Abstract
Both gelatin and polyvinyl alcohol polymers have good biocompatibility to be used in biomedical applications. Cell adhesion and growth activity of gelatin have made it unique in artificial cartilage preparation. Two separate solutions of them are prepared by mixing 10% (w/v) of each one in water. These two solutions are then mixed at a temperature range of 70–90°C, and a pH value of 2 is maintained and finally it is cast into films. The dried films are gamma irradiated (using 60Co) at different doses. Fourier transform infrared analysis of the films shows the presence of amide linkages formed in them. It is observed from thermogravimetric analysis that 2.6% of non-irradiated and 20% of irradiated samples retain after heating them up to 600°C. Mechanical properties of the irradiated films show better properties than that of non-irradiated one. The irradiated films show a decrease in water uptake than that of the non-irradiated films. Scanning electron microscopy does not show any significant difference in morphology of the films with increasing radiation doses.
Introduction
Cartilage is a flexible connective tissue found in many areas in the bodies of humans and animals, including the
When considering biodegradable polymeric replacement for cartilage, several parameters need to be addressed like sufficient mechanical properties, adequate lubrication, good biocompatibility, good adhesion to the surrounding tissue, and of course resisting wear and fatigue. Many synthetic polymers such as polyvinyl alcohol (PVA), polyacrylic acid, polymethacrylic acid, polyglycolic acid, and polylactic acid meet the above requisites except mechanical properties. 5 PVA is one of the first synthetic polymers used as artificial cartilage, although it does not have enough mechanical stability like other polymers. However, PVA offers several beneficial properties to be used as artificial cartilage. It has high solubility in water, excellent film forming, emulsifying and adhesive properties, moderate TS and flexibility, and relative biocompatibility. It can also be sterilized and molded into a desired shape. PVA has been investigated as an artificial cartilage replacement mostly in the form of hydrogels, because they have rubber elastic physical properties and can be manufactured having a TS in the cartilage range of 1–17 MPa and compressive modulus varying from 0.0012 MPa and 0.85 MPa, depending on the polymer concentration and number of cycles tested. 6,7 PVA cryogels (gel matrices formed at moderately frozen condition) have also been used as an artificial cartilage for the replacement of arthritic cartilage in shoulder joints. 8 Moreover, as a film, PVA becomes transparent and more resistant to tear and punctures. 9
Polymer blends exhibiting two-phase behavior can achieve mechanical compatibility as well as a range of characteristics between the component polymers and thus can guide to a large number of potentially useful and different products. 10,11 In many researches, PVA has been blended with several polymers, macromolecules, and ceramics such as gelatin, starch, 12 chitosan, 13 hyalauronan, 14 hydroxyapatite (HA), 15 tricalcium phosphate, 16 and so on. Among these blend materials, gelatin can be pointed out. It is a water soluble (at temperatures above solgel transition) animal source protein. Gelatin can be considered different from other proteins due to a noticeable absence of internal order. At molecular level, gelatin-gel formation involves protein re-structuration, with the transition of a disordered state to a more ordered structure, and the physical properties of these gels result from the formation degree of microcrystalline junctions; consequently, it has been widely used as a biomaterial in tissue engineering. 17 –19
The blend of PVA and gelatin has been reported by a few numbers of authors. Pal et al. prepared hydrogels by esterification of PVA with gelatin and successfully used them as wound dressing. 20 Gelatin as a sheath and PVA as a core in electrospun nanofiber demonstrated an increase in stiffness and TS and a decrease in plastic deformation, and they were also susceptible to be used for biomedical applications. 21 Wang et al. have mixed HA with gelatin and PVA by emulsification to create a cartilage scaffold for tissue engineering and studied this composite material in vivo by implanting it subcutaneously in the dorsal region of rats for 12 weeks. 22 The results indicated that the composite scaffold HA/PVA/gelatin is biocompatible and may serve as a cartilage scaffold for tissue engineering applications.
The present work is to prepare films from PVA and gelatin and the cross-linking of the films is to be conducted by gamma (γ) radiation. Chemical, thermal, and mechanical properties and cytotoxicity of the films are investigated for its potential use as artificial articular cartilage.
Experimental
Materials
Gelatin (bloom strength 185, pharmaceutical grade, Opsonin Pharma Ltd, Bangladesh) and PVA pellets of M W 70,000 g mol−1 (Merck, Germany) were used for the preparation of different films. Hydrochloric acid (HCl; Merck) and sodium hydroxide (NaOH; Merck) were also used to maintain different pH of the blend solutions.
Preparation of gelatin-PVA films
Two separate solutions of one with 10 g of PVA into 90 ml of water and another one with 10 g of gelatin into 90 ml of water were prepared in two separate conical flasks with constant stirring and heating at 70–90°C using hotplates until all the PVA and gelatin completely dissolved into water. These two solutions (gelatin solution and PVA solution) were then completely mixed with continuous stirring at 70–90°C. After cooling to room temperature at ambient condition, 20 ml of the blend solution was taken in each of nine beakers, maintaining pH from 1 to 9 by using 1 M HCl and 1 M NaOH. The nine beakers were allowed to stand for 1–2 h to observe that whether any layer formed in the mixture of PVA and gelatin. It was observed that at pH 1 and 2 the blend solution did not form any layers, while it happened at pH higher than 2 for the rest of the blends. The solubility of PVA and gelatin blend solution is pH dependent due to partially unfold situation of gelatin protein in acidic and alkaline condition. 23 The blend solution having pH 2 was further used to cast onto silicon cloths followed by ambient air drying to form the final films.
γ-Irradiation technique
γ-Radiation source-type T-T252 loaded with source Co-60 pellets (925 TBq model γ beam 650 is loaded with source GBS-98) that comprises of 36 double-encapsulated capsules was used as radiation sources in this study. The films obtained were irradiated with five γ-radiation doses including 0.5, 1.5, 2.5, 3.5, and 5 kGy for 1 h, and these five samples were identified as samples B, C, D, E, and F, respectively, for the rest of the article. The non-irradiated sample was named as sample A in the article.
