Abstract
The increased demand for the discovery of new conducting materials encourages the researchers for the development of advanced electronic technologies. In the present study, nanocomposites of poly (anthranilic acid-co-indole)
Keywords
Introduction
Conductive copolymer nanocomposites are an emerging area of research for the development of engineered composite materials that often exhibit high thermal stability, optical, electrical, magnetic properties and mechanical performance enhancement as compared to their non-hybrid counterparts.1–3 The mechanical properties of copolymer composites depend on the selective incorporation of nanoparticles into the copolymer matrix. The filler-polymer interactions have a major role in controlling the surface chemistry of the polymer composites and the relative size of the nanoparticles control the entropic interactions in the host polymeric domains. 4 Novel strategies are important for the preparation of novel conducting polymer nanocomposites at room temperature and are highly desirable to obtain conjugated polymeric systems for various applications. Two major synthetic strategies have been used for incorporating inorganic nanoparticles into copolymeric systems to develop copolymer-based nanocomposites. The first approach involves the grafting of polymeric substituents on the surface of nanoparticles which can change the surface chemistry of the polymer composites. 5 However, the self-orientation of preformed nanoparticles and the incompatibility between the copolymer segment and nanofiller lead to the agglomeration of nanoparticles in the copolymer. The second method involves the in-situ synthesis of nanoparticles within the copolymer domain where the synergistic interaction between the monomer and nanoparticles is maximum and therefore, the problem of aggregation of nanofiller in the polymer can be avoided by the in-situ polymerization method. 6
Magnetite (Fe3O4) is one of the most extensively studied nanomaterials due to its higher chemical stability, biocompatibility, high dielectric constant, magnetic susceptibility, high saturation magnetisation, easy to synthesis and inexpensive.7–10 The distinctive electrical and magnetic properties of Fe3O4 are due to the transfer of ions from Fe2+ ions to Fe3+ ions. In search of advanced conjugated conducting polymeric materials, copolymerization of indole with anthranilic acid monomers in the presence of Fe3O4 nanoparticles exhibits interesting properties which have not been much explored earlier. With the proper selection of a suitable conjugative polymer matrix, Fe3O4 nanoparticles can be integrated to form nanocomposites which result in higher mechanical strength, electrical conductivity, magnetic and dielectric properties. There are reports on the studies of magnetic and electrical properties of conductive polymer composites based heterocyclic groups containing polymers.11–15
Heterocyclic conjugated polymers having a nitrogen moiety have interesting properties because of their easy synthesis compared to other heteroatoms containing polymers. Polyindole (PIN) shows poor solubility, thermal and mechanical properties among the conducting polymers. 16 The demand for replacing polyaniline has increased due to its brittleness, poor processability and insolubility. These problems can be greatly overcome by the use of poly (anthranilic acid) (PANA) because of its easy synthesis and the reactive groups of carboxyl moiety in the polymer backbone helps the formation of intermolecular interaction with the polar conducting polymers like PIN.17,18 The copolymerization of different conducting monomers provides a significant route to altering the thermal, electrical, physical and chemical properties of the resultant copolymer.19–21 So, the present work focused on the synthesis of copolymer based on anthranilic acid and indole (poly (anthranilic acid-co-indole)) by a chemical oxidative polymerization technique. Herein, we report the formation of nanostructured conductive copolymer composites based on anthranilic acid and indole by incorporating magnetite nanoparticles within the polar segments of the copolymer using a simple in situ polymerization method for enhancing the magnetic, electrical and thermal properties of the poly (anthranilic acid-co-indole). The structural, morphological, and thermal properties of poly (anthranilic acid-co-indole)/Fe3O4 nanocomposites have been analysed by FTIR, UV, XRD, SEM, TEM, Differential scanning calorimetry (DSC) and TGA. The magnetic properties of the prepared composite were analysed by vibrating sample magnetometer (VSM) measurement. Finally, both AC and DC conductivity and dielectric properties of the copolymer nanocomposites as a function of various loadings of nanoparticles were also explored.
Experimental
Materials and methods
Indole (Fluka) was purified by double distillation under reduced pressure. Sodium dodecyl sulfate (SDS), ammonium persulfate (APS), sulfuric acid (H2SO4), anthranilic acid and methanol were procured from Merck (India). Double distilled water was used as a solvent for all the synthesis.
Synthesis of Magnetite Nanoparticles (Fe3O4)
The Fe3O4 nanoparticles were synthesised by a simple co-precipitation method. 22 Ferric chloride and ferrous chloride (1: 2 M ratio) were dissolved in de-ionized water in a reaction vessel and stirred for 30 min in an inert condition. Ammonia was added to this solution and regulated the PH up to 11. The whole solution was stirred for 6 h. at 50 0C. The precipitate formed was centrifuged at 3000 r/min. It was then washed with water, ethanol and dried to get a constant weight.
Synthesis of poly (anthranilic acid-co-indole) (PANA-Co-PIN)
The poly (anthranilic acid-co- indole) was prepared by the oxidative polymerization of indole and anthranilic acid with APS as an oxidant in an acid aqueous medium. Appropriate amounts of indole and anthranilic acid monomers were dissolved in 0.5
Synthesis of Poly (anthranilic acid-co-indole)/magnetite nanocomposites
Poly (anthranilic acid-co- indole)/magnetite nanocomposites were synthesized by in-situ polymerization of indole and anthranilic acid in an aqueous acidic solution containing different contents of Fe3O4 nanoparticles using APS. Magnetite nanoparticles (5, 10, 15 and 20 wt. %) were dispersed with SDS in distilled water. This solution was then added to the solution of indole and anthranilic acid dissolved in 0.5
Characterisation techniques
The UV-VIS absorption spectra of copolymer Fe3O4 nanocomposites in dimethyl sulfoxide were recorded on Hitachi 91-300 spectrophotometers. A Fourier transform infrared spectrometer (FT-IR series model JASCO 4100) was used for structural analysis of copolymer/metal oxide nanocomposites at a scanning rate of 20 and 4 cm−1 resolution from 400-4000 cm−1. X-Ray diffractometer (Bruker AXS, XRD) was employed to analyse the distribution of nanoparticles in the copolymer nanocomposites at a scanning rate of 1°/min. FE-SEM (Carl Zeiss, VP500, field emission scanning electron microscopy (SEM)) was used to study the morphology of fabricated composites. A high-resolution transmission electron microscopy (TEM) (JEM-2100F, HR-TEM, JEOL Co., Japan) was employed to analyse the size, shape and structure of copolymer nanocomposites. The samples for High-resolution transmission electron microscopy (HRTEM) analysis were obtained using the droplet of poly (anthranilic acid-co-indole)/Fe3O4 nanocomposites in solution on the surface of carbon-coated copper grid and then air dried for 2h. A vibrating sample magnetometer (VSM model LDJ 9600) was used to study the magnetic properties of the copolymer with different contents of Fe3O4 nanoparticles. The thermal decomposition pattern of copolymer/metal oxide nanocomposites was carried out in a Hitachi STA7200 thermogravimetric analyser at a heating rate of 10 °C per minute in an inert atmosphere. Shimadzu DSC-50 instrument was employed to find out the glass transition temperature of the materials at an optimum heating rate of 10 °C per minute. Electrical conductivity and dielectric properties of the composites were determined with the Hioki impedance analyzer (HIOKI 3570 model) over a frequency range window of 102–106 Hz at room temperature. The gas sensing characteristics of copolymer with Fe3O4 composites were measured by passing ammonia gas at room temperature. The pelletized composites were inserted into the glass chamber for passing ammonia gas. The ammonia gas (100 ppm) was passed into the glass chamber and the resistance of composites was measured as the sensitivity of the polymer. The ammonia gas was removed from the glass chamber for each cycle and the sensor was placed in atmospheric air for recovery. The sensor response is defined as: Response (%) = [(Rg - Ra)/Ra] ×100, where Ra is the sensor resistance tested in air, and Rg is in the target gas.
