Abstract
This work is mainly concentrated on the preparation and characterization of poly(propylene oxide) (PPO)/organic clay nanocomposites. The effects of the nature of organoclays and the method of preparation have been elucidated to evaluate their morphological, structural and thermal properties. These nanocomposites were characterized by X-ray diffraction (XRD), Fourier-transform infrared spectroscopy, scanning electron microscopy, transmission electron microscopy, thermal analysis (differential scanning calorimetry) and thermogravimetric analysis (TGA). This study involved the use of an Algerian clay called ‘maghnite’ which was modified by an incorporation method using different concentrations of the surfactant cetyltrimethylammonium bromide (CTAB). Propylene oxide (PO) was polymerized in the presence of ethylene glycol and various proportions of organic clay ranging from 1%, 5% and 10% by weight. The structural study by XRD showed, at different loading rates of maghnite and PO in the preparation of PPO/maghnite nanocomposites, different morphologies: intercalated/exfoliated blend and intercalated/agglomerated. TGA showed that the intercalation of PPO at the montmorillonite (MMT)-CTA interface would be more advantageous since the required amount of 5% organic clay (by weight) ensures high thermal stability of the nanocomposites, and the glass transition temperature (
Introduction
Nanocomposites are a relatively new class of materials, which are defined by the particle size of the dispersed phase having dimension less than 100 nm. 1 Nanocomposites have attracted great interest as promising advanced materials due to their superior properties compared to conventional polymers2,3 such as density, high resistance to surface treatment, high modulus of elasticity, non-flammability and thermomechanical/optoelectronic/magnetic properties.4–6 Indeed, the use of a polymer matrix by adding a well-defined percentage of clay as reinforcement leads to the improvement of the physicochemical properties of the resulting nanocomposite and increases the biodegradability of the polymer.7–11 Nanoclay (NC) is a versatile, naturally occurring, layered structured nanoparticle of clay. They have a high surface area and possess high cation exchange capacity (CEC) which improves its interfacial interaction with the host polymer. NCs are potentially capable to increase the mechanical properties even at low content. Also, it can enhance the thermal properties of the polymer such as heat deflection temperature. 12 Their two-dimensional structures in lamellar form and their electronegative colloidal charge make them the compounds of choice for various organic molecules, including cationic surfactant molecules. Nanoparticles have a higher surface area/volume ratio improving interaction with host polymers and then imparting properties. Nanoparticles mainly phyllosilicates, oxides and ceramics have been experienced in polymeric nanocomposites. 13 The resulting hybrid materials are used in various fields such as adsorption, barrier applications and supports for the development of nanocomposites. 14 Polyether elastomers are used in various industries. These elastomers must have specific properties such as good behaviour at different temperatures and high mechanical properties. The most commercially important elastomers are amorphous homopolymers of epichlorohydrin, ethylene oxide (EO) and propylene oxide (PO). Poly(propylene oxide) (PPO) oligomers are used for a wide range of applications and are bulk base materials. 15 This work deals with the preparation, characterization and use of an Algerian clay called ‘maghnite’. We first deal with the modification of maghnite-Na, using different concentrations of cetyltrimethylammonium bromide (CTAB). The clays thus obtained were then used as green nano-reinforcements for the preparation of nanocomposites PPO/orgnaophilic clay. In this study, we investigated the effect of different loading levels of modified NCs of 1CEC and 2CEC to evaluate the structural, morphological and thermal properties of the prepared materials.16,17 The detailed thermal and morphological properties were studied to get a good overview of the main parameters that can be controlled to determine the structure of the nanocomposite and therefore the properties of the nanocomposite PPO/clay.
