Abstract
In this paper, an experimental study was conducted to characterize industrial PVC pipes and to investigate the effect of hydrothermal aging on their physico-chemical, thermal, and mechanical behavior. Three temperature (25°C, 60°C and 90°C) and full immersion in distilled water were retained as accelerated hydrothermal conditions. Kinetic of water absorption was examined and Fickian behavior was observed. The aging temperature was found to influence the water uptake behavior of PVC samples. Thermogravimetric analysis (TGA) has proved that the pipe material is not pure, while it consists of PVC reinforced with calcium carbonate (CaCO3). After exposure to accelerated aging, TGA and FTIR analysis exhibit preliminary signs of degradation of PVC samples under the retained conditions. Changes affecting the shape and the color of aged samples were examined. Mechanical properties have been characterized, after immersion of 30 days, with an improvement of strength and stiffness of the aged samples, in particular at elevated aging temperature. However, the aging response is accompanied by a loss of ductility for the aged material. These results, even for brief exposure, could help to understand the behavior of PVC composite pipes under hydrothermal conditions.
Introduction
The use of polymeric pipes is conceivable in various applications due to both environmental, and economic benefits (low cost of installation and maintenance). Over the years, these polymer pipes were widely used for petroleum, gas and water transportation and distribution.1–3 From the used polymer materials, the thermoplastics are the best candidate, such as the polyvinyl chloride (PVC), 2 the polyethylene (PE),4–6 the polypropylene (PP). 7
However, the service life of these pipes remains dependent on the service conditions. They can be exposed to several factors or loadings reducing their service life, such as internal, or external pressure, attack of hot or cold fluid (gas or liquid). In this view, several studies were conducted about the investigation of the mechanical characterization of polymeric pipes and the evaluation of properties deterioration.2–4 Here, the main common goal is to enlarge their potential application. In 2005, Krishnaswamy 8 carried out creep testing on high-density polyethylene (HDPE) pipes, the results have shown that they are ductile and brittle materials. It is also to be noted that for most PVC applications, fillers are added to reduce formulation cost and to enhance the material performance. For instance, calcium carbonate (CaCO3) particles have been used as fillers in thermoplastics and especially in PVC. The purpose of adding CaCO3 particles is to lower the material costs of the finished products. 9 The CaCO3 particle size is generally comparable in size with particles of PVC and hence are capable of generating interparticle friction with PVC particles, increasing shear, and promoting gelation. 10 CaCO3 could also act as a fusion promoter. One possible explanation for the observed trend could be that the finer particles having a larger surface area would have a larger contact surface with the PVC particles, creating a higher shear heating and increasing the fusion speed. 11 CaCO3 is usually surface treated with stearic acid, producing a layer of calcium stearate on the filler surface. This results in improved processing and dispersion (mechanical properties) and improved moisture resistance. 12
In 2016, Guermazi et al. 2 have focused on the effects of the content of CaCO3 fillers in PVC matrix and environmental conditions on water absorption, thermal stability, mechanical properties, and wear properties of PVC/CaCO3 composite pipes. Besides, many researchers have focused on the study of the durability of the polymer pipes which were initially manufactured based on the service life of 50 years, as reported in Ref. 13 Here, the use of accelerated conditions is more suitable method to analyze the durability of pipes. In particular, various studies have retained hydrothermal aging methodology as aggressive conditions to provoke the rupture or failure of pipes damage. Different temperatures or/and different aging environments (humidity, distilled water, seawater) were widely used at laboratory scale to study materials aging.5,14
From the literature review, polymeric materials become plasticized, softened, and swelled when exposed or subjected to humidity. 15 Studies dealing with the effects of moisture absorption on the properties have revealed that moisture absorption affects the material strength, leading to the degradation of the material’s mechanical properties. 16
Recently, Mei et al. 17 have found that mechanical properties, in terms of average strength and average stiffness, of carbon fiber reinforced plastics (CFRP) composite decrease considerably as a function of aging time under 50°C hydrothermal environment. According to Larbi et al. 18 and Aldajah et al., 19 the hydrothermal aging of polymeric materials can induce a loss of rigidity and a loss of resistance to breaking, while a gain of ductility (larger elongation or strain at break) due to swelling phenomenon.
Bao 20 has recently shown that with the prolonging of hydrothermal aging period, the mechanical properties of CMR/PLA bio-composites were decreased. In particular, the bending and tensile strength characteristics of composites were dramatically affected by both time and aging temperature: the higher the temperature, the more significant decrease in the material performance. In general, absorption or diffusion of water into polymeric material degrades and weakens the strength material. Also, if the polymeric material plasticizes, mechanical properties will be affected but this depends on some parameters such as time, temperature, relative humidity, and external stress.21,22
Our goal through this work is (i) to conduct physico-chemical, thermal, and mechanical investigations of industrial PVC pipes and (ii) to investigate their properties under the combined effects of two parameters namely temperature and relative humidity.
Materials and techniques
Material and samples
The material used in the current study consists of polymeric pipes based on polyvinyl chloride (PVC) reinforced with calcium carbonate (CaCO3) fillers, and some other additives (plasticizer and stabilizer). In particular, the commercial dioctyl phthalate (DOP) was used in the formulation of pipe material as plasticizer.
The polymeric pipes, which used for water boreholes, are produced by the extrusion process making use of an extruder with twin-screw at counter-rotating mode. Other details were given in our previous paper. 23 Samples of PVC pipes were supplied for this study having a 200 mm external diameter and 8 mm of thickness. Samples, dedicated to characterization, were prepared by a DMTC-PX1-CNC milling machine according to ISO 2818:1994 after cutting strips from the pipe section according to ISO 6259-1:2015 standard.
Dimensions of tensile samples were chosen according to the ASTM D638-M-I specifications. Figure 1 illustrates the dimensions of the pipes, the dimensions of the samples, and how they were extracted.
