Abstract
Chemically reduced graphene (C-rGO) nanosheets were first prepared from graphene oxide (GO), and then the polypropylene (PP) composites synergistically reinforced–toughened by styrene–butadiene rubber (SBR), and C-rGO nanosheets were fabricated via melt blending. The mechanical properties of PP can be considerably improved by synergistically filling with C-rGO nanosheets and SBR, especially for the notched Izod impact strength (IS). The results from the X-ray diffraction, polarizing optical micrographs, scanning electron microscope, differential scanning calorimetric, dynamic mechanical analysis, and thermogravimetric analysis measurements reveal that: (1) the β-phase crystal structure of the PP is formed when the C-rGO and SBR are synergistically filled with PP and its formation plays a role for the enhancement of the impact strength for PP/SBR/C-rGO composites; (2) the dispersion of the C-rGO and SBR in the PP/SBR/C-rGO composites is homogeneous, indicating that synergistic incorporating method decreases the aggregation of nanosheets and thus increases the sites for dissipation of shock for impact energy in the PP/SBR/C-rGO composites; and (3) the thermal analysis shows high thermal stability for the PP/SBR/C-rGO composites.
Introduction
Polymers reinforced with particles of nanometer size, in at least one dimension, have shown superior thermal and mechanical properties and are being considered for advanced applications in many industrial fields. Since the development of layered filler (i.e. silicate) reinforced polymer composites by Toyota researchers in 1987, much interest has been focused on the preparation and characterization of polymer/layered fillers nanocomposites and much considerable progress has been achieved. 1 –3 Especially, with advantage of the high aspect ratio (width to thickness), excellent mechanical, electrical, and thermal conductivities of graphene sheets, 4,5 its homogeneous dispersion in a continuous polymer matrix improved greatly the properties of polymers, and even produced new properties that cannot be derived from other composites’ counterparts. 6 –9 Therefore, material scientists have paid more attention to the investigation on the applications of graphene in polymer/graphene composites and many interesting works have been extensively reported. 10 –13 However, due to high crystal lattice energy and van der Walls forces existed in graphene layers, complete separation and dispersion of these graphene layers to sheets is difficult. In order to overcome these problems, many convenient methods for preparation of polymer composite with excellent properties, in which graphene sheets disperse homogeneously in polymer matrix, are being an interesting project in the polymer science. Various approaches for making polymer/graphene composites have been reported, including blending polymers and graphite particles 14 and intercalating the polymers into graphite interlayer, mainly through in situ polymerization, 15,16 solution blending, 17 as well as melting compounding. 18 –20 Interestingly, chemical functionalization of graphene oxide (GO) using the oxygen-containing functional groups on the surface of GO, or hybridizing using nano-materials in different dimensions, has been proved to be an effective method to improve the dispersion of graphene, accompanied by a synergistic effect from hybrids can be achieved. 21,22
Poly(propylene) (PP) is one of the most important polyolefin. It has been widely used in many fields, such as building materials, furniture, automobiles, and toy industry. However, PP suffers from poor impact resistance at low temperature. Therefore, PP is traditionally modified with elastomers to improve its impact strength (IS), 23 –27 which enhances the toughness of PP/rubber blends, however, at the same time always accompanies the sacrifice of the stiffness. Alternatively, inorganic nanoparticle-filled composites have been proved to be an effective method to enhance both the stiffness and the toughness of polymer matrix simultaneously. 28,29 However, there exist several important issues when fabricating these polymer composites, such as the difficulty of achieving homogeneous dispersion of nanoparticles in a polymeric matrix due to the strong tendency of nanoparticles to agglomerate, and thus low loading level of the reinforcing fillers. 28,29 To solve these problems, many researchers focused on the studies of in situ polymerization of monomers in the presence of nanoparticles, 30,31 such as the sol–gel process and the intercalation polymerization technique. 32,33 Although nanoscale dispersion of the particles can be obtained using these methods, they are not suitable to produce nanocomposites in mass with low cost and ready applicability.
Recently, Zhang et al. reported that styrene–butadiene rubber (SBR) nanoparticles could be used to toughen PP. 34 However, the inhomogeneous dispersion and incorporation of low loading level of nanoparticles in PP only increase the toughness and stiffness of composites moderately. In Wang and Liu’s work, 35 the stiffness of inorganic silicon dioxide (SiO2) nanoparticles and the toughness of SBR nanoparticles were combined in PP matrix to overcome the disadvantages existing in the abovementioned methods. It is found that a certain interparticle distance or space could be formed between the two different nanoparticles when they are mixed together and incorporated into PP matrix, leading to PP can penetrate the agglomerated nanoparticles easily and thus greatly enhance the dispersion state of the nanoparticles, and consequently the mechanical and thermal properties of neat PP have been considerably improved by synergistically filling with SiO2 and SBR. However, excessive addition of silica will increase the rigidity and sacrifice the precious flexibility of the PP composite. Moreover, the stress transmission and distributive effects are limited because SBR and silica are in spherical particles. In view of this, we assume that combining two-dimensional flake filler and SBR to modify PP should improve the load transmission and dispersion effect, in order to obtain PP composite with better performance. In our previous work, 22 a facile strategy to synthesize SiO2 nanoparticles-coated graphene oxide (GO/SiO2) nano-hybrids (GOS) has been reported. Due to the space-layer barrier effects of SiO2 nanoparticles to GO sheets, GOS has been dispersed uniformly in the PP matrix. Tensile tests showed that GOS improved the tensile and IS in the full filler content range. Moreover, the strengthen–toughening effect of GOS is much better than the other fillers, GO and SiO2.
In this article, chemically reduced graphene (C-rGO) sheets were firstly prepared from GO, and then PP composites synergistically reinforced–toughened by styrene–butadiene rubber (SBR) and C-rGO nanosheets were fabricated via melt blending, and its microstructure and properties were investigated systematically.
Experimental
Materials
PP (T30S; Melting Index = 2.5–3.5 g/10 min) used in this study was provided by Dushanzi Fuel & Chemical Co. (Xinjiang, China). SBR (1502) were supplied by SINOPEC Lanzhou Fuel & Chemical Co. (Lanzhou, China). Natural graphite powder (NGP; SP-2, (C > 99%, D = 5 mm)) was purchased from Qingdao Tianhe Graphite Ltd, (Qingdao, China). Potassium permanganate (KMnO4; analytical reagent [AR]), sodium nitrate (AR), concentrated sulfuric acid (H2SO4; >96%), hydrogen peroxide, anhydrous ethanol (AR), tryptophan, ascorbic acid, and sodium hydroxide (NaOH) were kindly provided by KeLong Reagent Ltd, (Chengdu, China) and used as received.
Preparation of graphite oxide, GO, and chemical reduced graphene C-rGO sheets
Graphite oxide used in this research was synthesized from the NGP by graphite oxidation with KMnO4 in concentrated H2SO4 according to the procedures depicted in the study by Bian et al.. 17 The obtained graphite oxide was then sonicated to produce GO.
C-rGO was prepared by chemical reduction method. The detailed steps are depicted as follows: A certain amount of GO, tryptophan, ascorbic acid (vitamin C), and NaOH were firstly dispersed in 200-ml aqueous solution under ultra-sonication. Then above mixture was stirred for 24 h at 80°C to finish the reduction reaction. The suspension was then filtered and washed several times with anhydrous ethanol. Finally, the resulting product was dried at 80°C under vacuum for 24 h to obtain C-rGO.
