Abstract
In this study, ethylene propylene diene rubber (EPDM)/styrene–butadiene rubber (SBR) blend nanocomposites filled with alumina (Al2O3) nanoparticles were prepared. Al2O3 nanoparticles were used with or without surface treatment by the silane coupling agent bis[3 (triethoxysilyl)propyl]tetrasulfide, and Al2O3 nanoparticle–filled EPDM/SBR was compared with carbon black. The influences of the filler type, size, concentration, and surface property on the curing characteristics, degree of filler dispersion, mechanical properties, and morphological characteristics of the EPDM/SBR nanocomposites were investigated. We found that the mechanical and curing properties of the nanocomposites were enhanced owing to the presence of Al2O3 nanoparticles. The addition of the silane coupling agent further improved the properties of the nanocomposites.
Keywords
Introduction
Rubber blends based on nanomaterials have become a hot topic of research in recent years. Previously, nanomaterials, such as carbon nanotubes,1,2 halloysite nanotubes,3,4 nano-Al2O3,5,6 silica nanoparticles,7–9 graphene nanoplates,10,11 titanium dioxide nanoparticles,12,13 CaCO3 nanoparticles, 14 graphene nanosheets,15,16 and nano–zinc oxide17,18 have been utilized to prepare rubber nanocomposites.
Carbon black (CB) is a common filler in the rubber industry.19,20 However, owing to its nonrenewable petroleum origin, dark color, contamination, and pollution,7,21 alternative fillers are highly sought after. Thus, the use of nanofillers such as silica nanoparticles, nano-Al2O3, and other nanomaterials as substitutes for CB in rubber compounding has been suggested.5–7 Nanofillers outperformed conventional composites in terms of physical and mechanical properties in vulcanizates. However, the interaction between the rubber matrix and nanomaterials is a crucial parameter determining the attained properties of rubber nanocomposites.
Blending two or more polymers to produce the desired product has been widely performed in the manufacturing industry. Blending rubbers is known to improve the physical properties of the final vulcanized products. Rubber blends are frequently used in the rubber industry to achieve the best combination of compound properties, processability, and cost.9,22,23
Ethylene propylene diene rubber (EPDM)/styrene–butadiene rubber (SBR) blends are used in a wide range of industrial and commercial applications, such as tires, conveying belts, gaskets, seals, tubes, and electrical insulators.22,24–26 Therefore, their blends are suitably used as rubber matrices for different purposes. The surface modification of nonpolar fillers by grafting suitable functional groups on the macromolecular chains of rubbers to improve their adhesion to the filler surface (rubber–filler interaction) is an effective approach, which is widely used to prepare rubber composites.
Surface modification of fillers is a perfect way to improve their dispersive properties and surface activity and, thus, is widely used in the preparation of rubber composites.27,28 Most particulate fillers are inorganic, and their surfaces have poor compatibility with polymers. Various silane coupling agents have been mainly used to improve the compatibility between the filler and the hydrophobic rubber matrix.8,19,29 Silane coupling agents provide efficient reinforcement by improving the dispersion of the filler by reducing filler–filler interactions and increasing the compatibility of the filler with the rubber.8,30,31
Vishvanathperumal and Anand 8 studied the effect of nano-silica on the mechanical properties and swelling resistance of EPDM/SBR nanocomposites with and without (triethoxysilyl)propyl]tetrasulfide (TESPT). The results showed that the mechanical and swelling properties were improved for composites with TESPT than for those without TESPT. This improvement in the properties indicates stronger filler–rubber matrix interfacial adhesion and proper wetting of silica nanoparticles.
Lockhorn and Klüppel 31 investigated the mechanical performance of natural rubber (NR), synthetic polyisoprene rubber, and SBR composites filled with various silica/silane systems. The results showed that the coupling silane provides a strong chemical polymer–filler coupling, accompanied by improved filler–filler bonds for all the rubber types.
Previously, nano-Al2O3–based rubber composites had been fabricated using rubber-like silicone rubber, 32 NR,5,6,33 epoxidized natural rubber (ENR), 34 SBR,35,36 and EPDM.30,37
Mohamad et al. 34 studied the effects of filler loading and 3-aminopropyltriethoxysilane (SCA) on the physical characteristics of ENR–Al2O3 nanoparticle (ENRAN) composites. The filler loading varied from 10, 20, 30, 40, 50, and 60 phr in the absence and presence of 2 phr SCA in every formulation. The Al2O3 nanoparticles strongly interacted with the ENR matrix since they increase the glass transition temperature of the composites. The addition of SCA decreased the maximum torque (MH) and the torque difference (MH–ML) of ENRAN composites, accelerating the speed of the curing process. The spherical Al2O3 particles were dispersed in the ENR matrix, and a uniform particle distribution was observed under a scanning electron microscope, which enabled the torque values obtained from the composites.
Previous studies have shown that the addition of Al2O3 to rubber materials can improve the curing, mechanical, and morphological properties of their composites. However, the effects of Al2O3 on these properties and the optimal concentration of Al2O3 used as fillers in rubber blends have not been studied. Therefore, the main objective of this study was to investigate the effects of adding 2–10 phr of nano-Al2O3 and 5–25 phr of micro-Al2O3 to EPDM/SBR rubber blend nanocomposites on the curing characteristics, degree of filler dispersion, mechanical properties, and morphological characteristics of rubber blends.
Experimental
Materials
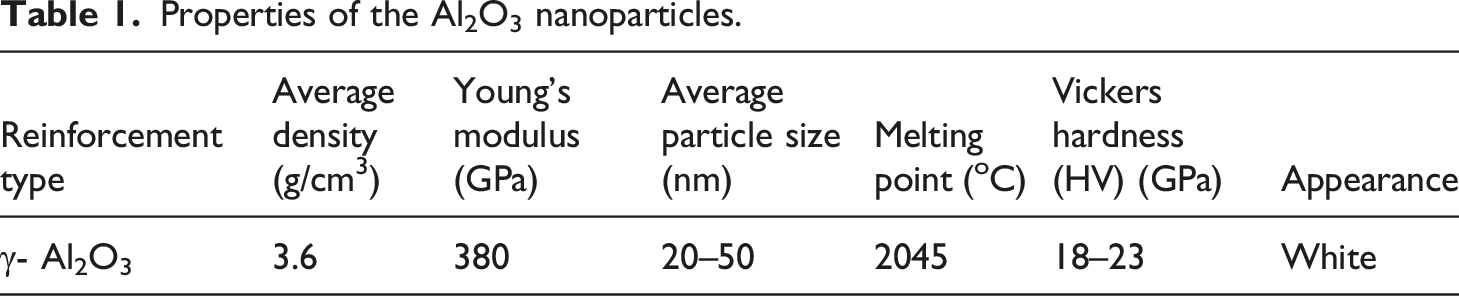
Properties of the Al2O3 nanoparticles.
Surface modification of nano-Al2O3 by silane coupling agents
Surface modification of Al2O3 was performed using TESPT as a chemical surface modifier following a process given by Thongsang and Sombatsompop. 38 TESPT was first combined with ethanol and stirred for 30 min. The TESPT solution was then mixed with Al2O3, and the mixture was stirred for 15 min. The TESPT-treated nano-Al2O3 was then dried for 12 h at 100°C to achieve consistent weight.
Preparation of EPDM/SBR blend nanocomposites
Compounding formulations for EPDM/SBR/filler blend nanocomposites in parts per hundred parts of rubber (phr).
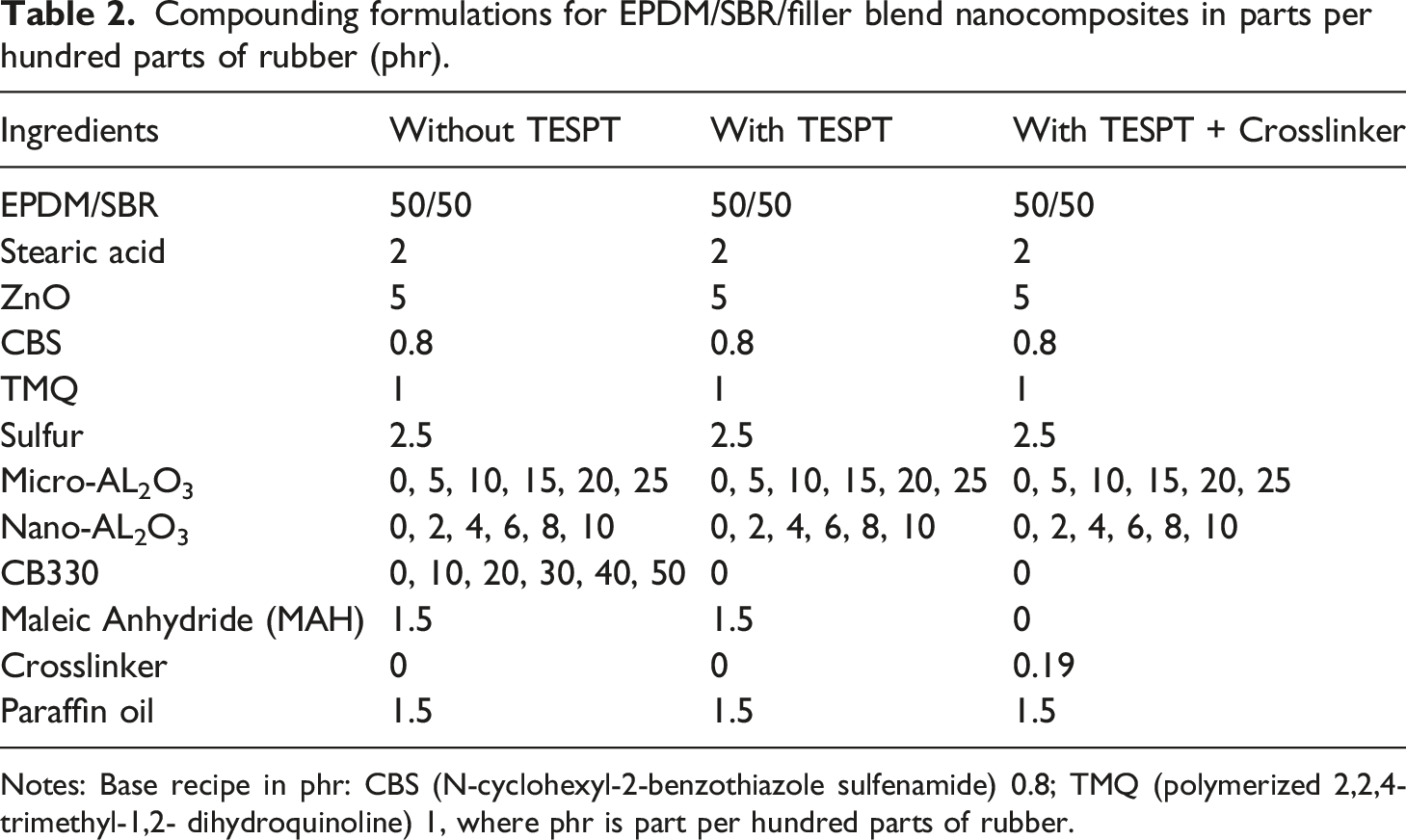
Notes: Base recipe in phr: CBS (N-cyclohexyl-2-benzothiazole sulfenamide) 0.8; TMQ (polymerized 2,2,4-trimethyl-1,2- dihydroquinoline) 1, where phr is part per hundred parts of rubber.