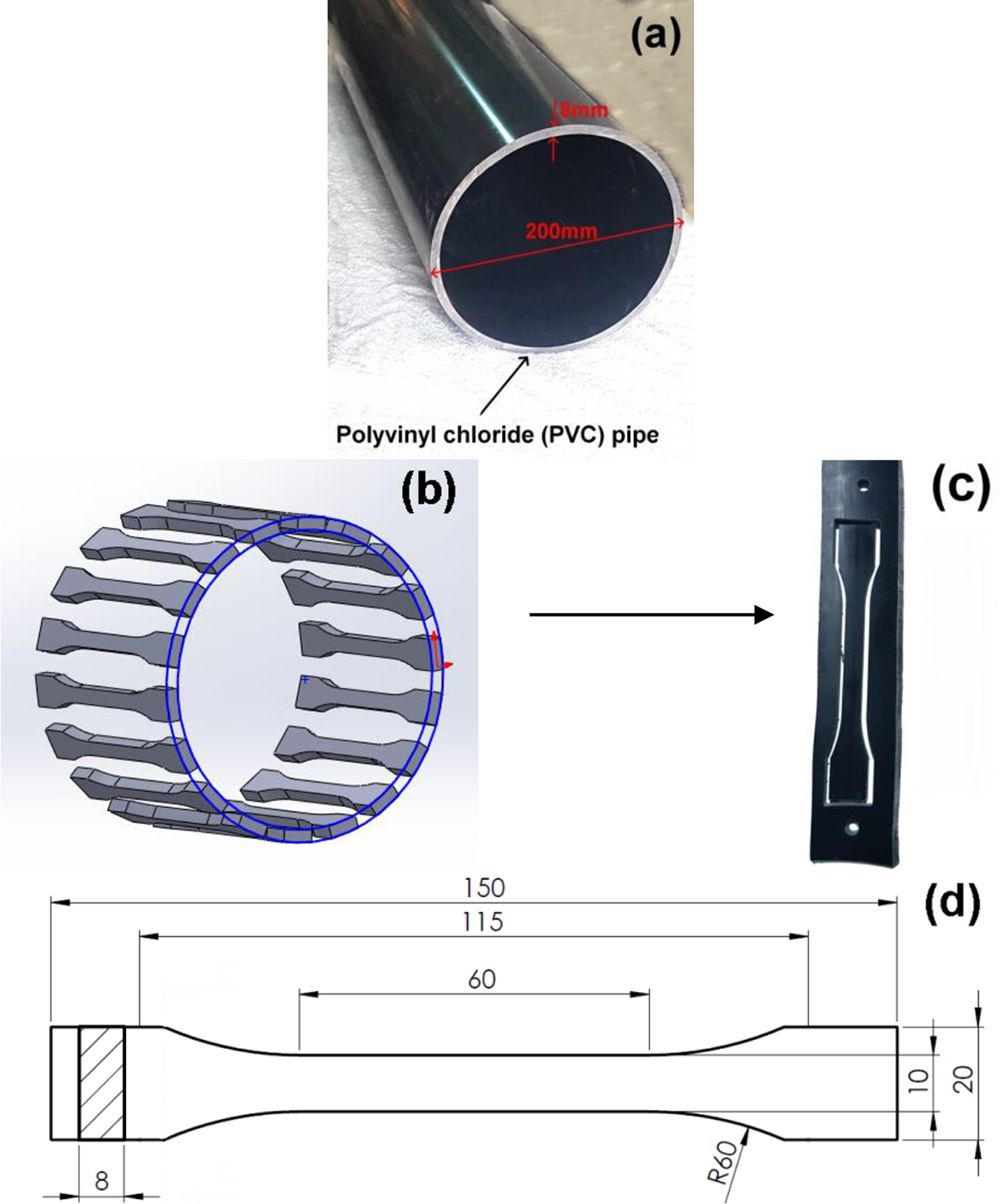
Extraction method and dimensions (in mm) of PVC pipe and samples. (a) Dimensions of pipe; (b) extraction of samples from tube; (c) sample and strip; and (d) sample dimensions.
Water uptake measurements
Here, a set of pipe specimens were undergone to accelerated hydrothermal aging. Haltered and flat samples were immersed in an aqueous environment (distilled water) and aged at three different temperatures (25°C, 60°C and 90°C) for around 90 days. Here, it is true that the maximum of service temperature for PVC pipes does not exceed 90°C, but such temperature was selected to simulate accelerated weathering conditions since the study of natural aging is time consuming. 14
The moisture diffusion into the sample was then measured. Water uptakes were measured through a gravimetric method in which samples were taken out from the water, wiped with filter paper to eliminate surface water, weighted at regular intervals with the digital balance of 10−4 g accuracy, and then immersed again in water.
The water uptake rate, (
where
Thermogravimetric analysis (TGA)
Thermogravimetric analysis (TGA) and derivative thermal gravimetry (DTG) were investigated by using a TGA-DTG (STA PT 1600, LINSEIS, GERMANY). The analysis was performed under nitrogen flow (20 ml/min) at a temperature interval of 25 at 700°C. The used weight was about 10–20 mg and the rate of heating is 20°C/min.
Fourier transform infrared spectroscopy (FTIR)
Fourier transform infrared spectroscopy (FTIR) spectra of the unaged and aged PVC samples were recorded using an FTIR (Cary 630 FTIR, Agilent technologies) in the range of 4000–400 cm−1 with a resolution of 4 cm−1. Thin films, which were between 0.5 and 1 mm in thickness, were cut from the tube sample for the analysis.
Tensile tests
Tensile tests were performed at room temperature using a universal testing machine of the type WDW-10 E with a crosshead rate of 5 mm/min. Five measurements were conducted and an average for the final result was considered.
Mechanical properties obtained from tensile tests of PVC samples were determined for different immersion temperatures (25°C, 60°C and 90°C) after regular intervals of time.
Results and discussion
Kinetics of water absorption
Figure 2 shows the behavior of PVC samples immersed in the water according to the aforementioned conditions. Curves show that the specimens immersed followed the same tendency of the weight change. The three curves exhibit the same behaviors with two stages of absorption: in the first one, the sorption curve is a linear function of the square root of time (Fick’s law of diffusion). Here, it is to note that such curve slope was used to estimate the diffusion coefficient (

where
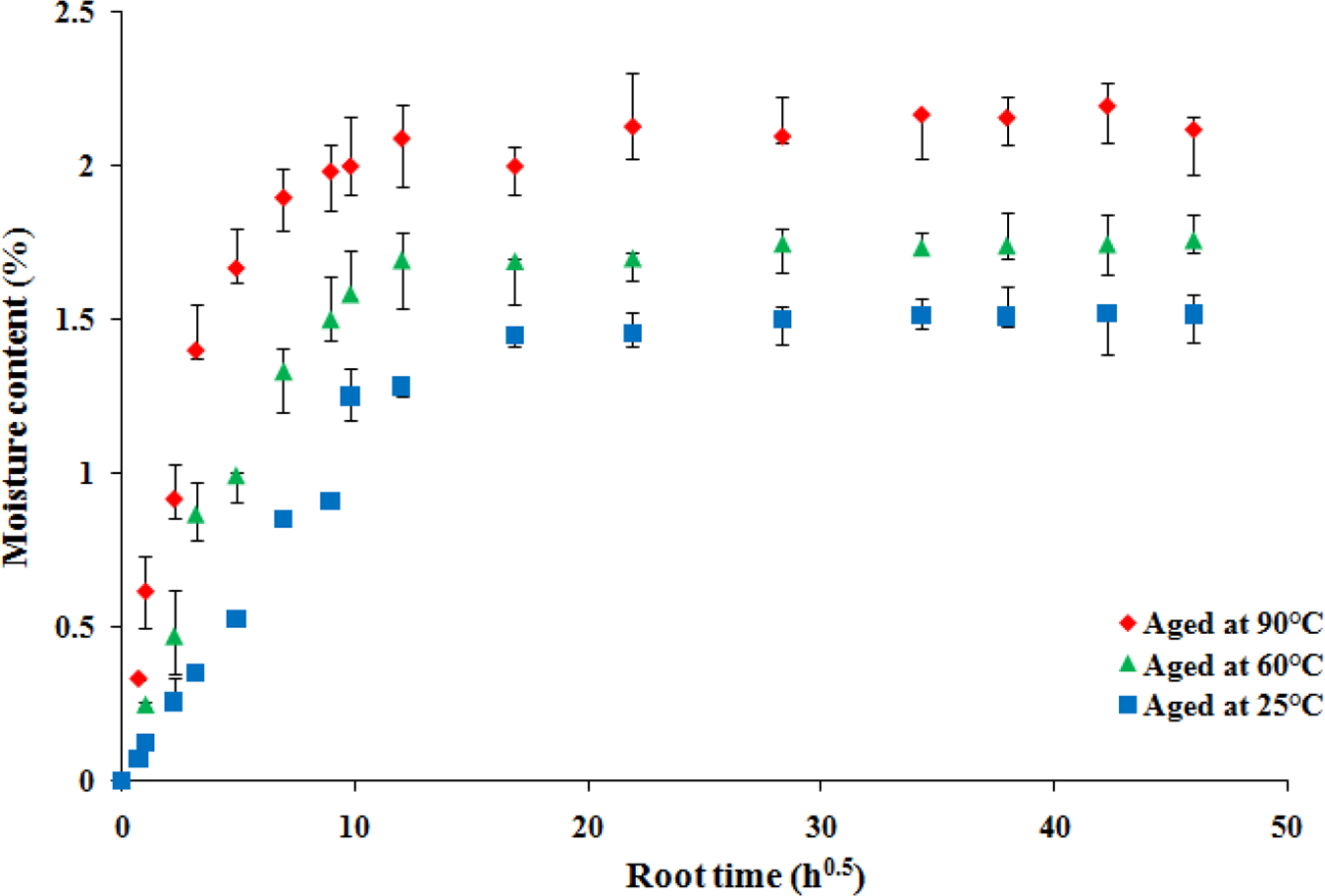
Moisture content (%) as function of immersion root time (
Afterward, in the second stage, the equilibrium water uptake seems to be reached and consequently, the sample is saturated. It is seen that the higher the immersion temperature, the greater the mass gain. According to Ref., 25 the temperature favors the movement of the polymer chains which facilitates the penetration of the water. Thereby, the temperature of the absorption process has sway on the water absorption curves.
