Abstract
Ultraviolet (UV) radiation present in natural sunlight degrades the chemical and mechanical properties of polymeric matrices in composites. Polyurea possess a unique set of chemical and mechanical properties due to its complex microstructure comprising of hard and soft segments (phases). The primary objective of the work presented here is to characterize the chemical and mechanical degradation of pure polyurea and polyurea - polyhedral oligomeric silsesquioxane (POSS) nanocomposites subjected to UV radiation exposure for 45 days. Control specimen in as-received condition (reference specimen) and UV-aged tensile test specimen are tested in the study. Analysis of Fourier transformed infrared (FTIR) spectra for UV-aged polyurea and polyurea-POSS composites reveals that the addition of POSS nanoparticles accelerate the deterioration of polyurea matrix by breaking the hard phase network and altering the mechanism of degradation for the soft phase present in polyurea microstructure. The deterioration of hard and soft segments cause a significant reduction in tensile properties of polyurea-POSS composites.
Keywords
Introduction
Polyurea is an extremely fast curing elastomer which makes it an excellent candidate for protective coating against malicious environmental effects for concrete and steel structures. Additional applications of polyurea include, but not limited to, its use as joint filler and caulking agent, abrasion resistant coating in automobile beds and open rail wagons, chemical protection linings for storage and delivery tanks and blast and fire resistance coating for strategically critical buildings and military personnel/delivery vehicles.1–6 However, one of its most important applications is the use of polyurea based composites as a blast and ballistics mitigation coatings for buildings and vehicles. Guo et al. studied the protective properties of polyurea coated concrete subjected to impact loading and reported that polyurea coating applied on the front face (loading face) was most effective in controlling the damage to the structure. 7 Additionally, polyurea coating have also been reported to effective in reducing the fragmentation due to blast and secondary damage by arresting the debris.8,9 In case of steel plates with polyurea coating, the polyurea compositions with higher ductility have been reported as more effective in controlling the fragmentation of specimen as compared to brittle polyurea.10,11 Overall, the polyurea coatings can improve the ballistic and blast resistance performance of engineering systems composed of concrete or steel. 12 For blast protection coatings, fire retardancy coupled with sufficient mechanical strength of the protective material is important. Polyurea composites with reinforced POSS and other nanoparticles have been reported to provide the desired combination of thermal properties/fire-resistance and mechanical strength.13–19 On the application side, these coatings get subjected to a variety of environmental conditions, including natural sunlight, during their service period. Therefore, it is pertinent to evaluate the extent of degradation of these polyurea protective layers under the deleterious effects of ambient environment.
The average bond dissociation energy for most of the common organic bonds e.g. C-C in alkanes and alkene, C-O in alcohols or C-N bond in amine or nitro groups, lies in the range of 60–105 kcal/mole. 20 The photon with 300 nm (nm) (approx.) wavelength has sufficient energy to break these bonds to start polymer degradation through photo-oxidation, photolysis or thermo-oxidative reactions. The photo-oxidative degradation of polymers can be started through various initiators e.g. direct photolysis of C−C and C−H bonds, the presence of residue from the catalyst used in polymerization, Norrish-I and Norrish-II type reactions due to the presence of a carbonyl group, the attack of oxygen on C−C/C=C bond, the presence of hydroperoxide and/or the photo-Fries reaction which leads to the formation of free radicals. 21 In general, UV radiation in ambient sunlight has wavelength in the range of 290 nm–400 nm. Therefore, the polymer degradation due to ambient sunlight exposure is primarily driven by the UV radiation. The degradation may start in the form of discoloration of polymeric objects, followed by surface cracking and, finally, the complete disintegration of the polymer. 22 However, different physical properties of polyurea are affected to different extent due to UV exposure. Whitten et al. reported that shear and elastic moduli of polyurea continue to decrease as duration of UV exposure increased, but the acoustic properties attenuation were found to exhibit minimal change. 23 They further underlined the importance of detailed chemical analysis of the top surface of polyurea specimen in order to understand the mechanism associated with the development of crazing/hairline cracks resulting due to UV exposure. Goodwin et al. investigated the extent of UV-induced degradation in dry and wet (rainy) condition of polyurethane–graphene oxide and polyurethane - graphene nanocomposites.24,25 Their results show that presence of graphene oxide in polyurea matrix is more effective under dry conditions than wet conditions in terms of controlling the matrix degradation. Che et al. also performed experiments to investigate the combined effects of moisture and UV exposure in a marine environment on the physical properties of polyurea. 26 They reported a drop in functional groups such as C–O–C, C=O and C–N bond and the extent of hydrogen bonding due to marine aging (moisture + UV). According to their results, the hard segment remained less damaged as compared to the soft segment of polyurea network. The higher degree of erosion of soft segment of specimen promoted the degree of microphase separation in polyurea. Mwaura et al. studied the UV degradation reaction in pyridyl-based aromatic polyurea using nuclear magnetic resonance (NMR) and UV/visual and fluorescence spectroscopy. 27 They reported that the exposure to UV radiation breaks the urea linkage and produces two primary amines as reaction products. It was also reported that the water present in the system participates in the reaction and promotes the disintegration of urea linkage presence of oxygen has virtually no effect on the photocleavage process. However, The goal of the research work presented herein is to characterize the UV aging behavior of the micro-segregated polyurea in the presence of POSS particles as additive/filler and its effect of the tensile performance of polyurea-POSS composites.
