Abstract
Poplar leaves graphene (PLG) was synthesized from the poplar leaves. Modified poplar leaves graphene (M-PLG) was obtained by graphene treated with silane coupling agent γ-aminopropyltriethoxysilane (KH550). The M-PLG-modified silicone rubber (methyl ethylene silicon rubber [MVQ]) composites were prepared by mechanical blending. The effects of M-PLG on the mechanical properties, thermal properties, and morphology structure of MVQ were investigated. The results showed that the carbon content and purity of the PLG were high. The M-PLG can be evenly dispersed into MVQ compared with the PLG, the mechanical properties of the M-PLG/MVQ composites were better than those of the PLG/MVQ when M-PLG content was 0.1 parts per hundred rubber (phr), the tensile strength and elongation at break are increased by 36.2% and 19.4% respectively, and wear resistance increased by 57.1%. The thermal stability of the M-PLG/MVQ was also better. This concept of combining PLG with MVQ demonstrated the potential of preparing novel green materials with excellent properties.
Introduction
Chemically and thermally stable silicone rubber (MVQ) 1,2 is widely used in various fields such as medical field, 3 advanced national defense technology, aerospace manufacturing, and fine chemical industry; 4 –7 because its mechanical strength is very low, it is difficult to meet the needs for industrial use. 8 Therefore, MVQ had to improve the wear resistance, fatigue resistance, strength, and other properties to meet the engineering requirements. 9,10 Graphene, with large specific surface area, tensile strength, elastic modulus, electron mobility, and excellent conductivity, 11 –14 has become one of the hot spots in the research of new materials all over the world. 15 –18 At present, the domestic and foreign scholars have tried to use the graphene-modified electric properties of rubber. 19 –25 As the graphene is expensive, the graphite is currently used as a raw material to prepare graphene. However, a large number of poplar grows in northern China, and their leaves fall into waste in autumn. The carbon content of poplar leaves is 57.27%, and the carbon content of graphite is 54.11%. Therefore, the resource rich poplar leaves were chosen as raw materials to prepare graphene. However, it is a bottleneck problem that the graphene is difficult to uniformly disperse into MVQ. 26 To improve the compatibility between graphene and MVQ, the graphene was treated with silane coupling agent KH550.
In this work, the poplar leaves graphene (PLG) was prepared by oxidation–reduction method of poplar leaves, and the properties of MVQ were improved with KH550-modified poplar leaves graphene (M-PLG). The design concept of combining PLG with MVQ, coupled with the coupling agent KH550, will possibly lead to new insights in developing green materials for environmental protection with potential applications in material engineering, industry, as well as aerospace field.
Experimental
Materials
Silicone rubber (MVQ, methyl ethylene silicon rubber, ethylene content, 0.09 wt%, M n, 5.8×106) was supplied by DC Chejue organic silicon aggregation group (China). Silica (industrial grade) was obtained from Qingdao sweet silica Co., Ltd (China). Coupling agent KH550 (H2N(CH2)3Si(OC2H5)3; industrial grade) was supplied by Tianjin Shengli trade Co., Ltd (China). The other agents (analytical grade) were all common commercially available materials and were used as purchased (China). Poplar leaves were produced from Qiqihar in northern China.
Sample preparation
Synthesis of M-PLG
First, washing and drying the poplar leaves with deionized water several times, and the dried poplar leaves (less than 1 wt% water content) were ground to powder, then poplar leaves powder was carbonized in a tube furnace at 800°C for 1 h under nitrogen (25 mL min−1). After that, the carbonized sample was activated to prepare acticarbon with phosphate treated, and the acticarbon was continuously treated with concentrated sulfuric acid, potassium permanganate, and hydrazine hydrate, washed and dried to obtain PLG, put the PLG in a beaker containing deionized water with ultrasonic processing for 30 min, and then the silane coupling agent KH550 was added in the PLG, stirred fully until uniform, and finally dried to obtain an M-PLG solid.
Preparation of M-PLG/MVQ composites
MVQ was mixed on a XK-160 two-roll mill (Tianjin Electrical Machinery Plant, China) with the nip gap about 2 mm, at 50°C for 20 min, and various other ingredients (MVQ 100 phr, silica 5 phr, DCP 2 phr, accelerant M 2 phr, antioxidant D 1.5 phr, stearic acid 2 phr, ZnO 3 phr, and sulfur 1 phr) were added, and then 0.1 phr of M-PLG was added with the full mixing. Finally, the mixture was cured in the form of films (1 mm thick) in an electrically heated hydraulic press (XLB-D350 × 350, Shanghai First Rubber Machinery Co., Ltd., China) at 170°C under a pressure of 1.2 MPa for 10 min, and post cured at 170°C under 1 atm for 2 hours. Heat aged test samples were obtained by putting samples in an aging box (401B, Jiangdu True Power Machinery Co., Ltd., China) at 200°C under 1 atmosphere of air for 2 h (used for testing tensile strength and elongation at break after heat aging). The preparation of M-PLG/MVQ composites was showed in Figure 1.