FTIR spectroscopy
All these six samples were grounded and pelletized using potassium bromide to be analyzed by attenuated total reflectance-Fourier transform infrared (FTIR; model no. 01831, SHIMADZU Corp, Japan). The spectra were recorded in the absorption band mode in the range of 4000–400 cm−1 and 20 scans for each sample.
Thermogravimetric analysis
Thermogravimetric analysis (TGA) of the six samples was conducted using a TGA-50 (Shimadzu Thermogravimetric Analyser; SHIMADZU Corp). Approximately 2.5 mg of each sample was placed in an aluminum cell and then heated from room temperature up to 600°C at a heating rate of 10°C min−1 and a nitrogen gas flow rate of 10 mL min−1.
DSC analysis
Differential scanning calorimetry (DSC) of the six films (around 3 mg) was conducted in an aluminum cell using a DSC machine (model: 00885; SHIMADZU Corp) at a nitrogen gas flow rate of 20 mL min−1 and a heating rate of 10°C min−1.
Tensile properties
TS, elongation at break % (Eb), and elastic modulus (Em) of the non-irradiated and irradiated films were investigated with Universal Testing Machine (H50ks-0404, RHi-50Z; Tinius Olsen Limited, Surrey, UK) according to DIN 53455 standard having efficiency within ±1%. 24 The films were cut into a rectangular size of dimension (6.5 × 1 cm2). For testing, crosshead speed of the machine was 100 mm min−1 with a gauze length and load of 40 mm and 500 N, respectively. Minimum 10 replicates were tested to take an average of any data.
Water uptake
Water uptake test was performed to observe the amount of water absorption into the films and, therefore, their sustainability in water. The films (around 0.3 g) were oven dried at 60°C for at least 24 h to remove the ambient moisture in the samples and were then used for water absorption according to ASTM test method D570-81. 25 The dried films were weighed for the initial value and immersed in water for different time periods such as 5, 15, 40, 80, 110, 140, and 170 min. It was measured by computerized moisture content analysis machine (KERN RH 120-3; maximum 120 g, Germany). An average of results from eight films was taken for each data of different time periods and of different radiation doses.
Scanning electron microscopy
Surface morphology of the six films was observed using scanning electron microscope (SEM; JEOL JSM-6490LA, Japan) at 20 kV with carbon coating onto the samples and under nitrogen atmosphere.
In vitro cytotoxicity study
