Abstract
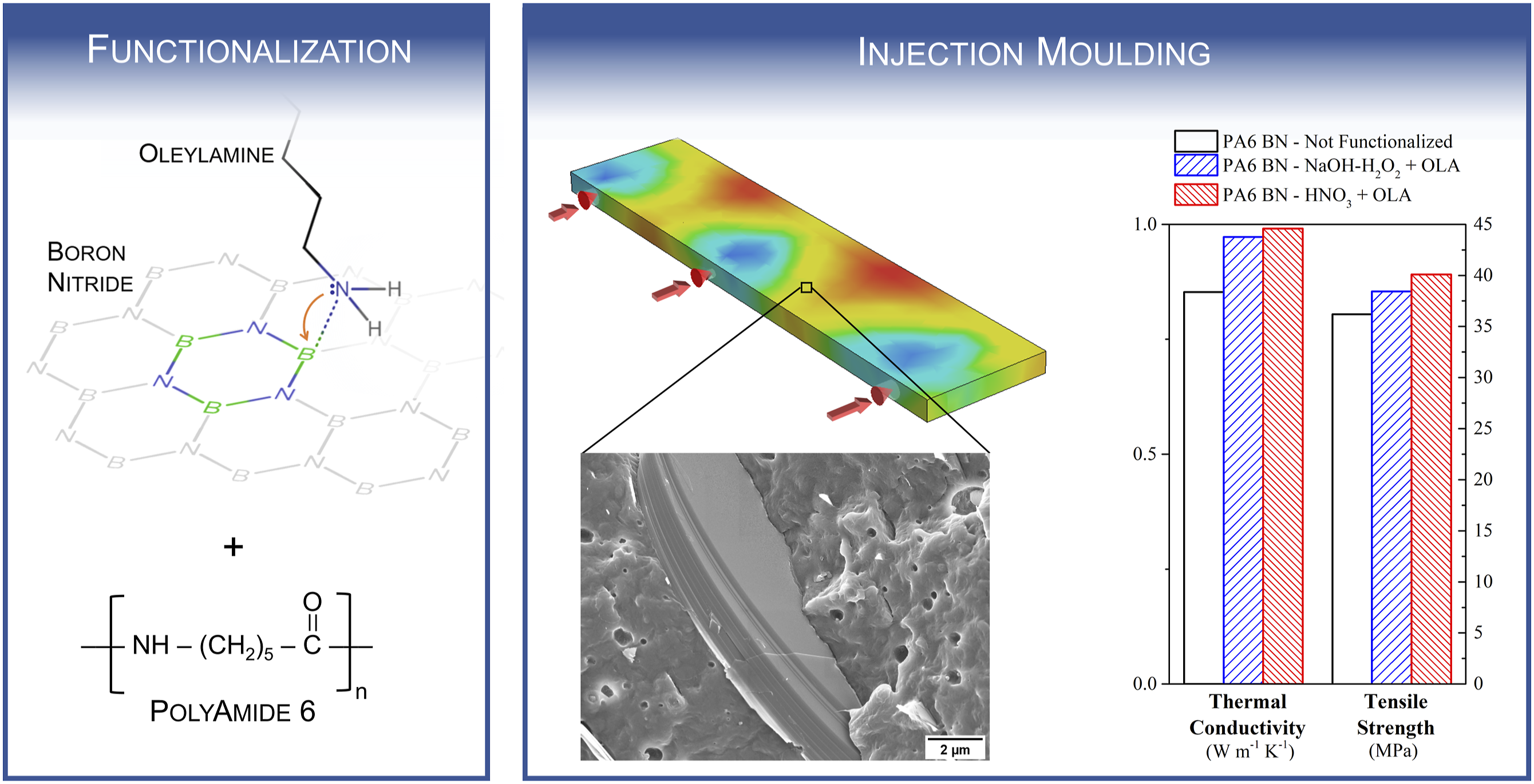
Boron nitride nano-platelets have been functionalized with oleylamine using a simple method without ultrasonication. The functionalization is effective with the formation of a coordination bonds between the boron nitride nano-platelets and the oleylamine molecules. The oleylamine functionalization allows for the production of 25% by weight loaded polyamide 6 – boron nitride composites having higher mechanical properties (15% tensile strength increase) and better through-plane thermal conductivity (0.85 to 0.99 W/m·K) with respect to the untreated samples. A better particle-matrix interaction is confirmed by SEM imaging and rheological analysis. The injection moulding process determines a filler orientation which influences the thermal conductivity.
Keywords
Introduction
Thermally conductive polymer matrix composites (PMCs) showing electrically insulating properties are innovative materials employed for thermal applications such as microelectronic packaging 1 and cooling of mechatronic components. 2 The thermal conductivity associated to electrical insulation can be achieved by adding to the polymer matrix thermally conductive ceramic fillers such as aluminum nitride, 3 aluminum oxide, 4 silicon carbide, 5 silicon nitride 6 and boron nitride. 7 Among these, hexagonal boron nitride (BN) particles seem to be one of the most promising candidate as filler for thermally conductive PMCs. BN shows a remarkable in-plane thermal conductivity (600 W m−1 K−1), 8 with low electrical conductivity and dielectric constant, high temperature resistance and low density.9,10 The resulting composite is a material where the heat conduction follows a phonon-based mechanism, 11 and the achievable thermal conductivity depends on matrix and filler thermal conductivities, as well as the amount, the geometry, the dimension and dispersion condition of the filler itself. 12 The formation of thermally conductive paths is favored by a wide particle size distribution of the fillers and, preferably, with a high aspect ratio such as to promote their orientation during production processes. In this frame, planar shape particles easily create conducting pathways for heat dissipation due to their ability to pack efficiently in the in-plane direction. 13 It was found that the thermal conductivity of BN-filled PMCs quickly decreases passing from monolayer particles (boron nitride nano-sheets, BNNSs) to multilayer particles (boron nitride nano-platelets, BNNPs), since the interlayer coupling facilitates out-of-plane vibration and thus phonon scattering. 14 In spite of the higher conductivity, BNNSs show the tendency to aggregate into nanoplatelets requiring additional exfoliating processes; hence BNNPs are preferred for industrial application.
