Abstract
The main element of electrophotographic (EP) printing and copying devices is a polymeric composite called toner and one of the most significant components of toner composite is an iron oxide (magnetite). Magnetite, which is applied as a colorant and additive for toner is the main mineral able to develop an electrical charge on the printing procedure. Although there are several ways to produce magnetite, given the dearth of resources and environmental aspect, it is safer to practice recycling and greener method. In the present study, an encouraging way to reuse the magnetite particles as a byproduct of the preparation of micro silica in the concrete industry was described. The obtained magnetite was then utilized as the charge control agent to produce magnetite/carbon black/styrene co-butyl acrylate composite microspheres by green emulsion aggregation method, which is used as toner in the printing procedure. Characterization of toner and recovered magnetite was done by X-ray Powder Diffraction (XRD), Atomic Gradient Force Magnetometry (AGFM), Particle Size Analysis (PSA), Differential Scanning Calorimetry (DSC), and Scanning Electron Microscopy with Energy Dispersive X-Ray (SEM-EDX). The results supported the fact that the produced toner composite by recycled waste magnetite show suitable characteristics comparing to an industrial toner.
Keywords
Introduction
Toner is a composite powder consist of a polymer, colorant, iron oxide, and additives, which is used as electrophotographic (EP) printing ink. Over the recent decades, this method of printing has grown into an equitable substitute to other print technologies. 1 Technically, the ink used in the EP printing, which is called toner needs to be magnetic. 2 Monochrome toner has been separated into two categories including non-magnetic and magnetic toner, which contains 0–15% and 30–50% of iron oxide, respectively. 3
As the strongest natural magnetic mineral Iron oxide occurs in many varieties. 4 Because of Magnetite (Fe3O4) magnetic assets, color, density, and hardness 5 it has scientific and industrial applications. 6 Magnetite is used as a pigment, in a building materials, and, exclusively, in printing toner to control the charge of the toner.7,8 There are currently several techniques to produce magnetic particles, such as micro-emulsions, 9 laser-pyrolysis, 10 sonochemical synthesis, 11 and chemical co-precipitation. 12 Due to a great amount of magnetite usage and the famine of resources and environmental aspect, it is important to determine the environmentally green and also clean way for this particle production. Developing countries are taking the major responsibility of recycling waste. 13 On the other word, recovering waste is a vital social issue in developing nations not only for environmental protection, but also for resource recycling. 14
Concrete is well-known as one of the most wieldy used materials in the construction business. Its development has been linked with attempts of optimizing its performance, which is typically achieved by changing its characteristics. 15 In a beginning stage, these adjustments result from the balance adjustment of the materials and, later, from the incorporation of new constituents. 16 Worldwidly, More than 500,000 MT of microsilica is sold to the construction industry, which used in fiber cement, oil-well drilling, refractories and concrete treatment. 17
Microsilica is obtained as a byproduct in the procedure of producing ferrosilicon. The process of manufacturing ferrosilicon is accompanied by the production of a relatively large amount of gases from the reaction zone (mainly contains the CO, SiO gases), which are cut along the loading surface of the furnace and converted to CO2 and dust containing SiO2. Amorphous micro silica is separated from the gases after a series of mid-stage procedure. Actually, for many years, micro silica remains as a waste of ferrosilicon production, only with the advent of technology and the finding of different functions for these wastes (particularly in concrete), now micro silica as a byproduct of this process is being traded. It should be mentioned that in the output process of ferrosilicon, per ton of ferrosilicon produce about 220 kg of micro silica. 18 As an impurity, Micro silica contains trace quantities of heavy metal oxides and organic deposits. 19 One of these metal oxides impurities and waste in micro silica is iron oxide.
In a series of earlier publications, the author studied the influence of agitation speed, 20 toner component, 21 and colorant 22 on the assets of the produced toners prepared by emulsion aggregation (EA) process. Likewise, the author produced ceramic and florescence toner by EA process. Emulsion aggregation (EA), the developed technique by the Xerox Corporation Ltd. was a chemically controlled process with the power to yield toners with smaller particle size, narrower dispersion and uniform shape. This technique is environmental-friendly concerning both the process and the final product 23 which has demonstrated a reduction in the amount of energy per pound of produced toner composite.
In the present research, the possibility of producing printing toner composite with iron oxide (magnetite) obtained as a waste in preparing and treatment of micro silica for the Concrete industry via green EA method was described. Accordingly, recovered magnetite and carbon black were incorporated into poly (styrene-co-acrylic acid) using an eco-friendly emulsion aggregation (EA) approach, with the ending application as an electrophotographic printing ink composite, named toner.
