Abstract
A simple method for determining the presence of cadmium (Cd) in thermoformed and biodegradable flexible films and flour and starch samples from cassava, using graphite furnace-atomic absorption spectrometry (GF-AAS), is proposed. The biopolymers used in contact with foods can contain Cd due to contamination from the origin of the material used to make them. The nitric: perchloric acid digestion with reflux was optimized, and the statistical quality parameters were standardized. The GF-AAS instrumental conditions and the graphite furnace heating program were adjusted through pyrolysis and atomization curves, and the optimal temperatures were 530 and 1750°C, respectively. In addition, under optimal conditions, the working range was from 0.1 µg/L to 1.0 µg/L, with limits of detection and quantification of 0.02 and 0.07 µg/L, respectively. The precision was evaluated using intermediate precision and repeatability of the method, which showed standard deviations of 1.6–4.8% and 2.1–4.8%, respectively. The percentage of recovery of Cd ranged from 96.2% to 97.3%. The results obtained support the suitability of the method for determining the presence of Cd in these samples. Cd concentrations in flexible films and starch were below the quantification limit. The other samples (thermoformed and flours) contained less than 1 mg/kg (maximum level set by the NTC 4096), indicating that these polymers meet this standard and therefore can be used for food containers and packaging for fresh fruits and vegetables.
Introduction
Thermoformed and biodegradable flexible films can be made from cassava flours and starches, by adding substances, such as fique fiber, gelatin, poly(butylene adipate-co-terephthalate), green composites, polylactic acid, glycerol, plasticizer, cellulose, pullulan, chitosan, clay, and natural extracts. 1 –17 Nowadays, industries are trying to improve their products from natural and renewable sources to store, packaging and wrap to food products, 13 –16,18 such as thermoformed and flexible films (biopolymers) obtained from agricultural sources (cassava). These constitute a new and environmentally friendly industrial alternative (composting) due to their fast and easy biodegradability upon completion of useful life, and main purpose is to replace petroleum-derived plastic polymers that people use regularly and the waste associated with it. 2,4,6,7,10,16,17,19,20 These characteristics greatly benefit different environmental ecosystems.
An usual method for thermoformed manufacture is the compression molding technique, in which the material is placed into an open mold to which pressure and heat are applied. Single screw extrusion is used for the manufacture of biodegradable flexible films, and by turning the screw and applying heat, the material is pushed along and melted. 5,11,14,21
In developing countries, the accelerated process of industrialization combined with a fast population growth and the use of intensive agricultural techniques, and the inappropriate management of waste have led to an increase in the levels of substances considered harmful or toxic to living beings, such as heavy metals. These metals can cause serious damage and can enter the human body from food (migration), water, air, soil, or skin absorption, from emissions to the environment, anthropogenic sources (treated sewage discharges, mining operations), through contact with industrial fallout as well as agricultural activities, such as pesticide formulations, urban traffic, and contamination from chemical fertilizers and irrigation with inadequate quality water. 15,16,22 –25 The heavy metal migration caused by materials in contact with foods is a negative interaction between packaging and food. Additionally, heavy metals are nonbiodegradable, and they persist and accumulate in the environment and in organisms over long periods of time because they are not metabolized. 22,26 –30
The manufacture of biodegradable polymers may produce heavy metal contamination due to contamination of the raw material used for their processing (cassava flour and starch, fique fiber, polylactic acid, glycerine, etc.) or cross-contamination during manufacture, for example, during petroleum and nonpetroleum activities, from pots used for cooking and storage, during drying techniques for moisture content reduction process, and from utensils or contaminated water. 23,31,32 For this reason, a tracking process during manufacturing is necessary to determine the presence of toxic heavy metals in the biopolymers for the purpose of safeguarding public health by limiting exposure. 26,28 Among these metals, cadmium (Cd) is considered a toxic heavy metal to both plants and animals (is widespread in atmosphere, soils, and water) that can cause serious health hazards, affecting the gastrointestinal, cardiovascular, musculoskeletal, nervous, renal, and reproductive systems, and long-term exposure may cause mitochondrial damage and death. Cd bioaccumulates not only in individual organisms 23,24,33 –36 but also through the future use of biodegradable polymers in the packing and packaging of dry foods and other degradable products, as it enters the food chain. The United States Department of Health and Human Services has stated that an excessive Cd accumulation in humans may even cause cancer because Cd and Cd salts are considered a “possible human carcinogen.” 23,34 Additionally, certain plants can accumulate Cd in their tissues and this increases in plants that are grown in peri-urban areas and zones with soil contamination by irrigation using wastewater and sewage sludge. 27,37,38,39,40
Currently, there are no studies reporting the presence of Cd in thermoformed and biodegradable flexible films, and literature reports the presence of Cd in the cortex of cassava tubers, 32 cassava food crops, 41 and contaminated rice that causes the Itai-itai disease 42 by exposure to Cd-contaminated water used for irrigation or from farms impacted with petroleum and nonpetroleum activities.
