Abstract
In the present study, zinc oxide nanoparticles (ZnO-NPs) were synthesized by a hydrothermal method followed by the fabrication of polylactic acid/polycaprolactone blend (PLA/PCL, 80/20 wt/wt) at various loadings of ZnO-NPs (2, 4, and 6 wt%) via melt mixing. FTIR and XRD patterns confirmed that the ZnO-NPs were successfully synthesized. The ZnO-NPs with an average diameter of about 46–73 nm were observed by the FESEM analysis. The effect of ZnO-NPs on morphological, thermal, UV absorption, mechanical, photochemical degradation, rheological and cell viability properties of PLA/PCL blend were investigated. FESEM micrographs of bionanocomposites demonstrated that polycaprolactone was dispersed as a droplet to the Polylactic acid matrix phase. DSC analysis showed that the addition of ZnO-NPs increased the degree of crystallinity and melting temperature of the PLA. Mechanical assessment of the bionanocomposites reveals that the addition of 2, 4 and 6 wt.% of ZnO-NPs into the blend sample leads to increase in the tensile modulus by about 5.4, 11 and 24%. The MTT assay results implied that cell viability of the both filled and unfilled samples is greater than 90% indicating their biocompatibility to the fibroblast cells. It is observed that the melt linear viscoelastic properties of the prepared bionanocomposites are under control of LA/PCL chain degradation and hydrodynamic nanoparticles interaction.
Introduction
Nowadays, considering ineffective systems in the disposal of waste petroleum polymers, the remaining waste of petroleum polymers as sources of pollution has made a serious concern in the world. In this regard, biodegradable and biocompatible polymers have been extensive attention by both researchers and industrials owing to the remarkable properties such as biodegradability, low toxicity, and low weight. Among these type of polymers, Polylactic acid (PLA) is a biodegradable thermoplastic and aliphatic polyester which is derived from renewable sources including corn and sugar. PLA represents favorable properties such as high strength, stiffness (at room temperature) and process-ability leading to wide applications in the biomedical and packaging industries.1,2 In contrast to these advantages, some of the crucial limitations in the usage of PLA are its brittleness, limited thermal stability, and low heat deflection temperature.3,4 The various strategies have been proposed to overcome PLA brittleness including plasticization, copolymerization, and melt blending with flexible polymers or fillers.5–9 Among the mentioned strategies, melt blending with flexible polymers has more advantageous environmental properties than that of other methods due to the absence of hazardous solvent and the cost-efficiency. Several kinds of researches have been made to improve the brittle properties of PLA by blending with flexible polymers, while the environmental pollution of petroleum-based polymers diminishes the biodegradability of final blend.10–12 Therefore, the use of biodegradable polymers is the best option to improve brittleness as well as biodegradation.13,14 Among the various a ductile and renewable polymers, polycaprolactone (PCL) is a thermoplastic, aliphatic polyester, semi-crystalline and low rate of degradation which is widely applied in biomedical applications including tissue engineering, drug carrier and food packaging industries.15–17 PLA and PCL is immiscible thermodynamically, but due to the advantages of the PLA/PCL blend, various aspects of it have been investigated, even have used the compatibilizers to reduce the incompatibility between PLA and PCL.18,19 Also the biocompatibility, biodegradability and renewability of PLA/PCL blend has been made it to be popular among blends in the medical application.20,21
The literature showed that the addition of nanoparticles to the PLA/PCL blend can completely change its properties. Wu et al. 22 Studied the ternary systems containing functionalized multiwalled carbon nanotube (MWCNTs) as compared with those of PCL/PLA blend. They believed that, due to the selective localization of MWCNTs in the PCL matrix phase and at in the interface, lower rheological and conductive percolation thresholds and the mechanical properties are better than the PCL/PLA blend. Also, with addition of MWCNTs, the size of the PLA domains in the continuous phase reduces remarkably. Another kinds of filler such as titanium dioxide (TiO2), 23 pure and impure magnetic nanoparticles (MPs), 24 graphite nano-platelets (GNP), 25 carbon nanotubes (CNTs) and organoclays 26 used as the third component in the PLA/PCL blend. Among these studies, no research on the zinc oxide nanoparticles (ZnO-NPs) as a third component, has been severely seen and felt. ZnO-NPs is a multifunctional material with effective antimicrobial, photocatalytic properties, biocompatibility as well as being nontoxic and inexpensive. Many studies have examined the effect of the presence of ZnO-NPs on other polymers, and of course PLA and PCL.27–33 Murariu et al. 34 have used the modified ZnO-NPs by triethoxycaprylylsilane in the PLA matrix, their results showed that triethoxycaprylylsilane can slow down the degradation process and improve the properties of bionanocomposites compared to the nanocomposite containing unmodified nanoparticles. Hydrolytic degradation of samples containing 1, 2 and 3% modified ZnO-NPs by triethoxycaprylylsilane, was also investigated by Benali and coworkers. 35 A review of the results over a 10-month period, showed that the use of modified ZnO-NPs, would slow down hydrolytic degradation of PLA.
The review of literature showed that bionanocomposites based PLA/PC have been less discussed. In this regard, the aim of this work was to understand effect of incorporated ZnO-NPs on behavior of bionanocomposites, and to investigate the impact of ZnO-NPs on final properties. For this purpose, ZnO-NPs were synthesized by hydrothermal method and then incorporated to the PLA/PCL blend as a third component by melt mixing method. All morphological, thermal, optical rheological, and cell viability properties as well as photochemical degradation behavior of bionanocomposites were studied and discussed in details. Despite the interesting nature of the research discussed above, this is one of the first studies concerning the characteristics of PLA/PCL/ZnO bionanocomposites, which helps us to get a better understanding of the properties of this bionanocomposites.
Experimental
Materials
The PLA grade 2003D was purchased from NatureWorks LLC product (USA) with molecular weight of 200,000 g/mol, and density 1.24 g/cm3. The PCL grade, Capa™ 6800 was purchased from Solvay Co. Ltd. (Belgium) with molecular weight of 80,000 g/mol, and density 1.29 g/cm3. Ammonia and zinc acetate were used for synthesis of ZnO, supplied by Merck (Germany).
Preparation of ZnO-NPs
The synthesis of ZnO-NPs was carried out as follows: first, 10 mL of 1.0
Preparation of PLA/PCL blend and its bionanocomposites
Sample codes and their corresponding compositions.
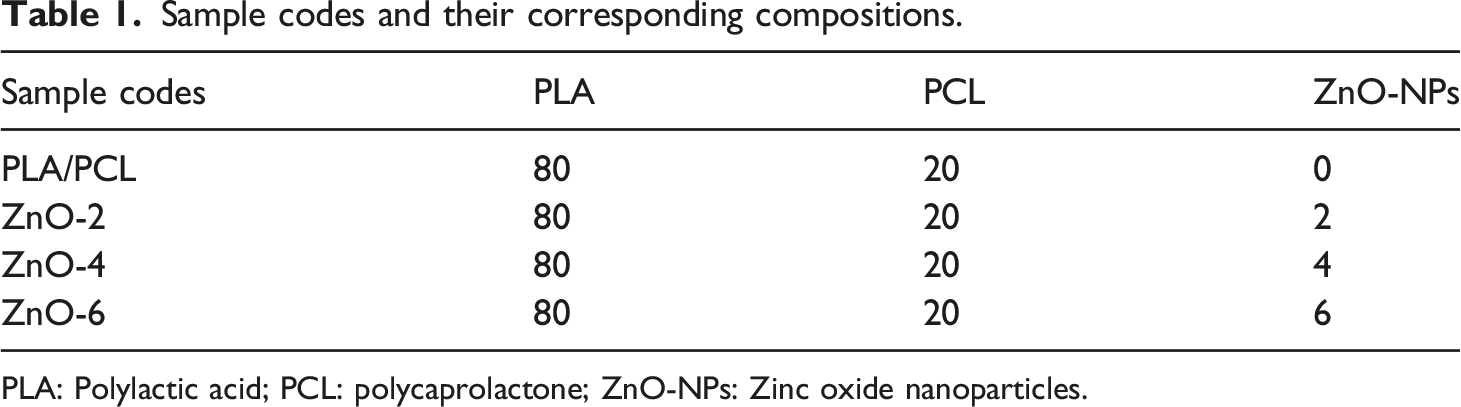
PLA: Polylactic acid; PCL: polycaprolactone; ZnO-NPs: Zinc oxide nanoparticles.