Material and preparation
Materials
A styrene-acrylic (R579; ResinFam Co., Iran) was used as a polymer in this study which, according to the supplier, has a medium pH value, Tg of 51°C, and mean particle size of 220 nm. A polyethylene wax (Kala Kar Co., Iran) and a carbon black (CB) pigment (Printex U, Degussa-Evonik, Germany) were also employed and Polyaluminum chloride (PAC) was used as a coagulating agent.
Micro silica is a byproduct of the ferrosilicon Production Process, which is obtained from the Ferrosilice Company (Iran).
Preparation
The magnetite recycling
In order to treat and prepare micro silica slurry for the concrete industry, it is necessary to use HCl 5% as ionic stabilizers Merck (Germany) and magnesium sulfate 2% as mineral salts Merck (Germany) to modify the micro silica surface. The above mentioned chemicals were all used as received. As a consequence, black sediments of waste iron oxide are produced in the container, which should be taken away from the micro silica slurry. Those particles have magnetism properties and are separated from the slurry by a magnet. Figure 1 illustrates the preparation scheme.
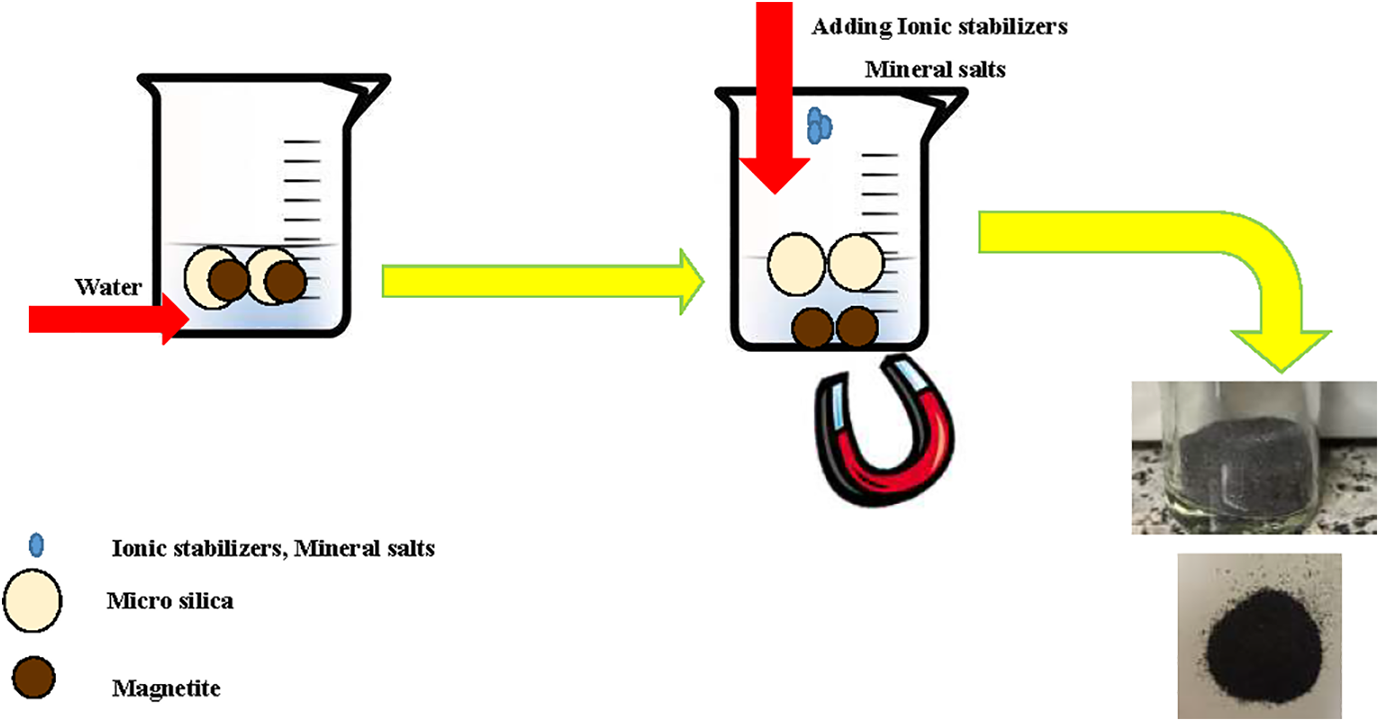
Schematic of magnetite recycling from micro silica.
Preparation of the printing toner composite
Toner particles were produced via a stepwise procedure, in accord with previous studies.24,25 First (step a), a 1-liter beaker was filled with 24.5 g styrene-acrylic latex, 2 g carbon black, 3 g wax, 3 g magnetite and 120 g deionized water and the contents were mixed manually at room temperature for about 15 min. In step b, the resulting suspension obtained in a mixed using a homogenizer for 5 min. Next step, that is c, was started by continuous mixing of ingredients at room temperature for about 1 h followed by addition of a solution of 0.6 g coagulation agent in nitric acid over 10 min until reaching the pH value of the mixture to 2. In this manner, a gel was seen to form, as a consequence of changing the viscoelastic nature of the suspension from a Newtonian water-like fluid to a shear-thinning paste-like gel. In step d, the temperature of the mixture was raised to 50°C for about 30 min while the gel was continually mixed. The mixture was held at this temperature for another 60 min in step e, where the temperature of the mixture was increased to 96°C for 30 min. The last step, denoted as g, was started by holding the product of step f, at 96°C for a further 60 min.