Preparation of PP/SBR/C-rGO composites
PP/SBR binary blends with different SBR contents were firstly prepared and tested in order to determine the optimum SBR content. After that, PP/SBR/C-rGO composites were then prepared by melting blending, according to the formulation listed in Table 1. Before processing, PP was dried under vacuum at 80°C for 6 h. In order to improve the dispersion of C-rGO in PP composites, a coating method reported in our previous work 36 was applied. In particular, C-rGO was firstly dispersed in anhydrous ethanol by sonication for 1 h, PP particles were then added to C-rGO solution, and the sonication was continued for 1 h. Finally, the solvent was evaporated at 80°C resulting in a complete coverage of PP particles with C-rGO. The solid mixtures, PP/SBR binary blends, were melt-blended at 180°C for 8 min in a compounder (HL-200 type, made in Jilin University, China). At last, the compounds were hot-pressed on a plate vulcanization machine (XLB type, made in Qingdao, China) to make dumbbell specimens (62.5 × 3.25 × 0.7 mm3) for tensile tests and rectangular sample (10 × 1.5 mm2) for impact tests. For comparison purpose, PP/C-rGO composites were prepared under the identical processing conditions as that of PP/SBR/C-rGO composites. The GO content is consistent with the optimal C-rGO content in PP/SBR/C-rGO composites.
The experimental formulation and typical mechanical properties.
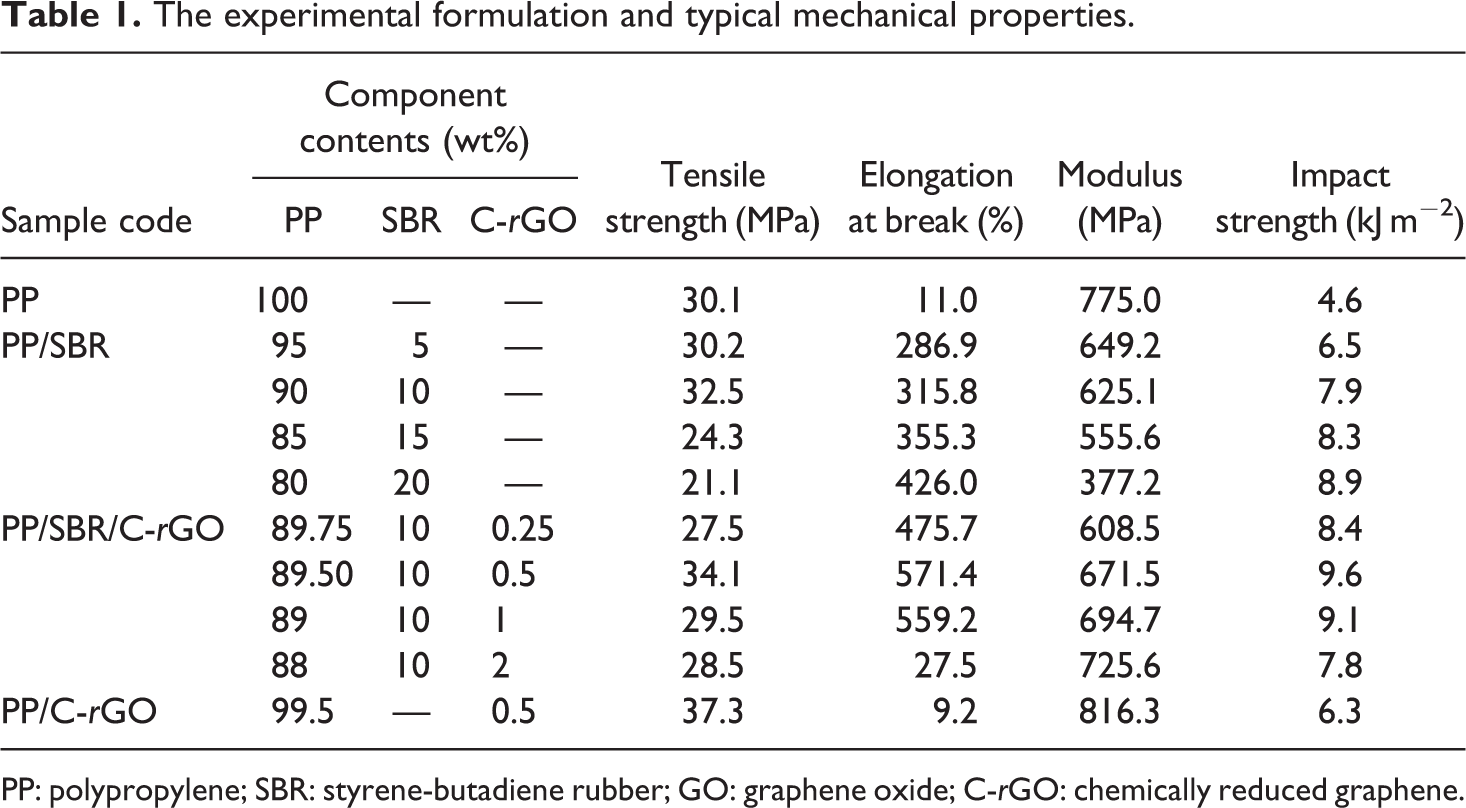
PP: polypropylene; SBR: styrene-butadiene rubber; GO: graphene oxide; C-rGO: chemically reduced graphene.
Characterization
X-ray diffraction (XRD) test was carried out at ambient temperature on a Rigaku D/max-1200X diffractometer (40 kV, 200 mA, Akishima-shi, Tokyo, Japan) with copper Kα (λ = 0.154 nm) irradiation. Scans were taken from 1.5° to 40° with a step of 0.02°. The NGP, GO, and GOS samples were in fine powder form, whereas PP and Pcomposites were from hot-pressed films (1 mm).
The microscopic images of samples were collected using a scanning electron microscope (SEM; JEOL JSM-820, Akishima-shi, Tokyo, Japan). Impact fracture surfaces of nanocomposites were coated with gold before SEM examination.
Differential scanning calorimetric (DSC) analysis was carried out on NETZSCH DSC-200PC under continuous N2 flow. Samples were heated from room temperature to 220°C at a rate of 20°C min−1 and held at that temperature for 5 min to eliminate the heat history. The samples were then cooled to room temperature at a rate of 20°C min−1. After keeping at room temperature for 5 min, samples were reheated to 220°C at a rate of 20°C min−1. The crystallization (T c) and melting (T m) temperatures were determined from the cooling and reheating scans, respectively. The relative crystallinity (X c, %) can be calculated by the following equation
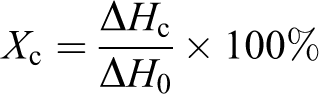
ΔH c is the enthalpy of crystallization and ΔH 0 is the crystallization enthalpy of 100% crystallized PP (209 J g−1). 37
Thermogravimetric analyses (TGAs) were performed on a NETZSCH STA449 F3 thermal analyzer (Selb, Germany) under argon flow at the rate of 20°C min−1. Mechanical properties were obtained using an INSTRON3365 electronic tensile tester (Boston, USA) with computer control systems. The rate of crosshead motion was 10 mm min−1 at room temperature. Impact performance was tested using impact testing machine under the room temperature. Five specimens of each composition were tested, and the average values were reported.
Polarizing optical micrographs (POM) were obtained using an XP-203 (made in Shanghai, China) with sample thickness of about 50 μm.
TGAs were performed on a NETZSCH STA449 F3 thermal analyzer under argon flow at the rate of 20°C min−1.
Mechanical properties were obtained using an INSTRON3365 electronic tensile tester with computer control systems. The rate of crosshead motion was 10 mm min−1 at room temperature. IS was tested using impact testing machine under the room temperature. The notched Izod IS was measured using an Izod impact tester (made in Chengde, China). The sample size of the rectangular specimens used for IS tests was 75 × 12 × 1 mm3 with a V-shaped notch (with tip radius of 0.25 mm and depth of 2 mm). The data reported here were averaged using the values from five specimens tested.
