Abstract
Poly(methyl methacrylate) (PMMA)/nanobarium titanate (NBT) composite has potential application in denture base materials. The denture base materials should be stable in the wet environment and exhibit good mechanical properties. This study aimed to evaluate the effectiveness of titanate coupling agent (TCA) on NBT behavior after soaking in the simulated body fluid (SBF) and to determine the effect of SBF exposure on fracture toughness of the PMMA nanocomposites. Silanated (Si-NBT), titanated (Ti-NBT), and pure NBT (Un-NBT) at 5% concentration (by mass) were incorporated in the PMMA matrix. NBT was sonicated in MMA prior to mixing with PMMA. SBF absorption, solubility, and leaching were measured, and fracture toughness of the PMMA nanocomposites was evaluated after soaking. The results showed that Titanated samples displayed lower SBF absorption capability and solubility values than the silanated ones. Moreover, the leachability of filler elements (Ba and Ti) was substantially reduced by titanation (54% and 61%, respectively), whereas the Si-NBT/PMMA revealed values of 12.3% and 7% respectively. Significant differences in fracture toughness were observed among the tested samples after 6 weeks of aging in SBF (
Introduction
Denture bases have been conventionally fabricated utilizing poly(methyl methacrylate) (PMMA) because of its favorable characteristics, ease of processing and repair, low cost, superior appearance, and excellent biocompatibility. However, PMMA is typically weak, moderately flexible, brittle on impact, and only fairly resistant to fatigue failure. In the oral aqueous environment, PMMA absorbs small amounts of water. This water significantly affects the mechanical and dimensional properties of the cured polymer. 1 PMMA dentures can gain weight from water uptake or lose weight from dissolution in the water when exposed to or stored in water. 2
The development of hydrolytically stable dental resin composites is an important goal in dental materials research. The interface stability in the oral environment and the capability to transfer stresses between the matrix and filler phases during mastication is especially significant properties for dental composites. Geerts et al. 3 reported that the degree of adhesion between filler and polymer affects the degree of reinforcement. Although silanation is commonly employed to promote adhesion of the dental resin matrix to the filler, the composites and their hydrolytic stability remain a concern.4–6 Furthermore, silane coupling agents form aggregates on the filler surface because of incomplete coating, thereby leading to unstable bonding between fillers and resin.7,8 The oral cavity is a harsh environment because of its wetness, temperature stresses from drinking and eating, and mechanical stress caused by dental occlusion. Therefore, enhancements are more apparent when dental composites are exposed to high humidity. However, exposing dental composites to an aqueous environment degrades their mechanical properties because of the failure of the polymer–filler bond. 4 and this failure could facilitate the release of the filler components. Signs of leaching from different silanated fillers has been reported,9–12 thereby negatively affecting the composites’ longevity and leading to health concerns. 9 This deterioration results in stress and cracks growth that results in a rapid increase in the amount of water absorbed by the composite material due to silane hydrolysis and degradation. Söderholm et al. 12 found that dental composites stored in distilled water leach filler components. Glass-modifying elements, such as Na, Ba, and Sr, increase the leaching rate which could cause health risks. An improved silane bond was fabricated via hydrosilylation after silanation, 13 and applying an acetone primer to the glass prior to silanation has been reported 14 ; however, such procedures do not solve the problem of slow silane degradation in oral fluids.
Titanate coupling agents (TCAs) could be favorable alternative coupling agents to silanes. TCAs are known to chemically bind inorganic filler and organic polymer via proton coordination on nonsilane reactive substrates without water for condensation and less void formation around the particles.15,16 TCA can react with oxides or hydroxyl groups on the surface of stainless steel to generate C–O or P–O bonds, to build “molecular bridges” between the adhesive and steel to enhance the adhesion strength.
17
Tham et al.
18
investigated the effect of TCAs on the mechanical and morphological properties of PMMA denture base composites and reported that TCAs could strengthen interfacial bonding, improve mechanical properties, enhance filler homogeneous dispersibility, and modify the rheological behavior of composites. Another work evaluated the hydrolytic stability of TCA and found that TCA-treated composite samples exhibited higher resistance to moisture ingress than the untreated composites.
19
In addition, the behavior of TCA as an alternative coupling agent to silane was studied in the dry environment.
6
The results showed that the fracture toughness of the titanated samples was higher than that of the untreated and silanated ones because of the improved compatibility and interfacial bonding between nanofiller and PMMA. The accurate selection of a coupling agent is critical to obtain high-performance composites for dental applications. Simulated body fluid (SBF) test, a well-recognized method to characterize
The introduction of nanomaterials offers a new promise for augmenting the mechanical properties of dental composites because of the high surface-area-to-volume ratio of these materials enhances their interfacial interaction with the resin matrix. The properties of polymer nanocomposites depend on the type of incorporated nanoparticles, their size, shape, and distribution, as well as the concentration and interaction with the polymer matrix. 21 When inorganic nanoparticles function as fillers in polymer materials, the superior characteristics of both polymer materials and nanomaterials integrate to enhance rigidity, hardness, fracture toughness, and other functional properties of the composites. Nanobarium titanate (NBT), as a class of ceramic systems, displays interesting features for biological applications because of its favorable mechanical properties. 22 Furthermore, barium titanate fillers have been incorporated as a radiopacifier in PMMA matrix to enhance its radiopacity characteristics because of the filler components’ high atomic number. Accordingly, this material can be detected by radiographic means; any delay in localizing or removing the foreign body may be life-threatening.23–25
In the current study, NBT is introduced as a new dental filler to enhance the properties of denture bases. The biocompatibility of TCAs and NBT has been reported26,27; however, the applications of such materials in dentistry are still limited. This topic, particularly in relation to denture base composites, has yet to be included in the dental literature. Therefore, this study was performed to evaluate the efficiency of titanated NBT compared with untreated and silanated NBT-filled PMMA after 6-week storage in SBF. The effect of titanated and silanated NBT on filler leachability, absorption, solubility, and fracture toughness was investigated after SBF exposure.
Materials and methods
The solid components consisted of PMMA (molecular weight: 996,000 as determined by gel permeation chromatography; GPC) and 0.5% benzoyl peroxide (BPO). The liquid component comprised MMA stabilized with 0.0025% hydroquinone plus the cross-linking agent 10% ethylene glycol dimethacrylate (EGDMA). Powder-form NBT constituted the filler. These materials were purchased from Sigma-Aldrich (USA).
NBT was divided into three groups. The first group denoted as Un-NBT was incorporated as an untreated filler. The second group referred to as Si-NBT was treated with the silane coupling agent 3-(trimethoxysilyl) propyl methacrylate (γ-MPS) (Figure 1a), which was also supplied by Sigma-Aldrich (USA). The last group denoted as Ti-NBT was treated with the TCA, KR 12, or isopropyl tri[di(octyl) phosphato] titanate (Figure 1b). This coupler was provided by Kenrich Petrochemicals (USA). SBF was supplied by B-Braun Medical Industries (Malaysia).
