Abstract
In this study, different amounts of boric acid (BA, 1.25, 2.5, 3.75 and 5.0 wt%) were used to enhance the effectiveness of an intumescent flame retardant (IFR) system composed of ammonium polyphosphate (APP) and pentaerythritol (PER) in polypropylene (PP) including 2 wt% montmorillonite nanoclay (MMT). Meanwhile, metaboric acid and boron oxide which were generated by the decomposition of BA appeared in the melt compounding and the burning processes, respectively. Extensive experimental studies were performed to investigate the effects of BA/boron oxide and MMT combinations on the properties of PP/IFR. The fire resistances of the composites were studied by UL 94, limiting oxygen index (LOI) and cone calorimetry tests. The thermal properties were determined by using thermogravimetric analysis, differential scanning calorimetry and thermal conductivity measurements. In addition, the mechanical properties of the composites were examined. The experimental results revealed that although the additions of 1.25 and 2.5 wt% BA with 2 wt% MMT significantly enhanced thermal and flame resistances of PP composites, 3.75 and 5.0 wt% BA additions generated antagonistic effects and deteriorated the fire resistance of the composites. The sample including 2.5 wt% BA addition achieved the best flame retardancy. The LOI value was increased from 18 to 31% with UL 94 V-0 rating. In addition, the peak heat release rate was reduced from 668.6 to 150.0 kW/m2 and the total heat release value was decreased from 247.9 to 98.4 MJ/m2. In the meantime, the thermal conductivity was increased from 0.22 up to 0.28 W/mK. Furthermore, CO, CO2 and the smoke productions were significantly decreased with respect to PP. NO generation was decreased with BA replacements. At the same time, although there was a slight decrease in the tensile strength, the flexural strength significantly increased with BA and MMT additions.
Introduction
Polymeric materials are widely preferred in many areas due to their superior properties. Among them, PP is extensively used in many fields, such as transportation, building, automotive, electronics-electric, packaging, textiles, and housing. Because, it has a good balance between the cost and the superior properties like good chemical resistance and high mechanical strength.1–3 However, PP is a flammable material with a LOI of about 18% and it burns in standard air conditions with a very hot and clean flame without leaving any char residue because of its wholly aliphatic hydrocarbon structure.4,5 Its inherent flammability restricts its usage in most applications. For this reason, flame retardants are usually added during compounding process to improve the fire retardancy of PP.6–12
The incorporation of flame retardant additives has proved to be an effective way to reduce their flammability. The halogenated organic compounds are good fire retardant additives for PP, especially synergizing with antimony trioxide. However, the halogenated flame retardant added PP composites have serious disadvantages such as the evolution of toxic gases and corrosive chemical fumes. 13 Thus, researchers have examined on halogen-free flame retardants. Morgan 14 reviewed existing flame retardant chemical technology and discussed the future of flame retardant polymers. In recent years, IFR systems have been investigated as the effective halogen-free flame retardants.15–30 In general, an IFR system mainly consists of three compounds, namely an acid source, a blowing agent, and a carbonization agent.31–33 Sometimes, a substance may have multiple effects. APP and PER have been widely used to synthesize an intumescent flame retardant system. APP acts both as an acid source and as a blowing agent during thermal degradation and burning of the materials. Meanwhile, PER that is easily dehydrated by the acid acts as a carbonizing agent. 34 An IFR generates a barrier effect of an intumescent carbonaceous char created by the combined action of the compounds.35,36 The char layer reduces heat transfer between the heat source and the material surface, and limits combustible gas transfer from the material toward the flame as well as the diffusion of oxygen into the material. In addition, the char layer may prevent dripping from the material.37–43 IFR systems have been added into polymer composite materials at different ratios, between 5 and 30 wt%. Researchers have worked to find optimum APP/PER ratio in IFR systems for different polymers. Demir et al. 44 investigated the incorporation of IFR systems composed of APP and PER in 4/1, 3/1, 2/1 and 1/1 ratios into PP. They reported that 3/1 ratio satisfied the best flame retardancy.
Some inorganic fillers such as zeolite, clay and boron compounds have been used to improve the effectiveness of IFR systems.45–48 In addition, the use of some fillers is a common practice in the plastic industry to reduce the production costs of products. Meanwhile, some fillers may enhance the mechanical properties of the polymers.49–51 Clay has been used with traditional flame retardants to generate synergistic effects on flame resistance of polymers.52,53 Among different clays, montmorillonite has been attracted much attention to improve the flame retardancy of PP and small amounts of montmorillonite could play the role of protecting the polymer matrix from heat and flame.54–62 Zhu et al. 54 studied effects of combination of montmorillonite and IFR system on the flame retardancy and the thermal properties of PP composites. The amounts of the montmorillonite incorporated into the system varied between 0.5 and 5.0 wt%. It was reported that increasing the montmorillonite content up to 3% improved the flame retardancy of PP. However, excessive montmorillonite deteriorated the flame retardancy. Rohlmann et al. 55 pointed out that the clay improves thermal and mechanical properties and flame resistance of polymers with reducing gas permeability. The clay transports from the interior of the polymer toward to the surface during combustion and accumulates at the surface of char layer. The clay enhances char layer during combustion and protects the inside resin. Furthermore, Liu et al. 57 studied effect of the montmorillonite on IFR. It was suggested that the montmorillonite could act as a promising flame retardant additive to PP systems.63–65 To enhance effectiveness of flame retardants, boron compounds have been used in the IFR systems. Boron compounds promote char formation with IFR systems in the burning process. It was revealed that boron substances could improve the flame retardant effectiveness and char-forming ability of IFR systems.16,66–68 The addition of boron containing substances shows a synergistic effect with IFR systems and increases LOI values of polymer composites.69–71 Jimenez et al. 72 studied intumescent fire protective coatings including APP and BA to protect steel in the case of fires. They reported that BA addition enhanced the intumescent behavior of the coating. In addition, the mechanism of action of BA and APP in a commercial intumescent epoxy-based formulation was explained by Jimenez et al. 73 It was reported that the carbonization agent increased the thermal degradation rate of APP resulting in reactions of the degradation products of BA and the coated APP. As a result, borophosphate was formed and it increased mechanical resistance of the char, which improve the fire resistance. Moreover, Jimenez et al. 74 analyzed and simulated the kinetics of pyrolysis of an epoxy-based intumescent coating including BA and APP. Xu et al. 75 investigated the combined effects of phosphorus and boron flame retardants on flame retardancy, and smoke suppression of coatings applied on wood substrates. It was reported that incorporation of BA into the coatings generated a high quality and compact intumescent char which insulated the heat and mass transfers. The effects of BA with kaolin clay on thermal degradation of IFR coating were investigated and it was reported that BA could improve the fire resistance performance of the coating by increasing the residual mass of char. 76 Also, Ullah et al. 77 reported the effects of APP and BA on char expansion, heat shielding, char morphology and char composition after a fire test.
Dogan and Bayramli 34 investigated the combustion and thermal properties of polypropylene composites containing an IFR system and clay with boron containing substances; namely zinc borate, borophosphate and boron silicon containing preceramic oligomer. Zinc borate acts as a source of boron oxide (B2O3) that helps to form a glassy layer that prevents further combustion of char formed by IFR systems.78–80 Ayrilmis et al. 81 and Kurt and Mengeloğlu 82 investigated effect of boron compounds; namely BA, borax, and zinc borate, on fire properties of wood-polypropylene composites. Borax and BA were used as flame retardants which have a synergistic effect in high density polyethylene composites. 83 Recently, BA was studied as a flame retardant and a smoke suppressant in wood-polypropylene, polyamide, natural rubber, epoxy resin, polyethylene.84–87 Demirel et al. 88 studied the synergistic effect of BA in IFR based polyester resin with glass fiber and it was found that BA could improve the fire resistance performance of the polyester resin. They also reported that when BA was added up to 30 wt% into the polyester resin matrix, the limiting oxygen index (LOI) value of the composite could reach up to 34.5%. When the material is heated, boric acid generates a glassy boron oxide (B2O3) barrier on the surface of material.75,89 Li et al. 90 studied BA as an acid source based on epoxy resin. In addition to all these, recently, different studies have been carried out to provide more uniform and compact char by using different materials such as ferrocene-based polymer, aluminum phosphate, organo-modified sepiolite, triazine derivative-modified nano-SiO2 and functionalized zirconium phosphate with IFR systems in PP composite materials.91–94
In this study, the effects of BA and MMT combinations were investigated to enhance the thermal degradation and flame retardancy of PP incorporated with an IFR system that is composed of APP and PER. To the best of the authors’ knowledge, although BA and MMT were used separately at different ratios in different polymers and PP composite materials, synergistic effects of BA and MMT combinations in PP / IFR composites have not been extensively studied in the literature. In addition, since BA was considered as an acid source and a blowing agent in IFR system, the amounts of BA (1.25, 2.5, 3.75 and 5.0 wt%) were carefully selected to find optimum value of BA in the system. The same amount of APP was reduced while adding the BA into the system. To the best of our knowledge, no study on this approach for PP has so far been reported in the literature. Extensive experimental studies were performed to investigate the effects of BA and MMT combinations on the flammability, burning, thermal and mechanical properties of PP/IFR system.
