Abstract
A high loading rice husk fibre (RHF) incorporated polyolefin blend of recycled polyethylene high-density/poly(ethylene terephthalate) (rPEHD/rPET) biocomposite was produced through extrusion and hot/cold press process. Interfacial modification of biocomposites was conducted through alkalization (sodium hydroxide solution) of fibres, compatibilization (using E-GMA) and/or coupling agent (MA-g-PE) to improve the interaction between components and the homogeneity of materials blending. Alkalization of fibres had improved the impact strength of biocomposites. The compatibilization and coupling effects on immiscible rPEHD/rPET blend and recycled polymer/RH filler, respectively, had led to an enhancement of mechanical performance and morphological stability. Scanning electron microscope micrograph revealed the improved matrix–matrix and filler–matrix interaction through interfacial modification. This investigation had shown that RHF mixed well with rPEHD/rPET/RHF to produce eco-friendly biocomposite products.
Introduction
Because of the many advantages of natural fibres, that is, abundant, lightweight, low cost, non-toxic and environmental friendly, agricultural wastes are becoming increasingly important reinforcement in composite material. 1 Rice husk (RH) is a kind of natural fibres from agricultural wastes which produced in large quantities, approximately 840,000 tons a year in Malaysia. 2 The characteristics of lightweight and adequate compressive strength (floor and wall applications) and low thermal conductivity (which is lower than other building materials such as plywood) can be the main driving forces to make RH-filled composite suitable for use as an alternative material in construction applications. 3,4 Nevertheless, the reinforcement effect of RH in the polymer material is prominently affected by their weak adhesion due to the difference in polarity with non-polar or less polar matrix. 5 Thus, the surface modification of fibre (i.e. RH) or polymer is essential to achieve good mechanical properties of final composite. 6 A better comprehension on the role of polyolefin blends as a matrix as well as the reinforcement effect of second filler on nature and composite performances is still an interesting field to be explored. 7
Commonly, compatibilizer is used to modify the interface of inherently immiscible (polar/non-polar) polymer blend. In general, compatibilizer provides three major effects of chemical modification on the functional groups of polymer blend matrix. Firstly, to reduce the interface tension. Secondly, to restrict the agglomeration and reduce the particle size through emulsification effect. Thirdly, to increase the adhesion between both phases. These three effects are interconnected to provide optimum compatibility. 8 Copolymer containing glycidyl methacrylate (GMA) with an epoxy functional unit is more effective than that of having maleic anhydride (MA) functional unit due to the higher reactivity of functional group, as reported in polypropylene/polyethylene terephthalate (PP/PET) blend. 9
Surface modification of fibres (chemical treatment, i.e. alkali treatment with sodium hydroxide, NaOH) 10 –12 and the use of coupling agent (CA) such as MA-grafted onto polypropylene (MA-g-PP) 13 and PE (MA-g-PE) 14 are commonly applied to improve the hydrophilic biofibres/hydrophobic polymer interface. NaOH solution treatment is mostly used to clean the fibre surface as well as to promote the activation of the –OH group that exists in the cellulose unit through the breakdown of hydrogen bonds. Wetting and chemical reactivity on the surface of alkali-treated fibres could enhance adhesion properties, thereby contributing to better composite mechanical properties. 15 In our previous research, it was stated that the CAs have a reactive group which can react with –OH functional group on the fibre surface, whereas its ethylene block is responsible for physical entanglement with polymer matrix, especially polyolefin, that is, MA-g-PE is often applied for PE-based composites. 16
In lignocellulose-reinforced composite system, many researches have been performed using an extruder of either single screw or twin screw with one-way or opposite screw rotation, different temperature profile, screw speed and residence time. 17 According to Yang et al., 18 it was evident from the tensile properties findings that the composite material prepared by twin-screw extrusion system was better than that of single-screw extrusion system. This proves that twin-screw extrusion system could increase the distribution of fibres in the matrix (the agglomeration can be avoided). The better distribution of the fibre results in homogenous stress distribution and thereby enhancing the mechanical properties. In this study, composite materials were produced through extrusion process using a twin-screw extruder. To obtain good mixing, a two-step extrusion process was conducted on the materials where recycled PE high-density (rPEHD) and recycled poly(ethylene terephthalate) (rPET) were first extruded, followed by the second time extrusion of polymer blend matrix (pre-extruded) and rice husk fibre (RHF).
In our previous studies, the optimization of rPEHD/rPET blend composition and compatibilizer loading from the aspect of morphological behaviour and mechanical properties 19 as well as the effects of RHF filler loadings on the mechanical and physical properties of biocomposites have been reported. 20 However, these two articles are not focused on the effect of the chemical modifications types on the interfacial interaction between each component, either chemical bonding formation or physical interaction, in which the interaction mechanism in rPEHD/rPET biocomposites is important to appeal. In this study, the effects of compatibilization in polyolefin blend matrix, the alkali treatment of RHF and the addition of CA in blend/RHF composites on the mechanical and microstructure properties were studied. The polymer–polymer and filler–polymer interfacial interactions were discussed in depth.
Materials and method
Raw materials
Recycled rPEHD (density: 923 kg/m3, melt flow index: 0.72 g/10 min at 190°C) and rPET (glass transition temperature: 74°C, melting temperature: 253°C) were used as polymer blend materials. Ethylene-glycidyl methacrylate (E-GMA, trade name: Lotader AX8840, melt index: 5 g/10 min at 190°C, GMA content: 8%) was added as a compatibilizer for the recycled polyolefin blend. The agricultural waste filler used in this study was RHF. MA-g-PE (melting temperature: 135.2°C) was introduced as a CA in RHF-filled composites. The rPEHD, rPET, RHF, E-GMA and MA-g-PE were obtained by Bio Composite Extrusion Sdn. Bhd. (Malaysia). A sodium hydroxide (NaOH) solution with concentration of 4% was used to modify RHF powder via alkali treatment.
