Abstract
The present work deals a new polymer electrolyte, that is, polyvinylpyrrolidone doped with salt ammonium iodide and plasticizer ethylene carbonate (EC) prepared via solution casting technique for supercapacitor application. Electrochemical impedance spectroscopy was carried out using CH instrument electrochemical work station which shows enhancement in conductivity (
Introduction
In the early 1970s, PV Wright et al. was the first to study polymer electrolytes for the conducting properties.1,2 Armand et al. later was the first to find polymer electrolytes as a material having a potential for high-energy batteries.3,4 In view of the fact that solid polymer electrolytes (SPEs) have been largely used by several groups5–9 for their applications in high-energy batteries. SPEs are the ionic conducting material produced by dissociation of ion species in a solvent with a suitable polymer which acts as a host material for the conduction of ions. SPEs systems are generally a mixed phase system which contains crystalline polymer complex (non-conducting or poorly conducting) and amorphous phase (conducting). In a system of SPEs, mostly the amorphous phase is responsible for conduction of ions.
10
In an amorphous phase, the chain of a polymer shows fast internal modes of bond rotations which produce larger segmental motion leading to elevated ionic conductivity. As the viscosity of macroscopic physical quantity of polymer in a polymer electrolyte is comparatively high, hence it behaves like solids. SPEs are essentially being studied because of their capability for technological applications. SPEs show enhanced properties compared to their counterpart’s conventional aqueous electrolytes-based supercapacitor with the advantages such as easy to prepare, easy to handle, mechanically stable, noncorrosive, inflammable, and can be molded in different shapes and sizes. However, the prime problem which is associated with the SPEs is low ionic conductivity. In the past, various methods have been used to enhance the ionic conductivity of SPEs.11–15 Since decades, researchers have reported solid polymer electrolytes for electrochemical applications due to its various advantages over liquid electrolytes16–19 such as no leakage problem, mechanically stable, large electro stability window, and long life,. making it a promising material for various applications such as dye-sensitized solar cells, batteries, and supercapacitor. A number of amorphous nature host polymers such as poly (vinylidene fluoride-
The problems and the limitations associated with SPEs pushed us to search a host polymer like polymer which should be of less crystallinity nature at ambient temperature, molecules of polymer should be adequately mobile to assist ion migration in a matrix, and it should acquire solvation and coordination sites for ions support ion pair dissociation. In the present work, we have chosen polyvinylpyrrolidone (PVP) as the host polymer, ammonium iodide (NH4I) as an ionic salt, and ethylene carbonate (EC) as a plasticizer in a host polymer to improve the performance of an electrical double-layer capacitor (EDLC) fabricated in our laboratory. In a scenario of energy storage devices, the uniqueness of the work is that the solid polymer electrolyte doped with EC plasticizer in PVP + NH4I-based system with effective supercapacitor application.
Experimental details
Materials
PVP, PVDF-HFP, NH4I, current collector (graphite sheet), methanol, and EC were purchased from Sigma-Aldrich (St Louis, Missouri, USA), whereas solvents were purchased from Qualikems Fine Chem Pvt. Ltd (Vadodara, Gujarat, India). Graphene oxide (GO) was prepared in our laboratory by electro-exfoliation method.
Synthesis of polymer electrolyte
To prepare an EC-doped polymer electrolyte, we have used a simple approach, that is, solution casting technique. Primarily we have chosen the optimized 40 wt% NH4I-doped PVP polymer film (reported elsewhere by our group). Then, plasticizer added to the host polymer film, that is (PVP + 40 wt% NH4I) ranging from 10 wt% to 80 wt%.
Initially PVP was dissolved in methanol in a sample bottle and stirrer for a 4 h. Now, in a solution, mixed 40 wt% NH4I, followed by mixing of EC plasticizer ranging 10–80 wt% in a solution to prepare an EC-doped polymer electrolyte films. In continuity of stirring a solution, mixture was kept for a whole night to form a homogeneous solution of PVP + 40 wt% NH4I + 30 wt% EC. Finally, the solution was poured into polypropylene petri dishes at ambient temperature, followed by vacuum drying to remove the remaining traces of methanol.
