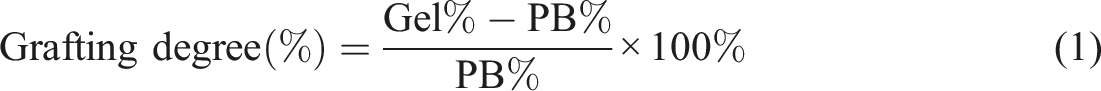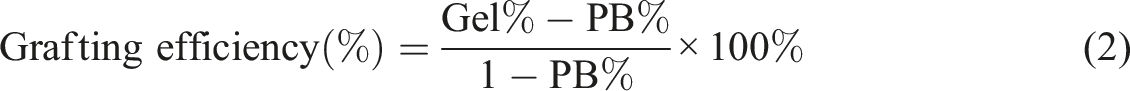Abstract
Submicron polybutadiene particles were prepared using four different methods: one-step batch emulsion polymerization, pressure agglomeration, acid agglomeration, and polymer latex agglomeration. Core-shell elastomers were synthesized by grafting styrene and methyl methacrylate on polybutadiene particles. Transparent methacrylate-acrylonitrile-butadiene-styrene resin was prepared by blending the elastomers with methacrylate-styrene-acrylonitrile matrix resin synthesized by bulk polymerization. The effects of the construction method of submicron rubber particles on core-shell elastomer structure and composite properties were investigated. Additionally, the relationship between the rubber phase structure and composite properties was studied. The obtained results showed that the construction method significantly affected the size distribution and morphology of the submicron rubber particles. The rubber particles prepared by one-step batch emulsion polymerization were grafted easily, with 90.2% grafting efficiency of the corresponding core-shell elastomer. The grafting efficiency of the rubber particles prepared by acid agglomeration was only 69.4%. The former was most suitable as the toughening core of transparent Acrylonitrile-butadiene-styrene resin, and the transmittance and haze values of the composite were 90% and 2.4, respectively. Impact and tensile strengths were 185.6 J/m and 39 MPa, respectively. The acid agglomeration method generated submicron rubber particles more quickly, and the transmittance and haze of composite were 90% and 2.9, respectively. However, the impact and tensile strengths were reduced to 170.4 J/m and 31 MPa, respectively.
Introduction
Acrylonitrile-butadiene-styrene (ABS) resin is an important engineering thermoplastic material, and is comprised of a rigid matrix resin with toughened rubber particles dispersed throughout.1–6 As a multi-component composite material, ABS resin has excellent comprehensive performance due to the complementary and cohesive advantages of each component. This material is widely employed in household appliances, automobile dashboards, construction pipes, medical equipment and electronic industries.7,8 Transparent methyl methacrylate-acrylonitrile-butadiene-styrene (MABS) resin is a special grade material based on simple ABS resin, and adheres to typical transparency requirements while exhibiting good mechanical properties. 9 Compared to other transparent materials that exhibit obvious disadvantages, for example, polystyrene (PS) with low mechanical properties, 10 polymethyl methacrylate (PMMA) with poor surface properties, 11 and polycarbonate (PC) with low surface hardness, 12 transparent MABS resin has become an excellent alternative due to its exceptional comprehensive performance.
ABS resin is a typical composite material, in which its opacity stems from the refraction and scattering of light at the interface of the two phases.13,14 It is known that there are two ways to produce ABS resin transparent: the first is that the refractive index of the rubber phase must be equal to that of the matrix resin,15–17 and the second is that the particle size must be small enough to avoid turbidity.18,19 However, the latter is not suitable to maintain the advantageous toughness of ABS resin, because the small sized rubber particles can not defend against crack propagation. 20 Therefore, matching the two-phase refractive index to improve transparency while maintaining excellent mechanical properties with sub-micron rubber particles is the greatest solution in the preparation of transparent ABS resin.
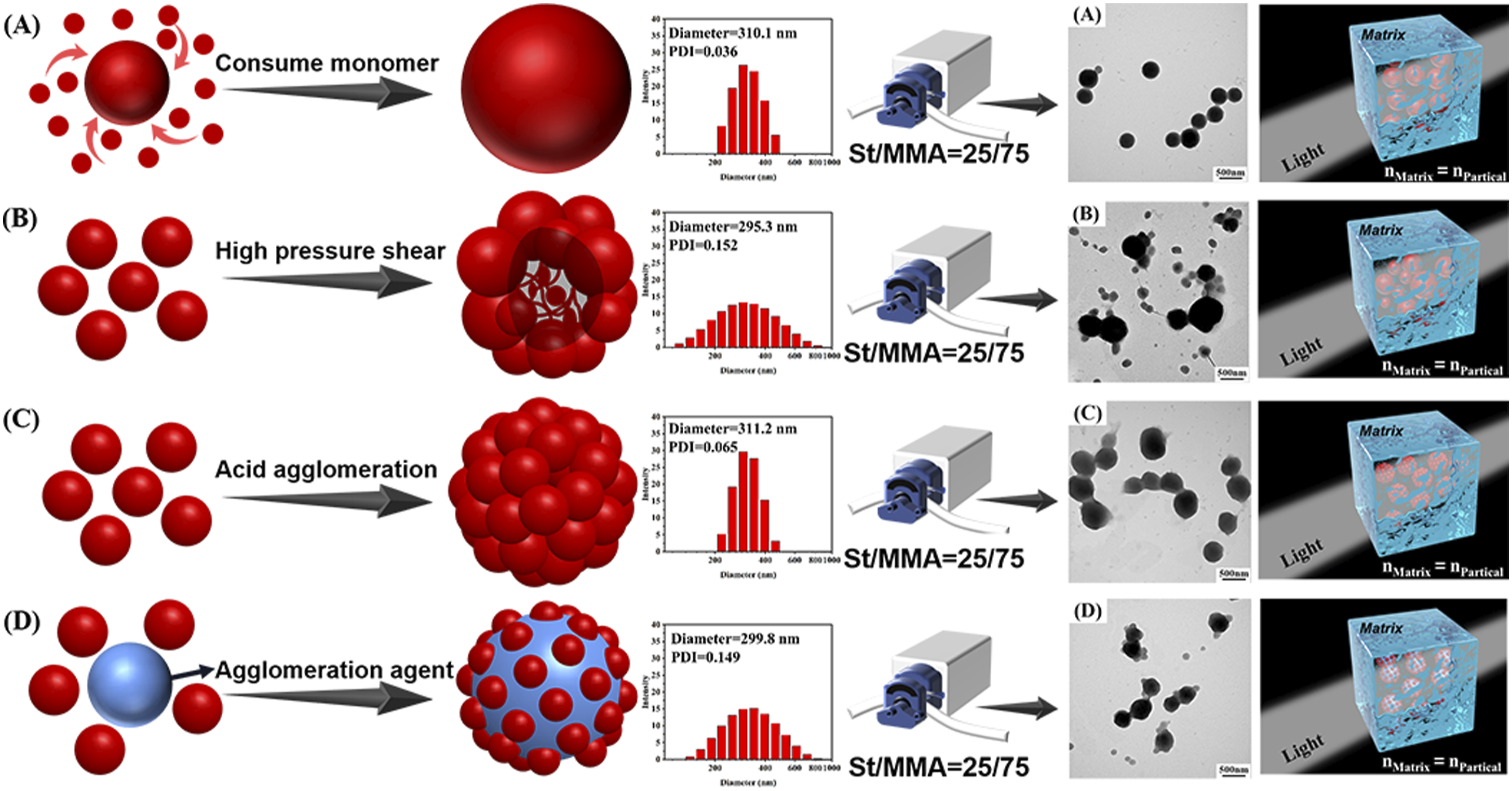
The common industrial preparation process of transparent ABS resin involves blending core-shell elastomer prepared by emulsion grafting and matrix resin synthesized by bulk polymerization. The preparation process of the matrix resin usually adopts bulk polymerization technology. However, the preparation process of submicron polybutadiene rubber particles that maintains excellent mechanical properties of transparent ABS resin is diverse. Currently, one-step polymerization, pressure agglomeration, acid agglomeration and polymer latex agglomeration are mature and widely used methods for the generation of large-size polybutadiene in industry. One-step batch emulsion polymerization technology produces submicron polybutadiene latex particles with narrow particle size distribution by controlling the ionic strength of the emulsion and interfacial tension between the polymer and reaction medium in the latex particles.21–23 Pressure agglomeration technology operates by passing small sized particles of polybutadiene latex through a homogenization valve with a narrow gap under high pressure. The latex particles are torn under high-pressure mechanical shearing force, where the latex quickly loses pressure after leaving the homogenization valve, and linear molecules of the rubber unprotected by the emulsifier quickly entangle together. As particle size increases, the emulsifier re-surrounds the particles to form a stable submicron polybutadiene latex.24,25 Acid agglomeration technology involves addition of weak acid to