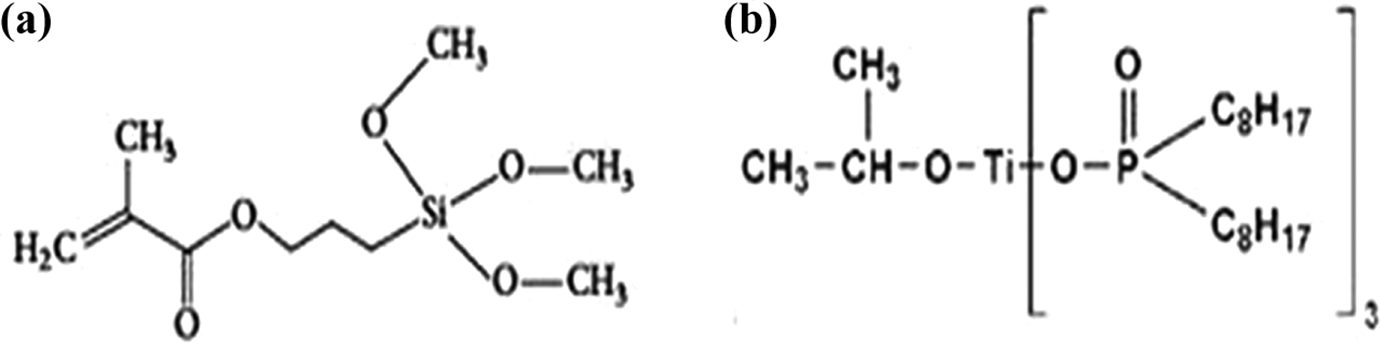
Chemical structure of coupling agents; (a) γ-MPS, (b) KR12.
Surface modification of NBT
A 70/30 solution of acetone/water was prepared in a glass beaker. A total of 5 g NBT was dispersed in the solution under stirring. Then, 10% (by mass) γ-MPS relative to the filler was added dropwise using a sterile syringe under rapid stirring. The beaker was covered by a plastic paraffin film and mixed for approximately 15 min under magnetic stirring. The slurry was sonicated for 20 min to disrupt agglomeration. The ultrasonication was conducted using an ultrasonic bath (Wisd, WUC-A03H, Korea). The resulting solution exhibited homogeneous white-color. The mixture was further stirred at 80°C until acetone completely evaporated to allow coupling reaction. Finally, the modified NBT was dried for 24 h in an oven at 120°C.
For the TCA, 5 g NBT was added to 100 mL of acetone followed by KR 12 under stirring for 15 min. The mixture was sonicated for 20 min at room temperature for deagglomeration. The slurry was stirred for 30 min, and then the temperature of the mixture was increased up to 80°C to remove the solvent. Finally, the modified NBT was dried for 24 h in an oven at 120°C. Sample preparation was performed as detailed elsewhere. 6
Field emission scanning electron microscopy (FESEM)
The morphology of the powder NBT was examined using FESEM (Zeiss, Supra 55VP, Germany) to determine the distribution of the particle size of the filler. Samples were prepared through dispersion, and one drop was pipetted onto a carbon support film on a 3 mm copper grid. The acceleration voltage was 5 kV. The image files were processed using more than 500 particles. FESEM was also used to characterize the fracture surface of NBT/PMMA nanocomposites. All surfaces were gold-coated to enhance image resolution and avoid electrostatic charging and to obtain better image resolution.
SBF absorption
This test was conducted according to ISO 1567. Six specimens were prepared for each material. The specimens were dried in a preconditioning oven at 37 ± 1°C for 24 h and kept in a desiccator containing silica gel for 24 h. They were weighed to an accuracy of ±0.00001 g utilizing an analytical balance (

where

where
Chemical composition of the simulated body fluid (SBF) as provided by the supplier.

SBF solubility
After soaking the samples in SBF for 6 weeks, they were placed in a drying oven at 37 ± 1°C to evaporate the absorbed moisture and kept in a desiccator for 24 h. A similar process to that of absorption repeated during desorption until there was no significant change in mass (
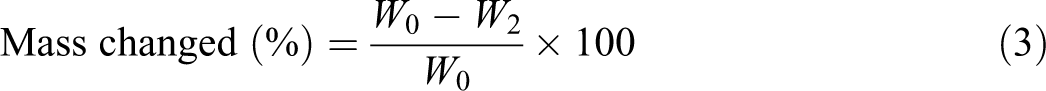
where W0 and W2 are the mass of the sample before and after immersion, respectively.
Leaching experiment
Each specimen was placed in a disposable vial containing 10 mL SBF. After 6-week soaking, droplets were picked up from the storage liquid and placed in an oven at 37°C for 24 h. The samples were then removed and evaluated through energy-dispersive X-ray spectroscopy to confirm the leaching of the filler’s elements into the storage medium during the immersion period after treatment with different coupling agents.
Fracture toughness (KIC)
The
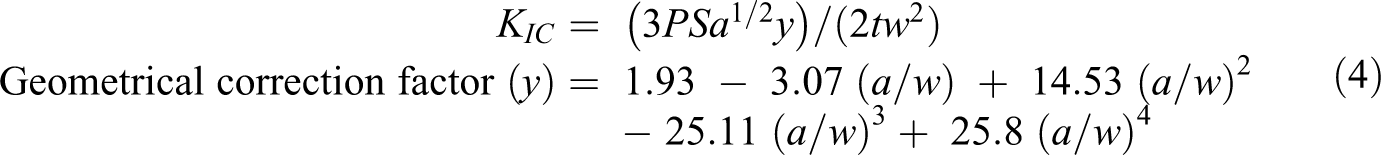
where

Equipment used fracture toughness test; (a) testing machine; (b) mold used in samples preparation.
Statistical analysis
Data of KIC were statistically analyzed using SPSS software (version 22, Statistical Package for Social Science, SPSS Inc., Chicago, IL, USA). To find the performance difference in each kind of tested materials, one-way analysis of variance (ANOVA) was performed to detect the significant effects of variables. Tukey’s multiple comparison test was used to compare the data at with significance of
Results and discussion
Dispersed phase characteristic
With the development of polymeric dental composites, several works have focused on the optimization of filler types, compositions, and loading. This optimization has led to the enhancement of the physical–mechanical properties of fillers. 16 Particle size is an important factor influencing the properties of the resulting composite. Small filler sizes are beneficial to good surface polishing and, consequently, high gloss. Size reduction allows for close particle packing, which facilitates the fabrication of resin composites with a high filler volume fraction and good appearance without compromising their mechanical and wear characteristics. 21
Nanocomposites reportedly possess improved mechanical, physical, and optical characteristics. A decrease in filler particle size with a constant filler volume fraction enlarges the surface area of the dispersed phase and then increases the probable constraint upon the matrix phase. Thus, stress is inversely proportional to filler particle size. Filler particles are constrained by the matrix and undergo translation and rotational movement, thereby relaxing stress within the material. 28 However, the same study presented that composites with small filler particles shrink significantly because of the effect of particle shape. One other advantage of nano-size reinforcing agents compared with micro-sized particles was the lower loading requirements.
Zha et al. 29 inferred that even the incorporation of a small amount of silica nanoparticles into the current dental composites could lead to much improvement of their mechanical and curing performances.
In the present study, NBT was used as a reinforcing filler to improve the properties of PMMA denture base materials. NBT particles are characterized by FESEM, and the physical structure of the filler is presented in Figure 3. Akbari et al. 30 reported that microscopy is the only method in which the sizes of individual particles are directly observed and measured. Nano-spherical-shaped particles are seen, and these particles could promote good mechanical adhesion between the filler’s particles and the matrix. Data on particle size and size distribution extracted from 513 particles are presented in Table 2 and Figure 4.