Dynamic mechanical analysis was carried out using a TA-Q800 (New Castle, USA) analyzer. Double cantilever shear mode, at a heating rate of 1°C min−1.
Results and discussion
Mechanical properties of PP/SBR binary blends
Table 1 shows the designed formulation and the mechanical properties of PP composites. From Table 1, it can be seen that the tensile strength (TS), the elongation at break (EB), the modulus, and the IS of pure PP are 30.1 MPa, 11%, 775 MPa, and 4.6 kJ m−2, respectively. For PP/SBR binary blends, increasing the content of SBR increases the EB and the IS, but decreases the modulus of blends. While the TS increases slightly firstly and then decreases with increasing SBR contents. The corresponding values of PP/SBR (90/10) blends are 32.5 MPa, 315.8%, 625.1 MPa, and 7.9 kJ m−2, respectively. Compared with those of pure PP, the TS and IS of PP/SBR (90/10) blends increased by 7.9% and 71.7%, respectively. These results indicate that SBR can improve the toughness of PP matrix. It should be noted that although PP/SBR (90/20) blends show the highest IS improvements (93.5%), its TS decreases (about 29.9%) obviously, in contrast to that of pure PP. Therefore, the optimization content of SBR is 10 wt% in PP/SBR binary blend. PP/SBR blends containing 10 wt% SBR were selected and studied in PP/SBR/C-rGO composites.
Mechanical properties of PP/SBR/C-rGO composites
Table 1 lists the TS, EB, modulus, and IS data of PP/SBR/C-rGO and PP/C-rGO composites. It can be seen that the TS, EB, modulus, and the IS data of PP/SBR/C-rGO (89.5/10/0.5) composite are 34.1 MPa, 571.4%, 671.5 MPa, and 9.6 kJ m−2, respectively. Compared with those of PP/SBR (90/10) binary blends, these typical mechanical properties of PP/SBR/C-rGO composites increased by 4.9%, 80.9%, 7.4%, and 21.5%, respectively. The corresponding values of PP/C-rGO (99.5/0.5) composites are 37.3 MPa, 9.2%, 816.3 MPa, and 6.3 kJ m−2, respectively. The TS and modulus of PP/C-rGO (99.5/0.5) composites are higher than those of PP, PP/SBR (90/10) binary blends, and PP/SBR/C-rGO (89.5/10/0.5) composite, as shown in Table 1. It can be attributed to the rigid nature of C-rGO nanosheets. The values of TS, EB, and the modulus of PP/SBR/C-rGO (89.5/10/0.5) composite are between those of the PP/SBR and PP/C-rGO composites, which is different from the case of the IS. The IS of PP/SBR/C-rGO (89.5/10/0.5) composite is higher than that of PP/SBR or PP/C-rGO composites, as shown in Table 1. It is attributed to the synergistic effect upon incorporating both rigid C-rGO nanosheets and flexible SBR. In summary, compared with neat PP, the C-rGO nanosheets and SBR can improve the toughness and stiffness of PP matrix simultaneously.
The toughening mechanisms synergistically using SBR and C-rGO nanosheets can be mainly attributed to two reasons. Firstly, SBR acts as stress concentration sites for dissipation of shock or impact energy by controlling and promoting matrix deformation. The addition of flexible SBR into PP leads to relaxation of the stress concentration and suppresses the formation of the matrix crazes or deformation. Secondly, the addition of C-rGO nanosheets further improves the fine dispersion of SBR and thus decreases the possibility of rubber cohesion into bulky phases during the mechanical mixing process. Figure 1 shows the preparation process of C-rGO and possible interactions between C-rGO and SBR. The strong π–π interfacial interactions between graphene and phenyl groups of SBR matrix are assumed to be beneficial to the dispersion of C-rGO and formation interval effects. As a result, the sites for dissipation of shock or impact energy (i.e. the IS) are greatly increased in the PP/SBR/C-rGO composites.
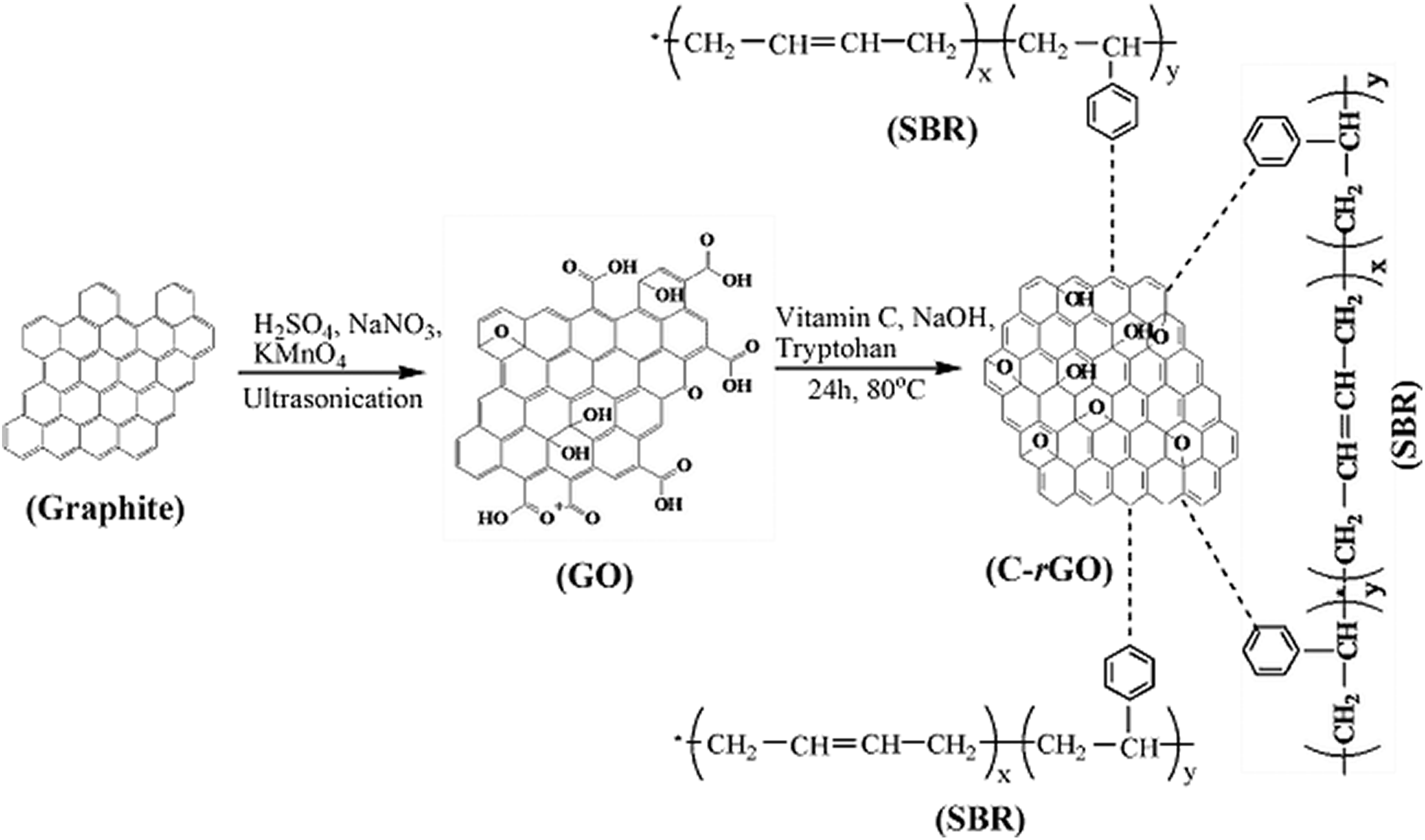
Schematic illustration of preparation process of C-rGO and possible interactions between C-rGO and SBR.