The safety of the materials that come into contact food is evaluated by the quantity of substances that migrate from the biopolymer into food and fulfil the requirements in the legislation on foods. The legislation does not allow the migration of toxics substances from reaching unacceptable levels as well as on maintaining the integrity of the foodstuff, thereby preventing health hazard, contamination, and changes in the composition and sensory properties of the food. 26 In the current Colombian regulation NTC 4096, 43 a maximum level of Cd of 1 mg/kg is allowed for plasticizers. Additionally, the Agency for Toxic Substances and Disease Registry 34 estimated that the average Cd intake in American is 30 µg/day and only one-tenth of this amount is absorbed into the tissues. Because of thermoformed and flexible films can be used as containers of foods, Cd levels should be monitored in these materials.
Numerous techniques have been used to determine the concentration of metals in different samples, such as graphite furnace–atomic absorption spectrometry (GF-AAS). GF-AAS is a good alternative for the determination of trace elements such as Cd due to its high sensitivity, with limits of detection in the order of micrograms per liter. 44 –47 Sample preparation involving acid digestion was employed in this study. 41
The purpose of this study is the quantification of Cd in samples of thermoformed and biodegradable flexible films and their raw materials from a standardized technique of GF-AAS, after a sample acid digestion treatment with nitric: perchloric acid mixture.
Experiment
Samples
Thermoformed (MBRA-383, MPER-183, CM 523-7, CM 7951-5, CM 4574-7, NATAIMA 31, HMC 1) and biodegradable flexible films (SM 707-17, SM 1498-4, and CM 7138-7, all pristine and hydrolyzed samples) were previously prepared from cassava flour and starch, respectively. Acronyms correspond to the name of the cassava varieties from which were made.
Prior to the analytical determinations, all samples were cut manually and thermoformed samples were macerated down to a particle size smaller than 1.135 mm (Sieve Newark USA Standard Series No. 18). Then, the water content was eliminated from thermoformed and biodegradable flexible films by drying at 70°C in a furnace (Fisher) for 4.5 and 4 h, respectively.
Reagents
All the reagents were of the highest analytical grade. Deionized water Millipore Milli-Q system was used throughout. Nitric acid (HNO3; 65%, Merck, Germany). Cd standard solution (1000 mg/mL, Titrisol Merck). Standard working solutions (0.1–1.0 µg/L for Cd) were prepared from the stock solution immediately before use.
Acid digestion with reflux
The process of digestion was performed using a mass of 1.0 g of the sample dissolved in 20 mL of a mixture containing HNO3 (65%, Merck): perchloric acid (48%, Merck) prepared in a 3:1 ratio. The solution was heated at 70°C for 3 h for the thermoformed and 50°C for 45 min for the biodegradable flexible films. After cooling off, they were filtered through a Gooch crucible (Schott Duran glass porosity 2), stored in polyethylene containers at 4°C, and finally, analyzed using GF-AAS (Thermo AA S4, Thermo Fisher Scientific). 30,32,37,45,47,48
Standardization of GF-AAS
The following statistical quality parameters were determined in order to carry out the standardization of the analytical method GF-AAS for the quantification of Cd in thermoformed and biodegradable flexible films: 49,50 (1) The linear range was evaluated by preparing a calibration curve of Cd concentration from 0.1 µg/L to 1.0 µg/L. Dilutions were prepared from a stock solution of Cd, 1000 mg/L (cadmium nitrate in HNO3, Merck). (2) The precision was evaluated at two levels: (a) for intermediate precision, seven calibration curves of Cd (0.1–1.0 µg/L) were analyzed over 7 days; (b) for repeatability, five calibration curves of Cd (0.1–1.0 µg Cd/L) were prepared and analyzed the same day. The concentration of Cd was determined using GF-AAS. (3) The sensitivity of the method was established by comparing the slopes of the calibration curves used to verify the precision. (4) For the limit of detection (LOD) and limit of quantification (LOQ), three calibration curves with Cd concentration ranging from 1.5 µg/L to 4.0 µg/L were prepared to obtain the standard deviation of the intercept and average of the slopes. 51 (5) The accuracy of the method was determined in terms of percent recovery. Known amounts (4, 8, and 12 µL) of a stock solution of Cd (1000 µg/L) were added to thermoformed (MPER 183) before the digestion treatment followed by GF-AAS analysis.
In addition to this standardization, (6) the stability was another parameter taken into account in order to optimize the method for the quantification of Cd in polymers using GF-AAS. Samples of a standard solution of Cd (0.8 µg/L) and a thermoformed (CM 4574-7) were analyzed using GF-AAS over 10 consecutive days.
Quantification of Cd
The quantification of Cd was carried out using atomic absorption spectrometry (Thermo AA S4) with a graphite furnace (GFS-97) and a deuterium background corrector. 20 µL of the concentrated sample, obtained from the acid digestion with reflux, was injected into the graphite cell. Previously, the optimal pyrolysis (500–800°C) and atomization (900–1800°C) temperatures were determined and programmed into the graphite furnace. Drying (100°C) and clean (2500°C) temperatures were programmed according to instructions in previous reports. 45,52 The technique used a normal electrographite cell (Thermo Elemental Solaar, Germany) with argon flowing at a rate of 0.2 L/min and 0.5-nm slit. No matrix interferences were encountered.