Materials and methods
Raw materials
The polypropylene (PP) (Petoplen MH-418) granulated product with a melt flow index (MFI, 2.16 kg, 230°C) of 4.5 g/10 min was supplied by Petkim Co. Ltd. (Turkey). Ammonium polyphosphate (APP, n>1000), Exolit AP 423, (average particle size 8 µm) was obtained from Clariant (Turkey). Pentaerythritol (PER) powder (Melting point, 261–263°C) was supplied by MKS Marmara Integrated Chemistry Industry Co. Ltd. (Turkey). Nanoclay (Montmorillonite nanoclay, MMT) (esanNANO 1-140) was provided by Eczacıbaşı Esan Co. Ltd. (Turkey). Boric acid (BA, H3BO3) from the boron products in the form of solid crystalline powder was obtained from Eti Mine Works General Directorate (Turkey).
Preparation of flame retardant PP samples
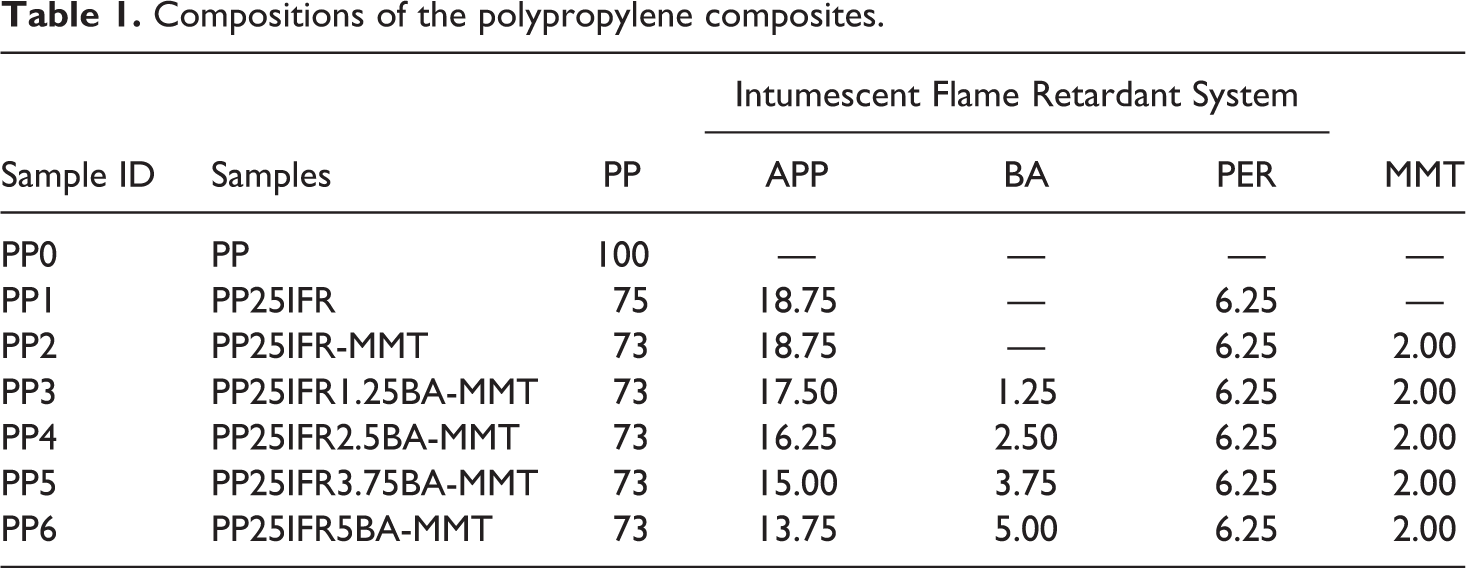
PP, APP, PER, MMT and BA were subjected to oven drying at 100°C for 24 h before extrusion production. PP and the other materials were melt-mixed in a twin-roller mill extruder (screw speed 200 rpm, feed rate 20 rpm, screw length/diameter L/d 1000/20 mm and production temperature 175–200°C) and cut into pellets using a granulator.41,80,91,95 The water vapor which was released from BA was thrown into the atmosphere during the melt compounding and the metaboric acid was generated. The loading of IFR system composed of APP and PER in the ratio of 3/1 by weight was fixed at 25 wt%.38–43 MMT as a filler and char reinforcer was added in 2 wt% by reducing the same amount of PP. 34 BA as an acid source and blowing agent was replaced with APP in different mass fractions of 1.25, 2.5, 3.75 and 5 wt%. The amount of APP was reduced by the amount of BA added. The compositions of the samples are given in Table 1. The ratios of BA in IFR systems were 5, 10, 15 and 20%. The composite materials in the form of extruded granules were dried in an oven at 100°C for 6 hours before the injection molding process. The samples used in UL 94, LOI, the cone calorimeter and the mechanical tests were produced by using a plastic injection machine and a special mold.
Compositions of the polypropylene composites.
Thermogravimetric analysis (TG/DTG)
Thermal degradation behaviors of PP and its composites were determined by using a Perkin Elmer Diamond thermogravimetric analysis system. Samples (approximately 10 mg) were tested in a nitrogen gas environment at a flow rate of 200 ml/min, at a temperature range of 30–1000°C with a temperature increase of 20°C/min, using ceramics pans.96,97
Differential scanning calorimetry (DSC)
The thermal behaviors of the composite materials were determined by using a Perkin Elmer Pyris 6 DSC instrument. Approximately 10 mg samples were tested in a nitrogen gas environment at a flow rate of 200 ml/min, within the temperature range of −50°C to +200°C with a temperature increase of 10°C/min, using aluminum pans.
79
Degree of crystallinity of PP varies from about 50% to about 70%. Degree of crystallinity (

where, ΔHm is the measured melting enthalpy and was obtained from the DSC test, ΔHm° is the theoretical specific melting heat of 100% crystalline PP (209 J/g) and wp is the weight fraction of PP in the composite material. 80
Thermal conductivity test
The thermal conductivities of the samples were measured by using a QTM-500 Quick Thermal Conductivity Meter by Kyoto according to ASTM C1113. The sample size was 100 mm × 100 mm × 20 mm. At least three measurements were performed for each sample and their mean values were calculated and presented.
Limiting oxygen index (LOI) and vertical burning tests (UL 94)
LOI and UL 94 vertical burning tests are widely used practical tests to evaluate fire resistance of polymers.98,99 The LOI values of the samples were measured by using a Qualitest LOI Analyzer. Dimensions of the samples were 100 mm × 6.5 mm × 3 mm according to ASTM D2863-17 standard. The UL 94 tests were carried out using a vertical burning instrument according to ASTM D 3801. The UL 94 vertical burning test results provide information on ignition, progression of combustion, dripping and extinguishing. The sample dimension was 125 mm × 12.7 mm × 3 mm. Materials shall be classified V-0, V-1, or V-2 on the basis of results obtained on small bar specimens. All samples were conditioned at 23 ± 2°C and a relative humidity of 50 ± 5% for 24 h prior to testing. The results were averaged from the three specimen tests.
Cone calorimeter tests
The cone calorimeter tests were carried out by using a cone calorimeter instrument in accordance with the ASTM E-1354 standard. Each specimen (100 mm × 100 mm × 6 mm) was wrapped in aluminum foil and exposed horizontally to an external heat flux of 35 kW/m2. All materials were conditioned at 23 ± 2°C and a relative humidity of 50 ± 5% for 24 h prior to testing. The emissions (CO2, CO, NO, O2), temperatures and mass losses were recorded in 1 s increment via a special software. The cone calorimeter results are averages of the three experiments.
Mechanical properties
Mechanical properties (tensile and flexural strengths) were measured by using an Instron tester 8801. The tensile strengths were determined according to standard ASTM D-638 at a testing crosshead speed of 50 mm/min. The flexural strengths were measured according to standard ASTM D-790 at a crosshead speed of 50 mm/min. The specimen dimension was 125 mm × 13 mm × 3 mm. All samples were conditioned at 23 ± 2°C and a relative humidity of 50 ± 5% for 24 h prior to testing. The results were the average values of at least five measurements.
Sample morphology
A scanning electron microscopy (SEM) (Zeiss gemini, Supra 40VP) equipped with EDX spectrometers was used to examine the dispersion of the additives and morphology of the fracture surfaces. SEM pictures were obtained at the voltage of 10 kV. The specimens were coated with gold before analysis.
Results and discussion
Limiting oxygen index (LOI) and UL 94 testing of PP and its composites
UL 94 vertical burning and LOI tests are practical tests and widely performed for acceptance or rejection of raw materials by the companies apart from researchers. In general, UL 94 V-0 rating is primarily requested for composite materials that will be used in industrial applications. 100 The results of LOI and UL 94 vertical burning tests of the samples are listed in Table 2. As it is known, the flammability properties of pure PP are poor.8,57,58 As it is expected that the LOI value of PP (PP0) was determined as 18% and it was not rated (NR) in the UL 94 test. 21 Among the samples, the four samples (PP1, PP2, PP3, PP4) satisfied UL 94 V-0 rating. Therefore, LOI tests of these samples were performed. Figures 1 and 2 show the images of the samples after UL 94 and LOI tests, respectively. The generated char layers at the ends of the samples exposed to the flame are clearly visible. Since PP0, PP5 and PP6 samples completely burned during the UL 94 tests, the images of their residues could not be given in Figure 1. PP1 which includes 25 wt% IFR satisfied UL 94 V-0 rating with LOI value of 29%. In addition, PP2 including 25 wt% IFR and 2 wt% MMT showed better flammability resistance in terms of LOI test with 30% due to stronger char layer generated by MMT.54,55,61
LOI and UL 94 values of PP and its composites.
