Abstract
In this study, 5%, 10%, and 15% vermiculite was added to the rigid polyurethane (PU) structure as a fire retardant. The scanning electron microscopy (SEM), energy-dispersive X-ray spectroscopy (EDS), X-ray diffraction (XRD), limiting oxygen index, thermal gravimetric analysis, differential scanning calorimetric (DSC) curves, and combustion behavior of the composite materials were investigated. When SEM/EDS analyses were examined, it was observed that surface morphology was affected by the amount of additive material. When XRD analyses were examined, peaks appeared in three different groups. Thermogravimetric analysis was used to determine the thermal stability of PU material with the addition of vermiculite. When the DSC analysis was examined, four temperature zones were obtained. The combustion behaviors of the vermiculite added PU samples with single-flame source and the flammability classes were determined with TS EN ISO 11925-2 standard. It can be said that the usage of inorganic mineral, which is harmless to the environment, can enhance the thermal stability of the material and also reduce the cost of production.
Introduction
Polyurethane (PU), which found by Otto Bayer and colleagues in 1937, is a polymer composed of a chain of organic units including urethane linkages. 1 Polyols and isocyanates are the basic materials for the production of PU. The properties of PUs are affected by the production technology and the type and the amount of chemicals.2,3 Nowadays, PUs are available in various forms, such as thermoplastic PU, flexible PU, rigid PU, and water-borne PU.4–6 Industrially produced PUs are used in construction, automotive, marine, sports, furniture, leather, textile, shoes, and adhesive furniture applications.7–13
PUs used in the industry have some common advantages and disadvantages. The most common advantages are excellent thermal insulation; thanks to closed-cell structure, low thermal conductivity (λ = 0.03–0.04 W/mK) owing to swelling agent trapped inside the cells and low density. 14 Also, they are the most efficient insulation material with the lowest thickness. So, these kinds of materials have a wide range of usage in industrial applications.15–17 Main disadvantages are low thermal decomposition and combustion resistance14,18–20 and poor mechanical strength. Studies reported that the mechanical strength can be eliminated by reducing the hollows in the PU 21 and the low combustion resistance can be increased with different additives (boron, borax, boric acid, clay, calcite, ammonium polyphosphate, and so on).22–29
Vermiculite which is obtained from volcanic magma sources is a layered silicate mineral.30,31 The volume of the vermiculite expands (up to 20–30 times), permeability increases, and density is significantly decreased (from 0.8 g/cm3 to 0.08 g/cm3) when exposed to high heat source. The decrease of the density is due to the quality of the vermiculite and the properties of the heater. 32 The structure of the material seems as exfoliated after the expansion. 33 The gas permeability of the material is decreased when the vermiculite was used as a filler. 34 The expanded vermiculite (EV) has high resistance to flame (1270–1370°C) and good heat and sound isolation. It is a promising material for the application in the field of energy-efficient buildings. 35 Vermiculite has advantages, such as lightweight, sterile, dry, compressible, high liquid absorption, thermal insulation, high anion–cation exchange, high capacity of air, and 6–9 pH value. Vermiculite is generally used in agriculture applications, protection of fire, production of the nanocomposites, and construction and packaging materials. 36 There are restricted studies in the literature about the vermiculite-reinforced PUs.37–39
A considerable amount of the literature has been published on rigid PU and additives/fillers. Xia and Wang 40 studied the effects of EV and melamine phenylphosphate (MPP) on thermal, combustion, and mechanical properties of the PU material. They added EV as the amount of 5–20 wt%. The addition of vermiculite increased the compressive and flexural strength ratio as 82% and 115%, respectively. They have reported that vermiculite enhanced the combustion behaviors of the PU. In addition, they have used MPP as flame retardant with EV and found that MPP has good synergistic effect in the view of thermal and combustion properties. Agrawal et al. 41 investigated the combustion, thermal, and mechanical properties of PU reinforced with feldspar and kaolinite clay. They have reported that the mechanical properties of the material significantly increased with the mineral fillers. The experimental results showed that thermal properties enhanced and combustion resistance increased with these two kinds of minerals. Wang et al. 42 evaluated the effects of zeolite on thermal, mechanical, and combustion behaviors of PU foam. The ratio of zeolite addition was given as 0–9 wt% into the rigid PU foam. It was found that compressive stress increased with increasing of zeolite addition. Similarly, thermal conductivity increased with zeolite addition. In addition, thermal resistance of the PU increased with the addition of zeolite. Shang et al. 43 investigated the effects of cuttlebone (CB) on the structure, thermal, and mechanical properties of the PU material. CB and PU showed great adhesion. In addition, mechanical and thermal properties of the PU were well improved with the addition of CB. Aydoğan and Usta compared the thermal and combustion behaviors of rigid PU foam reinforced with nanoclay. They added nanoclay and intumescent flame retardant with various ratios. Thermogravimetric analysis (TGA) and cone calorimeter tests were performed to investigate the thermal and combustion behaviors. It was found that nanoclay and intumescent flame retardant have great synergistic effect. Thermal degradation temperatures increased with the addition of nanoclay and degradation rates decreased. Cone calorimeter tests showed that nanoclay and intumescent flame retardant addition significantly increased combustion resistance of the foam. 44 Aydoğan and Usta investigated the effects of calcite on thermal and combustion behaviors of rigid PU foam in their other study. They added the calcite as the amount of 5–15 wt% as a filler. They performed TGAs, thermal conductivity, and UL94 tests. In addition, ammonium polyphosphate and pentaerythritol were used as an intumescent flame retardant. The results showed that thermal resistance of the rigid PU increased with calcite addition. Thermal conductivity slightly decreased with calcite but it increased by time. They found that calcite has no positive effect on combustion resistance, however, using calcite and intumescent flame retardant enhanced the combustion resistance. 45 Wu et al. 46 investigated the effects of silica (SiO2) on the thermal and mechanical behaviors of the ethylene–octene copolymer/polypropylene composites. They have reported that the addition of 2 wt% SiO2 showed the highest resistance to thermal degradation. However, the best mechanical properties were obtained for 3 wt% SiO2 addition.