Characterization methods
The surface morphology of the ZnO-NPs and PLA/PCL/ZnO bionanocomposites were investigated by field emission scanning electron microscopy (FESEM, MIRA3 TESCAN-XMU, Czech Republic) under an acceleration voltage of 20 KV. The samples were fractured in liquid nitrogen and coated by a very thin film of gold powder.
A differential scanning calorimeter (DSC, ZF-DSC-D2H, Finland) was used to study the nonisothermal crystallization and melting behavior of the samples. First, about 9 mg of samples was heated from 30 to 200°C at the rates of 10°C/min under nitrogen atmosphere and held for 5 min to eliminate the thermal history. Then, the samples were quenched to room temperature at the rates of 60°C/min and again heated to 180°C at 10°C/min rates. The degree of crystallinity (Xc) of PLA/PCL blends and PLA/PCL/ZnO bionanocomposites were calculated according to equation (1)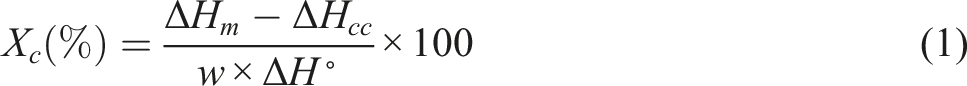
The tensile properties of the samples were determined using GOTECH Testing Machines (Model Al-3000, Taiwan) with 5 kN load cell, 20 mm/min crosshead speed at room temperature and gauge length 100 mm according to the ASTM D638. The dog-bone shaped specimens were prepared by injection molding (PAYA Co., Iran) at 170°C and pressure of 10 MPa. For each bionanocomposite, 5 samples were analyzed, and the reported values of mechanical properties are an average of 5 data from each sample.
X-ray diffraction (XRD, Philips PW 1800, Netherland) was preformed to characterize the phase and structure of the prepared nanoparticles using CuKα radiation (40 KV and 40 mA) at angles ranging from 20 to 70°.
Photochemical degradation of PLA/PCL bionanocomposites was studied by exposing to UV irradiation. Exposures took place by emitting mainly 400 nm radiation at room temperature and in air atmosphere. Times of the irradiation were set to be 0–50 h, followed by the measurement of tensile strength, tensile modulus and elongation at break of exposed samples for every 5 h.
FTIR analysis for the ZnO powder were done by using Bruker model Equinox55 (Germany) in the range of 200 cm−1–800 cm−1.
Optical absorption (A) measurements of the particles were carried out at room temperature using a PG Instrument spectrophotometer (United Kingdom) in the wavelength range of 300–800 nm. The band gap energy value was estimated from the UV- visible spectroscopic data.
The flow behavior and linear viscoelastic properties of the specimens were evaluated using rheometrics mechanical spectrometer (Paar Physica UDS200, Anton Paar GmbH, Austria) under nitrogen atmosphere at 170°C. In order to obtain the response of the materials in the linear viscoelastic region, the strain was adjusted to 1%.
The toxicity effects of fabricated sheets on fibroblast cells were evaluated by MTT assay and acridine orange (AO) fluorescence viability staining, according to Kazemi-Pasarvi et al. 38 Briefly, L929 fibroblast cells were obtained from Pasture Institute (Tehran-Iran) and then were cultured in dulbeco’s modified eagle’s medium (DMEM) supplemented with 10% fetal bovine serum (Gibco-Invitrogen) and 1% antibiotic mixture containing penicillin (Sigma-Alderich). 10 × 103 cells were seeded in each well of 96 well culture plate. After 24 h, cells were incubated with sterilized samples and then cell viability assay was investigated after 48 h. According to manufacturer’s protocol, cells were incubated with 100 μl DMEM supplemented with 20 μl MTT solution (5 mg/mL) at 37°C for 4 h. After that, formed formazan crystal of alive cells was dissolved with DMSO. The absorbance was recorded at wavelength of 570 nm using a plate reader (Awareness, Statfax 4300, USA). 39
Result and Discussion
FESEM, XRD, and FTIR results of prepared ZnO-NPs
Morphology of the nanoparticles is one of the important factors in the final properties of the bionanocomposites and can change many of their properties such as optical,
40
mechanical,
41
antimicrobial
42
and photo-catalytic activity.
43
Morphology and size of the ZnO-NPs were investigated by FESEM technique. Figure 1 shows the FESEM images of the prepared ZnO-NPs with two different magnifications. The FESEM images reveal that the ZnO-NPs are semi-spherical. As shown in Figure 1(c), the approximate diameter of the nanoparticles is 46–73 nm. FESEM images of the prepared ZnO-NPs at different magnifications: (a) X 30K, (b) and (c) X 60K.
Phase and crystalline structure of the prepared ZnO-NPs were investigated by XRD technique in ranging 2θ = 20–70°. Figure 2(a) shows the XRD pattern of the prepared ZnO-NPs. Seven strong peaks observed at around 31.8, 34.4, 36.3,47.5,56.5, 62.8, 66.3 and 67.9° are attributed to (100), (002), (101), (102), (110), (103), (112) and (201) planes of ZnO hexagonal structure, respectively.44,45 The strong XRD peaks indicate the well-crystalized structure of the prepared ZnO-NPs. No characteristic peak of impurities such as Zn and/or Zn(OH)2 was observed in the XRD pattern. The size of nanocrystals (D) was estimated using the Debye–Scherrer formula (Equation (2)) (a) XRD patterns and (b) FTIR spectra of the synthesized ZnO particles.
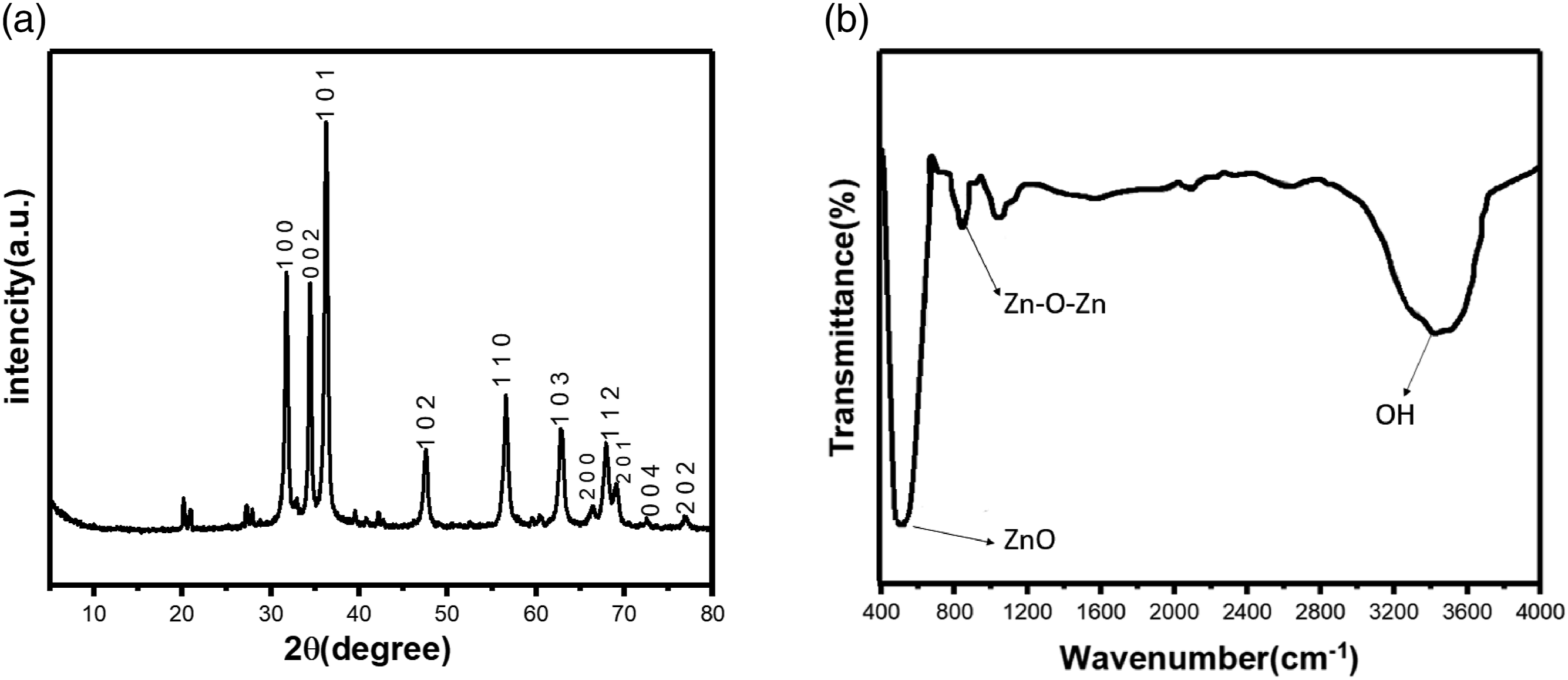
Figure 2(b) shows the FTIR absorption spectrum of the prepared ZnO-NPs. The strong and sharp absorption peak at around 400 is dedicated to the stretching vibration modes of Zn-O nanoparticles.47–49 The observed strong peak in the range of 3235 cm−1 can be attributed to the adsorbed water molecules in the KBr matrix or ZnO-NPs.44,50,51 The appeared peak at 828 cm−1 can be related to the stretching vibrations mode of Zn-O-Zn that probably is related to two zinc oxides that are linked together by removing a water molecule. 52
Thermodynamic evaluation of ZnO-NPs localization in blend bionanocomposites
Surface energy and interfacial tension are one of the most important factors in the selective localization of the nanoparticles. The interfacial tensions between the components in the blend, 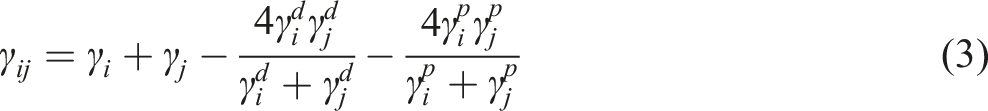
The interfacial tension between the ZnO-NPs and each blend component was calculated using the geometric mean equation of Wu (Equation (4)).