The ultimate mixture was neutralized with sodium hydroxide solution and cooled down to 25°C, after which the produced toner were isolated from the water, washed to remove divalent ions, filtered, and dried with a frizzed dryer. Figure 2 shows the variation of pH and temperature over the course of the synthesis process of the toner.
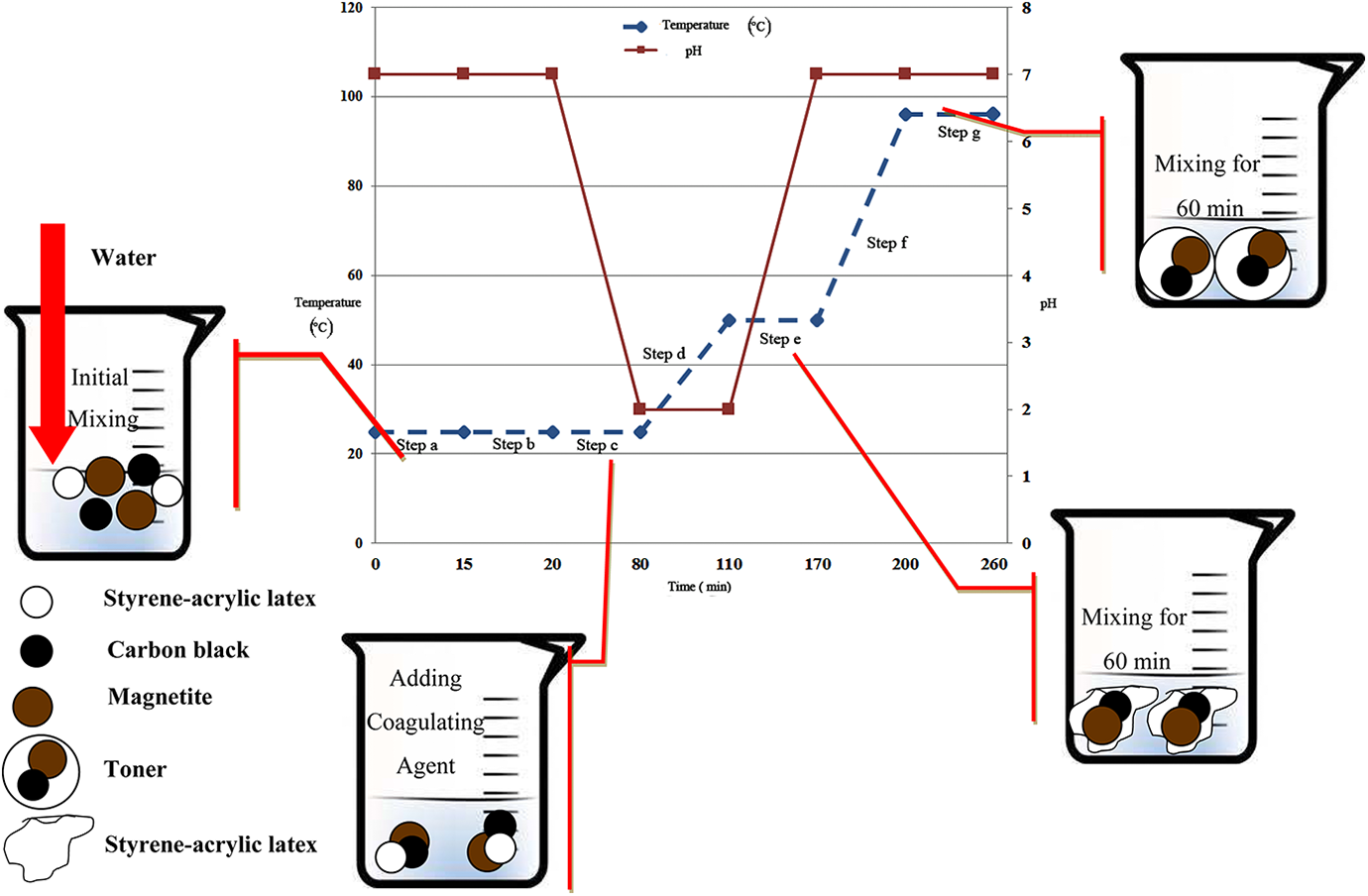
The graphics of composite toner production process.
The printability of the produced toner composite was checked by the printing in a controlled environment using a monochrome laser-jet printer (HP 1100). For comparison a toner composite without magnetite is also prepared.
