Abstract
In this study, a biocomposite material derived from rice husk filler (RHF) reinforced for polyvinyl chloride (PVC)/epoxidized natural rubber (ENR) blend was developed. RHF particles obtained as a byproduct from the agricultural industry were used as environmentally friendly fillers. ENR was applied as a plasticizer and compatibilizer to improve the compatibilization between PVC matrix and RHF. Regarding the influence of RHF content, RHF particle size, and ENR amount on the properties of such biocomposite, the mechanical and thermal properties were analyzed. The obtained results implied that the combination between PVC, ENR, and RHF in an optimized ratio of 90/10/20 (wt%) can produce a novel biocomposite with enhanced properties, which can offer good advantages for various applications in requirement of environmental protection.
Keywords
Introduction
The fast development of world population along with upgraded living conditions has been causing an impressive increase of waste material. In this regard, a large amount of nonbiodegradable plastic wastes are being produced every day without optimal solution for treatment or further usage. Polyvinyl chloride (PVC) is a major thermoplastic, which is annually manufactured more than 22 million tones. 1,2 Due to its easy process and thermal stability compared to that of polyethylene and polystyrene, the PVC-based products are currently common in various applications. 3 –6 For further enhancing polymer properties effective for specific application as well as reducing the use of polymer amount for minimizing environmental pollution, the combination of polymer matrix with fillers has been extensively announced as the economic approaches. 7 –10 Regarding the environmental protection and ecological sustainability, the polymer composites based on natural materials as biofillers have led to widespread concern. In this context, the use of composites containing natural fillers with high cellulose content, such as wood flour, hemp, bamboo fibers, kenaf, sisal, and rice husk filler (RHF), has been increasingly attracting the consideration of industry to reduce environmental degradation due to their advantages, such as abundant availability, versatility, low cost, biodegradability properties, and high mechanical properties. 11 –15 In addition, as compared to a polymer composite using inorganic fillers, the polymer composites from natural fillers have better rigidity, resistance, less density, and low hardness, thereby reducing wear and tear during processing. Among biofillers for polymer composites, RHF, a byproduct of the agricultural industry, is currently attracting as an interesting reinforcing filler to combine with the polymer in an attempt to enhance the mechanical properties of the resulting composites. 16 –18 In particular, the social concerns for the protection of trees from forest resources have implied RHF as a potential inexpensive substitute to replace traditional wood fillers in a wide number of composite applications. The combination between PVC matrix and RHF has been reported with high interest in composite fabrication because of their economic advantages and the protection of the environment. Chand et al. studied the dielectric behavior of PVC/RHF composites. 19 Kamel employed rice straw for the fabrication of PVC composite. 20 The influences of the pretreatment process for rice straw, PVC content, pressing pressure, and temperature on the mechanical behavior and water absorption of the resulting composites were investigated. The remaining issues for such types of composites are that the use of RHF in PVC matrix will lead to the reduction of tensile and flexural strength, strain and modulus due to the poor interfacial interactions between fillers and matrix. Therefore, the pretreatment of fillers or the use of compatibilizers is necessary to produce an efficient composite material for applications.
Previous literature reported that epoxidized oils have been widely used as nontoxic plasticizer and stabilizer for PVC. 21 –23 They have good compatibility with significance to improving the physical properties of PVC product. Moreover, the presence of epoxidized groups can adsorb the hydrogen chloride released from PVC during decomposition, thereby increasing the thermal stability of PVC. Among epoxidated elastomers, epoxidized natural rubber (ENR) has been emerging as a new polar polymeric plasticizer, which could generate good miscibility with the polar polymers as PVC. The compatibility between ENR and PVC is gaining acceptance since the epoxy groups in ENR gave a stiffer polymeric plasticizer for PVC in comparison to other macromolecules plasticizers. 24 The compatibility of ENR with PVC has been demonstrated to depend on the epoxidation degree, in which it is confirmed that ENR50 was well miscible throughout the composition range while ENR25 was only partially miscible. 25
In an effort to improve the properties of PVC/RHF composite, we employed ENR as a compatibilizer for the first time. The obtained result indicated that the use of the ENR can significantly enhance flexural and impact strength along with thermal properties of composite materials.
Materials and methods
Materials
PVC-SG660 was purchased from Thai Plastics and Chemicals Public Co. (Ho Chi Minh, Vietnam). NR latex (dry rubber content [DRC] = 60%) was provided by Van Thanh Co. (Ho Chi Minh, Vietnam). Polyethylene (PE) wax, stearic acid, and heat stabilizer (PVC GY-TM181) were provided by Go Yen Chemical Industrial Co. (Kaohsiung, Taiwan). Hydrogen peroxide, formic acid, ethanol (98%), and sodium carbonate were purchased from Samchun Co. (Seoul, Korea). Arkopal (nonylphenyl-polyethylene glycol), a nonionic surfactant agent and free emulsifiers for the emulsion polymerization, was purchased from Merck (Darmstadt, Germany). Rice husk powder with particle size from 30 μm to 50 μm was provided by Khac Hung Co. (Ho Chi Minh, Vietnam). The main chemical compositions of RHF were investigated by fourier-transform infrared spectroscopy (FTIR), as reported in our previous study. 11
ENR preparation
The epoxidation process of NR latex was performed using in-situ method in the presence of formic acid and hydrogen peroxide at 60°C for 5 h. In a specific procedure, 200 g of NR latex (60% of DRC) was firstly stabilized by 2 g Arkopal at 60°C followed by removing ammonia to obtain the solution with pH = 7. The solution was then added with 3 g of Arkopal (2.5%) and kept at 10°C. After that, 17.75 g of formic acid and 146.75 g of hydrogen peroxide were slowly added into the solution, respectively; 280 g of H2O was added into the reaction solution, which was then kept for the reaction at 60°C for 5 h. After the reaction finished, the temperature was reduced to room temperature. The acidic residue in the solution was neutralized by Na2CO3 (10%) to obtain a solution with pH = 7.