Characterization of rubber blend nanocomposites
Curing characteristics
The curing characteristics of the unvulcanized rubber compounds for different conditions, such as ML (minimum torque), MH (maximum torque), tc90 (time of 90% curing), ts2 (scorch time), and the cure rate index (CRI), were obtained using an oscillating disk rheometer (Monsanto 100, USA) at 152°C ± 1°C according to ASTM D 2084–01. 40 To get vulcanized rubber sheets, the blends were stored for 1 day before being vulcanized in an electrically heated press under a pressure of 4 MPa according to the optimum curing conditions.
Tensile properties
The tensile properties of the nanocomposites were analyzed using the Zwick/Roell Z010 tensile testing machine, Germany. The crosshead speed of the tensile test machine was maintained at 500 mm/min. Dumbbell samples cut from a compression-molding vulcanized lap sheet were used for tensile testing according to ASTM D 412-16. 41 The tensile properties of each sample, such as tensile strength (TS), stress at 50%, 100%, 200%, and 300% elongations (M50, M100, M200, and M300), and elongation at break (%), were determined. The average result of data collected from five samples was taken for each composite composition, and the results were tabulated and analyzed.
Morphology of EPDM/SBR/Al2O3
Transmission electronic microscopy
The particle size of the nanoparticles was measured by transmission electronic microscopy (TEM), where the TEM images were examined using JEOL JEM 2100 (Japan) with a micro-analyzer electron probe.
FT-IR spectroscopy
Fourier-transform infrared (FT-IR) spectroscopy, JASCOFTIR-6000 E (Japan), operated in the absorption mode was used to determine the functional groups in the samples. The analysis was conducted at wavelengths between 400 and 4000 cm−1 at room temperature, and the KBr tablet method was used to evaluate the functional groups of the samples by mixing with KBr (potassium bromide) disks. The spectra were obtained with a resolution of 4 cm−1 with Model ATR PRO450-S.
Scanning electron microscopy
The fractured surface of the rubber nanocomposite samples was examined using a scanning electron microscope (JEOL, JSM 6360LA, Tokyo, Japan) at 25 kV.
Results and discussions
Morphology of Al2O3 particles treated by TESPT
TEM results
The TEM images of the untreated/treated Al2O3 particles are shown in Figure 1. The Al2O3 particles agglomerated together, and the dispersibility was poor, as shown in Figure 1(a) and (c) for micro and nanoparticles, respectively. After Al2O3 particles were treated with TESPT, they showed improved dispersibility, although some smaller agglomerates remained, as shown in Figure 1(b) and (d), indicating that TESPT has modified the surface of Al2O3 particles. TEM images of alumina before and after modification with TESPT. (a) Untreated micro-alumina, (b) treated micro-alumina, (c) untreated nano-alumina, and (d) treated nano-alumina.
FT-IR spectroscopy
Figure 2 illustrates the FT-IR spectra of both treated and untreated Al2O3 particles. The results showed absorption peaks with different bands in the range of 400–4000 cm−1. This spectral region involved many important stretching modes, including carbon, hydrogen, oxygen, and Al2O3 bands. The spectra below 1000 cm−1 revealed the general features of Al2O3. The O–H vibrational mode of Al2O3 ranged from 3216 to 3644 cm−1. For untreated alumina, as shown in Figure 2(a) and (c), the stretching mode of surface hydroxyl groups was responsible for the broad peak around 3420.29 and 3393.53 cm−1 for micro- and nano-Al2O3 particles, respectively. Moreover, the absorption peak intensity decreased to 3333.45 and 3332.03 cm−1 for treated micro- and nano-Al2O3 particles with the addition of TESPT, respectively, as shown in Figure 2(b) and (d). IR spectra of alumina before and after modification with TESPT. (a) Untreated micro-alumina, (b) treated micro-alumina, (c) untreated nano-alumina, and (d) treated nano-alumina.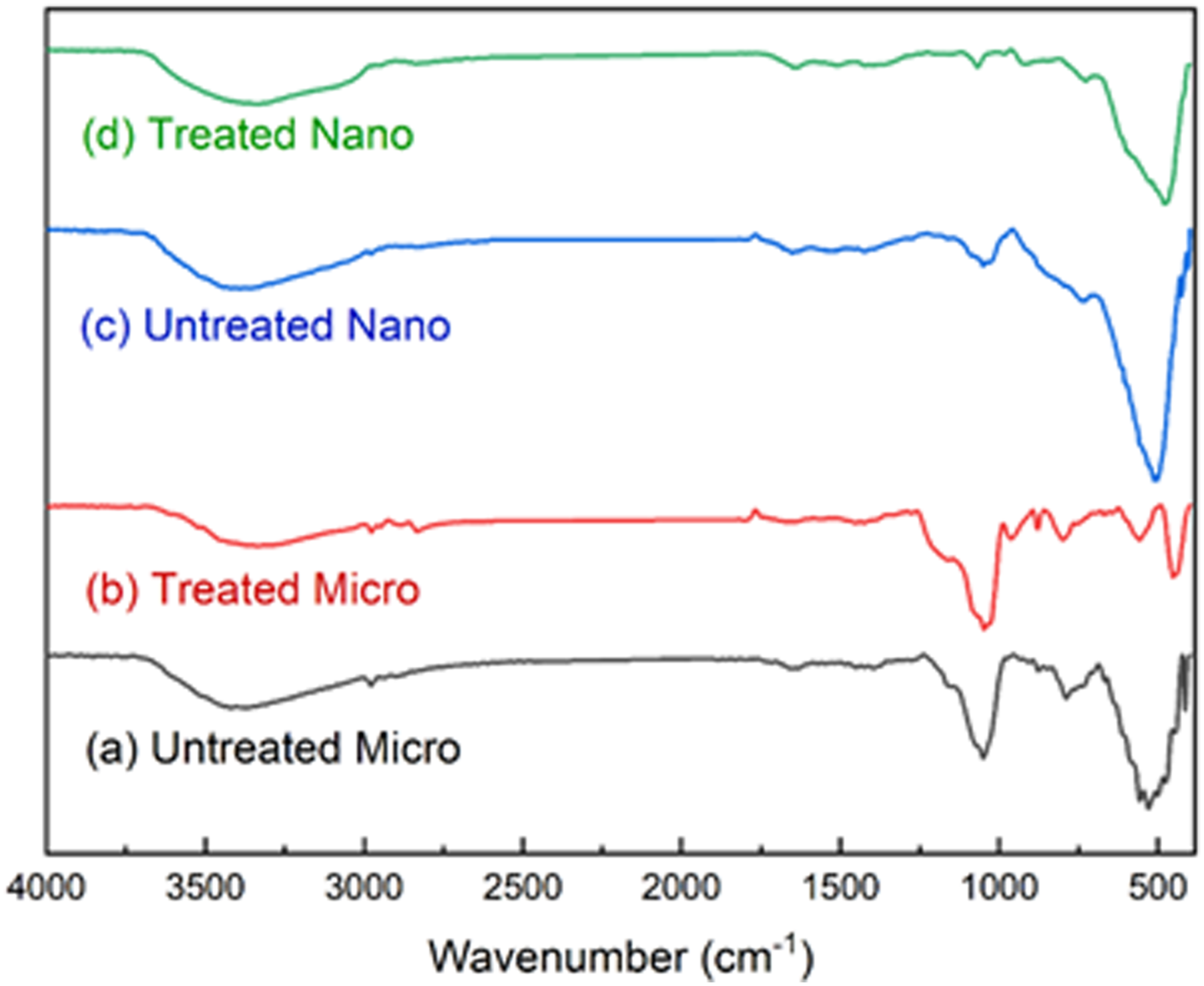
In the FT-IR spectra of surface-treated Al2O3, the intense peak of the surface hydroxyl group shifted toward the lower wavenumber region compared to the untreated one. This type of shifting was due to the effective interaction between the surface hydroxyl group of nano-Al2O3 and silane coupling agents. Therefore, FT-IR results suggested that the treatment of silane coupling agents successfully modified the surface hydroxyl groups of Al2O3.5,42,43 For surface-treated micro-Al2O3, some new bands were observed at 878.66, 962.47, and 2831.48 cm−1. Furthermore, a new band was observed at 911.78 cm−1 for the treated nano-Al2O3 particles For all samples, the wavenumber range of 2750–2990 cm−1 could be associated with C–H bonds.