After drying the films, we could easily pill out the free-standing polymer electrolyte films and characterize the measurement like conductivity, dielectric, X-ray diffraction (XRD), polarized optical microscopy (POM), and Fourier transform infrared (FTIR) analyses. Finally, we have fabricated a supercapacitor using maximum EC-doped polymer electrolyte film and GO as an active material.
Synthesis of GO
A pure graphite rod extracted from dry cell was used as a source material as an electrode and dilute sulfuric acid as an electrolyte. The electrochemical exfoliation method was used to extract GO from graphite rod. Graphite and platinum rods were dipped in an electrolyte with a distance of 0.5 + 0.1 cm. On applying a 10 V DC potential between the rods, exfoliation started and continued for 4 h. Finally, dispersion was washed and filtered several times to remove the impurities and lighter particles. The prepared GO powder was dried at 80°C for 24 h. 23
Supercapacitor fabrication
EDLC was prepared using polymer electrolyte having maxima and GO as an active material coated on a current collector graphite sheet in a configuration.
Electrode (GO)/polymer electrolyte film/electrode (GO)
To fabricate an EDLC, symmetric current collector graphite sheet was cut into 1 × 1 cm2. Approximately 1 mg GO was coated on a current collector with binder PVDF-HFP (dissolved in acetone) in a ratio of 90:10, followed by vacuum drying at 90°C of prepared electrodes in an oven. Maximum conducting EC-doped polymer electrolyte film was sandwiched in between GO-based electrodes to measure the cyclic voltammetry (CV) and low-frequency impedance spectroscopy to calculate the capacitance value of a cell.
Results and discussion
Impedance spectroscopy
The room temperature Cole–Cole impedance plot at high-frequency spectroscopy of different wt% ranging from 10 to 80 of EC plasticizer was done using CH instrument (model 604D; Austin, Texas, USA), as shown in Figure 1.
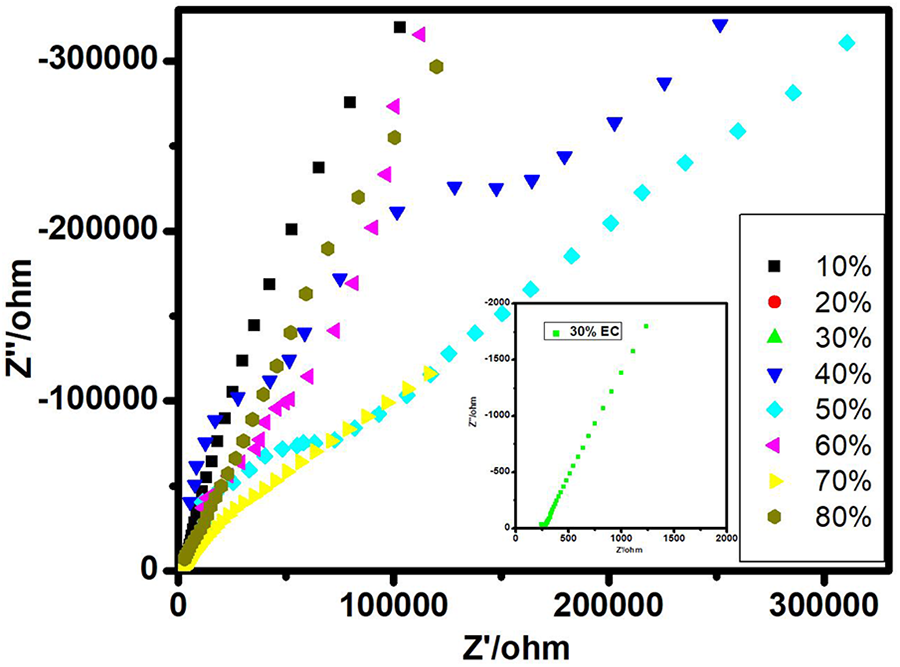
Room-temperature Nyquist plot of different wt% of EC in PVP + 40 wt% NH4I.
