Abstract
In this study, the effects of two different biodegradable plastic additives on the thermal and mechanical properties of isotactic polypropylene were investigated, analyzed, and presented. Biosphere biodegradable plastic additive and another oxidative biodegradable plastic additive, D2W, were used in 0.25, 0.5, 1.0, 1.5, and 2.0 wt% to prepare two sets of isotactic polypropylene composites. Differential scanning calorimeter and optical polarized light microscope were used to analyze the obtained composites thermally while the mechanical properties of these composites were studied by conducting tensile and impact testing. The morphological results reveal that only monoclinic α spherulites were obtained when the polypropylene crystallizes isothermally from the melt, it was found that both biodegradable plastic additives used decreases the size of the spherulites and increases their densities per unit volume. The incorporation of the Biosphere within the polypropylene matrix raises the crystallization temperature while the existence of D2W within the matrix delays the crystallization process. The addition of 2 wt% of Biosphere allows polypropylene to crystallize at about 13°C higher than neat polypropylene. It has been found that the addition of the biosphere increases Young’s modulus, while decreases the yield strength, the elongation at break, and the impact strength. On the other hand, D2W as an oxidative biodegradable plastic additive increases the elongation at break and the impact strength while decreases the yield strength and Young’s modulus.
Keywords
Introduction
Plastics are found almost everywhere; the applications of plastics are essentially limitless. Plastics have attained a unique position in the packaging industry because of their favorable features such as high strength, water resistance, lightweight, energy effectiveness, and low cost. Plastics are diversely used as insulators, ranging from single wires to entire buildings. As well, roofing materials, door and window frames, screws, and hardware all contain plastics. A variety of plastics are used in the manufacture of automobiles, trucks, and even airplanes. Fuel lines, brake linings, windshield wipers, tires, bumpers, speedometer gears, emission canisters, headlights, steering wheels, and fuel pumps all contain tough plastics. More flexible plastics are used for interior paneling, seats, and trim. Even the outer bodies of some cars are made of fiberglass-reinforced plastic. The wires of most electronic devices are encased in some type of plastic. As well, plastics are used in the outer casings of telephones, lighting fixtures, electric mixer housings, fans, radio cabinets, coffee makers, computers, and clocks.
Unfortunately, conventional plastics have their origin in the petrochemical industry making them non-biodegradable and non-renewable.1,2 The non-biodegradable and non-renewable nature of plastics has been a serious disadvantage to their application leading to huge municipal wastes and environmental degradation. Therefore, most users are faced with increasing pressure to minimize non-degradable waste and to switch to biodegradable materials. Despite this pressure, no biodegradable plastics combining all the features, functionalities, and economies have been developed. 1 Efforts to produce competitive biodegradable materials are often frustrated by the problem of finding an optimal balance between mechanical properties, cost, and acceptable level of biodegradability.
The use of commodity plastics, especially polyethylene, polypropylene, and polystyrene are growing day by day. Geyer 3 has estimated that around 343 million tons of plastic waste are produced every year. Every year 25 million tons of synthetic plastics are being accumulated in the sea coasts and terrestrial environment. In the marine environment alone, out of total marine waste, plastics share about 60–80% by mass. All the polypropylene and polyethylene waste along with other plastic wastes generated by the human activity finally enters into marine water through rivers, canals/channels, and municipal drainages. Therefore, the beaches were reported to be the excellent depository sites for the plastics wastes. 4 The main environmental disadvantage of plastic materials is that they do not readily break down in the environment and therefore can litter the natural environment. Using biodegradable plastics can reduce the amount of plastics in landfills. The use of biodegradable polymers is increasing at a rate of 30% per year in some markets worldwide.
Numerous studies have been conducted to characterize, analyze, and optimize the properties of biodegradable plastics. The most important properties in biodegradable plastic materials include mechanical and thermoforming properties, gas and water vapor permeability, transparency, and availability. While encouraging results have been reported, the development of a biodegradable plastic material that possesses all of the required properties has been an enormous challenge. Especially, there has been the issue of the product cost as well as technical challenges such as brittleness associated with high loads, and poor water and gas barrier properties. This in turn has limited the wide application of renewable biodegradable plastic materials.
Pérez et al. 5 studied the influence of botanical corn and potato starches content on thermal properties and morphology of polypropylene/starch blends. The authors used 7 and 15 wt% of starch content in the blends and concluded that the corn starch (CS) and potato starch (PS) content induces a retardant effect on the degradative process of the polypropylene, since it increases the initial decomposition temperature of the blend, acting as an antioxidant agent. Moreover, Huang et al. 6 noted that when starch-based bioplastics are treated with plasticizers are stored for a long time; their mechanical properties significantly deteriorate due to recrystallization. Mechanical properties and degradability of different mixtures of polyhydroxybutyrate and polypropylene were studied by Pachekoski et al. 7 Proportional to the amount of polypropylene in the blends, the polyhydroxybutyrate (PHB)/polypropylene (PP) composites showed a tendency for lower crystallinity and stiffness, rendering them less stiff and fragile. The degradation tests showed that both pure PHB and blends with 90% PHB and 10% PP were degraded, with loss of their mechanical properties and weight. In a ratio of 80/20, blends of high-density polyethylene (HDPE)/polylactic acid (PLA) with/without compatibilizer in 0.1 and 0.2 wt% pro-oxidant (cobalt stearate) were prepared by melt blending technique. 8 It is reported that all the prepared blends were able to biodegrade in the composting environment and the blend containing pro-oxidant was maximum degraded. A slight increase in the melting point (Tm) of HDPE was observed after incorporation of PLA in it but the negligible effect of adding CoSt was noticed on the Tm of HDPE and/or PLA. Percent crystallinity of PLA was decreased after pro-oxidant addition, which was probably due to the reorganization of the polymer chains in the blends caused by cobalt starch. The morphology of the blend showed increased formation of cavities, rough surface with large holes and increased droplet radius, the domain size of polymers was reduced ascribing to the in situ compatibilization. The influence of biodegradable polymers such as polylactic acid (PLA), polyhydroxybutyrate (PHB), and thermoplastic starch (TPS) in the recycled PP were analyzed by Samper et al.. 9 Their results revealed that the softening temperature is negatively affected by the presence of biodegradable polymers in recycled PP. Also, the melt flow index was negatively affected by PLA and PHB added to the blends. The presence of biodegradable polymers in recycled PP caused a significant loss of mechanical, thermomechanical as well as thermal properties, especially when using percentages of biodegradable polymers higher than 5 wt%. In another study, the thermal, mechanical, and biodegradation properties of polypropylene-based green composites containing 10, 20, 30, 40, and 50 wt% of rice straw cellulose fibers (CFs) were prepared by melt blend method and then investigated. 10 Young’s modulus and flexural properties of PP were improved by blending PP with CFs. The pure PP showed no degradability, but the PP/CFs composites degraded from about 3 to 23 wt%, depending on CFs content after being buried in soil for 50 days.
Tavanaie and Mahmudi 11 investigated the physical and structural characterization of polypropylene (PP) fibers blended with recycled polylactic acid (r-PLA) flakes. The mechanical and morphological properties, biodegradability, differential scanning calorimetry (DSC) analysis, and dyeing behavior were carried out. The results showed that the mechanical properties of the as-spun and cold drawn PP/r-PLA fibers were significantly changed for the fiber samples with 40% and 50% of r-PLA dispersed phase content. The crystallinity percentage of the modified PP fiber samples decreased with increasing of the dispersed r-PLA phase and the morphological observations showed that the best fibrillar structure belonged to the samples with 5% and 50% of r-PLA dispersed phase content, and the biodegradability of the blend fiber sample with 50% of r-PLA content was about 36.2%. Extrusion and injection molding methods were used to prepare bamboo fiber (BF) reinforced starch/polypropylene (PP) composites and then the mechanical and thermal properties were evaluated 12 and found that the contents of BF and starch resin had a significant influence on the properties of the composites. With more content of BF, the composite exhibited a better flexural property and biodegradation, there was a distinct decrease in weight loss and mechanical properties indicated the degradation caused by the microbes. After biodegradation, the thermal stability of the composites decreased while the crystallinity of PP increased, also the tensile properties of the composites decrease to some degree while bending property improves by increasing the content of BF. Using a twin-screw extruder, polylactic acid (PLA), poly (butylene adipate-co-terephthalate) (PBAT), and poly (propylene carbonate) polyurethane (PPCU) were blended and then, the PLA/PBAT/PPCU trinary blends were made into films via the blown film technique, the mechanical tests showed that the elongation at break of PLA/PBAT/PPCU films decreased from 240 to 226% in the machine direction and from 413 to 353% in the transverse direction. The tensile strength was increased in the machine direction from 40.2 to 46.5 MPa and from 28.5 to 42.8 MPa in the transverse direction. The tear strength was from 134.7 to 143.6 kN/m in the machine direction and from 137.3 to 147.8 kN/m in the transverse direction. 13 The effect of thermoplastic starch (TPS) on the properties of PP/PE and PP/PEg-MA/PE blends and the use of nanoclay as a reinforcing filler was studied by Hammache et al. 14 and concluded that the addition of 20% TPS causes a decrease in the melt flow index due to certain compatibility between PP/PE and thermoplastic starch while the addition of 30% has an antagonistic effect. Due to the reaction between the modifying agent of the clay and Maleic anhydride being presented in PE-g-MA or/and the hydroxide in the glycerol, the addition of nanoclay to the system decreased the melt flow index. The authors claimed that the addition of a small amount of TPS and organoclay to PP/PE improved PP/PE crystallites and decreases the impact strength of the blends. In a recent study, biodegradable polypropylene (PP) reinforced with short jute fiber was prepared by melt mixing followed by hot press molding. The results show that alkali-treated jute reinforced composites have much better properties than the untreated ones. The tensile strength, tensile modulus, flexural strengths, and flexural modulus were decreased with increasing of untreated fiber and increased with the increasing of treated fiber in PP-jute composites. 15 Biodegradable modified PP fibers containing 30% recycled polylactic acid were prepared by the melt spinning process and the biodegradability and the mechanical properties of the samples were evaluated. 16 After incubation in the soil for a long time, the initial moduli and tenacity of the modified fibers decreased up to 72% and 53%, respectively. The average molecular weight measurement showed a 28% decrease in average molecular weight after 80 days of soil burial. Mangaraj et al. 17 developed a polylactic acid-corn starch-based biodegradable films along with reagents benzyl peroxide and glycidylmethacrylate using commercial extrusion blown molding method. The tensile strength of the developed films varied from 56.49 to 24.18 MPa, the addition of corn starch decreased Tg from 65.26 to 59.71°C, Tm from 146.81 to 150.56°C with a slight increase in Tc from 106.45 to 107.7°C.
