Abstract
In this work, nanocomposites of polyvinyl chloride poly(epichlorohydrin-co-ethylene oxide)(ECO)/organoclay were prepared via melt processing and various parameters including nanoclay and rubber content (ECO) as well as rotor speed were tuned to find the optimum formulation for the highest thermal stability. The prepared products were characterized by X-ray diffraction as well as thermogravimetric analysis (TGA), derivative thermogravimetric (DTG) and differential thermal analysis XRD results showed that rotor speeds higher than 70 r/min are crucial for obtaining highly intercalated products with good thermal stability. From DTG analyses, it was observed that at lower concentrations of rubber, the rate of mass loss is higher which results in faster dehydrochlorination of the composite. The sample prepared with 2 phr OMMT, 30 phr rubber, and 70 r/min rotor speed showed the highest thermal stability. The selected nanocomposite showed the first weight loss at 294
Introduction
Since the development of the polymer nanocomposites in 1960s, 1 their extraordinary properties has attracted great interest. Due to unusual concurrent improvement in mechanical, thermal, physical, and flammability properties of organic and inorganic polymers by adding known values of silicate materials, this subject has been studied extensively in recent years.2–4 Montmorillonite (MMT), a naturally occurring layered silicate consisting 1 nm thin layers, with a central octahedral sheet of alumina fused between two external silica tetrahedral sheets is one of the attractive materials for this application. The hydrophilic nature of the interlayers in its pristine form, makes MMT only miscible with hydrophilic polymers. 5 One solution to this problem is using organophilic montmorillonite (OMMT) in which the alkali ions are exchanged with cationic organic agents such as alkylammoniums. One of the common thermoplastics which is generally used is polyvinyl chloride (PVC) which is intercalated between the layers of OMMT. The resulting PVC/clay nanocomposite usually shows high mechanical and thermal stability.
One important issue in preparation procedure of PVC/clay nanocomposites is poor heat resistance of PVC which undergoes dehydrochlorination at relatively low temperatures (<250
Although using OMMT has been a novel approach for improving thermal resistance of the nanocomposites, better dispersion of the organoclay in polymer matrix is still an issue. One of the proposed solutions to this problem is using elastomers as compatibilizers having interactions with organoclays. 15 Recently, elastomers have been also used to toughen polymer composites such as polyamide-6 (PA6). 16 Among different elastomers, polyepichlorohydrin has been mentioned as an outstanding rubber for its resistance against high temperature as well as ozone, fuel, and oil. Therefore, it is suitable for many applications such as production of emission tubes, automotive, vacuum hoses, air ducts, etc. 17 Polyepichlorohydrin elastomers are able to make chemical bonds with organic modifiers of nanoclays and therefore are miscible with polymer/OMMT nanocomposite. In our previous work, we used Poly(epichlorohydrin-co-ethylene oxide) (ECO) as an electrostatically dissipative compound to see its effect on fusion behavior of (PVC/ECO)/organoclay nanocomposites. 18 The results of our study, based on response surface method (RSM) showed that low levels of the ECO content, and low or medium levels of OMMT and rotor speed are the optimum conditions for lower fusion time (FT) and higher fusion factor (FF). In continuation of our previous work, herein, we are going to investigate the effect of ECO, OMMT, and rotor speed parameters on thermal stability of (PVC/ECO)/OMMT nanocomposite. To the best of our knowledge, this is the first study which shows thermal stability of this composition and the effect of reaction parameters in melt compounding.
Materials and methods
Materials and characterization
X-Ray diffraction (XRD) patterns were recorded by a Philips-X’Pert Pro, X-ray diffractometer using Ni-filtered Cu Kα radiation at scan range of 10°<2θ<80°. Scanning electron microscopy images were obtained on LEO-1455VP. Thermal analyses including thermogravimetric analysis (TGA), derivative thermogravimetry (DTG), and differential thermal analysis (DTA) were performed on a TG/DTA 3600 unit from EXSTAR with an aluminum cell, under a constant nitrogen flow of 200 mL/min. The experiments were performed at a temperature ramping of 10
PVC resin (suspension grade) with a K-value of 565 was purchased from Bandar-e-Imam petrochemicals (Iran). The number- and weight-average molecular weights of the resin were 40,000 and 90,000 g/mol, respectively. ECO rubber, a copolymer of epichlorohydrin and ethylene oxide, was provided by Robinson Rubber Products. The NC (under the commercial name of Cloisite 30B) was obtained from Southern Clay Products (USA). This organoclay was modified with methyl, tallow, bis-2-hydroxyethyl quaternary ammonium cation. The density, cation exchange capacity and gallery space of the nanofiller are 1.98 g/cm3, 90 meq/100 g, and 18.5 Å, respectively.
Sample preparation
Array of the parameters used to prepare composite samples.