Results and discussion
UV spectroscopy
The UV spectra of PANA-Co-PIN and their nanocomposites with Fe3O4 particles are provided in Figure 1. The UV spectrum of copolymer exhibits a weak reflection at 329 nm and a strong peak at 359 nm is the π-π* and n-π* transitions respectively. The UV spectrum of magnetite nanoparticles filled copolymer follows a different pattern as compared to that of PANA-Co-PIN. The prominent peak of the copolymer is slightly shifted in copolymer nanocomposite with an additional UV peak at 380 nm indicating the attachment of magnetite in the copolymer segments. It can be seen from the figure that the shift in the UV peak of nanocomposite increases with the loading of Fe3O4 nanoparticles up to 10 wt.% (from 359 nm to 332 nm) and this shift indicates that the resultant electronic changes are associated with the strong interaction between Fe3O4 and the copolymer. When the loading of filler in the copolymer increased beyond 10 wt. %, the absorption maxima are slightly shifted and this is due to the formation of aggregated structure in the copolymer. It can be suggested that the change in intensity of this electronic transition is associated with the formation of polarons and the generation of polarons depends on the content of Fe3O4 nanofillers. UV spectra of copolymer with different contents of magnetite nanoparticles.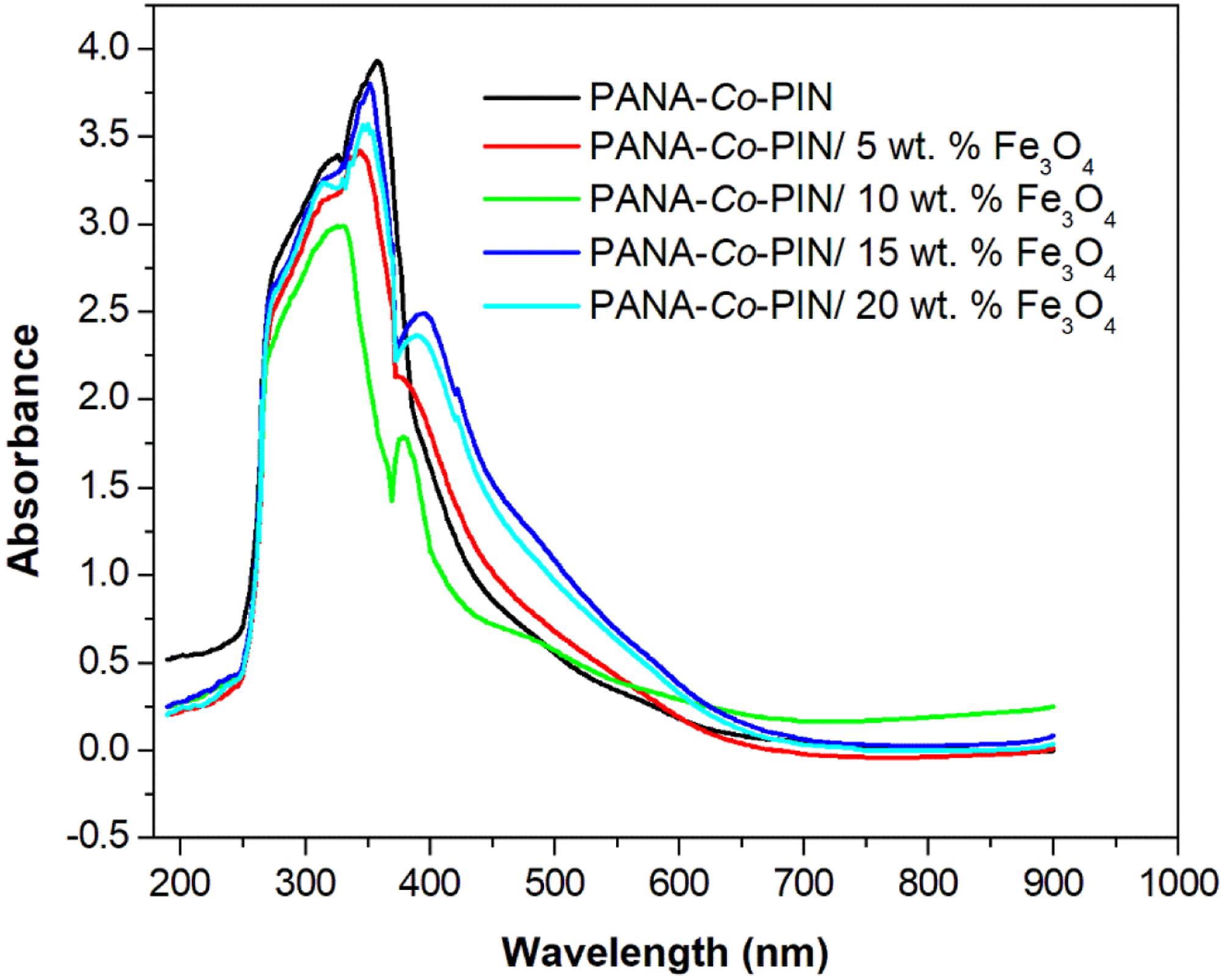
Fourier transform infrared spectroscopy
Figure 2 shows the FTIR spectra of Fe3O4, synthesised copolymer and its composites with different contents of Fe3O4 nanoparticles. The IR spectrum of nanoparticles shows a sharp absorption band at 577 cm−1 is the characteristic Fe–O bond in Fe3O4. The broad peak centered at 3417 cm−1 is the OH and Fe-OOH on the surface of nanoparticles.
22
The main absorption peak of the FTIR spectrum for the copolymer is observed in the broad range of 3400 to 2800 cm−1 bands which are attributed to the NH and O-H stretching of the copolymer.17,19 A sharp band around 1696 cm−1 corresponds to the strong stretching vibration of carbonyl group present in the poly (anthranilic acid). The absorption bands at 1308 and 1204 cm−1 are the CN and CH stretching vibrations of aromatic rings in the copolymer. Another prominent band at 1452 cm−1 indicates C-N stretching of indole ring in the copolymer. Similarly, the band at 1582 cm−1 shows the C = C stretching frequencies of the quinoid ring in the copolymer. These aforementioned inferences confirm the effective copolymerization of anthranilic acid and indole monomers. It is evident from the copolymer nanocomposites that the main absorption peak magnetite in the PANA-co-PIN is clearly visible at 589 cm−1 which is attributed to the Fe-O stretching of the nanofiller.
23
In comparison of the FT-IR spectra of PANA-co-PIN/Fe3O4 nanocomposites with that of the copolymer, the broadened region (3400–2800 cm−1) is greatly reduced by the insertion of Fe3O4 nanoparticles into the copolymer provides another proof for the formation of copolymer nanocomposites. In addition to this, the carbonyl group in the copolymer is slightly moved to a higher wavenumber i.e., from1696 cm−1 to 1705 cm−1. This indicates the strong intermolecular interaction between the polar segments of Fe3O4 with the polar groups of the copolymer and the schematic representation for the preparation and mechanism of copolymer nanocomposites is given in Figure 3. FTIR spectra of Fe3O4, PANA-Co-PIN and its composite with Fe3O4. Schematic representation for the preparation and mechanism of PANA-Co-PIN/Fe3O4 nanocomposites.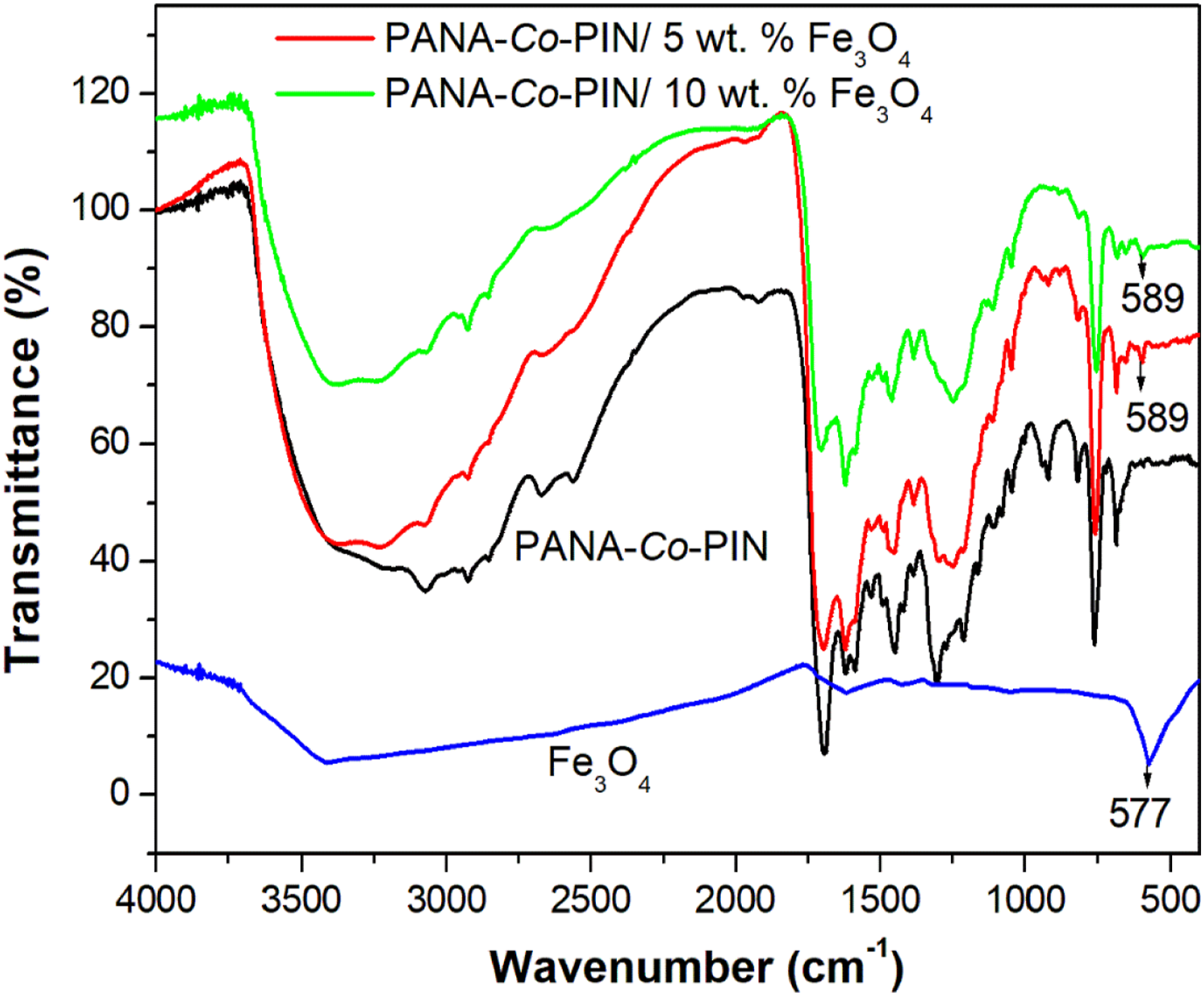
