests have been conducted on BM-GBC, Bn-GBC, BM-GBC + IFR, and Bn-GBC + IFR materials. The cone calorimeter experiments allow the evaluation of the following combustion parameters: Heat Release Rate (HRR), Total Heat Release (THR), Peak Heat Release Rate (PHRR), Effective Heat Combustion (EHC), Mass Loss Rate (MLR), CO2 and CO Production, Smoke Production and Time to Ignition (TTI). The analysis of all these parameters permit to evaluate the effect of natural fibre type and IFR coating on the flammability behaviour of GBCs.
Heat release results
Figure 8 displays the evolution of HRR (Figure 8(a)), THR (Figure 8(b)), EHC (Figure 8(c)), and PHRR (Figure 8(d)) of the investigated materials. Figure 8(a) shows that the HRR variation of the neat GBCs (BM-GBC and Bn-GBC without IFR coating) depict two stages. The first stage corresponds to the preheating stage where the material is heated up and the moisture and other volatile compound are released. During this stage, the HRR increases rapidly and reaches its maximum beyond which the materials start to decompose. By observing the HRR evolution of the neat GBC samples, it can be clearly noticed that the HRR of the BM-GBC sample (923 and 584 kW/m2 of PHRR) is thicker than that obtained by Bn-GBC sample (840 and 837 kW of PHRR). By comparing the HRR of both neat materials, one can distinguish that the materials behave differently. The Bn-GBC sample depicts a fully developed pattern, while the HRR of the BM-GBC sample depicts a decay pattern (the HRR decreases gradually toward second peak). All investigated materials start to ignite at the similar time (100 s) however, the burning time of BM-GBC sample (377 s) was longer than that of the Bn-GBC sample (275 s). PHRR of BM-GBC sample is higher than that of Bn-GBC, this is likely due to the higher burning rate of BM-GBC sample, which indicates that more heat is produced by the Bn-GBC sample decomposition (see Figure 8(d)). Furthermore, the Bn-GBC HRR has a thinner charring layer, which suggests it more prone to burning quickly and completely. In contrast, the BM-GBC sample exhibits a thicker charring layer which indicates that is more resistant to burning. From the above discussion, it can be noticed that the natural fibre type significantly affects the analysed combustion parameters and thus has an effect of the fire reaction behaviour of the studied materials. HRR (a), THR (b), EHC (c) and PHRR (d) evolution as function of time during the cone calorimeter tests.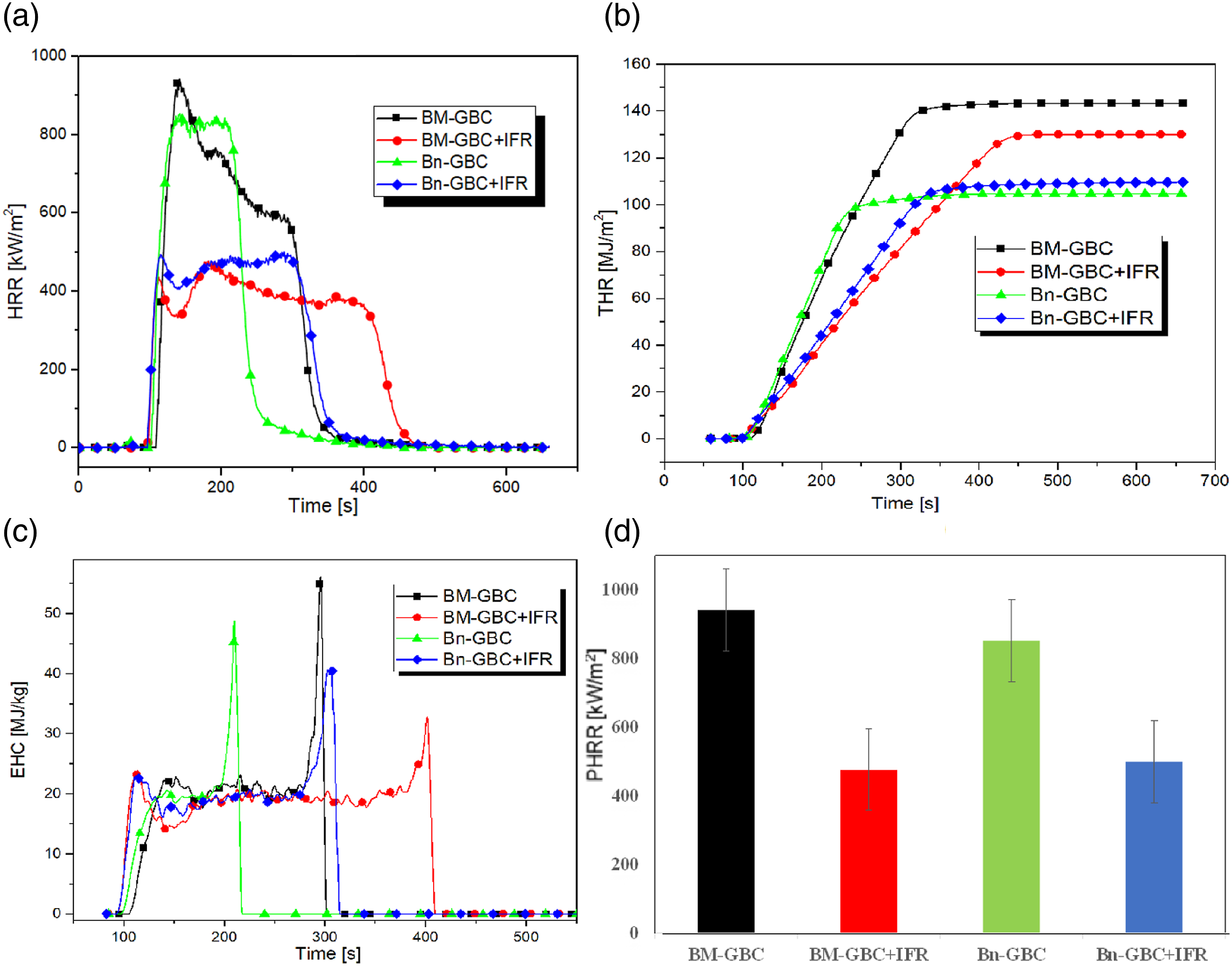
Regarding the coated GBC (BM-GBC + IFR and Bn-GBC + IFR), the HRR evolution depicts three stages. one remarks that the HRR of coated GBC sample is substantially below of that recorded for neat GBC samples. The pattern is a thick fully developed phase. The three consecutive HRR peaks of the three peaks of BM-GBC + IFR and Bn-GBC + IFR were recorded as 428, 470, and 380 kW/m2 and 483, 481, 488 kW/m2, respectively. This is due to the IFR coating which reduces the HRR amount by almost 50% comparing to that recorded by both neat GBC samples. In addition, Figure 8(a) shows thick charring on BM-GBC + IFR and Bn-GBC + IFR during burning process. The surface was cross-linked between the IFR and BM-GBC material which forms a carbonized layer and participates to block oxygen and heat from outside. 51 It is worth to notice that the heat release measurements can also be used to evaluate the efficiency of a product in terms of energy conservation. In Figure 8(d), the PHRR of the investigated materials is given. The highest HRR peak value is recorded on the BM-GBC sample while the lowest one is given by the BM-GBC + IFR sample. The lower HRR value of the BM-GBC + IFR sample refers to the weak heat release or to the fact that the sample takes longer to release the same amount of heat compared to the BM-GBC material under the same heating.
