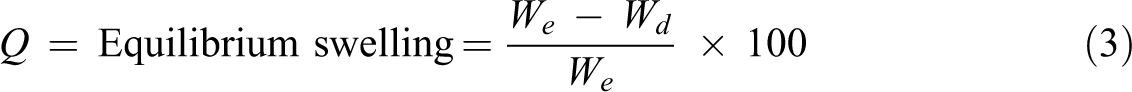Abstract
Various compositions of polyvinyl alcohol/acrylic acid Poly(PVA/AAc) hydrogels are prepared using of gamma radiation. The properties of the prepared poly(PVA/AAc) hydrogel and poly(PVA/AAc/magnesium oxide (MgO)) composite hydrogel were characterized by various techniques such as fourier transform infrared (FTIR), X-ray diffraction (XRD), transmission electron microscopy (TEM), and scanning electron microscopy. The TEM and XRD patterns of MgO nanoparticles indicate that the MgO with average particle size ranged from 18.11 nm to 45.6 nm. The gel (%) of the prepared poly(PVA/AAc) hydrogel with different contents of AAc increased with increasing both irradiation dose and AAc content. The swelling (%) of poly(PVA/AAc) hydrogel at different irradiation doses and different AAc contents found to be decreased with increasing both irradiation dose and AAc contents. The effect of incorporation of MgO nanoparticles into the poly(PVA/AAc) hydrogel matrix for absorption of boron from aqueous solution under various factors such as different concentrations of boron, different concentrations of MgO, and at different treatment times was investigated. From the study, the modified poly(PVA: AAc/MgO) composite hydrogel with composition (80:20 vol.%/5 wt%) has higher substitution, macroporous structure and characterized by better performance in swelling and boron uptake capacity than poly(PVA/AAc) hydrogel with composition (80/20 vol.%). The high absorption of boron indicates complex formation via ion exchange and chelation between poly(PVA/AAc/MgO) and boron due to the presence of different moieties provided by acrylic–acrylic, acrylic-PVA, and PVA-PVA.
Introduction
Boron is used in various industries and applications such as textile dyes, soap, detergent, solder, photography, nuclear field, glass fiber, and paper industry 1 and also in the production of glass, enamels, glazes, skin, synthetic detergents, and microfertilizers. 2 It is also necessary for humans and animals and affects crop productivity and plant growth but in small amount. 1 The recommended concentration of boron for drinking water according to the World Health Organization guidelines is up to 2.4 mg l−1,3,4 and the level of boron in drinking, irrigating water, and wastewaters discarded to the environment is 1.0 mg l−1.5,6 The high levels of boron are harmful and even lethal to plants. In humans, an excess of boron may lead to damage of the nervous system. 7
There are many methods and materials used for removal of boron, but there is no effective and concurrently economical method for removing boron from aqueous solution even of using low-cost abundant polysaccharides adsorbents. 7 The low removal of boron in the previously studied adsorbent limits their potential applications. 7
Boric acid can form complexes with numerous hydroxyl compounds.8–11 Polyvinyl alcohol (PVA) is widely used in the formation of cross-linked hydrogels. The carbon backbone of the PVA contains large number of hydroxyl groups, acrylic acid (AAc) which is a hydrophilic monomer and has reactive chelating carboxylic acid groups capable of chelating with boron. Therefore, this work aims to synthesize low-cost and highly chelating poly(PVA/AAc)12,13 and new poly(PVA/AAc/magnesium oxide (MgO)) composite which are prepared by gamma radiation that can be used in boron removal from its aqueous solution.
Gamma ray is a high-energy ionizing radiation in electromagnetic spectrum that easily penetrates most materials. The use of gamma radiation for preparing of different types of adsorbent polymers and hydrogels is considered as wide field of important applications of radiation technology that can be used in environmental applications.14–22
Experimental
Materials
AAc (99%) and boric acid (99%) were purchased from SD fine-Chem Limited, Mumbai, Maharashtra, India. Polyvinyl alcohol (DP 1700-1800), hydrolysis 98-99% (fully hydrolysed) was provided by Lobachemie Pvt. Ltd, Mumbai, Maharashtra, India. All other chemicals are analytical grads and used without further purification.
Preparation
Preparation of MgO nanoparticles
Stock solution of magnesium nitrate (1 M) and sodium hydroxide (1 M) were prepared. Fifty milliliter of sodium hydroxide was put in a beaker (250 ml) on a magnetic stirrer, and then, 50 ml of magnesium nitrate was added very slowly and drop wise, resulting in the formation of a white suspension instantly. The dispersion was left to settle down and the supernatant was removed by decantation; and the produced MgO particles were repeatedly washed by distilled water with subsequent redispersion in distilled water to remove excess salt. To ensure the formation of MgO oxide nanoparticles, the dry white precipitate was calcined in an oven at 450°C for 3 h. Then, the MgO oxide nanoparticles were removed and put in polyethylene bag.
Preparation of poly(PVA/AAc) hydrogel
Firstly, a stock solution of PVA was prepared by dissolving 3 g of solid PVA (fully hydrolyzed) in 100 ml of water and stirred with heating until complete dissolution. Various compositions of PVA/AAc were prepared by adding 5, 10, 15, and 20 vol.% of AAc into appropriate amounts of PVA to make 10-ml sample volume, each in a separate test tube and exposed them to various gamma radiation doses 10, 30, 40, and 50 kGy at a dose rate of 0.309 Gy s−1. The prepared poly(PVA/AAc) hydrogel samples with different compositions were first air-dried for about 24 h, and then, they were dried to have a constant weight in an oven at 60°C. The samples were kept in distilled water for 4 h by heating to remove soluble part of samples followed by drying for constant weight. The gel (%) was calculated gravimetrically using the following equation
where Wg and Wo are the weight of dried sample after and before extractions, respectively.
Preparation of poly(PVA/AAc/MgO) composite hydrogel
Different amounts of MgO nanoparticle (1, 2, 3, 4, and 5 wt%) prepared previously (see “Preparation of MgO nanoparticles” section) were added to poly(PVA/AAc) hydrogel with composition (80/20 vol.%) and then subjected to gamma irradiation of dose 30 kGy radiation kGy at a dose rate of 0.309 Gy s−1. The formed copolymer composite hydrogel then cut into similar and equal disks and left to dry at room temperature.
Characterization
Transmission electron microscopy
Transmission electron microscopy (TEM) was used to observe the morphology of the formed nanoparticles. The prepared MgO powder and Ag nano-dispersion were suspended in water, and a drop of the resultant mixture was deposited on an ultrathin carbon-supported copper (Cu) grid, and air-dried then examined by TEM) (JEM100CS, Jeol Electron Microscope, Japan) with an acceleration voltage of 80 kV.
X-ray diffraction
X-Ray diffraction (XRD) measurement was conducted for MgO nanoparticles, using X-ray diffraction operated at Cu K at wavelength of 1.54A, 30 nmA, and a voltage of 40 keV. The X-ray tube with Cu target operates at 40-kV voltage and 30-mA current. The X-ray data were recorded in the range from 4° to 80° (2θ) with continuous scanning mode and scanning speed 8° min−1. The particle size calculated from Scherer equation is as follows 23
where D is the particle size, K = 0.89 is the Scherer constant related to the shape and index of the crystals, λ is the wave length of the X-ray (Cu Kα, 1.54056 Å), θ is the diffraction angle, and β is the fall-width at half-maximum (in radian).