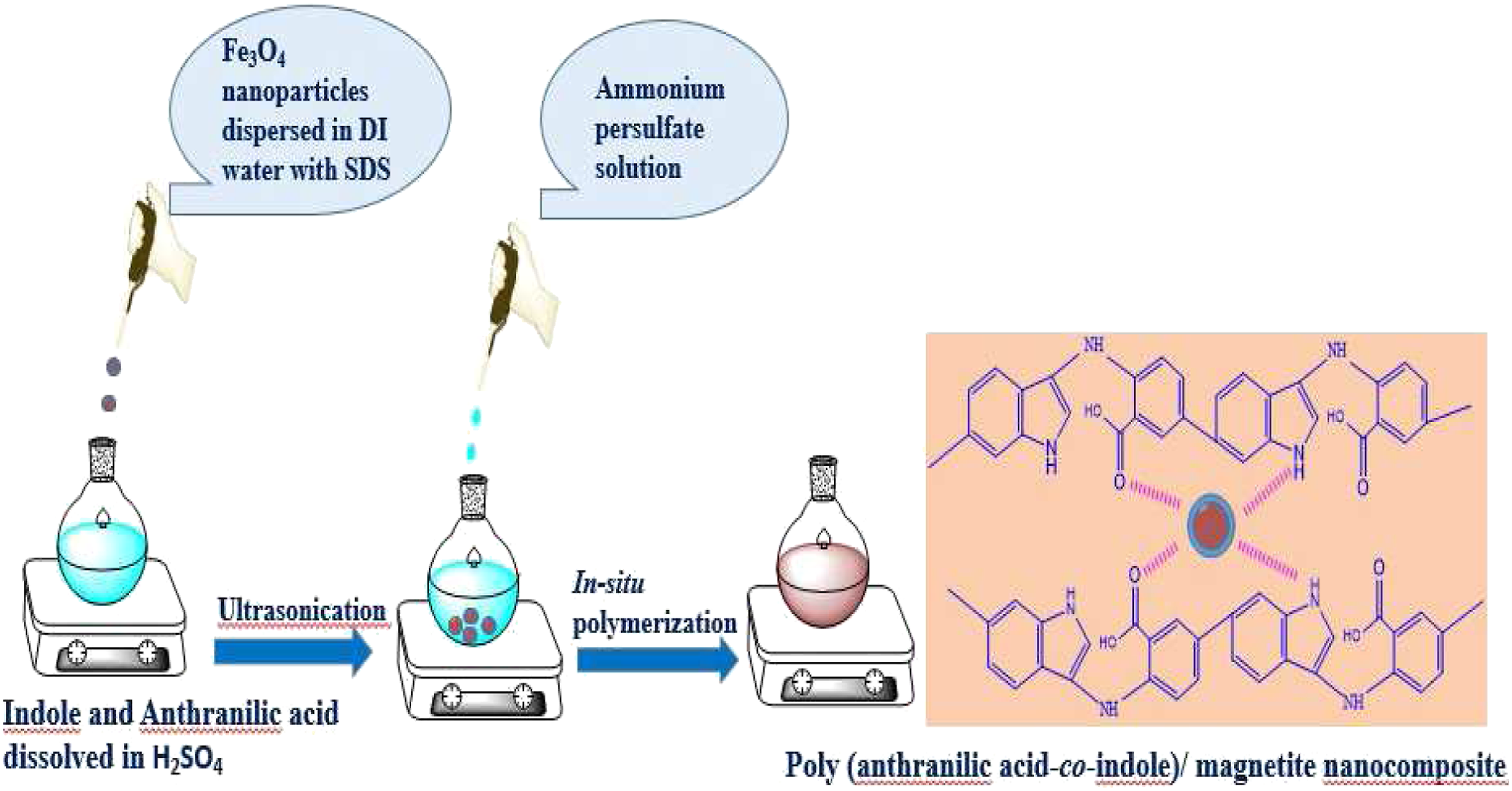
X-Ray diffraction pattern
The chemical identity and the arrangement of nanoparticles in the copolymer have been investigated with XRD analysis and the XRD result of PANA-Co-PIN and PANA-Co-PIN with different contents of Fe3O4 nanofillers are illustrated in Figure 4. The XRD profile of PANA-Co-PIN exhibits a broad amorphous peak at 2θ = 19.14° and a weak peak at 26.47° which suggests that the indole and anthranilic acid monomers are homogeneously incorporated in the network structure of the copolymer. From the XRD profile of Fe3O4 incorporated copolymer nanocomposites, it is evident that the amorphous region of the copolymer is slightly moved from 2θ = 19.14° to 19.77° and the existence of the crystalline peaks of nanoparticles are visible at 2θ = 36.25°, 42.11°, 57. 24° and 62. 66°. It has been observed that the intensity of crystalline peaks in the copolymer increases with the increase of loading of Fe3O4, which is a clear indication of the increase in crystallinity with increasing content of nanoparticles. This observation indicates an increase in long-range order of copolymer with the encapsulation of nanofillers.
24
The enhancement in crystallinity can be attributed to the strong chemical interaction between the copolymer and magnetite nanofillers. XRD of copolymer with different contents of magnetite nanoparticles.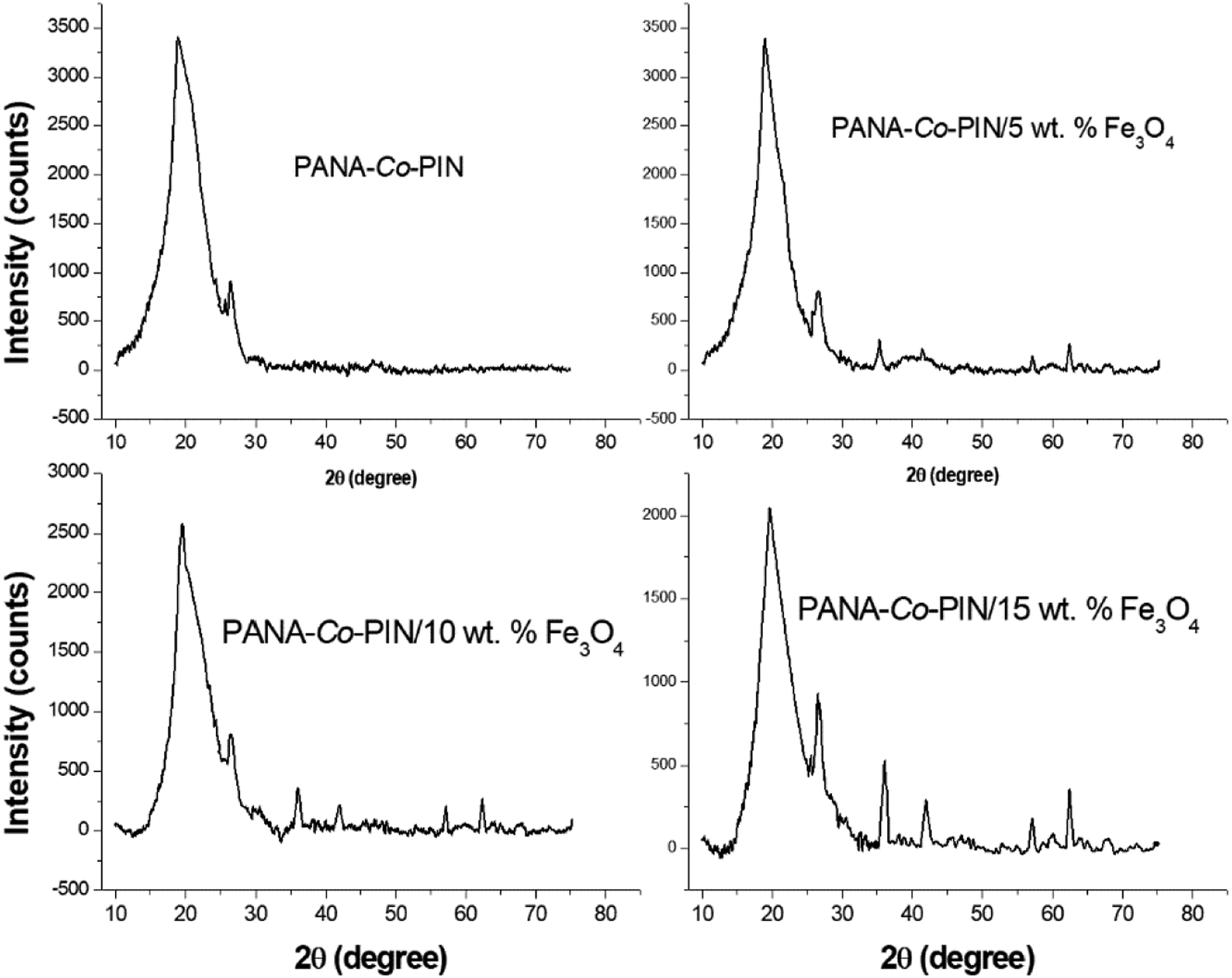
Scanning electron microscopy
The FESEM micrographs of Fe3O4 incorporated PANA-Co-PIN of nanocomposite is depicted in Figure 5 represents the distribution of nanoparticles in the copolymer matrix. It is clear that the structure of the pure copolymer is irregular with a rugged-flaky morphology. The rugs and flakes on the surface of copolymer has entirely changed into a uniform morphology with the incorporation of nanofillers. From these figures, it is also observed that the uniform dispersion of Fe3O4 in the copolymer is found to be increased with the loading of Fe3O4 nanoparticles. The copolymer nanocomposite of Fe3O4 at 10 wt. % in PANA-Co-PIN shows more compact and uniform morphology with spherically shaped particles than the other composites with higher concentrations of Fe3O4. This means that the intermolecular interaction between the copolymer and nanoparticles is maximum at this loading during the in-situ polymerization. However, at higher loading of fillers, the surface morphology of the copolymer composite changed into an irregular and non-homogenous structure, which arises from the weakened interfacial interaction that leads to the agglomeration of Fe3O4 in the copolymer. Thus, FESEM images confirm the reinforcement of Fe3O4 within the copolymer matrix and validate the results obtained from UV spectroscopy and XRD analysis. SEM images of (a) copolymer (b) 5 wt. % (c) 10 wt. % and (d) 15 wt. % magnetite incorporated copolymer.
