Abstract
In this study, the effects of alkali and silane coupling agents and coir fiber (CF) loading on the fundamental properties of the CF-filled polypropylene (PP) composites were investigated. Mechanical properties of the PP/CF composites, such as tensile strength, tensile modulus, impact strength, and water absorption were increased by the increase of the CF loading. The inclusion of 3-aminopropyl trimethoxy silane (ATS) and tetramethoxy orthosilicate (TOS) after the alkali pretreatment for the CF increased all the mechanical properties and water desorption of the resulting composites. This trend was more evident with the increase in CF loading. The best results were obtained for PP/TOS composites as compared to other composites. SEM images of fractured samples show improved adhesion between CF and PP matrix after treatment with ATS and TOS. The horizontal rate of combustion is significantly reduced with the inclusion of Mg(OH)2 in PP/ATS and PP/TOS composites. DSC results show improved crystallization temperature, melting temperature, and melting enthalpy as compared with virgin PP. The addition of ATS and TOS after the alkali pretreatment improved the thermal stability of the resultant composites. TOS-modified CF composites showed better resistance than ATS-modified CF composites in water medium.
Introduction
Recently, there has been a significant increase in interest in composite materials reinforced with natural fibers (NFs).1–3 Currently, NFs like jute, coir, flax, kenaf, sisal, pineapple fiber, etc. are widely used for making composites because of their easy availability, easy processing, low density, high hardness, high specific properties, lightweight, recyclable as renewable resources, low cost and above all, “eco-friendly” features. 4 NF-reinforced thermoplastic composites are gaining popularity in industrial packaging as well as applications for fencing, panel work, assembly boards, furniture, and construction with a few drawbacks.5,6 Extensive research work on various NF composites by Zaman and coworkers has proven that NF is an excellent alternative to potentially toxic synthetic ones.7–9
The NF under consideration here is coir, a fruit fiber derived from the outer shell or husk of the coconut (Cocos nucifera), which is widely grown in tropical countries. Due to its hardware quality, durability, and other advantages, it is used in making various types of floor furniture, yam, rope, etc. But these traditional coir products use a small portion of the potential total world production using coconut husk. Therefore, in addition to the conventional use of coir mentioned above, research and development efforts are underway to find new areas of application of coir, including the use of coir as a reinforcement in polymer composites.10–12 Low cost and high performance of coir are beneficial for the industry; on the other hand, its biodegradability contributes to a healthy ecosystem. NFs are lignocellulosic and highly hydrophilic. NF-reinforced composites are also hydrophilic. The hydrophilicity of the coir is extremely unfavorable as far as composite performance is concerned due to low adhesion to the hydrophobic elastomeric matrix.13,14 Coir efficiency can be improved as reinforcement by increasing the interfacial adhesion between the coir and the polymer matrix. This can be achieved either by modifying the surface topology of the coir by pre-treatment or by selecting the appropriate component of the bonding system. Surface modifications of NFs have improved the mechanical properties of composites, as several workers have recently reported.15–18
In this present study, coir fiber was used as the reinforcing agent and PP was used as the polymer matrix for polymer composites to reduce the content of synthetic PP keeping in mind the environmental consciousness. PP was chosen as the matrix of the composite because it has low specific density, excellent and desirable mechanical, physical, and thermal properties when used in normal temperature applications, and takes a large part in wood plastic composite markets. 19 One of the possible routes for making natural-synthetic polymer composites is PP through NFs. Weak adhesion between NFs and polymer matrix often prevents the possibility of NFs to act as fillers, resulting in weak dispersion, inadequate reinforcement, and low mechanical properties. Hence, NFs require the addition of coupling agents or chemical modifications for final application to composite materials. Chemical modification is an effective method of improving the physicomechanical properties of composites. Alkali pre-treatment and subsequent silane treatments were used to increase the adhesion between the fiber and the matrix which can significantly improve the composite performance.20,21 Alkali treatment removes impurities from the fiber, reduces moisture sorption, and activates mechanical bonding, and is effective in improving matrix-reinforcement interactions. 21 Various workers22,23 have made efforts to modify coir fibers for possible use in polymer composites. However, a comprehensive literature review reveals that the addition of ATS and TOS after the alkali pretreatment for the CF reinforced PP-based unidirectional composites have not yet been investigated. In this study, the effects of new silane treatments and fiber loading on the mechanical, morphological, flammability, and thermal properties of unidirectional CF reinforced PP composites. The comparison was made to test the effect of a coupling agent for the composites. The water absorption behavior of the composite was identified to understand the effect of chemical treatment on the hydrophilicity of the composite.
Experimental
Materials
CFs were collected from the husk of coconut (Cocos nucifera) fruit and obtained from local sources of Bangladesh. Liquid washing detergent powder was obtained from Linghai Zhanwang Biotechnology Co., Ltd. Liaoning, China. The diameter of the CF used was ∼ 0.25 ± 0.056 mm. PP was supplied by the Polyolefin Company, Pvt. Lt. Singapore. Aminopropyl trimethoxy silane (ATS), 97% from Sigma-Aldrich® and Tetramethoxy orthosilicate (TOS), 99% from Sigma-Aldrich® silane coupling agents were used for chemical modification. Magnesium hydroxide Mg(OH)2 as flame retardant was obtained from Fisher Science Education, Rochester, NY. Chemicals used in this study to treat coir were pure methanol (E. Merck, Germany) and NaOH (E. Merck, India Ltd).
Methods
Coir pith and unwanted components were isolated from the CF. It was then cut to a size of 100 mm in length. The well-isolated CFs were first polished with liquid washing detergent solution (2%) at 60°C for 1 h and then cleaned with purified water to eliminate extraneous elements. The fibers were then desiccated at 60°C for 24 h to adjust the humidity to 1–2% and then kept in a vacuum desiccator. The weight of the desiccated sample was taken through an analytical electrical balance.
Pretreatment of coir fibers
Washed and desiccated CFs were kept in a plastic tray. A 3% alkali (NaOH) solution in water was incorporated into the tray and the CFs were immersed in the solution at room temperature for about 1 h. The solution to the CF ratio was 20:1 in terms of weight percentage (wt%). Thereafter, the fibers were washed in de-ionized water until all NaOH has been eliminated, until the water indicates no more alkaline reaction. However, the final wash was done with 2% acetic acid to neutralize the last traces of NaOH. Finally, the fibers were again washed with fresh distilled water and dried in an oven at 80°C for 24 h.
Silane treatments
Silane treatment of alkaline pre-treated CFs was conducted with ATS and TOS. 3 wt% solutions of silane were prepared by mixing ATS and TOS in a methanol/water mixture ratio (50:50) for 1 h. Alkaline pre-treated CFs were immersed in this solution for 1 h at room temperature. The pH of the solution was maintained between 6 and 7 using Metrepack pHydrion buffer and pH indicator strip. After the coupling reaction was completed, the CFs were dried in a vacuum oven at 70°C for a fixed weight for 24 h.