Calculations
The optimization of the molecular structures of ammonium cation in organoclay having hydrogen bond interactions with two monomers of ECO polymer in two different bonding modes (O-H and Cl-H) in the ground state were optimized on the basis of density functional theory (Hartree-Fock) at the Default Spin (with 6-31G basis set) and by means of visual inspection using the GAUSSVIEW program (Version 5.0). All the calculations were performed using the GAUSSIAN 09 software package. 19
Results and discussion
Characterization of as-prepared samples
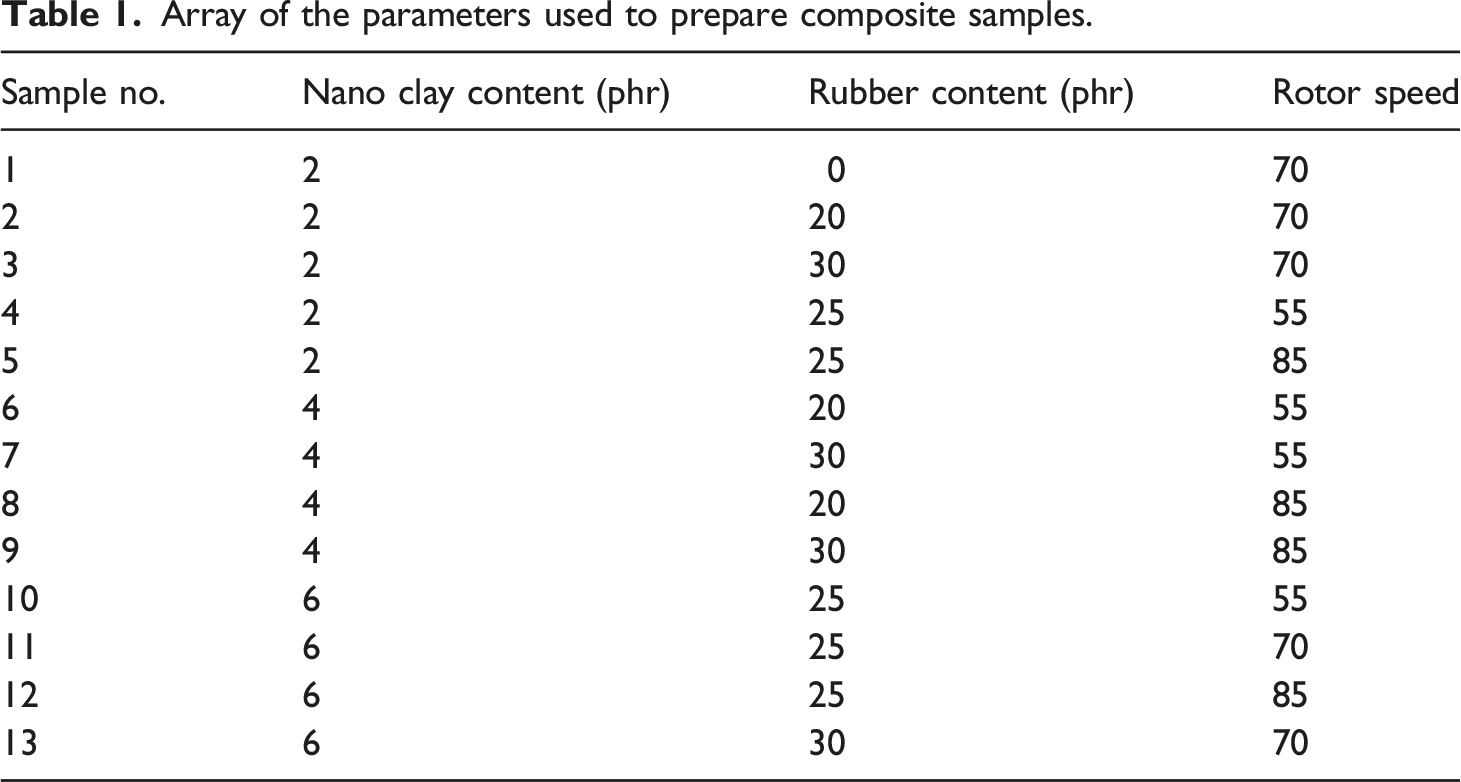
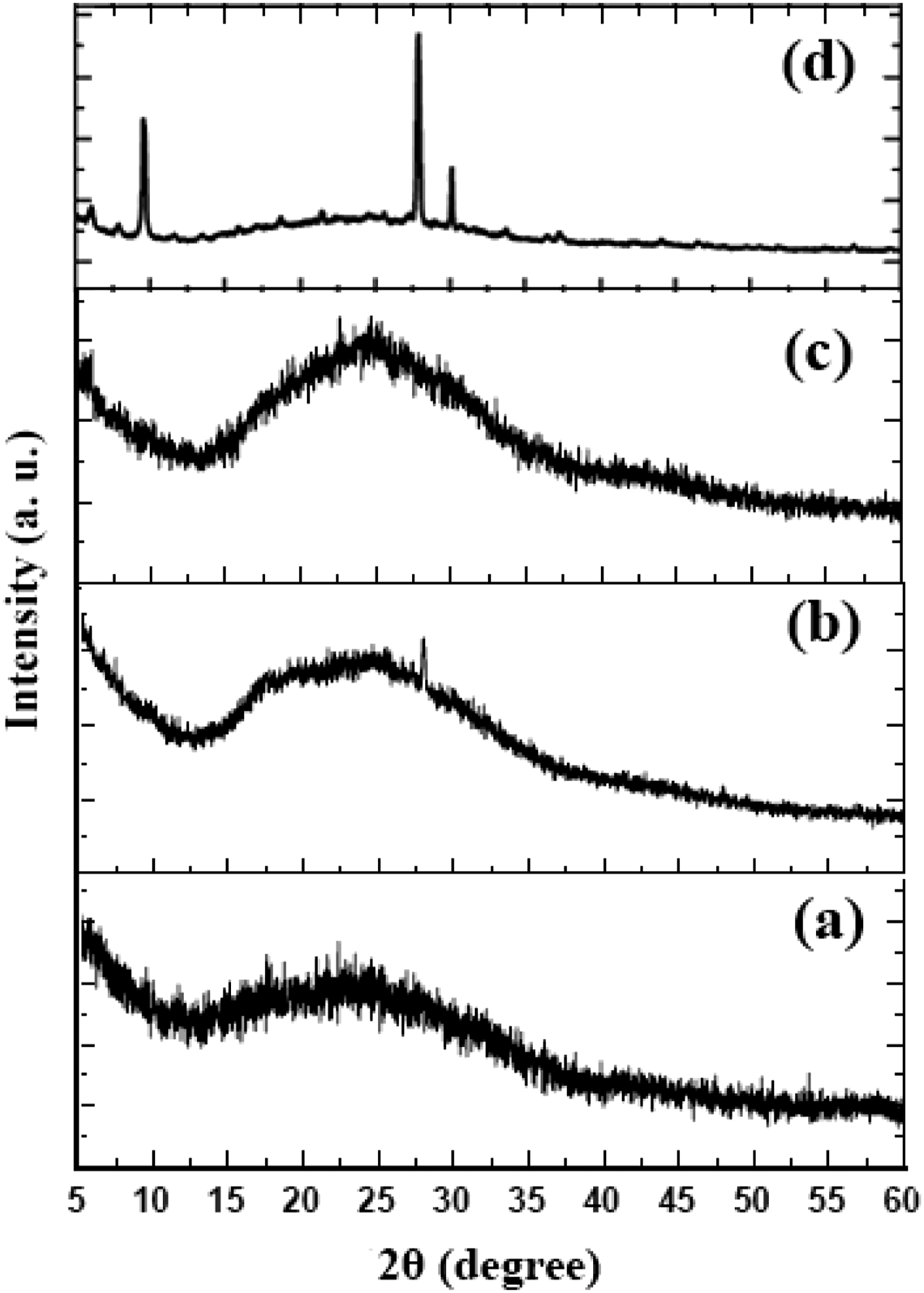
The phase of the samples was characterized by their X-ray diffraction patterns. Figure 1 and Figure 2-a-f show the XRD patterns of pure OMMT and samples no. 1 to no. 5, respectively which were prepared by adding 2 phr of OMMT with different amounts of rubber and rotor speeds. In all patterns, two peaks at 2θ = 10° and 28° representatives of OMMT are appeared with a slight shift towards lower angles. This shift generally occurs due to the intercalation of polymer chains in the OMMT interlayers ending up in increase of the d-spacing.