Transmission electron microscopy
The high resolution of TEM examination gives a clear idea about the specific details of nanostructured materials. HR-TEM images of PANA-Co-PIN/Fe3O4 nanocomposites containing 7 and 15 wt. % of magnetite is shown in Figure 6. The TEM image of 7 wt. % composite (Figure 6 (a)) revealed that the nanofillers are well spread in the copolymer with spherically shaped particles having a size ranging from 15-30 nm. The homogeneously dispersed nanoparticles in PANA-Co-PIN nanocomposite are due to the strong binding force from the strong interactions between Fe3O4 and copolymer. As the content of magnetite particles within the copolymer matrix increased to 15 wt. %, the aggregation of Fe3O4 is visible. This is due to the poor interaction of nanoparticles with the copolymer segments. TEM images of copolymer with (a) 7 wt. % and (b) 15wt.% magnetite.
Differential scanning calorimetry
Differential scanning calorimetry is used to study the glass transition temperature of Fe3O4 incorporated PANA-Co-PIN and the corresponding thermograms of the copolymer nanocomposites are displayed in Figure 7. The endothermic dip observed at 104. 4 °C indicates the glass transition temperature (Tg) and another weak endothermic peak at 239 °C is the melting temperature (Tm) of pristine PANA-Co-PIN. The copolymer with 5, 10 and 15 wt. % composites showed the Tg at 114.25 °C, 121.35 °C and 130.21 °C respectively. The increase in glass transition temperature of the copolymer nanocomposites with the increase in loading of Fe3O4 is due to the compact network structure developed in the copolymer matrix. This indicates that the segmental mobility of the copolymer chain decreases with an increase in the loading of filler in the copolymer. Similarly, the Tm values of the copolymer are found at 247 °C, 256 °C and 266 °C respectively for 5, 10 and 15 wt. % composites. As the amount of nanoparticles in the copolymer increases, the melting temperature of the copolymer nanocomposite improved indicating the obstruction of the copolymer mobility and rotation of the macromolecular chains. The proper insertion of Fe3O4 in PANA-Co-PIN resulted in a broadening of the endothermic peak indicating that the addition of nanoparticles boosts the crystallinity of copolymer nanocomposite. DSC of copolymer with different contents of magnetite.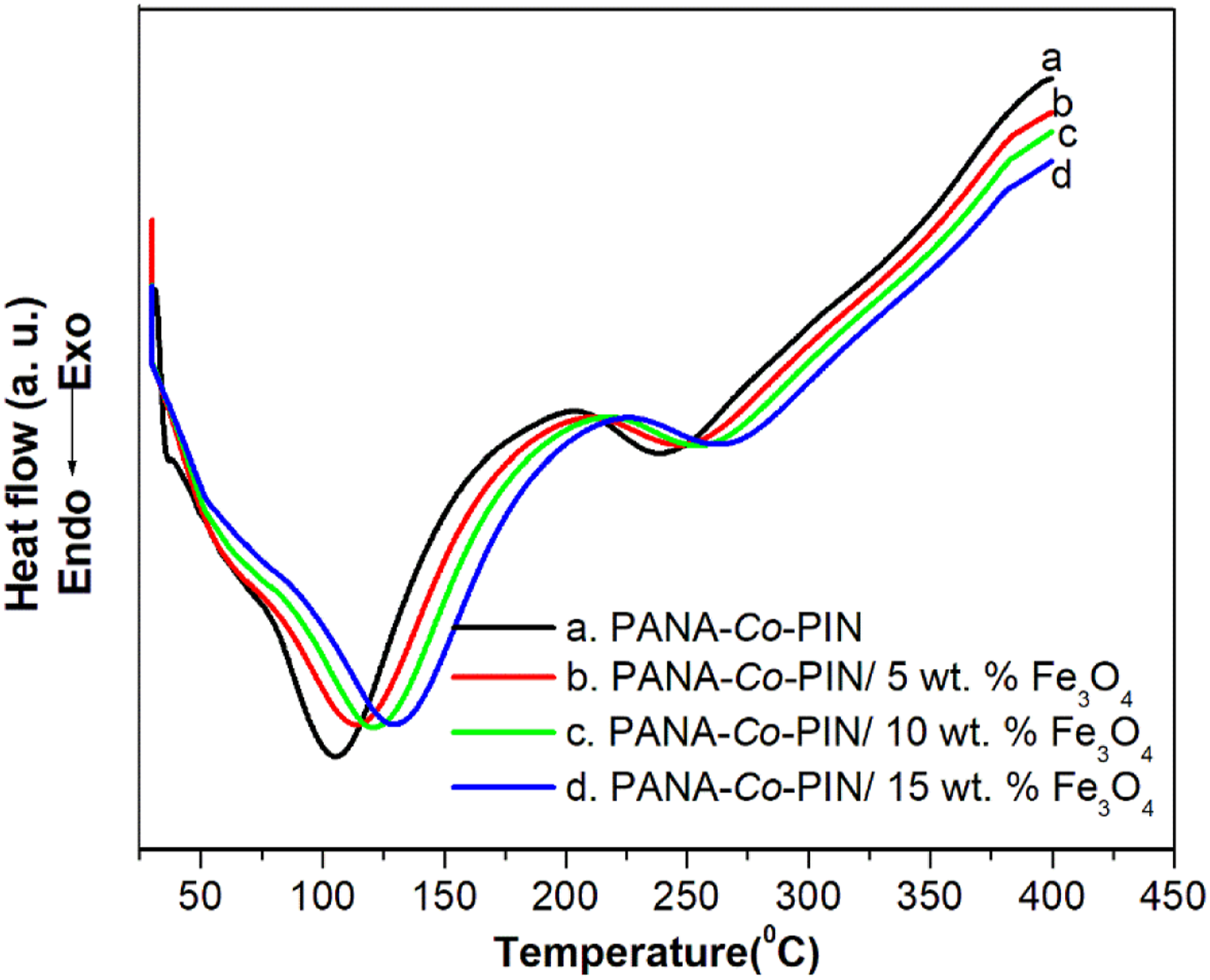
Thermogravimetric Analysis
The thermal stability of the synthesized copolymer nanocomposites is investigated by Thermogravimetric Analysis (TGA) and the thermograms of the nanocomposites are illustrated in Figure 8. It can be evident from the TGA curve that the thermal decomposition copolymer is greatly enhanced by the addition of nanofiller in the current system. The TGA profile of all samples shows two recognizable weight loss. The initial thermal decomposition temperatures for all the samples are found between 80 °C and 205 °C and this is the vaporization of water, dopant ions and solvents entrapped within the samples. The second and major weight loss of pure copolymer is observed at 244 °C is attributed to the decomposition of bond scission in the backbone of the copolymer. The copolymer nanocomposites with 5, 10 and 15 wt. % sample shows the major decomposition temperature at 268 °C, 279 °C and 297 °C respectively. The increased thermal stability of the copolymer nanocomposites denotes the strong interaction between filler particles and the copolymer matrix. The final char residue remaining for pure copolymer at 700°C is 28.8% while the composites with 5, 10 and 15 wt. % sample is increased to 33.11, 36.96 and 44.18% respectively. The metal oxide nanoparticles present in the copolymer reduced the formation of carbonaceous char layers and thereby preventing the thermal decomposition process.