Preparation of composites
For making one PP sheet, the PP granules were compression molded at 170°C into a 0.25 mm thick isotropic sheet. The compressive pressure was 7 MPa for 3 min and was kept at this pressure during cooling to ambient temperature. The CFs were fixed on two double face tapes which were glued to the top of a sheet of metal frame and distant with the required fiber length (Figure 1(A, a)). The CFs were fixed on the tape as uniform as possible and parallel to the required direction. Both untreated and treated composites were fabricated at different CF loading (say, 10–40 wt%) by stacking four layers of unidirectional aligned CF between five layers of pre-weighted PP sheet. The composite was made by pressing this stacking at 180°C for 5 min at a constant pressure of 10 MPa. (Figure 1(A, b)). The compositions of untreated and treated PP/CF composites are displayed in Table 1. The schematic illustration of the manufactured composite is shown in Figure 1(B).
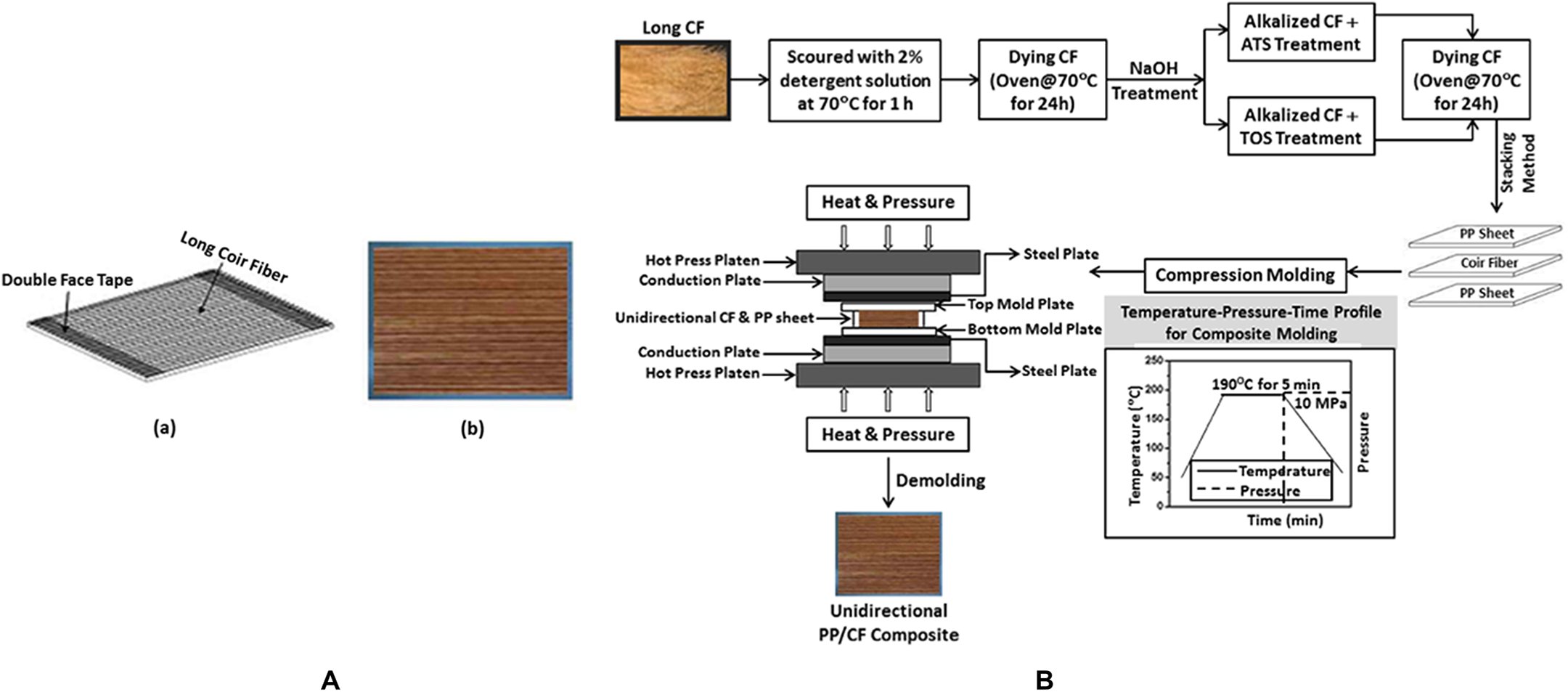
(A) Unidirectional coir fiber preparation (a); and unidirectional CF/PP composite (b). (B) The schematic illustration of the manufactured CF/PP composite and its processing profile.
Compositions of untreated and treated PP/CF composites.
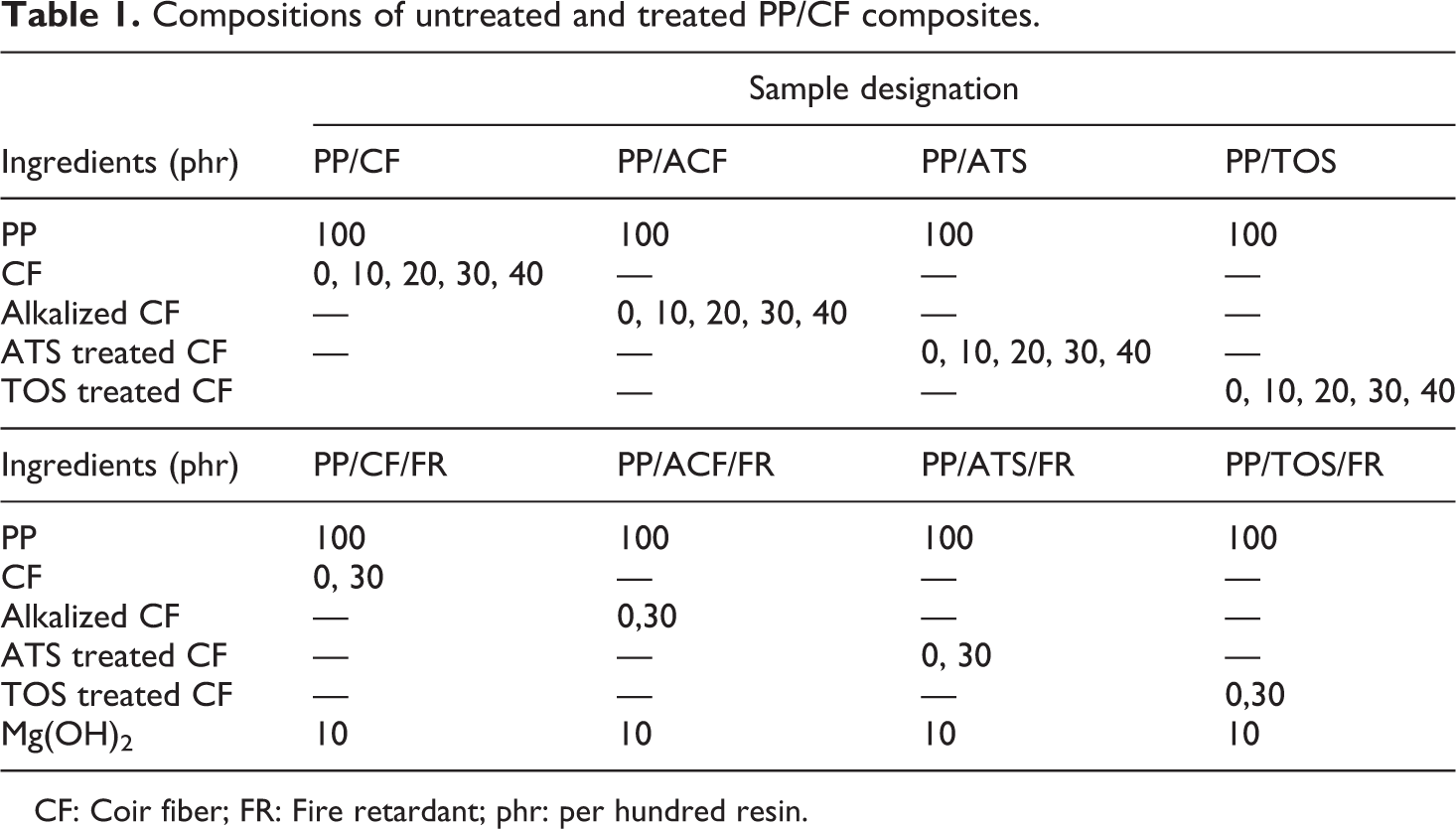
CF: Coir fiber; FR: Fire retardant; phr: per hundred resin.