Characterization
Fourier transform infrared (FTIR) spectra of samples were recorded by means of a PerkinElmer Spectrum One spectrometer. FTIR spectroscopy was carried out in diffuse mode using KBr pellets prepared from toner and magnetite.
X-ray diffraction (XRD) analyses of the toners and magnetite were performed using Cu-Kα (λ = 1.54056 Å) radiation over the diffraction angle (2θ) range of 10° to 100° with a typical step size of 0.026° by an X-ray diffractometer (STOEstadi P, Germany) using X’Pert Highscore software.
Scanning electron microscopy, in combination with energy-dispersive X-ray spectrometry (SEM/EDX S360, Cambridge Instruments Ltd., UK) was conducted to investigate the existence of the magnetite. The EDX measurements were conducted by using a primary electron beam with an acceleration voltage of 20 kV to detect.
The saturation magnetization was measured using an alternative gradient force magnetometer (AGFM-150)
The size and size distribution of the toner particles was determined using a Particle Size Analyzer (PSA, Mastersizer2000, Malvern, UK).
Differential scanning calorimetry (DSC, PerkinElmer, USA) was also used to investigate the thermal properties of the toner. Approximately 5 mg of each powder toner sample were loaded into pans and sealed with a covering lid. Measurements were conducted over a temperature range of 0–160°C at a heating rate of 10°C/min in an atmosphere of nitrogen.
Color measurement of the printed toner was conducted on a GretagMacbeth Color-Eye 7000A spectrophotometer (USA).
The optical density of the printed samples was measured using a spectrophotometer (S900; Ihara, USA) for a range of 400 nm to 800 nm with 0.45 geometry.
Results and discussion
In order to confirm the composition and structure of samples, FTIR spectra were measured. As shown in Figure 3, magnetite show typical bands centered at 585 and 635 cm−1, which are related to the Fe–O stretches and the absorption band at 1635cm−1 assigned to the O-H bending groups. FTIR spectra, provided by toner samples containing magnetite, CB and St/BA, proved the formation of composite particles of toner. The peaks shown in Figure 1 are as follows: (1) C-H of copolymer Aromatic ring in 3060 cm−1, (2) aliphatic C-H in 2926 cm−1 and 2960 cm−1, (3) the carbonyl group of the ester bond in 1733 cm−1, (4) C=C of copolymer Aromatic ring in 1601 cm−1, (5) C-O of ester group in 1150 cm−1, and (6) main body C-H2 of copolymer and butyl substituent in 1493 cm−1 and 1452 cm−1. 26
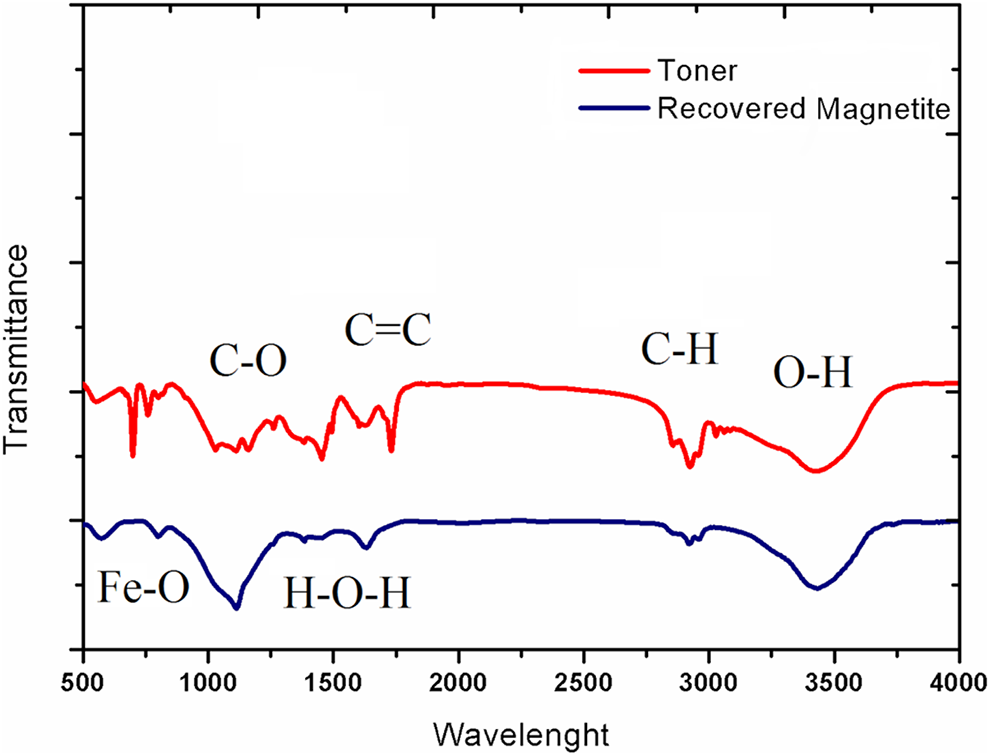
The FTIR diagram of produced composite toner and recovered magnetite.