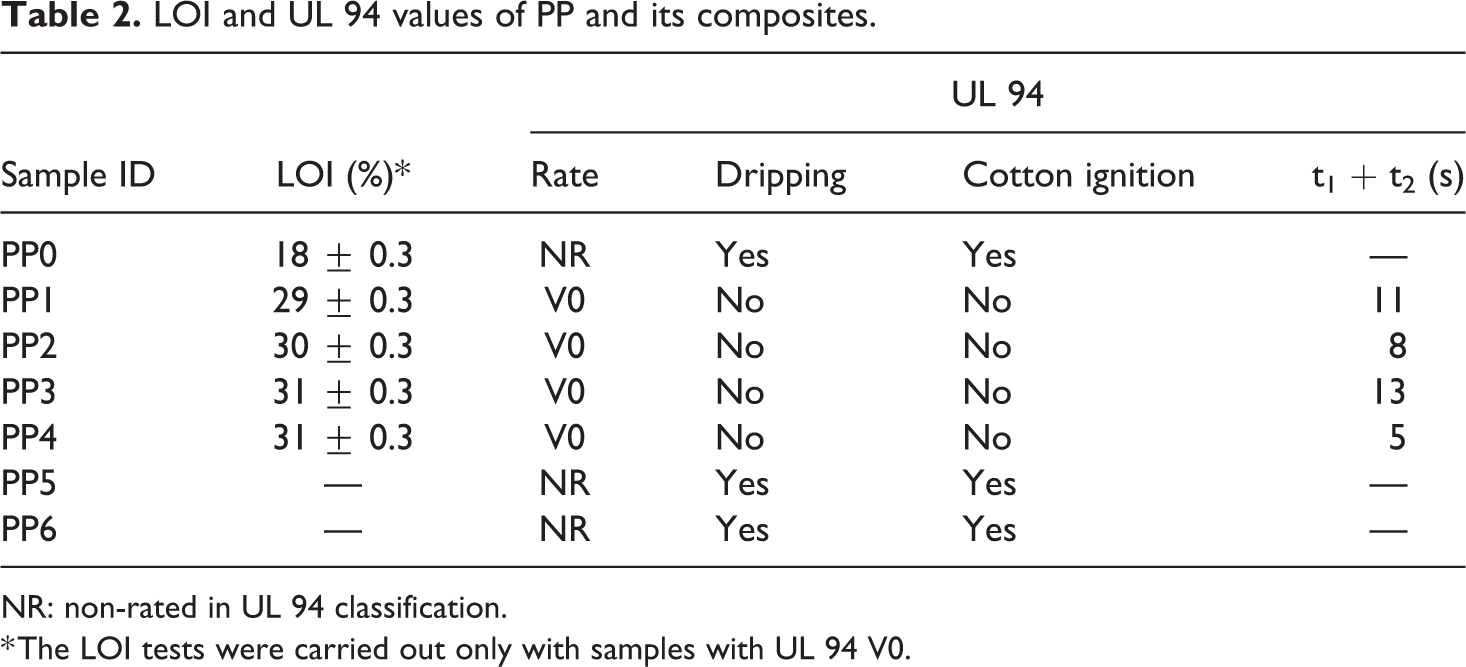
NR: non-rated in UL 94 classification.
* The LOI tests were carried out only with samples with UL 94 V0.

Images of PP and its composites after UL 94 vertical burning test.
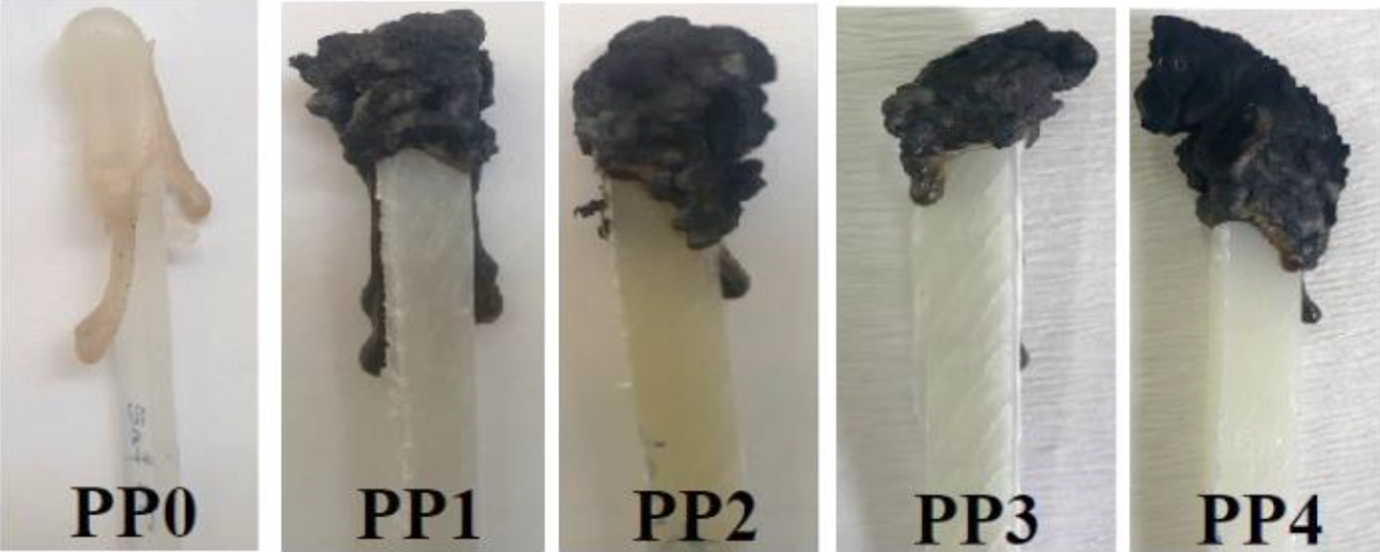
Images of PP and its composites after limiting oxygen index (LOI) test.
When 1.25 and 2.5 wt% BA were replaced with APP in the matrix, the LOI values of the samples PP3 (1.25 wt% BA) and PP4 (2.5 wt% BA) increased from 30% to 31%, and they also met UL 94 V-0 rating. However, the samples PP5 and PP6 which were obtained by replacing 3.75 and 5.0 wt% BA, respectively, completely burned and did not achieve UL 94 V-0 rating. They were not even rated (NR) in the UL 94 test. Although, the partial substitution of BA at low ratios (1.25 and 2.5 wt%) improved the fire resistance of the materials, this situation was reversed when the ratio was increased to 3.75 and 5 wt%. In other words, the effective char layers appeared at the ends of PP3 and PP4 samples were not generated in PP5 and PP6, and they completely burned. A similar situation was reported by Unlu et al. 69 The detailed discussion on this subject will be given in the following sections.
Thermogravimetric analysis (TG/DTG) of PP and its composites
Thermal decomposition behaviors of the raw materials (APP, PER, BA, MMT, IFR2.5BA-MMT PP) and the composites were investigated by thermogravimetric analysis. TG/DTG curves of the raw materials are presented in Figure 3.