Mechanical testing of composites
The tensile properties of the composites were measured according to the ASTM-D 638-03 standard method using a Shimadzu Universal Testing Machine (Model AG-1, Japan) with an electronic load cell of 5 kN. The crosshead speed was 10 mm/min and a gauge length was 20 mm. Izod impact strength of the samples having dimensions 63.5 × 12.7 × 3 mm3 was determined according to the ASTM-D 256 standard using an Izod impact machine (Toyo Seiki Co., Japan). All experiments were conditioned at 25°C ± 2°C and relative humidity at 55 ± 5%. A minimum of five samples was tested in each case to obtain an average value.
Fracture morphology analysis
SEM micrographs of untreated and treated composite samples were analyzed by a Zeiss, Evo 50 scanning electron microscope. The fracture edges of the samples were embedded in an aluminum spit and covered with a thin layer of gold to disperse the electric charge throughout the test.
Flammability test
The flammability of the samples was identified by a horizontal burning test according to ASTM D635 using a horizontal-vertical flame chamber instrument (Atlas, HVUL). In the horizontal burn test, the sample was held horizontally and a fire ignited by natural gas was applied to one end of the sample. The burning rate of the sample was calculated as follows:
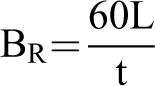
where BR is the burning rate in mm/min, L is the length of the flame travel and t is the time in second for flame travel. Five measurements were recorded in each sample.
Thermal characterization
The crystallization and melting behaviors of virgin PP and its composites were investigated by differential scanning calorimetry (DSC, Perkin Elmer DSC-7). Each sample (5–8 mg) was heated from 30°C to 200°C at a heating rate of 10°C/min under N2 atmosphere and then held at 200°C to confirm the duplicate thermal history. The sample was cooled down to 30°C at a cooling rate of 10°C/min. The thermal stability of virgin PP and its composites was determined using a thermogravimetric analyzer (TGA, TA Instruments Q500). Each sample (5–8 mg) was heated from 30°C to 600°C at a heating rate of 10°C/min under N2 atmosphere.
Water absorption measurement
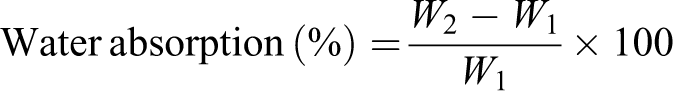
To determine the water absorption retention of the untreated and treated composites, rectangular samples of dimensions 30 × 10 × 2.5 mm3 were prepared. The water absorption tests were carried out by immersing the samples in a water bath for 24 h at room temperature. After immersion, the excess water was removed using a piece of a soft cloth and the final weight of the sample was taken. The water absorption was measured as follows:
where W2 and W1 are the weight of the sample after and before immersion, respectively.
Results and discussion
Tensile properties
Figure 2(A) displays the tensile strength of untreated and treated PP/CF composites as a function of CF loading. The tensile strength of virgin PP was 27.3 MPa. It was observed that the tensile strength of PP/CF composite gradually increased up to 30 wt% CF loading and then decreased with CF loading. According to our previous works,8,24 the tensile strength increased with CF loading. This increase was due to an initial reinforcement effect by the CFs, which allowed stress distribution from the nonpolar PP matrix to the polar CF. At lower levels of fiber content (10 wt%), the composites show weak tensile properties due to poor fiber populations and low load transfer capacity to each other. As a result, stress gets accumulated at certain points of the composites and highly localized strains occur in the matrix. At the intermediate level of loading (30 wt%), the fiber population is just right for maximum orientation and the fibers actively participate in stress transfer. At high levels of loading (40 wt%), the composites show poor tensile properties due to low mixing of coir and PP, and the amount of PP will not be sufficient to disperse the CF thoroughly, and consequently tensile strength is reduced.