RHF pretreatment
The alkaline treatment of RHF is to remove some unexpected substances, such as lignin and pectin, to enhance the compatibilization between the cellulose phase in RHFs and the PVC polymer matrix. In a particular procedure, RHF was soaked in NaOH solution (2%) at room temperature for 24 h. After that, RHF was collected and cleaned many times with acetic acid and water, respectively, to achieve pH = 7, followed by drying at 80°C for 24 h before using for further experiments. In fact, the morphology of RHF before and after alkaline treatment was observed by scanning electron microscopic (SEM) images, as reported in our previous study. 11 In this context, before treating in NaOH, the RHF presented clustered impurities, including lignin and hemicellulose; however, these substances were removed to leave a clean surface of RHF after NaOH treatment.
Preparation of PVC/ENR/RHF composites
In a particular procedure, the heat stabilizer (2 wt% by wt of PVC), PE wax (0.3 wt% by wt of PVC), and stearic acid (0.8 wt% by wt of PVC) based on PVC content were preliminarily added to PVC powder and mixed using a blender with a rotation rate of 15,000 r min−1 at room temperature for 2 min. The samples of PVC, PVC/ENR, and PVC/NBR/RHF were prepared by melting process using an internal mixer (Haake Polydrive, Karlsruhe, Germany) at 170°C and 50 r min−1 for 6 min. Then, the molten mixture was placed and shaped in an aluminum mold (10 × 10 × 0.2 cm3) at 180°C and 1500 lbf in−2 for 3 min by a hydraulic press. The samples were then cooled down to room temperature using the cooling water system to avoid warping. These samples were cut into smaller specimens according to the standard for further characterization. The compositions for preparing all samples were listed in Table 1.
The composition of PVC/ENR/RHF composites.
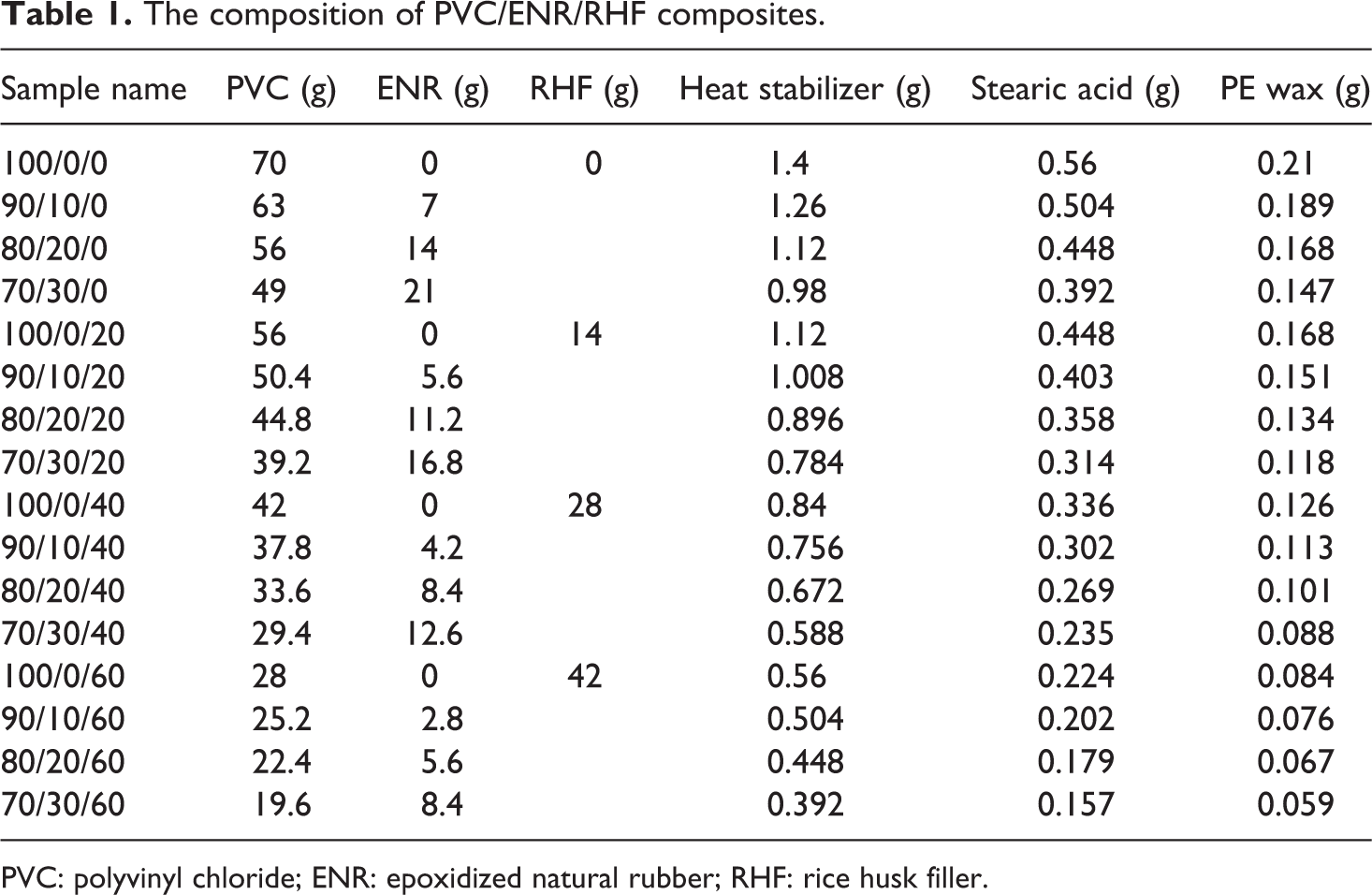
PVC: polyvinyl chloride; ENR: epoxidized natural rubber; RHF: rice husk filler.