Table 1 regroups the obtained data of diffusivity (
Characteristics extracted from absorption curves for PVC pipe (8 mm).
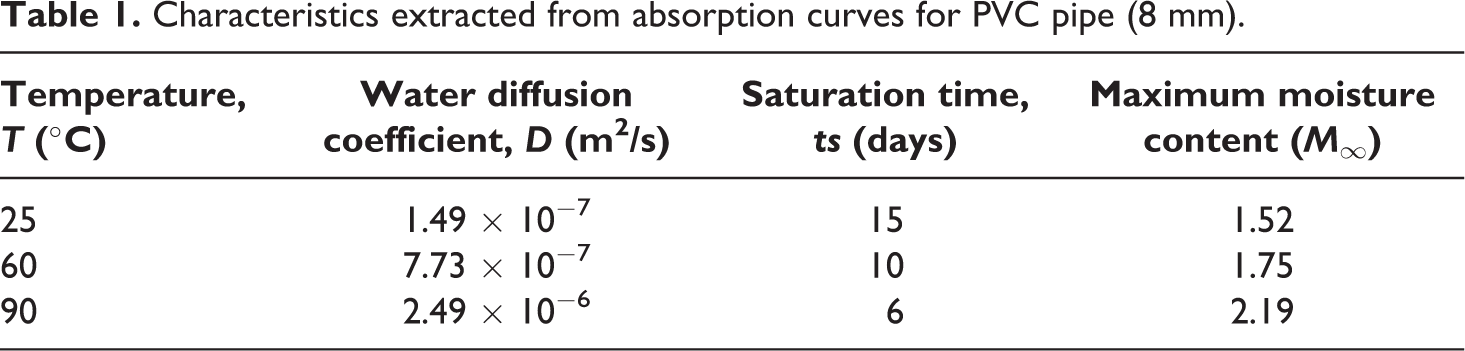
As has been pointed from Table 1, the maximum moisture content increased considerably when hydrothermal aging temperatures increased. This tendency agrees with the result obtained by Bao when studying the hydrothermal aging behavior of CMR/PLA biocomposites at different temperatures. 20 In addition, the diffusion coefficient increases significantly with the immersion temperature and reaches a remarkable value at 90°C. This confirms that the diffusion is a thermally activated process and the diffusivity is very sensitive to temperature. 26 The increase of aging temperature improves segmental mobility which induces a greater zone of activation, consequently, the rate of water absorption raises.
In other words, the diffusion mechanism in polymer materials is shown to be highly dependent on the conditioning temperature.27–29 Indeed, the temperature plays a role of activator of the absorption mechanism obeying the law of Arrhenius whose activation energy “

where
Figure 3 shows a plot of ln
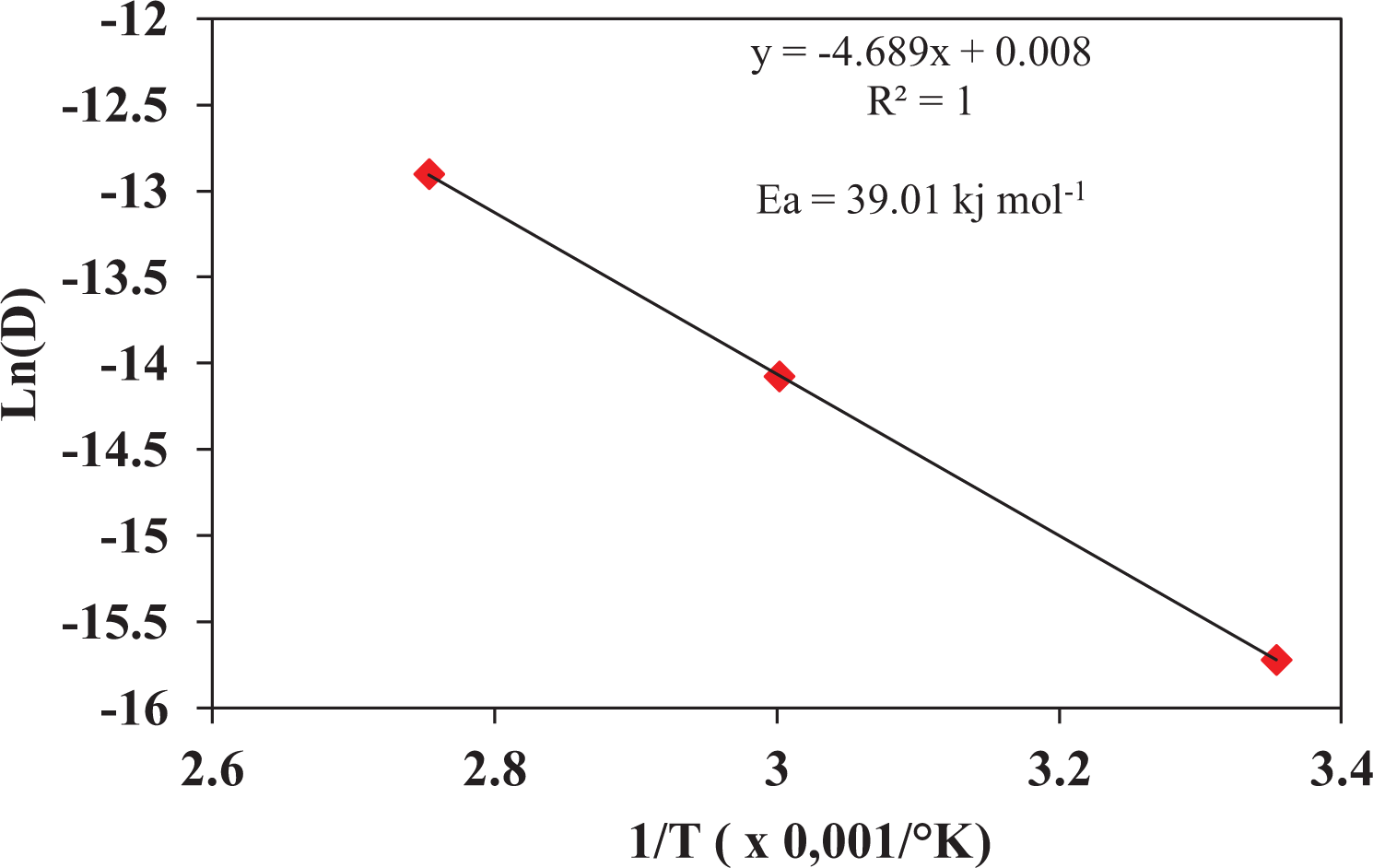
Arrhenius plot for PVC samples aged in distilled water at three temperatures (25, 60 and 90°C).