Crystalline structure of C-rGO nanosheets and PP/SBR/C-rGO composites
The crystalline structure of natural graphite, GO, and C-rGO is shown in Figure 2. It can be seen that the graphite shows its features at the 2θ = 26.6° (002) diffraction peaks, corresponding to the graphite layer distance of 0.335 nm. But after oxidation, the graphite (002) diffraction peak shifts to lower angle, the diffraction peak appears at 2θ = 11.2°, attributable to its characteristics of the GO in the (001) diffraction peaks. The introduction of a large number of oxygen-containing functional groups increases the layer spacing of the graphite from 0.335 nm to 0.77 nm. By chemical reduction, a 2θ = 26.2° appeared in the C-rGO with layer spacing of 0.341 nm, indicating that GO was reduced effectively by vitamin C.
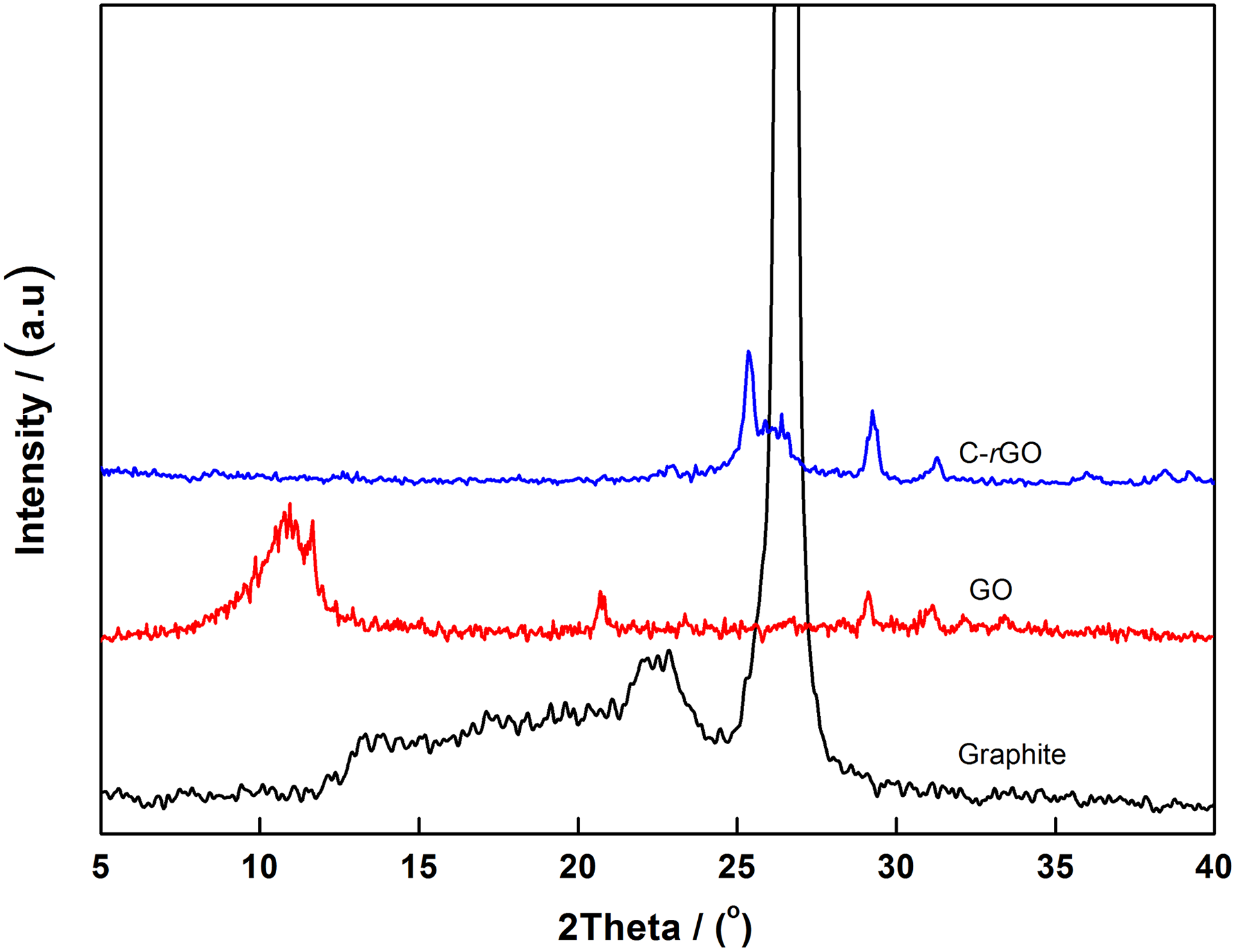
XRD patterns of graphite, GO, and C-rGO.
The XRD patterns of neat PP, PP/SBR (90/10), PP/C-rGO (99.5/0.5), and PP/SBR/C-rGO (89.5/10/0.5) composites are shown in Figure 3. The diffractive peaks at 2θ = 14.1°, 16.8°, 18.5°, and 21.2° correspond to (110), (040), (130), and the overlapped (131, 041) planes, respectively, which are characteristics of a-type monoclinic crystal structures of PP. 22 It indicates that all the samples contain a-type crystals. However, in comparison to neat PP, it can be clearly seen that a new reflection at 2θ = 16.2° appears in all the three composites, especially for the PP/SBR/C-rGO system. This new peak is assigned to (300) reflection of the β-type hexagonal crystal structure of PP. Moreover, the fraction of β-phase in the crystalline samples can be roughly estimated from the ratio of the height of (300) reflection of β-phase to the sum of the heights of the four reflections, that is, (110), (040), and (130) of the a-phase and (300) of the β-phase, as proposed by Turner-Jones et al. 38 The intensity of β-phase diffractive peak in PP/SBR/C-rGO sample is higher than those of PP/C-rGO and PP/SBR samples, probably due to the synergistic nucleating effect of SBR and C-rGO in PP matrix. The appearance of β-type hexagonal crystals obviously improves the IS of PP because the β-type hexagonal crystals have higher ductility and strength than a-type monoclinic crystals. These results are also confirmed by the mechanical measurements.