The concentration of Cd was determined by building a calibration curve with concentrations of Cd ranging from 0.1 µg/L to 1.0 µg/L and reading the maximum absorbance at a wavelength of 228.8 nm. These dilutions were prepared from a stock solution of Cd (1000 µg/L) in 0.2% HNO3 in a 5-mL volumetric flask. All samples were measured in quatriplicate, and the mean values were expressed as µg Cd/kg.
Statistics
The statistical SPSS analysis, using version 11.5 Windows and Microsoft Office Excel 2007, support the results of the standardization and implementation. Initially, the Shapiro–Wilk test and the test of equality of variances using Levene were applied. Finally, each calibration curve was statistically evaluated by applying one-way analysis of variance, Pearson’s correlation, the coefficient of determination and analysis of the relation. 53
Results and discussion
Thermoformed samples showed moisture values (after drying at 70°C in a furnace for 3 h) of between 3.71% and 5.80% (RSD lower than 3.4%). Biodegradable flexible films presented higher values of moisture (after drying at 50°C in a furnace for 45 min) at 7.81–10.35%, with relative standard deviation (RSD) lower than 1.96%, these levels being similar to those reported by Costa et al. 6 and Cha et al. 54
The graphite furnace heating program was optimized through pyrolysis and atomization curves. Figure 1(a) shows the change in absorbance at several pyrolysis temperatures and exhibits a maximum absorbance when the pyrolysis temperature was 530°C, with an acceptable coefficient of variation (CV) of 1.2%. Figure 1(b) illustrates absorbance against different temperatures at which atomization was performed; the coefficients of variation ranged from 1.0% to 43.7%. Maximum absorbance was observed when the atomization temperature was 1750°C, with an acceptable CV of 2.1%.
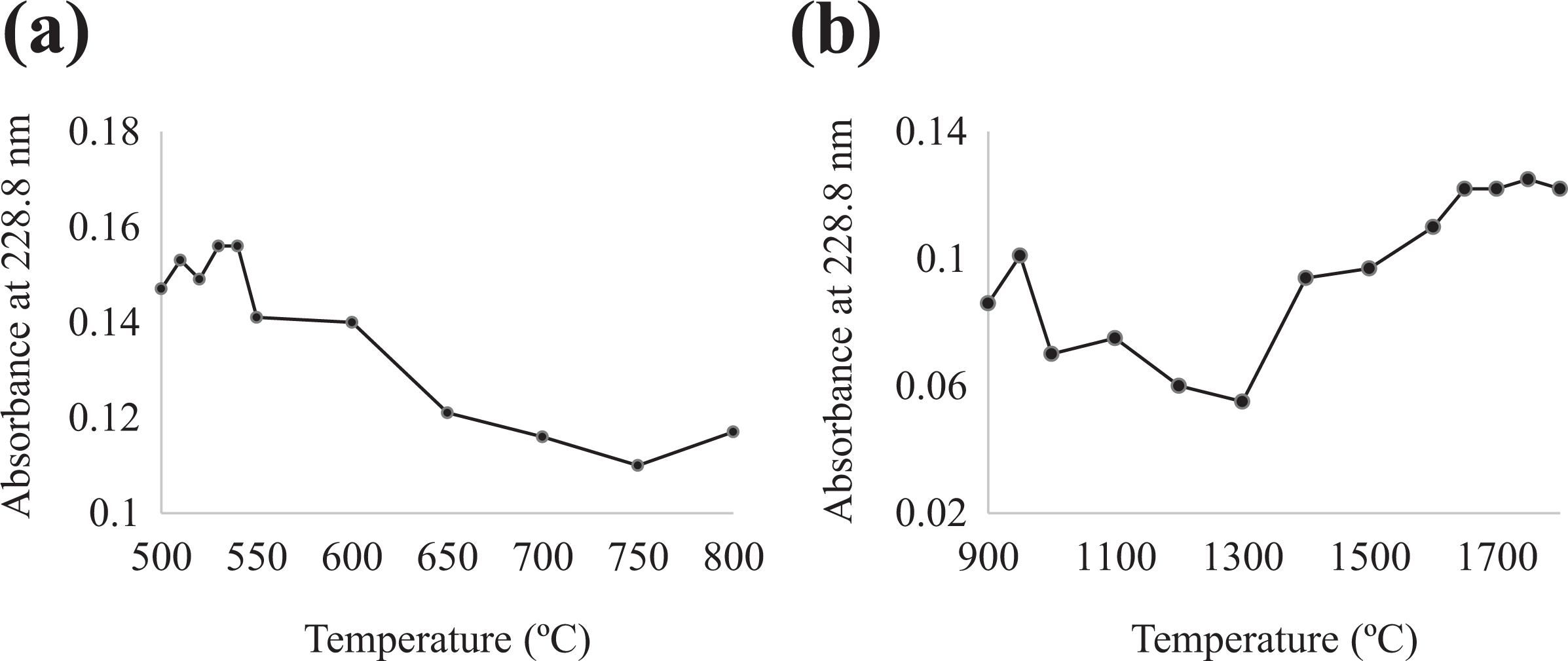
Optimization of graphite furnace temperature for Cd: (a) pyrolysis and (b) atomization. Cd: cadmium.