Alkali treatment of RHF
Before blending, RHF powder was dried at 100°C for 24 h. Raw RHF is labelled as untreated RHF (uRHF). The raw RHF powder was immersed in 4% NaOH solution at ambient temperature for 1 day. After 1 day, the soaked RHF powder was washed with water to eliminate excess NaOH. RHF powder was subsequently oven-dried at 90°C overnight. The treated RHF powder with NaOH solution is abbreviated as tRHF.
Preparation of biocomposite-reinforced RHF
The schematic preparation of RHF-reinforced biocomposites with different interfacial modifications is illustrated in Figure 1. The rPEHD and rPET were blended using the lab-scale Thermo Prisma TSE 16PC twin-screw extruder (Thermo Electron Corporation, Germany) having a diameter screw of 16 mm and a length/diameter ratio of 24. The extruder barrel temperature profile was applied at 250°C, 270°C, 240°C and 190°C, from feed zone to die zone, respectively, at a screw rotation rate (r/min) of 30. For untreated polymer blend (uPB, without E-GMA), the blend composition ratio of rPEHD/rPET was selected at 75:25 (wt%). Meanwhile, 5% of E-GMA compatibilizer was incorporated into rPEHD/rPET blend with the same blend ratio, and this modified polymer blend was named as the treated polymer blend (tPB).
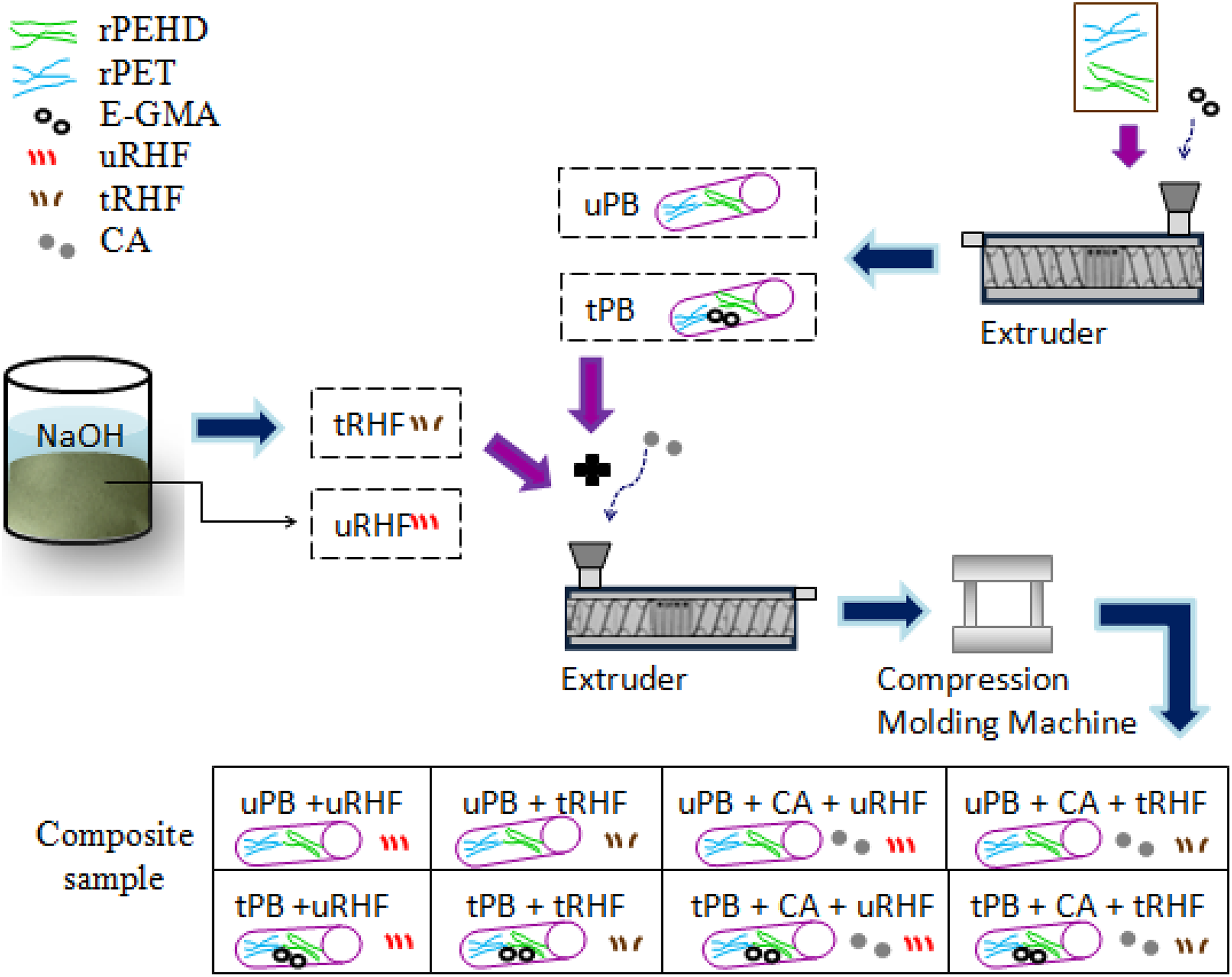
Preparation scheme of biocomposite-reinforced RHF flour with different interface modifications.