To the best of the author’s knowledge, there are very limited studies about vermiculite in the view of thermal properties. In this study, thermal and fire behaviors and the structure of the PU foams added vermiculite were investigated. Vermiculite was added into the PU foam as 5, 10, and 15 wt%. The scanning electron microscopy (SEM) analyses, X-ray diffraction (XRD), limiting oxygen index (LOI), TGA, differential scanning calorimeter (DSC) analyses, and vertical flame tests were performed and compared to neat PU foam.
Experimental
Materials
Rigid PU (polyol-senta 77/HC, isocyanate-senta 77/SG) raw materials were obtained from OSA Kimya, Turkey. The density of the polyol at 25°C is 1.00–1.15 g/cm3. The density of the isocyanate at 25°C is 1.10–1.20 g/cm3. The cream time and gelling time at 22°C are 40 and 100 s, respectively. Vermiculite used in the experiments was provided by Organik Madencilik Corporation in Sivas-Yıldızeli region, Turkey. The properties of vermiculite are given in Table 1.
Properties of vermiculite.

LBD: Loose Bulk Density (kg/m3).
Preparation of rigid PU
Two-component production was applied with polyol and isocyanate. The 1:1 mixture ratio was used while PU production. Vermiculite was added to the total mass by 5, 10, and 15%. Sufficient mixing time was provided in the manufacturing process to obtain a homogeneous mixture after the addition of the additive material into the polyol. Then, the isocyanate was added and poured into the silicone molds after mixing (1000 rpm, 60 s). Samples that were kept in the mold for 2 h curing period and removed from the mold. And then, the samples were kept in the laboratory conditions to complete the curing process for 48 h. The pictures of the produced PUs are shown in Figure 1. The abbreviations and the contents of the samples are given in Table 2.

The pictures of the produced polyurethanes: (a) Pure, (b) VMT05, (c) VMT10, and (d) VMT15.
Nomenclature of samples.

Characterization
The morphology of PU and the approximate content of each element were carried out with a Thermo Scientific™ Quanta™ (FEG-250) model SEM/energy-dispersive X-ray spectroscopy (EDS).The XRD was carried out using a Bruker XRD system (AXS D8 Advance) X-ray diffractometer in the Mehmet Akif Ersoy University central laboratory. The LOI test was conducted with YY 907 type oxygen index test instrument. LOI test was performed according to ASTM D 2863 standard. TGA (differential thermal analyzer (DTA)) was conducted with Seiko SII TG/DTA 7200 in nitrogen and the samples were heated to 20–800°C at a heating rate of 10°C /min. DSC analysis was performed using a Perkin Elmer DSC 400 tester in the Mehmet Akif Ersoy University central laboratory. The process was carried out at 20–300°C. The vertical burning flame propagation test was carried out with TS EN ISO 11925-2 standard.