In recent years, there has been some interest in the literature towards the use of BN, both as BNNP and exfoliated BNNS, to improve the thermal conductivity of polyamides (e.g. Polyamide-6 (PA6)) for automotive and electronics applications. Polyamide-BN composites have been prepared via extrusion mixing and then processed via traditional moulding.15–17 or additive manufacturing18,19 techniques. Despite the increase of thermal conductivity, 20 a general decrease in mechanical strength is reported due to the low chemical compatibility of the filler with the matrix. In this case, the bond between BN and matrix is insufficient and the applied mechanical stress cannot be effectively transferred from the polymer matrix to the filler, decreasing the tensile strength. 21 By improving the polymer/filler compatibility, for instance performing a surface functionalization, the PMC tensile strength increases, since stronger bonds between particles and matrix are reached. Moreover, the thermal conductivity can also increase as the phonon scattering at the interface is reduced.11,22
Generally, the boron sites in the BN lattice are activated by performing oxidation using strong oxidizer solutions (i.e nitric acid, 23 sulphuric acid, 24 sodium hydroxide 25 also combined with hydrogen peroxide23,26) in order to obtain lattice defects and/or to generate hydroxyl groups at the BN surfaces. 27 Subsequently, the defect-induced BN can be further modified using a surface coupling agent. BN has been functionalized by grafting silanizing agents23–25,27 or amine groups (i.e. ammonia plasma irradiation, 28 urea grafting, 29 covalent grafting via azide precursor, 30 and functionalization with long chain amine (ODA) 30 ). In addition, the use of natural amino acid, such as Glycine (NH2-CH2-COOH), has been investigated. 31 When employed as filler in PMCs, the functionalized BN particles have shown an increase of thermal conductivity due to enhanced interfacial interaction between filler and matrix.27,31,32 This can be explained considering that often, during functionalization, the electron-deficient boron acts as Lewis acid at defect and edge sites of the BN sheet. 33 These sites are prone to interact with electron-rich Lewis bases such as amino-terminated molecules.
Among amine-based molecules, oleylamine (OLA, C18H35NH2) seems to be a good candidate for the functionalization of boron nitride. OLA is an unsaturated fatty amine belonging to the long chain alkylamine family, showing a double bond (C=C) in the middle of the chemical structure. OLA represents an interesting option as functionalizing agent thanks to (i) its amine termination, which allows for the formation of a coordinate covalent bond with the boron atom in BN particles, (ii) its long linear carbon chain, which enables an improved interaction between BN and the hydrophobic polymer matrix, (iii) its lower commercial cost compared to other alkylamines, and (iv) the fact that it is liquid at room temperature, facilitating the functionalization and washing procedures. One very recent work deals with the OLA functionalization of BN. 34 The authors synthesized OLA-functionalized BNNS from bulk boron nitride crystals by using OLA as an exfoliating/functionalizing agent and dichlorobenzene (DCB) as solvent.
In this paper, commercially available BN nanoplatelets have been functionalized using oleylamine as coupling agent employing a simple procedure which does not involve ultrasonication, in order to produce thermoplastic-based composites. The absence of ultrasonication to exfoliate the BNNPs could represent a more easily applicable method in industrial scale polymer processing. Polyamide-6 was chosen as polymeric matrix because of its wide use in a variety of industrial applications such as automotive, 35 electronics and electrical devices 36 thanks its excellent mechanical, sliding and wear properties. The OLA-functionalized powders were then used as filler in injection moulded PA6-based thermally conductive composite samples, which have been characterized using a range of techniques.
Experimental
Boron nitride surface activation
Boron Nitride nanoplatelets HeBoFill ® 501 (BN) were provided by Henze Boron Nitride Products AG. The powder was dried in a vacuum oven at 80°C for 24 h in order to remove moisture. Two different activations to generate lattice defects on the BN surfaces were performed: the first one involving nitric acid (HNO3) while the second one involving a sodium hydroxide and hydrogen peroxide (NaOH-H2O2) solution. The nitric acid treatment was carried out by etching the BN powder in concentrated nitric acid solution (HNO 3 65% Carlo Erba Reagents) in the BN:HNO3 molar ratio 1:25. The etching was carried out in pyrex flask at a temperature of 150°C, under constant magnetic stirring for 24 h. The second etching has been performed adding the BN powder to a solution in the volume ratio 50:50 made of 40% w/v sodium hydroxide (NaOH Sigma Aldrich) and hydrogen peroxide solution (H 2 O 2 30% Sigma Aldrich). The treatment was carried out in pyrex flask at a temperature of 150°C, under constant magnetic stirring for 24 h. After treatment, both etched powders have been filtered (PTFE filter, 0.2 μm pores, Sartorius), washed with ethanol (EtOH 95% Sigma Aldrich) and dried in oven at 80°C for 24 h.
Oleylamine functionalization
The functionalization treatment was performed by adding the etched BN powders in oleylamine (Oleylamine, Sigma Aldrich) in the BN:OLA molar ratio 1:1.5. The treatment was carried out in pyrex flask at a temperature of 180°C for 24 h. Successively, the powder has been vacuum filtered (PTFE filter, 0.2 μm pores, Sartorius) and washed multiple times with excess ethanol (EtOH 95% Sigma Aldrich) on the filter. The collected powders have been dried in oven at 80°C for 24 h and stored in vacuum desiccator.
Characterizations of functionalized powders
Fourier-transform Infrared Spectroscopy (FT-IR) has been carried out on the functionalized powders using a Jasco FT/IR-6600 spectrometer, equipped with ATR PRO ONE Single-reflection accessory and diamond prism. The spectra were acquired in the range 4000-400 cm−1, with 4 cm−1 resolution averaging 32 scans for each spectrum. FT-IR spectra have been acquired on functionalized powder with different number of washings in ethanol in order to compare the washing effectiveness. Untreated BN and OLA-BN powders were analysed via thermogravimetric analysis (TGA) (Perkin Elmer Pyris 1 TGA) in the temperature range 25–700°C, setting a heating rate of 10°C min−1, under nitrogen flow. The dispersibility of BN powders in a non-polar solvent (dichloromethane, DCM, Sigma Aldrich) has been qualitatively assessed. With this aim, a suspension (1 mg/mL) was prepared by mechanical stirring at 700 r/min for 2 h and the settling rate of the particles has been visually observed up to 7 days.
Polyamide 6 BN composite compounding and injection moulding
Composition of the composite materials.
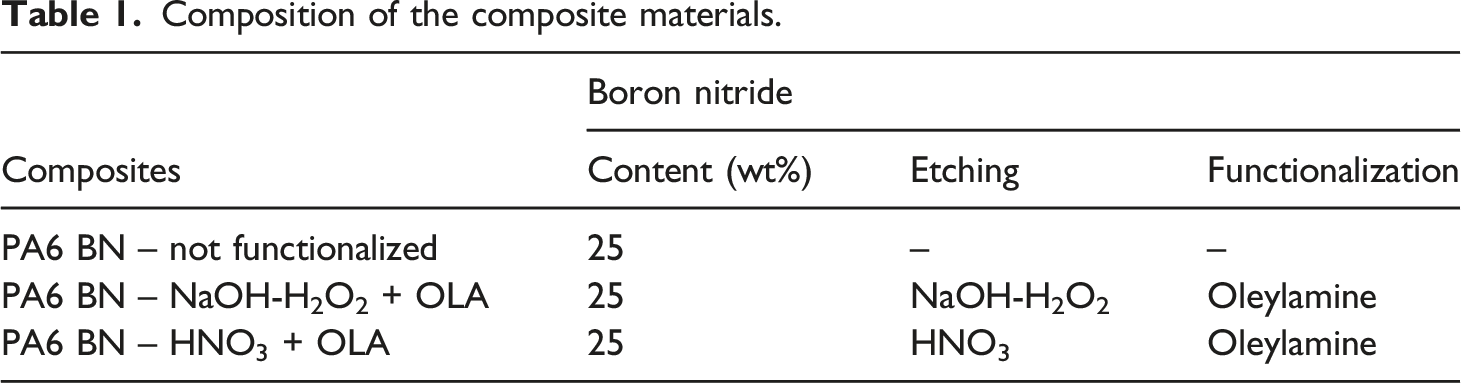
The PA6 BN pellets have been injection moulded into bar specimen with dimensions 65 × 15 × 2.5 mm3 and then milled to produce tensile specimens according to ASTM D 638 type V. The injection moulding process has been carried out using a Megatech H7/18 (Tecnica Duebi) machine with the following conditions: nozzle temperature 250°C, barrel temperature 250°C, mould temperature 40°C, clamp pressure 160 bar, injection pressure 65 bar, holding pressure 65 bar, holding time 2 s, cooling time 10 s.