On the other hand, by linking the HRR measurement to that of THR (Figure 8(b)), which allows to evaluate the fire hazard of materials and to develop effective fire safety strategies. THR provides an indication of the material potential to contribute to fire growth and development. This can lead to more rapid-fire growth, higher temperatures, and greater risk to life and property.
By analysing the obtained results on the THR, the BM-GBC sample gives the highest values at the end of the cone calorimetry tests comparing to the other samples. A higher THR value indicates that the material is more flammable and will release more heat during combustion. One can notice that the IFR coating works effectively on the BM-GBC sample. This fact can be seen from the THR curves where a decrease of 14% is obtained by applying the IFR coating. Contrarily, the interaction between the BnF and the IFR coating (see THR of Bn-GBC + IFR) seems to be less efficient in terms of the THR reduction. In fact, THR of Bn-GBC + IFR is 5% higher than that of Bn-GBC. The THR of Bn-GBC + IFR increased because of the Bn fibre chemical properties, in particular the lignin amount. To gain more comprehensive understanding, a comparison between BM-based and Bn-based sample should be conducted. According to the literature, the two fibres contain nearly similar amount of cellulose, while there is a big difference on lignin content (BM fibre has 32% of lignin, vs. 5% for Bn fibre). 45 Consequently, the high percentage of lignin in bamboo fibre affects the thermal properties of BM-GBC + IFR by ameliorating its thermal stability therefore, THR curve of BM-GBC + IFR is lower even if the sample is coated by IFR. In fact, lignin has a complex structure composed of aromatic units linked by various functional groups such as hydroxyl, methoxyl, aldehyde and carboxylic acid groups. The aromatic rings in the lignin structure are known to have good thermal stability preventing the thermal degradation of the material.
The EHC of the studied samples is depicted in Figure 8(c). it allows to evaluate the amount of the heat released per unit mass of the material during the combustion process. From Figure 8(c), one notices that the EHC decreases by almost 50% and 20% due to the IFR coating on BM-GBC and Bn-GBC, respectively. In fact, the APP content in IFR mixture releases ammonia and water which dilutes the gaseous phases and lead to a carbonaceous residue formation. 52 The higher value of EHC corresponds to higher energy density, generally material with a higher energy density tends to be more flammable or have a higher potential for combustion. However, there are other factors that can also influence the flammability, such as MLR, ignition temperature, reactivity with oxygen, and smoke production.
Mass residue and mass loss rate
Figure 9 displays respectively the mass percentage (Figure 9(a)) and the MLR (Figure 9(b)) of the investigated materials. It is seen that the Bn-GBC sample starts to decompose at 100 s and it is completely degraded at 250 s, while the BM-GBC sample starts to decompose at the same time with a full decomposition at almost 300 s. The BM-GBC sample has a higher mass residue than that recorded by the Bn-GBC sample (almost 2%). This is due to the difference on the lignin content which is higher for the BnF compared to the BMF. This result is coherent with the MLR curves which shows that the Bn-GBC sample has a thin charring, while the BM-GBC sample has a thick charring which gradually decreases the mass loss rate. It might be because both natural fibres consist of lignocellulosic materials of around 22.37% lignin and 55.48% holocellulose.