Materials and testing equipment
Materials
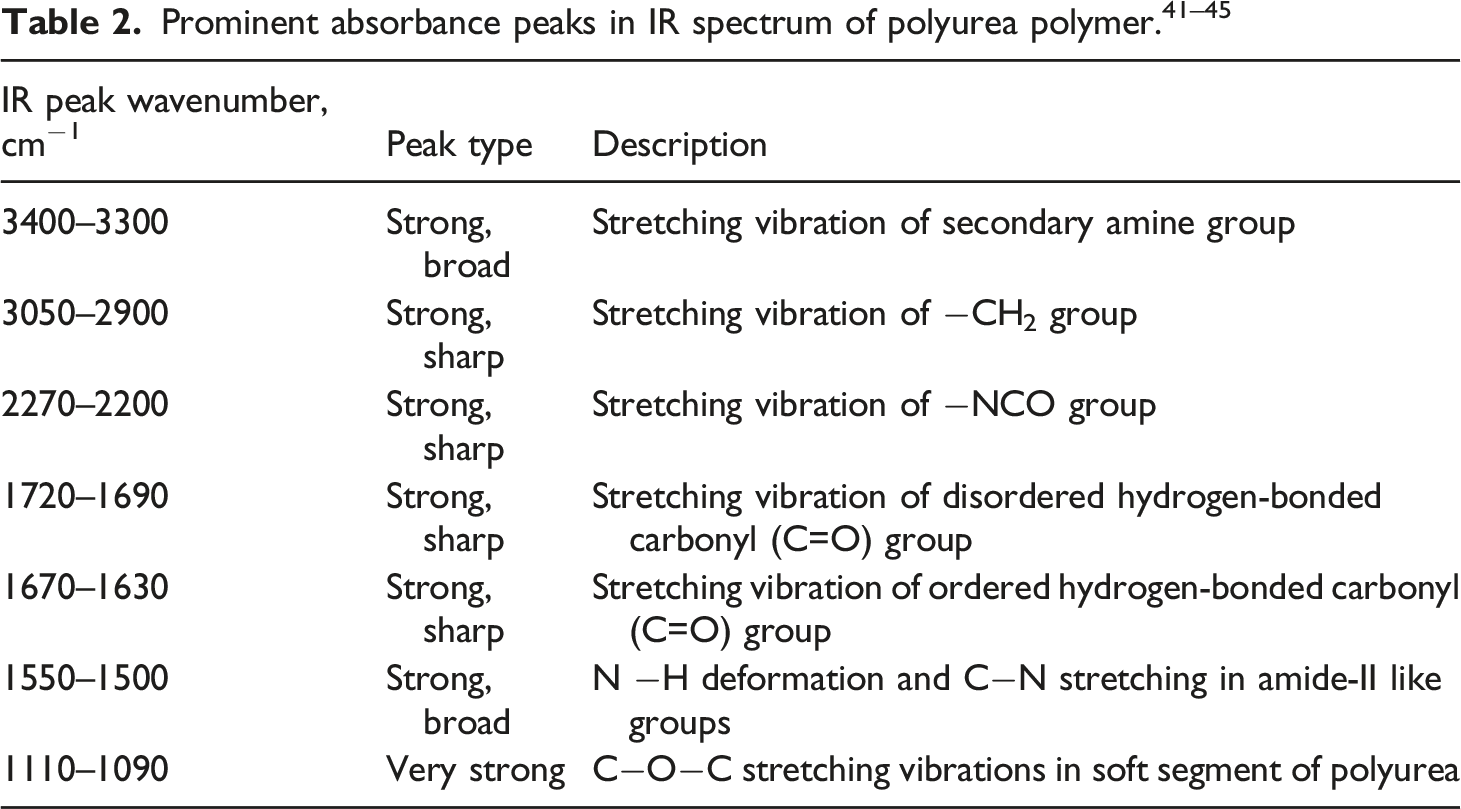
Polyurea monomer [chemical formula: (NH2)2CO] is a product of reaction between organic diamine (moiety with –NH2 group) and organic isocyanate (moiety with NCO group). The polyurea copolymer (PU-1) used in this work is the product of chemical reaction between aromatic 4,4′-Methylenediphenyldiisocyanate “Isonate 143L” (NCO-R-NCO) (isocyanate equivalent weight: 144.5) from Dow Inc. And oligomeric poly(tetramethylene oxide) (PTMO) diamines “Versalink P-1000” (H2N-R′-NH2) from Air Products (see Figure 1).28,29 In case of Versalink P-1000, the number 1000 represents the average molecular weight of PMTO repeating units (approx. 13–14 units). POSS, PM1285, used as reinforcement for composites, is supplied by Hybrid Plastics Inc., USA. Chemically, PM1285 comprises of Si atoms functionalized with a vinyl group [chemical formula: (R*SiO
Composition of polyurea-POSS nanocomposites used in present study.

Preparation of composite materials
The composite sheets used in this work were manufactured in the form of 3.18 mm (approx.) thick sheets via a closed die casting technique at Nano Infrastructure Research Group (NIRG), University of Mississippi, USA. In order to fabricate the composite sheet, 0.160 kg Versalink P-1000 diamine was mixed with required amount of POSS PM1285 in a 0.24 L mixing cup in multiple steps. In each step, the mixture was heated to 80°C and vigorously mixed using mechanical stirrer for 30 min. After adding POSS particles in VP1000 diamine, 0.04 kg Isonate 143L (di-isocyanate) was added in the mixture and stirred for 1 min. The final mixture was transferred to Teflon mold and covered with top plate using clamps. The clamped mold was placed in the oven at 80°C for 4 h to complete the curing of composite sheet. At the end of curing period, the composite sheet was carefully removed from mold for testing.
UV aging of polyurea (PU-1) and PUPM3PO composite
The dog-bone shaped PU-1 and PUPM3PO tensile testing specimen were exposed to 45 UV-aging cycles in accordance with procedure specified in ASTM standard G154-06. 31 Each UV aging cycle was 24 h long comprising of 16 h of exposure to 302 nm UV radiation followed by immersion in water (50°C) in a dark box for 8 h. The FTIR spectra and tensile testing for PU-1 and PUPM3PO specimens were measured at the intervals of 7 days, 15 days, 30 days and 45 days during the UV-aging.
Test equipment
UV aging experiment details
The ultraviolet aging chamber was fabricated in the NIRG laboratory (see Figure 2). For UV aging, a 0.48 m × 0.24 m × 0.18 m wooden box was fitted with 6.35 mm (approx.) thick UV filter sheet and UV lamp. The UV lamp produced, depending on the settings deployed, 365, 302 or 254 nm wavelength UV light. Among these, 302 nm wavelength UV light representing UV-B radiation was used in the research work presented herein. The distance between UV lamp and sample was maintained at approximately 38 mm such that the intensity of incident UV radiation was 1000 μW/cm2 on upper surface of the specimens. UV test box set up and UV lamp used in the experiments.
Tensile testing
Tensile testing of as-received (un-aged) and UV-aged specimen was carried out in accordance with the procedure specified in ASTM D412-16.
32
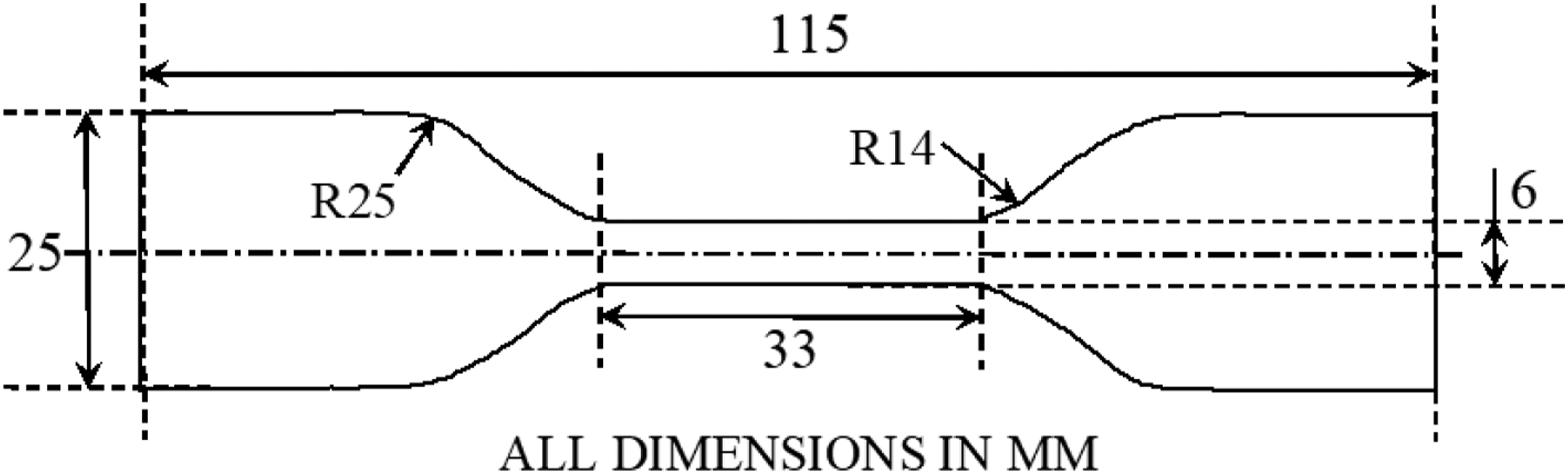
Dog-bone shaped samples as per specimen dimensions prescribed in the standard were cut from 3.18 mm thick nanocomposite sheets (as mentioned earlier) using a cutting die (see Figure 3). A total of three specimens from each scenario (composition combined with aging duration) were tested at the grip separation rate of 500 mm/minute using Instron 5982 dual column floor model test machine equipped with video extensometer. Dog bone tensile testing specimen used in the present work ASTM D 412-C–16.