Results and discussion
Surface morphology and composition analysis
SEM images were used to determine the surface morphology of rigid PU added vermiculite. The SEM images of VMT05, VMT10, VMT15, and Pure are shown in Figure 2. Vermiculites have an exfoliated structure. A coating was formed on the surface of the PU with vermiculite particles. Therefore, it had an agglomerated appearance and a hollow structure in the surface. As the rate of addition was increased, the formation of hollows in the structure markedly increased. 33 EDS spectra of the samples are given in Table 3. When Table 3 was examined, no impurity atoms were detected in the structure, and the given values were only for vermiculite. 47
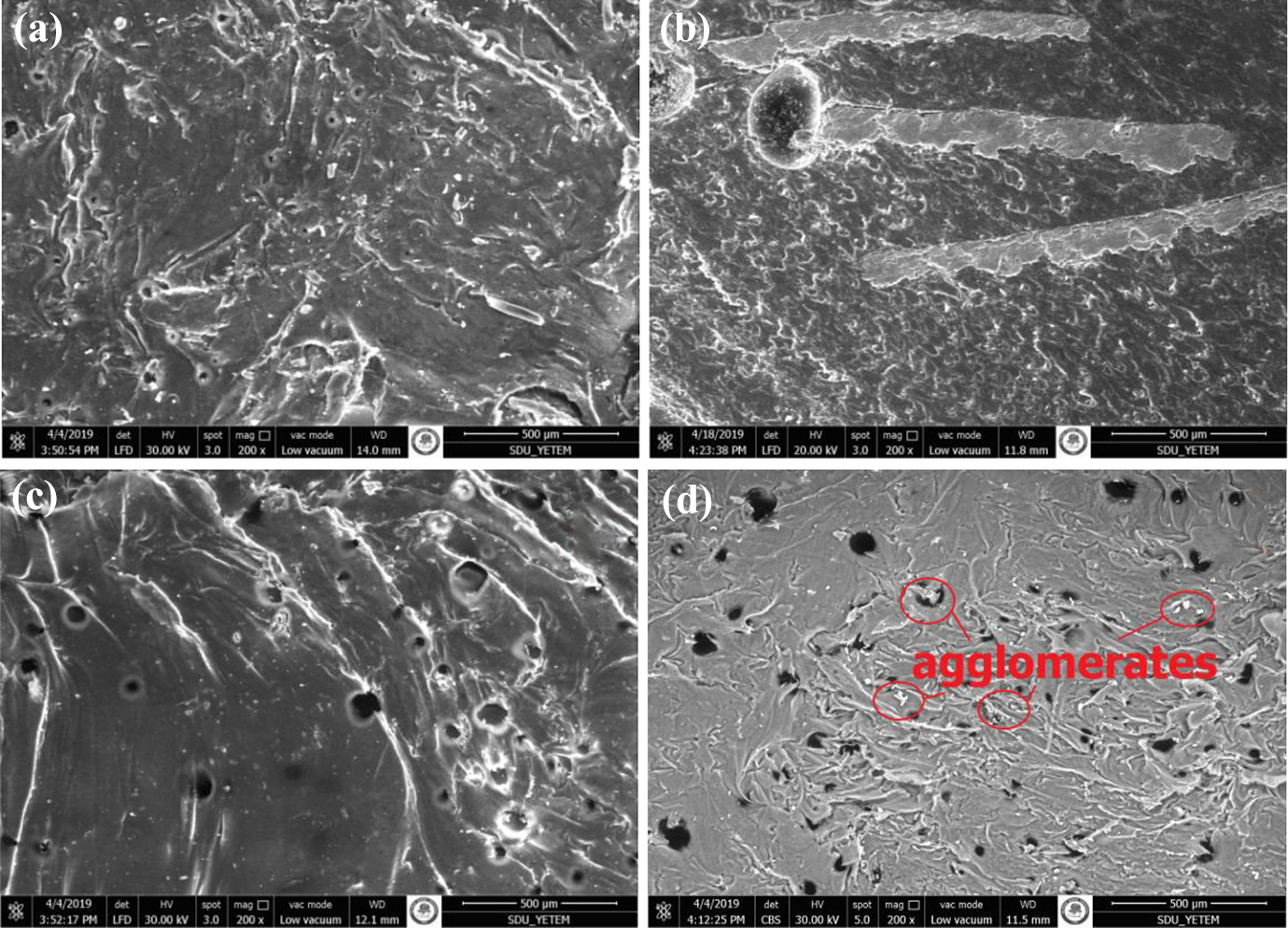
The SEM images of (a) pure, (b) VMT05, (c) VMT10, and (d) VMT15.
EDS results of pure and vermiculite added polyurethane.