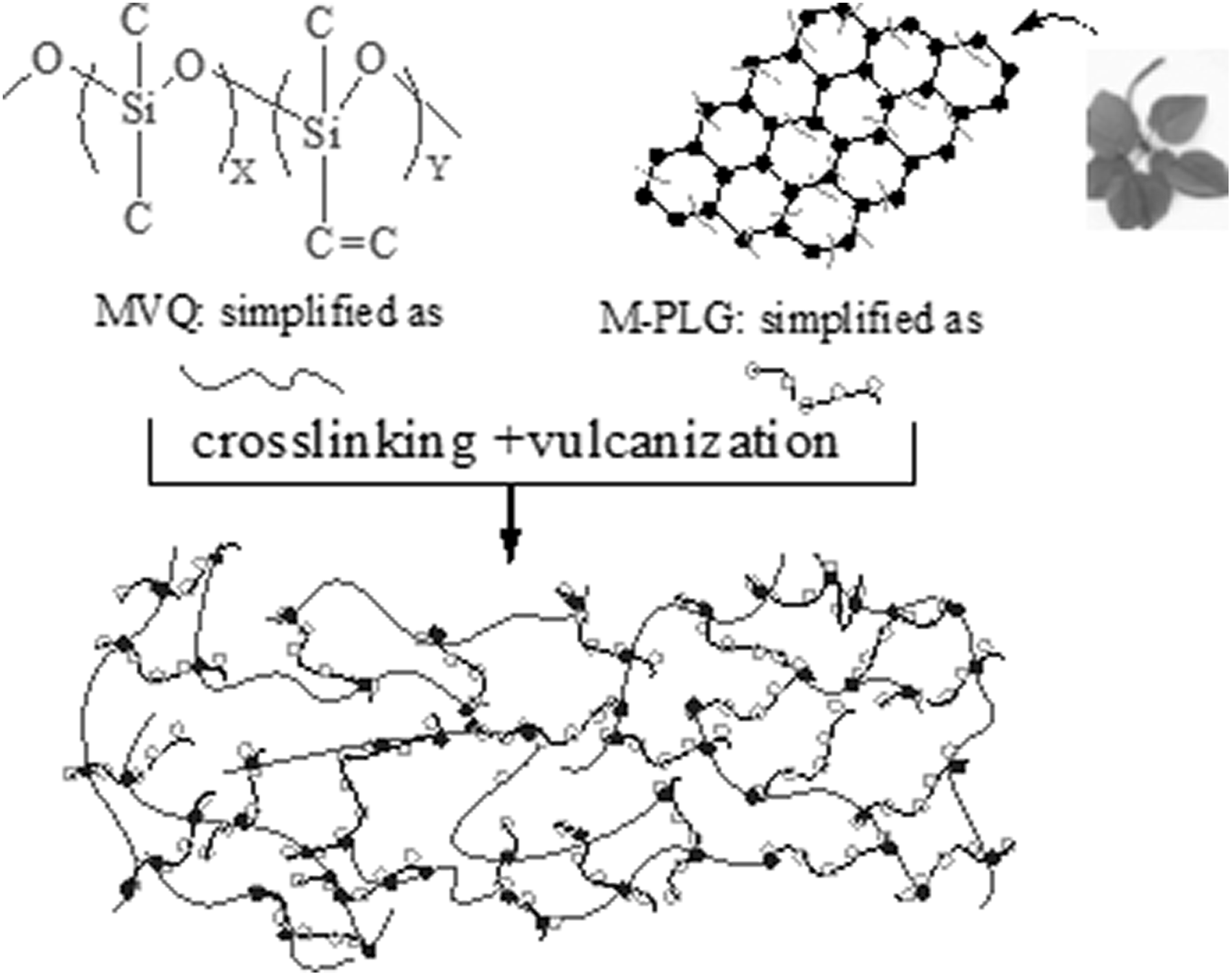
Preparation of M-PLG/MVQ.
Characterization
Organic element analysis test was carried on PE2400 analyzer (Perkin Elmer Co. Ltd., Waltham Massachusetts, USA); the temperatures of the combustion and the reduction tube are 925°C and 640°C, respectively.
XPS was recorded by using an X-ray photoelectron spectrometer (ESCALAB-250XI, Thermo Co., Waltham Massachusetts, USA) with an aluminum (mono) K α source. The aluminum K α source was operated at 15 kV and 10 mA. All core level spectra were referenced to the C1 s neutral carbon peak at 284.8 eV.
X-ray diffraction (XRD) analysis was performed with SmartLab XRD meter (Rigaku Corporation, Japan). The diffraction angle (2θ) is from 5° to 90°, the operating voltage is 45 kV, and the electric current is 200 mA.
Atomic force microscopy (AFM) analysis was carried out by a MultiMode 8 AFM (Bruker Instruments Inc., Germany) in tapping mode at a scan rate of 0.7–1 Hz.
The morphological investigation was done using transmission electron microscopy (TEM, H-7650, Hitachi Co., Japan) at an accelerating voltage of 100 kV.
The mechanical properties of the samples, such as tensile strength and elongation at break, were determined by a universal testing instrument (model CSS-2200, Zhongji Application Technical Institute, China) with a strain rate of 100 mm min−1 according to the ISO37 method. The effect of heat aging of the samples on the mechanical properties was measured according to ISO188. Abrasion tests were measured with a roller abrasion tester (High-Speed Rail Testing Instrument Co., Ltd, Qingdao, Shandong province, China) according to ISO 4649:2002, 2.5 N load. For each of the measurements, with the average reported for at least five samples, errors in the measurement of mechanical properties were within 10%.
Microstructural of vulcanizate specimen and elemental analysis of PLG were performed using a scanning electron microscope (SEM, S-4300, Hitachi Co.), in which the vulcanizate specimen was fractured in liquid nitrogen and the section surface was sputtered with gold.
The infrared spectra of the samples were recorded by using a Fourier transform infrared (FTIR spectrometer; model Spectrum One, Perkin Elmer Co. Ltd.). The samples were made by grinding 1–2 mg samples and 200 mg KBr together (both be dried), put in the mold, and pressed into slice with a hydraulic press under 5–10 MPa. FTIR spectra were collected with 256 scans at 2 cm−1 resolution in the region of 4000–500 cm−1 at room temperature.
The samples, the obtained vulcanizates, were cut into a size of 5 × 5 × 1 mm3 slices and were tested with a thermogravimetric (TG) analyzer (Netzsch Co. Ltd., STA449F3 Jupiter, Germany) in the temperature range from 20°C to 600°C using a heating rate of 5°C min−1 under nitrogen.