To establish the performance of the method 50 for the accurate quantification of Cd in thermoformed and biodegradable flexible films using GF-AAS, the following statistical quality parameters were determined: linear range, precision, sensitivity, LOD and LOQ, and accuracy.
To verify the linearity, a calibration curve was built for aqueous standard solutions of Cd with concentrations of 0.1, 0.2, 0.4, 0.6, 0.8, and 1.0 µg/L. Figure 2 shows values for the maximum absorbance at 228.8 nm versus concentration of Cd. This calibration curve exhibits a linear behavior for different concentration solutions of Cd ranging from 0.1 µg/L to 1.0 µg/L with relative standard deviations of 0.7–4.6%; the Pearson correlation coefficient was determined to be 0.998.
Table 1 represents the concentration (in µg/L) of Cd for each standard solution prepared and analyzed for intermediate precision and repeatability as follows: (a) The seven calibration curves were analyzed to determine the intermediate precision and were statistically evaluated using the Shapiro–Wilk and Levene tests. They did not show significant differences among them, and the dispersion of the data was less than 4.0% (CV). This indicates that the proposed method for the determination of Cd using GF-AAS provides a good intermediate precision. The average linear equation was y = 0.136 (±0.004)x + 0.0095 (±0.001) with r2 of 0.998. The Pearson correlation coefficient was determined to be 0.9993. In all cases, the standard deviation was less than 0.005 µg/L of Cd. (b) The Shapiro–Wilk and Levene tests showed that each concentration of Cd in the five calibration curves was from the same population, presenting a dispersion with a variation less than 5.0%. This indicates that this method has a good repeatability, with an average linear equation of y = 0.136 (±0.004)x + 0.010 (±0.002) with r2 0.9975. The Pearson correlation coefficient was determined to be 0.9987. The precision is adequate according to the acceptance criteria set by INMETRO (2007). 55
The sensitivity of the method was calculated from the calibration curve’s steepest slope with concentrations of Cd ranging from 0.1 µg/L to 1.0 µg/L. These values correspond to 0.135 and 0.136 for intermediate precision and repeatability, respectively. This method provides an increased sensitivity when the analysis is carried out on the same day, indicating that the proposed method for sample preparation is adequate. Therefore, the sensitivity of the method was 0.136.
The LOD of Cd was 0.02 µg/L (0.4 µg/kg) and corresponds to the minimum amount of Cd derived from the lowest analytical signal that can be detected with reasonable certainty. The LOQ of Cd was 0.07 µg/L (1.4 µg/kg) and represents the minimum concentration that can be measured with precision and accuracy. The LOD and LOQ are adequate for the quality control of biopolymers.
Certified biopolymers were unavailable, so the validity of the method was evaluated by addition-recovery tests. The accuracy of the method (Table 2) takes into account sample preparation, digestion treatment, and analysis of samples using GF-AAS. The percent recovery of Cd ranged from 96.23% to 97.31% for the MPER 183 thermoformed material. Comparing percent recoveries of Cd in the thermoformed with 100% recovery (using a Student’s t test), it was found that some significant differences can be attributed to random errors such as preparation of solutions and analyte loss during the digestion treatment. These values (96.2 and 97%) can be accepted as good percent recovery according to the criteria set by INMETRO (2007) 55 ; therefore, this method exhibits excellent accuracy for the determination of Cd in these samples. The values of the standardization parameters studied demonstrate quality assurance when using GF-AAS for the determination of Cd in thermoformed and flexible films.
The statistical analysis of a solution of 0.80 µg/L of Cd and a thermoformed (CM 4574-7) showed high stability over 10 consecutive days. For the standard solution, the average concentration was 0.81 µg/L ± 0.01, while the coefficients of variation for the thermoformed was less than 4.6%.