20
The broad peak appeared in the range of 2θ = 20–35° is assigned to PVC polymer. Comparing sample no. 1 with no rubber content with sample no. 2 and 3 with 20 and 30 phr rubber contents, respectively, it can be seen that increasing the rubber content results in decreasing the interlayer distance of the nanoclay since the nanoclay peak at 2θ = 28° for sample no. 1 has slightly moved to higher angles in sample no.3. It has been reported that the increased amount of rubber results in partial aggregation of rubber in PVC matrix and therefore less PVC is intercalated in nanoclay interlayers.
21
Selecting 25 phr rubber content and changing the rotor speed to 55 and 85 r/min shows that higher rotor speed has resulted in better intercalation of PVC and rubber in OMMT interlayers since the OMMT sharp peaks are distinguished from PVC background broad band. Because of higher affinity between ECO chains and organoclay, it is more probable for ECO chains to be intercalated between the clay galleries. Similar result has been reported in previous studies.
22
XRD pattern of pure PVC. XRD pattern of (a) pure OMMT and different samples containing 2 phr nanoclay, samples (b) no. 1 (c) no. 2 (d) no. 3 (e) no. 4 (f) no. 5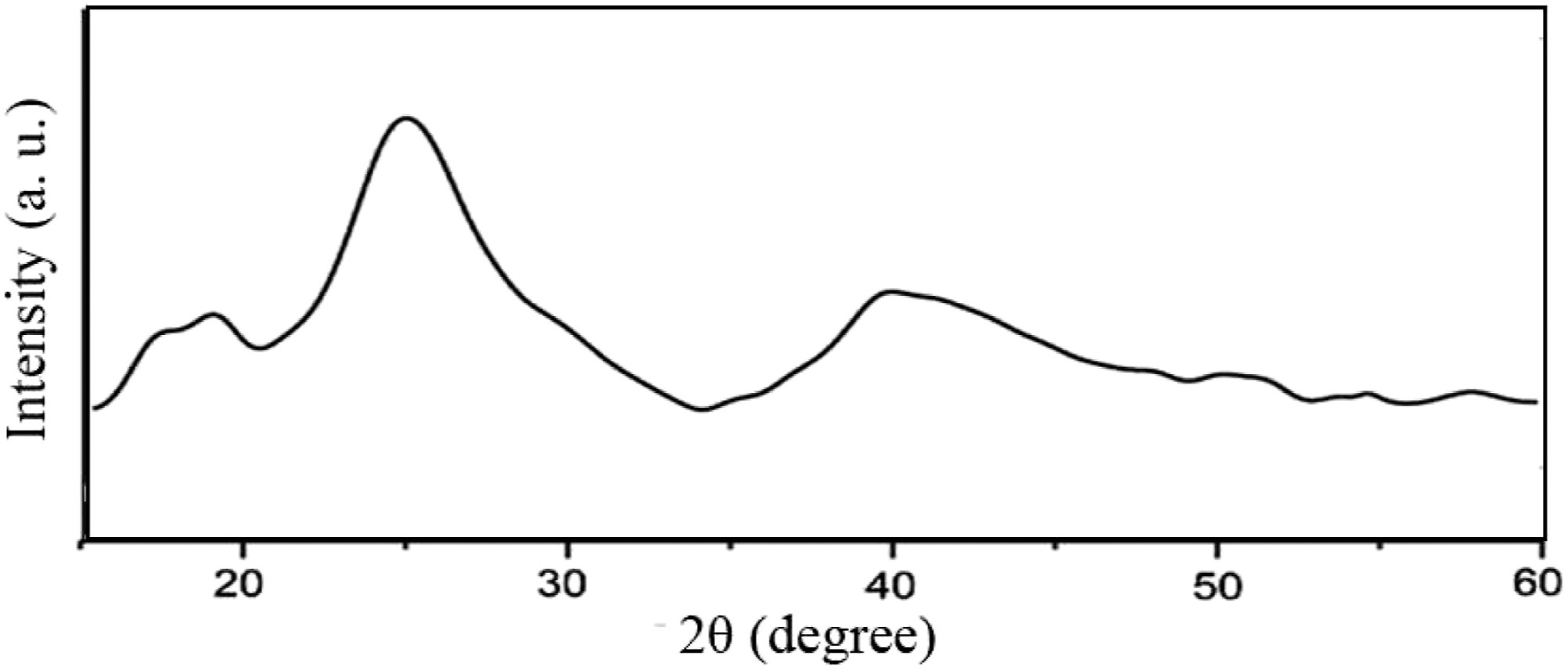