The plots of complex impedance in between
Conductivity
The ionic conductivity of an EC doped on polymer electrolyte (PVP + 40 wt% NH4I) reported elsewhere was carried out using impedance spectroscopy using formula.
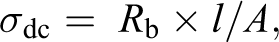
where
Conductivity with an increased amount of EC concentration in (PVP + 40 wt% NH4I) polymer electrolyte films.
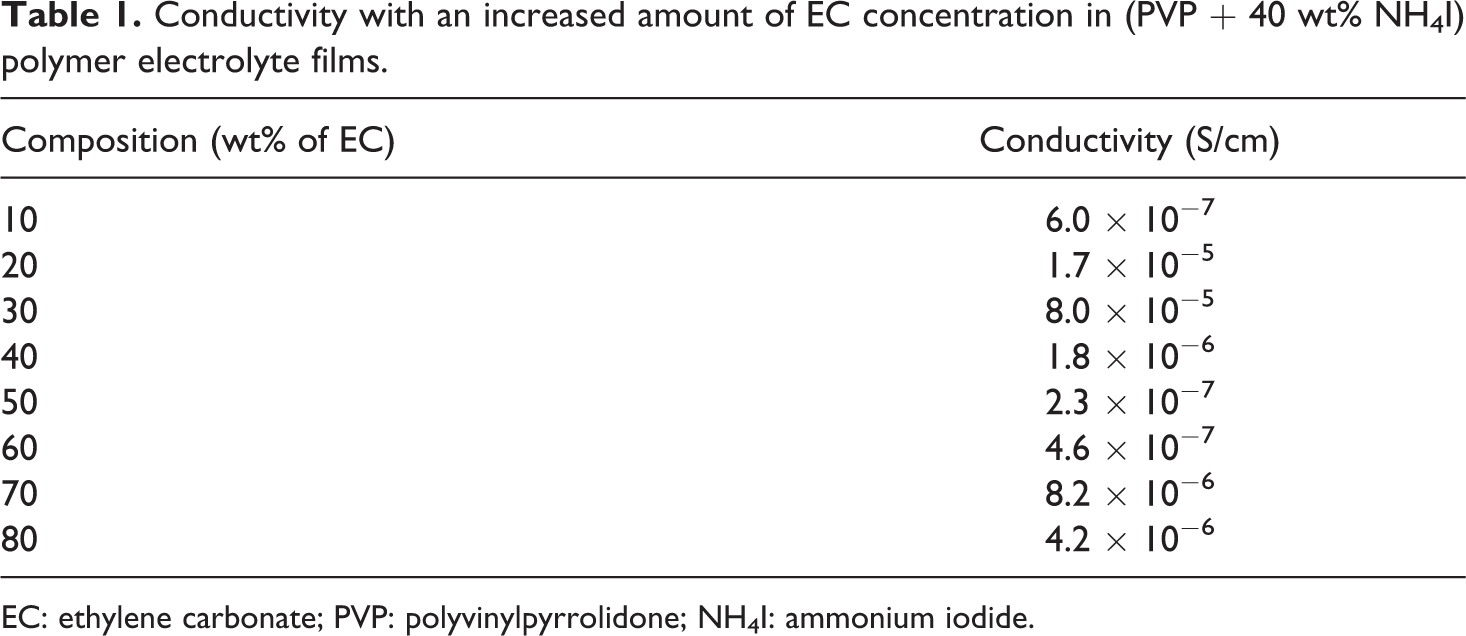
EC: ethylene carbonate; PVP: polyvinylpyrrolidone; NH4I: ammonium iodide.