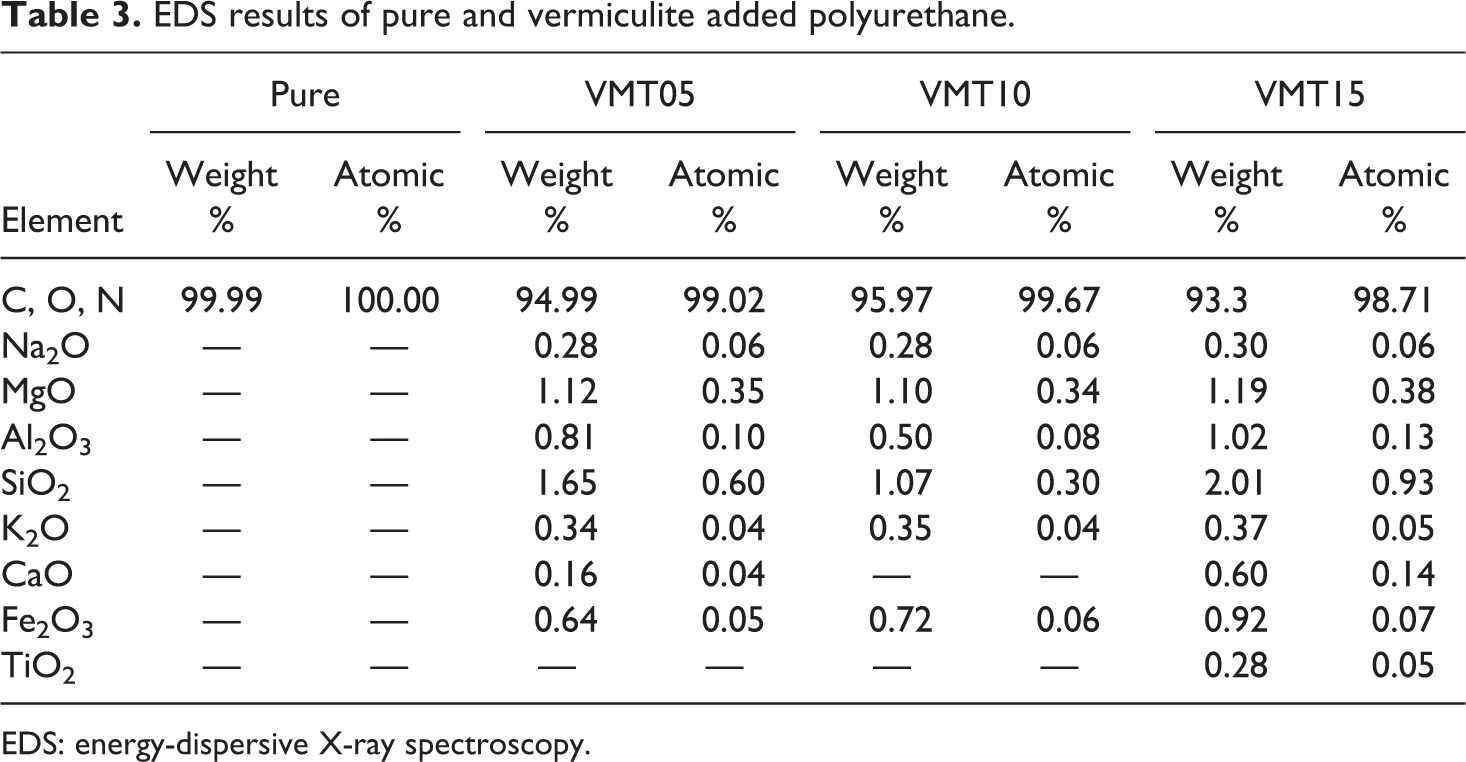
EDS: energy-dispersive X-ray spectroscopy.
Structural analysis
XRD results of vermiculite added PUs are shown in Figure 3. The peak value for all samples was obtained in the 2θ = 19.5° position. The peaks obtained at 2θ = 28.8°, 34.3°, 36.9°, and 60.3° positions by the addition are characteristic peak points of vermiculite.33,48 When the amount of vermiculite was increased, the intensity of the peaks increased. This situation may be explained with the transition from amorphous structure to crystal structure. Ercenk has identified these peaks as spinel (19.5°), fluorphlogopite (28.8°), and phlogopite (34.3°) phases. The peak values obtained are consistent with the literature. 49
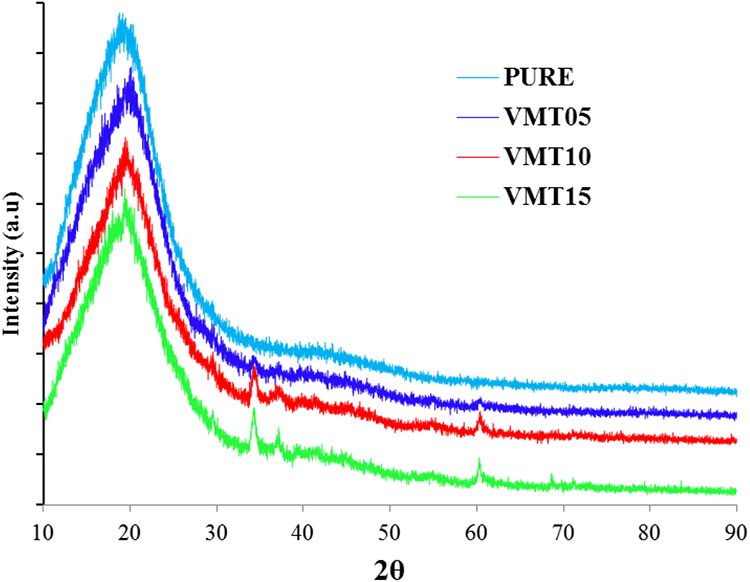
The X-ray diffraction patterns of pure and vermiculite added polyurethane.