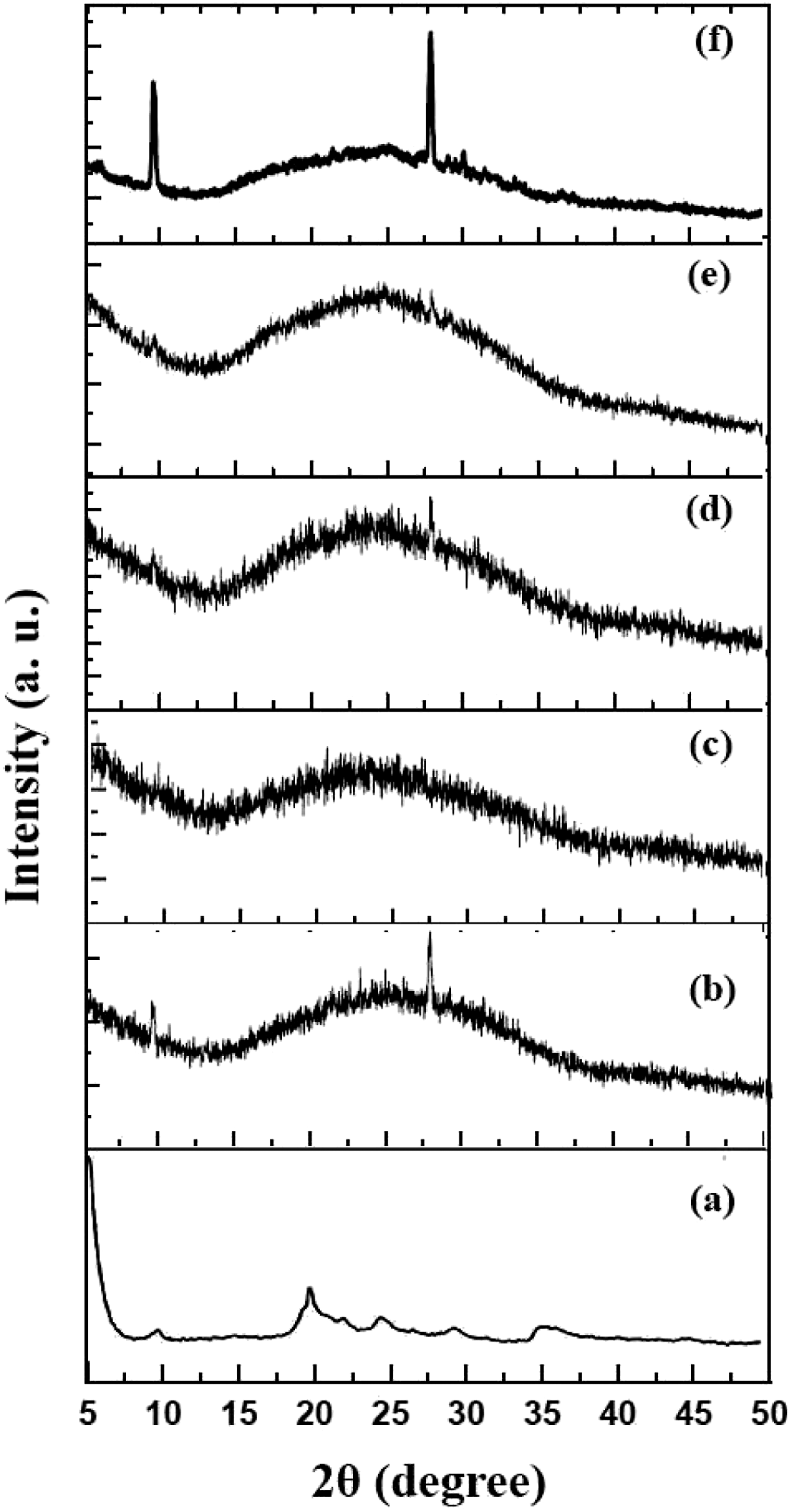
Figure 3 and Figure 4 show the XRD patterns of the samples containing 4 and 6 phr nanoclay, respectively. In Figures 3(a) to (d), the effect of increasing rubber content in two different rotor speeds (55 and 85 r/min, respectively) is shown. In Figures 4(a) to (c), the XRD patterns of three different samples prepared under various rotor speeds (55, 70, and 85 r/min, respectively) are shown. It is seen that a regular trend is not observed here and three parameters including OMMT content, rubber content, and rotor speed are affecting the crystal structure of the composite. However, it is clearly observed that at 4 phr OMMT, the tuned parameters do not induce huge change in the final structure of the sample and at 6 phr OMMT, the significant change is observed in sample no. 13 which gives a different crystal structure showing OMMT peaks. From the XRD analyses, it is concluded that the rotor speed is a more important parameter in producing nanocomposites with more intercalated structures. As observed here, the rotor speeds lower than 70 r/min are