32
Fourier transformed infrared (FTIR) spectroscopy
FTIR spectroscopic studies of the un-aged and aged specimens were carried out using an Agilent Cary 630 ATR-FTIR spectroscope with diamond window in ATR-FTIR mode. The spectroscope was set to work in the spectral range of 750–4000 cm−1 with a resolution of 2 cm−1, controlled using Microlab software from Agilent. The surfaces of all the specimens were cleaned with ethyl alcohol before ATR-FTIR spectroscopy. The infrared absorbance peak data obtained from specimens was analyzed using Spekwin32 software, available free for research and education purposes (https://www.effemm2.de/spekwin/index_en.html).
Atomic force microscope
Nanoscope IIIa multimode scanning probe microscope was used for the imaging of hard and soft phases present in polyurea microstructure. The AFM was operated at ambient conditions. Silicon AFM cantilever probes (Tap150-Al-G from Budget Sensors) with spring constant of 5 N/m and approximate resonance frequency of 150 kHz with reflective aluminum coating on the back were used in tapping mode to generate phase images of polyurea.
Results
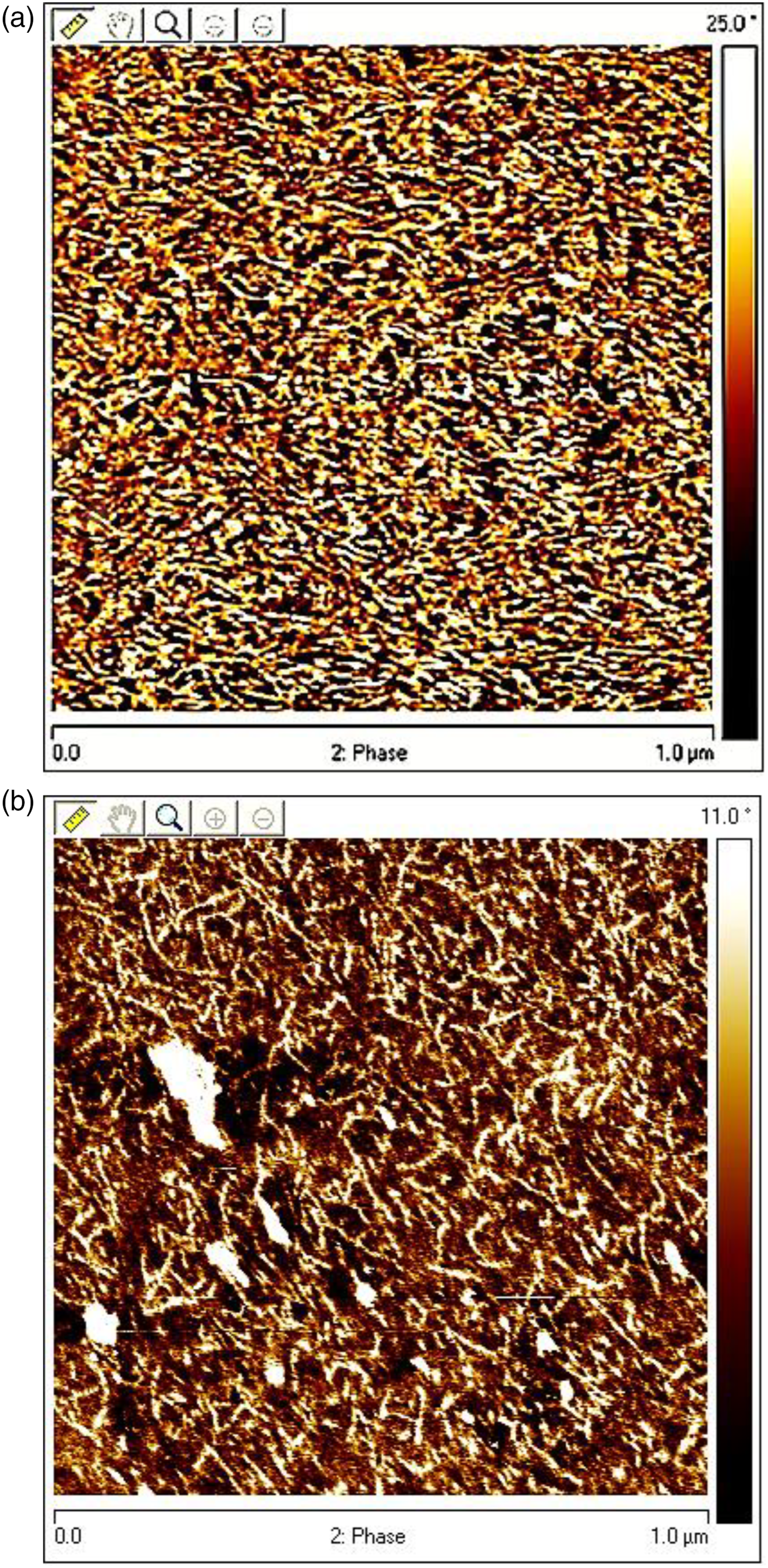
Atomic force microscopy of polyurea (PU-1)
In the beginning of the chemical reaction, diamine and diisocyanate molecules react together and produce a polyurea macromolecule as shown below. Phase-based atomic force microscopy image of (a) unaged PU-1 polyurea (b) unaged PUPM3PO composite.