25
In other words, the magnetite nanoparticles restrict the mobility of the copolymeric chains and delay the heat transfer and thereby increasing the fire resistance nature of the composite materials. TGA of copolymer with different contents of magnetite.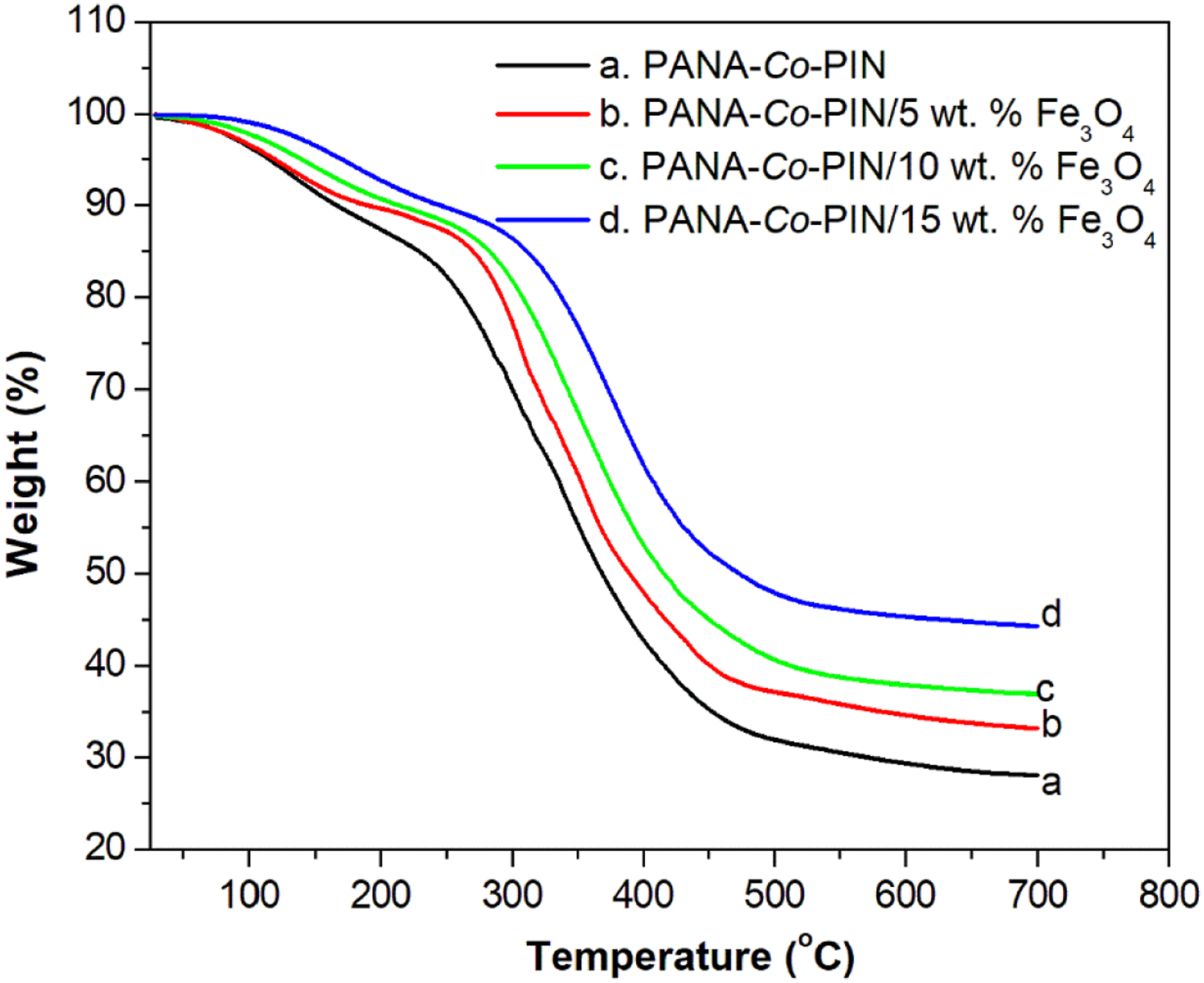
Vibrating Sample Magnetometer (VSM)
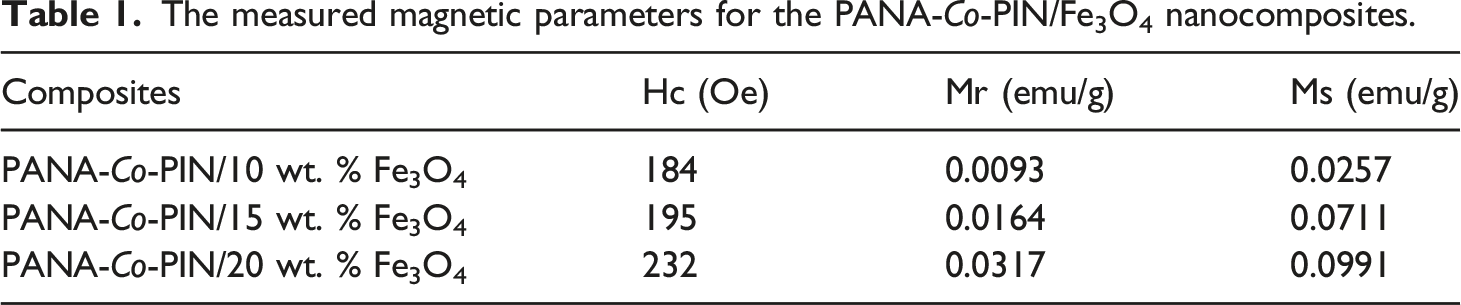
The magnetization curves of PANA-Co-PIN nanocomposites with magnetite concentrations of 10, 15 and 20 wt.% loadings are shown in Figure 9 and the magnetic properties obtained from these curves are summarized in Table 1. It can be seen that the remnant magnetization (Mr) increases with the addition of Fe3O4 nanoparticles. Interparticle interaction developed in the copolymer matrix is responsible for the remnant magnetization. The coercivity (Hc) and saturation magnetization (Ms) are clearly visible in all the magnetic hysteresis curves, which indicates their superparamagnetic behaviour. It can be seen from the curves that the ferromagnetic property is considerably enhanced in which the saturation magnetization value jumped from 0.025 to 0.099 emu/g. This implies that increasing the magnetite nanoparticles creates ion vacancies in the copolymer and thereby boosts the ferromagnetic ordering in the polymer matrix.
26
Homogenous reinforcement of magnetic Fe3O4 nanofillers over the non-magnetic copolymer matrix can be confirmed from the hysteresis loop. The change in magnetic properties is attributed to the dipolar nature and the magnetic interactions between the adjacent nanoparticles in the copolymer. Corroborating the electrical, dielectric and magnetic properties, devices with excellent EMI shielding properties can be developed from these soft magnetic materials. VSM characteristics of magnetite and magnetite incorporated copolymer. The measured magnetic parameters for the PANA-Co-PIN/Fe3O4 nanocomposites.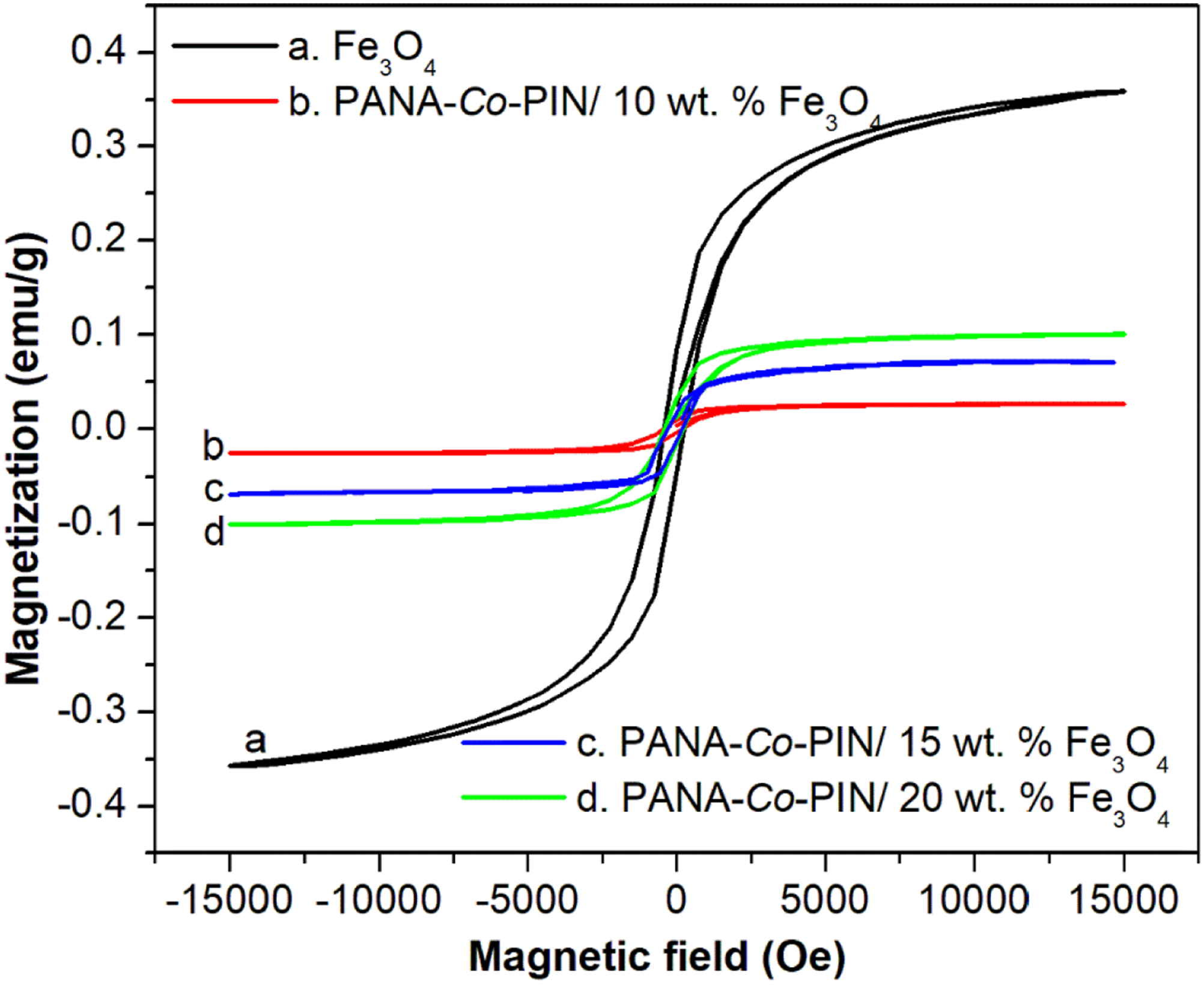
Dielectric constant
Figure 10 shows the frequency dependence of relative permittivity or dielectric constant of PANA-Co-PIN with different contents of magnetite nanocomposites over a broad frequency range of 100 Hz to 106 Hz. The dielectric value of magnetite particles filled copolymer is greater than the pure copolymer which enables them for capacitive energy device fabrication The dielectric constant is found to decrease exponentially with a rise in applied frequency. The large value of dielectric constant at lower frequency originates from the well-known Maxwell–Wagnor-Sillar interfacial polarization effect.