Thermogravimetric analysis
TGA measurements are used to summarize the combustion behavior and thermal stability of polymer-based materials.50,51 Thermal decomposition starts from the weakest link forming the PU. The TGA curves and the detailed results of TGA analyses were given in Figure 4 and Table 4, respectively. Decomposition temperature increases with increasing the amount of vermiculite into the PU foam. The initial decomposition temperatures of pure and VMT15 were 80°C and 92°C, respectively. This phenomena can be attributed to the high decomposition resistance of the vermiculite. 52 When the Figure 4 was mentioned that T5 wt% and T50 wt% temperatures increased with the increasing amount of vermiculite into the PU. T50 wt% which is mentioned as the main mass loss temperature, increased about 32°C for the addition of 15 wt% when compared to pure. Yu et al reported a similar increase in their study with the addition of boron nitride into the epoxy resin. 53 Vermiculite has a layered structure, which prevents heat transfer. Heat release is decreased owing to this layered structure. In addition, vermiculite particles act like a barrier, which hinder leaving the volatile gases from the material and retard the decomposition processes. 54
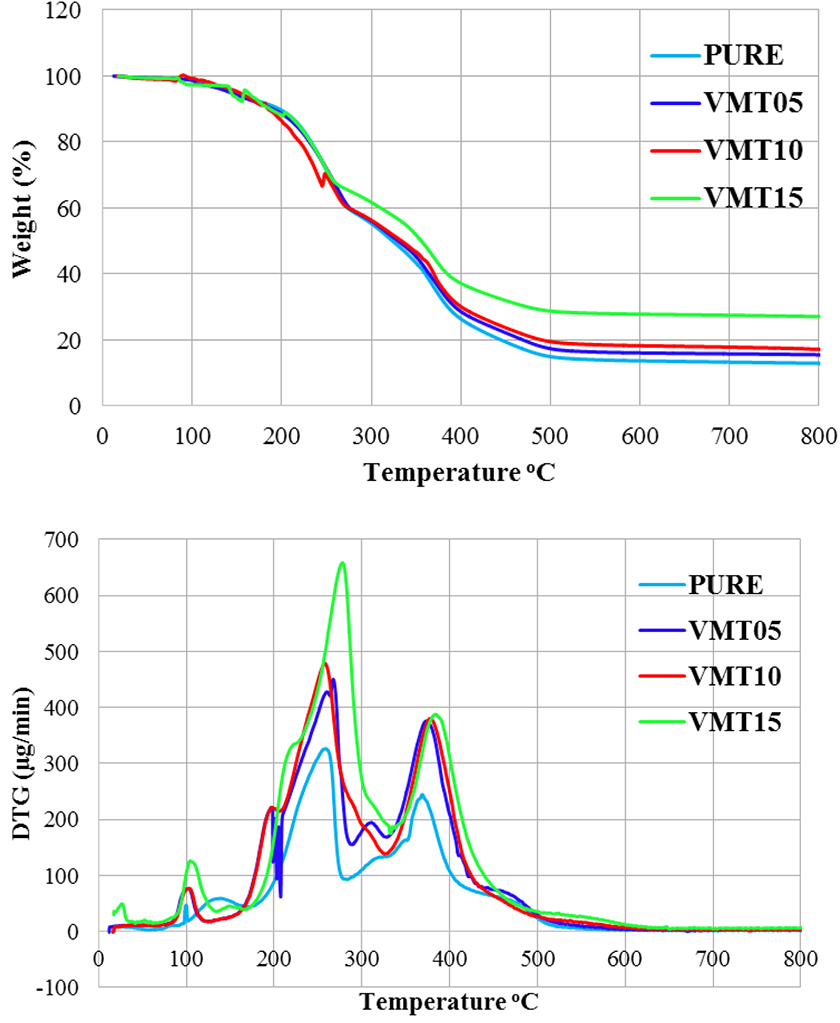
TGA-DTG diagram of pure and VMT added polyurethane.
TGA-DTG data for pure and VMT added polyurethane.