Tensile testing of control specimen
Figure 5 shows the effect of amount of POSS added on the tensile strength of polyurea-POSS nanocomposites. The tensile strength of pure polyurea (PU-1) specimen is equal to 27.6 MPa. The tensile strengths of PUPM1PO and PUPM3PO composites are comparable to that of PU-1, specifically 28.9 MPa and 26.7 MPa, respectively. However, as POSS content in increased beyond 3 phr, the average tensile strengths of the composites reduces significantly by 22.8% (21.3 MPa) in case of PUPM5PO and by 41.4% (16.2 MPa) for PUPM10PO. Therefore, based on the un-aged tensile properties comparable to PU-1 and high POSS content (required for improving the fire retardancy), PUPM3PO is used as candidate material for UV-aging investigation along with PU-1 (FTIR spectroscopy and Tensile testing) as presented in the following sections. Tensile strength results for various unaged polyurea-POSS nanocomposites.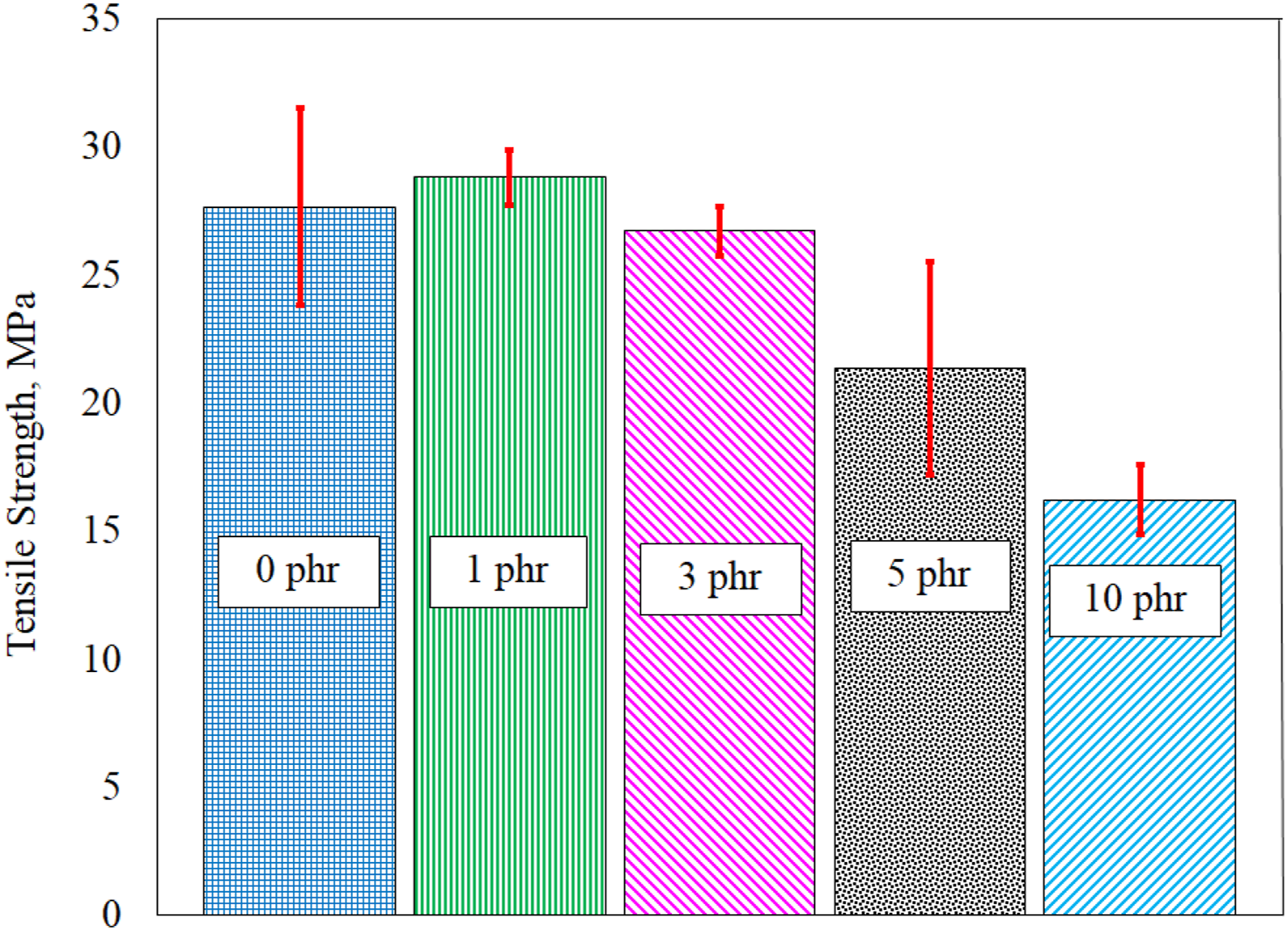
Fourier transformed infrared (FTIR) spectroscopy of un-aged polyurea and polyurea-POSS composites
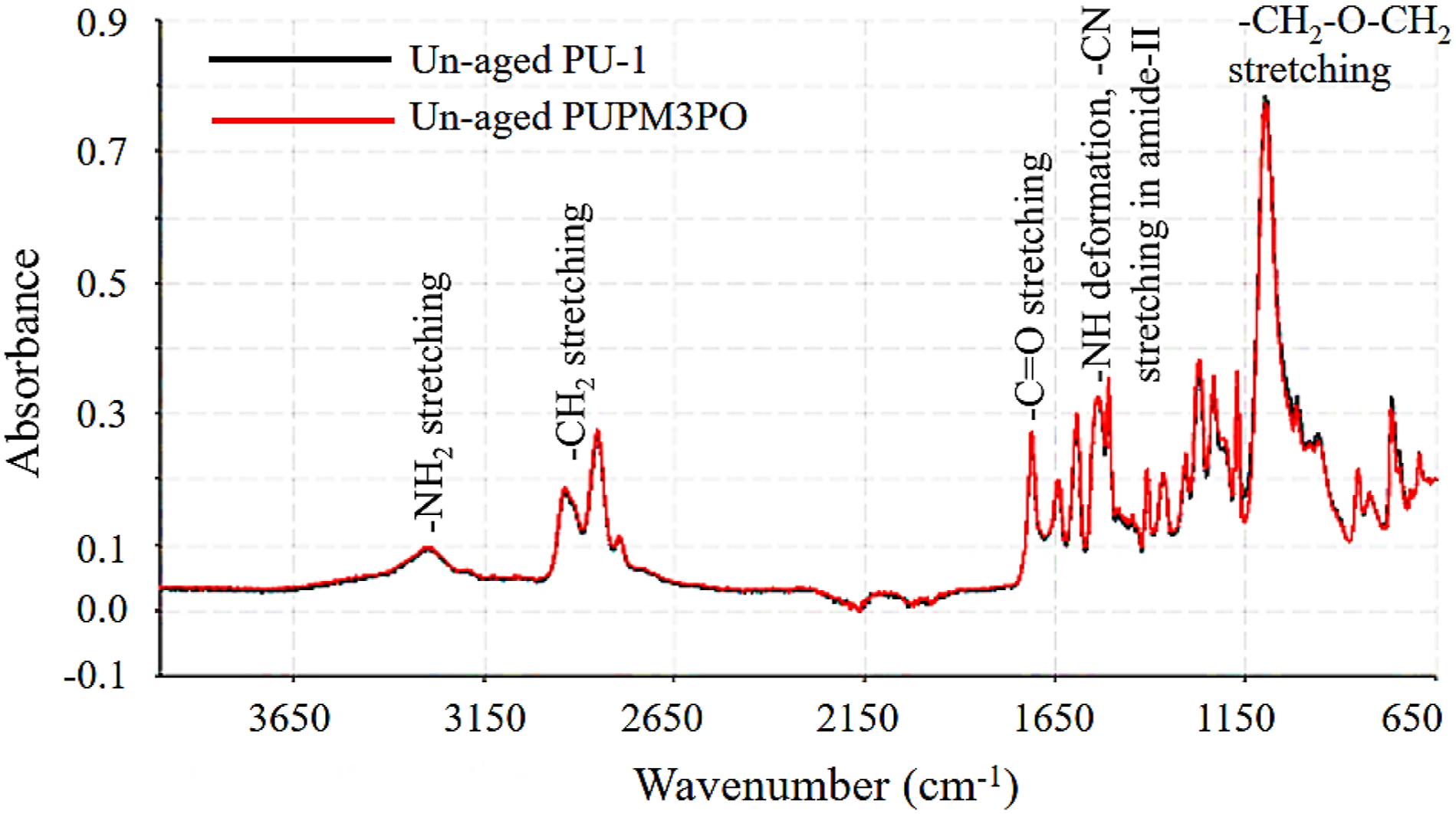
Comparison of FTIR spectra of un-aged polyurea (PU-1) and polyurea-POSS nanocomposite (PUPM3PO).