FTIR spectrum in Figure 3 also shows that the H-O-H bending vibration at about 1000–1600 cm−1. Additionally, the second absorption band, between 900 cm−1 and 1000 cm−1, corresponds to bending vibration associated to the O – H bond. 27 These first two bands correspond to the hydroxyl groups attached by the hydrogen bonds in the iron oxide surface, as well as the water molecules chemically adsorbed to the magnetic particle surfaces. 28
Figure 4 presents The XRD of the produced toner, recovered magnetite and CB/St/BA (toner composite without magnetite). It is clear that the recovered magnetite is the combination of Iron Oxide (Fe3O4), Hematite (Fe2O3), and Silicon Oxide (SiO2), which correspond to JCPDS 01-075-0033, JCPDS 00-033-0664, and JCPDS card 01-086-1629, respectively. For the Fe3O4 particles, the diffraction peaks at 2θ of 30.206°, 35.565°, 43.199°, 53.579°, 57.121°, and 62.734° correspond to the inverse spinel structure of Fe3O4. These peaks were attributed to the diffraction faces of the crystals at 220, 311, 400, 422, 511, and 440, correspondingly. 29 The peak position of recovered magnetite was unaffected before and after encapsulation by St/BA and CB. This result showed that the crystal structure of recovered magnetite is not changed during the toner preparation. The broad diffraction peaks of CB/St/BA microspheres between 10° and 30° were attributed to the amorphous polymer in the microspheres. 27
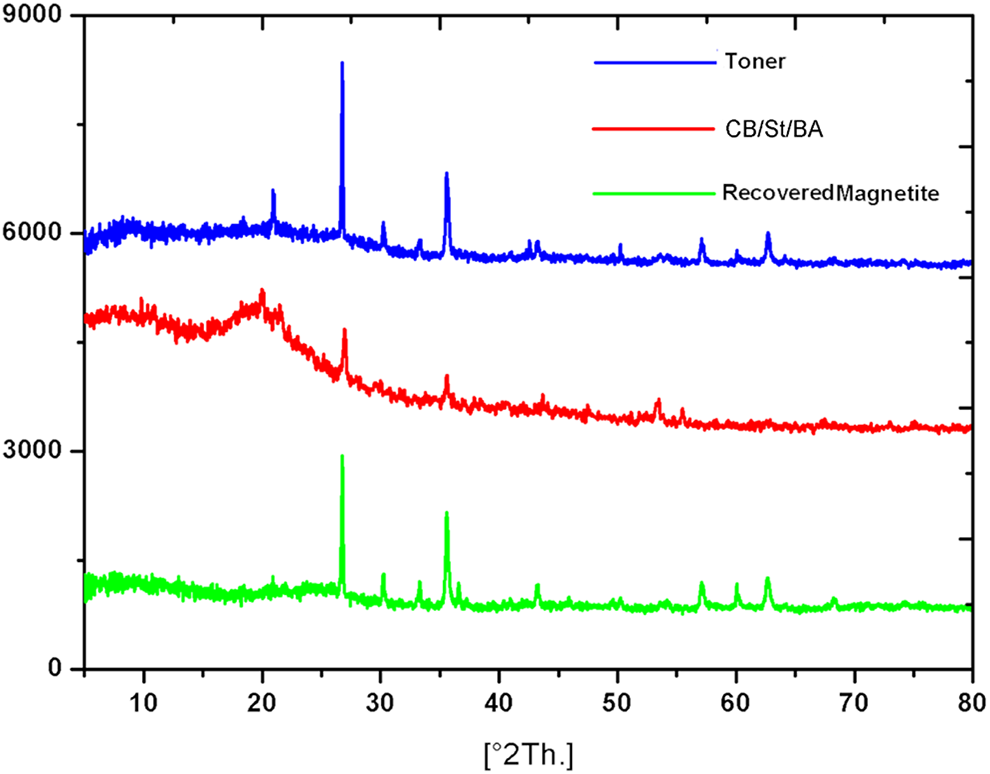
The XRD pattern of produced composite toner, recovered magnetite and CB/St/BA (toner composite without magnetite).
Figure 5 shows SEM images and EDX analysis of the composite toner and recovered magnetite particles. It can be seen that mostly toner particles are nearly spherical in shape with a diameter in the range of a few micrometers to dozens of micrometers. Also, some amounts of magnetite particles present in the exterior of the microspheres and some were embedded in the surface of toner and also micro silica particles are seen beside magnetite particle. The EDX spectrum of the recovered magnetite indicated the detection of oxygen (49.5%), carbon (30.5%), silicon (6.9%), and iron (3.1%) element.