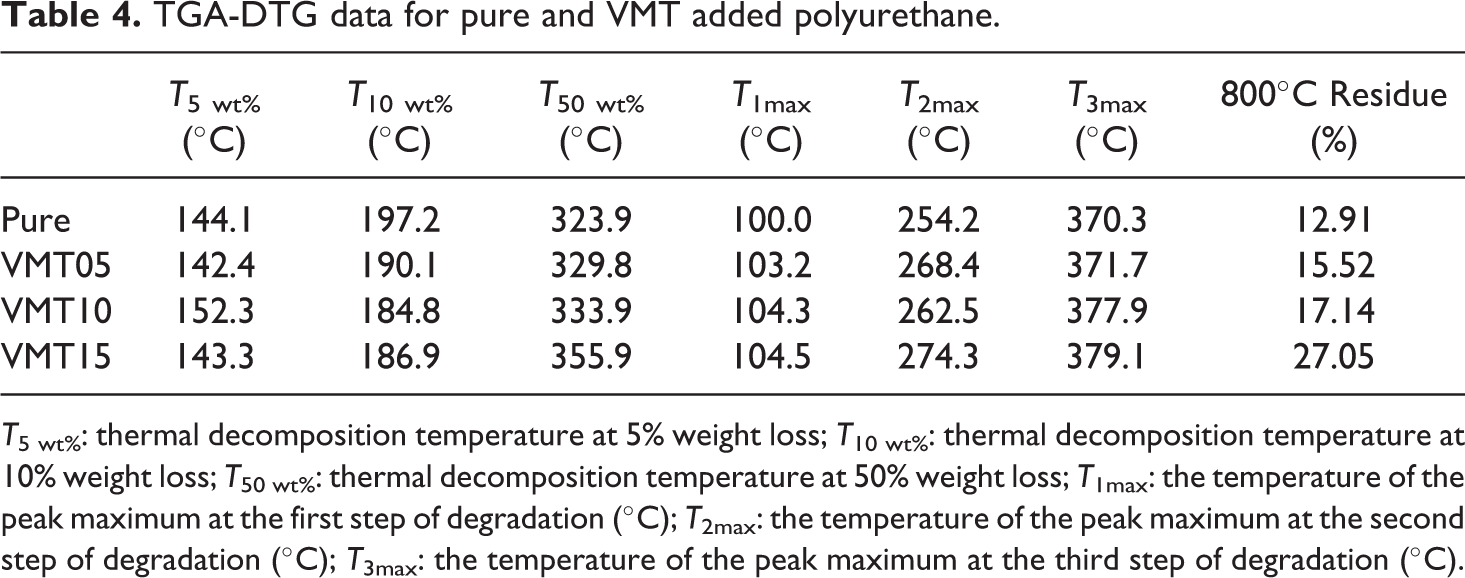
T5 wt%: thermal decomposition temperature at 5% weight loss; T10 wt%: thermal decomposition temperature at 10% weight loss; T50 wt%: thermal decomposition temperature at 50% weight loss; T1max: the temperature of the peak maximum at the first step of degradation (°C); T2max: the temperature of the peak maximum at the second step of degradation (°C); T3max: the temperature of the peak maximum at the third step of degradation (°C).
Figure 4 shows that three decomposition processes, which were between 80°C and 125°C, 190°C and 300°C, 320°C and 450°C for pure, VMT05, VMT10, and VMT15, respectively, were determined. The maximum temperature of the first decomposition step (T1max) of pure was 100°C and it was increased with the increase of vermiculite addition into the PU and the corresponding value was obtained as 104.5°C for VMT15. This may be explained with the decomposition of the weakest links and the evaporation of the structural water. The maximum temperature of the second decomposition step (T2max) was obtained as 254.2°C for pure. T2max increased about 26°C with the addition of 15 wt% vermiculite into the PU. The decomposition of isocyanate and the dissociation of the alcohols occur in the second decomposition step. 55 The third decomposition step proceeds slower and the decomposition of the hard segments occurs. 56 The maximum temperature of the third decomposition step (T3max) was 367.3°C and 380.8°C for pure and VMT15, respectively. This situation may also be attributed to the high thermal stability of the vermiculite. The carbon residues at 800°C increased with increasing the amount of vermiculite and were 12.91% and 27.05% for pure and VMT15, respectively. Shi et al. had similar results for char residue at TGA when they increased the amount of the additive. 57
To further understand the effect of VMT on the thermal stability of the PU, it is assumed that the RCcal (calculated residue char at 800°C) of pure/VMT added PU follows the linear mixing rule, as shown in equation (1) 57
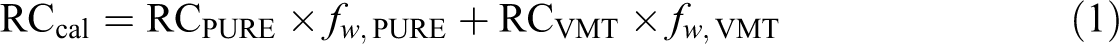
where RCpure and RCVMT show the residual yield of pure and VMT added PU, respectively, and fw,pure and fw,VMT, respectively, represent the weight fraction of pure and VMT added PU.
The char yield of pure and VMT added PU is presented in Table 5. According to Table 5, char residue VMT15 increased 27.05 wt% with respect to 12.91 wt% pure. The experimental char residue (RCepx) of VMT added PUs is higher than the calculated char residue (RCcal). The magnitude of increase in the RCepx relative to RCcal (ΔRC) is between 9% and 60%. The obtained values show that the presence of VMT considerably raised the thermal stability and charring capability of PU. Huang et al. 58 reported the enhancement in charring capability that contributes to reducing the release of flammable gas and heat during combustion, and in this way increasing the flame retardancy of PU.
RC data for pure and VMT added PU.