In vitro, cytotoxicity test was performed using brine shrimp lethality bioassay method. 26 Brine shrimps (Artemia salina) were hatched using brine shrimp eggs in a conical shaped vessel (1 L), filled with sterile artificial seawater, and pH was adjusted at 8.5 using 0.1 N NaOH under constant aeration for 48 h. After hatching, active nauplii free from egg shells were collected from brighter portion of the hatching chamber and used for the assay. The films were dissolved in artificial seawater at 0.20 mg mL−1 concentration and were taken in petri dishes where the active nauplii were inoculated. After overnight incubation, the nauplii were counted; 0.5 mg mL−1 of vincristine sulfate (an anticancer drug) was considered as positive control.
Results and discussion
FTIR characterization
In all the spectra of the films (A, B, C, D, E, and F), as shown in Figure 1, peaks at around 3300, 1640 (1550–1500), and 2900 cm–1 are due to –NH stretching of secondary amide, C=O stretching, –NH bending, and C–H stretching, respectively, for the presence of gelatin. 27 The characterizing peaks of PVA for hydroxyl groups (around 3300 cm–1) and C–H stretching (around 2900 cm–1) are overlapped on the peaks of gelatin in the same range. 28 The IR absorption peak at around 1070 cm−1 is due to the presence of C–O bond of secondary alcoholic groups and the ester formed between the polymers used. 27 The weak peak at 1070 cm−1 due to C–O bond in sample A is also observed and this is because of the acid initiated esterification of the polymers happened. There is a shoulder shape, occurred at 2900 cm− 1 for the sample A and this shoulder became a clearly visible peak (from samples B to F) as the radiation doses increase from 0.5 kGy to 5 kGy due to corresponding increase in cross-linking of PVA with gelatin. 29 The peak at 2947 cm–1 indicates the presence of a hydrocarbon chromophore in the esterified product. Similarly, a peak at 3300 cm–1 in the spectra has been shifted to a slightly lower wavelength (and also increase in intensity) from the samples A to F might have been a consequence of more intermolecular or intramolecular hydrogen bonds. 29 However, there may be some signals related to the eventual stretching of the OH groups of the absorbed water molecules in this region. 30 These three kinds of peaks (1070, 2900, and 3300 cm–1) happen to be more prominent with the increase of irradiation dose especially in samples E and F. These observations confirm the probable cross-linking of between gelatin and PVA. Moreover, the typical IR peaks of the triple helix structure (1660 cm− 1) and the coil structure (1633 cm− 1) of gelatin were not observed in the spectra obtained, which may also be an indication of the cross-linking reaction, since cross-linked polymers normally lose the capacity to return to their native forms. 29
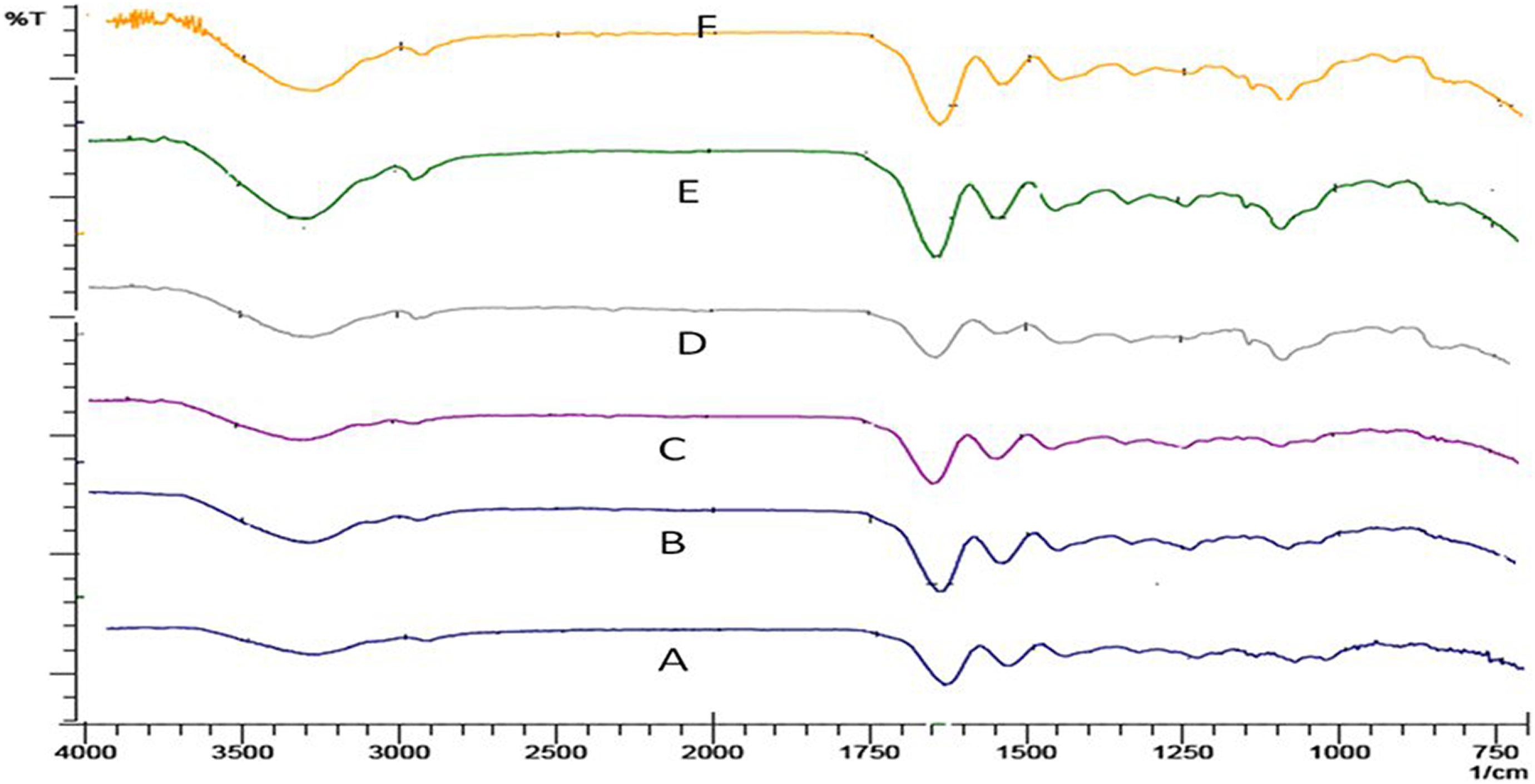
FTIR spectra of the films A, B, C, D, E, and F.
TG analysis