Equilibrium swelling
The hydrogels discs were soaked in distilled water at room temperature. Swollen hydrogels were removed from water, dried, and weighed for several times. The measurements were continued until a constant weight is reached for each sample. The swelling percent (Q) at equilibrium was calculated using the following equation
where Wd is the weight of dried hydrogels, and We is the weight of the swelling gel at equilibrium.
Fourier transform infrared spectroscopic measurements
The fourier transform infrared (FTIR) spectra of poly(PVA/AAc) hydrogel and poly(PVA/AAc/MgO) composite hydrogel over the range 4000–400 cm−1 were recorded using FTIR (FTIR-Vertex 70) spectrophotometer (Bruker, Germany).
Scanning electron microscopy
Scanning electron microscopic (SEM, JSM-5400, Japan) analysis was used to investigate the morphology of the surfaces of the poly(PVA/AAc) hydrogel and poly(PVA/AAc/MgO) composite hydrogel. A sputter coater was used to pre-coat conductive gold onto the fractured surfaces before observing the micrographs at 30 kV.
Application
Batch adsorption of boron
The batch adsorption technique of boron was carried out by adding 1 g of poly(PVA/AAc) hydrogel with composition (80/20 vol.%) and poly(PVA/AAc/MgO) composite hydrogel with different MgO contents in 100 ml of 0.1-M H3BO3 solution in a beaker under stirring at 25°C for 24 h. The hydrogel was removed from the mixture for each sample, and the residual boron concentration of the solution was determined by ultraviolet (UV)-spectrometer. Finally, the equilibrium boron uptake capacity of the prepared hydrogels (q) was characterized by the mmol of boron adsorbed per gram of hydrogel. The adsorption capacity (Q) of the boron is calculated according to the following equation
where Co and Ce (mg l−1) are the initial and equilibrium concentration of boron in the solution, respectively, V is the volume of the solution (l), m is the mass of dry hydrogels (g), and M is the mole weight of H3BO3 (g mol−1).
UV spectroscopy
T60 UV-Vis spectrophotometer (PG instruments) with a scan range of 190–1100 nm was used for the detection of removal of boron by PVA/AAc hydrogel with composition (80/20 vol.%) and PVA/AAc/MgO composite hydrogel with different MgO contents. The determination of boron concentration was carried out at λmax = 280 nm. Calibration curve of boron at different concentrations was constructed.
Field-emission scanning electron microscope
Scanning Electron Microscope- Field Emission for poly(PVA: AAc/MgO) composite hydrogel with composition (80:20 vol.%/5 wt%) and poly(PVA/AAc/MgO/B) composite was carried out using SEM Model NRC Quanta FEG 250 (Field Emission Gun) attached with energy-dispersive X-ray (EDX) unit (EDX analyses), Netherlands. The samples were sputtered with gold. The analysis was performed in the National Research Center (Dokki, Giza, Egypt).
Results and discussion
Characterization of MgO nanoparticles
The TEM and XRD patterns of MgO nanoparticles, which are prepared chemically and followed by calcination method, are shown in Figure 1(a) and (b), respectively. From XRD patterns, the MgO nanoparticles show sharp diffraction peaks appeared at 2θ = 29°, 43°, and 62° which are characteristic of nano-MgO nanoparticles.24,25 Also from Figure 1(a) and (b), both TEM and XRD patterns of the MgO nanoparticles have the lowest average particle size ranged from 18.11 nm to 45.6 nm, indicating that successful preparation of MgO nanoparticles described in this study.
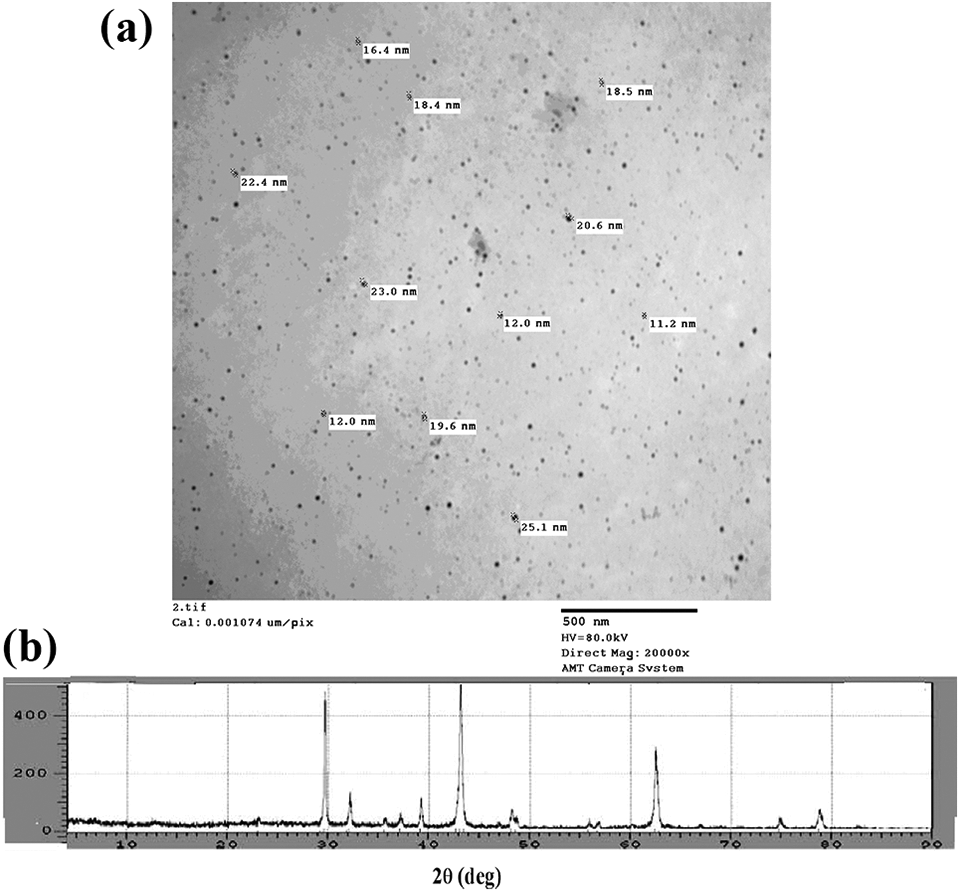
(a) Transmission electron microscopy and (b) X-ray diffraction of magnesium oxide nanoparticles.
Preparation and gel percent of poly(PVA/AAc) hydrogels at different irradiation doses
Figure 2 shows the gel (%) of poly(PVA/AAc) hydrogels containing different contents of AAc (5, 10, 15, and 20 vol.%) prepared at different gamma irradiation doses (10, 20, 30, 40, and 50 kGy). It is noted that for all poly(PVA/AAc) hydrogel compositions the gel (%) increases with increasing AAc contents. This is due to the increase of AAc content which leads to the formation of more free radical that increases the copolymerization process with the PVA and thereby increasing the cross-linking, leading to the formation of nondissolved cross-linked matrix and increased gel (%). Also, as irradiation dose increases, the cross-linking of the formed poly(PVA/AAc) hydrogel increases up to 50 kGy and the gel (%) increases as shown in Figure 2. The cross-linking is formed through the formation of both the physical hydrogen bonds between –OH groups of PVA and –COOH groups of AAc and through the radiation technique which induced chemical covalent bonds formation responsible for achieving stable hydrogels network with high gel (%) ranged from 84% to 96% at doses 10 kGy to 50 kGy, respectively.