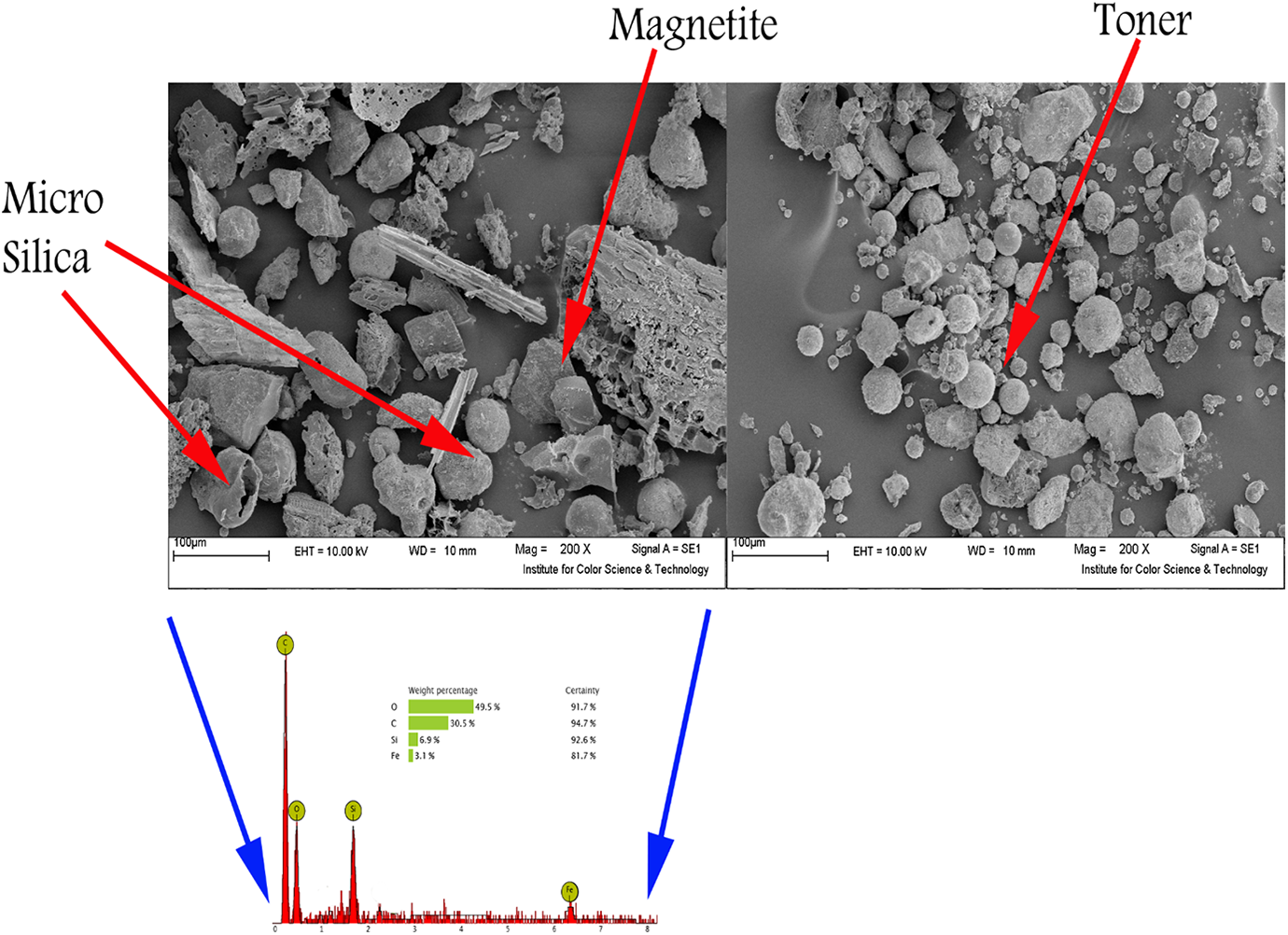
The SEM micrograph and EDX analysis of composite toner and recovered magnetite.
The toner and recovered magnetite magnetic characterization was done using vibrating sample magnetometer (VSM) in the physical property measurement system (PPMS) of Quantum Design, at room temperature, with a magnetic field in the range of −10000 to 10000 Oe. Figure 6 showed the hysteresis loop of the toner, recovered magnetite, and original toner on the market that exhibits a low remanent magnetization value similar to superparamagnetic behavior. The obtained saturation magnetization (Ms) for toner, recovered magnetite, and original toner at room temperature were 58, 72 emu/g, and 60 respectively. The comparison between the produced toner and Original Equipment Manufacturer (OEM) toners showed that the recovered magnetite from the waste of micro silica could be an appropriate replacement for market magnetite.