Figure 1 illustrated the reactions happening during the preparation of the PVC/ENR/RHF composites. It was composed of the chemical reactions between PVC and ENR on high temperature to form ether crosslinks network between two polymers. This result was previously confirmed by FTIR with an increase of ether peak intensity of blend at high temperature and a decrease in epoxy ring peak of ENR. 25,26 Moreover, the Monsanto rheography during melt mixing also confirmed the chemical crosslinking of the PVC/ENR blends through an increase in torque that was not observed for a monopolymer counterpart. 25 Furthermore, Figure 1 also indicates the formation of hydrogen bonding between the surface hydroxyl groups of RHF and epoxy groups of the ENR.
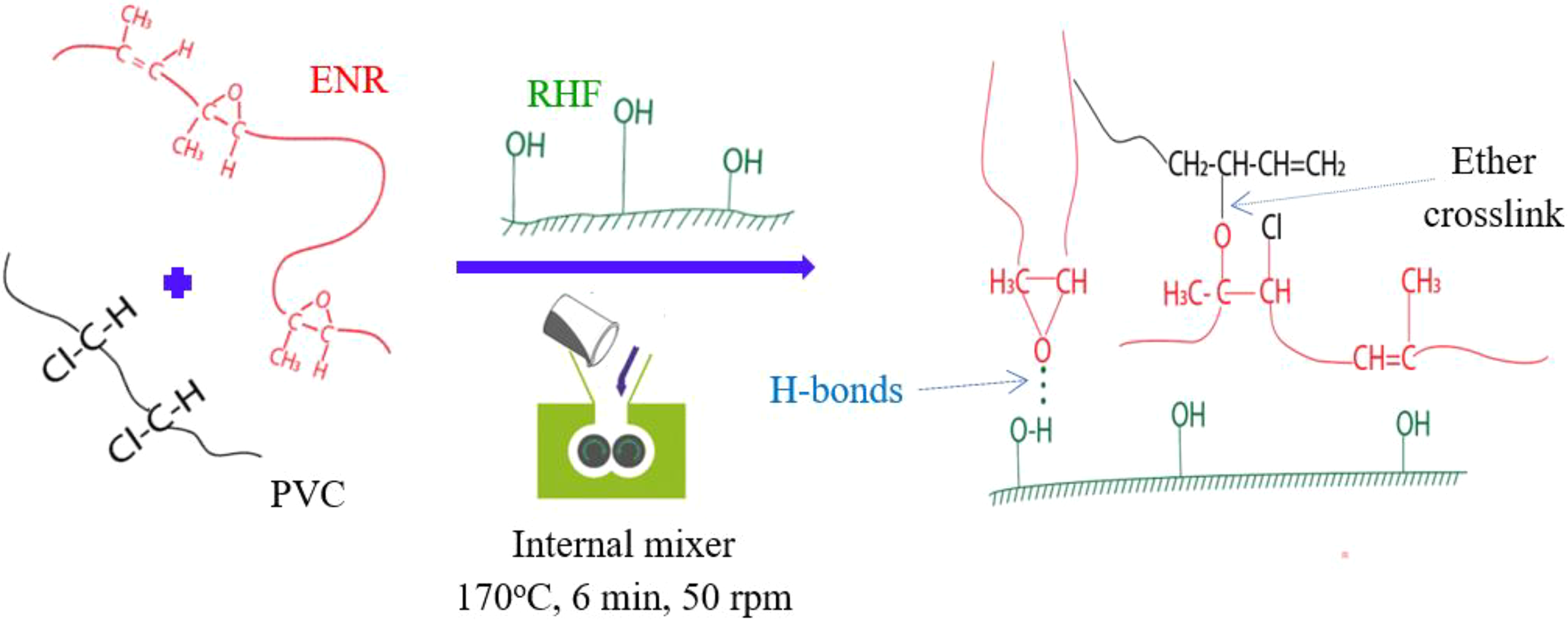
Schematic illustration for the synthesis of the PVC/ENR/RHF blend with the formation of the hydrogen bonding between the surface hydroxyl groups of RHF and epoxy groups of the ENR, and the ether crosslinks network between PVC and ENR.
Characterizations
The fractured surfaces of samples were observed using an SEM (Hitachi S-4800, Hitachi Co., Japan) to examine the phase structure of materials. Differential scanning calorimetric (DSC) measurements were investigated on DSC 1 equipment (Mettler Toledo Inc., Ohio,USA) at a heating rate of 10°C min−1 under nitrogen flow rate of 30 cm3 min−1. The thermal stability of the materials was conducted by thermogravimetric analysis (TGA) with a TGA Q500 analyzer (TA Instruments Ltd., New Castle, USA) under nitrogen atmosphere in the temperature range of 25–600°C at a heating rate of 10°C min−1. The flexural strength of all materials was evaluated by an AG-Xplus Series Precision Universal Tester (Shimadzu Inc., Japan) according to ASTM D790 standard. The impact strength of materials was evaluated by an IT504 Impact Tester (Tinius Olsen Inc., Horsham, Pennsylvania, USA) according to ASTM D256 standard. All samples were stabilized in a desiccator under vacuum environment for 24 h before testing. Five specimens were tested for each sample to achieve average values of mechanical parameters.