26

The non-uniform distribution of fillers within a polymer blend with matrix/dispersed morphology can be predicted by the wetting coefficient 
Therefore, using the abovementioned relations and calculating the value of
Surface tensions of PLA, PCL, and ZnO-NPs at 170°C.
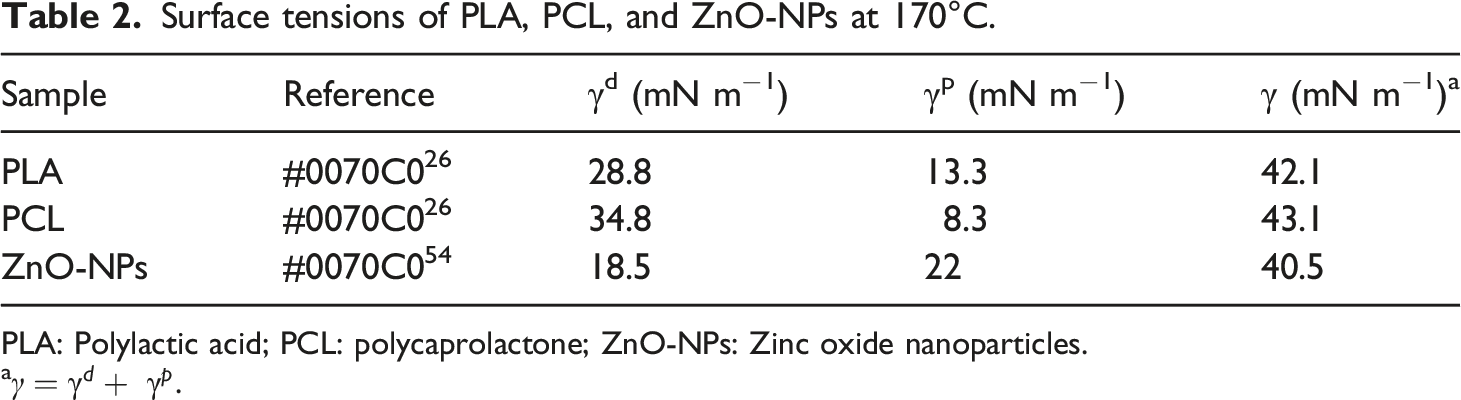
PLA: Polylactic acid; PCL: polycaprolactone; ZnO-NPs: Zinc oxide nanoparticles.
a
Calculated values of interfacial tension for various sample pairs.
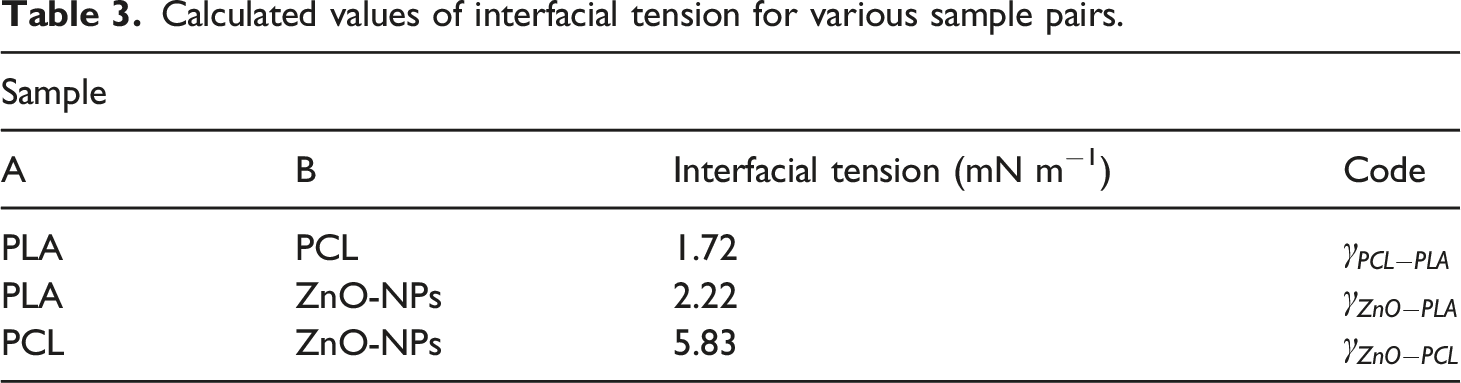
With respect to calculated wetting coefficient,
Morphological observation of blend bionanocomposites
Figure 3 shows the FESEM images of surface fractured in liquid nitrogen of PLA/PCL (80/20), and PLA/PCL/ZnO with various loadings of ZnO-NPs. Figure 3(a) shows that droplets of PCL distributed in PLA matrix. Several basic parameters affecting the evolution of binary polymer blends morphology. They are briefly: blend composition, viscosity ratio, interfacial tension, shear stress, and elasticity ratio. The literature showed if the viscosity of component B is lower than that of component A and/or component B is the major component, at processing temperature above melting point of both components (T > TmA > TmB), phase inversion may take place having component B as the continuous phase and component A as the dropltest.56,57 Therefore, despite the greater viscosity of PLA than that of PCL (see rheological characterization section), it seems the dominant composition of PLA has a key role in the developed morphology of the prepared blends in which PLA and PCL are matrix and dispersed phase, respectively. FESEM images for (a) the PLA/PCL(80:20) blend, (b) ZnO-2, (c) ZnO-4, and (d) ZnO-6.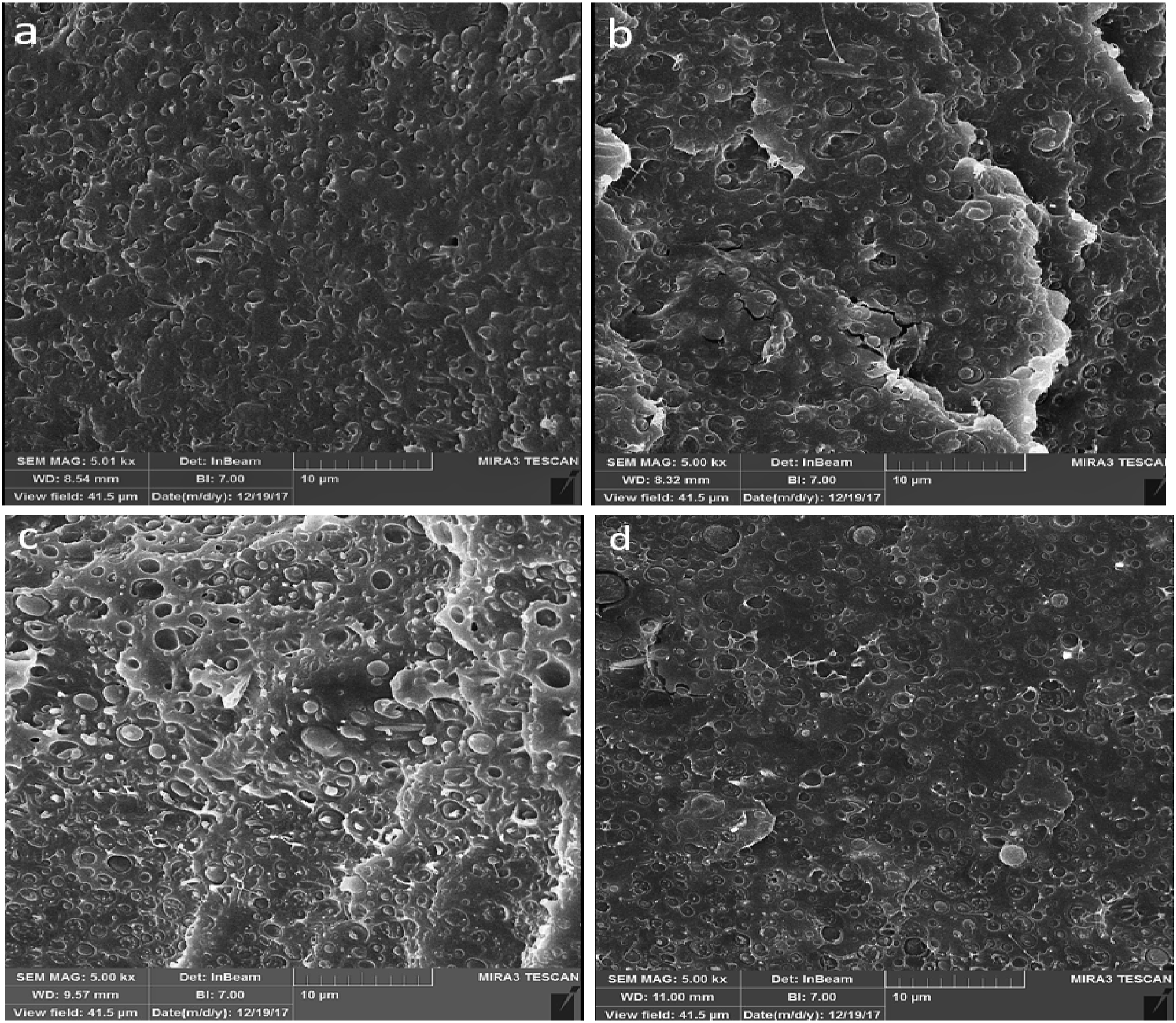
The phase morphologies of bionanocomposites are shown in Figures 3(b)–(d). As it is seen, the addition of ZnO-NPs to the PLA/PCL blend caused an increase in droplet size. According to FESEM images and taking 100 domains, the number average radii (
Mechanical properties of samples
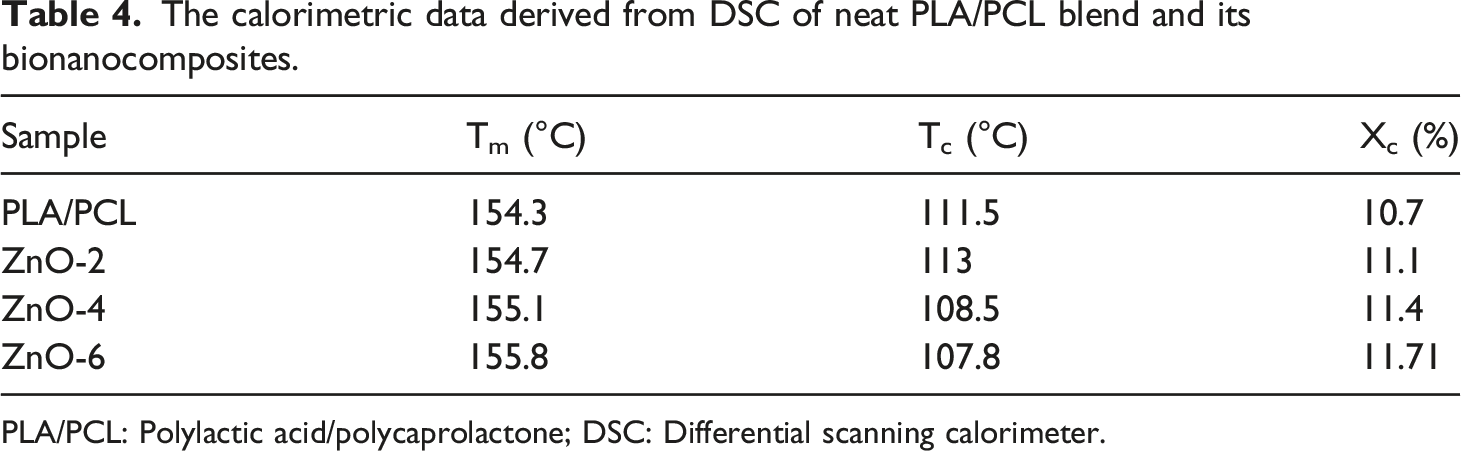