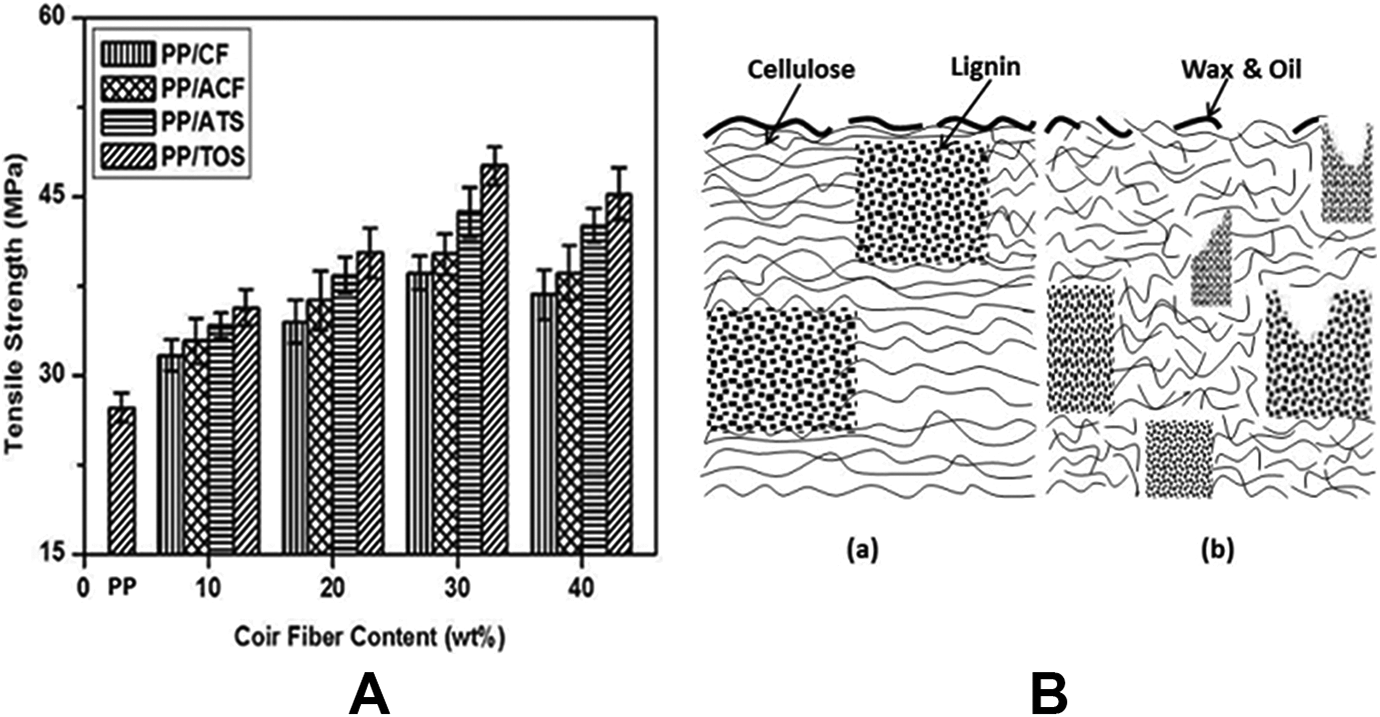
(A) Tensile strength of PP/CF, PP/ACF, PP/ATS, and PP/TOS composites. (B) Characteristic structure of (a) untreated and (b) alkaline cellulosic fibers.
Figure 2(A) shows the tensile strength of PP/CF composites with different surface modifications. It can be seen that the tensile strength of PP/ACF, PP/ATS, and PP/TOS composites increased by 4%, 13%, and 23% at 30 wt% CF loading, respectively, as compared to PP/CF composite. For the PP/ACF composite, the increase in tensile strength was attributed to the removal of natural and artificial impurities (e.g. lignin, hemicellulose, pectin, waxed substances, and natural oils) covering the outer surface of the fiber cell wall (Figure 2(B, a)) and developed microporosity with many pits and cavities on the fiber surface. This indicates that alkalization depolymerizes the native cellulose I molecular structure to form short-length crystallites (Figure 2(B, b)). Furthermore, alkalization reduces the fiber diameter, increasing the aspect ratio which leads to the development of a rough surface topography resulting in better fiber-matrix interface adhesion and mechanical properties. 25 The reaction of NaOH with cellulose is shown in Figure 3(a). At the loading level of 30 wt% CF, the tensile strength of the PP/ACF composites treated with 3 wt% ATS and TOS was 13% and 23% higher than that of the PP/CF composite. In the presence of moisture, the hydrolyzable alkoxy group leads to silanol formation (Figure 3(b)). After hydrolysis, the alkoxysilanes go to the stage of condensation and covalent bond formation and can form polysiloxane structures by reaction with the -OH group of CFs. Reactions are shown in Figure 3(c) and (d). Four methoxys (OCH3) groups of TOS can form silanol by hydrolysis, whereas three methoxys (OCH3) groups of ATS can form silanol by hydrolysis. For this reason, TOS may form a tighter silane crystalline complex with the CF than ATS. In addition, the aminopropyl group of ATS may have a more hydrophilic nature than the OCH3 group or -OH group without the chemical reactions of TOS. Therefore, TOS has better compatibility with the PP matrix than ATS. This indicates the effectiveness of silane treatment in increasing the fiber/matrix bond, which will improve the tensile strength of the composite.
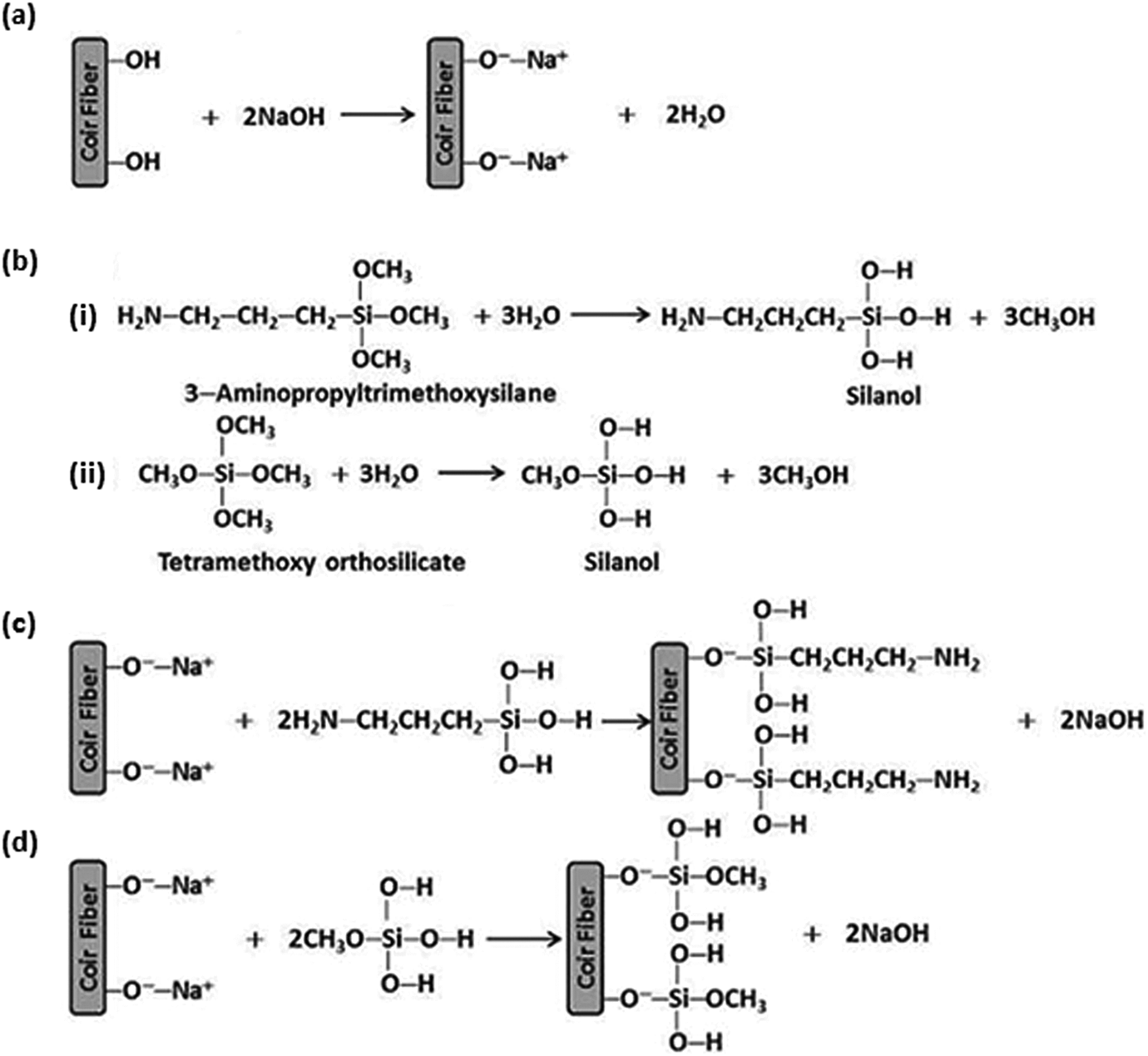
(a) The role of sodium hydroxide in the formation of an alleged intermediate species with CFs; (b) hydrolysis of silanes (i) 3-aminopropyl trimethoxy silane (ii) tetramethoxy orthosilicate; potential reaction mechanisms between cellulosic –OH groups and (c) 3-aminopropyl trimethoxy silane (d) tetramethoxy orthosilicate.