In the case of PU-1 polyurea, the carbonyl group (C=O), present in Isonate 143L (diisocyanate), is the first important chemical group to identify. The carbonyl groups are part of the hard segment in polyurea. These segments create an extensive network of inter-chain hydrogen bonding and participate in most of the aging reactions. The presence of hydrogen bonding imparts unique mechanical properties to polyurea. The peak in the region of wavenumbers 1720–1690 cm−1 designates disordered hydrogen bonded carbonyl group, and the absorption peak between wavenumbers 1670–1630 cm−1 represents the presence of ordered hydrogen bonded carbonyl groups. 26 In case of PU-1 and its composites, the comparatively strong peak at wavenumber 1710 cm−1 indicates the presence of primarily amorphous microstructure. The next important peak in polyurea IR spectra appears around wavenumber 3300 cm−1. It is a medium-low intensity broad peak with a shoulder band and is characteristic of the stretching vibration of the secondary amine (–NH) group. The broadness of the peak is attributed to intermolecular hydrogen bonding among amine groups. On the right side of amine peak (3300 cm−1), a pair of sharp and medium-strong peaks appear around wavenumber 2940 cm−1 representing the stretching vibration of the C−H bonds for aliphatic methylene (−CH2) group located in the soft segment of the polyurea. 40 The last critical IR absorption peak of interest appears as a strong absorption peak around wavenumber 1090 cm−1 representing stretching vibrations of the C−O−C group (ether group) which works as an inter-molecular link for chain extender molecules in the soft segment of polyurea.
The absence of an IR absorption peak belonging to a particular chemical group is as important as the presence of any IR peak. In both the cases (PU-1 and PUPM3PO), the absence of any strong absorbance peak between wavenumbers 2270–2200 cm−1 indicates the absence of any unreacted diisocyanate group in the specimen indicating the complete consumption of reactants.
Effect of addition of POSS nanoparticles on UV-induced degradation of hard segment of polyurea chain
Figure 7 compares the FTIR spectra for un-aged PU-1, UV-aged PU-1 and UV-aged PUPM3PO composites (aged for 45 days) showing the effects of POSS addition on the UV-induced chemical degradation mechanism. Summary comparison of FTIR spectra of 45 days UV-aged polyurea (PU-1) and polyurea-POSS nanocomposite (PUPM3PO).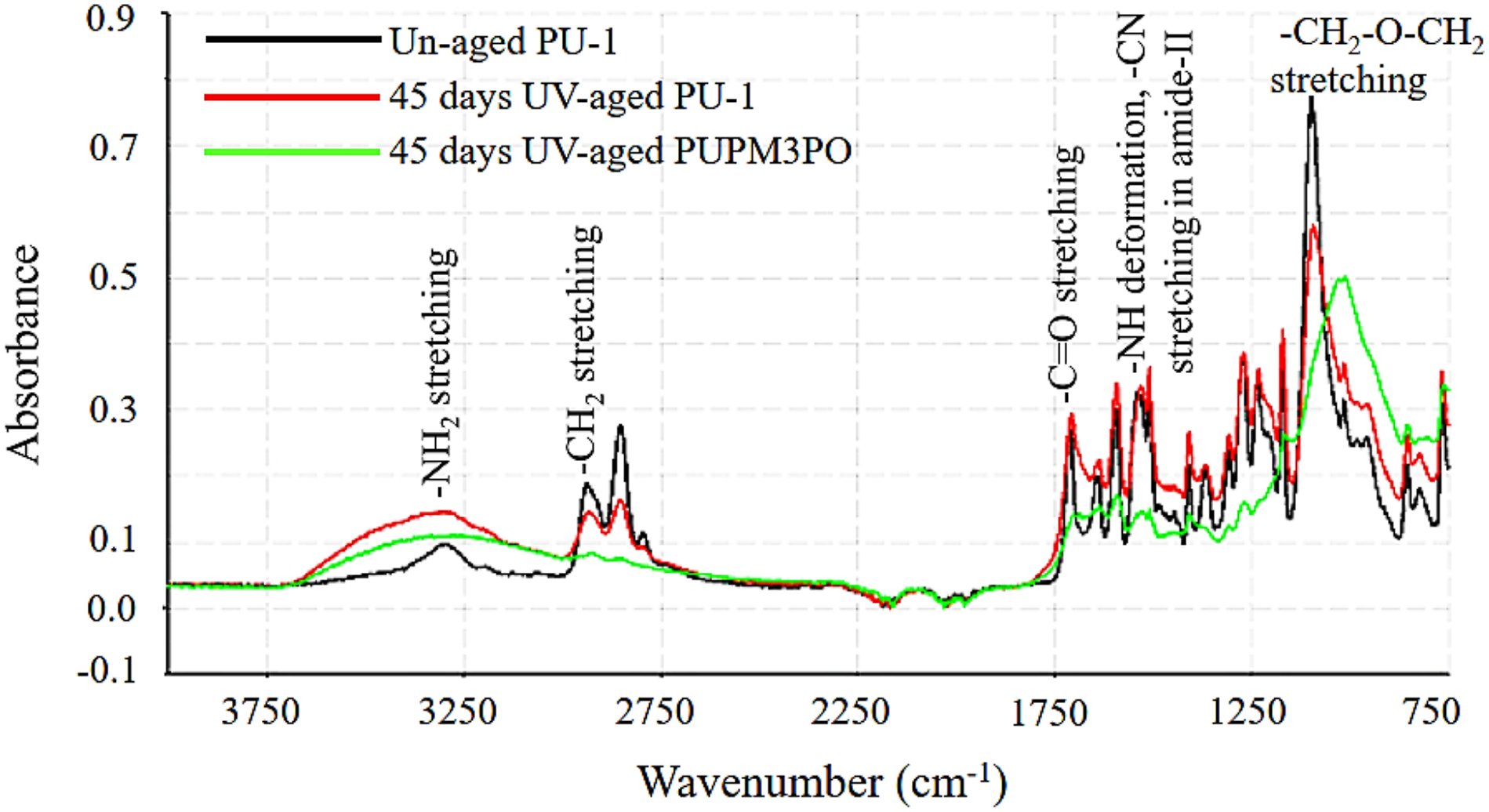
Generally, the degradation of the hard segments of the polyurea due to UV exposure can be studied by monitoring the change in the intensity of absorption peaks representing the deformation of the carbonyl group (C=O, 1720–1630 cm−1) and the amine (N−H, 1540 cm−1), as reported in Figure 8.24,25 A significant difference in the UV-aging of the two materials (PU-1 and PUPM3PO) can be observed as the IR absorption peaks around wavenumbers 1720–1690 cm−1 and 1670–1630 cm−1 are compared. Initially, the strength of these peaks increases with the aging time for both the cases. As the aging progresses, the relative strength of these IR peaks remains unchanged for PU-1 specimen. However, the same pair of IR peaks reduce in strength for PUPM3PO