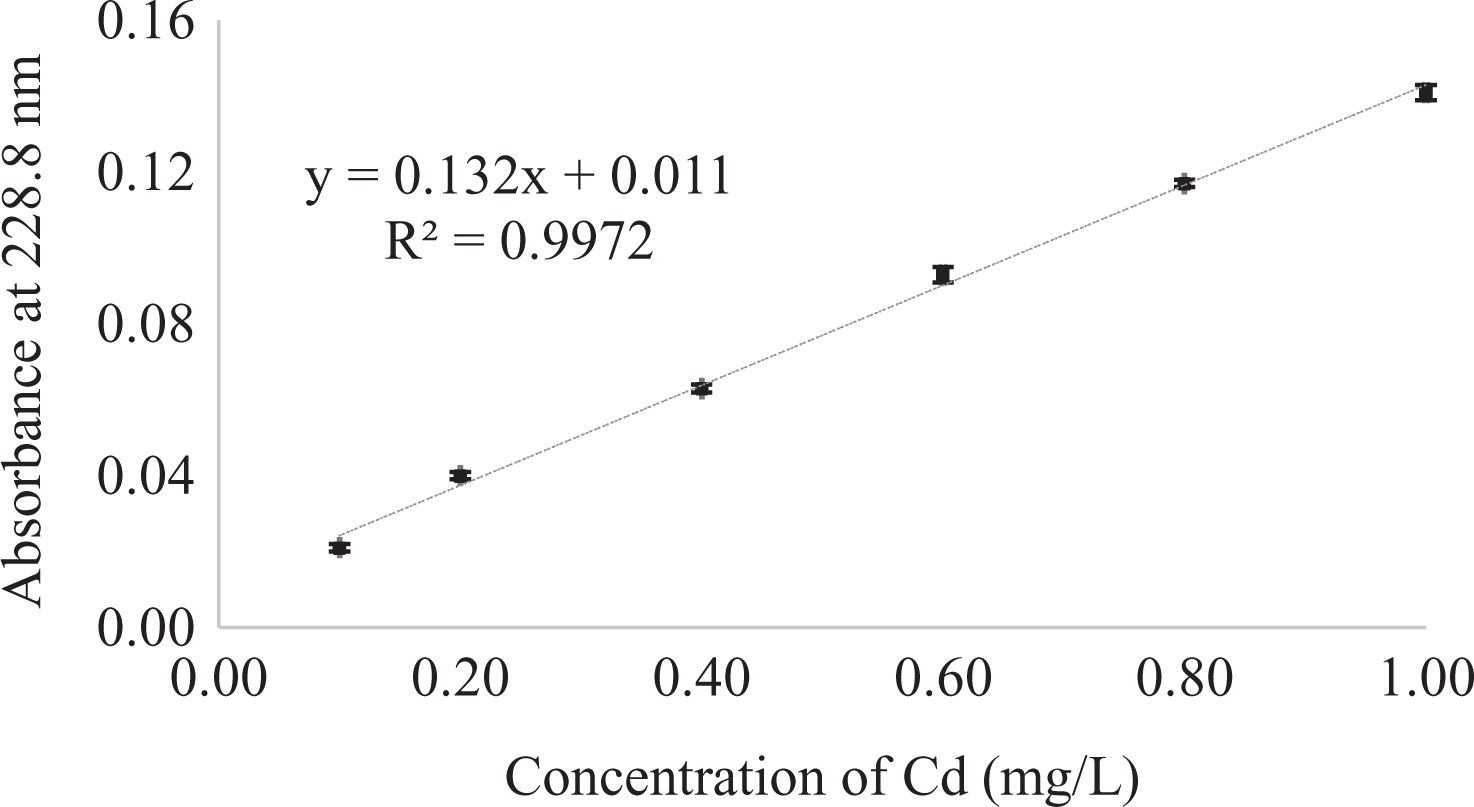
Calibration curve of cadmium obtained for the linear range (N = 4).
Average absorbance (±standard deviation) of calibration curve for precision: Intermediate precision and repeatability.a
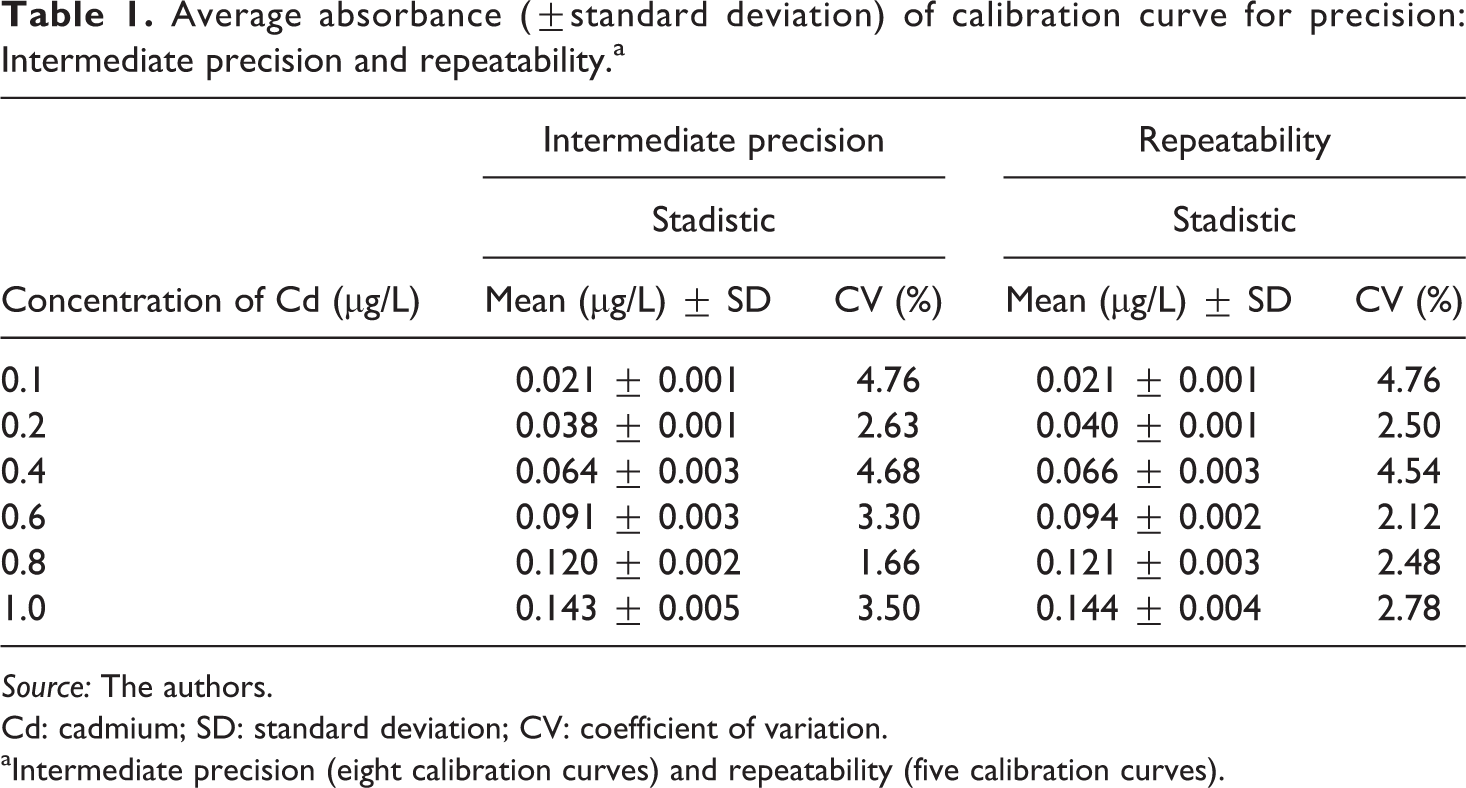
Source: The authors.