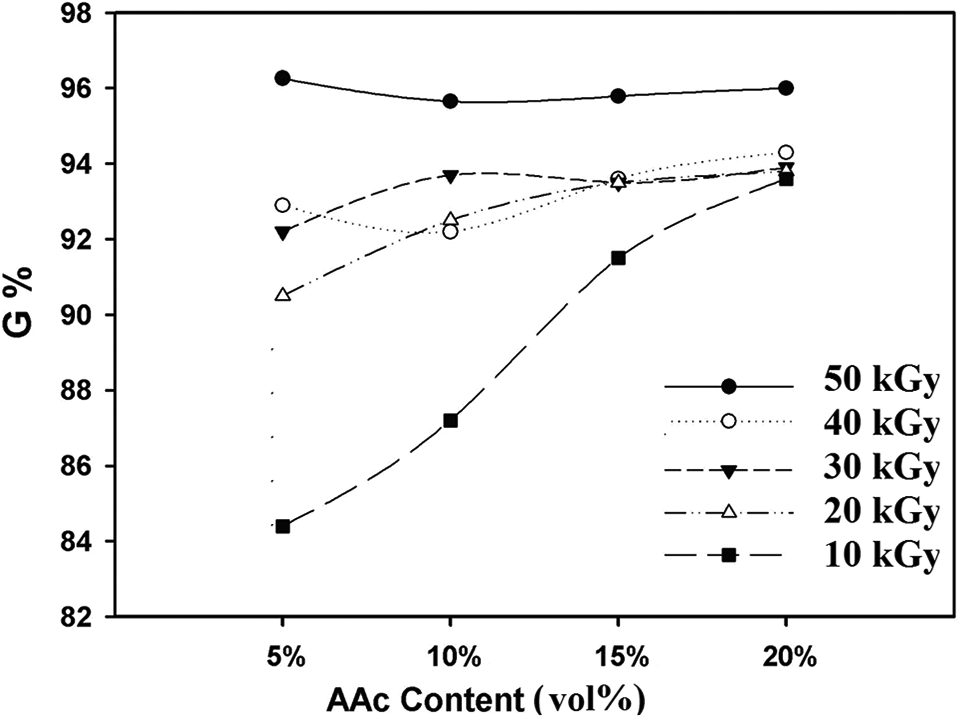
Gel (%) of poly(PVA/AAc) hydrogel at different irradiation doses.
FTIR analysis
FTIR spectroscopy was employed to confirm the chemical structures of PVA, poly(PVA/AAc) hydrogel with composition (80/20 vol.%), and poly(PVA/AAc/MgO) composite hydrogel with composition (80:20 vol.%/5 wt%), the spectra of which are represented in Figure 3(a), (b), and (c), respectively. The characteristic peak for the PVA as shown in Figure 3(a) is appeared at 3261, 2918, and 1089 cm−1. The broad peak appeared at 3261 cm−1 due to the hydroxyl group (O–H) stretching vibration of PVA; the peak appeared at 2918 cm−1 due to aliphatic C–H stretching vibration; and a peak observed at 1089 cm−1 corresponds to C–O symmetrical stretching of the PVA backbone. The characteristic peaks of poly(PVA/AAc) hydrogel represented in Figure 3(b) are similar to the peaks of PVA in Figure 3(a) beside two new peaks appeared at 1722 cm−1 due to carboxylic group (–COOH) of AAc in poly(PVA/AAc) hydrogel and the second peak appeared at 1189 cm−1 which correspond to C–O–C group. 26 Also the new broad peak appeared at 3253 cm−1 corresponds to –OH groups resulted from overlapping between hydroxyl groups (–OH) of PVA with the –OH group of carboxylic group of AAc. The previous peaks prove the copolymerization of PVA with the AAc.
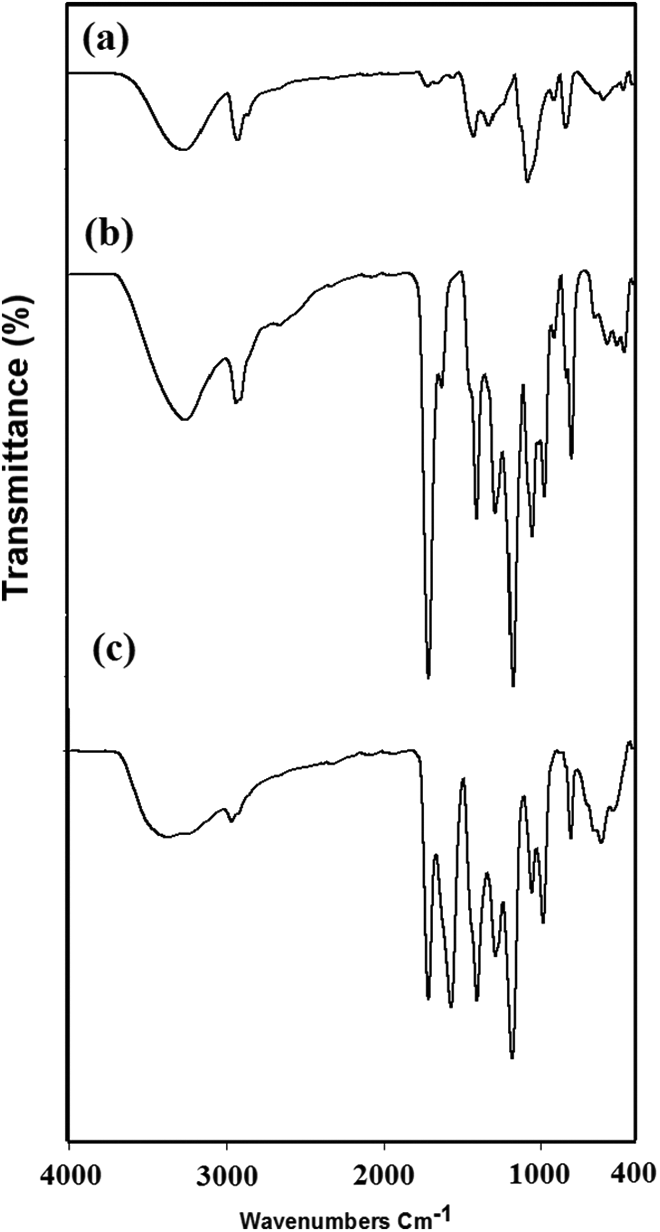
FTIR spectra of (a) PVA, (b) poly(PVA/AAc) hydrogel with composition (80/20 vol.%), and (c) poly(PVA: AAc/MgO) composite hydrogel with composition (80:20 vol.%/5 wt%) prepared at 30 kGy.