The pre-extruded polymer blend (uPB or tPB) pellets were blended with 70 wt% RHF powder (uRHF or tRHF) together with or without 3% CA 2 using the same extruder at 30 rpm. The fixed RHF powder loading at 70 wt% was determined from our previous studies. 20 A extrusion barrel temperature profile was applied at 170°C, 215°C, 210°C and 195°C. A hot/cold pressing was carried out using compression moulding machine (model LP50) at 200°C and 6.9 MPa. The periods of preheating, ventilation, full hot and cold pressing were set for 15 min.
Characterizations
Tensile and flexural mechanical tests were performed by a Testometric M350-10CT (Testometric materials testing machines, UK) following to ASTM D638-03 (Type I) and ASTM D790-03, respectively. In this study, a 5 mm/min speed was applied. The Izod (Impact System of Ray-Ran Universal Pendulum) testing was carried out at 3.46 ms−1 and 2.675 J. Five replicates for each tested composition were done to obtain the average values of mechanical test results.
Scanning electron microscope (SEM; model LE 1450 VP) was run to examine the surface morphologies of composite specimen fractured from tensile testing after coating with gold. Fourier transform infrared attenuated total reflectance (FTIR-ATR) spectroscopy was conducted on the powder (uRHF and tRHF) and solid/pelletized (E-GMA and other composite) samples with a wavelength from 650 to 4000 cm−1 (three zones: <1500, 1500–2000, 2500–4000 cm−1) using a PerkinElmer (Perkin Elmer Inc., USA) Spectrum 400 FTIR. An average of four scans is tested for the samples with the resolution of 2 cm−1.
Results and discussion
Mechanical properties
Figure 2(a) to (c) shows the tensile and flexural strength and flexural modulus of recycled polyolefin blend–based biocomposites with different interfacial modifications. Generally, biocomposites produced without MA-g-PE and E-GMA showed low mechanical properties due to the lack of interaction between the immiscible polymer blend (rPEHD and rPET) and RHF filler with great different polarity. The addition of CA and alkalization increased the tensile strength up to 95%. Comparing the types of blend matrix (uPB and tPB), it was noted that tPB-based biocomposites exhibited an increment in strength (approximately by 89.3% for tensile and 51.7% for flexural) and flexural modulus (approximately by 31.6%). This is ascribed to the strong interactions and chemical bonding formed between the non-polar rPEHD and polar rPET components via the addition of E-GMA compatibilizer (see Figure 3(b)). The epoxy functional group from GMA could react with the hydroxyl and carbonyl functional groups from rPET components, while ethylene segment (as backbone) from the E-GMA was compatible with PEHD components where a good wetting could be achieved. 8

(a) Tensile strength, (b) flexural strength, (c) flexural modulus, (d) impact strength and (e) elongation at break of biocomposite-recycled polymer blend/RHF with different interface modifications.
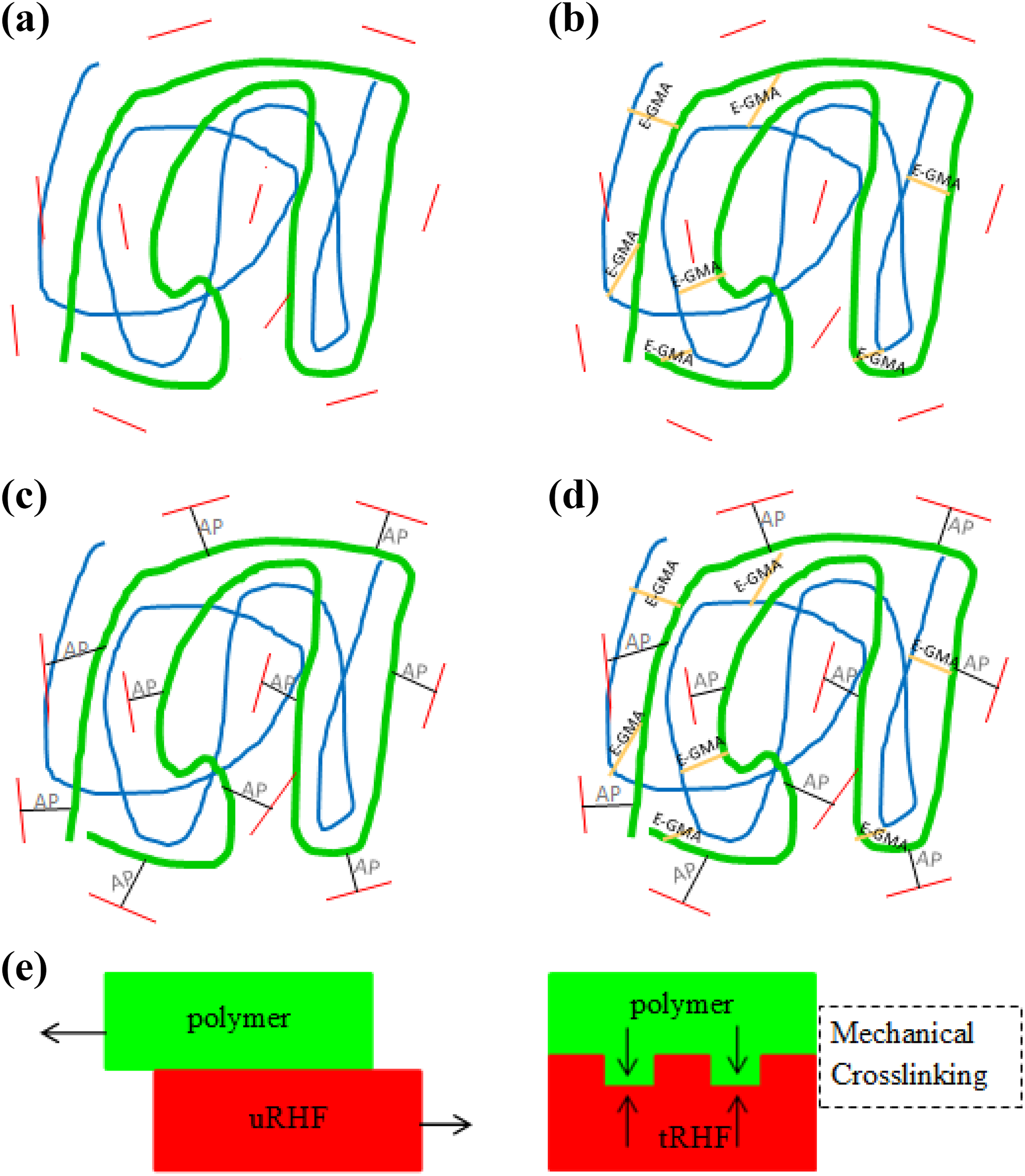
Representative schematic of chemical structure for biocomposite-reinforced RHF (a) based on uPB matrix, (b) based on tPB, (c) CA addition effect, (d) tPB + CA and (e) alkali treatment effect. Coarse curved lines indicate recycled PEHD; fine curved lines indicate recycled PET; straight line indicate rice husk fibre; CA: MA-g-PE link; E-GMA: E-GMA link.