Figure 4 shows that, with the addition of ZnO-NPs into the PLA/PCL blend, the mean and standard deviations values of tensile strength and modulus were promoted while those of elongation at break decreased. The incorporation of 2, 4, and 6 wt.% ZnO-NPs into PLA/PCL decreased elongation at break from 77.97 ± 9.91 to 12.33 ± 3.96, 3.08 ± 1.05, to 2.22 ± 0.88, respectively. The tensile results indicated that the neat blends break in ductile regime while in the sample containing ZnO-NPs the ductility were significantly decreased. It needs to explain that, during tensile testing, some of the tensile bars broke around the neck of the test bar, which was far away from the gate. Therefore, the standard deviation values for the elongation at break of the virgin specimens was greater than those of other specimens. Mechanical properties of PLA/PCL blends and its bionanocomposites: (a) stress-elongation curve, (b) tensile modulus, (c) elongation at break, and (d) tensile strength. The error bars indicate the standard deviation.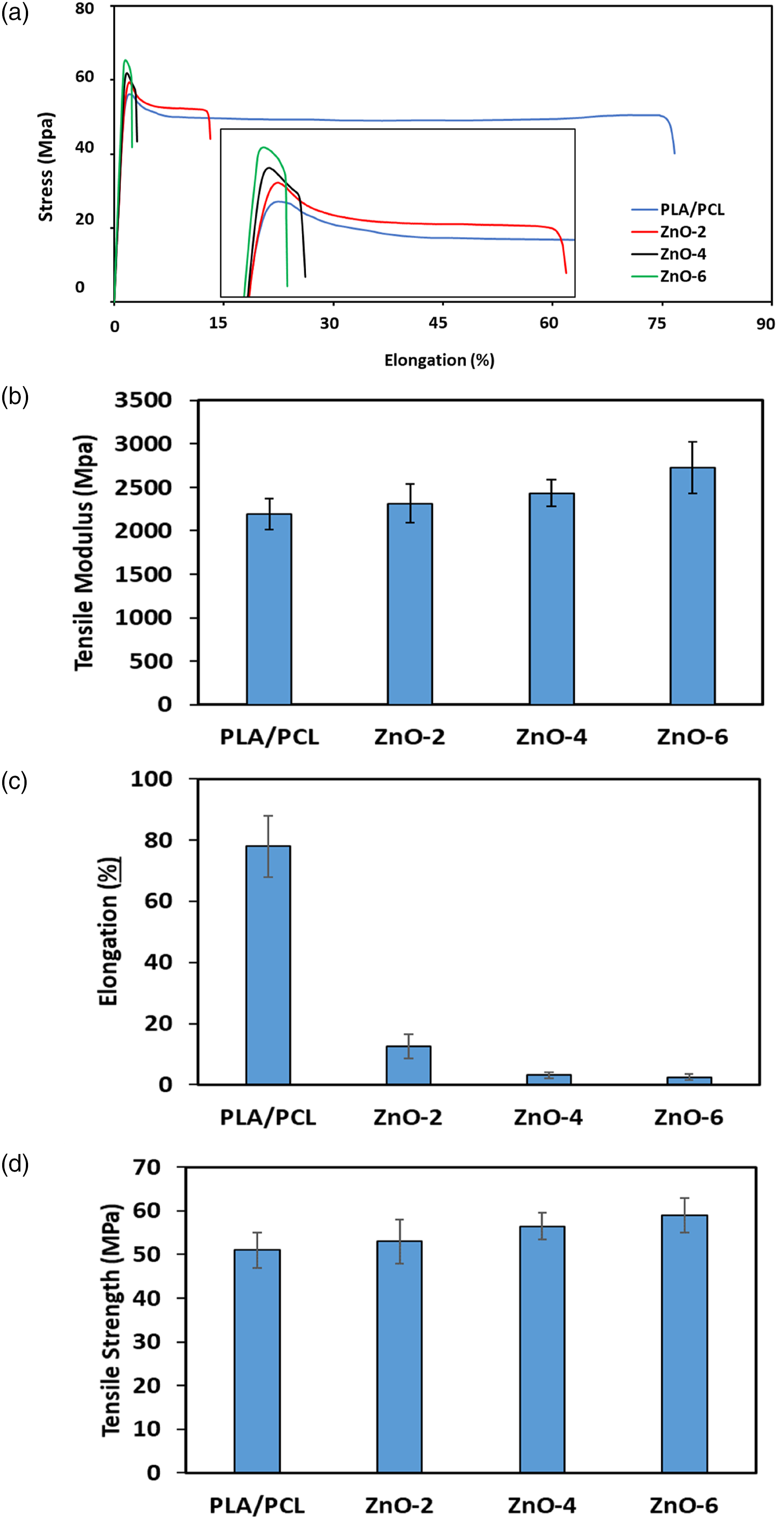
Reinforcement ability of ZnO-NPs is associated with the reduction in the mobility of polymer chains due to interacting with nanofillers61,62 The incorporation of 2, 4, and 6 wt.% ZnO-NPs into PLA/PCL increased the tensile strength from 50.88 ± 4.05 by about 52.73 ± 5.09, 56.12 ± 3.08, to 58.89 ± 3.85, respectively. Improvement of tensile strength is believed to be due to the localization of some part of ZnO-NPs in the interface between two phases, leading to the higher efficiency of stress transfer through interphase and consequently, higher ultimate strength. 22 The localization of ZnO-NPs in PLA/PCL is governed by two sets of parameters including thermodynamic and kinetic parameters. Because high viscosity of blend components, the diffusion coefficient of ZnO-NPs is extremely low. Therefore, the kinetic parameters play a role in the ZnO-NPs localization. To more explain in thermodynamic point of view based on wetting parameter the localization of ZnO-NPs would be in the PLA phase while in the kinetic point of view it is possible to trap some part of nanoparticles in the interface of the prepared binary blends. 53
Thermal properties of samples
Figure 5 shows the DSC results of the virgin PLA/PCL and PLA/PCL bionanocomposites containing various content of ZnO-NPs. The calorimetric data obtained from DSC results are shown in Table 4. Tg is the glass transition, Tc is the cold crystallization temperature peak, Tm is the melting temperature, and Xc% is the degree of cold crystallinity of the PLA of the samples. The glass transition temperature (Tg) of pure PLA is about 60°C. In the blend studied here, Tg is very close to the melting temperature of PCL (∼60°C). Hence, this overlap made it relatively hard to study the influence of nanoparticles on the glass transition temperature of PLA in the compounds. The results of thermal characterization reveal that, as the ZnO-NPs content increased in bionanocomposites, Xc and Tm increased. Incorporation of 6 wt.