FESEM image of the NBT nanospheres at the magnification of 200000×.
Descriptive analysis of particle size data extracted from the FESEM images.

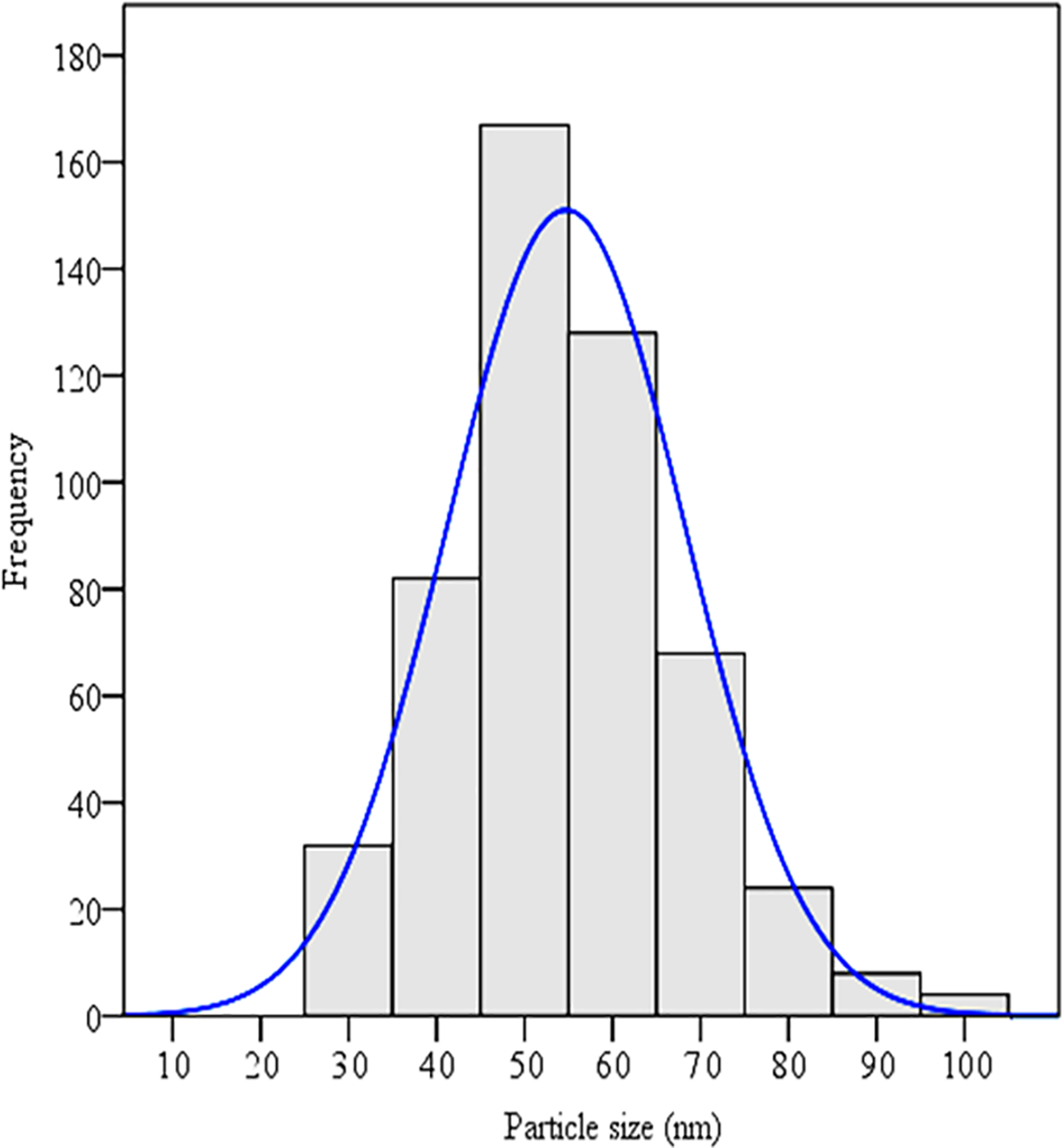
Number–frequency histograms showing the particle size distribution of NBT. The particle size data is based on the FESEM image analysis of more than 500 particles.
The NBT filler is composed of a fine powder that contains spherical particles. These particles promote good mechanical adhesion between the filler particles and the matrix. Satterthwaite et al. 28 described that spherical filler particles display better property enhancement and lower shrinkage–stress values than irregular filler particles. Spherical particles also provide uniform distribution, which maintains dimensional stability. In addition, the capability of the dispersed phase to move within the matrix and relax stress increases as the sphericity of the dispersed phase increases. Another study reported that spherical filler particles affects the microfracture mechanisms of dental resin composites and increases the bending strength and fracture toughness at a high rate for elastic modulus. 31 Data on particle size and size distribution can be represented in either a tabular or graphical form. Data on particle size were obtained after counting more than 500 particles distributed on approximately 18 FESEM images (Table 2). Particle size distribution was plotted on the basis of the particle size extracted from the FESEM images. A number–frequency histogram presents particle size and its distribution. Figure 4 shows the number of frequency histograms of particle size data. Hundreds of particles could be measured to present statistically reliable mean size data. The particle size curve is not skewed and exhibits a normal distribution. For instance, measuring 500–1000 grains for an optimum sample size has been proposed. Brittain 32 reported that powder distributions described via the normal distribution function would be advantageous because all of the statistical procedures developed for Gaussian distributions could be used to describe the properties of the sample.
Filler/matrix interaction
Coupling agents should be hydrolytically stable, not deteriorate in an aqueous environment, and provide a waterproof bond at the interface of two dissimilar materials. Under humid conditions, water can initiate corrosion and aging mechanisms in inorganic material-filled organic composites. TCAs offer superior hydrolytic stability in a wet environment. This TCA property must be considered in fabricating dental composites. However, information on the hydrolytic stability of TCAs is insufficient. Monte 33 stated that after exposing a reinforced composite to boiling water, the silanes lose more than 70% of their original strength, whereas titanates maintained more than 80% of their original properties. Titanate proton coordination covers 100% of the surface and resists aging. A distinct property of titanates is their capability to be coupled in the phase of the matrix resin because of the three functional radicals of the coupling agent and transesterification reaction (Figure 5a). By contrast, the silane coupling agent creates only one functional radical, as shown in Figure 5b, leading to poor silane adhesion. Figure 6 illustrates the FESEM images of of the fracture surface of NBT-filled PMMA. In the absence of a coupling agent (Figure 6a), brittle failure mode was contributed by the formation of voids at the interfacial area because of incompatibility of Un-NBT/PMMA and the affinity of nanoparticles to agglomerate. In case of silanated NBT (Figure 6b), nanofiller particles were incompletely wetted by the matrix which indicates weak filler–matrix interaction, leading to debonding prior to the full development of plastic deformation. Figure 6c shows that the titanated NBT were embedded in the matrix with good interfacial bonding. In addition, the vacuoles and agglomeration of the filler in the fractured surface were much less. The introduction of TCA into the system promoted polymer–filler bonding; thus, it improved the performance of the composite. Consequently, the chemical bonding between the filler and TCA considerably strengthened compared with that in silane.