By adding the CA into the manufacture of RHF/polymer blend biocomposites, the improvement of tensile strength and flexural properties shown can be explained by the enhanced fibre–matrix interface bonding. In which the chemical reaction occurred between the MA-g-PE with the RHF and the polymer matrix is illustrated in Figure 3(c). The anhydride functional group from MA-g-PE tended to react with hydroxyl functional groups from hydrophilic lignocellulosic filler and caused a consequent improvement in adhesion, 21 whereas the ethylene blocks of MA-g-PE promoted the polymerization with polymer matrix, particularly hydrophobic PEHD. 16
For alkali treatment of RHF (tRHF), the tensile strength and flexural properties of tRHF-filled biocomposites based on uPB and tPB without the presence of CA were higher than those of tRHF-filled biocomposites. The alkali treatment removes wax, impurities and hemicellulose from the fibre surface, resulting in a rough surface of RHF (as shown in Figure 3(e) on tRHF surface) and fibrillation. This increases the accessibility of available RHF surface onto the polymer matrix, thereby increasing the filler–matrix adhesion via mechanical interlocking mechanism (Figure 3(e)). 11 On the contrary, the smooth surface of uPB has resulted in a slippage between matrix polymer and RHF due to the weak interfacial adhesion (Figure 3(e)). Rahman et al. 22 had reported that PE/alkali-treated RHF composites exhibited a significant improvement in mechanical properties as compared to composites containing RHF treated in acidic and neutral medium. However, it can be seen that alkali treatment was only effective in biocomposites without the presence of CA. This is because the addition of CA could provide the chemical bonding between the fibres in which this was more effective than the alkali treatment that physically modified the fibre surface by mechanical interlocking.
The incorporation of compatibilizer (E-GMA) in the recycled polymer blend (rPEHD/rPET) and the addition of CA (MA-g-PE) into the polymer blend/RHF biocomposites promoted a positive effect in both impact strength (Figure 2(d)) and elongation at break (Figure 2(e)), which was similar to the increasing trend in tensile and flexural properties (Figure 2(a) to (c)). This finding is correlated to the improved miscibility, compatibility and interfacial bonding. Meanwhile, for biocomposites based on tPB matrix, tRHF filler provided higher resistance to impact and elongation. This is attributed to tRHF filler could mechanically interact with the matrix in comparison to the individual uRHF filler that tends to act as an initiator of microcracks.
Morphological observation
Figure 4 shows the fractured surface morphology of polymer blend/RHF biocomposites with various interfacial modifications. In general, the effects of three types of interfacial modifications on the morphological behaviours of composites comprising the same polymer blend/filler ratio (30/70 wt/wt %) were compared, which are first, the effect of adding E-GMA compatibilizer into the rPEHD/rPET by comparing Figure 4(a) and (b) with Figure 4(e) and (f) and Figure 4(c) and (d) with Figure 4 (g) and (h); second, the effect of introducing the MA-g-PE into the polymer blend/RHF composites by comparing Figure 4(a) and (b) with Figure 4(c) and (d) and Figure 4(e) and (f) with Figure 4(g) and (h); third, the effect of alkali treatment on RHF by comparing Figure 4(a, c, e, g) with Figure 4(b, d, f, h).

SEM micrographs of surface fracture of biocomposite sample (a) uPB + uRHF, (b) uPB + tRHF, (c) uPB + CA + uRHF, (d) uPB + CA + tRHF, (e) tPB + uRHF, (f) tPB + tRHF, (g) tPB + CA + uRHF and (h) tPB + CA + tRHF.
Compared to Figure 4(a) to (d) and Figure 4(e) to (h), biocomposites made from tPB matrix look more compact morphology structure than those of uPB-biocomposites. This observation can be explained by the absence of adhesion between the non-polar rPEHD and polar rPET, where both polymers were clearly visible in the form of two segregated phases.