not much effective in structural intercalation of the components. XRD pattern of different samples containing 4 phr nanoclay, samples (a) no. 6 (b) no. 7 (c) no. 8 (d) no. 9 XRD pattern of different samples containing 6 phr nanoclay, samples (a) no. 10 (b) no. 11 (c) no. 12 (d) no. 13.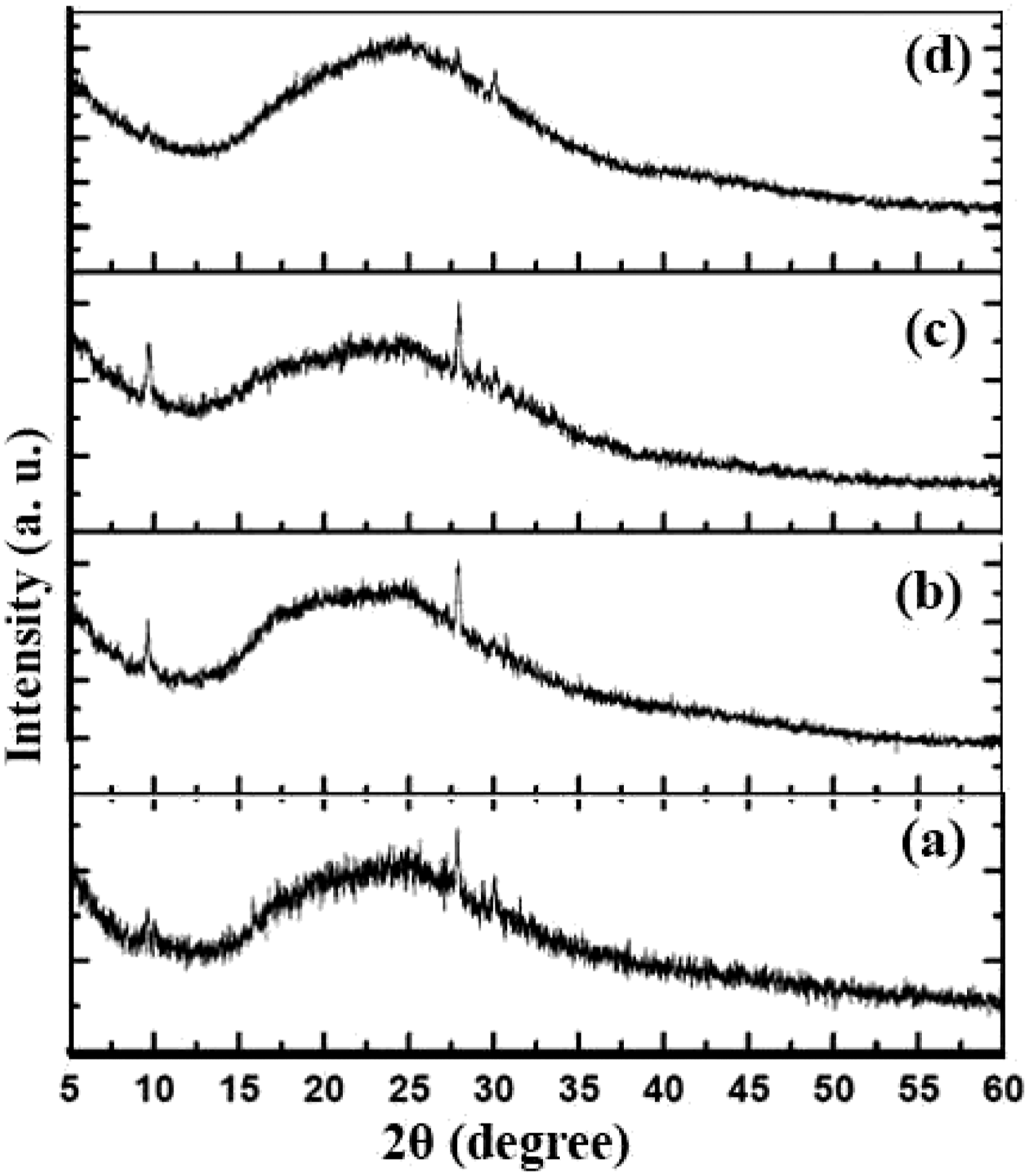
The thermal decomposition of the samples was studied by using DTG/DTA analyses. Figures 5 to 7 show the corresponding diagrams for the samples containing 2, 4, and 6 phr of nanoclay. Moreover, TGA analysis was performed for all the samples. The obtained data from DTG, DTA, and TGA analyses are available in Table 2. (a) DTG (b) DTA curves versus time for different samples containing 2 phr nanoclay, samples no. 1, 2, 3, 4, and 5 (a) DTG (b) DTA curves versus time for different samples containing 4 phr nanoclay, samples no. 6, 7, 8, and 9 (a) DTG (b) DTA curves versus time for different samples containing 6 phr nanoclay, samples no. 10, 11, 12, and 13. Data collected from TG, DTG, and DTA of different samples with 2, 4, and 6 phr OMMTs.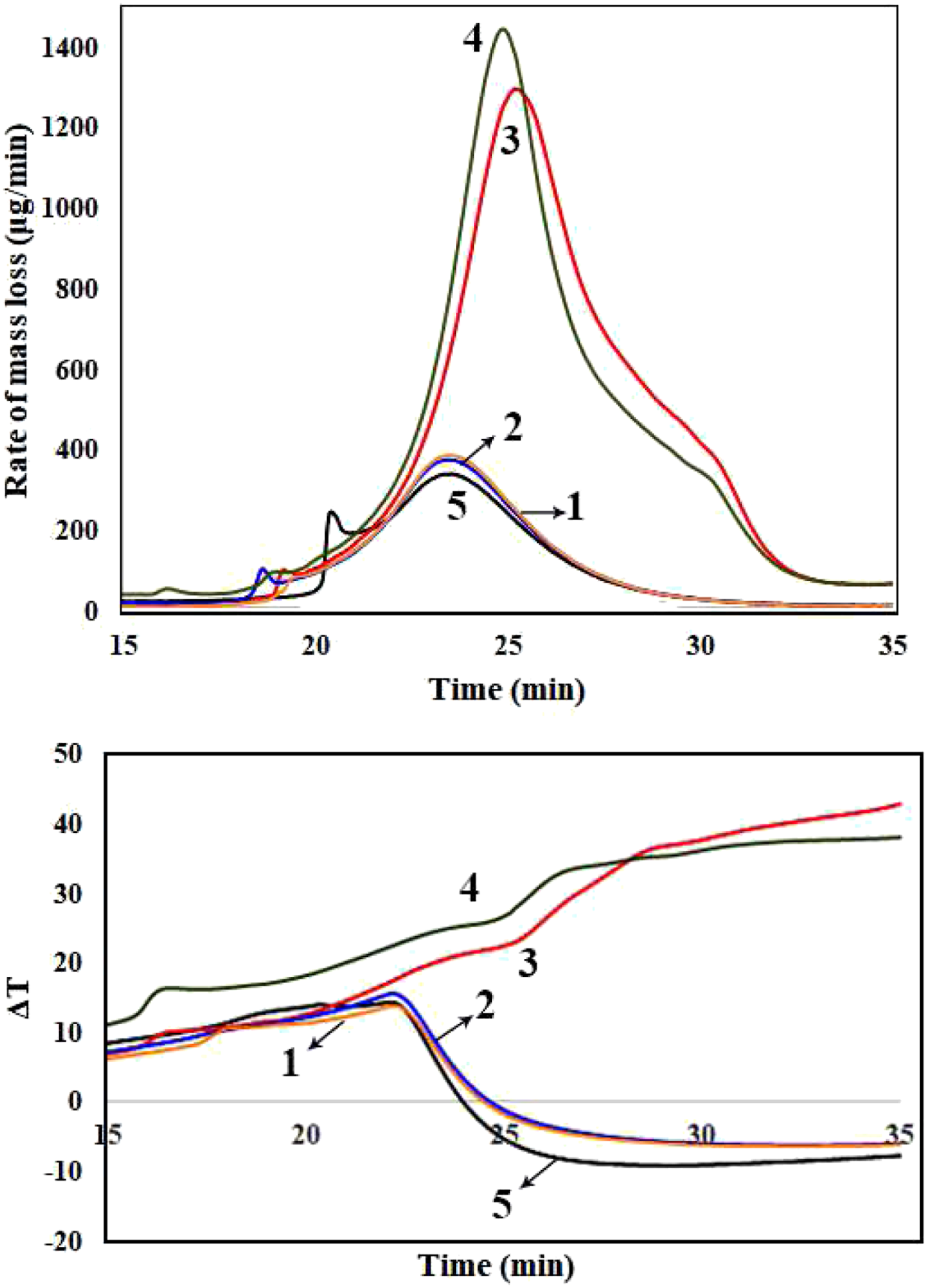
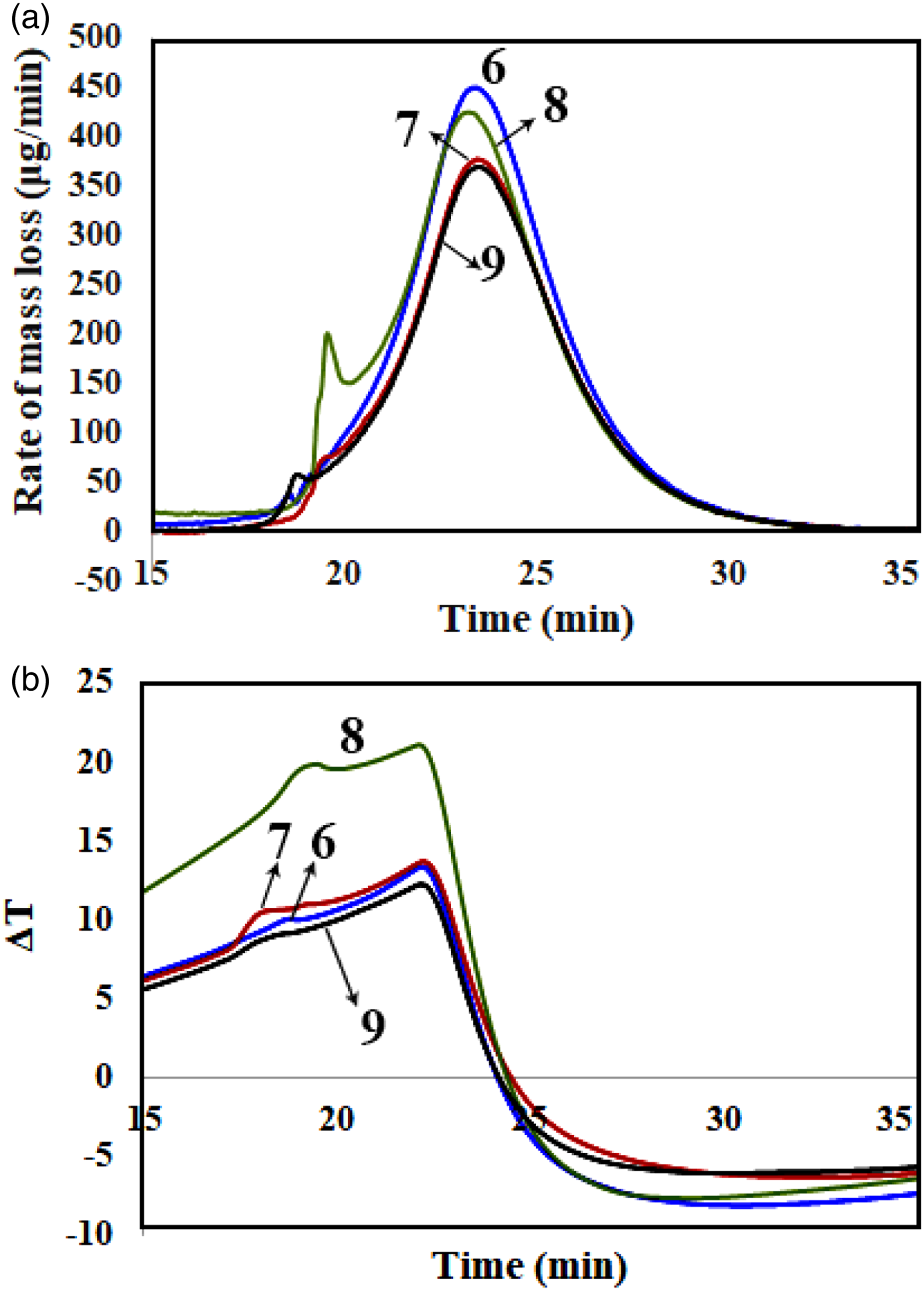
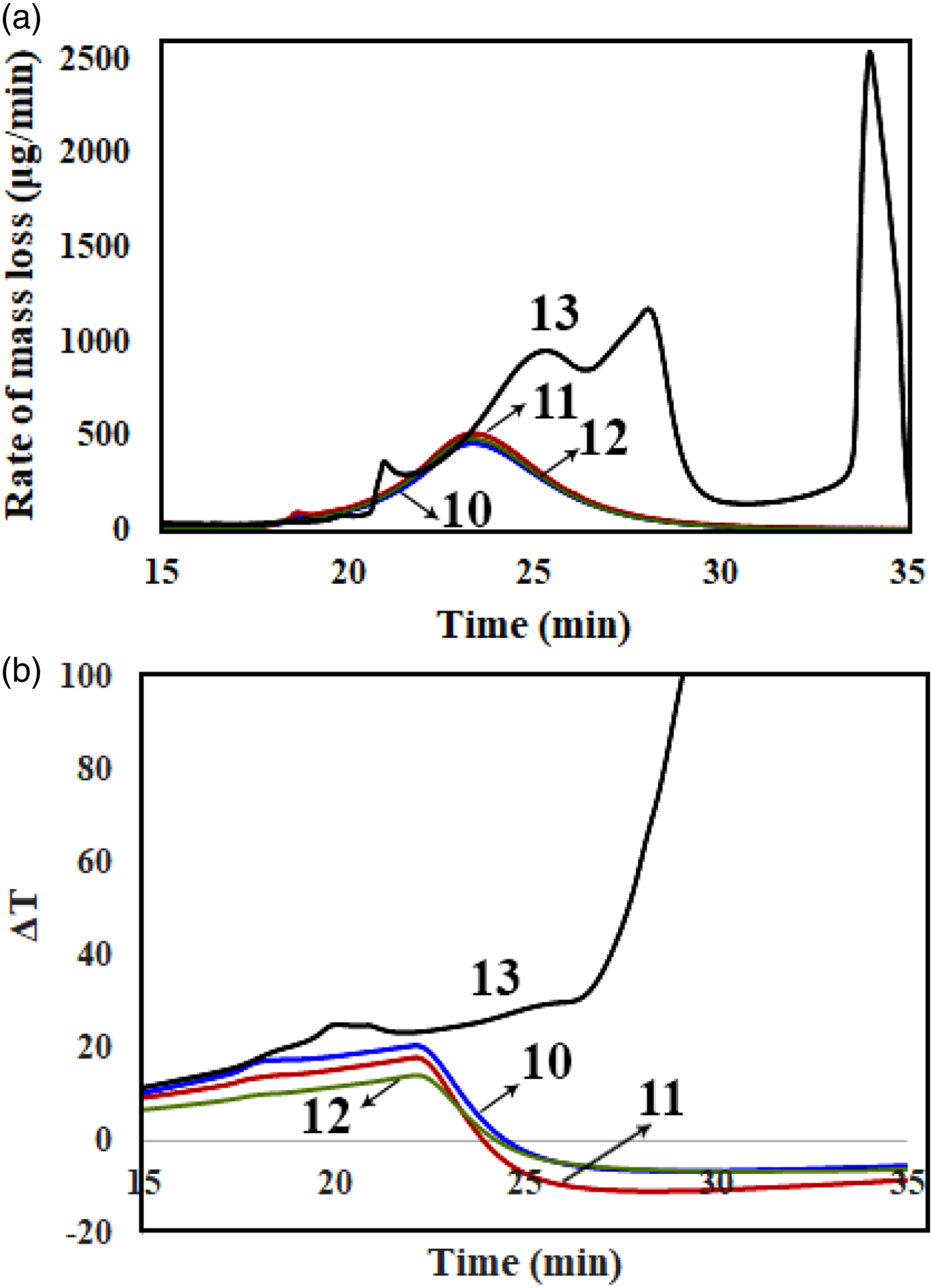