Cure characteristics
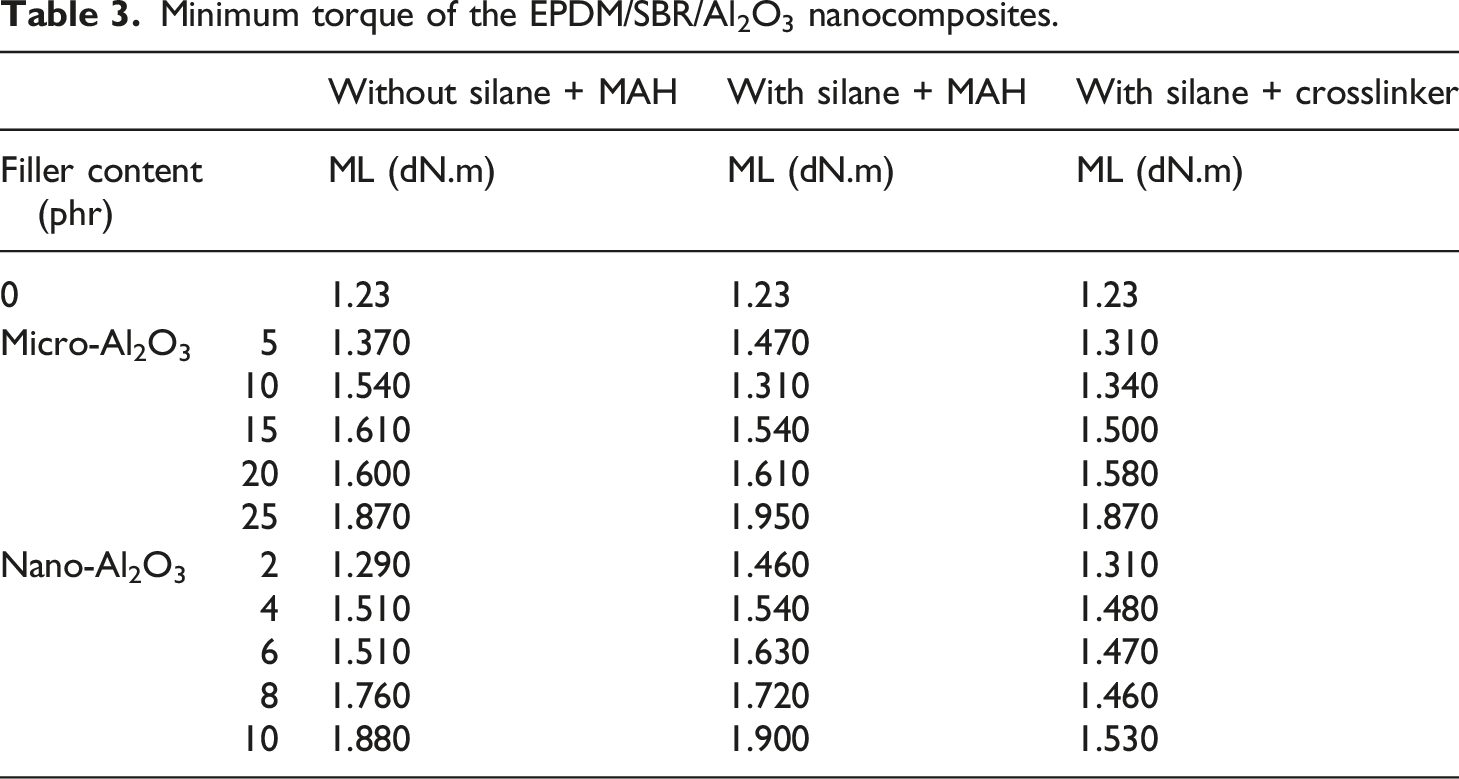
Minimum torque of the EPDM/SBR/Al2O3 nanocomposites.
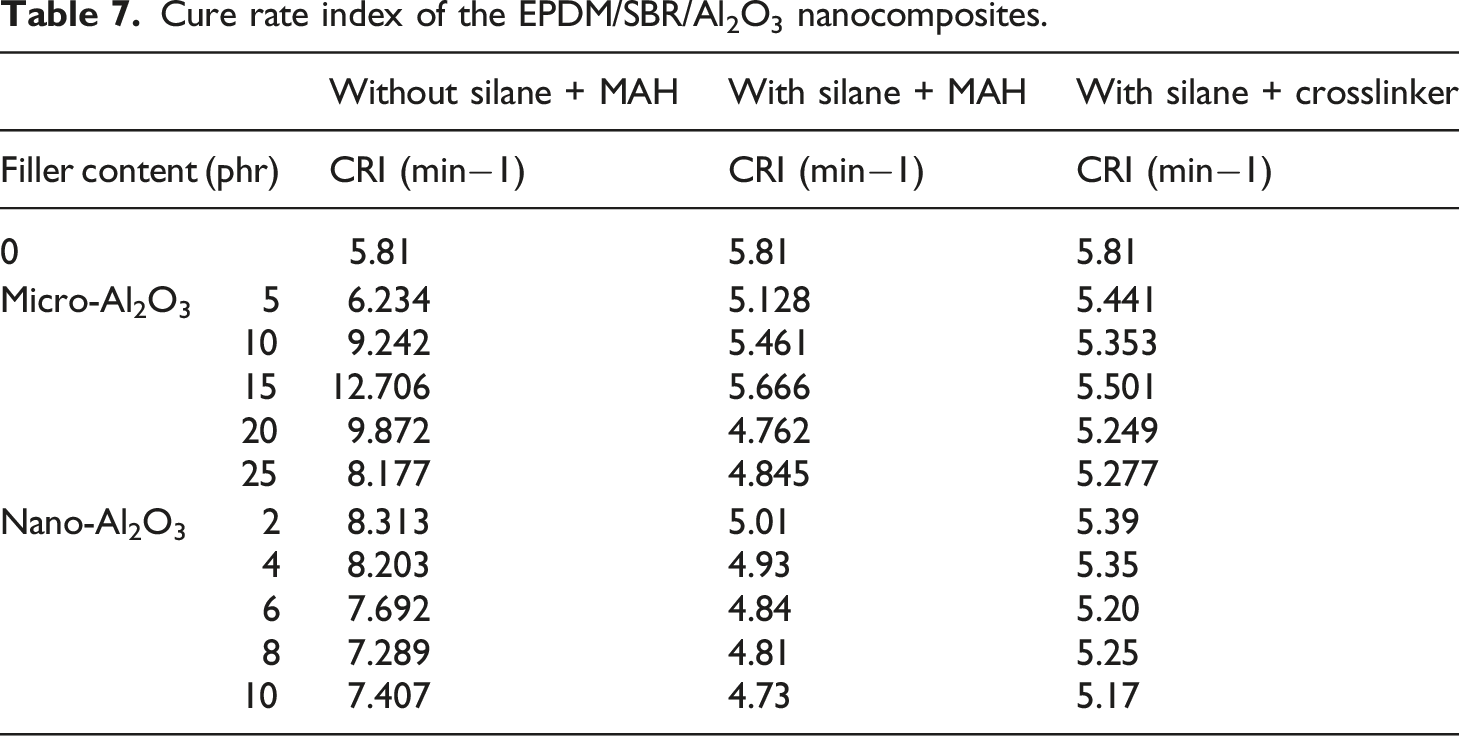
Maximum torque of the EPDM/SBR/Al2O3 nanocomposites.
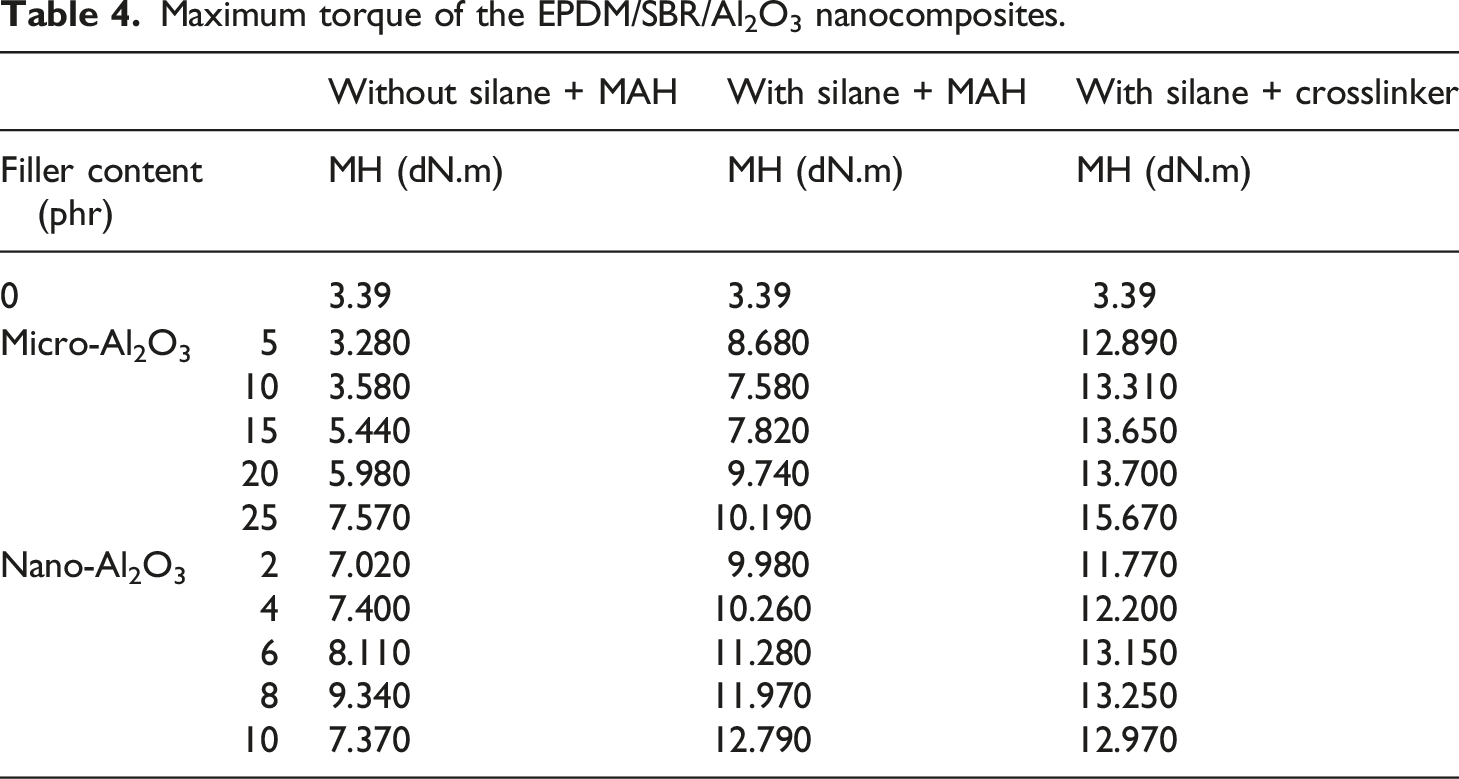
Scorch time of the EPDM/SBR/Al2O3 nanocomposites.