The FTIR analysis of poly(PVA/AAc/MgO) composite hydrogel is shown in Figure 3(c), and the characteristic peaks appeared are similar to both PVA and poly(PVA/AAc) hydrogel. The MgO nanoparticles caused changes in the FTIR spectra of poly(PVA/AAc) hydrogel which include shift in some peaks due to shift in functional group of poly(PVA/AAc) hydrogel to form poly(PVA/AAc/MgO) and also to cause decrease and broadening in the intensities of the peaks as other metal oxides incorporated into polymer matrix. 27
Swelling behavior
Equilibrium swelling
Swelling behavior is one of the most important properties of adsorbents that affect final efficiency on removal of metal ions such as boron and other metal ions.14,15 For this purpose, the swelling behavior of the prepared poly(PVA/AAc) hydrogel at different AAc contents and at different irradiation doses (10, 20, 30, 40 and 50 kGy) was studied, and the results are shown in Figure 4. It is found that the swelling (%) decreased with the increasing amounts of AAc. This is due to increase of both hydrogen bonding and cross-linking structure by increasing contents of AAc 28 and hence the gel (%) increased as shown in Figure 2, leading to decrease of the swelling (%) as shown in Figure 4.
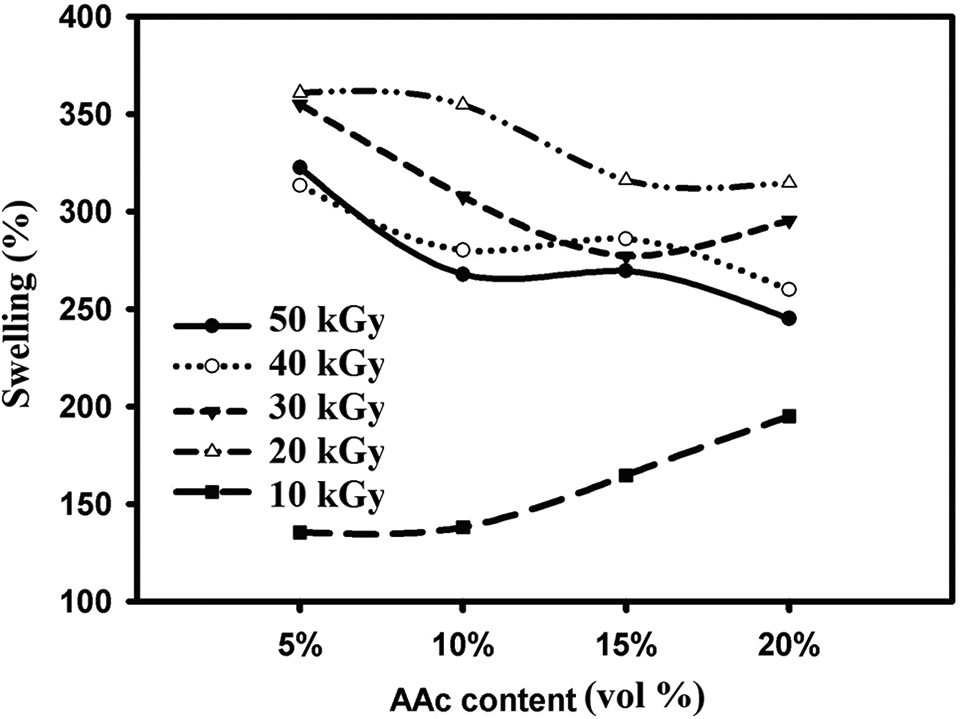
Equilibrium swelling of poly(PVA/AAc) hydrogel with different compositions at different irradiation doses.
In the case of the effect of irradiation dose shown in Figure 4, it is found that the swelling (%) decreased with increasing irradiation doses due to increased cross-linking density formation within the hydrogel structure that leads to decrease in the swelling behavior.
Equilibrium swelling study at different pH
Most of the hydrogels containing carboxylic groups of AAc are sensitive to the pH changes where these groups are ionized with increasing pH. From Figure 5 in case lower pH 3 and pH 5 protonation of carboxylic groups occurs which resulted in narrowing of the pore size and reduces the free spaces available to water penetration 15 that leads to disappearance of the swelling osmotic pressure inside the hydrogel and hence decreases swelling (%). 29 While as pH of the medium increases to pH 7, the swelling (%) increases due to ionization of carboxylic groups, leading to the generation of swelling osmotic pressure inside the hydrogel of poly(PVA/AAc) hydrogel for all contents of AAc which increases swelling (%) with increasing pH. Therefore, the prepared poly(PVA/AAc) hydrogel containing different contents of AAc from 5% to 20% of AAc exhibits sensitive respond with the pH of the environment. The poly(PVA/AAc) hydrogel with composition (80/20 vol.%) has higher swelling behavior than other hydrogels which can be used in different applications such as removal of boron, and therefore, it is selected.
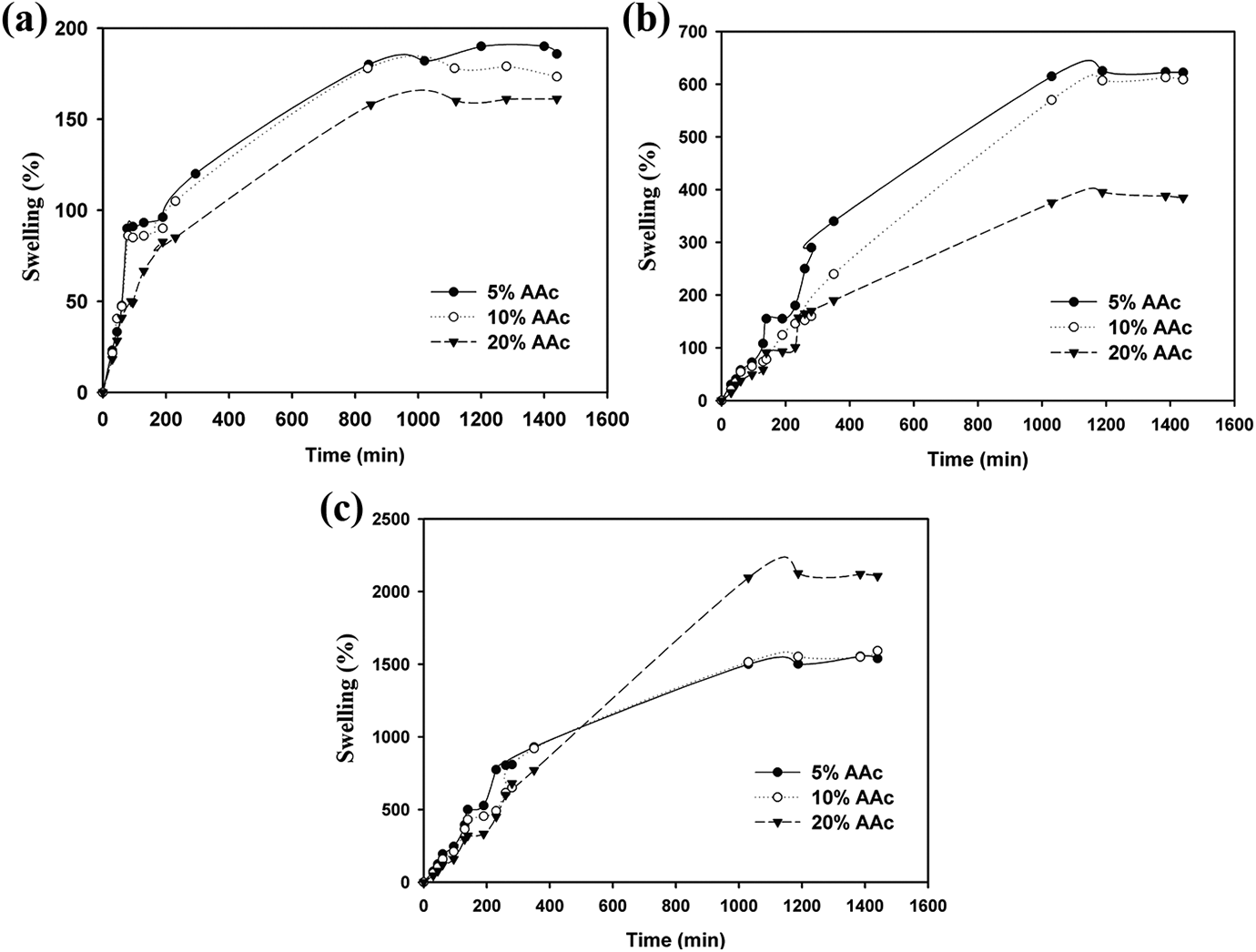
Swelling of poly(PVA/AAc) hydrogel at (a) pH3, (b) pH5, and (c) pH7.