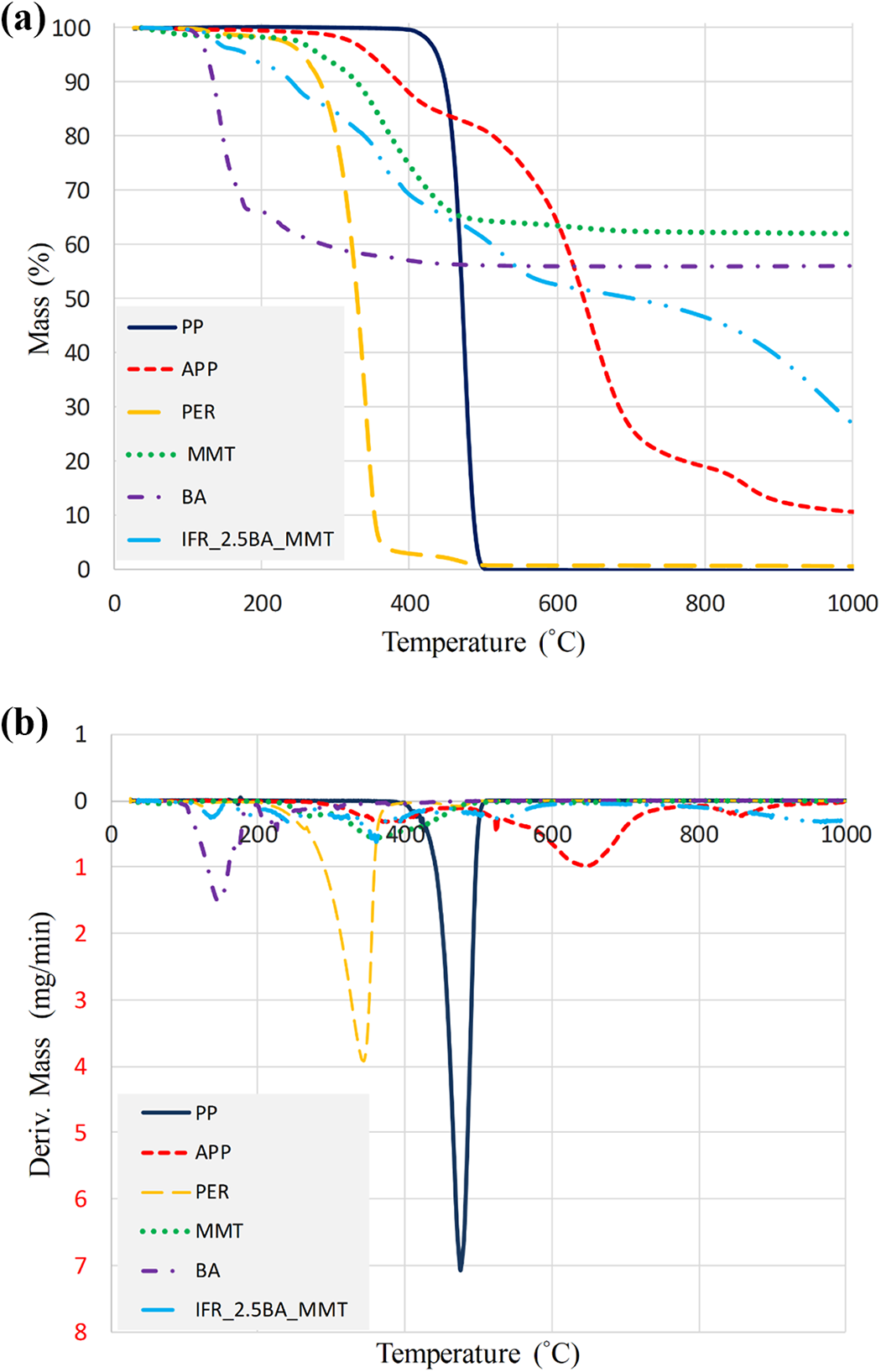
(a) TG and (b) DTG thermograms of PP, APP, PER, MMT, BA and IFR2.5BA-MMT.
PP has a low melting point (164°C) polymer that cannot withstand at higher temperatures. Due to its aliphatic hydrocarbon structure, polypropylene burns very rapidly. During the heating of polypropylene, a decrease in molecular weight is observed between 230 and 250°C while volatilization becomes significant above 300°C. 101 In the case of neat PP, the thermal decomposition behavior exhibited a single stage with almost 100% weight loss around 500°C. For APP, two main weight loss stages centered at 375°C and 645°C were observed. The first step was attributed to the elimination of NH3 and H2O and the formation of cross-linked polyphosphoric acids. 31 The second weight loss stage was due to the evaporation of polyphosphoric acids and/or dehydration to phosphorus pentoxide (P4O10).31,102 PER was completely degraded in a step starting at about 142°C and completing at 500°C and maximum degradation temperature was determined as 345°C.
MMT exhibited a maximum decomposition temperature of about 364°C between 255°C and 450°C. The residue of MMT at 1000°C was 62 wt%. The decomposition of BA (H3BO3) started at approximately 100°C with evaporation and release of water resulting of metaboric acid (HBO2) formation. It completed at approximately 600°C. Consequently, BA lost all its water, and it was entirely transformed to boron oxide (B2O3).103,104 In addition, the interaction between components of the flame retardant IFR2.5BA-MMT (APP/BA/PER/MMT: 16.25/2.5/6.25/2) is clearly seen in Figure 3.
Figure 4 compares the thermal decomposition behaviors of PP and its composites. The data of the char residue and the decomposition temperatures are summarized in Table 3. There are 5% and 50% weight loss temperatures (T5 wt% and T50 wt%), amounts of the char residues at 1000°C, maximum weight loss temperature (Tmax) and maximum weight loss rate (Rmax).
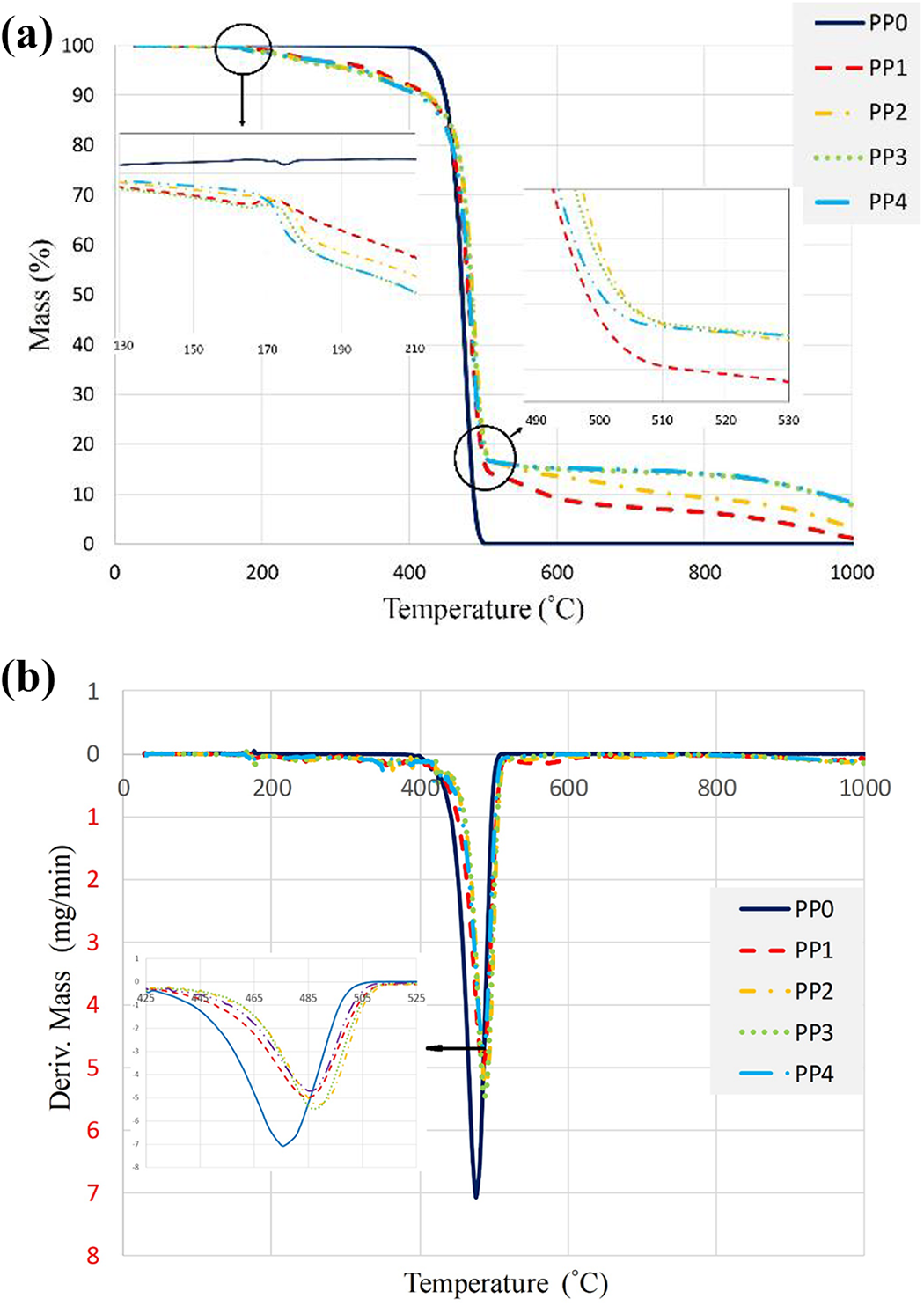
(a) TG and (b) DTG thermograms of PP and its composites.
Thermal degradation values of PP and its composites under nitrogen.