Characterizations of pellets and injection moulded samples
Thermogravimetric Analysis (TGA) was carried out on injection moulded samples using a Perkin Elmer Pyris 1 TGA instrument, under nitrogen atmosphere. The measurements were performed heating the samples from room temperature (RT) to 700°C with a heating rate of 10 °C/min.
Density of the pellets have been measured via buoyancy method (density determination apparatus for analytical balance, Sartorius) in distilled water (ASTM D792 standard). Expected theoretical density have been computed via rule of mixtures according to equation (1), where 
Differential scanning calorimetry (DSC, Netzsch DSC 214 Polyma, Nitrogen flow, from 25°C to 300°C, using a heating and cooling rate of 10°C min−1) was performed on injection moulded samples. Equation (2) was used to calculate the degree of crystallinity
Results and discussion
Functionalized BN powder
The FT-IR spectra of untreated BN powders and oleylamine are reported in Figure 1(a). The untreated BN powder spectrum (black line) shows the typical peaks at 1330 cm−1 and 745 cm−1 corresponding to B-N bond vibrations.
38
The oleylamine spectrum (green line) shows several absorption bands, the most characteristic are: the peaks at 2925 and 2854 cm−1 attributable to the asymmetric stretching of C-H bonds of CH2 and terminal CH3 groups, and the peak at 3679 cm−1 due to N-H stretching.
34
FT-IR transmittance spectra of (a and b) BN powders and oleylamine; and (c) TGA curve on BN powders.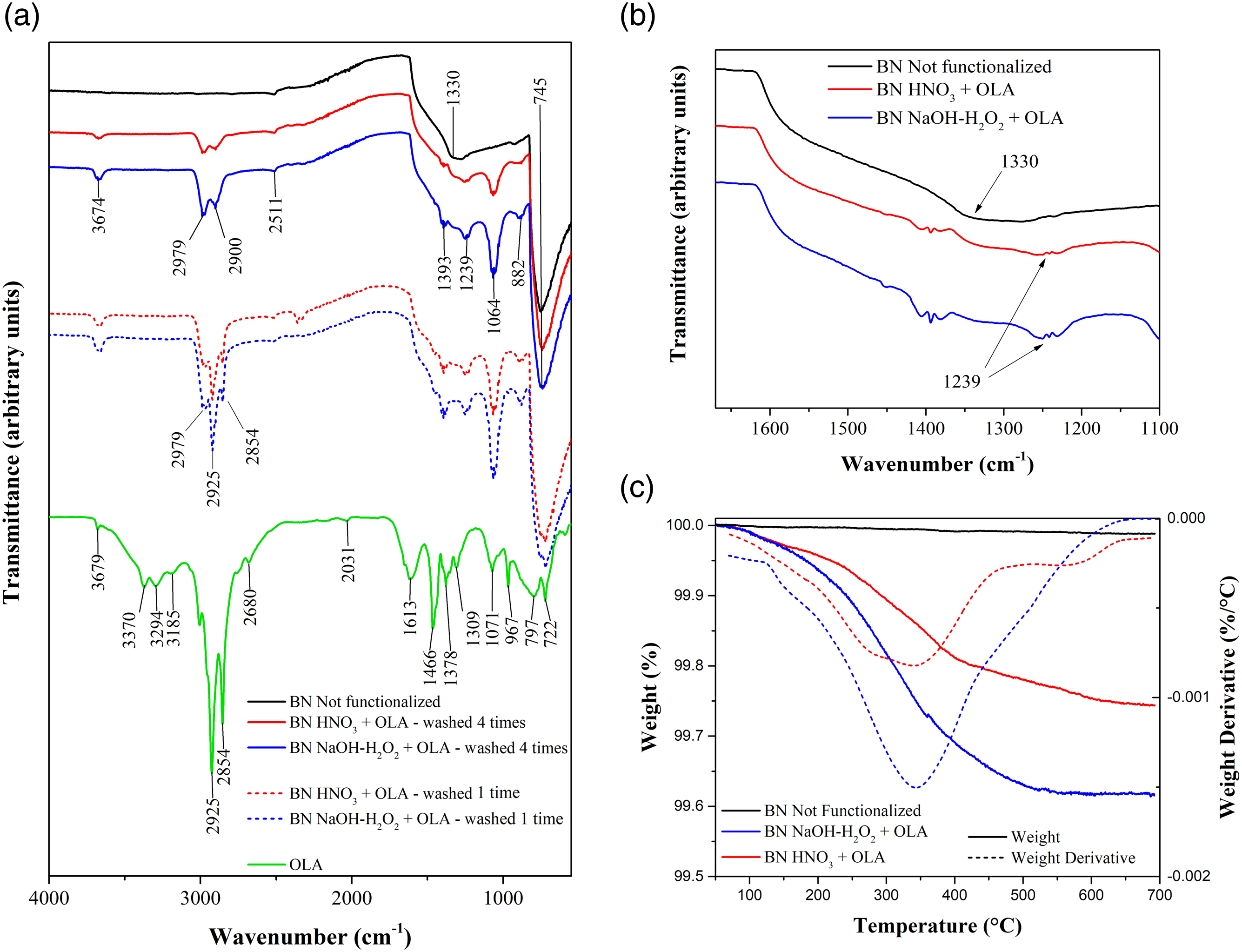
Figure 1(a) reports the FT-IR spectra of functionalized BN powders washed one and four times in ethanol. No variation in the spectra is appreciated performing further washings (spectra not reported here). The OLA-functionalized BN samples washed 4 times in ethanol show, in addition to the typical bands of boron nitride, new peaks indicating the presence of the primary fatty amine: at 3674 cm−1 (NH stretching), at 2979 and 2900 cm−1 (CH2 and CH3 stretching), all shifted with respected to the spectrum of pure OLA, suggesting an interaction between the amine and the BN particles. This interaction is further confirmed by the shift of the peak related to the stretching of B-N bonds with respect to the untreated BN powder (from 1330 to 1239 cm−1, Figure 1(b). It is well known that boron atoms in BN are Lewis acid centers (lone pair acceptors) that easily react with a Lewis base (lone pair donor), such as nitrogen atom of the amino group of OLA, forming a coordinate covalent bond (N: → B) (Figure 2(a)). Comparing the spectra of the powders washed one and four times, a significant decrease of the intensity of the peaks associated to CH2/CH3 bond vibrations of the OLA (2979, 2925 and 2854 cm−1) is observed. Moreover, the most intense peak for the powders washed one time is the peak at 2925 cm−1, as in the case of pure OLA, while with four washings the peak at 2979 cm−1 results the most intense. These results suggest that four washings are effective in removing the excess non-grafted OLA and that the remaining signals are presumably associated to the chemically bonded OLA. (a) Schematic of the OLA-BN bond; (b) Stability of the suspension in DCM of (i) BN Not functionalized, (ii) BN NaOH-H2O2 + OLA and (iii) BN HNO3 + OLA.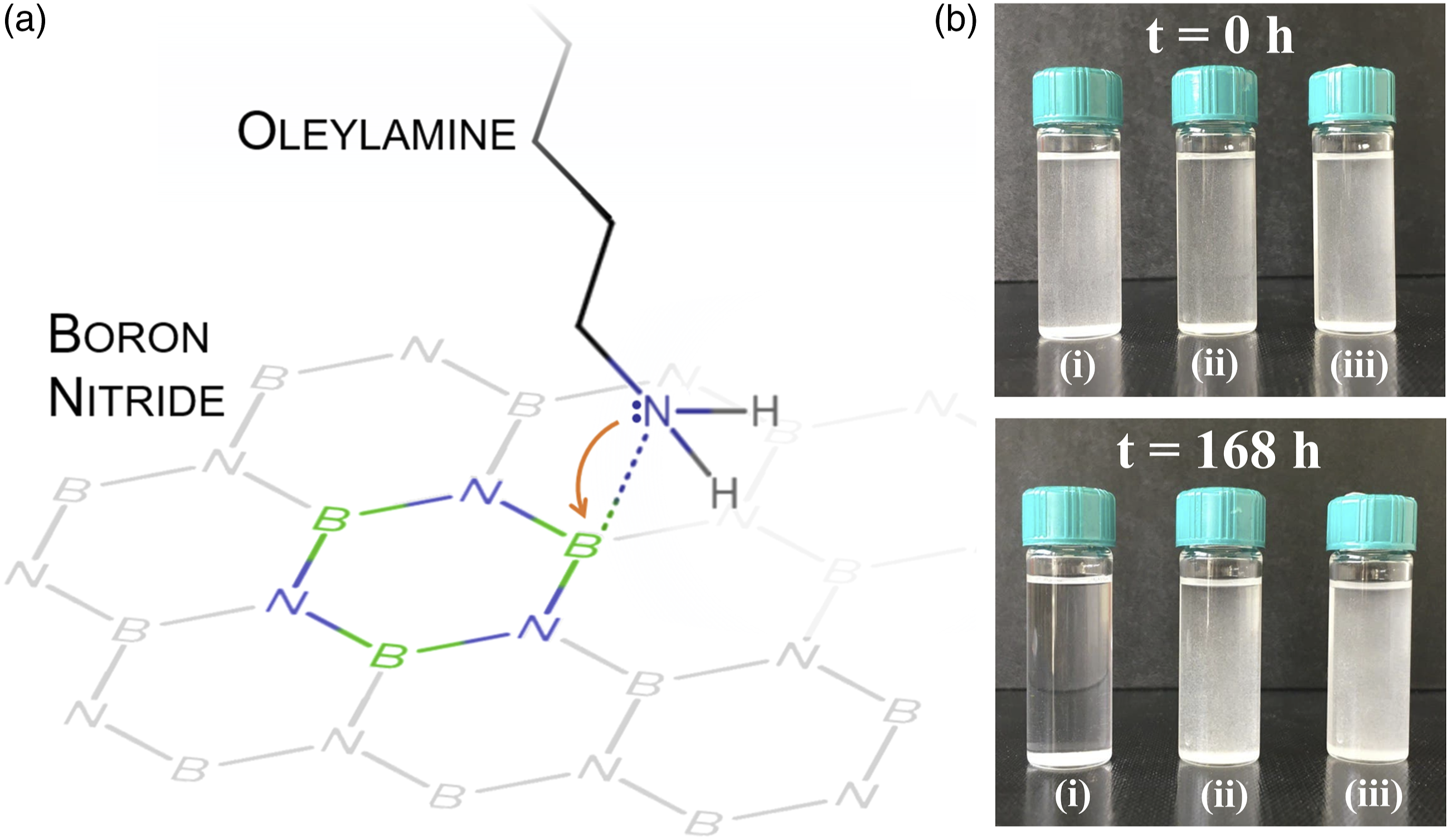
Therefore, powder washed five times have been employed in this work.
The suspension stability in the non-polar solvent (Figure 2(b)) shows that, while the untreated powder quickly sediments, the functionalized BN powders exhibit good stability over a period of several days. This effect suggests that the functionalized BNNPs should have good compatibility with the nonpolar PA6 matrix, and the OLA effect should lower the particle tendency to aggregate during the melt compounding process. 39
The thermogravimetric analysis curves are reported in Figure 1(c). As expected, the untreated BN shows no weight variation up to 700°C due to its outstanding chemical and thermal stability. On the other hand, the OLA functionalized samples show a weight loss in correspondence of 370°C, which can be ascribed to the boiling point of oleylamine as reported by the producer. The mass loss at 700°C is directly ascribable to the oleylamine content in the functionalized powders, which results to be 0.26% and 0.38% for BN HNO3 and BN NaOH-H2O2, respectively.
Thermal, rheological, mechanical, and morphological properties of BN composites
The results of thermogravimetric analysis (TGA) performed on the composite pellets are displayed in Figure 3(a) and Table 2. All the composites present, under nitrogen atmosphere, a one-step thermal decomposition with degradation temperatures, taken as the peak in weight derivative, in the range 470–480°C. The non-oxidative thermal decomposition is governed by chain scission mechanisms via several reactions,40,41 which include predominantly homolytic scission of peptide and alkylamide bonds into decomposition products which further decompose by random chain scission into lower molecular weight hydrocarbons.