small particle size polybutadiene latex. Reduction of pH value in the system changes the charge distribution of the emulsified layer on the surface of the rubber particles, which promotes unbalance of the steady state of the electric double layer. The unstable particles collide and agglomerate through Brownian motion to form sub-micron polybutadiene rubber particles, and finally, via addition of the lye regeneration emulsifier, the stability of the emulsion is restored.26,27 Polymer latex agglomeration technology consists of the addition of unsaturated carboxylic acid copolymer latex containing hydrophilic segments into the small particle size polybutadiene latex. Molecular groups containing carboxyl groups can penetrate the emulsion layer of small particle size polybutadiene. These particles aggregate to form submicron particles due to bonding between the carboxyl groups and the unsaturated bonds of polybutadiene.28–31 Therefore, different processes in the preparation of polybutadiene latex essentially change the construction of the submicron rubber particles, as shown in Figure 1. Since equally matching the refractive index of the two phases is almost impossible, the difference in the microstructure of the phase region inevitably affects the performance of transparent ABS resin. Hence, selecting a suitable preparation process for submicron polybutadiene rubber particles is very important for the industrial production of transparent ABS resin. Preparation process of submicron polybutadiene particles. (a): One-step batch emulsion polymerization; (b): Pressure agglomeration; (c): Acid agglomeration; (d): Polymer latex agglomeration).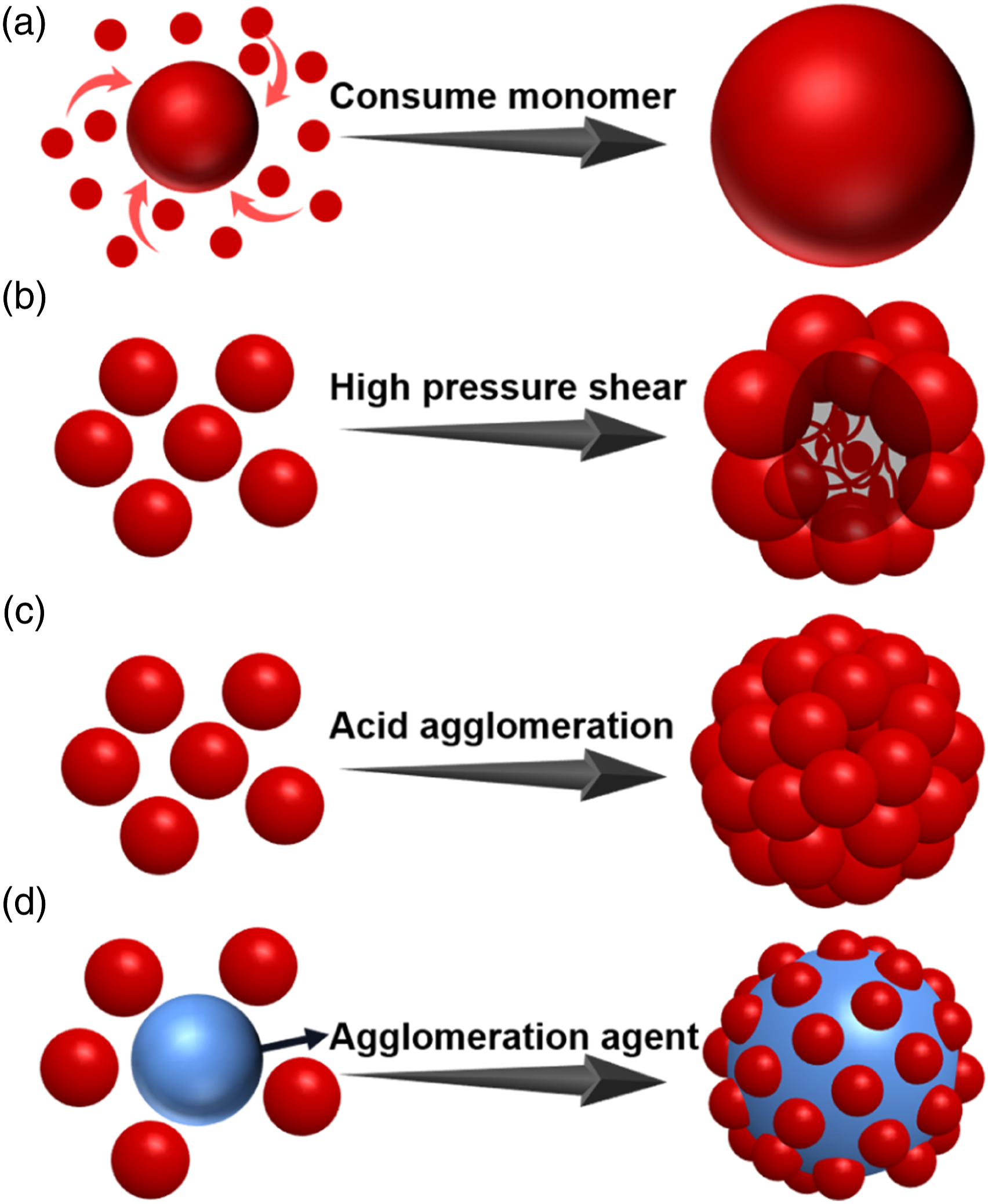
In this work, submicron polybutadiene rubber particles were prepared using four different architectural approaches. The microstructure of submicron polybutadiene particles was observed, and the effect of construction methods on the core-shell elastomers microstructure and transparent ABS resin properties were studied. Additionally, the most suitable preparation process of submicron polybutadiene latex was selected for industrial transparent ABS resin production. Finally, the relationship between the rubber phase microstructure and composite material properties was analyzed, which provided a reference for the structural design of the toughened rubber phase of the transparent ABS resin.
Materials and methods
Material
Butadiene (Bd), styrene (St), acrylonitrile (AN), methyl methacrylate (MMA) and tert-dodecyl mercaptan (TDDM), cumene hydroperoxide (CHP), disproportionate potassium rosinate (solid content 50%) and fatty acid soap (solid content 10%) used in this study were supplied by the Jilin Chemical Industry Group synthetic resin factory (Jilin, China). Sodium pyrophosphate (SPP), potassium persulfate (KPS), dextrose (DX), iron (II) sulfate (FeSO4), potassium carbonate (K2CO3), Dt-butyl peroxide (DTBP), butyl acrylate (BA) and potassium oleate were purchased from Shanghai Aladdin Bio-chem Technology Co., Ltd. Sulfuric acid, toluene and acetone were produced by Beijing Chemical Plant (Beijing, China).
Synthesis of methyl methacrylate-styrene-acrylonitrile (MSAN) resin
Bulk polymerization of MSAN resin was carried out in a 2 L glass tank reactor under nitrogen atmosphere. MMA, St, AN, DTBP and toluene were added to the reactor. The system was heated using externally circulating heated silicon oil to 130°C at a heating rate of 2°C/min for polymerization. The reaction was performed for 4 h with a stirring speed of 120 rpm. The product was dried in a vacuum oven at 200°C to remove residual solvent. The initial weight ratio of St and AN was 3/1. The initial weight percent of MMA was 78% of the total monomer mass.
Synthesis of small particle size polybutadiene latex
The polybutadiene latex was synthesized by emulsion polymerization in a 3 L reactor. Bd monomer, disproportionate potassium rosinate (2.0 wt%), potassium oleate (2.0 wt%), K2CO3 (0.8 wt%), KPS (0.3 wt%), TDDM (1.0 wt%) and deionized water were added to the reactor. Emulsion polymerization was performed at 68°C under constant stirring for 12 h.
Synthesis of submicron polybutadiene latex particles
One-step batch emulsion polymerization method: The polybutadiene latex was synthesized by emulsion polymerization in a 3 L reactor. Bd monomer, disproportionate potassium rosinate (2.0 wt%), fatty acid soap (0.4 wt%), K2CO3 (1.4 wt%), KOH (0.04 wt%), KPS (0.35 wt%), TDDM (0.25 wt%) and deionized water were added to the reactor. Emulsion polymerization was performed at 68°C under constant stirring for 27 h. The finally obtained latex was named PBL-Onestep.
Pressure agglomeration method: The small particle size latex is amplified into submicron latex after passing through a homogenization valve with a narrow gap under high pressure. The finally obtained latex was named PBL-Pressure.
Acid agglomeration method: The acid agglomeration was carried out in a 2 L glass flask at 40°C. 5 wt% aqueous solution of acetic anhydride was added dropwise to small particle size PBL latex (1000 g) under uniform stirring at a speed of 22.4 g/min. After 7 min, potassium hydroxide solution (40 g) with a concentration of 20 wt% was added. The finally obtained latex was named PBL-Acid.