Cd: cadmium; SD: standard deviation; CV: coefficient of variation.
aIntermediate precision (eight calibration curves) and repeatability (five calibration curves).
Percent of cadmium recovery (µg/L; N = 4) in the analysis of thermoformed (MPER 183).
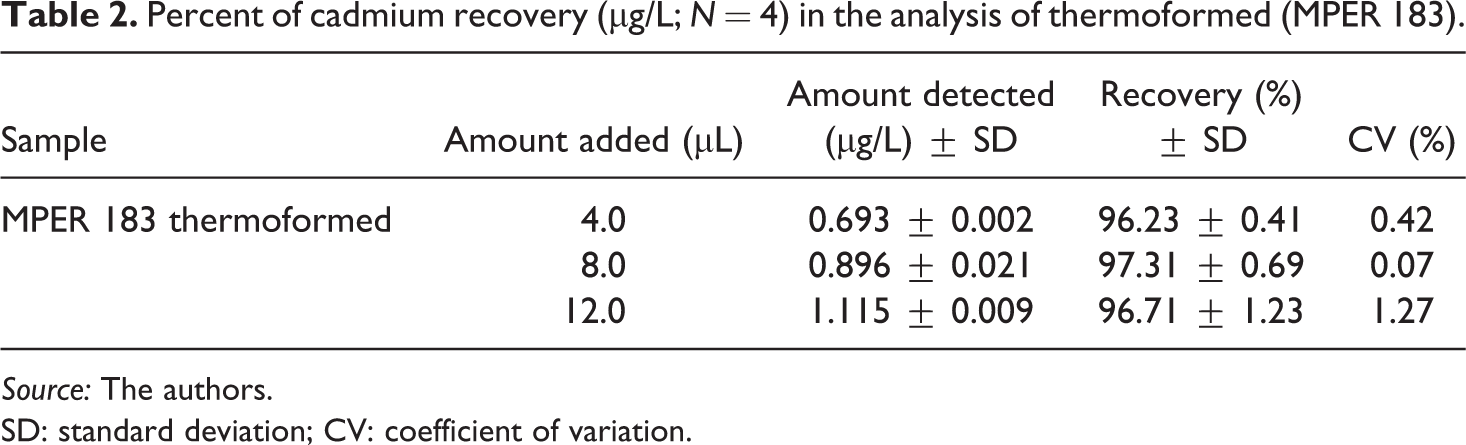
Source: The authors.
SD: standard deviation; CV: coefficient of variation.