Results and discussion
Composition of poplar leaves and PLG
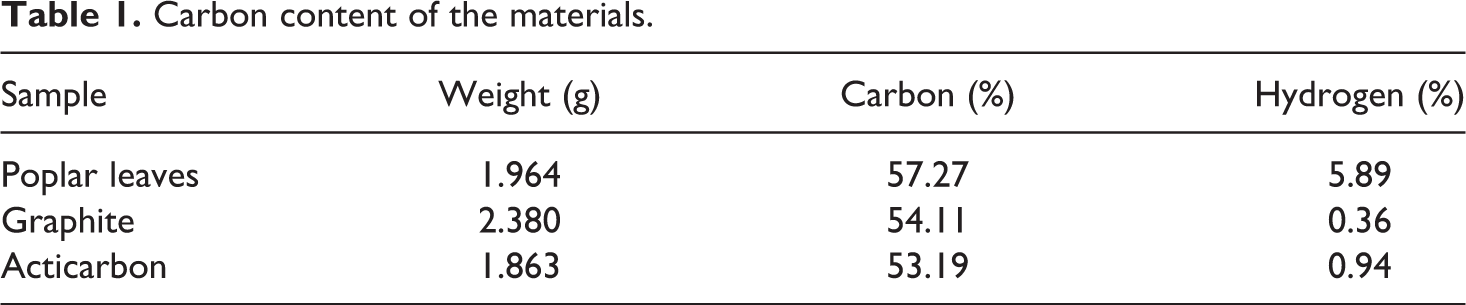
The carbon content of poplar leaves was studied as shown in Table 1. The carbon content of poplar leaves and graphite were 57.27% and 54.11%, respectively; the carbon content of poplar leaves was 3.16% higher than that of graphite; and the hydrogen content in poplar leaves was very high. After the poplar leaves were made into acticarbon, the hydrogen content decreased and the carbon content changed no significantly. It indicated that the graphene can be prepared from poplar leaves.
Carbon content of the materials.
Moreover, to further prove the element types of the three materials, X-ray photoelectron spectroscopy analysis was performed on the materials (in Figure 2(a)). The poplar leaves has the peaks of Si2p, C1 s, O1 s, K2p, Cl2p, and Ca2p, which indicated that there were Si, C, O, K, Cl, and Ca elements in the poplar leaves. The activated carbon had the peaks of Si2p, C1 s, O1 s, and N1 s, which indicated that there were Si, C, O, and N elements in the acticarbon, the mixed elements in poplar leaves disappeared after the acticarbon was made, and the carbon content had not changed much. The PLG prepared by the redox method contained the peaks of Si2p, C1 s, and O1 s, demonstrating that the surface of PLG contained only C, Si, and O element, and the content of carbon element was more.
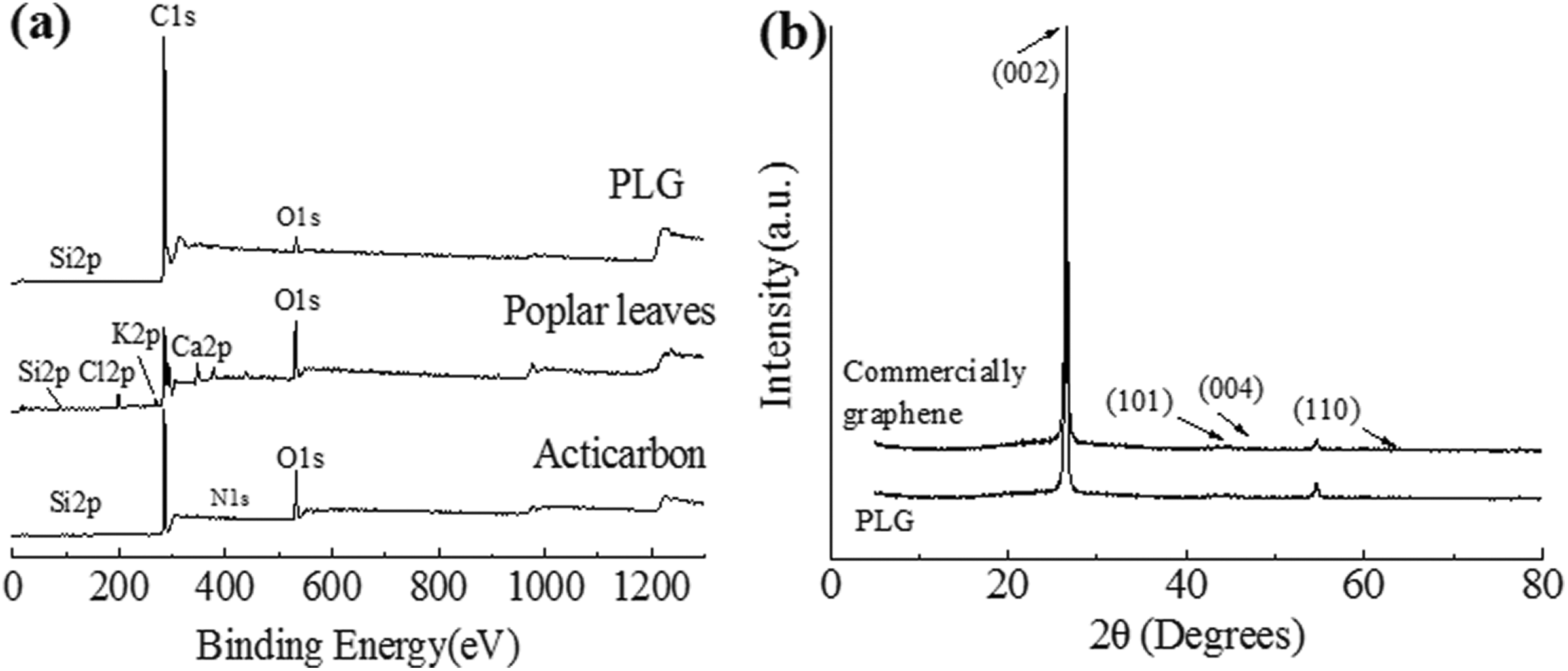
(a) XPS spectra of acticarbon, poplar leaves, and PLG; (b) XRD pattern of the PLG and commercially graphene.