Thermogravimetric analysis of PVC pipes
Thermogravimetric analysis (TGA) was conducted to assess the thermal stability of the samples at different temperatures. Dynamic thermogravimetric and the derivative thermogravimetric (DTG) curves are shown in Figures 4 and 5.
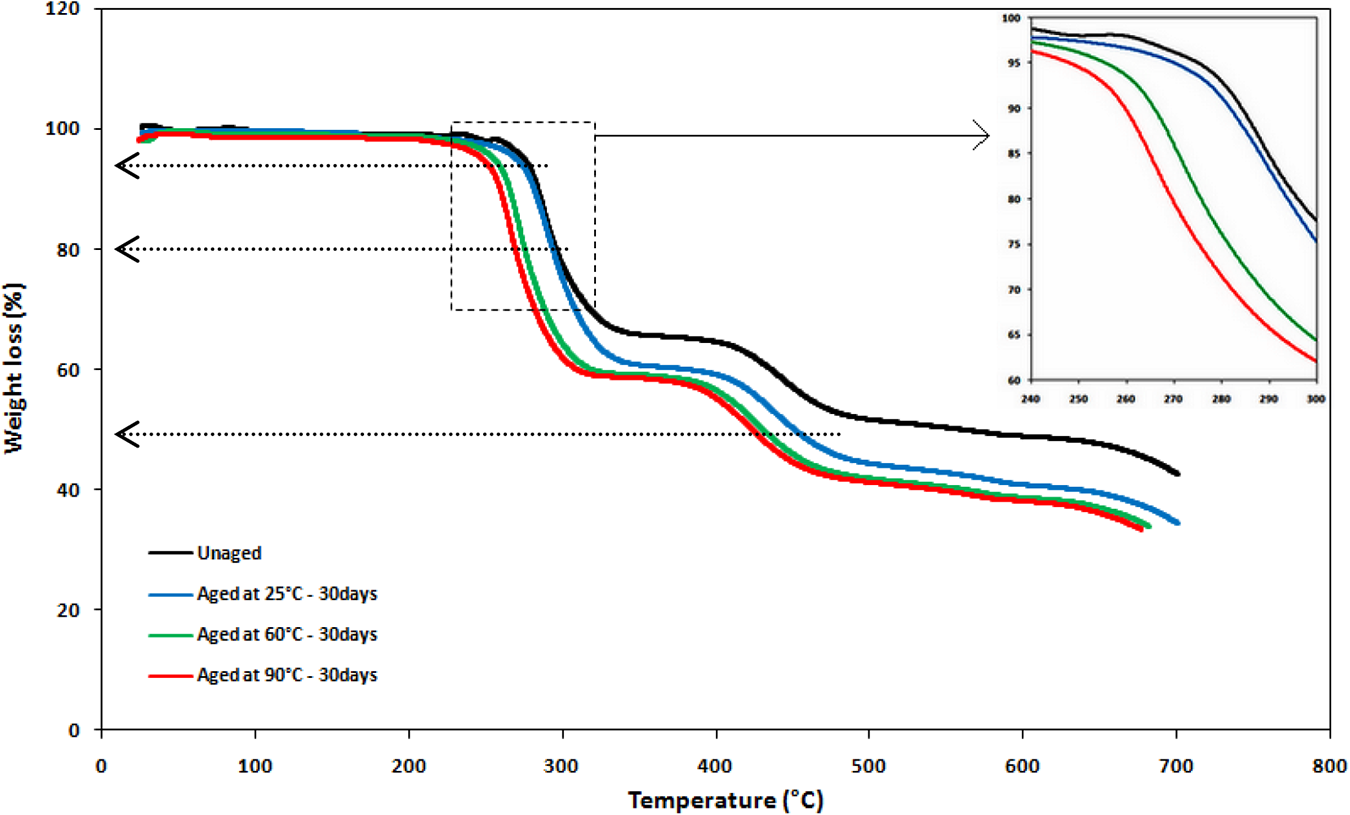
TGA curves of unaged and aged PVC samples after 30 days of immersion in distilled water.
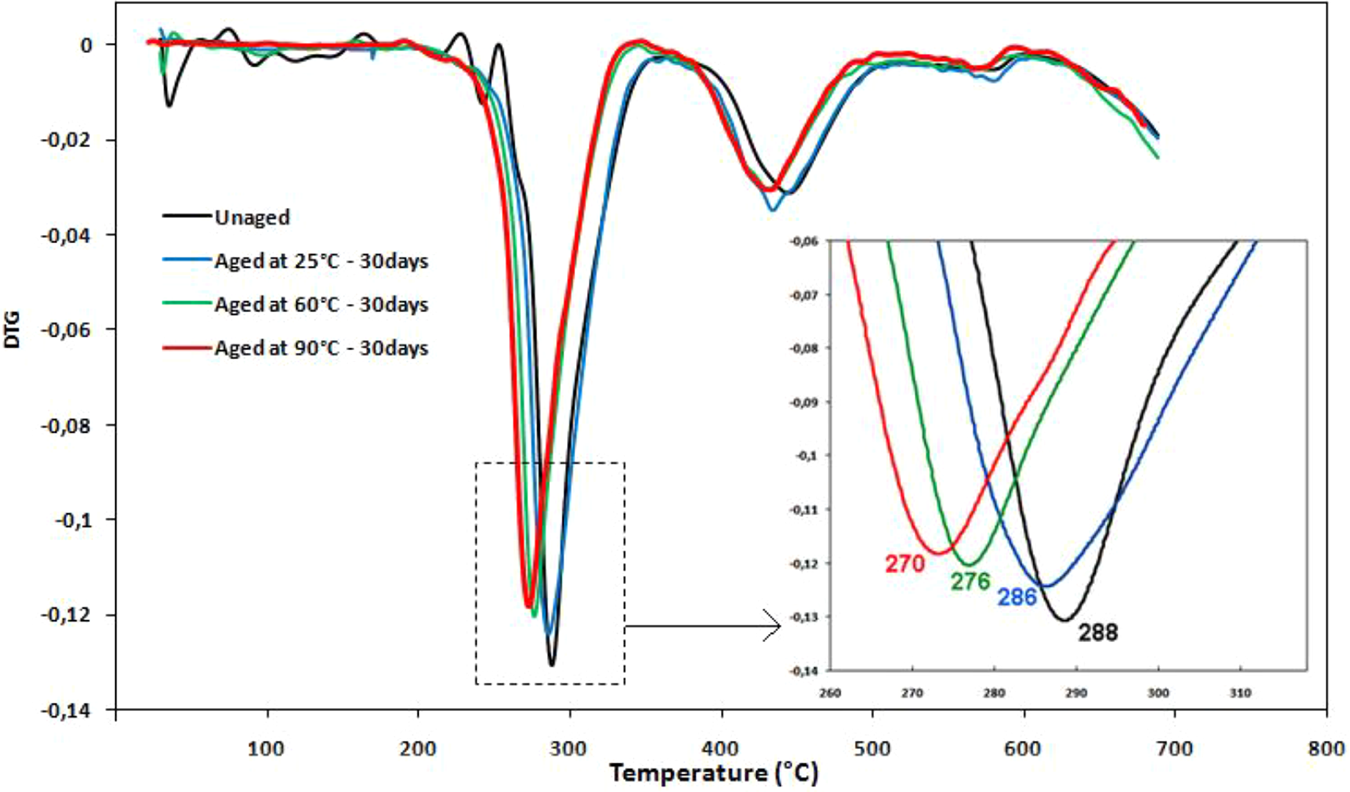
DTG curves of unaged and aged PVC samples after 30 days of immersion in distilled water.