Results and discussion
NR is naturally abundant polymer having good physical properties and elasticity. The epoxidation of NR can create ENR structure with many epoxy functional groups on carbon chains, which can display as an effective compatibilizer in polymer composites. 27 –29 ENR material can be formed by the treatment of NR latex with a mixture of formic acid and H2O2 at 60°C for 24 h. 30 Generally, the percentage of epoxy groups can be evaluated using DSC analysis, which indicated that the glass transition (T g) value of the NR will be increased approximately 1°C if its epoxy groups increase 1%. 24 As shown in Figure 2, T g value of the NR was found to be around −62.7°C. After the epoxidation process, an additional second-order transition appears at −47.01°C, implying a large amount of epoxy group already attached on NR structure to produce ENR with approximately 16%.
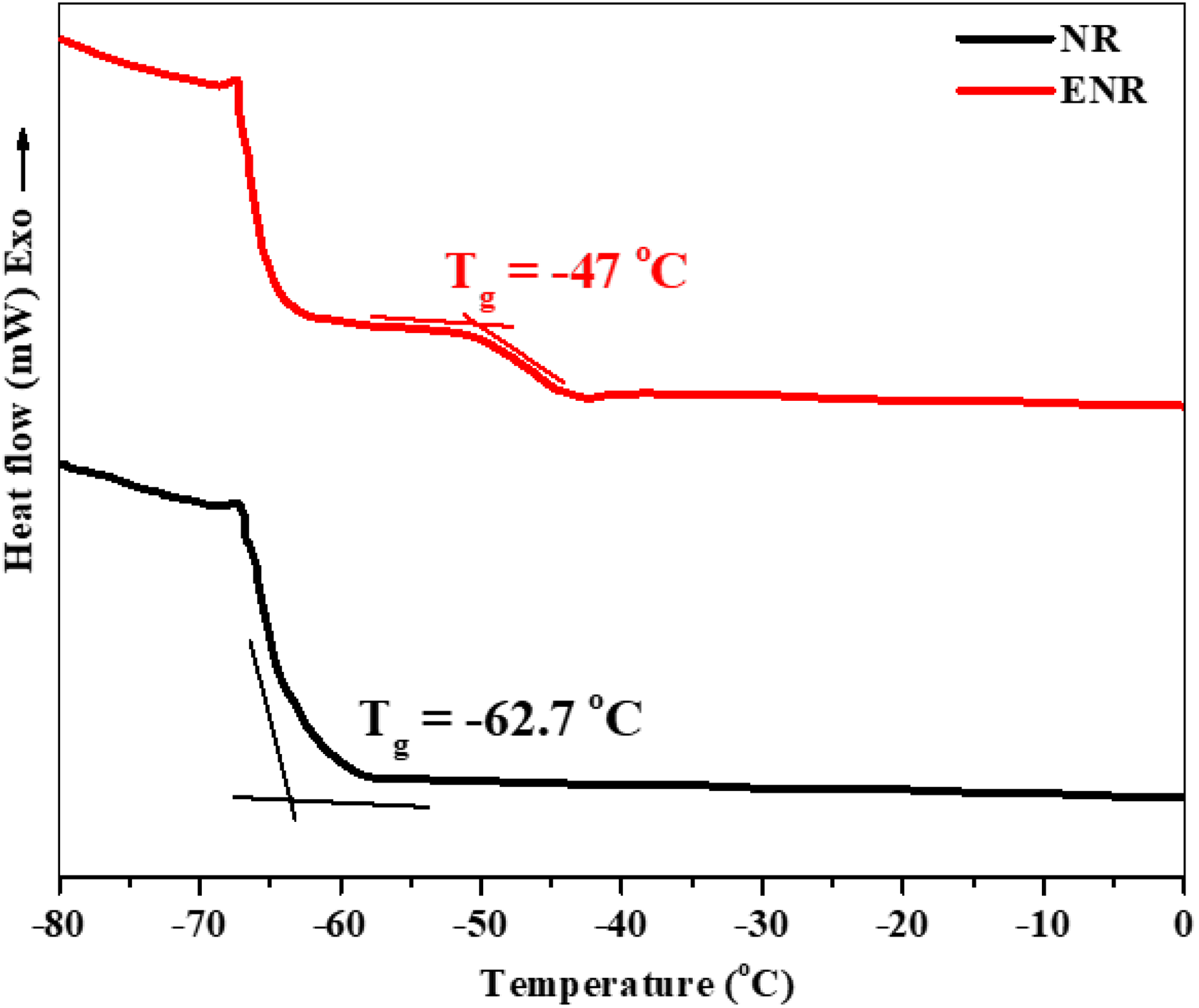
DSC measurements of the NR and ENR.
Mechanical properties of composites
The flexural properties of various composites materials were investigated to evaluate the influence of ENR and RHF content on mechanical performance of composite materials. Figure 3(a) shows the flexural modulus of materials. Results showed that the composites based on PVC/RHF possess higher modulus values as compared to PVC/ENR/RHF composites. This was due to the presence of soft material like ENR in PVC/ENR/RHF composites, which acted not only as a compatibilizer but also as a plasticizer, leading to the significant reduction of modulus value. The increase of ENR content on composite materials caused a significant decrease of modulus. Regarding the PVC/ENR/RHF composites, one can observe that the PVC/ENR (90/10) sample with the use of RHF at 20 wt% can produce the best modulus value as compared to other materials. The further increase of RHF content to 40 and 60 wt% led to the reduction of modulus. This indicates that RHF cannot well disperse into PVC, thus reducing interactions between fillers and matrix. Flexural stress depicts a similar trend to flexural modulus (Figure 3(b)). The addition of ENR into PVC/RHF composites decreased of stress value for all composite materials due to the plasticization of ENR. In addition, the increase of RHFs also generated the reduction of flexural stress. This was due to the different nature of hydrophilic fillers and hydrophobic polymer matrix. The availability of ENR at 10 wt% can enhance the compatibility between RHF and PVC to obtain a balance value of stress, especially for PVC/RHF (90/20) composite, which is much better than others.