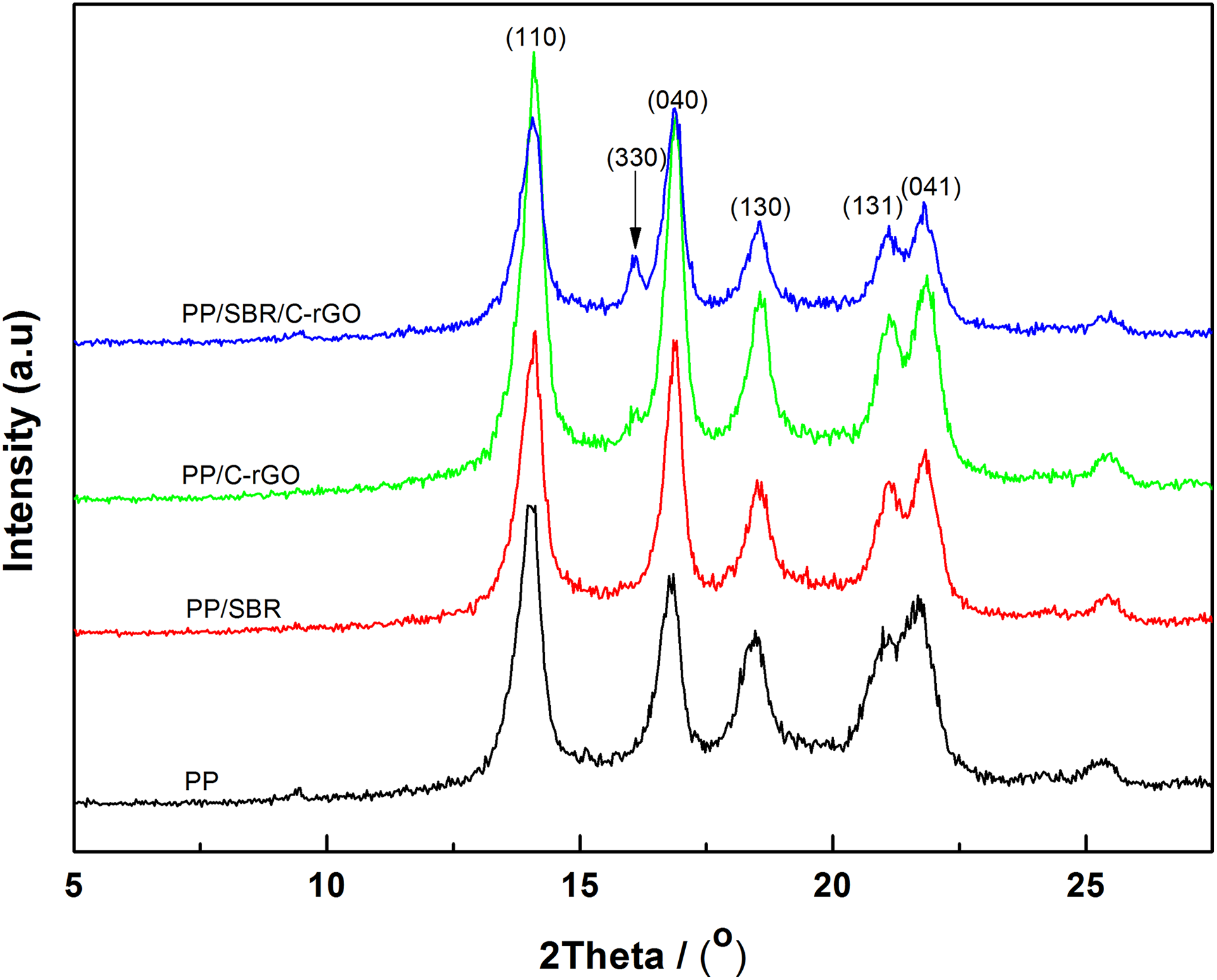
XRD patterns of PP, PP/SBR (90/10), PP/C-rGO (99.5/0.5), and PP/SBR/C-rGO (89.5/10/0.5) composites.
Figure 4 shows the POM images of the composites. Figure 4(a), (b), and (c) are for pure PP, PP/SBR (95/5), and PP/SBR (90/10) binary blends, respectively. From Figure 4(a), it clearly shows that the spherulites of pure PP is relatively perfect or much more developed. With the addition of SBR, the spherulite is becoming imperfect and the spherulite grain boundaries are blurred, as shown in Figure 4(b) and (c). Figure 4(d) is the POM image of PP/C-rGO composite with 0.5 wt% C-rGO, it can be seen that the spherulite is much more perfect than those of PP/SBR (90/10) blends, but more fine than that of pure PP, indicating that C-rGO nanosheets play a role of heterogeneous nucleating agent, resulting in increased crystallization rate of PP. Figure 4(e) to (h) is for PP/SBR/C-rGO composites. It can be seen that with increasing C-rGO contents, the spherulites of PP in PP/SBR/C-rGO composites are imperfect or much less developed, and the spherulite size significantly decreases when compared with those of PP/C-rGO composite (Figure 4(d)) and PP/SBR (Figure 4(b) and (c)) blends. It thus indicates that incorporating C-rGO and SBR into PP makes the spherulites imperfect and also induces the formation of the β-type hexagonal crystals of PP. And the increase in the β-type crystal fraction is favorable to the improvement of the toughness of PP matrix.

POM images of different composites: (a) pure PP, (b) PP/SBR (95/5), (c) PP/SBR (90/10), (d) PP/SBR (80/20), (e) PP/C-rGO (99.5/0.5), (f) PP/SBR/C-rGO (89.75/10/0.25), (g) PP/SBR/C-rGO (89.5/10/0.5), and (h) PP/SBR/C-rGO (88/10/2).
Morphologies of PP composites
Figure 5 shows the fractured surfaces of the samples. Pure PP shows a single continuous phase and the section has a certain plastic deformation, as shown in Figure 5(a), which shows that PP has certain impact toughness. In Figure 5(b), when the SBR is added into PP, PP/SBR binary blends show a homogeneous PP continuous phase, and the plastic deformation of the cross section is more obvious, indicating that the addition of SBR improved the impact toughness of the pure PP (as shown in Table 1). Figure 5(c), (d), (e), and (f) is for PP/SBR/C-rGO composites containing C-rGO of 0.25, 0.5, 1, and 2 wt%, respectively. Both of them show a continuous PP matrix in which C-rGO and SBR are dispersed homogeneously. The small round particles observed on the fracture surface are SBR particles which are finely dispersed without noticeable agglomeration. The C-rGO nanosheets are also homogeneously dispersed in PP matrix at relatively low C-rGO contents. Therefore, the synergistically filled C-rGO and SBR particles into PP can improve their dispersions due to the special interval effects and strong π–π interfacial interactions between graphene and phenyl groups of SBR matrix, 39,40 as shown in Figure 1. The fine dispersion of C-rGO and SBR particles is favorable to the improvements in mechanical properties of resultant PP composites. However, at high C-rGO incorporation (Figure 5(f)), poor dispersion of C-rGO nanosheets in some regions of the fractured surface can also be observed in PP matrix, and the existence of C-rGO cluster results in the destruction of matrix when PP/SBR/C-rGO composite is deformed by IS. Therefore, the mechanical properties of PP/SBR/C-rGO composite at higher C-rGO contents are much worse than those at lower C-rGO contents.

SEM micrographs of the composites: (a) pure PP, (b) PP/SBR (90/10), and (c) to (f) PP/SBR/C-rGO composites containing C-rGO of 0.25, 0.5, 1.0, and 2.0 wt%.
Thermal analysis of PP composites
Figure 6 shows the DSC melting/cooling curves for pure PP, PP/SBR, PP/C-rGO, and PP/SBR/C-rGO composites. The DSC results are listed in Table 2. It can be seen that the melting peaks of PP/SBR samples shift toward lower temperatures in contrast to that of neat PP. This is probably because SBR plays a role of nucleating agent and thus leads to the formation of small and imperfect PP crystals. This effect is improved by adding a small amount of C-rGO, and it is more obvious in PP/SBR/C-rGO composites at higher C-rGO contents. The synergistic incorporation of C-rGO and SBR into PP improves their respective dispersion among the matrix, thus results in the formation of more crystalline nuclei and promotes the formation of PP crystals. It can also be seen that the melting endotherm has a slight broadening in the DSC curves of the composites. This indicates that the presence of C-rGO and SBR acting as nucleation agent induces the formation of β-type crystals of PP, and therefore the coexistence of the α-type and β-type crystals in the composites results in the broadening of melting endotherm. From Figure 6(b), it can be seen that the crystalline peaks shift toward higher temperatures for PP/SBR/C-rGO and PP/C-rGO samples. This effect is more obvious in PP/SBR/C-rGO composites at higher C-rGO contents in contrast to that of neat PP and PP/SBR binary blends. As shown in Table 2, the crystallinity (X c) of PP/SBR/C-rGO sample is higher than those of PP/SBR and pure PP samples, probably due to the formation of more β-type crystals in PP/SBR/C-rGO sample. It is found that adding SBR and C-rGO into PP simultaneously is much more beneficial to improve the crystallinity of PP composites in contrast to other research works. 22,34,35 The above results from thermal analysis are consistent with those obtained from XRD.