Removal of boron from its aqueous solution
In the point of view, water sources like lakes, sea, groundwater, and so on are becoming polluted by different kinds of contaminants, including toxic heavy metals such as boron.
The sources of these pollutants in surface waters are commercial and industrial establishments. The resulting environmental hazards are undesirable, and therefore, heavy metal ions such as boron must be appropriately removed using new improved techniques.
In our novel research work, both poly(PVA/AAc) with composition (80/20 vol.%) and poly(PVA: AAc/MgO) hydrogel composite, containing different MgO contents 1, 3, and 5 wt%, were selected and employed for the removal of boron due to its good chemical stability and its higher swelling behavior as shown in Figures 2 and 5, respectively.
The effect of different conditions in removal of boron by poly(PVA/AAc) hydrogel and poly(PVA/AAc/MgO) composite hydrogel with different contents of MgO nanoparticles ranged from 1 wt% to 5 wt% is investigated and shown in Figure 6(a) to (c). From Figure 6(a), it is observed that the adsorption capacity of poly(PVA/AAc) hydrogel and poly(PVA/AAc/MgO) composite hydrogel toward boron increased with increasing of boron concentration from 1 mg ml−1 to 5 mg ml−1 then decreased at high initial concentration of boron at 6 mg ml−1. The increase of the adsorption capacity with the increasing initial boron concentration was supposed to be due to the increase of the probability of poly(PVA/AAc) hydrogel and poly(PVA/AAc/MgO) composite hydrogel chelating moieties to react with boron with higher diffusion rate up to 5 mg ml−1. The lower adsorption capacity of poly(PVA/AAc) hydrogel and poly(PVA/AAc/MgO) composite hydrogel toward boron at high initial concentration of boron 6 mg ml−1 indicates that the boron adsorption is mostly from the complex reaction therefore, the higher concentration 6 mg ml−1 lowers the complex formation between boron and poly(PVA/AAc) hydrogel and poly(PVA/AAc/MgO) composite hydrogel. 8
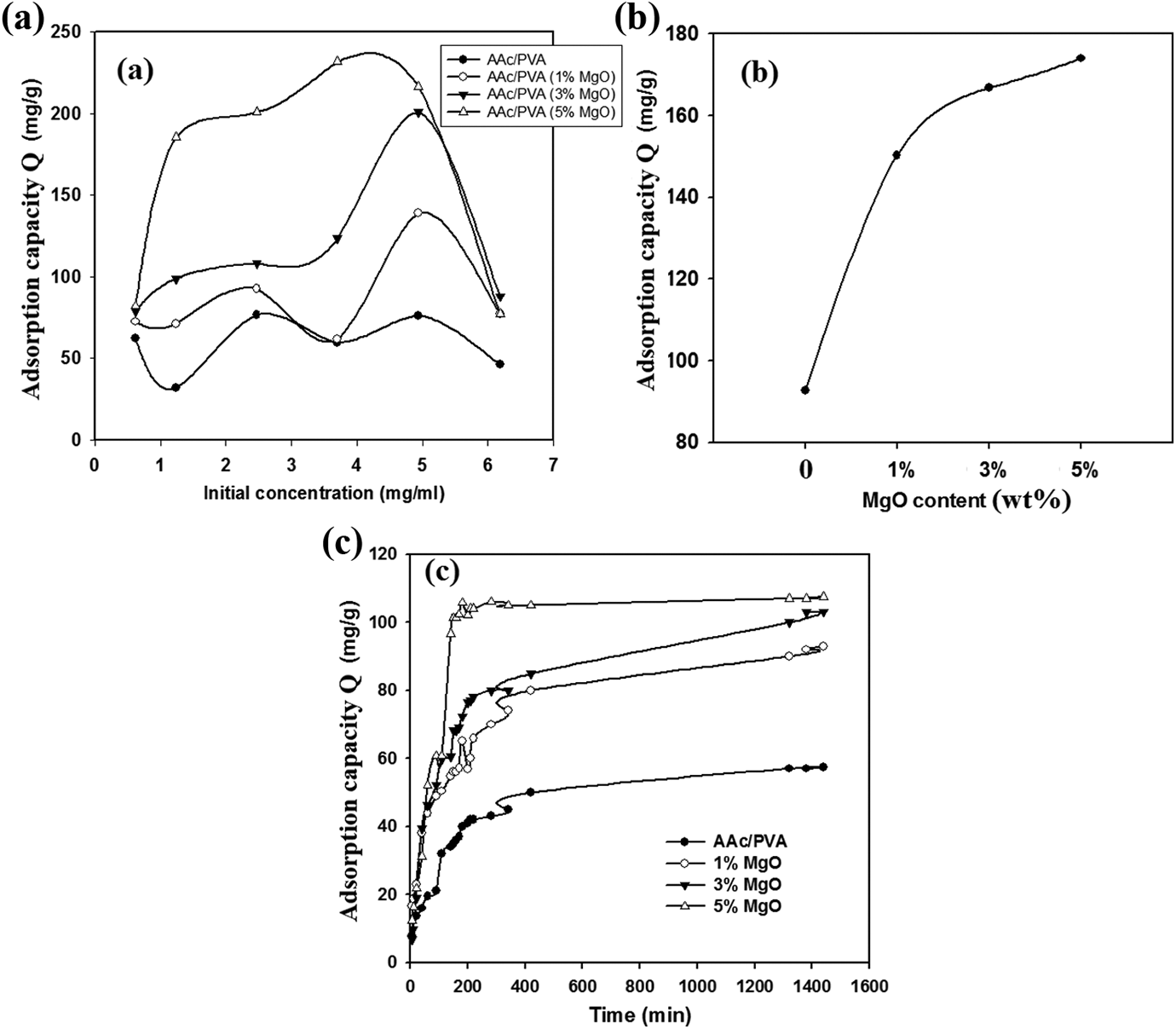
Effect of different conditions in removal of boron (a) at different concentrations of boron, (b) at different concentrations of MgO, and (c) at different treatment times.