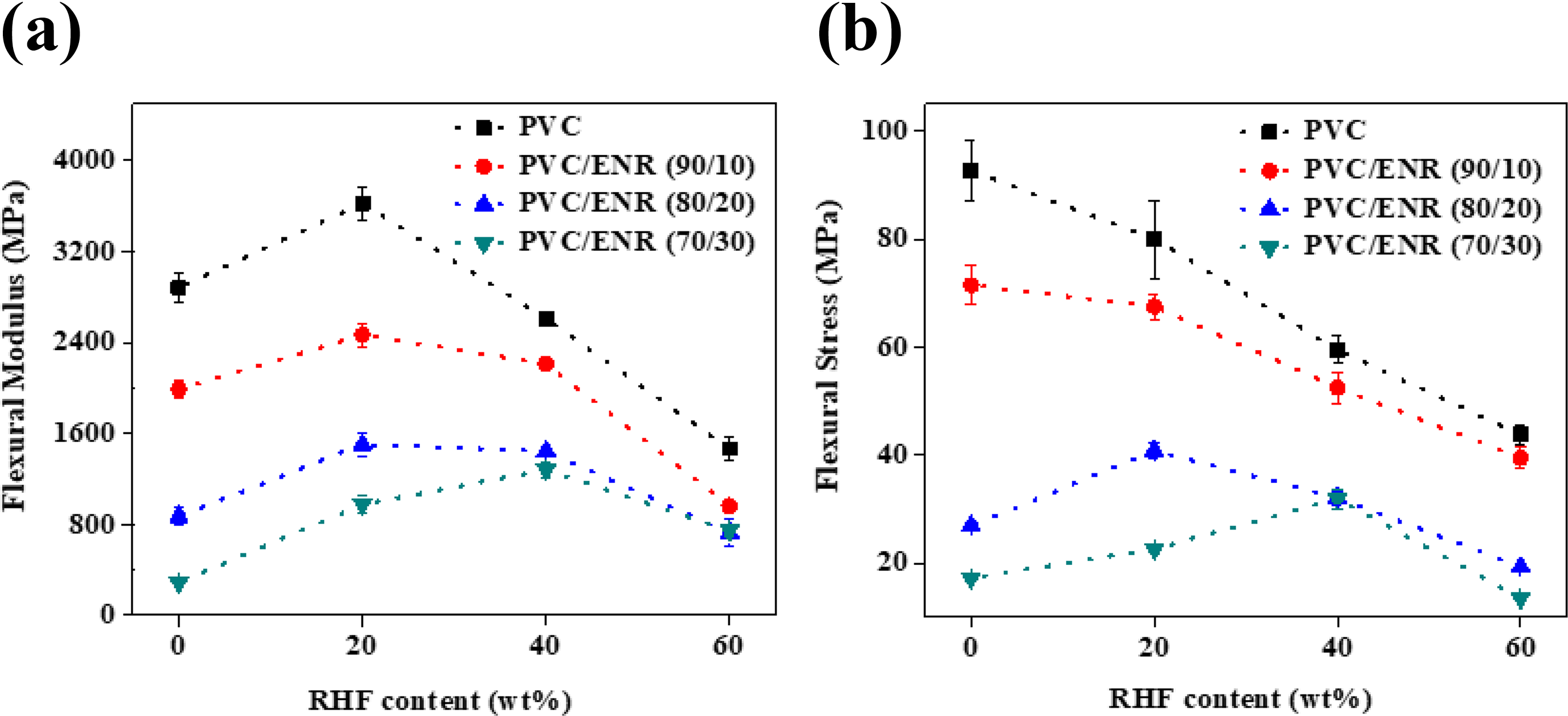
The flexural strength of different composite materials.
As shown in Figure 4, the impact strength of PVC/RHF composites shows lower value as compared to that of the PVC/ENR and PVC/ENR/RHF composites. The addition of RHF into PVC/ENR blends led to the noticeable reduction of the impact strength of the materials. It can be seen that the use of 20 wt% of RHF in PVC/ENR (70/30) showed better behavior than others. This is due to the formation of systems, in which a thermoplastic matrix is blended with an appropriate amount of elastomer and filler, leading to suitable compatibility and interfacial adhesion between the components in hybrid, thus resulting in a good resistance under impact loading. The blend systems with a suitable amount of rubber content and a good dispersion of rubber in the polymer matrix lead to promote elasticity in the blend. The ENR particles act as the nucleus to absorb energy caused by an external load and then free this energy to the surrounding elastic molecules, resulting in hardly breaking before the occurrence of matrix plastic deformation, thereby enhancing the impact strength of materials. 31,32 The balance values of impact strength for PVC/ENR/RHF can be achieved at 20 wt% of RHFs. The increase of RHF amount led to a significant decrease in impact strength in blend due to the poor dispersion of RHF in the matrix (Figure 5(d)), leading to the agglomeration of RHF in the matrix and the reduction of interfacial adhesion between RHF and matrix. This is due to the fact that ENR did not well support the dispersion of RHF into the matrix. This leads to hindrance of the energy adsorption/desorption of ENR, thereby reducing the impact strength.