Experimental
Materials
PO (Reagent plus, ≥99%), ethylene glycol (EG) and CTAB have been supplied by Aldrich Chemical (sigma-Aldrich, Saint-louis, Missouri, USA) and have been used without further purification. Raw maghnite:montmorillonite (MMT) clay is used as a catalyst and comes from a local company (ENOF Maghnia, Western Algeria). Maghnite belonging to the MMT family has an interfoliar space of 10 Å in its raw state and a CEC = 90 meq/100 g. 18
Preparation of maghnite-Na+
Sodium ions (Na+) are frequently used as compensating ions due to their better hydration and promote the swelling and dispersion of MMT in water. A mass of raw maghnite (10 g) is introduced into a volume of distilled water (40 ml), this mixture is kept in suspension for 2 h under magnetic stirring and at room temperature. After 2 h, a volume (60 ml) of sodium chloride solution (NaCl, 0.2M) is poured into the previous mixture and placed under stirring for 48 h. Subsequently, the maghnite-Na+ is filtered and washed many times with distilled water to remove Cl− ions. After filtration, the maghnite-Na+ is dried in the oven for 24 h at a temperature of 105°C, then finely ground and stored in a dry place.19,20
Organic modification of clay
Cationic substitution takes place in an aqueous medium, since the swelling of maghnite in water promotes the insertion of alkylammonium ions within the interfacial galleries. A quantity of 10 ml of 1N hydrochloric acid was added to a 1-l volumetric flask, and the volume was then made up with distilled water. The resulting solution is then introduced into a conical flask equipped with a magnetic stirrer, and this acid solution is then brought to a temperature of 80°C for process optimization. When the temperature is stabilized, 10−2 mol of CTAB are added to the mixture. After 3 h of stirring at 80°C, the amine is then dissolved and ionized. We then integrated 5 g of sodium maghnite. After 3 h of cation exchange, the modified maghnite was collected and rinsed six times in a row with distilled water at 80°C to remove the mineral cations. The verification of the rinsing efficiency was done by adding a few drops of silver nitrate to the residue. The physisorbed alkylammonium ions had been extracted by a mixture of water previously heated to 75°C. The organophilic maghnite produced was dried at 120°C and crushed. This MMT is called maghnite-CTA throughout this study.20–22
Preparation of PPO/clay nanocomposites
Different PO-clay nanocomposites have been prepared using different amounts of modified clays corresponding to 1%, 5% and 10% by weight based on the initial amount of PO, and PO without clay has also been prepared under the same conditions as the standard. Each tube contained a given amount of EG (0.086 mol), PO (10 g, 0.172 mol) and an amount of maghnite-CTA (1%, 5%, 10% and 15%); the tubes were purged with nitrogen for 20 min and then the mixture was ultrasonically agitated for 4 h to ensure good dispersion of the clay in the PO. The polymerization is then carried out at room temperature for 24 h with a stirring of 400 r min−1 to obtain PPO nanocomposites (PPONC), and the nanocomposites will then be designated as PPONC1, PPONC3, PPONC5 and PPONC10 according to the clay they contain (Table 1).
Experimental conditions for the preparation of PPO/maghnite-organophilic nanocomposites by in situ polymerization.
PPO: poly(propylene oxide); CTA: cetyltrimethylammonium; CEC: cation exchange capacity.
Characterization of nanocomposites
Infrared spectra are plotted on a Perkin Elmer (FTIR, Digilab, Cambridge, MA, USA) IR TWO spectrophotometer instrumented with an ATR in the range 400–4000 cm−2. X-ray diffraction (XRD) analysis was performed using a Bruker (XRD, STADI P, Karlsruhe, Germany) D8 Advance X-ray diffractometer (40 kV, 30 mA) at room temperature. The different phase morphologies of the nanocomposites were determined using a Philips CM 120 (Tokyo, Japan) transmission electron microscope (TEM) with an accelerating voltage of 120 kV. The surface morphology of these elaborated materials has been examined by scanning electron microscopy (SEM) ‘JEOL 7001F, FEG-SEM’ (Tokyo, Japan). The various thermograms have been carried out on a differential scanning calorimetry (DSC) 204 F1 Phoenix. The thermal behaviour of PPO and PPO/maghnite nanocomposites was examined with the DSC (TA Q100 series Instruments, Tokyo, Japan). The thermal stability of the materials was achieved with Shimadzu TGA 51 (Tokyo, Japan) equipment, using an airflow rate of 50 ml min−1 and a heating rate of 10°C min−1.