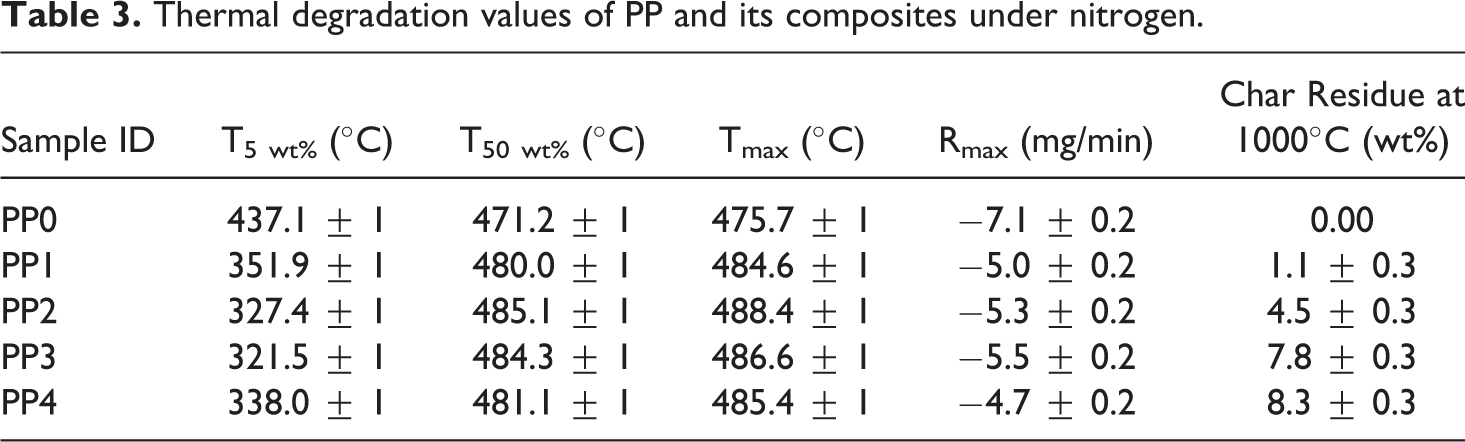
PP began to decompose at 400°C in nitrogen and the weight loss increased rapidly until all PP diminished at about 500°C, with no residual char left. The addition of IFR into PP (PP1) resulted in early decomposition of the composite material and the generation of a carbonaceous charred layer. This layer is a physical protective barrier on the surface of the materials that can insulate the underlying PP matrix from the heat source and slow down heat and mass transfers between the gas and condensed phases.60–63 However, the effect of the carbonaceous charred layer decreases at the high temperatures, the residues were 6.3 wt% at 800°C and 1.1 wt% at 1000°C. The incorporation of MMT (2 wt%) into PP1 (PP/IFR) did not significantly modify the decomposition trend, however, the amount of the char residue of PP2 (PP/IFR/MMT) was higher than that of PP1. The char residue increased from 1.1 wt% to 4.5 wt% at 1000°C.
Meanwhile, BA was added to PP2 (PP/IFR/MMT) by reducing APP content from the composite material. Although the additions of BA did not cause significant changes in the decomposition of the material, 1.25 wt% and 2.5 wt% BA additions resulted in increasing of char residues up to 7.8 wt% and 8.3 wt% at 1000°C, respectively. This suggests that the synergistic effect exists between MMT/IFR and MMT/BA/IFR, which can promote the composite to form a thermally stable barrier to prevent heat and gas transformation.60,105 The increase of thermal stability is attributed to the hindered diffusion of volatile decomposition products within the composites.54,56
Differential scanning calorimetry test (DSC) of PP and its composites
DSC scanning processes were carried out in the temperature range between −50°C and 200°C. The results of PP composites are presented in Table 4. The melting temperature (Tm) of PP was determined as approximately 164.4°C. By analyzing the thermograms of the composite samples, it could be concluded that IFR, MMT and BA additions did not significantly change Tm of PP. IFR, MMT and BA filled PP composites were found to have slightly higher crystallization temperatures than the polypropylene. These results show that IFR, MMT and BA act as nucleating agents increasing the crystallization temperature of PP. 106 The melting enthalpy (ΔHm) and crystallization enthalpy (ΔHc) of PP were 105.8 J/g and 112.2 J/g. ΔHm and ΔHc decreased with additions of IFR, MMT and BA. This result indicates that thermal stability of PP increased with increasing the filler content. 53 Glass transition temperature (Tg) of the samples decreased with incorporation of IFR, MMT and BA.
Thermal parameters of PP and its composites during melting and crystallization.
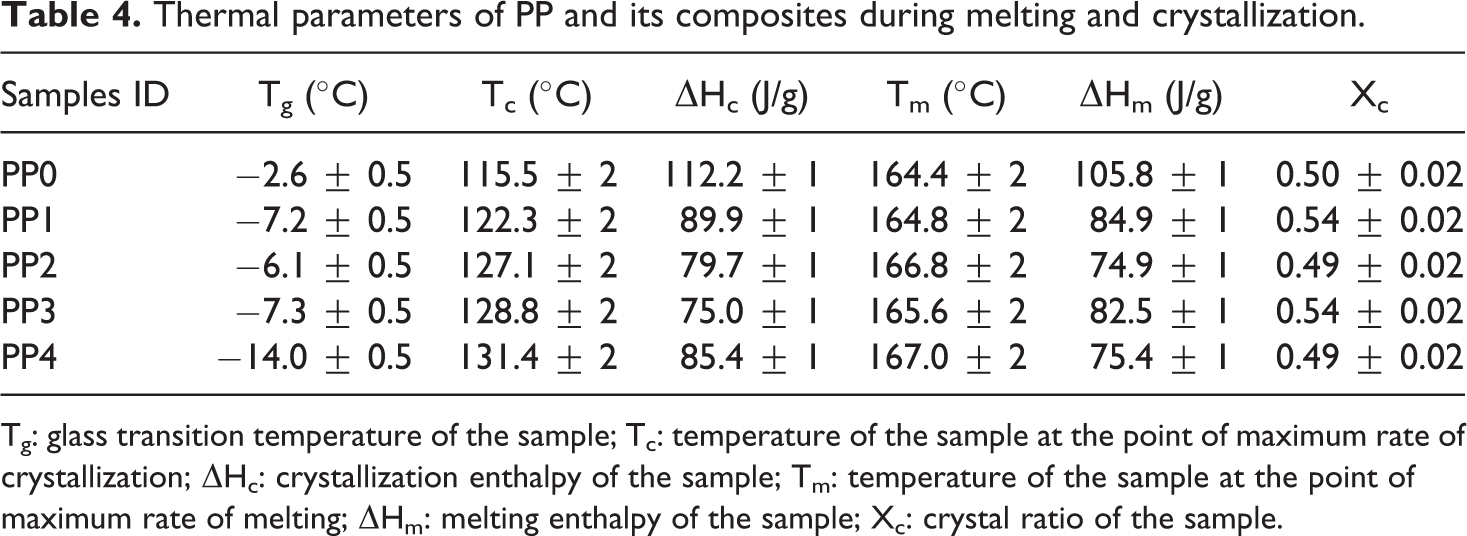
Tg: glass transition temperature of the sample; Tc: temperature of the sample at the point of maximum rate of crystallization; ΔHc: crystallization enthalpy of the sample; Tm: temperature of the sample at the point of maximum rate of melting; ΔHm: melting enthalpy of the sample; Xc: crystal ratio of the sample.
Cone calorimeter tests of PP and its composites
Cone calorimeter tests can give detailed information about the fire retardancy of the composites and the results may be very valuable to understand why a material passes or fails a particular UL 94 rating. 107 All the samples were tested in the cone calorimeter under 35 kW/m2 heat flux and the changes of heat release rate (HRR), total heat release value (THR), extinction coefficient, CO, CO2 and NO emissions with respect to the time were determined. As it was shown in the presentation of UL 94 test results, there were synergistic and antagonistic effects of BA on the flame retardancy of the intumescent flame retardant polypropylene composites. Although, the partial substitution of BA (PP3 and PP4) with APP in IFR system at lower ratios (1.25 and 2.5 wt%) had synergistic effect on the flame retardancy, 3.75 and 5 wt% BA substitutions (PP5 and PP6) exhibited an antagonistic effect on the flame retardancy. Initially, PP3 and PP4 which exhibited synergistic effects on the flame retardancy are compared with PP0, PP1 and PP2 in Figure 5. Since the cone calorimeter system can measure all parameters at every second, the lines of calculated and measurement parameters could be drawn precisely. However, the markers were added at 25 seconds intervals for the sake of clarity. Meanwhile, the time to ignition (TTI), the peak heat release rate (PHRR), the effective heat of combustion (EHC), the average mass loss rate between 10% and 90% of the mass loss (MLRav,10-90) and the total heat release values (THR) for 1000s are presented in Table 5.