42
(a) TGA; (b) rheological and (c) DSC curves 25 wt% loaded PA6-BN composite samples; (d) XRD pattern of PA6- BN non functionalized injection moulded sample. Density, TGA and MFI results for the composite pellets (with sample standard deviation).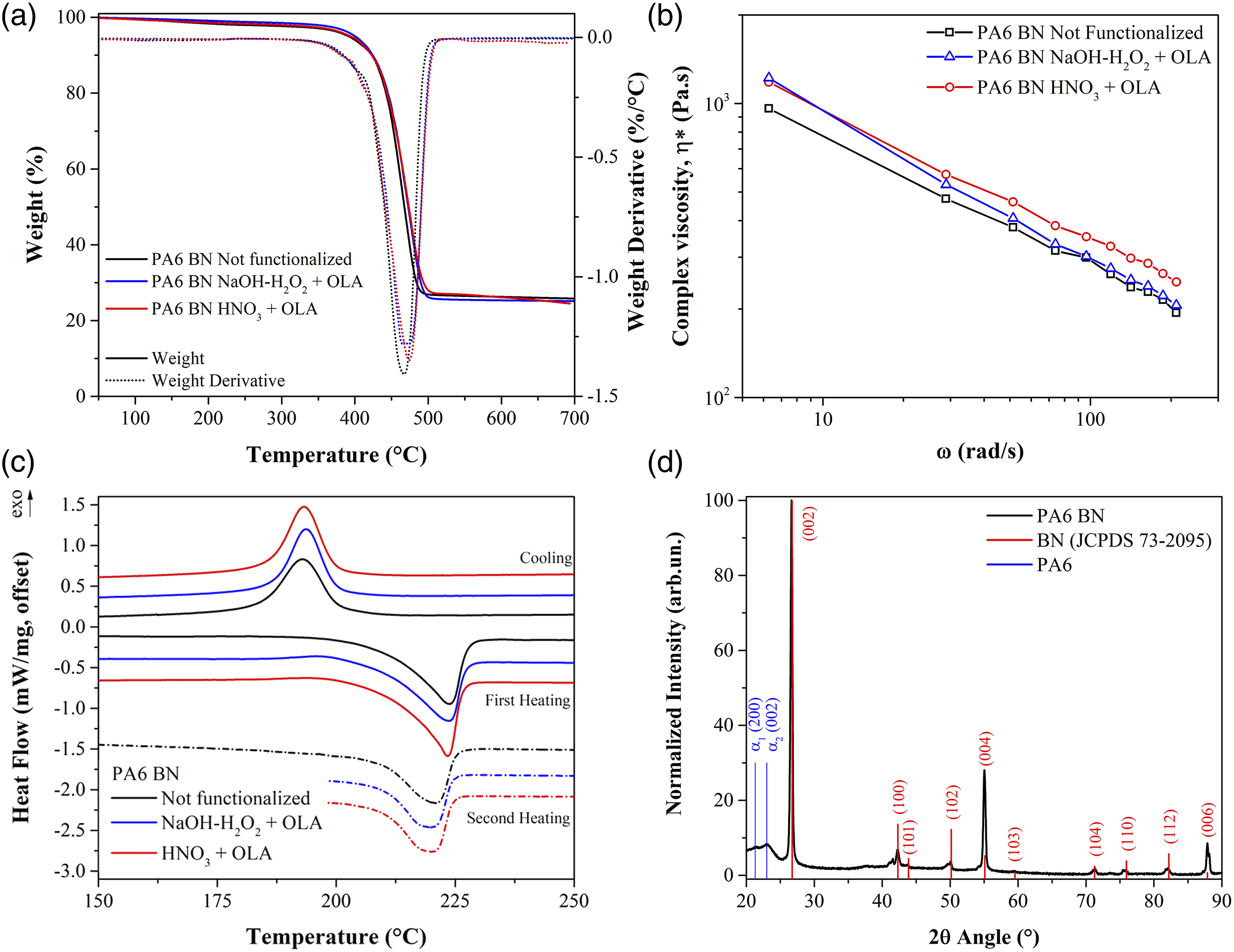
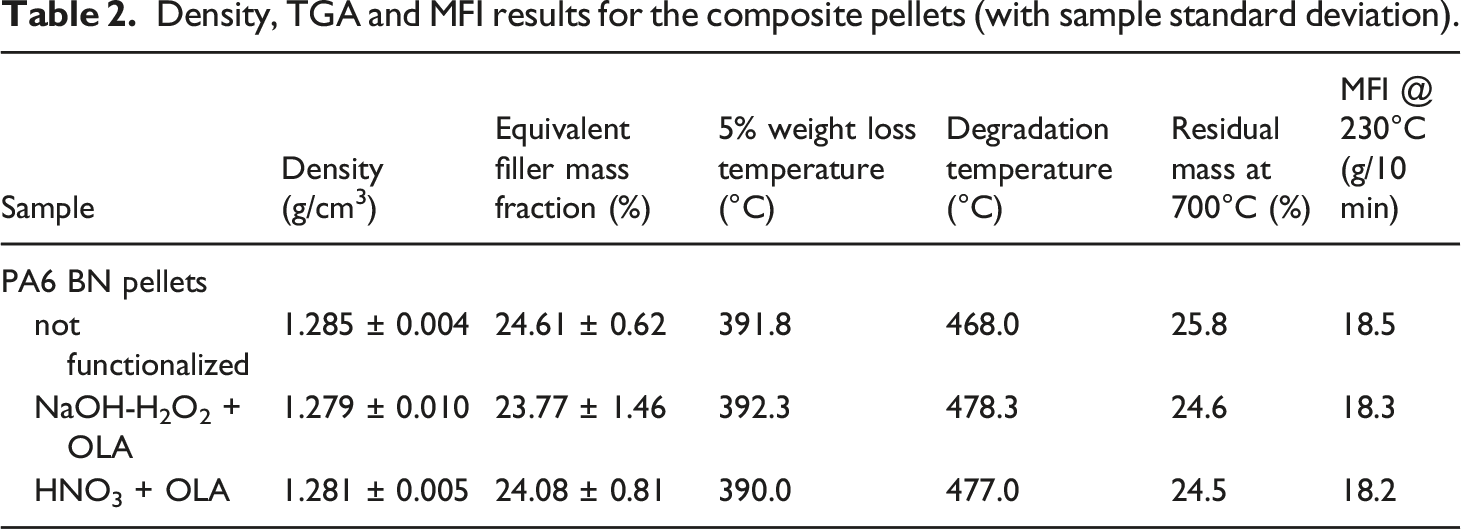
Comparing the different composite formulations, no significant variation has been recorded for the degradation temperature and the 5% weight loss temperature showing that the functionalization treatment does not alter the composite thermal stability and decomposition kinetics. The results achieved are in agreement with other research.43,44 As expected, the PA6 matrix underwent full pyrolysis, showing a residual mass of about 25% at 700°C, which matches with the weight amount of BN in the composite composition. The measured densities on the composite pellets result to be in accordance, within the sample standard deviation, with the expected value of 1.287 g/cm3, showing that a very low degree of porosity is present in the material. Moreover, the computed equivalent filler fraction is compatible with the nominal BN content and the TGA residue at 700°C.