Figure 4(a) shows the correlation between the tensile modulus and the CF loading with and without the alkali and silane (ATS and TOS) treatments. The tensile modulus of virgin PP was 455 MPa. It was found that the tensile modulus of PP/CF, PP/ACF, PP/ATS, and PP/TOS composites increased to the tune of 255%, 272%, 313%, and 320% at 30 wt% CF loading, respectively, as compared to virgin PP. The tensile modulus increased with fiber loading by other researchers.26,27 The increase in tensile modulus may be highly related to the increase in composite stiffness due to CF loading. PP/ACF, PP/ATS and PP/TOS composites received higher tensile modulus than PP/CF composites. At the loading level of 30 wt% CF, the tensile modulus of the PP/ACF, PP/ATS, and PP/TOS composites was 5%, 16%, and 18.3% higher than that of PP/CF composite, respectively. It can be seen that the PP/TOS composites exhibit better tensile modulus than other composites due to some strong chemical bonding between the hydrophobic part of the silane of the coil surface and the PP matrix.
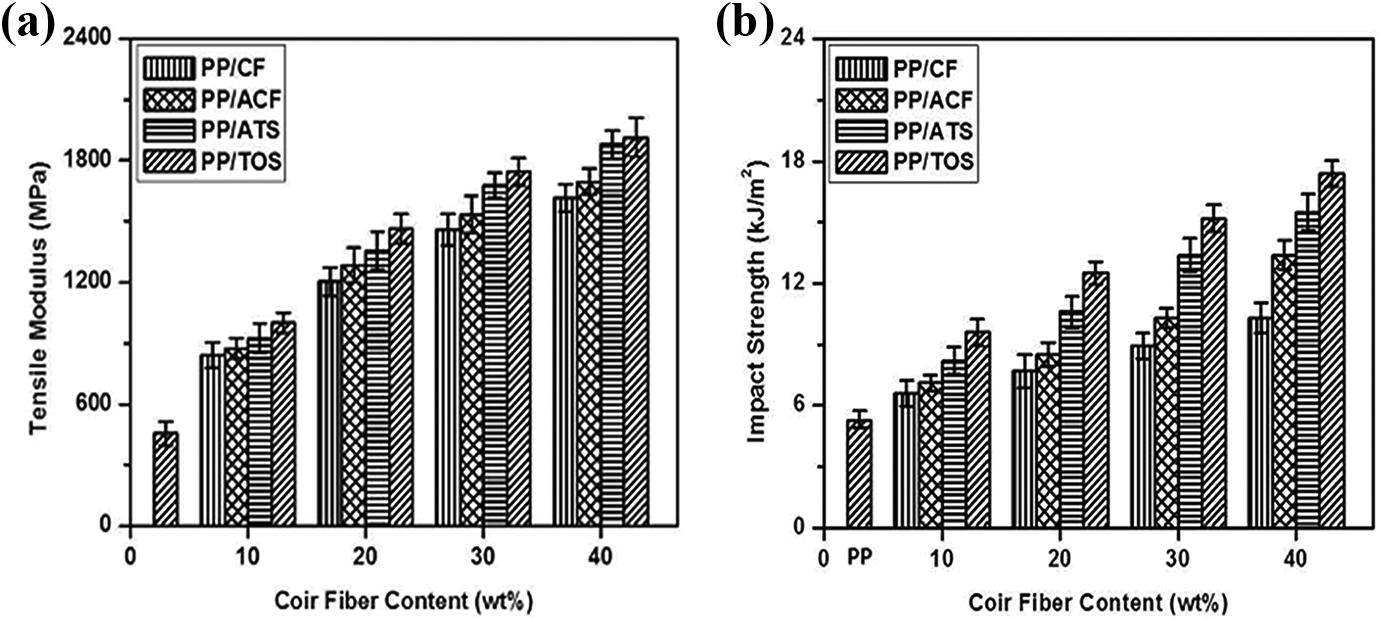
(a) Tensile modulus and (b) impact strength of PP/CF, PP/ACF, PP/ATS, and PP/TOS composites.
Impact toughness results
As shown in Figure 4(b), the Izod impact strength of virgin PP was 5.3 kJ/m2. As the loading level of the CF increased from 10 to 40 wt%, the Izod impact strength of the PP/CF composites was considerably higher than that of virgin PP. At the loading level of 40 wt% CF, the impact strength of the PP/ACF, PP/ATS, and PP/TOS composites was 30%, 50%, and 69% higher than that of the PP/CF composite. These results are because the fiber was able to absorb energy because of the strong interfacial bond between the fiber and the matrix. As the load on the fiber increases, a larger force is required to pull-out the fibers. Significantly higher impact strength for PP/ATS and PP/TOS composites is probably due to the favorable interaction between the treated CF and the hydrophobic PP chain of the matrix. The so-called fiber pullout and fiber agglomerations may be responsible for the low impact strengths of the PP/CF composite.
Rockwell hardness properties
The hardness of a composite material refers to its resistance to change in shape when the force is applied to it. Composites generally exhibit less rigidity due to the presence of a more flexible matrix. 28 The addition of both untreated and treated CF to the PP matrix reduced the flexibility of the matrix, resulting in a more rigid composite. The effect of CF content of PP/CF, PP/ACF, PP/ATS, and PP/TOS composites on Rockwell hardness is illustrated in Figure 5(A). As the CF loading increases, the hardness of both untreated and untreated composites increases. The inclusion of CF in the PP matrix has reduced the mobility of the polymer chain in rigid composites. At the loading level of 40 wt% CF, the hardness of the PP/ACF, PP/ATS, and PP/TOS composites was 2%, 6%, and 8% higher than that of the PP/CF composite. This can be attributed to the better dispersion of CFs in the PP matrix so that the voids are reduced and the strong interfacial adhesion between the PP matrix and the CF.