53
Mass percentage (a) and MLR (b) evolution as function of time for the investigated samples.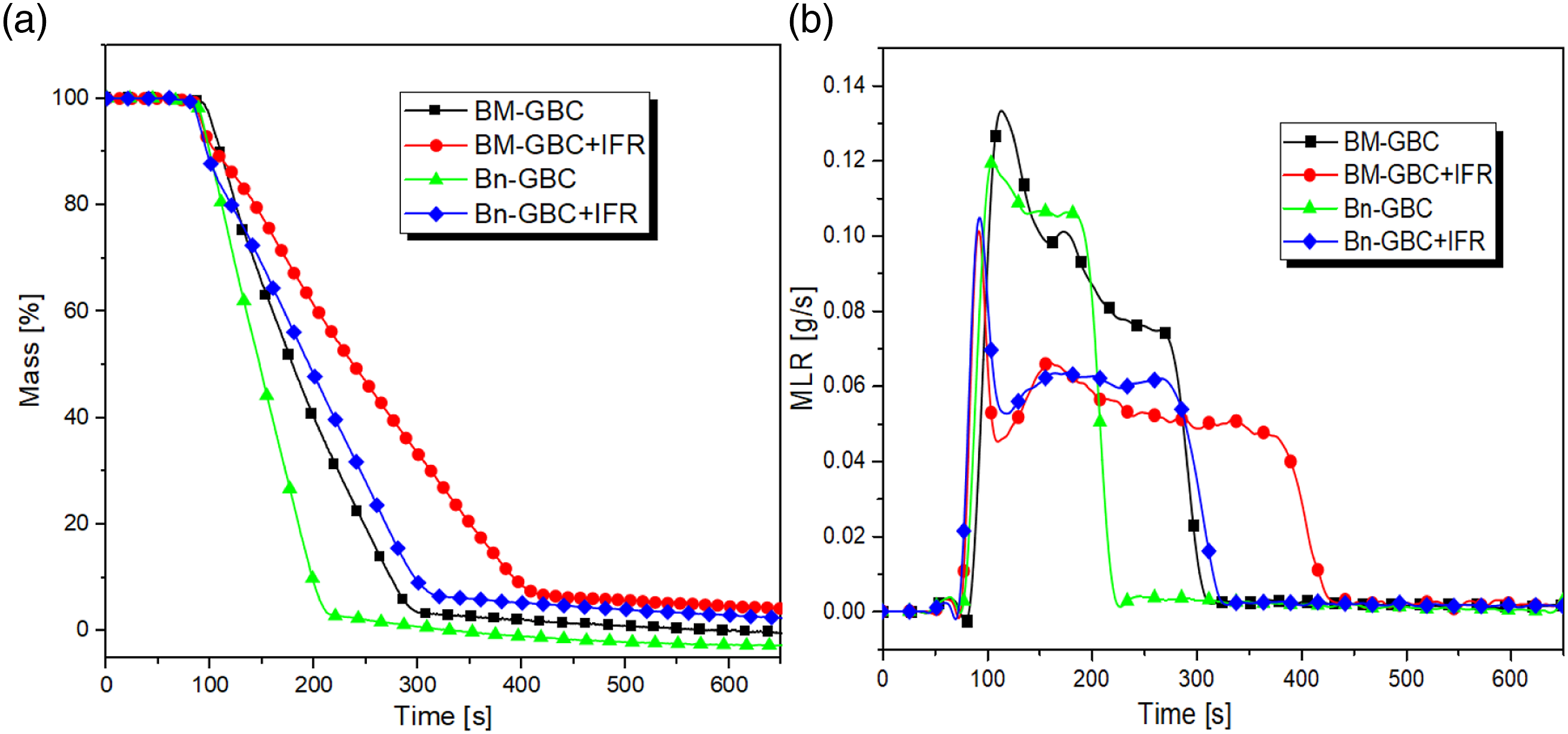
From the above discussion, one can conclude that the IFR coating on BM-GBC material is more effective that that applied on Bn-GBC material, in terms of delaying the thermal decomposition, because of its capacity to increase the burning time (see Figure 9(b)). On the other hand, the synergic effect of APP-THEIC and boric acid decelerates mass loss of BM-GBC + IFR by 8% which is in agreement with the mass loss trend discussed in the TGA subsection. After the initial sharp rising peak, a descending mass loss rate was observed for BM-GBC coated with APP-THEIC and boric acid (Figure 9(b)). This is maybe due to the thermoset Green-epoxy resin which creates covalently cross-linked networks with the synergism of borophosphate, since the IFR coating is maintaining the mass loss around 7-8% of each GBCs. The material was able to retain 50% the mass loss rate even when maximal level of fire developed and provided endurance against fire for 120–180 seconds. Both coated materials (BM-GBC + IFR and Bn-GBC + IFR) show thick charring. This behaviour indicates that the IFR coating protects the material surface by building a char residue on the surface. In addition, the IFR coating reduces the MLR of both GBCs, a high MLR can be an indication that the material is more flammable or has a lower thermal stability, which can be an important information for assessing the fire safety of the material.