Over the analyzed samples, until 250°C, all curves remain constant and there is no variation in weight, this means the samples are thermally stable in this range of temperature.
However, for the unaged PVC sample, which is considered as reference, it can be observed that there are two weight loss stages. In the first stage, the decomposition starts at about 253°C and ends around 330°C, as summed up in Table 2. From 400 to 500°C, a second decomposition stage is observed, much shorter than the first one, and corresponding to polyacetylene cracking. 32 Finally, a stable residue can be formed when the temperature is beyond 550°C.
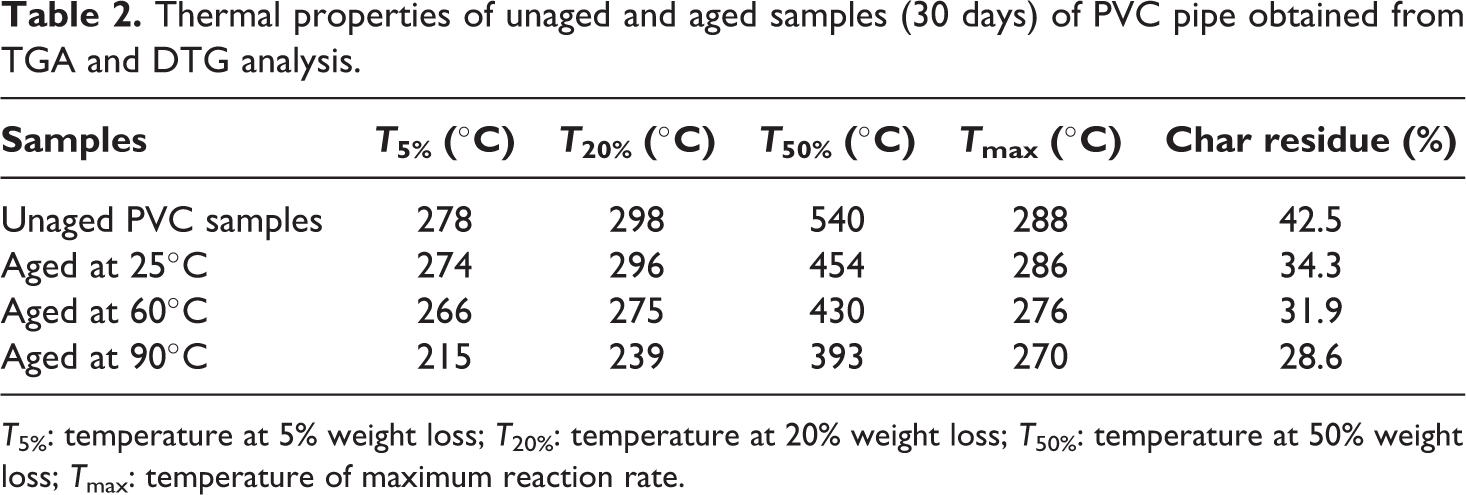
Thermal properties of unaged and aged samples (30 days) of PVC pipe obtained from TGA and DTG analysis.
It is to note that the DTG curves show a peak in the range of 253–330°C, which corresponds to the “temperature of maximum rate degradation.” Such peak arises at a bass temperature for aged samples. This can be explained by the effect of water on the polymer structure. The later seems to be softer with the absorption of water, which directly affects the thermal stability.
The decrease in the thermal stability of the PVC after long-term water immersion was attributed to the network structure change, driven by plasticization which allows migration of the stabilizers. This finding is conforming well to those found in Ref. 33
Also, it can be observed from Figure 4, even at high temperature, 700°C, a percentage of material samples remains intact, around 40%. It is probably an inorganic material in the PVC pipes.
This suggests an inorganic filler used in the PVC matrix. According to the literature, 34 such residual mass can correspond to the carbonate of calcium (CaCO3). Therefore, it will be important to confirm this point in the following section.
The results referred to in Table 2 show a lowering in the thermal parameters (
Thermal stability and HCl release reduction in PVC formulation is improved by using fillers (CaCO3). 36 From Table 2, we note that the char rate decreases as exposure temperature increased indicating the decrease of the amount of filler in PVC after migration caused by the mobility of macromolecules. This explains the lowering of the stability of the polymeric samples.
Infrared spectroscopy characterization of PVC pipes
Figure 6 represents the FTIR spectrum for the unaged PVC sample. The absorption bands observed at 610, 635, and 690 cm−1 are attributed to the stretching vibration modes of the (C–Cl) bond. The peaks at 720 and 729 cm−1 reveal that all the previous structures belong to long-chain hydrocarbon of four or more methylene units. These vibrations have been identified by others to belong to the long aliphatic chains existing in the lead-based stabilizer as reported in Refs.37–39
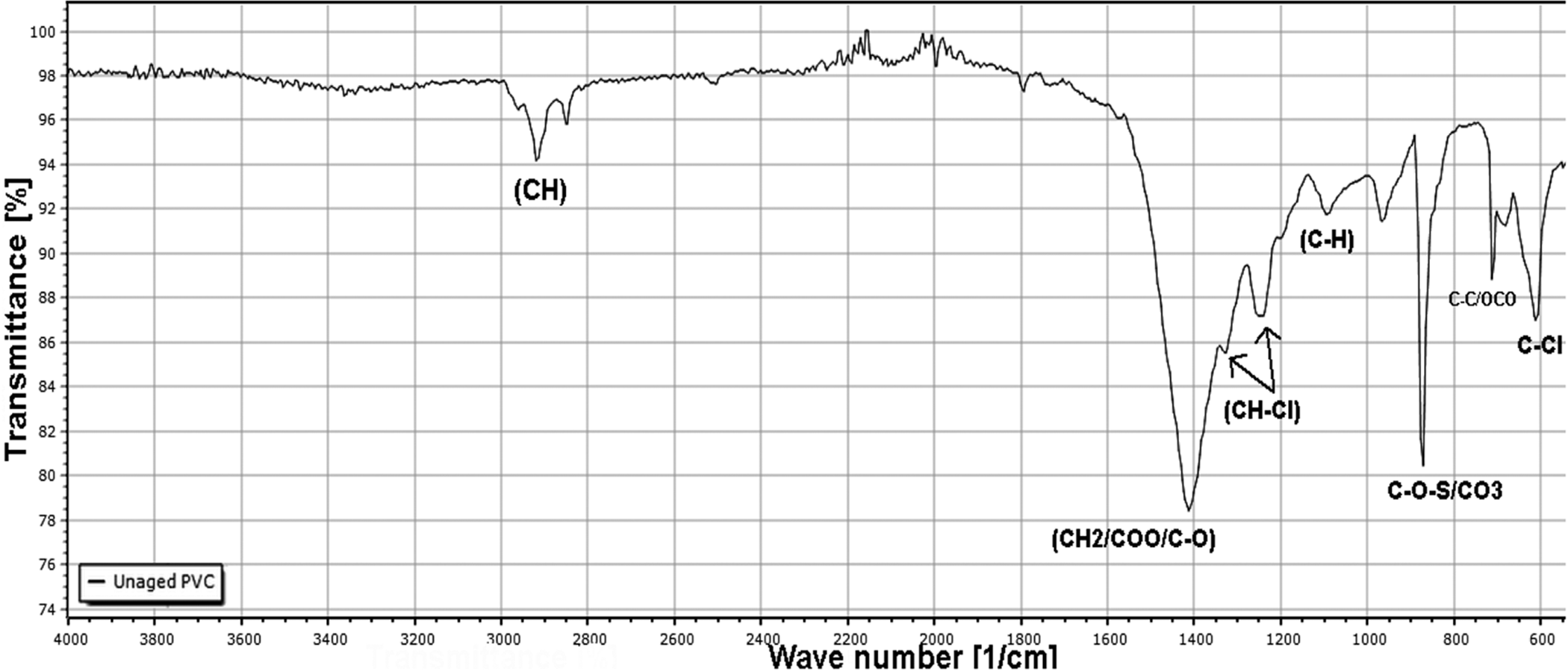
FTIR spectra of unaged PVC samples.