The increase or decrease in the conductivity of the polymer electrolyte films as a function of EC is presented in Table 1. It is a well-known phenomenon that ionic conductivity is directly proportional to the number of charge carriers (

From Table 1, it is clear that with the increase wt% of EC in (PVP + 40 wt% NH4I), conductivity increases up to a mark 30 wt%, that is, 8.08 × 10−5 S/cm, which is the maxima of the composition of plasticizer. Then it continuously decreases up to mark of 60 wt% EC. Fascinatingly, again increases slightly up to 80 wt% EC.
Initially, the addition of EC in PVP + 40 wt% NH4I projected toward the increases in the degree of salt dissociation, hence, increases the ions in the electrolyte. Incorporation of EC plasticizer in a polymer matrix gives ascends to the amorphicity and decreases the viscosity which is the most favorable condition for ionic conduction in a polymer matrix. Additional plasticizer also plays a role in rising the change in the polymer chain by coordinating with the solvent and ion conducting interfacial phase flexibility. In the plasticized electrolyte, the NH4+ ions become coordinated to both the polymer and the plasticizer EC, which results in increase in the conductivity up to 30 wt% EC plasticized with the polymer electrolyte.
After 30 wt% EC on PVP + 40 wt% NH4I, conductivity decreases up to 60 wt% plasticizer. This decrease in the conductivity is probably due to the steric hindrance in between ions which causes obstacle in movement of ions toward respective electrodes. Hence, the overall conductivity decreases up to 60 wt% EC. With further increase in the concentration of EC in PVP + NH4I, conductivity slightly increases of one order up to 80 wt% EC which is probably due to the secondary interaction (hydrogen bonding). After 80 wt% EC concentration, film could not be pilled out from the petri dish.
Dielectric constant
To set up a relation between the dielectric response and the ionic conductivity, we have also calculated the dielectric constant of different wt% EC on polymer host (PVP + NH4I) at three different frequencies, that is, 8301 Hz, 99,610 Hz, and 825,200 Hz (marked with black, red, and blue) colors, respectively, as shown in Figure 2. The values of dielectric response were calculated by using the formula
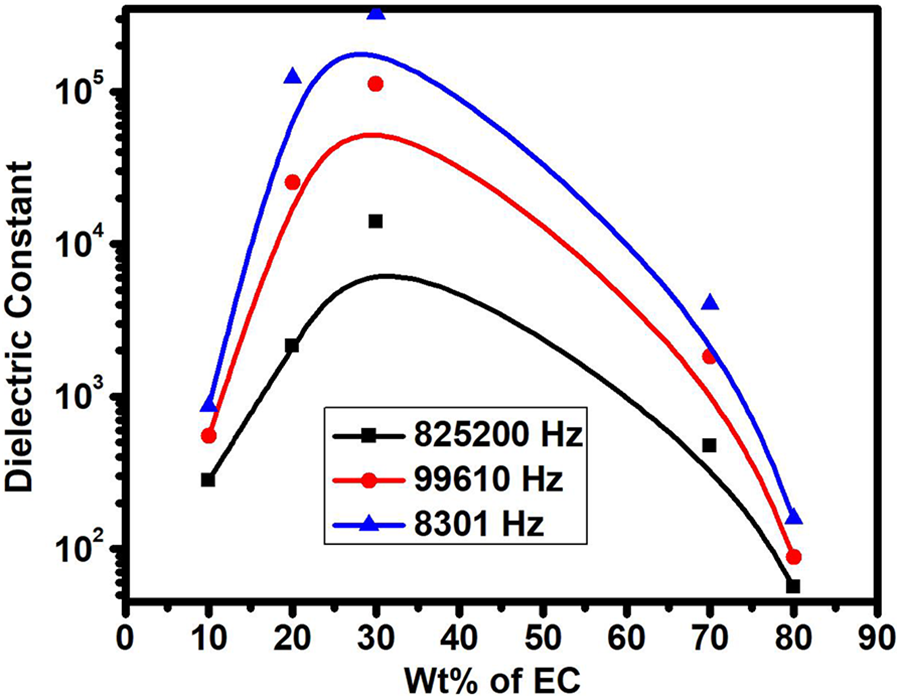
Dielectric constant versus EC concentration in PVP + 40 wt% NH4I at different frequencies.