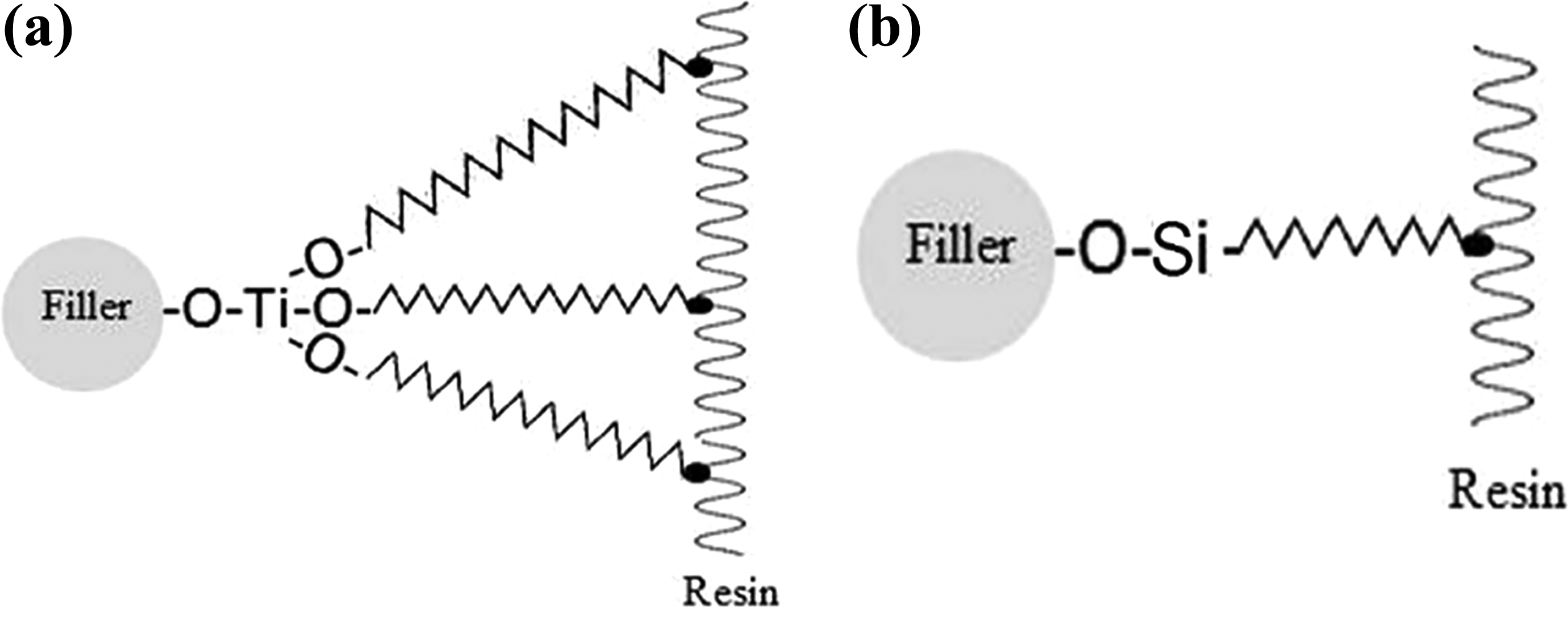
Reaction scheme for different coupling agents; (a) titanate-treated composite showing three reaction sites; (b) silane-treated composite showing one reaction site

FESEM images of the fracture surface of (a) Un-NBT/PMMA, (b) Si-NBT/PMMA, and (c) Ti-NBT/PMMA.
Titanates not only aid the mixing of two phases but also help improve the particle dispersion because of the hydrophobic particle surfaces. The efficacy of surface modification of NBT by TCA and the formation of a monomolecular film on the surface of NBT powder have been established. 6 Filler dispersion results from the application of electrochemical and mechanical forces to the interface of the inorganic filler/polymer, leading to the removal of air and water voids and enhances hydrophobicity, ultimately resulting in deagglomeration of filler particles and the creation of a true continuous inorganic/organic composition. 16 By replacing water present at the filler surface with the organo-functional titanate, the filler–matrix interface will become compatible, enhancing the dispersion of filler particles. Consequently, this phenomenon results in a more uniform dispersion and deagglomeration of the talc fillers in the matrix. The dispersibility of filler nanoparticles in PMMA matrix before and after titanation is schematically presented in Figure 7. As previously reported, the porosity level is remarkably decreased after titanation of NBT. This behavior was attributed to enhanced adhesion between the NBT and PMMA matrix. 6 Except for silanes, titanate coupling agents have not been explored to be used in dental composite materials.
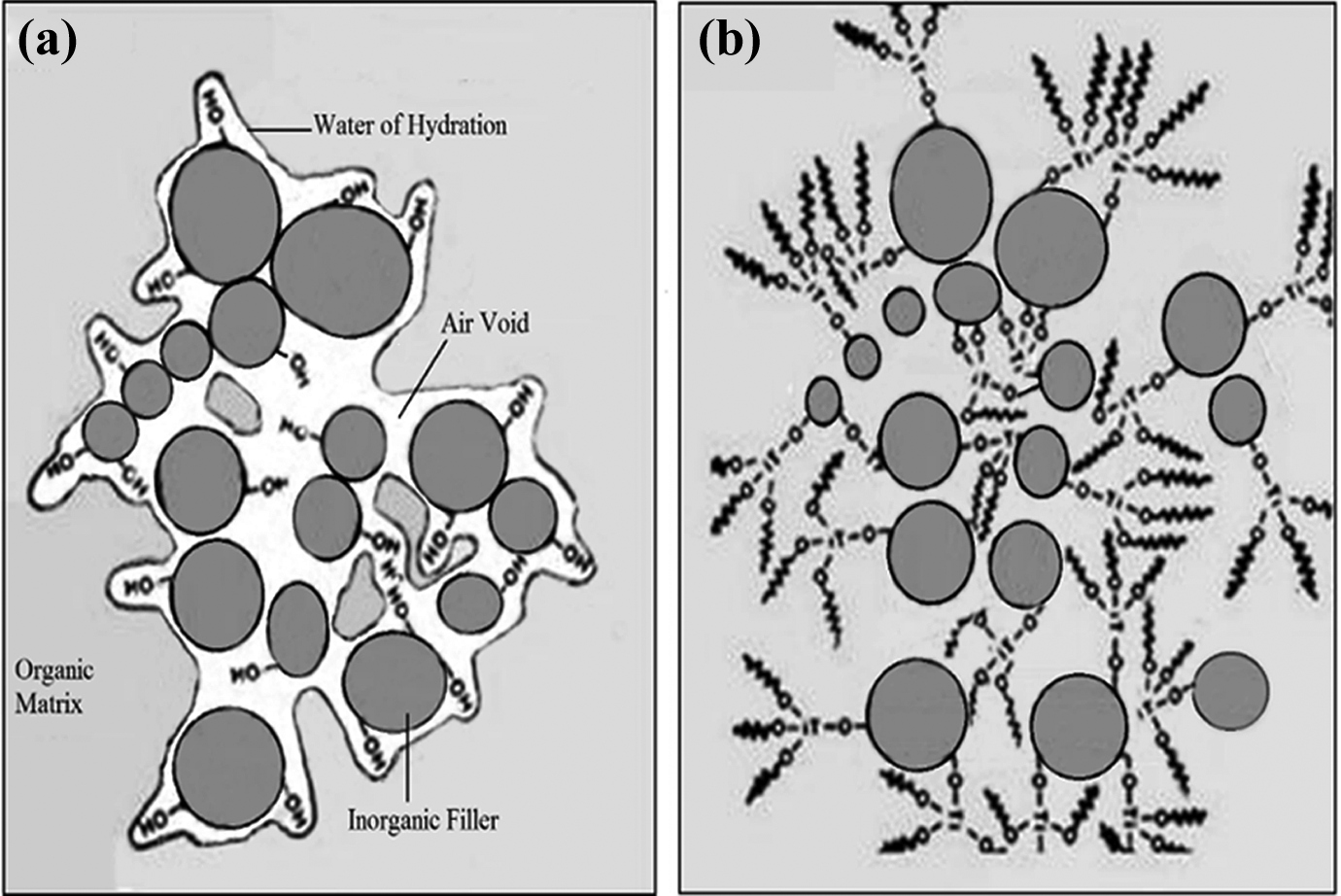
Illustration of the dispersion effect of coupling a titanate monolayer on an agglomerated inorganic material in an organic phase: (a) before; (b) after titanation.