27
The combined effect of interfacial polarization and space charge polarization operates in the lower frequency region due to its long relaxation period. The outstanding increase in the dielectric performance of the copolymer nanocomposite can be ascribed to the polarizable nature of nanoparticles. The polarizable functionalities within the copolymer matrix respond quickly to the applied electric field.
28
Among the nanocomposites, the maximum dielectric constant value of 15 wt.% sample indicates the higher dispersion of Fe3O4 nanofillers into the copolymer segments. A decrease in dielectric performance is observed beyond 15wt. % composite is due to the non-uniform coverage of Fe3O4 at higher loading results in hindering the polarization phenomena. Dielectric constant of magnetite incorporated copolymer.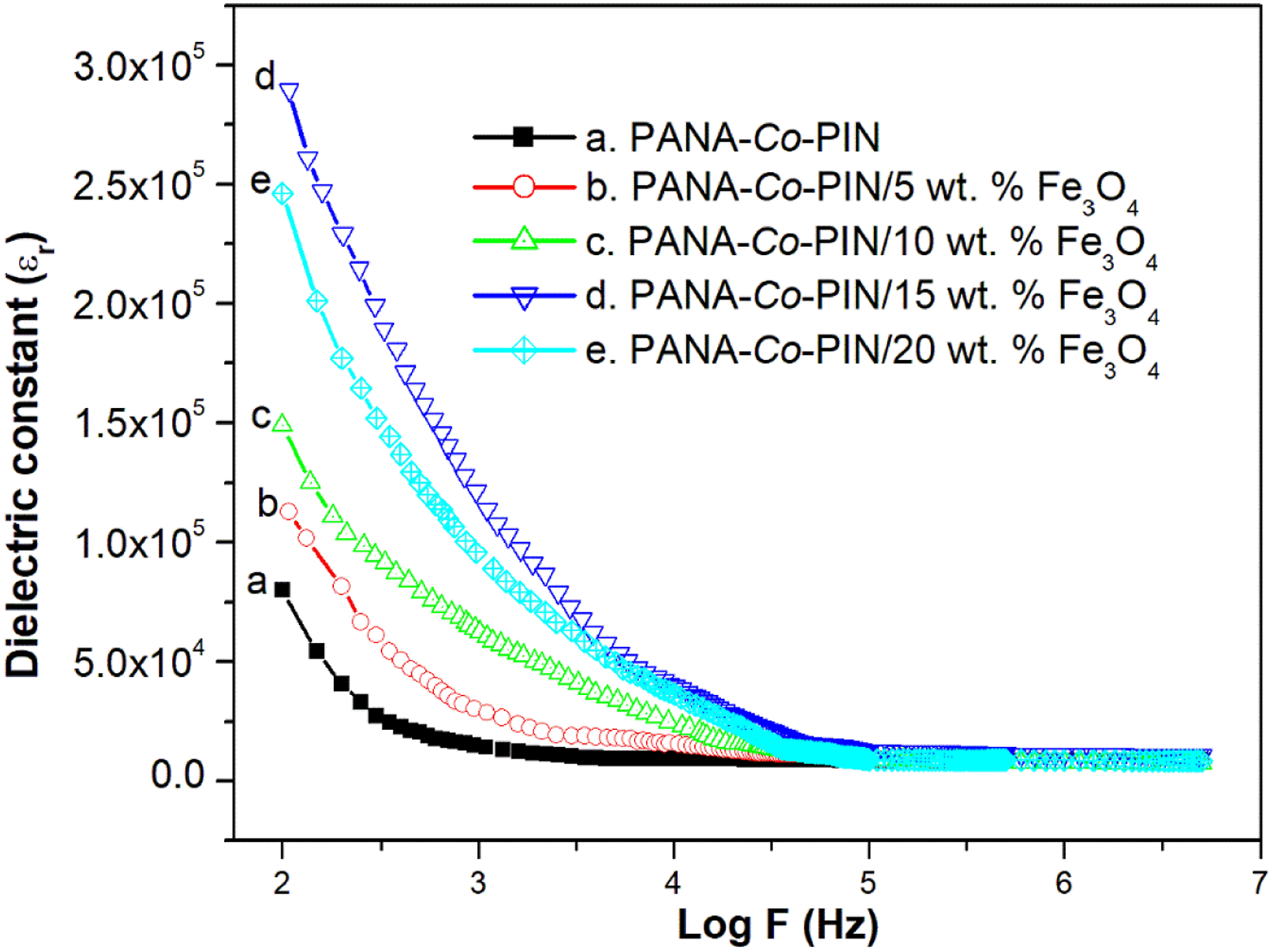
AC conductivity
The change in AC conductivity (σac) with the various frequencies of copolymer with different contents of magnetite nanocomposites are illustrated in Figure 11. It can be seen that the value of log σac increases with an increase in frequency for all the samples. The enhancement in conductivity with frequency indicates the semiconducting nature of the materials. The conductivity of a disordered material generally depends on the hopping mechanism.