Melt flow index values of extruded pellets are reported in Table 2 and the complex viscosities as function of frequency are displayed in Figure 3(b). The complex viscosity represents the viscoelastic resistance of the material during flow, while the melt flow index is a measure of the ease of flow of the polymer melt through a nozzle under constant pressure. All the samples show a decrease in viscosity with increasing shear rate, highlighting a shear thinning behaviour expected for particle-filled composites.45,46 This behaviour can be ascribed to the rupture of filler agglomerates and the consequent aggregate-aggregate slippage at increasing shear rates. 47 The viscosity of composites is indeed directly related to the nature, the content and the dimension of included filler, due to energy dissipation through the sliding and squeezing motion of the melt between the particles and the particle-particle interactions. 48 Moreover, the filler dispersion plays a crucial role in determining the rheological behaviour. 49 Both the composites containing functionalized boron nitride particles display a higher value of melt viscosity with respect to the not functionalized BN, in accordance with a slight decrease of MFI. It is generally reported that a higher viscosity implies a stronger adhesion between the filler and matrix. 50 These results confirm the effectiveness of the coupling agent in promoting Van der Waals bonds between the matrix and the filler, thus affecting the rheological properties as a better interfacial bonding is achieved.
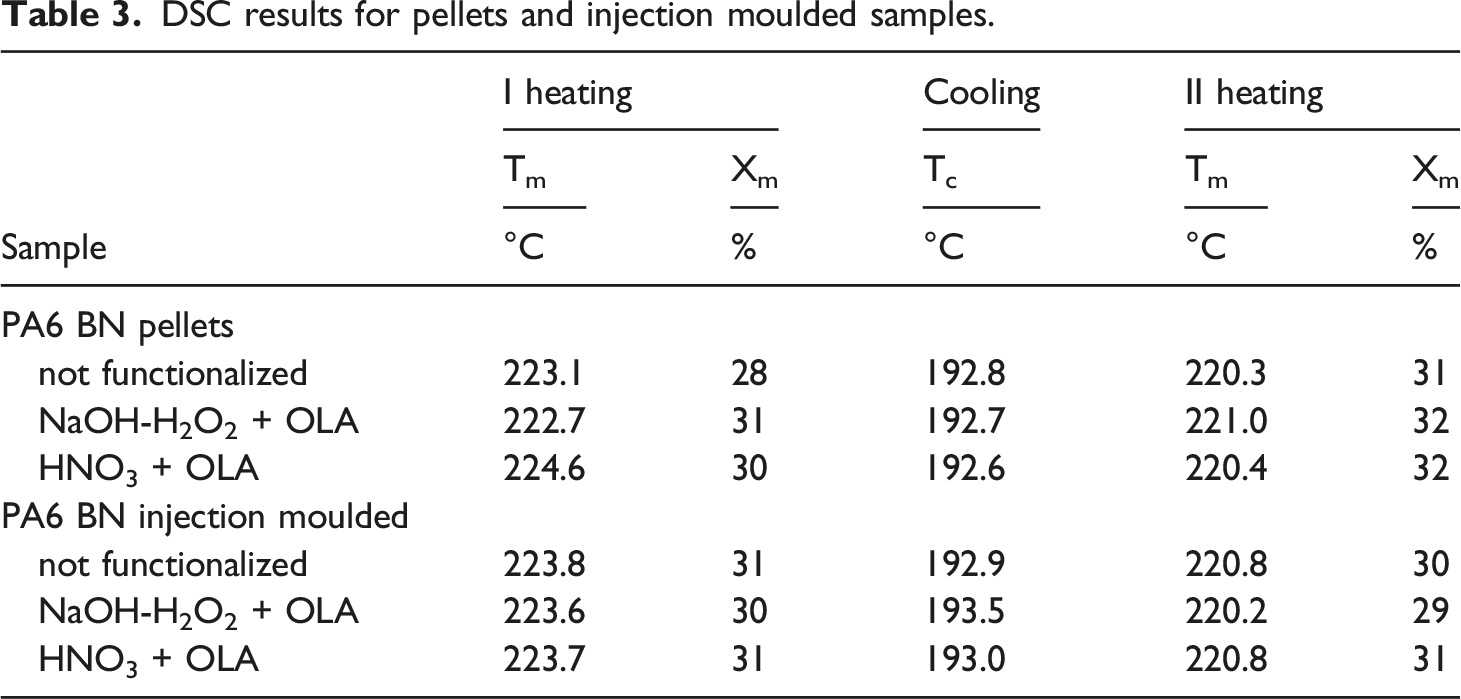
DSC results for pellets and injection moulded samples.
All the injection moulded samples show comparable XRD patterns, hence the normalized XRD pattern of injection moulded PA6-BN not functionalized is displayed in Figure 3(d) as representative. The pattern shows the presence of peaks ascribable to hexagonal BN (JCPDS 73-2095) corresponding to the Miller’s indices reported in the figure. Comparing the relative peak intensities of the injection moulded sample pattern to the BN powder with randomly oriented grains reported in the JCPDS card, a preferred orientation can be detected. Indeed, the ratio Ihkl/I100 for the (002), (004), and (006) planes for the measured pattern results in values of 14.5, 4.1 and 0.28, respectively, while the reference values, in absence of texturing, are 7.3, 0.39 and 0.06. These ratios indicate a preferred {002} crystallographic orientation, which is ascribable to the arrangement of the BN nanoplatelets during the injection moulding process, and will be later discussed in paragraph 3.3. The two broad peaks at 2θ 21° and 24° are associated with the (200) and (002) reflections of the α-phase of PA6, 55 respectively. In accordance with the DSC results, XRD analysis has not revealed the presence of γ-phase. 56
SEM micrographs of cryo-fractured injection moulded samples are reported in Figure 4. A remarkable difference can be observed comparing not functionalized and OLA-functionalized PA6 BN composites. In Figure 4(a) and (b) it is possible to appreciate how the BNNPs are not wetted by the polymeric matrix, as a gap ranging from 100 nm to few microns is clearly visible between the particle and the polymer. This detachment is ascribable to the lack of polymer-filler chemical compatibility. On the other hand, both the NaOH-H2O2 + OLA (Figure 4(c) and (d)) and HNO3 + OLA (Figure 4(e) and (f)) BNNPs result to be well wetted by the matrix with no evident detachment between the two components. This behavior is due to the enhanced chemical compatibility of the two phases which leads to stronger physical (Van der Waals) interactions. Despite the difference in the interfaces, all the composites show a good dispersion of the filler, without any extensive clusters or agglomeration of particles. SEM micrographs of PA6 BN not functionalized (a and b), NaOH-H2O2 + OLA; (c and d) and HNO3 + OLA; (e and f) cryo-fractured injection moulded samples.