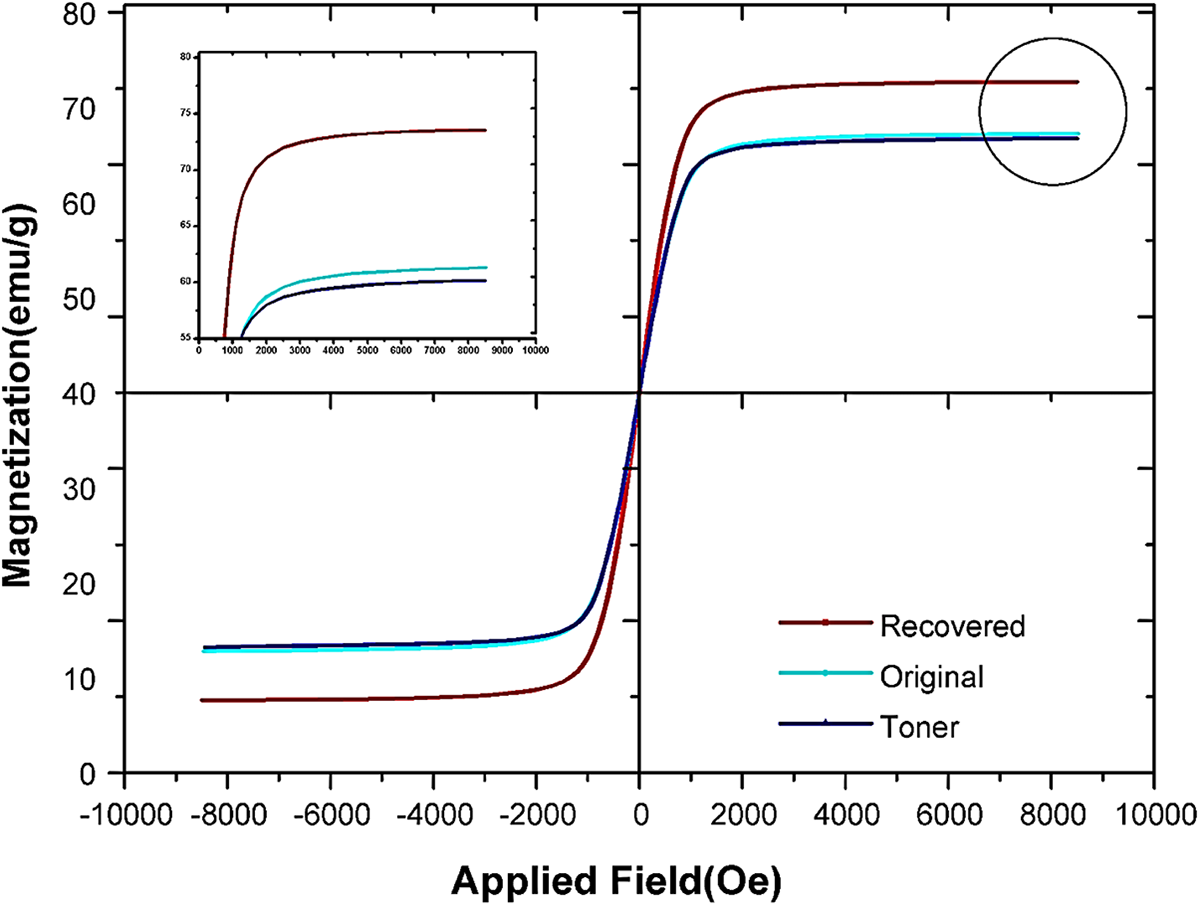
The VSM micrograph of composite toner, recovered magnetite, and original toner magnetite.
The thermal characteristics of the toner, especially Tg, has a direct effect on fixing the properties of the toner on the substrate. Tg determines the fusing temperature of the toner. Generally, a moderate Tg value is required for the toner to have appropriate fixing properties. Too high Tg results in high energy consumption during the printing process. Conversely, if Tg is too low, the toner will stick to the printer cartridge. Suitable fixing properties for Original Equipment Manufacturer (OEM) toners for energy-efficient laser printing requires a Tg value in the range of 50°C to 80°C. 30 Figure 7a shows the results of differential scanning calorimetry analysis of the printing toners. The appropriate amount of Tg may be related to the Tg of the polymer component (styrene-acrylic resin) used in toner formulation (Tg = 51.12°C). In other words, the Tg value of the toner sample show that the synthesized toner has the thermal characteristics of an OEM toners for use in EP printing.