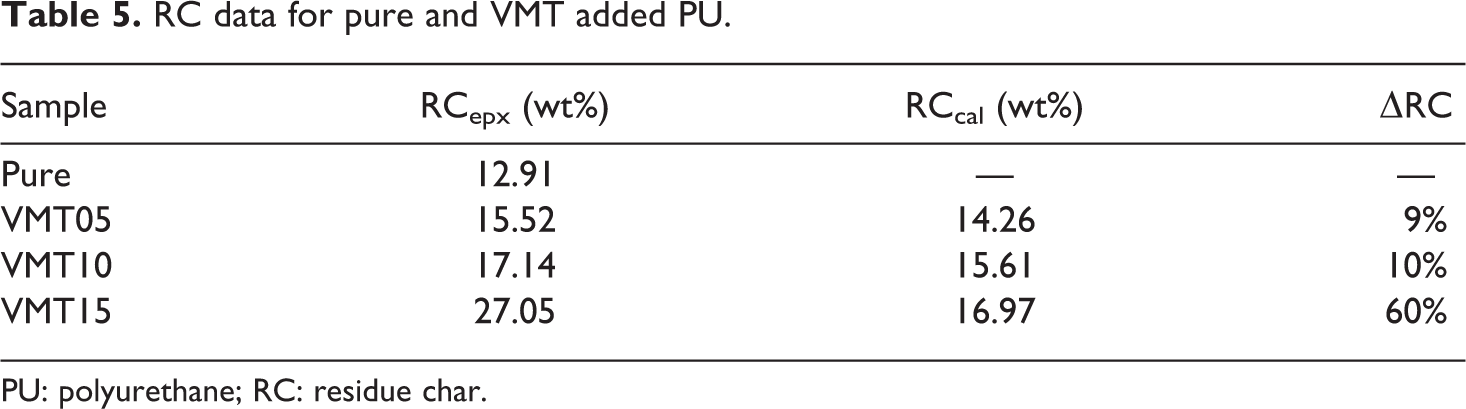
PU: polyurethane; RC: residue char.
The residue is obtained at 800°C. RCepx and RCcal, respectively, refer to experimental values and calculated ones of residue chars, and ΔRC means the magnitude of increase in the RCepx relative to RCcal.
Differential scanning calorimeter
When DSC thermal behavior was examined, four temperature zones were obtained. This temperature value increases with the addition of vermiculite minerals. As seen from Figure 5, vermiculite acted as an active flame retardant. There was an increase in peak values as seen from Table 6, hence, the material has become more endothermic. There has been a temperature shift with the addition of vermiculite as an additive compared to pure PU. This situation may be explained with the fire resistance of the vermiculite. 59
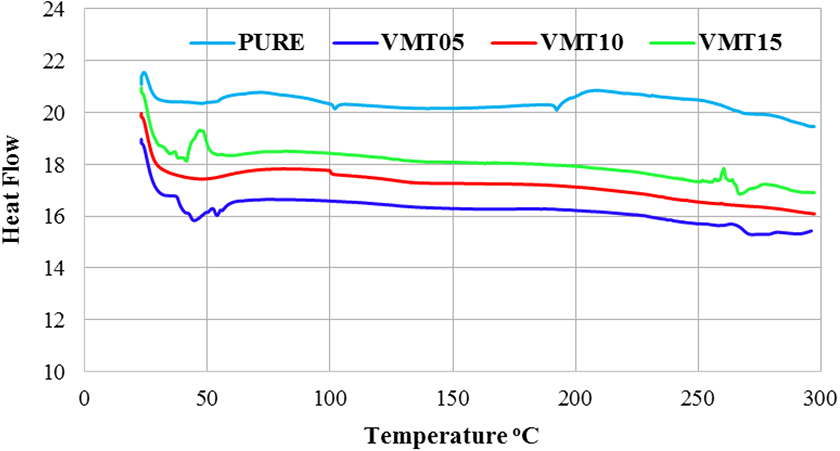
DSC curves of pure and VMT added polyurethane.
DSC data for pure and VMT added polyurethane.