Thermograms obtained after TG analysis of the films (A, B, C, D, E, and F) are presented in Figure 2. The thermograms of PVA/gelatin blend showed four stages of degradation. The first stage involved dehydration and occurred at temperatures about 100°C with an initial weight loss of about 4–5%; then in the second step, degradation took place by ring scission and decomposition reactions. 13 The second one occurred in the range from 250°C to 380°C and then the third decomposition happened at 420–550°C. Further increase in temperature increases gradual decomposition of them and the decomposition continues till 600°C (the maximum temperature applied). The total degradation was 98 and 80% up to the final temperature of 600°C for samples A and F, respectively. Compared to the TGA values of sample A with samples B, C, D, E, and F, it is clearly observed that with the increase of irradiation doses to the films, the weight loss % decreased. This may be due to the amount of cross-linking developed in its structure after irradiation of different doses.
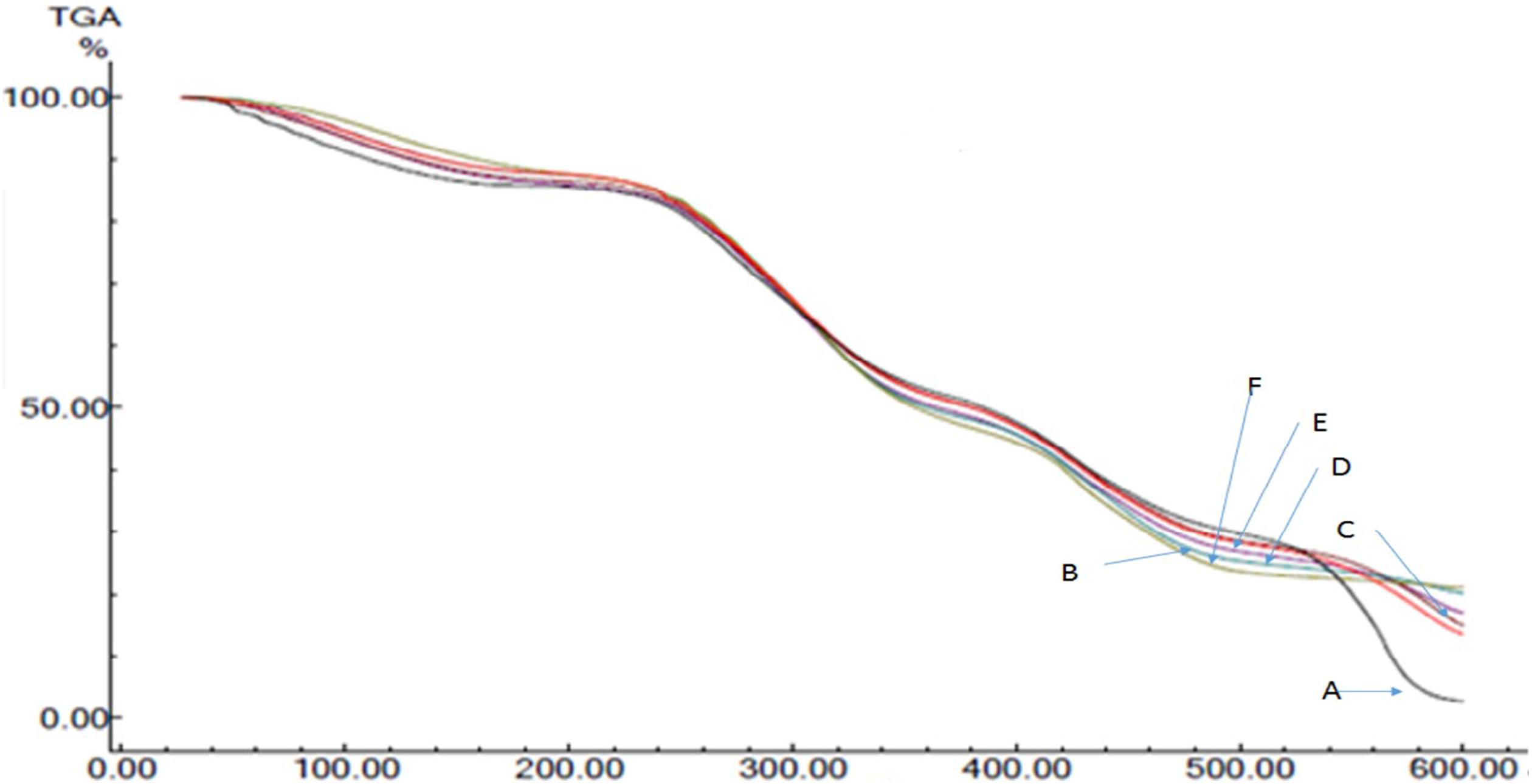
TG thermograms of the films A, B, C, D, E, and F.
DSC analysis
DSC is the technique used to determine the quantity of heat either absorbed or released when substances undergo physical or chemical changes. DSC thermograms for the six samples (A, B, C, D, E, and F) did not show any significant change due to irradiation applied in them (shown in Figure 3). The DSC thermograms of all the films obtained showed a glass transition temperature at 50–60°C due to the amorphous part of them. Pure PVA melts at 209°C and pure gelatin melts at 101–104°C. 31,32 A small endothermic peak occurring at the lower temperature (110°C) was due to the melting of the gelatin crystals; those were not cross-linked by the process. 29,30,33 The other endothermic peak observed at about 218°C was due to the melting of residual PVA crystals. Similar results were found in Sobral et al., where the films based on gelatin/PVA blends showed two melting peaks: one at 90–120°C and another at 172–209°C due to fusion of gelatin and PVA crystals, respectively. 17 Sobral et al. concluded that this is because the polymers were not thermodynamically miscible in the blend, therefore, emerging phase separation during DSC applied. 17 Gelatin is coiled in structure, while PVA has a planar zigzag structure. Because of this disparity in crystallinity of the two polymers, their blend behaves as an amorphous polymer complex and this is why, there is no sharp melting point rather a melting range at 250–270°C was observed in the thermograms of all samples. 27 From all the peaks observed in the DSC thermograms of the films, there is no significant change noticed. The possible cross-linking reactions were presented in Figure 4.
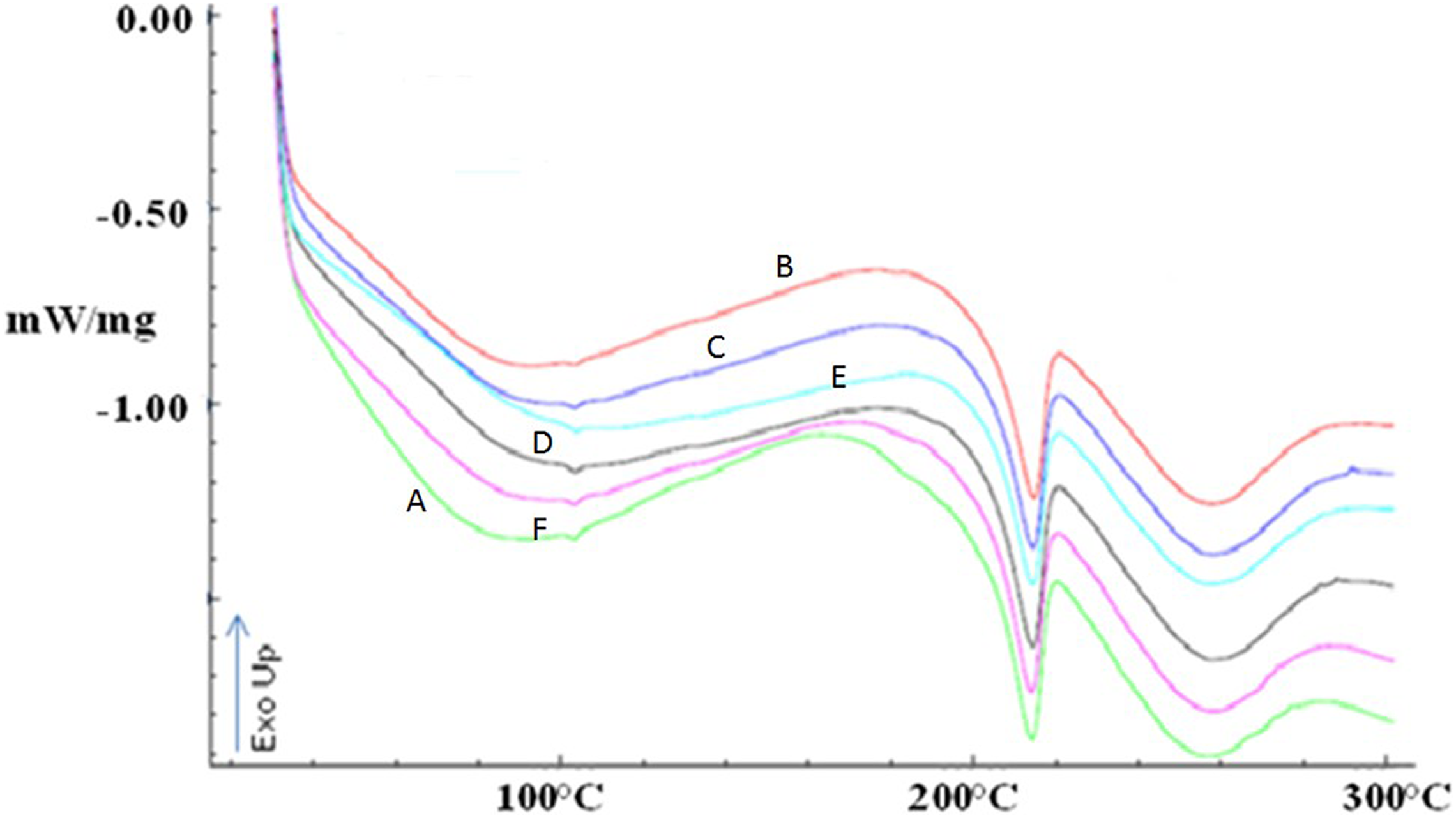
DSC thermograms of the films A, B, C, D, E, and F.