Without the presence of CA in uPB-based biocomposites, the existence of a small gap is noted between the filler phase and the polymer phase (see Figure 4(a)). Meanwhile, Figure 4(c) displays the effect of uRHF breakage where this phenomenon indicates good interfacial interaction and stress distribution between the filler–polymer matrix as the filler pullout corresponds to poor interfacial bonding. For tPB-based biocomposites irrespective of uRHF or tRHF, the addition of CA had led to more homogeneous and smoother surface morphologies. This can be ascribed to the effective improvement of interactions between all components in composite materials where CA acted as an interface modifying agent for polymer/natural fibres, 23 besides the incorporation of E-GMA as compatibilizer in rPEHD/rPET blend. 8,19 This is evidenced by the improved mechanical properties.
By comparing the effect of the alkali treatment applied on the RHF filler, composites reinforced with tRHF exhibited a more coarse morphological structure with the obvious pulled fracture impact of the rPEHD component in the SEM micrographs (Figure 4(b, d, f, h)). The coarse structure is believed to be resulted from the strong mechanical interlocking between the tRHF and rPEHD-rich blend matrix as compared to uRHF filler. 10 Nevertheless, in the presence of CA, it can be observed that the tRHF fibre tended to agglomerate as shown in Figure 4(d) and (h). This observation suggests the encapsulation of tRHF by the CA (MA-g-PE) and weak interfacial bonding which contributing to the decreased tensile and flexural properties of biocomposites filled with tRHF and coupled with CA.
FTIR-ATR analysis
Figure 5 (a) and (b) describes FTIR spectra for biocomposites-reinforced uRHF and tRHF based on (a) uPB, (b) uPB + CA, (c) tPB, and (d) tPB + CA in the wavenumber range of 650–3850 cm−1, respectively. Generally, all polymer blend/RHF biocomposites showed absorption band at 3333–3340 cm−1 (O–H stretching vibration corresponds to functional group in cellulose fibre), 2,24 2916 and 2849 cm−1 (asymmetric, symmetric C–H stretching of a –CH2 from ethylene unit), 1715–1718 cm−1 (C=O bending vibration for ester groups in PET and E-GMA), 25 1099–1096 cm−1 and 1023–1020 cm−1 (C–O vibration for PET and RHF, respectively) and 690–900 cm−1 (C–H bending). 26
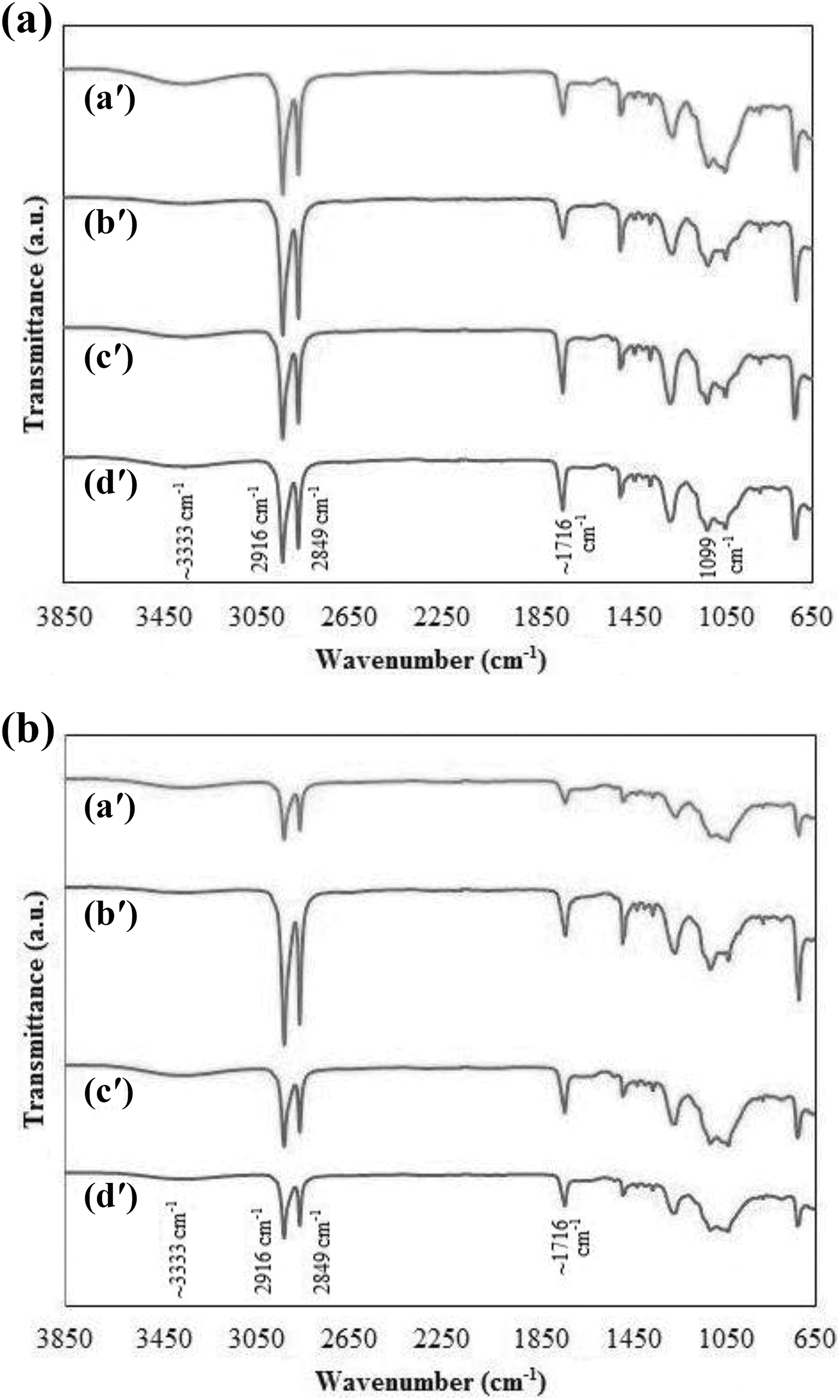
FTIR spectra of biocomposites-reinforced (a) uRHF and (b) tRHF based on (a′) uPB, (b′) uPB + CA, (c′) tPB and (d′) tPB + CA.