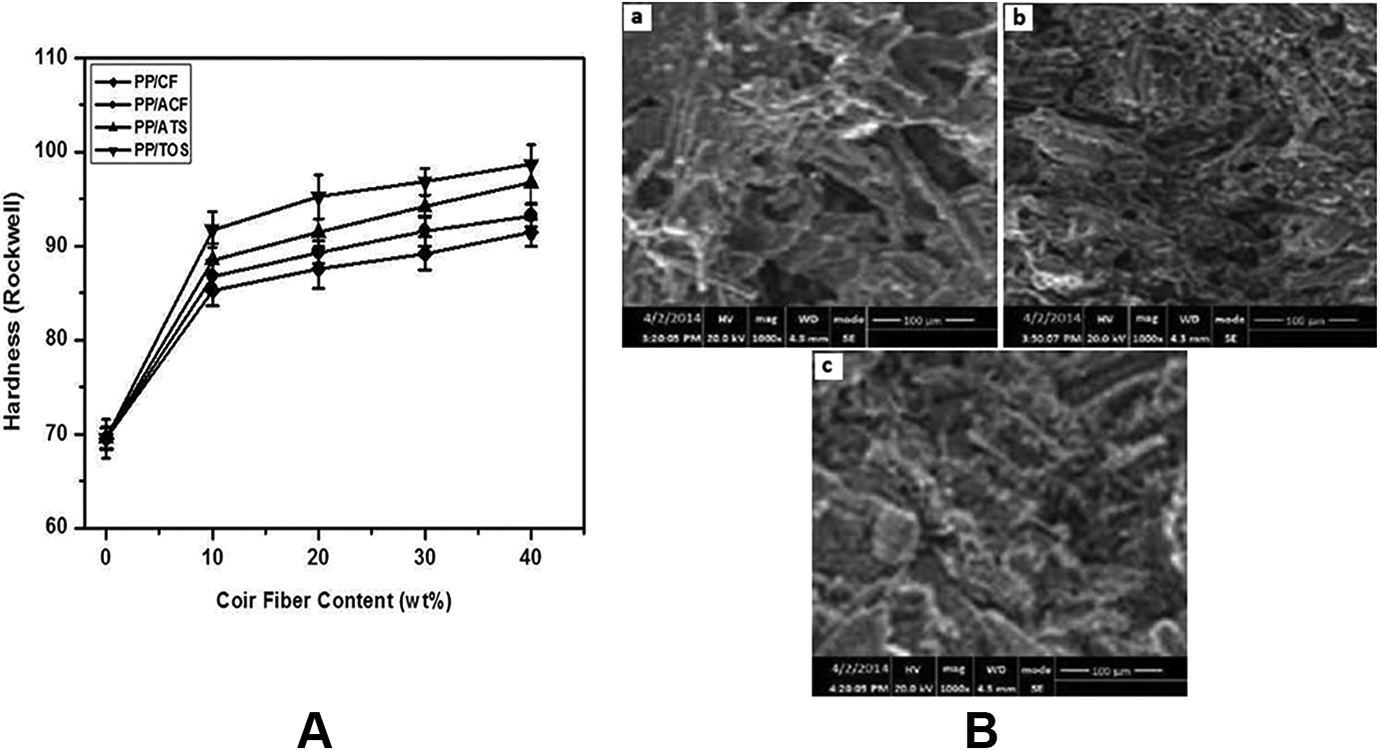
(A) Rockwell hardness of PP/CF, PP/ACF, PP/ATS and PP/TOS composites; and (B) SEM images of (a) PP/CF (b) PP/ATS and (c) PP/TOS composites at 40 wt% fiber content.
SEM analysis of fracture surfaces
SEM images of PP/CF, PP/ATS, and PP/TOS composites prepared with 40 wt% CF are displayed in Figure 5(B, a–c), respectively. The SEM images of the PP/CF composite show several fiber agglomerations and fiber pullout trace in the composites (Figure 5(B, a)). These properties suggest fiber-fiber interaction and weak interfacial bonding between the hydrophilic CF and the hydrophobic PP matrix. At this stage, the interfacial structure of this composite could not effectively stress transfer. This observation was consistent with the low tensile strength values described in Figure 2(A). As can be seen from the PP/ATS composite (Figure 5(B, b)), the CFs were slightly pullout during the fracture process and most of the PP matrix was attached to the surface of the CFs and adhered. Fiber pullout seems to be lower for PP/ATS composites than for PP/CF composites. This result confirmed a good interfacial bonding between CF and PP. However, for PP/TOS composite (Figure 5(B, c)), CFs were broken without complete pullout during the fracture process, and there was much PP matrix still coating the CFs. This result confirmed that interfacial bonding between the CF and the PP matrix has become much more favorable for PP/TOS composite compared to that of the PP/ATS composite.
Flammability of the composites
The flammability of virgin PP and PP/CF composites with and without alkali and silane (ATS and TOS) after alkali pretreatment, as influenced by the addition of fire retardant (FR), is demonstrated in Figure 6(a). It has been reported that magnesium hydroxide Mg(OH)2 can act as a flame retardant in plastics and will be effective at a higher loading. 29 It was observed that PP/CF composites show a higher rate of burning than the virgin PP, which indicated a higher sensitivity of the CF to flame. However, with the inclusion of 10 wt% of Mg (OH)2, the rate of burning of PP/CF/FR composite reduced to 13 wt%. This confirms the effectiveness of Mg(OH)2 as a flame retardant additive, by releasing a significant amount of water at high temperatures thereby diluting the amount of fuel available to sustain the combustion during a fire. In addition, it is also assumed that Mg(OH)2 absorbs heat from the combustion zone, reducing the likelihood of continuous combustion and creating a char during combustion resulting in more flame retardant protection and less smoke generation. The inclusion of alkali and silane (ATS and TOS) after alkali pretreatment showed 50%, 63%, and 68% reduction in the burning rate of PP/ACF/FR, PP/ATS/FR, and PP/TOS/FR composites, respectively. PP/TOS/FR composite showed better improvement in flame retardancy than PP/ATS/FR composite because PP/TOS/FR composite achieved better compatibility with the PP matrix than PP/ATS/FR composite.