Results and discussion
The Fourier-transform infrared (FTIR) spectra of nanocomposites obtained by mass polymerization contain both the characteristic absorption bands of PO and modified clay. The different characteristic bands and their allocations are grouped in Table 2.
Band assignments of FTIR spectra of PPO and PPONC.
FTIR: Fourier transform infrared; PPO: poly(propylene oxide); PPONC: PPO nanocomposites; CTAB: cetyltrimethylammonium bromide.
The FTIR spectra of pure PPO, organic clay and a nanocomposite sample are shown in Figure 1. PPO shows a large broadband of asymmetric –CH2 stretching between 3200 cm−1 and 2800 cm−1 with the two narrow bands at 2700 and 2680 cm−1. In the nanocomposite, the band broadens between 3100 cm−1 and 2870 cm−1, whereas the narrow peaks disappear. In this region, the characteristic peaks of PPO which appear at 2915.8 and 2861.61 cm−1 are shifted to 2850 and 2936 cm−1 (Table 2). The band at 3600 cm−1 characterizing the free OH groups in the octahedral layers of the Mag-CTA disappears, indicating the possibility of hydrogen bond formation with PPO chains and a high attenuation of the OH group absorption bands of water molecules (at 3400 cm−1) indicating the replacement of water molecules by CTA molecules. In the region of 1300–900 cm−1, in particular the 1141.37 cm−1 peak of PPO which is for asymmetrical stretching of the C–O–C groups widens, while the 1024.48 cm−1 peak moves at a lower frequency of 951 cm−1 (Figure 1). 23 Through these characteristic absorption bands, it can be deduced that the prepared nanocomposites confirm the incorporation of the clay into the PPO matrices.
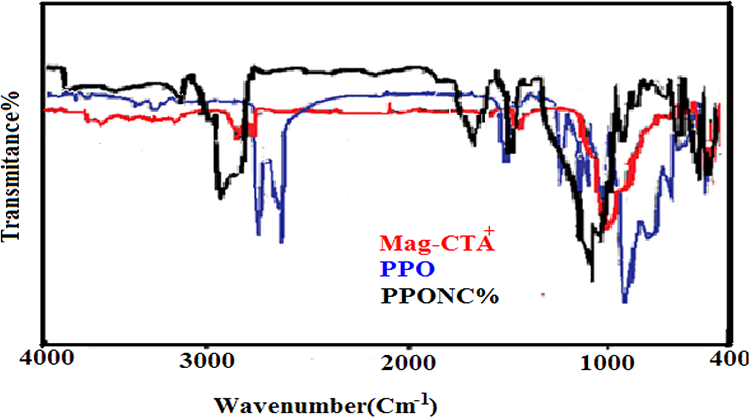
FTIR spectra of CTA-Mag, PPO/clay nanocomposite containing 5 wt% clay and pure PPO.