Figure 5(a) shows DTG diagram of samples no. 1–5. Tmax1 which is related to the dehydrochlorination of the PVC matrix is measured to be 265, 266, 294, 288, and 265
Figures 6(a) and (b) show DTG and DTA diagrams of the samples with 4 phr OMMT, respectively. From Figure 6(a), although Tmax1 for all samples were determined 265–266
Figures 7(a) and (b) belongs to samples containing 6 phr which shows DTG and TGA curves, respectively. In Figure 7(a), it is seen than samples 10–12 have similar thermal stability behaviors with Tmax1 of 266
Comparing thermal analyses of all samples showed that samples no. 3, 4, and 13 show better thermal stabilities among all. In order to elucidate the thermal gravimetric behavior of our best nanocomposites, TGA analysis was performed on these samples. Figure 8 shows TGA diagrams of samples no. 3, 4, and 13. As expected, the first weight loss for samples no. 3, 4 and 13 observed at 294, 288, and 292 TGA curves versus temperature for samples no. 3, 4, and 13.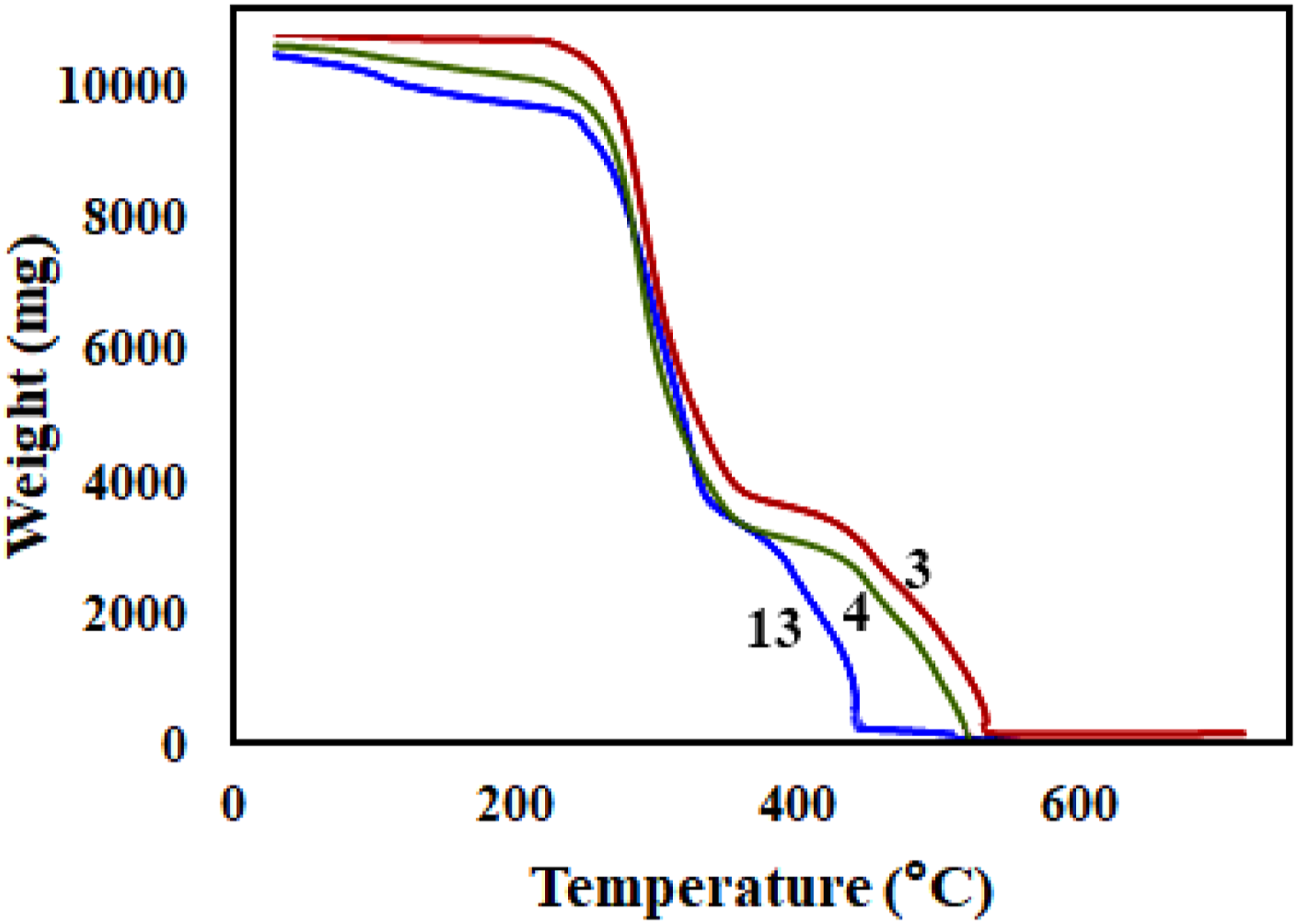