Smoke production
The amount and type of smoke produced during combustion can be toxic and reduce the visibility during a fire. Figure 10 represents the following smoke production parameters: Total Smoke Production (TSP) (Figure 10(a)), the Specific Extension Area (SEA) (Figure 10(b)), the Smoke Production Rate (SPR) (Figure 10(c)) and finally the Time To Ignition (TTI) (Figure 10(d)). From the TSP curves, one notices that the BM-GBC sample produces the highest total smoke, while the lowest TSP is recorded by the Bn-GBC sample. by applying the IFR coating on both neat GBC surfaces, TSP is decreased by 5% and 10% for Bn-GBC and BM-GBC samples, respectively. TSP reduction has a significant impact on reducing CO2 and CO production (Smoke Production). In fact, the IFR coating presence reduces the TSP which may be due to the reduction of the amount of fuel available for combustion. This behaviour conducts to a less smoke generation, or to the heat transfer reduction which could correspond to a slower combustion. IFR presence leads to increase the time around 100–150 s to spread the smoke around the air. From the above discussion, it can be said that the IFR coating seems to be very effective due to the synergism of APP-THEIC and boric acid which build up borophosphate during the pyrolysis. Smoke production properties (a) TSP ;(b) SEA) ; (c) SPR; (d) TTI. 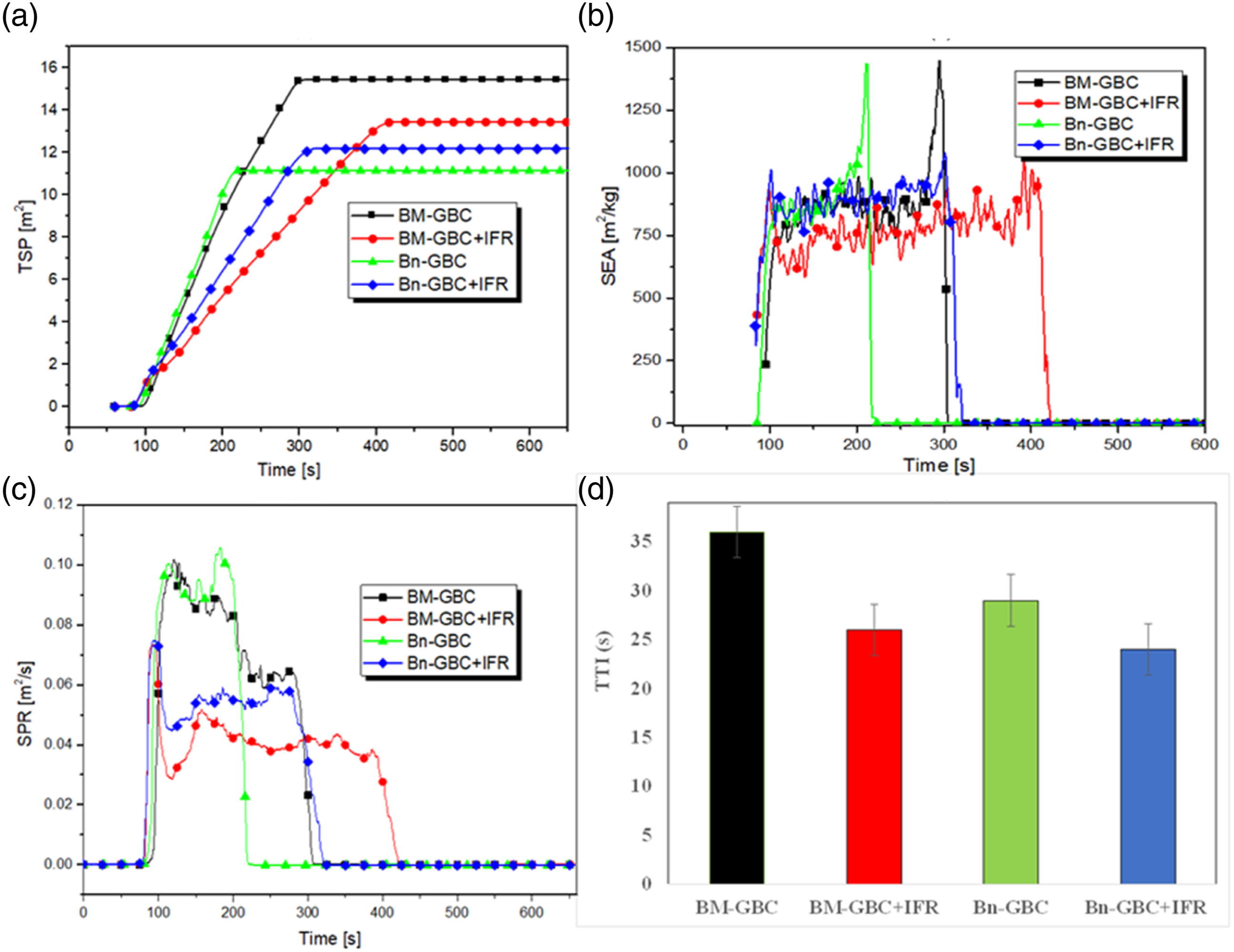
Figure 10(b) which represents the SEA evolution as function of time, confirms the noticed behaviour on the TSP results. This fact demonstrates that the BM-GBC sample has a larger SEA than that recorded by the Bn-GBC sample. In addition, the applied IFR on the surface reduces the average value of SEA. Consequently, the IFR coating leads to a decrease of the smoke production and delay the thermal decomposition of the BM-GBC sample. The SEA value of BM-GBC + IFR is 1194.9 at 391 s. This value is reduced by 26% compared to the uncoated BM-GBC sample. The reduction in SEA value is likely due to the improve surface properties of the coated BM-GBC + IFR, which reduces friction between the surface and surrounding air, resulting in improving the aerodynamic performance. In Figure 10(c), the SPR measurements as function of time is represented. High SPR values indicate that the material produces a large amount of smoke when it burns. Therefore, a high value of SPR may indicate that a material poses a greater health risk due to the toxic and cause respiratory problems. In similar result with TSP and SEA curves, SPR curves gives the information that BM-GBC has the highest value of smoke production rate. After IFR coating, the smoke production is significantly reduced by almost 40% and by 30% for the BM-GBC and Bn-GBC samples, respectively. Figure 10(d) shows the comparison of time to ignition (TTI) of all material, it can be seen that the Bn-GBC sample records lower TTI than that obtained by the BM-GBC sample. This observation indicates that the Bn-GBC is more thermally reactive than the BM-GBC sample. When the samples are coated with IFR, a decrease on TTI is noticed. This may be due to the topcoat contained higher amount of epoxy that has a low ignition temperature. The ignition time of Bn-GBC + IFR is 2–5 minutes, which is much higher than that of the uncoated Bn-GBC sample.