There are many other published articles in which the mechanical and thermal properties of other biodegradable polymers have been investigated. Pirsa et al. 18 prepared a biodegradable film composed of starch/tragacanth gum/nanoclay and studied their physicochemical and mechanical properties and found that the addition of tragacanth and nanoclay improved the physical and mechanical properties of the starch film. Another biodegradable/antioxidant active film based on potato starch was provided by Pirsa et al. 19 and reported that the potato starch composite film containing Zedo gum and salvia essential oil showed excellent physical and antioxidant properties.
Noting the high market potential for bioplastic materials, it is evident that there are great opportunities ahead. However, some challenges need to be overcome to improve the competitiveness of bioplastic materials. As a result, significant attention must be paid to understand fully how to manipulate final material properties for various applications. The present work is an extension of previous work 20 in which the effect of Biosphere Biodegradable Plastic (BSP) on the nucleation intensity and spherulite growth rates were studied. The current work focuses on the effects of biodegradable plastic additives (BSA and D2W) on the nucleation, morphology, and crystallization temperatures of isotactic polypropylene spherulites, in addition to that, the mechanical properties (yield strength, Yong’s modulus, elongation at break, and impact strength) will be investigated and discussed. Biosphere Biodegradable Plastic (BSP) and another masterbatch that is designed to control the life of ordinary plastic products called D2W were used as polypropylene additives to accelerate its degradation. Both of these additives, BSP and D2W, were added during the polypropylene extrusion process.
Materials and methods
Materials
Isotactic polypropylene (iPP) homopolymer grade SABIC PP 575P for Injection Molding was used in this study. This PP is manufactured to be used for injection molding and its typical applications include housewares articles, caps, closures, containers, and toys. It has a density of 905 kg/m3 and a melt flow index of 11 g/10 min. SABIC PP 575P is free of any nucleating agent and has a processing temperature within the range of 220 to 240°C.
Two biodegradable plastic additives in pellets form were supplied by Biosphere Plastic (BSP) LLC-USA and D2W additive produced by Symphony Environmental-UK. Biosphere plastic has a bulk density of 500–1000 kg/m3 and a melting temperature range between 110 to 120°C. D2W additive has polypropylene as a carrier resin, a melt flow index of 5, and a bulk density of 550 g/l. 21
The manufacturer of BioSphere’s biodegradable plastic additive claims that this additive will give microbes the ability to secrete acids and consume plastics products, turning the products into CH4, CO2, biomass, and water. BioSphere’s biodegradable plastic additive enhances the ability for microbes to consume the plastic at a much faster rate by adding in key features to the polymer and this in turn enhances the biodegradability of plastics. A 1% addition of the Biosphere can increase the rate of degradation of a plastic product by 100–200 times when compared to unmodified plastics. On the other hand, the producer of D2W additive claims that the addition of D2W to polypropylene causes the final product to degrade abiotically and then biodegrade after its useful life without affecting the functionality of the product, and at the end of its useful service life, the process of oxo-biodegradation starts, and the plastic will eventually be broken down into the water, carbon dioxide and biomass. The plastic will degrade and then biodegrade in the dark or sunlight, heat or cold, land or sea, leaving no fragment, no methane, no harmful residues thus avoiding pollution and damage to the environment and wildlife.
Preparation of iPP/biodegradable plastic additive composites
To ensure good mixing BSP and D2W particles were mixed dry with SABIC iPP 575P then fed to the extruder’s hopper by direct addition. A single screw extruder (Axon ab10 Mini Extruder-Sweden) with a 10 mm screw diameter, 20×D L/D-ratio was used to prepare the iPP/BPA composites of different compositions. The weight fractions of biodegradable plastic additive for both BSP and D2W covered in this study are 0.25, 0.5, 1.0, 1.5, and 2.0; the recommended weight fraction by Biosphere Plastic is 2.0 while the recommended weight fraction of D2W is 1.0. Extruder’s temperature zones were set at 170°C near the feeder, 185 and 195°C in the middle zones, and 210°C at the die. 22 On exit from the extruder’s die (the point that gives the final shape), the extrudate passed through a water trough at a temperature of 25°C for further cooling and solidification. The cooled laces are chopped into small granules using a pelletizing machine (Axon Pelletizer) equipped with a steel blade. 23 The obtained PP/BPA composites granules were dried in an oven at 100°C to remove any moisture before they were used for the thermal and mechanical characterization.
Polarized light microscopy studies
Polarized light microscopy (PLM) (ML9430-Meiji Techno-Japan microscope) equipped with a (Mettle FB82-USA) hot stage and a Sony digital camera were used to study the morphology of melt-crystallized iPP and iPP/BPA composites. Polarized light microscope samples were cut from the prepared iPP/BPA composites and then sandwiched between two microscope cover glasses. Samples were first melted at a heating rate of 10°C/min passing the measured melting temperature of SABIC PP 575P (Tm ≈ 164°C), pressed into a thin film, and then kept for 3 minutes at a temperature of 200°C to erase melt memory effects. On termination of the 3 minutes, the molten samples were cooled rapidly to the required crystallization temperature at a rate of 40°C/min, the temperature was maintained during the period required to complete the crystallization process.24–26 A Sony digital camera fixed on top of the microscope tube and connected to the PC by a TV card along with a video recorder software were used to record the crystallization process. On completion of the crystallization process, images were captured and then analyzed to study the morphology of composites.
Differential scanning calorimetry (DSC)
DSC experiments were performed using a power-compensation-based differential scanning calorimeter DSC-7 (Perkin–Elmer). The instrument was operated in conjunction with the cryogenic cooling accessory CCA-7, ensuring a constant temperature of the heat sink. The sample and reference furnaces were purged with nitrogen gas at a flow rate of 40 ml/min. Calibration of the sensor temperature and the heat-flow rate was done according to standard procedures, using indium and tin as calibrants. Circular and flat samples with a mass of about 5 mg were cut from the nanocomposites strips and placed in the aluminum pans (Mettler-Toledo).
Samples weight of approximately 5.0 ± 0.1 mg were crimped in aluminum pans and loaded at 25°C to the DSC, heated up rapidly (40°C/min) to 220°C, and maintained at this temperature for 3 min to remove thermal history. The molten samples were then cooled non-isothermally down to room temperature at a constant cooling rate of 10°C/min.
Preparation of tensile testing samples
Six sets of each iPP/BSP and iPP/D2W composites (0, 0.25, 0.5, 1, 1.5, and 2 wt % BPA) for the impact testing were prepared by a hot compression molding (Carver Auto Series Press). Each processing cycle gives 14 tensile samples using a copper square mold. Overhead projector sheets were used to prevent the composite tensile testing samples from sticking to the machine surface. The hot press molding temperature was set at 220°C and the hot pressing cycle lasts for 5 min to ensure complete melting of the composite particles. Molten samples under compression were left to solidify and cool naturally. Figure 1 shows the exact dimensions of the tensile testing sample used in the present study. 27
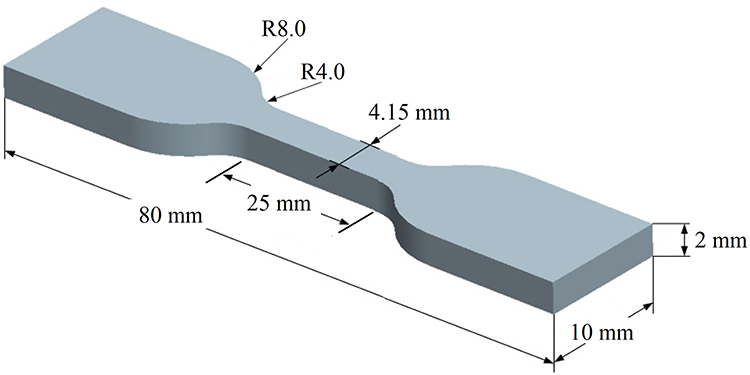
Dimensions of the tensile testing samples.
The prepared tensile testing samples were analyzed via a Hounsfield H50 K-S UTM tensile testing machine using a transducer load force of 5 kN and a testing speed of 10 mm/min. Approximately 10 replicates of each group were tested; the average of these results was used to represent the final result.
Preparation of impact testing samples
Six sets of each iPP/BSP and iPP/D2W composites (0, 0.25, 0.5, 1, 1.5, and 2 wt % BSP or D2W) for the impact testing were prepared by the same machine and procedure used to prepare the tensile testing samples. Using a steel square mold 14 impact rectangular samples were obtained in each hot pressing cycle. Figure 2 shows the exact dimensions of the impact testing samples used in this study according to ASTM 256. 28
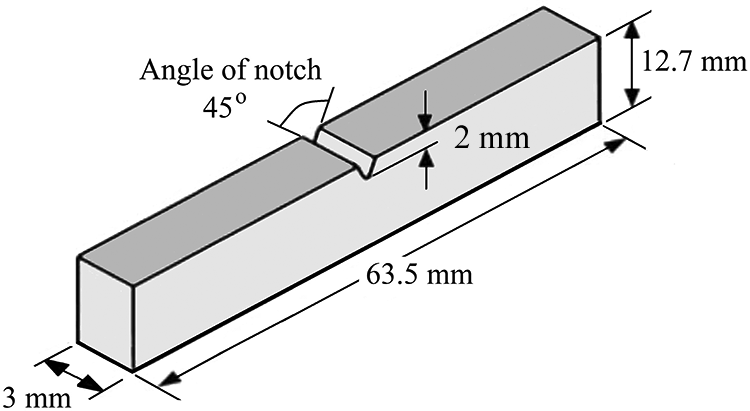
Dimensions of the impact testing samples.