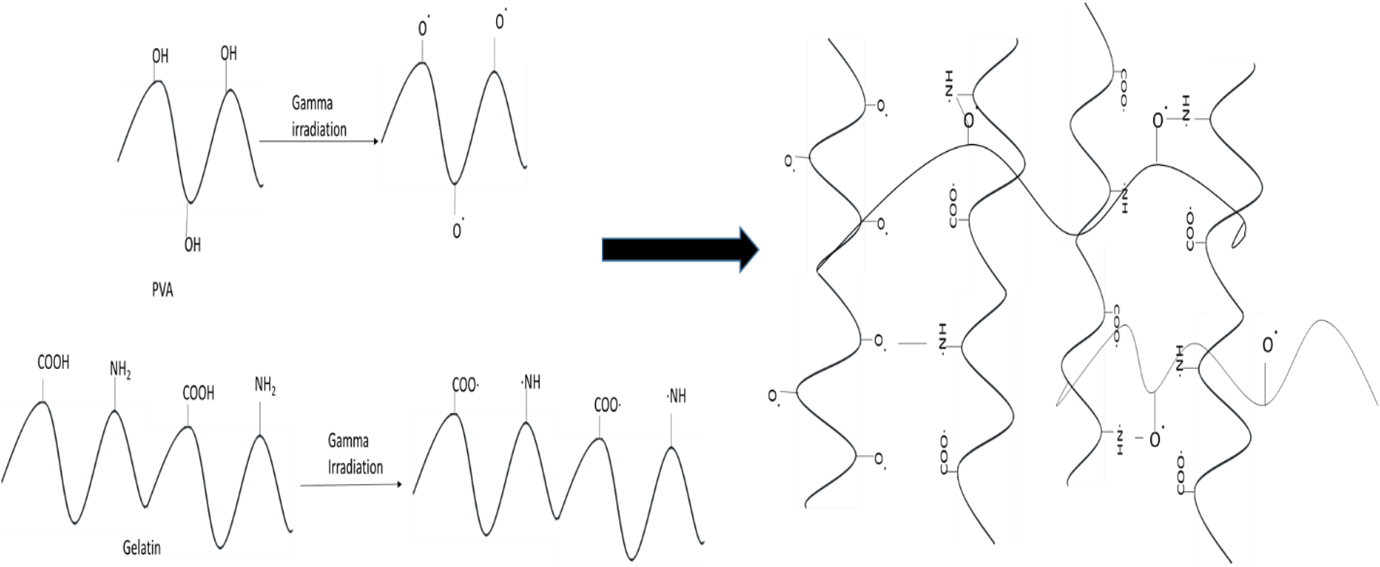
Proposed radiation induced cross-linking reactions between PVA and gelatin.
Mechanical properties
TS and Eb (%) of the films A, B, C, D, E, and F after different radiation doses applied were measured and presented in Figure 5. It is observed from Figure 5 that TS is increasing with increasing radiation doses. This is probably due to the reaction happened between PVA and gelatin with the influence of increasing radiation doses which results an increase in cross-linking. However, the difference of TS of non-irradiated (A) and irradiated samples (B, C, D, E, and F) is not very high. Sample F shows noticeably higher TS (16 MPa) among those of the other films (p value is less than 0.05), which may be for higher amount of cross-linking formation between the polymers because of the higher dose of radiation applied. It is usual that with the increase in TS when more cross-linking occurs between the polymers, the Eb (%) decreases. Thus, Eb (%) of the films shows a decreasing trend with increasing radiation doses. The non-irradiated sample A shows the highest Eb around 295% and the sample F irradiated with the highest dose shows the lowest Eb of about 72%. Em of the films A, B, C, D, E, and F obtained is 323 ± 16, 281 ± 17, 312 ± 12, 318 ± 18, 389 ± 18, and 437 ± 18 MPa, respectively. It is observed from Figure 6 that Em of the films increases with increasing radiation doses applied. The sample B shows a little decrease in Em than that of the non-irradiated film; however, their Ems are within the standard deviations. The maximum Em has been shown by the sample F, which is about 75% higher in amount than that of non-irradiated sample A. The higher Em value of sample F results from the increased TS and decreased Eb value of it due to an increase in cross-linking occurred at higher radiation dose applied in sample F.