specimen after prolonged UV aging. The reason behind such behavior may be described as following. It the beginning of UV-aging, the amount of organic peroxide radicals (produced during propagation step) increases which contributes to the strengthening of these two peaks. The oxidation of loose ends of broken polyurea chains also promotes the strengthening of peaks at 1720–1690 cm−1 and 1670–1630 cm−1. As sufficient aging time passes (45 days for PUPM3PO specimen), a portion of these dangling peroxide ends may get further degraded by yielding CO2 gas. It may explain the drop in strength for both the peaks in PUPM3PO specimen at the end of 45 days of UV-aging. The observation indicates that the presence of POSS nanoparticles hastens the UV-induced degradation of polyurea. Further, the amount of relative larger drop in strength of IR peak at 1720–1690 cm−1 reveals that the carbonyl groups located in the amorphous region of polyurea microstructure could be the preferred oxidation sites during UV-aging for polyurea-POSS composites. Time evolution of IR absorption peak around 1710 cm−1 and 1540 cm−1 for (a) UV aged PU-1, and (b) UV-aged PUPM3PO specimen.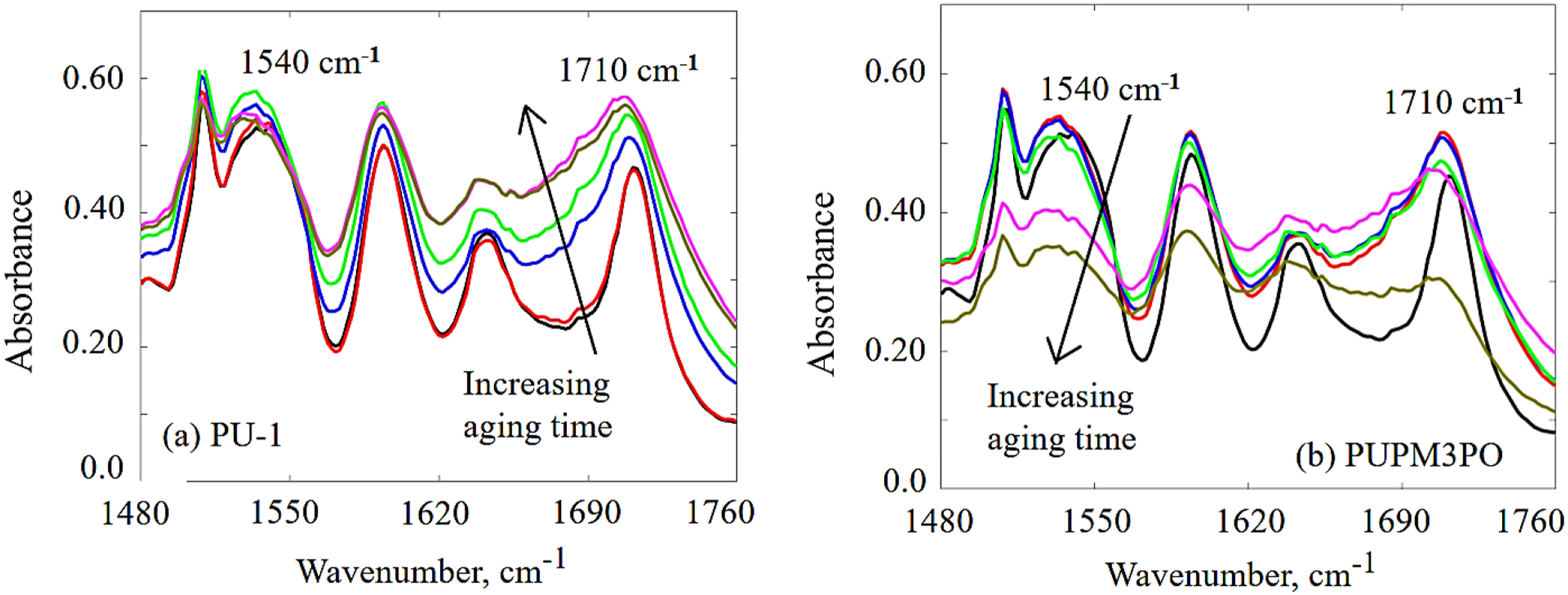
The next significant change in the FTIR spectra of aged specimens with respect to unaged PU-1 appears around wavenumber 3300 cm−1(Figure 7). 46 In both the aged specimens, the height and spread of this peak increases to different extent as a result of aging. During the photolysis of polyurea, peroxide and acid free radicals are generated as reaction intermediates and form intermolecular hydrogen bonds. The hydrogen bonding between the peroxide and acid free radicals spreads the band at wavenumber 3300–3700 cm−1. However, at the same time, the intensity of the band representing the carbonyl group (1710 cm−1) decreases while its spread increases with aging time for both the aged samples. This implies that the oxygen atom at the end of the chain extender gets converted into –OH group and increases the amount of hydrogen bonding in the system. This also helps in explaining the increase in the spread of band at 3300 cm−1 which extends up to 3700 cm−1 reaching the range of the IR band representing the –OH group. Therefore, changes in the IR absorption peak at 3300 cm−1 cannot be entirely attributed to hydrogen bonding among the amine group. Further, the modulation in intensity of the peak at 1540 cm−1 for PU-1 and PUPM3PO holds another key information (Figure 8). The height of this IR peak remains unchanged for PU-1 as the aging progresses while the same first increases and then decreases with aging time in PUPM3PO specimen. It is likely that due to intensified UV-aging in PUPM3PO the a fraction of urea linkages gets converted into primary amines after dissociation which further get oxidized and yields NH3 gas. It also helps in explaining the reduced height of peak at 3300 cm−1 in PUPM3PO as compared to that in PU-1 specimen. Further, collective comparison of absorption bands at 1540 cm−1, 1710 cm−1 and 3300 cm−1 in the FTIR spectra for aged PUPM3PO and PU-1 specimens suggests that the UV radiation attack on the hard segment of polyurea chains gets intensified in the presence of POSS nanoparticles.