CO2 and CO production
Figure 11 represents the evolution of CO2 and CO production percentage as function of time. the results show that the BM-GBC and Bn-GBC samples have the higher CO2 production than the coated samples. For BM-GBC reduces around 54% and Bn-GBC reduces around 48%. For all material comparation, the highest CO2 is produced by BM-GBC. However, the CO production indicates that small thin peak is observed in the initial ignite time and undergoes decrease at 150 s. In this case, Bn-GBC + IFR has a higher CO production in the earlier stage than BM-GBC + IFR. The CO2 production is followed the order BM-GBC > Bn-GBC > Bn-GBC + IFR > BM-GBC + IFR, similar trend is noticed for the CO production beyond 150 s. It corresponds to a better fire resistance after IFR coating, the earlier stage might be due to the GBCs content of resin combined with the APP-THEIC. As mentioned above, CO2 and CO production are correlated with TSP (Total Smoke Production), which is mentioned in Smoke Poduction. The TSP reduction gives the slower combustion reaction can also result in reduced production of other combustion product such CO and CO2. This is because of the slower combustion reaction which provides more time for gases to be consumed by the reaction, rather than being emitted into atmosphere. CO2 (a) and Co (b) gas production as function of time.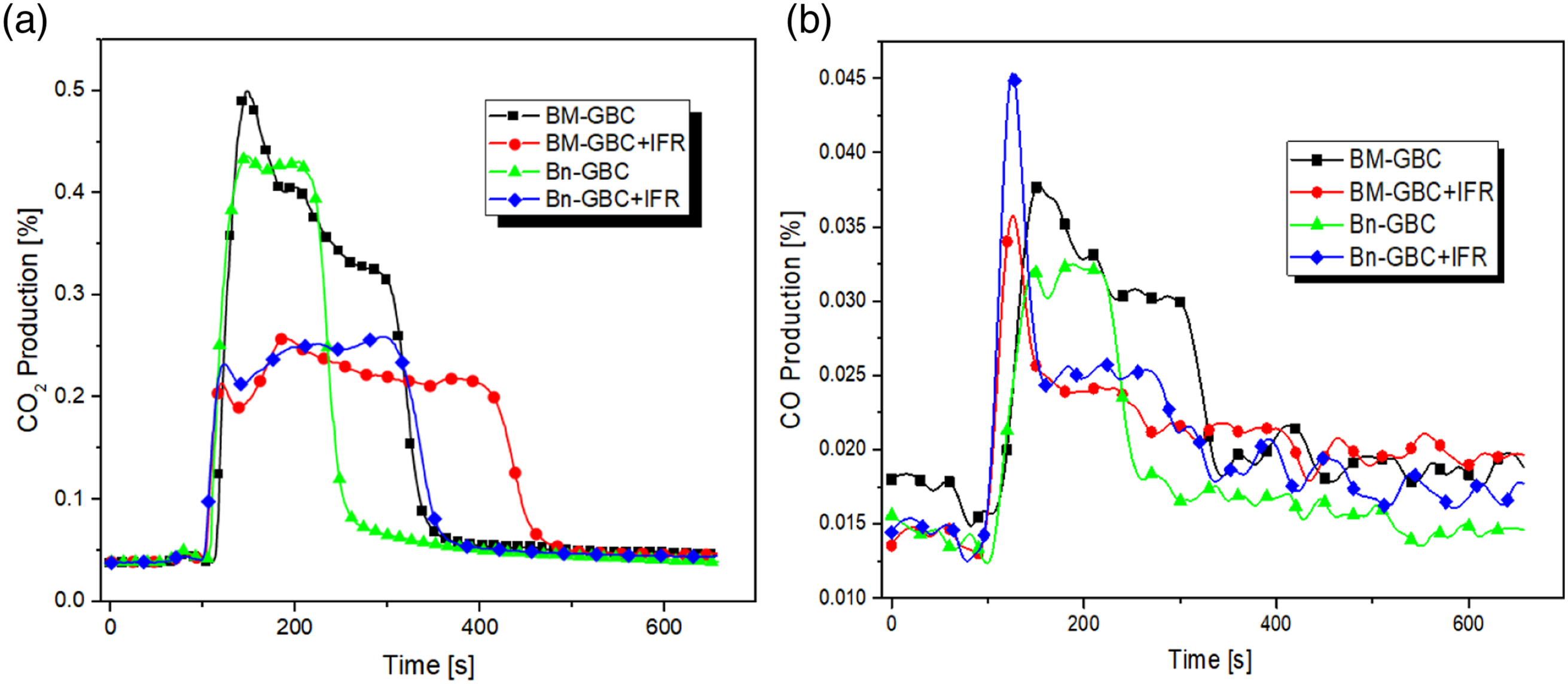
The parameter on cone calorimeter.