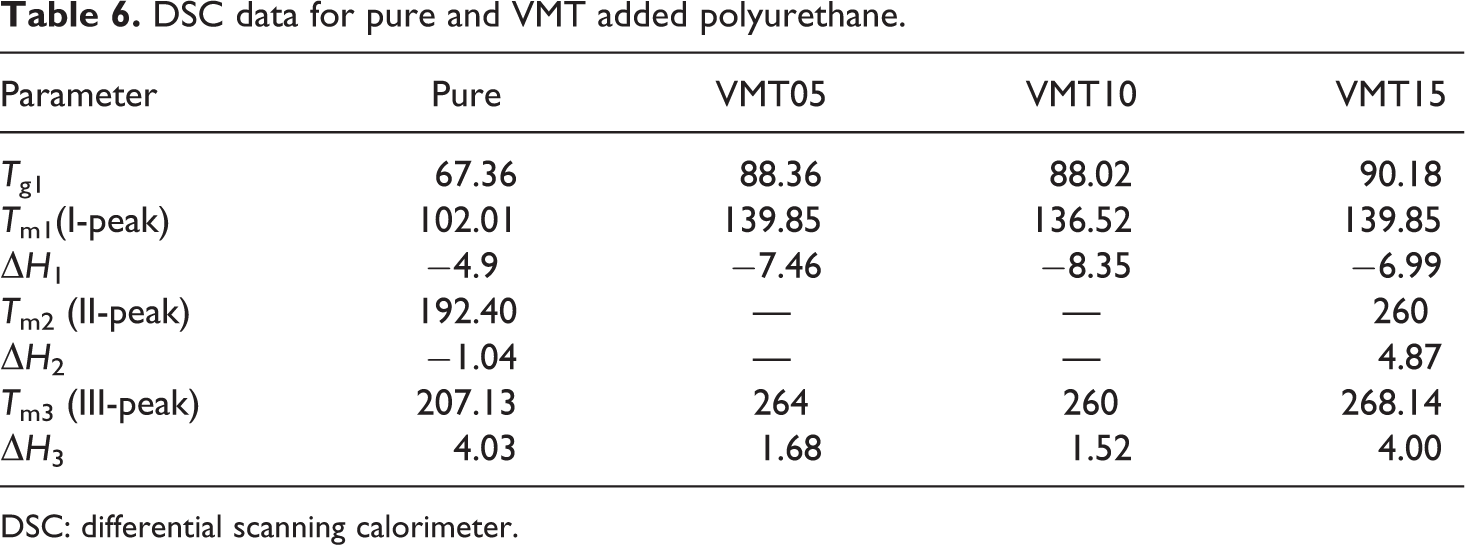
DSC: differential scanning calorimeter.
Vertical burning
The vertical burning tests were carried out according to the standard TS EN ISO 11925-2. Samples prepared at 90 × 250 mm dimensions were exposed to a single source flame at 45° angle for 30 s (total test time 60 s). The vertical burning images of pure and vermiculite added PU samples are shown in Figure 6.

Vertical burning images of pure and VMT added polyurethane: (a) pure, (b) VMT05, (c) VMT10, and (d) VMT15.
It can be seen from Figure 6 that sample integrity was not disturbed and the combustion progression did not reach to 150 mm in all samples. In the pure sample, the combustion proceeds to the depths, and the surface integrity of the sample is impaired. On the surfaces of vermiculite-added samples, it was observed that the mineral absorbs heat, the sample surface retains its form, and the flame does not advance. The flammability class of the samples added vermiculite was determined as D according to TS EN ISO 11925-2 standard.
Limiting oxygen index
Every material needs different oxygen fraction to continue burning. The minimum oxygen necessity is named as LOI. The materials have LOI value of below 21% in which the atmosphere contains easily burn. On the contrary, the materials that have LOI value of above 21% cannot burn until it reaches this value. LOI test is usually used to evaluate the flammability of materials, especially for determining the effects of the flame retardants in the materials. 60 The LOI analyses of the PU materials added vermiculite were performed according to the ASTM D 2863 standard and the results were given in Table 7. The LOI values of the samples differ between 18% and 24.3%. The flame resistance of the sample which contains 15% vermiculite was increased about 35% when compared to pure PU. This situation may be explained with that the vermiculite addition into the material can act as a physical barrier and prevent oxygen penetration. The low permeability coefficients for all gases and the aspect ratio of silicate layers reduce the possibility of more combustion. 61
Limiting oxygen index of VMT-doped PU.

PU: polyurethane.
Conclusion
In this article, 5%, 10%, and 15% vermiculite was added into the rigid PU to determine the internal structure, thermal and combustion resistance of the materials. The results are listed as follows: According to the analysis of the SEM images, a coating was formed on the surface of the PU. It was found that there had been agglomerated appearance and a hollow structure on the surface. The hollowed structure has increased with the increasing of vermiculite addition. The XRD graph of the pure rigid PU sample shows an amorphous structure. As the amount of vermiculite addition was increased, the intensity of the peaks increased. This situation may be explained with the transition from amorphous structure to crystal structure. Thermal stabilities of the samples were determined with TGAs, which give prior knowledge about the combustion behaviors of the polymer-based materials. It was found that the addition of vermiculite delayed the decomposition temperature and the residue at 800°C increased about 15% with VMT15 compared to pure. When the thermal behavior of DSC was examined, the four temperature zones appear prominently. There was a temperature shift with the addition of vermiculite particles into the PU. According to DSC, the VMT15 shifted the temperature approximately 61°C compared to pure. In the vertical combustion test, it was observed that the surface temperature was maintained with the addition of vermiculite, and the combustion progress was stopped. According to TS EN ISO 11925-2 standard, the flammability class of the samples added vermiculite is determined as D. The LOI value of the PU foam increased with the addition of 5% VMT, which is the lowest ratio of the addition. The highest LOI value was determined as 24.3% when the VMT addition increased to 15%.