The prepared impact samples were analyzed via a 6545 Ceast Impact Tester. The sample was cantered for testing, and a 7.5 J pendulum hammer was used to hit the sample. The energy needed for breaking each of the samples was recorded for further calculations; the results are expressed in energy lost per unit of the thickness (J/m) at the notch or energy lost per unit cross-sectional area at the notch (J/m2). Approximately 10 replicates of each sample were tested; the average of these results was used to represent the final result.
Testing statistical hypothesis and significance test
To determine the probability distribution of the means (μ) and standard deviations (σ) of the mechanical properties obtained in the current work, statistical hypothesis testing was employed. Mainly, the hypothesis testing for the probability distribution consists of two steps
29
: Stating a null hypothesis H0: the probability distribution of the mechanical properties of iPP/BPA composites follows either a normal, log-normal, or Weibull distribution. Deciding whether to reject or accept hypothesis 0: the decision is often taken with a probability threshold, α, which is called the significance level. The smaller the p-value is, the stronger the evidence against hypothesis 0
30
: Very strong evidence against hypothesis 0 if p-value < 0.01. Strong evidence against hypothesis 0 if 0.1 < p-value < 0.05. Weak evidence against hypothesis 0 if 0.05 < p-value < 0.1. Little or no evidence against hypothesis 0 if p-value > 0.1.
Testing methods depend on the normality of the data, the two-sample student’s t-test is the most commonly used method to evaluate the differences in the mean of two data sets. The t-test can be performed knowing just the means, standard deviation, and the number of data points. The t value is a ratio between the difference between two groups and the difference within the groups and can be estimated using the following equation:
The larger the t score, the more difference there is between groups, and the smaller the t score, the more similarity there is between groups. The bigger the t value, the more likely it is that the results are repeatable. Every t value has a p-value to go with it. A p-value is a probability that the results from the sample data occurred by chance. Low p-values are good; They indicate that the data did not occur by chance. In most cases, a p-value of 0.05 (5%) is accepted to mean the data is valid.
Results and discussions
The effects of the addition of biodegradable plastic additives (BPA) on the morphology, the crystallization temperature, and some mechanical properties will be presented and discussed in the following sections:
Effects of biodegradable plastic additives on iPP morphology
It is well known that both thermodynamic and kinetic are important factors in the polymer crystallization process. The physical, chemical, and mechanical properties of crystalline polymers depend on the morphology, crystalline structure, and degree of crystallinity. To reveal the effect of the BPA on the morphology of the iPP spherulites, isothermal crystallization was performed by rapidly cooling the sample from the melt temperature to the crystallization temperature and the whole crystallization process was monitored by the aid of a controlled hot stage and a polarized light microscope. Spherulites morphology of neat isotactic polypropylene (iPP) and iPP/BPA composites was investigated by conducting isothermal crystallization in a temperature range lies between 145 and 130°C.
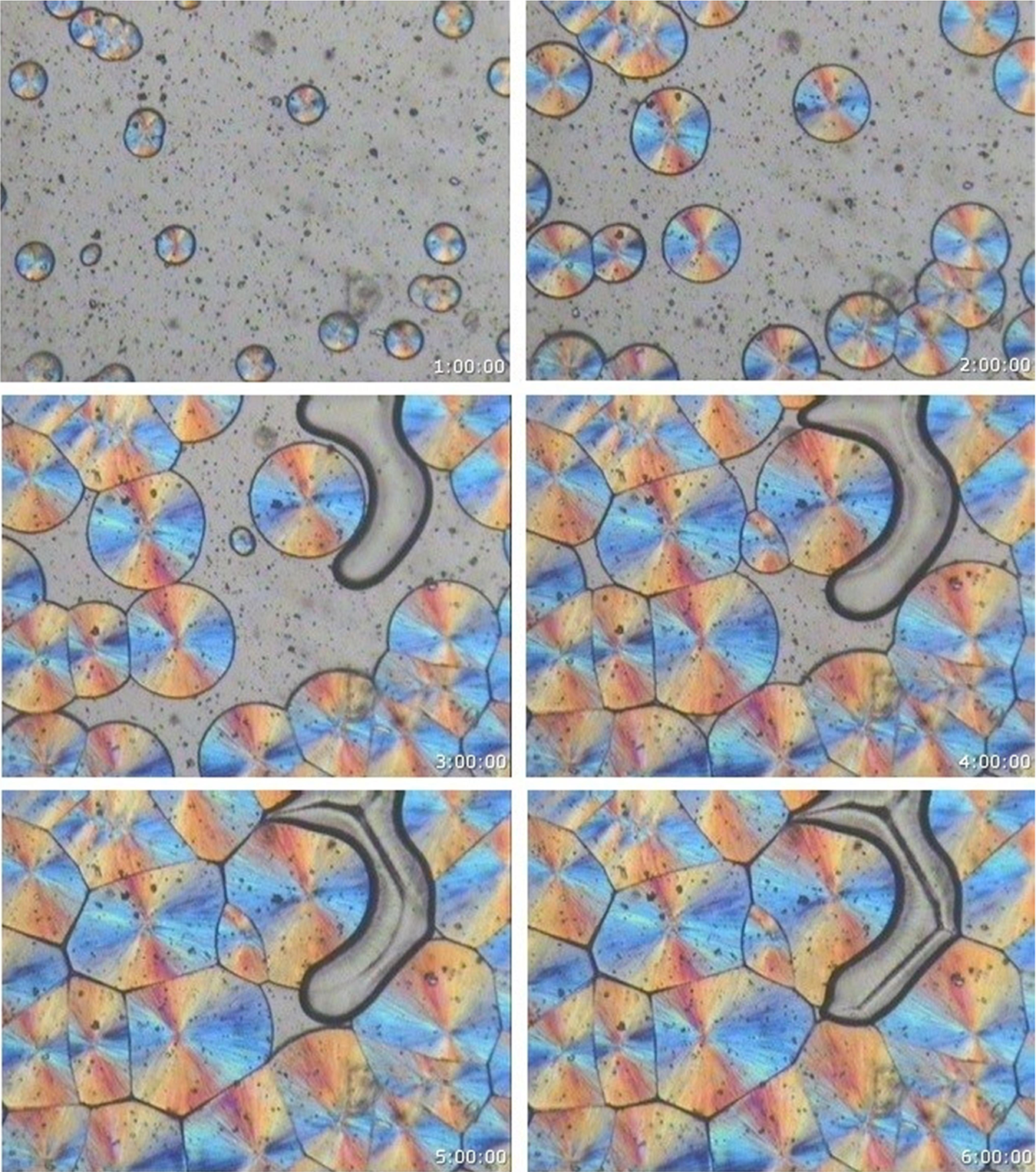
From the morphological and spherulitic growth isothermal crystallization experiments, it is observed that only the monoclinic α phase spherulites were obtained for the neat iPP, iPP/BSA, and iPP/D2W composites at all crystallization temperatures covered in the current study. It was not possible to obtain any hexagonal β spherulites at any of the covered crystallization temperatures. Figures 3 to 6 show neat iPP, 98 wt% iPP/2 wt% BSP, and 98 wt% iPP/2 wt% D2W samples during isothermal crystallization at 140°C, in all these figures, only the monoclinic phase can be observed. For other crystallization temperatures (145, 135, and 130), Figure 7 shows that only the monoclinic phase is obtained for the neat iPP and the iPP/BPA composites.

PLM photomicrographs of neat iPP during isothermal crystallization at 140°C [magnification = 133×].
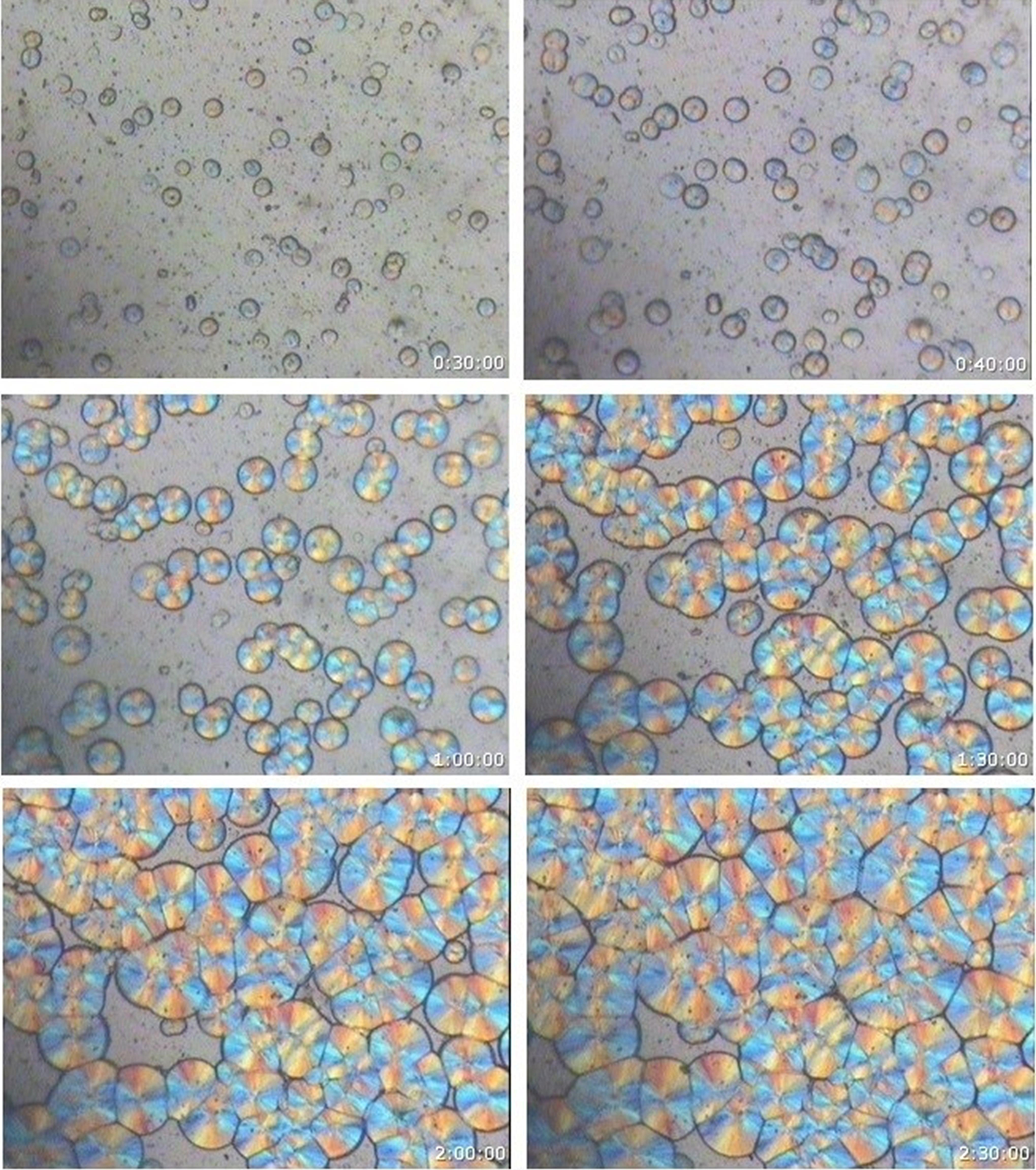
PLM photomicrographs of iPP/2 wt% BSP composite during isothermal crystallization at 140°C [magnification = 133×].