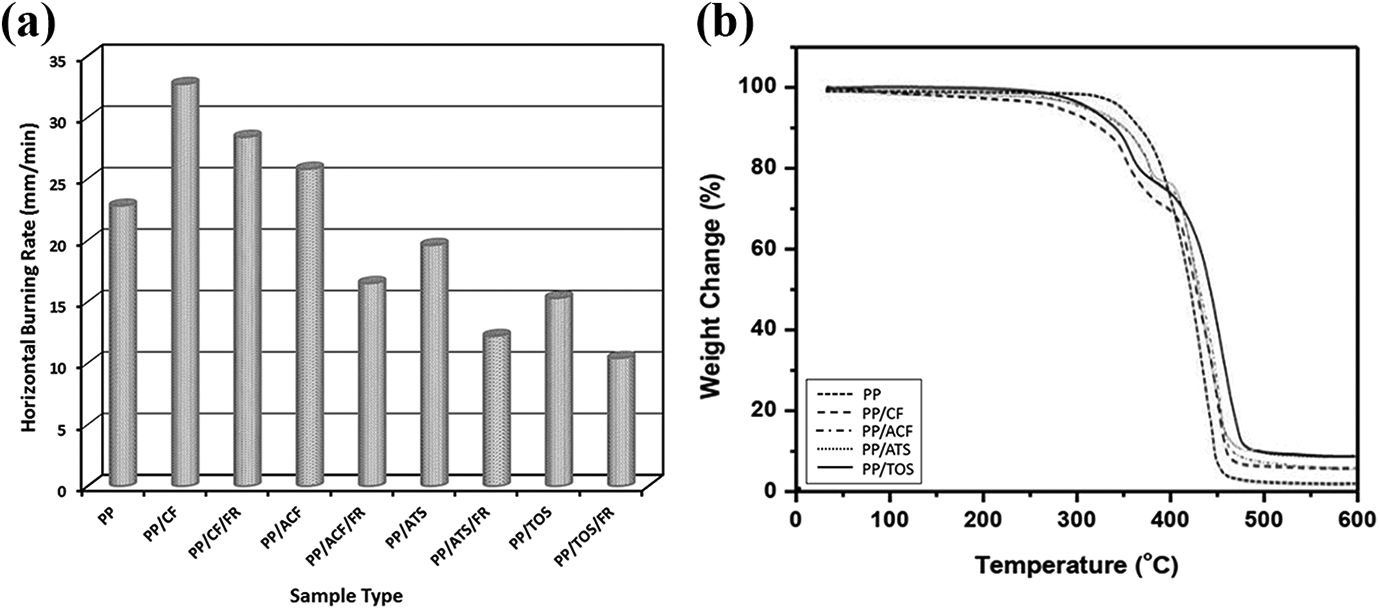
(a) Horizontal burning rate of virgin PP and their composites with and without fire retardant; (b) TGA analysis curve for thermal analysis.
Thermogravimetric analysis (TGA)
The thermal stability of the virgin PP and PP/CF, PP/ACF, PP/ATS, and PP/TOS composites was investigated using the TGA curve is presented in Figure 6(b). The TGA thermogram showed that the thermal degradation of virgin PP started from 350°C with a final degradation temperature of 460°C. It was also observed that the weight loss of virgin PP occurred in a one-step degradation process from 350°C to 450°C, after which a very small amount of PP residue was left in the gaseous products due to the thermal degradation of PP at a higher temperatures. However, in the case of PP/CF composites with and without ATS and TOS, a two-stage degradation process was observed where the weight loss at 340°C coincides with the degradation of CF whereas the second stage at 400°C indicated the degradation of the PP matrix. However, the thermal degradation temperature in both the stages for the PP/CF composites in presence of ATS and TOS was comparatively higher (approximately 11.5°C and 14.9°C) than that of the composites without silane, indicating higher thermal stability. This behavior is probably due to the increase in the molecular weight by cross-linking reaction between PP matrix and CF or molecular chain-extension of the matrix itself. 30 The increased thermal degradation may be highly related to the increased inorganic silane content on the surface of CF.
Differential scanning calorimetry (DSC)
The thermal transition of PP, crystallization temperature (Tc), melting temperature (Tm), melting enthalpy (▵Hm), and crystallinity (Xc) for the PP phase in the composites with 40% CF is determined from the DSC thermograms and are shown in Figure 7(a–d), respectively. Figure 7(a) shows that the Tc of virgin PP is 116.3°C, and the addition of 40% CF to virgin PP results in an increase of Tc of about 1.5°C for composites without coupling agents. This result may be due to the increase in the nucleation process in the presence of CF. The addition of ATS and TOS to PP/CF composites increased Tc to 3.1−5.3°C, respectively, indicating a further increase in the nucleation process in the presence of coupling agents. The ester linkage between the PP matrix and CF produced by the addition of ATS and TOS might have further resulted in increased nucleation of the PP matrix.
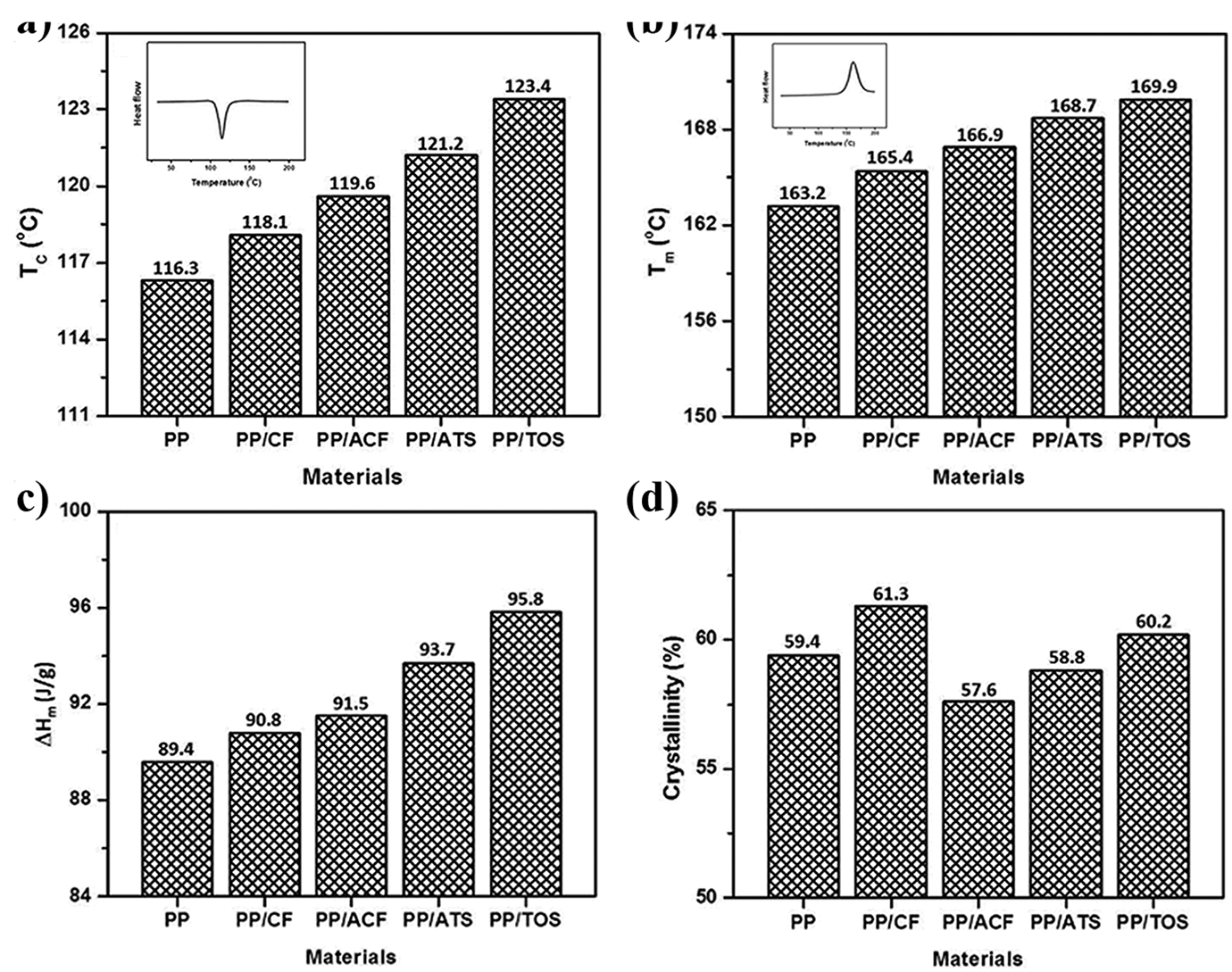
(a) Crystallization temperature, (b) melting temperature, (c) melting enthalpy, and (d) crystallinity of PP/CF, PP/ACF, PP/ATS and PP/TOS composites.