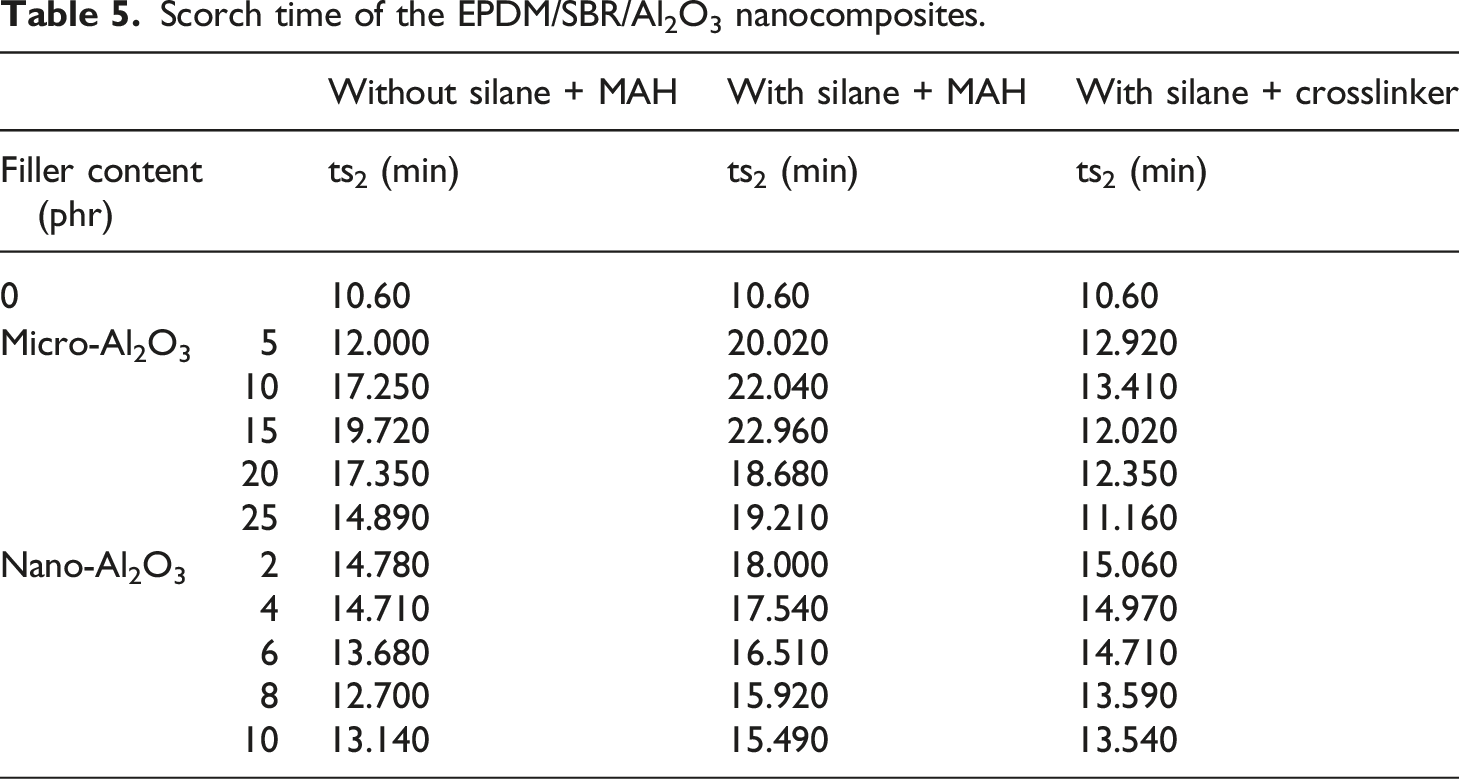
Optimum cure time of the EPDM/SBR/Al2O3 nanocomposites.
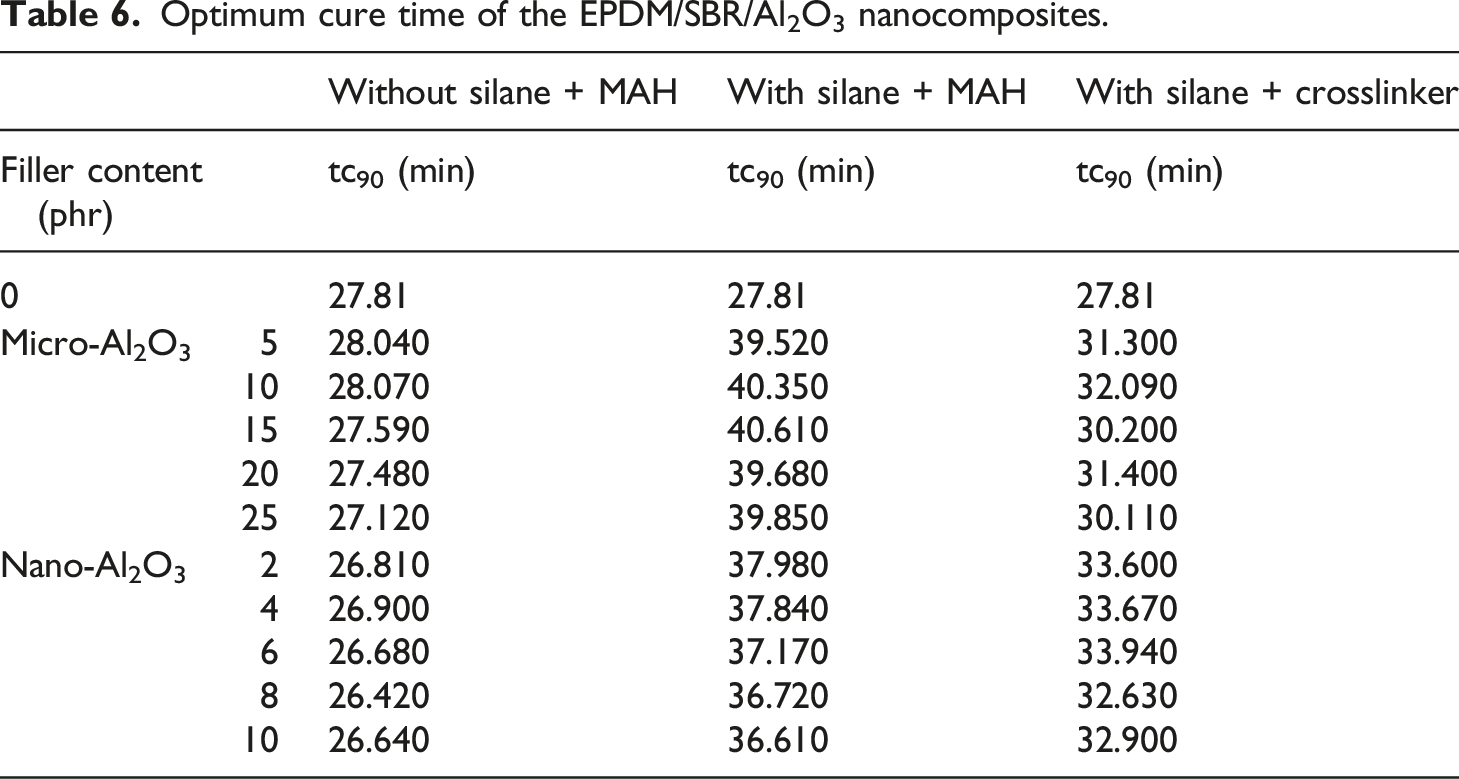
Cure rate index of the EPDM/SBR/Al2O3 nanocomposites.
Rheometric characteristic of the EPDM/SBR/CB nanocomposites.