To compare the purity of PLG and commercial graphene, the samples were analyzed. XRD patterns of PLG and commercial graphene were illustrated in Figure 2(b). Diffraction peaks at 26.43°, 44.70°, and 54.54° are the typical peaks of orthorhombic phase of graphene, which were assigned to the (002), (101), and (004) of graphene, respectively (PDF card no. 41--1478). Both graphene had a strong absorption peak at 2θ = 26.5°, and the absorption peaks of PLG were weaker than those of the commercially graphene, which demonstrated that the regularity and crystallinity of graphene were greatly improved during oxidation–reduction process and the PLG had high purity.
Morphology and structure of PLG
In order to understand the microstructure of PLG and M-PLG, the surface morphology of graphene was observed by AFM as shown in Figure 3. The surface on the PLG sample presented a round thin sheet as shown in Figure 3(a) and there was irregular and thick sheet on the surface of the M-PLG as shown in Figure 3(b), which suggested that the surface of the PLG was attached with coupling agent KH550.
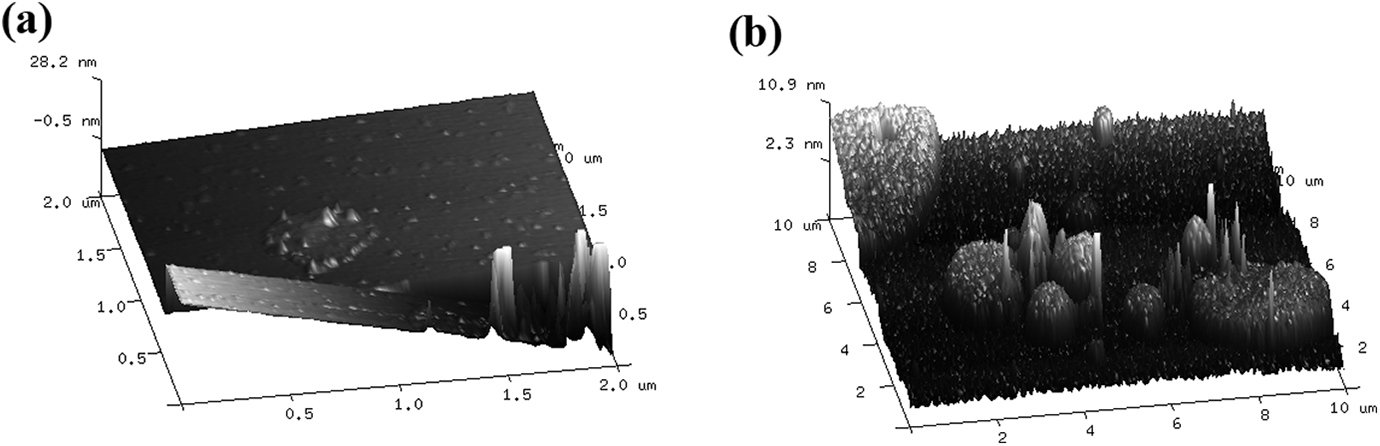
AFM analysis of (a) PLG and (b) M-PLG.
The EDS analysis was conducted as shown in Figure 4 to prove whether the adhesive material on the M-PLG surface was KH550. Figure 4(a) was unmodified PLG, and Figure 4(b) was M-PLG. It was obvious from the EDS that the PLG contains only C element and the M-PLG contains C, Si, and O element, which further proved that the silane coupling agent KH550 had been successfully attached on the PLG surface.
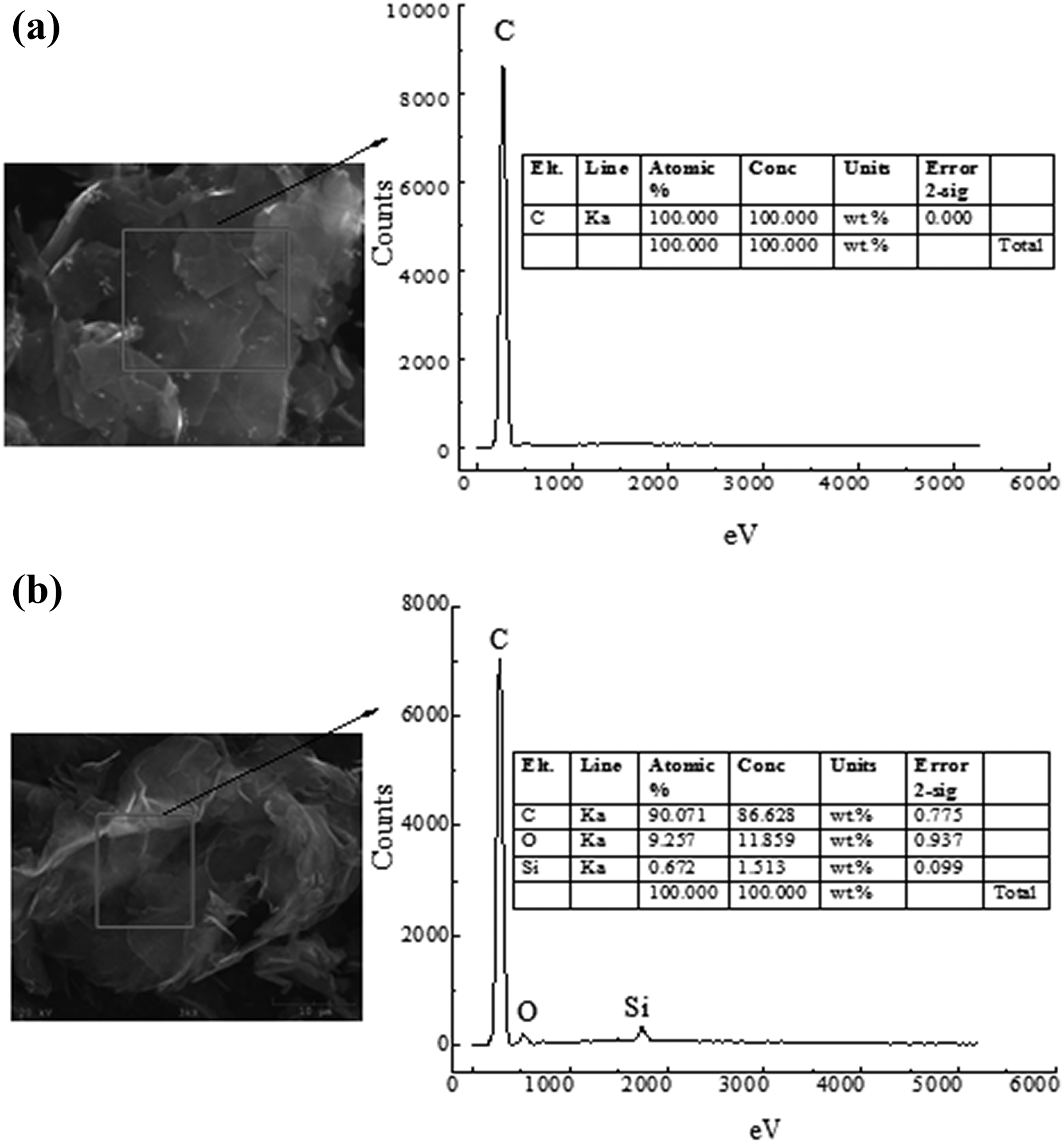
Energy-dispersive spectrometer (EDS) spectra of (a) PLG and (b) M-PLG.
Mechanical properties of PLG/MVQ