Behavior in SBF
When the polymer–resin composite was soaked in an aqueous environment, two phenomena occurred. First was a rapid elution of uncured monomer. Simultaneously, SBF was absorbed by the composite, predominantly through diffusion, into the polymer matrix leading to failure of filler–matrix bonding thereby inferior mechanical properties. 11 In general, the absorption rate is represented by the diffusion coefficient. Thus, the solvent uptake of the dental composite is an important property that must be investigated. Another event is solubility, which is described as the maximum amount of solute that can dissolve in a specific solvent under a given temperature. The water sorption and solubility phenomena are associated with physical changes (such as plasticization and swelling) and chemical changes (such as oxidation and hydrolysis). 5
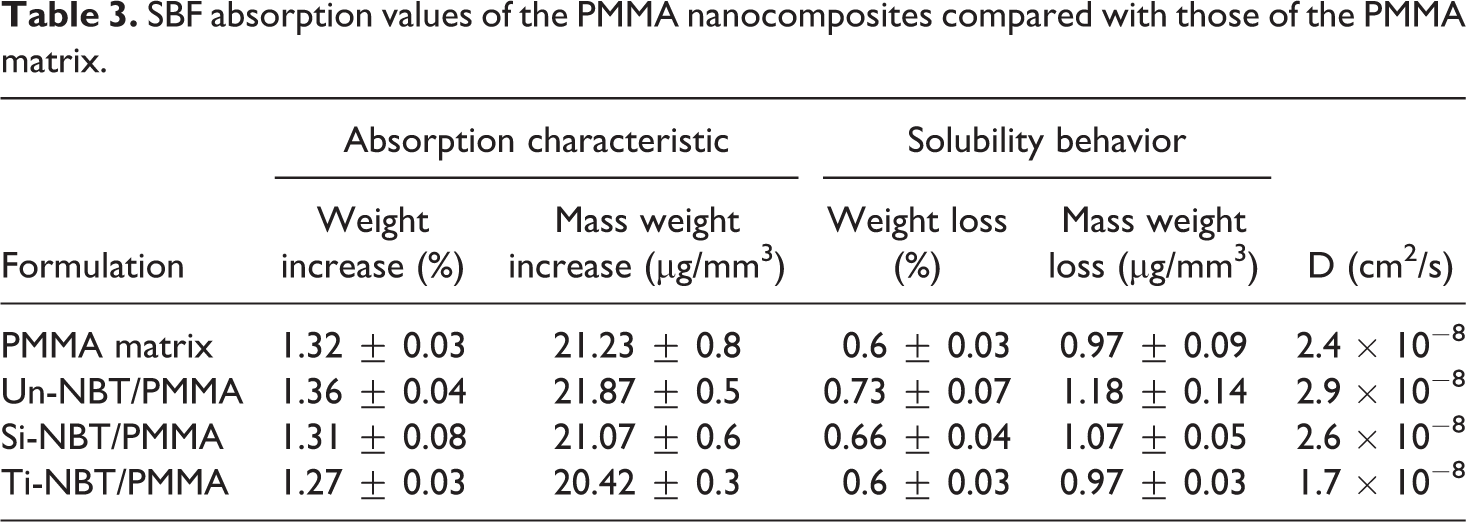
Table 3 shows the SBF absorption solubility and diffusion coefficient values of the NBT-filled PMMA and PMMA matrix. The SBF behavior for all formulations was remarkably lower than the ISO standard, which requires that the absorption and solubility of all materials must be less than or equal to 32 μg/mm3 and 1.6 μg/mm3, respectively. The behavior of SBF absorption of pure PMMA and NBT/PMMA nanocomposites is presented in Figure 8. The SBF uptake increases steadily with time until it reaches the saturation limit after 45 days of immersion. Moreover, the samples notably showed significant SBF gain in the first 2 weeks and remained stable after the fourth week of immersion. The PMMA matrix showed higher SBF absorption capacity than the Ti-NBT/PMMA. This behavior can be attributed to the fact that the presence of polar and hydrophilic functional groups in acrylic resins may increase their polarity and facilitate the uptake of storage solution to form primary and secondary hydration shells around the polar groups via hydrogen bonding. Al-Bader et al. 2 stated that water molecules are able to diffuse through the interchain spaces of the resin matrix because of their small radius size, which is less than 0.158 nm and smaller than the interchain spaces of the matrix. In addition, the Un-NBT/PMMA samples exhibited the highest values of SBF uptake and solubility because of their poorly bonded area and weak interfacial adhesion between the NBT and PMMA, leading to accumulation of water molecules at the interfacial region. The high moisture absorption of the filler occurs due to hydrogen bonding of water molecules to the hydroxyl groups on the filler surface, leading to a moisture build-up in the fiber–matrix interface. 34 This behavior could facilitate filler leaching into surrounding media, leading to weight loosening after dehydration.
SBF absorption values of the PMMA nanocomposites compared with those of the PMMA matrix.