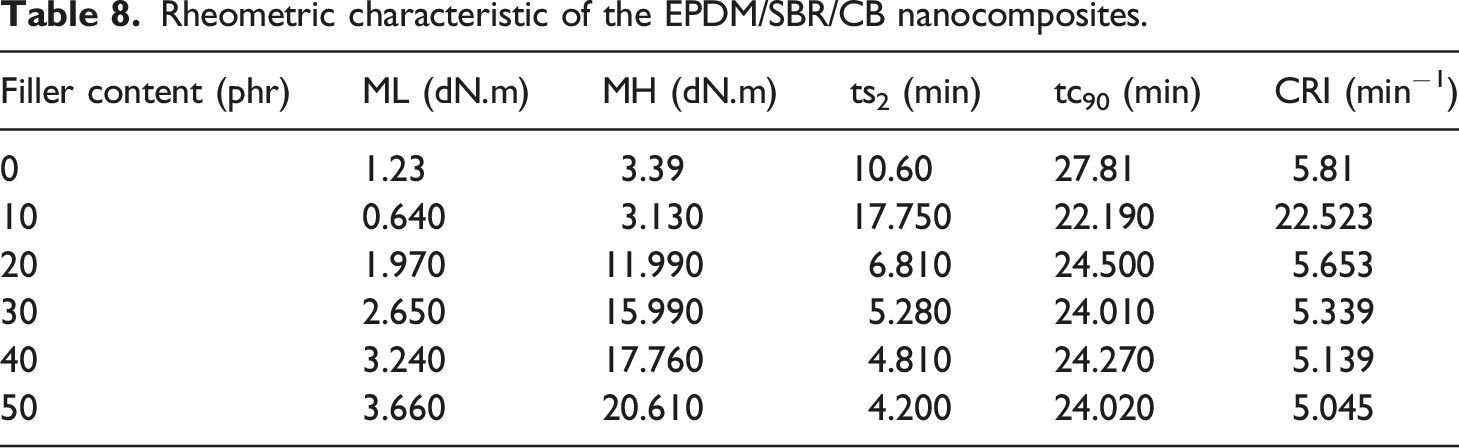
Table 3 shows the minimum torque of nanocomposites. EPDM/SBR/Al2O3 nanocomposites’ minimum torque was higher than that without Al2O3 particles. The minimum torque of EPDM/SBR/Al2O3 nanocomposites with the crosslinker was lower than that of Al2O3 with/without TESPT and MAH compounds. The minimum torque reflects both the filler–filler agglomeration and the viscosity of the rubber compound. A lower minimum torque indicates lower viscosity and weaker filler–filler interactions.7,45 The enhancement in the minimum torque for the EPDM/SBR composite was due to the formation of some premature crosslinks during compounding. TESPT is an active ingredient for the EPDM/SBR composite. The Al2O3 filler and TESPT interacted and reacted chemically forming another type of cross-link. In this case, TESPT could be considered a curing agent for the EPDM/SBR composite. Our findings are in agreement with those of previous studies. 5
The crosslinker decreased the minimum torque, facilitating the dispersion of the Al2O3 filler. In this case, the crosslinker could be considered an internal plasticizer for the EPDM/SBR composite. 46
Table 4 shows the maximum torques of nano-Al2O3–filled EPDM/SBR compounds with/without TESPT and crosslinker. The maximum torque correlates with the measurement of stock modulus. 8 The maximum torque of nano-Al2O3–filled EPDM/SBR compounds of all the rubber compounds was higher than that of their control ones. As a result, above a loading level of 8 phr of untreated nano-Al2O3, the maximum torque of the EPDM/SBR system dropped noticeably. Several researchers have demonstrated that the agglomeration tendency of nanofillers within the rubber matrix reduces the maximum torque in rubber nanocomposites. 5
The maximum torques of the nanocomposites increased with the addition of the silane coupling agent. This may be due to the breakdown of agglomerates by the silane coupling agent.5,8,45 Also, the value of the maximum torque of Al2O3-filled EPDM/SBR composites with the crosslinker was higher compared to those with and without TESPT. The mobility of the rubber chain in the composite reduces with an increase in the crosslinker in the presence of treated nano-Al2O3 and, thus, increases its maximum torque. The improvement in the maximum torque was because of the more significant interaction among Al2O3 and the EPDM/SBR blend owing to the presence of the cross-linker. TESPT and the crosslinker could be considered a compatibilizer for the Al2O3-filled EPDM/SBR composites. 46
The premature vulcanization in the composite was measured by scorch. Table 5 presents the scorch time of nanocomposites. For different Al2O3 ratio-cured EPDM/SBR nanocomposites with and without TESPT, it was seen that the incorporated Al2O3 increased the scorch time. TS2 of nano-Al2O3–filled EPDM/SBR blends was higher compared with that of the EPDM/SBR gum because highly polar nano-Al2O3 interacts with ZnO during vulcanization, reducing the activity of ZnO to boost the accelerator during vulcanization. 44 The scorch time showed that the adsorption of curing agents on Al2O3 resulted in delayed scorch time in the EPDM/SBR composite with TESPT. The analysis also indicated that the TESPT-induced composite enhanced growth in scorch time compared with the composite without TESPT. This is due to the uniform dispersion of nano-silica particles and TESPT. Higher scorch time showed that nanocomposites with Al2O3 particles and TESPT have better processability. 44
The optimum curing time was measured to determine the minimum time required to meet the maximum torque of about 90%. It is used to measure the time required for the complete curing of the rubber compound. Table 6 indicates the optimum cure time of the rubber blend nanocomposites. For different untreated Al2O3 nanoparticle ratio-cured EPDM/SBR composites, it was seen that the incorporated Al2O3 nanoparticles decreased the optimum curing time. This result indicated that untreated Al2O3 nanoparticles could activate the EPDM/SBR matrix. 5 The optimum curing time of nanocomposites filled with treated Al2O3 nanoparticles was higher than that of the similar composite with untreated Al2O3 nanoparticles. An increase in curing time with the concentration of treated nano-Al2O3 particles is due to the adsorption of the curing agents on the Al2O3 particles.5,44 However, the increase in the content of treated/untreated Al2O3 particles did not significantly influence curing time. This is due to the interfacial interactions between Al2O3 and EPDM/SBR rubber blends through hydrogen bonding between OH groups and surface silanols of Al2O3.
The CRIs of nanocomposites are presented in Table 7. The CRI increased with the increased content of Al2O3 without TESPT. The increase in the CRI indicates the maximum curing rate and is attributed to the presence of Al2O3 and crosslinking agents in EPDM/SBR nanocomposites. The EPDM/SBR composite with Al2O3 and TESPT exhibited the lowest CRI. Overall, adding Al2O3 and TESPT increased torque and times compared to the neat EPDM/SBR composite. These results are in good agreement with those of a previous study by Roy and Potiyaraj 5 who studied the effect of nano-Al2O3 on the curing properties of nano-Al2O3-based NR nanocomposites.
Table 8 shows the rheometric characteristic of the EPDM/SBR/CB nanocomposites. Overall, it can be clearly seen that the rheometric characteristic of blend nanocomposites improved with increasing CB content which was in agreement with the work reported by many considerable studies.20,47,48 This increase indicated that the incorporation of CB enhanced the viscosity of the EPDM/SBR blend matrix. The higher MH for filled rubber blend indicated that stronger interactions between CB and the EPDM/SBR rubber matrix are obtained. In addition, the ts2 and the tc90 of the samples are decreased as the CB loading increased as shown in Table 8. The lower ts2 of the sample represented better processing ability. The decrease in the ts2 is due to the presence of more crosslinkers in the rubber composites, whereas the decrease in tc90 is attributed to the higher energy input and greater heat buildup during mixing due to higher viscosity.49,50
Figure 3 shows the rheometric characteristic of the EPDM/SBR nanocomposites for different filler types at 10 phr. As shown in Figure 3, the highest values of ML and MH in EPDM/SBR/Al2O3 composites at 10 phr may originate from higher rigidity and the more number of bonds produced between Al2O3 and the rubber blend matrix chains at the interface. Aside from the nature of TESPT bonds, the presence of a large amount of sulfur (40% of the total sulfur content of the compound) in the interfacial region of the EPDM/SBR/Al2O3 composite and the high activity of TESPT in the vulcanization reaction are associated with a higher number of these bonds at the interface. Therefore, the increased number of stiff bonds resulted in a rigid interface in the EPDM/SBR/Al2O3 composite with TESPT and increased the stress transfer between the rubber and Al2O3 nanoparticles, leading to higher torques (ML and MH).