29
On increasing the frequency, the hopping rate of charge carriers increases thereby increasing the AC conductivity. The conductivity of all the copolymer composites are higher than the pure copolymer and the poor conductivity of copolymer is due to the disordered structure of the copolymer segments. The maximum AC conductivity is obtained for the sample with 15 wt. % composite. The uniform dispersion of conductive metal oxide nanofillers facilitates both interchain and intrachain electron transfer thereby, improving the AC conductivity of the copolymer matrix. However, the Fe3O4 nanoparticles are irregularly arranged in the copolymer matrix at higher loading of fillers and therefore the conductivity values are found to decrease. Due to the agglomeration of fillers in the copolymer, the polymer-filler interaction is lower than the filler-filler interaction which retards the hopping between the chains, resulting in a poor conductivity at higher loadings of fillers. AC conductivity of magnetite incorporated copolymer.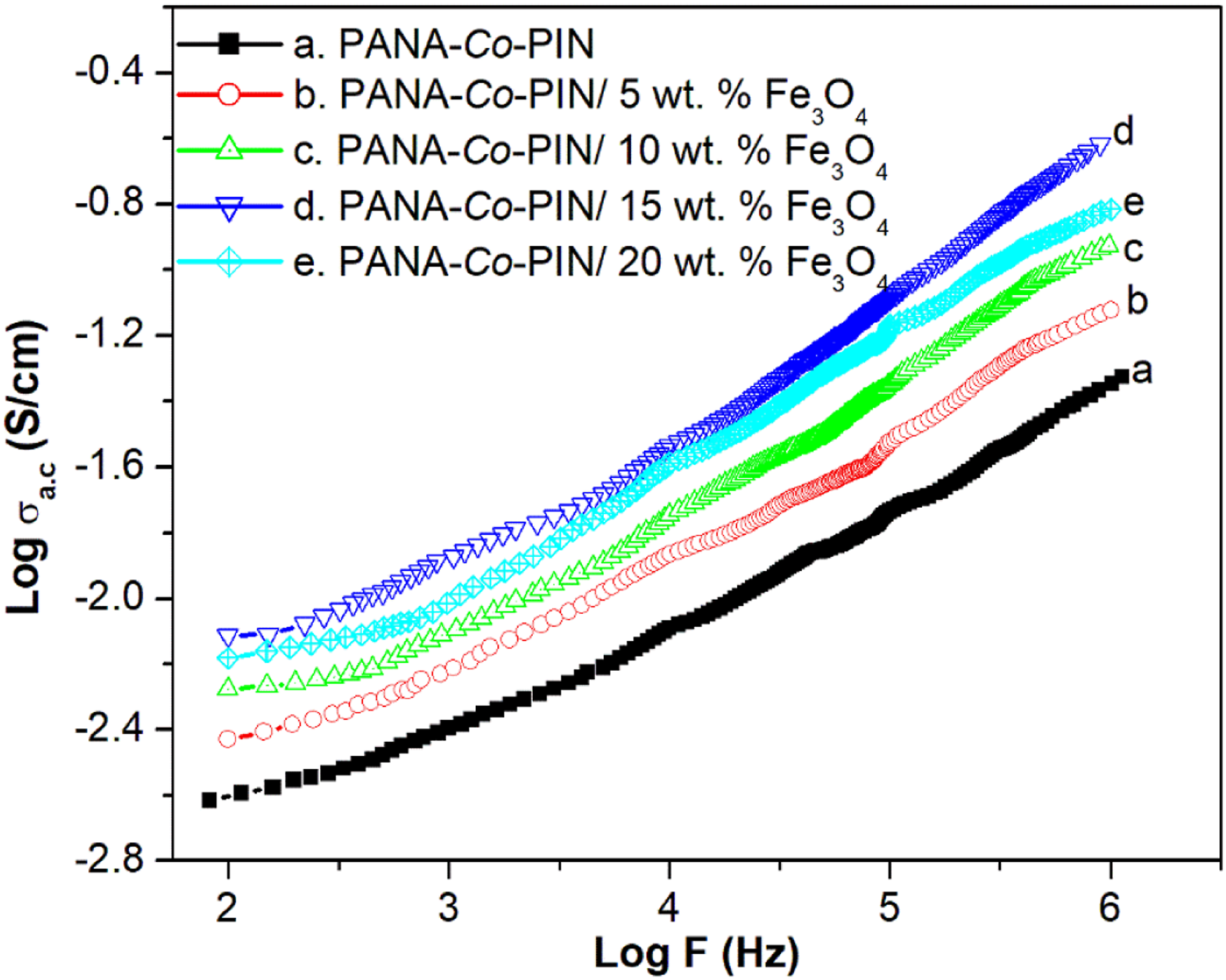
DC electrical conductivity
Figure 12 shows the I-V characteristics of Fe3O4 incorporated PANA-Co-PIN. The I-V plot of all samples follows a straight line and this represents the conducting nature of the copolymeric system. The conduction current of the pure copolymer is lower than the sample with Fe3O4 nanoparticles. This implies that the addition of nanoparticles enhances the movement of charge in the bulk material.
30
The DC conductivity of PANA-Co-PIN nanocomposites is significantly increased with the incorporation of Fe3O4 nanofillers. The successful interaction between the copolymer and Fe3O4 nanofillers can be ascribed to the enhanced DC conductivity of polymer nanocomposites. The increase in conductivity with the addition of Fe3O4 nanoparticles indicates that the tunnelling mechanism is operating in the copolymer matrix due to the increased number of conductive Fe3O4 particles. DC conductivity of magnetite incorporated copolymer.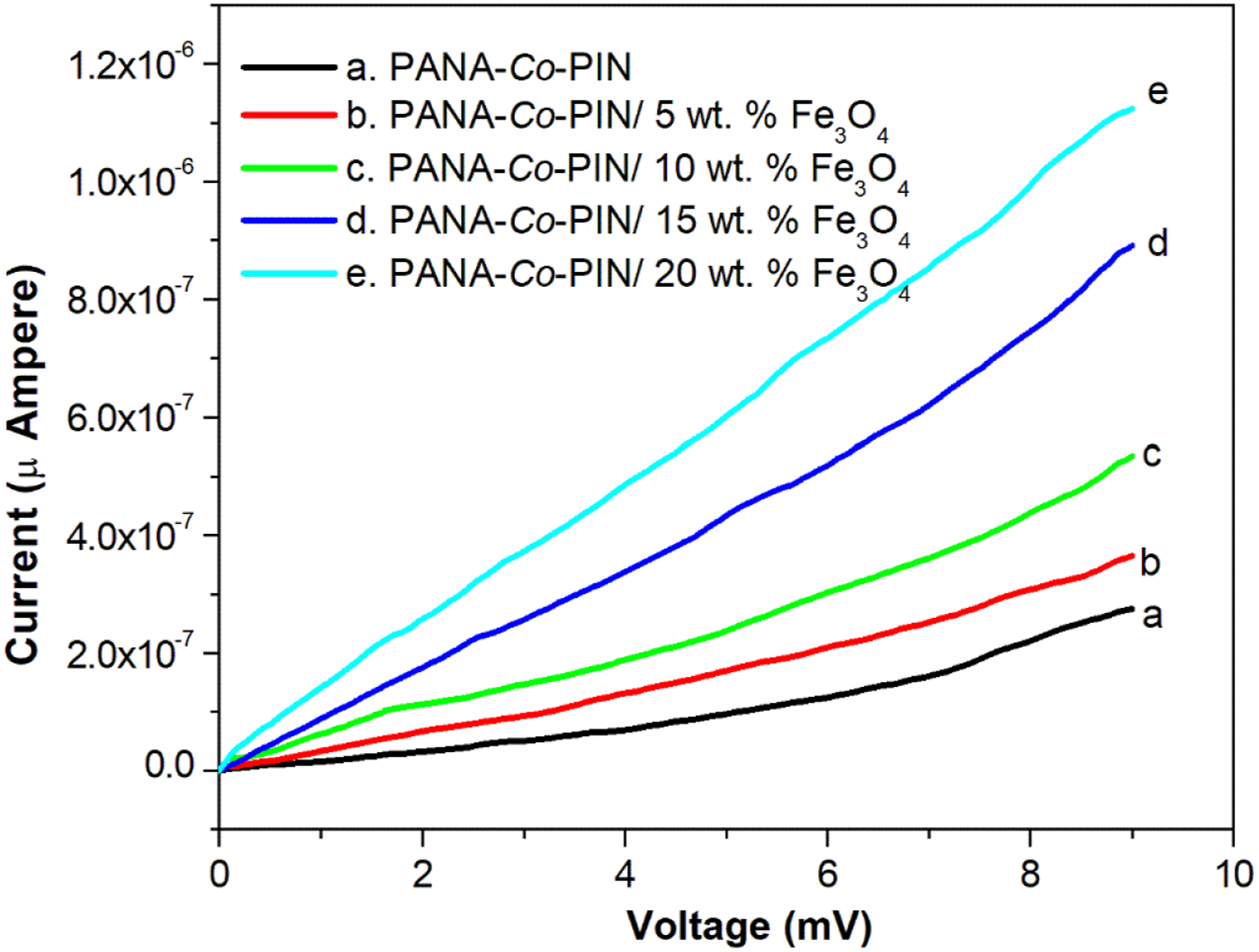
Gas sensing applications
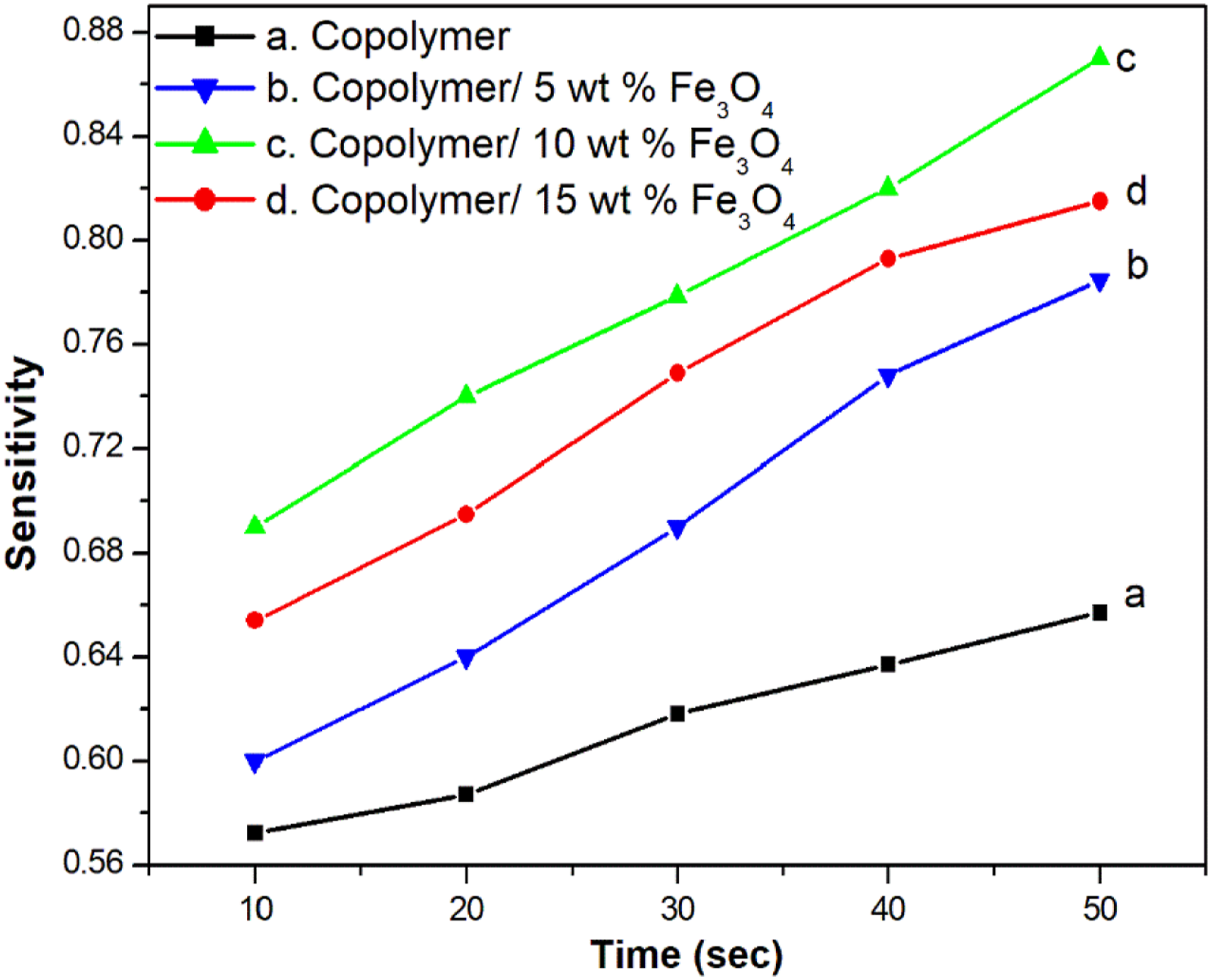
The gas response and recovery of copolymer nanocomposites towards 100 ppm ammonia gas is illustrated in Figure 13. Here the electrical resistance is measured for the copolymer and its nanocomposites during the passage of ammonia gas. Pure copolymer exhibits a higher electrical resistance and this may be due to the interaction between the disordered copolymer chain and ammonia molecules. In other words, the copolymeric segments get lone pair of electrons from ammonia gas, resulting in a higher resistance copolymer. The composite with 10 wt. % sample shows higher gas sensitivity. The strong intermolecular interaction between copolymer and magnetite nanoparticles results in an ordered structure of the composites, which is less available for the passage of ammonia gas into the