The dielectric constant values increase with the increases in the wt% EC and increase in the conductivity up to a mark 30 wt% EC and follow the similar trend as we have observed in conductivity data. Hence, Figure 2 follows the similar trend of conductivity data, that is, on increasing the wt% of EC the dielectric constant increases up to a mark 30 wt% EC which is due to the increases in the degree of salt dissociation, ascends to the amorphicity and decreases the viscosity, then decreases up to 80 wt% of EC. It is well-known that increase in the dielectric constant leads to an increase in the ionic conductivity and decrease in the electronic conductivity. The more the dielectric constant values, the larger the ionic conductivity.
X-Ray diffraction
To check the nature of the polymer electrolyte to be either crystalline or amorphous in nature, we have performed XRD as shown in Figure 3. The host polymer, that is, PVP + 40 wt% NH4I shows a peak at 2 Θ value of 23.18°C, some well-known semicrystalline nature of polymer electrolyte film (Figure 3, black line). On the other side, adding plasticizer in the polymer matrix, that is, PVP + 40 wt% NH4I + 30 wt% EC shows a broad hollow at same 2
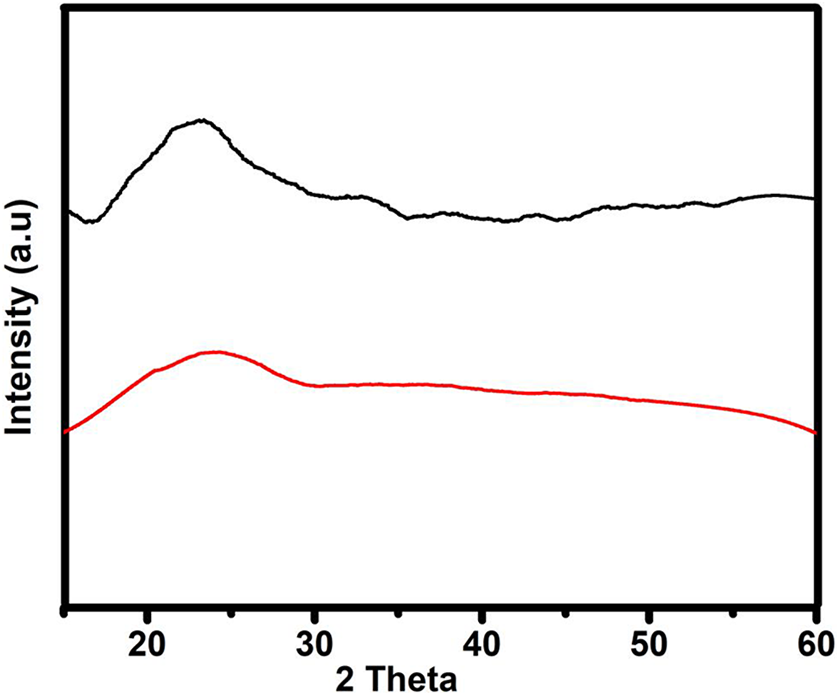
XRD pattern of PVP + 40 wt% NH4I (black line) and PVP + 40 wt% NH4I + 30 wt% EC (red line) polymer electrolyte films.
Polarized optical microscopy
Polarized optical micrograph (POM) images of pure PVP, PVP + 40 wt% NH4I, 25 and PVP + 40 wt% NH4I + 30 wt% EC were recorded using POM instrument (Motic-model no. BA310 Pol, Carlsbad, California, USA) at 40× magnification. The micrographs of pure PVP and PVP + 40 wt% NH4I are presented in Figure 4. We have seen that the pure PVP film exhibits the rough crystalline nature and PVP + 40 wt% NH4I exhibits more dark region, suggesting the amorphous nature of electrolyte (Figure 4(a) and (b)). Interestingly, the maximum conductivity of the polymer electrolyte (PVP + 40 wt% NH4I + 30 wt% EC) shows highly dense amorphous morphology (more blackish nature) which affirms reduction in crystalline nature (more amorphous). As we know from the reported literature that amorphous morphology of a polymer film leads to the high ionic conductivity23,24–27 and hence POMs confirm our results obtained in XRD measurement.