51
Rheometric characteristic of the EPDM/SBR nanocomposites for different filler types at 10 phr: (a) Minimum torque, (b) maximum torque, (c) scorch time, and (d) optimum cure time.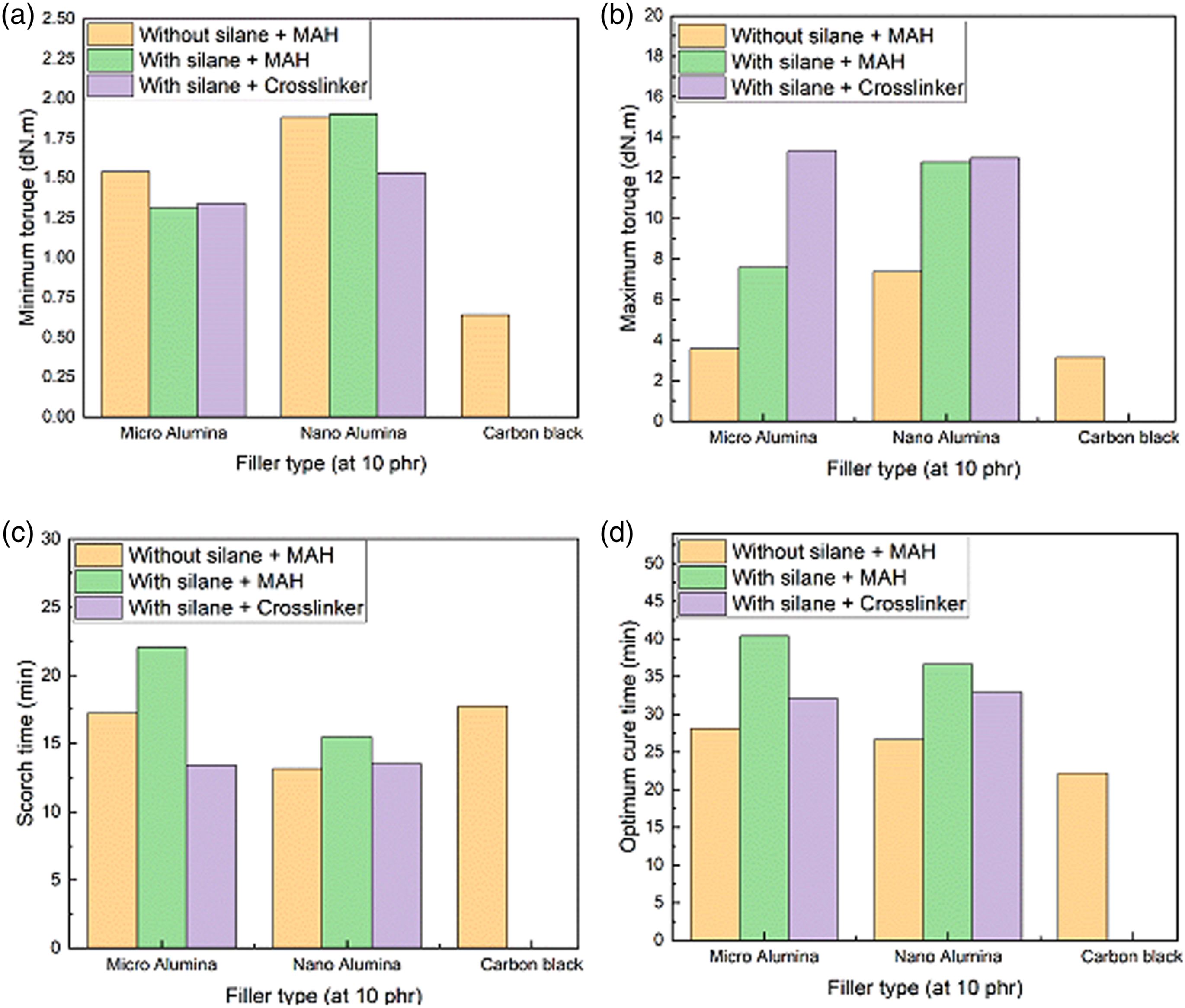
Degree of filler dispersion in EPDM/SBR/filler–rubber blend nanocomposites
The degree of reinforcement of EPDM/SBR
The L values of alumina in the rubber systems are shown in Figure 4. At particular filler loading, a lower value of L means a better degree of filler dispersion. As can be seen, the L value of the EPDM/SBR Filler dispersion parameters of EPDM/SBR/filler rubber blend nanocomposites: (a) Micro-Al2O3, (b) nano-Al2O3, (c) carbon black, and (d) different filler types at 10 phr.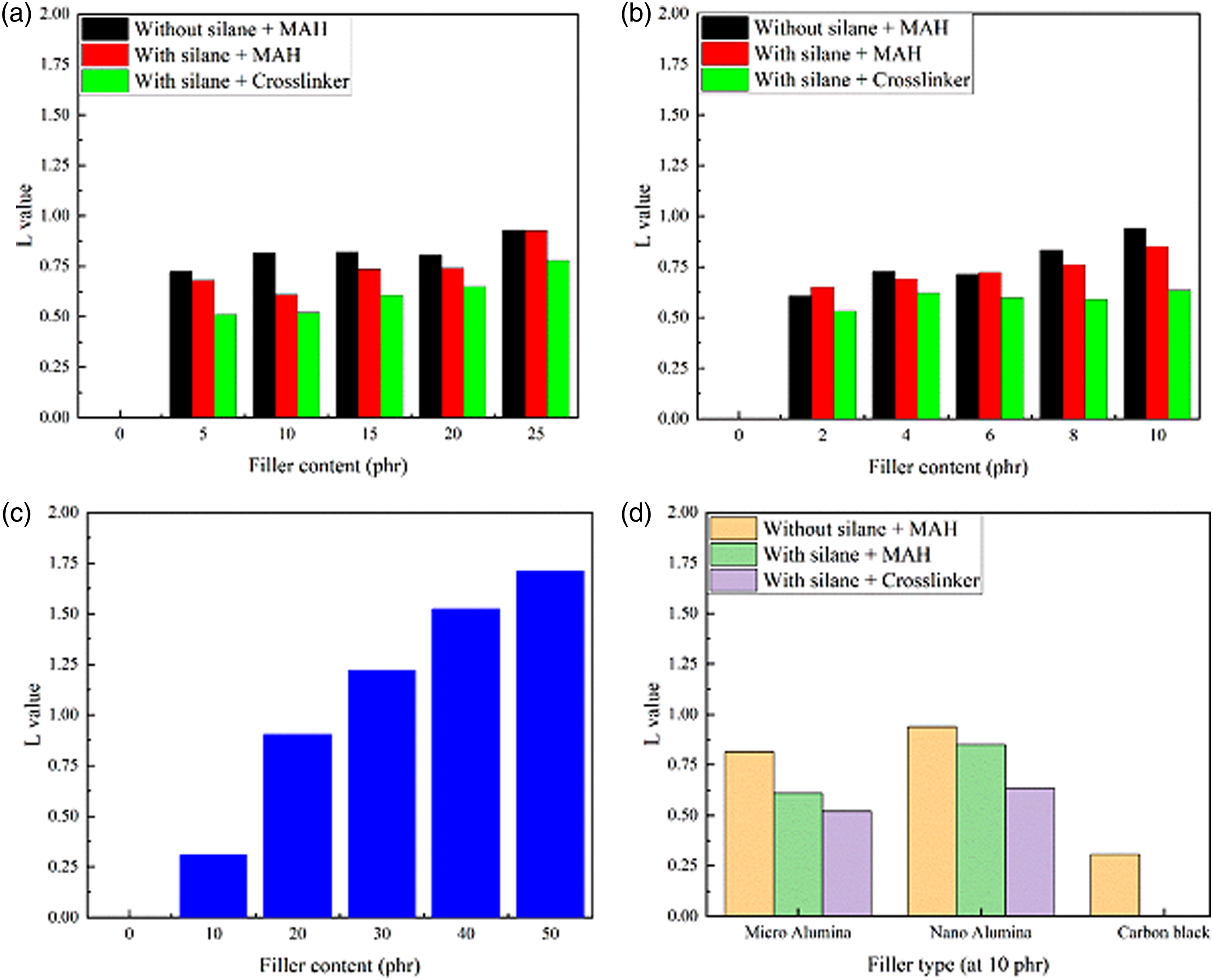
Tensile properties
The mechanical characteristics of the EPDM/SBR/Al2O3 nanocomposites were investigated in comparison with the Al2O3 nanoparticle-cured EPDM/SBR rubber blends with/without TESPT and the crosslinker. The TS and elongation at break (%) of the cured rubber blend nanocomposites concerning the concentration of treated/untreated Al2O3 nanoparticles and CB are presented in Figures 5 and 6. The TSs of the treated Al2O3 nanoparticle–filled vulcanizates with MAH were higher than those of the untreated Al2O3 nanoparticle–filled vulcanizates and treated Al2O3 nanoparticle–filled vulcanizates without MAH, showing the significant effect of Al2O3 nanoparticle surface modification on improving the mechanical properties of Al2O3 nanoparticle–reinforced vulcanizates. Tensile strength of the EPDM/SBR nanocomposites: (a) Micro-Al2O3, (b) nano-Al2O3, and (c) carbon black. Elongation at break (%) of the EPDM/SBR nanocomposites: (a) Micro-Al2O3, (b) nano-Al2O3, and (c) carbon black.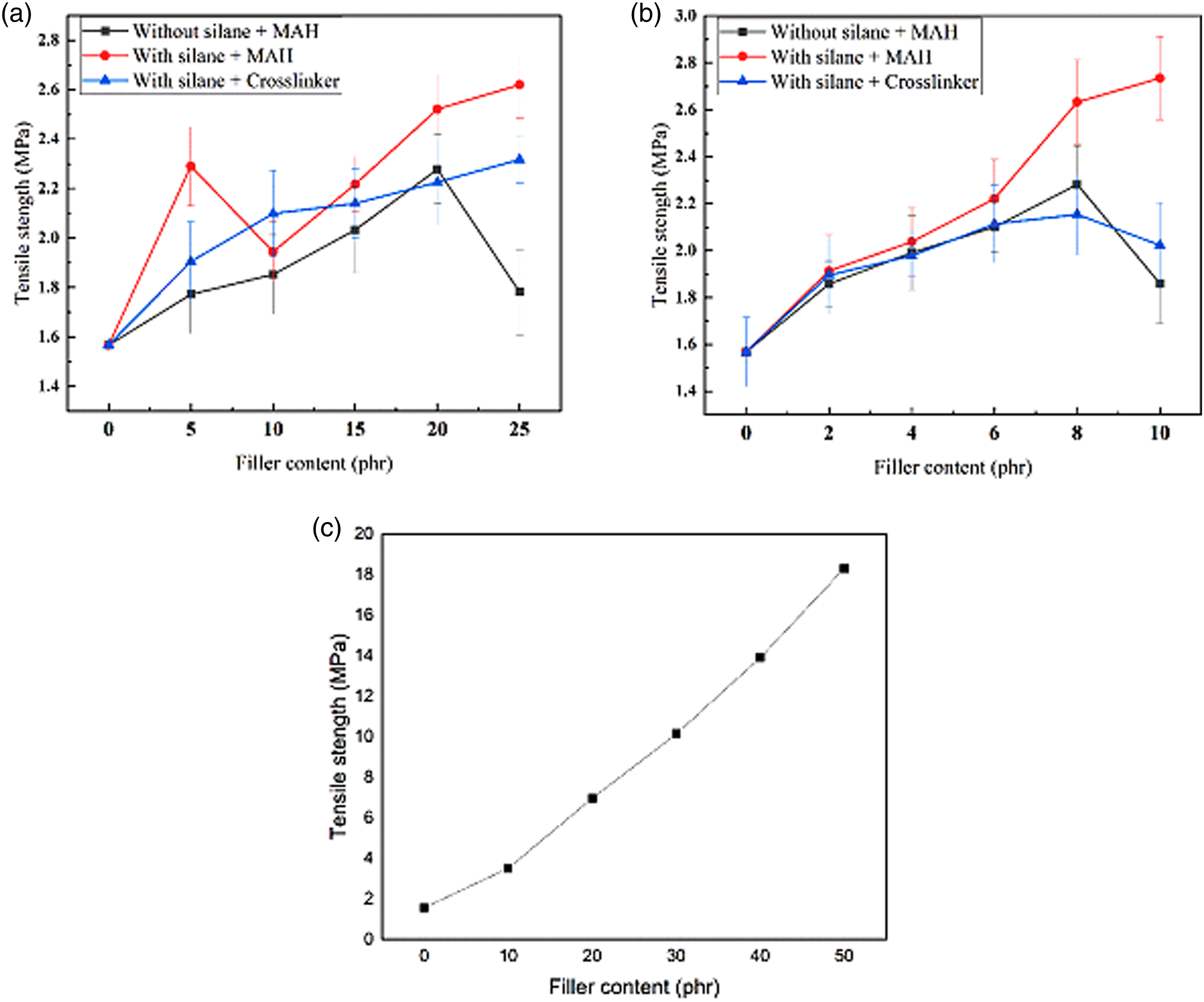
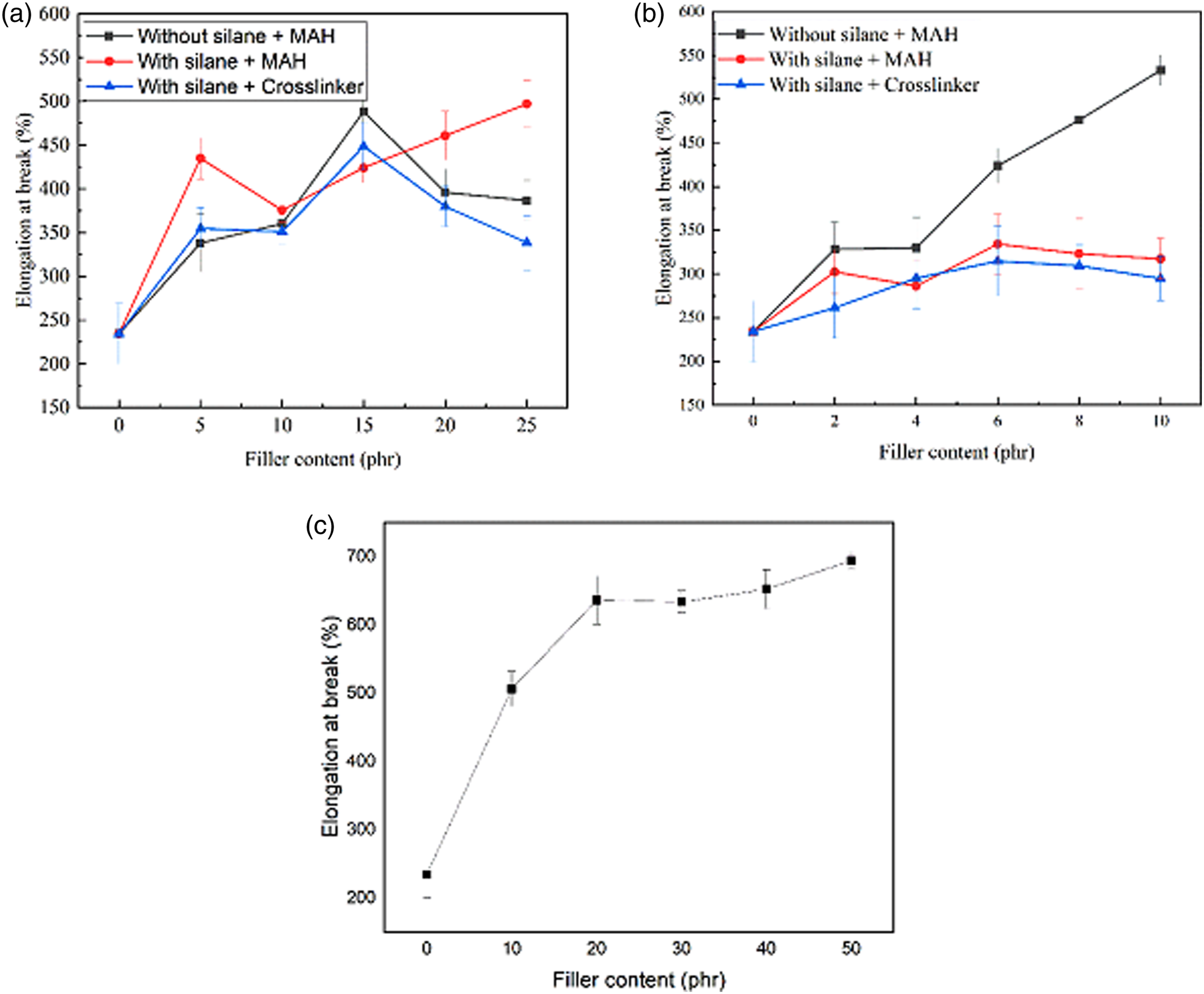
Figure 5(a) shows that the TSs of the nanocomposites with treated Al2O3 significantly improved in comparison with those of untreated Al2O3. However, alumina content is the same in both nanocomposites. So, the use of the silane coupling agent further improved rubber–filler interactions and reduced filler–filler interactions, evident from SEM investigations. Moreover, the use of the organosilane provides additional crosslinks through its organofunctional group. These effects may have led to the improved tensile properties of the nanocomposites with treated Al2O3 compared with the nanocomposites with untreated alumina, which is reflected in the stress–strain study. 