PLM photomicrographs of iPP/2 wt% D2W composite during isothermal crystallization at 140°C [magnification = 133×].
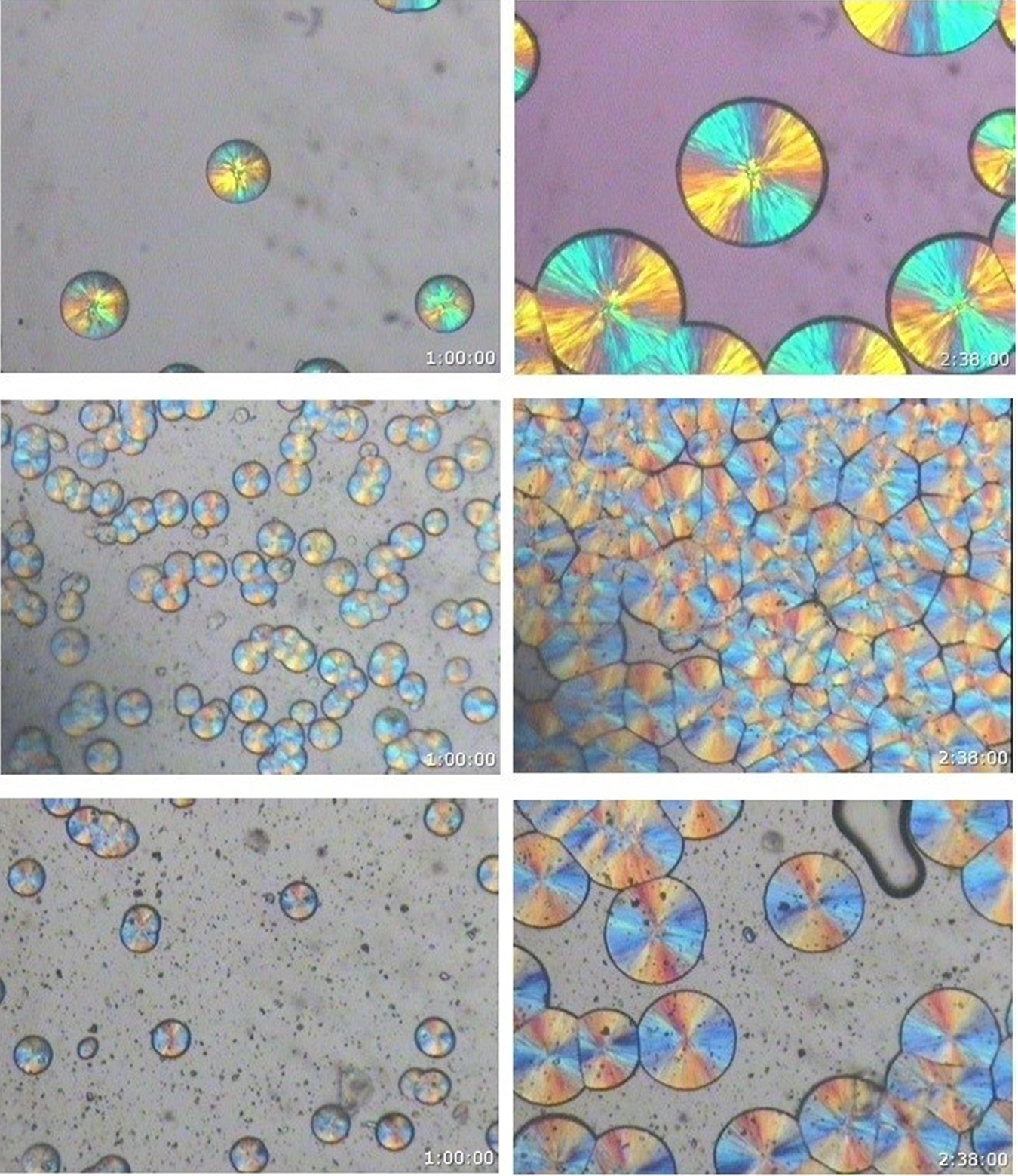
PLM photomicrographs of neat iPP (top) iPP/2 wt% BSP (middle) and iPP/2 wt% D2W (bottom) show the intensity of crystals during isothermal crystallization at 140°C [magnification = 133×].

PLM photomicrographs of neat iPP (A, B, C, and D) 98 wt% iPP/2 wt% BSP composites (E, F, G, and H) and 98 wt% iPP/2 wt% D2W composites (I, J, K, and L) during isothermal crystallization at 130 (A, E, I) 135 (B, F, J) 140 (C, G, K) and 145 (D, H, L) [magnification = 133×].
It is clearly seen that the biodegradable plastic additives used in this study work as nucleating agents with different nucleation efficiencies. In comparison with the neat iPP (Figure 3), iPP/BSA and iPP/D2W composites isothermally crystallized at the same temperature have higher spherulites density as shown in Figures 4 and 5. Biosphere Plastic has higher nucleation efficiency when compared with D2W additive, the number of spherulites that exist per same unit area is higher and the final size of these spherulites is much smaller than those spherulites grow in neat iPP or iPP/D2W composite. Obviously, these two biodegradable plastic additives play a rule of nucleating agents with different nucleation strength, they both enhance the high nucleation rate and increase the intensity of the spherulite.31–33 As the number of nuclei increases, the spherulites will impinge early in a limited space, this will end up in a smaller spherulite size without a well-defined spherulite structure and boundaries, and this can be clearly seen in Figure 7E, F, and I.
Generally, for melt crystallization, higher crystallization temperatures result in a decrease in the degree of supercooling and consequently a decrease of nucleation and growth rate. This can be observed in Figure 7A, B, C, and D as an example for neat iPP, the four samples were isothermally crystallized at 130, 135, 140, and 145°C respectively, and it is visible the differences in sizes and the number of spherulites per unit area for the samples. It can be observed that, with the increase of crystallization temperature, the spherulite growth rate of the sample decreases rapidly. This is because the crystallization rate of growth depends on the chains in the crystal diffusion and structured deposits. With the increase of crystallization temperature, the melt viscosity decreases, increasing the chain segment mobility, the crystal diffusion rate of the chains increases. The addition of biodegradable plastic additives (BSP and D2W) increases the nucleation and hence smaller spherulites were obtained at the same crystallization temperature as shown in Figure 7D, H, and L. At all crystallization temperatures covered in this study, the number of spherulites created in the iPP/BSP > iPP/D2W > neat iPP while the size of the final spherulites of neat iPP > iPP/D2W > iPP/BSP.
El-Hadi 34 found that the addition of Poly (3-hydroxy butyrate) (PHB) and cellulose nanocrystal (CNC) resulted in a decrease in the final size of spherulites in the poly (lactic acid) (PLLA) biocomposites. It is also concluded that the addition of CNC and PHB to PLLA leads to an increase in the number of spherulites with a diameter of less than 1 µm and the crystallization of PLLA is accelerated. Jia et al. 35 studied the morphology of graphene oxide (GO)/polyethylene glycol (PEG)/polylactic acid (PLA) matrix and found that GO works as a nucleating agent for PLA crystallization, while PEG can be an efficiency plasticizer that accelerates the motion of PLA molecular chains. Celli et al. 36 reported that, in isothermal conditions, for crystallization temperatures varying between 123 and 138°C, the number of spherulites per unit area does not depend on crystallization time and temperature. Instead, at small undercooling, the total number of nuclei per unit area remains independent of crystallization time but decreases with increasing temperature.
Effect of BPA on iPP crystallization temperature
DSC cooling thermograms at 10°C/min for neat iPP and iPP/BPA composites are shown in Figures 8 and 9. It can be observed that iPP/BSP composites start to crystallize at a higher temperature compared to the neat iPP and iPP/D2W composites. The difference between neat iPP crystallization temperature and iPP/BSP composites increases as the weight fractions of BSP increases. This obviously indicates that the Biosphere polypropylene additive acts as a strong nucleating agent for the iPP. Although the addition of D2W additive to iPP increases the nucleation and hence the number of spherulites per unit volume the crystallization temperatures of iPP/D2W composites are slightly lower than those of the neat iPP as shown in Figure 9. This reduction can be attributed to the increase in the viscosity of the composite by increasing the weight fraction of D2W, the melt flow index of the neat iPP is 11 g/10 min while the melt flow index of D2W additive is 5 and this may lead to this reduction in the viscosity of the final composite and hence delay the nucleation process.