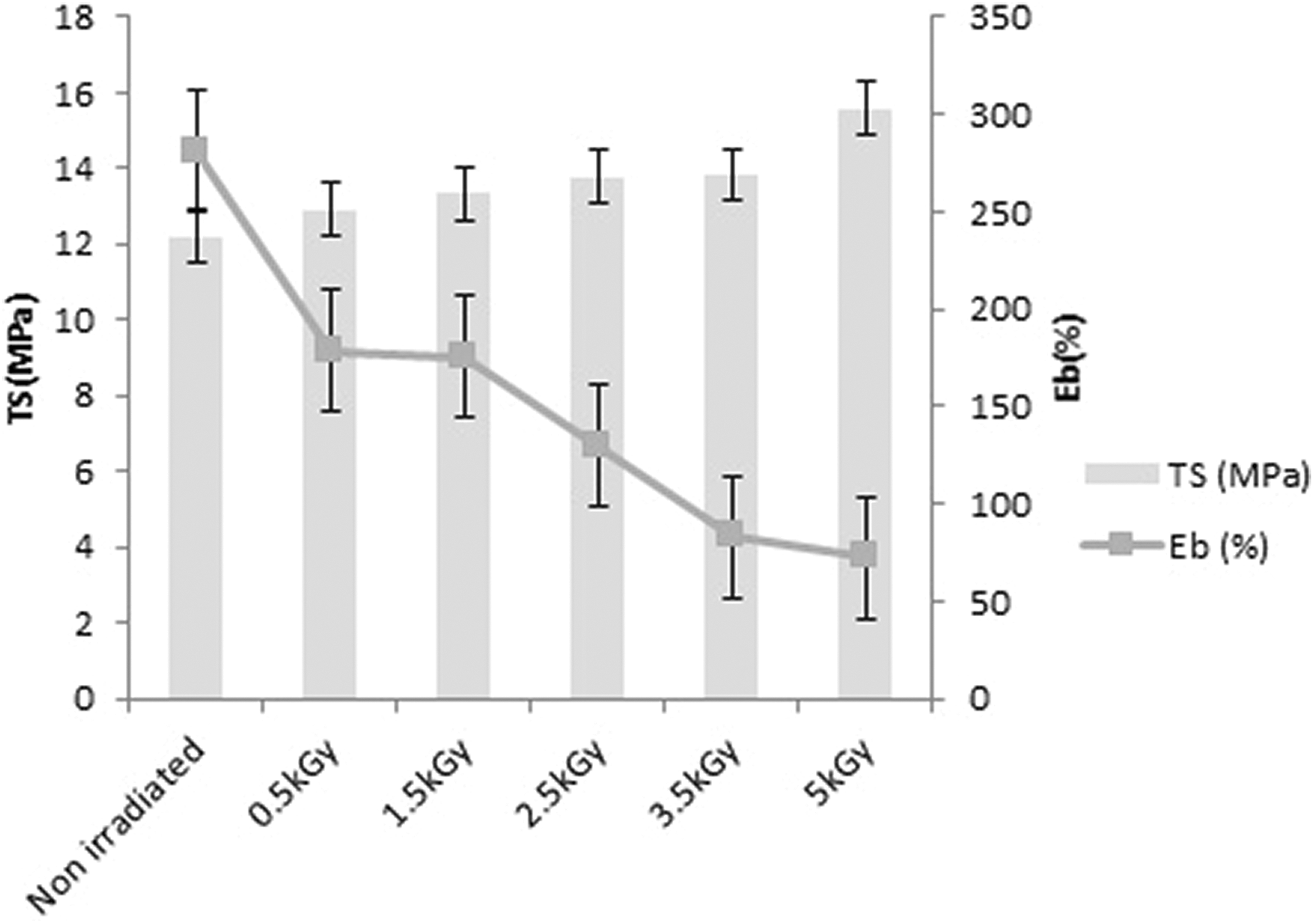
TS and Eb% of the films (A, B, C, D, E, and F) irradiated at different doses.
Water uptake
The water uptake data have been represented in Figure 7. All the curves primarily show increase in water uptake and then they become plateau due to attain equilibrium point of it. Both gelatin and PVA are hydrophilic in nature. This is why, maximum water uptake initially occurs into all the films and the saturation is reached in around 15–20 min for all of them. Then, the films gradually release water and the data are taken up to 7 days. However, it is noticeable that non-irradiated film A uptakes more water than the irradiated films (B, C, D, E, and F) because of the presence of free carboxylic and amino groups of gelatin and hydroxyl group of PVA. 27 These functional groups are essentially responsible for gelatin and PVA being hydrophilic. After 7 days, samples B, C, D, and E show water uptake around 240 wt%, while sample F contains about 300 wt%. It is discussed in earlier sections that in sample F, more cross-linking happened between polymers due to higher amount of radiation doses applied and this is so, sample F can retain much amount of water into the cross-linked structure of it compared to the other irradiated samples.