copolymer composites leading to lower resistance.31,32 The gas sensitivity versus time for PANA-Co-PIN/Fe3O4 nanocomposites is also given in Figure 14. The sensitivity response of the composite increases linearly with the passage of ammonia gas. The copolymer exhibits lower gas sensitivity and the gas sensitivity of the copolymer increases with increases in loading of magnetite nanoparticles up to 15 wt. % loading. This suggests that the interaction between the nanofiller and copolymer is maximum at 15 wt. % loading. The decrease in sensitivity beyond 15 wt. % loading is due to the agglomeration of nanoparticles in the copolymer matrix. Hence, it can be suggested that PANA-Co-PIN/Fe3O4 nanocomposite sensor can be utilized for detecting ammonia gas. Gas sensing response for ammonia gas of copolymer with various contents of magnetite. The variation in gas sensing response of copolymer with different contents of magnetite.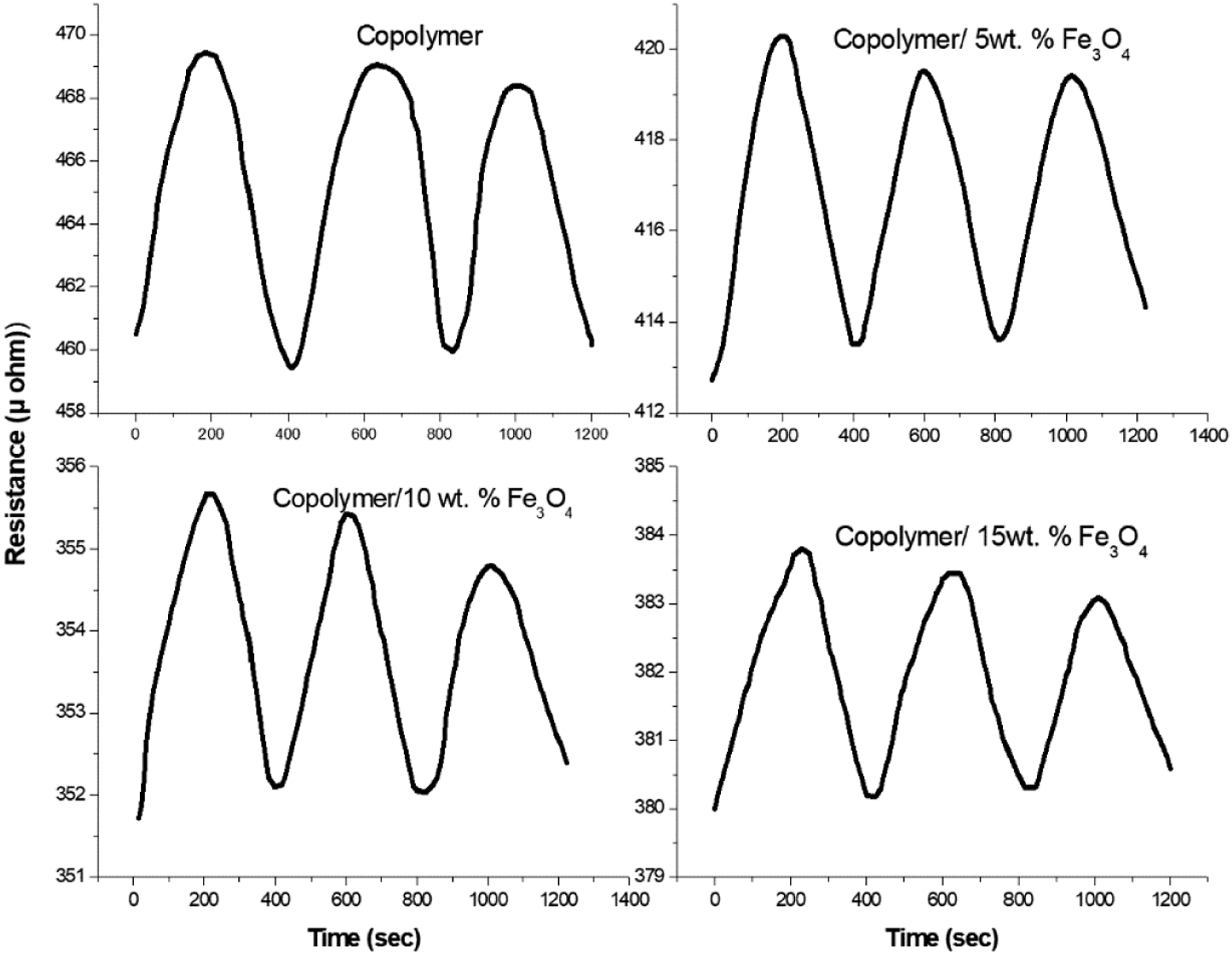
Conclusions
Poly (anthranilic acid-co-indole) with magnetite nanocomposites were successfully synthesised by in-situ polymerization of anthranilic acid and indole in the presence of various contents of synthesised Fe3O4 nanoparticles. FTIR spectra of nanocomposites showed the signals of magnetite particles due to the in-situ polymerization of copolymer with the nanoparticles. The absorbance peak of 15 wt. % sample shifted to the lower wavelength region from UV confirmed the interaction of magnetite nanoparticles with the copolymer. The FESEM images showed that magnetite nanoparticles were well dispersed in the copolymer matrix with uniformly shaped spherical particles indicating the structural changes taking place during the polymerization process. HRTEM images revealed the nanoscale dispersion of magnetite in the copolymer with a size distribution of 15–30 nm. Magnetization measurements showed that the in-situ polymerization of Fe3O4 with the copolymer increases the saturation of magnetization. The VSM result proved that the in-situ polymerization of Fe3O4 with the copolymer nanocomposites has a superparamagnetic characteristic feature. The TGA showed that network polymerization increased the higher thermal stability of the nanocomposites compared to the pure copolymer. Differential scanning calorimetry analysis showed that the glass transition temperature of copolymer nanocomposite increased with the addition of Fe3O4. The higher dielectric properties of polymer composites with increased AC and DC conductivity by the addition of Fe3O4 in copolymer matrix indicated the uniform dispersion of nano-filler due to increased surface area and enhanced displacement of electrons in the conductive network of composite materials. The synthesised copolymer nanocomposite with increased conductivity and magnetic properties may find application in various electronic devices.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