27 The TSs of EPDM/SBR/Al2O3 nanocomposites increased with the favorable loading of nano-Al2O3 up to 8 phr and then decreased gradually, as shown in Figure 5(b). This may be due to the poor interaction of nano-Al2O3–EPDM/SBR rubber blends caused by the hydrophilic nature of Al2O3 nanoparticles, leading to higher loading conditions in the formation of aggregates.44,45 However, in the case of using MAH as a compatibilizer, the tensile strengths of the nanocomposites increased with the favorable loading of nano-Al2O3. These results are in good agreement with those of a previous study 5 ; silane coupling agents were very effective to enhance mechanical properties such as modulus and tensile strength of nano-Al2O3–based NR nanocomposites.
In Figure 6, the elongation at break (%) of EPDM/SBR nanocomposites is shown. The elongation at break (%) increased with the increase in nano-Al2O3 loading. The increase in elongation at break (%) suggests a good interfacial adhesion between the filler and the rubber and proper wetting of nano-Al2O3. The reduction in elongation at break (%) indicates the restriction of movement in polymeric molecular chains. 45 The elongation at break (%) of EPDM/SBR nanocomposites with TESPT decreased compared with the nanocomposites without TESPT, indicating that the dispersion of nano-silica particles in the rubber matrix has improved. In addition, TESPT and nano-Al2O3 particles are non-deformable and inflexible, restricting the free movement of molecular chains. 45 For the neat EPDM/SBR rubber blends, the addition of 10 phr of nano-Al2O3 with TESPT resulted in a roughly 74% increase in tensile strength, from 1.57 MPa to 2.74 MPa, and a 40.49% decrease in elongation at break (%), from 533.33% to 317.41%, as shown in Figures 5 and 6. From the tensile strength analysis, the presence of TESPT can increase the dispersion capability of Al2O3 nanoparticles and their interfacial interaction with the EPDM/SBR matrix blend.
Figure 7 shows the tensile properties of the EPDM/SBR nanocomposites for different filler types at 10 phr. However, in the EPDM/SBR/CB vulcanizate, the tensile properties were appropriate due to the higher quality of filler–polymer interactions compared with the EPDM/SBR/Al2O3 vulcanizate, as shown in Figure 7.