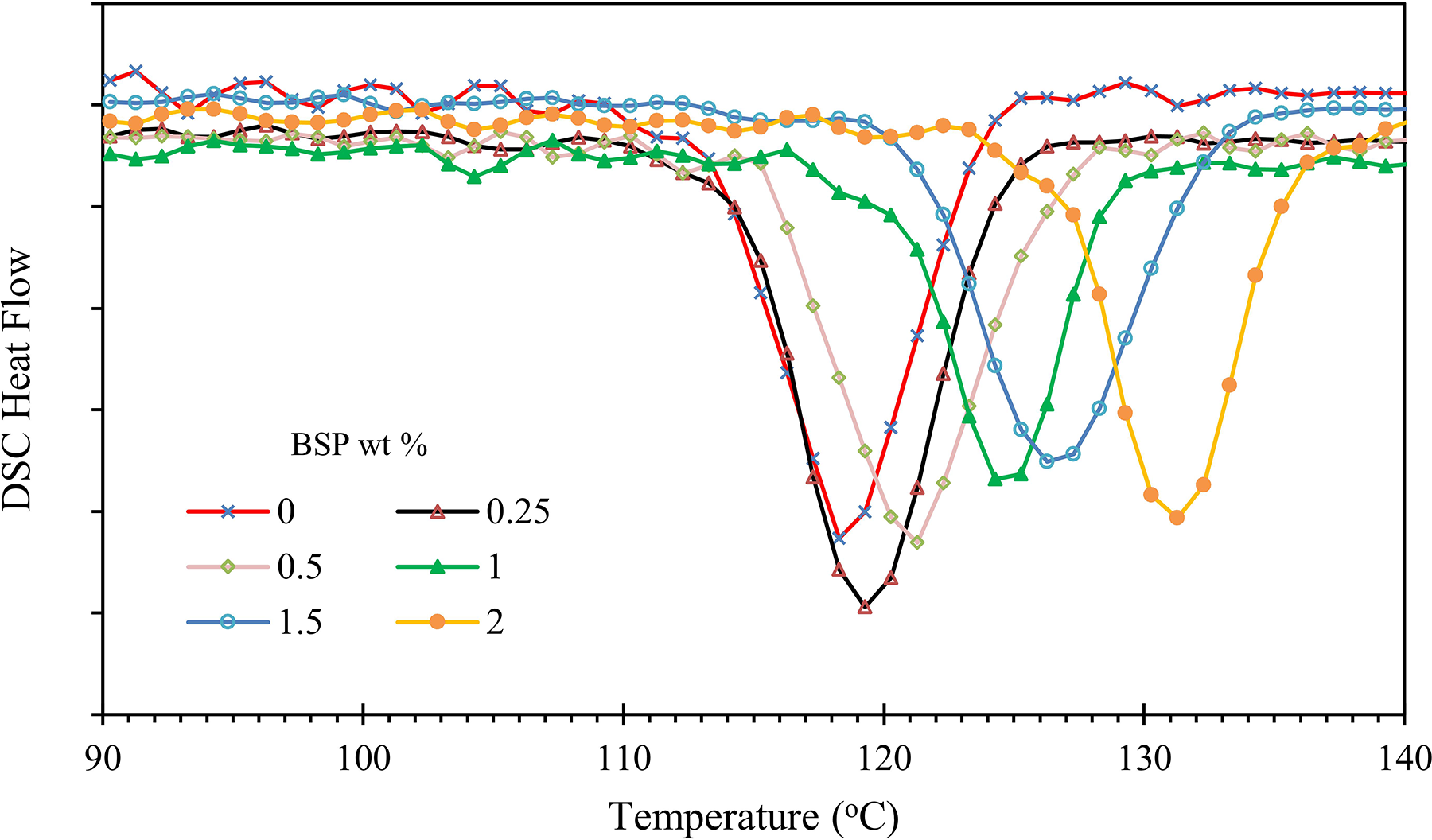
DSC cooling thermograms for iPP/BSP composite cooled non-isothermally at 10°C/min.
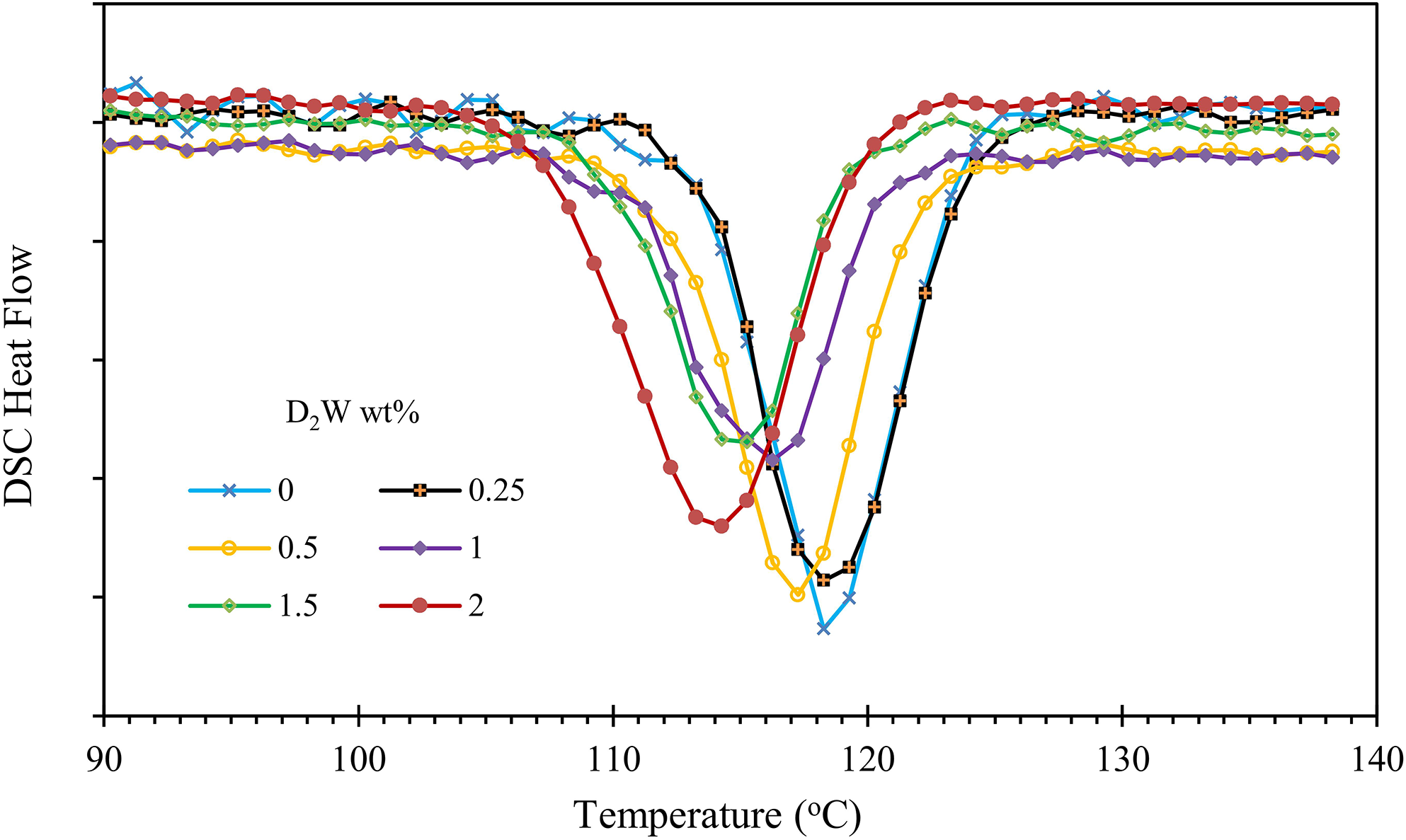
DSC cooling thermograms for iPP/D2W composite cooled non-isothermally at 10°C/min.
The addition of 1.0 wt% of BSP to iPP increased its crystallization temperature by 5°C while the addition of the same weight fraction of D2W decreased the temperature by 2°C. On the other hand, the addition of 2 wt% of BSP increased the crystallization temperature by 8.5°C and the addition of a similar quantity of D2W decreased the temperature by 3.2°C as shown in Figure 10. This proves that BSP has a stronger effect on the thermal properties of iPP compared with D2W.