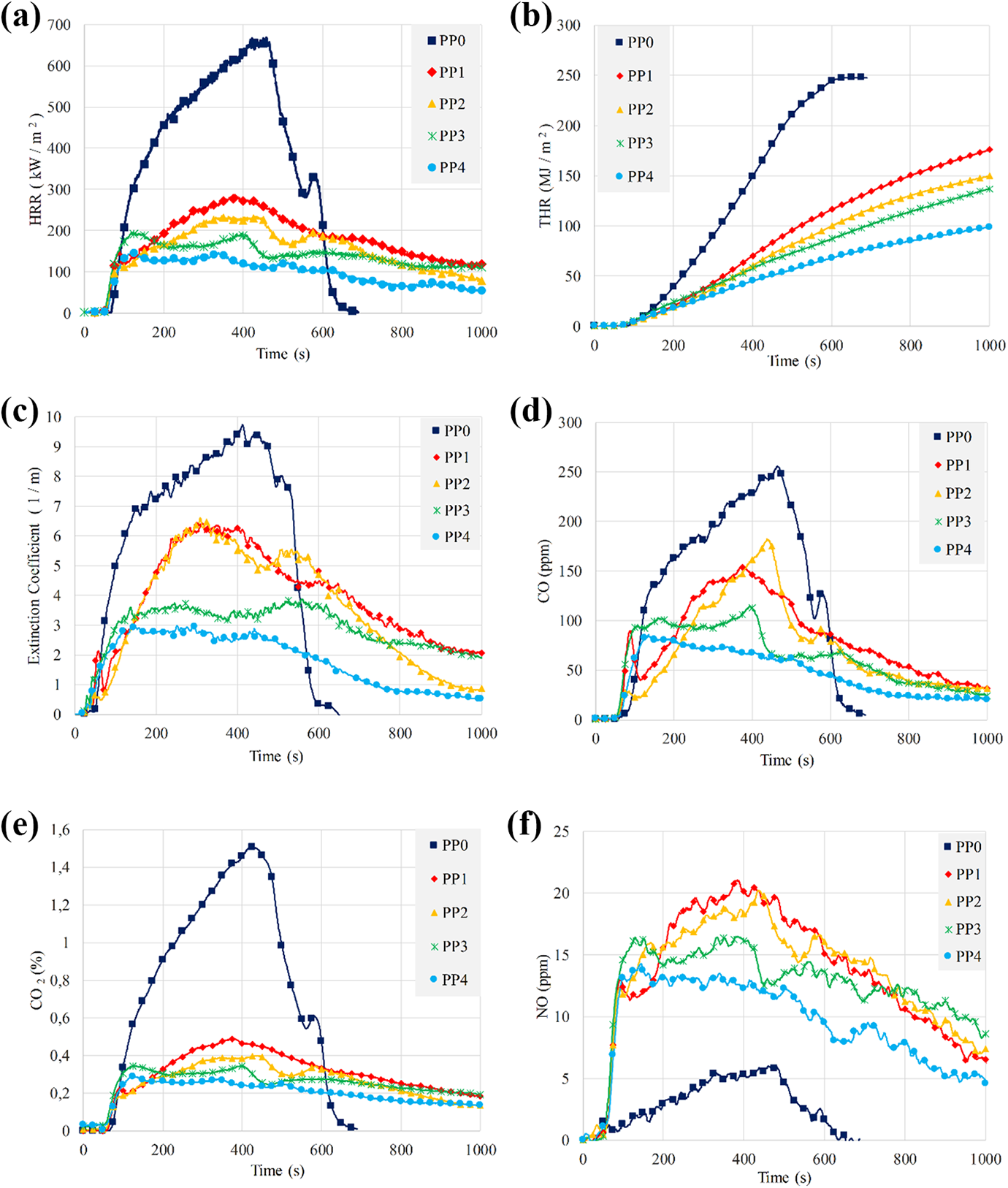
Cone calorimeter results of PP and its composites: (a) HRR, (b) THR, (c) extinction coefficient, (d) CO, (e) CO2, and (f) NO.
Cone calorimeter results of PP and its composites.
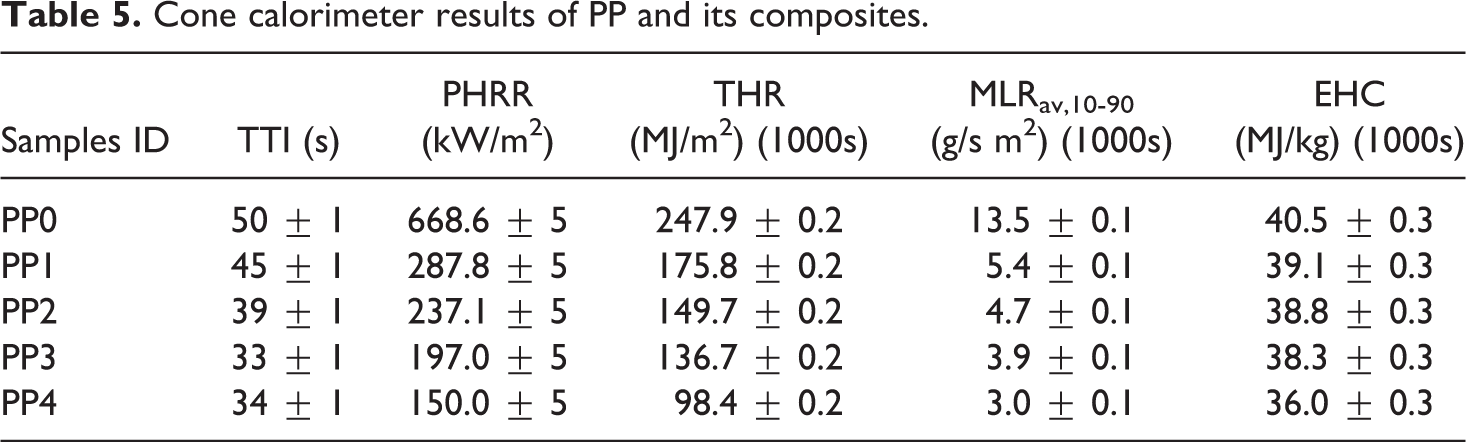
PP rapidly burned after the ignition, and PHRR and THR values reached to 668.6 kW/m2 and 247.9 MJ/m2, respectively, with no residual char left.8,21,57 It can be seen from Figure 5(a), significant reductions in HRR values and maximum heat release rate values were achieved with the addition of 25 wt% IFR (PP1). TTI advanced approximately 5 s due to the earlier decomposition of IFR as expected.36,37 Meanwhile, the decomposition of IFR produced a char layer that could partially prevent the decomposition of the composite material and distinguish the burning substrates. It reduced the intensity of the flame and the rate of flame spread leading to ceasing the burning. Consequently, PHRR and THR decreased to 287.8 kW/m2 and 175.8 MJ/m2, respectively (Figure 5(b)). The addition of MMT into PP1 composite resulted in some more decreases in PHRR (237.1 kW/m2) and THR (149.7 MJ/m2). MMT can be used as a synergist for intumescent fire retardant and heat absorber.60,20 MMT has two different effects on composite materials. First, it forms a layer to prevent the material from heating from the surface; the latter catalyzes the degradation of the polymer matrix. 108 In addition, it was determined that the burning times of the composites were extended in comparison with that of PP. 71
When 1.25 wt% BA was replaced with the same amounts of APP in the matrix, the fire slowed down, and PHRR and THR values decreased to 197 kW/m2 and 136.7 MJ/m2, respectively. PHRR and THR continued to decrease to 150 kW/m2 and 98.4 MJ/m2 values when the amount of BA was increased to 2.5 wt%. The decreases in the MLRav,10-90 and the EHC values with the incorporation of BA are also shown in Table 5. In other words, 1.25% BA and 2.5 wt % BA revealed a better synergistic effect and enhanced the fire resistance of the polypropylene.
BA released water vapor in the endothermic reactions between 100°C and 200°C generating the metaboric acid (HBO2) and then a glassy boron oxide (B2O3) appeared between 200°C and 600°C due to the water vapor released from the metaboric acid.103,104 The released water vapor acted as a heat sink; meanwhile it diluted oxygen and combustible gases. Moreover, the glassy boron oxide reinforced the char layer.34,71,75,89,109,110 It is thought that the reinforced char layer effectively enhanced the fire resistance of the composites.111,112 Jimenez et al.72,73 reported that BA acts as an intumescent agent and the dehydration of BA leads the formation of a “glass-like” material (B2O3). This increases the viscosity of the melt and prevents the gaseous decomposition products escaping to feed the flame. Also, the reactions between BA and APP partially produced borophosphate which is a hard material and can satisfy good thermal stability. In addition, Ullah et al. 77 reported that the addition of boric acid reduces the char expansion, while it enhances the adhesion of the char to the material.
Soot and CO emissions are considered as emissions causing poisoning in cases of fires. 40 Soot and CO emissions are generated due to incomplete combustion of materials. 43 Soot, CO and CO2 emissions of PP and its composites are shown in Figure 5(c), (d) and (e). Addition of IFR, MMT and BA into PP caused significant reductions in the soot, CO, and CO2 emissions due to enhancing the charring ratio. 57 NO emissions of PP and its composites are shown in Figure 5(f). IFR additions increased NO emissions of PP due to nitrogen content of APP. However, BA substitutions slightly decreased NO emissions due to decreasing of APP in the matrix. Figure 6 shows the views of the samples after the cone calorimeter tests. Although PP completely burned, its composites partially burned. The char residues of the composites are clearly seen in Figure 6.

Images of PP and its composites residues after the cone calorimeter tests: (a) PP0, (b) PP1, (c) PP2, (d) PP3, and (e) PP4.
To examine the antagonistic effect of 3.75 and 5 wt% BA substitution (PP5 and PP6), HRR variations of PP5 and PP6 are compared with those of other samples in 300 s in Figure 7. It is seen that there were some delays in the decomposition and the ignition of PP5 and PP6 with respect to PP3 and PP4. Therefore, the effective char layers of PP5 and PP6 were not generated in time as those of PP3 and PP4. In other words, the effective char layers which could reduce the intensity of the flame and the rate of flame spread leading to ceasing the burning were not formed. PHRR values of PP5 and PP6 reached to 327.8 kW/m2 and 431.0 kW/m2, respectively.