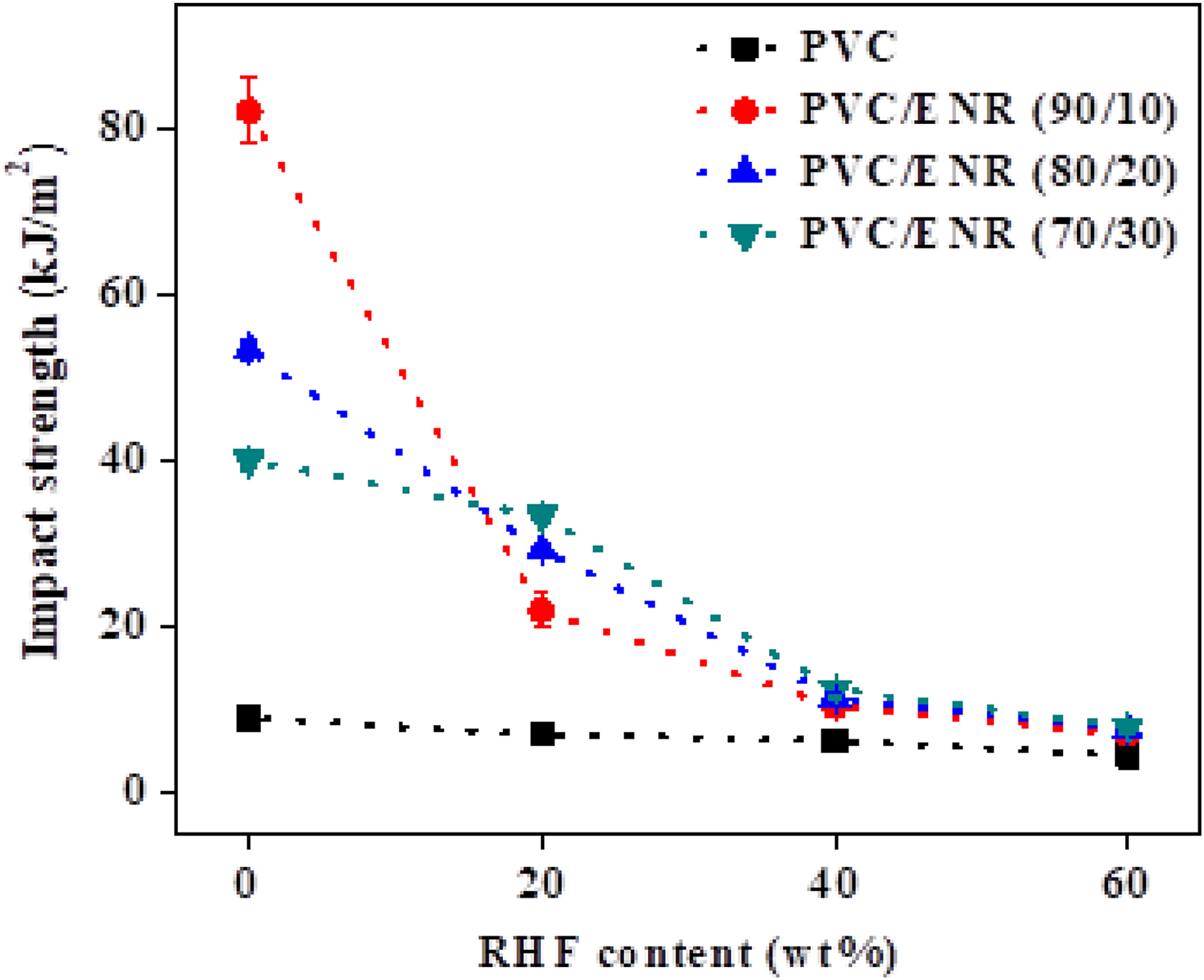
The impact strength of different composite materials.

SEM images of (a) PVC/ENR (90/10), (b) PVC/RHF (80/20), (c) PVC/ENR/RHF (90/10/20), and (d) PVC/ENR/RHF (90/10/40) composites.
Morphology
Figure 5(a) shows an SEM image of the PVC/ENR (90/10) blend in which small rubber particles are well dispersed into the polymer matrix. It is noted that the epoxidation degree of ENR decides the compatibility between ENR and PVC. 24 In this study, with 16% epoxidation degree, ENR possesses incomplete compatibility with PVC matrix, 24 resulting in the presence of ENR at the size of the microparticles, as observed by SEM image. However, due to the phenomenon of partial compatibility, ENR also dispersed to PVC matrix and acts as a nucleus to absorb energy and then free this energy to the surrounding particles under external load before to be broken. Therefore, the ENR particles effectively played a critical role in contributing the enhanced impact strength of composite materials. Meanwhile, SEM image of PVC/RHF (80/20) composite displayed a highly rough surface, which contained a large number of voids due to the detachment of RHF from PVC surface, implying low interactions between RHF and PVC polymer matrix (Figure 5(b)). The use of ENR as a compatibilizer for PVC/RHF composites could effectively connect between RHFs and PVC matrix, thereby promoting the dispersion of filler into the matrix (Figure 5(c)). In addition, the detachment behavior of RHF particles was reduced as seen at the fractured surface, implying the enhanced interactions between RHF and PVC matrix. However, when RHF content was increased in the PVC/ENR/RHF (90/10/40) composite, Figure 5(d) shows the significant agglomeration of RHF. This implied that ENR did not have enough ability to well disperse RHF into PVC matrix, thereby leading to poor dispersion of RHF along with the reduced strength properties of the composites.