The effects of PLG and M-PLG on mechanical properties of MVQ were studied, as the addition of the PLG and M-PLG, the tensile strength, and the elongation at break of the composites increased first and then decreased as shown in Figure 5(a) and (d). It indicated that the two kinds of graphene had an improved effect on the MVQ. When 0.1 phr PLG was added, the tensile strength and the elongation at break of the PLG/MVQ composites showed the best value as shown in Figure 5(a), which suggested that the PLG was dispersed evenly in the MVQ and the mechanical properties were improved. When the content of PLG was more than 0.1 phr, the decrease of the tensile strength and the elongation at break was due to the aggregation of PLG, resulting in uneven dispersion in the MVQ and the worse compatibility between PLG and MVQ. The Shore A hardness of PLG/MVQ composites increased with the increasing amount of PLG, and the Shore A hardness of the composites gradually increase, it seemed that the more the PLG content was, the more cross-linking points between rubber and PLG were, and the harder the material. In addition, the tensile strength and the elongation at break after heat aging were basically the same as those before heat aging, as shown in Figure 5(b), but the Shore A hardness was higher than that before aging due to heat embrittlement of the rubber. Similarly, it can be explained that the wear of the composites decreased first and then increased with the addition of PLG.
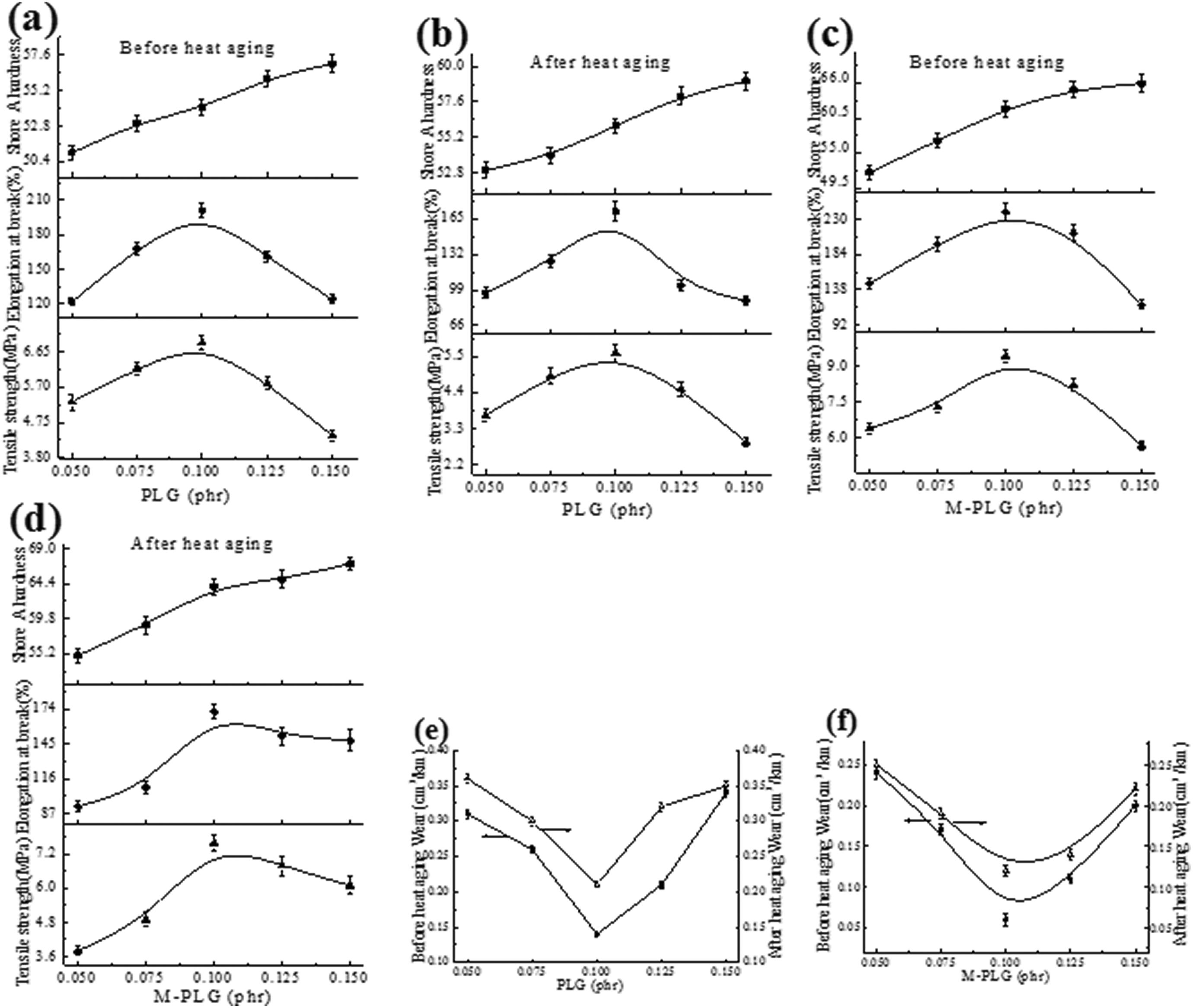
Mechanical properties of PLG/MVQ (a) before heat aging and (b) after heat aging, M-PLG/MVQ (c) before heat aging (d) after heat aging, and wear of (e) PLG/MVQ and (f) M-PLG/MVQ.