The compatibilizing effects of polymer blend interface via the addition of E-GMA, as shown in Figure 6. The epoxy glycidyl group that served as the functional group for E-GMA can be observed by the appearance of the absorption band at 997, 912 and 846 cm−1. Referring to uPB as a reference, the disappearance of absorption bands at 912 cm−1and 846 cm−1 was obviously visible in tPB where this phenomenon has confirmed the opening reaction of epoxy ring with the terminal groups from PET, which are the hydroxyl (−OH) and carboxyl (−COOH) groups. It was also found that the characteristics peak of PET, which is –OH group, was seen at around 3280–3318 cm−1 in uPB but disappeared in tPB. These observations confirmed the grafting reaction of CA onto PET and thereby enhancing the interfacial between PEHD-PET and the mechanical properties of biocomposites based on tPB.
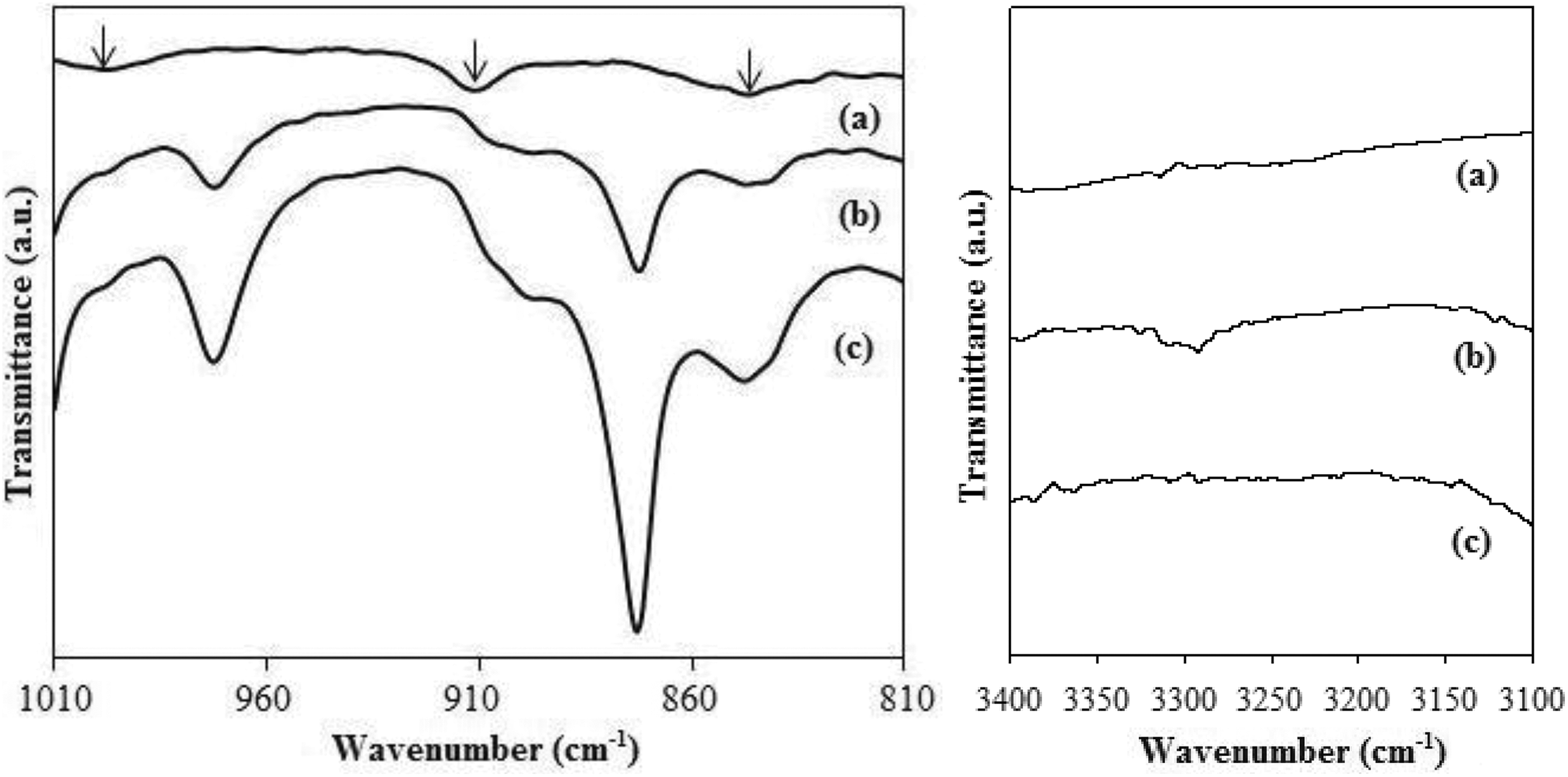
FTIR spectra of (a) E-GMA, (b) uPB and (c) tPB.