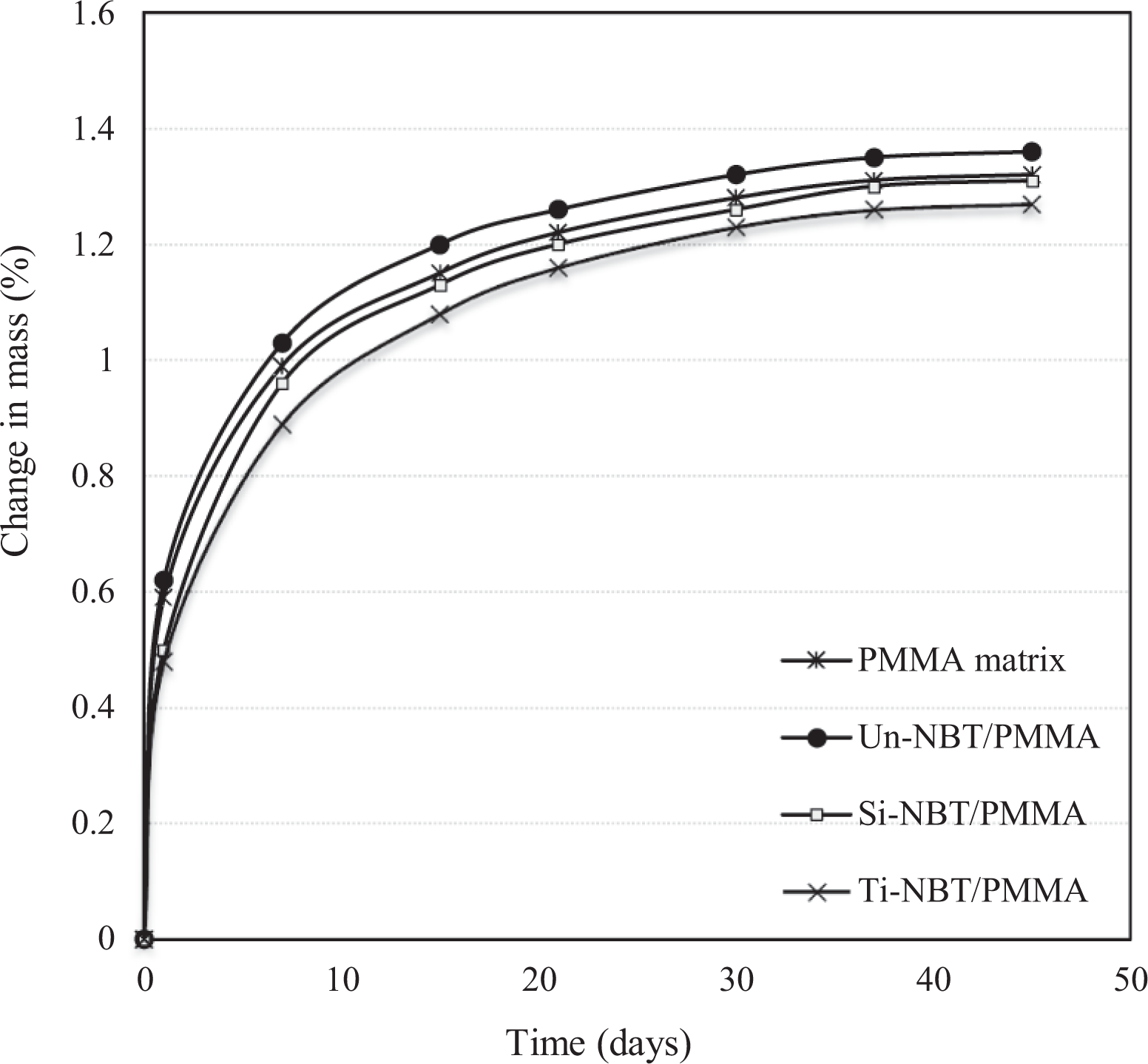
Dependence of water absorption on time for pure PMMA and untreated, silanated and titanated NBT/PMMA nanocomposites.
However, lower SBF uptake, solubility, and diffusion coefficient values were recorded from the treated sample than the Un-NBT/PMMA samples because of the presence of the coupling agent, which induced the hydrophobicity of the NBT and enhanced the adhesion between the filler and PMMA matrix; this condition inhibited the capability of the composites to absorb water. 20 Moreover, the weight gain and diffusion coefficient of the Si-NBT/PMMA samples were higher than those of the titanated ones. This observation can be explained by the absorbed moisture hydrolyzing the silane interface and opening up an extra pathway for SBF diffusion. The filler porosity of the NBT agglomerations provided a fast diffusion path. According to Al-Bader et al., 2 water absorption and the diffusion coefficient depend on the structure of the polymer, the nature of the solvent, the filler composition, the presence of air-filled voids within the matrix, and interaction between polymeric matrix and filler. Zanchi et al. 5 reported that artificial aging of silanated filler surfaces could break siloxane bonds, and the hydrolysis induces debonding of the filler particles, increasing the mass loss of the composite and leading to a shortened service life of dental restorations.
The equilibrium moisture content and the diffusion coefficient are depending on the type of coupling agent in NBT/PMMA nanocomposites. In Ti-NBT/PMMA, the lowest absorption, solubility, and diffusion coefficient values were attained because of the increase in compatibility between the NBT and PMMA matrix, resulting in inhibition of filler leakage. The mechanism through which water diffuses into polymeric materials can be explained as either infiltration into the free space (i.e., microvoids and other morphological defects) or specific molecular interaction being controlled by the available hydrogen bond at hydrophilic sites. The slight decrease of the diffusion coefficient for Ti-NBT compared with Si-NBT imply the reduction of voids for water accumulation and a hydrophobic behavior due to the better interaction of NBT filler and PMMA matrix via titanation of NBT. This observation is consistent with other researchers34,35 who reported that a high contact angle and reduced hydroxyl groups on titanated fibers favor the improved hydrophobicity of the composite over the treatment, leading to increased moisture resistance. In addition, the surface free energy of untreated and silanated fibers is almost similar. Meanwhile, lowering of the surface free energy has been reported when the fiber surface is modified with TCA. 35 This observation indicates that titanates protected the interface from hydrolytic deterioration, implying that titanates can provide better hydrolytic stability than silanes. Krysztafkiewicz et al. 36 concluded that the highest degree of hydrophobicity was observed for silicas modified with titanates, whereas hydrophobicity slightly decreased after modification with silane. These findings show that although the sorption and solubility are multifactorial processes, they are dependent mostly on the hydrophobic nature and chemical composition of the dental composites.
Leaching experiment
In an aqueous environment, composites not only absorb water but also elute filler elements, unreacted monomers, and polymerization promoters. Consequently, parts of the material may be lost into the oral fluids either via diffusion or ion exchange, which is commonly referred to as leaching. In addition, water may cause debonding of filler particles from the matrix or even hydrolytic degradation of filler particles, leading to a shortened service life of composite restorations.
The results of the leaching experiment, depicted in Figures 9a to 9c, confirm the leaching of the filler elements. The existence of Ba and Ti is clearly evident in the immersing media because of the filler leaching via ion exchange. 9 The values of leached filler elements in SBF solution are shown in Table 4. The highest level of the filler constituents (Ba, Ti) leakage was presented by Un-NBT/PMMA samples (34.61, 9.52 respectively) because of the higher tendency of the filler components to leach compared with the treated samples because of a lack of filler surface functionalization and weak adhesion in the composite’s phase. This behavior could accelerate the leaching of the filler components in SBF and lead to deleterious effects in the polymeric network of the material.