It can also be seen that the tensile strength and elongation at break of the M-PLG/MVQ composites increased first and then decreased at 0.1 phr of M-PLG as the addition of M-PLG as shown in the Figure 5(c) and (d). Compared to the PLG/MVQ composite with 0.1 phr of PLG, the tensile strength and the elongation at break of M-PLG/MVQ composite increased by 36.2% and 19.4%, respectively, which showed that the effect of M-PLG-reinforced MVQ was much better than that of PLG. It was considered that PLG was easily agglomerated and had poor compatibility with MVQ. Therefore, the coupling agent KH550 was used as the interface phase between PLG and MVQ, and the surface of the PLG was treated to disperse evenly in the MVQ matrix. The trend of Shore A hardness of M-PLG/MVQ composites increases first and then tends to be constant, and the wear decreases and then increases as shown in Figure 5(e) and (f), and the best mechanical properties appeared at 0.1 phr of PLG or M-PLG. Compared with PLG/MVQ composites, the hardness was increased by 20.4%, and the wear resistance was increased by 57.1%.
Morphology and structure of PLG/MVQ
To compare the dispersion of PLG and M-PLG in MVQ, SEM and transmission electron microscopic analyses of the PLG/MVQ and M-PLG/MVQ composites were shown in Figure 6. It was found that PLG/MVQ composites had a rough surface, many holes, and large gaps as shown in Figure 6(a), which indicated that binding force between PLG and MVQ was weak. The fracture surface of M-PLG/MVQ composites had no stratification and was closely connected with MVQ, as showed in Figure 6(b), which suggested that the binding force between the M-PLG and the MVQ was significantly enhanced. Compared Figure 6(a) and (b), it can also be seen that the M-PLG was dispersed in the MVQ relatively uniformly.
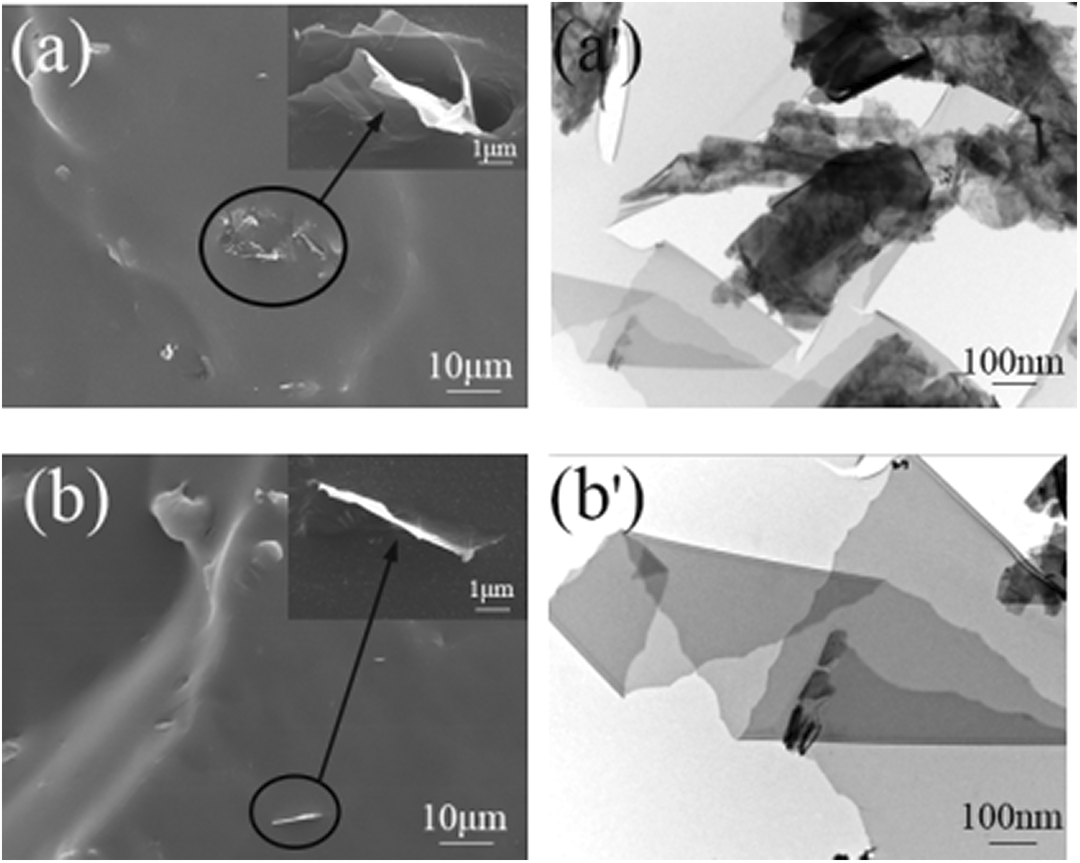
SEM micrographs of (a) PLG/MVQ and (b) M-PLG/MVQ and TEM micrographs (c and d) of the same specimens.