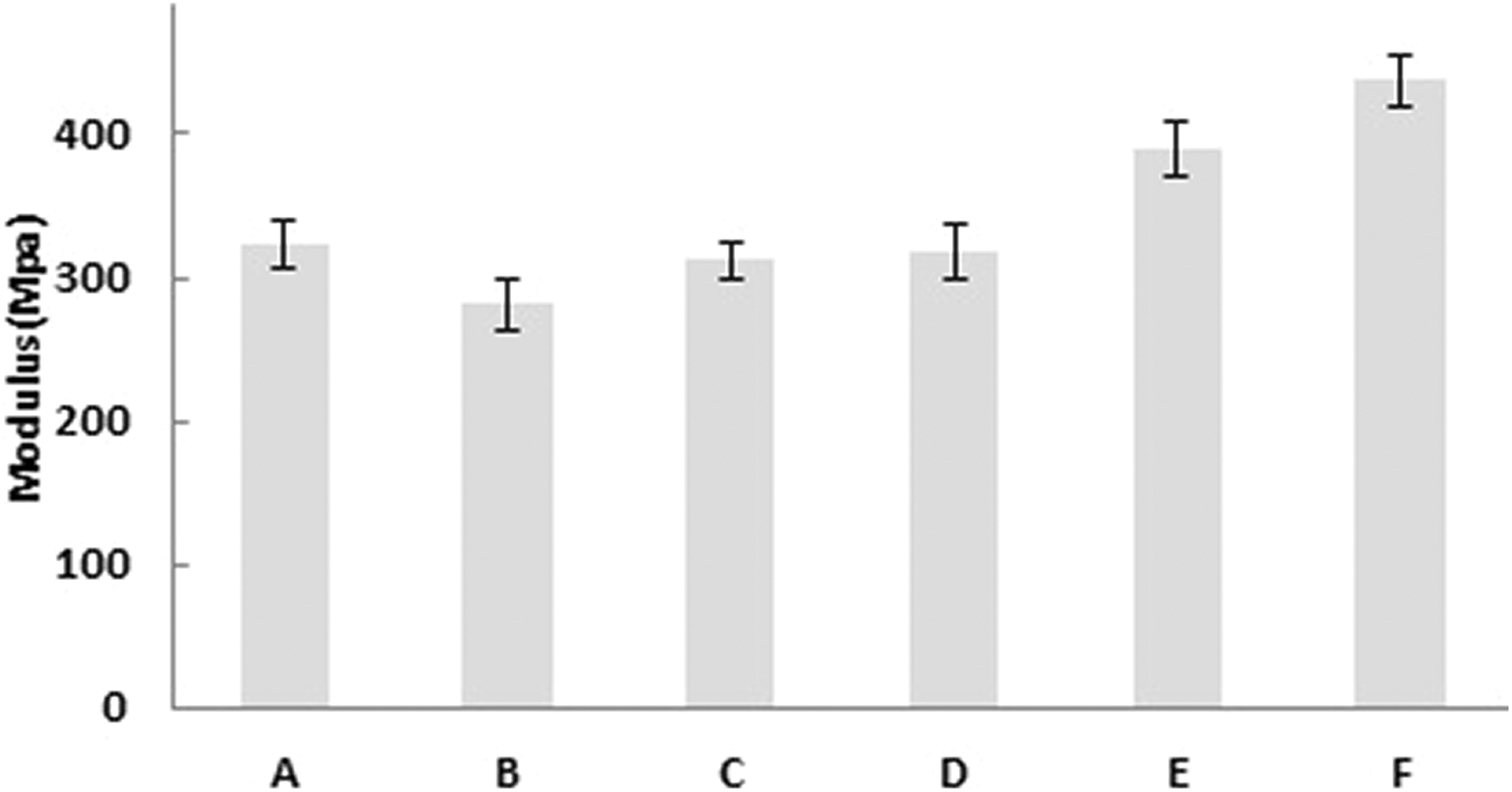
Em of the films (A, B, C, D, E, and F) irradiated at different doses.
In vitro cytotoxicity study
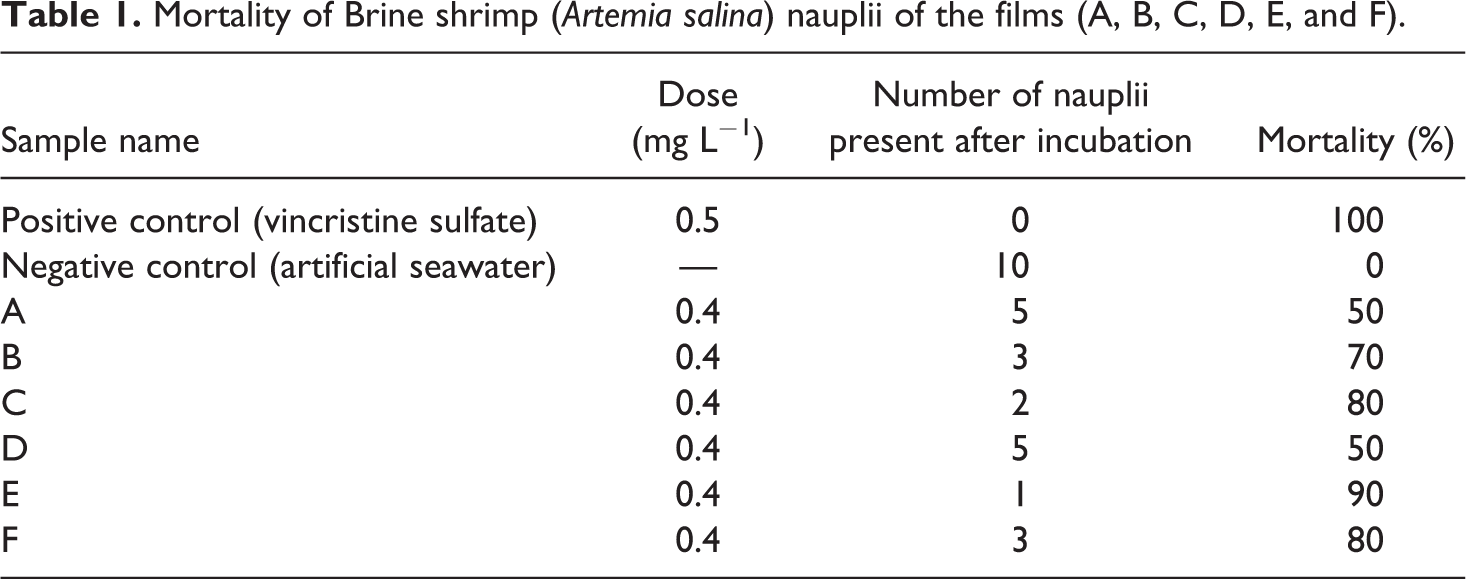
One method to evaluate cell and tissue response is to measure in vitro cytotoxicity or its quality of being toxic to cell. Cell toxicity is determined by cell lysis (death) or the inhibition of cell proliferation. Prior to investigating a material in vivo, cytotoxicity can provide insight to any potential issues with the local tissue response. In this work, the possibility of death of nauplii due to toxicity is shown in Table 1. The number of death of nauplii varies insignificantly. Although the number of death of nauplii is the highest for sample E, there is no significant trend is observed in the samples due to the irradiation used into them. There may be three reasons for nauplii’s death: (a) the films may have cytotoxic effect, (b) dissolved oxygen concentration of the saline water may have decreased, and (c) a viscous layer of PVA may be formed on the gills of nauplii. 34 The possibility of death of nauplii due to toxicity is maximum 50% for the case of samples A and D, which explains that irradiation did not make the films toxic. Moreover, both gelatin and PVA are biocompatible and nontoxic; thus, the most possible reason for the death of nauplii is the formation of PVA layer on their gills that eventually inhibit oxygen permeability to the gills and causes death. 35
Mortality of Brine shrimp (Artemia salina) nauplii of the films (A, B, C, D, E, and F).