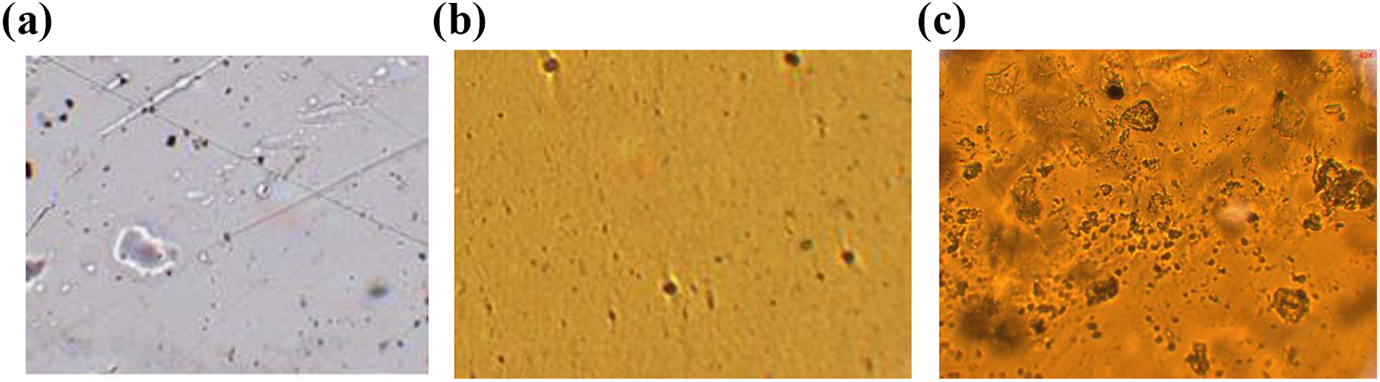
POM of pure PVP matrix: (a) PVP + 40% NH4I; (b) PVP + 40 wt% NH4I + 30 wt% EC; and (c) polymer electrolyte films.
FTIR analysis
In Figure 5, the spectrum of PVP (black color) contains the peaks at 1737, 1640, 1495, 1424, 1443, 1462, 1294, 1320, 1294, and 844 which corresponds to C=O stretch, –C=C– stretch, C–C stretch, C–H bend, C–N stretch, and C–O stretch. The FTIR spectra of PVP + 40 wt% NH4I have been reported elsewhere by our group 27 with peak assignment. On adding 30 wt% EC on the host polymer (PVP + 40 wt% NH4I), no extra peak was observed which affirms the composite nature of the plasticized polymer electrolyte film.
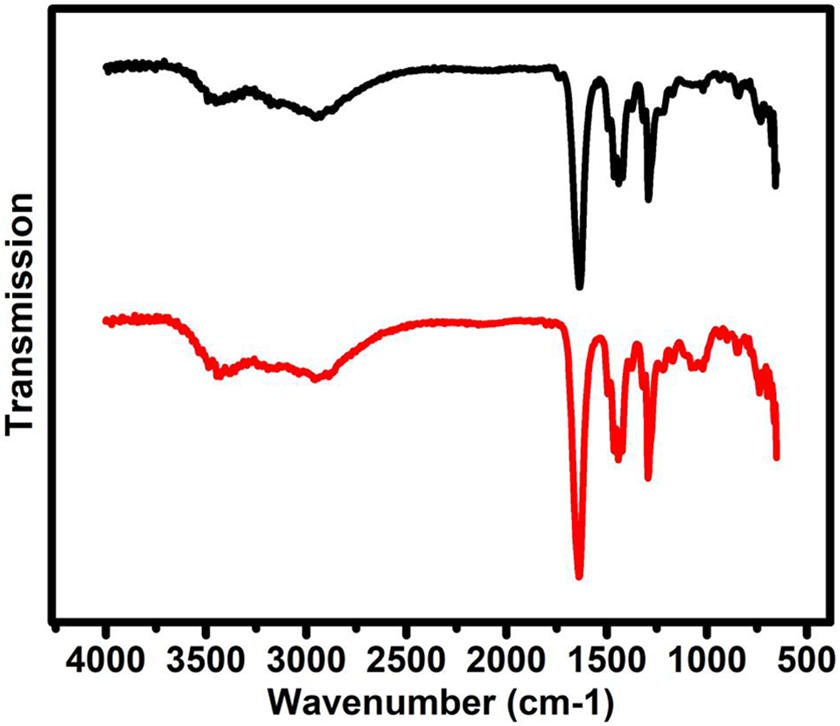
FTIR spectra of pure PVP (black) and maximum conducting PVP + 30 wt% EC (red) polymer electrolyte films.