It can be seen from Figure 6(c) that the PLG had agglomerated and it was difficult to uniformly disperse in the silicone rubber matrix, and the M-PLG could be dispersed in the MVQ on its own, without piling up as shown in Figure 6(d); the results were consistent with those of the SEM, so the compatibility of KH550-modified PLG with MVQ was better than that of PLG.
The IR spectra of the PLG/MVQ composites retained the absorption peaks of OH (3430 cm−1), CH (2956 cm−1), C=O (1257 cm−1), and Si–O (1100 cm−1) as shown in Figure 7, which was seemed to be the characteristic peaks of the MVQ and silica. A new absorption peak of NH bonds at 1631 cm−1 appeared in the M-PLG/MVQ curve, which was believed the KH 550 reacted with the composites and generated NH group that promoted the cross-linking of the composites. So KH 550 plays a good cross-linking role.
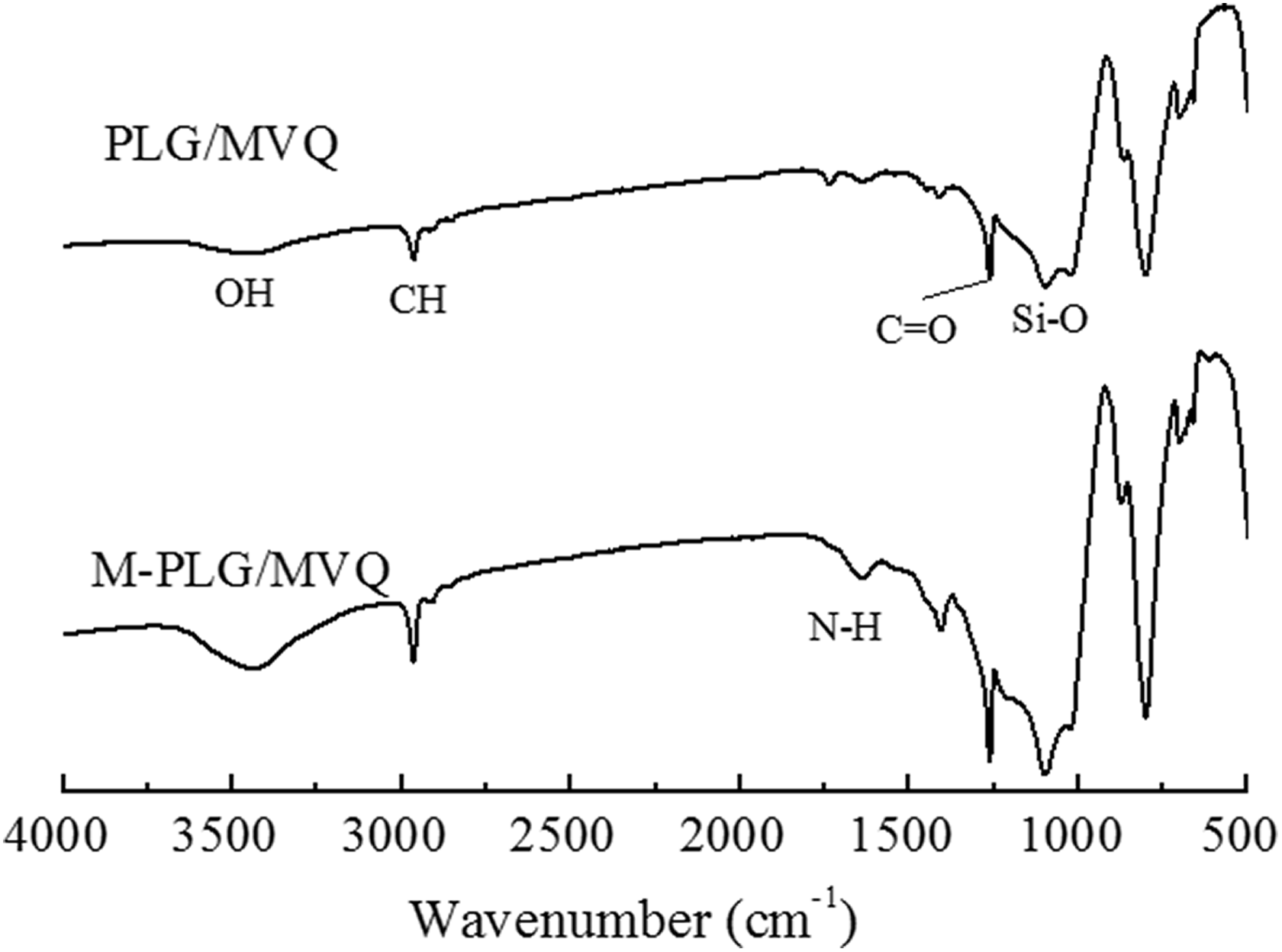
FTIR spectra of PLG/MVQ and M-PLG/MVQ.