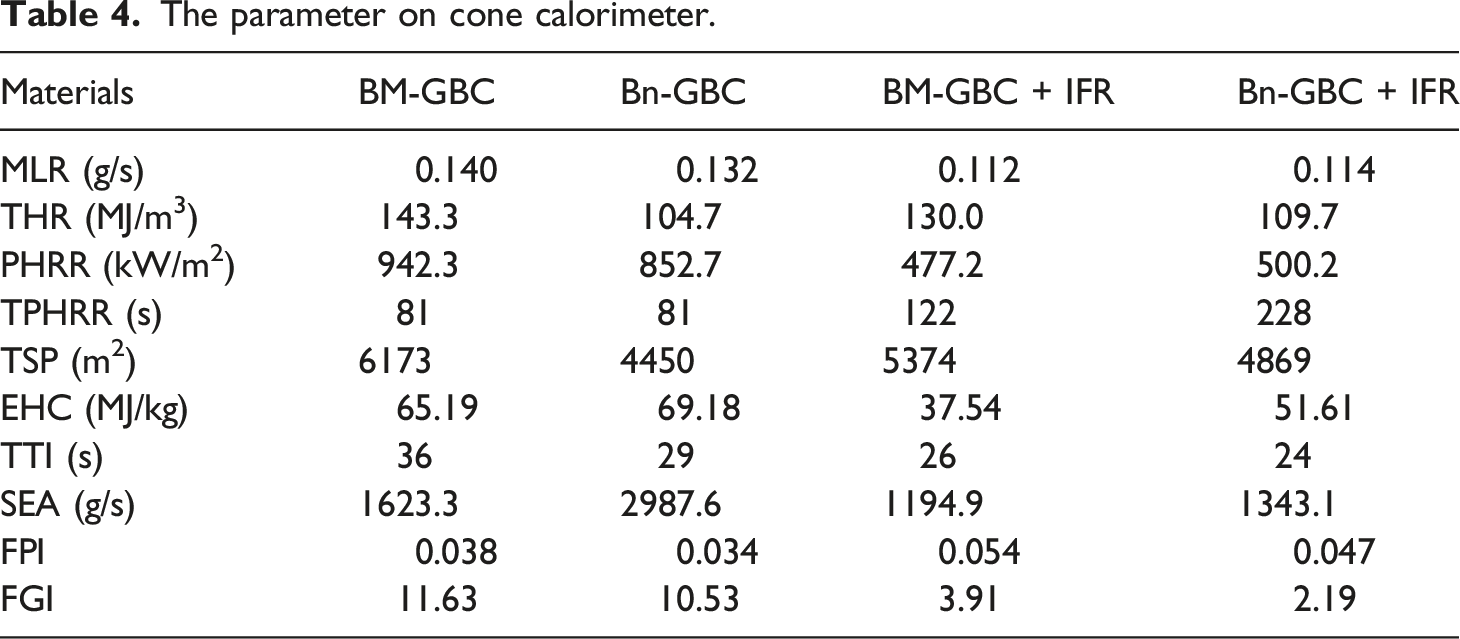
The visual appearance before and after (char) cone calorimeter test displays in Figure 12, it’s clearly seen that the presence of IFR build a foam at the surface of the material to protect the material from the fire exposition. The IFR addition improves flame retardancy that was attributed to forming an intact char layer (see Figure 12). The fire retardant develops a barrier during the combustion process called thermal shielding that slows the burning reaction. This shows that IFR coating is an effective fire-retardant material which can be protect material from fire, it can be proved by the SEM, EDS and FTIR analysis in the Char Residue Analysis. The materials before and after test cone calorimeter.
Char residue analysis
The SEM images of the char produced from Bn-GBC and Bn-GBC + IFR are displayed in the Figure 13(a) and (b), respectively. The char of Bn-GBC displays numerous spongy-like (hollows) morphology due to its micro-porous nature. The appeared hollows have an average length of 6.07 μm. The length of all appeared hollows is obtained using ImageJ software. The formation of surface voids can be attributed to the release of an excessive gas amount during combustion process, which leds to the near-total consumption of the Bn-GBC sample. Whereas the SEM micrograph of Bn-GBC + IFR (Figure 13(b)) reveals a decrease in the amount, size and thickness of the holes (cavities reef-like), indicating that IFR works effectively in sealing the porosities of Bn-GBC. Additionally, IFR tends to form agglomerates into a foam and reduce the average hollow length into 4.4 μm. This is due to the IFR degradation mechanism, which is a combination of APP-THEIC and boric acid. This interaction leads to generate borophosphate creating foam during the combustion. The protection mechanism is based on the char layer acting as a physical barrier, which slows down heat and mass transfer between the gas and condensed phases.
54
This suggest that IFR acts as a protective layer and enhances the fire resistance of Bn-GBC. This aspect will be more detailed in the next subsection. The SEM micrographs of (a) Bn-GBC. (b) Bn-GBC + IFR.
Figure 14(a) and (b) show the morphology of BM-GBC and BM-GBC + IFR char under SEM observation. The BM-GBC char shows clearly the multiples irregular holes resulted from the burning process despite this fibre has higher density than that Bn-GBC. The average diameter spherical holes before IFR coating is 34 μm. Whereas after IFR coated (Figure 14(b)), the holes are disappeared and becomes an intact and compact layer. It means that IFR coating can cover the surface completely by building a foam layer (similar with Bn-GBC + IFR sample) and brings them likely graphite structure char. The SEM micrographs of (a) BM-GBC. (b) BM-GBC + IFR.
The EDS result analysis of char all investigated material.