The XRD concretely realized reveals an insertion of the alkyl chains of the surfactant ‘CTA’ in the maghnite galleries. In fact, for a concentration level of 1CEC, the interfoliar distance of Mag-Na+ increased from 12.27 Å to 18.16 Å for Mag-CTA, corresponding to a ‘bilayer’ organization. Nevertheless, for a concentration of 2CEC, a bimodal profile for Mag-CTA was recorded, centred on two values, 19.27 Å and 33.53 Å (Figure 2(a)). The higher relative intensity of the peaks in the plane (001) results from a narrower distribution of interlamellar spacing due to the incorporation of CTA ions into clay sheets. 24
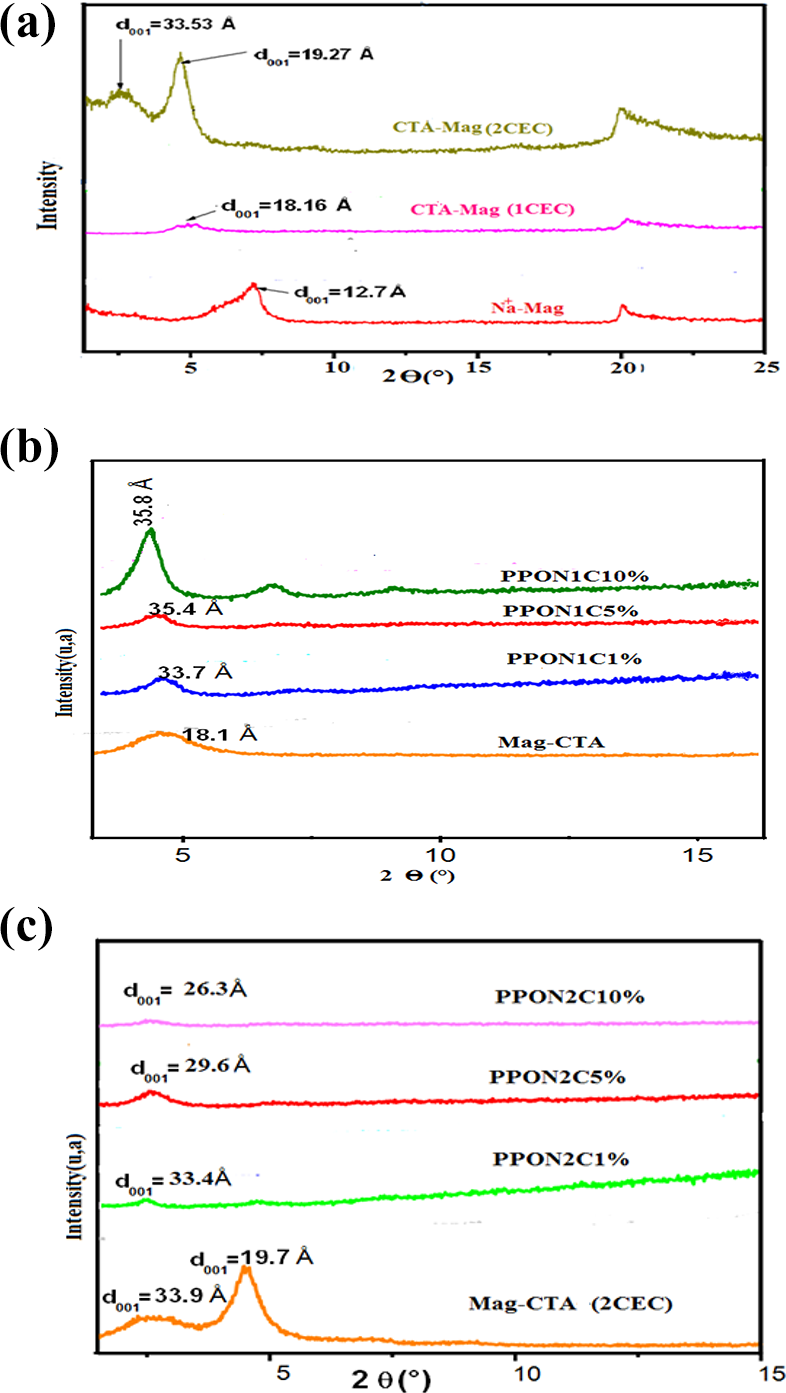
XRD patterns of the (a) maghnite-Na+, maghnite-CTA (1CEC) and maghnite-CTA (2CEC), (b) PPO/CTA-Mag (1CEC) nanocomposites and (c) PPO/CTA-Mag (2CEC) nanocomposites.
The realization of the nanocomposites by Mag-CTA (1CEC) revealed the displacement of the basal peak
Morphology of nanocomposites PPO/Mag-CTA+ (1CEC).

PPO: poly(propylene oxide); CTA: cetyltrimethylammonium; CEC: cation exchange capacity.
In the case of the PPO/Mag-CTA (2CEC) sample, the basal spacing of the clay is between 36 Å and 29.3 Å, respectively, as the diffraction angle shifts from 2
Morphology of PPO/Mag-CTA+ (2CEC) nanocomposite.