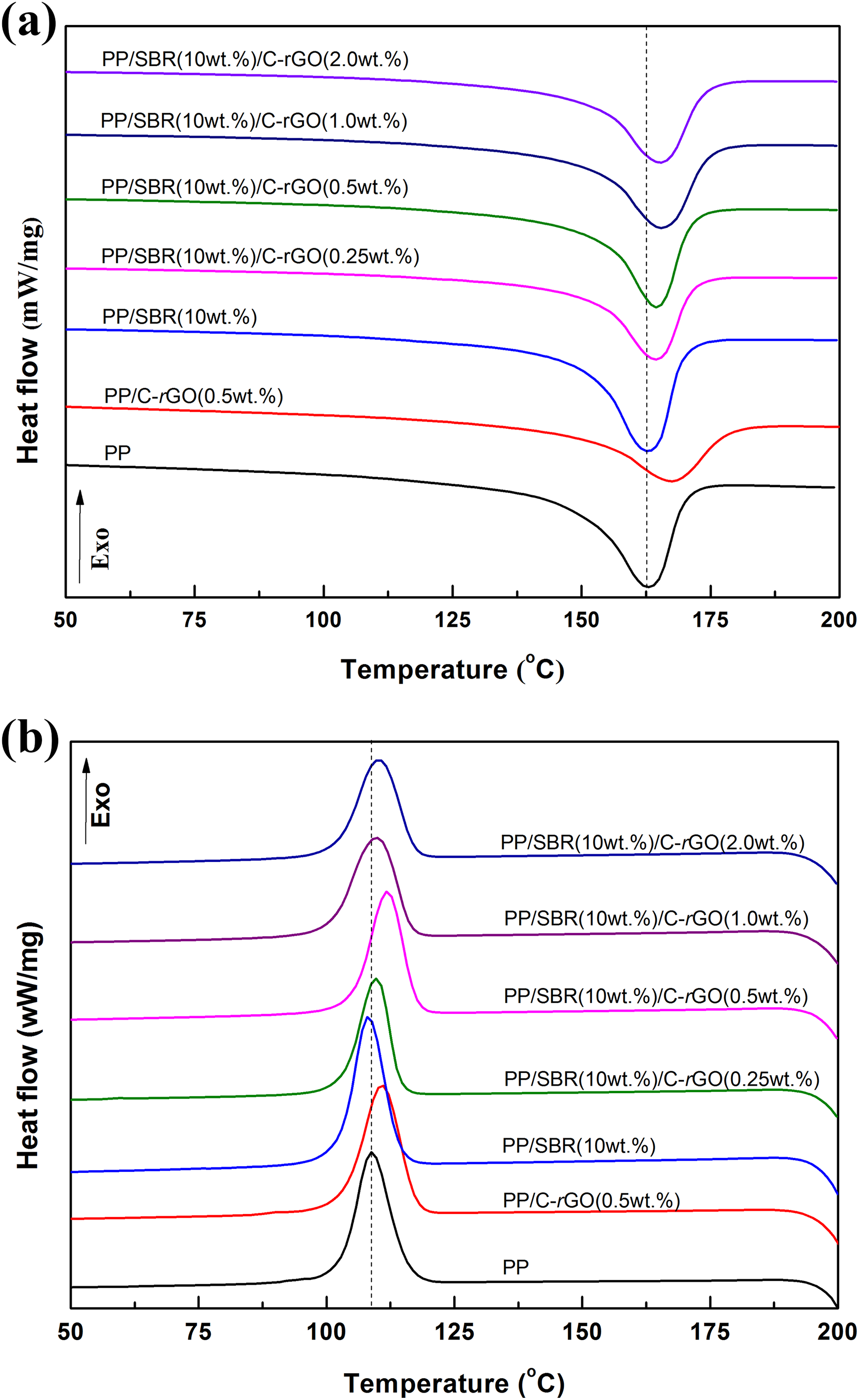
DSC curves for pure PP, PP/SBR, PP/C-rGO, and PP/SBR/C-rGO composites: (a) melting and (b) cooling.
Melting and crystallinity behavior of neat PP, PP/SBR, PP/C-rGO, and PP/SBR/C-rGO composites.
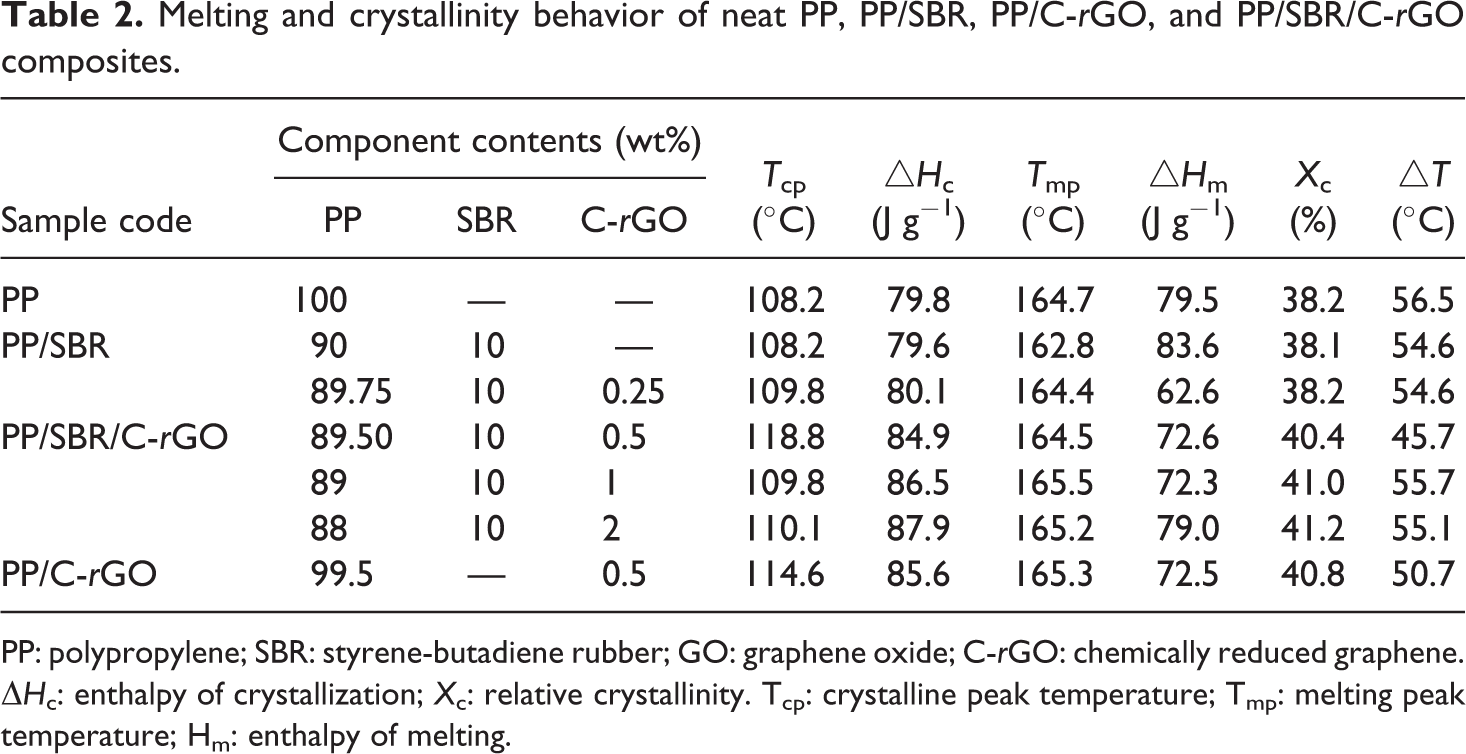
PP: polypropylene; SBR: styrene-butadiene rubber; GO: graphene oxide; C-rGO: chemically reduced graphene. ΔH c: enthalpy of crystallization; X c: relative crystallinity. Tcp: crystalline peak temperature; Tmp: melting peak temperature; Hm: enthalpy of melting.