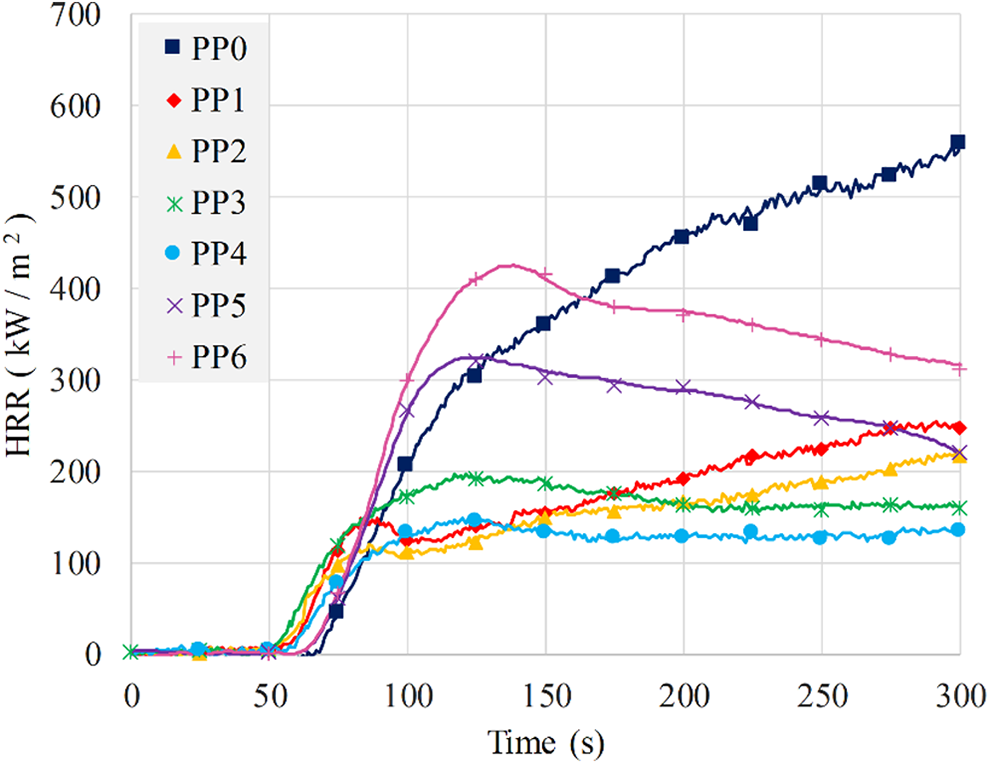
HRR of the samples in the first 300 s.
The burning processes of PP5 and PP6 were compared with PP4 in Figure 8. Although the char layer was formed during the burning of PP4, it was not generated in the burning of PP5 and PP6. In other words, after the ignition, the effective char layers could not be quickly generated in the burning of PP5 and PP6. The burning process was stopped at the time of 300 s after the samples were exposed to the heat flux. The pictures of the residues are shown in Figure 9. Although the char layer is clearly seen in the pictures of PP4, similar char layers are not seen in the pictures of PP5 and PP6.

Pictures of burning processes of (a) PP4, (b) PP5 and (c) PP6 in cone calorimeter test.

The residue pictures of (a) PP4, (b) PP5 and (c) PP6 at the end of 300 s.
The explanation of antagonistic effects of BA (3.75 and 5.0 wt% substitution) on the flame retardancy of PP/IFR composites may be explained with late-forming weak char layers. When the amount of BA was increased from 2.5 wt% to 3.75 and 5.0 wt% in IFR systems, the amount of boron oxide also increased in the melts. The excessive amount of boron oxide resulted in higher viscosity of the melt, and this prevented the effective char formation and expansion (see Figures 8 and 9). 77 Similar results were obtained by Unlu et al. 69 They reported that although 1 wt% BA addition generated synergistic effects in terms of flame retardancy of epoxy based intumescent coating, 3 and 5 wt% BA additions reduced the protective effect of the intumescent coating. This antagonistic effect was explained with the reduction of the char expansion and the poor mechanical strength of the char.
Thermal conductivity of PP and its composites
Figure 10 shows the thermal conductivities of PP and its composites. There are many factors affecting the thermal conductivity such as temperature, processing condition, polymer crystallinity and chain structure. 109 PP is a polymer with a low thermal conductivity. The thermal conductivity of PP was measured as approximately 0.22 W/mK.113,114 The addition of IFR, MMT and BA increased the thermal conductivity values of the composites. The thermal conductivity values of PP1 and PP2 were 0.25 W/mK and 0.24 W/mK, respectively. When 1.25 and 2.5% BA were added into the composites, the thermal conductivity values reached to 0.27 W/mK and 0.28 W/mK, respectively.115–117
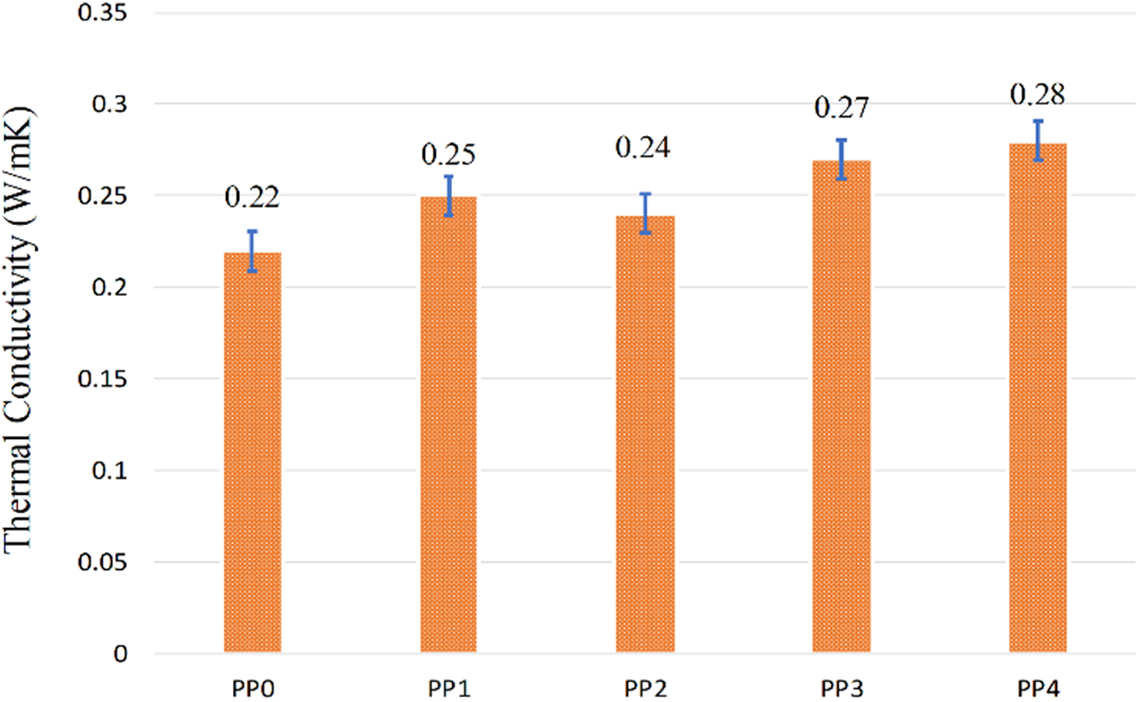
Thermal conductivities of PP and its composites.
Mechanical properties of PP and its composites
Beside the thermal and the fire resistance, the mechanical properties are also crucial indicators for evaluating the usage of PP composites in industrial applications.36,50,118,119 The tensile and the flexural strengths of PP and its composites are shown in Figure 11. The tensile and flexural strengths of PP (PP0) is 41.3 MPa and 44.2 MPa, respectively. The addition of IFR caused slight decreases in the mechanical properties. When IFR was added to PP0, the tensile and flexural strengths of PP1 were decreased by 8.7% and 5.7%, respectively, due to the partially poor compatibility, poor dispersion, and agglomeration of IFR in the polypropylene matrix.36,120–122 However, 2% MMT addition into PP1(PP/IFR) enhanced the mechanical properties. The tensile strength of PP2 became 41.3 MPa and the flexural strength increased to 54.7 MPa. It is thought that MMT could help the homogeneous dispersion of IFR in the matrix like a coupling agent. In addition, since MMT is nano-sized and, APP and PER are micron-sized, MMT could improve the mixing of APP and PER by filling the gaps between them. 123 Meanwhile, MMT could act as a plasticizer in the PP matrix, which is beneficial for the process properties of PP/IFR composites. 36 Manoharan and Selvakumar 124 reported that MMT layers serve as the short fasciculus and reinforce the PP matrix resulting in increases in tensile and flexural strength of the composites. With the loading of BA, although the tensile strengths of PP3 and PP4 slightly decreased, the flexural strengths of them increased by 8.6% and 6.4% increments compared with that of PP2, respectively. It is known that when the bending load is applied on the composite specimen, its top side is exposed to compressive stress while the bottom side is exposed to tensile stress. Since the tensile strengths of PP3 and PP4 decreased slightly, the flexural strength of the composites increased harmoniously. 120
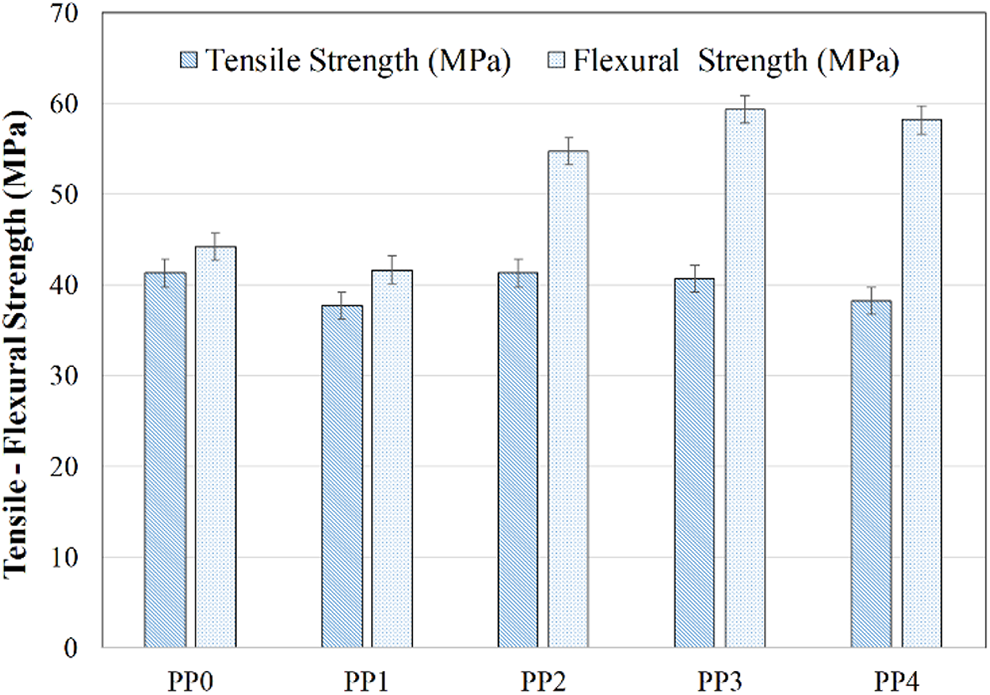
Tensile and flexural strengths of PP and its composites.