The chemical compatibility between matrix and filler plays a crucial role in the mechanical behavior of a composite material. Indeed, the tensile strength heavily depends on several factors, especially particles dimensions and quality of the interface between polymer and filler. If the particles are well bonded, the mechanical load can be effectively transferred from the matrix to the reinforcing filler, leading to an increase in tensile strength.21,57 In our case, the OLA-functionalization is effective in improving the ultimate tensile strength (UTS) of the injection moulded samples, from 36.2 MPa, for the not functionalized composites, up to 40.1 MPa, with an increase of UTS of 10–15% (Figure 5(a)). The elongation at break also increases on the treated samples. It is known that the addition of micrometric particles in a polymeric matrix also entails a weakening effect due to stress concentration which could generate the fracture for lower loads and elongations, in part contrasting the strengthening effect. In this work, the fact that the latter effect prevails is again an evidence of the improved chemical compatibility of the functionalized particles. The elongation at break of the NaOH-H2O2 + OLA composites appears to be slightly increased with respect to HNO3 + OLA, although with an overlap in error bars indicating almost no statistical significance. However, this effect could be ascribed to the higher content of OLA grafted onto the NaOH-H2O2 BN particles, as measured from the TGA analysis, which can allow the polymer chain segments to move and rotate more freely. On the other hand, the elastic modulus is the same for all composites, with a value of 2.847 ± 0.055 GPa, which is higher than neat PA6 found in literature.
18
This result is expected from the composite theory since a rigid ceramic filler is added in the thermoplastic matrix. (a) Mechanical properties and (b) DMA curves for the PA6 BN injection moulded samples.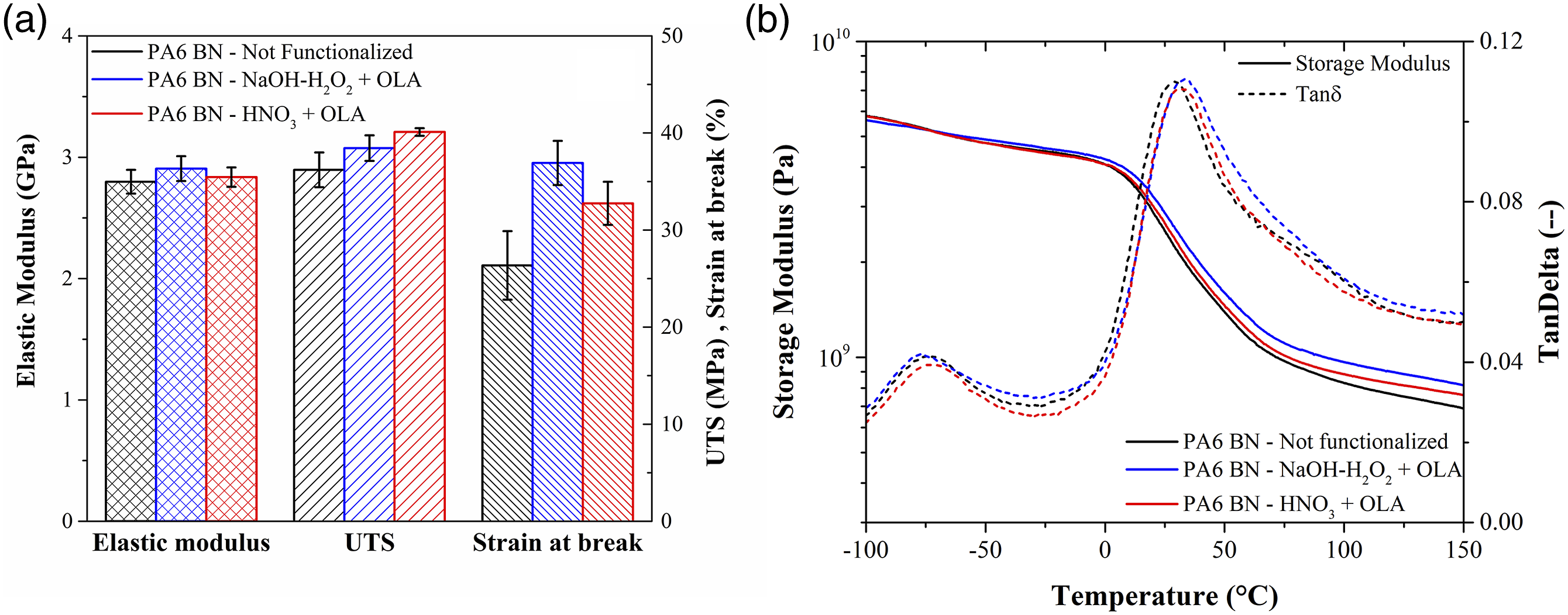
DMA measurements on the composite specimens (Figure 5(b)) show comparable storage modulus (E’) values for all the samples, with an E’ of 2.67 ± 0.19 GPa at room temperature. All investigated materials show two tanδ peaks. The first one, at about −75°C, is related to a β relaxation, which is generally associated with the motion of small portions of the backbone chain, vibration of 1 or 2 monomeric units, fluctuation holes formation, and polymeric chain rearrangements. 58 In the case of PA6, the β relaxation is related to the coordinated motion or rotation of chain portions on the order of the Kuhn length, such as CH2 or amide groups. 59 The second peak, instead, represents the α transition and it is associated to the glass transition of the polymer, related to segmental motion of the polymer chain. The glass transition temperature (Tg) is almost unchanged among the three samples, with a value of about 30°C.
Thermal conductivity of injection moulded samples
Thermal conductivity of injection moulded samples.

Literature comparison between achieved thermal conductivity in PA6-based composites with BN particles.

BN = Boron Nitride, AlN = Aluminium Nitride, IM = injection moulded, CM = compression moulded, FFF = Fused Filament Fabrication.
It is known that the thermal conductivity of a polymer, governed by phonon propagation, is heavily influenced by the presence of discontinuity in the microstructures. The phonon propagation is more efficient in ordered and defect-free systems, while presents scattering at any discontinuity such as the particle-matrix interface or the amorphous phase in semi-crystalline polymers. 12 The BNNP particles functionalization acts in reducing the filler-matrix interface and therefore leads to a decrease in resistance to the phonon propagation60,63,64 resulting in an improved thermal conductivity (from 0.85 to 0.97–0.99 Wm−1K−1) corresponding to a 14–16% increase. These values are compatible to other works where functionalized BN (even with exfoliation process) have been employed in a similar content (i.e. 10–30 wt%) as conductive filler in several polymeric matrices refs. 27, 65–67 showing an increase in the range 13–19% with respect to the non-functionalized composite.
Even though the functionalization successfully improves the composite thermal conductivity, all the compositions (not functionalized BN included) show a slightly lower conductivity in comparison with previous works on PA6 with the same filler content of pristine BN processed by compression moulding.