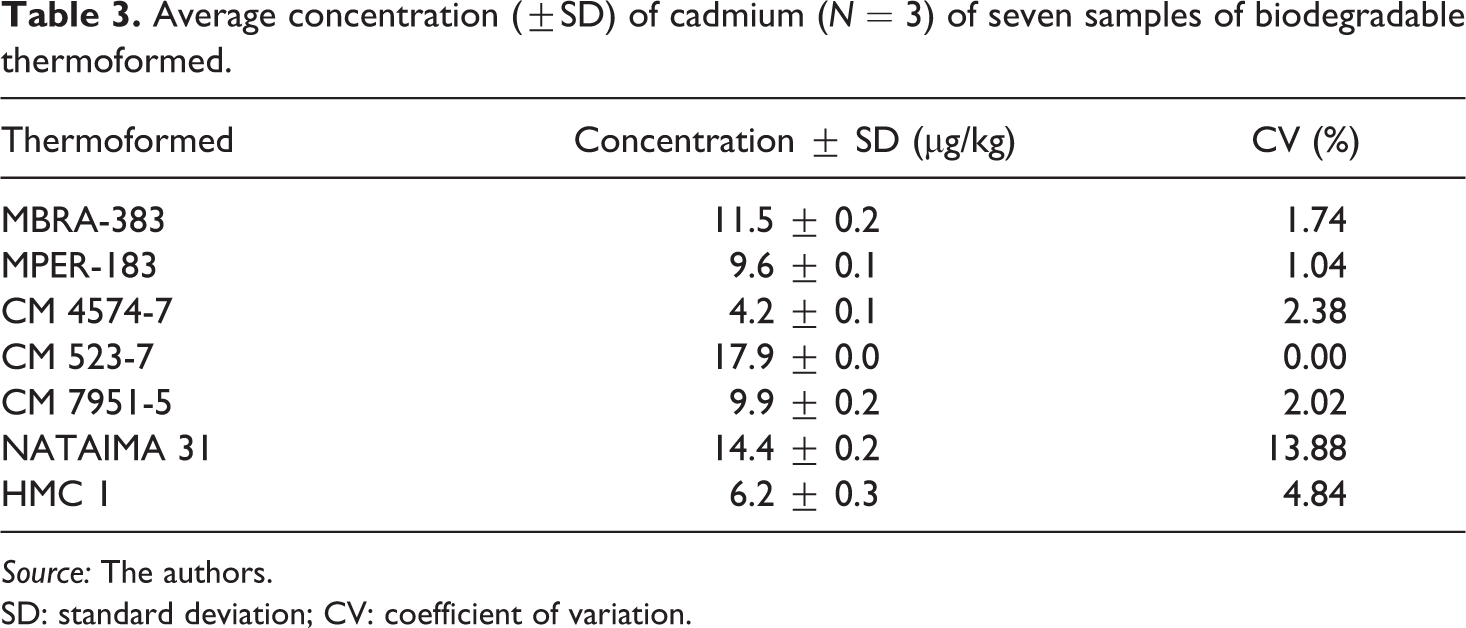
Once the analytical technique of GF-AAS was standardized for the determination of Cd in biopolymers, the implementation was performed. Table 3 presents the concentrations of Cd in µg/kg that were quantified in seven thermoformed samples. The concentrations typically ranged from 4.2 µg/kg to 17.9 µg/kg. This variation in Cd may be attributed to the quality of the raw materials used in the elaboration of biopolymers.
Average concentration (±SD) of cadmium (N = 3) of seven samples of biodegradable thermoformed.
Source: The authors.
SD: standard deviation; CV: coefficient of variation.
Other authors found mercury, arsenic, and selenium present in these thermoformed samples. Del Castillo et al. 56 determined mercury ranging from undetectable to 1343.4 µg/kg, Alvira et al. 57 determined arsenic with concentrations of less than the LOQ (39.2 µg/kg), and Rada-Mendoza et al. 58 found selenium ranging from undetectable to 1240.0 µg/kg.
As given in Table 4, for all flexible films in the pristine and hydrolyzed forms, the detected amounts of Cd were below the LOQ. This corresponds to a concentration of a trace level of Cd less than 1.4 µg/kg. In these flexible films, mercury was reported to be undetectable to 127.3 µg/kg, 56 arsenic was undetected, 57 and selenium was reported to be undetectable to 122 µg/kg. 58 Other authors reported that selenium can decrease the Cd concentrations and increase the growth in vegetables. 59
Estimated average concentration of cadmium (N = 3) in six samples of biodegradable flexible films.

Source: The authors.
CV: coefficient of variation; N.D.: not detectable.
In thermoformed and biodegradable flexible films, the values of heavy metals accumulated were higher for mercury than for selenium, Cd, and arsenic, suggesting that absorption and bioaccumulation depend upon the availability of metals.
In Europe, Asia, the United States, and Brazil, there is no legislation for regulating the content of heavy metals in biopolymers. The Colombian regulation NTC-4096 43 establishes 1.0 mg/kg as the maximum level of Cd allowed in Dioctyl adipate (DOA) and Dioctyl phthalate (DOP) plasticizers used in the manufacture of plastics that are in contact with food which indicates that Cd concentrations in the range of 4.2–17.9 µg/kg found in the thermoformed material (Table 3) and in lower concentrations than the LOQ found in flexible films (Table 4); all are below the limit established by the regulation. Therefore, it is expected that there is no incidence of this metal in the final product.