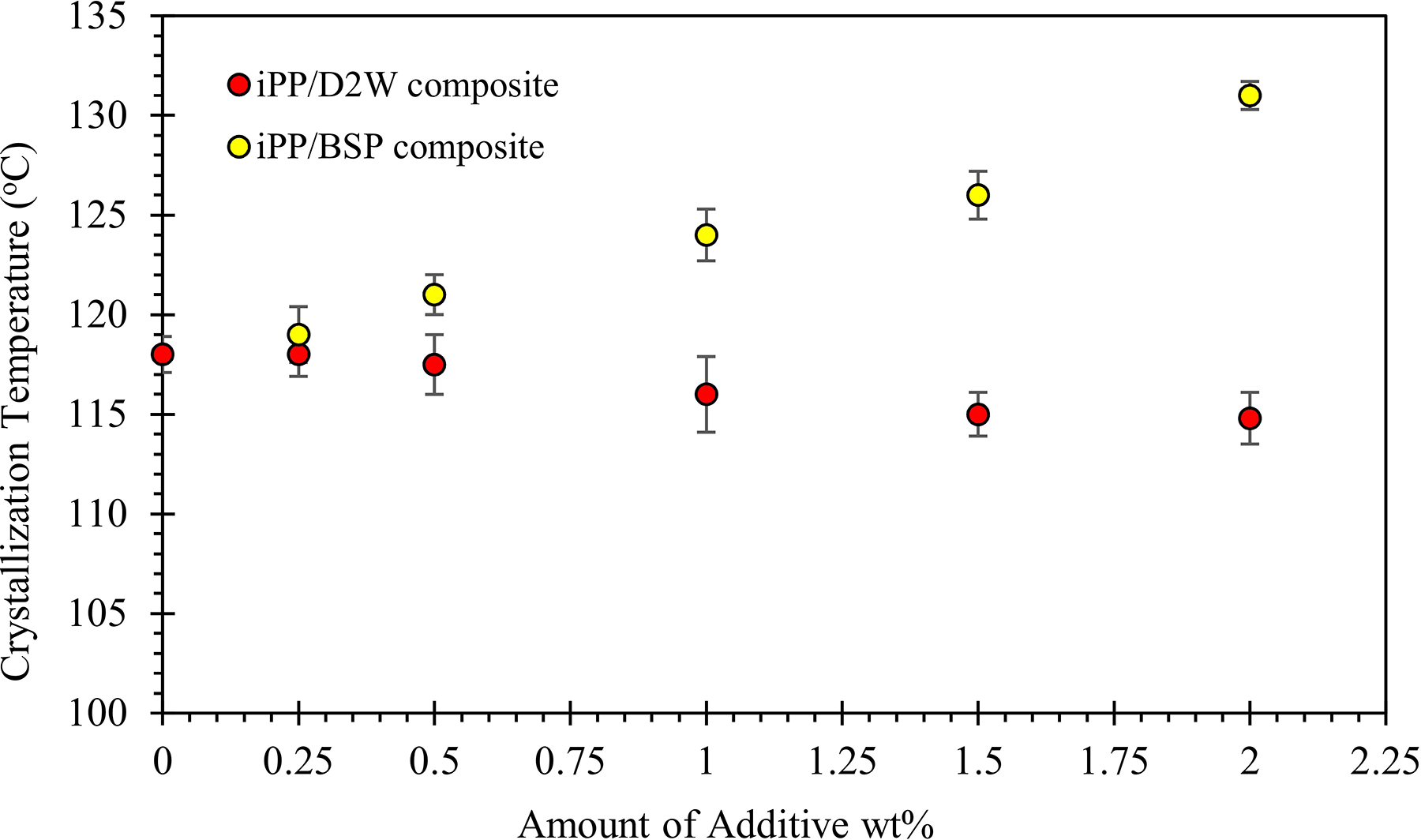
Isotactic polypropylene crystallization temperature as a function of biodegradable plastic additives weight fraction.
An et al. 37 studied the effects of phosphate nucleating agent (NA), carboxylate nucleating agent (MD), rosin type nucleating agent (WA) and sorbitol nucleating agent (NX) on the crystallization behavior of isotactic polypropylene and reported that these nucleating agents significantly increase the crystallization temperature of polypropylene as a result of the reduction in nucleation free energy. The authors claimed that the sorbitol nucleating agent has a better effect on improving the crystallization of the polypropylene and led to a high crystallization temperature. In another study, 38 crystallization temperature for polypropylene component compatibilized with different amounts of maleated thermoplastic elastomer decreased from 120.5 to 115°C when the weight fraction of the thermoplastic elastomer increased from 0 to 30%.
Mechanical properties of iPP composites
The mechanical properties of neat iPP and iPP/BPA composites are presented in Figures 11, 12, 13, and 14 where the yield strength, Young’s modulus, elongation at break, and impact strength are shown respectively. Yield strength is a measure of the force that a material can withstand before it suffers macroscopic plastic deformation. For plastics, it is taken as the peak of the stress–strain curve, as that is simpler to measure. Several factors determine the yield strength of plastics, these include, the volume fraction of filler added, the surface area of the filler, particle shape, the level of adhesion between the filler and polymer, as well as the thickness and nature of the interphase between the two phases. 39 The yield strength of iPP decreased by the addition of both BSP and D2W, 26% reduction was obtained by the addition of 2 wt% of D2W while only 14% reduction is shown by the addition of similar weight fraction of BSP. The obtained results are in good agreement with Mustafa’s results 40 where the author studied the effect of kaolin addition to PP/LDPE composites on the yield strength and reported a reduction from 3.4 to about 2.1 MPa by the addition of 14 wt% kaolin to the matrix. Usually, it is considered that spherical fillers such as calcium carbonate, dolomite, and talc do not reinforce at all, and in fact, they usually reduce the yield strength of the material. 41
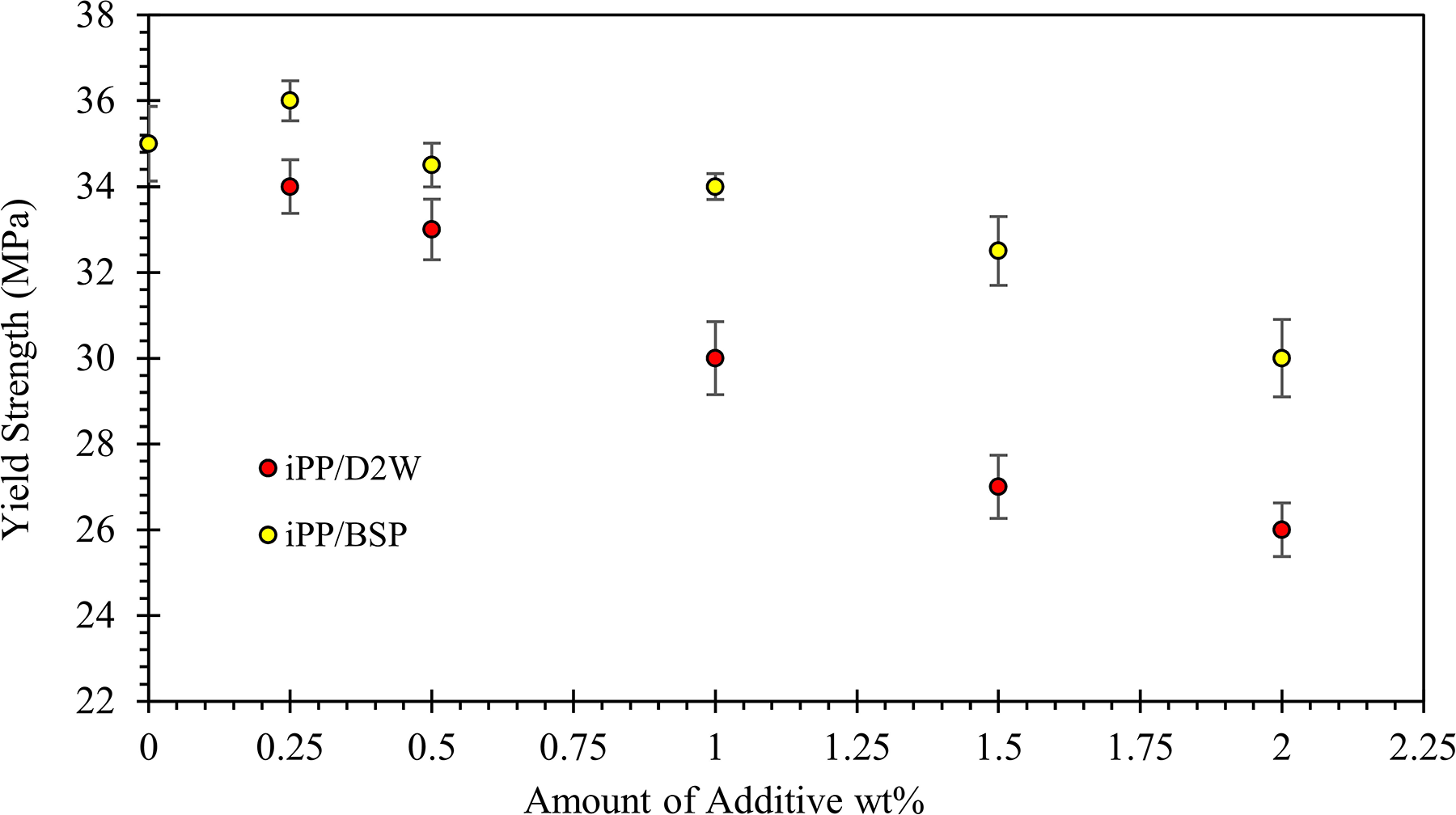
Yield strength of iPP as a function of the weight fraction of biodegradable plastic additives.