Thermal properties of the PLG/MVQ
As can be seen from Figure 8, the weightlessness of the M-PLG/MVQ, PLG/MVQ, and MVQ occurred above 300°C due to the evaporation of water and decomposition of some impurities after heating. The weightlessness rate of MVQ increased obviously above 400°C. The weightlessness rate of PLG/MVQ was faster than that of M-PLG/MVQ above 500°C, which indicated that the coupling agent KH550 played a good cross-linking role on PLG and the thermal stability of M-PLG/MVQ in high temperature range was better than unmodified PLG.
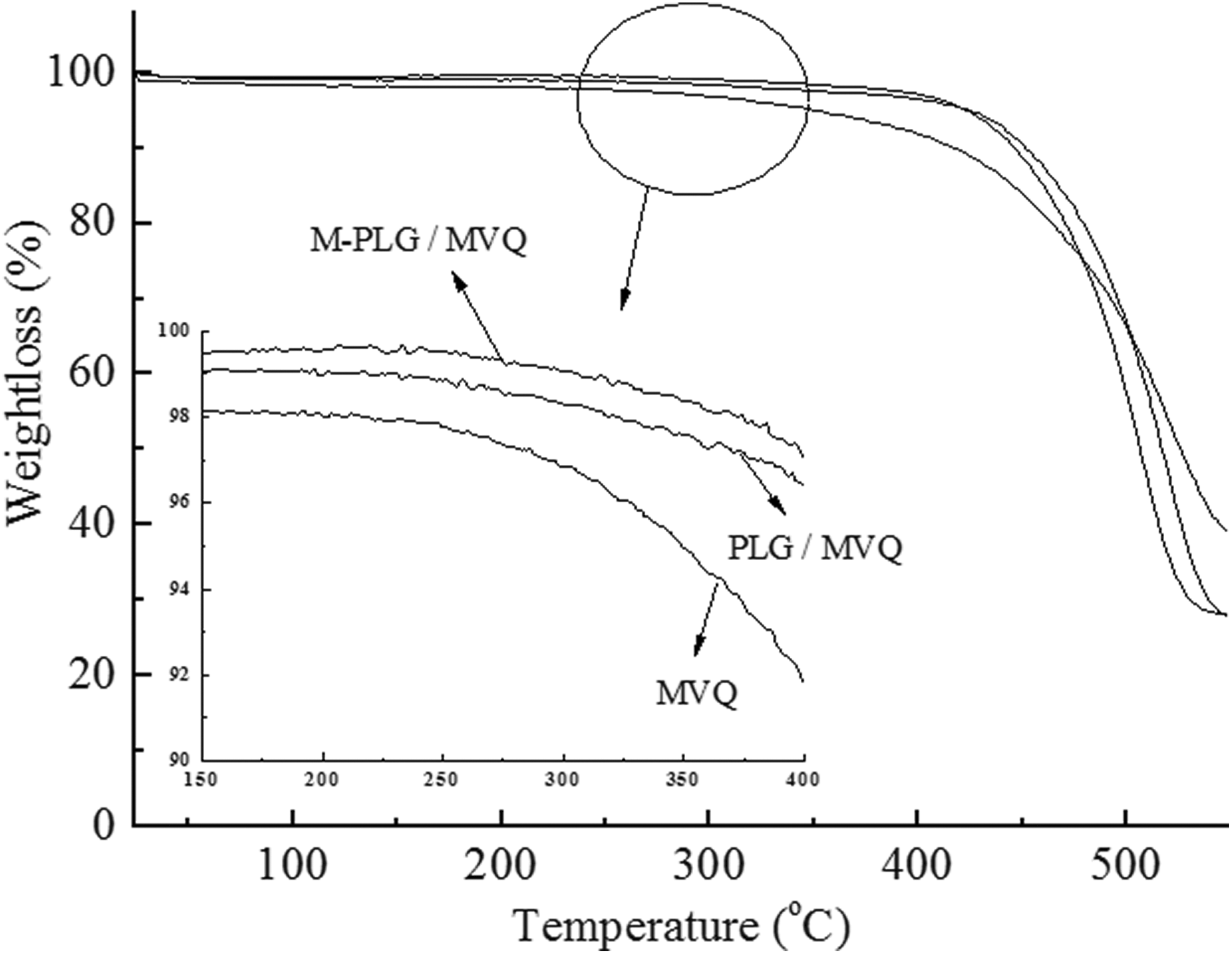
TG curves of MVQ, PLG/MVQ, and M-PLG/MVQ.
Conclusions
The PLG with nanometer level had been successfully prepared, the content and purity of carbon element were high, and the absorption peak was at 26.5°. The silane coupling agent KH550 had been attached on the PLG surface to prepare M-PLG. The M-PLG could be better dispersed in the MVQ than PLG. When the content of M-PLG was 0.1 phr, the mechanical properties of the M-PLG/MVQ composites were the best. Compared to the PLG/MVQ composites, the tensile strength and the elongation at break increased by 36.2% and 19.4%, respectively, and wear resistance increased by 57.1%. The thermal stability of the M-PLG/MVQ was also better than that of the PLG/MVQ. The design concept of combining PLG with MVQ would possibly lead to new insights in material engineering, industry, as well as aerospace field.
Footnotes
Funding
The author(s) disclosed financial support for the research, authorship, and/or publication of this article: This work was supported by provincial higher education fund basic research business expenses research project of Heilongjiang Provincial, China (No. YSTSXK201864), university student innovation and entrepreneurship training of Heilongjiang Province, China (No. 201810232061), and 2018 graduate student innovation research of Qiqihar University, China (No. YJSCX2018-038X).