Cyclic voltammetry
Cyclic voltammetry (CV) was carried out using a CH Instrument workstation (model 604D; Austin, Texas, USA) to calculate the capacitive behavior of (PVP + 40 wt% NH4I + 30 wt% EC) optimized polymer electrolyte film. Figure 6 depicts the CV profile of supercapacitor using optimized polymer electrolyte in our laboratory and GO as an active material. Figure 6 clearly shows that the hysteresis curve shape predicts the capacitive behavior of a cell. No ideal rectangular shape is obtained in a CV graph, which suggests that the solid polymer electrolyte in a supercapacitor has poor interfacial contact with electrodes.
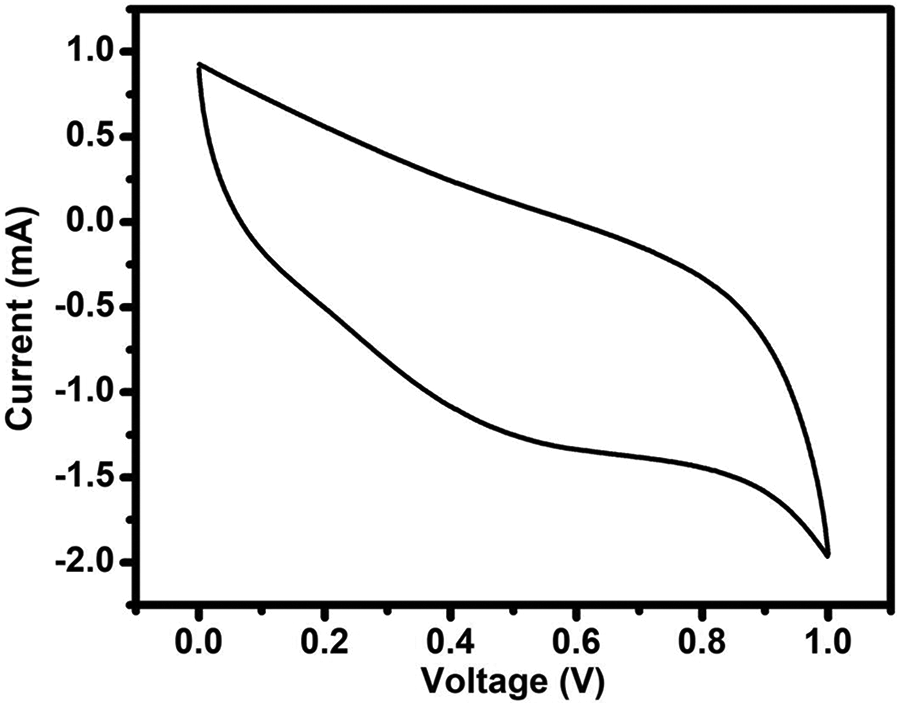
Cyclic voltammogram of EDLC fabricated using the maximum conducting (PVP + 40 wt% NH4I + 30 wt% EC) film recorded at a scan rate of 0.05 mV/s.