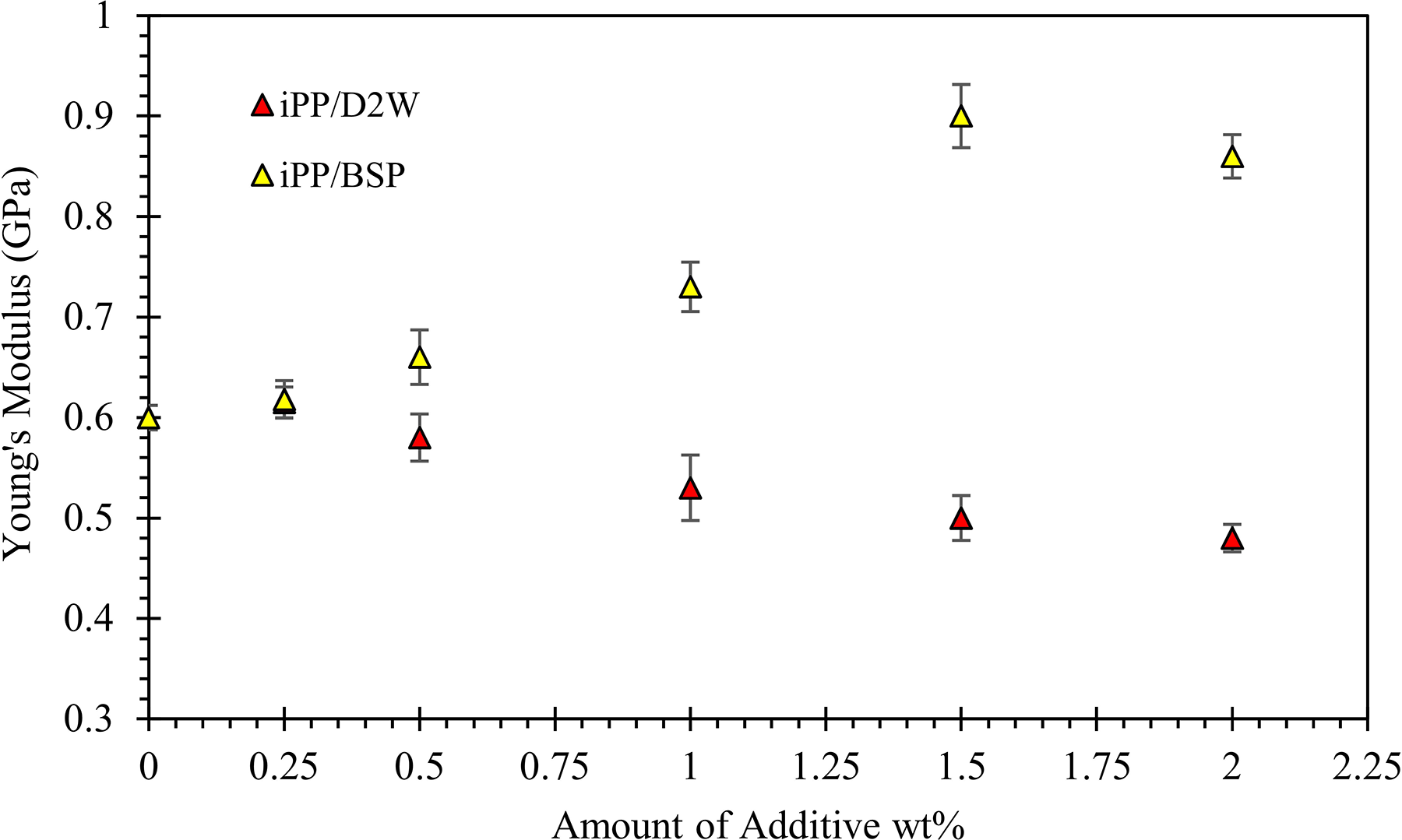
Young’s modulus of iPP as a function of the weight fraction of biodegradable plastic additives.
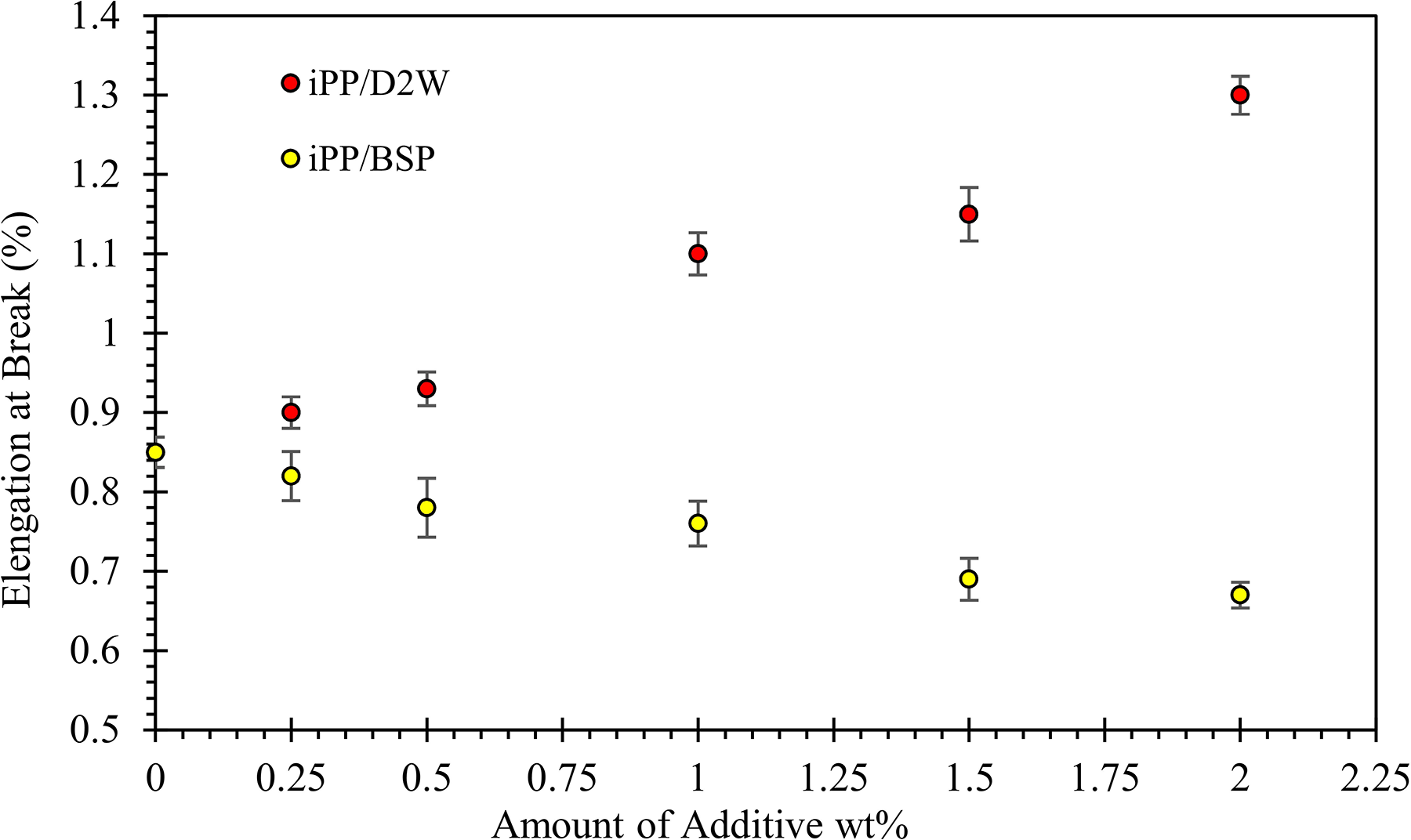
Elongation at break of iPP as a function of the weight fraction of biodegradable plastic additives.
The Young’s modulus which reflects the strength of the interaction between the polymer and the additives is presented in Figure 12. The figure shows that Young’s modulus of iPP increases and then decreases as the weight fraction of the BSP increases, this means that there is an optimum value for BSP addition into the iPP matrix, beyond this optimum value, Young’s modulus drops down. The maximum Young’s modulus value obtained is for iPP filled with 1.5 wt% BSP. It is noticed that iPP/BSP composites become brittle as the percentage of the BSP increases. During the tensile testing, iPP/BSP composites with a high weight fraction of BSP showed little extension and the elongation at break was lower when compared with neat iPP samples as shown in Figure 13. On the contrary, more than 50% increment in the elongation at break can be achieved by the addition of 2 wt% of D2W, but Young’s modulus values were lower than those iPP/BSP composites.
In general, by increasing the weight fraction of BSP Young’s modulus increases while the elongation at break decreases, and this is expected and can be attributed to a large number of nuclei sites present within the sample as the BSP amount increases. A large number of nuclei will produce more fine spherulites and as a result of impingement of the spherulites with each other, more grain boundaries will be created and hence producing materials with good strength but brittle with low elongation. 42 The other biodegradable plastic additive (D2W) shows the opposite, its presence in the iPP matrix decreases Young’s modulus and increases the elongation at break, and this can be attributed to the lower number of nuclei sites created during the crystallization stage which leads to larger spherulites.
An increase in Young’s modulus for PP/LDPE composite from 175 to almost 345 MPa by the addition of 14 wt% kaolin was reported by Mustafa. 40 On the contrary, Ammar et al. 43 found that talc has a positive effect on the stiffness of polypropylene but a deleterious effect on the tensile strength and ductility. Micron-sized glass fibers, talc, and CaCO3 were added into the recycled polypropylene granules (RPP) and characterized by Yetgin et al. 44 The authors reported that the yield strength of RPP polymer decreased by 15.3 and 26.9% with the addition of 30 wt% talc and CaCO3, respectively, and that was attributed to easing of separation of talc and CaCO3 additives from the matrix due to the existence of a weak customization interface. On the other hand, the yield strength of GF reinforced composites found to be higher and explained by the creation of strong bonds with the PP matrix at the same contribution rate, the yield strength of RPP + 30% GF polypropylene composite increased by 142% compared to the RPP polymer. The modulus of elasticity increased with talc, CaCO3, and glass fiber inclusion into the RPP, elastic modulus increased by 42, 52, and 300% with the addition of 30 wt% of talc, CaCO3, and GF, respectively.
Figure 14 represents the effect of the biodegradable plastic additives on the impact properties of iPP composites. While the addition of BSP decreased the impact strength of iPP, the inclusion of D2W as a biodegradable plastic additive to iPP increased its impact strength slightly. The addition of 1.0 wt% of BSP reduced iPP impact strength by about 43% and by doubling the weight fraction of BSP the reduction approached a percentage of 55%. On the other hand, the incorporation of 1.0 wt% of D2W into the polypropylene matrix resulted in a slight increase in the impact strength, and by doubling the weight fraction added, the impact strength increased by about 25%. At very low weight fractions of BSP (<0.5), the impact strength of the composites presented little increase before their impact strength dropped by increasing the amount of BSP. It seems that the incorporation of the BSP improves the tensile strength of iPP but reduces the toughness leading to poorer impact strength. 45 It is also noticed that iPP/2 wt% BSP composites were brittle compared to the composites with less BSP weight fraction or when compared with the neat iPP and iPP/D2W composites.