The vibrations at (847, 874 cm−1) are those of the bonds of the (C–O–S) structure.40,41 In the same region, there is the CO3 (875 cm−1) belonging to calcium carbonate (CaCO3).42,43 This funding confirms that the material of the PVC tube is not pure. It consists of a PVC matrix containing CaCO3 used as fillers.
An intense peak at 1425 cm−1 assigned to (CH2, COO) of the stabilizer system, and (C–O) of calcium carbonate (CaCO3). Similarly, this result indicates that the material of the PVC pipe also contains a lead-based stabilizer system. Finally, the absorption bands at (2850–2918 and 2874–2956 cm−1) belong to symmetric stretching vibration and anti-symmetric stretching vibration of the (C–H) bond. Such observed peak was already described in a similar study. 44
The spectrum also shows the typical doublet of the phthalates at 1580 cm−1. The bands corresponding to the carbonyl and ester group, as well as those corresponding to the aromatic and aliphatic C–H bonds can also be observed and Table 3 shows these bands, according to Refs.45,46
Characteristic bands for the used plasticizer.

The spectrum of PVC before and after aging was compared. This allowed the identification of some characteristic bands which are related to the migration of plasticizer present in the formulation. The intensity of characteristic bands will change. An increase in the intensity corresponds to the absorption of liquid through the PVC samples and inversely, a decrease in intensity corresponds to the migration of one or more components in the water. 47
There is a decrease in absorbance after aging. This indicates that the migration of plasticizer occurred at both temperatures aging. In the case of high temperature, it seems that the penetration of the water which was observed in the rate of mass variation favored the migration of additives. Indeed, the lowest absorbance were obtained for the formulation aged at 90°C; therefore, the highest rates of migration.
The superposition of the spectra of unaged and aged samples in Figure 7 shows that all the bands present in the unaged one are also recorded in the aged specimen but with some difference in intensity of absorbance.
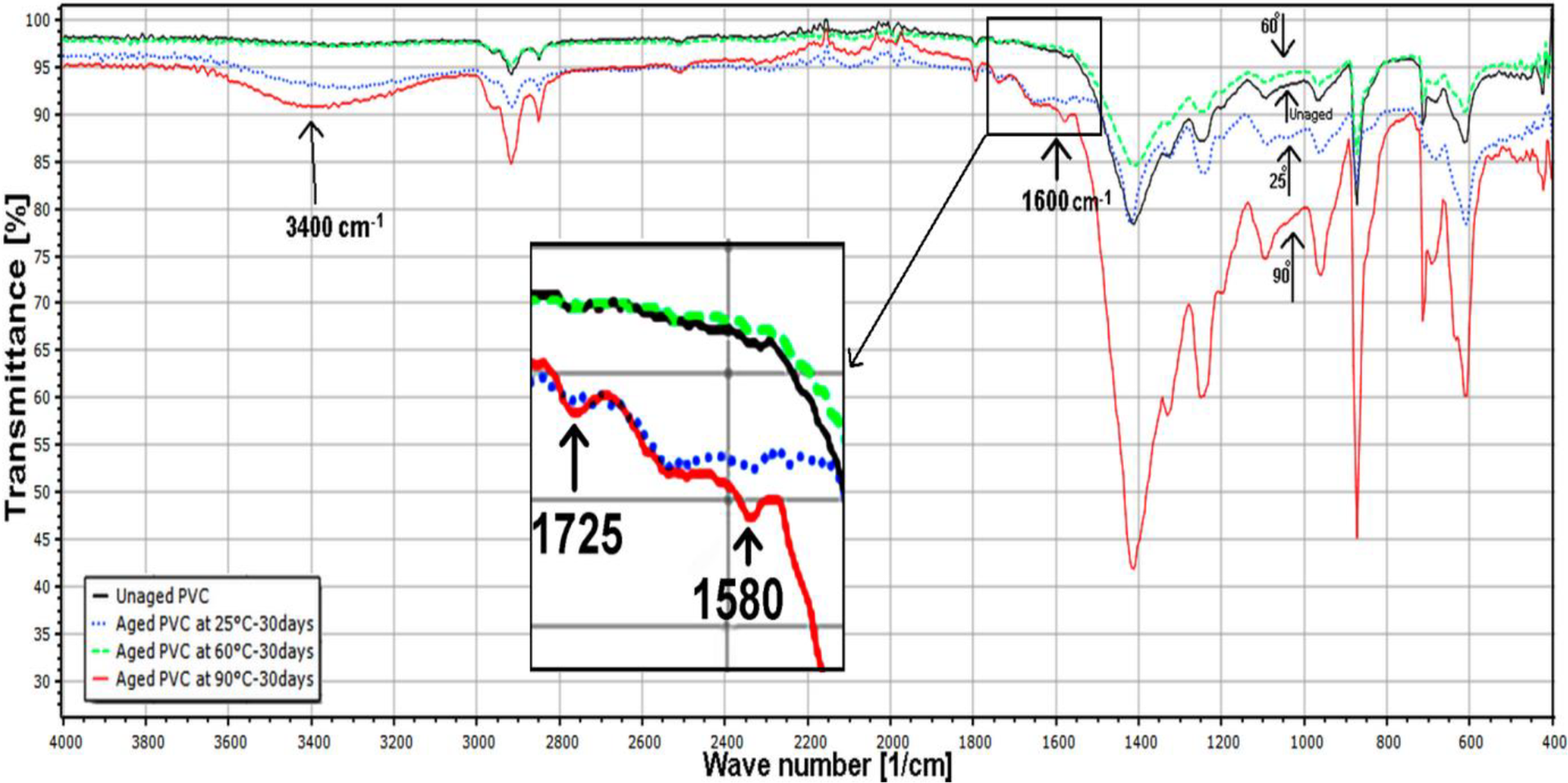
FTIR spectra of unaged and aged PVC samples.