Polymer latex agglomeration method: The polymer latex agglomerate was synthesized by emulsion polymerization in a three-necked flask at 70°C. BA (21.4 g), K2CO3 (0.15 g), SDS (0.8 g), KPS (0.3 g), TDDM (0.12 g), and deionized water (140 g) were added to the reactor, and the reaction was carried out under nitrogen protection for 30 min. BA (32 g) and MAA (6.6 g) were mixed and added dropwise to the reactor at a rate of 0.45 g/min. The agglomerates were obtained by reacting at 80°C for 1 h. The polymer agglomeration process was carried out in a 2 L three-necked flask at 45°C. Pour agglomeration agent (20 g) into small particle size polybutadiene latex (500 g) and react for 1 h to obtain submicron polybutadiene latex, which is named PBL-Polymer.
Synthesis of core-shell elastomers
Polybutadiene-styrene-methyl methacrylate graft polymer (PBL-g-SM) core-shell elastomer was synthesized via seed emulsion graft polymerization. First, the polybutadiene latex, emulsifier (4 wt%), SPP (1 wt%), DX (1 wt%), FES (0.02 wt%), CHP (0.8 wt%) and deionized water were added to the glass reactor and stirred for 10 min under nitrogen atmosphere. Then, the monomers were added via continuous feeding to the glass reactor in two-stages over 80 min in the order of St and then MMA. The weight ratio of polybutadiene latex to monomer was 6/4. The weight ratio of St and MMA was 1/3. The polymers were isolated from the emulsion by coagulation and dried in a vacuum oven at 60°C for 24 h. The core-shell elastomers corresponding to PBL-Onestep, PBL-Pressure, PBL-Acid, and PBL-Polymer grafted with monomers were named PBL-g-SM-Onestep, PBL-g-SM-Pressure, PBL-g-SM-Acid, and PBL-g-SM-Polymer, respectively.
Preparation of MABS resin
MABS resin was prepared by blending the matrix resin with PBL-g-SM at a weight ratio of 75/25 on a two-roll mill at 170°C for 5 min. Transparent ABS resins with PBL-g-SM-Onestep, PBL-g-SM-Pressure, PBL-g-SM-Acid, and PBL-g-SM-Polymer as toughening agents are named ABS-Onestep, ABS-Pressure, ABS-Acid, and ABS-Polymer in turn.
Characterization and measurements
The 1H NMR spectrum of the MSAN resin was recorded on a nuclear magnetic resonance instrument (NMR, Avance III 400M, Bruker, Germany) using deuterated chloroform (CDCl3) as the solvent and tetramethylsilane (TMS) as an internal standard.
The mean particle size and size distribution of the polybutadiene latex were characterized by the dynamic light scattering particle size analyzer (DLS, Zetasizer- ZS90, Malvern, UK). 1 mg/mL polybutadiene latex was dispersed in deionized water and automatically measured by DLS.
The morphology of the polybutadiene latex was observed using a transmission electron microscope (TEM, JEOL-1021, Japan) operating at an acceleration voltage of 100 KV. 32
The prepared PBL-g-SM (0.1 g) was dissolved in 30 g of acetone and shaken for 8 h at 23°C. The solution was centrifuged at 10000 rpm in a GL-21M ultracentrifuge for 20 min for 2 times. The ungrafted styrene-methyl methacrylate polymer was collected through pouring the supernatant into an appropriate amount of ethanol under intensive stirring and the acetone insoluble part was the grafted styrene-methyl methacrylate polymer. Grafting rate and grafting efficiency are calculated according to the formula.
33
The transmittance and haze of each sample were measured using a photometer (model WGW-1, Shanghai Shanke Instrument Factory, China). Samples with a thickness of 1 mm were obtained from a compression-molded sheet for this measurement.
The notched Izod impact strength was obtained using an Izod impact tester (model AJU-22, Chengde Tester Machinery Factory, China) at 23°C according to ASTM D-256. Tensile tests were performed according to ASTM D-638 at 23°C using an Instron tester with a crosshead speed of 50 mm/min.
The MABS were compression moldedto obtain specimen that are suitable for dynamic mechanical thermal analysis (DMA) measurements. These specimen were sized 50 mm × 6 mm × 1 mm. The DMA testing was carried out with the Netzsch DMA242 (Germany) in a single-can tilever mode. The dynamic mechanical measurements were performed over a temperature range from −110–150°C at a constant heating rate of 3°C/min, and at a frequency of 1 Hz.
The impact fracture surface morphology was observed by scanning electron microscopy (SEM, Model Japan JSM-6510) after coating the broken surface with a thin gold layer using a JFC-1600 Auto Fine Coater (Japan).
Results and discussion
Composition and refractive index of matrix resin
In order for ABS resin to be transparent, the refractive indices of the two phases must be close. The refractive index of polybutadiene is 1.515,
34
which requires the refractive index of the matrix resin to be similar. 1H NMR spectra of MSAN resin are shown in Figure 2. The multiplet at 6.6–7.6 ppm corresponded to protons of the aromatic ring.
35
The chemical shifts at 3.6 ppm were related to the methoxy protons. The broad singlet peak at 2.7–3.0 ppm corresponded to the proton of -CHCN-.
36
Each component proportion of MSAN was calculated from the peak area of the aforementioned protons.
37
The weight percentages of methyl methacrylate, styrene, and acrylonitrile in MSAN resin were 66.71%, 20.79%, and 12.5%, respectively. The refractive indexes of PMMA, PS and PAN were determined as 1.492, 1.591 and 1.52, respectively. According to the additivity principles and the Lorentz-Lorentz mixing rule,
38
the refractive index of the blends was 1.5158 and calculated as follows: 1H NMR spectra of MSAN resin.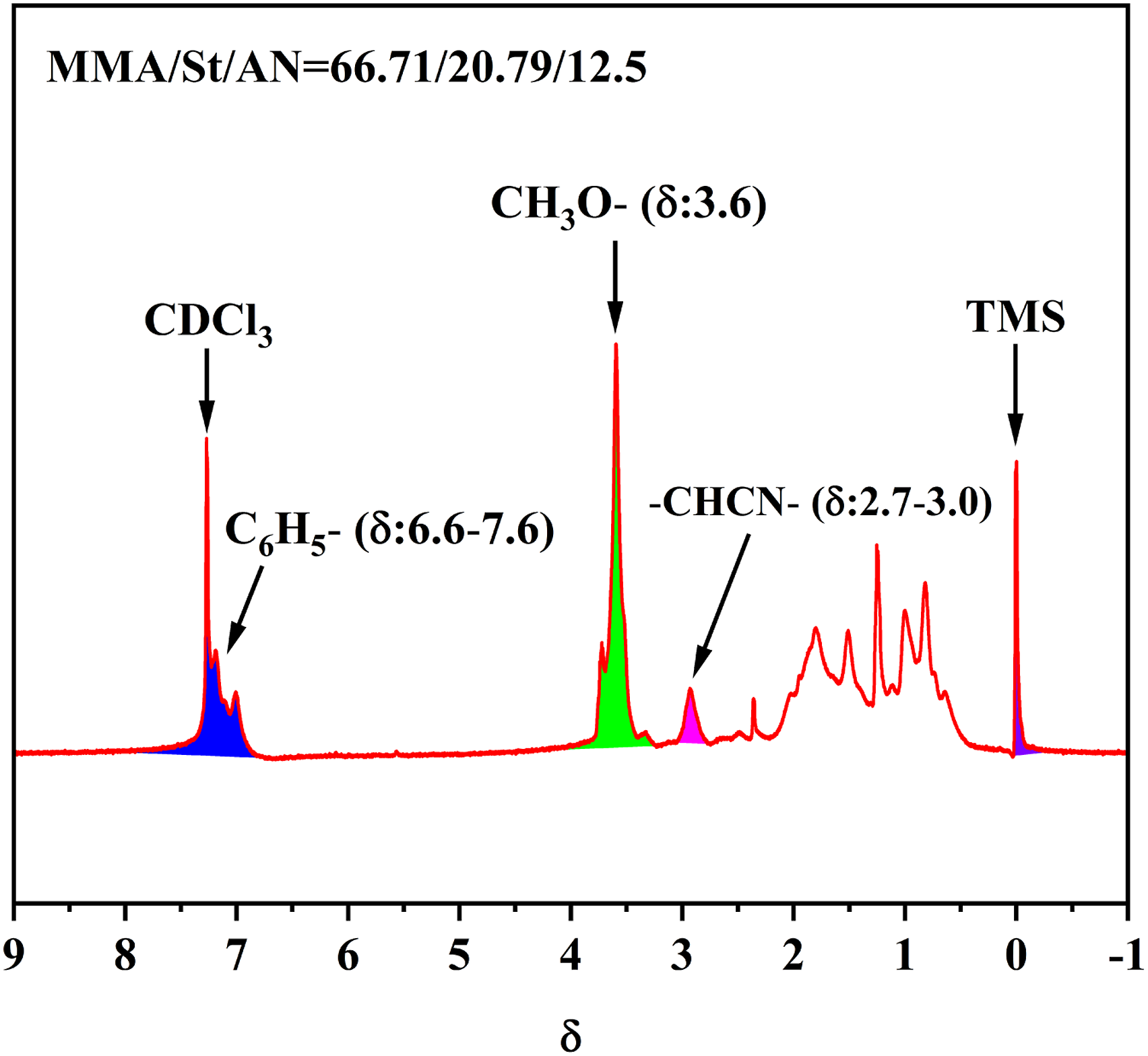
Size and size distribution of submicron polybutadiene latex particles
The particle size of polybutadiene latex is approx 300 nm, which is commonly used in industry.