Effect of addition of POSS nanoparticles on UV-induced degradation of soft segment of polyurea chain
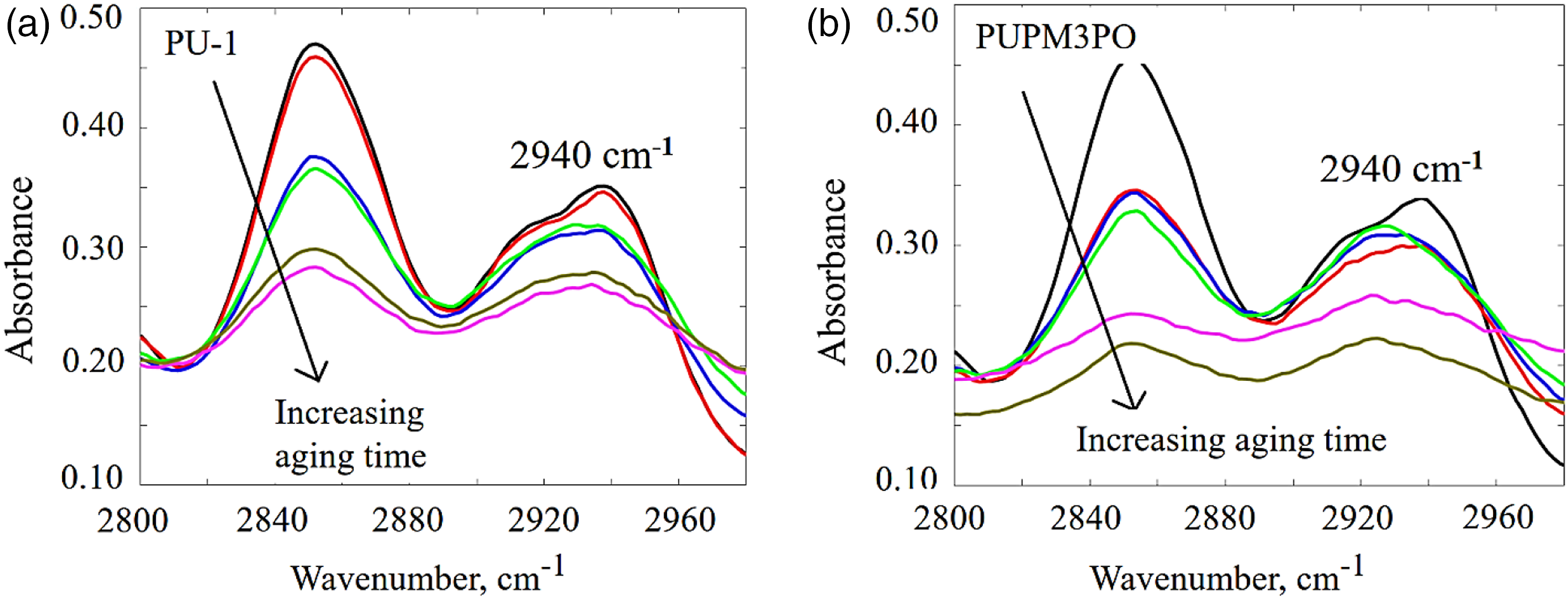
Figure 7 also shows a drop in the absorption strength for peaks located around wavenumber 1090 cm−1 and 2940 cm−1 for UV-aged PU-1 and aged PUPM3PO. These peaks represent the presence of chain extender ether (-C-O-C-) and methylene (-CH2) chemical groups, respectively, belonging to the soft segments in the polyurea chains network (as explained in the previous section). The drop in their absorbance confirms the disintegration of polyurea chain in the soft phase regions as well. In the case of PUPM3PO, the peak at 1090 cm−1 not only decreases in intensity but also it shifts towards a lower wavenumber while increasing in spread whereas the same peak for PU-1 decreases in intensity only as aging progresses. The movement of any IR absorption peak towards a lower wavenumber with a simultaneous increase in its spread indicates the increasing amount of hydrogen bonding of larger lengths among the chemical group represented by the corresponding IR peak. With that in mind, these changes in the absorption peaks at 2940 cm−1 and 1090 cm−1 suggest that exposure to UV radiation damages aliphatic chain extender in soft segment and converts it into a free radical which reacts with the Oxygen present in the environment and yields a chain peroxide radical. These peroxide radicals are highly reactive, and as the aging time progresses they get converted into –COOH group. Presence of –COOH group in the soft segment of polyurea chain enables hydrogen bonding with more neighboring chains simultaneously. This evidence helps in explaining the reduction in strength for IR peaks at 2940 cm−1 and 1090 cm−1 and increase in the spread of peak at 1090 cm−1 in PUPM3PO specimen. Finally, a direct comparison of Figures 9 and 10 show that the drop in intensity for both of the absorption IR peaks is significantly more in PUPM3PO than PU-1 implying that PUPM3PO experiences higher degree of UV-induced degradation than PU-1 due to the presence of POSS nanoparticles. Time evolution of IR absorption peak around 1090 cm−1 for (a) UV aged PU-1, and (b) UV-aged PUPM3PO specimen. Time evolution of IR absorption peak around 2940 cm−1 for (a) UV aged PU-1, and (b) UV-aged PUPM3PO specimen.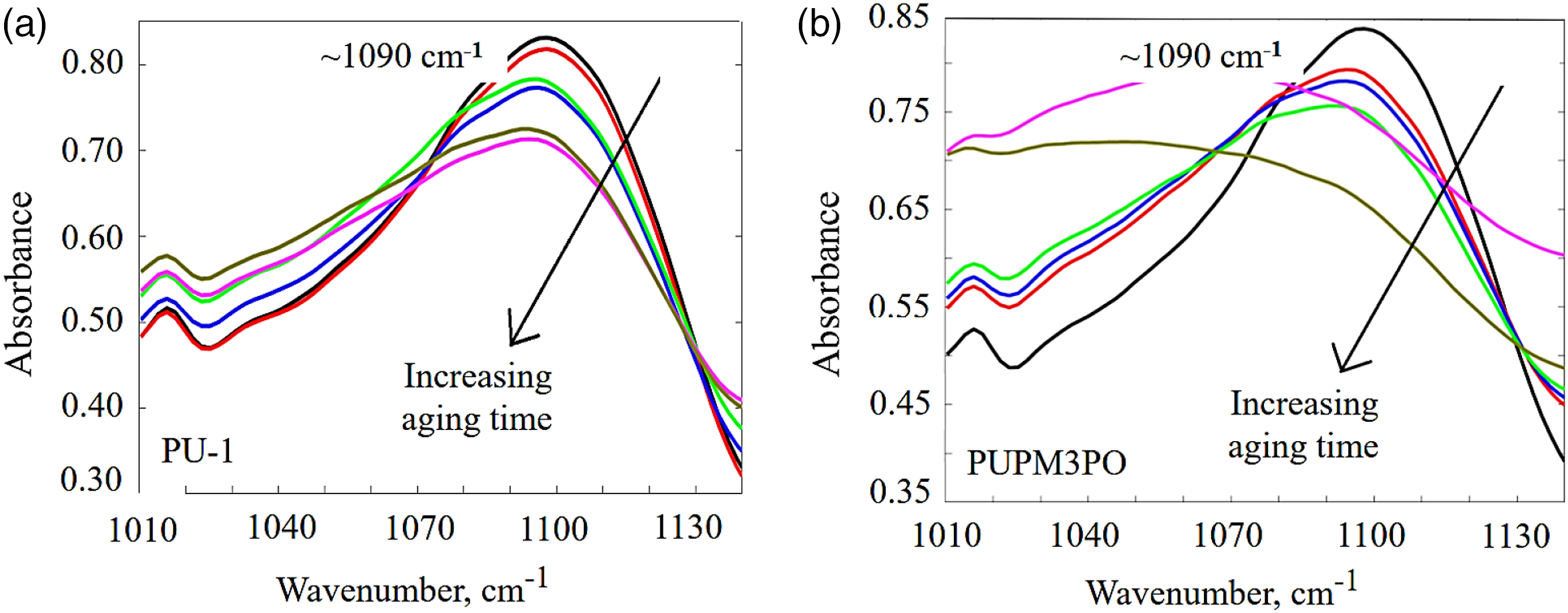
Summary of degradation mechanism
Based on earlier observations, the following schematics involved in the UV-induced degradation of POSS-polyurea composites can be described in the following manner.
Degradation of hard segment
In case of POSS/polyurea composites, the degradation of hard segment may begin due to the conversion of the carbonyl group (C=O) in the peroxide free radical due to the absorption of the photon energy. As the aging proceeds, the peroxide radicals get reduced by yielding CO2 gas as supported by the drop in peaks intensity at wavenumbers 1720–1690 cm−1 and 1670–1630 cm−1 after 45 days. The relative intensities of the same peaks for PU-1 and PUPM3PO specimen indicate the contribution of POSS nanoparticles in accelerating the degradation for PUPM3PO specimen. However, the similarity between the shapes and the respective locations of these peaks implies that the presence of POSS particles may not alter the mechanism of photo-degradation of polyurea hard segment. Further, it is likely that the intensified UV-aging in PUPM3PO damages the urea linkages and converts them in primary amines which later may get oxidized and yield NH3 gas.