It was noticed, from Figures 6 and 7, that the intensity of the (C–Cl) peaks at (635 and 690 cm−1) and those of (CH, CH2) at (955, 1197, 1253, 1329, 1425 cm−1) decrease with increasing the immersion temperature (from 25 to 60°C). Further, Figure 7 indicates that the peak intensity between 1600–1680 cm−1 increases when the aging temperature grows. This region corresponds to the stretching vibration of the C=C conjugated group and the C=C in conjugated group stretching vibration. 48 This can be explained by a dehydrochlorination process. 44
The increase in peak intensity (875, 965, 1101, and 2853 cm−1) for the sample aged at 90°C can be explained by the fact that the additives (plasticizer and stabilizer) have migrated from the core of the samples to the surface. The same result was found by Jakubowicz et al., 49 when studying the effects of accelerated and natural aging on plasticized polyvinyl chloride (PVC). They have confirmed the mass loss of PVC samples by a dominant loss of plasticizer.
The spectrum of the aged PVC specimen at 25°C and 90°C present specific characteristics of absorbed water put in evidence by the picks at 3400 and 1600 cm−1. That is obvious by the presence of the stretching vibrations and the bending modes of the water molecules as reported by Hammiche and their collaborators. 25 These results correlate well with the water uptake ones as described in the absorption section.
Mechanism of yellowing and shape change
In this context, the identification of the mechanism of PVC samples yellowing and shape change was studied after 60 days of immersion at different temperatures. This investigation displays that there are obvious signs of degradation of PVC samples on view to temperature and the time of immersion, including the color and shape changes as illustrated in both Figures 8 and 9, respectively.

Discoloration of aged PVC after immersion in distilled water for 30 days at different temperatures.

Shape change of aged PVC after immersion in distilled water for 30 days at different temperatures.
As it is clear from Figure 8, at room temperature (25°C) and after medium immersion periods, PVC samples do not change its color. However, when exposed to high temperatures (90°C) in full immersion PVC samples change its color. So, extended exposure can cause yellowing too. Based on the literature, 50 such discoloration can result from the interaction of additives or compounds in the polymer formulation and their accumulation on the material surface. It equally reveals a degradation process (breaking and rearranging of chemical bonds). Beside, such discoloration endures after the air drying. This result was in good agreement with the finding of Krauklis and Echtermeyer 51 in the case of the hydrothermal aging of epoxy. These authors have observed a change in the color of aged versus unaged epoxy and have confirmed that the turn in color was irreversible. Similarly, Simar et al. 22 have highlighted polymer color changes during hydrothermal aging (yellow, brown, and dark) and they have explained this by the network chemical modifications provoked by oxidation or hydrolysis phenomena.
Additionally, there is another sign of degradation: it consists of a change of the shape of the aged sample, especially at the higher temperature (see in Figure 9). This underlies the mechanism of shape change in PVC pipes; thus, our studied material can be considered a shape-changing polymer under hydrothermal conditions. Here, one can conclude that, depending on the conditions and extent of hydrothermal exposure, the observed aging effects affecting the color and the shape of aged samples are considered as irreversible changes that provoke permanent property alterations within the thermoplastic. These findings are in good accordance with the dramatic symptoms of degradation (deformation and discoloration) previously reported by Guermazi et al., 2 in the case of PVC/CaCO3 composites exposed to hydrothermal aging. Also, the severity of the exposing conditions is found to increase with the energy when increasing the aging temperature, as reported by Grammatikos et al. 52
Mechanical properties of PVC pipes
To confirm the results of the previous sections, the effect of the aging temperature on mechanical behavior was investigated by tensile tests.
Superposed stress-strain curves of unaged samples and aged ones at 25, 60, and 90°C are subjected in Figure 10. All the curves present three steps, the first step of the deformation corresponds to the elastic linear deformation. After the limit point of elasticity, the stress goes through a maximum corresponding to ultimate stress “σu” followed by a plastic deformation until rupture.53,54
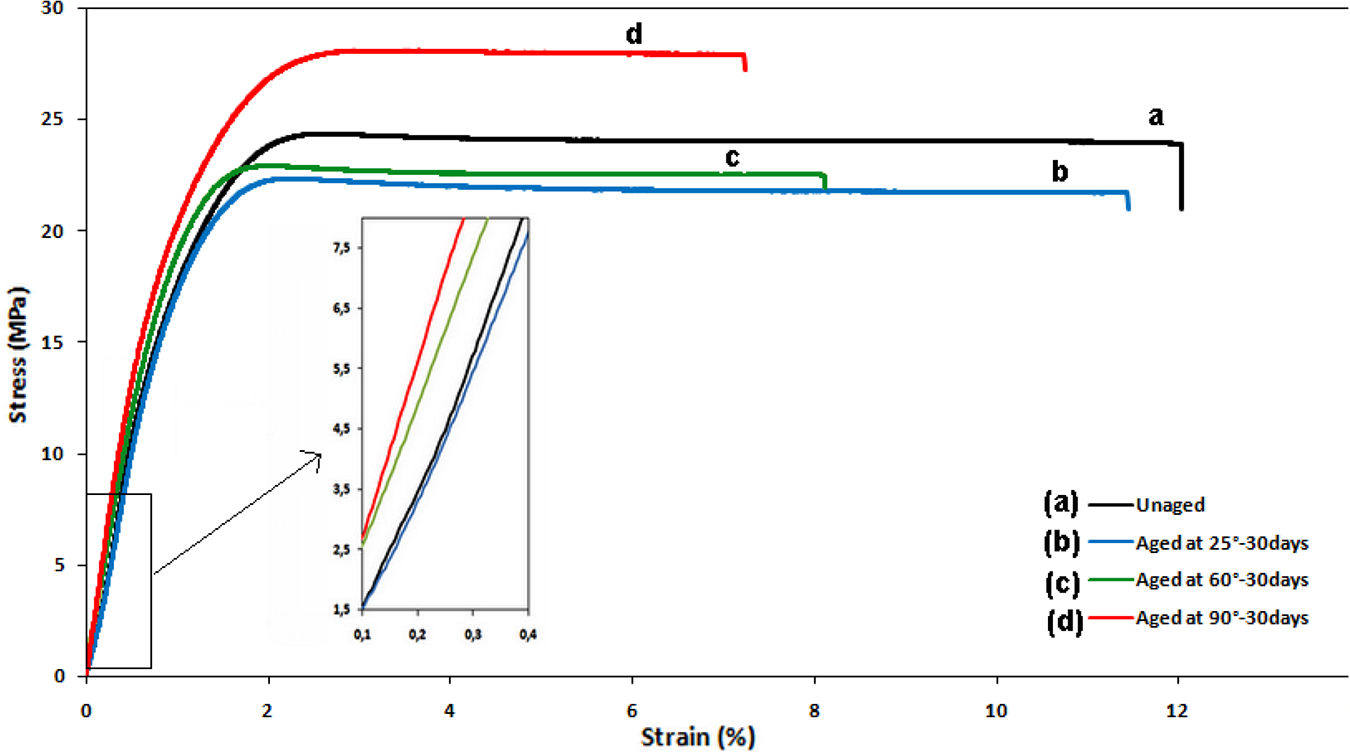
Stress-strain curves of unaged and aged samples of PVC.