40
The particle size and distribution of submicron polybutadiene latex particles prepared by different construction methods are shown in Figure 3. Significant differences in the size distribution of the polybutadiene latex particles was observed. The particle size diagrams of the submicron polybutadiene latex particles prepared by one-step method and acid agglomeration method showed narrow dispersion, and the dispersion indices were 0.036 and 0.065, respectively. The particle size diagrams of the particles prepared by pressure agglomeration method and polymer latex agglomeration method showed broad distribution, and the dispersion indices were 0.152 and 0.149, respectively. Average particle size and size distribution of submicron polybutadiene latex particles. (a): One-step batch emulsion polymerization; (b): Pressure agglomeration; (c): Acid agglomeration; (d): Polymer latex agglomeration).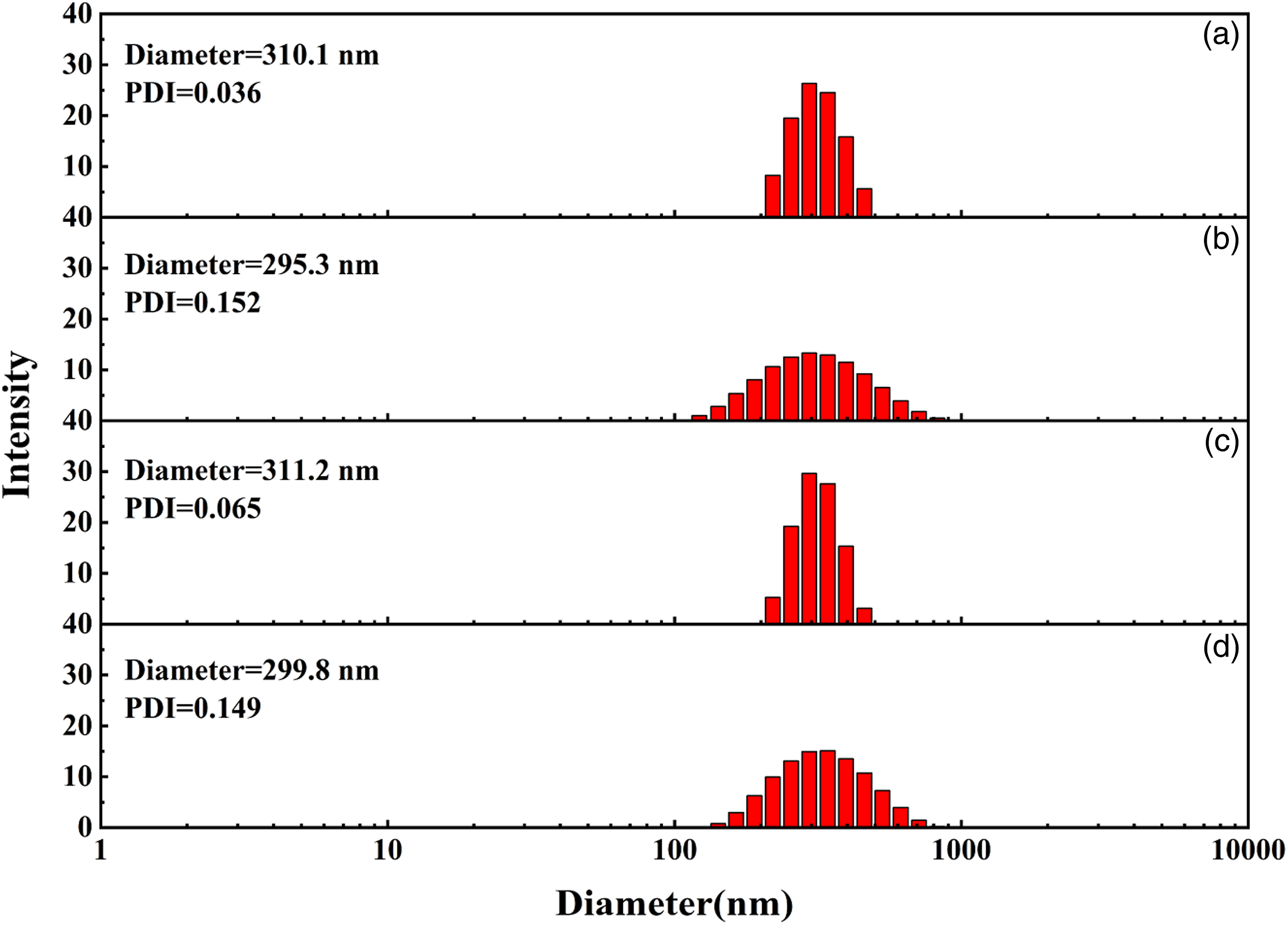
Morphology of core-shell elastomers
The transmission electron micrographs intuitively show the size distribution and appearance of the core-shell elastomers PBL-g-SM-Onestep, PBL-g-SM-Pressure, PBL-g-SM-Acid, and PBL-g-SM-Polymer in Figure 4. The size distribution of the core-shell elastomer depended on the size distribution of the submicron polybutadiene latex particles. PBL-g-SM-Onestep possesses an isolated full regular spherical shape due to the direct synthesis of submicron polybutadiene latex particles. Reports have shown that the increase in latex particles size occurs when the monomer droplets continuously enter the interior of the latex particles.
41
In our case, the elastomer was regular in shape and uniformly covered by the graft layer due to the lack of particle collision. The size distribution of PBL-g-SM-Pressure was broad and the rubber particles had an irregular spherical shape. The rubber particles were deformed due to high pressure used when they pass through the homogenizing valve, and the emulsified layer was destroyed due to the shearing effect. The linear molecules of the rubber particles were entangled and formed large-sized particles when the pressure was removed. The rapid reorganization of rubber particles under physical activity resulted in wide size distribution and irregular edges of latex particles. PBL-g-SM-Acid exhibited spherical shape with irregular edges. The introduction of free acid changed the charge distribution of the emulsion layer on the surface of the rubber particles, which disrupted the local stability of the emulsion. Thus, the unstable rubber particles collided through Brownian motion, promoting agglomeration. The interfacial tension between particles and water provided the driving force for particle fusion to decrease with increasing particle size. These weak forces were responsible for the irregular edges on the submicron polybutadiene. There are many small particles adhering to the surface of PBL-g-SM-Polymer. Agglomerates bearing numerous carboxyl hydrophilic segments on the surface, aggregate small polybutadiene particles into large particles via chemical bonding. The agglomeration capacity of the agglomerator depended on the hydrodynamic shell thickness and extension of the agglomerator’s hydrophilic segment. When the agglomeration agent reached saturation, some small particles remained unfused, which formed bulges on the surface as shown in Figure 4(d). Transmission electron micrographs of PBL-g-SM-Onestep (a), PBL-g-SM-pressure (b), PBL-g-SM-acid (c) and PBL-g-SM-polymer (d).