The melting temperature (Tm) of virgin PP was observed at 163.2°C (Figure 7(b)). The addition of 40 wt% CF to virgin PP increased the Tm by 1.3°C. This is probably due to fiber-fiber interactions as well as fiber-polymer interactions that contribute to the marginal increase in the melting point of PP in PP/CF composites. Moreover, the addition of ATS and TOS in PP/CF composites showed 3.3−4.5°C higher than the PP/CF composites. Tm may increase due to good compatibility between the two materials. The melting enthalpy (▵Hm) of the PP was 89.4 J/g (Figure 7(c)). The inclusion of 40 wt% CF to virgin PP increased the ▵Hm to 90.8°C. The incorporation of ATS and TOS to the PP/CF composites showed higher ▵Hm than the PP/CF composites. These results indicate that the composites with coupling agents require higher energy to be molten. The TOS provides higher ▵Hm than ATS for the PP/CF composites.
The crystallinity (Xc) value in Figure 7(d) shows that the degree of crystallinity increased with the inclusion of CF. CF acts as the nucleation site of PP and therefore accelerates the rate of crystallization and increases the degree of crystallization. The addition of ATS and TOS reduces the Xc value of PP/ATS and PP/TOS which can be attributed to the increased interaction between the fiber and ATS and TOS which inhibits the crystallization phase of PP and reduces the crystallization when compared to PP/CF composite. However, with the addition of ATS and TOS, the intermolecular interaction between PP and CF increased. As a result, the crystallization of the PP phase in the ATS and TOS is hindered.
Water absorption (WA) behavior
The results of WA values of PP/CF, PP/ACF, PP/ATS, and PP/TOS composites against fiber loading are shown in Figure 8 as a function of soaking time (24 h) in water. The soaking time and CF content are significant factors, which affect the WA of the composite. After 24 h of water soaking, the WA of the PP/40 wt% CF composite was 8% higher than the PP/30 wt% CF composite due to the higher availability of cellulose –OH groups that can absorb water. On the other hand, alkali-treated PP/CF composite (PP/ACF) presented a higher WA (0.81%) than PP/ATS (0.70%) and PP/TOS (0.62%) composites indicating that this alkaline treatment was incapable to remove the –OH groups on the surface of treated fibers causing higher water retention. Conversely, the surface modification by silane promoted a better mechanical interlocking between the fiber and matrix, and thus water absorption was lower for PP/ATS and PP/TOS samples.
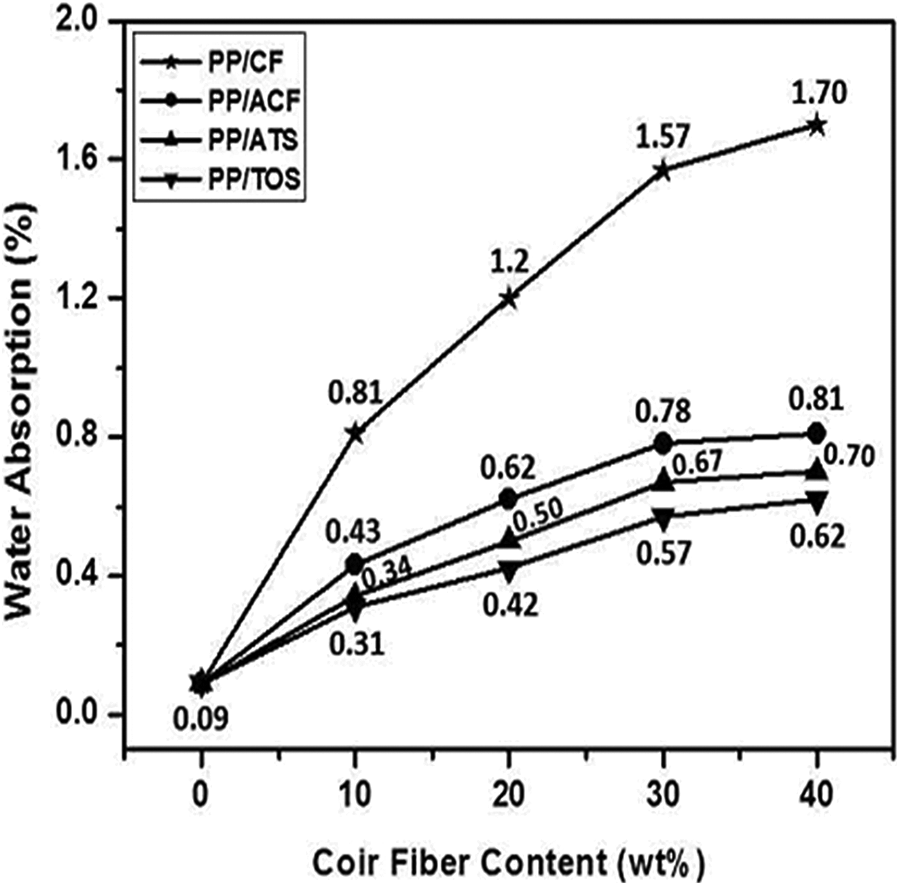
Variation of WA of PP/CF, PP/ACF, PP/ATS, and PP/TOS composites at different fiber loadings.
Conclusions
In this experiment, the effects of alkaline and silane treatment of CFs on the basic properties of PP/CF composites were investigated and analyzed. Based on the experimental results, the following conclusions can be drawn from the current investigation: Mechanical properties such as tensile strength, tensile modulus, impact strength, and hardness of PP/CF composite were improved by increasing the amount of fiber in the optimum fiber content of 40 wt%. However, 40 wt% filler load composites had lower tensile strength than 30 wt%. The mechanical properties of PP/ATS and PP/TOS composites were higher than those of untreated composites. SEM investigations of fractured surfaces of composites have revealed rich bonds between PP and CFs. The inclusion of Mg(OH)2 in the PP/CF composite significantly reduced the horizontal flaming rate. The thermal properties of PP/CF composites were increased with the addition of ACF, ATS, and TOS. In contrast, the degree of crystallinity was reduced by adding ACF, ATS, and TOS. Alkaline and silane treatment of CF composites reduced WA compared to untreated composites.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