SEM analysis
No significant change in the surface morphology of the non-irradiated and irradiated films is observed from SEM images (Figure 8). Some imprints are observed on the samples which are the artefacts produced during casting onto silicon cloths. This can be concluded from the images that the γ-irradiation did not impart any change in the morphology of the films.
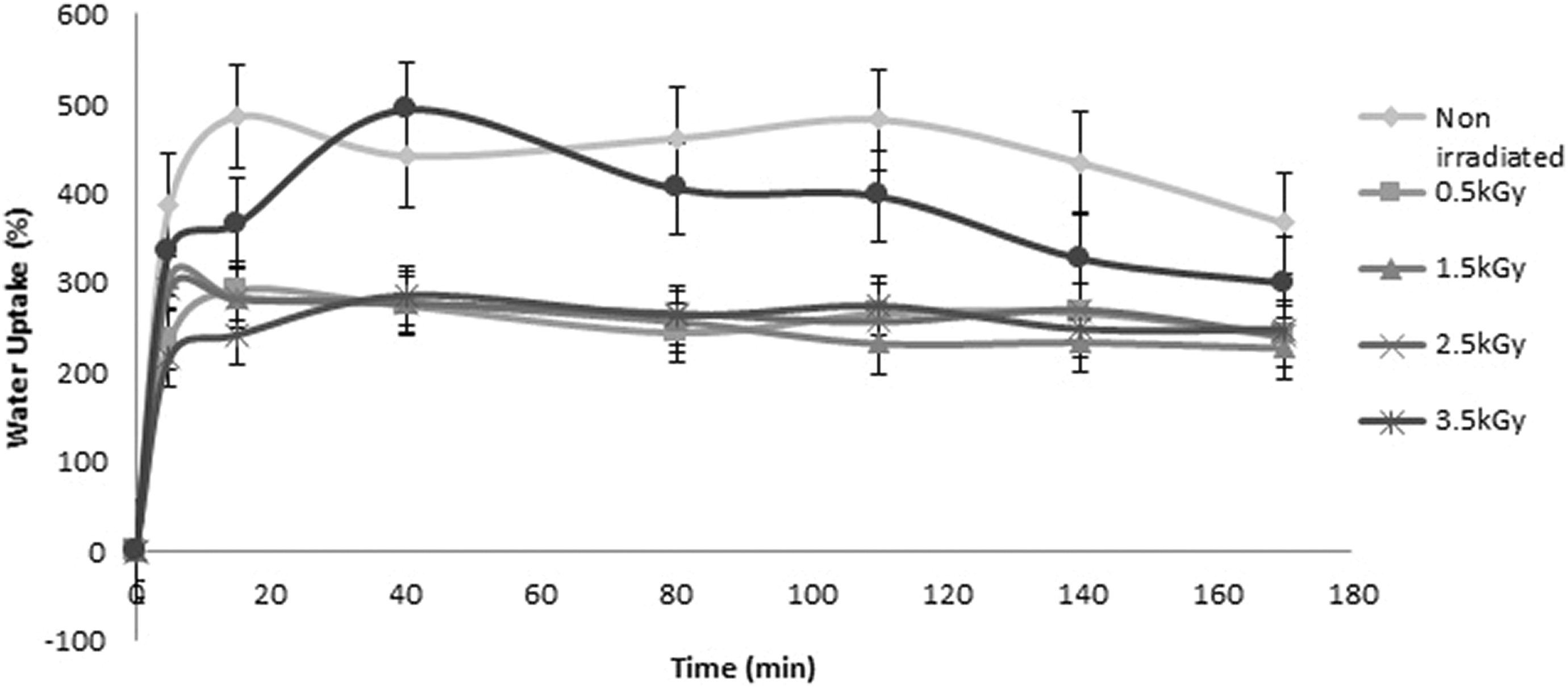
Water uptake (wt%) of the films (A, B, C, D, E, and F) up to 7 days.

(a) and (b) The SEM images of non-irradiated and irradiated samples, respectively.
Conclusion
Gelatin-PVA films were prepared successfully by mixing both gelatin and PVA at pH 2 by casting and then irradiated at different doses (0.5, 1.5, 2.5, 3.5, and 5 kGy). FTIR analysis confirms cross-linking of gelatin and PVA by esterification after using γ-irradiation. TGA analysis determines that the 5 kGy irradiated sample retains around 20% after heating them up to 600°C, which is much higher in amount than that of the non-irradiated sample. DSC analysis reveals that there is a significant amount of cross-linking present in both the non-irradiated and irradiated films along with a noticeable volume of free PVA and gelatin in them. TS and Em of the irradiated films showed simultaneous increase with the increase of radiation dose. Water uptake is also improved for the irradiated samples. In vitro cytotoxicity test clarifies that the non-irradiated samples are not cytotoxic. SEM confirms no change in the morphology of the films even after irradiation. It can be inferred from this work that γ-irradiated gelatin-PVA film can be a potential nontoxic material with increased thermal and mechanical properties along with good water retention capacity to be used as artificial articular cartilage.
Footnotes
Acknowledgement
The authors are thankful to the University Grants Commission, Bangladesh for funding this research.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The author(s) received a financial support from the University Grants Commission, Bangladesh, for the research, of this article.