Although the CV of the present work did not show the complete rectangular shape but still showing the highest capacitance of 27.6 F/g at 50 mV/s scan rate using the following formula:
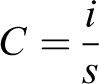
where
Low-frequency impedance spectroscopy
Low-frequency impedance spectroscopy was carried out using a CH Instrument workstation (model 604D) to calculate the capacitance at low frequency, that is, of PVP + 40 wt% NH4I + 30 wt% EC optimized polymer electrolyte film using the following fomula:
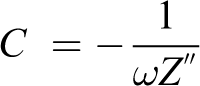
where
Figure 7 depicts the low-frequency impedance spectroscopy of supercapacitor using optimized polymer electrolyte film and GO as an active material coated on a current collector. From the graph it is clear that at higher frequency a semicircular spur is obtained, which reveals
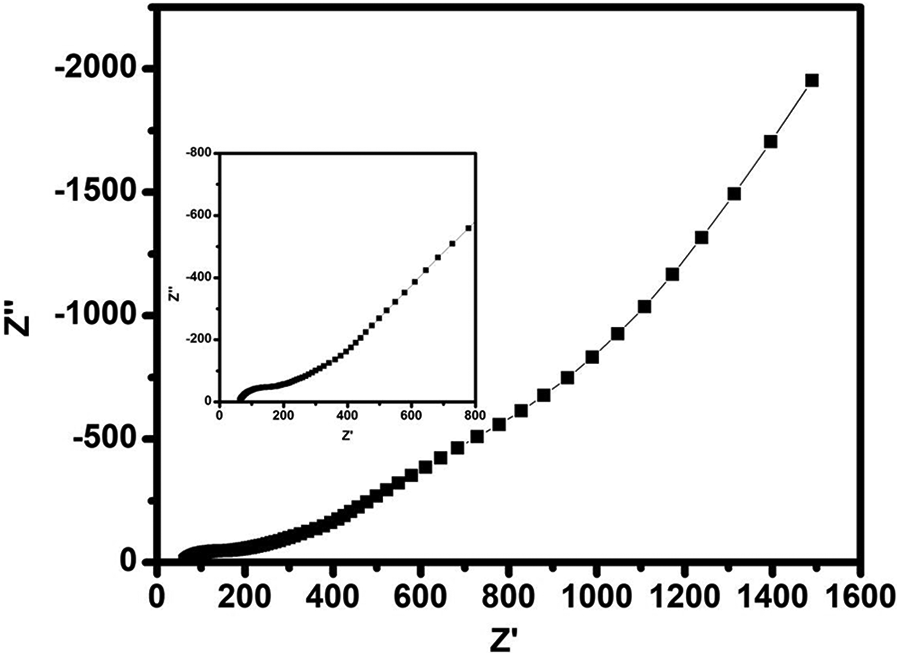
Low-frequency impedance spectra of EDLC containing maximum conducting (PVP + 40 wt% NH4I + 30 wt% EC) polymer electrolyte film.
Conclusion
EC-added optimized polymer electrolyte (PVP + 40 wt% NH4I) was prepared by a simple approach solution casting technique. On adding EC ranging from 10% to 80%, conductivity enhances up to 30 wt% EC on (PVP + 40 wt% NH4I) and reaches maximum 8.08 × 10−5. Additionally, dielectric studies follow the same trend as shown in the conductivity graph, which predicts that the ionic conductivity is directly proportional to the dielectric constant. XRD and POM analyses reveal the amorphousity on adding EC in PVP + 40 wt% NH4I. FTIR spectra affirm the composite nature of the 30 wt% EC-doped film on PVP. Supercapacitor based on maximum EC-doped polymer electrolyte showed better performance at the highest capacitance of 27.6 F/g at 50 mV/s.
Footnotes
Acknowledgement
Prof. Nanda G Sahoo acknowledges the financial assistance from NMHS research grant, GBPIHED, Kosi-Katarmal, Almora.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the NMHS research grant [NMHS/MG-2016/002/8503/455/73/290], GBPIHED, Kosi-Katarmal, Almora.