DSC analysis
DSC measurement confirmed that pure PVC material has a T g value around 84.3°C (Figure 6(a)). The combination of PVC matrix with different amounts of ENR led to significant reductions of the gall transition temperature T g. In general, a T g value of approximately 80.4°C was found for PVC/ENR blends, which is 4°C lower than that of pure PVC. In addition, the increase in ENR amount up to 20 and 30 wt% does not cause a further decrease of T g for PVC matrix, implying that the use of 10 wt% of ENR is like to produce an optimal compatibilization between rubber phase and PVC matrix along a good dispersion of ENR particles into the polymer matrix. Therefore, the PVC polymer chains would be flexible and promote the dispersion of RHFs. In this regard, the addition of RHFs (20 wt%) into PVC/ENR (90/10) led to a slight reduction of T g for PVC/ENR blend (Figure 6(b)), indicating that the dispersion of RHF particles into matrix promoted the flexibility of PVC chains, thus generating a decrease of T g for matrix.
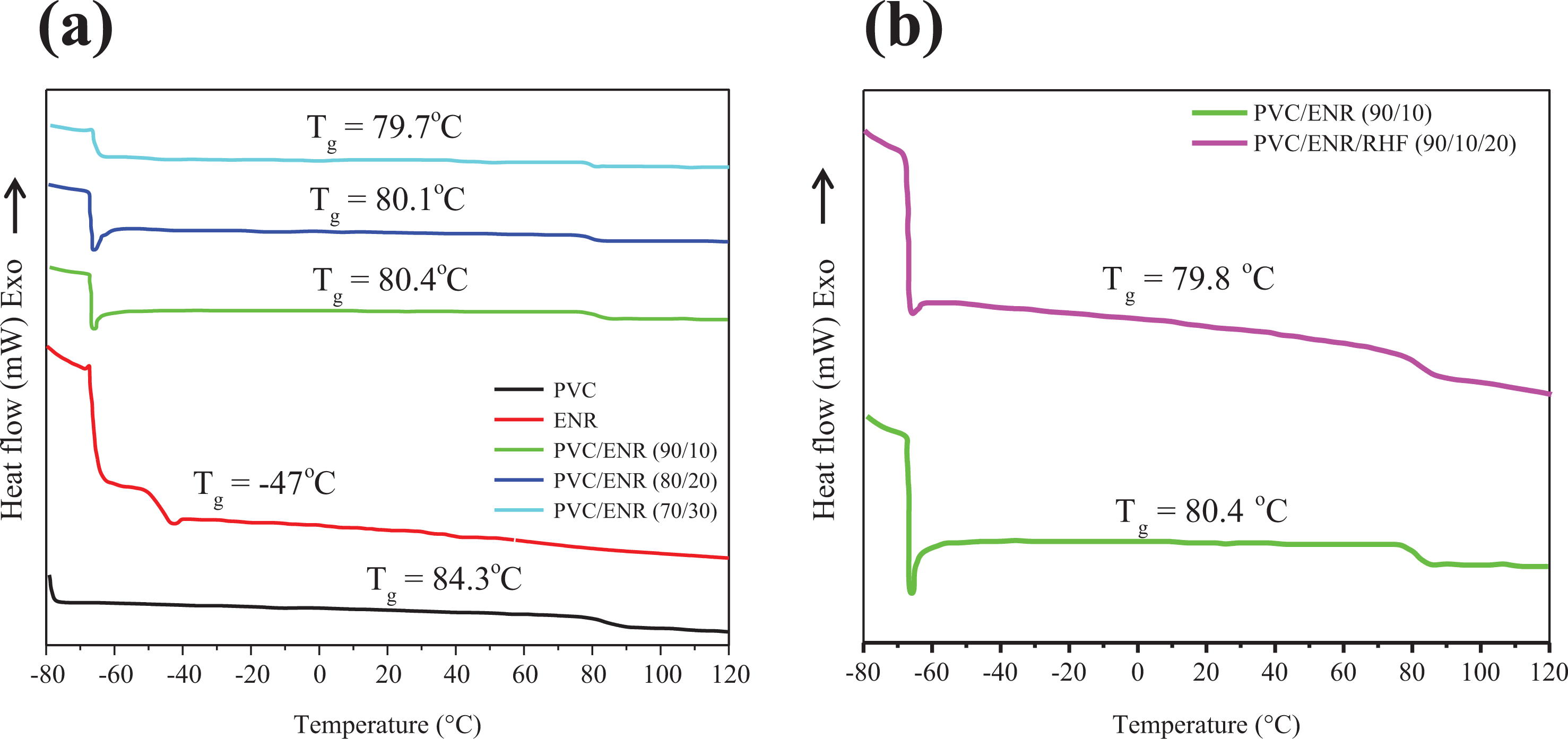
(a and b) DSC analyses thermograms of the pure PVC, pure ENR, PVC/ENR blends, and PVC/ENR/RHF (90/10/20) composite.
Thermal stability
Figure 7 plots the mass loss of different materials as a function of temperature for a constant heating rate of 10°C min−1. In the case of a pure PVC polymer, the thermal decomposition occurred through two steps. The first step happened in the temperature range 250–350°C, consistent with the weight loss of around 63.7%. This first step is due to the production of HCl molecules to generate (–CH=CH-CH=CH–). The second, occurring between 410°C and 480°C, is attributed to the degradation of (–CH=CH-CH=CH–). Meanwhile, the TGA of ENR indicated a slight weight loss in the range from 170°C to 350°C, due to evaporation of water and decomposition of epoxy rings. The maximum weight loss, around 400°C, is attributed to the decomposition of carbon chains. Due to the high amount of PVC matrix, the behavior of the PVC/ENR (90/10) composite is similar to that of pure PVC. The first weight loss of 2% at temperature from around 180°C to 250°C resulted from water evaporation and decomposition of epoxy rings. Then, the further weight loss of 60% observed in the range from 250°C to 350°C denotes the degradation of the PVC matrix. Finally, the weight loss of 30.5% in the range from 410°C to 470°C is assumed to be due to the degradation of polymer chains from remaining PVC and ENR. Regarding the PVC/ENR/RHF (90/10/20) composite, the weight loss starts at 50°C and extends up to 460°C and its behavior is almost similar to that of PVC and PVC/ENR composite.