In the present study, two different binding modes of ammonium cation with ECO polymer are considered. To simplify the theoretical calculations, only two monomers of ECO were selected and the Tallow part of the ammonium cation was summarized to one methyl group. Figure 9 a and b show two different modes of hydrogen bonds with chlorine atom, and with oxygen atom of ECO, respectively. The obtained summary details of the calculations are available in Table 3. The total energy of the molecule after optimization is −1596.33 and −1596.80 Kcal.mol−1 for the molecules with Cl....H and O....H bonds, respectively. Although the molecular energies are very close, Cl....H bond is more preferred since it results in lower energy. Hydrogen bond lengths were also measured and it was shown that Cl....H and O....H bonds are 2.81 and 1.85 Å, respectively. The spatial positioning of two molecules does not allow two simultaneous bonds with one repeat unit. As mentioned before, the organoclay used in this work is modified with bis-(2-hydroxyethyl) methyl tallow alkyl ammonium cations which contain hydroxyl groups. ECO polymer also has oxygen groups in its repeating unit. According to Figure 9 b, hydrogen bonds are possible to form between polymer chains of ECO and hydroxyl groups of the modifier which can be considered as physical crosslinks. Possible molecular structures of ammonium cation in nanoclay in interaction with ECO polymer and hydrogen bond of ammonium cation with (a) chlorine atom, and (b) oxygen atom in ECO polymer. (To simplify theoretical calculations, only one carbon atom of Tallow molecule in Cloisite 30B is considered.) Data from theoretical calculation of two bonding modes of hydrogen atom from ammonium cation in nanoclay and oxygen or chlorine atoms in ECO polymer.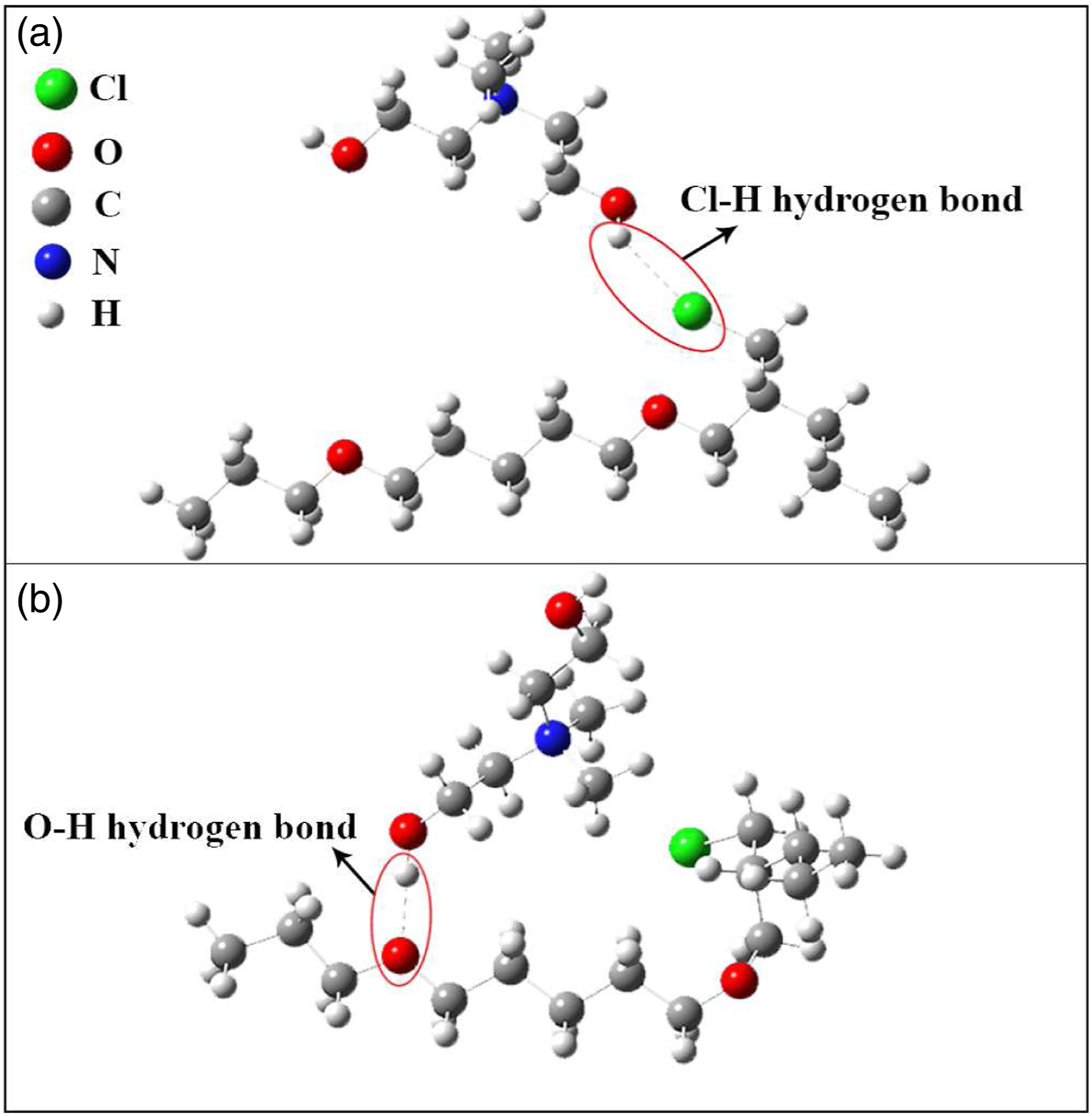
Conclusions
In conclusion, investigating the effect of ECO, OMMT, and rotor speed parameters on thermal stability of (PVC/ECO)/OMMT nanocomposites showed that rotor speeds higher than 70 r/min are more effective in intercalation of the components. DTG analyses confirmed that at lower concentrations of rubber the rate of mass loss is higher than the other samples which results in faster dehydrochlorination of the composite. The best thermal stability was observed for sample no. 3 prepared with 2 phr OMMT, 30 phr rubber, and 70 r/min rotor speed. The selected nanocomposite showed the first weight loss at 294
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