Grafting rate and grafting efficiency
Figure 5 shows the grafting ratio and grafting efficiency of submicron polybutadiene latex particles synthesized by different construction methods. The submicron toughened particles synthesized by one-step method exhibited the highest grafting rate and grafting efficiency, 60.1% and 90.2%, respectively. The grafting effect of submicron polybutadiene particles obtained by agglomeration of small particles varied greatly, in which the grafting rate and grafting efficiency of PBL-g-SM-Pressure were 57.7% and 86.6%, respectively. The presence of large rubber particles reduced the specific surface area of the rubber particles and led to a decreased grafting effect. The submicron toughened particles synthesized by acid anhydride agglomeration had the lowest grafting rate and grafting efficiency, 46.2% and 69.4%, respectively. This may be related to the significant looseness of the particle structure. The grafting rate and grafting efficiency of PBL-g-SM-Polymer were 54.4% and 81.6%, respectively. The bonding between the carboxyl group and the unsaturated double bond reduced the amount of grafting sites, which reduced the overall grafting effect. Grafting rate and grafting efficiency of PBL-g-SM-Onestep, PBL-g-SM-Pressure, PBL-g-SM-Acid and PBL-g-SM-Polymer.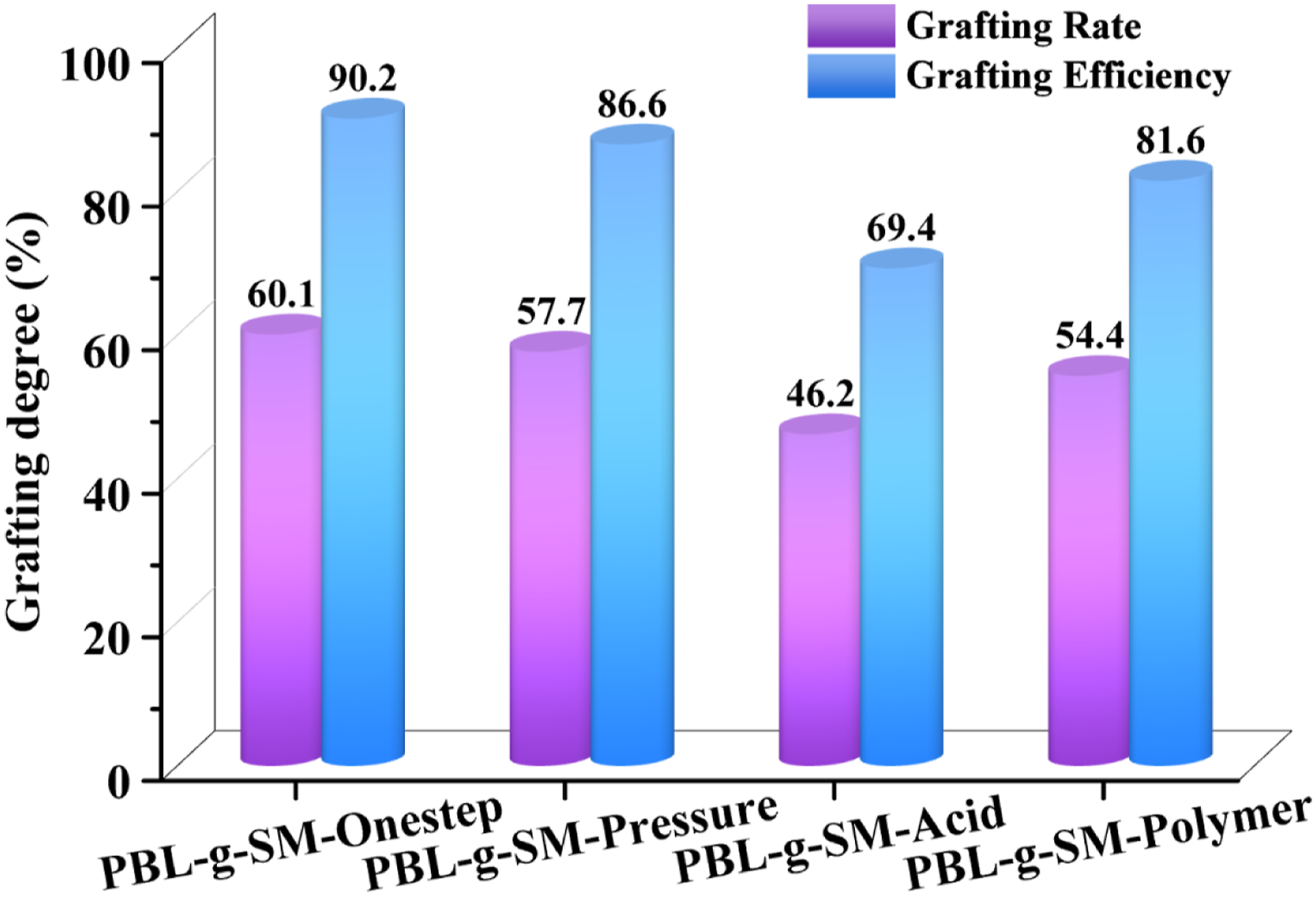
Optical properties of transparent ABS resin
Figure 6 shows the transmittance and haze values of the transparent ABS resin with different toughened cores. The transmittance of ABS-Onestep and ABS-Acid were determined as 90%, however, their haze values were 2.4 and 2.9, respectively. The irregular particle morphology resulted in enhanced light scattering. The transmittance and haze value of ABS-Pressure was 89% and 4.4, respectively. The reason for the poor optical properties stemmed from broad distribution of toughened particles. According to Rayleigh’s law, large rubber particles caused more scattering of incident light at the interface.
42
The data obtained from the haze value of ABS resin and size distribution of the toughened particles revealed that as the size distribution of the toughened particles widened, the haze value of the transparent ABS resin increased. The transmittance and haze value of ABS-Polymer was 88.5% and 3.7, respectively. The observed optical deterioration of the transparent ABS resin occurred due to the introduction of the polymer agglomeration agent. The polymer agglomerator consisted of butyl acrylate (BA) and methacrylic acid (MAA). Incorporation of other components changed the refractive index of rubber phase, which led to more refraction and scattering of the incident light at the interface of the two phases. In general, toughened particles prepared by different construction methods allowed transparent ABS resin to obtain good visibility. The particle structure had a significant influence on the haze of transparent ABS resin. The haze of the transparent ABS resin was more affected by the structure of the toughened particles. The transmittance of the transparent ABS resin was more affected by the difference in refractive index between the two phases. Compared with the PMMA/SAN/MBS blends prepared by Chen and Ren et al., the transmittance has been improved.43,44 Transmittance and haze value of ABS-Onestep, ABS-Pressure, ABS-Acidand ABS-Polymer.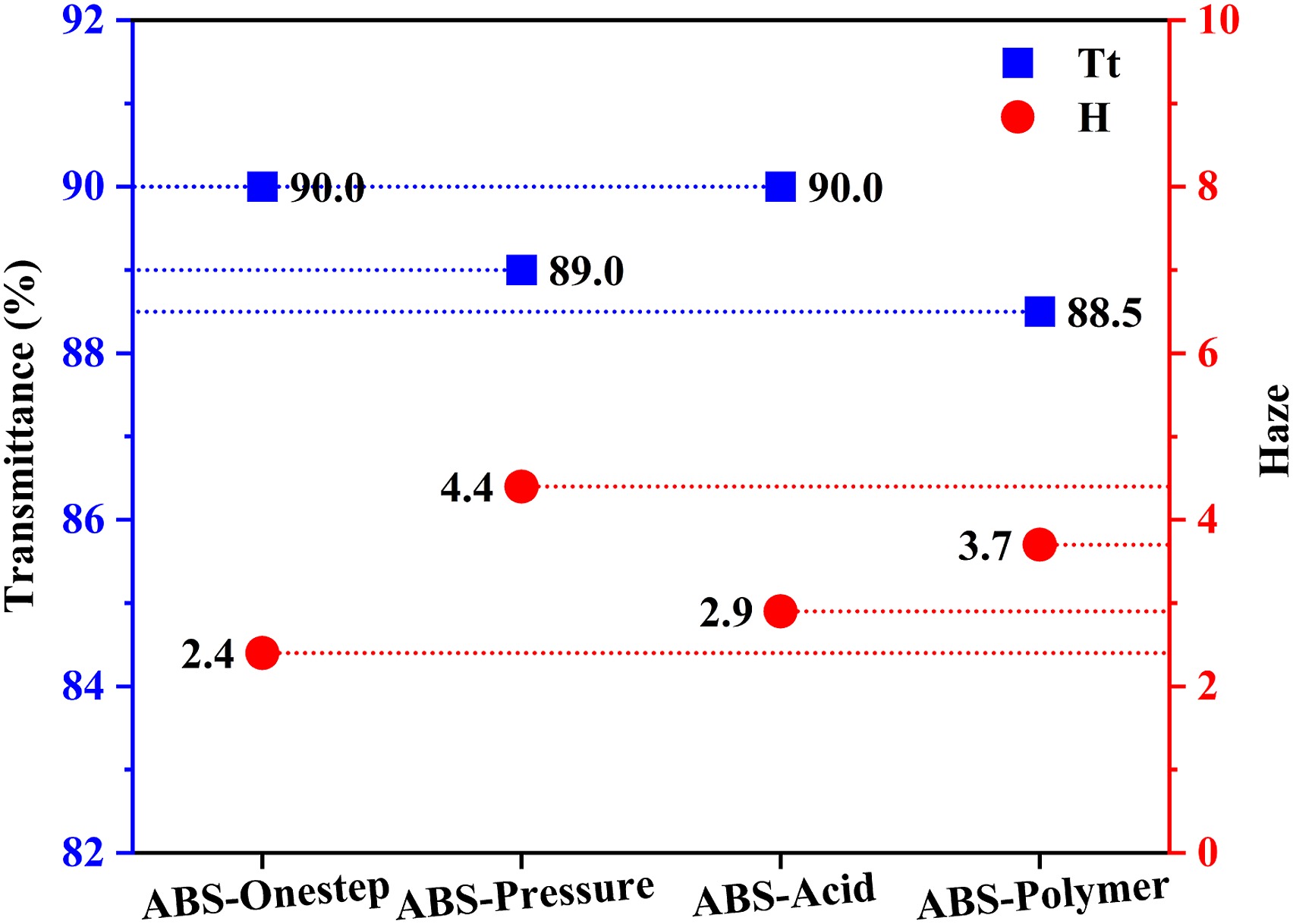
Mechanical properties of transparent ABS resin
The notched impact strengths of the transparent ABS resins with core-shell particles prepared by different construction methods were above 170 J/m, as shown in Figure 7. The toughness of the transparent ABS resin was enhanced compared to that prepared by Song et al.20,45 The high impact strength of the transparent ABS resin depended on the high AN content in the matrix resin and excellent toughening of rubber particles. The submicron rubber toughened particles acted as stress concentrators to facilitate the dissipation of impact energy. The matrix resin was more conducive to toughening by rubber particles due to the highly polar groups on the molecular chain of acrylonitrile. The impact strengths of ABS-Onestep and ABS-Polymer were slightly higher, at 185.6 J/m and 181.7 J/m, respectively. The highest impact strength of ABS-Onestep was due to the excellent grafting effect and compact particle structure. The high graft ratio enhanced the interaction between the rubber particles and the matrix and promoted stress transfer between the two. The entanglement of the rubber chains by the polymer agglomerates led to the transfer of stress and energy dissipation. The notched impact strength of ABS-Pressure was 175.6 J/m. The wide distribution of rubber particle size contributed to an excellent toughening effect. Large rubber particles better induce silver streaks, and a larger number of small rubber particles resulted in shear deformation as they absorb more energy and effectively inhibit the growth of crazes.