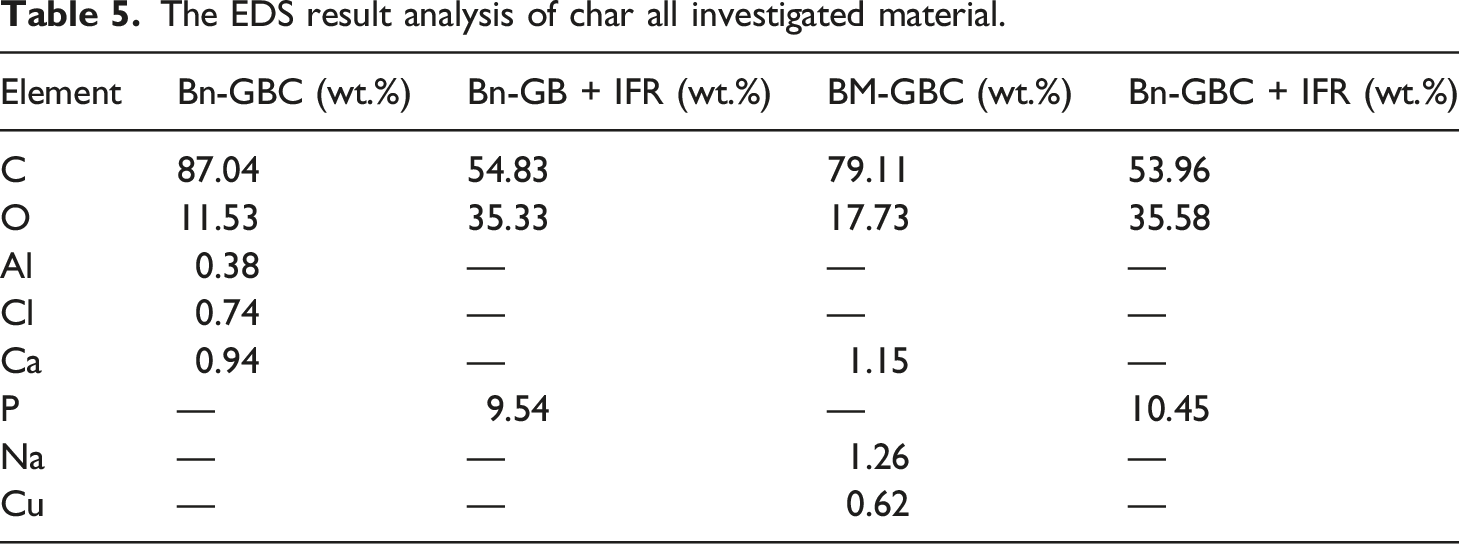
After applying IFR coating, both of Bn-GBC + IFR and BM-GBC + IFR char exhibit three abundant components: C, O and Phosphorus (P). Indeed, the presence of P component correspond to the combustion of APP-THEIC due to the control sample there is no P component. The high amount of C in the char residue confirms that a carbonized layer was formed during combustion by the IFR. This point has been confirmed by Figure 12 which depicts the Bn-GBC + IFR and BM-GBC + IFR char residue. Despite this, the EDS analysis did not reveal the presence of component B, likely due to the negligible weight percentage of boric acid in Bn-GBC + IFR and BM-GBC + IFR (merely 1%). These findings suggest that the IFR is working effectively, as it can form a protective carbonized layer during combustion.
Fire retardant mechanism
The cone calorimetry method is used to investigate the fire-retardant mechanism of materials over time, as demonstrated in Figure 15. The BM-GBC + IFR and Bn-GBC + IFR samples ignited after 100 seconds. Then, IFR influence can be observed at 200 seconds when the samples begin to suppress the horizontally spreading fire on their top surface. It was observed that both samples formed a char foam approximately at 300 s. However, at this point, Bn-GBC + IFR sample degrades faster and contains less fire compared to the BM-GBC + IFR sample. The height of the char measured of 4.2 cm and 4.5 cm for Bn-GBC + IFR and BM-GBC + IFR, respectively. The char layer acts as an insulator and plays a crucial role in enhancing the fire resistance of materials. These findings confirm the fire-retardant mechanism and prove the synergistic between APP and boric acid. Digital photos of (a) BM-GBC + IFR (b) Bn-GBC + IFR under cone calorimeter test.