Degradation of soft segment
The UV-induced degradation of soft segment can be understood by change in intensities of FTIR peaks at wavenumber 1090 cm−1, representing ether group, and at 2940 cm−1, representing methylene group. The incident photon breaks the aliphatic chain extender molecule, which in presence of Oxygen gets converted to –OH group leading to extended hydrogen bonding. This explains the drop in intensities of above peaks and broadening of peak at 1090 cm−1 (due to presence of –COOH group in neighboring chains) belonging to soft segment in PUPM3PO specimen. In the fingerprinting region, the IR peak at wavenumber ∼860 cm−1 represents O-O stretching vibration in organic peroxides. The strength gain of this peak as aging progresses suggests the increasing presence organic peroxides which are intermediaries products during the initiation and propagation stages of photo-oxidation reaction.
Tensile testing of UV aged polyurea and polyurea-POSS composites
Dog-bone shaped specimens cut from PU-1 and PUPM3PO sheets are provided cyclic UV exposure (as described in Section 2.1.2) for 45 days to study of the degradation in the tensile strength due to UV-exposure. Both PU-1 and PUPM3PO specimens are tested for tensile strength after the 0 days (un-aged), 15 days, 30 days and 45 days of UV exposure. Results of tensile testing of PU-1 and PUPM3PO specimens are compared in Figure 11. Effect of prolonged (45 days) UV exposure on tensile strength of polyurea and polyurea-POSS composite specimens.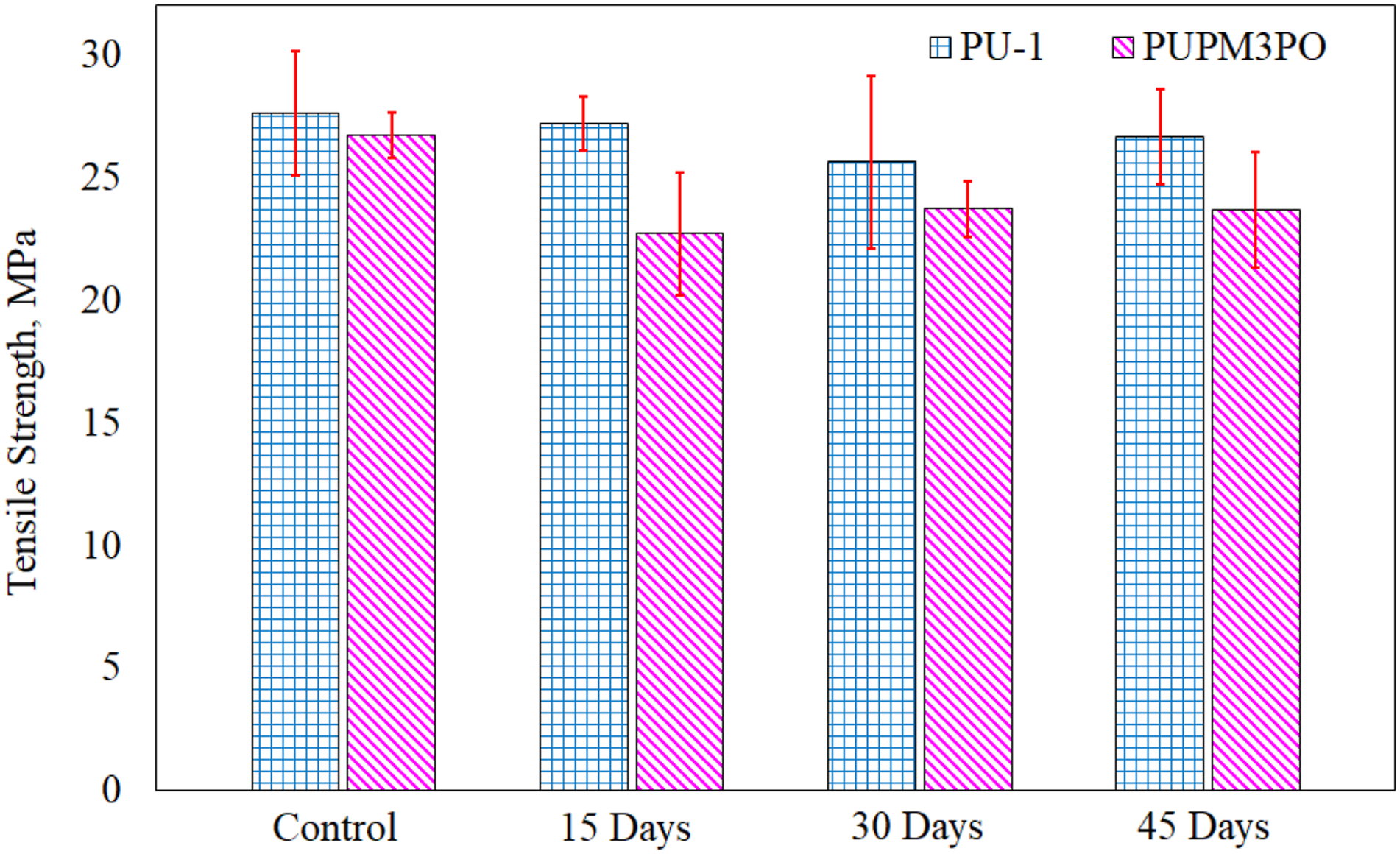
From these results, it is observed that 45 days of UV exposure has only a minimal effect (3.8% drop, from 27.7 MPa to 26.6 MPa) on the tensile strength of the PU-1 specimen. However, the tensile strength of PUPM3PO gets reduced by almost 12% (from 26.7 MPa to 23.5 MPa) after 45 days of UV exposure. The severe degradation of POSS nanocomposites can be attributed to the presence of POSS particles in the polyurea matrix which may be treated as trace impurity particles functioning as initiation sites for various photo-degradation chemical reactions.
Conclusions
Polyurea and polyurea-POSS nanocomposites are subjected to UV aging for 45 days. Both un-aged and aged polyurea-POSS nanocomposites are subjected to FTIR spectroscopy in order to study chemical changes in the polyurea matrix that result from UV exposure. Careful observation of the FTIR spectra for UV-aged polyurea (PU-1) and polyurea composite with 3 phr POSS (PUPM3PO) reveals that for the PUPM3PO undergoes more severe degradation than the PU-1 specimen due to enhanced deterioration of hard and soft phases of polyurea microstructure. Detailed analysis of the FTIR spectra of UV-aged PU-1 and PUPM3PO shows that the degradation of the PUPM3PO nanocomposite specimen occurs at a comparatively faster rate than that of the PU-1 specimen (intensity reduction for IR peaks at 3300 cm−1 and 1090 cm−1). From these evidences, it can be reported that the presence of POSS nanoparticles promote the degradation of polyurea matrix while also altering the degradation mechanism of the polyurea soft segment. The extent of degradation of hard segment of polyurea chain in PUPM3PO specimen is also higher than that in PU-1 specimen. However, the degradation mechanism remains unchanged for both the materials. The severe UV-induced degradation of polyurea matrix network in aged PUPM3PO results in a higher loss of the tensile strength.
Footnotes
Author contributions
GP: Conceptualization, Experiments, Manuscript preparation; AO: Conceptualization, Funding, Manuscript review; XL: Experiments, Manuscript preparation; HA: Experiments; AC: Funding, Manuscript review.
Declaration of conflicting interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the financial support received under a subcontract from the Department of Homeland Security-sponsored Southeast Region Research Initiative (SERRI) at the Department of Energy’s Oak Ridge National Laboratory for this research work. We acknowledge the support of Dr Ikhlas Khan and Dr Ahmed Galal in providing FTIR facility and necessary training at National Center for Natural Product Research, The University of Mississippi.