40
The notched impact strength of ABS-Acid was relatively low at 170.4 J/m due to the low graft ratio and soft monodisperse rubber particles. A large amount of free copolymer hindered impact properties. The notched impact strength of ABS-Onestep, ABS-Pressure, ABS-Acid and ABS-Polymer.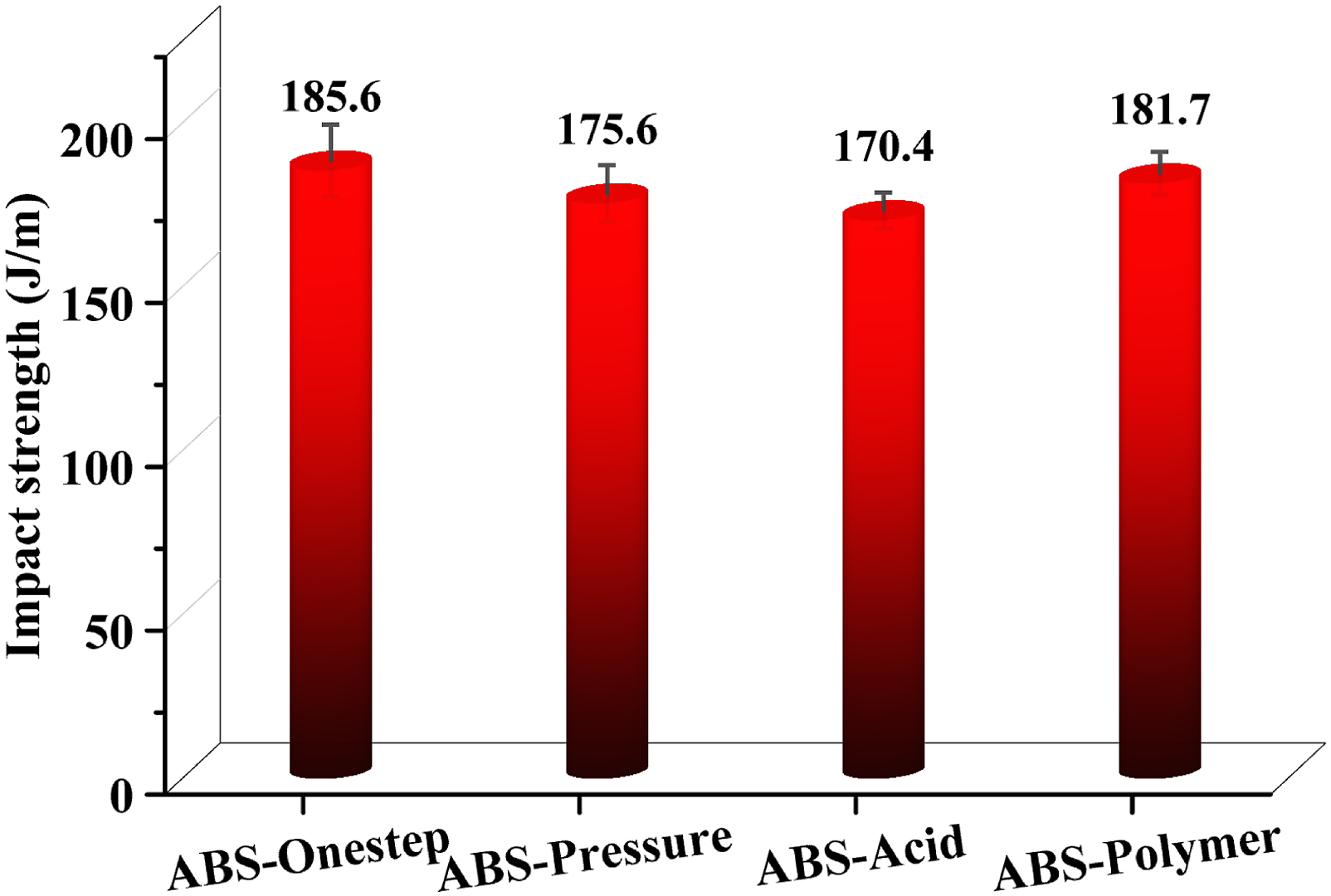
Figure 8 shows the stress-strain curve of transparent ABS resin with core-shell toughened particles prepared using different construction methods. ABS-Onestep displayed the highest yield and breaking strength, 39 MPa and 35 MPa, respectively. The high graft ratio enhanced the bonding strength between the rubber particles and matrix, which benefited the tensile properties. The regular spherical particles reduced the defects of the material and enhanced tensile strength of the transparent ABS resin.