PPO: poly(propylene oxide); CTA: cetyltrimethylammonium; CEC: cation exchange capacity.
Figure 3(a) shows the TEM images of the various PPONC elaborated. These observations suggest that the fillers were mostly homogeneously distributed in the PPO matrix. Regardless of the filler, the inorganic layers are parallel to the surface, mainly well distributed in the PO matrix. The intercalated/exfoliated blend of PPO/Mag-CTA clay-containing nanocomposites is fully confirmed for PPO/Mag-CTA (1CEC) nanocomposites at different weight percentage rates. The TEM image of the PPON1C% nanocomposite shows an intercalated morphology that corresponds to the results obtained by XRD in the presence of tactoids (Figure 3(a)).

TEM images of (a) Mag-CTA and PPON1C% nanocomposites and (b) Mag-CTA and PPON2C% nanocomposites.
Figure 3(b) of the nanocomposite PPON2C5% and PPON2C10% shows a degree of disorder in the morphology of the clay. The dark lines correspond to the silicate monolayers and the silicate nanoplaquettes are oriented more or less parallel to each other but are separated enough so that they do not interact. The TEM image of PPON2C5% and PPON2C5% nanocomposite shows an intercalated and agglomerated morphology, which corresponds to the results found by XRD with the presence of small tactoids. We note the presence of some aggregates (Figure 3(b)).
Figure 4(a) describes that the more organoclay dispersed in the PPO matrix increases the more irregular and porous appearance of the nanocomposites. Micrographs of the PPON1C% nanocomposite show the presence of tortuous crack propagation lines indicating a porous appearance. It has been traced in the literature that when silicate polymers are well dispersed, many non-linear cracks are built and tend to evaluate themselves until they interfere with each other. 25 The stresses at the ends of the fracture lines interact with each other and hinder the growth of these stresses. Since the strength of the polymeric material is closely related to crack formation at the molecular level, the more numerous these tortuous cracks are during the failure process, the more energy will have to be absorbed to damage the material (Figure 4(a)).

SEM images of (a) Mag-CTA (1CEC) and PPON1C% nanocomposites and (b) Mag-CTA (2CEC) and PPON2C% nanocomposites.
Figure 4(b) illustrates the surface morphology of the PPO/CTA-Mag (2CEC) nanocomposite sample, this latter has a conventional alloy where the dispersed MMT-CTA particles are well anchored to the PPO matrix, even it reveals the presence of clay entities in the form of stacked sheets or aggregates well dispersed in the PPO matrix. For the 5% and 10% loading rate, the existence of large aggregates, the number and size of aggregates observed at high loading rates increases and these composite structures appear to be dependent on the loading rate. Figure 4(b) shows a characteristic morphology consisting of dense non-woven micro agglomerates, the topography is heterogeneous. A thickening of the sample on 50 μm shows a more porous structure. The SEM image of the nanocomposite PPON2C5% and PPON2C10% shows the presence of large tactoids.
The DSC analysis was performed to study the influence of the loading rate on the
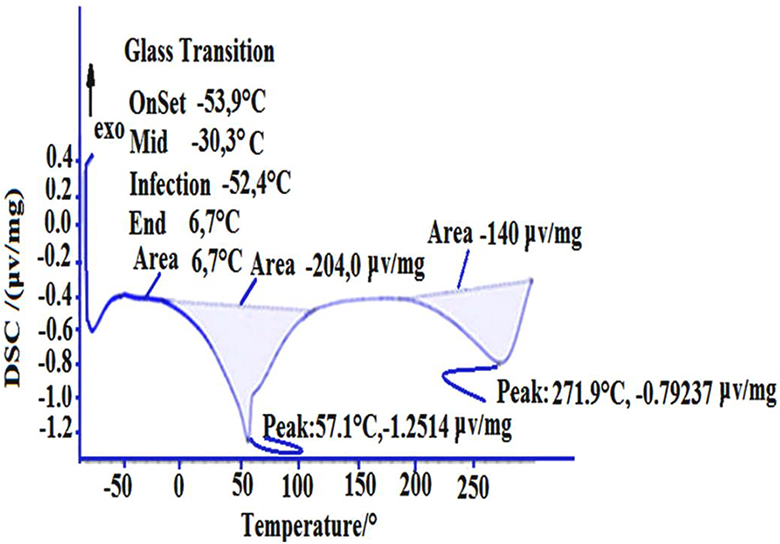
DSC for PPONC (5 wt%).