Figure 6(b) represents the adsorption capacity of poly(PVA/AAc) hydrogel and poly(PVA/AAc/MgO) composite hydrogel with different contents of MgO toward boron. It is observed that as MgO content increased from 1 wt% to 5 wt%, the adsorption capacity toward boron increased due to high porous structure of the poly(PVA/AAc/MgO) composite hydrogel as shown in Figure 7(b) than poly(PVA/AAc) hydrogel which increases complex formation between poly(PVA/AAc/MgO) composite and boron compared to poly(PVA/AAc) hydrogel. 30 This is due to the porous structure that increased the surface area of poly(PVA/AAc/MgO) composite hydrogel, facilitating the adsorption of boron. 4 Figure 6(c) represents the adsorption capacity as function of treatment time toward boron by poly(PVA/AAc) hydrogel and poly(PVA/AAc/MgO) composite hydrogel with different contents of MgO. It is noted that as treatment time increased, the amount of boron adsorption increased due to the increase of removal efficiency of boron by increasing complex formation between boron and poly(PVA/AAc) hydrogel and poly(PVA/AAc/MgO) composite hydrogel.
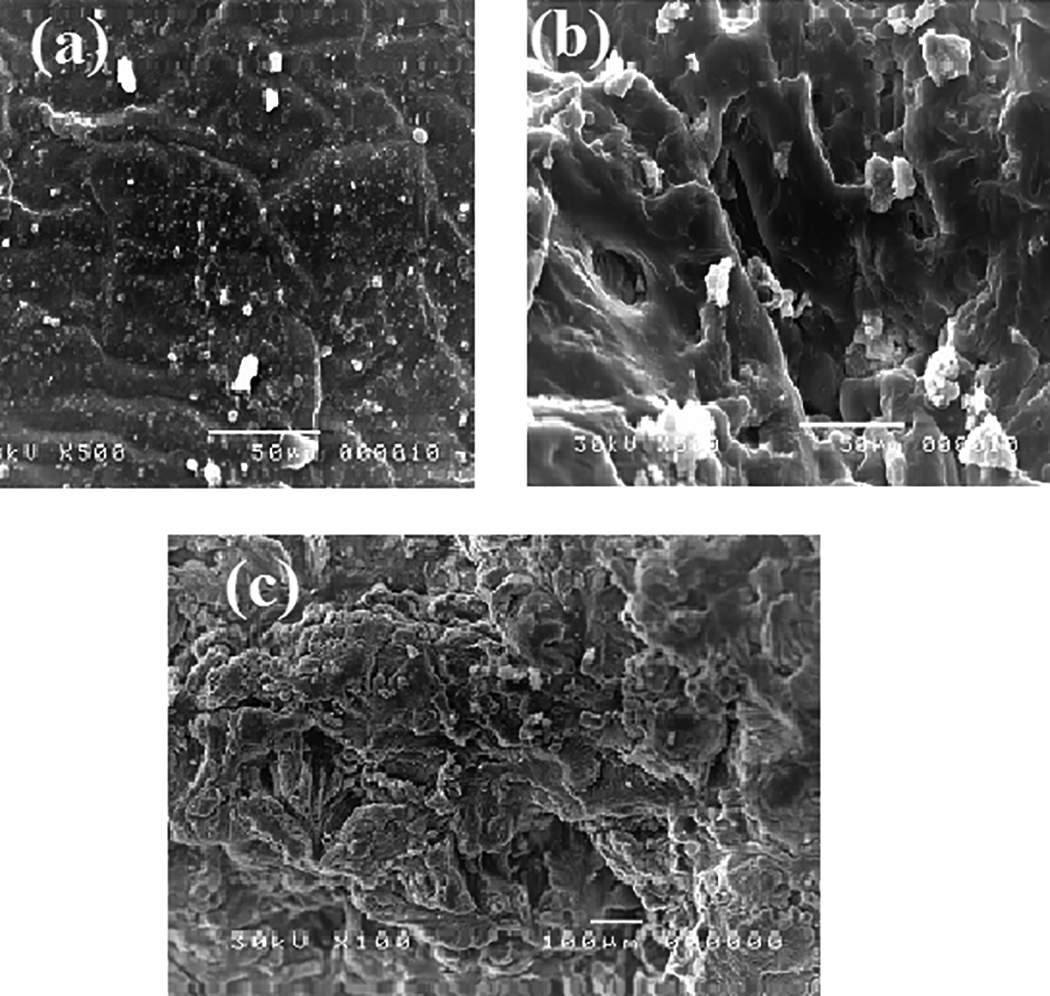
Scanning electron microscopic images of (a) poly(PVA/AAc) hydrogel with composition (80/20 vol.%), (b) poly(PVA: AAc/MgO) composite hydrogel with composition (80:20 vol.%/5 wt%), and (c) poly(PVA/AAc/MgO/B) hydrogel with composition loaded with boron.
Also the the amount adsorbed of boron increased with increasing content of MgO. This is due to the formation of high porous structure with large average pore size diameter formed in poly(PVA/AAc/MgO) composite hydrogel as shown in Figure 7(b). The higher swelling properties of poly(PVA/AAc/MgO) composite hydrogel affect the removal efficiency of metal ions such as boron and other metal ions14,15 than poly(PVA/AAc) hydrogel with composition (80/20 vol.%) as shown in swelling studies.