% ZnO-NPs into PLA/PCL increased the Xc from 10.7 to 11.71%, representing an increase of about 55%. Differential scanning calorimeter of neat PLA/PCL blend and its bionanocomposites. The calorimetric data derived from DSC of neat PLA/PCL blend and its bionanocomposites. PLA/PCL: Polylactic acid/polycaprolactone; DSC: Differential scanning calorimeter.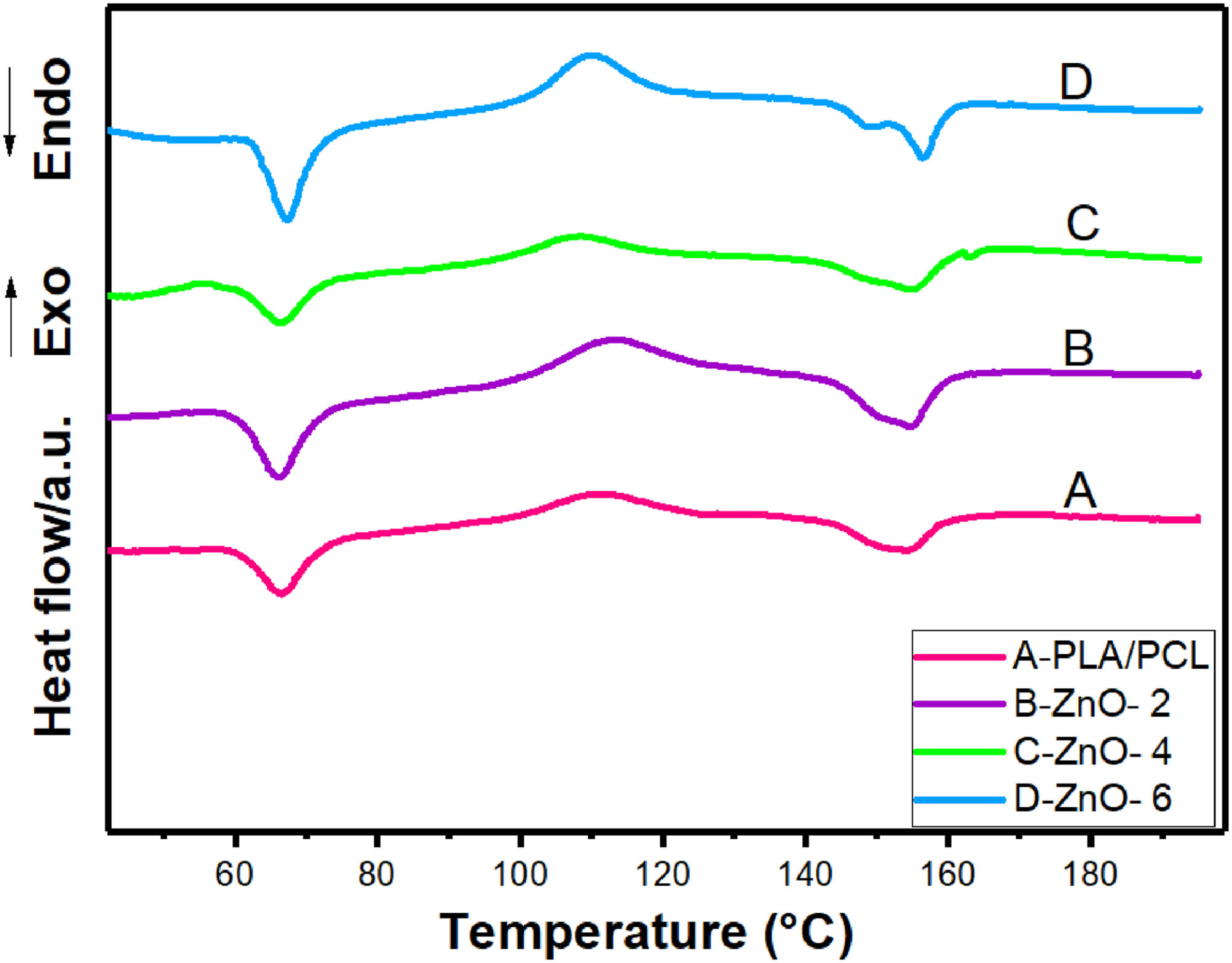
The two melting peaks around 155°C correspond to the two types of PLA crystals. One is the melting of the crystalline phase with less organized crystals and the other is the better crystalline phase at the higher temperature.26,63
The results showed while the addition of 2 wt% of ZnO-NPs led to the elevating the cold-crystallization temperature, bionanocomposite having high loading of nanofillers showed less Tc, even less than that of pure PLA/PCL blend. These observations can be correlated to the nucleating and degrading role of added nanoparticles. Results suggest that, they are acting in a same direction for low amount of ZnO-NPs nanofillers and for higher amount of loading, they operate in an opposing manner. Meaning that, incorporation of ZnO-NPs can be considered that is associated with two inducing mechanisms on the cold-crystallization of PLA chains. First, ZnO-NPs increases the nucleation sites and, as a result, capability of crystallinity. Second, since PLA having a slow rate of crystallization, lower molecular weight caused by degradation of ZnO-NPs facilitates the motion of polymer chain toward the nuclei.64–66 At 2 wt% of ZnO-NPs, since the level of degradation is limited, both of these mechanisms favor the higher degree of crystallinity. Nevertheless, when the nanofiller loading increased, degradation extent was intensified and average number of molecular weight decreased which results in the higher mobility of polymer chains. It can be suggested that, while this higher degree of mobility resulted in the decrease of Tc, assisted in the elevated capability of polymer chain moving toward nuclei and advanced degree of crystallinity, manifested by higher Xc and Tm.