In the comparison from the literature studies, the chemical bond of APP-THEIC (NH4)3PO4) can be represented: Nitrogen from the ammonium (NH4+) ion forms a covalent bond with the oxygen (O) atom from the phosphate (PO4)3− ion. Then, the nitrogen and oxygen atoms are linked by a shared pair of electrons to form a nitrogen-oxygen (N-O) bond. The remaining oxygen atoms of the phosphate ion are bonded to the nitrogen atom and to each by covalent bonds. This creates a repeating pattern of nitrogen and oxygen atoms, linked by covalent bonds, to form the structure of ammonium polyphosphate. While chemical bond boric acid can be presented from Boron (B) atom forms a covalent bond with three oxygen (O) atoms. Then, the boron atom shares a pair of electrons with each of the three oxygen atoms to form boron-oxygen (B-O) bond. The remaining oxygen atom are bonded to hydrogen (H) atoms by covalent bond. The chemical structure of APP and boric acid display in Figure 16. Chemical structure of APP (a) and boric acid (b).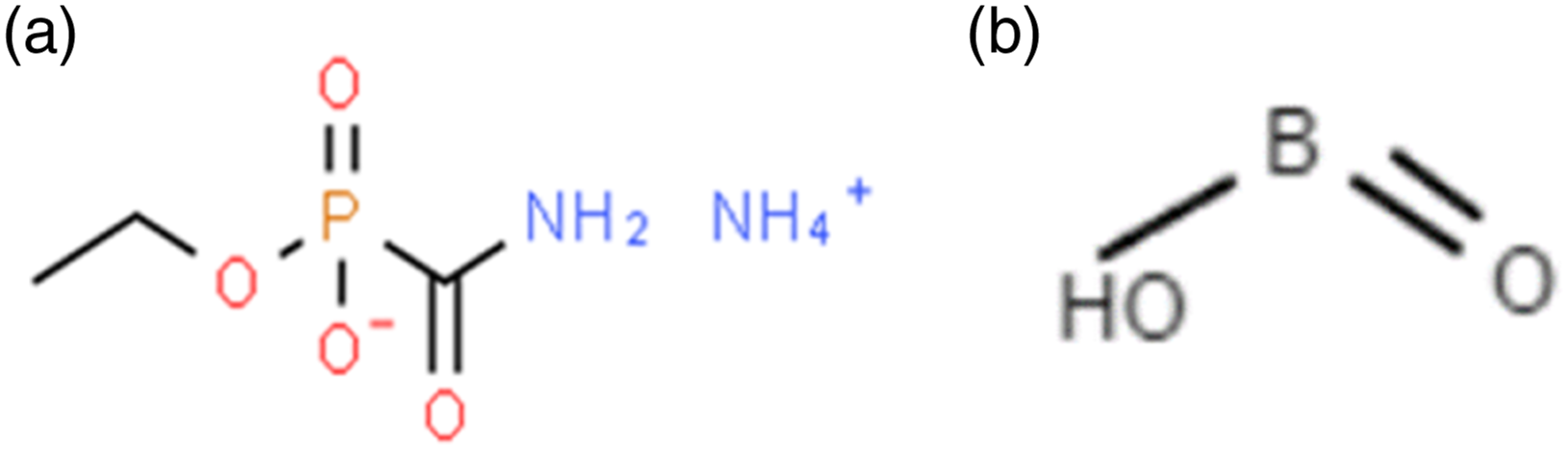
The synergism of (NH4)3PO4 (APP-THEIC) and boric acid (H3BO3) as IFR provides a reaction which occurs between the two compound to form new compounds. The reaction can be shows as follows: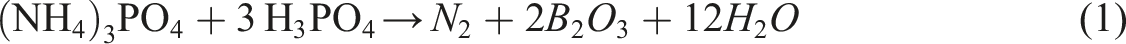
Moreover APP-THEIC in Exolit IFR36 acts as flame retardant (blowing) and boric acid function as a smoke suppressant. The combination of two components helps to reduce the mass loss of BM-GBC by decreasing the flammability and smoke production of the materials. This agrees with the result of the TGA analysis, which showed a decrease in mass loss of BM-GBC when two components were combined. The IFR coating was able to provide a high level of flammability, as it was able to retain 50% of the mass even when exposed to a maximal level of fire. The coating was also able to provide endurance against fire for 3 minutes.
The combination of Exolit IFR-36 and boric acid in IFR systems has good impact on enhancing the flame retardancy of biocomposite and delay decomposition process. It was proven by changes from two-steps reactions into three-steps mechanism reaction.


Since the TGA test using the temperature 30–900°C, there are three step temperature occurs at 140°C boric acid (HBO3) with loss of one water molecules (equation (2)) then 180°C undergoes to the reaction (equation (3)) which replaces another water molecule forming tetra boric-acid (proboric-acid/H2B4O7) then at 530°C continues to the reaction (equation (4)) which leads to boron trioxide.52,55 Furthermore, the synergism of boric acid and APP-THEIC involves the chemical routes of APP-THEIC decomposes to polyphosphoric acid and build up polyphosphate. 56 After combining with boric acid it undergoes to build borophosphate57,58 that expand when exposed to heat, forming a protective layer (as mentioned at equation (3)). This layer insulates the material from heat and slows the spread of fire. APP-THEIC on Exolit IFR26 produce function as a blowing fire-retardant agent, while boric acid act as a char-forming agent, which help to form a protective char layer on the surface of material.
Conclusion
This study was carried out to evaluate the thermal degradation and to improve the flame retardancy of two newly developed GBCs i.e. BM-GBC and Bn-GBC. A thermal decomposition study employing TGA revealed that thermal degradation pattern of Bm-GC and Bn-GBC was approximately same. A two-stage mass loss profile demonstrated similar onset and peak decomposition temperatures. When the two GBCs were coated with Exolit IFR 36 and boric acid IFR, the thermal decomposition became slower and higher amounts of char residue was recorded for both GBCs. Cone calorimeter fire test was conducted to characterize the fire performance and to evaluate the fire reaction behaviour of BM-GBC, Bn-GBC and their corresponding fire-retardant coated counterparts. Overall, BM-GBC exhibited slower thermal degradation and better fire reaction behaviour as compared to Bn-GBC. BM-GBC performed better might be due the presence of BMF, which contains higher lignin content as compared to BnF. When the two GBCs were coated with Exolit IFR 36 and boric acid, the thermal decomposition of both materials became slower and higher amount of residual char was recorded. The IFR coating also reduced the HRR, PHRR, TTI, EHC, TSP, and MLR of both materials BM-GBC and Bn-GBC, which indicates a reduction in the flammability of both materials. The effectiveness of IFR works better in the Bn-GBC according to the smoke production result.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Authors are grateful for the funding support BIB-MORA 5000 Doctor Scholarship in collaboration with Indonesia Endowment Fund for Education (LPDP) and Greensprink Laboratory INSA Centre Val De Loire France which gives high support for experimental studies and this work was supported by Lembaga Pengelola Dana Pendidikan (84).