Finally, Scheme 1 represents the type and the mechanism of chelating of boron by the prepared poly(PVA/AAc) hydrogel and poly(PVA/AAc/MgO) composite hydrogel between different moieties acrylic–acrylic, acrylic-PVA, and PVA-PVA which describe the reason of the high removal properties of the prepared poly(PVA/AAc) hydrogel and poly(PVA/AAc/MgO) composite hydrogel than other previously prepared materials described in Introduction part.1,2,4,7–13
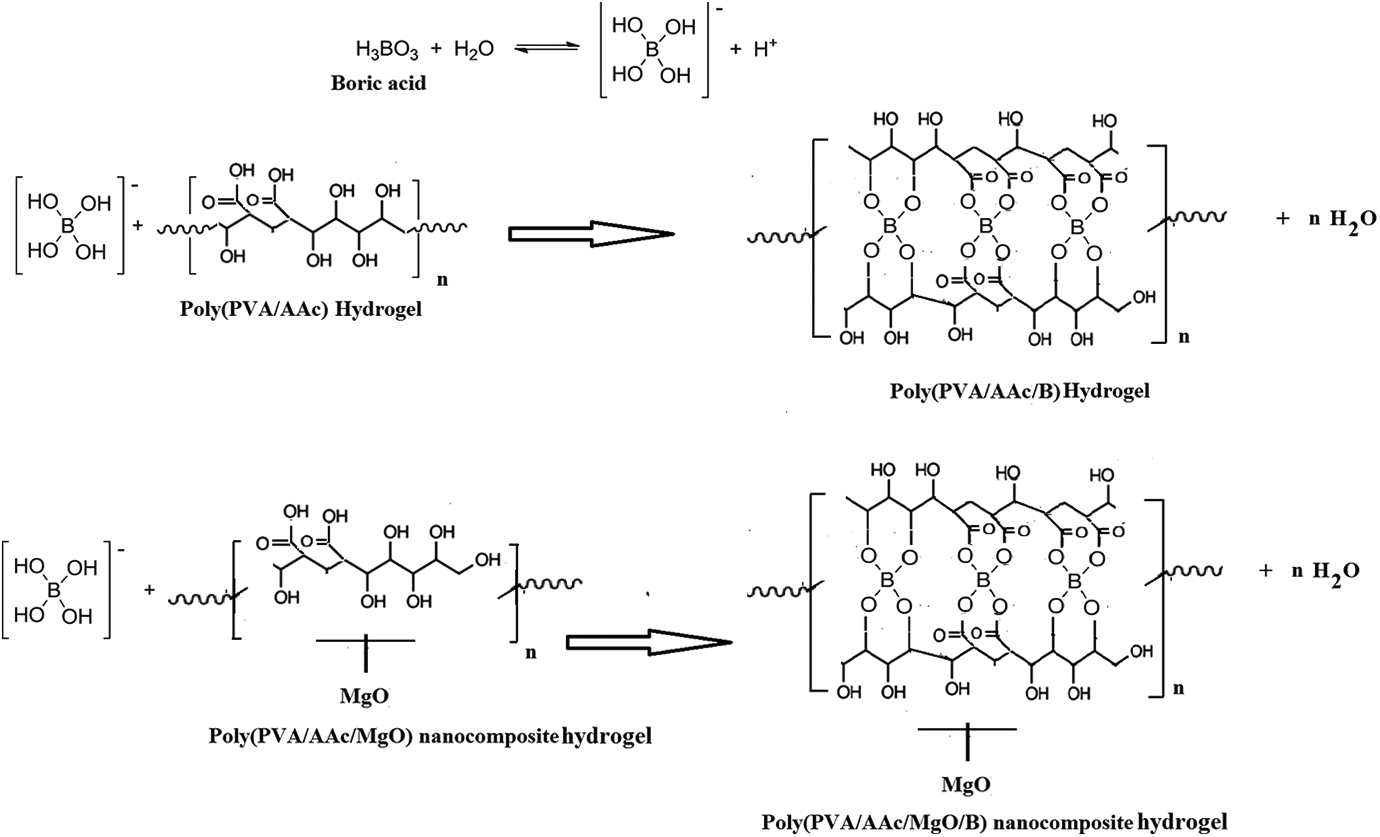
Removal of boron.
Scanning electron microscopy
Figure 7(a) to (c) Finally for Poly(PVA/AAc/MgO/B) hydrogel composite represents the SEM morphology photo of poly(PVA/AAc) hydrogel with composition (80/20 vol.%), poly(PVA: AAc/MgO) composite hydrogel with composition (80:20 vol.%/5 wt%), and poly(PVA/AAc/MgO/B) composite. The SEM images showed that there are significant changes on the morphology of the prepared hydrogels according to its different chemical structures. It is clear that the surface of poly(PVA/AAc) hydrogel has low diameter pores, while the surface of poly(PVA: AAc/MgO) composite hydrogel with composition (80:20 vol.%/5 wt%) before adsorption of boron characterized by high porous structure with increasing the average pore size diameter than poly(PVA/AAc) hydrogel. Composite the surface were highly coated with boron after batch absorption technique.
Adsorption mechanism
The adsorption mechanism is the interactions occurring between the adsorbent/adsorbate interfaces. In the present study, the boron adsorption onto the surface of poly(PVA: AAc/MgO) composite hydrogel with composition (80:20 vol.%/5 wt%) is assumed to occur through complex formation via ion exchange and chelation14,15,31–34 between positively charged boron and ionized and non-ionized carboxylic groups and hydroxyl group within the hydrogels. Schematic images showing removal of boron (Scheme 1), SEM images (Figure 7), and FTIR spectra (Figure 8) of the Poly(PVA/AAc/MgO) composite hydrogel before and after boron adsorption were investigated to confirm the abovementioned adsorption mechanism.
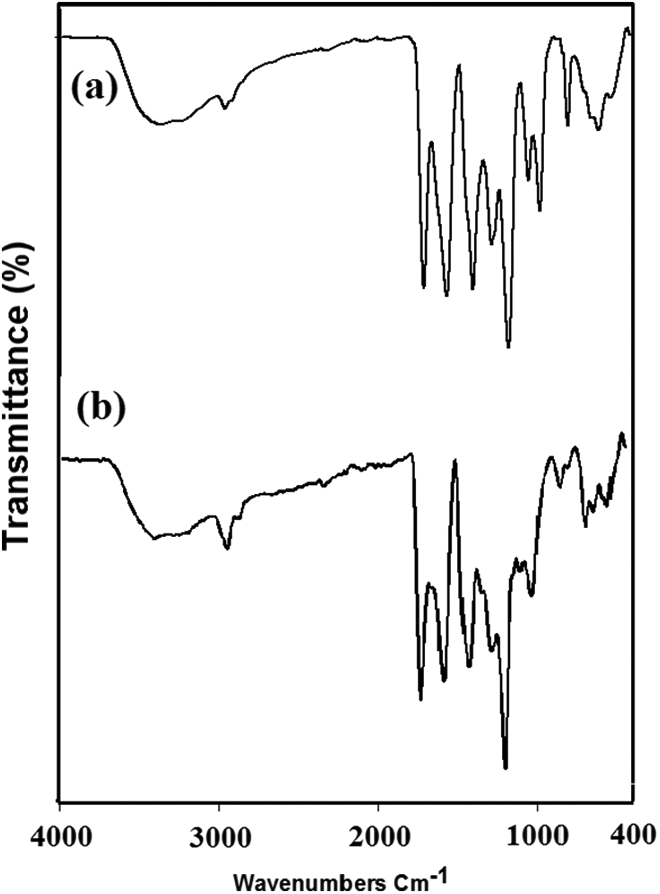
FTIR spectra of (a) poly(PVA: AAc/MgO) composite hydrogel with composition (80:20 vol.%/5 wt%) and (b) poly(PVA /AAc/MgO/B) hydrogel.
In the FTIR spectra (Figure 8), the presence of broad absorption peak at 3253 cm−1, corresponding to the overlap of –OH stretching vibrations of PVA and –OH stretching band of acrylic in poly(PVA/AAc/MgO) composite hydrogel was broadened and shifted to peak at 3329 cm−1 after adsorption of boron. The presence of peak at 1722 cm−1, which was ascribed to the stretching vibration of –COOH groups, was shifted to 1714 cm−1 after boron adsorption. The absorption peak at 1569 cm−1, corresponding to asymmetric stretching vibration of C–O–C group in the carboxylate groups, was shifted to 1409 cm−1, and the peak at 1483 cm−1, related to the symmetric stretching vibration of –CO in carboxylate groups, were shifted to 1462 cm−1 after adsorption of boron. Therefore, the adsorption of boron onto the surface of poly(PVA/AAc/MgO) composite hydrogel is assumed to occur through complex formation of ion exchange and chelation of boron14,15,31–34 between different moieties acrylic–acrylic, acrylic-PVA, and PVA-PVA as shown in Scheme 1.