Photochemical degradation
This degradation and chain scission of polymers show up directly with the reduction of their mechanical properties. Therefore, samples will be exposed to UV light for 1–50 h. Tensile modulus and strength under exposure UV are considerably reduced, which is in direct relation with the time of exposure and ZnO-NPs concentration. In the same conditions after 50 h, the modulus of ZnO-6 has reached 10% compared to first day. Considering the Figure 6, after some time, in some samples, we saw an increase in the elongation at break, which is probably due to the reduction of chain entanglements due to the degradation and breaking of polymeric chains. Mechanical properties of PLA/PCL blend and its bionanocomposites after exposure to UV light, (a) Module, (b) elongation at break, and (c) Tensile strength.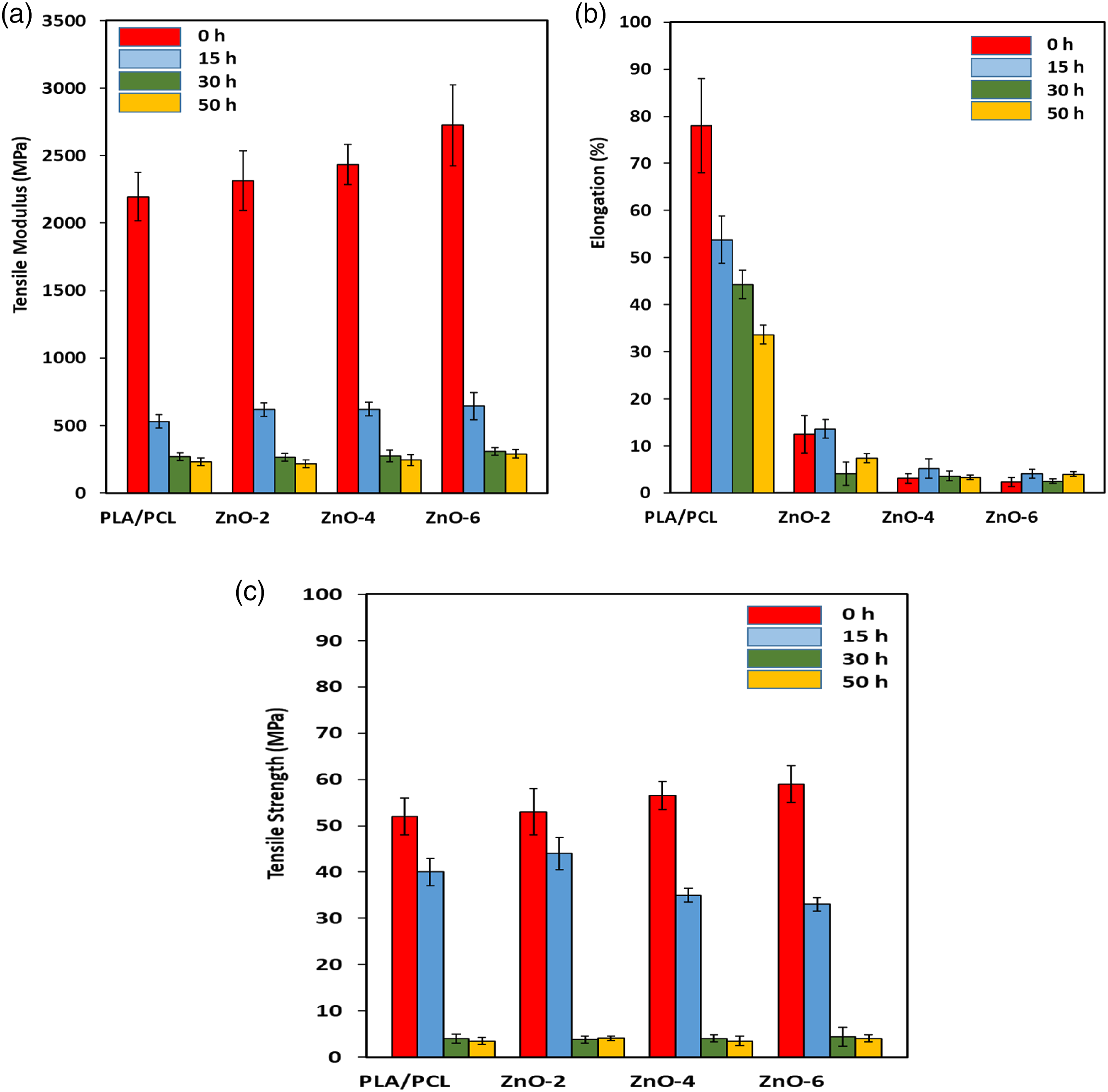
UV-Vis Analysis
Figure 7(a) shows the UV-Vis optical absorption spectra of the PLA/PCL blend and its bionanocomposites with different percentages of the ZnO-NPs. The observed strong absorption peak at 220 nm (UV-C region) is related to the band gap energy of the PLA/PCL blend. As can be seen, with increasing the ZnO-NPs loading in the bionanocomposite samples, in addition to the observation of absorption peak at around 220–230 nm, a strong and sharp absorption peak at 370 nm (UV-A region) is appeared. The observed peak at 370 nm is a characterization peak of ZnO-NPs arising from their band gap energy. Although the absorption intensity in the UV-C and UV-A regions obviously increases with increasing the ZnO-NPs loading in the bionanocomposite samples, but this increasing is considerably more sever in the UV-A region, indicating a good distribution/dispersion of the ZnO-NPs in the polymer matrix. Besides, the observation a very sharp absorption edge at 370 nm for bionanocomposite samples containing 4 and 6 wt% of nanoparticles indicates a narrow size distribution of the ZnO-NPs and confirms a very good dispersion of ZnO-NPs in the polymer matrix with a minimum agglomeration. In the absorption spectrum of 2 wt% of ZnO-NPs in the bionanocomposite samples, the intensity of peak at 370 nm is very weak, indicating the lack of sufficient amount of the ZnO-NPs and as a result, a poor dispersion of the ZnO-NPs in the bionanocomposite samples. (a) UV-Vis absorbation spectra and (b) (αhν)
2
versus photon energy (hν) of of neat PLA/PCL blend and its bionanocomposites.