46
The yield strength and breaking strength of ABS-Pressure and ABS-Polymer were similar, approx 35 MPa and 33 MPa, respectively. The good grafting effect provides the ABS resin with good tensile strength. The higher elongation at the break of ABS-Polymer was due to the tight toughened particle structure. ABS-Acid had the lowest yield strength and breaking strength, 31 MPa and 29 MPa, respectively. The low graft ratio made it easier for the grafted chains to unravel upon subjection to external stress, and thus yielding at very low stress. The irregularly shaped toughened particles were also responsible for their poor tensile properties. Combined with the morphology and tensile strength analysis of the toughened particles, it was determined that the regular and compact spherical particles were more conducive to reducing material defects that benefited the tensile properties and elongation at the break of the transparent ABS resin. The stress-strain curve of ABS-Onestep, ABS-Pressure, ABS-Acid and ABS-Polymer.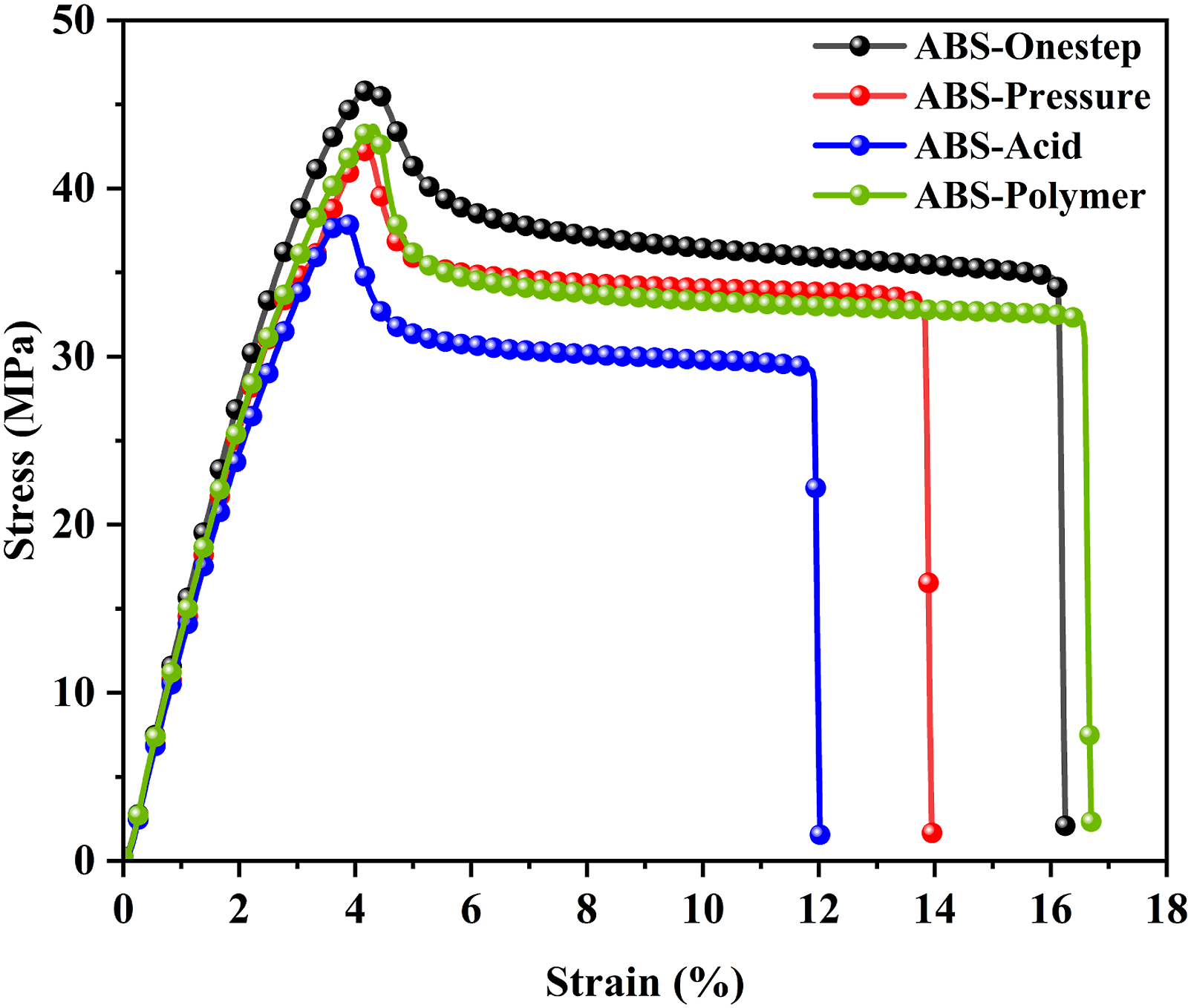
Dynamic mechanical thermal analysis
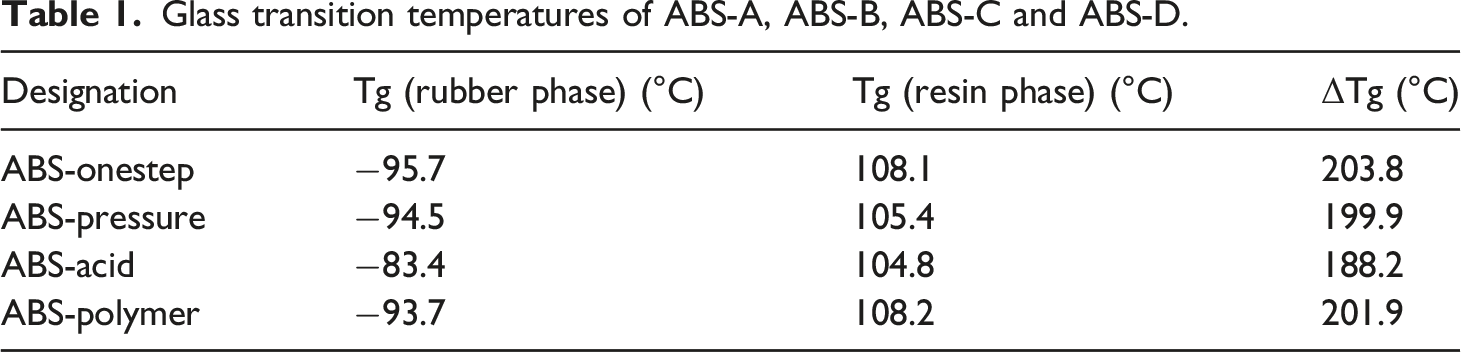
The glass transition behavior and dynamic storage modulus of the samples were measured by DMA. The tanδ curves and storage modulus of all samples (ABS-Onestep, ABS-Pressure, ABS-Acid and ABS-Polymer.) are exhibited in Figures 9 and 10, respectively. Due to the incompatibility of the rubber phase and matrix phase, ABS resin gave two glassy loss peaks for the rubber phase and matrix phase, respectively. The low- and high-temperature regions represented the glass transition process of the rubber phase and matrix phase, respectively. The corresponding temperature of the maximum tanδ was defined as the glass transition temperature (Tg). The two glassy loss peaks revealed the heterogeneous nature of the two phases and the difference values (ΔTg) of tanδ peak temperatures of both phases indicated the degree of compatibility between the phases. The glass transition temperatures of each phase are listed in Table 1. The best two-phase compatibility was obtained from ABS-Acid, which was attributed to the loose structure of the toughened particles. ABS-Acid was easily penetrated by MMA during the grafting process, which promoted two-phase compatibility. The tanδ peak of the rubber was shifted towards high temperature due to the penetration of monomers. The heterogeneity of the rubber phase increased the phase transition width of the corresponding tanδ peak. The tanδ shift of the rubber phase of ABS-Polymer was towards high temperature due to the agglomerates present in the rubber phase. The level of tanδ represented the dynamic transition amplitude of one component of the composite material. The maximum value of tanδ of the rubber phase first increased and then decreased with increasing graft ratio. The increase in the grafting ratio effectively dissipated the applied energy, resulting in increased height of tanδ. The observed decrease in the maximum tanδ value at the highest degree of grafting stemmed from the highly grafted chain plug restricting the movement of the rubber. Dynamic mechanical properties of ABS-Onestep, ABS-Pressure, ABS-Acid and ABS-Polymer. Storage modulus of ABS-Onestep, ABS-Pressure, ABS-Acid and ABS-Polymer. Glass transition temperatures of ABS-A, ABS-B, ABS-C and ABS-D.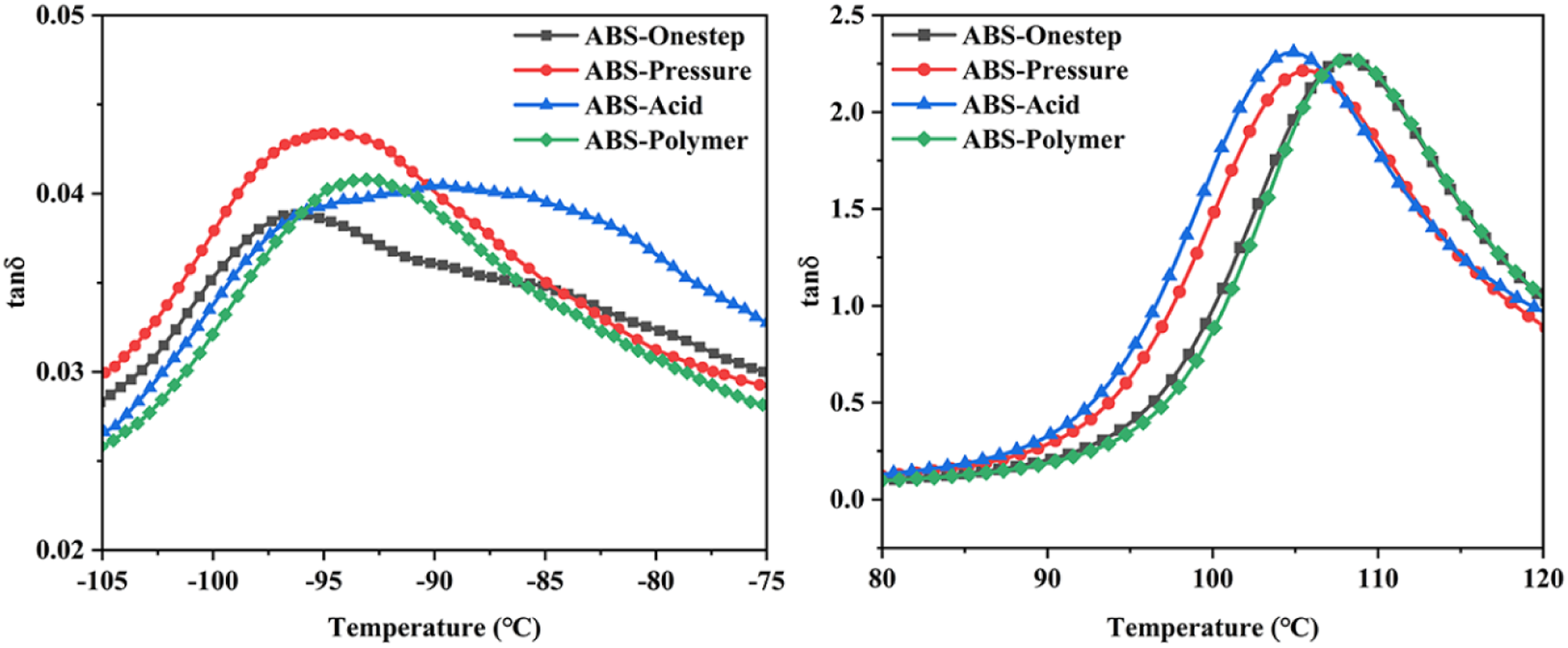
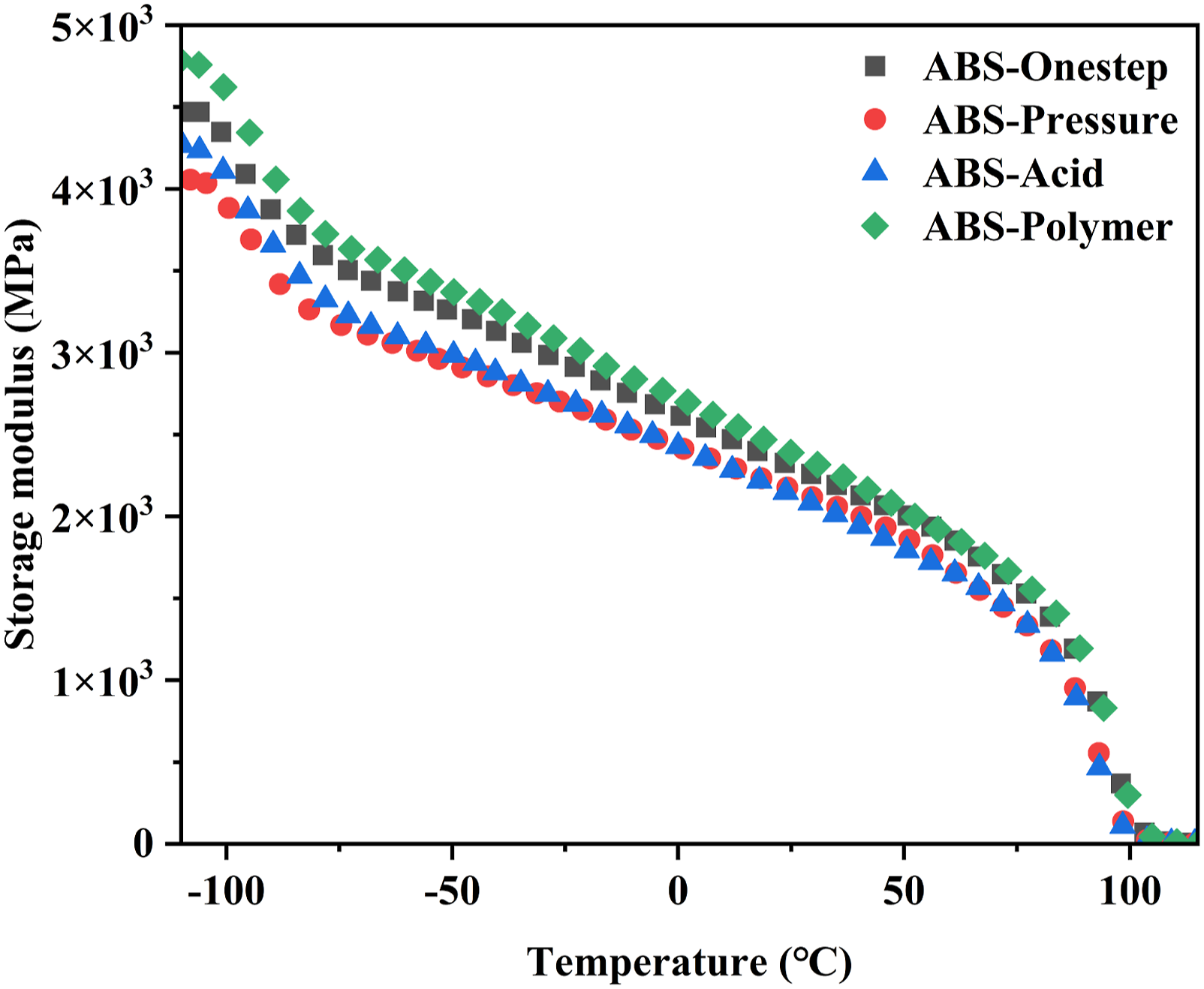
The dynamic storage modulus (E′) is the most important property to assess the load-bearing capability of materials and characterize the stiffness of materials. 