The thermal stability of the PPO/Mag-CTA nanocomposites was investigated by TGA. Figure 6 shows the thermograms of PPO/Mag-CTA and the pure PPO compound. Thermal degradation of the PPO polymer occurs over a fairly narrow temperature range, with the onset of decomposition at 140°C. It is important to note that the trace of TGA recorded on the PPO/Mag-CTA material shows that the onset of PPO decomposition is about 30°C higher and that the rate of degradation of the polymer matrix in this nanocomposite structure is significantly reduced. At about 265°C, the weight loss is quantitative. In the case of the intercalated PPO/Mag-CTA sample, the nanocomposites show a degradation temperature of about 200°C, while the intercalated composites/intercalated aggregates reach a degradation temperature of about 265°C (Figure 6). The results obtained demonstrated a gain in the stability that is due to the formation of a charred protective layer; in addition, the amount of organic clay required to prepare a nanocomposite PPO/Mag-CTA (1CEC) with high thermal stability is 5% (by weight). The formation of this layer is stimulated by the fine dispersion of interspersed MMT particles that act as an inorganic support. 26
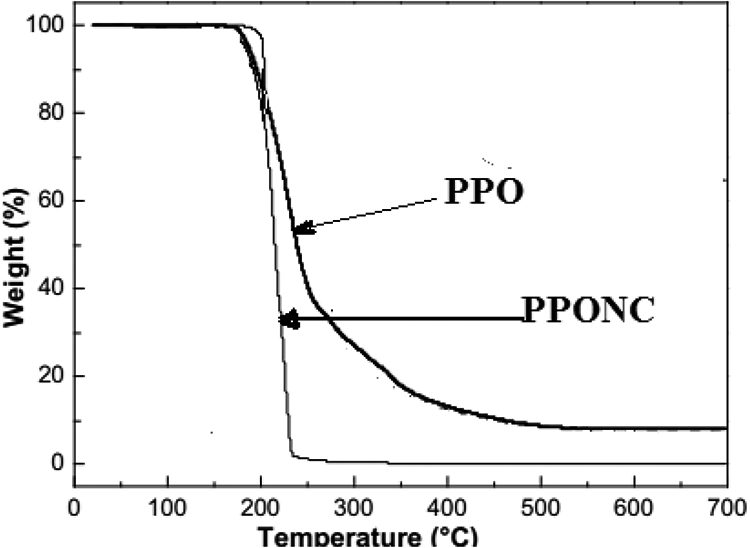
Thermogrammes (TGA) of PPO and PPONC.
Conclusion
The PPO/maghnite nanocomposites were synthesized on the basis of an organophilic clay by mass intercalation. XRD analyses of the PPO/Mag-CTA (1CEC) nanocomposites showed that the majority of the obtained nanocomposites have an intercalated/exfoliated blend morphology resulting in an expansion of the space between the layers, and the TEM and SEM images obtained are consistent with the XRD data. The IR spectroscopic study shows the incorporation of the filler within the polymer matrix. PPO/Mag-CTA (2CEC) composites based on maghnite-CTA were also obtained. The structural study by XRD showed at different loading rates and different amounts of PPO in the preparation of PPO/Mag composites, the existence of different morphologies, intercalated and agglomerated, and the TEM and SEM images obtained are compatible with the XRD data. Thermogravimetry results show that composites have higher thermal stability than pure polymer PPO. This is due to the interactions that come into play at the level of all the polymer chains and the organic compounds of the modified clays. The DSC analysis revealed a slight increase in the