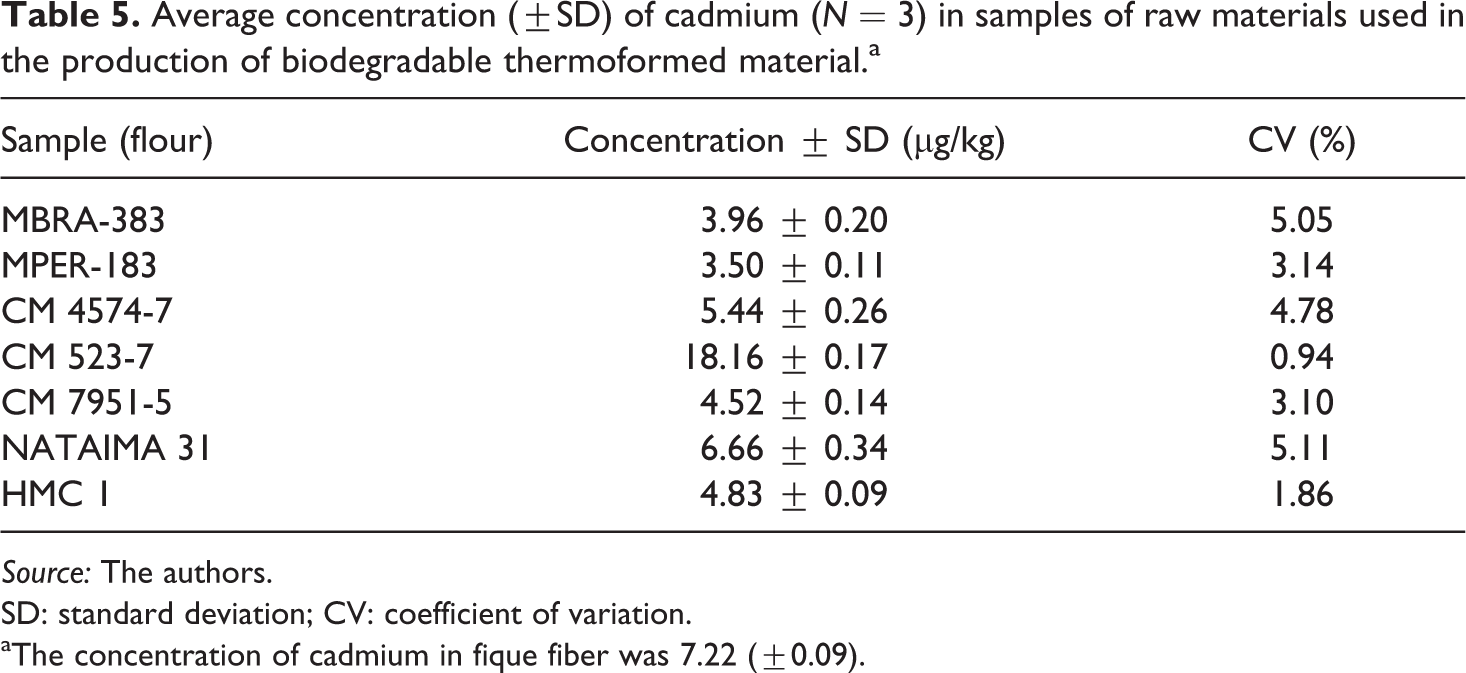
However, it is necessary to investigate the source of the Cd in these samples. In this case, Cd was detected in raw materials (cassava flour and starch and fique fiber) used in both the thermoformed and flexible films. Table 5 presents the concentration in µg/kg of Cd in different samples of the raw materials used in the production of biodegradable thermoformed films. Of these, the thermoformed had concentrations in the range of 3.5–18.2 µg/kg of Cd, with the fique fiber having the concentration of 7.2 µg/kg; therefore, this source contributes the most Cd to the thermoformed film.
Average concentration (±SD) of cadmium (N = 3) in samples of raw materials used in the production of biodegradable thermoformed material.a
Source: The authors.
SD: standard deviation; CV: coefficient of variation.
aThe concentration of cadmium in fique fiber was 7.22 (±0.09).
Cd found in the flexible films made from cassava starch, the raw material used for producing this biopolymer, was not quantified (concentrations less than LOQ).
With the results given in Tables 3 and 4, it can be inferred that Cd found in thermoformed material comes principally from cassava flour and fique fiber. The conclusion here is that traces of Cd detected in the studied biopolymers were low, indicating non-absorption and contamination from anthropogenic sources, meaning that these materials are not toxic and may be employed as food wrappers and containers, conserving food 1,2,60 and extending its shelf-life, without potential health risks to the consumer.
Cd uptake from soil is highest in plants that are grown in areas with high heavy metal concentrations, 27 and so it is very important to take precautions when obtaining and handling raw materials, as was mentioned previously, because Cd is released into the atmosphere (30,000 t/year) and can contaminate the flour, starch, and fique fiber through the disposal of sewage sludges as soil amendments, exposure to smoke from cigarettes, emissions from industrial processes (nickel–Cd battery industry), 23 use of phosphate fertilizers, soils containing heavy metals, and the irrigation of vegetables with contaminated water. 27,32,34,45,52,61 –63 Additionally, it is very important to note that the best way to reduce Cd contamination is through the control of raw materials and a thorough understanding of the manufacturing process.
Conclusions
The developed GF-AAS method was efficient (high sensitivity and acceptable in terms of accuracy and reliability) for Cd quantification in thermoformed and biodegradable flexible films and flour samples from Cassava. Cd content found in thermoformed films is associated with a prior contamination of the raw material (during cultivation, pretreatment, and/or transportation) and reducing the impact of the manufacturing process on films, which used new machines.
Additionally, the present study provide new data to the food safety authorities and broaden knowledge of the contribution made by raw materials on the concentrations of Cd in biopolymers.
Finally, this study demonstrates the immense potential of using biopolymers in packaging and food conservation, and how these materials add value to agricultural activity and help to reduce nonbiodegradable plastics in the environment.
Footnotes
Acknowledgements
The authors would like to acknowledge the University of Cauca (Industrial Analysis Unit, BICAMSA and QPN laboratories) and the Ministry of Agriculture and Rural Development for funding the projects on which this work is based.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the University of Cauca and the Ministry of Agriculture and Rural Development of Colombia.