Scanning electron microscopy (SEM) of PP and its composites
SEM images of the fracture surfaces of PP and its composites are shown in Figure 12(a) to (e). It can be easily seen that the surfaces of the pure PP (PP0) are smooth and featureless. 124 However, the surfaces of PP composites are significantly different from that of PP0. Small agglomerations of IFR appear on the surface of PP1. Meanwhile, the additions of MMT particles improved the distribution of IFR. The particles were homogeneously distributed in the matrix of PP2, PP3 and PP4. There are no significant agglomerations on the surfaces of PP2, PP3 and PP4. It is thought that the homogeneous distribution is an important factor preventing deterioration of the mechanical properties and enhancing of the fire resistance. In addition, the chemical composition results determined by using EDX are given in Figure 12(f). It shows that carbon, oxygen, boron, nitrogen, phosphorus, and silicon are present in PP4.
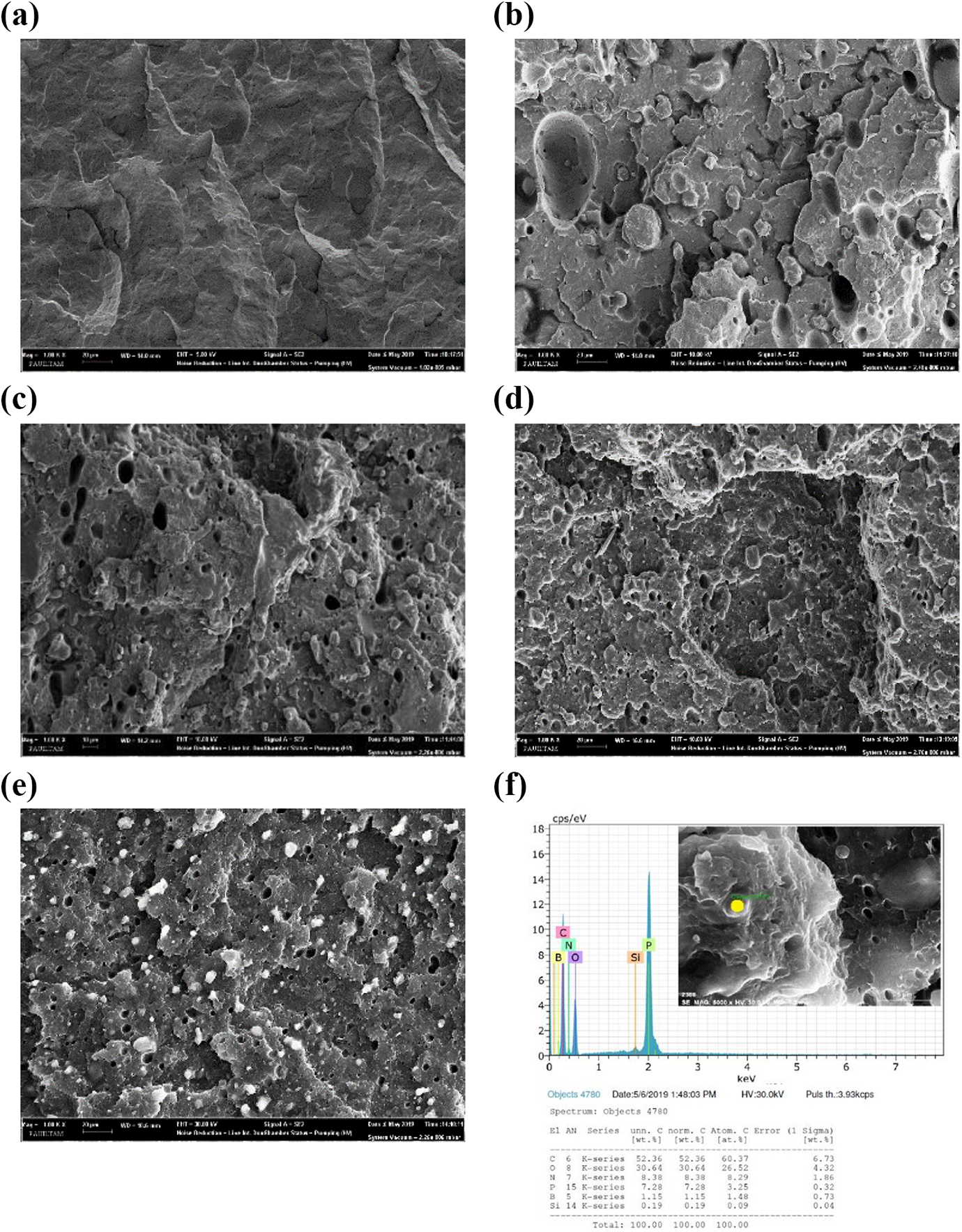
SEM micrographs of (a) PP0, (b) PP1, (c) PP2, (d) PP3, and (e) PP4, and (f) EDX results of PP4.
Conclusions
In this research, different amounts of BA (1.25, 2.5, 3.75 and 5.0 wt%) were used to improve the effectiveness of IFR system composed of APP/PER (3/1 mass ratio) in PP including 2 wt% MMT. The combinations of 1.25 and 2.5 wt% BA with 2 wt% MMT (PP3 and PP4) achieved better thermal and fire resistance than PP1(PP/IFR) and PP2 (PP/IFR/MMT). BA released water vapor acted as a heat sink and diluted oxygen/combustible gases in endothermic reactions generating the metaboric acid and the glassy boron oxide that reinforced the char layer. The reinforced char layer effectively enhanced the thermal degradation and the fire resistance of the composites. The combinations satisfied UL 94 V0 rating with 31% LOI value. In the cone calorimetry tests, it was determined that although PP sample completely burned in approximately 600 s, IFR/MMT/BA additions resulted in decreases in PHRR values, which indicated decreasing of the fire intensity and the extension of the burning time with more residues. The combinations of 1.25 and 2.5 wt% BA with 2 wt% MMT (PP3 and PP4) decreased PHRR in approximately 31.5% and 47.8% ratios compared to PP1(PP/IFR), respectively. Meanwhile, the total heat release values were decreased by approximately 22.2% and 44.0% in 1000s duration.
In parallel with the improvements in the fire resistance, CO, CO2, and the smoke emissions were significantly decreased with the additions of BA and MMT. IFR additions increased NO emissions of pure polypropylene due to nitrogen content of APP. However, BA and MMT additions also resulted in decreases in NO emissions compared to PP1 (PP/IFR). Although the additions of BA and MMT did not cause significant changes in the decomposition of PP1 (PP/IFR) composite, 1.25 wt% and 2.5 wt% BA with 2 wt% MMT additions (PP3 and PP4) resulted in increasing of char residues up to 7.8 wt% and 8.3 wt% at 1000°C, respectively. This suggests that more stable and strong char layers were generated with BA/MMT additions.
The intumescent flame retardants including 1.25% and 2.5% BA increased the thermal conductivity by 22.7% and 27.3%, respectively. This is also an advantage for heat transfer related applications such as anti-corrosion heat exchanger, thermal interface materials and heat sinks in electronic devices. Although only IFR addition (PP1) reduced the tensile and flexural strengths approximately by 8.7% and 5.7, respectively, it was determined that BA and MMT additions enhanced the tensile and the flexural strengths.
Footnotes
Acknowledgments
The authors would like to thank “Pamukkale University Scientific Research Council” and “The Scientific and Technological Research Council of Turkey (TUBITAK)” for supporting this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by “Pamukkale University Scientific Research Council” and “The Scientific and Technological Research Council of Turkey (TUBITAK)” under projects contract no. 2016FEBE015 and 108T246, respectively.