47 A high storage modulus indicated excellent stiffness of the material. Since polybutadienes are amorphous polymers, their storage moduli decreased dramatically at higher test temperature compared to Tg value, as shown in Figure 10. The difference in modulus depended on the different construction methods used. ABS-Polymer had the highest modulus because of the molecular bonding of the polybutadiene with the coagulant and the presence of PBA and MAA in the rubber phase. The high modulus of ABS-Onestep was attributed to its compact particle structure and high graft ratio. The low storage modulus of ABS-Acid was due to its loose particle structure and low grafting ratio. The reduction of the entanglement between the two phases easily led to intermolecular slippage, resulting in reduced storage modulus of the blends. The lowest storage modulus of ABS-Pressure stemmed from the larger rubber particles. According to the literature, larger size rubber particles are detrimental to the stiffness of the blend. Mechanical loss factor (tanδ), the ratio of storage modulus and loss modulus, is a measure of the balance between the stiffness and toughness. Combined with the impact toughness and storage modulus analysis, the balance of stiffness and toughness of the transparent ABS resin with the toughened core prepared by the one-step method was the best.
Fracture surface analysis
SEM photographs shown in Figure 11 exhibit the impact-fractured surfaces of the transparent ABS resin. In general, the toughness of polymer blends was visually observed in the obtained SEM photographs. A rough impact-fractured surface corresponded to the ductile polymer blends with a high impact strength and elongation at the break. The addition of low-modulus PBL-g-SM rubber particles deviated the direction of the crack propagation within the matrix and caused rough fracture surfaces. Hence, PBL-g-SM rubber particles significantly improved the material’s toughness. The fractured surfaces of MABS resins showed many grooves, indicating that shear bands were formed and plenty of energy was absorbed during impact testing. Additionally, a certain amount of holes were observed on the fracture surfaces of transparent ABS resins, which stemmed from the deformation of the rubber particles during the impact test. The size of the holes was much larger than that of the rubber particles, indicating the cavitation of the few rubber particles, as shown in Figures 11(a) and (b). The impact fracture surfaces of ABS-Acid and ABS-Polymer had no visible rubber particle cavitation, as shown in Figures 11(c) and (d). The submicron rubber particles prepared by different construction methods had different toughening models. The rubber particles prepared by the one-step method and pressure agglomeration method were more likely to induce cavitation due to their relatively relaxed rubber segments. The cavitation phenomenon of ABS-Acid resin was not obvious. It is difficult to induce cavitation in submicron rubber particles prepared by polymer agglomeration. MS resin and PBA inside the rubber particles restricted the free movement of the rubber segments, which may be the reason it was not easy to induce cavitation.! SEM micrographs of the impact-fractured surfaces for samples: ABS-Onestep (a), ABS-Pressure (b), ABS-Acid (c), and ABS-Polymer (d).
Conclusion
According to the obtained results, there were obvious differences in the size distribution and appearance of the submicron rubber particles prepared using four different methods: one-step batch emulsion polymerization, pressure agglomeration, acid agglomeration, and polymer latex agglomeration. Polybutadiene latex particles with monodisperse size and full shape were more suitable as the toughening core of transparent ABS resin. Significant differences in the size of rubber particles or deviations in the refractive index of the two phases led to diminished optical properties. The tight particle structure and high degree of entanglement facilitate the toughening of rubber particles. Therefore, the submicron latex particles prepared by one-step batch polymerization method were the most suitable as the toughening core of transparent ABS resin. The transmittance and haze value of ABS resin was 90% and 2.4, respectively. The impact and tensile strengths were 185.6 J/m and 39 MPa, respectively. Considering the economic cost and preparation time, the submicron rubber particles prepared by acid agglomeration method were also suitable as the toughening core of the transparent ABS resin, where the transmittance and haze of ABS resin were 90% and 2.9, respectively. The impact and tensile strengths were 170.4 J/m and 31 MPa, respectively.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (U21A2088).