51
Tensile properties of the EPDM/SBR nanocomposites for different filler types at 10 phr: (a) Tensile strength (MPa); (b) elongation at break (%); (c) M50 (MPa); (d) M100 (MPa); M200 (MPa); and M300 (MPa).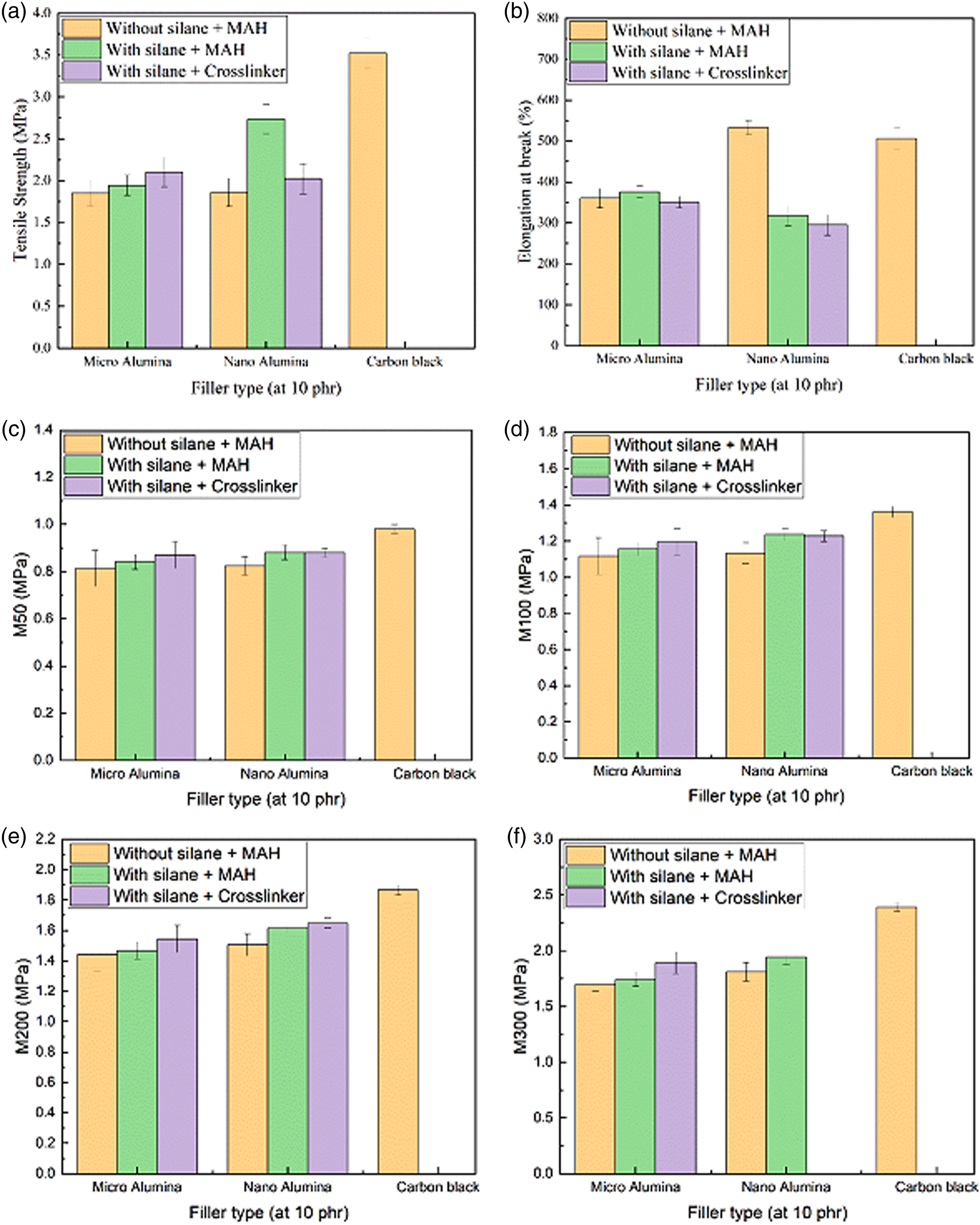
Morphology of EPDM/SBR/Al2O3
SEM analysis
The mechanical properties of EPDM/SBR/Al2O3 nanocomposites were determined by the dispersion of nanofillers in the rubber blend matrix. The uniform distribution of nanofillers enhanced the properties of the nanocomposite.7,44 The aggregated nanofillers developed a stress concentration point, deteriorating the properties of the composite materials.
45
Figure 8 shows the SEM images of the fracture surfaces of composite rubbers reinforced with CB and Al2O3 without or with surface treatment. The size of untreated Al2O3 agglomerates in the EPDM/SBR/Al2O3 nanocomposites was much larger than that in vulcanizates with treated Al2O3, as shown in Figure 8(b) and (c). The smaller aggregate size in the latter correlates well with the effect of interface modification on decreasing filler–filler interactions (caused by polar hydroxyl groups) and increasing filler–polymer interactions, thereby obtaining a better dispersion of treated alumina compared with that of the untreated alumina in the EPDM/SBR rubber matrix. These findings are in agreement with previous studies.
30
SEM images of (a) EPDM/SBR (50/50) blend without alumina, (b) EPDM/SBR/untreated micro-Al2O3 (50/50/25) blend, (c) EPDM/SBR/treated micro-Al2O3 (50/50/25) blend, (d) EPDM/SBR/CB (50/50/10) blend, and (e) EPDM/SBR/treated micro-Al2O3 + crosslinker (50/50/25) blend.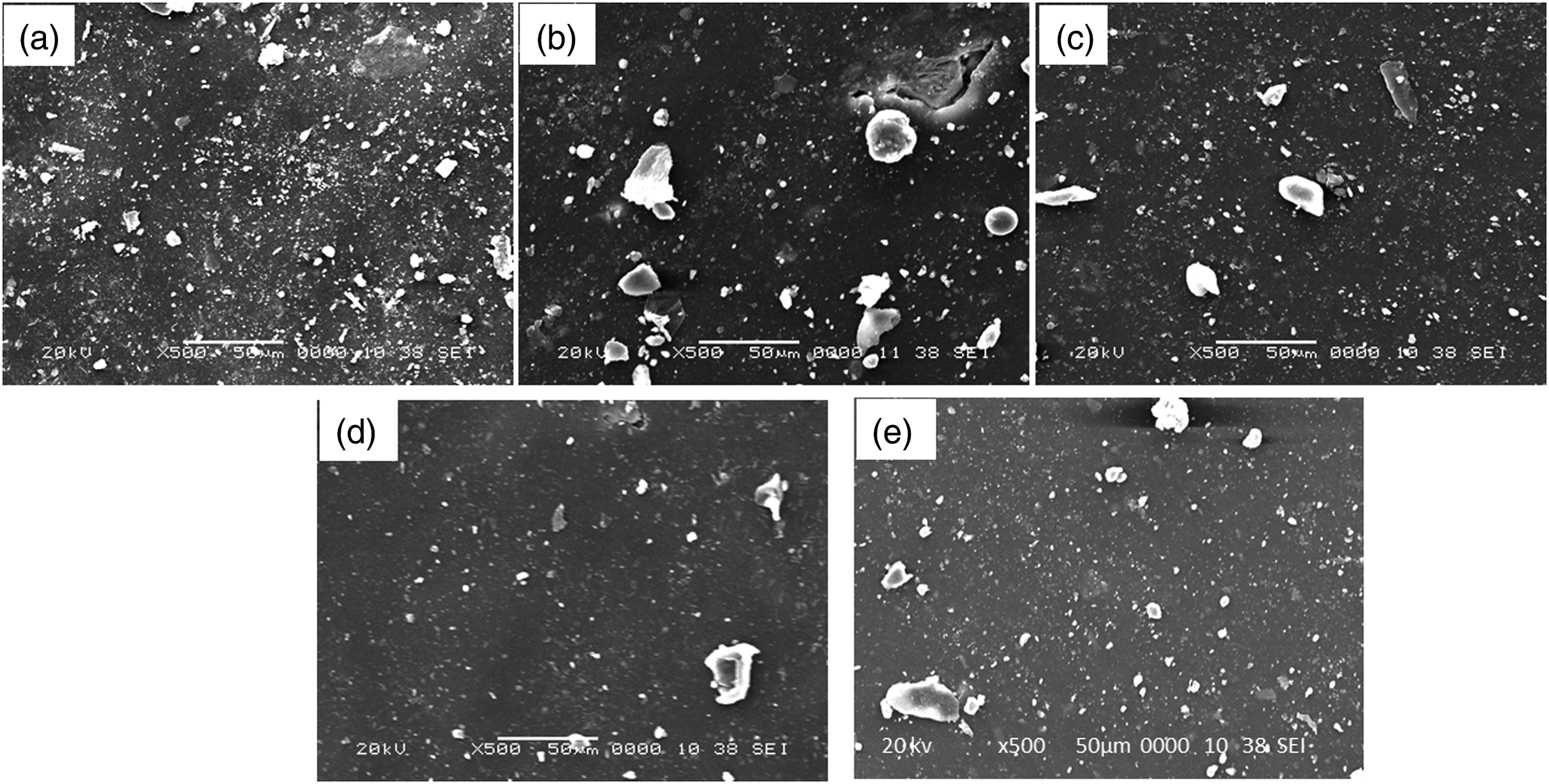
Silane modification is a well-accepted strategy to improve filler dispersion and filler–polymer interfacial bonding. TESPT reduces friction among the Al2O3 particles.5,27 In the case of the CB-filled EPDM/SBR/CB vulcanizate, although the state of dispersion was worse than that of the treated alumina-filled vulcanizates, as shown in Figure 8(d), it was better than that of the untreated alumina-filled vulcanizate, as shown in Figure 8(b). This result is due to more filler–polymer interactions at the CB surface than at the untreated Al2O3 surface, controlled by the hydrocarbon-based surface groups and the higher surface roughness of CB, causing more engagement between CB and rubber.51,53
Conclusion
In the current study, we prepared EPDM/SBR/Al2O3 nanocomposites by introducing nano-Al2O3 particles into the EPDM/SBR matrix. The nanocomposites with and without the silane coupling agent were compared based on the curing characteristics, mechanical properties, and morphological characteristics of EPDM/SBR nanocomposites.
From the experimental findings, the following conclusions were derived: 1. Silane-treated Al2O3 was much more effective in increasing the maximum rheometric torque (MH) of EPDM/SBR nanocomposites than the untreated Al2O3. 2. The crosslinker decreased the minimum torques of the treated Al2O3-filled EPDM/SBR rubber compounds. 3. TESPT also increased the maximum torques of the Al2O3-filled EPDM/SBR rubber compounds. The plasticizing and compatibilization effects of TESPT were more pronounced in Al2O3-filled EPDM/SBR rubber compounds, while the curing effect of TESPT was more pronounced in Al2O3-filled EPDM/SBR rubber compounds. 4. The plasticizing, compatibilization, and curing effects of the crosslinker were higher compared to those of TESPT on the Al2O3-filled EPDM/SBR compounds. 5. The tensile characteristics such as strength and elongation at break (%) of the EPDM/SBR nanocomposites without TESPT increased with the increase in the content of nano-Al2O3. 6. At 8 phr, the nanocomposites without the silane coupling agent exhibited a 45.29% and 103.32% increase in tensile strength and elongation at break (%), respectively, compared with the neat rubber blend.
Footnotes
Acknowledgements
The authors would like to thank the Polymers and Pigments Department, National Research Centre, Cairo, Egypt, and also the Mechanical Engineering Department, Benha Faculty of Engineering, Benha University, Benha, Egypt.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