The thermal stability of neat PP and PP/SBR, PP/C-rGO, and PP/SBR/C-rGO composites, estimated using TGA over the temperature range of 20–800°C, is shown in Figure 7. The data of initial degradation temperature (T on) and the maximum weight loss degradation temperature (T max) of all the samples are summarized in Table 3. Pure PP shows the T on and T max of 438.0°C and 454.8°C, respectively. For PP/C-rGO, the T on and T max are tested as 444.9°C and 462.7°C, respectively. It can be clearly seen that the thermal stability of PP/C-rGO system is only slightly improved in comparison to that of pure PP, probably due to the inorganic nature of C-rGO nanosheets. The presence of inorganic C-rGO nanosheets hinders the out diffusion of the volatile degraded products. For PP/SBR, it shows lower T on, but higher T max value than those of neat PP. However, the thermal degradation temperatures of PP/SBR/C-rGO system are significantly increased, and the most significant increase in the T on (by about 30.2°C) and T max (by about 23.0°C) at 1.0 wt% C-rGO content is observed for PP/SBR/C-rGO system, due to the synergistic incorporation of both C-rGO and SBR into PP matrix. The improvement on the thermal stability is greater than that of our previous works. 22,41
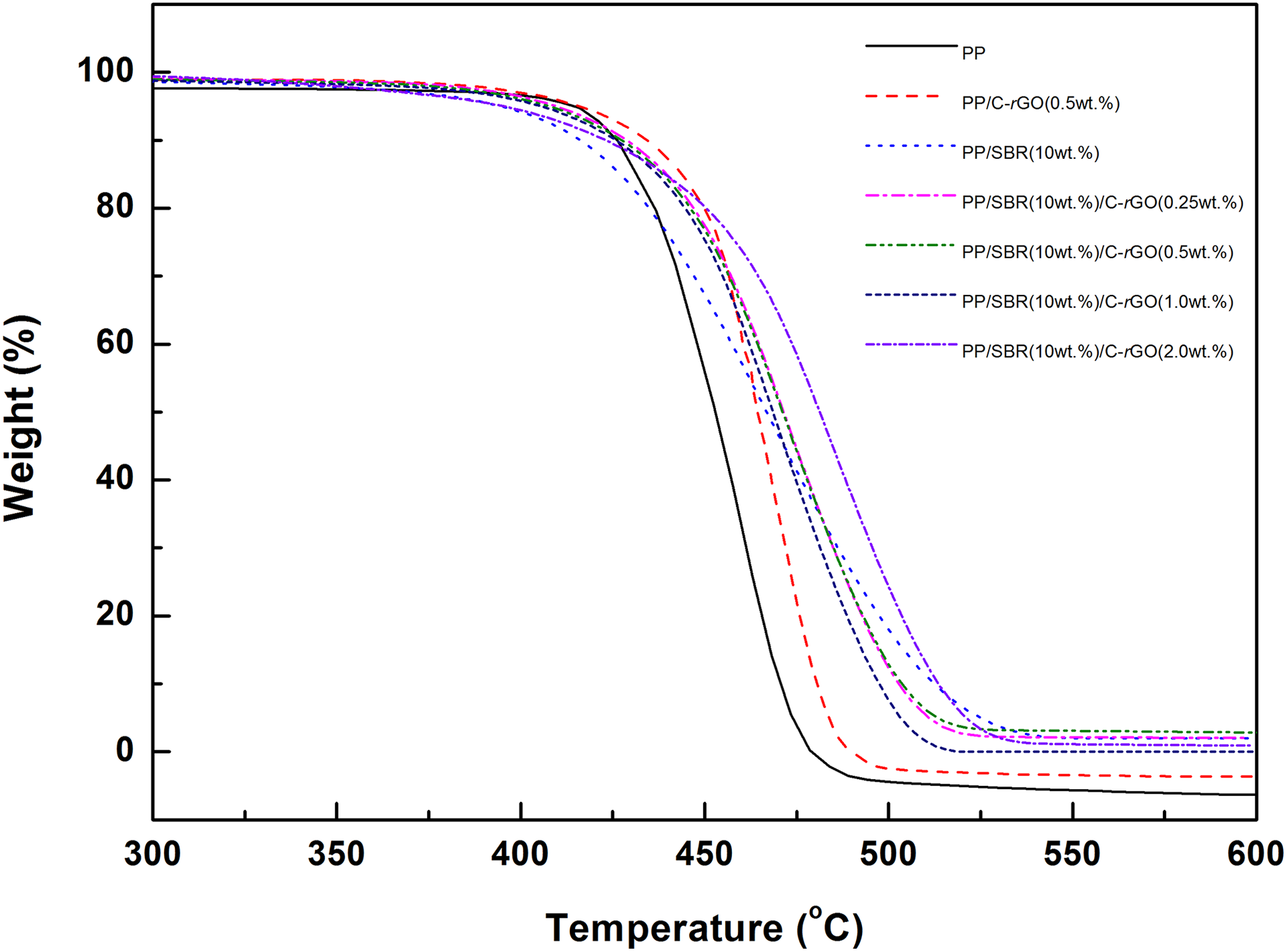
TGA curves of samples.
Typical TGA testing results.
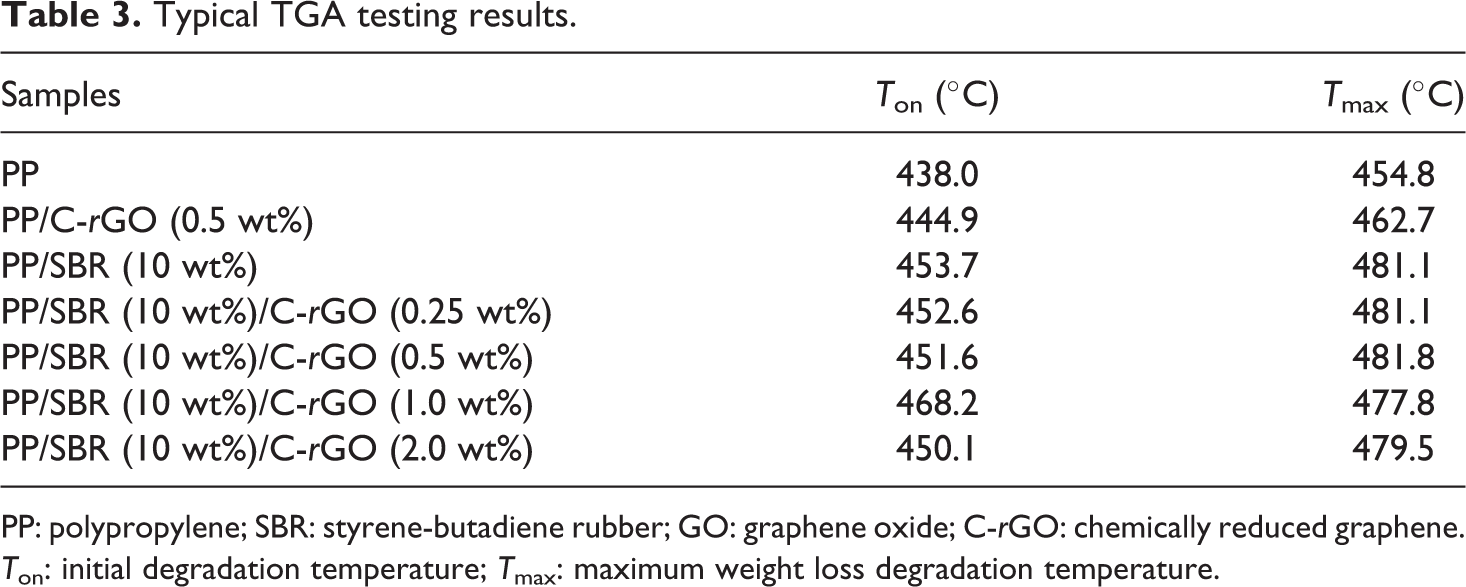
PP: polypropylene; SBR: styrene-butadiene rubber; GO: graphene oxide; C-rGO: chemically reduced graphene. T on: initial degradation temperature; T max: maximum weight loss degradation temperature.