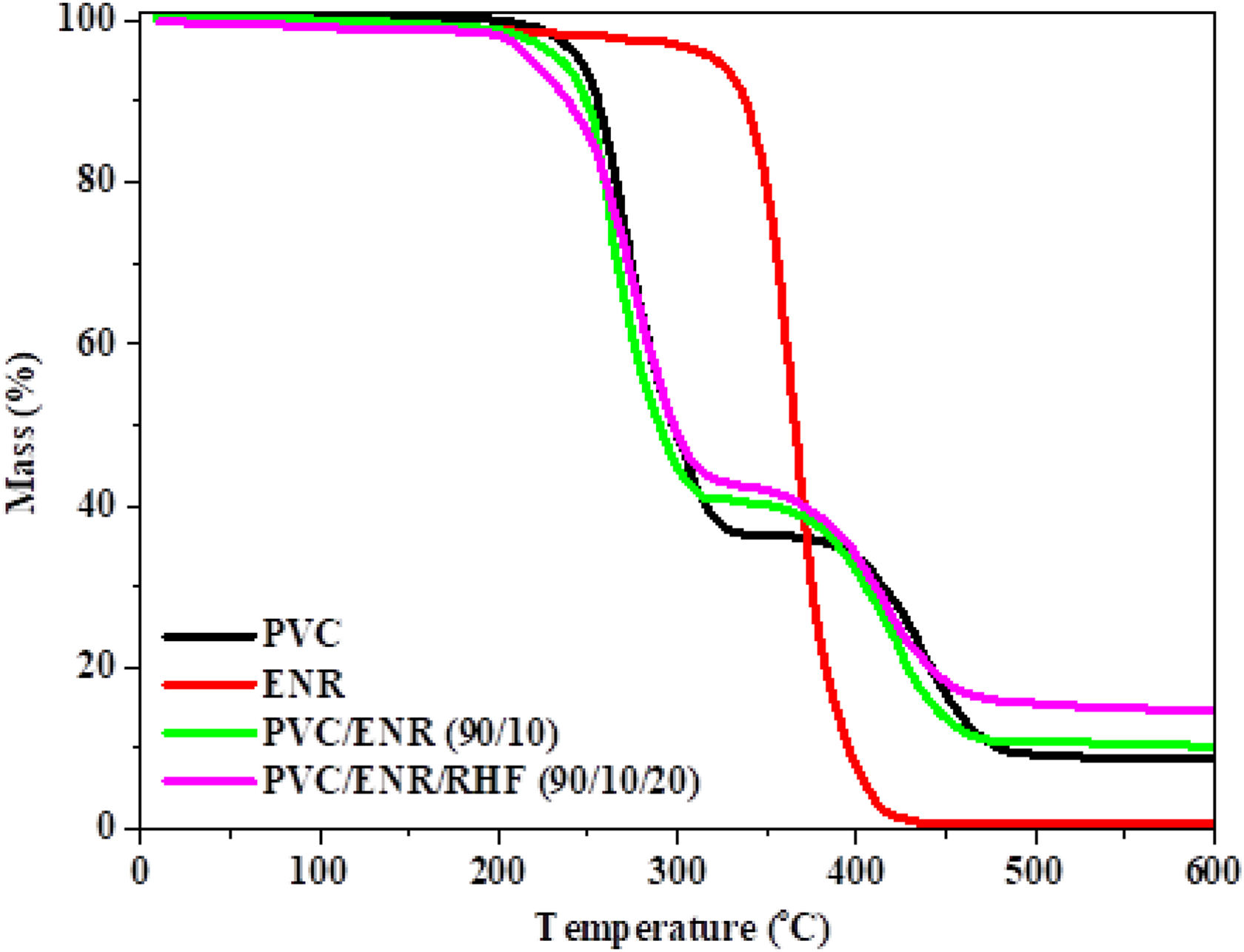
TGA curves of ENR, pure PVC, PVC/ENR (90/10), and PVC/ENR/RHF (90/10/20) composite materials.
It could be observed that the addition of ENR to PVC did not affect the thermal stability of PVC matrix although the thermal stability of ENR is higher than that of PVC, as mentioned above. Its thermal decomposition curve was similar to that of PVC because PVC occupies a higher proportion in the blend and the partial compatibility between PVC and ENR via the ether crosslinking was not enough to effect on the thermal property of this blend. However, after adding RHF to PVC/ENR, the first thermal decomposition state started earlier than that of PVC due to the disruption of the PVC/ENR blend crosslinking chain and the low compatibility between the PVC/ENR matrix and RHF. The second state of thermal decomposition for this blend started at the same decomposition temperature of PVC. However, the residue after decomposition was much higher than that of PVC/ENR due to the presence of inorganic components, such as silica in RHF that could not decompose during heating process.
Conclusion
The morphological structure, mechanical properties, and thermal stability of the composite materials based on PVC, ENR, and RHF were investigated. The presence of ENR displayed as a good compatibilizer to enhance interactions between RHF and PVC matrix. The PVC/ENR/RHF composite at a ratio of 90/10/20 provided the best balance of mechanical properties. This research may open up a new approach for the manufacture of a biocomposite with suitable properties in artificial wood field.
Footnotes
Acknowledgements
This study was supported by the Ton Duc Thang University (Ho Chi Minh city, Vietnam) and the LGPM and Chaire de Biotechnologie de CentraleSupélec, Université Paris-Saclay, France.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