Figure 7 demonstrates the FTIR spectra of uRHF, CA which is MA-g-PE and uPB biocomposites-filled uRHF with and without the presence of CA. There were two observations that explained the reaction between RHF and polymer blend matrix via the addition of CA. Firstly, in the wavenumber of 3000–3600 cm−1, the shown absorption peak at 3340 cm−1 which characterizes the −OH group from uRHF to remarkably decline in the uPB/uRHF biocomposites coupled with CA as compared to those without CA. Secondly, in the wavenumber of 1650–1800 cm−1, MA-g-PE CA exhibited MA functional group by symmetry stretching of C=O at 1790 cm−1 where this peak was not visible in uPB/uRHF biocomposites. This may be due to the chemical interaction between the polymer and fillers as well as possibly because of the low amount of CA used in this study. However, as can be observed, the broad absorption band peak at 1716 cm−1 with lower intensity for uPB/uRHF biocomposites coupled with CA. This phenomenon indicates that CA could interact with RHF via the formation of covalent and ester bond between MA groups (from CA) and hydroxyl groups (on RHF surface). 27
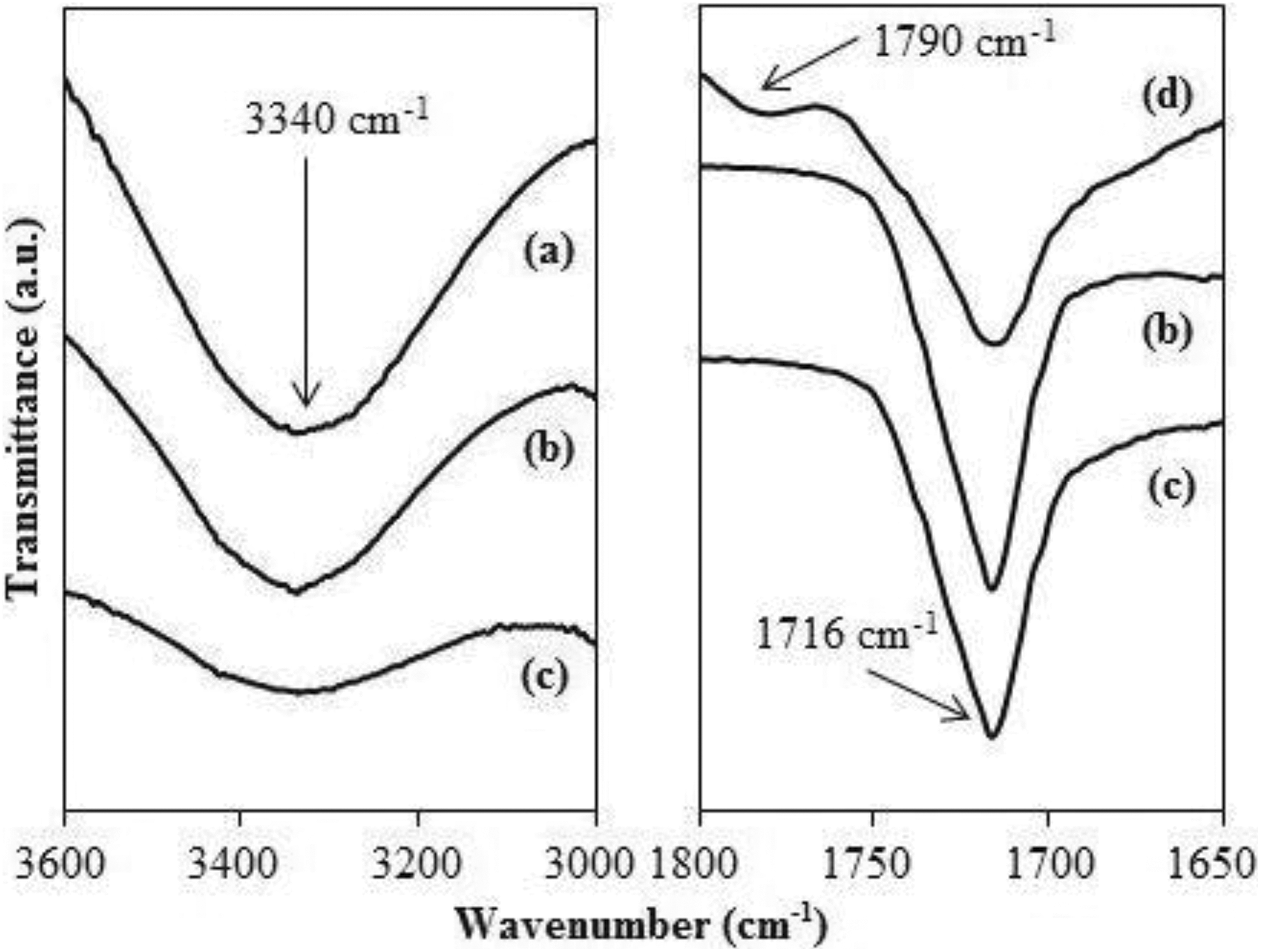
FTIR spectra of (a) uRHF, (b) uPB + uRHF biocomposite, (c) uPB + CA + uRHF biocomposite and (d) CA.
The FTIR spectra for raw RHF (uRHF), treated RHF (tRHF) and uPB-based biocomposites with both RHF are illustrated in Figure 8. Compared to uRHF, the FTIR spectrum for tRHF showed an increase in absorption band of O–H group (located at 3340 and 1660 cm−1) and C–O stretching (located at 1045 cm−1). 15 The elimination of RHF surface impurities causing more O–H groups to access 24 could be the possible reason for this phenomenon. The absorption band of O–H group at 3340 cm−1 and other main absorption peak for biocomposites filled with tRHF have a lower intensity as compared to those biocomposites containing uRHF. These results could be explained by phenomena occurred on the highly accessible hydroxyl group on tRHF surface (tRHF has a greater amount of –OH groups than that of uRHF); (1) chemical reaction between tRHF and CA, (2) the mechanical interlocking occurred between matrix and tRHF which had a rougher surface upon the removal of impurities and extractives on the exterior of RHF. In which these phenomena caused to the lesser amount of –OH groups in tRHF biocomposites.