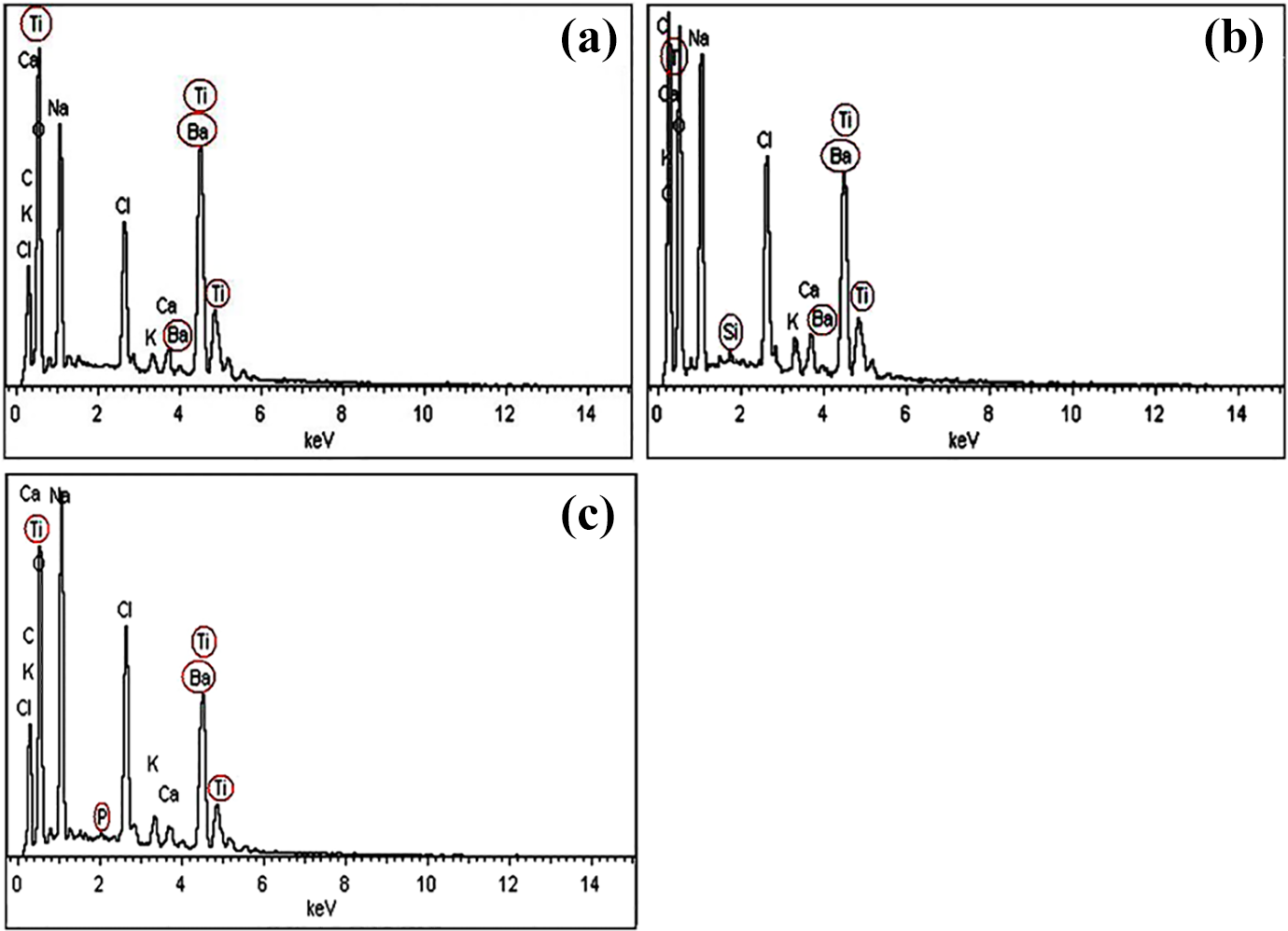
EDX graphs of the storage liquid showing the leached NBT elements: (a) Un-NBT; (b) Si-NBT; (c) Ti-NBT.
Percentages of leached elements in the SBF solution.

However, the leakage of Ba and Ti is reduced by 76% after titanation, indicating the enhancement of the material stability in moisture. Although the treated sample filler showed slightly lower value of filler leakage than unfilled PMMA, the Si-NBT/PMMA showed higher leached elements (30.82, 8.84) than the Ti-NBT (22.47, 5.91 for Ba, Ti respectively) because of Si instability in moisture and poor interfacial bonding with dental matrix due to incomplete silane coverage, leading to filler leaching and forming a porous structure in dental composites. Si–O–Si bonds start to break as a result of the increased hydroxyl ion concentration. The production and concentration of hydroxyl ions during hydrolytic degradation of siloxane layer lead to an autocatalytic reaction that can result in the following: degradation of the interface and subsequent leaching of ingredients, micro-cracks at the interface, particle deboning, and reduced fatigue resistance and mechanical properties. Salivary esterases and organic solvents existing in the oral cavity may cause more destructive effects than water, particularly under cyclic loading. 15 The presence of the Si group (0.81%) also confirms that the hydrolytic degradation of silane, and leaking into the surrounding liquid occurred because of the slow development of superficial flaws related to the preexistent corrosive process. 9 Such a process can be explained by the assumption that, during interaction with water, the Si–O–Si structure becomes negatively charged. These negatively charged filler particles will restrict the amount of cations that can leave the filler surface. However, if positive ions diffuse through the matrix and interact with the negatively charged Si–O–Si surface, and then some of the original filler cations can easily leave the filler particles and diffuse into the storage medium. Such ion exchange process implies that the original filler ions have been substituted by ions from the solution. Varying works have been done with identical results, indicating that aging in artificial saliva resulted in an ion charge balance, which allowed for an elevated and continuous release of Si.11,12 Söderholm et al. 12 concluded that storage solution, filler composition, and total time in the storage solution had strong effects on leachability. The silanated Ba-containing filler leached Si faster in artificial saliva than in distilled water and roughly twice as much as the quartz filler. The release of Si ion was the greatest for all of the composites and fairly consistent between the four different composites due to the weakening of the bond between the resin matrix and the glass filler. 11 Ion exchange processes occurred at the solid–liquid interface after dental composites were aged in a storage solution. When an ion is released from the surface of a dental composite, the leaching process will induce a charge imbalance at the solid–liquid interface, delaying further ion release. However, other ions of the same charge, such as Na, K, and Ca, in the SBF solution (Table 1) will help re-establish a charge balance at the interface. These changes may result from chemical breakdown through hydrolysis, leaching of the interface, and loss of strength due to corrosion.10,37
However, the Ti-NBT/PMMA samples exhibited the lowest values of leached elements. The concentration of Ti in titanated samples is higher than silanated ones because of Ti-based coupling agent, but the amount of ion released was notably less at 6 weeks of immersion. The high stability of Ti-NBT/PMMA in an aqueous environment can be attributed to the alteration of the filler composition because of TCA and its tendency to form –Ti–O–Ti–O– bonds leading to enhanced chemical compatibility. This behavior could inhibit the leaching process. Thus, chemical compatibility between the dispersed phase and the coupling agent should be considered. 24 These results are consistent with the findings of a previous study which determined that the treatment of TiO2 with the effective TCA not only enhances the bond between TiO2 and monomer but may also prevent the leaching process of the TiO2 composites in oral environments. 38 In addition, TCA application changes the hydrophilic character of the NBT to hydrophobic. Such coupling agent improves the compatibility of two different phases, and interpenetration of TCA and matrix lead to their mixing on a molecular scale which can prevent leaching by limiting water penetration along the interfaces and decreasing the hydrolytic degradation of composite material. 15
Effect of SBF exposure on fracture toughness
The mechanical properties of dental composites are dependent on the component properties, composition, structure, and filler–matrix interaction. Improved mechanical properties strongly depend on the stress transfer between the particles and the matrix. For well-bonded particles, the applied stress can be effectively transferred to the particles from the polymer matrix resin. 39 Many authors found that fracture toughness is a suitable measure of the effects of resin modifications and that this parameter is affected by filler surface treatment.15,37 Although the treatment of the filler surface could enhance the fracture toughness of the composites, the coupling agent should protect the interface from hydrolytic deterioration and offer reliable bonding in the wet environment.