The aging temperature has an impact on mechanical properties. The slope of the elastic part of the curve increases remarkably from 25°C to 90°C, showing a stiffening arise of the material. The ultimate strength also increased with the immersion temperature and reaches the maximum value at 90°C.
The experimental tensile properties, extracted from Figure 10, for PVC samples are given in Table 4 with standard deviations. Here, it is to note that the average values of each characteristic are based on the results of three tensile samples at least.
Tensile properties of unaged and aged samples (30 days) of PVC pipe.

E: Young’s modulus; σu: ultimate strength; εb: strain at break; σb: stress at break.
To better show the impact of the aging temperature on mechanical behavior, the variation of E, σu, εb, and σb are presented in Figure 11(a) and (b).
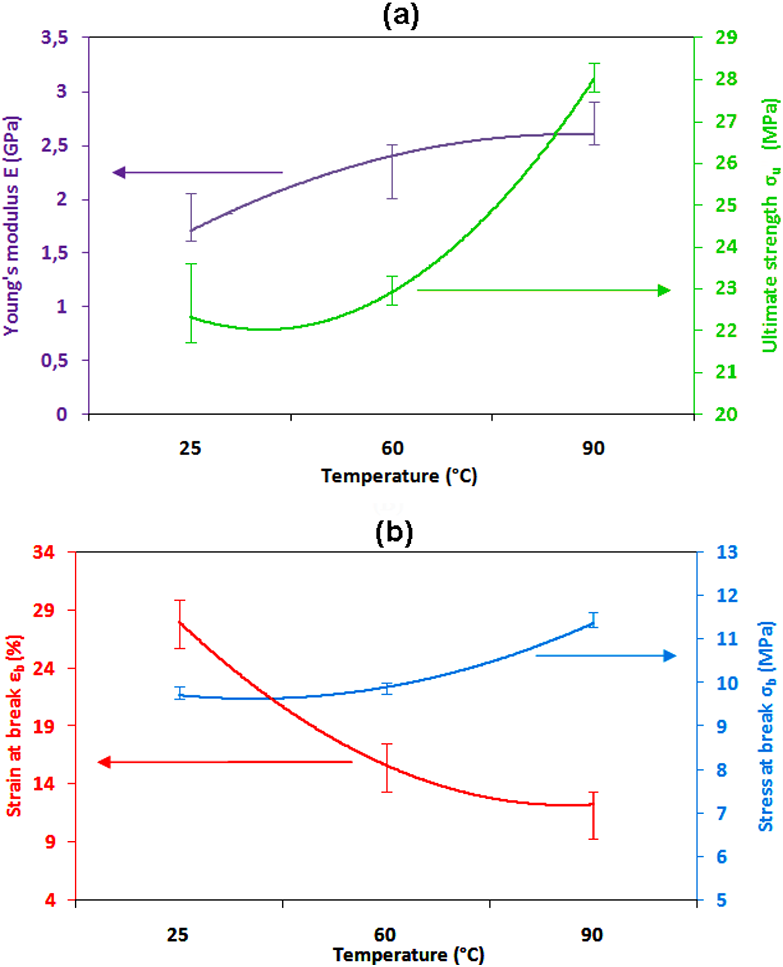
Tensile properties: (a) E, σu and (b) εb, σb of PVC versus aging temperature.
It can be observed that E increased with aging temperature. The increase was clear from 25°C to 90°C with a rate of 34.61%.The σu evolved similarly to E with the increasing aging temperature. From 25°C to 60°C the increasing rate of σu was 2.62%, while from 60°C to 90°C it was 18.21%. The increasing of E and σu reveals the change of the elastic character of the material, which tends to be more stiffen. In the same way, we have studied the evolution of εb and σb as a function of the aging temperature for 30 days. From Figure 11(b), it was seen that the stress at break σb tends to increase with the aging temperature. These results correlate well those found by Gumargalieva et al., 55 in their investigation about plasticized poly(vinyl chloride) samples aged in natural and artificial conditions. The latter have found that the stress at break σb and the strain at break εb, increases from 25 to 30 MPa for 24 months and from 60 to 25%, respectively.
However, Figure 11(b) displays a remarkable decrease of εb with the temperature of aging. In fact, from 25°C to 90°C, the reduction rate is about 36.84%. This can be explained by the fact that the material became more brittle. This result is consistent with that reported by Simar et al., 22 the latter has noted that the resin behavior becomes brittle due to hydrothermal aging (based on the significant reduction of the failure strain).The same behavior was observed by Merah et al., 53 when studying the physical and chemical properties of the powders and films of PVC. Indeed, the later have found that there is a significant effect of the temperature on the fracture behavior.
To more explain the variation of εb, some photographs of the samples before, during, and after tensile tests have been performed, as seen in Figure 12. The failure of the tested samples takes place with brittle rupture. Indeed, there is no significant reduction in the loaded section (thickness × width). This can be explained by the loss of additives as previously noted in the FTIR section. Perhaps, these compounds or additives migrate, during the immersion period, from the core of material to its surface, as described in the previous section (discoloration and shape change). These results correlate well with several studies that have investigated the effect of the rate of plasticizers on the mechanical properties of PVC.56–58 In particular, Ito and Nagai, 57 have shown that the stepwise flow out of inorganic components and plasticizer can be considered the main mechanism of the degradation of plasticized PVC under weathering conditions. Accordingly, we can consider that the loss of inorganic components and plasticizer leads to reduce the elongation at break, whereas it enhances the rigidity and the resistance of aged PVC samples. Finally, although the aging temperature, in this study, significantly affects the failure strain of PVC material and tends to improve its strength and stiffness, longer exposure durations can also significantly affect material behavior than brief exposure. The question of whether obtained tendencies of mechanical properties changes will remain the same even in the long-term is then crucial. This point will be developed in future work.

Illustration of the decrease of the strain at break εb during 30 days.
Conclusions
The work presented in this manuscript is based on the assessment of the physico-chemical, thermal, and mechanical characteristics of industrial PVC pipes before and after hydrothermal aging. This characterization was evaluated by several techniques involving water absorption, TGA, FTIR, and mechanical measurements. Based on the obtained results, during the retained conditions, the following conclusions can be drawn: The pipe material has been identified, it is based essentially on thermoplastic matrix (PVC) reinforced with CaCO3 fillers, and mixed with a commercial plasticizer (DOP). Fickian behavior was observed for PVC samples immersed in water at 25°C, 60°C and 90°C. Both of maximum water absorption and the rate of water absorption increased dramatically with increasing temperature. Both of TG-DTG and FTIR analysis show preliminary signs of degradation of PVC samples under the retained conditions. The adopted hydrothermal aging induces discoloration and shape changes of the aged samples. Higher strength and stiffness were obtained for the aged samples, particularly at elevated aging temperatures. However, the aged material losses its ductility, compared to the reference one.
In future work, it will be interesting to conduct a more detailed mechanical and tribological characterization of PVC pipes before and after long exposure to hydrothermal aging.
Footnotes
Acknowledgment
Many thanks are owed to the Engineer Mr Jamel Lachkhem, the Head of Tuyauplast company (Gafsa, Tunisia) for his technical support in this work.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