Figure 8 shows the storage modulus (Figure 8(a)), loss modulus (Figure 8 (b)), and tan δ (Figure 8(c)) of PP, PP/SBR, and PP/SBR/C-rGO composites. It can be seen that compared with pure PP, the storage modulus of PP/SBR and PP/SBR/C-rGO composites increased, especially for PP/SBR/C-rGO composites containing 0.5 wt% C-rGO. When the content of C-rGO is 2.0 wt%, the storage modulus of the composite decreased, probably due to the aggregation of C-rGO. The loss modulus of PP/SBR and PP/SBR/C-rGO composites is higher than that of pure PP, which is attributed to the high elastic modulus of C-rGO, contributing to the reinforcing effect of the matrix. Further analysis from Figure 8(c), it can be seen that after doping with C-rGO, the tan δ loss peak induced by α relaxation moves to the higher temperature region. This phenomenon is more obvious at high C-rGO contents. The α relaxation described the movement process of PP segment, C-rGO nanoparticles restricted the molecular chain segment movements by anchoring mechanism, resulting in the increasing energy required for overall segment motions, leading to the tan δ loss peak shifts to higher temperature area.
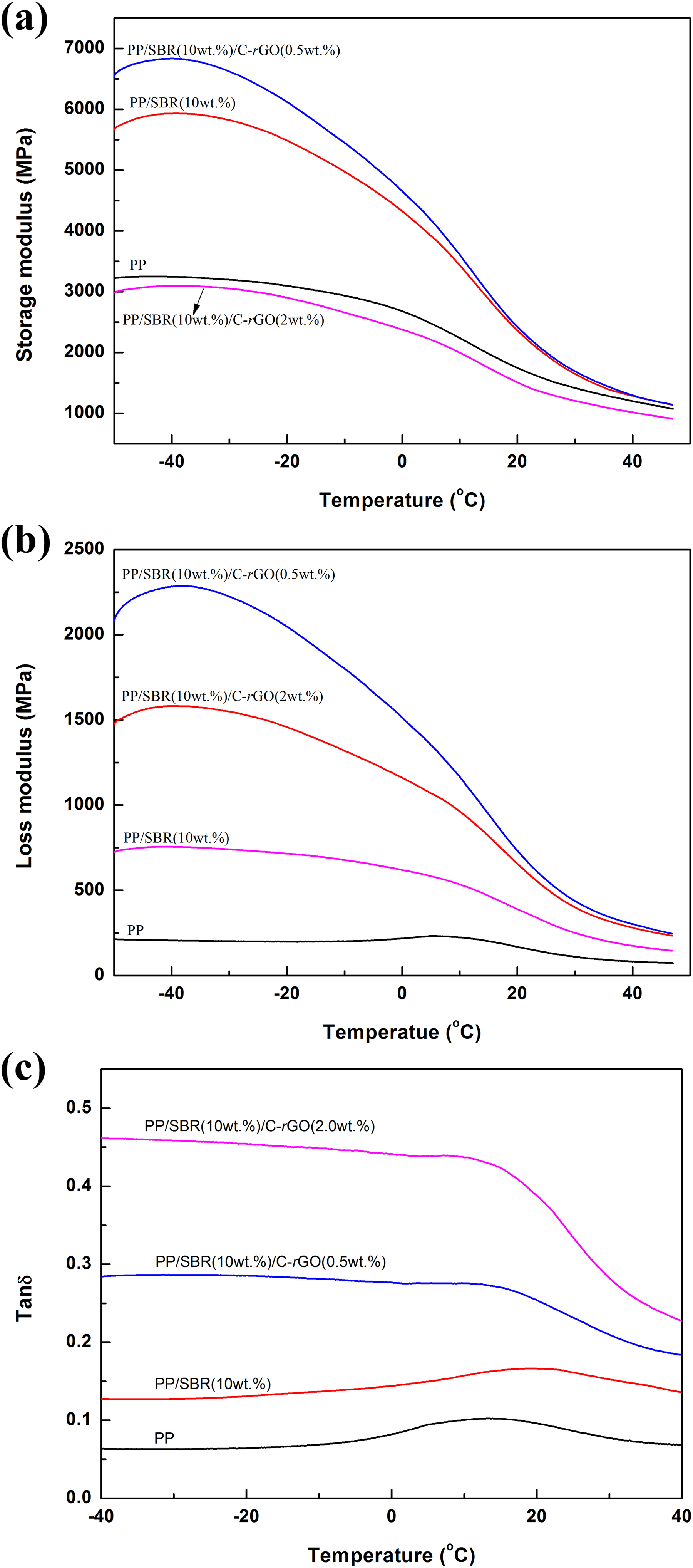
DMA curves of PP, PP/SBR, and PP/SBR/C-rGO composites: (a) storage modulus, (b) loss modulus, and (c) tan δ.
Conclusions
In this work, PP composites synergistically reinforced–toughened by SBR and C-rGO nanosheets have been fabricated via melt blending. We found that the mechanical properties of PP can be considerably improved by synergistically filling with C-rGO nanosheets and SBR, especially for the notched Izod IS, which is enhanced by 108.7% compared with that of pure PP when 0.5 wt% C-rGO is incorporated. The XRD data show that the formation of β-type crystals plays a role for the enhancement of the IS for PP/SBR/C-rGO composites. SEM observations on the fractured surfaces of the composites demonstrate that the dispersion of C-rGO and SBR in PP/SBR/C-rGO composite is homogeneous. The synergistic filling approach greatly enhances the dispersion state of nanoparticles among the matrix thus increasing the sites for dissipation of shock for impact energy in PP/SBR/C-rGO composites. The DSC analysis indicates that the crystalline peaks shift toward higher temperatures for PP/SBR/C-rGO (118.0°C) and PP/C-rGO (114.6°C) compared with that of pure PP (108.2°C), while the crystallinity also increases from 38.2% of pure PP to 40.4% of PP/SBR/C-rGO at the C-rGO loading of 0.5 wt%. The TGA characterization shows that the initial degradation and the maximum weight loss degradation temperatures increase by about 30.2°C and 23.0°C, respectively, at 1.0 wt% C-rGO content for the PP/SBR/C-rGO composites in contrast to those of pure PP. Thermal mechanical analysis results show that PP/SBR/C-rGO composite possesses higher thermal stability and storage modulus than those of PP.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by the Cooperation project of Chunhui plan of the ministry of education of China (Z2017070), open research fund project of key laboratory of automotive high-performance materials and forming technology of universities in Sichuan province (szjj2017-066); Scientific research project of Sichuan education department (17ZB0422); The national innovation and entrepreneurship training program for college students (201810623007), Sichuan Science and Technology Program (2019JDTD0024), and “Xihua Cup” Innovation and Entrepreneurship Project for College Students in Xihua University (2019-051) for the financial supports of this work.