The present study was originally planned to investigate the hydrolytic stability of denture base nanocomposites by determining the effect of 6-week SBF storage on KIC. The results revealed statistically significant differences in KIC among the experimented samples in the four groups (
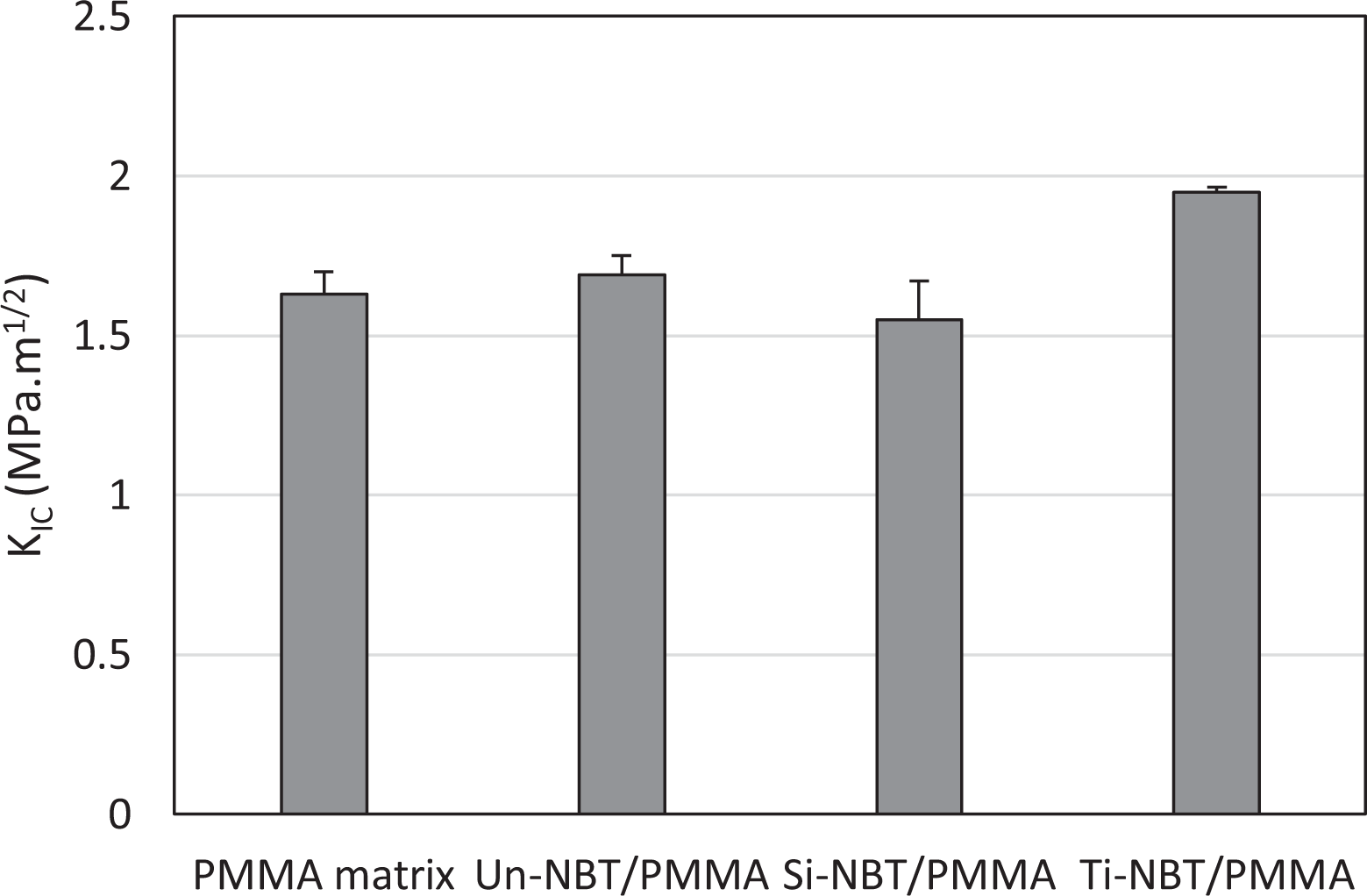
Fracture toughness of the PMMA matrix, Un-NBT/PMMA, Si-NBT/PMMA, and Ti-NBT/PMMA nanocomposites after 6-week immersion in SBF.

FESEM micrographs of the fracture surfaces of (a) PMMA matrix, (b) Un-NBT/PMMA, (c) Si-NBT/PMMA, and (d) Ti-NBT/PMMA after 6-week soaking in SBF at 37°C.
The fracture surface of the uncoupled NBT-filled PMMA matrix (Figure 11b) exhibits randomly propagated smooth fractured surface, which indicates the brittle behavior induced by the PMMA matrix. Brittle failure would be aggravated by the formation of voids due to the incompatibility of the Un-NBT/PMMA and the poor dispersive effect of the filler in the matrix that leads to the accumulation of water molecules at the interface. The filler nanoparticles are small and spherical, causing very high surface energy, leading to the formation of a high nanoparticles-nanoparticles interaction.
41
In addition, the absence of coupling agents could impair the tendency of the mineral to dissolve partly in the SBF, thereby affecting the mechanical performance of the nanocomposite.
20
Although wet samples exhibited a slightly higher KIC, their toughness was lower than the titanated ones (
A decrease in
Although the deteriorative effect of SBF on the strength of dental composites was reported.7,20 others concluded that aging in aqueous solutions may have a beneficial effect on dental composites due to internal stress relaxation that results in enhanced toughness.
40
However, the Ti-NBT/PMMA samples had higher
According to the manufacturer’s description, this type of titanate is hydrolytically stable and has a greater affinity for filler protons than water molecules, and this hypothesis was in agreement with this finding. Furthermore, the enhanced filler–matrix adhesion can be because of pyrophosphate, which acts as a binder group and offers substantial hydrolytic stability. Thus, the chemical structure of the coupling agent could effectively affect the hydrolytic behavior of dental composites. Cheng et al. 42 reported that couplers containing phosphate have gained increasing attention as a monomer alternative to silane coupling agents due to its enhanced bonding and hydrolytic stability.
Conclusions
Coupling agents display considerable influence on the examined properties. With respect to the behavior of the titanate coupling agent, TCA was more effective than silane and exhibited better stability in moisture by minimizing the amounts of leached elements and decreasing SBF absorption and solubility. Moreover, the titanation of the NBT enhanced the fracture toughness after aging in SBF because of its excellent interfacial bonding. FESEM images showed that the titanated filler nanoparticles were well-bonded to the PMMA matrix, and no vacuoles can be observed. These results indicate that TCA effectively reacts to develop the hydrolytic stability of dental composites. Overall, the Ti-NBT/PMMA nanocomposites examined in this study demonstrated adequate fracture toughness and higher stability in the wet environment leading to the promotion of the clinical longevity and make this material an important area for further research.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by Universiti Kebangsaan Malaysia (UKM) and the Ministry of Education, Malaysia via the research sponsorship of DIP-2016-001. The authors gratefully acknowledge the Center for Research and Instrumentation Management (CRIM), UKM for their support and excellent testing equipment.