Tauc’s equation was used for determination of the band gap energy of the samples. Figure 7(b) shows the plot of the (αhυ) 2 versus the photon energy (hυ) for the prepared bionanocomposites at different percentages of the ZnO-NPs. The band gap energy of the PLA/PCL blend is 4.87 eV while with increasing ZnO-NPs percentage in the bionanocomposite samples, band gap energy values clearly shift to higher wavelengths (lower energy/red shift) due to the band gap energy of the ZnO-NPs in the polymer matrix. As can be seen, the band gap energy of the prepared bionanocomposite samples with 4 and 6 wt% of ZnO-NPs are 3.33 and 3.21 eV, respectively. These values are related to band gap energy of the ZnO-NPs, indicating a low agglomeration and a very good dispersion of ZnO-NPs in the matrix. If there was a high agglomeration of ZnO-NPs in the prepared bionanocomposite samples, either the sharp absorption edge of the ZnO-NPs is not observed or a broad absorption edge with the band gap energy smaller than that of the bulk ZnO-NPs is obtained.
Rheological characterization
Figure 8 shows the storage modules and complex viscosity of PLA/PCL blends and its bionanocomposites with different amount of ZnO-NPs as a function of frequency. Since the viscosity ratio is about 16, PLA shows more viscosity than PCL in all frequencies. It can be observed that storage modulus and complex viscosity of bionanocomposites decreased with increasing ZnO-NPs, and also reduced slope of complex viscosity. This can be attributed to the role of the ZnO-NPs to degradation of the polyester matrix via transesterification reactions. Due to degradation, the molecular weight is reduced and, we see a decline in elasticity. This molecular weight loss reduces viscosity. It is true that, the temperature and shear during the melt mixing method itself are causes degraded, and this is unavoidable. But this factor is minimized by the use of nitrogen. As a result, this decrease in viscosity is more due to the presence of ZnO-NPs.
67
(a) Storage modulus and (b) complex viscosity as a function of frequency for PLA, PCL, PLA/PCL blend, and its bionanocomposites.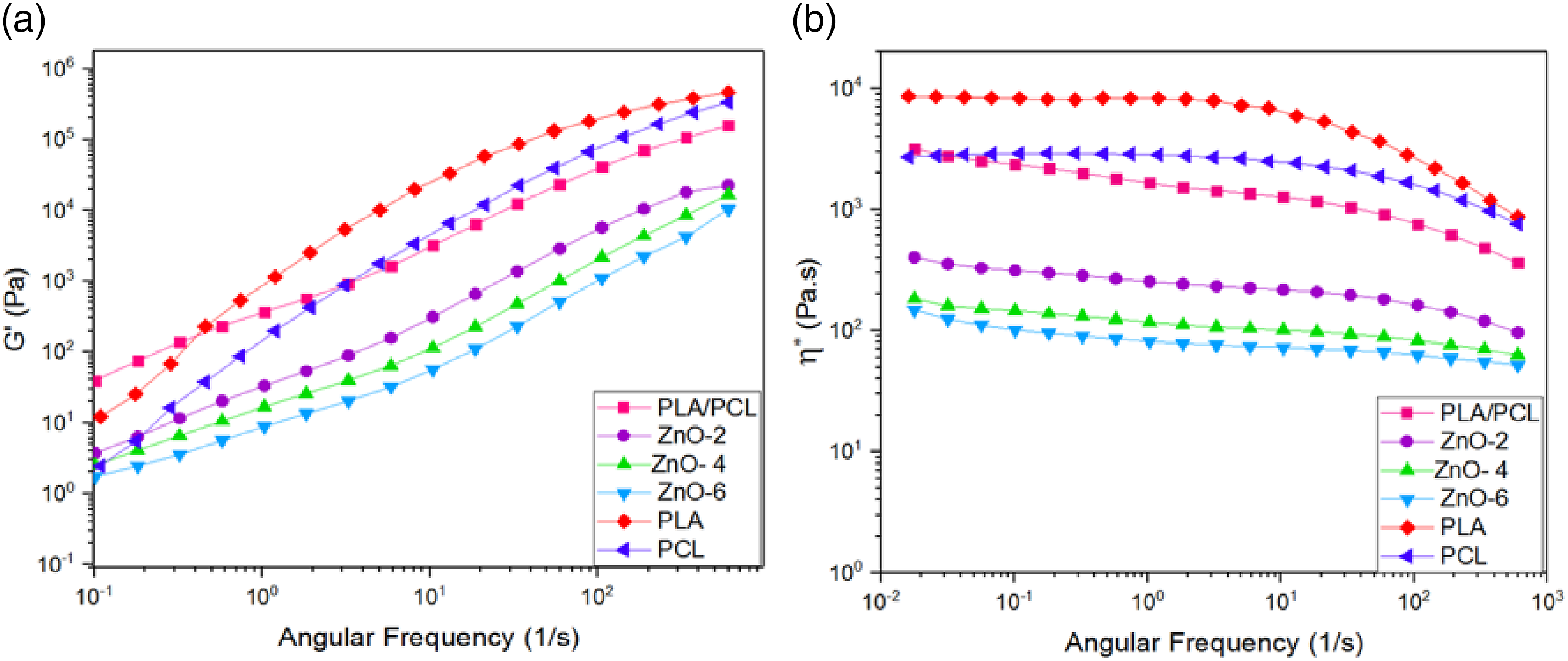
It is also interesting to note while the complex viscosity and storage modulus of the blends decreased with incorporation ZnO-NPs, their tensile modulus and strength increased (see the mechanical properties section). It seems detrimental effect of degradation of ZnO-NPs is more pronounced in the case of rheological properties and some minor degradation of polymer chains occurred at low time of compounding was in the range of sensitivity of rheological assessments. One may notice that beyond a certain value of molecular weight, numbers of repeating unit in polymer chain has no notable effect on the mechanical properties 68 and therefore decreasing in molecular weight of polymer in PLA/PC blends at the presence of ZnO-NPs due to degradation has no adverse effects in mechanical properties.
Figure 9 shows plotting complex viscosity (η*) against complex modulus (G*). The increase in the slope of η*- G*and the suddenly shift in the graphs represent the formation of a three-dimensional physical structure. So that the threshold of rheological accumulation can be determined using this graph.