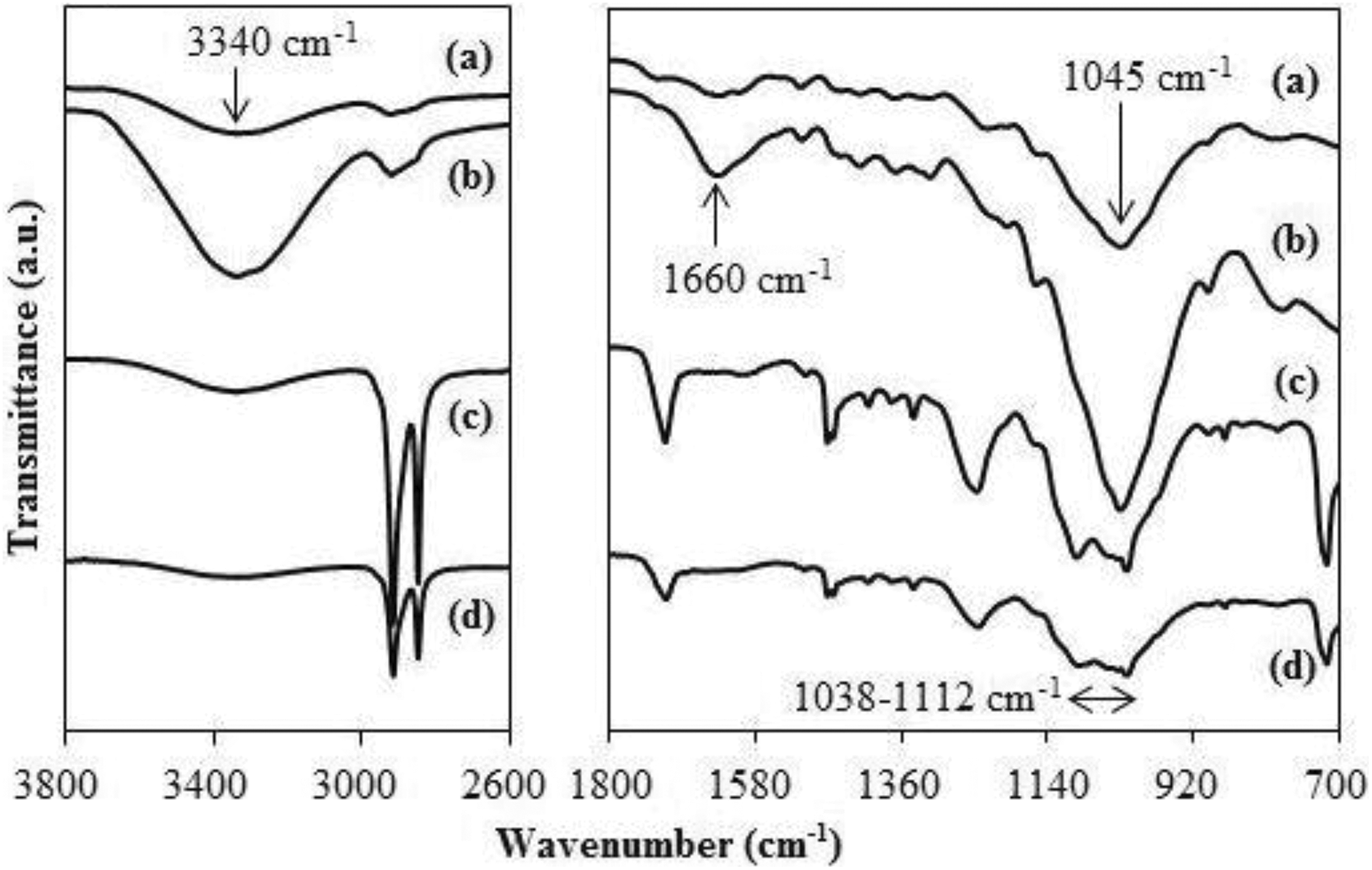
FTIR spectra of (a) uRHF, (b) tRHF (c) uPB + uRHF biocomposite, (c) uPB + tRHF biocomposite.
Conclusions
The rPEHD/rPET biocomposites incorporated with RHF at a high loading of 70 wt% were produced via a melt-compounding method, which was first extrusion for rPEHD/rPET and followed by a second extrusion of premixed polymer blend with RHF. With this preparation method, a homogeneous composite material was fabricated. The tensile, flexural and impact properties of rPEHD/rPET blend–based biocomposites were found to increase with the incorporation of E-GMA. The interfacial modification between recycled polymer and RHF through the addition of CA was effective to improve the mechanical performance, whereas the alkali treatment of fibres was only effective to enhance the impact strength of biocomposites based on tPB matrix. In conclusion, RH acts as a good candidate for being reinforcing filler in the blend (rPEHD/rPET) which to be used in automotive, furniture and construction applications.
Footnotes
Acknowledgements
The authors thank Electron Microscopy Unit and Center for Research and Instrumentation Management (CRIM), Universiti Kebangsaan Malaysia (UKM) and Bio Composites Extrusion Sdn Bhd for their support in terms of finance and materials.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was financially supported by Electron Microscopy Unit and Center for Research and Instrumentation Management (CRIM), Research Grant DIP-2016-023, GGPM-2018-061 and FRGS/1/2018/STG07/UKM/01/2 from Universiti Kebangsaan Malaysia (UKM).