18
This effect can be ascribed to the anisotropy of the nanoplatelets within the polymeric matrix. A SEM micrograph on a cryo-fractured bar sample (Figure 6(a)), indeed, clearly shows how the BN nanoplatelets present different orientation through the sample thickness. The particles are oriented in a parabolic profile in the centre of the sample (Figure 6(b), particles coloured in yellow), while the top and bottom surfaces present a layer (about 300 μm in thickness) with BNNPs oriented parallelly to the outside surface (Figure 6(c)). Since the Hot Disk measurement involves a heat wave travelling from the top surface to the bottom surface of the sample, the observed morphology results in the thermal conductivity being measured mostly across through-plane oriented particles. Hexagonal BN conductivity is, indeed, highly anisotropic due to its chemical bonds, with values of about 600 Wm−1 K−1 in the in-plane (002) direction and only 1.5–2.5 W m−1 K−1 in the through-plane (100) direction. Therefore, a reduction in thermal properties for an oriented composite is expected when compared to a isotropic system, as also found in literature.20,67,68 The composite morphology, and especially the presence of the parallelly oriented surface layers, is also responsible for the {002} texturing, as previously discussed for the XRD patterns. SEM micrograph of the cross-section of a cryo-fractured injection moulded sample (a), with details (b and c) and drawing; (d) showing the BNNPs orientation.
The orientation of anisotropic particles in a polymer flow is known to be governed by the rheological behaviour of the melt, mould cavity geometry and injection parameters. During the injection three flow regimes are usually recognised: (i) a radial diverging flow near the injection points, (ii) a shear-governed flow through the cavity thickness, (iii) fountain flow at the fluid front.
69
Within the second and third regimes, the prevalent flow phenomena that determine particles orientation are, respectively, the high shearing across the cavity thickness, and the in-plane deformation at fluid front.
70
The mould geometry in the present work is expected to present no in-plane deformation, and thus the shear flow regime is the governing factor in the BNNPs orientation. The particles therefore align parabolically across the sample thickness, with a frozen layer at the mould walls where they are aligned along the boundaries following the flow direction, as confirmed both numerically and experimentally in literature.
71
Figure 7 shows a qualitative flow profile of the polymer melt (in terms of fill time, defined as the time after the injection start in which the flow front reaches each point). The colour map in the 3D view illustrates the radial diverging flow from the injection points, while in the cross-section the flow proceeds with a parabolic front (white curves), explaining the filler texturing imaged by SEM analysis. Qualitative polymer injection schematic in terms of fill time, showing the polymer flow in the cross-section.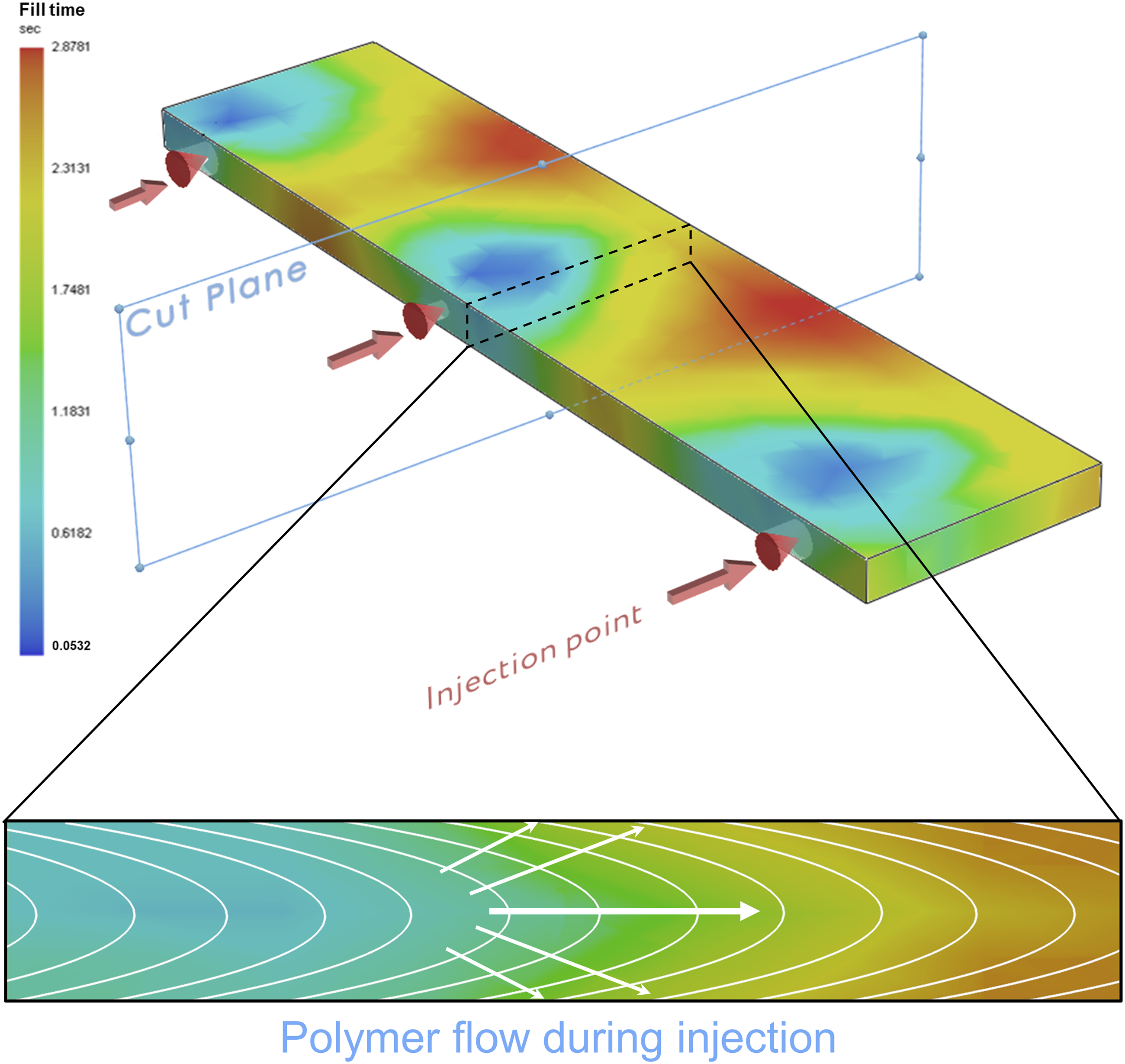
Conclusions
In this paper BN nanoplatelets have been functionalized using oleylamine to produce PA6 BN thermally conductive composites via injection moulding. FT-IR and TGA analysis showed the successful grafting of OLA onto the BN surface regardless of the chemical etching performed prior to the functionalization. The produced PA6 functionalized composites display higher melt viscosity and improved mechanical strength, both ascribable to better interaction between particles and matrix thanks to the coupling agent presence. The SEM microstructural characterization confirmed the improved wettability and adhesion of the polymer onto the BNNPs, as well as particle orientation due the injection moulding process. Finally, the thermal conductivity of functionalized PA6 BN composites is higher due to reduced phonon scattering as consequence of better chemical compatibility.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