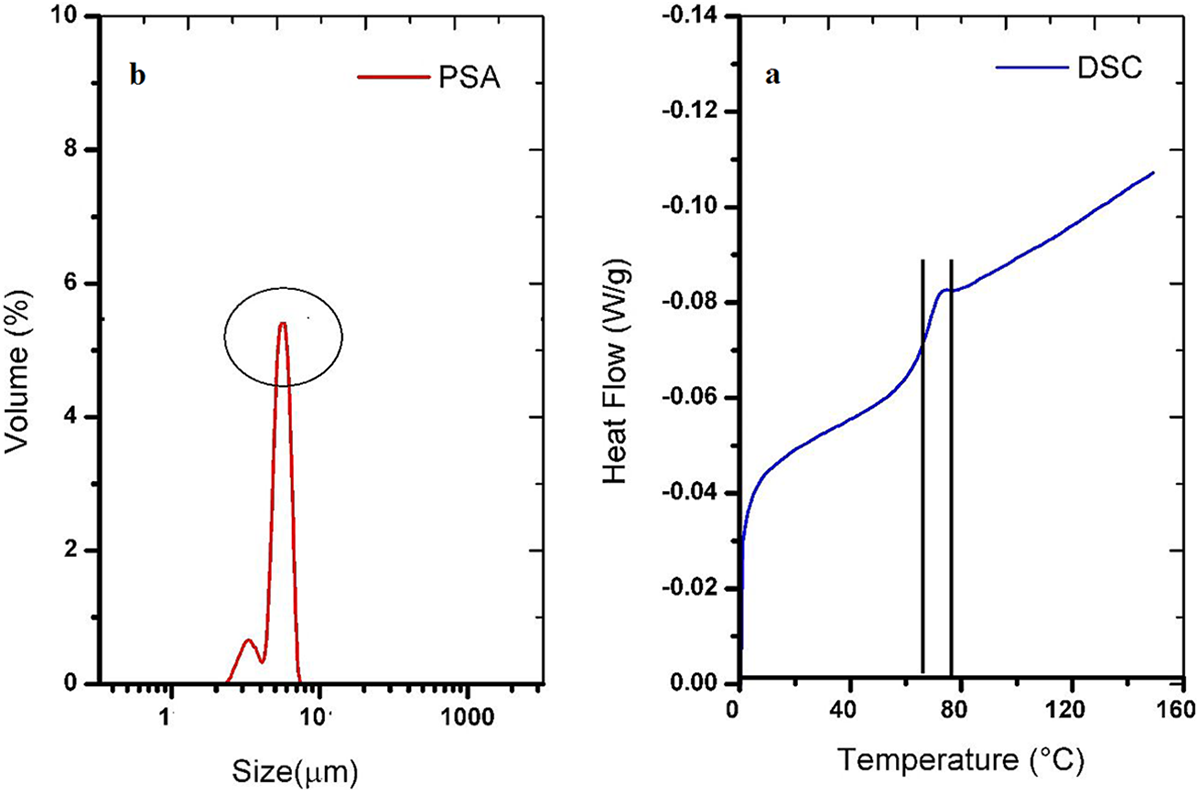
The DSC (a) and PSA (b) micrograph of composite toner.
EA method can produce toner with various ranges of particle size from 3 to 10 µm with a very narrow particle size distribution. 31 For creating images with high-resolution and uniformity in color, these two characteristics are all-important. Figure 7b shows the PSA measurements of the toner. The results indicate that the produced toner has an appropriate particle size and distribution for printing (9 µm).
The spectral characteristics of toner were measured and then converted into colorimetric coordinates L*, a*, b* using the CIE standard illuminant D65. An increase in the L* shows a higher lightness. The a* axis are green at one extremity −a* and red at the other +a*, while the b* axis has blue and yellow extremes. 32 Despite the change in lightness values, toner had a* and b* near zero, which offered the proper blackness of the samples which situate in the range of original toner.
The following logarithm mathematically represents the relationship between the quantities of light reflected from a stage along the ink film to the quantities of light falling on the same point which is named as the ink optical density (Eq.1):
where LW is the intensity of light reflected from the white region of the paper, and LR is the intensity of light reflected from the process ink region printed on the paper. 33
Broadly, the better the ink film causes the higher the optical density reading. The average optical density results for printed toner (0.9) were found to place in the range original printed toner (1.2).
Conclusions
Moved over the dearth of resources and environmental aspect, this study for the first time, investigated the possibility of applying the recycled magnetite particles from the waste of micro silica treatment in the concrete industry as a charge control agent in the printing industry. The obtained magnetite as a byproduct from the waste of micro silica treatment was applied as the charge control agent to produce magnetite/carbon black/styrene co-butyl acrylate composite microspheres by the green emulsion aggregation method which is used as toner in the printing procedure. XRD results show the presentation of Fe2O3, Fe3O4 or SiO2 in the recovered magnetite particles. VSM results showed that the recycled magnetite is super paramagnetic with Ms of ∼60 which is in the appropriate range as a charge control agent for printing toner. Produced toner had an applicable particle size (9 µm) with a spherical shape and appropriate thermal properties for printing.