69
As the nanoparticles increase, the graph shows a divergence, and the slope of the graph became steeper. This can be attributed to the 3D-network structure due to hydrodynamic interaction between both particle-particle and/or polymer-particle. Plots of η∗ versus G∗ for PLA, PCL, PLA/PCL blend, and its bionanocomposites.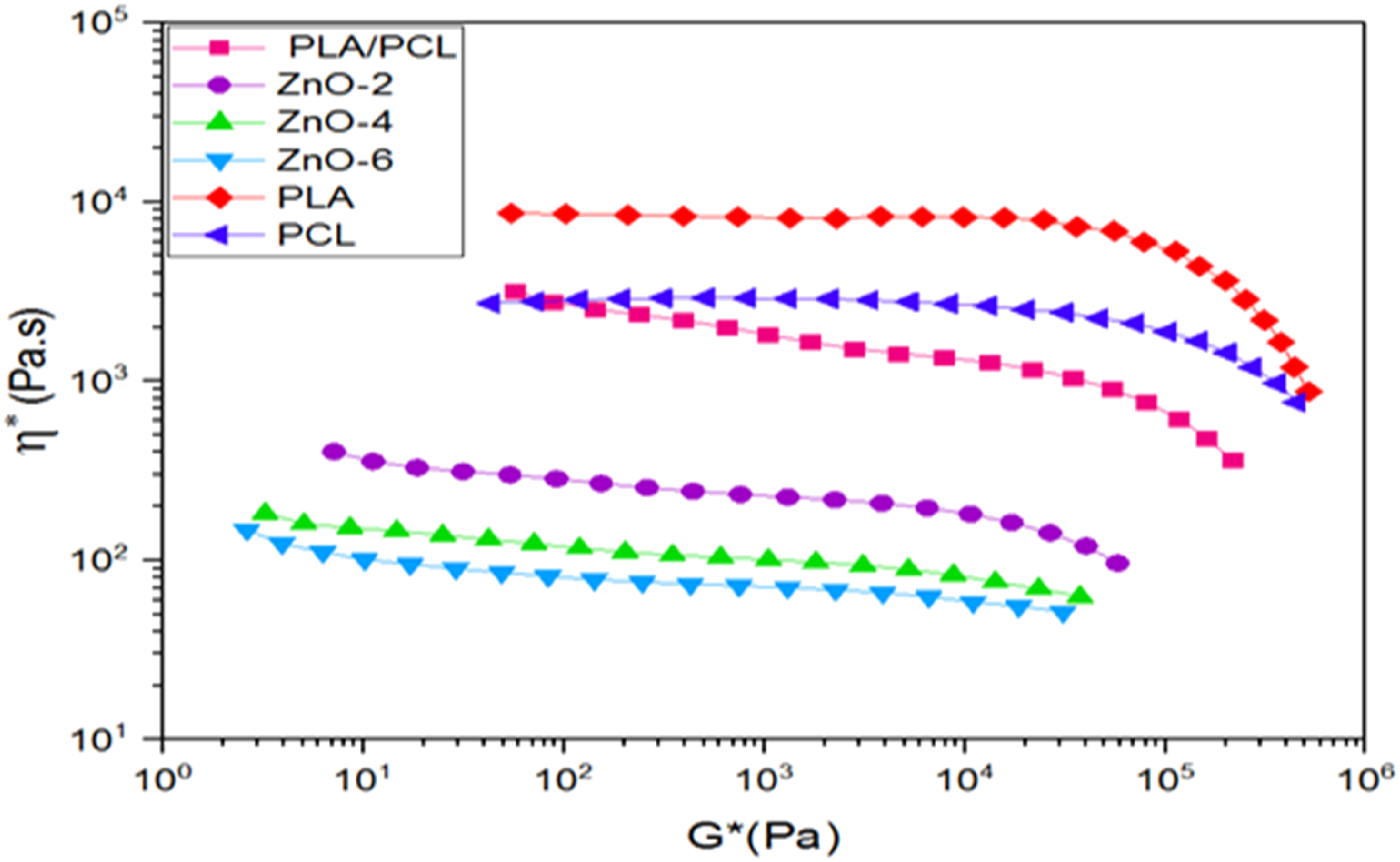
MTT assay results
An in vitro cytotoxicity assay was carried out for consideration of the harmful effect of samples. The viability of samples in the exposure of the normal fibroblast cells after 1, 3 and 5 days are shown in the Figure 10. As expected, there are no significant change in the cell viability of PLA/PCL blend at experiment days due to its biocompatibility and non-toxicity. The MTT assay results demonstrated that the cell viability of bionanocomposites slightly reduced by addition the ZnO-NPs in the PLA/PCL blend as well as time of incubation compared to control sample. It presumably comes from the release of zinc ions which can bond with oxygen and hydrogen atoms present in the intracellular amino compounds leading to ROS generation. Subsequently, it causes serious damages in the membrane protein which consequently lead to the cell death. It is interesting that, the value of cell viability on all bionanocomposites were greater than 90% which represents outstanding biocompatibility of the bionanocomposites to the normal fibroblast cells. MTT assays of cell viability of PLA/PCL and its bionanocomposites in days 1, 3, and 5.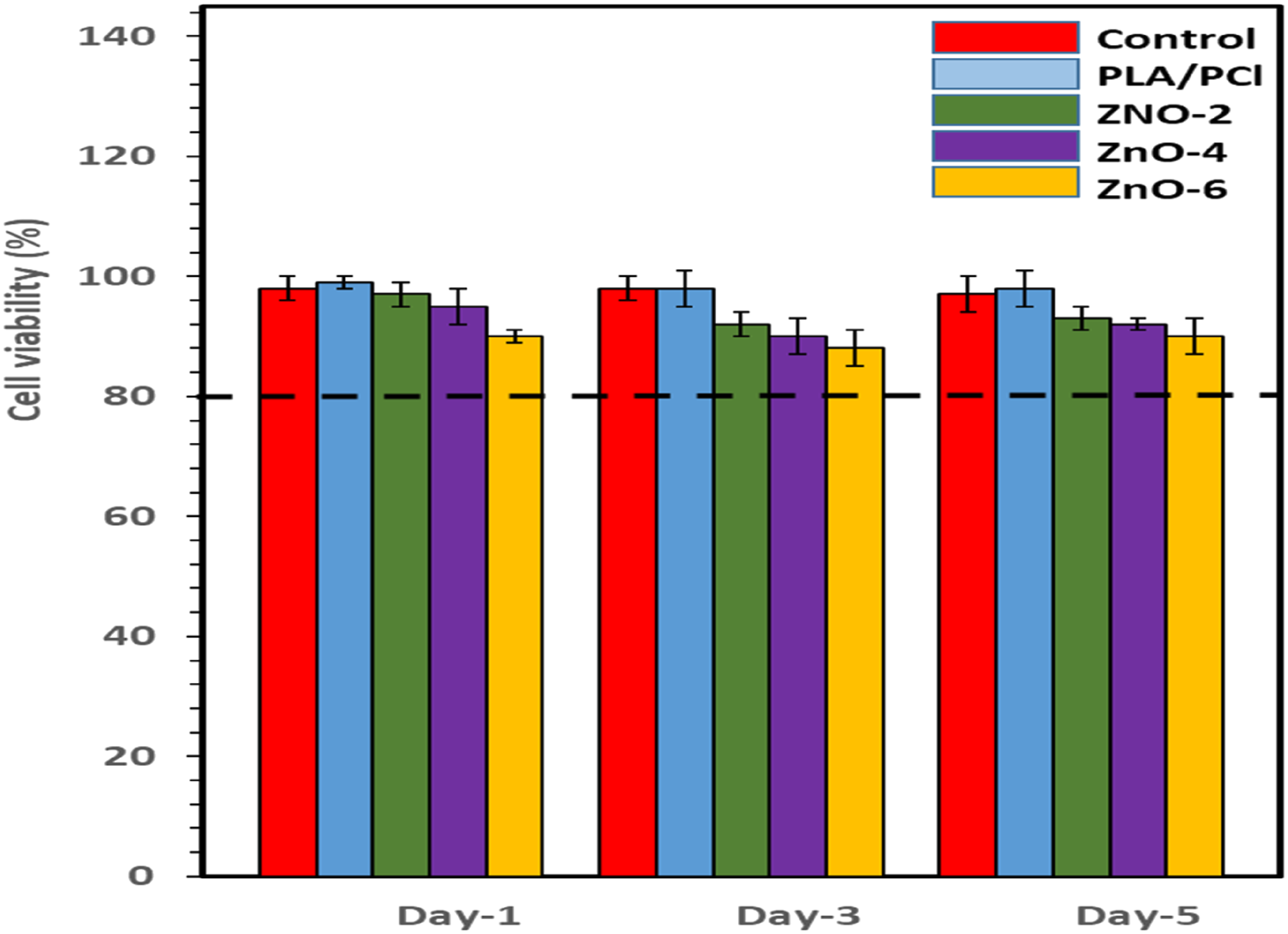
Conclusions
In this work, ZnO-NPs having the hexagonal structure were successfully synthesized using hydrothermal method. FE-SEM results showed that the average diameter of the obtained ZnO-NPs was about 46–73 nm. The PLA/PCL (80/20 wt/wt) bionanocomposties containing were 2, 4, and 6 wt% of ZnO-NPs were prepared via melt mixing. The calculated wetting parameter showed that the ZnO-NPs is thermodynamically preferred to be located in PLA matrix. The morphological analysis illustrated that incorporation of ZnO-NPs into PLA/PCL blend increased interestingly the PCL droplet size. The young modulus and tensile strength of the bionanocomposites increased with increasing the ZnO-NPs loadings as a results of reinforcement effects. The retarded mobility of polymer chains at the presence of ZnO-NPs led to decrease in elongation at break of blends. The cold crystallization temperature of PLA in the blend decreased at the presence of high loadings of ZnO-NPs due to the enhanced moving capability of PLA toward nuclei as a consequence of degradation. Degradation of the PLA/PCL chains via transesterification reactions due to ZnO-NPs resulted in decreasing the storage modulus and complex viscosity over the entire range of frequencies. It was observed that the photochemical degradation of the samples was accelerated with incorporation of the ZnO-NPs. The fabricated bionanocomposites did not show any toxicity into the fibroblast cell and therefore can be applied in biomedical applications.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author contribution
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Amir Babaei, Mehdi Haji Abdolrasouli. The first draft of the manuscript was written by Amir Babaei and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