Field-emission scanning electron microscopy
Field-emission scanning electron microscopic analysis of poly(PVA: AAc/MgO) composite hydrogel with composition (80:20 vol.%/5 wt%) and poly(PVA/AAc/MgO/B) composite was performed and represented in Figure 9. It can be seen that the surface structure of the poly(PVA/AAc/MgO) composite hydrogel was characterized by the presence of MgO particles with an average particle size of 28 µm due to the accumulation of the MgO in the surface of the poly(PVA/AAc/MgO) composite. After adsorption of boron, the surface becomes more coated with boron and the composite surface becomes more dense from the interior to the surface of the composite with high numbers of boron particles.

Scanning electron microscope–field emission of (a) poly(PVA: AAc/MgO) composite hydrogel with composition (80:20 vol.%/5 wt%) and with magnification 250 times, (b) poly(PVA/AAc/MgO) composite hydrogel with magnification 1000 times, (c) poly(PVA/AAc/MgO/B) composite with magnification 250 times, and (d) poly(PVA/AAc/MgO/B) composite with magnification 12,000 times.
EDX analysis
The EDX analysis of poly(PVA: AAc/MgO) composite hydrogel with composition (80:20 vol.%/5 wt%) and poly(PVA/AAc/MgO/B) composite was performed and represented in Figure 10(a) and (b) to determine the presence of the magnesium in poly(PVA/AAc/MgO) composite hydrogel and boron in the poly(PVA /AAc/MgO/B) composite. As shown in Figure 10(a), the EDX analysis confirms the presence of magnesium within the hydrogel matrix of poly(PVA/AAc/MgO) composite hydrogel. Also, EDX results prove the presence of boron and magnesium in the poly(PVA/AAc/MgO/B) composite hydrogel.
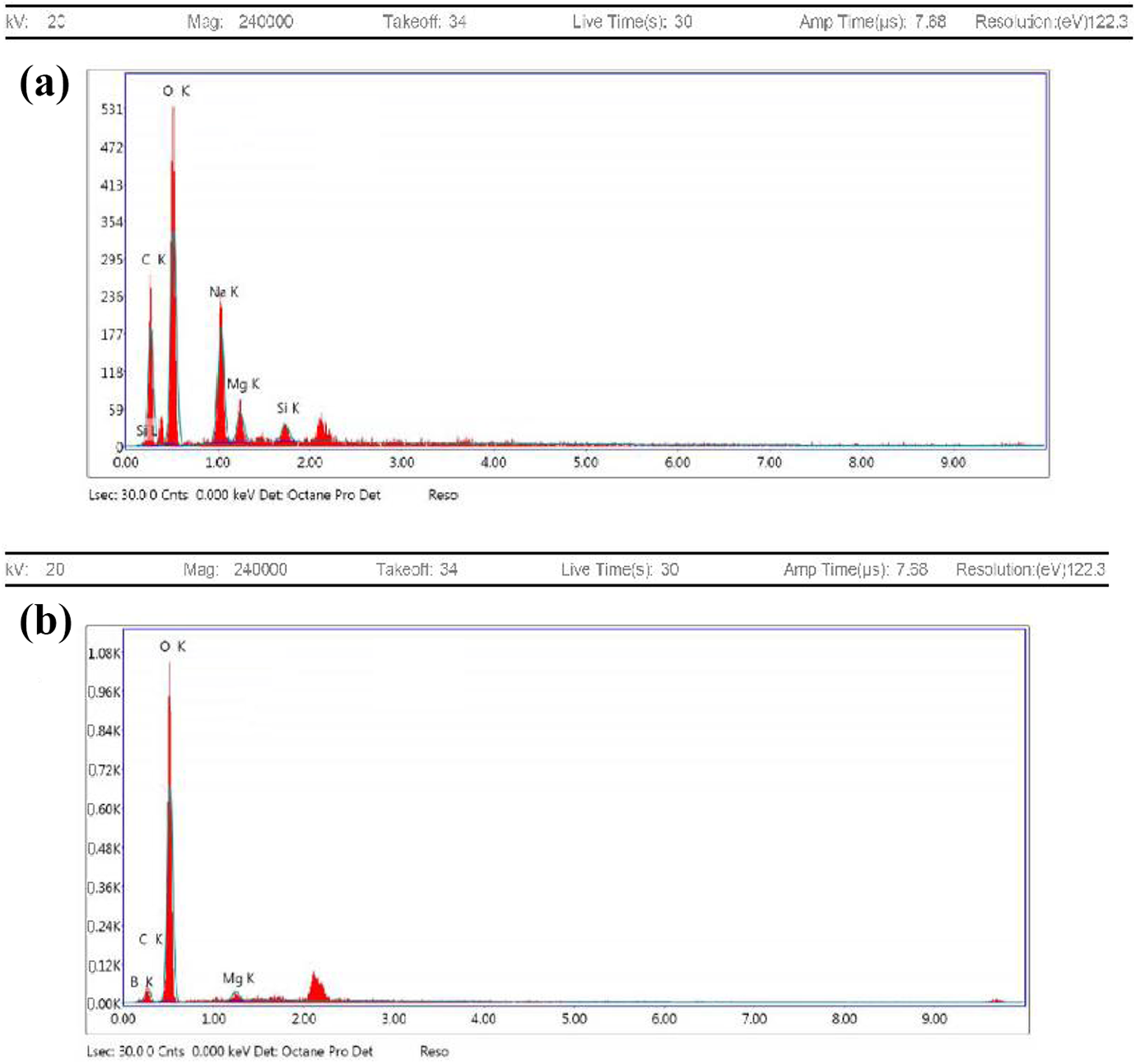
EDX of (a) poly(PVA: AAc/MgO) composite hydrogel with composition (80:20 vol.%/5 wt%) and (b) poly(PVA/AAc/MgO/B) composite.
Conclusions
Successful preparation of MgO nanoparticles was described in this study and confirmed by XRD pattern and TEM. The use of gamma radiation as eco-friendly technique for the preparation of both poly(PVA/AAc) and poly(PVA: AAc/MgO) composite hydrogel was found to be suitable technique without using initiator, catalysis, or any another chemicals or additives. Therefore, the prepared hydrogels are free from undesirable chemical impurities such as residues from initiators, retarders, and/or accelerators for initiation than the cross-linking reaction used in chemical cross-linking methods. There are several review papers describing removal methods of boron from either saline waters or aqueous solutions, but there is no effective and concurrently economical method for removing boron from aqueous solution even of using low-cost abundant polysaccharides adsorbents. The low-cost poly(PVA: AAc/MgO) composite hydrogel with composition (80:20 vol.%/5 wt%) was characterized by good properties than other hydrogels and has higher adsorption capacity toward boron than any other previously reported review. The absorption of boron indicates complex formation via ion exchange and chelation between poly(PVA/AAc/MgO) and boron due to the presence of different moieties acrylic–acrylic, acrylic-PVA, and PVA-PVA as shown in Scheme (1).