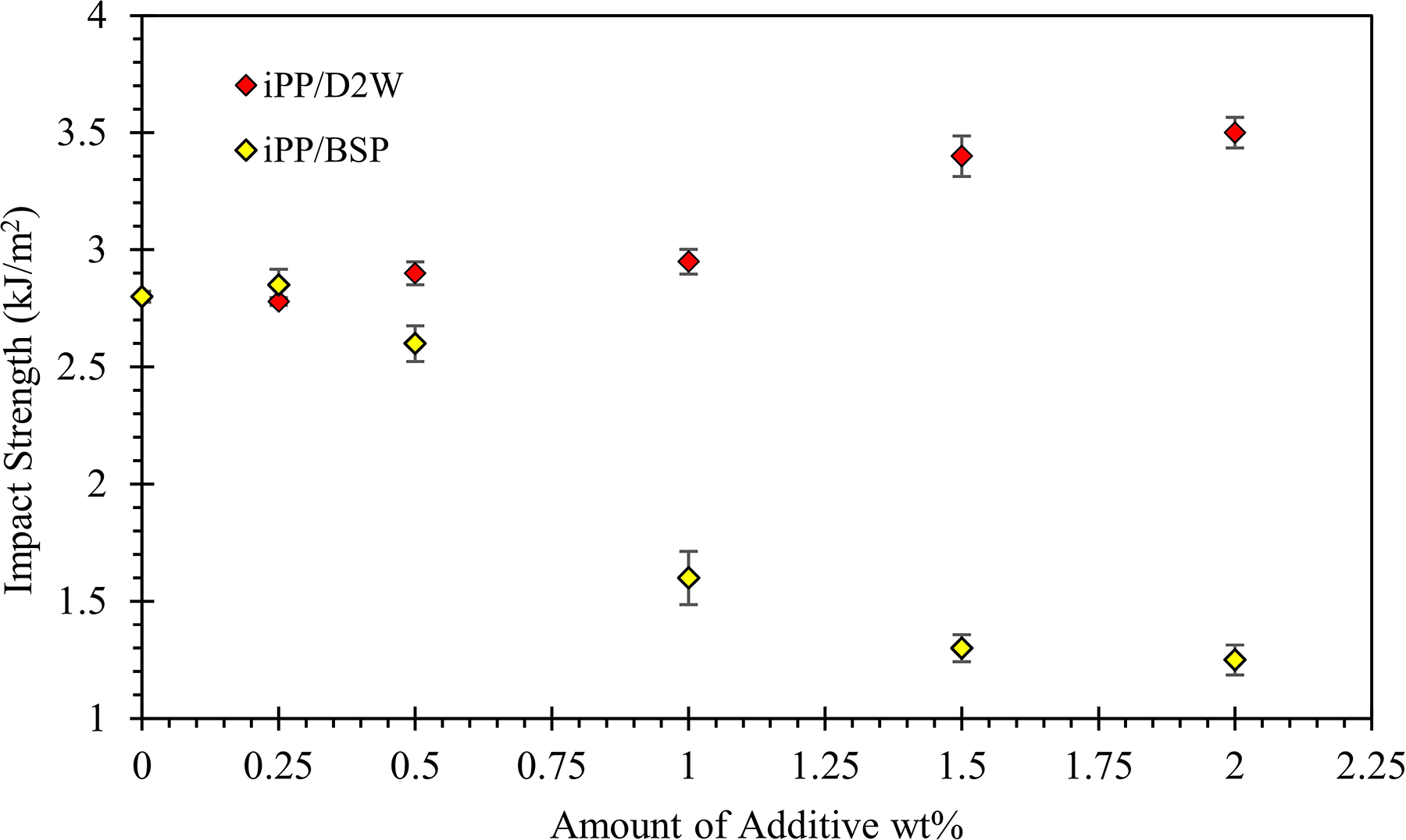
Izod impact strength of iPP as a function of the weight fraction of biodegradable plastic additives.
The impact strength of RPP polymer decreased with the addition of 30 wt% of talc and CaCO3 while the addition of the same weight fraction of GF increased RPP impact strength. 44 Mubarak and Abu-Halimeh 46 reported a strong enhancement of polypropylene’s impact strength by the addition of styrene-butadiene-styrene into the matrix, the addition of 40 wt% of SBS to polypropylene increased its impact strength by 320%.
Statistical analysis of iPP/BPA composites mechanical properties
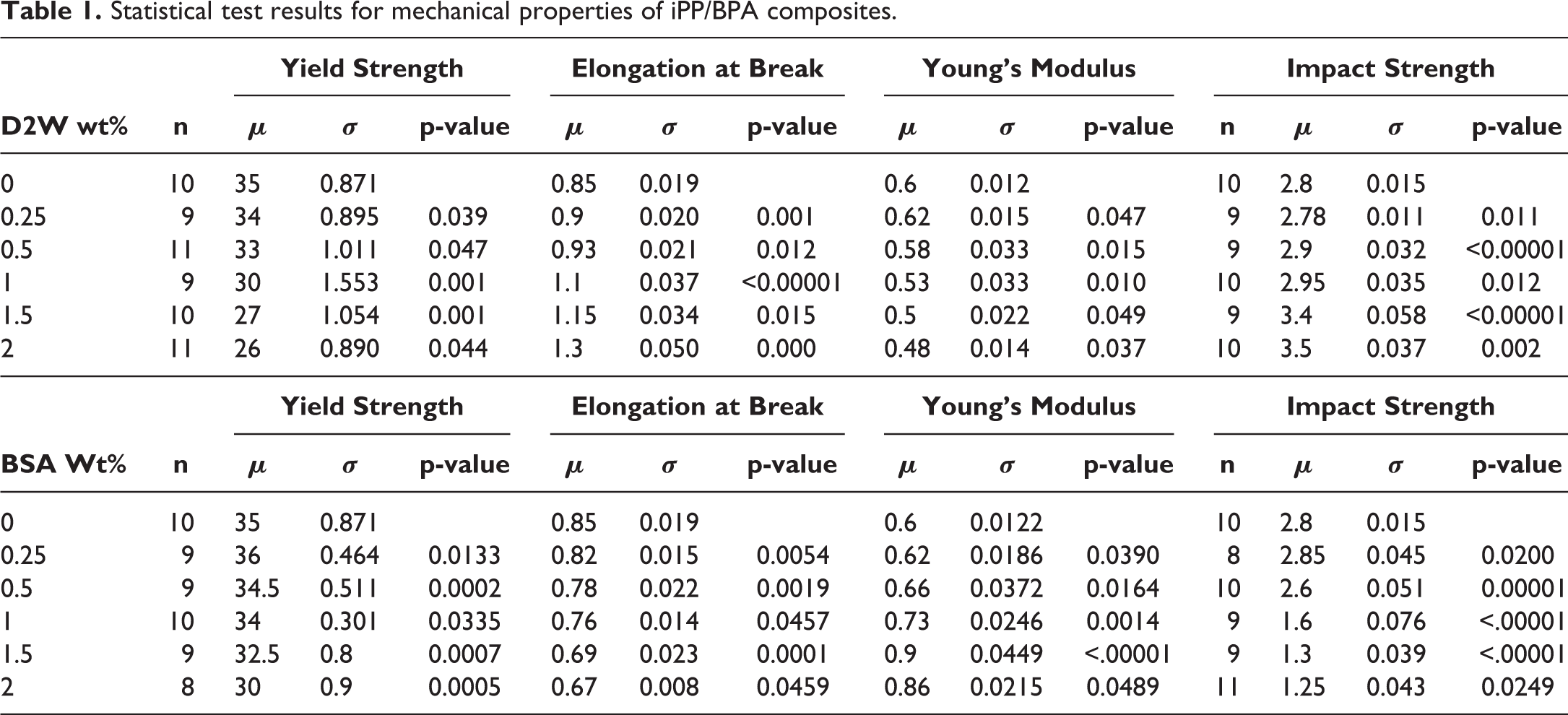
The purpose of the significance test is to evaluate the significance of the differences in the mean values of the mechanical properties obtained for iPP at different BSP or D2W weight fractions. This allows determining if the additive content added to the iPP influence the mechanical properties or not. Table 1 summarizes the mean and standard deviation of the studied mechanical properties in addition to the p-values obtained from the two-sample student’s t-test. Table 1 shows that differences between the mean values of yield strength, Young’s modulus, elongation at break, and impact strength are all significant at α = 0.05 for all iPP/BSP and iPP/D2W composites. It is known that the smaller the p-value, the stronger the evidence against the null hypothesis. It can be observed from Table 1 that p-values were less than 0.049 for all the test results, this indicates that the differences between the mean values of the data sets were significant at α = 0.05 which indicates that the results are repeatable and the data did not occur by chance.
Statistical test results for mechanical properties of iPP/BPA composites.
Conclusions
It is concluded that the biodegradable plastic additives used in this study (BSP and D2W) behave the same as nucleating agents when added to isotactic polypropylene matrix, both increased the number of nucleation sites and hence the number of spherulite per unit volume during isothermal crystallization. Compared with the neat iPP, the density of spherulites per unit area is greater for iPP/BSP and iPP/D2W composites at all crystallization temperatures covered in the current study. Also, the size of the final spherulites is found to be much smaller for the iPP/BSP composites.
The incorporation of BSP into the iPP matrix enhances the crystallization process and allow the nuclei to be born at a higher temperature, while the presence of D2W delays the born of the nuclei. By the addition of 2 wt%, BSP raised the crystallization temperature from 118 to 131°C, while, the addition of the same weight fraction of D2W delayed the crystallization by about 3.2°C. It seems that the addition of D2W increases the viscosity of molten polypropylene composites and hence reduces the mobility of polypropylene chains and consequently delays the initiation of crystallization.
The yield strength of iPP decreased from 35 to 30 and 26 MPa with the addition of 2 wt% of BSP and D2W, respectively. Young’s modulus increased with the addition of BSP by 43% and decreased by 20% by the addition of 2 wt% of D2W into the iPP composites. More than 50% increment in the elongation at break can be achieved by the addition of 2 wt% of D2W while a brittle behavior was observed for iPP/BSP composite with lower extension compared to neat iPP samples. Also, the addition of BSP to iPP increased the brittleness of the resulting composites and hence decreased the impact strength. On the contrary, the addition of 2 wt% of D2W into the iPP matrix increased its ductility and hence its impact strength by 25%.
Footnotes
Acknowledgement
This work has been carried out during sabbatical leave granted to the author Yousef Mubarak from the University of Jordan during the academic year 2019–2020.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
